Is There an Interplay between Environmental Factors, Microbiota Imbalance, and Cancer Chemotherapy-Associated Intestinal Mucositis?
Abstract
1. Introduction
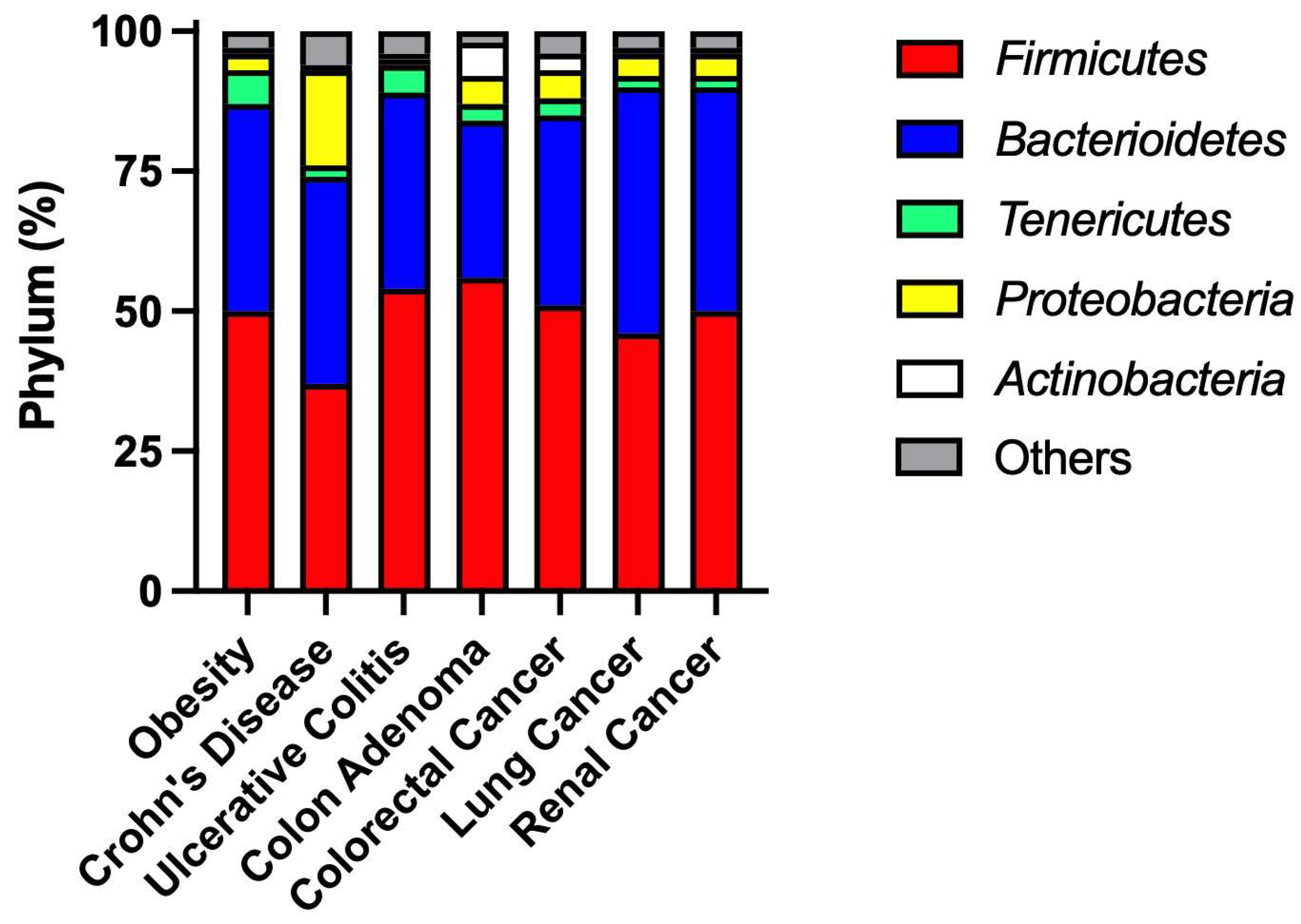
2. Environmental Factors Alter Intestine Microbiome Composition
2.1. Diet
2.2. Xenobiotics
3. Intestinal Mucositis: Definition and Pathophysiology
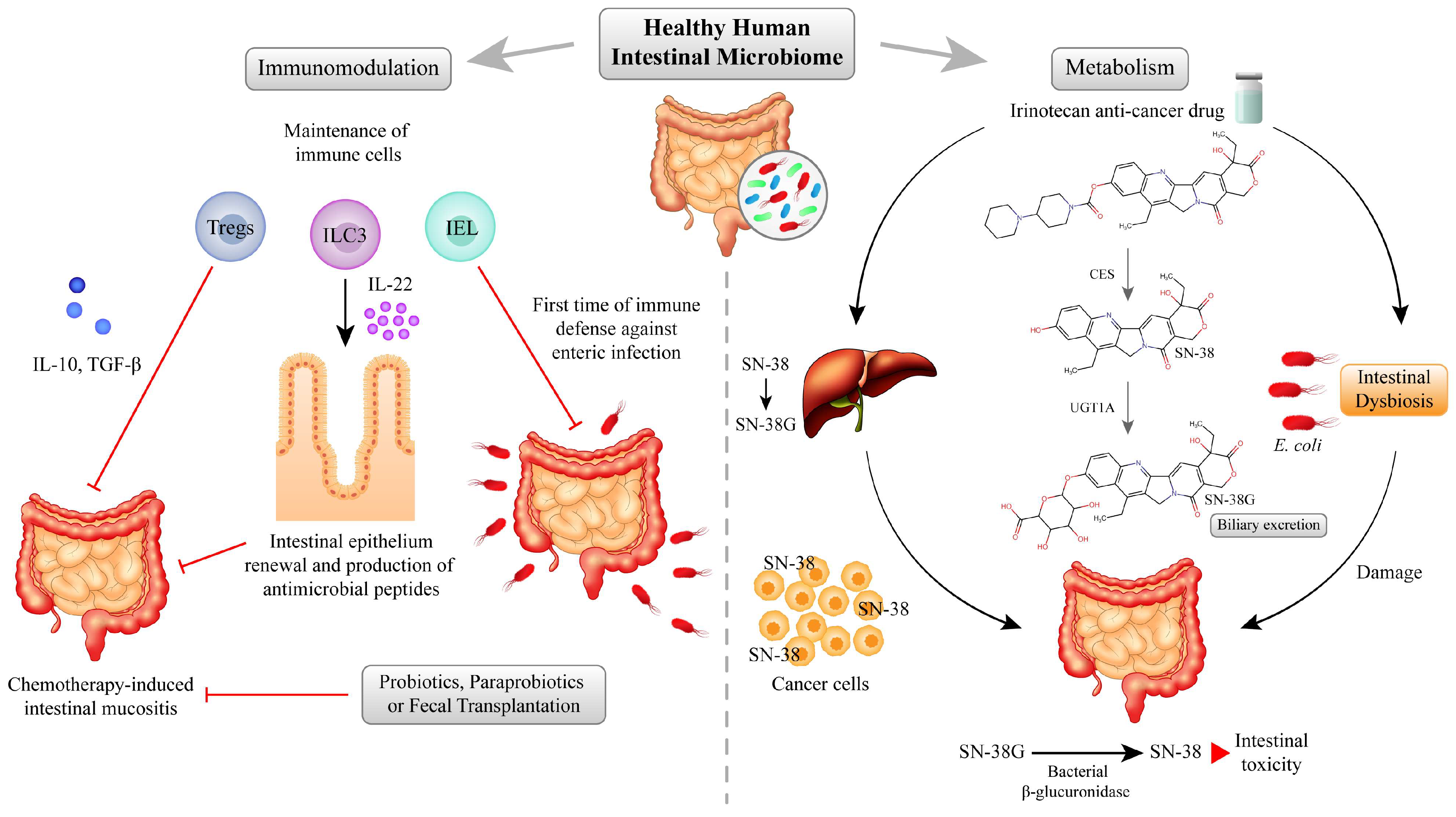
4. The Interplay between Microbiome and Chemotherapy Toxicity in the Intestine
4.1. Immunomodulation
4.2. Microbiota Effects on the Metabolism of Cancer Chemotherapy Drugs
5. Perspectives
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Weng, L.; Zhang, L.; Peng, Y.; Huang, R.S. Pharmacogenetics and Pharmacogenomics: A Bridge to Individualized Cancer Therapy. Pharmacogenomics 2013, 14, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Doestzada, M.; Vila, A.V.; Zhernakova, A.; Koonen, D.P.Y.; Weersma, R.K.; Touw, D.J.; Kuipers, F.; Wijmenga, C.; Fu, J. Pharmacomicrobiomics: A Novel Route towards Personalized Medicine? Protein Cell 2018, 9, 432–445. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.-K.; Xie, R.-L.; You, R.; Liu, Y.-P.; Chen, X.-Y.; Chen, M.-Y.; Huang, P.-Y. The Role of the Bacterial Microbiome in the Treatment of Cancer. BMC Cancer 2021, 21, 934. [Google Scholar] [CrossRef] [PubMed]
- Kundu, P.; Blacher, E.; Elinav, E.; Pettersson, S. Our Gut Microbiome: The Evolving Inner Self. Cell 2017, 171, 1481–1493. [Google Scholar] [CrossRef] [PubMed]
- Bäckhed, F.; Fraser, C.M.; Ringel, Y.; Sanders, M.E.; Sartor, R.B.; Sherman, P.M.; Versalovic, J.; Young, V.; Finlay, B.B. Defining a Healthy Human Gut Microbiome: Current Concepts, Future Directions, and Clinical Applications. Cell Host Microbe 2012, 12, 611–622. [Google Scholar] [CrossRef] [PubMed]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet Rapidly and Reproducibly Alters the Human Gut Microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef] [PubMed]
- Dethlefsen, L.; Huse, S.; Sogin, M.L.; Relman, D.A. The Pervasive Effects of an Antibiotic on the Human Gut Microbiota, as Revealed by Deep 16S RRNA Sequencing. PLoS Biol. 2008, 6, e280. [Google Scholar] [CrossRef] [PubMed]
- Sivan, A.; Corrales, L.; Hubert, N.; Williams, J.B.; Aquino-Michaels, K.; Earley, Z.M.; Benyamin, F.W.; Lei, Y.M.; Jabri, B.; Alegre, M.-L.; et al. Commensal Bifidobacterium Promotes Antitumor Immunity and Facilitates Anti-PD-L1 Efficacy. Science 2015, 350, 1084–1089. [Google Scholar] [CrossRef] [PubMed]
- Freedberg, D.E.; Toussaint, N.C.; Chen, S.P.; Ratner, A.J.; Whittier, S.; Wang, T.C.; Wang, H.H.; Abrams, J.A. Proton Pump Inhibitors Alter Specific Taxa in the Human Gastrointestinal Microbiome: A Crossover Trial. Gastroenterology 2015, 149, 883–885.e9. [Google Scholar] [CrossRef]
- Forslund, K.; Hildebrand, F.; Nielsen, T.; Falony, G.; Le Chatelier, E.; Sunagawa, S.; Prifti, E.; Vieira-Silva, S.; Gudmundsdottir, V.; Pedersen, H.K.; et al. Disentangling Type 2 Diabetes and Metformin Treatment Signatures in the Human Gut Microbiota. Nature 2015, 528, 262–266. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Xie, C.; Wang, G.; Wu, Y.; Wu, Q.; Wang, X.; Liu, J.; Deng, Y.; Xia, J.; Chen, B.; et al. Gut Microbiota and Intestinal FXR Mediate the Clinical Benefits of Metformin. Nat. Med. 2018, 24, 1919–1929. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, M.; Zimmermann-Kogadeeva, M.; Wegmann, R.; Goodman, A.L. Separating Host and Microbiome Contributions to Drug Pharmacokinetics and Toxicity. Science 2019, 363, eaat9931. [Google Scholar] [CrossRef] [PubMed]
- Chau, J.; Yadav, M.; Liu, B.; Furqan, M.; Dai, Q.; Shahi, S.; Gupta, A.; Mercer, K.N.; Eastman, E.; Hejleh, T.A.; et al. Prospective Correlation between the Patient Microbiome with Response to and Development of Immune-Mediated Adverse Effects to Immunotherapy in Lung Cancer. BMC Cancer 2021, 21, 808. [Google Scholar] [CrossRef] [PubMed]
- Weersma, R.K.; Zhernakova, A.; Fu, J. Interaction between Drugs and the Gut Microbiome. Gut 2020, 69, 1510–1519. [Google Scholar] [CrossRef] [PubMed]
- Waring, R.H.; Harris, R.M.; Mitchell, S.C. Drug Metabolism in the Elderly: A Multifactorial Problem? Maturitas 2017, 100, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Markle, J.G.M.; Frank, D.N.; Mortin-Toth, S.; Robertson, C.E.; Feazel, L.M.; Rolle-Kampczyk, U.; von Bergen, M.; McCoy, K.D.; Macpherson, A.J.; Danska, J.S. Sex Differences in the Gut Microbiome Drive Hormone-Dependent Regulation of Autoimmunity. Science (1979) 2013, 339, 1084–1088. [Google Scholar] [CrossRef] [PubMed]
- Allen, J.M.; Berg Miller, M.E.; Pence, B.D.; Whitlock, K.; Nehra, V.; Gaskins, H.R.; White, B.A.; Fryer, J.D.; Woods, J.A. Voluntary and Forced Exercise Differentially Alters the Gut Microbiome in C57BL/6J Mice. J. Appl. Physiol. 2015, 118, 1059–1066. [Google Scholar] [CrossRef] [PubMed]
- Montassier, E.; Gastinne, T.; Vangay, P.; Al-Ghalith, G.A.; Bruley des Varannes, S.; Massart, S.; Moreau, P.; Potel, G.; de La Cochetière, M.F.; Batard, E.; et al. Chemotherapy-driven Dysbiosis in the Intestinal Microbiome. Aliment. Pharmacol. Ther. 2015, 42, 515–528. [Google Scholar] [CrossRef] [PubMed]
- Ervin, S.M.; Ramanan, S.V.; Bhatt, A.P. Relationship Between the Gut Microbiome and Systemic Chemotherapy. Dig. Dis. Sci. 2020, 65, 874–884. [Google Scholar] [CrossRef]
- McQuade, R.M.; Stojanovska, V.; Abalo, R.; Bornstein, J.C.; Nurgali, K. Chemotherapy-Induced Constipation and Diarrhea: Pathophysiology, Current and Emerging Treatments. Front. Pharmacol. 2016, 7, 414. [Google Scholar] [CrossRef] [PubMed]
- Lalla, R.V.; Bowen, J.; Barasch, A.; Elting, L.; Epstein, J.; Keefe, D.M.; McGuire, D.B.; Migliorati, C.; Nicolatou-Galitis, O.; Peterson, D.E.; et al. MASCC/ISOO Clinical Practice Guidelines for the Management of Mucositis Secondary to Cancer Therapy. Cancer 2014, 120, 1453–1461. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Sun, L.; Chen, S.; Guo, S.; Yue, T.; Hou, Q.; Feng, M.; Xu, H.; Liu, Y.; Wang, P.; et al. The Administration of Escherichia Coli Nissle 1917 Ameliorates Irinotecan-Induced Intestinal Barrier Dysfunction and Gut Microbial Dysbiosis in Mice. Life Sci. 2019, 231, 116529. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.; Yan, J.; Liu, F.; Ding, P.; Chen, B.; Lu, Y.; Sun, Z. Probiotics in Preventing and Treating Chemotherapy-Induced Diarrhea: A Meta-Analysis. Asia Pac. J. Clin. Nutr. 2019, 28, 701–710. [Google Scholar] [CrossRef] [PubMed]
- Reyna-Figueroa, J.; Barrón-Calvillo, E.; García-Parra, C.; Galindo-Delgado, P.; Contreras-Ochoa, C.; Lagunas-Martínez, A.; Campos-Romero, F.H.; Silva-Estrada, J.A.; Limón-Rojas, A.E. Probiotic Supplementation Decreases Chemotherapy-Induced Gastrointestinal Side Effects in Patients with Acute Leukemia. J. Pediatr. Hematol./Oncol. 2019, 41, 468–472. [Google Scholar] [CrossRef] [PubMed]
- Bowen, J.M.; Gibson, R.J.; Coller, J.K.; Blijlevens, N.; Bossi, P.; Al-Dasooqi, N.; Bateman, E.H.; Chiang, K.; de Mooij, C.; Mayo, B.; et al. Systematic Review of Agents for the Management of Cancer Treatment-Related Gastrointestinal Mucositis and Clinical Practice Guidelines. Support. Care Cancer 2019, 27, 4011–4022. [Google Scholar] [CrossRef] [PubMed]
- Elad, S.; Cheng, K.K.F.; Lalla, R.V.; Yarom, N.; Hong, C.; Logan, R.M.; Bowen, J.; Gibson, R.; Saunders, D.P.; Zadik, Y.; et al. MASCC/ISOO Clinical Practice Guidelines for the Management of Mucositis Secondary to Cancer Therapy. Cancer 2020, 126, 4423–4431. [Google Scholar] [CrossRef] [PubMed]
- Robles Alonso, V.; Guarner, F. Linking the Gut Microbiota to Human Health. Br. J. Nutr. 2013, 109, S21–S26. [Google Scholar] [CrossRef] [PubMed]
- Gomaa, E.Z. Human Gut Microbiota/Microbiome in Health and Diseases: A Review. Antonie Van Leeuwenhoek 2020, 113, 2019–2040. [Google Scholar] [CrossRef] [PubMed]
- Rothschild, D.; Weissbrod, O.; Barkan, E.; Kurilshikov, A.; Korem, T.; Zeevi, D.; Costea, P.I.; Godneva, A.; Kalka, I.N.; Bar, N.; et al. Environment Dominates over Host Genetics in Shaping Human Gut Microbiota. Nature 2018, 555, 210–215. [Google Scholar] [CrossRef]
- Levy, M.; Kolodziejczyk, A.A.; Thaiss, C.A.; Elinav, E. Dysbiosis and the Immune System. Nat. Rev. Immunol. 2017, 17, 219–232. [Google Scholar] [CrossRef]
- Leocádio, P.C.L.; Lopes, S.C.; Dias, R.P.; Alvarez-Leite, J.I.; Guerrant, R.L.; Malva, J.O.; Oriá, R.B. The Transition From Undernutrition to Overnutrition Under Adverse Environments and Poverty: The Risk for Chronic Diseases. Front. Nutr. 2021, 8, 676044. [Google Scholar] [CrossRef] [PubMed]
- Simpson, H.L.; Campbell, B.J. Review Article: Dietary Fibre-Microbiota Interactions. Aliment. Pharmacol. Ther. 2015, 42, 158–179. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.K.; Deehan, E.C.; Zhang, Z.; Jin, M.; Baskota, N.; Perez-Muñoz, M.E.; Cole, J.; Tuncil, Y.E.; Seethaler, B.; Wang, T.; et al. Gut Microbiota Modulation with Long-Chain Corn Bran Arabinoxylan in Adults with Overweight and Obesity Is Linked to an Individualized Temporal Increase in Fecal Propionate. Microbiome 2020, 8, 118. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Long, W.; Zhang, C.; Liu, S.; Zhao, L.; Hamaker, B.R. Fiber-Utilizing Capacity Varies in Prevotella- versus Bacteroides-Dominated Gut Microbiota. Sci. Rep. 2017, 7, 2594. [Google Scholar] [CrossRef] [PubMed]
- Parada Venegas, D.; De la Fuente, M.K.; Landskron, G.; González, M.J.; Quera, R.; Dijkstra, G.; Harmsen, H.J.M.; Faber, K.N.; Hermoso, M.A. Short Chain Fatty Acids (SCFAs)-Mediated Gut Epithelial and Immune Regulation and Its Relevance for Inflammatory Bowel Diseases. Front. Immunol. 2019, 10, 277. [Google Scholar] [CrossRef]
- Chambers, E.S.; Byrne, C.S.; Morrison, D.J.; Murphy, K.G.; Preston, T.; Tedford, C.; Garcia-Perez, I.; Fountana, S.; Serrano-Contreras, J.I.; Holmes, E.; et al. Dietary Supplementation with Inulin-Propionate Ester or Inulin Improves Insulin Sensitivity in Adults with Overweight and Obesity with Distinct Effects on the Gut Microbiota, Plasma Metabolome and Systemic Inflammatory Responses: A Randomised Cross-over Trial. Gut 2019, 68, 1430–1438. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.M.; Howitt, M.R.; Panikov, N.; Michaud, M.; Gallini, C.A.; Bohlooly-y, M.; Glickman, J.N.; Garrett, W.S. The Microbial Metabolites, Short-Chain Fatty Acids, Regulate Colonic Treg Cell Homeostasis. Science (1979) 2013, 341, 569–573. [Google Scholar] [CrossRef]
- Haghikia, A.; Jörg, S.; Duscha, A.; Berg, J.; Manzel, A.; Waschbisch, A.; Hammer, A.; Lee, D.-H.; May, C.; Wilck, N.; et al. Dietary Fatty Acids Directly Impact Central Nervous System Autoimmunity via the Small Intestine. Immunity 2015, 43, 817–829. [Google Scholar] [CrossRef] [PubMed]
- Lupp, C.; Robertson, M.L.; Wickham, M.E.; Sekirov, I.; Champion, O.L.; Gaynor, E.C.; Finlay, B.B. Host-Mediated Inflammation Disrupts the Intestinal Microbiota and Promotes the Overgrowth of Enterobacteriaceae. Cell Host Microbe 2007, 2, 204. [Google Scholar] [CrossRef] [PubMed]
- Thim-Uam, A.; Makjaroen, J.; Issara-Amphorn, J.; Saisorn, W.; Wannigama, D.L.; Chancharoenthana, W.; Leelahavanichkul, A. Enhanced Bacteremia in Dextran Sulfate-Induced Colitis in Splenectomy Mice Correlates with Gut Dysbiosis and LPS Tolerance. Int. J. Mol. Sci. 2022, 23, 1676. [Google Scholar] [CrossRef] [PubMed]
- Islam, J.; Koseki, T.; Watanabe, K.; Ardiansyah; Budijanto, S.; Oikawa, A.; Alauddin, M.; Goto, T.; Aso, H.; Komai, M.; et al. Dietary Supplementation of Fermented Rice Bran Effectively Alleviates Dextran Sodium Sulfate-Induced Colitis in Mice. Nutrients 2017, 9, 747. [Google Scholar] [CrossRef] [PubMed]
- Vrieze, A.; Out, C.; Fuentes, S.; Jonker, L.; Reuling, I.; Kootte, R.S.; van Nood, E.; Holleman, F.; Knaapen, M.; Romijn, J.A.; et al. Impact of Oral Vancomycin on Gut Microbiota, Bile Acid Metabolism, and Insulin Sensitivity. J. Hepatol. 2014, 60, 824–831. [Google Scholar] [CrossRef] [PubMed]
- Palleja, A.; Mikkelsen, K.H.; Forslund, S.K.; Kashani, A.; Allin, K.H.; Nielsen, T.; Hansen, T.H.; Liang, S.; Feng, Q.; Zhang, C.; et al. Recovery of Gut Microbiota of Healthy Adults Following Antibiotic Exposure. Nat. Microbiol. 2018, 3, 1255–1265. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, J.G. Clinical Practice. Antibiotic-Associated Diarrhea. N. Engl. J. Med. 2002, 346, 334–339. [Google Scholar] [CrossRef] [PubMed]
- Willing, B.P.; Russell, S.L.; Finlay, B.B. Shifting the Balance: Antibiotic Effects on Host-Microbiota Mutualism. Nat. Rev. Microbiol. 2011, 9, 233–243. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Young, C.; Neu, J. Molecular Modulation of Intestinal Epithelial Barrier: Contribution of Microbiota. BioMed Res. Int. 2010, 2010, 305879. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Huang, Y.; Wang, Y.; Wang, P.; Song, H.; Wang, F. Antibiotics Induced Intestinal Tight Junction Barrier Dysfunction Is Associated with Microbiota Dysbiosis, Activated NLRP3 Inflammasome and Autophagy. PLoS ONE 2019, 14, e0218384. [Google Scholar] [CrossRef] [PubMed]
- von Itzstein, M.S.; Gonugunta, A.S.; Sheffield, T.; Homsi, J.; Dowell, J.E.; Koh, A.Y.; Raj, P.; Fattah, F.; Wang, Y.; Basava, V.S.; et al. Association between Antibiotic Exposure and Systemic Immune Parameters in Cancer Patients Receiving Checkpoint Inhibitor Therapy. Cancers 2022, 14, 1327. [Google Scholar] [CrossRef] [PubMed]
- Chi, L.; Bian, X.; Gao, B.; Tu, P.; Ru, H.; Lu, K. The Effects of an Environmentally Relevant Level of Arsenic on the Gut Microbiome and Its Functional Metagenome. Toxicol. Sci. 2017, 160, 193–204. [Google Scholar] [CrossRef] [PubMed]
- Chi, L.; Tu, P.; Ru, H.; Lu, K. Studies of Xenobiotic-Induced Gut Microbiota Dysbiosis: From Correlation to Mechanisms. Gut Microbes 2021, 13, 1921912. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Zhou, C.; Wu, C.; Guo, X.; Hu, G.; Wu, Q.; Xu, Z.; Li, G.; Cao, H.; Li, L.; et al. Subchronic Oral Mercury Caused Intestinal Injury and Changed Gut Microbiota in Mice. Sci. Total Environ. 2020, 721, 137639. [Google Scholar] [CrossRef] [PubMed]
- Pinto, D.V.; Raposo, R.S.; Matos, G.A.; Alvarez-Leite, J.I.; Malva, J.O.; Oriá, R.B. Methylmercury Interactions with Gut Microbiota and Potential Modulation of Neurogenic Niches in the Brain. Front. Neurosci. 2020, 14, 576543. [Google Scholar] [CrossRef] [PubMed]
- Kittle, R.P.; McDermid, K.J.; Muehlstein, L.; Balazs, G.H. Effects of Glyphosate Herbicide on the Gastrointestinal Microflora of Hawaiian Green Turtles (Chelonia mydas) Linnaeus. Mar. Pollut. Bull. 2018, 127, 170–174. [Google Scholar] [CrossRef] [PubMed]
- Joly, C.; Gay-Quéheillard, J.; Léké, A.; Chardon, K.; Delanaud, S.; Bach, V.; Khorsi-Cauet, H. Impact of Chronic Exposure to Low Doses of Chlorpyrifos on the Intestinal Microbiota in the Simulator of the Human Intestinal Microbial Ecosystem (SHIME) and in the Rat. Environ. Sci. Pollut. Res. Int. 2013, 20, 2726–2734. [Google Scholar] [CrossRef] [PubMed]
- Nasuti, C.; Coman, M.M.; Olek, R.A.; Fiorini, D.; Verdenelli, M.C.; Cecchini, C.; Silvi, S.; Fedeli, D.; Gabbianelli, R. Changes on Fecal Microbiota in Rats Exposed to Permethrin during Postnatal Development. Environ. Sci. Pollut. Res. Int. 2016, 23, 10930–10937. [Google Scholar] [CrossRef] [PubMed]
- Montassier, E.; Batard, E.; Massart, S.; Gastinne, T.; Carton, T.; Caillon, J.; Le Fresne, S.; Caroff, N.; Hardouin, J.B.; Moreau, P.; et al. 16S RRNA Gene Pyrosequencing Reveals Shift in Patient Faecal Microbiota during High-Dose Chemotherapy as Conditioning Regimen for Bone Marrow Transplantation. Microb. Ecol. 2014, 67, 690–699. [Google Scholar] [CrossRef] [PubMed]
- Motoori, M.; Yano, M.; Miyata, H.; Sugimura, K.; Saito, T.; Omori, T.; Fujiwara, Y.; Miyoshi, N.; Akita, H.; Gotoh, K.; et al. Randomized Study of the Effect of Synbiotics during Neoadjuvant Chemotherapy on Adverse Events in Esophageal Cancer Patients. Clin. Nutr. 2017, 36, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Yang, W.; Liu, H.; Duan, J.; Zhang, Y.; Liu, M.; Li, H.; Hou, Z.; Wu, K.K. Effect of High-Dose Methotrexate Chemotherapy on Intestinal Bifidobacteria, Lactobacillus and Escherichia Coli in Children with Acute Lymphoblastic Leukemia. Exp. Biol. Med. 2012, 237, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Rajagopala, S.V.; Singh, H.; Yu, Y.; Zabokrtsky, K.B.; Torralba, M.G.; Moncera, K.J.; Frank, B.; Pieper, R.; Sender, L.; Nelson, K.E. Persistent Gut Microbial Dysbiosis in Children with Acute Lymphoblastic Leukemia (ALL) During Chemotherapy. Microb. Ecol. 2020, 79, 1034–1043. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.B.; Dieleman, L.A.; Ketabi, A.; Bibova, I.; Sawyer, M.B.; Xue, H.; Field, C.J.; Baracos, V.E.; Gänzle, M.G. Irinotecan (CPT-11) Chemotherapy Alters Intestinal Microbiota in Tumour Bearing Rats. PLoS ONE 2012, 7, e39764. [Google Scholar] [CrossRef] [PubMed]
- Stringer, A.M.; Gibson, R.J.; Logan, R.M.; Bowen, J.M.; Yeoh, A.S.J.; Keefe, D.M.K. Faecal Microflora and Beta-Glucuronidase Expression Are Altered in an Irinotecan-Induced Diarrhea Model in Rats. Cancer Biol. Ther. 2008, 7, 1919–1925. [Google Scholar] [CrossRef] [PubMed]
- Zeng, M.Y.; Inohara, N.; Nuñez, G. Mechanisms of Inflammation-Driven Bacterial Dysbiosis in the Gut. Mucosal Immunol. 2017, 10, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-H.; Ko, J.-L.; Liao, J.-M.; Huang, S.-S.; Lin, M.-Y.; Lee, L.-H.; Chang, L.-Y.; Ou, C.-C. D-Methionine Alleviates Cisplatin-Induced Mucositis by Restoring the Gut Microbiota Structure and Improving Intestinal Inflammation. Ther. Adv. Med. Oncol. 2019, 11, 1758835918821021. [Google Scholar] [CrossRef] [PubMed]
- Klaassen, C.D.; Cui, J.Y. Review: Mechanisms of How the Intestinal Microbiota Alters the Effects of Drugs and Bile Acids. Drug Metab. Dispos. 2015, 43, 1505–1521. [Google Scholar] [CrossRef] [PubMed]
- Jaye, K.; Li, C.G.; Bhuyan, D.J. The Complex Interplay of Gut Microbiota with the Five Most Common Cancer Types: From Carcinogenesis to Therapeutics to Prognoses. Crit. Rev. Oncol. Hematol. 2021, 165, 103429. [Google Scholar] [CrossRef]
- Kaźmierczak-Siedlecka, K.; Daca, A.; Fic, M.; van de Wetering, T.; Folwarski, M.; Makarewicz, W. Therapeutic Methods of Gut Microbiota Modification in Colorectal Cancer Management—Fecal Microbiota Transplantation, Prebiotics, Probiotics, and Synbiotics. Gut Microbes 2020, 11, 1518–1530. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, R.A.; Wanderley, C.W.S.; Wong, D.V.T.; Mota, J.M.S.C.; Leite, C.A.V.G.; Souza, M.H.L.P.; Cunha, F.Q.; Lima-Júnior, R.C.P. Irinotecan- and 5-Fluorouracil-Induced Intestinal Mucositis: Insights into Pathogenesis and Therapeutic Perspectives. Cancer Chemother. Pharmacol. 2016, 78, 881–893. [Google Scholar] [CrossRef] [PubMed]
- Sougiannis, A.T.; VanderVeen, B.N.; Davis, J.M.; Fan, D.; Murphy, E.A. Understanding Chemotherapy-Induced Intestinal Mucositis and Strategies to Improve Gut Resilience. Am. J. Physiol. Gastrointest. Liver Physiol. 2021, 320, G712–G719. [Google Scholar] [CrossRef]
- Dranitsaris, G.; Maroun, J.; Shah, A. Severe Chemotherapy-Induced Diarrhea in Patients with Colorectal Cancer: A Cost of Illness Analysis. Support Care Cancer 2005, 13, 318–324. [Google Scholar] [CrossRef]
- Mayo, B.J.; Stringer, A.M.; Bowen, J.M.; Bateman, E.H.; Keefe, D.M. Irinotecan-Induced Mucositis: The Interactions and Potential Role of GLP-2 Analogues. Cancer Chemother. Pharmacol. 2017, 79, 233–249. [Google Scholar] [CrossRef]
- Bailly, C. Irinotecan: 25 Years of Cancer Treatment. Pharmacol. Res. 2019, 148, 104398. [Google Scholar] [CrossRef] [PubMed]
- Wong, D.V.T.; Lima-Júnior, R.C.P.; Carvalho, C.B.M.; Borges, V.F.; Wanderley, C.W.S.; Bem, A.X.C.; Leite, C.A.V.G.; Teixeira, M.A.; Batista, G.L.P.; Silva, R.L.; et al. The Adaptor Protein Myd88 Is a Key Signaling Molecule in the Pathogenesis of Irinotecan-Induced Intestinal Mucositis. PLoS ONE 2015, 10, e0139985. [Google Scholar] [CrossRef] [PubMed]
- Wong, D.V.T.; Holanda, R.B.F.; Cajado, A.G.; Bandeira, A.M.; Pereira, J.F.B.; Amorim, J.O.; Torres, C.S.; Ferreira, L.M.M.; Lopes, M.H.S.; Oliveira, R.T.G.; et al. TLR4 Deficiency Upregulates TLR9 Expression and Enhances Irinotecan-Related Intestinal Mucositis and Late-Onset Diarrhoea. Br. J. Pharmacol. 2021, 178, 4193–4209. [Google Scholar] [CrossRef] [PubMed]
- van Vliet, M.J.; Harmsen, H.J.M.; de Bont, E.S.J.M.; Tissing, W.J.E. The Role of Intestinal Microbiota in the Development and Severity of Chemotherapy-Induced Mucositis. PLoS Pathog. 2010, 6, e1000879. [Google Scholar] [CrossRef] [PubMed]
- Newton, K.; Dixit, V.M. Signaling in Innate Immunity and Inflammation. Cold Spring Harb. Perspect. Biol. 2012, 4, a006049. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.S.; Ryan, E.J.; Doherty, G.A. Gastro-Intestinal Toxicity of Chemotherapeutics in Colorectal Cancer: The Role of Inflammation. World J. Gastroenterol. 2014, 20, 3751–3761. [Google Scholar] [CrossRef] [PubMed]
- Lima-Júnior, R.C.P.; Freitas, H.C.; Wong, D.V.T.; Wanderley, C.W.S.; Nunes, L.G.; Leite, L.L.; Miranda, S.P.; Souza, M.H.L.P.; Brito, G.A.C.; Magalhães, P.J.C.; et al. Targeted Inhibition of IL-18 Attenuates Irinotecan-Induced Intestinal Mucositis in Mice. Br. J. Pharmacol. 2014, 171, 2335–2350. [Google Scholar] [CrossRef] [PubMed]
- Arifa, R.D.N.; Madeira, M.F.M.; De Paula, T.P.; Lima, R.L.; Tavares, L.D.; Menezes-garcia, Z. Inflammasome Activation Is Reactive Oxygen Species Dependent and Mediates Irinotecan-Induced Mucositis through IL-1 b and IL-18 in Mice. Am. J. Pathol. 2014, 184, 2023–2034. [Google Scholar] [CrossRef] [PubMed]
- Wong, D.V.T.; Ribeiro-Filho, H.V.; Wanderley, C.W.S.; Leite, C.A.V.G.; Lima, J.B.; Assef, A.N.B.; Cajado, A.G.; Batista, G.L.P.; González, R.H.; Silva, K.O.; et al. SN-38, the Active Metabolite of Irinotecan, Inhibits the Acute Inflammatory Response by Targeting Toll-like Receptor 4. Cancer Chemother. Pharmacol. 2019, 84, 287–298. [Google Scholar] [CrossRef] [PubMed]
- Secombe, K.R.; Crame, E.E.; Tam, J.S.Y.; Wardill, H.R.; Gibson, R.J.; Coller, J.K.; Bowen, J.M. Intestinal Toll-like Receptor 4 Knockout Alters the Functional Capacity of the Gut Microbiome Following Irinotecan Treatment. Cancer Chemother. Pharmacol. 2022, 89, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yang, M.; Tang, L.; Wang, F.; Huang, S.; Liu, S.; Lei, Y.; Wang, S.; Xie, Z.; Wang, W.; et al. TLR4 Regulates RORγt(+) Regulatory T-Cell Responses and Susceptibility to Colon Inflammation through Interaction with Akkermansia muciniphila. Microbiome 2022, 10, 98. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, C.; Wanderley, C.W.S.; Silva, C.M.S.; Muniz, H.A.; Teixeira, M.A.; Souza, N.R.P.; Cândido, A.G.F.; Falcão, R.B.; Souza, M.H.L.P.; Almeida, P.R.C.; et al. Role of Regulatory T Cells in Irinotecan-Induced Intestinal Mucositis. Eur. J. Pharm. Sci. 2018, 115, 158–166. [Google Scholar] [CrossRef] [PubMed]
- Ohnmacht, C.; Park, J.-H.; Cording, S.; Wing, J.B.; Atarashi, K.; Obata, Y.; Gaboriau-Routhiau, V.; Marques, R.; Dulauroy, S.; Fedoseeva, M.; et al. MUCOSAL IMMUNOLOGY. The Microbiota Regulates Type 2 Immunity through RORγt+ T Cells. Science 2015, 349, 989–993. [Google Scholar] [CrossRef] [PubMed]
- Cervantes-Barragan, L.; Chai, J.N.; Tianero, M.D.; Di Luccia, B.; Ahern, P.P.; Merriman, J.; Cortez, V.S.; Caparon, M.G.; Donia, M.S.; Gilfillan, S.; et al. Lactobacillus reuteri Induces Gut Intraepithelial CD4(+)CD8αα(+) T Cells. Science 2017, 357, 806–810. [Google Scholar] [CrossRef] [PubMed]
- Hepworth, M.R.; Monticelli, L.A.; Fung, T.C.; Ziegler, C.G.K.; Grunberg, S.; Sinha, R.; Mantegazza, A.R.; Ma, H.-L.; Crawford, A.; Angelosanto, J.M.; et al. Innate Lymphoid Cells Regulate CD4+ T-Cell Responses to Intestinal Commensal Bacteria. Nature 2013, 498, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Cerf-Bensussan, N.; Gaboriau-Routhiau, V. The Immune System and the Gut Microbiota: Friends or Foes? Nat. Rev. Immunol. 2010, 10, 735–744. [Google Scholar] [CrossRef] [PubMed]
- Mazmanian, S.K.; Round, J.L.; Kasper, D.L. A Microbial Symbiosis Factor Prevents Intestinal Inflammatory Disease. Nature 2008, 453, 620–625. [Google Scholar] [CrossRef] [PubMed]
- Dalile, B.; Van Oudenhove, L.; Vervliet, B.; Verbeke, K. The Role of Short-Chain Fatty Acids in Microbiota-Gut-Brain Communication. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 461–478. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Wu, W.; Liu, Z.; Cong, Y. Microbiota Metabolite Short Chain Fatty Acids, GPCR, and Inflammatory Bowel Diseases. J. Gastroenterol. 2017, 52, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Visekruna, A.; Luu, M. The Role of Short-Chain Fatty Acids and Bile Acids in Intestinal and Liver Function, Inflammation, and Carcinogenesis. Front. Cell Dev. Biol. 2021, 9, 703218. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhang, H.; Chen, T.; Shi, L.; Wang, D.; Tang, D. Regulatory Role of Short-Chain Fatty Acids in Inflammatory Bowel Disease. Cell Commun. Signal. 2022, 20, 64. [Google Scholar] [CrossRef] [PubMed]
- Venkatraman, A.; Ramakrishna, B.S.; Pulimood, A.B. Butyrate Hastens Restoration of Barrier Function after Thermal and Detergent Injury to Rat Distal Colon in Vitro. Scand. J. Gastroenterol. 1999, 34, 1087–1092. [Google Scholar] [CrossRef] [PubMed]
- Kelly, D.; Campbell, J.I.; King, T.P.; Grant, G.; Jansson, E.A.; Coutts, A.G.P.; Pettersson, S.; Conway, S. Commensal Anaerobic Gut Bacteria Attenuate Inflammation by Regulating Nuclear-Cytoplasmic Shuttling of PPAR-Gamma and RelA. Nat. Immunol. 2004, 5, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Pandey, S.; Kawai, T.; Akira, S. Microbial Sensing by Toll-like Receptors and Intracellular Nucleic Acid Sensors. Cold Spring Harb. Perspect. Biol. 2014, 7, a016246. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Wen, X.-S.; Xian, C.J. Chemotherapy-Induced Intestinal Microbiota Dysbiosis Impairs Mucosal Homeostasis by Modulating Toll-like Receptor Signaling Pathways. Int. J. Mol. Sci. 2021, 22, 9474. [Google Scholar] [CrossRef] [PubMed]
- Buela, K.-A.G.; Omenetti, S.; Pizarro, T.T. Cross-Talk between Type 3 Innate Lymphoid Cells and the Gut Microbiota in Inflammatory Bowel Disease. Curr. Opin. Gastroenterol. 2015, 31, 449–455. [Google Scholar] [CrossRef] [PubMed]
- Crellin, N.K.; Trifari, S.; Kaplan, C.D.; Satoh-Takayama, N.; Di Santo, J.P.; Spits, H. Regulation of Cytokine Secretion in Human CD127(+) LTi-like Innate Lymphoid Cells by Toll-like Receptor 2. Immunity 2010, 33, 752–764. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Mumm, J.B.; Herbst, R.; Kolbeck, R.; Wang, Y. IL-22 Increases Permeability of Intestinal Epithelial Tight Junctions by Enhancing Claudin-2 Expression. J. Immunol. 2017, 199, 3316–3325. [Google Scholar] [CrossRef] [PubMed]
- Yeung, C.-Y.; Chan, W.-T.; Jiang, C.-B.; Cheng, M.-L.; Liu, C.-Y.; Chang, S.-W.; Chiang Chiau, J.-S.; Lee, H.-C. Amelioration of Chemotherapy-Induced Intestinal Mucositis by Orally Administered Probiotics in a Mouse Model. PLoS ONE 2015, 10, e0138746. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Chiang Chiau, J.-S.; Cheng, M.-L.; Chan, W.-T.; Jiang, C.-B.; Chang, S.-W.; Yeung, C.-Y.; Lee, H.-C. SCID/NOD Mice Model for 5-FU Induced Intestinal Mucositis: Safety and Effects of Probiotics as Therapy. Pediatr. Neonatol. 2019, 60, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Quintanilha, M.F.; Miranda, V.C.; Souza, R.O.; Gallotti, B.; Cruz, C.; Santos, E.A.; Alvarez-Leite, J.I.; Jesus, L.C.L.; Azevedo, V.; Trindade, L.M.; et al. Bifidobacterium Longum Subsp. Longum 5(1A) Attenuates Intestinal Injury against Irinotecan-Induced Mucositis in Mice. Life Sci. 2022, 289, 120243. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.-W.; Lee, H.-C.; Li, L.-H.; Chiang Chiau, J.-S.; Wang, T.-E.; Chuang, W.-H.; Chen, M.-J.; Wang, H.-Y.; Shih, S.-C.; Liu, C.-Y.; et al. Fecal Microbiota Transplantation Prevents Intestinal Injury, Upregulation of Toll-Like Receptors, and 5-Fluorouracil/Oxaliplatin-Induced Toxicity in Colorectal Cancer. Int. J. Mol. Sci. 2020, 21, 386. [Google Scholar] [CrossRef] [PubMed]
- Wardill, H.R.; van der Aa, S.A.R.; da Silva Ferreira, A.R.; Havinga, R.; Tissing, W.J.E.; Harmsen, H.J.M. Antibiotic-Induced Disruption of the Microbiome Exacerbates Chemotherapy-Induced Diarrhoea and Can Be Mitigated with Autologous Faecal Microbiota Transplantation. Eur. J. Cancer 2021, 153, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Nobre, L.M.S.; da Silva Lopes, M.H.; Geraix, J.; Cajado, A.G.; Silva, J.M.R.; Ribeiro, L.R.; Freire, R.S.; Cavalcante, D.I.M.; Wong, D.V.T.; Alves, A.P.N.N.; et al. Paraprobiotic Enterococcus faecalis EC-12 Prevents the Development of Irinotecan-Induced Intestinal Mucositis in Mice. Life Sci. 2022, 296, 120445. [Google Scholar] [CrossRef] [PubMed]
- Nobre, L.M.S.; Fernandes, C.; Florêncio, K.G.D.; Alencar, N.M.N.; Wong, D.V.T.; Lima-Júnior, R.C.P. Could Paraprobiotics Be a Safer Alternative to Probiotics for Managing Cancer Chemotherapy-Induced Gastrointestinal Toxicities? Braz. J. Med. Biol. Res. 2023, 55, e12522. [Google Scholar] [CrossRef] [PubMed]
- Mego, M.; Chovanec, J.; Vochyanova-Andrezalova, I.; Konkolovsky, P.; Mikulova, M.; Reckova, M.; Miskovska, V.; Bystricky, B.; Beniak, J.; Medvecova, L.; et al. Prevention of Irinotecan Induced Diarrhea by Probiotics: A Randomized Double Blind, Placebo Controlled Pilot Study. Complement. Ther. Med. 2015, 23, 356–362. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; He, J.; Jia, W. The Influence of Gut Microbiota on Drug Metabolism and Toxicity. Expert Opin. Drug Metab. Toxicol. 2016, 12, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Chamseddine, A.N.; Ducreux, M.; Armand, J.-P.; Paoletti, X.; Satar, T.; Paci, A.; Mir, O. Intestinal Bacterial β-Glucuronidase as a Possible Predictive Biomarker of Irinotecan-Induced Diarrhea Severity. Pharmacol. Ther. 2019, 199, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Takasuna, K.; Hagiwara, T.; Hirohashi, M.; Kato, M.; Nomura, M.; Nagai, E.; Yokoi, T.; Kamataki, T. Involvement of Beta-Glucuronidase in Intestinal Microflora in the Intestinal Toxicity of the Antitumor Camptothecin Derivative Irinotecan Hydrochloride (CPT-11) in Rats. Cancer Res. 1996, 56, 3752–3757. [Google Scholar] [PubMed]
- Kehrer, D.F.; Sparreboom, A.; Verweij, J.; de Bruijn, P.; Nierop, C.A.; van de Schraaf, J.; Ruijgrok, E.J.; de Jonge, M.J. Modulation of Irinotecan-Induced Diarrhea by Cotreatment with Neomycin in Cancer Patients. Clin. Cancer Res. 2001, 7, 1136–1141. [Google Scholar]
- Pedroso, S.H.S.P.; Vieira, A.T.; Bastos, R.W.; Oliveira, J.S.; Cartelle, C.T.; Arantes, R.M.E.; Soares, P.M.G.; Generoso, S.V.; Cardoso, V.N.; Teixeira, M.M.; et al. Evaluation of Mucositis Induced by Irinotecan after Microbial Colonization in Germ-Free Mice. Microbiology 2015, 161, 1950–1960. [Google Scholar] [CrossRef] [PubMed]
- Vermes, A.; Kuijper, E.J.; Guchelaar, H.-J.; Dankert, J. An in Vitro Study on the Active Conversion of Flucytosine to Fluorouracil by Microorganisms in the Human Intestinal Microflora. Chemotherapy 2003, 49, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Alexander, J.L.; Wilson, I.D.; Teare, J.; Marchesi, J.R.; Nicholson, J.K.; Kinross, J.M. Gut Microbiota Modulation of Chemotherapy Efficacy and Toxicity. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Viaud, S.; Saccheri, F.; Mignot, G.; Yamazaki, T.; Daillère, R.; Hannani, D.; Enot, D.P.; Pfirschke, C.; Engblom, C.; Pittet, M.J.; et al. The Intestinal Microbiota Modulates the Anticancer Immune Effects of Cyclophosphamide. Science (1979) 2013, 342, 971–976. [Google Scholar] [CrossRef]
- Berman, H.M.; Westbrook, J.; Feng, Z.; Gilliland, G.; Bhat, T.N.; Weissig, H.; Shindyalov, I.N.; Bourne, P.E. The Protein Data Bank. Nucleic Acids Res. 2000, 28, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Oriá, R.B.; Empadinhas, N.; Malva, J.O. Editorial: Interplay Between Nutrition, the Intestinal Microbiota and the Immune System. Front. Immunol. 2020, 11, 1758. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernandes, C.; Miranda, M.C.C.; Roque, C.R.; Paguada, A.L.P.; Mota, C.A.R.; Florêncio, K.G.D.; Pereira, A.F.; Wong, D.V.T.; Oriá, R.B.; Lima-Júnior, R.C.P. Is There an Interplay between Environmental Factors, Microbiota Imbalance, and Cancer Chemotherapy-Associated Intestinal Mucositis? Pharmaceuticals 2024, 17, 1020. https://doi.org/10.3390/ph17081020
Fernandes C, Miranda MCC, Roque CR, Paguada ALP, Mota CAR, Florêncio KGD, Pereira AF, Wong DVT, Oriá RB, Lima-Júnior RCP. Is There an Interplay between Environmental Factors, Microbiota Imbalance, and Cancer Chemotherapy-Associated Intestinal Mucositis? Pharmaceuticals. 2024; 17(8):1020. https://doi.org/10.3390/ph17081020
Chicago/Turabian StyleFernandes, Camila, Mahara Coelho Crisostomo Miranda, Cássia Rodrigues Roque, Ana Lizeth Padilla Paguada, Carlos Adrian Rodrigues Mota, Katharine Gurgel Dias Florêncio, Anamaria Falcão Pereira, Deysi Viviana Tenazoa Wong, Reinaldo Barreto Oriá, and Roberto César Pereira Lima-Júnior. 2024. "Is There an Interplay between Environmental Factors, Microbiota Imbalance, and Cancer Chemotherapy-Associated Intestinal Mucositis?" Pharmaceuticals 17, no. 8: 1020. https://doi.org/10.3390/ph17081020
APA StyleFernandes, C., Miranda, M. C. C., Roque, C. R., Paguada, A. L. P., Mota, C. A. R., Florêncio, K. G. D., Pereira, A. F., Wong, D. V. T., Oriá, R. B., & Lima-Júnior, R. C. P. (2024). Is There an Interplay between Environmental Factors, Microbiota Imbalance, and Cancer Chemotherapy-Associated Intestinal Mucositis? Pharmaceuticals, 17(8), 1020. https://doi.org/10.3390/ph17081020








