NSAIDs in the Acute Treatment of Migraine: A Review of Clinical and Experimental Data
Abstract
:1. Introduction
| Migraine without aura | |
| A. At least 5 attacks fulfilling criteria B–D | |
| B. Headache attacks lasting 4–72 hours (untreated or unsuccessfully treated) | |
| C. Headache has at least 2 of the following characteristics: | |
| -Unilateral location | |
| -Pulsating quality | |
| -Moderate or severe pain intensity | |
| -Aggravation by or causing avoidance of routine physical activity | |
| D. During headache at least 1 of the following: | |
| -Nausea and/or vomiting | |
| -Photophobia and phonophobia | |
| E. Not attributed to another disorder | |
| Migraine with aura | |
| A. At least 2 attacks fulfilling criteria B–D | |
| B. Aura consisting of at least one of the following, but no motor weakness: | |
| -fully reversible visual symptoms including positive features (e.g., flickering lights, spots or lines) and/or negative features ( i.e. , loss of vision) | |
| -fully reversible sensory symptoms including positive features ( i.e. , pins and needles) and/or negative features ( i.e. , numbness) | |
| -fully reversible dysphasic speech disturbance | |
| C. Art least two of the following: | |
| -homonymous visual symmptoms1 and/or unilateral sensory symptoms | |
| -at least one aura symptom develops gradually over ≥5 minutes and/or different aura symptoms occur in succession over ≥5 minutes | |
| -each symptom lasts ≥5 and <60 minutes | |
| D. Headache fulfilling criteria B–D for migraine without aura begins during the aura or follows aura within 60 minutes | |
| E. Not attributed to another disorder | |
2. Pharmacology
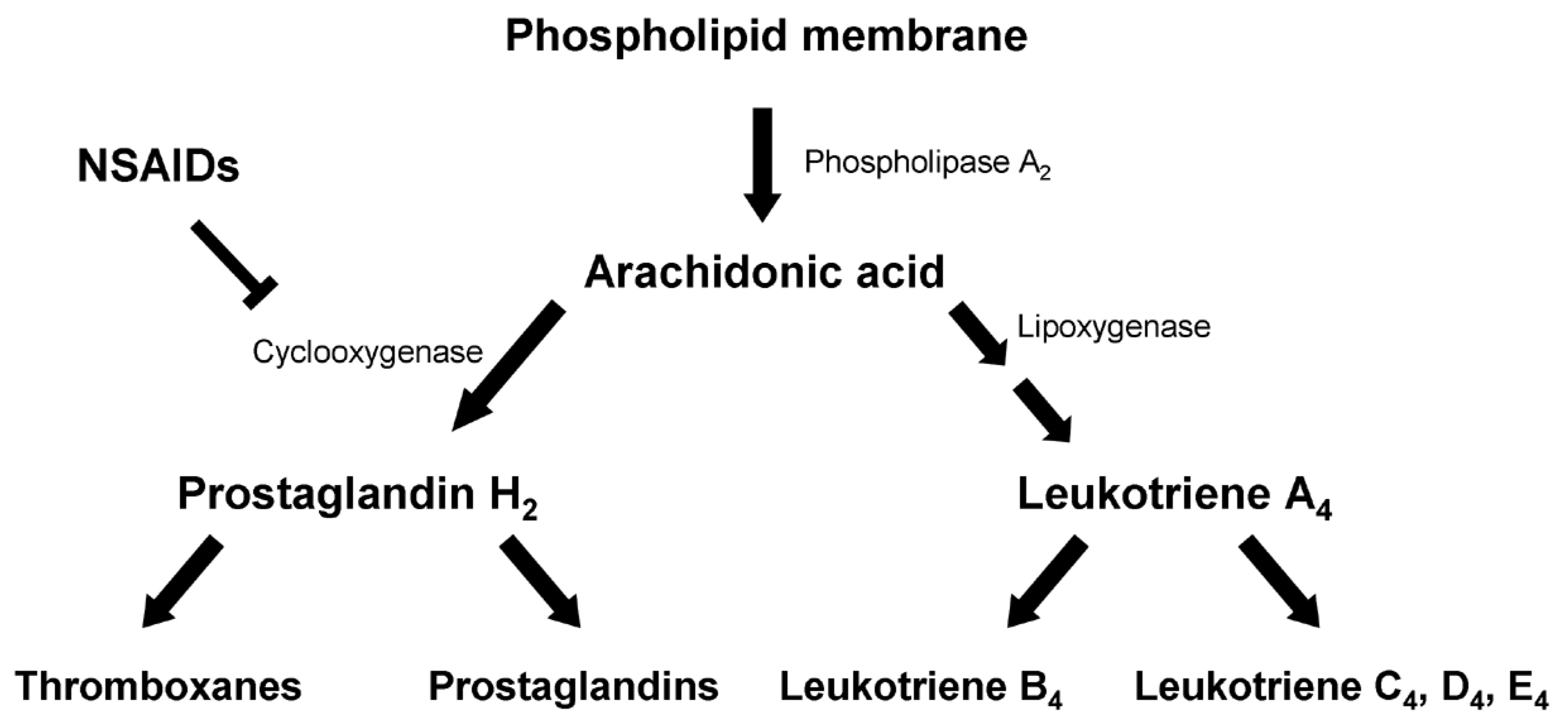
2.1. NSAIDs and Migraine Pathophysiology
2.1.1. Experimental data
2.1.1.1. NSAIDs and peripheral nociceptors in the trigeminovascular system
2.1.1.2. NSAIDs and 2nd order trigeminal nociceptors
2.1.1.3. NSAIDs and migraine "generators"
2.1.1.4. Cortical spreading depression
2.1.2. Clinical data
2.2. Pharmacokinetics
3. Results of Randomized Controlled Trials
3.1. Placebo Controlled Trials
| Authors | Drugs | N° patients | Results |
|---|---|---|---|
| Tfelt-Hansen et al., 1984 [67] | ASA 650+Met 10 / ASA 650/ Pl | 85 | Need for rescue medication: ASA+Met (63/92) = ASA (51/86) < Pl (75/95) |
| Effect on pain: Met + ASA = ASA > Pl | |||
| Henry et al., 1995 [61] | ASA 900+Met 10 / Pl | 303 | Success rate: ASA + Met (57%) > Pl (19%) |
| Lange et al., 2000 [68] | ASA 1000 / Pl | 343 | 2 h response: ASA (55%) > Pl (37%) p < 0.001 |
| 2 h pain free: ASA (29%) > Pl (17%) p = 0.007 | |||
| MacGregor et al., 2002 [69] | ASA 900 / Pl | 101 | 2 h response: ASA (48%) > Pl (19%) p = 0.0005 |
| 2 h pain free: ASA (14%) = Pl (5%) | |||
| 3 h pain free: ASA (18%) > Pl (5%) p < 0.05 | |||
| Tfelt-Hansen et al., 1995 [65] | ASA 900+ Met / Suma 100 / Pl | 421 | Success rate 1st attack: ASA + Metocl (57%) = Sum (53%) > Pl (24%) |
| Diener et al., 2004 [76] | ASA 1000 / Suma 50 / Ibu 400/ Pl | 312 | 2 h relief : Ibu (60.2%) = Suma (55.8%) = ASA (52.5%) > Pl |
| 2 h pain free: Suma (37.1%) = Ibu (33.2%) > ASA (27.1%) > Pl (12.6%) | |||
| Diener et al., 1999 [66] | Lys-ASA 1000 iv / Suma 6 s.c. | 275 | 2 h relief: Suma (91%) > Lys-ASA (74%) > Pl (24%) |
| 2 h pain free: Suma (76.3%) > Lys-ASA (43.7%) > Pl (14.3%) | |||
| Havanka-Kanniainen 1989 [ 71] | Ibu 800 (+ 400) / Pl | 27 | Decrease of attack duration: Ibu (5 h) > Pl (11 h) |
| Mild attacks: Ibu (33%) > Pl (7%) | |||
| Kloster et al., 1992 [72] | Ibu 1200 / Pl | 25 | Headache severity: Ibu (1,78) > Pl (2,33) |
| Need for rescue medication: Ibu (25.6%) > Pl (57.5%) | |||
| Sandrini et al., 1998 [73] | IbuArg 400 / Pl | 29 | Pain reduction: IbuArg > Pl |
| Kellstein et al., 2000 [74] | Ibuliq 200, 400, 600 / Pl | 735 | 2 h relief: Ibuliq (64%, 72%, 72%) > Pl (50%) |
| 2 h pain free: Ibuliq (25%, 28%, 29%) > Pl (13%) | |||
| Codispoti et al., 2001 [75] | Ibu 200, 400 / Pl | 460 | 2 h response: Ibu 400 (41%) = Ibu 200 (42%) > Pl (28%) |
| Misra et al., 2007 [76] | Ibu 400 / Riza 10 / Pl | 155 | 2 h relief: Riza (73%) > Ibu (54%) > Pl (8%) |
| Hämäläinen et al., 1997 [78] | Ibu 10/kg / Ace 15/kg / Pl (children 4–16 yo) | 88 | Ibu > Ace > Pl |
| Lipton et al., 2000 [80] | Ace 1000 / Pl | 140 | 2 h response: Ace (58%) > Pl (39%) |
| 2 h pain free: Ace (22%) > Pl (11%) | |||
| Boureau et al., 1994 [82] | ASA 1000 / Ace 400+Cod 25 | 198 | Success rate: ASA (52%) = Ace + Cod (50%) > Pl (30%) |
| Leinisch et al., 2005 [83] | Ace 1000 iv / Pl | 60 | 2 h pain free: Ace (10%) = Pl (13%) |
| 2 h relief: Ace (30%) = Pl (20%) | |||
| Hakkarainen et al., 1979 [84] | Tol 200 / Erg 1 / ASA 500 / Pl | 20 | Duration of attacks: Tol (3,2) = Erg (3,8) = ASA (4,2) > Pl (7,1) |
| Myllyla et al., 1998 [85] | Tol 200 / Suma 100 | 141 | 2 h response: Tol (77%) = Sum (79%) > Pl (29%) |
| Tokola et al., 1984 [86] | Tol 200 / Tol 200+Met 10/ Tol 200+Caff 100 / Pl | 49 | Tol + Met > Tol = Tol + Caff > Met = Caf = Pl |
| Nestvold, et al., 1985 [88] | Napro 750 (+205/500) / Pl | 32 | Headache relief: Napro > Pl |
| Need for rescue medication: Napro (24%) > Pl (46%) | |||
| Andersson et al., 1989 [89] | Napro 750 (up to 1250) / Pl | 32 | 2 h relief: Napro > Pl |
| Severity for the whole attack: Napro (2,2) = Pl(2,2) | |||
| Johnson et al., 1985 [90] | NaproNa 825 (up to 1375 / Pl | 61 | Relief: NaproNa (3,8) > Pl (5,0) |
| Need for rescue medication: NaproNa (44%) < Pl (67%) | |||
| Smith et al., 2005 [93] | NaproNa 500 / Suma 50 / NaproNa 500 + Suma 50 / Pl | 972 | 2h relief: NaproNa + Suma (65%) > Suma (49%) = NaproNa (46%) > Pl (27%) |
| 2h pain free: NaproNa + Suma (34%) > Suma (20%) = NaproNa (18%) > Pl (6%) | |||
| Brandes et al., 2007 [91] | NaproNa 500 / Suma 85 / NaproNa 500 + Suma 85 / Pl (2 studies) | 1461/1495 | 2h relief: NaproNa + Suma (65/57%) > Suma (55/50%) > NaproNa (44/43%) > Pl (28/29%) |
| 2h pain free: NaproNa + Suma (34/30%) > Suma (25/23%) = NaproNa (15/16%) > Pl (9/10%) | |||
| Bussone et al., 1999 [94] | DicloK 50, 100 / Suma 100 / Pl | 156 | 2 h relief: DicloK 50 (-17) = Diclo 100 (-18.6) = Suma 100 (-14.5) > Pl |
| Dahlöf et al., 1993 [95] | DicloK 50, 100 / Pl | 73 | 2 h relief: DicloK 100 = DicloK 50 > Pl |
| Peroutka et al., 2004 [97] | Diclo 100 / Diclo 100 + Caff 100 / Pl | 72 | 1 h relief: Diclo + Caff (41%) > Diclo (27%) > Pl (14%) |
| Need for rescue medication: Diclo + Caff (33%) = Diclo (30%) > Pl (63%) | |||
| Massiou et al., 1991 [64] | Diclo 50 (100) / Pl | 91 | 2 h pain free: Diclo (27%) > Pl (19%) |
| Need for rescue medication: Diclo (54%) > Pl (66%) | |||
| Del Bene et al., 1987 [98] | Diclo 75 im / Pl | 32 | Response to treatment: Diclo > Pl |
| Kinnunen et al., 1988 [100] | Pirpro 200 (500) / Erg 2 (5) / Pl | 55 | Pain relief: Pirpro = Erg > Pl |
| Need for rescue medication: Pirpro (18/58) = Erg (18/59) < Pl (32/60) | |||
| Awidi et al., 1982 [101] | Flurbi 100 (300) / Pl | 19 | Relief score: Flurbi (3,2) > Pl (0,7) |
| Dib et al., 2002 [102] | Keto 75, 150 / Zolmi 2, 5 / Pl | 235 | 2 h relief: Zolmi (67%) = Keto 150(62%) = Keto 75 (63%) > Pl |
| Silberstein et al., 2004 [104] | Rof 25, 50 / Pl | 557 | 2 h relief: Rof 50 (57%) = Rof 25 (54%) > Pl (34%) |
3.2. Comparative Trials
| Trial | Drug | N° patients | Results: |
|---|---|---|---|
| Geraud et al., 2002 [107] | ASA 900+Met 10 / Zolmi 2,5 | 666 | 2 h relief: ASA + Met (32.9%) = Zol (33.4%) |
| 2 h pain free: Zolmi (10.7%) > ASA + Met (5.3%) | |||
| Tfelt-Hansen et al., 1995 [65] | ASA 900+ Met / Suma 100 / Pl | 421 | Success rate 1st attack: ASA + Metocl (57%) = Sum (53%) > Pl (24%) |
| Diener et al., 2004 [76] | ASA 1000 / Suma 50 / Ibu 400/ Pl | 312 | 2 h relief : Ibu (60.2%) = Suma (55.8%) = ASA (52.5%) > Pl |
| 2 h pain free: Suma (37.1%) = Ibu (33.2%) > ASA (27.1%) > Pl (12.6%) | |||
| Diener et al., 1999 [66] | Lys-ASA 1000 iv / Suma 6 s.c. | 275 | 2 h relief: Suma (91%) > Lys-ASA (74%) > Pl (24%) |
| 2 h pain free: Suma (76.3%) > Lys-ASA (43.7%) > Pl (14.3%) | |||
| Misra et al., 2007 [76] | Ibu 400 / Riza 10 / Pl | 155 | 2 h relief: Riza (73%) > Ibu (54%) > Pl (8%) |
| Hämäläinen et al., 1997 [78] | Ibu 10/kg / Ace 15/kg / Pl (children 4–16 yo) | 88 | Ibu > Ace > Pl |
| Karachalios et al., 1992 [109] | Diclo 75 im / Ace im. | 86 | 30 min pain free: Dicl (88%) > Ace (17.5%) |
| Bussone et al., 1999 [94] | DicloK 50, 100 / Suma 100 / Pl | 156 | 2 h relief: DicloK 50 (-17) = Diclo 100 (-18.6) = Suma 100 (-14.5) > Pl |
| Schoenen et al., 2008 [99] | Aceclo 100 + Almo 12.5 / Almo 12.5 + Pl | 112 | 2 h relief: Aceclo + Almo (69%) > Almo + Pl (57.9%) |
| 2 h pain free: Aceclo + Almo (40.7%) > Almo + Pl (29.1%) | |||
| Hakkarainen et al., 1979 [84] | Tol 200 / Erg 1 / ASA 500 / Pl | 20 | Duration of attacks: Tol (3,2) = Erg (3,8) = ASA (4,2) > Pl (7,1) |
| Myllyla et al., 1998 [85] | Tol 200 / Suma 100 | 141 | 2 h response: Tol (77%) = Sum (79%) > Pl (29%) |
| Larsen et al., 1990 [63] | Tol 200,400 / Ace 500,1000 | 83 | 2 h effect on pain: Tol > Ace |
| Pradalier et al., 1985 [111] | Napro 825 / Erg 2+Caff 91.5+Cyclizine 50 | 114 | If taken within 2 h of onset: Napro > Erg + Caff + C |
| Treves et al., 1992 [112] | Napro / Erg | 42 | Overall patients' satisfaction: Napro > Erg |
| Duration and severity reduction: Napro = Erg | |||
| Sargent et al., 1988 [113] | Napro 825 / Erg 1+Caff 100 | Attack abortion: Napro = Erg + Caff > Pl | |
| Nausea reduction: Napro > Erg + Caff | |||
| Smith et al., 2005 [93] | NaproNa 500 / Suma 50 / NaproNa 500+Suma 50 / Pl | 972 | 2 h relief: NaproNa + Suma (65%) > Suma (49%) = NaproNa (46%) > Pl (27%) |
| 2 h pain free: NaproNa + Suma (34%) > Suma (20%) = NaproNa (18%) > Pl (6%) | |||
| Brandes et al., 2007 [91] | NaproNa 500 / Suma 85 / NaproNa 500+Suma 85 / Pl (2 studies) | 1461/1495 | 2 h relief: NaproNa + Suma (65/57%) > Suma (55/50%) > NaproNa (44/43%) > Pl (28/29%) |
| 2 h pain free: NaproNa + Suma (34/30%) > Suma (25/23%) = NaproNa (15/16%) > Pl (9/10%) | |||
| Kinnunen et al., 1988 [100] | Pirpro 200 (500) / Erg 2 (5) / Pl | 55 | Pain relief: Pirpro = Erg > Pl |
| Need for rescue medication: Pirpro(18/58) = Erg (18/59) < Pl (32/60) | |||
| Karabetsos et al., 1997 [103] | Keto 100 im / Ace 500 im | 64 | 40 min relief: Keto (82.5%) > ace (17.5%) |
| Dib et al., 2002 [102] | Keto 75, 150 / Zolmi 2,5 / Pl | 235 | 2 h relief: Zolmi (67%) = Keto 150 (62%) = Keto 75 (63%) > Pl |
| Larkin et al., 1992 [114] | Ketorolac 30 im / Meperidine 75 im | 31 | 1 h relief: Meperidine > Ketorolac |
| Davis et al., 1995 [115] | Ketorolac 60 im / Meperidine 75+ promethazine 25 im | 42 | 30 min response: Meperidine + promethazine (68%) = Ketorolac (55%) |
| Shrestha et al., 1996 [116] | Ketorolac 60 im / Chlorpromazine 25 iv | 47 | 30 min-2h relief: Ketorolac = Chlorpromazine |
| Seim et al., 1998 [117] | Ketorolac 30 iv / Prochlorperazine 10 iv | 64 | 1 h relief: Prochlorperazine > Ketorolac |
| Meredith et al., 2003 [118] | Ketorolac 30 iv / Suma 20 nasal | 29 | 1 h relief: Ketorolac > Suma |
| Sandrini et al., 2007 [119] | Indo 25+Prochlor 2+Caff 75 / Suma 50 | 297 | 2 h relief: I + P + C (57%) = Suma (57%) |
| 2 h pain free: I + P + C (32%) = Suma (36%) | |||
| Di Monda et al., 2003 [120] | Indo 25 + Prochlor 2 + Caff 75 rectal / Suma 25 rectal | 88 | 2 h relief: I + P + C (67%) = Suma (63%) |
| 2 h pain free: I + P + C (47%) > Sum (35%) |
3.3. Summary of Efficacy Data
3.4. Adverse Effects
4. Conclusions
References
- Diener, H.C.; Katsarava, Z.; Limmroth, V. Current diagnosis and treatment of migraine. Schmerz 2008, 22 (Suppl. 1), 51–58. [Google Scholar]
- Headache Classification Subcommittee of the International Headache Society. The international classification of headache disorders. Cephalalgia 2004, 24 (Suppl. 1), 9–160. [CrossRef] [PubMed]
- Lipton, R.B.; Diamond, S.; Reed, M.; Diamond, M.L.; Stewart, W.F. Migraine diagnosis and treatment: Results from the American Migraine Study II. Headache 2001, 41, 638–645. [Google Scholar]
- Lipton, R.B.; Scher, A.I.; Steiner, T.J.; Bigal, M.E.; Kolodner, K.; Liberman, J.N.; Stewart, W.F. Patterns of health care utilization for migraine in England and in the United States. Neurology 2003, 60, 441–448. [Google Scholar]
- Tulunay, F.C. NSAIDs: Behind the mechanisms of action. Funct. Neurol. 2000, 15 (Suppl. 3), 202–207. [Google Scholar] [PubMed]
- Hardman, J.G.; Limbird, L.E.; Goodman Gilman, Alfred, cons. Insel PA Analgesic-antipyretics and antiinflammatory agents and drug employed in the treatment of gout. In Goodman and Gilman's: The Pharmacological Basis for Therapeutics, 9th ed; McGraw-Hil: New York, NY, USA, 1996; pp. 617–657. [Google Scholar]
- Obrien, J.R. Effects of salicylates on human-platelets. CC/Clin. Pract. 1980, 51, 18. [Google Scholar]
- Ebersberger, A.; Averbeck, B.; Messlinger, K.; Reeh, P.W. Release of substance P, calcitonin gene-related peptide and prostaglandin E2 from rat dura mater encephali following electrical and chemical stimulation in vitro. Neuroscience 1999, 89, 901–907. [Google Scholar] [CrossRef] [PubMed]
- Goadsby, P.J.; Edvinsson, L.; Ekman, R. Release of vasoactive peptides in the extracerebral circulation of humans and the cat during activation of the trigeminovascular system. Ann. Neurol. 1988, 23, 193–196. [Google Scholar]
- Williamson, D.J.; Hargreaves, R.J. Neurogenic inflammation in the context of migraine. Microsc. Res. Tech. 2001, 53, 167–178. [Google Scholar]
- Markowitz, S.; Saito, K.; Moskowitz, M.A. Neurogenically mediated plasma extravasation in dura mater: Effect of ergot alkaloids. A possible mechanism of action in vascular headache. Cephalalgia 1988, 8, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Williamson, D.J.; Hargreaves, R.J.; Hill, R.G.; Shepheard, S.L. Intravital microscope studies on the effects of neurokinin agonists and calcitonin gene-related peptide on dural vessel diameter in the anaesthetized rat. Cephalalgia 1997, 17, 518–524. [Google Scholar]
- Strassman, A.M.; Raymond, S.A.; Burstein, R. Sensitization of meningeal sensory neurons and the origin of headaches. Nature 1996, 384, 560–564. [Google Scholar]
- Blau, J.N.; Dexter, S.L. The site of pain origin during migraine attacks. Cephalalgia 1981, 1, 143–147. [Google Scholar]
- Zhang, X.C.; Kainz, V.; Jakubowski, M.; Burstein, R.; Strassman, A.; Levy, D. Localization of COX-1 and COX-2 in the intracranial dura mater of the rat. Neurosci. Lett. 2009, 452, 33–36. [Google Scholar]
- Ellrich, J.; Schepelmann, K.; Pawlak, M.; Messlinger, K. Acetylsalicylic acid inhibits meningeal nociception in rat. Pain 1999, 81, 7–14. [Google Scholar]
- Levy, D.; Zhang, X.C.; Jakubowski, M.; Burstein, R. Sensitization of meningeal nociceptors: Inhibition by naproxen. Eur. J. Neurosci. 2008, 27, 917–922. [Google Scholar]
- Buzzi, M.G.; Sakas, D.E.; Moskowitz, M.A. Indomethacin and acetylsalicylic-acid block neurogenic plasma-protein extravasation in rat dura mater. Eur. J. Pharmacol. 1989, 165, 251–258. [Google Scholar]
- Schuh-Hofer, S.; Tayefeh, M.; Reuter, U.; Dirnagl, U.; Arnold, G. Effects of parecoxib on plasma protein extravasation and c-fos expression in the rat. Headache 2006, 46, 276–285. [Google Scholar]
- Burstein, R.; Yamamura, H.; Malick, A.; Strassman, A.M. Chemical stimulation of the intracranial dura induces enhanced responses to facial stimulation in brain stem trigeminal neurons. J. Neurophysiol. 1998, 79, 964–982. [Google Scholar]
- Burstein, R.; Yarnitsky, D.; Goor-Aryeh, I.; Ransil, B.J.; Bajwa, Z.H. An association between migraine and cutaneous allodynia. Ann. Neurol. 2000, 47, 614–624. [Google Scholar]
- Jenkins, D.W.; Langmead, C.J.; Parsons, A.A.; Strijbos, P.J. Regulation of calcitonin gene-related peptide release from rat trigeminal nucleus caudalis slices in vitro. Neurosci. Lett. 2004, 366, 241–244. [Google Scholar] [CrossRef] [PubMed]
- Beiche, F.; Scheuerer, S.; Brune, K.; Geisslinger, G.; Goppelt-Struebe, M. Up-regulation of cyclooxygenase-2 mRNA in the rat spinal cord following peripheral inflammation. FEBS Lett. 1996, 390, 165–169. [Google Scholar]
- Goppelt-Struebe, M.; Beiche, F. Cyclooxygenase-2 in the spinal cord: Localization and regulation after a peripheral inflammatory stimulus. Adv. Exp. Med. Biol. 1997, 433, 213–216. [Google Scholar]
- Kaube, H.; Hoskin, K.L.; Goadsby, P.J. Intravenous acetylsalicylic-acid inhibits central trigeminal neurons in the dorsal horn of the upper cervical spinal-cord in the cat. Headache 1993, 33, 541–544. [Google Scholar]
- Tassorelli, C.; Greco, R.; Armentero, M.T.; Blandini, F.; Sandrini, G.; Nappi, G. A role for brain cyclooxygenase-2 and prostaglandin-E2 in migraine: Effects of nitroglycerin. Int. Rev. Neurobiol. 2007, 82, 373–382. [Google Scholar]
- Tassorelli, C.; Joseph, S.A.; Nappi, G. Neurochemical mechanisms of nitroglycerin-induced neuronal activation in rat brain: A pharmacological investigation. Neuropharmacology 1997, 36, 1417–1424. [Google Scholar]
- Pardutz, A.; Szatmari, E.; Vecsei, L.; Schoenen, J. Nitroglycerin-induced nNOS increase in rat trigeminal nucleus caudalis is inhibited by systemic administration of lysine acetylsalicylate but not of sumatriptan. Cephalalgia 2004, 24, 439–445. [Google Scholar]
- Varga, H.; Pardutz, A.; Vamos, E.; Plangar, I.; Egyud, E.; Tajti, J.; Bari, F.; Vecsei, L. Cox-2 inhibitor attenuates NO-induced nNOS in rat caudal trigeminal nucleus. Headache 2007, 47, 1319–1325. [Google Scholar]
- Varga, H.; Pardutz, A.; Vamos, E.; Bohar, Z.; Bago, F.; Tajti, J.; Bari, F.; Vecsei, L. Selective inhibition of cyclooxygenase-2 attenuates nitroglycerin-induced calmodulin-dependent protein kinase II alpha in rat trigeminal nucleus caudalis. Neurosci. Lett. 2009, 451, 170–173. [Google Scholar]
- Yang, G.Y.; Lee, M.K.; Bae, Y.C.; Ahn, D.K. Intracisternal administration of COX inhibitors attenuates mechanical allodynia following compression of the trigeminal ganglion in rats. Prog. Neuropsychopharmacol. Biol. Psychiatry 2009, 33, 589–595. [Google Scholar]
- Diener, H.C. Positron emission tomography studies in headache. Headache 1997, 37, 622–625. [Google Scholar]
- Lance, J.W.; Lambert, G.A.; Goadsby, P.J.; Duckworth, J.W. Brainstem influences on the cephalic circulation: Experimental data from cat and monkey of relevance to the mechanism of migraine. Headache 1983, 23, 258–265. [Google Scholar]
- Weiller, C.; May, A.; Limmroth, V.; Juptner, M.; Kaube, H.; Schayck, R.V.; Coenen, H.H.; Diener, H.C. Brain stem activation in spontaneous human migraine attacks. Nat. Med. 1995, 1, 658–660. [Google Scholar]
- Breder, C.D.; Smith, W.L.; Raz, A.; Masferrer, J.; Seibert, K.; Needleman, P.; Saper, C.B. Distribution and characterization of cyclooxygenase immunoreactivity in the ovine brain. J. Comp. Neurol. 1992, 322, 409–438. [Google Scholar]
- Breder, C.D.; Dewitt, D.; Kraig, R.P. Characterization of inducible cyclooxygenase in rat brain. J. Comp. Neurol. 1995, 355, 296–315. [Google Scholar]
- Shin, M.C.; Jang, M.H.; Chang, H.K.; Kim, Y.J.; Kim, E.H.; Kim, C.J. Modulation of cyclooxygenase-2 on glycine- and glutamate-induced ion currents in rat periaqueductal gray neurons. Brain Res. Bull. 2003, 59, 251–256. [Google Scholar]
- Vaughan, C.W. Enhancement of opioid inhibition of GABAergic synaptic transmission by cyclo-oxygenase inhibitors in rat periaqueductal grey neurones. Br. J. Pharmacol. 1998, 123, 1479–1481. [Google Scholar]
- Leith, J.L.; Wilson, A.W.; Donaldson, L.F.; Lumb, B.M. Cyclooxygenase-1-derived prostaglandins in the periaqueductal gray differentially control C- vs. A-fiber-evoked spinal nociception. J. Neurosci. 2007, 27, 11296–11305. [Google Scholar] [PubMed]
- Leao, A.A.P. Spreading depression of activity in cerebral cortex. J. Neurophysiol. 1944, 7, 359–390. [Google Scholar]
- Lauritzen, M.; Jorgensen, M.B.; Diemer, N.H.; Gjedde, A.; Hansen, A.J. Persistent oligemia of rat cerebral cortex in the wake of spreading depression. Ann. Neurol. 1982, 12, 469–474. [Google Scholar]
- Lauritzen, M. Pathophysiology of the migraine aura. The spreading depression theory. Brain 1994, 117, 199–210. [Google Scholar] [CrossRef] [PubMed]
- Moskowitz, M.A.; Nozaki, K.; Kraig, R.P. Neocortical spreading depression provokes the expression of c-fos protein-like immunoreactivity within trigeminal nucleus caudalis via trigeminovascular mechanisms. J. Neurosci. 1993, 13, 1167–1177. [Google Scholar]
- Bolay, H.; Reuter, U.; Dunn, A.K.; Huang, Z.; Boas, D.A.; Moskowitz, M.A. Intrinsic brain activity triggers trigeminal meningeal afferents in a migraine model. Nat. Med. 2002, 8, 136–142. [Google Scholar]
- Kaube, H.; Goadsby, P.J. Anti-migraine compounds fail to modulate the propagation of cortical spreading depression in the cat. Eur. Neurol. 1994, 34, 30–35. [Google Scholar]
- Wiedemann, M.; de Lima, V.; Hanke, W. Effects of antimigraine drugs on retinal spreading depression. Naunyn Schmiedebergs Arch. Pharmacol. 1996, 353, 552–556. [Google Scholar] [PubMed]
- Shibata, M.; Leffler, C.W.; Busija, D.W. Pial arteriolar constriction following cortical spreading depression is mediated by prostanoids. Brain Res. 1992, 572, 190–197. [Google Scholar]
- Koistinaho, J.; Chan, P.H. Spreading depression-induced cyclooxygenase-2 expression in the cortex. Neurochem. Res. 2000, 25, 645–651. [Google Scholar]
- Miettinen, S.; Fusco, F.R.; Yrjanheikki, J.; Keinanen, R.; Hirvonen, T.; Roivainen, R.; Narhi, M.; Hokfelt, T.; Koistinaho, J. Spreading depression and focal brain ischemia induce cyclooxygenase-2 in cortical neurons through N-methyl-D-aspartic acid-receptors and phospholipase A2. Proc. Natl. Acad. Sci. USA 1997, 94, 6500–6505. [Google Scholar]
- Yokota, C.; Inoue, H.; Kuge, Y.; Abumiya, T.; Tagaya, M.; Hasegawa, Y.; Ejima, N.; Tamaki, N.; Minematsu, K. Cyclooxygenase-2 expression associated with spreading depression in a primate model. J. Cereb. Blood Flow Metab. 2003, 23, 395–398. [Google Scholar]
- Bergström, S.; Carlson, L.A.; Ekelund, L.G.; Orö, L. Cardiovascular and metabolic response to infusions of prostaglandin E1 and to simultaneous infusions of noradrenaline and prostaglandin E1 in man. Prostaglandin and related factors 35. Acta Physiol. Scand. 1965, 64, 332–339. [Google Scholar] [CrossRef] [PubMed]
- Carlson, L.A.; Ekelund, L.G.; Orö, L. Clinical and metabolic effects of different doses of prostaglandin E1 in man. Prostaglandin and related factors. Acta Med. Scand. 1968, 183, 423–430. [Google Scholar] [PubMed]
- Fitzgerald, G.A.; Friedman, L.A.; Miyamori, I.; Ogrady, J.; Lewis, P.J. Double-blind placebo controlled crossover study of prostacyclin in man. Life Sci. 1979, 25, 665–672. [Google Scholar]
- Peatfield, R.C.; Gawel, M.J.; Rose, F.C. Effect of infused prostacyclin in migraine and cluster headache. Headache 1981, 21, 124–124. [Google Scholar]
- Wienecke, T.; Olesen, J.; Ashina, M. Prostaglandin I(2) (epoprostenol) triggers migraine-like attacks in migraineurs. Cephalalgia 2010, 30, 179–190. [Google Scholar]
- Gobel, H.; Ernst, M.; Jeschke, J.; Keil, R.; Weigle, L. Acetylsalicylic-acid activates antinociceptive brain-stem reflex activity in headache patients and in healthy-subjects. Pain 1992, 48, 187–195. [Google Scholar]
- Jurna, I.; Brune, K. Central effect of the nonsteroid antiinflammatory agents, indomethacin, ibuprofen, and diclofenac, determined in c-fiber-evoked activity in single neurons of the rat thalamus. Pain 1990, 41, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Rosslee, L.M.; Eadie, M.J.; Heazlewood, V.; Tyrer, J.H. The absorption of aspirin in migraine, and the effect of metoclopramide. Clin. Exp. Pharmacol. Physiol. 1983, 10, 669–669. [Google Scholar]
- Volans, G.N. Effect of metoclopramide on absorption of effervescent aspirin in migraine. Br. J. Clin. Pharmacol. 1975, 2, 57–63. [Google Scholar]
- Gillis, J.C.; Brogden, R.N. Ketorolac—A reappraisal of its pharmacodynamic and pharmacokinetic properties and therapeutic use in pain management. Drugs 1997, 53, 139–188. [Google Scholar]
- Henry, P.; Hiesseprovost, O.; Dillenschneider, A.; Ganry, H.; Insuasty, J. Efficacy and tolerance of effervescent aspirin metoclopramide association in the treatment of migraine attack. Presse Med. 1995, 24, 254–258. [Google Scholar]
- Kangasniemi, P.; Kaaja, R. Ketoprofen and ergotamine in acute migraine. J. Inter. Med. 1992, 231, 551–554. [Google Scholar]
- Larsen, B.H.; Christiansen, L.V.; Andersen, B.; Olesen, J. Randomized double-blind comparison of tolfenamic acid and paracetamol in migraine. Acta Neurol. Scand. 1990, 81, 464–467. [Google Scholar]
- Massiou, H.; Serrurier, D.; Lasserre, O.; Bousser, M.G. Effectiveness of oral diclofenac in the acute treatment of common migraine attacks—A double-blind-study vs. placebo. Cephalalgia 1991, 11, 59–63. [Google Scholar] [PubMed]
- Tfelthansen, P.; Henry, P.; Mulder, L.J.; Scheldewaert, R.G.; Schoenen, E.; Chazot, G. The effectiveness of combined oral lysine acetylsalicylate and metoclopramide compared with oral sumatriptan for migraine. Lancet 1995, 346, 923–926. [Google Scholar]
- Diener, H.C. Efficacy and safety of intravenous acetylsalicylic acid lysinate compared to subcutaneous sumatriptan and parenteral placebo in the acute treatment of migraine. A double-blind, double-dummy, randomized, multicenter, parallel group study. Cephalalgia 1999, 19, 581–588. [Google Scholar] [CrossRef] [PubMed]
- Tfelthansen, P.; Olesen, J. Effervescent metoclopramide and aspirin (Migravess) vs. effervescent aspirin or placebo for migraine attacks—A double-blind-study. Cephalalgia 1984, 4, 107–111. [Google Scholar] [PubMed]
- Lange, R.; Schwarz, J.A.; Hohn, M. Acetylsalicylic acid effervescent 1000 mg (Aspirin (R)) in acute migraine attacks; a multicentre, randomized, double-blind, single-dose, placebo-controlled parallel group study. Cephalalgia 2000, 20, 663–667. [Google Scholar] [PubMed]
- MacGregor, E.A.; Dowson, A.; Davies, P.T.G. Mouth-dispersible aspirin in the treatment of migraine: A placebo-controlled study. Headache 2002, 42, 249–255. [Google Scholar]
- Kirthi, V.; Derry, S.; Moore, R.A.; McQuay, H.J. Aspirin with or without an antiemetic for acute migraine headaches in adults. Cochrane Database Syst. Rev. 2010, 4, CD008041. [Google Scholar]
- Havankakanniainen, H. Treatment of acute migraine attack—Ibuprofen and placebo compared. Headache 1989, 29, 507–509. [Google Scholar]
- Kloster, R.; Nestvold, K.; Vilming, S.T. A double-blind-study of ibuprofen vs. placebo in the treatment of acute migraine attacks. Cephalalgia 1992, 12, 169–171. [Google Scholar]
- Sandrini, G.; Franchini, S.; Lanfranchi, S.; Granella, F.; Manzoni, G.C.; Nappi, G. Effectiveness of ibuprofen-arginine in the treatment of acute migraine attacks. Int. J. Clin. Pharm. Res. 1998, 18, 145–150. [Google Scholar]
- Kellstein, D.E.; Lipton, R.B.; Geetha, R.; Koronkiewicz, K.; Evans, F.T.; Stewart, W.F.; Wilkes, K.; Furey, S.A.; Subramanian, T.; Cooper, S.A. Evaluation of a novel solubilized formulation of ibuprofen in the treatment of migraine headache: A randomized, double-blind, placebo-controlled, dose-ranging study. Cephalalgia 2000, 20, 233–243. [Google Scholar] [CrossRef] [PubMed]
- Codispoti, J.R.; Prior, M.J.; Fu, M.; Harte, C.M.; Nelson, E.B. Efficacy of nonprescription doses of ibuprofen for treating migraine headache. A randomized controlled trial. Headache 2001, 41, 665–679. [Google Scholar] [CrossRef] [PubMed]
- Misra, U.K.; Kalita, J.; Yadav, R.K. Rizatriptan vs. ibuprofen in migraine: A randomised placebo-controlled trial. J. Headache Pain 2007, 8, 175–179. [Google Scholar] [CrossRef] [PubMed]
- Diener, H.C.; Bussone, G.; de Liano, H.; Eikermann, A.; Englert, R.; Floeter, T.; Gallai, V.; Gobel, H.; Hartung, E.; Jimenez, M.D.; Lange, R.; Manzoni, G.C.; Mueller-Schwefe, G.; Nappi, G.; Pinessi, L.; Prat, J.; Puca, F.M.; Titus, F.; Voelker, M. Placebo-controlled comparison of effervescent acetylsalicylic acid, sumatriptan and ibuprofen in the treatment of migraine attacks. Cephalalgia 2004, 24, 947–954. [Google Scholar]
- Hamalainen, M.L.; Hoppu, K.; Valkeila, E.; Santavuori, P. Ibuprofen or acetaminophen for the acute treatment of migraine in children: A double-blind, randomized, placebo-controlled, crossover study. Neurology 1997, 48, 103–107. [Google Scholar] [PubMed]
- Lewis, D.W.; Kellstein, D.; Dahl, G.; Burke, B.; Frank, L.M.; Toor, S.; Northam, R.S.; White, L.W.; Lawson, L. Children's ibuprofen suspension for the acute treatment of pediatric migraine. Headache 2002, 42, 780–786. [Google Scholar]
- Lipton, R.B.; Baggish, J.S.; Stewart, W.F.; Codispoti, J.R.; Fu, M. Efficacy and safety of acetaminophen in the treatment of migraine—Results of a randomized, double-blind, placebo-controlled, population-based study. Arch. Inter. Med. 2000, 160, 3486–3492. [Google Scholar] [CrossRef]
- Dexter, S.L.; Graham, A.N.; Johnston, E.S.; Ratcliffe, D.M.; Wilkinson, M.I.P. Double-blind controlled-study of paramax in the acute treatment of common and classical migraine. Br. J. Clin. Practice 1985, 39, 388–392. [Google Scholar]
- Boureau, F.; Joubert, J.M.; Lasserre, V.; Prum, B.; Delecoeuillerie, G. Double-blind comparison of an acetaminophen 400 mg codeine 25 mg combination vs. aspirin 1000 mg and placebo in acute migraine attack. Cephalalgia 1994, 14, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Leinisch, E.; Evers, S.; Kaempfe, N.; Kraemer, C.; Sostak, P.; Jurgens, T.; Straube, A.; May, A. Evaluation of the efficacy of intravenous acetaminophen in the treatment of acute migraine attacks: A double-blind, placebo-controlled parallel group multicenter study. Pain 2005, 117, 396–400. [Google Scholar]
- Hakkarainen, H.; Vapaatalo, H.; Gothoni, G.; Parantainen, J. Tolfenamic acid is as effective as ergotamine during migraine attacks. Lancet 1979, 2, 326–328. [Google Scholar]
- Myllyla, V.V.; Havanka, H.; Herrala, L.; Kangasniemi, P.; Rautakorpi, I.; Turkka, J.; Vapaatalo, H.; Eskerod, O. Tolfenamic acid rapid release versus sumatriptan in the acute treatment of migraine: Comparable effect in a double-blind, randomized, controlled, parallel-group study. Headache 1998, 38, 201–207. [Google Scholar] [PubMed]
- Tokola, R.A.; Kangasniemi, P.; Neuvonen, P.J.; Tokola, O. Tolfenamic acid, metoclopramide, caffeine and their combinations in the treatment of migraine attacks. Cephalalgia 1984, 4, 253–263. [Google Scholar] [PubMed]
- Hakkarainen, H.; Parantainen, J.; Gothoni, G.; Vapaatalo, H. Tolfenamic acid and caffeine—A useful combination in migraine. Cephalalgia 1982, 2, 173–177. [Google Scholar]
- Nestvold, K.; Kloster, R.; Partinen, M.; Sulkava, R. Treatment of acute migraine attack—Naproxen and placebo compared. Cephalalgia 1985, 5, 115–119. [Google Scholar]
- Andersson, P.G.; Hinge, H.H.; Johansen, O.; Andersen, C.U.; Lademann, A.; Gotzsche, P.C. Double-blind-study of naproxen vs. placebo in the treatment of acute migraine attacks. Cephalalgia 1989, 9, 29–32. [Google Scholar] [PubMed]
- Johnson, E.S.; Ratcliffe, D.M.; Wilkinson, M. Naproxen sodium in the treatment of migraine. Cephalalgia 1985, 5, 5–10. [Google Scholar]
- Brandes, J.L.; Kudrow, D.; Stark, S.R.; O'Carroll, C.P.; Adelman, J.U.; O'Donnell, F.J.; Alexander, W.J.; Spruill, S.E.; Barrett, P.S.; Lener, S.E. Sumatriptan-naproxen for acute treatment of migraine: A randomized trial. JAMA 2007, 297, 1443–1454. [Google Scholar]
- Landy, S.; Freitag, F.; Smith, T.; Burch, S.; White, J.D.; Runken, M.C. Assessing workplace productivity and activity time in migraineurs treated with a single-tablet of sumatriptan formulated with RT technology (TM) and naproxen sodium (SumaRT/NAP) or placebo. Neurology 2007, 68, A265. [Google Scholar]
- Smith, T.R.; Sunshine, A.; Stark, S.R.; Littlefield, D.E.; Spruill, S.E.; Alexander, W.J. Sumatriptan and naproxen sodium for the acute treatment of migraine. Headache 2005, 45, 983–991. [Google Scholar]
- Bussone, G.; Grazzi, L.; D'Amico, D.; Manzoni, C.; Granella, F.; Cortelli, P.; Pierangeli, G.; Canal, N.; Colombo, B.; Frediani, F.; Maltempo, C.; Peruzzi, E.; Edson, K.; Ezzet, N.; Huels, J. Acute treatment of migraine attacks: Efficacy and safety of a nonsteroidal anti-inflammatory drug, diclofenac-potassium, in comparison to oral sumatriptan and placebo. Cephalalgia 1999, 19, 232–240. [Google Scholar] [PubMed]
- Dahlof, C.; Bjorkman, R. Diclofenac-K (50 and 100 mg) and placebo in the acute treatment of migraine. Cephalalgia 1993, 13, 117–123. [Google Scholar]
- McNeely, W.; Goa, K.L. Diclofenac-potassium in migraine—A review. Drugs 1999, 57, 991–1003. [Google Scholar]
- Peroutka, S.J.; Lyon, J.A.; Swarbrick, J.; Lipton, R.B.; Kolodner, K.; Goldstein, J. Efficacy of diclofenac sodium softgel 100 mg with or without caffeine 100 mg in migraine without aura: A randomized, double-blind, crossover study. Headache 2004, 44, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Delbene, E.; Poggioni, M.; Garagiola, U.; Maresca, V. Intramuscular treatment of migraine attacks using diclofenac sodium—A crossover clinical-trial. J. Inter. Med. Res. 1987, 15, 44–48. [Google Scholar]
- Schoenen, J.; De Klippel, N.; Giurgea, S.; Herroelen, L.; Jacquy, J.; Louis, P.; Monseu, G.; Vandenheede, M. Almotriptan and its combination with aceclofenac for migraine attacks: A study of efficacy and the influence of auto-evaluated brush allodynia. Cephalalgia 2008, 28, 1095–1105. [Google Scholar]
- Kinnunen, E.; Erkinjuntti, T.; Farkkila, M.; Palomaki, H.; Porras, J.; Teirmaa, H.; Freudenthal, Y.; Andersson, P. Placebo-controlled double-blind trial of pirprofen and an ergotamine tartrate compound in migraine attacks. Cephalalgia 1988, 8, 175–179. [Google Scholar]
- Awidi, A.S. Efficacy of flurbiprofen in the treatment of acute migraine attacks—A double-blind crossover study. Curr. Ther. Res. 1982, 32, 492–497. [Google Scholar]
- Dib, M.; Massiou, H.; Weber, M.; Henry, P.; Garcia-Acosta, S.; Bousser, M.G. Efficacy of oral ketoprofen in acute migraine—A double-blind randomized clinical trial. Neurology 2002, 58, 1660–1665. [Google Scholar]
- Karabetsos, A.; Karachalios, G.; Bourlinou, P.; Reppa, A.; Koutri, R.; Fotiadou, A. Ketoprofen vs. paracetamol in the treatment of acute migraine. Headache 1997, 37, 12–14. [Google Scholar] [PubMed]
- Silberstein, S.; Tepper, S.; Brandes, J.; Diamond, M.; Goldstein, J.; Winner, P.; Venkatraman, S.; Vrijens, F.; Malbecq, W.; Lines, C.; Visser, W.H.; Reines, S.; Yuen, E. Randomized, placebo-controlled trial of rofecoxib in the acute treatment of migraine. Neurology 2004, 62, 1552–1557. [Google Scholar]
- Hakkarainen, H.; Gustafsson, B.; Stockman, O. Comparative trial of ergotamine tartrate, acetyl salicylic-acid and a dextropropoxyphene compound in acute migraine attacks. Headache 1978, 18, 35–39. [Google Scholar]
- Hakkarainen, H.; Quiding, H.; Stockman, O. Mild analgesics as an alternative to ergotamine in migraine—A comparative trial with acetylsalicylic-acid, ergotamine tartrate, and a dextropropoxyphene compound. Eur. J. Clin. Pharmacol. 1980, 20, 590–595. [Google Scholar]
- Geraud, G.; Compagnon, A.; Rossi, A. Zolmitriptan vs. a combination of acetylsalicylic acid and metoclopramide in the acute oral treatment of migraine: A double-blind, randomised, three-attack study. Eur. Neurol. 2002, 47, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Pearce, I.; Frank, G.J.; Pearce, J.M.S. Ibuprofen compared with paracetamol in migraine. Practitioner 1983, 227, 465–467. [Google Scholar]
- Karachalios, G.N.; Fotiadou, A.; Chrisikos, N.; Karabetsos, A.; Kehagioglou, K. Treatment of acute migraine attack with diclofenac sodium—A double-blind-study. Headache 1992, 32, 98–100. [Google Scholar]
- Peatfield, R.C.; Petty, R.G.; Rose, F.C. Double-blind comparison of mefenamic-acid and acetaminophen (Paracetamol) in migraine. Cephalalgia 1983, 3, 129–134. [Google Scholar]
- Pradalier, A.; Rancurel, G.; Dordain, G.; Verdure, L.; Rascol, A.; Dry, J. Acute migraine attack therapy—Comparison of naproxen sodium and an ergotamine tartrate compound. Cephalalgia 1985, 5, 107–113. [Google Scholar]
- Treves, T.A.; Streiffler, M.; Korczyn, A.D. Naproxen sodium vs. ergotamine tartrate in the treatment of acute migraine attacks. Headache 1992, 32, 280–282. [Google Scholar] [CrossRef] [PubMed]
- Sargent, J.D.; Baumel, B.; Peters, K.; Diamond, S.; Saper, J.R.; Eisner, L.S.; Solbach, P. Aborting a migraine attack—Naproxen sodium v ergotamine plus caffeine. Headache 1988, 28, 263–266. [Google Scholar]
- Larkin, G.L.; Prescott, J.E. A randomized, double-blind, comparative-study of the efficacy of ketorolac tromethamine vs. meperidine in the treatment of severe migraine. Ann. Emerg. Med. 1992, 21, 919–924. [Google Scholar] [CrossRef] [PubMed]
- Davis, C.P.; Torre, P.R.; Williams, C.; Gray, C.; Barrett, K.; Krucke, G.; Peake, D.; Bass, B. Ketorolac vs. meperidine-plus-promethazine treatment of migraine headache—Evaluations by patients. Amer. J. Emerg. Med. 1995, 13, 146–150. [Google Scholar] [CrossRef]
- Shrestha, M.; Singh, R.; Moreden, J.; Hayes, J.E. Ketorolac vs. chlorpromazine in the treatment of acute migraine without aura—A prospective, randomized, double-blind trial. Arch. Intern. Med. 1996, 156, 1725–1728. [Google Scholar] [PubMed]
- Seim, M.B.; March, J.A.; Dunn, K.A. Intravenous ketorolac vs. intravenous prochlorperazine for the treatment of migraine headaches. Acad. Emerg. Med. 1998, 5, 573–576. [Google Scholar] [CrossRef] [PubMed]
- Meredith, J.T.; Wait, S.; Brewer, K.L. A prospective double-blind study of nasal sumatriptan vs. IV ketorolac in migraine. Amer. J. Emerg. Med. 2003, 21, 173–175. [Google Scholar] [CrossRef]
- Sandrini, G.; Cerbo, R.; Del Bene, E.; Ferrari, A.; Genco, S.; Grazioli, I.; Martelletti, P.; Nappi, G.; Pinessi, L.; Sarchielli, P.; Tamburro, P.; Uslenghi, C.; Zanchin, G. Efficacy of dosing and re-dosing of two oral fixed combinations of indomethacin, prochlorperazine and caffeine compared with oral sumatriptan in the acute treatment of multiple migraine attacks: A double-blind, double-dummy, randomised, parallel group, multicentre study. Int. J. Clin. Pract. 2007, 61, 1256–1269. [Google Scholar] [PubMed]
- Di Monda, V.; Nicolodi, M.; Aloisio, A.; Del Bianco, P.; Fonzari, M.; Grazioli, I.; Uslenghi, C.; Vecchiet, L.; Sicuteri, F. Efficacy of a fixed combination of indomethacin, prochlorperazine, and caffeine versus sumatriptan in acute treatment of multiple migraine attacks: A multicenter, randomized, crossover trial. Headache 2003, 43, 835–844. [Google Scholar] [CrossRef] [PubMed]
- Lanas, A.; Garcia-Rodriguez, L.A.; Arroyo, M.T.; Gomollon, F.; Feu, F.; Gonzalez-Perez, A.; Zapata, E.; Bastida, G.; Rodrigo, L.; Santolaria, S.; Guell, M.; de Argila, C.M.; Quintero, E.; Borda, F.; Pique, J.M. Risk of upper gastrointestinal ulcer bleeding associated with selective cyclo-oxygenase-2 inhibitors, traditional non-aspirin non-steroidal anti-inflammatory drugs, aspirin and combinations. Gut 2006, 55, 1731–1738. [Google Scholar] [PubMed]
- Lanas, A.; Garcia-Rodriguez, L.A.; Arroyo, M.T.; Gomollon, F.; Zapata, E.; Bujanda, L.; Feu, F.; Quintero, E.; Castro, M.; Santolaria, S. Coxibs, NSAIDs, aspirin, PPIs and the risks of upper GI bleeding in common clinical practice. Gastroenterology 2005, 128, A95. [Google Scholar]
- Pradalier, A.; Clapin, A.; Dry, J. Treatment review—Non-steroid anti-inflammatory drugs in the treatment and long-term prevention of migraine attacks. Headache 1988, 28, 550–557. [Google Scholar]
- Bombardier, C.; Laine, L.; Reicin, A.; Shapiro, D.; Burgos-Vargas, R.; Davis, B.; Day, R.; Ferraz, M.B.; Hawkey, C.J.; Hochberg, M.C.; Kvien, T.K.; Schnitzer, T.J. Comparison of upper gastrointestinal toxicity of rofecoxib and naproxen in patients with rheumatoid arthritis. VIGOR study group. N. Engl. J. Med. 2000, 343, 1520–1528. [Google Scholar] [PubMed]
- Konstam, M.A.; Weir, M.R.; Reicin, A.; Shapiro, D.; Sperling, R.S.; Barr, E.; Gertz, B.J. Cardiovascular thrombotic events in controlled, clinical trials of rofecoxib. Circulation 2001, 104, 2280–2288. [Google Scholar]
- Farkouh, M.E.; Greenberg, B.P. An evidence-based review of the cardiovascular risks of nonsteroidal anti-inflammatory drugs. Am. J. Cardiol. 2009, 103, 1227–1237. [Google Scholar]
- Bigal, M.E.; Serrano, D.; Buse, D.; Scher, A.; Stewart, W.F.; Lipton, R.B. Acute migraine medications and evolution from episodic to chronic migraine: A longitudinal population-based study. Headache 2008, 48, 1157–1168. [Google Scholar]
- Limmroth, V.; Katsarava, Z. Medication overuse headache. Curr. Opin. Neurol. 2004, 17, 301–306. [Google Scholar]
- Alahurula, V.; Myllyla, V.V.; Hokkanen, E.; Tokola, O. Tolfenamic acid and ergotamine abuse. Headache 1981, 21, 240–242. [Google Scholar]
- Mathew, N.T. Amelioration of ergotamine withdrawal symptoms with naproxen. Headache 1987, 27, 130–133. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an Open Access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pardutz, A.; Schoenen, J. NSAIDs in the Acute Treatment of Migraine: A Review of Clinical and Experimental Data. Pharmaceuticals 2010, 3, 1966-1987. https://doi.org/10.3390/ph3061966
Pardutz A, Schoenen J. NSAIDs in the Acute Treatment of Migraine: A Review of Clinical and Experimental Data. Pharmaceuticals. 2010; 3(6):1966-1987. https://doi.org/10.3390/ph3061966
Chicago/Turabian StylePardutz, Arpad, and Jean Schoenen. 2010. "NSAIDs in the Acute Treatment of Migraine: A Review of Clinical and Experimental Data" Pharmaceuticals 3, no. 6: 1966-1987. https://doi.org/10.3390/ph3061966
APA StylePardutz, A., & Schoenen, J. (2010). NSAIDs in the Acute Treatment of Migraine: A Review of Clinical and Experimental Data. Pharmaceuticals, 3(6), 1966-1987. https://doi.org/10.3390/ph3061966




