Metformin and Breast Cancer: Current Findings and Future Perspectives from Preclinical and Clinical Studies
Abstract
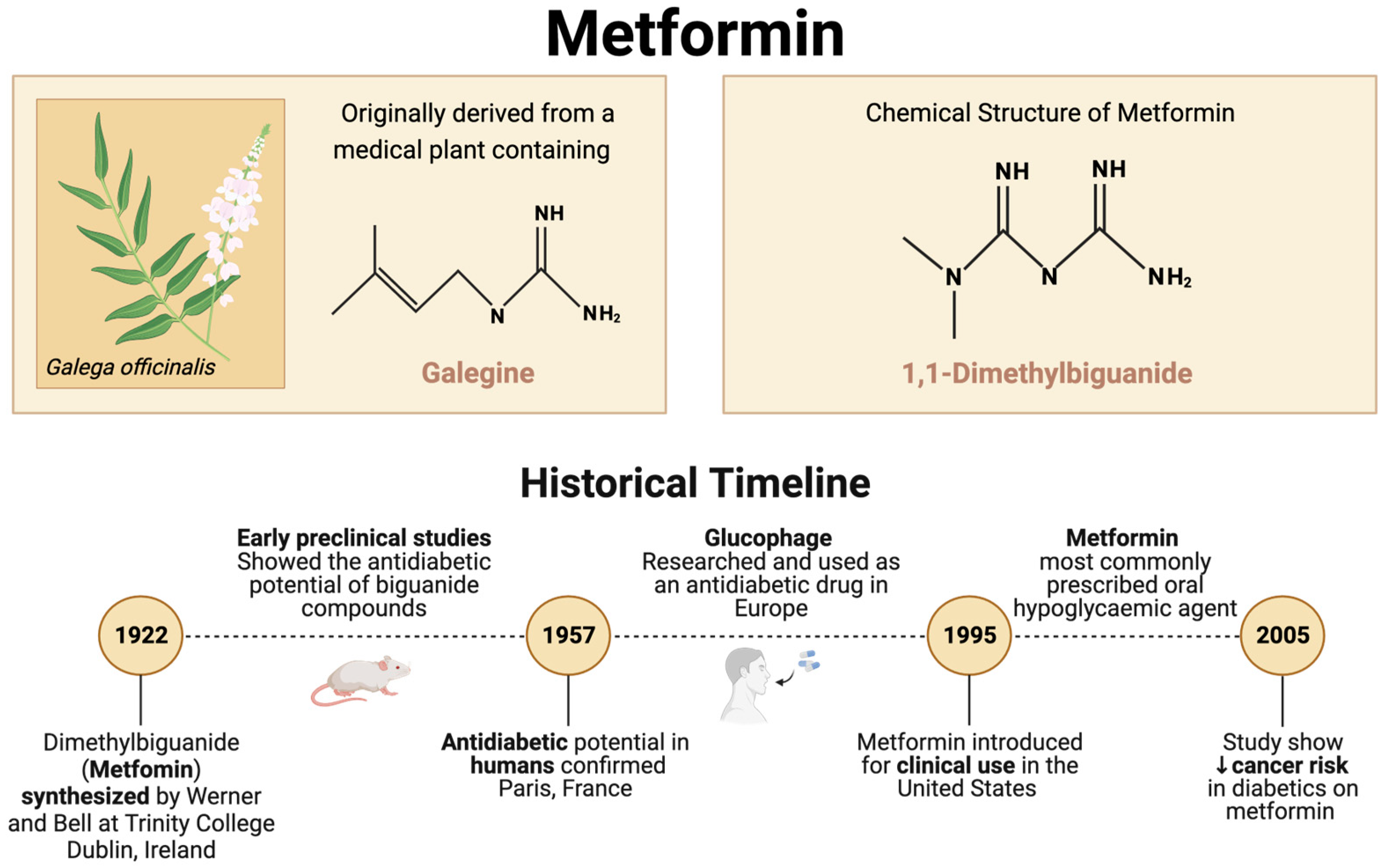
1. Introduction
2. Preclinical Evidence: Findings from In Vitro and In Vivo Models
2.1. Mechanisms of Action
2.2. Metformin: Impact on Breast Cancer Outcomes in Preclinical Models
2.3. Metformin: Anticancer Effects Are Influenced by Glycemic Status/Metabolic Status
2.4. Metformin Dose Modulates Cancer Prevention and Treatment Efficacy
2.5. Membrane Transporters Determine Metformin Efficacy
| Study | Model | Metformin Treatment | Impact of Metformin |
|---|---|---|---|
| Anisimov et al., 2005 [31] | Female Transgenic FVB/N mice carrying HER-2/neu mammary cancer gene | Dose: 1200 mg/L in drinking water; 5 d/wk Duration: from 2 months of age to natural death | ↑ life span by 8% vs. control ↑ tumor latency vs. control ↓ mean tumor diameter vs. control |
| Checkley et al., 2017 [32] | Female Wistar rats MNU-induced ER+ mammary tumors Diet: high-fat (45% kcal fat) | Dose: 2 mg/mL in drinking water Duration: 8 wks | 2/3 of tumors had ↓ size ↓ tumor proliferative index (Ki67) |
| Giles et al., 2018 [33] | Female Wistar rats; Ovariectomized MNU-induced ER+ mammary tumors Diet: high-fat (45% kcal fat) | Dose: 2 mg/mL in the drinking water Duration: 8 wks | ↓ tumor size vs. control ↓ new tumor formation ↓ aromatase+, CD68+ MΦ in tumor microenvironment vs. control + D4:D6 |
| Song et al., 2023 [35] | BALB/c-nu nude mice with MDA-MB-231 cells to form tumors | Dose: 22 mM Duration: 7 days | ↓ tumor size & weight vs. control |
| Thompson et al., 2015 [36] | Model 1: Female Sprague Dawley rats MNU-induced ER+ mammary tumors Diet: low fat (8% kcal fat) | Doses (2): 50 or 150 mg/kg BW/d (gavage) Duration: 121 days | No significant effect on tumor outcomes vs. controls |
| Model 2: MMTV-Neu+/−/p53 KO+/− mouse model (ER- mammary tumors) Diet: low fat (8% kcal fat) | Dose: 1500 mg/kg diet Duration: 60 days of age until ~11 months of age | No significant effect on tumor outcomes vs. controls | |
| Model 3: ER- orthotopic mammary tumors (cells from C3(1)Tag tumor–bearing mice implanted in female SCID mice) | Doses (2): 100 or 150 mg/kg BW/d (i.p. injection) Duration: once tumors reached 125 mg, daily for 14 d | Neither dose significantly inhibited the tumor growth | |
| Zhu et al., 2015 [37] | Female Sprague Dawley rats MNU-induced ER+ mammary tumors Diet: AIN-93G | Dose: 9.3 mmol/kg diet Duration: ~6 wks (started MET 1 wk after carcinogen) | No significant effect on tumor outcomes vs. controls |
| Bojkova et al., 2009 [38] | Female Sprague Dawley rats MNU-induced ER+ mammary tumors | Dose1: 50 μg/mL Dose2: 500 μg/mL Duration: ~20 wks | No significant effect on tumor outcomes vs. controls |
| Zhu et al., 2011 [42] | Female Sprague Dawley rats MNU-induced ER+ mammary tumors Diet: AIN-93G ad libitum or 40% calorie restriction (Expt 3 only) | Experiment 1 Dose1: loading 0.5%, then 0.05% Dose2: loading 1.0%, then 0.25% (w/w) in the diet Duration: 5 d loading + 28 d maintenance dose | Dose1: No significant effect on tumor outcomes vs. controls Dose2: ↓ tumor weight & multiplicity ↑ tumor latency vs. control |
| Experiment 2 Dose: 0.3% (w/w) in the diet Duration: started 7 d post carcinogen, for 9 wks | No significant effect on tumor outcomes vs. controls, suggesting that early events in the carcinogenic process are more susceptible to high dose metformin | ||
| Experiment 3 Dose: 0.25% (w/w) + 40% calorie restriction Duration1: 10 wks Duration2: 8 wks treatment, last 2 weeks of 10 wks removed from 0.25% (w/w) + 40% calorie restriction | 40% caloric restriction (CR) alone or combined with MET were equally effective in inhibiting mammary carcinogenesis ↓ tumor weight & multiplicity vs. controls ↑ tumor latency vs. control Stopping both CR & MET: - retained benefits on preventing new tumors - lost benefit on suppressing growth of existing tumors | ||
| Zhuang et al., 2014 [43] | Female Balb/C mice injected with 4T1 cells Diet: control (24% kcal fat) vs. ketogenic (4.6% pr, 93.4% fat, 2% cho—also calorically restricted) | Dose: 2 mg/d (i.p) Duration: ~2 wks | Control diet: MET had no effect on tumor volume Ketogenic diet: ↓ tumor volume vs. control MET + ketogenic diet: ↓ tumor volume vs. control & vs. ketogenic diet alone |
| Marini et al., 2016 [44] | Female BALB/c mice + syngeneic 4T1 cells (TNBC) Diet: chow (12% fat) +/− short-term starvation (2 × 48 h periods on days 5–7 & 12–14) | Dose: 3 mg/mL in the drinking water Duration: 14 days | MET ↓ tumor volume vs. chow control Short-term starvation + MET further ↓ tumor growth |
| Zhu et al., 2014 [50] | Female FVB/N-Tg MMTV-ErbB2 transgenic mice Diet: estrogen-free AIN-93G diet | Premalignant stage experiment Dose: 250 mg/kg injected i.p. Duration: 10 weeks | ↓ lateral branching & alveolar structures ↓ CD61high/CD49fhigh tumor-initiating cells |
| Syngeneic grafting of MET pretreated 78617 tumor cells (derived from MMTV-ErbB2 tumors) into MMTV-ErbB2 mice | In vitro pretreatment + tumor grafts experiment Dose: 1 mmol/L in the media (in vitro) Duration: 72 h Tumors monitored for 14 d post injection | MET pretreatment ↓ tumor volume | |
| Barbieri et al., 2015 [51] | Female NOD-SCID mice (non-obese diabetic severe combined immunodeficient) injected with cancer stem cell like cells from mammary canine tumors. | Dose: 360 mg/Kg BW/day in the drinking water Duration: 6 months | ↓ tumor weight ↓ tumor Ki-67 & mitotic index |
| Study | Glucose | Metformin | Outcomes |
|---|---|---|---|
| Wahdan-Alaswad et al., 2013 [39] | 5 mM 10 mM 17 mM | 5 mM | Most cell lines: ↓ proliferation |
| 5 mM 10 mM 17 mM | 10 mM | ↓ proliferation in TNBC but no other cell lines | |
| Varghese et al., 2019 [40] | 5.5 mM | 25–100 µM | no change: MDA-MB-231 no change: MDA-MB-468 |
| 500 µM–10 mM | ↓ proliferation: MDA-MB-231: ↓ proliferation: MDA-MB-468: | ||
| 25 mM | 25 µM | no change: MDA-MB-231 no change: MDA-MB-468 | |
| 100 µM | ↑ proliferation: MDA-MB-231 ↑ proliferation: MDA-MB-468 | ||
| 250, 500 uM | ↑ proliferation: MDA-MB-231 ↓ proliferation: MDA-MB-468 | ||
| 1–10 mM | no change: MDA-MB-231 ↓ proliferation: MDA-MB-468 | ||
| Zhu et al., 2011 [42] | Not stated | 0.02–0.2 mM | no change: MDA-MB-468 |
| 0.02–1 mM | no change: MCF7 | ||
| 0.02–2 mM | no change: BT-20 no change: MDA-MB-453 | ||
| 0.02–5 mM | no change: BT-549 no change: MDA-MB-231 | ||
| 0.02–10 mM | No change: SK-BR-3 | ||
| 1–20 mM | ↓ proliferation: MDA-MB-468 | ||
| 2–20 mM | ↓ proliferation: MCF7 | ||
| 5–20 mM | ↓ proliferation: BT-20 ↓ proliferation: MDA-MB-453 | ||
| 10–20 mM | ↓ proliferation: BT-549 ↓ proliferation: MDA-MB-231 | ||
| 20 mM | ↓ proliferation: SK-BR-3 |
2.6. Metformin and Breast Cancer Stem Cells
2.7. Metformin: Modulation of microRNAs and Long Non-Coding RNAs in Breast Cancer
2.7.1. MicroRNAs (miRNAs)
2.7.2. Long Non-Coding RNAs
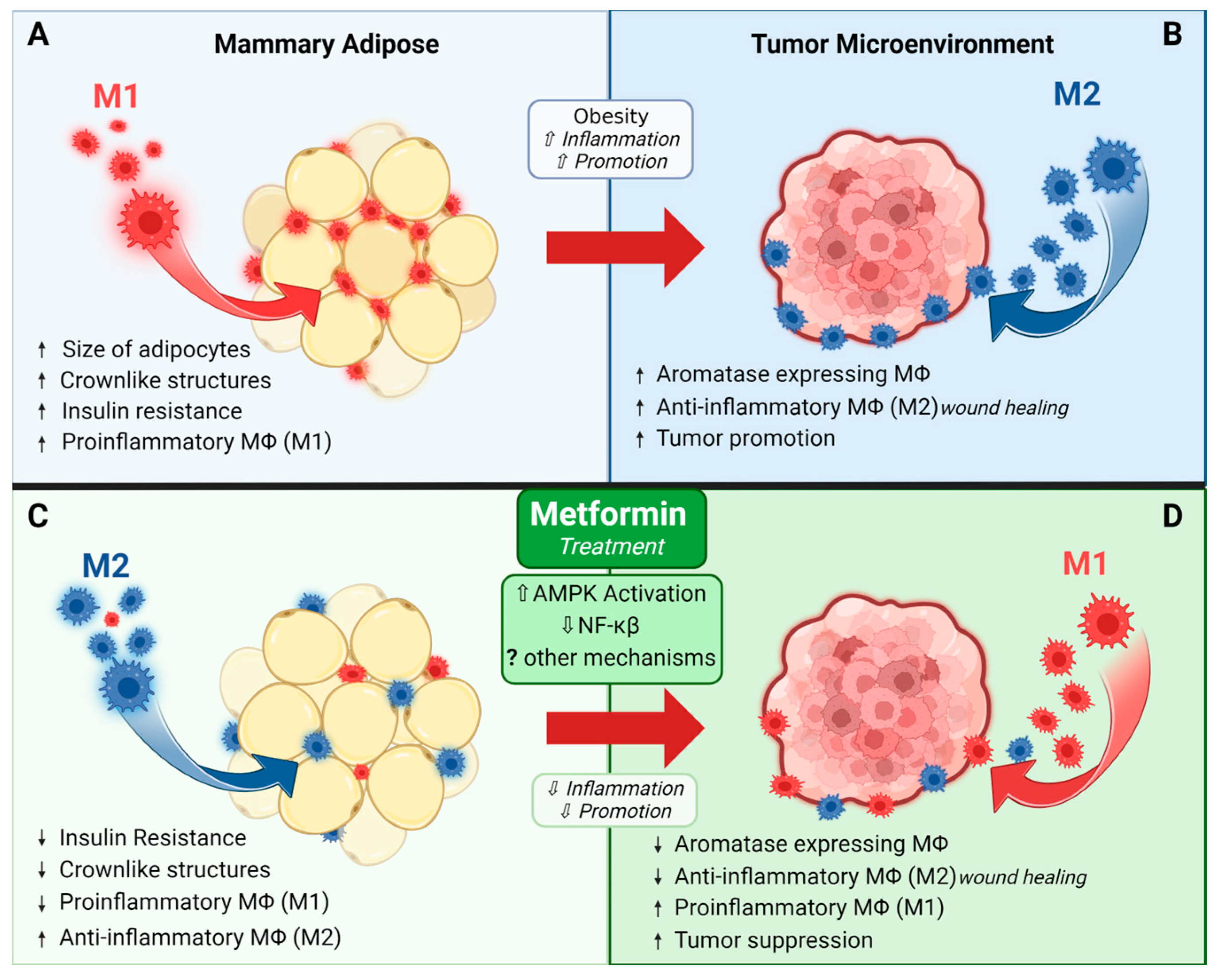
2.8. Metformin and Immune Modulation
3. Translating Metformin to the Clinic
3.1. Epidemiological Evidence
3.2. Clinical Findings: Window of Opportunity Trials
| Study Type | ID ClinicalTrials.gov | Population/Cancer Details | Intervention(s) | Primary Outcome(s) | Related Publications | Key Findings |
|---|---|---|---|---|---|---|
| Completed Studies | ||||||
| RCT | NCT01302379 | Postmenopausal breast cancer survivors w/BMI > 25 kg/m2 | 6-month intervention (4 groups) (1) placebo + lifestyle intervention (2) MET + lifestyle intervention (3) placebo + standard printed dietary guidelines (4) MET + standard printed dietary guidelines MET dosing: wk 1: 500 mg/day (PM) wks 2–4: 1000 mg/day (PM) wks 5+: 1500 mg/day (500 mg AM + 1000 mg PM) | Change (baseline to 6 mos) for: - insulin, - glucose, - C-reactive protein, - bioavailable testosterone, - sex hormone binding globulin | Nwanaji-Enwerem et al., 2021 [110], Bellerba et al., 2022 [111] | MET + Lifestyle Intervention ↓ insulin −21.8% (CI −29.7 to −13.0) ↓ C-reactive protein −21.4% (CI −38.9 to 1.0) MET + Standard Dietary Guidelines ↓ insulin −13.2% (CI −21.7 to −3.7) ↓ C-reactive protein −9.2% (CI −29.0 to 16.1) |
| Phase 2 | NCT00930579 | Newly diagnosed early invasive BC | Pre-surgical intervention MET 1500 mg/day for a minimum of 2 wks prior to surgery | Kalinsky et al., 2014 [107], Kalinsky et al., 2017 [112] | No change in tumor proliferation (Ki-67) ↓ BMI, cholesterol, and leptin MET modulated proteins involved in apoptosis/cell cycle, cell signaling, & invasion/motility, including: - ↑ tumor Raptor, C-Raf, Cyclin B1, Cyclin D1, TRFC, and Syk - ↓ tumor pMAPKpT202, Y204, JNKpT183, pT185, BadpS112, PKC.alphapS657, and SrcpY416 | |
| NCT02028221 | Premenopausal w/BMI > 25 kg/m2 and metabolic syndrome | BC prevention 12-mo intervention (1) Placebo (2) MET wks 1–4 850 mg/day, wks 5 + 1700 mg/day | Change in breast density at 6 and 12 mos | Martinez et al., 2016 [113], Tapia et al., 2021 [114] | ↓ waist circumference and waist-to-hip ratio No change in % breast density or dense breast volume Non-significant (p = 0.07).↓ in non-dense breast volume | |
| NCT01310231 | Metastatic or unresectable locally advanced BC on 1st-4th line chemotherapy w/anthracycline, taxane, platinum, capecitabine or vinorelbine based regimens w/o diabetes | Intervention continued until disease progression (1) placebo + standard chemotherapy (2) MET 850 mg/2× day + standard chemotherapy | Progression-free survival | Pimentel et al., 2019 [115] | No effect on progression-free survival, overall survival, and response rate | |
| NCT02431676 | Individuals that have survived solid malignant tumors including breast, prostate, lung, colon, skin melanoma, endometrial, liver, pancreatic, rectal, kidney, other solid malignant tumors. | Secondary prevention study evaluating the effect of the interventions on IGF-1 (1) Self-directed weight loss (2) Coach-directed behavioral weight loss (3) MET up to 2000 mg/day | IGF-1 at 6 mos IGF1/IGFBP3 molar ratio levels at 6 and 12 mos | Juraschek et al., 2018 [116], Mueller et al., 2021 [117], Hu et al., 2021 [118], Tilves et al., 2022a [119], Tilves et al., 2022b [120] | ↓ BMI ↑ butyrate, acetate, and valerate at 6 months Altered microbiota composition: ↑ Escherichia coli and Ruminococcus torques, ↓ Intestinibacter bartlettii, R. faecis and R. intestinalis | |
| Phase 3 | NCT01101438 | Women w/o diabetes w/high-risk nonmetastatic BC | 5 Year Intervention (1) Placebo + standard treatment (2) MET + standard treatment MET dosing: wks 1–4 850 mg/day, wks 5+ 850 mg/2× day | Invasive disease-free survival | Goodwin et al., 2022 [121], Goodwin et al., 2023 [122] | No change in invasive disease-free survival or the risk of developing new cancers. |
| Window of opportunity | NCT00897884 | w/o diabetes w/newly diagnosed untreated BC | Window-of-opportunity neoadjuvant study, intervention 2–3 wks prior to surgical removal of tumor MET 500 mg/3× day | Tumor proliferation rate (pre vs. post treatment) | Dowling et al., 2015 [123] | ↓ insulin receptor expression in tumors ↓ phosphorylation status of protein kinase B (PKB)/Akt (S473), extracellular signal-regulated kinase 1/2 (ERK1/2, T202/Y204), AMPK (T172) and acetyl coenzyme A carboxylase (S79) in tumors |
| Phase 2 | NCT01340300 | Stage I-III CRC & BC survivors, >2 mo from completing standard therapy (excluding hormone rx or trastuzumab) | Randomized to 12 wks of: (1) Control educational materials (2) MET (3) Exercise (4) MET + Exercise MET dosing: wks 1–2 1/day, wks 3+ 2/day Exercise: Training w/exercise physiologist 2× wk | Change in fasting insulin (baseline to 3 mos) | Brown et al., 2020 [124], Meyerhardt et al., 2020 [125] | MET ↓ insulin from baseline −1.16 mU/L ± 1.18 ↓ leptin from baseline −2.56 ng/mL ± 1.33 ↓ IGF1 from baseline −2.66 ng/mL ± 3.28 MET + Exercise ↓ insulin from baseline −2.47 mU/L ± 1.07 ↓ leptin from baseline −5.09 ng/mL ± 1.21 ↓ IGF1 from baseline −1.29 ng/mL ± 2.98 |
| Early Phase 1 | NCT01980823 | Newly diagnosed operable invasive BC or DCIS No prior treatment | Window-of-opportunity intervention ~2 wks prior to surgical removal of tumor MET 1500 mg/day + Atorvastatin 80 mg/day | Change in tumor Ki-67 (proliferation; baseline to 2 wks) | N/A | No results reported. |
| NCT01793948 | Postmenopausal w/elevated risk for breast cancer w/BMI ≥ 25 w/o diabetes | BC prevention study MET (850 mg/ 2× day) for 12 cycles vs. placebo | Changes in protein phosphorylation after MET exposure from baseline to 12 mos | N/A | No results reported. | |
| Phase 1 | NCT02882581 | Adults >50 years w/BC | Radiation: 11C-metformin | MET uptake in BC | N/A | No results reported. |
| NCT01650506 | TNBC w/o diabetes | Single arm phase 1 study Intervention: Erlotinib + Metformin MET dosing: Dose titrated from 850 mg/2× day to 850 mg/3× day Erlotinib dosing: 150 mg/day | Max tolerated dose of MET in combination with a 150 mg erlotinib/day | N/A | No results reported. | |
| NCT02278965 | Pre- and postmenopausal w/history of early stage BC w/BMI ≥ 25 w/o diabetes | Single group assignment, intervention for 12 mos MET 850 mg/2× day + Omega-3 fatty acids 560 mg/2× day | # of participants completing the 1-year intervention | N/A | No results reported. | |
| NCT00933309 | Postmenopausal w/history HR+ BC and evidence metastasis w/BMI ≥ 25 | Duration: as long as the disease is stable and/or responding (1) Exemestane 25 mg/day (2) Exemestane 25 mg/day + Avandamet (MET 500 mg + Rosiglitazone 2 mg)/day | Dose-limiting toxicity | N/A | No results reported. | |
| NCT00659568 | Metastatic or unresectable solid tumor (breast, endometrial, kidney, lung, unspecified) or lymphoma | Determine max tolerated dose of MET when administered with temsirolimus Intervention: MET + temsirolimus | Max tolerated and recommended dose of MET when administered w/temsirolimus | N/A | No results reported. | |
| NCT02145559 | Adults w/solid tumors that is metastatic or unresectable and standard or palliative measures are not an option (breast, lung, liver, lymphoma, kidney) | Evaluate the pharmacodynamic markers sirolimus + metformin therapy (1) MET XR up to 1000 mg/day + Sirolimus (2) Delayed MET (no MET for 2 wks then titrated up to 1000 mg/day) + Sirolimus | Pharmacodynamic biomarker p70S6K | N/A | No results reported. | |
| Phase 2 | NCT01266486 | Early stage BC w/o diabetes | Single group assignment Intervention (14–21 days): Extended-release MET 1500 mg/day | Phosphorylation of S6K, 4E-BP-1 and AMPK | Lord et al., 2018 [126] Lord et al., 2020 [108], Ralli et al., 2022 [127] | ↑ in genes that regulate fatty acid oxidation ↓ mitochondrial metabolites, activates mitochondrial metabolic pathways, and ↑ 18-FDG flux in tumors Tumor heterogeneity: - Mitochondrial response to MET dictates response - Identified OXPHOS transcriptional response (OTR) signature in tumors that were resistant to MET - Tumors that ↑ OXPHOS genes had ↑ proliferation score |
| NCT04143282 | MBC w/o diabetes | Chemotherapy alone vs MET + chemotherapy | Radiologic response rate at 3 mos Overall & progression-free survival at 6 mos | Rabea et al., 2021 [128] | Improved radiologic response, ↓ mortality & ↓ disease progression but overall survival & progression-free survival not significantly affected | |
| NCT04170465 | BC w/o metastasis and w/o diabetes | RCT, intervention 6 mos (1) MET 850 mg/2× day + AC-T neoadjuvant chemotherapy (2) AC-T neoadjuvant chemotherapy alone | Tumor apoptosis and chemotherapy toxicities at 6 mos | Serageldin et al., 2023 [129] | MET + AC-T: ↓ peripheral neuropathy incidence & severity ↓ oral mucositis ↓ fatigue ↓ fatty liver incidence preserved cardiac function | |
| NCT05053841 | Postmenopausal women w/BC w/o diabetes | Parallel assignment, 6-mo intervention, randomized: (1) Control: women w/obesity (n = 15) letrozole alone (2) Women w/obesity (n = 15) letrozole + MET 2000 ± 500 mg/day (3) Women w/o obesity (n = 15) letrozole alone | Change in serum biomarkers from baseline to 6 mos | El-Attar et al., 2023 [130] | ↓ estradiol, leptin, fasting glucose, insulin, osteocalcin serum levels, and HOMA-IR | |
| NCT02488564 | Patients w/operable BC or locally advanced BC that is HER2+ w/o diabetes | Single group assignment, trial duration 36 mos Intervention: Liposomal doxorubicin + Docetaxel + Trastuzumab + MET 1000 mg/2× day | Pathologic complete response rate | N/A | No results reported. | |
| NCT01589367 | Postmenopausal w/ER+ BC w/o diabetes | 1:1 randomized clinical (1) Letrozole 2.5 mg/day + MET (2) Letrozole 2.5 mg/day + placebo MET dosing 1 wk 500 mg/2× day, followed by 1 wk (1000 mg AM|500 mg PM)/day, followed by 22 wks 1000 mg/2× day | Clinical response rate from baseline to 24 wks | Kim et al., 2014 [131] | No results reported. | |
| NCT05351021 | Early stage BC w/o diabetes receiving adjuvant paclitaxel | Randomized, parallel assignment study to test MET as a preventive for paclitaxel-induced peripheral neuropathy (1) MET 850 mg/2× day + adjuvant paclitaxel (2) placebo + adjuvant paclitaxel | Incidence of grade II or > peripheral neuropathy at end of paclitaxel treatment | Bakry et al., 2023 [132] | ↓ paclitaxel-induced peripheral neuropathy ↑ quality of life | |
| NCT00909506 | Women w/operable BC w/overweight or pre-DM | Randomized, parallel assignment, 24 wk intervention (1) placebo (2) MET 500 mg/day. | Weight loss at 6 mos | N/A | No results reported. | |
| NCT01885013 | HER2− MBC w/o diabetes | Randomized, parallel assignment, study 24 mos + 12-mo follow-up (1) MET 1000 mg/2× day + Myocet + Cyclophosphamide (2) Myocet + Cyclophosphamide | Progression-free survival | Gennari et al., 2020 [133] | no effect on IGF-1R expression or circulating tumor cell count | |
| Phase 4 | (breast, lung, liver, lymphoma, kidney) NCT05840068 | MBC w/o diabetes | Randomized, parallel assignment, intervention 6 mos (1) MET 500 mg/2× day + chemotherapy (2) Control chemotherapy alone | IGF-1 levels at 6 mos | N/A | No results reported. |
| Active—Not Recruiting | ||||||
| Phase 2 | NCT04248998 | Stage I-III TNBC w/fasting glucose ≤ 250 mg/dL | Randomized, parallel assignment (1) Chemotherapy + fasting-mimicking diet (2) Chemotherapy + fasting-mimicking diet + MET 1700 mg/day | Rate of pathologic complete responses | N/A | Pathologic complete response is higher in diabetic patients who utilize neoadjuvant chemotherapy and take metformin. |
| NCT04300790 | ER+ and/or PgR+, HER2− advanced BC w/centrally confirmed PI3KCAMut on an aromatase inhibitor | Single Group Assignment (1) Normoglycemic patients: Alpelisib + MET (up to 100 mg/2× day) + endocrine therapy (2) Pre-diabetic patients: Alpelisib + MET (up to 100 mg/2× day) + endocrine therapy (3) Insulin naïve type 2 diabetic patients: Alpelisib + MET (up to 100 mg/2× day) + endocrine therapy | Development of treatment-induced hyperglycemia (Alpelisib + Endocrine Therapy) | N/A | No results reported. | |
| NCT02874430 | Localized breast, uterine, or cervical cancer | Single Group Assignment, intervention 6 wks Doxycycline + MET | Change in % of stromal cells expressing Caveolin-1 at an intensity of 1+ or greater | N/A | No results reported. | |
| Phase 3 | NCT01905046 | Women w/high risk for BC (such as w/atypical hyperplasia, LCIS, DCIS, family history, etc.) | BC prevention, randomized, crossover assignment, (1) MET 850 mg/2× day for 24 mos (2) Placebo 12 mos, may crossover to MET mos 13–24 | Presence or absence of cytological atypia in unilateral or bilateral RPFNA aspirates after 12 and 24 mos | N/A | No results reported. |
| Active—Recruiting | ||||||
| Observational | NCT02695121 | Adults w/type 2 diabetes exposed to dapagliflozin and other antidiabetic treatments (including MET) | Observational cohort study | Incidence of bladder and breast cancer | N/A | No results reported. |
| Phase 1 | NCT03006172 | Locally advanced or metastatic PIK3CA-mutant solid tumors including BC | Non-randomized, sequential assignment (1) Inavolisib single agent (2) Inavolisib + palbociclib + letrozole (3) Inavolisib + letrozole (4) Inavolisib + fulvestrant (5) Inavolisib + palbociclib + fulvestrant (6) Inavolisib + palbociclib + fulvestrant + MET (7) Inavolisib + trastuzumab + pertuzumab | % of participants w/dose limiting toxicities Recommended phase II dose of inavolisib % of participants w/adverse and serious adverse events | N/A | No results reported. |
| Phase 2 | NCT05023967 | Early BC w/o diabetes | Randomized, parallel assignment, intervention 4–6 wks (1) Fasting (≥16 h every night) + continuous glucose monitoring + nutritional counseling + MET extended-release (2) Usual dietary pattern + continuous glucose monitoring | Frequency of dose-limiting toxicity Change cell proliferation pre-post treatment (Ki67) | N/A | No results reported. |
| NCT05660083 | HER2− metaplastic BC and/or TNBC | Single group assignment Combination of an iNOS inhibitor + nab-paclitaxel + alpelisib To prevent deep venous thrombosis & hypertension: aspirin + amlodipine To reduce risk of severe hyperglycemia: MET up to 1000 mg/2× day | Recommended phase II dose of Alpelisib + standard + nab-paclitaxel + L-NMMA Objective response rate | N/A | No results reported. | |
| NCT01042379 | Adults w/BC | I-SPY Trial: Randomized to one of 36 experimental agents (compared to standard therapy) One group contained MET: MET + Ganitumab (AMG 479; an anti-IGF-1R antibody) | Does adding experimental agents to standard neoadjuvant medications ↑ the probability of pathologic complete response over standard neoadjuvant chemotherapy | Wang and Yee 2019 [134], Yee et al., 2021 [135] | Numerous publications, only 1 metformin related Metformin + ganitumab + paclitaxel (PGM) - metformin not sufficient to control drug-induced hyperglycemia | |
| Active—Pre-Recruitment | ||||||
| Early Phase 1 | NCT05680662 | Early BC, MBC, TNBC | Randomized, parallel assignment (1) Adjuvant quadruple therapy (quercetin 500 mg/day + zinc sulfate 50 mg/day + EGCG 300 mg/day + MET 850 mg/day) + standard chemotherapy (2) Control only standard chemotherapy | Invasive disease-free survival | N/A | No results reported. |
| Phase 4 | NCT05507398 | Non-MBC | Randomized, parallel assignment (1) Placebo (2) MET 1000 mg/day (3) Atorvastatin 20 mg/day | Improvement in overall response rate and pathological response | N/A | No results reported. |
| Terminated | ||||||
| Early Phase 1 | NCT01302002 | Early BC w/o diabetes | Metformin pre-surgery, non-randomized, single-group assignment MET 500 mg/2× day for 3 wks | Cell proliferation (Ki67), apoptosis (TUNEL), fosforilate, AKT, CD1a CD83, CD68, F40/80, arginase, iNOS, T cells [CD4(+),CD45RA(+), CD 45RO, CD4, CD8 and FOXP3(+)] | Withdrawn—no enrollment | No results reported. |
| Phase 2 | NCT01477060 | HER2−, ER+ and/or PgR+, MBC w/o diabetes | Randomized, parallel assignment, intervention until disease progression or other circumstance that mitigates termination (1) hormonal therapy + lapatinib 1250 mg/day (2) hormonal therapy + MET 1500 mg/day (3) hormonal therapy + lapatinib 1250 mg/day + MET 1500 mg/day | Progression-free survival | Terminated; low accrual | No results reported. |
| NCT01627067 | Postmenopausal w/BMI ≥ 25 w/HR+ BC and metastatic disease | Single group assignment Everolimus 10 mg/day + Exemestane 25 mg/day + MET 1000 mg/2× day | Progression-free survival (compare between overweight and obese patients) | Terminated; funding issues Yam et al., 2019 [136] | MET + everolimus + exemestane - was safe - moderate clinical benefit in patients with both overweight and obesity - 5/22 = partial response; 7/22 = stable disease for ≥24 weeks --> clinical benefit rate of 54.5% | |
| NCT02360059 | Adults w/invasive BC w/o diabetes to undergo paclitaxel chemotherapy | MET for paclitaxel neurotoxicity, randomized, parallel assignment (1) MET 1000 mg 2× day for 12 wks during paclitaxel treatment (2) Placebo for 12 wks during paclitaxel treatment | Mean change in neuropathy | Terminated; low accrual | No results reported. | |
| NCT02472353 | Adults w/BC w/o diabetes needing neoadjuvant or adjuvant therapy w/doxorubicin | MET to reduce cardiac toxicity in BC, randomized, parallel assignment (1) Standard care w/doxorubicin (2) Standard care w/doxorubicin + MET | # of patients w/≤ 5% ↓ in left ventricle ejection fraction on echocardiogram | Terminated; low accrual | No results reported. | |
| NCT04899349 | Adults w/HR+, HER2− advanced BC | Randomized, parallel assignment (1) Alpelisib + fulvestrant + dapagliflozin + MET XR (500–2000 mg/day) (2) Alpelisib + fulvestrant + MET XR (500–2000 mg/day) | # of patients w/hyperglycemia grade ≥ 3 over the 1st 8 wks of alpelisib + fulvestrant | Terminated; low accrual | No results reported. | |
| Phase 3 | NCT02201381 | Adults w/cancer | Single group assignment Atorvastatin up to 80 mg/day + MET up to 1000 mg/2× day + doxycycline 100 mg/day + mebendazole 100 mg/day | Overall survival | Withdrawn Agrawal et al., 2023 [137] | No MET BC results reported. |
| Phase 4 | NCT04741204 | Non-Hispanic white or black females w/BMI ≥ 25 w/newly diagnosed BC | Non-randomized, single-group assignment, compare outcomes between black & white women MET XR release 750 mg/2× day | Tumor progression | Withdrawn—Staffing issues | No results reported. |
| NA | NCT00984490 | Stage I or II BC that can be surgically removed w/o diabetes | Single group assignment, intervention 7–21 days prior to surgery MET 850 mg/2× day | Change cell proliferation (Ki67) from baseline to post-treatment | Terminated; low accrual | No results reported. |
| Status Unknown | ||||||
| Phase 2 | NCT04559308 | w/o diabetes w/BC receiving neoadjuvant chemotherapy | Randomized, parallel assignment (1) 4 cycles (doxorubicin+cyclophosphamide), then 12 cycles paclitaxel+ MET 1000 mg/2× day, then surgery (2) 4 cycles (doxorubicin + cyclophosphamide), then 12 cycles paclitaxel, then surgery | Clinical benefit rate (tumor size) at 8 mos | N/A | No results reported. |
| NCT03238495 | HER2+ BC | Randomized, parallel assignment (1) Chemotherapy only (TCH + P) (2) Chemotherapy (TCH + P) + MET 850 mg/2× day | Pathologic complete response | N/A | No results reported. | |
| NCT02506777 | Locally advanced BC | Randomized, parallel assignment (1) Conventional chemotherapy—fluoruracil, doxorubicin, cyclophosphamide (FDC) × 6 cycles + MET 850 mg/2× day (2) Conventional chemotherapy FDC × 6 cycles + melatonin 3 mg/day (3) Conventional chemotherapy FDC × 6 cycles | Response rate and pathomorphological response after 6 mos | N/A | No results reported. | |
| NCT03192293 | HR+ HER2− MBC | Single group assignment MET 850 mg/2× day + Simvastatin + Fulvestrant | Clinical benefit rate after 24 wks | N/A | No results reported. | |
| NCT02506790 | ER+ locally advanced BC | Randomized, parallel assignment (1) Toremifene 60 mg/day + MET 850 mg/2× day (2) Toremifene 60 mg/day + melatonin 3 mg/day (3) Toremifene 60 mg/day | Response rate and pathomorphological response after 4 mos | N/A | No results reported. | |
| NCT01566799 | HR+, HER2− BC w/o diabetes | Single group assignment Standard chemotherapy + MET 500 mg/day for 24 wks | Pathologic complete response after 24 wks | N/A | No results reported. | |
| NCT04001725 | Melanoma, lung or BC w/brain metastasis requiring high-dose dexamethasone treatment | Randomized, parallel assignment (1) Dexamethasone min dose of 8 mg/day (2) Dexamethasone min dose of 8 mg/day + MET up to max 2550 mg/day | MET preventing precocious dexamethasone-induced diabetes after 14 days | Green et al., 2022 [138] | No MET BC results reported. | |
| NCT01929811 | BC | Randomized, parallel assignment (1) Standard chemotherapy + MET 500 mg/3× day (2) Standard chemotherapy | Pathologic complete response rate after 5 mos | Huang et al., 2023 [139] | No change in pathological complete response or disease outcome w/MET No difference in proliferation (Ki67) w/MET MET prevented the ↑ in total cholesterol and LDL-C after standard treatment | |
| Phase 2/3 | NCT04387630 | Early BC w/o diabetes | Randomized, parallel assignment (1) MET max 2550 mg/day + standard treatment (2) Placebo + standard treatment | Clinical response rate after 3 mos of therapy | N/A | No results reported. |
| Unknown | NCT01666171 | BC patients concurrently enrolling or previously enrolled in MA.32 study w/breast density ≥ 25% | (1) MET 850 mg/2× day for 5 years (2) placebo | % change mammographic breast density in contralateral (unaffected) breast from baseline to 1 year | N/A | No results reported. |
| NCT01286233 | BC patients eligible for randomization to MA.32 study | (1) MET 850 mg/2× day for 5 years (2) placebo | Patient-reported fatigue, stress, sleep, depression, general quality of life, comorbid conditions, and behavioral risks Biological correlates of fatigue DNA polymorphisms Changes in RNA gene expression | N/A | No results reported. | |
3.3. Has the Translation of Metformin to the Clinic as an Anticancer Agent Failed?
4. Future Perspectives and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Soranna, D.; Scotti, L.; Zambon, A.; Bosetti, C.; Grassi, G.; Catapano, A.; La Vecchia, C.; Mancia, G.; Corrao, G. Cancer risk associated with use of metformin and sulfonylurea in type 2 diabetes: A meta-analysis. Oncologist 2012, 17, 813–822. [Google Scholar] [CrossRef]
- Gong, Z.; Aragaki, A.K.; Chlebowski, R.T.; Manson, J.E.; Rohan, T.E.; Chen, C.; Vitolins, M.Z.; Tinker, L.F.; LeBlanc, E.S.; Kuller, L.H.; et al. Diabetes, metformin and incidence of and death from invasive cancer in postmenopausal women: Results from the women’s health initiative. Int. J. Cancer 2016, 138, 1915–1927. [Google Scholar] [CrossRef]
- Bailey, C.J.; Campbell, I.W.; Chan, J.C.N.; Davidson, J.A.; Howlett, H.C.S.; Ritz, P. Metformin—The Gold Standard: A Scientific Handbook; Wiley: Hoboken, NJ, USA, 2007. [Google Scholar]
- Bailey, C.J. Metformin: Historical overview. Diabetologia 2017, 60, 1566–1576. [Google Scholar] [CrossRef]
- Samuel, S.M.; Varghese, E.; Kubatka, P.; Triggle, C.R.; Büsselberg, D. Metformin: The Answer to Cancer in a Flower? Current Knowledge and Future Prospects of Metformin as an Anti-Cancer Agent in Breast Cancer. Biomolecules 2019, 9, 846. [Google Scholar] [CrossRef]
- Werner, E.A.; Bell, J. CCXIV.—The preparation of methylguanidine, and of ββ-dimethylguanidine by the interaction of dicyanodiamide, and methylammonium and dimethylammonium chlorides respectively. J. Chem. Soc. Trans. 1922, 121, 1790–1794. [Google Scholar] [CrossRef]
- Cejuela, M.; Martin-Castillo, B.; Menendez, J.A.; Pernas, S. Metformin and Breast Cancer: Where Are We Now? Int. J. Mol. Sci. 2022, 23, 2705. [Google Scholar] [CrossRef]
- Evans, J.M.; Donnelly, L.A.; Emslie-Smith, A.M.; Alessi, D.R.; Morris, A.D. Metformin and reduced risk of cancer in diabetic patients. BMJ 2005, 330, 1304–1305. [Google Scholar] [CrossRef] [PubMed]
- Bowker, S.L.; Majumdar, S.R.; Veugelers, P.; Johnson, J.A. Increased cancer-related mortality for patients with type 2 diabetes who use sulfonylureas or insulin. Diabetes Care 2006, 29, 254–258. [Google Scholar] [CrossRef]
- Chlebowski, R.T.; McTiernan, A.; Wactawski-Wende, J.; Manson, J.E.; Aragaki, A.K.; Rohan, T.; Ipp, E.; Kaklamani, V.G.; Vitolins, M.; Wallace, R.; et al. Diabetes, metformin, and breast cancer in postmenopausal women. J. Clin. Oncol. 2012, 30, 2844–2852. [Google Scholar] [CrossRef] [PubMed]
- Col, N.F.; Ochs, L.; Springmann, V.; Aragaki, A.K.; Chlebowski, R.T. Metformin and breast cancer risk: A meta-analysis and critical literature review. Breast Cancer Res. Treat. 2012, 135, 639–646. [Google Scholar] [CrossRef]
- He, X.; Esteva, F.J.; Ensor, J.; Hortobagyi, G.N.; Lee, M.H.; Yeung, S.C. Metformin and thiazolidinediones are associated with improved breast cancer-specific survival of diabetic women with HER2+ breast cancer. Ann. Oncol. 2012, 23, 1771–1780. [Google Scholar] [CrossRef] [PubMed]
- Libby, G.; Donnelly, L.A.; Donnan, P.T.; Alessi, D.R.; Morris, A.D.; Evans, J.M.M. New Users of Metformin Are at Low Risk of Incident Cancer: A cohort study among people with type 2 diabetes. Diabetes Care 2009, 32, 1620–1625. [Google Scholar] [CrossRef]
- Skuli, S.J.; Alomari, S.; Gaitsch, H.; Bakayoko, A.; Skuli, N.; Tyler, B.M. Metformin and Cancer, an Ambiguanidous Relationship. Pharmaceuticals 2022, 15, 626. [Google Scholar] [CrossRef] [PubMed]
- Naseri, A.; Sanaie, S.; Hamzehzadeh, S.; Seyedi-Sahebari, S.; Hosseini, M.S.; Gholipour-Khalili, E.; Rezazadeh-Gavgani, E.; Majidazar, R.; Seraji, P.; Daneshvar, S.; et al. Metformin: New applications for an old drug. J. Basic Clin. Physiol. Pharmacol. 2023, 34, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Najafi, F.; Rajati, F.; Sarokhani, D.; Bavandpour, M.; Moradinazar, M. The Relationship between Metformin Consumption and Cancer Risk: An Updated Umbrella Review of Systematic Reviews and Meta-Analyses. Int. J. Prev. Med. 2023, 14, 90. [Google Scholar] [CrossRef] [PubMed]
- Lv, Z.; Guo, Y. Metformin and Its Benefits for Various Diseases. Front. Endocrinol. 2020, 11, 191. [Google Scholar] [CrossRef]
- Heckman-Stoddard, B.M.; De Censi, A.; Sahasrabuddhe, V.V.; Ford, L.G. Repurposing metformin for the prevention of cancer and cancer recurrence. Diabetologia 2017, 60, 1639–1647. [Google Scholar] [CrossRef]
- Rena, G.; Hardie, D.G.; Pearson, E.R. The mechanisms of action of metformin. Diabetologia 2017, 60, 1577–1585. [Google Scholar] [CrossRef]
- Saraei, P.; Asadi, I.; Kakar, M.A.; Moradi-Kor, N. The beneficial effects of metformin on cancer prevention and therapy: A comprehensive review of recent advances. Cancer Manag. Res. 2019, 11, 3295–3313. [Google Scholar] [CrossRef]
- Salani, B.; Del Rio, A.; Marini, C.; Sambuceti, G.; Cordera, R.; Maggi, D. Metformin, cancer and glucose metabolism. Endocr. Relat. Cancer 2014, 21, R461–R471. [Google Scholar] [CrossRef]
- Marini, C.; Salani, B.; Massollo, M.; Amaro, A.; Esposito, A.I.; Orengo, A.M.; Capitanio, S.; Emionite, L.; Riondato, M.; Bottoni, G.; et al. Direct inhibition of hexokinase activity by metformin at least partially impairs glucose metabolism and tumor growth in experimental breast cancer. Cell Cycle 2013, 12, 3490–3499. [Google Scholar] [CrossRef]
- Lei, Y.; Yi, Y.; Liu, Y.; Liu, X.; Keller, E.T.; Qian, C.-N.; Zhang, J.; Lu, Y. Metformin targets multiple signaling pathways in cancer. Chin. J. Cancer 2017, 36, 17. [Google Scholar] [CrossRef]
- Alimova, I.N.; Liu, B.; Fan, Z.; Edgerton, S.M.; Dillon, T.; Lind, S.E.; Thor, A.D. Metformin inhibits breast cancer cell growth, colony formation and induces cell cycle arrest in vitro. Cell Cycle 2009, 8, 909–915. [Google Scholar] [CrossRef]
- Vazquez-Martin, A.; Oliveras-Ferraros, C.; Menendez, J.A. The antidiabetic drug metformin suppresses HER2 (erbB-2) oncoprotein overexpression via inhibition of the mTOR effector p70S6K1 in human breast carcinoma cells. Cell Cycle 2009, 8, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Li, D.; Li, B.; Chu, X.; Kong, B. STAT3 as a therapeutic target in the metformin-related treatment. Int. Immunopharmacol. 2023, 116, 109770. [Google Scholar] [CrossRef]
- Deng, X.S.; Wang, S.; Deng, A.; Liu, B.; Edgerton, S.M.; Lind, S.E.; Wahdan-Alaswad, R.; Thor, A.D. Metformin targets Stat3 to inhibit cell growth and induce apoptosis in triple-negative breast cancers. Cell Cycle 2012, 11, 367–376. [Google Scholar] [CrossRef] [PubMed]
- Kasznicki, J.; Sliwinska, A.; Drzewoski, J. Metformin in cancer prevention and therapy. Ann. Transl. Med. 2014, 2, 57. [Google Scholar] [CrossRef] [PubMed]
- Yeh, H.C.; Maruthur, N.M.; Wang, N.Y.; Jerome, G.J.; Dalcin, A.T.; Tseng, E.; White, K.; Miller, E.R.; Juraschek, S.P.; Mueller, N.T.; et al. Effects of Behavioral Weight Loss and Metformin on IGFs in Cancer Survivors: A Randomized Trial. J. Clin. Endocrinol. Metab. 2021, 106, e4179–e4191. [Google Scholar] [CrossRef]
- Lero, M.W.; Shaw, L.M. Diversity of insulin and IGF signaling in breast cancer: Implications for therapy. Mol. Cell. Endocrinol. 2021, 527, 111213. [Google Scholar] [CrossRef] [PubMed]
- Anisimov, V.; Egormin, P.; Bershtein, L.; Zabezhinskii, M.; Piskunova, T.; Popovich, I.; Semenchenko, A. Metformin decelerates aging and development of mammary tumors in HER-2/neu transgenic mice. Bull. Exp. Biol. Med. 2005, 139, 721–723. [Google Scholar] [CrossRef]
- Checkley, L.A.; Rudolph, M.C.; Wellberg, E.A.; Giles, E.D.; Wahdan-Alaswad, R.S.; Houck, J.A.; Edgerton, S.M.; Thor, A.D.; Schedin, P.; Anderson, S.M. Metformin accumulation correlates with organic cation transporter 2 protein expression and predicts mammary tumor regression in vivo. Cancer Prev. Res. 2017, 10, 198–207. [Google Scholar] [CrossRef]
- Giles, E.D.; Jindal, S.; Wellberg, E.A.; Schedin, T.; Anderson, S.M.; Thor, A.D.; Edwards, D.P.; MacLean, P.S.; Schedin, P. Metformin inhibits stromal aromatase expression and tumor progression in a rodent model of postmenopausal breast cancer. Breast Cancer Res. 2018, 20, 50. [Google Scholar] [CrossRef] [PubMed]
- Caciolla, J.; Bisi, A.; Belluti, F.; Rampa, A.; Gobbi, S. Reconsidering aromatase for breast cancer treatment: New roles for an old target. Molecules 2020, 25, 5351. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Du, J.; Han, L.; Lin, X.; Fan, C.; Chen, G. The Effect of Metformin on Triple-Negative Breast Cancer Cells and Nude Mice. Altern. Ther. Health Med. 2023, 29, 389–395. [Google Scholar] [PubMed]
- Thompson, M.D.; Grubbs, C.J.; Bode, A.M.; Reid, J.M.; McGovern, R.; Bernard, P.S.; Stijleman, I.J.; Green, J.E.; Bennett, C.; Juliana, M.M.; et al. Lack of effect of metformin on mammary carcinogenesis in nondiabetic rat and mouse models. Cancer Prev. Res. 2015, 8, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Jiang, W.; Thompson, M.D.; Echeverria, D.; McGinley, J.N.; Thompson, H.J. Effects of metformin, buformin, and phenformin on the post-initiation stage of chemically induced mammary carcinogenesis in the rat. Cancer Prev. Res. 2015, 8, 518–527. [Google Scholar] [CrossRef]
- Bojkova, B.; Orendas, P.; Garajova, M.; Kassayova, M.; Kutna, V.; Ahlersova, E.; Ahlers, I. Metformin in chemically-induced mammary carcinogenesis in rats. Neoplasma 2009, 56, 269. [Google Scholar] [CrossRef] [PubMed]
- Wahdan-Alaswad, R.; Fan, Z.; Edgerton, S.M.; Liu, B.; Deng, X.S.; Arnadottir, S.S.; Richer, J.K.; Anderson, S.M.; Thor, A.D. Glucose promotes breast cancer aggression and reduces metformin efficacy. Cell Cycle 2013, 12, 3759–3769. [Google Scholar] [CrossRef]
- Varghese, S.; Samuel, S.M.; Varghese, E.; Kubatka, P.; Büsselberg, D. High Glucose Represses the Anti-Proliferative and Pro-Apoptotic Effect of Metformin in Triple Negative Breast Cancer Cells. Biomolecules 2019, 9, 16. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, Q.; Yang, J.; He, W.; Jiang, Y.; Chen, Y.; Wang, Y. Metformin combined with glucose starvation synergistically suppress triple-negative breast cancer by enhanced unfolded protein response. Biochem. Biophys. Res. Commun. 2023, 675, 146–154. [Google Scholar] [CrossRef]
- Zhu, Z.; Jiang, W.; Thompson, M.D.; McGinley, J.N.; Thompson, H.J. Metformin as an energy restriction mimetic agent for breast cancer prevention. J. Carcinog. 2011, 10, 17. [Google Scholar] [PubMed]
- Zhuang, Y.; Chan, D.K.; Haugrud, A.B.; Miskimins, W.K. Mechanisms by which low glucose enhances the cytotoxicity of metformin to cancer cells both in vitro and in vivo. PLoS ONE 2014, 9, e108444. [Google Scholar] [CrossRef] [PubMed]
- Marini, C.; Bianchi, G.; Buschiazzo, A.; Ravera, S.; Martella, R.; Bottoni, G.; Petretto, A.; Emionite, L.; Monteverde, E.; Capitanio, S.; et al. Divergent targets of glycolysis and oxidative phosphorylation result in additive effects of metformin and starvation in colon and breast cancer. Sci. Rep. 2016, 6, 19569. [Google Scholar] [CrossRef] [PubMed]
- Stambolic, V.; Woodgett, J.R.; Fantus, I.G.; Pritchard, K.I.; Goodwin, P.J. Utility of metformin in breast cancer treatment, is neoangiogenesis a risk factor? Breast Cancer Res. Treat. 2009, 114, 387–389. [Google Scholar] [CrossRef]
- Liang, X.; Giacomini, K.M. Transporters Involved in Metformin Pharmacokinetics and Treatment Response. J. Pharm. Sci. 2017, 106, 2245–2250. [Google Scholar] [CrossRef]
- Chen, S.; Zhou, J.; Xi, M.; Jia, Y.; Wong, Y.; Zhao, J.; Ding, L.; Zhang, J.; Wen, A. Pharmacogenetic variation and metformin response. Curr. Drug. Metab. 2013, 14, 1070–1082. [Google Scholar] [CrossRef]
- Samodelov, S.L.; Kullak-Ublick, G.A.; Gai, Z.; Visentin, M. Organic Cation Transporters in Human Physiology, Pharmacology, and Toxicology. Int. J. Mol. Sci. 2020, 21, 7890. [Google Scholar] [CrossRef]
- Cai, H.; Zhang, Y.; Han, T.K.; Everett, R.S.; Thakker, D.R. Cation-selective transporters are critical to the AMPK-mediated antiproliferative effects of metformin in human breast cancer cells. Int. J. Cancer 2016, 138, 2281–2292. [Google Scholar] [CrossRef]
- Zhu, P.; Davis, M.; Blackwelder, A.J.; Bachman, N.; Liu, B.; Edgerton, S.; Williams, L.L.; Thor, A.D.; Yang, X. Metformin selectively targets tumor-initiating cells in ErbB2-overexpressing breast cancer models. Cancer Prev. Res. 2014, 7, 199–210. [Google Scholar] [CrossRef]
- Barbieri, F.; Thellung, S.; Ratto, A.; Carra, E.; Marini, V.; Fucile, C.; Bajetto, A.; Pattarozzi, A.; Würth, R.; Gatti, M.; et al. In vitro and in vivo antiproliferative activity of metformin on stem-like cells isolated from spontaneous canine mammary carcinomas: Translational implications for human tumors. BMC Cancer 2015, 15, 228. [Google Scholar] [CrossRef]
- Feng, F.; Zhang, J.; Fan, X.; Yuan, F.; Jiang, Y.; Lv, R.; Ma, Y. Downregulation of Rab27A contributes to metformin-induced suppression of breast cancer stem cells. Oncol. Lett. 2017, 14, 2947–2953. [Google Scholar] [CrossRef]
- Shi, P.; Liu, W.; Tala; Wang, H.; Li, F.; Zhang, H.; Wu, Y.; Kong, Y.; Zhou, Z.; Wang, C.; et al. Metformin suppresses triple-negative breast cancer stem cells by targeting KLF5 for degradation. Cell Discov. 2017, 3, 17010. [Google Scholar] [CrossRef]
- Lee, H.; Park, H.J.; Park, C.S.; Oh, E.T.; Choi, B.H.; Williams, B.; Lee, C.K.; Song, C.W. Response of breast cancer cells and cancer stem cells to metformin and hyperthermia alone or combined. PLoS ONE 2014, 9, e87979. [Google Scholar] [CrossRef]
- Venkatesh, J.; Wasson, M.D.; Brown, J.M.; Fernando, W.; Marcato, P. LncRNA-miRNA axes in breast cancer: Novel points of interaction for strategic attack. Cancer Lett. 2021, 509, 81–88. [Google Scholar] [CrossRef]
- Wu, D.; Thompson, L.U.; Comelli, E.M. MicroRNAs: A Link between Mammary Gland Development and Breast Cancer. Int. J. Mol. Sci. 2022, 23, 15978. [Google Scholar] [CrossRef]
- Gholami, M.; Klashami, Z.N.; Ebrahimi, P.; Mahboobipour, A.A.; Farid, A.S.; Vahidi, A.; Zoughi, M.; Asadi, M.; Amoli, M.M. Metformin and long non-coding RNAs in breast cancer. J. Transl. Med. 2023, 21, 155. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Zhang, Y.; Lu, J. The roles of long noncoding RNAs in breast cancer metastasis. Cell Death Dis. 2020, 11, 749. [Google Scholar] [CrossRef]
- Abdalla, F.; Singh, B.; Bhat, H.K. MicroRNAs and gene regulation in breast cancer. J. Biochem. Mol. Toxicol. 2020, 34, e22567. [Google Scholar] [CrossRef] [PubMed]
- Fridrichova, I.; Zmetakova, I. MicroRNAs Contribute to Breast Cancer Invasiveness. Cells 2019, 8, 1361. [Google Scholar] [CrossRef] [PubMed]
- Cabello, P.; Pineda, B.; Tormo, E.; Lluch, A.; Eroles, P. The Antitumor Effect of Metformin Is Mediated by miR-26a in Breast Cancer. Int. J. Mol. Sci. 2016, 17, 1298. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Li, G.; Chen, Y.; Fang, L.; Guan, C.; Bai, F.; Ma, M.; Lyu, J.; Meng, Q.H. Metformin Inhibits Tumorigenesis and Tumor Growth of Breast Cancer Cells by Upregulating miR-200c but Downregulating AKT2 Expression. J. Cancer 2017, 8, 1849–1864. [Google Scholar] [CrossRef]
- Pulito, C.; Mori, F.; Sacconi, A.; Goeman, F.; Ferraiuolo, M.; Pasanisi, P.; Campagnoli, C.; Berrino, F.; Fanciulli, M.; Ford, R.J.; et al. Metformin-induced ablation of microRNA 21-5p releases Sestrin-1 and CAB39L antitumoral activities. Cell Discov. 2017, 3, 17022. [Google Scholar] [CrossRef] [PubMed]
- Blandino, G.; Valerio, M.; Cioce, M.; Mori, F.; Casadei, L.; Pulito, C.; Sacconi, A.; Biagioni, F.; Cortese, G.; Galanti, S.; et al. Metformin elicits anticancer effects through the sequential modulation of DICER and c-MYC. Nat. Commun. 2012, 3, 865. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Cai, S.; Yu, S.; Lin, H. Metformin induces ferroptosis by targeting miR-324-3p/GPX4 axis in breast cancer. Acta Biochim. Biophys. Sin. 2021, 53, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Wahdan-Alaswad, R.S.; Cochrane, D.R.; Spoelstra, N.S.; Howe, E.N.; Edgerton, S.M.; Anderson, S.M.; Thor, A.D.; Richer, J.K. Metformin-induced killing of triple-negative breast cancer cells is mediated by reduction in fatty acid synthase via miRNA-193b. Horm. Cancer 2014, 5, 374–389. [Google Scholar] [CrossRef]
- Cheng, L.; Zhang, X.; Huang, Y.Z.; Zhu, Y.L.; Xu, L.Y.; Li, Z.; Dai, X.Y.; Shi, L.; Zhou, X.J.; Wei, J.F.; et al. Metformin exhibits antiproliferation activity in breast cancer via miR-483-3p/METTL3/m(6)A/p21 pathway. Oncogenesis 2021, 10, 7. [Google Scholar] [CrossRef]
- Golshan, M.; Khaleghi, S.; Shafiee, S.M.; Valaee, S.; Ghanei, Z.; Jamshidizad, A.; Dashtizad, M.; Shamsara, M. Metformin modulates oncogenic expression of HOTAIR gene via promoter methylation and reverses epithelial-mesenchymal transition in MDA-MB-231 cells. J. Cell. Biochem. 2021, 122, 385–393. [Google Scholar] [CrossRef]
- Jiang, Y.; Qian, T.; Li, S.; Xie, Y.; Tao, M. Metformin reverses tamoxifen resistance through the lncRNA GAS5-medicated mTOR pathway in breast cancer. Ann. Transl. Med. 2022, 10, 366. [Google Scholar] [CrossRef]
- Li, J.; Kim, S.G.; Blenis, J. Rapamycin: One drug, many effects. Cell Metab. 2014, 19, 373–379. [Google Scholar] [CrossRef]
- Chen, J.; Qin, C.; Zhou, Y.; Chen, Y.; Mao, M.; Yang, J. Metformin may induce ferroptosis by inhibiting autophagy via lncRNA H19 in breast cancer. FEBS Open Bio 2022, 12, 146–153. [Google Scholar] [CrossRef]
- Huang, Y.; Zhou, Z.; Zhang, J.; Hao, Z.; He, Y.; Wu, Z.; Song, Y.; Yuan, K.; Zheng, S.; Zhao, Q. lncRNA MALAT1 participates in metformin inhibiting the proliferation of breast cancer cell. J. Cell. Mol. Med. 2021, 25, 7135–7145. [Google Scholar] [CrossRef]
- Tao, S.; Zhao, Z.; Zhang, X.; Guan, X.; Wei, J.; Yuan, B.; He, S.; Zhao, D.; Zhang, J.; Liu, Q.; et al. The role of macrophages during breast cancer development and response to chemotherapy. Clin. Transl. Oncol. 2020, 22, 1938–1951. [Google Scholar] [CrossRef]
- Qiu, S.-Q.; Waaijer, S.J.H.; Zwager, M.C.; de Vries, E.G.E.; van der Vegt, B.; Schröder, C.P. Tumor-associated macrophages in breast cancer: Innocent bystander or important player? Cancer Treat. Rev. 2018, 70, 178–189. [Google Scholar] [CrossRef]
- Huang, X.; Cao, J.; Zu, X. Tumor-associated macrophages: An important player in breast cancer progression. Thorac. Cancer 2022, 13, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Zhang, J.; Liu, S.; Zhou, Z. Effects of metformin on the polarization and Notch 1 expression of RAW264.7 macrophages. Zhonghua Yi Xue Za Zhi 2015, 95, 1258–1261. [Google Scholar] [PubMed]
- Hu, X.; Luo, H.; Dou, C.; Chen, X.; Huang, Y.; Wang, L.; Xue, S.; Sun, Z.; Chen, S.; Xu, Q.; et al. Metformin Triggers Apoptosis and Induction of the G0/G1 Switch 2 Gene in Macrophages. Genes 2021, 12, 1437. [Google Scholar] [CrossRef] [PubMed]
- Qing, L.; Fu, J.; Wu, P.; Zhou, Z.; Yu, F.; Tang, J. Metformin induces the M2 macrophage polarization to accelerate the wound healing via regulating AMPK/mTOR/NLRP3 inflammasome singling pathway. Am. J. Transl. Res. 2019, 11, 655–668. [Google Scholar]
- Keuper, M. On the role of macrophages in the control of adipocyte energy metabolism. Endocr. Connect. 2019, 8, R105–R121. [Google Scholar] [CrossRef] [PubMed]
- Iyengar, N.M.; Brown, K.A.; Zhou, X.K.; Gucalp, A.; Subbaramaiah, K.; Giri, D.D.; Zahid, H.; Bhardwaj, P.; Wendel, N.K.; Falcone, D.J.; et al. Metabolic Obesity, Adipose Inflammation and Elevated Breast Aromatase in Women with Normal Body Mass Index. Cancer Prev. Res. 2017, 10, 235–243. [Google Scholar] [CrossRef]
- Deng, T.; Lyon, C.J.; Bergin, S.; Caligiuri, M.A.; Hsueh, W.A. Obesity, Inflammation, and Cancer. Annu. Rev. Pathol. 2016, 11, 421–449. [Google Scholar] [CrossRef]
- Jing, Y.; Wu, F.; Li, D.; Yang, L.; Li, Q.; Li, R. Metformin improves obesity-associated inflammation by altering macrophages polarization. Mol. Cell. Endocrinol. 2018, 461, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Kurelac, I.; Umesh Ganesh, N.; Iorio, M.; Porcelli, A.M.; Gasparre, G. The multifaceted effects of metformin on tumor microenvironment. Semin. Cell Dev. Biol. 2020, 98, 90–97. [Google Scholar] [CrossRef]
- Chiang, C.F.; Chao, T.T.; Su, Y.F.; Hsu, C.C.; Chien, C.Y.; Chiu, K.C.; Shiah, S.G.; Lee, C.H.; Liu, S.Y.; Shieh, Y.S. Metformin-treated cancer cells modulate macrophage polarization through AMPK-NF-κB signaling. Oncotarget 2017, 8, 20706–20718. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Liang, G.; Yao, Z.; Zhang, J.; Liu, R.; Chen, H.; Zhou, Y.; Wu, H.; Yang, B.; He, Q. Metformin prevents cancer metastasis by inhibiting M2-like polarization of tumor associated macrophages. Oncotarget 2015, 6, 36441–36455. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Lee, D.; Lee, K.J.; Yoon, J.E.; Kwon, J.H.; Seo, Y.; Kim, J.; Chang, S.Y.; Park, J.; Kang, E.A.; et al. Tumor-Suppressive Effect of Metformin via the Regulation of M2 Macrophages and Myeloid-Derived Suppressor Cells in the Tumor Microenvironment of Colorectal Cancer. Cancers 2022, 14, 2881. [Google Scholar] [CrossRef]
- Bodmer, M.; Meier, C.; Krähenbühl, S.; Jick, S.S.; Meier, C.R. Long-term metformin use is associated with decreased risk of breast cancer. Diabetes Care 2010, 33, 1304–1308. [Google Scholar] [CrossRef] [PubMed]
- Calip, G.S.; Yu, O.; Elmore, J.G.; Boudreau, D.M. Comparative safety of diabetes medications and risk of incident invasive breast cancer: A population-based cohort study. Cancer Causes Control 2016, 27, 709–720. [Google Scholar] [CrossRef]
- Hosio, M.; Urpilainen, E.; Marttila, M.; Hautakoski, A.; Arffman, M.; Sund, R.; Puistola, U.; Läärä, E.; Jukkola, A.; Karihtala, P. Association of antidiabetic medication and statins with breast cancer incidence in women with type 2 diabetes. Breast Cancer Res. Treat. 2019, 175, 741–748. [Google Scholar] [CrossRef]
- Dankner, R.; Agay, N.; Olmer, L.; Murad, H.; Keinan Boker, L.; Balicer, R.D.; Freedman, L.S. Metformin Treatment and Cancer Risk: Cox Regression Analysis, With Time-Dependent Covariates, of 320,000 Persons With Incident Diabetes Mellitus. Am. J. Epidemiol. 2019, 188, 1794–1800. [Google Scholar] [CrossRef]
- Vicentini, M.; Ballotari, P.; Giorgi Rossi, P.; Venturelli, F.; Sacchettini, C.; Greci, M.; Mangone, L.; Pezzarossi, A.; Manicardi, V. Effect of different glucose-lowering therapies on cancer incidence in type 2 diabetes: An observational population-based study. Diabetes Res. Clin. Pract. 2018, 143, 398–408. [Google Scholar] [CrossRef]
- Löfling, L.L.; Støer, N.C.; Andreassen, B.K.; Ursin, G.; Botteri, E. Low-dose aspirin, statins, and metformin and survival in patients with breast cancers: A Norwegian population-based cohort study. Breast Cancer Res. 2023, 25, 101. [Google Scholar] [CrossRef]
- Tang, G.H.; Satkunam, M.; Pond, G.R.; Steinberg, G.R.; Blandino, G.; Schünemann, H.J.; Muti, P. Association of Metformin with Breast Cancer Incidence and Mortality in Patients with Type II Diabetes: A GRADE-Assessed Systematic Review and Meta-analysis. Cancer Epidemiol. Biomark. Prev. 2018, 27, 627–635. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Hajjar, A.; Cryns, V.L.; Trentham-Dietz, A.; Gangnon, R.E.; Heckman-Stoddard, B.M.; Alagoz, O. Breast cancer risk for women with diabetes and the impact of metformin: A meta-analysis. Cancer Med. 2023, 12, 11703–11718. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Mushashi, F.; Son, S.; Bhatti, P.; Dummer, T.; Murphy, R.A. Diabetes medications and cancer risk associations: A systematic review and meta-analysis of evidence over the past 10 years. Sci. Rep. 2023, 13, 11844. [Google Scholar] [CrossRef] [PubMed]
- Yu, O.H.Y.; Suissa, S. Metformin and Cancer: Solutions to a Real-World Evidence Failure. Diabetes Care 2023, 46, 904–912. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Chan, J.C. Comment on: Suissa and Azoulay. Metformin and the risk of cancer: Time-related biases in observational studies. Diabetes Care 2012, 35, 2665–2673. [Google Scholar] [CrossRef][Green Version]
- Heer, E.; Harper, A.; Escandor, N.; Sung, H.; McCormack, V.; Fidler-Benaoudia, M.M. Global burden and trends in premenopausal and postmenopausal breast cancer: A population-based study. Lancet Glob. Health 2020, 8, e1027–e1037. [Google Scholar] [CrossRef] [PubMed]
- Bosco, J.L.; Antonsen, S.; Sørensen, H.T.; Pedersen, L.; Lash, T.L. Metformin and incident breast cancer among diabetic women: A population-based case-control study in Denmark. Cancer Epidemiol. Biomarkers Prev. 2011, 20, 101–111. [Google Scholar] [CrossRef]
- García-Esquinas, E.; Guinó, E.; Castaño-Vinyals, G.; Pérez-Gómez, B.; Llorca, J.; Altzibar, J.M.; Peiró-Pérez, R.; Martín, V.; Moreno-Iribas, C.; Tardón, A.; et al. Association of diabetes and diabetes treatment with incidence of breast cancer. Acta Diabetol. 2016, 53, 99–107. [Google Scholar] [CrossRef]
- Niraula, S.; Dowling, R.J.O.; Ennis, M.; Chang, M.C.; Done, S.J.; Hood, N.; Escallon, J.; Leong, W.L.; McCready, D.R.; Reedijk, M.; et al. Metformin in early breast cancer: A prospective window of opportunity neoadjuvant study. Breast Cancer Res. Treat. 2012, 135, 821–830. [Google Scholar] [CrossRef]
- Hadad, S.; Iwamoto, T.; Jordan, L.; Purdie, C.; Bray, S.; Baker, L.; Jellema, G.; Deharo, S.; Hardie, D.G.; Pusztai, L.; et al. Evidence for biological effects of metformin in operable breast cancer: A pre-operative, window-of-opportunity, randomized trial. Breast Cancer Res. Treat. 2011, 128, 783–794. [Google Scholar] [CrossRef]
- Hadad, S.M.; Coates, P.; Jordan, L.B.; Dowling, R.J.O.; Chang, M.C.; Done, S.J.; Purdie, C.A.; Goodwin, P.J.; Stambolic, V.; Moulder-Thompson, S.; et al. Evidence for biological effects of metformin in operable breast cancer: Biomarker analysis in a pre-operative window of opportunity randomized trial. Breast Cancer Res. Treat. 2015, 150, 149–155. [Google Scholar] [CrossRef]
- DeCensi, A.; Puntoni, M.; Guerrieri-Gonzaga, A.; Cazzaniga, M.; Serrano, D.; Lazzeroni, M.; Vingiani, A.; Gentilini, O.; Petrera, M.; Viale, G.; et al. Effect of Metformin on Breast Ductal Carcinoma In Situ Proliferation in a Randomized Presurgical Trial. Cancer Prev. Res. 2015, 8, 888–894. [Google Scholar] [CrossRef] [PubMed]
- DeCensi, A.; Puntoni, M.; Gandini, S.; Guerrieri-Gonzaga, A.; Johansson, H.A.; Cazzaniga, M.; Pruneri, G.; Serrano, D.; Schwab, M.; Hofmann, U.; et al. Differential effects of metformin on breast cancer proliferation according to markers of insulin resistance and tumor subtype in a randomized presurgical trial. Breast Cancer Res. Treat. 2014, 148, 81–90. [Google Scholar] [CrossRef]
- Bonanni, B.; Puntoni, M.; Cazzaniga, M.; Pruneri, G.; Serrano, D.; Guerrieri-Gonzaga, A.; Gennari, A.; Trabacca, M.S.; Galimberti, V.; Veronesi, P.; et al. Dual effect of metformin on breast cancer proliferation in a randomized presurgical trial. J. Clin. Oncol. 2012, 30, 2593–2600. [Google Scholar] [CrossRef] [PubMed]
- Kalinsky, K.; Crew, K.D.; Refice, S.; Xiao, T.; Wang, A.; Feldman, S.M.; Taback, B.; Ahmad, A.; Cremers, S.; Hibshoosh, H.; et al. Presurgical trial of metformin in overweight and obese patients with newly diagnosed breast cancer. Cancer Investig. 2014, 32, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Lord, S.R.; Collins, J.M.; Cheng, W.C.; Haider, S.; Wigfield, S.; Gaude, E.; Fielding, B.A.; Pinnick, K.E.; Harjes, U.; Segaran, A.; et al. Transcriptomic analysis of human primary breast cancer identifies fatty acid oxidation as a target for metformin. Br. J. Cancer 2020, 122, 258–265. [Google Scholar] [CrossRef]
- Cazzaniga, M.; DeCensi, A.; Pruneri, G.; Puntoni, M.; Bottiglieri, L.; Varricchio, C.; Guerrieri-Gonzaga, A.; Gentilini, O.D.; Pagani, G.; Dell’Orto, P.; et al. The effect of metformin on apoptosis in a breast cancer presurgical trial. Br. J. Cancer 2013, 109, 2792–2797. [Google Scholar] [CrossRef]
- Nwanaji-Enwerem, J.C.; Chung, F.F.; Van der Laan, L.; Novoloaca, A.; Cuenin, C.; Johansson, H.; Bonanni, B.; Hubbard, A.E.; Smith, M.T.; Hartman, S.J.; et al. An epigenetic aging analysis of randomized metformin and weight loss interventions in overweight postmenopausal breast cancer survivors. Clin. Epigenetics 2021, 13, 224. [Google Scholar] [CrossRef]
- Bellerba, F.; Chatziioannou, A.C.; Jasbi, P.; Robinot, N.; Keski-Rahkonen, P.; Trolat, A.; Vozar, B.; Hartman, S.J.; Scalbert, A.; Bonanni, B.; et al. Metabolomic profiles of metformin in breast cancer survivors: A pooled analysis of plasmas from two randomized placebo-controlled trials. J. Transl. Med. 2022, 20, 629. [Google Scholar] [CrossRef]
- Kalinsky, K.; Zheng, T.; Hibshoosh, H.; Du, X.; Mundi, P.; Yang, J.; Refice, S.; Feldman, S.M.; Taback, B.; Connolly, E.; et al. Proteomic modulation in breast tumors after metformin exposure: Results from a “window of opportunity” trial. Clin. Transl. Oncol. 2017, 19, 180–188. [Google Scholar] [CrossRef]
- Martinez, J.A.; Chalasani, P.; Thomson, C.A.; Roe, D.; Altbach, M.; Galons, J.P.; Stopeck, A.; Thompson, P.A.; Villa-Guillen, D.E.; Chow, H.H. Phase II study of metformin for reduction of obesity-associated breast cancer risk: A randomized controlled trial protocol. BMC Cancer 2016, 16, 500. [Google Scholar] [CrossRef] [PubMed]
- Tapia, E.; Villa-Guillen, D.E.; Chalasani, P.; Centuori, S.; Roe, D.J.; Guillen-Rodriguez, J.; Huang, C.; Galons, J.P.; Thomson, C.A.; Altbach, M.; et al. A randomized controlled trial of metformin in women with components of metabolic syndrome: Intervention feasibility and effects on adiposity and breast density. Breast Cancer Res. Treat. 2021, 190, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Pimentel, I.; Lohmann, A.E.; Ennis, M.; Dowling, R.J.O.; Cescon, D.; Elser, C.; Potvin, K.R.; Haq, R.; Hamm, C.; Chang, M.C.; et al. A phase II randomized clinical trial of the effect of metformin versus placebo on progression-free survival in women with metastatic breast cancer receiving standard chemotherapy. Breast 2019, 48, 17–23. [Google Scholar] [CrossRef]
- Juraschek, S.P.; Plante, T.B.; Charleston, J.; Miller, E.R.; Yeh, H.C.; Appel, L.J.; Jerome, G.J.; Gayles, D.; Durkin, N.; White, K.; et al. Use of online recruitment strategies in a randomized trial of cancer survivors. Clin. Trials 2018, 15, 130–138. [Google Scholar] [CrossRef]
- Mueller, N.T.; Differding, M.K.; Zhang, M.; Maruthur, N.M.; Juraschek, S.P.; Miller, E.R., 3rd; Appel, L.J.; Yeh, H.C. Metformin Affects Gut Microbiome Composition and Function and Circulating Short-Chain Fatty Acids: A Randomized Trial. Diabetes Care 2021, 44, 1462–1471. [Google Scholar] [CrossRef]
- Hu, J.R.; Yeh, H.C.; Mueller, N.T.; Appel, L.J.; Miller, E.R., 3rd; Maruthur, N.M.; Jerome, G.J.; Chang, A.R.; Gelber, A.C.; Juraschek, S.P. Effects of a Behavioral Weight Loss Intervention and Metformin Treatment on Serum Urate: Results from a Randomized Clinical Trial. Nutrients 2021, 13, 2673. [Google Scholar] [CrossRef]
- Tilves, C.; Yeh, H.C.; Maruthur, N.; Juraschek, S.P.; Miller, E.R.; Appel, L.J.; Mueller, N.T. A behavioral weight-loss intervention, but not metformin, decreases a marker of gut barrier permeability: Results from the SPIRIT randomized trial. Int. J. Obes. 2022, 46, 655–660. [Google Scholar] [CrossRef] [PubMed]
- Tilves, C.; Yeh, H.C.; Maruthur, N.; Juraschek, S.P.; Miller, E.; White, K.; Appel, L.J.; Mueller, N.T. Increases in Circulating and Fecal Butyrate are Associated With Reduced Blood Pressure and Hypertension: Results From the SPIRIT Trial. J. Am. Heart Assoc. 2022, 11, e024763. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, P.J.; Chen, B.E.; Gelmon, K.A.; Whelan, T.J.; Ennis, M.; Lemieux, J.; Ligibel, J.A.; Hershman, D.L.; Mayer, I.A.; Hobday, T.J.; et al. Effect of Metformin vs Placebo on Invasive Disease-Free Survival in Patients With Breast Cancer: The MA.32 Randomized Clinical Trial. JAMA 2022, 327, 1963–1973. [Google Scholar] [CrossRef]
- Goodwin, P.J.; Chen, B.E.; Gelmon, K.A.; Whelan, T.J.; Ennis, M.; Lemieux, J.; Ligibel, J.A.; Hershman, D.L.; Mayer, I.A.; Hobday, T.J.; et al. Effect of Metformin Versus Placebo on New Primary Cancers in Canadian Cancer Trials Group MA.32: A Secondary Analysis of a Phase III Randomized Double-Blind Trial in Early Breast Cancer. J. Clin. Oncol. 2023, 41, 5356–5362. [Google Scholar] [CrossRef]
- Dowling, R.J.; Niraula, S.; Chang, M.C.; Done, S.J.; Ennis, M.; McCready, D.R.; Leong, W.L.; Escallon, J.M.; Reedijk, M.; Goodwin, P.J.; et al. Changes in insulin receptor signaling underlie neoadjuvant metformin administration in breast cancer: A prospective window of opportunity neoadjuvant study. Breast Cancer Res. 2015, 17, 32. [Google Scholar] [CrossRef]
- Brown, J.C.; Zhang, S.; Ligibel, J.A.; Irwin, M.L.; Jones, L.W.; Campbell, N.; Pollak, M.N.; Sorrentino, A.; Cartmel, B.; Harrigan, M.; et al. Effect of Exercise or Metformin on Biomarkers of Inflammation in Breast and Colorectal Cancer: A Randomized Trial. Cancer Prev. Res. 2020, 13, 1055–1062. [Google Scholar] [CrossRef]
- Meyerhardt, J.A.; Irwin, M.L.; Jones, L.W.; Zhang, S.; Campbell, N.; Brown, J.C.; Pollak, M.; Sorrentino, A.; Cartmel, B.; Harrigan, M.; et al. Randomized Phase II Trial of Exercise, Metformin, or Both on Metabolic Biomarkers in Colorectal and Breast Cancer Survivors. JNCI Cancer Spectr. 2020, 4, pkz096. [Google Scholar] [CrossRef]
- Lord, S.R.; Cheng, W.C.; Liu, D.; Gaude, E.; Haider, S.; Metcalf, T.; Patel, N.; Teoh, E.J.; Gleeson, F.; Bradley, K.; et al. Integrated Pharmacodynamic Analysis Identifies Two Metabolic Adaption Pathways to Metformin in Breast Cancer. Cell Metab. 2018, 28, 679–688.e674. [Google Scholar] [CrossRef]
- Ralli, G.P.; Carter, R.D.; McGowan, D.R.; Cheng, W.C.; Liu, D.; Teoh, E.J.; Patel, N.; Gleeson, F.; Harris, A.L.; Lord, S.R.; et al. Radiogenomic analysis of primary breast cancer reveals [18F]-fluorodeoxglucose dynamic flux-constants are positively associated with immune pathways and outperform static uptake measures in associating with glucose metabolism. Breast Cancer Res. 2022, 24, 34. [Google Scholar] [CrossRef]
- Rabea, H.; Hassan, A.; Elberry, A.A. Metformin as an Adjuvant Treatment in Non-Diabetic Metastatic Breast Cancer. Bahrain Med. Bull. 2021, 43, 477–481. [Google Scholar]
- Serageldin, M.A.; Kassem, A.B.; El-Kerm, Y.; Helmy, M.W.; El-Mas, M.M.; El-Bassiouny, N.A. The Effect of Metformin on Chemotherapy-Induced Toxicities in Non-diabetic Breast Cancer Patients: A Randomised Controlled Study. Drug Saf. 2023, 46, 587–599. [Google Scholar] [CrossRef] [PubMed]
- El-Attar, A.A.; Ibrahim, O.M.; Alhassanin, S.A.; Essa, E.S.; Mostafa, T.M. Effect of metformin as an adjuvant therapy to letrozole on estradiol and other biomarkers involved in the pathogenesis of breast cancer in overweight and obese postmenopausal women: A pilot study. Eur. J. Clin. Pharmacol. 2023, 79, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lim, W.; Kim, E.K.; Kim, M.K.; Paik, N.S.; Jeong, S.S.; Yoon, J.H.; Park, C.H.; Ahn, S.H.; Kim, L.S.; et al. Phase II randomized trial of neoadjuvant metformin plus letrozole versus placebo plus letrozole for estrogen receptor positive postmenopausal breast cancer (METEOR). BMC Cancer 2014, 14, 170. [Google Scholar] [CrossRef] [PubMed]
- Bakry, H.M.; Mansour, N.O.; ElKhodary, T.R.; Soliman, M.M. Efficacy of metformin in prevention of paclitaxel-induced peripheral neuropathy in breast cancer patients: A randomized controlled trial. Front. Pharmacol. 2023, 14, 1181312. [Google Scholar] [CrossRef]
- Gennari, A.; Foca, F.; Zamarchi, R.; Rocca, A.; Amadori, D.; De Censi, A.; Bologna, A.; Cavanna, L.; Gianni, L.; Scaltriti, L.; et al. Insulin-like growth factor-1 receptor (IGF-1R) expression on circulating tumor cells (CTCs) and metastatic breast cancer outcome: Results from the TransMYME trial. Breast Cancer Res. Treat. 2020, 181, 61–68. [Google Scholar] [CrossRef]
- Wang, H.; Yee, D. I-SPY 2: A Neoadjuvant Adaptive Clinical Trial Designed to Improve Outcomes in High-Risk Breast Cancer. Curr. Breast Cancer Rep. 2019, 11, 303–310. [Google Scholar] [CrossRef]
- Yee, D.; Isaacs, C.; Wolf, D.M.; Yau, C.; Haluska, P.; Giridhar, K.V.; Forero-Torres, A.; Jo Chien, A.; Wallace, A.M.; Pusztai, L.; et al. Ganitumab and metformin plus standard neoadjuvant therapy in stage 2/3 breast cancer. NPJ Breast Cancer 2021, 7, 131. [Google Scholar] [CrossRef]
- Yam, C.; Esteva, F.J.; Patel, M.M.; Raghavendra, A.S.; Ueno, N.T.; Moulder, S.L.; Hess, K.R.; Shroff, G.S.; Hodge, S.; Koenig, K.H.; et al. Efficacy and safety of the combination of metformin, everolimus and exemestane in overweight and obese postmenopausal patients with metastatic, hormone receptor-positive, HER2-negative breast cancer: A phase II study. Investig. New Drugs 2019, 37, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, S.; Vamadevan, P.; Mazibuko, N.; Bannister, R.; Swery, R.; Wilson, S.; Edwards, S. A New Method for Ethical and Efficient Evidence Generation for Off-Label Medication Use in Oncology (A Case Study in Glioblastoma). Front. Pharmacol. 2019, 10, 681. [Google Scholar] [CrossRef] [PubMed]
- Green, B.J.; Marazzini, M.; Hershey, B.; Fardin, A.; Li, Q.; Wang, Z.; Giangreco, G.; Pisati, F.; Marchesi, S.; Disanza, A.; et al. PillarX: A Microfluidic Device to Profile Circulating Tumor Cell Clusters Based on Geometry, Deformability, and Epithelial State. Small 2022, 18, e2106097. [Google Scholar] [CrossRef]
- Huang, J.; Tong, Y.; Hong, J.; Huang, O.; Wu, J.; He, J.; Chen, W.; Li, Y.; Chen, X.; Shen, K. Neoadjuvant docetaxel, epirubicin, and cyclophosphamide with or without metformin in breast cancer patients with metabolic abnormality: Results from the randomized Phase II NeoMET trial. Breast Cancer Res. Treat. 2023, 197, 525–533. [Google Scholar] [CrossRef]
- Liubota, R.; Cheshuk, V.; Zotov, O.; Vereshchako, R.; Anikusko, M.; Liubota, I.; Gur’yanov, V. Metformin in neoadjuvant systemic therapy of breast cancer patients with metabolic syndrome. Arch. Oncol. 2018, 24, 1–5. [Google Scholar] [CrossRef][Green Version]
- Nanni, O.; Amadori, D.; De Censi, A.; Rocca, A.; Freschi, A.; Bologna, A.; Gianni, L.; Rosetti, F.; Amaducci, L.; Cavanna, L.; et al. Metformin plus chemotherapy versus chemotherapy alone in the first-line treatment of HER2-negative metastatic breast cancer. The MYME randomized, phase 2 clinical trial. Breast Cancer Res. Treat. 2019, 174, 433–442. [Google Scholar] [CrossRef] [PubMed]
- Barakat, H.E.; Hussein, R.R.S.; Elberry, A.A.; Zaki, M.A.; Ramadan, M.E. The impact of metformin use on the outcomes of locally advanced breast cancer patients receiving neoadjuvant chemotherapy: An open-labelled randomized controlled trial. Sci. Rep. 2022, 12, 7656. [Google Scholar] [CrossRef]
- Zhao, Y.; Gong, C.; Wang, Z.; Zhang, J.; Wang, L.; Zhang, S.; Cao, J.; Tao, Z.; Li, T.; Wang, B.; et al. A randomized phase II study of aromatase inhibitors plus metformin in pre-treated postmenopausal patients with hormone receptor positive metastatic breast cancer. Oncotarget 2017, 8, 84224. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Ma, X.; Long, J.; Du, X.; Pan, B.; Mao, H. Metformin and survival of women with breast cancer: A meta-analysis of randomized controlled trials. J. Clin. Pharm. Ther. 2022, 47, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Lusica, P.M.M.; Eugenio, K.P.Y.; Sacdalan, D.B.L.; Jimeno, C.A. A systematic review and meta-analysis on the efficacy and safety of metformin as adjunctive therapy among women with metastatic breast cancer. Cancer Treat. Res. Commun. 2021, 29, 100457. [Google Scholar] [CrossRef]
- Goodwin, P.J.; Parulekar, W.R.; Gelmon, K.A.; Shepherd, L.E.; Ligibel, J.A.; Hershman, D.L.; Rastogi, P.; Mayer, I.A.; Hobday, T.J.; Lemieux, J.; et al. Effect of metformin vs placebo on and metabolic factors in NCIC CTG MA.32. J. Natl. Cancer Inst. 2015, 107, djv006. [Google Scholar] [CrossRef]
- Goodwin, P.J.; Dowling, R.J.O.; Ennis, M.; Chen, B.E.; Parulekar, W.R.; Shepherd, L.E.; Gelmon, K.A.; Whelan, T.J.; Ligibel, J.A.; Hershman, D.L.; et al. Cancer Antigen 15-3/Mucin 1 Levels in CCTG MA.32: A Breast Cancer Randomized Trial of Metformin vs Placebo. JNCI Cancer Spectr. 2021, 5, pkab066. [Google Scholar] [CrossRef] [PubMed]
- Pimentel, I.; Chen, B.E.; Lohmann, A.E.; Ennis, M.; Ligibel, J.; Shepherd, L.; Hershman, D.L.; Whelan, T.; Stambolic, V.; Mayer, I.; et al. The Effect of Metformin vs Placebo on Sex Hormones in Canadian Cancer Trials Group MA.32. J. Natl. Cancer Inst. 2021, 113, 192–198. [Google Scholar] [CrossRef]
- Ko, K.P.; Ma, S.H.; Yang, J.J.; Hwang, Y.; Ahn, C.; Cho, Y.M.; Noh, D.Y.; Park, B.J.; Han, W.; Park, S.K. Metformin intervention in obese non-diabetic patients with breast cancer: Phase II randomized, double-blind, placebo-controlled trial. Breast Cancer Res. Treat. 2015, 153, 361–370. [Google Scholar] [CrossRef]
- El-Haggar, S.M.; El-Shitany, N.A.; Mostafa, M.F.; El-Bassiouny, N.A. Metformin may protect nondiabetic breast cancer women from metastasis. Clin. Exp. Metastasis 2016, 33, 339–357. [Google Scholar] [CrossRef]
- Giles, E.D.; Singh, G. Role of insulin-like growth factor binding proteins (IGFBPs) in breast cancer proliferation and metastasis. Clin. Exp. Metastasis 2003, 20, 481–487. [Google Scholar] [CrossRef]
- Sonnenblick, A.; Agbor-Tarh, D.; Bradbury, I.; Di Cosimo, S.; Azim, H.A., Jr.; Fumagalli, D.; Sarp, S.; Wolff, A.C.; Andersson, M.; Kroep, J.; et al. Impact of Diabetes, Insulin, and Metformin Use on the Outcome of Patients With Human Epidermal Growth Factor Receptor 2-Positive Primary Breast Cancer: Analysis From the ALTTO Phase III Randomized Trial. J. Clin. Oncol. 2017, 35, 1421–1429. [Google Scholar] [CrossRef]
- Dyatlova, N.; Tobarran, N.V.; Kannan, L.; North, R.; Wills, B.K. Metformin-Associated Lactic Acidosis (MALA). In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2023. [Google Scholar]
- Davies, G.; Lobanova, L.; Dawicki, W.; Groot, G.; Gordon, J.R.; Bowen, M.; Harkness, T.; Arnason, T. Metformin inhibits the development, and promotes the resensitization, of treatment-resistant breast cancer. PLoS ONE 2017, 12, e0187191. [Google Scholar] [CrossRef]
- Lee, J.O.; Kang, M.J.; Byun, W.S.; Kim, S.A.; Seo, I.H.; Han, J.A.; Moon, J.W.; Kim, J.H.; Kim, S.J.; Lee, E.J.; et al. Metformin overcomes resistance to cisplatin in triple-negative breast cancer (TNBC) cells by targeting RAD51. Breast Cancer Res. 2019, 21, 115. [Google Scholar] [CrossRef]
- Zhang, H.H.; Guo, X.L. Combinational strategies of metformin and chemotherapy in cancers. Cancer Chemother. Pharmacol. 2016, 78, 13–26. [Google Scholar] [CrossRef]
- Morio, K.; Kurata, Y.; Kawaguchi-Sakita, N.; Shiroshita, A.; Kataoka, Y. Efficacy of Metformin in Patients With Breast Cancer Receiving Chemotherapy or Endocrine Therapy: Systematic Review and Meta-analysis. Ann. Pharmacother. 2022, 56, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Krall, A.S.; Mullen, P.J.; Surjono, F.; Momcilovic, M.; Schmid, E.W.; Halbrook, C.J.; Thambundit, A.; Mittelman, S.D.; Lyssiotis, C.A.; Shackelford, D.B.; et al. Asparagine couples mitochondrial respiration to ATF4 activity and tumor growth. Cell Metab. 2021, 33, 1013–1026.e1016. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Rourk, K.; Bernier, A.; de Lartigue, G. Non-Nutritive Sweetened Beverages Impair Therapeutic Benefits of Metformin in Prediabetic Diet-Induced Obese Mice. Nutrients 2023, 15, 2472. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Corleto, K.A.; Strandmo, J.L.; Giles, E.D. Metformin and Breast Cancer: Current Findings and Future Perspectives from Preclinical and Clinical Studies. Pharmaceuticals 2024, 17, 396. https://doi.org/10.3390/ph17030396
Corleto KA, Strandmo JL, Giles ED. Metformin and Breast Cancer: Current Findings and Future Perspectives from Preclinical and Clinical Studies. Pharmaceuticals. 2024; 17(3):396. https://doi.org/10.3390/ph17030396
Chicago/Turabian StyleCorleto, Karen A., Jenna L. Strandmo, and Erin D. Giles. 2024. "Metformin and Breast Cancer: Current Findings and Future Perspectives from Preclinical and Clinical Studies" Pharmaceuticals 17, no. 3: 396. https://doi.org/10.3390/ph17030396
APA StyleCorleto, K. A., Strandmo, J. L., & Giles, E. D. (2024). Metformin and Breast Cancer: Current Findings and Future Perspectives from Preclinical and Clinical Studies. Pharmaceuticals, 17(3), 396. https://doi.org/10.3390/ph17030396





