Renaissance of Phoenix Drug Thalidomide—New Insights into Practical Clinical Application and Optimization Strategies for Managing Adverse Effects in Digestive Diseases
Abstract
1. Introduction
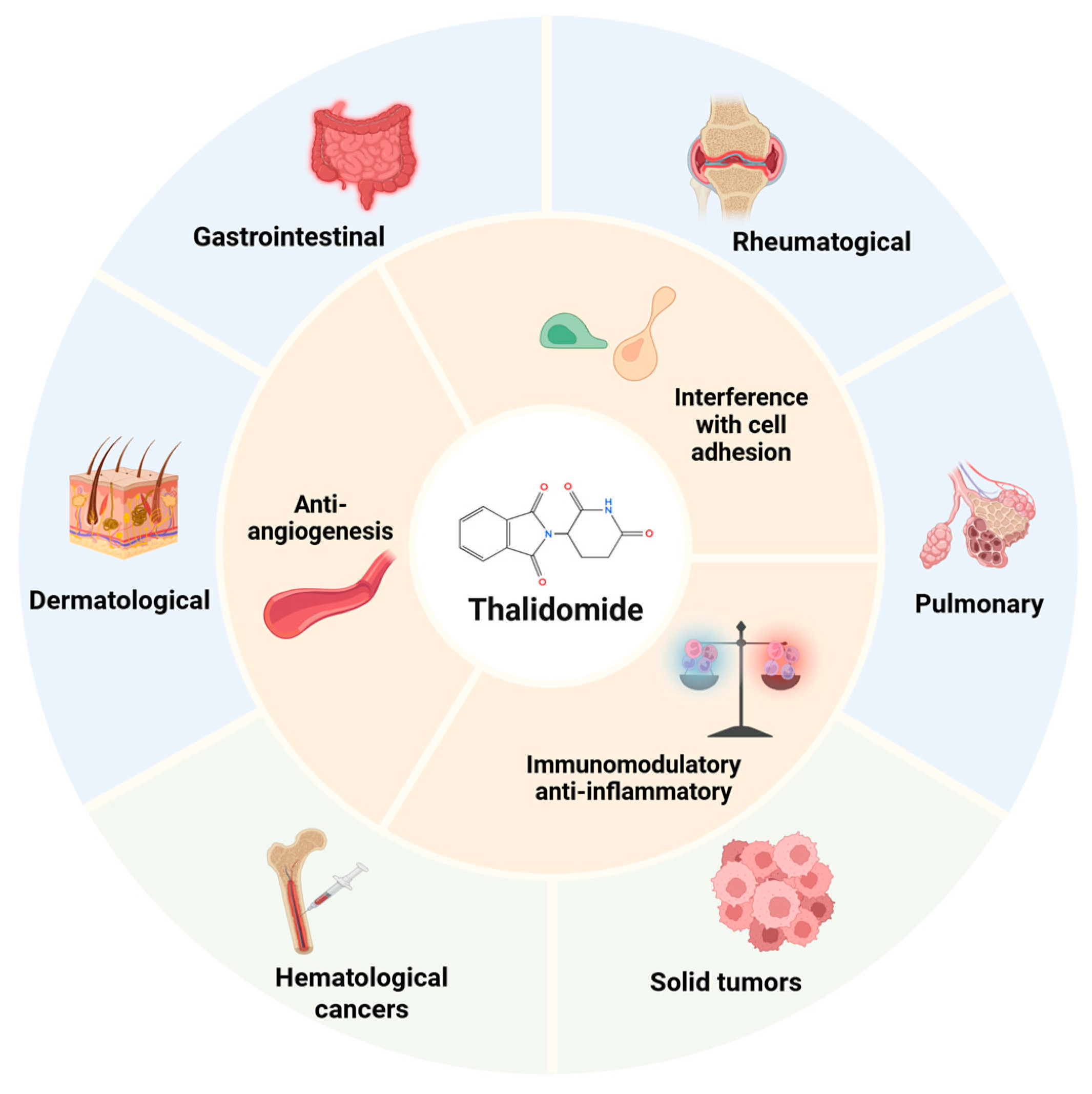
2. Mechanisms of Thalidomide
3. Thalidomide in Digestive Diseases: Clinical Applications and Limitations
3.1. Immune-Related Digestive Diseases
3.1.1. Intestinal Inflammatory Diseases
Ulcerative Colitis and Crohn’s Disease in Adults
Pediatric and Very-Early-Onset Inflammatory Bowel Disease
Intestinal Behcet’s Disease
3.1.2. IgG4-Related Digestive Diseases
3.1.3. Oral Mucosal Diseases
3.2. Non-Immune-Related Digestive Diseases
3.2.1. Gastrointestinal Bleeding
Gastrointestinal Bleeding Due to Angiodysplasia
Secondary Gastrointestinal Bleeding
3.2.2. Digestive Malignant Tumors
4. Strategies for the Clinical Use of Thalidomide
4.1. Alternative Therapy
4.2. Salvage Therapy
4.3. Combination Therapy
5. Adverse Effects of Thalidomide and Strategies to Mitigate Its Toxicity
5.1. Adverse Effects
5.2. Strategies for Optimizing Thalidomide Therapy and Reducing Its Toxicity
5.2.1. Dose and Risk Management of Thalidomide in Clinical Practice
5.2.2. Structural Modifications to Thalidomide to Reduce Toxicity
5.2.3. Targeted and Novel Delivery Systems
6. Strengths and Limitations
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| VEGF | vascular endothelial growth factor |
| GI | gastrointestinal |
| IBD | inflammatory bowel disease |
| BD | Behcet’s disease |
| IgG4 | Immunoglobulin G4 |
| TNF | tumor necrosis factor |
| IFN | interferon |
| IL | interleukin |
| CRBN | cereblon |
| UPS | ubiquitin–proteasome system |
| UC | ulcerative colitis |
| CD | Crohn’s disease |
| RCT | randomized controlled trial |
| allo-HSCT | allogeneic hematopoietic stem cell transplantation |
| VEOIBD | very-early-onset inflammatory bowel disease |
| EULAR | European League Against Rheumatism |
| MDS | myelodysplastic syndrome |
| GCs | glucocorticoids |
| GIAD | gastrointestinal angiodysplasia |
| SIA | small-intestinal angiodysplasia |
| TACE | transcatheter arterial chemoembolization |
| AZA | azathioprine |
| IFX | infliximab |
| FMT | fecal microbiota transplantation |
| DSS | dextran sulfate sodium salt |
| REMS | Risk Evaluation and Mitigation Strategy |
References
- D’Amato, R.J.; Loughnan, M.S.; Flynn, E.; Folkman, J. Thalidomide is an inhibitor of angiogenesis. Proc. Natl. Acad. Sci. USA 1994, 91, 4082–4085. [Google Scholar] [CrossRef]
- McBride, W.G. Thalidomide and Congenital Abnormalities. Lancet 1961, 278, 1358. [Google Scholar] [CrossRef]
- Burley, D.M.; Lenz, W. Thalidomide and Congenital Abnormalities. Lancet 1962, 279, 271–272. [Google Scholar] [CrossRef]
- Lewis, R.J., Sr. (Ed.) Sax’s Dangerous Properties of Industrial Materials; Wiley-Interscience, Wiley & Sons, Inc.: Hoboken, NJ, USA, 2004; p. 3430. [Google Scholar]
- Teo, S.K.; Resztak, K.E.; Scheffler, M.A.; Kook, K.A.; Zeldis, J.B.; Stirling, D.I.; Thomas, S.D. Thalidomide in the treatment of leprosy. Microbes Infect. 2002, 4, 1193–1202. [Google Scholar] [CrossRef] [PubMed]
- Dredge, K.; Marriott, J.B.; Macdonald, C.D.; Man, H.W.; Chen, R.; Muller, G.W.; Stirling, D.; Dalgleish, A.G. Novel thalidomide analogues display anti-angiogenic activity independently of immunomodulatory effects. Br. J. Cancer 2002, 87, 1166–1172. [Google Scholar] [CrossRef] [PubMed]
- Lebrin, F.; Srun, S.; Raymond, K.; Martin, S.; van den Brink, S.; Freitas, C.; Bréant, C.; Mathivet, T.; Larrivée, B.; Thomas, J.-L.; et al. Thalidomide stimulates vessel maturation and reduces epistaxis in individuals with hereditary hemorrhagic telangiectasia. Nat. Med. 2010, 16, 420–428. [Google Scholar] [CrossRef]
- Folkman, J.; Rogers, M.S. Thalidomide for multiple myeloma. N. Engl. J. Med. 2006, 354, 2389–2390. [Google Scholar]
- Mercurio, A.; Adriani, G.; Catalano, A.; Carocci, A.; Rao, L.; Lentini, G.; Cavalluzzi, M.M.; Franchini, C.; Vacca, A.; Corbo, F. A Mini-Review on Thalidomide: Chemistry, Mechanisms of Action, Therapeutic Potential and Anti-Angiogenic Properties in Multiple Myeloma. Curr. Med. Chem. 2017, 24, 2736–2744. [Google Scholar] [CrossRef]
- Eleutherakis-Papaiakovou, V.; Bamias, A.; Dimopoulos, M.A. Thalidomide in cancer medicine. Ann. Oncol. 2004, 15, 1151–1160. [Google Scholar] [CrossRef]
- Franks, M.E.; Macpherson, G.R.; Figg, W.D. Thalidomide. Lancet 2004, 363, 1802–1811. [Google Scholar] [CrossRef]
- Sherman, M.A.; Srinivasalu, H.; Kirkorian, A.Y.; Cardis, M.A. Clinical Images: Chronic, refractory childhood-onset cutaneous lupus erythematosus of the face responsive to lenalidomide. Arthritis Rheumatol. 2023, 75, 120. [Google Scholar] [CrossRef]
- Zhao, M.; Ma, L.; Duan, X.; Huo, Y.; Liu, S.; Zhao, C.; Zheng, Z.; Wang, Q.; Tian, X.; Chen, Y.; et al. Tofacitinib versus thalidomide for mucocutaneous lesions of systemic lupus erythematosus: A real-world CSTAR cohort study XXVII. Lupus 2024, 33, 1109–1115. [Google Scholar] [CrossRef]
- Jian, Y.; Wang, F.; Zhao, M.; Han, X.; Wang, X. Efficacy and safety of thalidomide for recurrent aphthous stomatitis: A systematic review and meta-analysis of randomized controlled trials. BMC Oral Health 2024, 24, 1149. [Google Scholar] [CrossRef] [PubMed]
- Hatemi, G.; Christensen, R.; Bang, D.; Bodaghi, B.; Celik, A.F.; Fortune, F.; Gaudric, J.; Gul, A.; Kötter, I.; Leccese, P.; et al. 2018 update of the EULAR recommendations for the management of Behçet’s syndrome. Ann. Rheum. Dis. 2018, 77, 808–818. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Rodríguez, O. Thalidomide. A promising new treatment for rheumatoid arthritis. Arthritis Rheum. 1984, 27, 1118–1121. [Google Scholar] [CrossRef]
- Sampaio, E.P.; Sarno, E.N.; Galilly, R.; Cohn, Z.A.; Kaplan, G. Thalidomide selectively inhibits tumor necrosis factor alpha production by stimulated human monocytes. J. Exp. Med. 1991, 173, 699–703. [Google Scholar] [CrossRef] [PubMed]
- Moreira, A.L.; Sampaio, E.P.; Zmuidzinas, A.; Frindt, P.; Smith, K.A.; Kaplan, G. Thalidomide exerts its inhibitory action on tumor necrosis factor alpha by enhancing mRNA degradation. J. Exp. Med. 1993, 177, 1675–1680. [Google Scholar] [CrossRef]
- McHugh, S.M.; Rifkin, I.R.; Deighton, J.; Wilson, A.B.; Lachmann, P.J.; Lockwood, C.M.; Ewan, P.W. The immunosuppressive drug thalidomide induces T helper cell type 2 (Th2) and concomitantly inhibits Th1 cytokine production in mitogen- and antigen-stimulated human peripheral blood mononuclear cell cultures. Clin. Exp. Immunol. 1995, 99, 160–167. [Google Scholar] [CrossRef]
- Moller, D.R.; Wysocka, M.; Greenlee, B.M.; Ma, X.; Wahl, L.; Flockhart, D.A.; Trinchieri, G.; Karp, C.L. Inhibition of IL-12 production by thalidomide. J. Immunol. 1997, 159, 5157–5161. [Google Scholar] [CrossRef]
- Keifer, J.A.; Guttridge, D.C.; Ashburner, B.P.; Baldwin, A.S., Jr. Inhibition of NF-kappa B activity by thalidomide through suppression of IkappaB kinase activity. J. Biol. Chem. 2001, 276, 22382–22387. [Google Scholar] [CrossRef]
- Rafiee, P.; Stein, D.J.; Nelson, V.M.; Otterson, M.F.; Shaker, R.; Binion, D.G. Thalidomide inhibits inflammatory and angiogenic activation of human intestinal microvascular endothelial cells (HIMEC). Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 298, G167–G176. [Google Scholar] [CrossRef]
- Yang, C.; Singh, P.; Singh, H.; Le, M.L.; El-Matary, W. Systematic review: Thalidomide and thalidomide analogues for treatment of inflammatory bowel disease. Aliment. Pharmacol. Ther. 2015, 41, 1079–1093. [Google Scholar] [CrossRef]
- Dsouza, N.N.; Alampady, V.; Baby, K.; Maity, S.; Byregowda, B.H.; Nayak, Y. Thalidomide interaction with inflammation in idiopathic pulmonary fibrosis. Inflammopharmacology 2023, 31, 1167–1182. [Google Scholar] [CrossRef]
- Cheng, J.; Jiang, J.; He, B.; Lin, W.J.; Li, Y.; Duan, J.; Li, H.; Huang, X.; Cai, J.; Xie, J.; et al. A phase 2 study of thalidomide for the treatment of radiation-induced blood-brain barrier injury. Sci. Transl. Med. 2023, 15, eabm6543. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Wu, S.; Tang, M.; Zhao, R.; Zhang, Q.; Dai, Z.; Gao, Y.; Yang, S.; Li, Z.; Du, Y.; et al. Thalidomide for Recurrent Bleeding due to Small-Intestinal Angiodysplasia. N. Engl. J. Med. 2023, 389, 1649–1659. [Google Scholar] [CrossRef] [PubMed]
- Monteonofrio, L.; Virdia, I.; Pozzi, S.; Quadri, R.; Amendolare, A.; Marzano, F.; Braile, M.; Sulfaro, V.; Paroni, M.; Tullo, A.; et al. Molecular mechanisms of thalidomide effectiveness on COVID-19 patients explained: ACE2 is a new ΔNp63α target gene. J. Mol. Med. 2024, 102, 1371–1380. [Google Scholar] [CrossRef]
- Ito, T.; Ando, H.; Suzuki, T.; Ogura, T.; Hotta, K.; Imamura, Y.; Yamaguchi, Y.; Handa, H. Identification of a primary target of thalidomide teratogenicity. Science 2010, 327, 1345–1350. [Google Scholar] [CrossRef]
- Fischer, E.S.; Böhm, K.; Lydeard, J.R.; Yang, H.; Stadler, M.B.; Cavadini, S.; Nagel, J.; Serluca, F.; Acker, V.; Lingaraju, G.M.; et al. Structure of the DDB1-CRBN E3 ubiquitin ligase in complex with thalidomide. Nature 2014, 512, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Waters, M.F.; Laing, A.B.; Ambikapathy, A.; Lennard-Jones, J.E. Treatment of ulcerative colitis with thalidomide. Br. Med. J. 1979, 1, 792. [Google Scholar] [CrossRef]
- He, Y.; Mao, R.; Chen, F.; Xu, P.P.; Chen, B.L.; Wu, Y.; Qiu, Y.; Zhang, S.H.; Feng, R.; Zeng, Z.R.; et al. Thalidomide induces clinical remission and mucosal healing in adults with active Crohn’s disease: A prospective open-label study. Ther. Adv. Gastroenterol. 2017, 10, 397–406. [Google Scholar] [CrossRef]
- Lazzerini, M.; Martelossi, S.; Magazzù, G.; Pellegrino, S.; Lucanto, M.C.; Barabino, A.; Calvi, A.; Arrigo, S.; Lionetti, P.; Lorusso, M.; et al. Effect of thalidomide on clinical remission in children and adolescents with refractory Crohn disease: A randomized clinical trial. JAMA 2013, 310, 2164–2173. [Google Scholar] [CrossRef]
- Lazzerini, M.; Martelossi, S.; Magazzù, G.; Pellegrino, S.; Lucanto, M.C.; Barabino, A.; Calvi, A.; Arrigo, S.; Lionetti, P.; Lorusso, M.; et al. Effect of Thalidomide on Clinical Remission in Children and Adolescents with Ulcerative Colitis Refractory to Other Immunosuppressives: Pilot Randomized Clinical Trial. Inflamm. Bowel Dis. 2015, 21, 1739–1749. [Google Scholar] [CrossRef] [PubMed]
- Sayarlioglu, M.; Kotan, M.C.; Topcu, N.; Bayram, I.; Arslanturk, H.; Gul, A. Treatment of recurrent perforating intestinal ulcers with thalidomide in Behçet’s disease. Ann. Pharmacother. 2004, 38, 808–811. [Google Scholar] [CrossRef] [PubMed]
- Yasui, K.; Uchida, N.; Akazawa, Y.; Nakamura, S.; Minami, I.; Amano, Y.; Yamazaki, T. Thalidomide for treatment of intestinal involvement of juvenile-onset Behçet disease. Inflamm. Bowel Dis. 2008, 14, 396–400. [Google Scholar] [CrossRef] [PubMed]
- Yasui, K.; Misawa, Y.; Shimizu, T.; Komiyama, A.; Kawakami, T.; Mizoguchi, M. Thalidomide therapy for juvenile-onset entero-Behçet disease. J. Pediatr. 2003, 143, 692–694. [Google Scholar] [CrossRef]
- Chen, Y.; Ye, C.; Yang, P.; Zhang, W.; Dai, L.; Wei, W.; Wu, R.; Ding, S.; Chen, L.; Wu, X.; et al. Thalidomide can effectively prevent relapse in IgG4-related disease outweighing its side effects: A multicentre, randomised, double-blinded, placebo-controlled study. Ann. Rheum. Dis. 2025, 84, 1246–1252. [Google Scholar] [CrossRef]
- Deng, Y.; Wei, W.; Wang, Y.; Pan, L.; Du, G.; Yao, H.; Tang, G. A Randomized controlled clinical trial on dose optimization of thalidomide in maintenance treatment for recurrent aphthous stomatitis. J. Oral Pathol. Med. 2022, 51, 106–112. [Google Scholar] [CrossRef]
- Liang, L.; Liu, Z.; Zhu, H.; Wang, H.; Wei, Y.; Ning, X.; Shi, Z.; Jiang, L.; Lin, Z.; Yan, H.; et al. Efficacy and safety of thalidomide in preventing oral mucositis in patients with nasopharyngeal carcinoma undergoing concurrent chemoradiotherapy: A multicenter, open-label, randomized controlled trial. Cancer 2022, 128, 1467–1474. [Google Scholar] [CrossRef]
- Ge, Z.Z.; Chen, H.M.; Gao, Y.J.; Liu, W.Z.; Xu, C.H.; Tan, H.H.; Chen, H.Y.; Wei, W.; Fang, J.Y.; Xiao, S.D. Efficacy of thalidomide for refractory gastrointestinal bleeding from vascular malformation. Gastroenterology 2011, 141, 1629–1637.e4. [Google Scholar] [CrossRef]
- Craanen, M.E.; van Triest, B.; Verheijen, R.H.; Mulder, C.J. Thalidomide in refractory haemorrhagic radiation induced proctitis. Gut 2006, 55, 1371–1372. [Google Scholar] [CrossRef]
- Heo, Y.; Park, H.S.; Shin, C.S.; Yoo, K.C.; Kim, D.; Lee, T. Successful Treatment of Life-Threatening Small Bowel Bleeding with Thalidomide After Living Donor Kidney Transplantation: A Case Report. Transplant. Proc. 2019, 51, 3092–3098. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Saenz, M.; Romero-Vazquez, J.; Caunedo-Alvarez, A.; Maldonado-Perez, B.; Gutierrez, J.M. Beneficial effects and reversion of vascular lesions by thalidomide in a patient with bleeding portal hypertensive enteropathy. Dig. Liver Dis. 2010, 42, 232–233. [Google Scholar] [CrossRef]
- Lv, J.; Liu, N.; Liu, K.W.; Ding, A.P.; Wang, H.; Qiu, W.S. A Randomised Controlled Phase II Trial of the Combination of XELOX with Thalidomide for the First-Line Treatment of Metastatic Colorectal Cancer. Cancer Biol. Med. 2012, 9, 111–114. [Google Scholar] [CrossRef]
- Zhong, N.; Yu, Y.; Yang, P. Effect of CAPEOX combined with thalidomide in the treatment of elderly patients with colon cancer: A single-center report. Asian J. Surg. 2024, 47, 3746–3747. [Google Scholar] [CrossRef]
- Cao, D.D.; Xu, H.L.; Liu, L.; Zheng, Y.F.; Gao, S.F.; Xu, X.M.; Ge, W. Thalidomide combined with transcatheter artierial chemoembolzation for primary hepatocellular carcinoma: A systematic review and meta-analysis. Oncotarget 2017, 8, 44976–44993. [Google Scholar] [CrossRef]
- Li, J.; Lv, B.; Song, L.; Zhang, X.; Zhang, J.; Ding, X.; Liu, Y.; Ye, T.; Guo, M.; Fang, T.; et al. Survival in patients with unresectable hepatocellular carcinoma: TCC cocktail plus TACE vs TACE alone prospective randomized clinical trial. J. Transl. Med. 2025, 23, 812. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Smith, P.J. Horizon scanning: New and future therapies in the management of inflammatory bowel disease. eGastroenterology 2023, 1, e100012. [Google Scholar] [CrossRef]
- Wettstein, A.R.; Meagher, A.P. Thalidomide in Crohn’s disease. Lancet 1997, 350, 1445–1446. [Google Scholar] [CrossRef]
- Vasiliauskas, E.A.; Kam, L.Y.; Abreu-Martin, M.T.; Hassard, P.V.; Papadakis, K.A.; Yang, H.; Zeldis, J.B.; Targan, S.R. An open-label pilot study of low-dose thalidomide in chronically active, steroid-dependent Crohn’s disease. Gastroenterology 1999, 117, 1278–1287. [Google Scholar] [CrossRef]
- Ehrenpreis, E.D.; Kane, S.V.; Cohen, L.B.; Cohen, R.D.; Hanauer, S.B. Thalidomide therapy for patients with refractory Crohn’s disease: An open-label trial. Gastroenterology 1999, 117, 1271–1277. [Google Scholar] [CrossRef] [PubMed]
- Bramuzzo, M.; Ventura, A.; Martelossi, S.; Lazzerini, M. Thalidomide for inflammatory bowel disease: Systematic review. Medicine 2016, 95, e4239. [Google Scholar] [CrossRef]
- Barkin, J.A.; Schonfeld, W.B.; Deshpande, A.R. Successful use of thalidomide for refractory esophageal Crohn’s disease. Am. J. Gastroenterol. 2013, 108, 855–857. [Google Scholar] [CrossRef]
- Ginsburg, P.M.; Hanan, I.; Ehrenpreis, E.D. Treatment of severe esophageal Crohn’s disease with thalidomide. Am. J. Gastroenterol. 2001, 96, 1305–1306. [Google Scholar] [CrossRef]
- Hegarty, A.; Hodgson, T.; Porter, S. Thalidomide for the treatment of recalcitrant oral Crohn’s disease and orofacial granulomatosis. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2003, 95, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Kolivras, A.; De Maubeuge, J.; André, J.; Song, M. Thalidomide in refractory vulvar ulcerations associated with Crohn’s disease. Dermatology 2003, 206, 381–383. [Google Scholar] [CrossRef]
- Weinstein, T.A.; Sciubba, J.J.; Levine, J. Thalidomide for the treatment of oral aphthous ulcers in Crohn’s disease. J. Pediatr. Gastroenterol. Nutr. 1999, 28, 214–216. [Google Scholar] [CrossRef]
- Phillips, F.; Verstockt, B.; Sladek, M.; de Boer, N.; Katsanos, K.; Karmiris, K.; Albshesh, A.; Erikson, C.; Bergemalm, D.; Molnar, T.; et al. Orofacial Granulomatosis Associated with Crohn’s Disease: A Multicentre Case Series. J. Crohn’s Colitis 2022, 16, 430–435. [Google Scholar] [CrossRef]
- Wagner, C.; Dachman, A.; Ehrenpreis, E.D. Mesenteric Panniculitis, Sclerosing Mesenteritis and Mesenteric Lipodystrophy: Descriptive Review of a Rare Condition. Clin. Colon Rectal Surg. 2022, 35, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Ginsburg, P.M.; Ehrenpreis, E.D. A pilot study of thalidomide for patients with symptomatic mesenteric panniculitis. Aliment. Pharmacol. Ther. 2002, 16, 2115–2122. [Google Scholar] [CrossRef] [PubMed]
- Simon, M.; Pariente, B.; Lambert, J.; Cosnes, J.; Bouhnik, Y.; Marteau, P.; Allez, M.; Colombel, J.F.; Gornet, J.M. Long-term Outcomes of Thalidomide Therapy for Adults with Refractory Crohn’s Disease. Clin. Gastroenterol. Hepatol. 2016, 14, 966–972.e962. [Google Scholar] [CrossRef]
- Xu, S.; Zu, X.M.; Feng, R.; Zhang, S.H.; Qiu, Y.; Chen, B.L.; Zeng, Z.R.; Chen, M.H.; He, Y. Thalidomide in refractory Crohn’s disease: Long-term efficacy and safety. Zhonghua Nei Ke Za Zhi 2020, 59, 445–450. [Google Scholar] [CrossRef]
- Peng, X.; Lin, Z.W.; Zhang, M.; Yao, J.Y.; Zhao, J.Z.; Hu, P.J.; Cao, Q.; Zhi, M. The efficacy and safety of thalidomide in the treatment of refractory Crohn’s disease in adults: A double-center, double-blind, randomized-controlled trial. Gastroenterol. Rep. 2022, 10, goac052. [Google Scholar] [CrossRef]
- Petruzzelli, M.G.; Margari, L.; Ivagnes, S.; Palumbi, R.; Margari, F. Early onset first-episode psychosis during treatment with thalidomide for refractory ulcerative colitis: A case report. J. Med. Case Rep. 2019, 13, 175. [Google Scholar] [CrossRef]
- Liu, M.; Lin, X.; Wang, L.; He, Y.; Chen, M.; Mao, R. Thalidomide-induced sinus bradycardia in Crohn’s disease: Case report and literature review. J. Int. Med. Res. 2019, 47, 2228–2233. [Google Scholar] [CrossRef]
- Sriskandarajah, P.; Scott, F.; Muthalali, S.; Mercieca, J. Life-threatening bowel perforation while on thalidomide-based triplet regimen for multiple myeloma: A retrospective case series. Br. J. Haematol. 2016, 173, 786–788. [Google Scholar] [CrossRef] [PubMed]
- Zitomersky, N.L.; Levine, A.E.; Atkinson, B.J.; Harney, K.M.; Verhave, M.; Bousvaros, A.; Lightdale, J.R.; Trenor, C.C., 3rd. Risk factors, morbidity, and treatment of thrombosis in children and young adults with active inflammatory bowel disease. J. Pediatr. Gastroenterol. Nutr. 2013, 57, 343–347. [Google Scholar] [CrossRef] [PubMed]
- Martín-Noguerol, E.; Martinez-Alcalá, C.; Gonzalez-Santiago, J.M.; Vara-Brenes, D.; Molina-Infante, J. High-dose thalidomide for severe idiopathic obscure gastrointestinal bleeding in a patient at high-thrombotic risk. Gastroenterol. Hepatol. 2013, 36, 35–38. [Google Scholar] [CrossRef] [PubMed]
- McClay, H.; Cervi, P. Thalidomide and bowel perforation: Four cases in one hospital. Br. J. Haematol. 2008, 140, 360–361. [Google Scholar] [CrossRef]
- Kaur, M.; Budania, A.; Agrawal, A.; Lahoria, U. Severe postural hypotension and sinus bradycardia with thalidomide in patients with erythema nodosum leprosum. BMJ Case Rep. 2024, 17, e256303. [Google Scholar] [CrossRef]
- Zhu, Z.; Li, M.; Shu, X.; Bai, A.; Long, S.; Liu, D.; Lu, N.; Zhu, X.; Liao, W. Thalidomide is a therapeutic agent that is effective in inducing and maintaining endoscopic remission in adult CD patients. Clin. Res. Hepatol. Gastroenterol. 2017, 41, 210–216. [Google Scholar] [CrossRef]
- Kane, S.; Stone, L.J.; Ehrenpreis, E. Thalidomide as “salvage” therapy for patients with delayed hypersensitivity response to infliximab: A case series. J. Clin. Gastroenterol. 2002, 35, 149–150. [Google Scholar] [CrossRef]
- Vernier-Massouille, G.; Balde, M.; Salleron, J.; Turck, D.; Dupas, J.L.; Mouterde, O.; Merle, V.; Salomez, J.L.; Branche, J.; Marti, R.; et al. Natural history of pediatric Crohn’s disease: A population-based cohort study. Gastroenterology 2008, 135, 1106–1113. [Google Scholar] [CrossRef] [PubMed]
- Van Limbergen, J.; Russell, R.K.; Drummond, H.E.; Aldhous, M.C.; Round, N.K.; Nimmo, E.R.; Smith, L.; Gillett, P.M.; McGrogan, P.; Weaver, L.T.; et al. Definition of phenotypic characteristics of childhood-onset inflammatory bowel disease. Gastroenterology 2008, 135, 1114–1122. [Google Scholar] [CrossRef]
- Kugathasan, S.; Baldassano, R.N.; Bradfield, J.P.; Sleiman, P.M.; Imielinski, M.; Guthery, S.L.; Cucchiara, S.; Kim, C.E.; Frackelton, E.C.; Annaiah, K.; et al. Loci on 20q13 and 21q22 are associated with pediatric-onset inflammatory bowel disease. Nat. Genet. 2008, 40, 1211–1215. [Google Scholar] [CrossRef]
- Heyman, M.B.; Kirschner, B.S.; Gold, B.D.; Ferry, G.; Baldassano, R.; Cohen, S.A.; Winter, H.S.; Fain, P.; King, C.; Smith, T.; et al. Children with early-onset inflammatory bowel disease (IBD): Analysis of a pediatric IBD consortium registry. J. Pediatr. 2005, 146, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Turunen, P.; Ashorn, M.; Auvinen, A.; Iltanen, S.; Huhtala, H.; Kolho, K.L. Long-term health outcomes in pediatric inflammatory bowel disease: A population-based study. Inflamm. Bowel Dis. 2009, 15, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Sawczenko, A.; Sandhu, B.K. Presenting features of inflammatory bowel disease in Great Britain and Ireland. Arch. Dis. Child. 2003, 88, 995–1000. [Google Scholar] [CrossRef]
- Lazzerini, M.; Villanacci, V.; Pellegrin, M.C.; Martelossi, S.; Magazzù, G.; Pellegrino, S.; Lucanto, M.C.; Barabino, A.; Calvi, A.; Arrigo, S.; et al. Endoscopic and Histologic Healing in Children with Inflammatory Bowel Diseases Treated with Thalidomide. Clin. Gastroenterol. Hepatol. 2017, 15, 1382–1389.e1381. [Google Scholar] [CrossRef]
- Wang, L.; Xue, A.; Zheng, C.; Zhou, Y.; Wang, Y.; Huang, Y. Long-term outcomes of thalidomide in pediatric Crohn’s disease. J. Gastroenterol. Hepatol. 2020, 35, 1124–1129. [Google Scholar] [CrossRef]
- Dotson, J.L.; Crandall, W.V.; Zhang, P.; Forrest, C.B.; Bailey, L.C.; Colletti, R.B.; Kappelman, M.D. Feasibility and validity of the pediatric ulcerative colitis activity index in routine clinical practice. J. Pediatr. Gastroenterol. Nutr. 2015, 60, 200–204. [Google Scholar] [CrossRef]
- Diamanti, A.; Knafelz, D.; Panetta, F.; De Angelis, P.; Candusso, M.; Bracci, F.; Papadatou, B.; Francalanci, P.; Monti, L.; Torre, G. Thalidomide as rescue therapy for acute severe ulcerative colitis. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 1690–1693. [Google Scholar]
- Dipasquale, V.; Tropeano, A.; Caime, F.; Romano, C. Thalidomide for de novo Crohn’s disease after ileal pouch anal anastomosis for ulcerative colitis. J. Clin. Pharm. Ther. 2020, 45, 819–821. [Google Scholar] [CrossRef] [PubMed]
- Giardino, S.; Bava, C.; Arrigo, S.; Pierri, F.; Gandullia, P.; Coccia, C.; Faraci, M. Thalidomide as treatment of crohn-like disease occurred after allogeneic hematopoietic stem cell transplantation in a pediatric patient. Pediatr. Transplant. 2021, 25, e13941. [Google Scholar] [CrossRef] [PubMed]
- Muise, A.M.; Snapper, S.B.; Kugathasan, S. The age of gene discovery in very early onset inflammatory bowel disease. Gastroenterology 2012, 143, 285–288. [Google Scholar] [CrossRef] [PubMed]
- Uhlig, H.H.; Schwerd, T.; Koletzko, S.; Shah, N.; Kammermeier, J.; Elkadri, A.; Ouahed, J.; Wilson, D.C.; Travis, S.P.; Turner, D.; et al. The diagnostic approach to monogenic very early onset inflammatory bowel disease. Gastroenterology 2014, 147, 990–1007.e1003. [Google Scholar] [CrossRef]
- Kelsen, J.R.; Baldassano, R.N.; Artis, D.; Sonnenberg, G.F. Maintaining intestinal health: The genetics and immunology of very early onset inflammatory bowel disease. Cell. Mol. Gastroenterol. Hepatol. 2015, 1, 462–476. [Google Scholar] [CrossRef]
- Benchimol, E.I.; Guttmann, A.; Griffiths, A.M.; Rabeneck, L.; Mack, D.R.; Brill, H.; Howard, J.; Guan, J.; To, T. Increasing incidence of paediatric inflammatory bowel disease in Ontario, Canada: Evidence from health administrative data. Gut 2009, 58, 1490–1497. [Google Scholar] [CrossRef]
- Bramuzzo, M.; Giudici, F.; Arrigo, S.; Lionetti, P.; Zuin, G.; Romano, C.; Graziano, F.; Faraci, S.; Alvisi, P.; Signa, S.; et al. Efficacy and Tolerance of Thalidomide in Patients with Very Early Onset Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2023, 30, 20–28. [Google Scholar] [CrossRef]
- Ye, Z.; Qian, L.; Hu, W.; Miao, S.; Wang, Y.; Lu, J.; Zhou, Y.; Lu, X.; Zhang, Y.; Zheng, C.; et al. Clinical outcome of infantile-onset inflammatory bowel disease in 102 patients with interleukin-10 signalling deficiency. Aliment. Pharmacol. Ther. 2022, 55, 1414–1422. [Google Scholar] [CrossRef]
- Liew, W.K.; Pacak, C.A.; Visyak, N.; Darras, B.T.; Bousvaros, A.; Kang, P.B. Longitudinal Patterns of Thalidomide Neuropathy in Children and Adolescents. J. Pediatr. 2016, 178, 227–232. [Google Scholar] [CrossRef]
- Fleming, F.J.; Vytopil, M.; Chaitow, J.; Jones, H.R., Jr.; Darras, B.T.; Ryan, M.M. Thalidomide neuropathy in childhood. Neuromuscul. Disord. 2005, 15, 172–176. [Google Scholar] [CrossRef]
- Bramuzzo, M.; Stocco, G.; Montico, M.; Arrigo, S.; Calvi, A.; Lanteri, P.; Costa, S.; Pellegrino, S.; Magazzù, G.; Barp, J.; et al. Risk Factors and Outcomes of Thalidomide-induced Peripheral Neuropathy in a Pediatric Inflammatory Bowel Disease Cohort. Inflamm. Bowel Dis. 2017, 23, 1810–1816. [Google Scholar] [CrossRef]
- Ruemmele, F.M.; Veres, G.; Kolho, K.L.; Griffiths, A.; Levine, A.; Escher, J.C.; Amil Dias, J.; Barabino, A.; Braegger, C.P.; Bronsky, J.; et al. Consensus guidelines of ECCO/ESPGHAN on the medical management of pediatric Crohn’s disease. J. Crohn’s Colitis 2014, 8, 1179–1207. [Google Scholar] [CrossRef] [PubMed]
- Hamza, M.H. Treatment of Behçet’s disease with thalidomide. Clin. Rheumatol. 1986, 5, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Hamuryudan, V.; Mat, C.; Saip, S.; Ozyazgan, Y.; Siva, A.; Yurdakul, S.; Zwingenberger, K.; Yazici, H. Thalidomide in the treatment of the mucocutaneous lesions of the Behçet syndrome. A randomized, double-blind, placebo-controlled trial. Ann. Intern. Med. 1998, 128, 443–450. [Google Scholar] [CrossRef]
- Hello, M.; Barbarot, S.; Bastuji-Garin, S.; Revuz, J.; Chosidow, O. Use of thalidomide for severe recurrent aphthous stomatitis: A multicenter cohort analysis. Medicine 2010, 89, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Skef, W.; Hamilton, M.J.; Arayssi, T. Gastrointestinal Behçet’s disease: A review. World J. Gastroenterol. 2015, 21, 3801–3812. [Google Scholar] [CrossRef]
- Hatemi, I.; Hatemi, G.; Çelik, A.F. Gastrointestinal Involvement in Behçet Disease. Rheum. Dis. Clin. N. Am. 2018, 44, 45–64. [Google Scholar] [CrossRef]
- Postema, P.T.; den Haan, P.; van Hagen, P.M.; van Blankenstein, M. Treatment of colitis in Behçet’s disease with thalidomide. Eur. J. Gastroenterol. Hepatol. 1996, 8, 929–931. [Google Scholar]
- Larsson, H. Treatment of severe colitis in Behçet’s syndrome with thalidomide (CG-217). J. Intern. Med. 1990, 228, 405–407. [Google Scholar] [CrossRef]
- Terrin, G.; Borrelli, O.; Di Nardo, G.; Pacchiarotti, C.; Cucchiara, S. A child with aphthae and diarrhoea. Lancet 2002, 359, 316. [Google Scholar] [CrossRef]
- Brik, R.; Shamali, H.; Bergman, R. Successful thalidomide treatment of severe infantile Behçet disease. Pediatr. Dermatol. 2001, 18, 143–145. [Google Scholar] [CrossRef] [PubMed]
- Shek, L.P.; Lee, Y.S.; Lee, B.W.; Lehman, T.J. Thalidomide responsiveness in an infant with Behçet’s syndrome. Pediatrics 1999, 103, 1295–1297. [Google Scholar] [CrossRef] [PubMed]
- Hatemi, I.; Hatemi, G.; Pamuk, O.N.; Erzin, Y.; Celik, A.F. TNF-alpha antagonists and thalidomide for the management of gastrointestinal Behçet’s syndrome refractory to the conventional treatment modalities: A case series and review of the literature. Clin. Exp. Rheumatol. 2015, 33, S129–S137. [Google Scholar]
- Mitsunaga, K.; Inoue, Y.; Naito, C.; Ogata, H.; Itoh, Y.; Natsui, Y.; Saito, T.; Tomiita, M. A case of A20 haploinsufficiency in which intestinal inflammation improved with thalidomide. Rheumatology 2022, 62, e193–e195. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Su, G.; Zhou, Z.; Lai, J. Clinical characteristics and genetic analysis of A20 haploinsufficiency. Pediatr. Rheumatol. Online J. 2021, 19, 75. [Google Scholar] [CrossRef]
- Zhang, C.; Yu, Z.; Gao, S.; Ma, M.; Gou, L.; Wang, C.; Wang, L.; Li, J.; Zhong, L.; Zhou, Y.; et al. Efficacy and safety of thalidomide in children with monogenic autoinflammatory diseases: A single-center, real-world-evidence study. Pediatr. Rheumatol. Online J. 2023, 21, 124. [Google Scholar] [CrossRef]
- Wei, Q.; Zhang, X.; Peng, Y.; Chen, K.; Xu, S.; Huang, X.; Zhang, L.; Xie, Q.; He, Y.; Li, Y.; et al. Successful treatment by thalidomide therapy of intestinal Behçet’s disease associated with trisomy 8 myelodysplastic syndrome. Rheumatology 2021, 60, e200–e202. [Google Scholar] [CrossRef]
- Yilmaz, U.; Ar, M.C.; Esatoglu, S.N.; Bavunoglu, I.; Erzin, Y.Z.; Hatemi, A.I.; Yildirim, S.; Hatemi, G.; Celik, A.F. How to treat myelodysplastic syndrome with clinical features resembling Behçet syndrome: A case-based systematic review. Ann. Hematol. 2020, 99, 1193–1203. [Google Scholar] [CrossRef]
- Chen, Y.; Shen, Y.; Ma, H.F.; Cai, J.F.; Hua, Y.Q.; Zou, J.; Guan, J.L. Infliximab associated with life-threatening lung infection in a patient with Behcet disease with intestinal and hematopoietic system involvement: A case report. Medicine 2017, 96, e9202. [Google Scholar] [CrossRef]
- Zhang, X.; Zhao, Y.; Pan, Y.; Jin, J.; Fan, Z.; Yu, H. Trisomy 8 presentation by inflammatory manifestations and its response to thalidomide: Two case reports and narrative review. Front. Pediatr. 2024, 12, 1431511. [Google Scholar] [CrossRef]
- Wu, S.; Wang, H. IgG4-related digestive diseases: Diagnosis and treatment. Front. Immunol. 2023, 14, 1278332. [Google Scholar] [CrossRef]
- Zeng, Q.; Shi, X.; Yang, J.; Yang, M.; Zhao, W.; Zhao, X.; Shi, J.; Zhou, H. The efficacy and safety of thalidomide on the recurrence interval of continuous recurrent aphthous ulceration: A randomized controlled clinical trial. J. Oral Pathol. Med. 2020, 49, 357–364. [Google Scholar] [CrossRef]
- Liang, L.; Gan, M.; Miao, H.; Liu, J.; Liang, C.; Qin, J.; Ruan, K.; Zhu, H.; Zhong, J.; Lin, Z. Thalidomide attenuates radiation-induced apoptosis and pro-inflammatory cytokine secretion in oral epithelial cells by promoting LZTS3 expression. J. Transl. Med. 2024, 22, 863. [Google Scholar] [CrossRef]
- Junquera, F.; Saperas, E.; de Torres, I.; Vidal, M.T.; Malagelada, J.R. Increased expression of angiogenic factors in human colonic angiodysplasia. Am. J. Gastroenterol. 1999, 94, 1070–1076. [Google Scholar] [CrossRef]
- Feng, N.; Chen, H.; Fu, S.; Bian, Z.; Lin, X.; Yang, L.; Gao, Y.; Fang, J.; Ge, Z. HIF-1α and HIF-2α induced angiogenesis in gastrointestinal vascular malformation and reversed by thalidomide. Sci. Rep. 2016, 6, 27280. [Google Scholar] [CrossRef]
- Paciullo, F.; Fierro, T.; Calcinaro, F.; Zucca Giucca, G.; Gresele, P.; Bury, L. Long-term treatment with thalidomide for severe recurrent hemorrhage from intestinal angiodysplasia in Glanzmann Thrombasthenia. Platelets 2021, 32, 288–291. [Google Scholar] [CrossRef]
- Alam, M.A.; Sami, S.; Babu, S. Successful treatment of bleeding gastro-intestinal angiodysplasia in hereditary haemorrhagic telangiectasia with thalidomide. BMJ Case Rep. 2011, 2011, bcr0820114585. [Google Scholar] [CrossRef]
- Kirkham, S.E.; Lindley, K.J.; Elawad, M.A.; Blanshard, C.; Shah, N. Treatment of multiple small bowel angiodysplasias causing severe life-threatening bleeding with thalidomide. J. Pediatr. Gastroenterol. Nutr. 2006, 42, 585–587. [Google Scholar] [CrossRef]
- Tang, C.T.; Zhang, Q.W.; Wu, S.; Tang, M.Y.; Liang, Q.; Lin, X.L.; Gao, Y.J.; Ge, Z.Z. Thalidomide targets EGFL6 to inhibit EGFL6/PAX6 axis-driven angiogenesis in small bowel vascular malformation. Cell. Mol. Life Sci. 2020, 77, 5207–5221. [Google Scholar] [CrossRef]
- Song, K.; He, K.; Yan, X.; Pang, K.; Tang, R.; Lyu, C.; Yang, D.; Zhang, Y.; Wu, D. Efficacy and safety of thalidomide in gastrointestinal angiodysplasias: Systematic review and meta-analysis with trial sequential analysis of randomized controlled trials. Ther. Adv. Gastroenterol. 2024, 17, 17562848241255295. [Google Scholar] [CrossRef]
- Singh, S.; Mohan, B.P.; Sanaei, O.; Vinayek, R.; Dutta, S.; Dahiya, D.S.; Bhat, I.; Sharma, N.; Adler, D.G. Safety and efficacy of thalidomide in treatment of gastrointestinal bleeding secondary to angioectasias: A systematic review and meta-analysis. Scand. J. Gastroenterol. 2024, 59, 781–787. [Google Scholar] [CrossRef]
- Ugur, M.C.; Baysal, M.; Umit, E.G. The Role of Thalidomide and Its Analogs in the Treatment of Hereditary Hemorrhagic Telangiectasia: A Systematic Review. J. Clin. Med. 2024, 13, 5404. [Google Scholar] [CrossRef]
- Kim, K.T.; Chae, H.S.; Kim, J.S.; Kim, H.K.; Cho, Y.S.; Choi, W.; Choi, K.Y.; Rho, S.Y.; Kang, S.J. Thalidomide effect in endothelial cell of acute radiation proctitis. World J. Gastroenterol. 2008, 14, 4779–4783. [Google Scholar] [CrossRef]
- Karajeh, M.A.; Hurlstone, D.P.; Stephenson, T.J.; Ray-Chaudhuri, D.; Gleeson, D.C. Refractory bleeding from portal hypertensive gastropathy: A further novel role for thalidomide therapy? Eur. J. Gastroenterol. Hepatol. 2006, 18, 545–548. [Google Scholar] [CrossRef]
- Lopez-Talavera, J.C.; Cadelina, G.; Olchowski, J.; Merrill, W.; Groszmann, R.J. Thalidomide inhibits tumor necrosis factor alpha, decreases nitric oxide synthesis, and ameliorates the hyperdynamic circulatory syndrome in portal-hypertensive rats. Hepatology 1996, 23, 1616–1621. [Google Scholar] [CrossRef]
- Qi, F.; Zhang, J.; Li, J.; Li, D.; Gao, N.; Qi, Z.; Kong, X.; Yu, Z.; Fang, Y.; Cui, W.; et al. Synergistic immunochemotherapy targeted SAMD4B-APOA2-PD-L1 axis potentiates antitumor immunity in hepatocellular carcinoma. Cell Death Dis. 2024, 15, 421. [Google Scholar] [CrossRef]
- Lichtenstein, G.R.; Loftus, E.V.; Isaacs, K.L.; Regueiro, M.D.; Gerson, L.B.; Sands, B.E. ACG Clinical Guideline: Management of Crohn’s Disease in Adults. Am. J. Gastroenterol. 2018, 113, 481–517. [Google Scholar] [CrossRef]
- Tas, S.W.; Maracle, C.X.; Balogh, E.; Szekanecz, Z. Targeting of proangiogenic signalling pathways in chronic inflammation. Nat. Rev. Rheumatol. 2016, 12, 111–122. [Google Scholar] [CrossRef]
- Deban, L.; Correale, C.; Vetrano, S.; Malesci, A.; Danese, S. Multiple pathogenic roles of microvasculature in inflammatory bowel disease: A Jack of all trades. Am. J. Pathol. 2008, 172, 1457–1466. [Google Scholar] [CrossRef]
- Wang, L.; Wang, S.; Xue, A.; Shi, J.; Zheng, C.; Huang, Y. Thalidomide Inhibits Angiogenesis via Downregulation of VEGF and Angiopoietin-2 in Crohn’s Disease. Inflammation 2021, 44, 795–807. [Google Scholar] [CrossRef]
- Hisamatsu, T.; Naganuma, M.; Pinton, P.; Takeno, M. Behçet’s disease: Incidence, prevalence, and real-word data on the use of biologic agents in Japan. J. Gastroenterol. 2024, 60, 294–305. [Google Scholar] [CrossRef]
- Shen, Y.; Ma, H.F.; Yang, Y.L.; Guan, J.L. Ulcerative intestinal tuberculosis case as a complication of treatment by infliximab for intestinal Behçet’s disease: A case report. Medicine 2019, 98, e17652. [Google Scholar] [CrossRef]
- Wang, L.; Hong, Y.; Wu, J.; Leung, Y.K.; Huang, Y. Efficacy of thalidomide therapy in pediatric Crohn’s disease with evidence of tuberculosis. World J. Gastroenterol. 2017, 23, 7727–7734. [Google Scholar] [CrossRef]
- Leite, M.R.; Santos, S.S.; Lyra, A.C.; Mota, J.; Santana, G.O. Thalidomide induces mucosal healing in Crohn’s disease: Case report. World J. Gastroenterol. 2011, 17, 5028–5031. [Google Scholar] [CrossRef]
- Guo, J.; Fei, X.S.; Zhang, X.; Lin, F.S. A case of hematogenous disseminated tuberculosis in a patient with contradictory reactions treated with thalidomide. Zhonghua Jie He He Hu Xi Za Zhi 2024, 47, 455–459. [Google Scholar] [CrossRef]
- Haslett, P.A.; Corral, L.G.; Albert, M.; Kaplan, G. Thalidomide costimulates primary human T lymphocytes, preferentially inducing proliferation, cytokine production, and cytotoxic responses in the CD8+ subset. J. Exp. Med. 1998, 187, 1885–1892. [Google Scholar] [CrossRef]
- Fu, L.M.; Fu-Liu, C.S. Thalidomide and tuberculosis. Int. J. Tuberc. Lung Dis. 2002, 6, 569–572. [Google Scholar]
- Li, T.; Qiu, Y.; Li, X.; Zhuang, X.; Huang, S.; Li, M.; Feng, R.; Chen, B.; He, Y.; Zeng, Z.; et al. Thalidomide Combined with Azathioprine as Induction and Maintenance Therapy for Azathioprine-Refractory Crohn’s Disease Patients. Front. Med. 2020, 7, 557986. [Google Scholar] [CrossRef]
- Chen, J.R.; Mai, L.; Sun, J.C.; Peng, X.; Zhang, M.; Zhi, M. Efficacy and safety of low-dose thalidomide combined with mesalazine in the treatment of refractory ulcerative colitis in adults. Gastroenterol. Rep. 2022, 10, goac032. [Google Scholar] [CrossRef]
- Sabate, J.M.; Villarejo, J.; Lemann, M.; Bonnet, J.; Allez, M.; Modigliani, R. An open-label study of thalidomide for maintenance therapy in responders to infliximab in chronically active and fistulizing refractory Crohn’s disease. Aliment. Pharmacol. Ther. 2002, 16, 1117–1124. [Google Scholar] [CrossRef]
- Li, Y.; Han, Z.; Wang, X.; Mo, Z.; Zhang, W.; Li, A.; Liu, S. Combination therapy of infliximab and thalidomide for refractory entero-Behcet’s disease: A case report. BMC Gastroenterol. 2013, 13, 167. [Google Scholar] [CrossRef]
- Bao, H.F.; Hou, C.C.; Ye, B.; Zou, J.; Luo, D.; Cai, J.F.; Shen, Y.; Guan, J.L. Predictors of infliximab refractory intestinal Behçet’s syndrome: A retrospective cohort study from the Shanghai Behçet’s syndrome database. Mod. Rheumatol. 2023, 33, 207–216. [Google Scholar] [CrossRef]
- Guo, X.H.; Zhu, Y.L.; Yang, L.; Li, W.J.; Du, X.F. The Effects of Multi-Donor Fecal Microbiota Transplantation Capsules Combined with Thalidomide on Hormone-Dependent Ulcerative Colitis. Infect. Drug Resist. 2022, 15, 7495–7501. [Google Scholar] [CrossRef]
- Clark, T.E.; Edom, N.; Larson, J.; Lindsey, L.J. Thalomid (Thalidomide) capsules: A review of the first 18 months of spontaneous postmarketing adverse event surveillance, including off-label prescribing. Drug Saf. 2001, 24, 87–117. [Google Scholar] [CrossRef]
- Bastuji-Garin, S.; Ochonisky, S.; Bouche, P.; Gherardi, R.K.; Duguet, C.; Djerradine, Z.; Poli, F.; Revuz, J. Incidence and risk factors for thalidomide neuropathy: A prospective study of 135 dermatologic patients. J. Investig. Dermatol. 2002, 119, 1020–1026. [Google Scholar] [CrossRef]
- Palumbo, A.; Palladino, C. Venous and arterial thrombotic risks with thalidomide: Evidence and practical guidance. Ther. Adv. Drug Saf. 2012, 3, 255–266. [Google Scholar] [CrossRef]
- Ghobrial, I.M.; Rajkumar, S.V. Management of thalidomide toxicity. J. Support. Oncol. 2003, 1, 194–205. [Google Scholar]
- Vargesson, N. Thalidomide-induced teratogenesis: History and mechanisms. Birth Defects Res. Part C Embryo Today 2015, 105, 140–156. [Google Scholar] [CrossRef]
- Zajączkowska, R.; Kocot-Kępska, M.; Leppert, W.; Wrzosek, A.; Mika, J.; Wordliczek, J. Mechanisms of Chemotherapy-Induced Peripheral Neuropathy. Int. J. Mol. Sci. 2019, 20, 1451. [Google Scholar] [CrossRef]
- van Heeckeren, W.J.; Sanborn, S.L.; Narayan, A.; Cooney, M.M.; McCrae, K.R.; Schmaier, A.H.; Remick, S.C. Complications from vascular disrupting agents and angiogenesis inhibitors: Aberrant control of hemostasis and thrombosis. Curr. Opin. Hematol. 2007, 14, 468–480. [Google Scholar] [CrossRef]
- Yamamoto, J.; Ito, T.; Yamaguchi, Y.; Handa, H. Discovery of CRBN as a target of thalidomide: A breakthrough for progress in the development of protein degraders. Chem. Soc. Rev. 2022, 51, 6234–6250. [Google Scholar] [CrossRef]
- Mao, J.; Chao, K.; Jiang, F.L.; Ye, X.P.; Yang, T.; Li, P.; Zhu, X.; Hu, P.J.; Zhou, B.J.; Huang, M.; et al. Comparison and development of machine learning for thalidomide-induced peripheral neuropathy prediction of refractory Crohn’s disease in Chinese population. World J. Gastroenterol. 2023, 29, 3855–3870. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, J.B.; Dredge, K.; Dalgleish, A.G. The evolution of thalidomide and its IMiD derivatives as anticancer agents. Nat. Rev. Cancer 2004, 4, 314–322. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Millan, B.; Diaz de la Guardia, R.; Roca-Ho, H.; García-Herrero, C.M.; Lavoie, J.R.; Rosu-Myles, M.; Gonzalez-Rey, E.; O’Valle, F.; Criado, G.; Delgado, M.; et al. Therapeutic effect of the immunomodulatory drug lenalidomide, but not pomalidomide, in experimental models of rheumatoid arthritis and inflammatory bowel disease. Exp. Mol. Med. 2017, 49, e290. [Google Scholar] [CrossRef]
- Paumgartten, F.J.R. The tale of lenalidomide clinical superiority over thalidomide and regulatory and cost-effectiveness issues. Cienc. Saude Coletiva 2019, 24, 3783–3792. [Google Scholar] [CrossRef]
- Lacy, M.Q.; McCurdy, A.R. Pomalidomide. Blood 2013, 122, 2305–2309. [Google Scholar] [CrossRef]
- Abdallah, A.E.; Eissa, I.H.; Mehany, A.B.M.; Celik, I.; Sakr, H.; Metwaly, K.H.; El-Adl, K.; El-Zahabi, M.A. Discovery of New Immunomodulatory Anticancer Thalidomide Analogs: Design, Synthesis, Biological Evaluation and In Silico Studies. Chem. Biodivers. 2024, 22, e202401768. [Google Scholar] [CrossRef] [PubMed]
- Saleh Al Ward, M.M.; Abdallah, A.E.; Zayed, M.F.; Ayyad, R.R.; Abdelghany, T.M.; Bakhotmah, D.A.; El-Zahabi, M.A. New immunomodulatory anticancer quinazolinone-based thalidomide analogs: Design, synthesis and biological evaluation. Future Med. Chem. 2024, 16, 2523–2533. [Google Scholar] [CrossRef]
- Song, T.; Gu, K.; Wang, W.; Wang, H.; Yang, Y.; Yang, L.; Ma, P.; Ma, X.; Zhao, J.; Yan, R.; et al. Prolonged Suppression of Neuropathic Pain by Sequential Delivery of Lidocaine and Thalidomide Drugs Using PEGylated Graphene Oxide. J. Pharm. Sci. 2015, 104, 3851–3860. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, Q.; Zou, H.; Chou, C.K.; Chen, X. Exosome-Modified Liposomes Targeted Delivery of Thalidomide to Regulate Treg Cells for Antitumor Immunotherapy. Pharmaceutics 2023, 15, 1074. [Google Scholar] [CrossRef]
- Shen, Y.; Li, S.; Wang, X.; Wang, M.; Tian, Q.; Yang, J.; Wang, J.; Wang, B.; Liu, P.; Yang, J. Tumor vasculature remolding by thalidomide increases delivery and efficacy of cisplatin. J. Exp. Clin. Cancer Res. 2019, 38, 427. [Google Scholar] [CrossRef]
- Fakhoury, M.; Coussa-Charley, M.; Al-Salami, H.; Kahouli, I.; Prakash, S. Use of artificial cell microcapsule containing thalidomide for treating TNBS-induced Crohn’s disease in mice. Curr. Drug Deliv. 2014, 11, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Meng, Z.; Fu, B.; Yang, Z.; Xu, Y.; Huang, H.; Bai, Y.; Fang, X.; Shen, S.; Yang, J.; Yong, J.; et al. Polydopamine-coated thalidomide nanocrystals promote DSS-induced murine colitis recovery through Macrophage M2 polarization together with the synergistic anti-inflammatory and anti-angiogenic effects. Int. J. Pharm. 2023, 630, 122376. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, L.; Xu, M.; Zhao, T.; Kuang, L.; Hua, D. Smart Oral Administration of Polydopamine-Coated Nanodrugs for Efficient Attenuation of Radiation-Induced Gastrointestinal Syndrome. Adv. Healthc. Mater. 2020, 9, e1901778. [Google Scholar] [CrossRef] [PubMed]
- Bousvaros, A.; Mueller, B. Thalidomide in gastrointestinal disorders. Drugs 2001, 61, 777–787. [Google Scholar] [CrossRef] [PubMed]
- Xi, Z.; Jie, W.; Long, Z.; Shasha, S. A review of thalidomide and digestive system related diseases. Front. Oncol. 2025, 15, 1543757. [Google Scholar] [CrossRef]
- Takagi, T.; van Bennekom, C.; Amann, S.; Hattori, C.; Shirakuni, Y.; Sato, T.; Nasu, M. Risk Management of Teratogenic Drugs~The Current States of Practice in Europe, US and Japan~. Yakugaku Zasshi 2015, 135, 1161–1168. [Google Scholar] [CrossRef]




| Disease | Common Adverse Effects | Grade of Recommendations in Guidelines or Consensus a | Dose Used in Clinical Studies | Dose Used in Case Reports | |
|---|---|---|---|---|---|
| Inflammatory intestinal diseases | IBD in adults | Drowsiness, constipation, rash, and peripheral neuropathy | —— | 50–100 mg/d [31] | —— |
| Pediatric and very-early-onset IBD | Peripheral neuropathy | —— | 1.5–2.5 mg/kg/d * [32,33] | —— | |
| Intestinal Behcet’s disease | Peripheral neuropathy | C [15] | 100 mg/d [34]; 2–3 mg/kg/d (in children) [35,36] | ||
| IgG4-related disease | IgG4-related digestive disease | Limb numbness, dizziness, edema, and fatigue | —— | 25–75 mg/d * [37] | |
| Oral mucosal diseases | Recurrent aphthous stomatitis | Peripheral neuropathy, thromboembolic events, constipation, and somnolence | —— | 25 mg/d * [38] | —— |
| Radiation-induced oral mucositis | Dizziness and constipation | —— | 75 mg/d * [39] | —— | |
| Gastrointestinal bleeding | Gastrointestinal bleeding due to angiodysplasia | Constipation, somnolence, limb numbness, peripheral edema, dizziness, and elevated liver-enzyme levels | —— | 50 mg/d *; 100 mg/d * [26,40] | —— |
| Hemorrhagic radiation proctitis | —— | —— | —— | 50–100 mg/d [41] | |
| GI bleeding after kidney transplantation | —— | —— | —— | 100 mg/d [42] | |
| Bleeding portal hypertensive gastropathy and enteropathy | —— | —— | —— | 100 mg/d [43] | |
| Digestive malignant tumors | Colorectal cancer | Constipation and lethargy | —— | 200 mg/d * [44] | 100 mg/d [45] |
| Hepatocellular carcinoma | Rash | —— | 200–400 mg/d * [46]; 50 mg/d (TCC cocktail) * [47] | —— | |
| Adverse Effect | Proposed Mechanism |
|---|---|
| Teratogenicity | Anti-angiogenic activity interferes with embryonic vascular development, leading to ischemia of developing tissues and limb buds |
| Peripheral neuropathy | Secondary ischemia and hypoxia of nerve fibers due to anti-angiogenic effects; downregulation of TNF-α and inhibition of NF-κB reduce neuronal survival and neurotrophic signaling |
| Thromboembolic events | Enhanced platelet aggregation and procoagulant activity |
| Constipation | Autonomic neuropathy affects intestinal motility |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yan, X.; Guo, Z.; Lyu, C.; Tang, R.; Li, R.; Wang, H.; Song, K.; Chen, W.; He, K.; Wu, D. Renaissance of Phoenix Drug Thalidomide—New Insights into Practical Clinical Application and Optimization Strategies for Managing Adverse Effects in Digestive Diseases. Pharmaceuticals 2025, 18, 1689. https://doi.org/10.3390/ph18111689
Yan X, Guo Z, Lyu C, Tang R, Li R, Wang H, Song K, Chen W, He K, Wu D. Renaissance of Phoenix Drug Thalidomide—New Insights into Practical Clinical Application and Optimization Strategies for Managing Adverse Effects in Digestive Diseases. Pharmaceuticals. 2025; 18(11):1689. https://doi.org/10.3390/ph18111689
Chicago/Turabian StyleYan, Xiaxiao, Ziqi Guo, Chengzhen Lyu, Rou Tang, Rutong Li, Hongwei Wang, Kai Song, Wangyang Chen, Kun He, and Dong Wu. 2025. "Renaissance of Phoenix Drug Thalidomide—New Insights into Practical Clinical Application and Optimization Strategies for Managing Adverse Effects in Digestive Diseases" Pharmaceuticals 18, no. 11: 1689. https://doi.org/10.3390/ph18111689
APA StyleYan, X., Guo, Z., Lyu, C., Tang, R., Li, R., Wang, H., Song, K., Chen, W., He, K., & Wu, D. (2025). Renaissance of Phoenix Drug Thalidomide—New Insights into Practical Clinical Application and Optimization Strategies for Managing Adverse Effects in Digestive Diseases. Pharmaceuticals, 18(11), 1689. https://doi.org/10.3390/ph18111689






