Pharmacovigilance Insights into Ibuprofen’s Neuropsychiatric Safety: A Retrospective Analysis of EudraVigilance Reports
Abstract
1. Introduction
2. Results
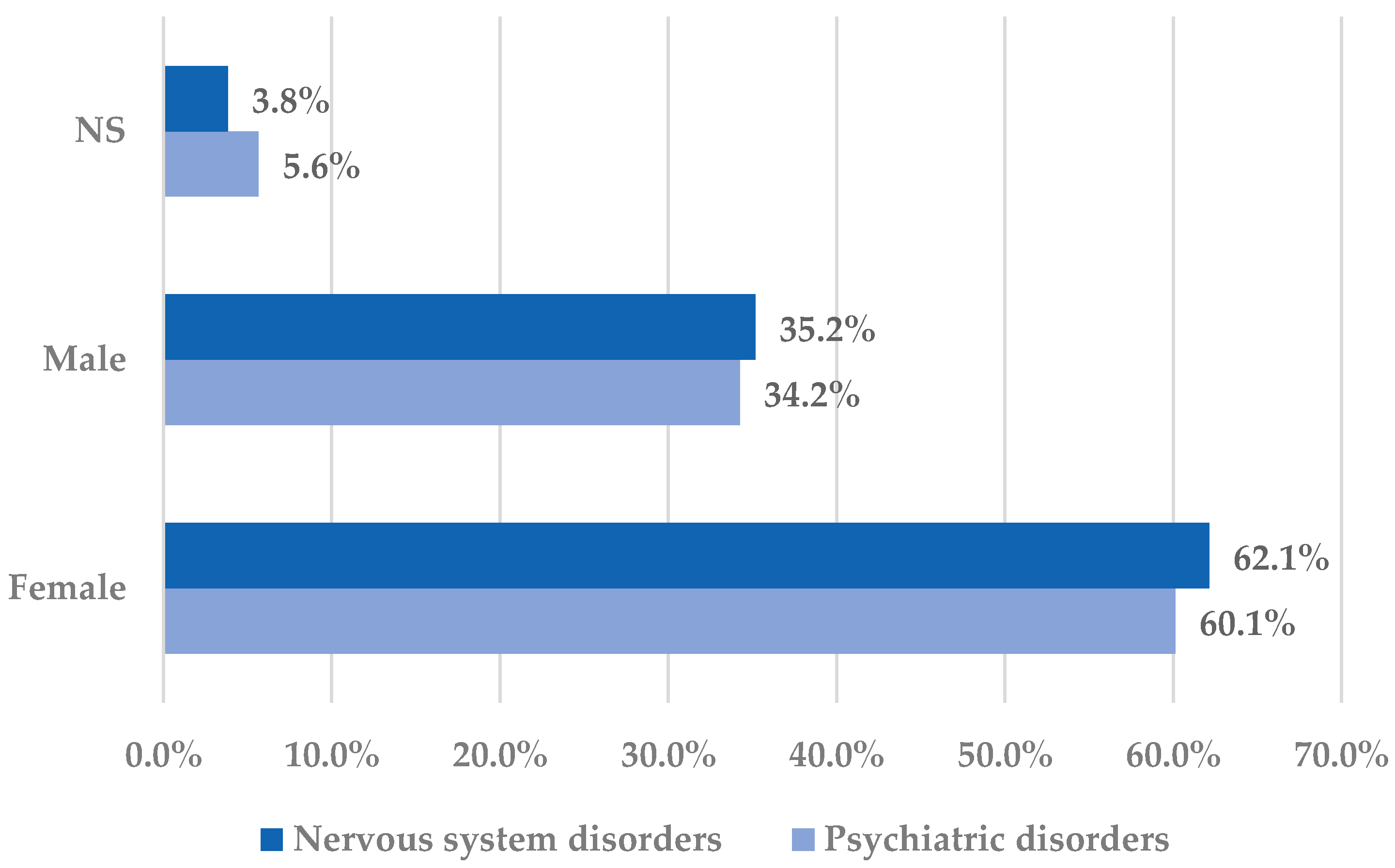
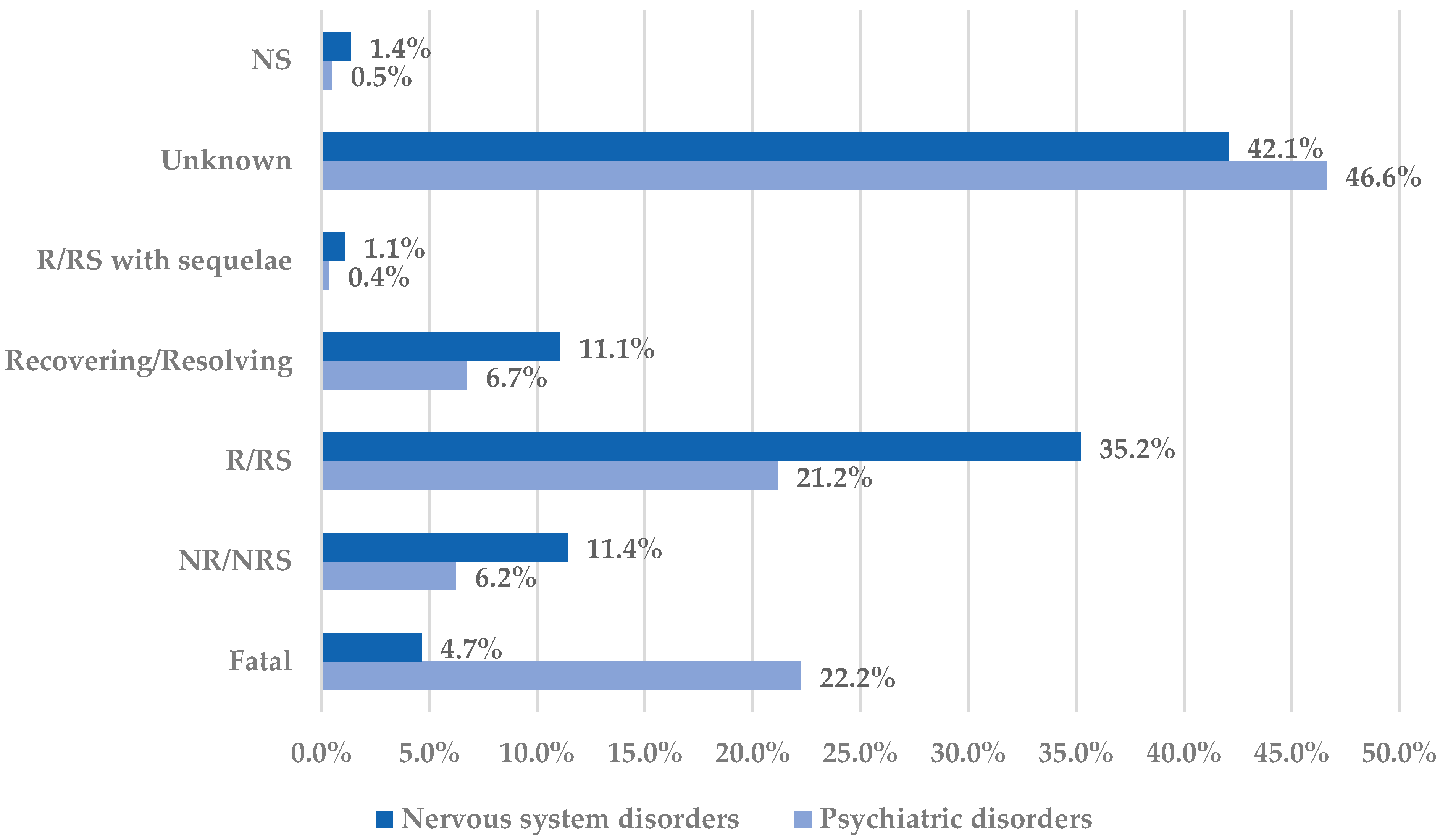
2.1. Descriptive Analysis
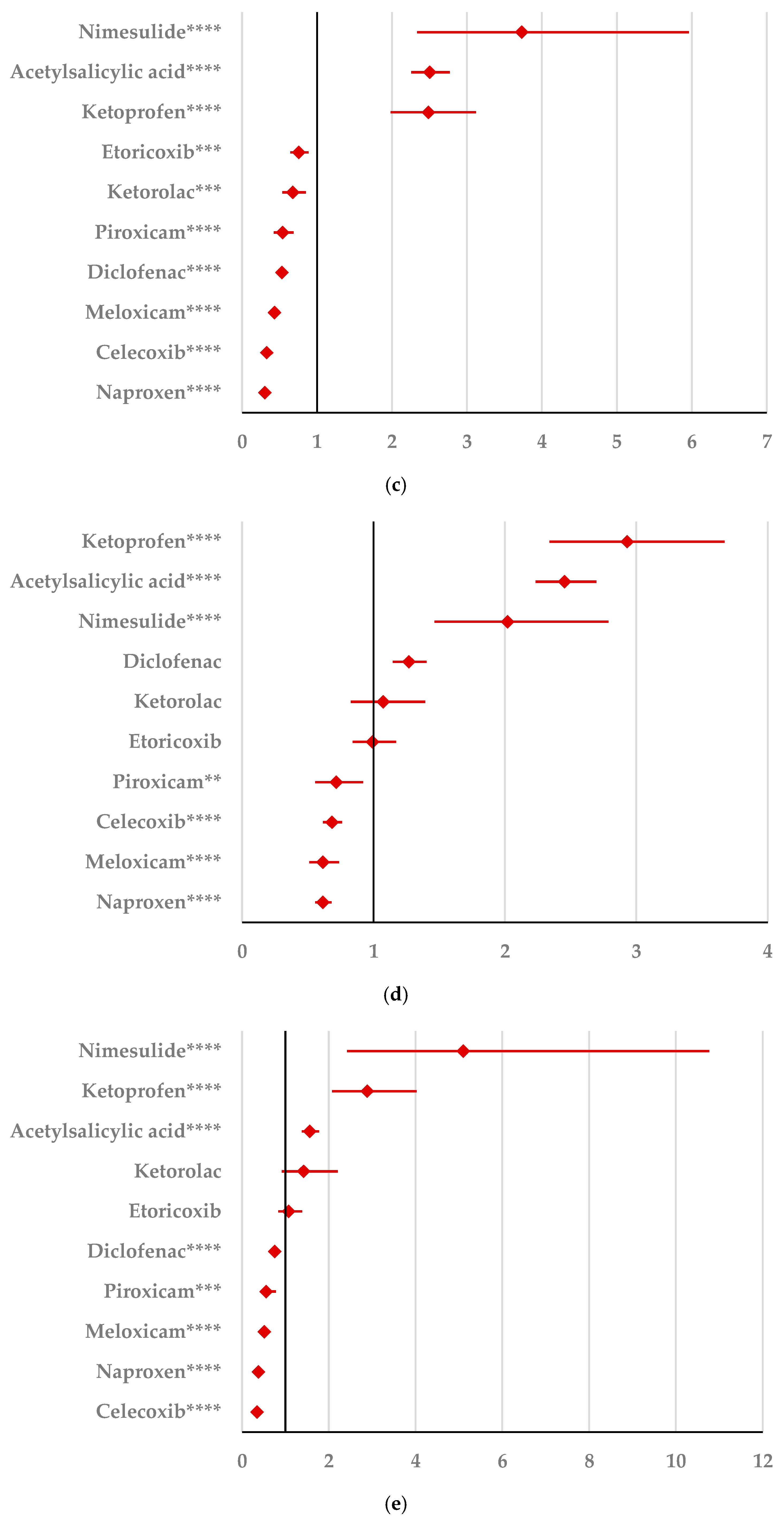
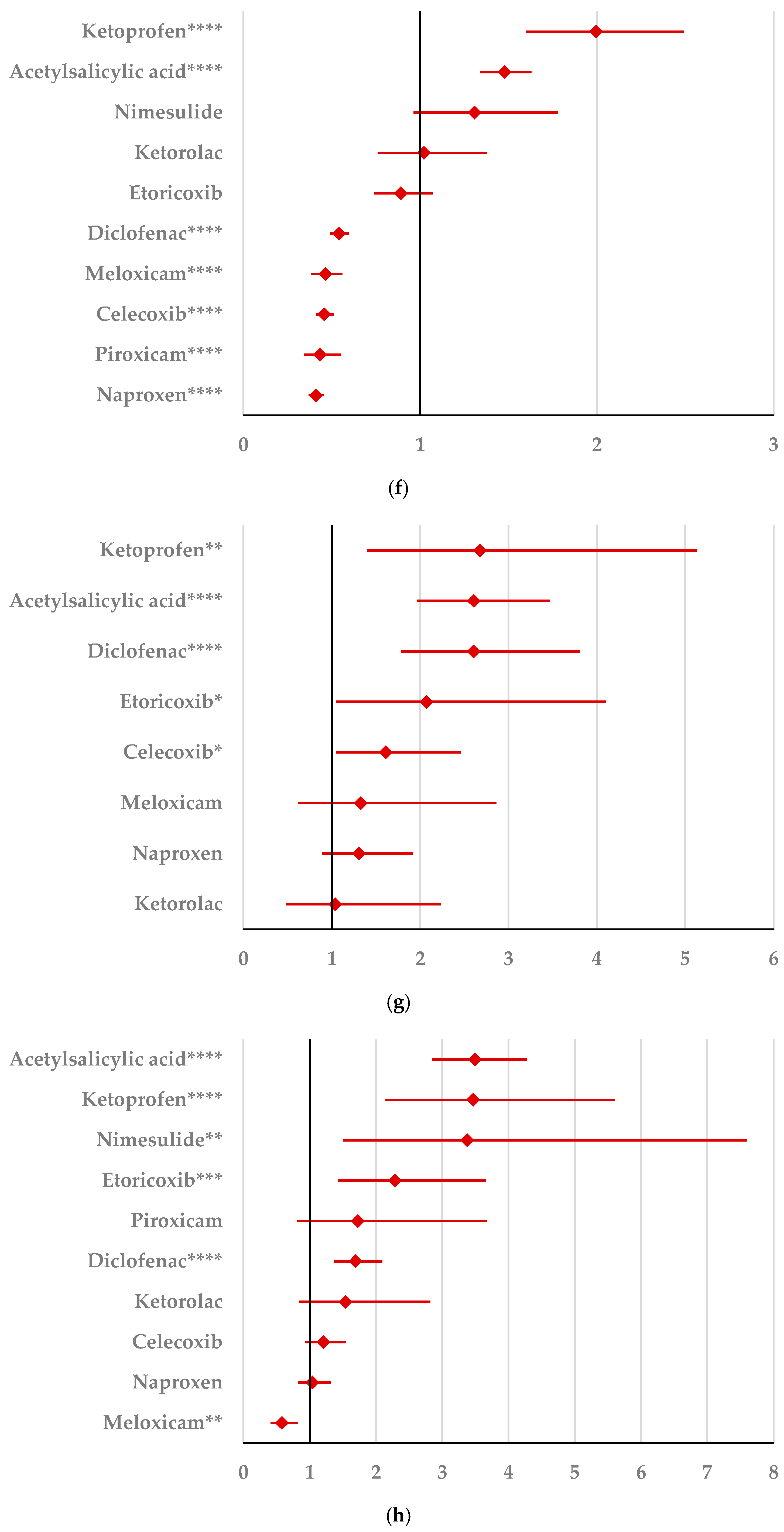
2.2. Disproportionality Analysis
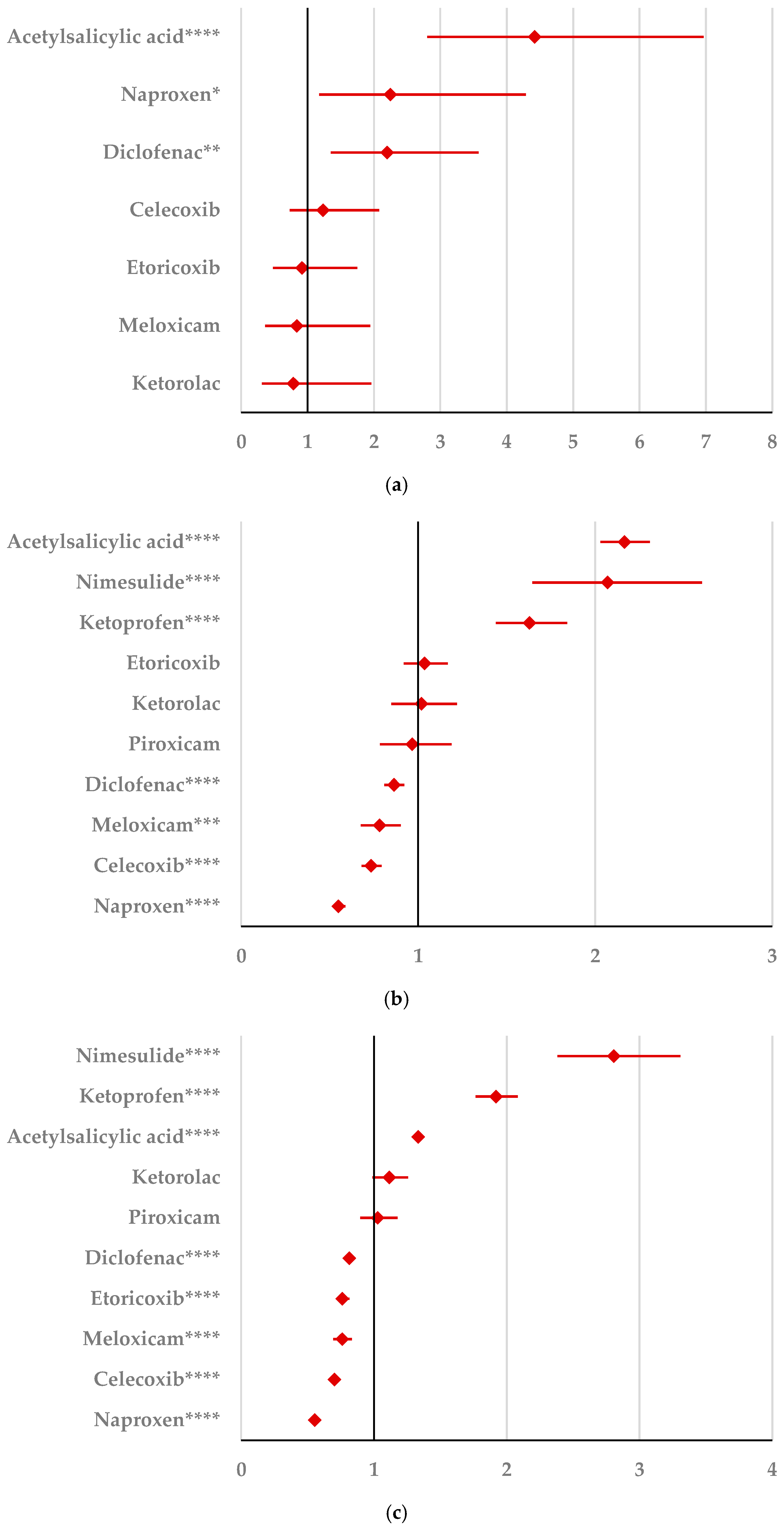
2.2.1. Psychiatric Disorders
2.2.2. Nervous System Disorders
3. Discussions
Strengths and Limitation of the Study
4. Materials and Methods
4.1. Study Design
4.2. Material
4.3. Descriptive and Disproportionality Analysis
4.4. Ethics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Varrassi, G.; Pergolizzi, J.V.; Dowling, P.; Paladini, A. Ibuprofen Safety at the Golden Anniversary: Are All NSAIDs the Same? A Narrative Review. Adv. Ther. 2020, 37, 61–82. [Google Scholar] [CrossRef] [PubMed]
- Rainsford, K.D. Ibuprofen: From Invention to an OTC Therapeutic Mainstay. Int. J. Clin. Pract. 2013, 67, 9–20. [Google Scholar] [CrossRef]
- Katz, J.N.; Smith, S.R.; Collins, J.E.; Solomon, D.H.; Jordan, J.M.; Hunter, D.J.; Suter, L.G.; Yelin, E.; Paltiel, A.D.; Losina, E. Cost-Effectiveness of Nonsteroidal Anti-Inflammatory Drugs and Opioids in the Treatment of Knee Osteoarthritis in Older Patients with Multiple Comorbidities. Osteoarthr. Cartil. 2016, 24, 409–418. [Google Scholar] [CrossRef] [PubMed]
- Tan, E.; Braithwaite, I.; Mckinlay, C.J.D.; Dalziel, S.R. Comparison of Acetaminophen (Paracetamol) with Ibuprofen for Treatment of Fever or Pain in Children Younger Than 2 Years: A Systematic Review and Meta-Analysis. JAMA Netw. Open 2020, 3, e2022398. [Google Scholar] [CrossRef]
- Chen, Z.; Li, S.; Choonara, I.; Zou, K.; Zeng, L.; Huang, L.; Jia, Z.J.; Cheng, G.; Jiang, Y.; Tang, Y.; et al. Access to Medicines for Children in China. BMJ Paediatr. Open 2022, 6, 1635. [Google Scholar] [CrossRef]
- Moore, R.A.; Derry, S.; Straube, S.; Ireson-Paine, J.; Wiffen, P.J. Faster, Higher, Stronger? Evidence for Formulation and Efficacy for Ibuprofen in Acute Pain. Pain 2014, 155, 14–21. [Google Scholar] [CrossRef]
- Agboola, A.A.; Nowak, A.; Duchnik, W.; Kucharski, Ł.; Story, A.; Story, G.; Struk, Ł.; Antosik, A.K.; Ossowicz-Rupniewska, P. Emulsion-Based Gel Loaded with Ibuprofen and Its Derivatives. Gels 2023, 9, 391. [Google Scholar] [CrossRef] [PubMed]
- Prasaja, B.; Harahap, Y.; Sandra, M.; Iskandar, I.; Lusthom, W.; Cahyaningsih, P. Rectal Administration of Ibuprofen: Comparison of Enema and Suppository Form. Drug Res. 2022, 72, 18–22. [Google Scholar] [CrossRef]
- Bookstaver, P.B.; Miller, A.D.; Rudisill, C.N.; Norris, L.B. Intravenous Ibuprofen: The First Injectable Product for the Treatment of Pain and Fever. J. Pain Res. 2010, 3, 67. [Google Scholar] [CrossRef]
- Pratama, D.E.; Hsieh, W.C.; Elmaamoun, A.; Lee, H.L.; Lee, T. Recovery of Active Pharmaceutical Ingredients from Unused Solid Dosage-Form Drugs. ACS Omega 2020, 5, 29147–29157. [Google Scholar] [CrossRef]
- Young, C.; Eggleston, W. Ibuprofen. Encycl. Toxicol. Fourth Ed. 2024, 5, 443–447. [Google Scholar] [CrossRef]
- Chang, R.W.; Tompkins, D.M.; Cohn, S.M. Are NSAIDs Safe? Assessing the Risk-Benefit Profile of Nonsteroidal Anti-Inflammatory Drug Use in Postoperative Pain Management. Am. Surg. 2021, 87, 872–879. [Google Scholar] [CrossRef]
- Yancy, C.W.; Jessup, M.; Bozkurt, B.; Butler, J.; Casey, D.E.; Drazner, M.H.; Fonarow, G.C.; Geraci, S.A.; Horwich, T.; Januzzi, J.L.; et al. 2013 ACCF/AHA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation 2013, 128, 16. [Google Scholar] [CrossRef]
- Lee, T.; Lu, N.; Felson, D.T.; Choi, H.K.; Dalal, D.S.; Zhang, Y.; Dubreuil, M. Use of Non-Steroidal Anti-Inflammatory Drugs Correlates with the Risk of Venous Thromboembolism in Knee Osteoarthritis Patients: A UK Population-Based Case-Control Study. Rheumatology 2016, 55, 1099–1105. [Google Scholar] [CrossRef]
- Ruschitzka, F.; Borer, J.S.; Krum, H.; Flammer, A.J.; Yeomans, N.D.; Libby, P.; Lüscher, T.F.; Solomon, D.H.; Husni, M.E.; Graham, D.Y.; et al. Differential Blood Pressure Effects of Ibuprofen, Naproxen, and Celecoxib in Patients with Arthritis: The PRECISION-ABPM (Prospective Randomized Evaluation of Celecoxib Integrated Safety Versus Ibuprofen or Naproxen Ambulatory Blood Pressure Measurement) Trial. Eur. Heart J. 2017, 38, 3282–3292. [Google Scholar] [CrossRef] [PubMed]
- Bally, M.; Dendukuri, N.; Rich, B.; Nadeau, L.; Helin-Salmivaara, A.; Garbe, E.; Brophy, J.M. Risk of Acute Myocardial Infarction with NSAIDs in Real World Use: Bayesian Meta-Analysis of Individual Patient Data. BMJ 2017, 357, j1909. [Google Scholar] [CrossRef] [PubMed]
- Zoubek, M.E.; Lucena, M.I.; Andrade, R.J.; Stephens, C. Systematic Review: Ibuprofen-Induced Liver Injury. Aliment. Pharmacol. Ther. 2020, 51, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Kowalski, M.L.; Asero, R.; Bavbek, S.; Blanca, M.; Blanca-Lopez, N.; Bochenek, G.; Brockow, K.; Campo, P.; Celik, G.; Cernadas, J.; et al. Classification and Practical Approach to the Diagnosis and Management of Hypersensitivity to Nonsteroidal Anti-Inflammatory Drugs. Allergy Eur. J. Allergy Clin. Immunol. 2013, 68, 1219–1232. [Google Scholar] [CrossRef] [PubMed]
- Shao, Q.H.; Yin, X.D.; Liu, H.X.; Zhao, B.; Huang, J.Q.; Li, Z.L. Kidney Injury Following Ibuprofen and Acetaminophen: A Real-World Analysis of Post-Marketing Surveillance Data. Front. Pharmacol. 2021, 12, 750108. [Google Scholar] [CrossRef]
- Bahta, M.; Ogbaghebriel, A.; Russom, M.; Tesfamariam, E.H.; Berhe, T. Impact of Adverse Reactions to First-Generation Antipsychotics on Treatment Adherence in Outpatients with Schizophrenia: A Cross-Sectional Study. Ann. Gen. Psychiatry 2021, 20, 27. [Google Scholar] [CrossRef]
- Pereira Pires, S.A.; Lemos, A.P.; Nunes Pereira, E.P.M.; da Silva Vilar Maia, P.A.; de Sousa, J.P. Ibuprofen-Induced Aseptic Meningitis: A Case Report. Rev. Paul. De Pediatr. 2019, 37, 382–385. [Google Scholar] [CrossRef]
- Finsterer, J. Transient Tunnel Vision and Altered Consciousness after a Single Dose of Ibuprofen. J. Int. Med. Res. 2022, 50, 03000605221126660. [Google Scholar] [CrossRef]
- Auriel, E.; Regev, K.; Korczyn, A.D. Nonsteroidal Anti-Inflammatory Drugs Exposure and the Central Nervous System. Handb. Clin. Neurol. 2014, 119, 577–584. [Google Scholar] [CrossRef]
- Murphy, P.J.; Badia, P.; Myers, B.L.; Boecker, M.R.; Wright, K.P. Nonsteroidal Anti-Inflammatory Drugs Affect Normal Sleep Patterns in Humans. Physiol. Behav. 1994, 55, 1063–1066. [Google Scholar] [CrossRef] [PubMed]
- Browning, C.H. Nonsteroidal Anti-Inflammatory Drugs and Severe Psychiatric Side Effects. Int. J. Psychiatry Med. 1996, 26, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Knights, K.M.; Mangoni, A.A.; Miners, J.O. Defining the COX Inhibitor Selectivity of NSAIDs: Implications for Understanding Toxicity. Expert. Rev. Clin. Pharmacol. 2010, 3, 769–776. [Google Scholar] [CrossRef] [PubMed]
- Saad, J.; Pellegrini, M.V. Nonsteroidal Anti-Inflammatory Drugs Toxicity; StatPearls: Treasure Island, FL, USA, 2023. [Google Scholar]
- Clark, D.W.J.; Ghose, K. Neuropsychiatric Reactions to Nonsteroidal Anti-Inflammatory Drugs (NSAIDs): The New Zealand Experience. Drug Saf. 1992, 7, 460–465. [Google Scholar] [CrossRef]
- Hoppmann, R.A.; Peden, J.G.; Ober, S.K. Central Nervous System Side Effects of Nonsteroidal Anti-Inflammatory Drugs: Aseptic Meningitis, Psychosis, and Cognitive Dysfunction. Arch. Intern. Med. 1991, 151, 1309–1313. [Google Scholar] [CrossRef]
- Rainsford, K.D. Ibuprofen: Pharmacology, Efficacy and Safety. Inflammopharmacology 2009, 17, 275–342. [Google Scholar] [CrossRef]
- Lucas, S.; Ailani, J.; Smith, T.R.; Abdrabboh, A.; Xue, F.; Navetta, M.S. Pharmacovigilance: Reporting Requirements throughout a Product’s Lifecycle. Ther. Adv. Drug Saf. 2022, 13, 20420986221125010. [Google Scholar] [CrossRef]
- European Medicines Agency. EudraVigilance—European Database of Suspected Adverse Drug Reaction Reports; European Medicines Agency: Amsterdam, The Netherlands, 2021. [Google Scholar]
- Coca, J.R.; Coca-Asensio, R.; Esteban Bueno, G. Socio-Historical Analysis of the Social Importance of Pharmacovigilance. Front. Sociol. 2022, 7, 974090. [Google Scholar] [CrossRef]
- Khan, M.A.A.; Sara, T.; Babar, Z.U.D. Pharmacovigilance: The Evolution of Drug Safety Monitoring. J. Pharm. Policy Pract. 2024, 17, 2417399. [Google Scholar] [CrossRef]
- Alomar, M.; Palaian, S.; Al-tabakha, M.M. Pharmacovigilance in Perspective: Drug Withdrawals, Data Mining and Policy Implications. F1000Research 2019, 8, 2109. [Google Scholar] [CrossRef]
- Serious Adverse Reaction|European Medicines Agency (EMA). Available online: https://www.ema.europa.eu/en/glossary-terms/serious-adverse-reaction (accessed on 20 July 2025).
- Botea, M.; Bedreag, O.; Dejeu, G.; Maghiar, O. Improving Perisurgical Pain Control: Ten Mistakes to Be Avoided. Eur. J. Anaesthesiol. 2020, 37, 251–253. [Google Scholar] [CrossRef]
- Muralidharan, A.; Smith, M.T. Pain, Analgesia and Genetics. J. Pharm. Pharmacol. 2011, 63, 1387–1400. [Google Scholar] [CrossRef]
- Grezenko, H.; Ekhator, C.; Nwabugwu, N.U.; Ganga, H.; Affaf, M.; Abdelaziz, A.M.; Rehman, A.; Shehryar, A.; Abbasi, F.A.; Bellegarde, S.B.; et al. Epigenetics in Neurological and Psychiatric Disorders: A Comprehensive Review of Current Understanding and Future Perspectives. Cureus 2023, 15, e43960. [Google Scholar] [CrossRef]
- Onder, G.; Pellicciotti, F.; Gambassi, G.; Bernabei, R. NSAID-Related Psychiatric Adverse Events: Who Is at Risk? Drugs 2004, 64, 2619–2627. [Google Scholar] [CrossRef] [PubMed]
- Le, T.T.; Kuplicki, R.; Yeh, H.W.; Aupperle, R.L.; Khalsa, S.S.; Simmons, W.K.; Paulus, M.P. Effect of Ibuprofen on BrainAGE: A Randomized, Placebo-Controlled, Dose-Response Exploratory Study. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 2018, 3, 836–843. [Google Scholar] [CrossRef] [PubMed]
- Farkouh, A.; Riedl, T.; Gottardi, R.; Czejka, M.; Kautzky-Willer, A. Sex-Related Differences in Pharmacokinetics and Pharmacodynamics of Frequently Prescribed Drugs: A Review of the Literature. Adv. Ther. 2020, 37, 644–655. [Google Scholar] [CrossRef] [PubMed]
- Roalsø, E.S.; Klonteig, S.; Kraft, B.; Skarstein, S.; Aalberg, M.; Jonassen, R. Associations between Over-the-Counter Analgesics Usage and Symptoms of Anxiety and Depression in Adolescents: A Network Analysis. BMC Psychiatry 2024, 24, 366. [Google Scholar] [CrossRef]
- Fischer, M.A.; Jan, A. Medication-Overuse Headache; StatPearls: Treasure Island, FL, USA, 2023. [Google Scholar]
- Stopping the Vicious Cycle of Rebound Headaches—Harvard Health. Available online: https://www.health.harvard.edu/blog/stopping-the-vicious-cycle-of-rebound-headaches-2019110718180 (accessed on 18 June 2025).
- Lehrer, S.; Rheinstein, P.H. Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) Reduce Suicidal Ideation and Depression. Discov. Med. 2019, 28, 205–212. [Google Scholar] [PubMed]
- Berk, M.; Dean, O.; Drexhage, H.; McNeil, J.J.; Moylan, S.; O’Neil, A.; Davey, C.G.; Sanna, L.; Maes, M. Aspirin: A Review of Its Neurobiological Properties and Therapeutic Potential for Mental Illness. BMC Med. 2013, 11, 74. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.L.; Xiang, D.C.; Li, Y.Y.; Ge, M.L.; Deng, A.P. An Exploratory Study Evaluating the 20 Medications Most Commonly Associated with Suicidal Ideation and Self-Injurious Behavior in the FAERS Database. BMC Pharmacol. Toxicol. 2025, 26, 24. [Google Scholar] [CrossRef] [PubMed]
- Wood, D.M.; Monaghan, J.; Streete, P.; Jones, A.L.; Ivor Dargan, P. Fatality after Deliberate Ingestion of Sustained-Release Ibuprofen: A Case Report. Crit. Care 2006, 10, R44. [Google Scholar] [CrossRef]
- Easley, R.B.; Altemeier, W.A. Central Nervous System Manifestations of an Ibuprofen Overdose Reversed by Naloxone. Pediatr. Emerg. Care 2000, 16, 39–41. [Google Scholar] [CrossRef]
- Hall, A.H.; Smolinske, S.C.; Conrad, F.L.; Wruk, K.M.; Kulig, K.W.; Dwelle, T.L.; Rumack, B.H. Ibuprofen Overdose: 126 Cases. Ann. Emerg. Med. 1986, 15, 1308–1313. [Google Scholar] [CrossRef]
- Ibuprofen Tablets, USP (400 Mg, 600 Mg and 800 Mg) Rx Only. Available online: https://dailymed.nlm.nih.gov/dailymed/fda/fdaDrugXsl.cfm?setid=17577b3b-e755-45d1-8b53-775d058578b2&type=display (accessed on 31 July 2025).
- PRAC Recommends Updating Advice on Use of High-Dose Ibuprofen|European Medicines Agency (EMA). Available online: https://www.ema.europa.eu/en/news/prac-recommends-updating-advice-use-high-dose-ibuprofen (accessed on 31 July 2025).
- Jiang, H.K.; Chang, D.M. Non-Steroidal Anti-Inflammatory Drugs with Adverse Psychiatric Reactions: Five Case Reports. Clin. Rheumatol. 1999, 18, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Köhler, O.; Petersen, L.; Mors, O.; Gasse, C. Inflammation and Depression: Combined Use of Selective Serotonin Reuptake Inhibitors and NSAIDs or Paracetamol and Psychiatric Outcomes. Brain Behav. 2015, 5, e00338. [Google Scholar] [CrossRef]
- Kapulsky, L.; Christos, P.; Ilagan, J.; Kocsis, J. The Effects of Ibuprofen Consumption on the Incidence of Postpartum Depression. Clin. Neuropharmacol. 2021, 44, 117–122. [Google Scholar] [CrossRef]
- NSAIDS Reduce Risk of Postoperative Delirium—Surgery International. Available online: https://surgery.international/nsaids-reduce-risk-of-postoperative-delirium/ (accessed on 18 June 2025).
- Gao, Z.; Zhang, J.; Nie, X.; Cui, X. Effectiveness of Intravenous Ibuprofen on Emergence Agitation in Children Undergoing Tonsillectomy with Propofol and Remifentanil Anesthesia: A Randomized Controlled Trial. J. Pain Res. 2022, 15, 1401–1410. [Google Scholar] [CrossRef] [PubMed]
- Gengo, F. Effects of Ibuprofen on Sleep Quality as Measured Using Polysomnography and Subjective Measures in Healthy Adults. Clin. Ther. 2006, 28, 1820–1826. [Google Scholar] [CrossRef]
- Koonalintip, P.; Phillips, K.; Wakerley, B.R. Medication-Overuse Headache: Update on Management. Life 2024, 14, 1146. [Google Scholar] [CrossRef]
- Radu, B.M.; Epureanu, F.B.; Radu, M.; Fabene, P.F.; Bertini, G. Nonsteroidal Anti-Inflammatory Drugs in Clinical and Experimental Epilepsy. Epilepsy Res. 2017, 131, 15–27. [Google Scholar] [CrossRef]
- Rainsford, K.D. Ibuprofen in Prevention of Neurodegenerative Diseases. In Ibuprofen: Discovery, Development and Therapeutics; Wiley Online Library: Hoboken, NJ, USA, 2015; pp. 547–570. [Google Scholar] [CrossRef]
- Leoutsakos, J.M.S.; Muthen, B.O.; Breitner, J.C.S.; Lyketsos, C.G. Effects of Non-Steroidal Anti-Inflammatory Drug Treatments on Cognitive Decline Vary by Phase of Pre-Clinical Alzheimer Disease: Findings from the Randomized Controlled Alzheimer’s Disease Anti-Inflammatory Prevention Trial. Int. J. Geriatr. Psychiatry 2012, 27, 364–374. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency. European Medicines Agency Policy on Access to EudraVigilance Data for Medicinal Products for Human Use (EudraVigilance Access Policy); European Medicines Agency: Amsterdam, The Netherlands, 2025. [Google Scholar]
- Medicines Agency, European. Guideline on Good Pharmacovigilance Practices (GVP)—Module VI—Collection, Management and Submission of Reports of Suspected Adverse Reactions to Medicinal Products (Rev 2); European Medicines Agency: London, UK, 2017. [Google Scholar]
- Popa Ilie, I.R.; Dobrea, C.M.; Butuca, A.; Homorodean, C.; Morgovan, C.; Vonica-Tincu, A.L.; Gligor, F.G.; Ghibu, S.; Frum, A. Real-Life Data on the Safety of Pasireotide in Acromegaly: Insights from EudraVigilance. Pharmaceuticals 2024, 17, 1631. [Google Scholar] [CrossRef] [PubMed]
- English|MedDRA. Available online: https://www.meddra.org/how-to-use/support-documentation/english/welcome (accessed on 20 July 2025).
- MedDRA Hierarchy|MedDRA. Available online: https://www.meddra.org/how-to-use/basics/hierarchy (accessed on 20 July 2025).
- MedDRA. Introductory Guide MedDRA Version 24.1 Notice to Reader MedDRA Introductory Guide; MedDRA: McLean, VA, USA, 2021. [Google Scholar]
- Fusaroli, M.; Salvo, F.; Begaud, B.; AlShammari, T.M.; Bate, A.; Battini, V.; Brueckner, A.; Candore, G.; Carnovale, C.; Crisafulli, S.; et al. The Reporting of a Disproportionality Analysis for Drug Safety Signal Detection Using Individual Case Safety Reports in PharmacoVigilance (READUS-PV): Development and Statement. Drug Saf. 2024, 47, 575. [Google Scholar] [CrossRef]
- Screening for Adverse Reactions in EudraVigilance. Available online: www.ema.europa.eu/contact (accessed on 2 January 2023).
- MedCalc Software Ltd. Odds Ratio Calculator, version 22.001; MedCalc Software Ltd.: Ostend, Belgium, 2025; Available online: https://www.medcalc.org/calc/odds_ratio.php (accessed on 14 May 2023).
- Vonica, R.C.; Morgovan, C.; Butuca, A.; Pumnea, M.; Cipaian, R.C.; Frum, A.; Dobrea, C.M.; Vonica-Tincu, A.L.; Pacnejer, A.M.; Batar, F.; et al. Real-World Evidence of Bevacizumab and Panitumumab Drug Resistance and Drug Ineffectiveness from EudraVigilance Database. Cancers 2025, 17, 663. [Google Scholar] [CrossRef] [PubMed]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buciuman, C.A.; Dobrea, C.M.; Butuca, A.; Frum, A.; Gligor, F.G.; Botea, M.O.; Vicaș, L.G.; Mureșan, M.E.; Gligor, O.; Maghiar, F.; et al. Pharmacovigilance Insights into Ibuprofen’s Neuropsychiatric Safety: A Retrospective Analysis of EudraVigilance Reports. Pharmaceuticals 2025, 18, 1301. https://doi.org/10.3390/ph18091301
Buciuman CA, Dobrea CM, Butuca A, Frum A, Gligor FG, Botea MO, Vicaș LG, Mureșan ME, Gligor O, Maghiar F, et al. Pharmacovigilance Insights into Ibuprofen’s Neuropsychiatric Safety: A Retrospective Analysis of EudraVigilance Reports. Pharmaceuticals. 2025; 18(9):1301. https://doi.org/10.3390/ph18091301
Chicago/Turabian StyleBuciuman, Cristina Anamaria, Carmen Maximiliana Dobrea, Anca Butuca, Adina Frum, Felicia Gabriela Gligor, Mihai O. Botea, Laura Grațiela Vicaș, Mariana Eugenia Mureșan, Octavia Gligor, Florin Maghiar, and et al. 2025. "Pharmacovigilance Insights into Ibuprofen’s Neuropsychiatric Safety: A Retrospective Analysis of EudraVigilance Reports" Pharmaceuticals 18, no. 9: 1301. https://doi.org/10.3390/ph18091301
APA StyleBuciuman, C. A., Dobrea, C. M., Butuca, A., Frum, A., Gligor, F. G., Botea, M. O., Vicaș, L. G., Mureșan, M. E., Gligor, O., Maghiar, F., Manole, A., & Morgovan, C. (2025). Pharmacovigilance Insights into Ibuprofen’s Neuropsychiatric Safety: A Retrospective Analysis of EudraVigilance Reports. Pharmaceuticals, 18(9), 1301. https://doi.org/10.3390/ph18091301









