Rational Design, Synthesis, and Evaluation of Fluorescent CB2 Receptor Ligands for Live-Cell Imaging: A Comprehensive Review
Abstract
1. Introduction
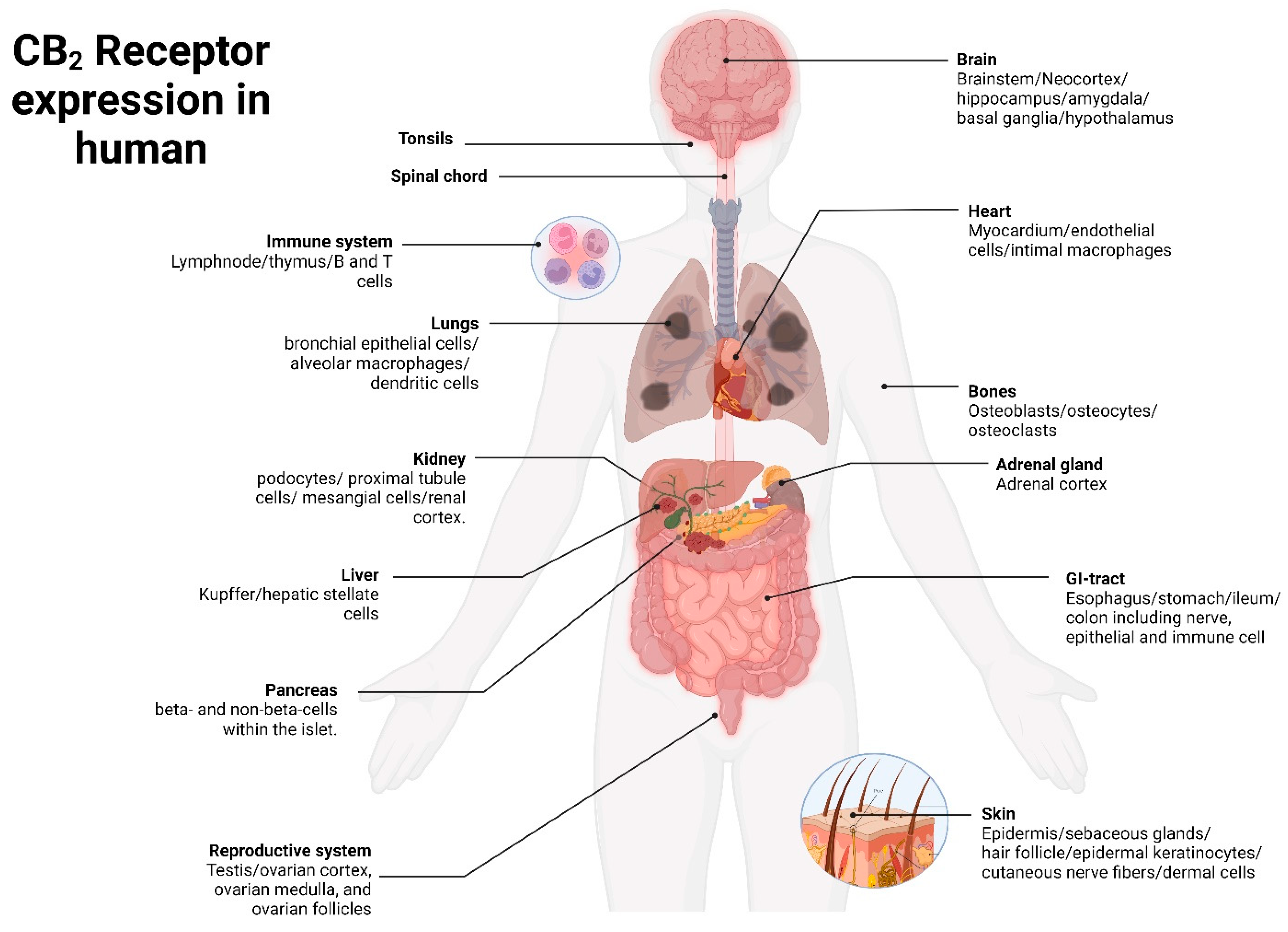
2. CB2 Receptor: Molecular Characterization and Spatial Distribution
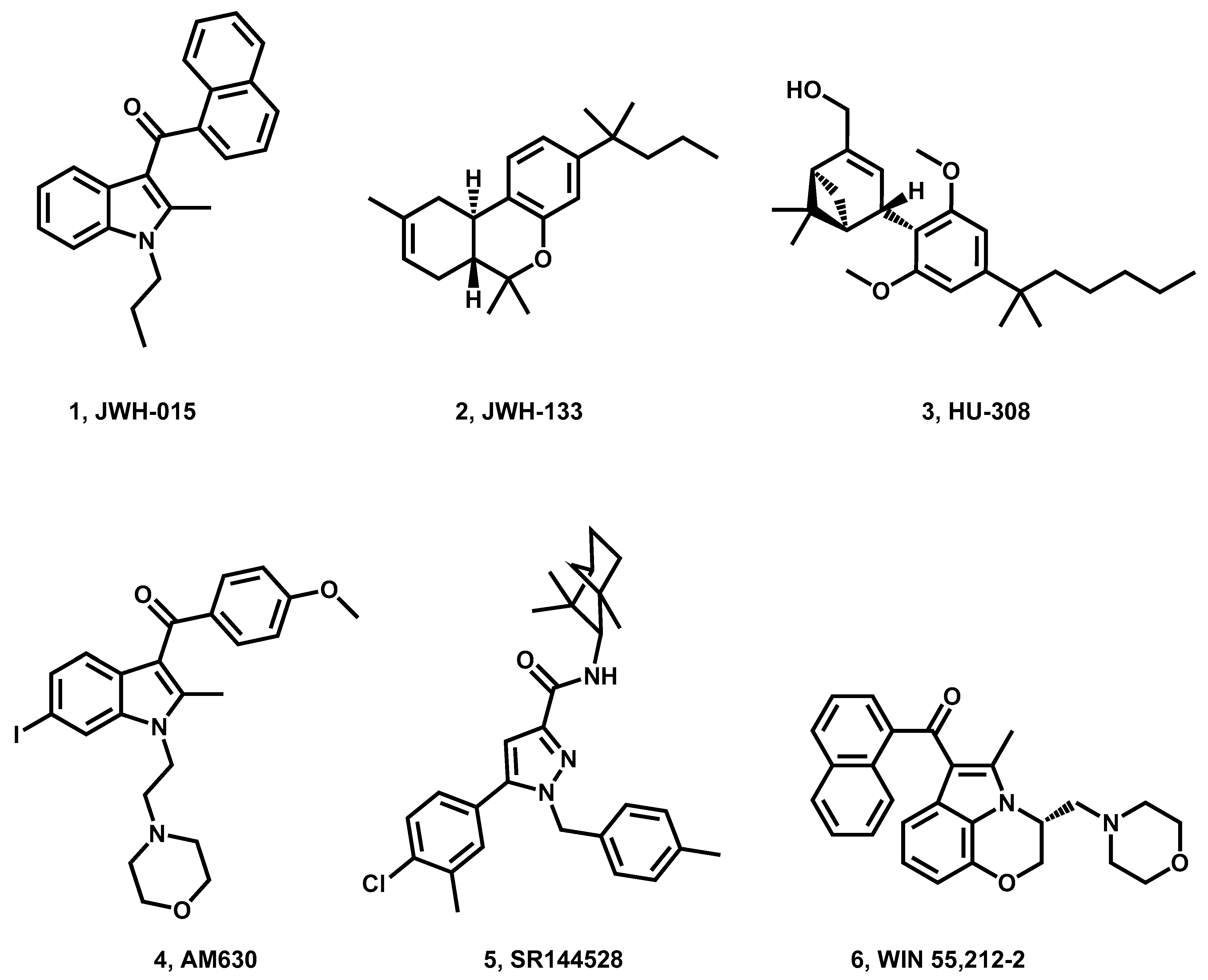
3. Fluorescent Probe Design Principles
4. CB2R Targeting Fluorescent Probes
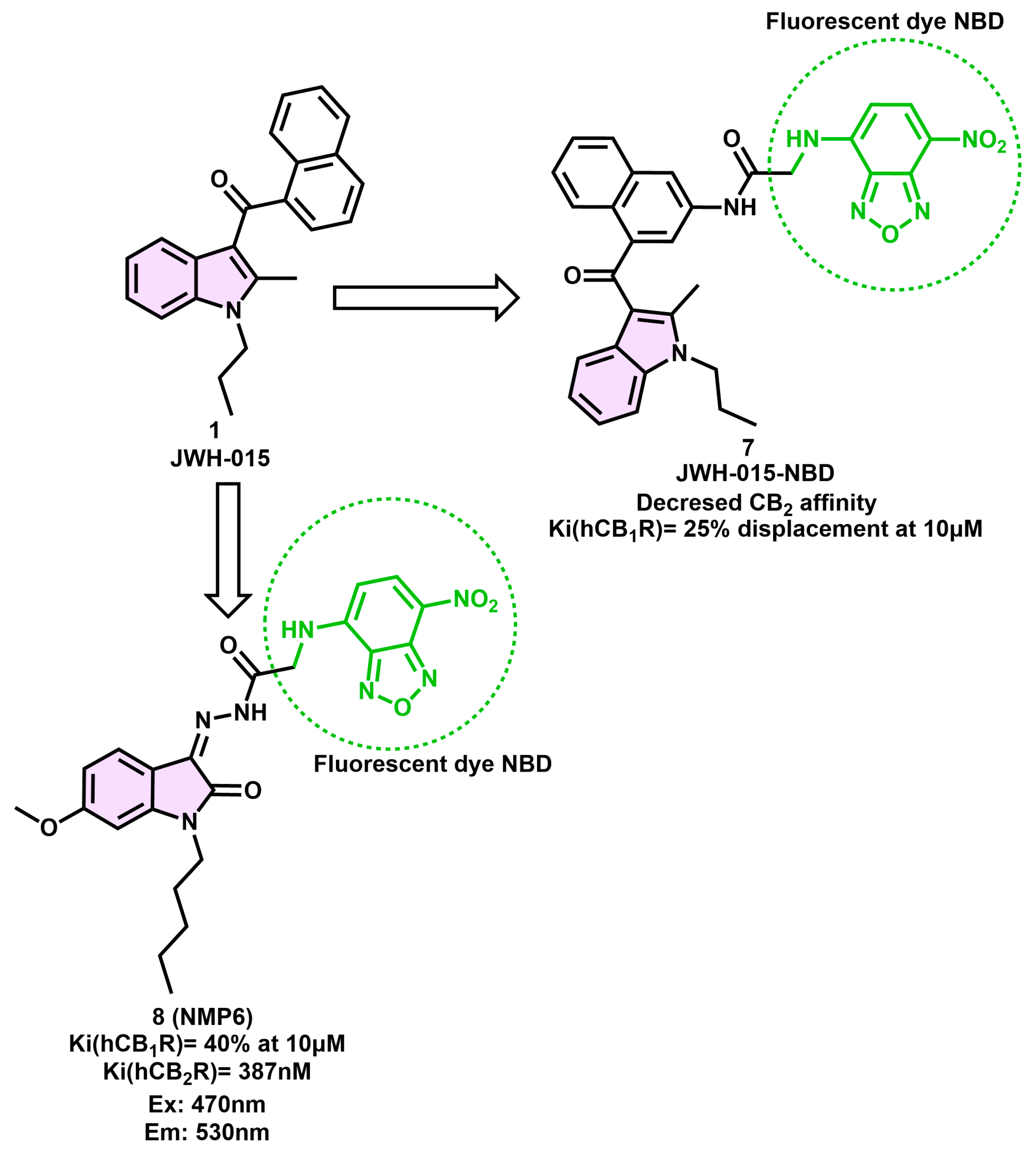
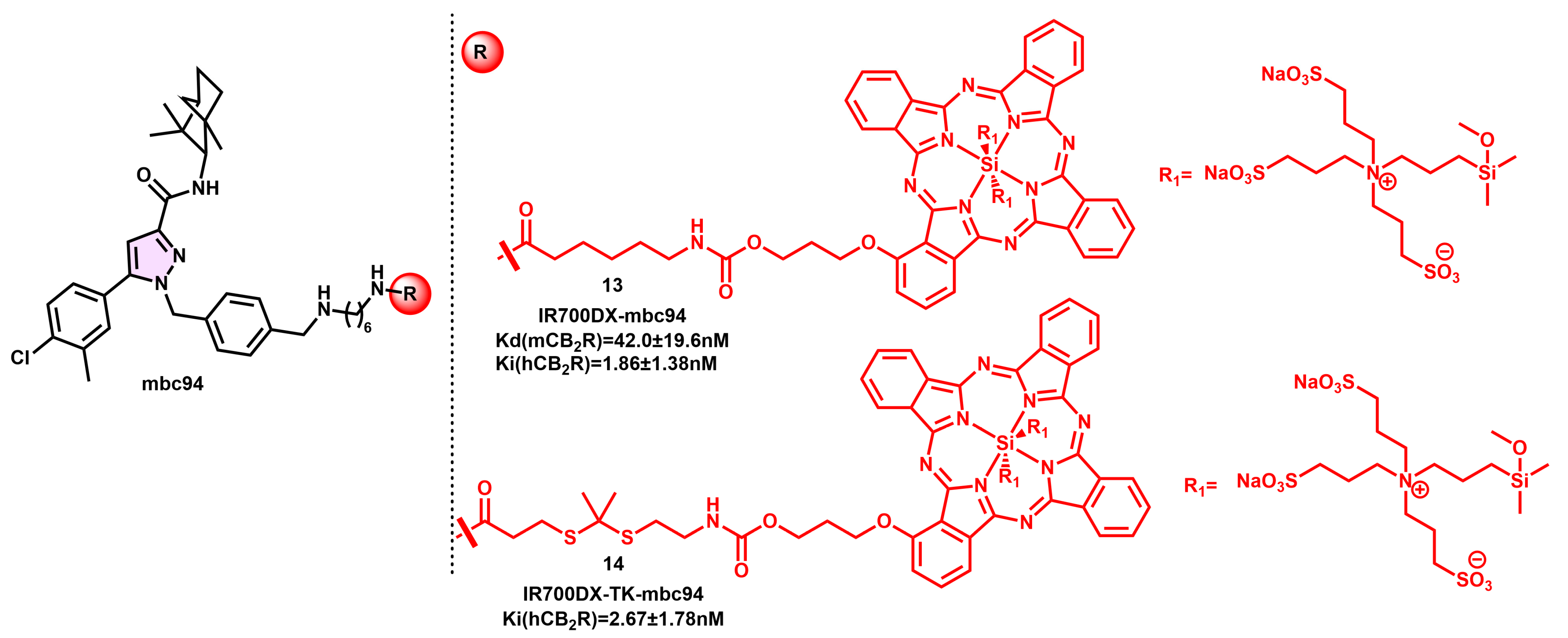
4.1. Fluorescent Indole Derivatives
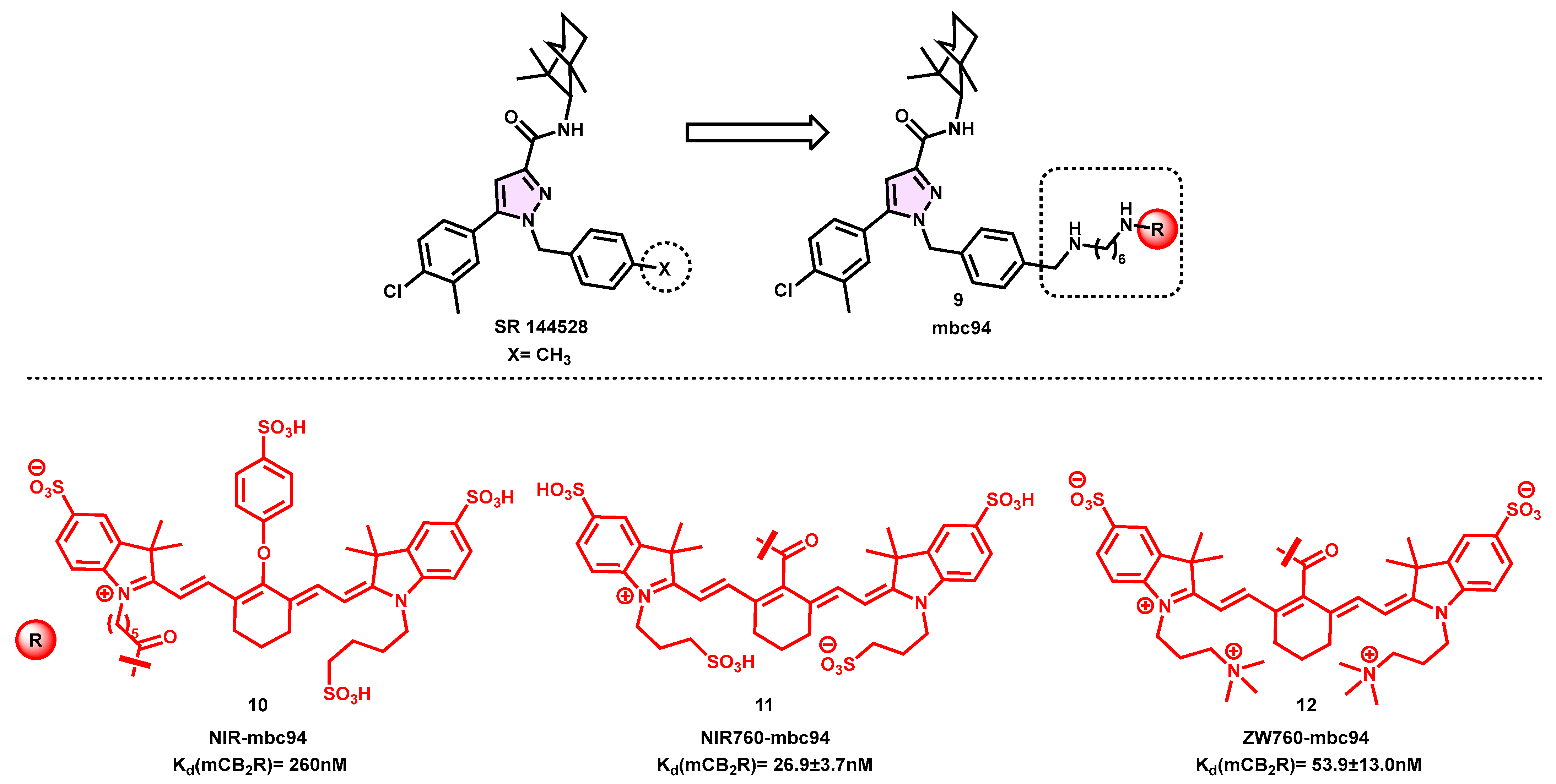
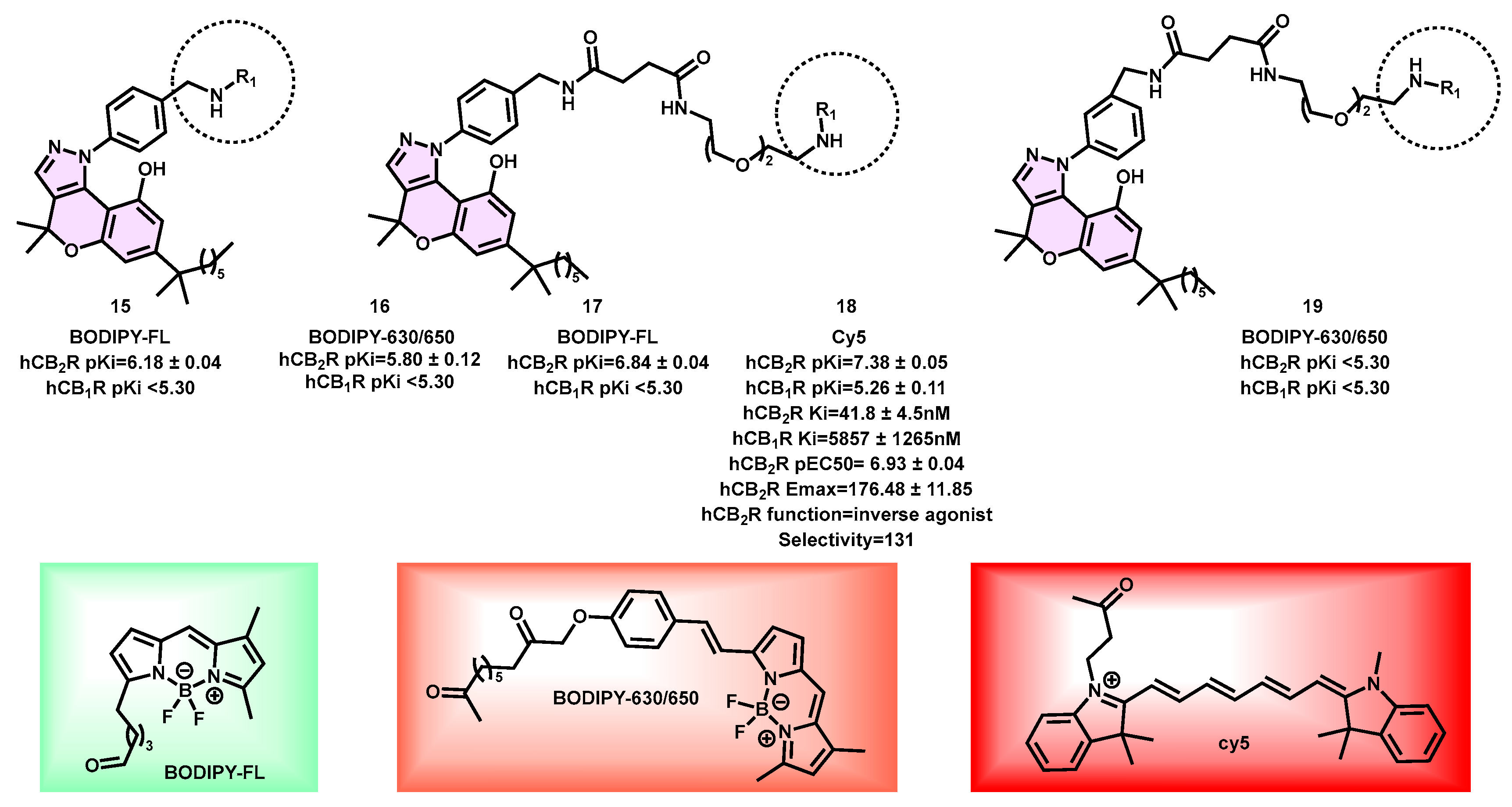
4.2. Pyrazole-Based Fluorescent Probes
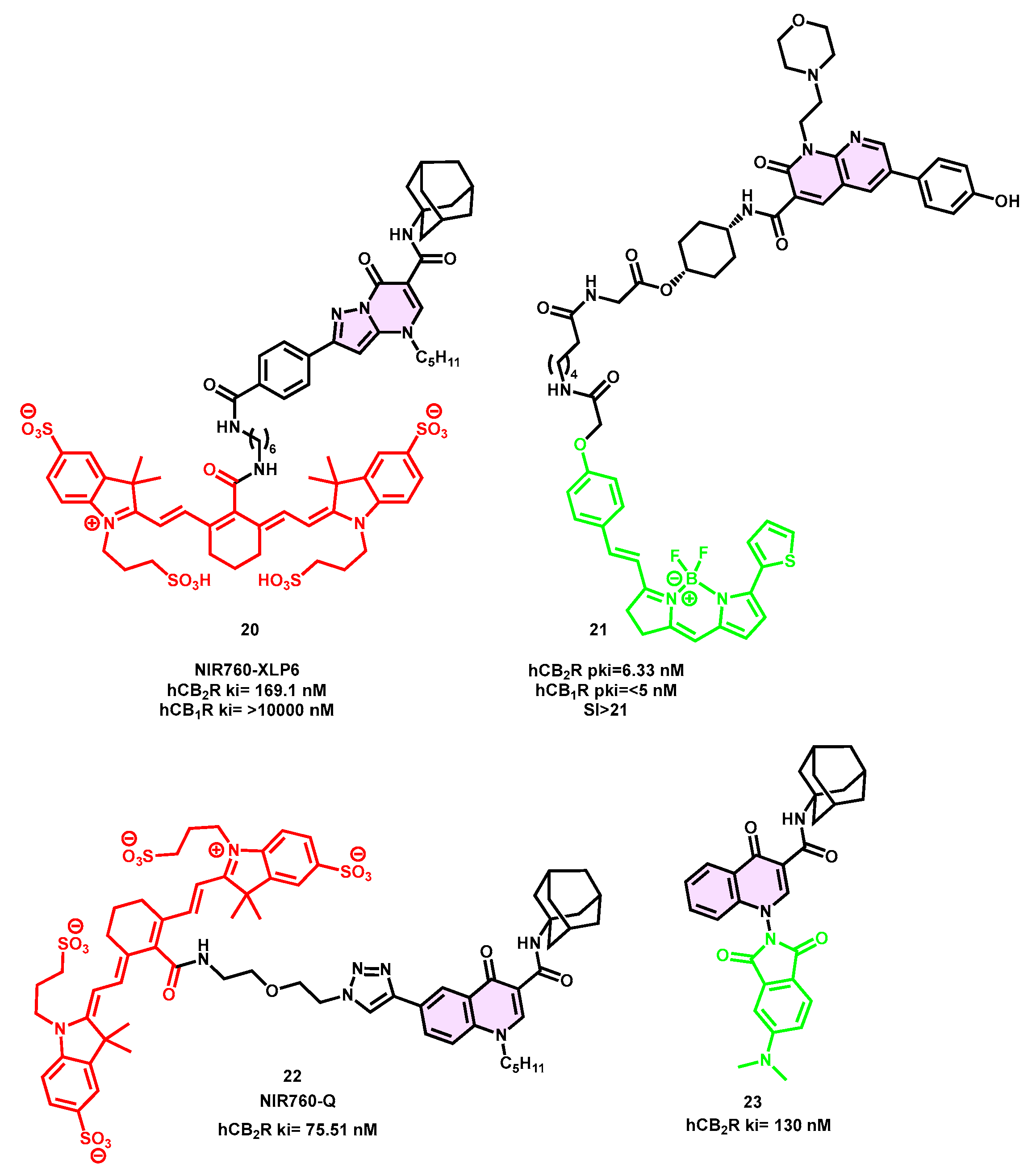
4.3. Fluorescent Oxoquinoline Derivatives
4.4. Miscellaneous Fluorescent Probes
5. Future Perspective and Critical Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Manglik, A.; Kobilka, B.K.; Steyaert, J. Nanobodies to Study G Protein-Coupled Receptor Structure and Function. Annu. Rev. Pharmacol. Toxicol. 2017, 57, 19–37. [Google Scholar] [CrossRef]
- Rosenbaum, D.M.; Rasmussen, S.G.F.; Kobilka, B.K. The Structure and Function of G-Protein-Coupled Receptors. Nature 2009, 459, 356–363. [Google Scholar] [CrossRef]
- Weis, W.I.; Kobilka, B.K. The Molecular Basis of G Protein-Coupled Receptor Activation. Annu. Rev. Biochem. 2018, 87, 897–919. [Google Scholar] [CrossRef]
- Marinissen, M.J.; Gutkind, J.S. G-Protein-Coupled Receptors and Signaling Networks: Emerging Paradigms. Trends Pharmacol. Sci. 2001, 22, 368–376. [Google Scholar] [CrossRef]
- Lagerström, M.C.; Schiöth, H.B. Structural Diversity of G Protein-Coupled Receptors and Significance for Drug Discovery. Nat. Rev. Drug Discov. 2008, 7, 339–357. [Google Scholar] [CrossRef]
- Katritch, V.; Cherezov, V.; Stevens, R.C. Structure-Function of the G Protein-Coupled Receptor Superfamily. Annu. Rev. Pharmacol. Toxicol. 2013, 53, 531–556. [Google Scholar] [CrossRef] [PubMed]
- Pacher, P.; Bátkai, S.; Kunos, G. The Endocannabinoid System as an Emerging Target of Pharmacotherapy. Pharmacol. Rev. 2006, 58, 389–462. [Google Scholar] [CrossRef]
- Pacher, P.; Kunos, G. Modulating the Endocannabinoid System in Human Health and Disease—Successes and Failures. FEBS J. 2013, 280, 1918–1943. [Google Scholar] [CrossRef]
- Lu, H.-C.; Mackie, K. Review of the Endocannabinoid System. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 2021, 6, 607–615. [Google Scholar] [CrossRef]
- Munro, S.; Thomas, K.L.; Abu-Shaar, M. Molecular Characterization of a Peripheral Receptor for Cannabinoids. Nature 1993, 365, 61–65. [Google Scholar] [CrossRef]
- Bellocchio, L.; Soria-Gómez, E.; Quarta, C.; Metna-Laurent, M.; Cardinal, P.; Binder, E.; Cannich, A.; Delamarre, A.; Häring, M.; Martín-Fontecha, M.; et al. Activation of the Sympathetic Nervous System Mediates Hypophagic and Anxiety-like Effects of CB1 Receptor Blockade. Proc. Natl. Acad. Sci. USA 2013, 110, 4786–4791. [Google Scholar] [CrossRef]
- Rodríguez de Fonseca, F.; Del Arco, I.; Bermudez-Silva, F.J.; Bilbao, A.; Cippitelli, A.; Navarro, M. The Endocannabinoid System: Physiology and Pharmacology. Alcohol Alcohol. 2005, 40, 2–14. [Google Scholar] [CrossRef]
- Liu, Q.R.; Pan, C.H.; Hishimoto, A.; Li, C.Y.; Xi, Z.X.; Llorente-Berzal, A.; Viveros, M.P.; Ishiguro, H.; Arinami, T.; Onaivi, E.S.; et al. Species Differences in Cannabinoid Receptor 2 (CNR2 Gene): Identification of Novel Human and Rodent CB2 Isoforms, Differential Tissue Expression and Regulation by Cannabinoid Receptor Ligands. Genes Brain Behav. 2009, 8, 519–530. [Google Scholar] [CrossRef] [PubMed]
- Onaivi, E.S.; Ishiguro, H.; Gong, J.-P.; Patel, S.; Perchuk, A.; Meozzi, P.A.; Myers, L.; Mora, Z.; Tagliaferro, P.; Gardner, E.; et al. Discovery of the Presence and Functional Expression of Cannabinoid CB2 Receptors in Brain. Ann. N. Y. Acad. Sci. 2006, 1074, 514–536. [Google Scholar] [CrossRef]
- Roche, M.; Finn, D.P. Brain CB2 Receptors: Implications for Neuropsychiatric Disorders. Pharmaceuticals 2010, 3, 2517–2553. [Google Scholar] [CrossRef]
- Wu, Y.-R.; Tang, J.-Q.; Zhang, W.-N.; Zhuang, C.-L.; Shi, Y. Rational Drug Design of CB2 Receptor Ligands: From 2012 to 2021. RSC Adv. 2022, 12, 35242–35259. [Google Scholar] [CrossRef]
- Gasperi, V.; Guzzo, T.; Topai, A.; Gambacorta, N.; Ciriaco, F.; Nicolotti, O.; Maccarrone, M. Recent Advances on Type-2 Cannabinoid (CB2) Receptor Agonists and Their Therapeutic Potential. Curr. Med. Chem. 2023, 30, 1420–1457. [Google Scholar] [CrossRef]
- Li, X.; Hua, T.; Vemuri, K.; Ho, J.-H.; Wu, Y.; Wu, L.; Popov, P.; Benchama, O.; Zvonok, N.; Locke, K.; et al. Crystal Structure of the Human Cannabinoid Receptor CB2. Cell 2019, 176, 459–467.e13. [Google Scholar] [CrossRef]
- Hua, T.; Vemuri, K.; Nikas, S.P.; Laprairie, R.B.; Wu, Y.; Qu, L.; Pu, M.; Korde, A.; Jiang, S.; Ho, J.-H.; et al. Crystal Structures of Agonist-Bound Human Cannabinoid Receptor CB1. Nature 2017, 547, 468–471. [Google Scholar] [CrossRef]
- Li, X.; Chang, H.; Bouma, J.; de Paus, L.V.; Mukhopadhyay, P.; Paloczi, J.; Mustafa, M.; van der Horst, C.; Kumar, S.S.; Wu, L.; et al. Structural Basis of Selective Cannabinoid CB2 Receptor Activation. Nat. Commun. 2023, 14, 1447. [Google Scholar] [CrossRef]
- Dhopeshwarkar, A.; Mackie, K. CB2 Cannabinoid Receptors as a Therapeutic Target-What Does the Future Hold? Mol. Pharmacol. 2014, 86, 430–437. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Thatte, J.; Buzard, D.J.; Jones, R.M. Therapeutic Utility of Cannabinoid Receptor Type 2 (CB(2)) Selective Agonists. J. Med. Chem. 2013, 56, 8224–8256. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, A.J.; Payne, A.D.; Mocerino, M.; Gunosewoyo, H. Imaging Cannabinoid Receptors: A Brief Collection of Covalent and Fluorescent Probes for CB. Aust. J. Chem. 2021, 74, 416–432. [Google Scholar] [CrossRef]
- Guberman, M.; Kosar, M.; Omran, A.; Carreira, E.M.; Nazaré, M.; Grether, U. Reverse-Design toward Optimized Labeled Chemical Probes—Examples from the Endocannabinoid System. Chimia 2022, 76, 425. [Google Scholar] [CrossRef]
- Giepmans, B.N.G.; Adams, S.R.; Ellisman, M.H.; Tsien, R.Y. The Fluorescent Toolbox for Assessing Protein Location and Function. Science 2006, 312, 217–224. [Google Scholar] [CrossRef]
- Evens, N.; Bormans, G.M. Non-Invasive Imaging of the Type 2 Cannabinoid Receptor, Focus on Positron Emission Tomography. Curr. Top. Med. Chem. 2010, 10, 1527–1543. [Google Scholar] [CrossRef]
- Basagni, F.; Rosini, M.; Decker, M. Functionalized Cannabinoid Subtype 2 Receptor Ligands: Fluorescent, PET, Photochromic and Covalent Molecular Probes. ChemMedChem 2020, 15, 1374–1389. [Google Scholar] [CrossRef]
- Soethoudt, M.; Stolze, S.C.; Westphal, M.V.; van Stralen, L.; Martella, A.; van Rooden, E.J.; Guba, W.; Varga, Z.V.; Deng, H.; van Kasteren, S.I.; et al. Selective Photoaffinity Probe That Enables Assessment of Cannabinoid CB2 Receptor Expression and Ligand Engagement in Human Cells. J. Am. Chem. Soc. 2018, 140, 6067–6075. [Google Scholar] [CrossRef] [PubMed]
- Stoddart, L.A.; Kilpatrick, L.E.; Briddon, S.J.; Hill, S.J. Probing the Pharmacology of G Protein-Coupled Receptors with Fluorescent Ligands. Neuropharmacology 2015, 98, 48–57. [Google Scholar] [CrossRef]
- Kherlopian, A.R.; Song, T.; Duan, Q.; Neimark, M.A.; Po, M.J.; Gohagan, J.K.; Laine, A.F. A Review of Imaging Techniques for Systems Biology. BMC Syst. Biol. 2008, 2, 74. [Google Scholar] [CrossRef]
- Singh, S.; Oyagawa, C.R.M.; Macdonald, C.; Grimsey, N.L.; Glass, M.; Vernall, A.J. Chromenopyrazole-Based High Affinity, Selective Fluorescent Ligands for Cannabinoid Type 2 Receptor. ACS Med. Chem. Lett. 2019, 10, 209–214. [Google Scholar] [CrossRef]
- Galiègue, S.; Mary, S.; Marchand, J.; Dussossoy, D.; Carrière, D.; Carayon, P.; Bouaboula, M.; Shire, D.; Le Fur, G.; Casellas, P. Expression of Central and Peripheral Cannabinoid Receptors in Human Immune Tissues and Leukocyte Subpopulations. Eur. J. Biochem. 1995, 232, 54–61. [Google Scholar] [CrossRef]
- Patel, K.D.; Davison, J.S.; Pittman, Q.J.; Sharkey, K.A. Cannabinoid CB(2) Receptors in Health and Disease. Curr. Med. Chem. 2010, 17, 1393–1410. [Google Scholar] [CrossRef]
- Van Sickle, M.D.; Duncan, M.; Kingsley, P.J.; Mouihate, A.; Urbani, P.; Mackie, K.; Stella, N.; Makriyannis, A.; Piomelli, D.; Davison, J.S.; et al. Identification and Functional Characterization of Brainstem Cannabinoid CB2 Receptors. Science 2005, 310, 329–332. [Google Scholar] [CrossRef]
- Cabral, G.A.; Raborn, E.S.; Griffin, L.; Dennis, J.; Marciano-Cabral, F. CB2 Receptors in the Brain: Role in Central Immune Function. Br. J. Pharmacol. 2008, 153, 240–251. [Google Scholar] [CrossRef]
- López, A.; Aparicio, N.; Pazos, M.R.; Grande, M.T.; Barreda-Manso, M.A.; Benito-Cuesta, I.; Vázquez, C.; Amores, M.; Ruiz-Pérez, G.; García-García, E.; et al. Cannabinoid CB2 Receptors in the Mouse Brain: Relevance for Alzheimer’s Disease. J. Neuroinflammation 2018, 15, 158. [Google Scholar] [CrossRef]
- Ferranti, A.S.; Foster, D.J. Cannabinoid Type-2 Receptors: An Emerging Target for Regulating Schizophrenia-Relevant Brain Circuits. Front. Neurosci. 2022, 16, 925792. [Google Scholar] [CrossRef]
- Schmöle, A.-C.; Lundt, R.; Gennequin, B.; Schrage, H.; Beins, E.; Krämer, A.; Zimmer, T.; Limmer, A.; Zimmer, A.; Otte, D.-M. Expression Analysis of CB2-GFP BAC Transgenic Mice. PLoS ONE 2015, 10, e0138986. [Google Scholar] [CrossRef]
- Kibret, B.G.; Ishiguro, H.; Horiuchi, Y.; Onaivi, E.S. New Insights and Potential Therapeutic Targeting of CB2 Cannabinoid Receptors in CNS Disorders. Int. J. Mol. Sci. 2022, 23, 975. [Google Scholar] [CrossRef]
- Yang, P.; Wang, L.; Xie, X.-Q. Latest Advances in Novel Cannabinoid CB(2) Ligands for Drug Abuse and Their Therapeutic Potential. Future Med. Chem. 2012, 4, 187–204. [Google Scholar] [CrossRef]
- Smoum, R.; Grether, U.; Karsak, M.; Vernall, A.J.; Park, F.; Hillard, C.J.; Pacher, P. Editorial: Therapeutic Potential of the Cannabinoid CB2 Receptor. Front. Pharmacol. 2022, 13, 1039564. [Google Scholar] [CrossRef]
- Bie, B.; Wu, J.; Foss, J.F.; Naguib, M. An Overview of the Cannabinoid Type 2 Receptor System and Its Therapeutic Potential. Curr. Opin. Anaesthesiol. 2018, 31, 407–414. [Google Scholar] [CrossRef]
- Morales, P.; Hernandez-Folgado, L.; Goya, P.; Jagerovic, N. Cannabinoid Receptor 2 (CB2) Agonists and Antagonists: A Patent Update. Expert Opin. Ther. Pat. 2016, 26, 843–856. [Google Scholar] [CrossRef]
- Berque-Bestel, I.; Soulier, J.-L.; Giner, M.; Rivail, L.; Langlois, M.; Sicsic, S. Synthesis and Characterization of the First Fluorescent Antagonists for Human 5-HT4 Receptors. J. Med. Chem. 2003, 46, 2606–2620. [Google Scholar] [CrossRef]
- Middleton, R.J.; Briddon, S.J.; Cordeaux, Y.; Yates, A.S.; Dale, C.L.; George, M.W.; Baker, J.G.; Hill, S.J.; Kellam, B. New Fluorescent Adenosine A1-Receptor Agonists That Allow Quantification of Ligand-Receptor Interactions in Microdomains of Single Living Cells. J. Med. Chem. 2007, 50, 782–793. [Google Scholar] [CrossRef]
- Middleton, R.J.; Kellam, B. Fluorophore-Tagged GPCR Ligands. Curr. Opin. Chem. Biol. 2005, 9, 517–525. [Google Scholar]
- Höltke, C.; von Wallbrunn, A.; Kopka, K.; Schober, O.; Heindel, W.; Schäfers, M.; Bremer, C. A Fluorescent Photoprobe for the Imaging of Endothelin Receptors. Bioconjug. Chem. 2007, 18, 685–694. [Google Scholar]
- Iliopoulos-Tsoutsouvas, C.; Kulkarni, R.N.; Makriyannis, A.; Nikas, S.P. Fluorescent Probes for G-Protein-Coupled Receptor Drug Discovery. Expert Opin. Drug Discov. 2018, 13, 933–947. [Google Scholar]
- Leopoldo, M.; Lacivita, E.; Berardi, F.; Perrone, R. Developments in Fluorescent Probes for Receptor Research. Drug Discov. Today 2009, 14, 706–712. [Google Scholar]
- McCarron, S.T.; Chambers, J.J. Modular Chemical Probes for Visualizing and Tracking Endogenous Ion Channels. Neuropharmacology 2015, 98, 41–47. [Google Scholar] [CrossRef]
- Ma, Z.; Du, L.; Li, M. Toward Fluorescent Probes for G-Protein-Coupled Receptors (GPCRs). J. Med. Chem. 2014, 57, 8187–8203. [Google Scholar] [CrossRef] [PubMed]
- Briddon, S.J.; Kellam, B.; Hill, S.J. Design and Use of Fluorescent Ligands to Study Ligand-Receptor Interactions in Single Living Cells. Methods Mol. Biol. 2011, 746, 211–236. [Google Scholar] [PubMed]
- Sarott, R.C.; Westphal, M.V.; Pfaff, P.; Korn, C.; Sykes, D.A.; Gazzi, T.; Brennecke, B.; Atz, K.; Weise, M.; Mostinski, Y.; et al. Development of High-Specificity Fluorescent Probes to Enable Cannabinoid Type 2 Receptor Studies in Living Cells. J. Am. Chem. Soc. 2020, 142, 16953–16964. [Google Scholar] [CrossRef]
- Gazzi, T.; Brennecke, B.; Atz, K.; Korn, C.; Sykes, D.; Forn-Cuni, G.; Pfaff, P.; Sarott, R.C.; Westphal, M.V.; Mostinski, Y.; et al. Detection of Cannabinoid Receptor Type 2 in Native Cells and Zebrafish with a Highly Potent, Cell-Permeable Fluorescent Probe. Chem. Sci. 2022, 13, 5539–5545. [Google Scholar] [PubMed]
- Zhang, H.-Y.; Shen, H.; Jordan, C.J.; Liu, Q.-R.; Gardner, E.L.; Bonci, A.; Xi, Z.-X. CB2 Receptor Antibody Signal Specificity: Correlations with the Use of Partial CB2-Knockout Mice and Anti-Rat CB2 Receptor Antibodies. Acta Pharmacol. Sin. 2019, 40, 398–409. [Google Scholar] [CrossRef]
- Cécyre, B.; Thomas, S.; Ptito, M.; Casanova, C.; Bouchard, J.-F. Evaluation of the Specificity of Antibodies Raised against Cannabinoid Receptor Type 2 in the Mouse Retina. Naunyn Schmiedebergs Arch. Pharmacol. 2014, 387, 175–184. [Google Scholar]
- Cooper, A.; Singh, S.; Hook, S.; Tyndall, J.D.A.; Vernall, A.J. Chemical Tools for Studying Lipid-Binding Class A G Protein-Coupled Receptors. Pharmacol. Rev. 2017, 69, 316–353. [Google Scholar]
- Contino, M.; Abate, C.; Colabufo, N.A.; Leonetti, F.; Stefanachi, A. Cannabinoid Receptor Subtype 2 (CB2R): Features and Targets for Medical Applications. In Medicinal Usage of Cannabis and Cannabinoids; Elsevier: Amsterdam, The Netherlands, 2023; pp. 319–333. [Google Scholar]
- Yates, A.S.; Doughty, S.W.; Kendall, D.A.; Kellam, B. Chemical Modification of the Naphthoyl 3-Position of JWH-015: In Search of a Fluorescent Probe to the Cannabinoid CB2 Receptor. Bioorg. Med. Chem. Lett. 2005, 15, 3758–3762. [Google Scholar] [CrossRef] [PubMed]
- Petrov, R.R.; Ferrini, M.E.; Jaffar, Z.; Thompson, C.M.; Roberts, K.; Diaz, P. Design and Evaluation of a Novel Fluorescent CB2 Ligand as Probe for Receptor Visualization in Immune Cells. Bioorg. Med. Chem. Lett. 2011, 21, 5859–5862. [Google Scholar] [CrossRef][Green Version]
- Cooper, A.G.; MacDonald, C.; Glass, M.; Hook, S.; Tyndall, J.D.A.; Vernall, A.J. Alkyl Indole-Based Cannabinoid Type 2 Receptor Tools: Exploration of Linker and Fluorophore Attachment. Eur. J. Med. Chem. 2018, 145, 770–789. [Google Scholar]
- Rinaldi-Carmona, M.; Barth, F.; Millan, J.; Derocq, J.M.; Casellas, P.; Congy, C.; Oustric, D.; Sarran, M.; Bouaboula, M.; Calandra, B.; et al. SR 144528, the First Potent and Selective Antagonist of the CB2 Cannabinoid Receptor. J. Pharmacol. Exp. Ther. 1998, 284, 644–650. [Google Scholar]
- Sexton, M.; Woodruff, G.; Horne, E.A.; Lin, Y.H.; Muccioli, G.G.; Bai, M.; Stern, E.; Bornhop, D.J.; Stella, N. NIR-Mbc94, a Fluorescent Ligand That Binds to Endogenous CB(2) Receptors and Is Amenable to High-Throughput Screening. Chem. Biol. 2011, 18, 563–568. [Google Scholar]
- Bai, M.; Sexton, M.; Stella, N.; Bornhop, D.J. MBC94, a Conjugable Ligand for Cannabinoid CB 2 Receptor Imaging. Bioconjug. Chem. 2008, 19, 988–992. [Google Scholar] [CrossRef]
- Zhang, S.; Shao, P.; Bai, M. In Vivo Type 2 Cannabinoid Receptor-Targeted Tumor Optical Imaging Using a near Infrared Fluorescent Probe. Bioconjug. Chem. 2013, 24, 1907–1916. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Shao, P.; Ling, X.; Yang, L.; Hou, W.; Thorne, S.H.; Beaino, W.; Anderson, C.J.; Ding, Y.; Bai, M. In Vivo Inflammation Imaging Using a CB2R-Targeted near Infrared Fluorescent Probe. Am. J. Nucl. Med. Mol. Imaging 2015, 5, 246–258. [Google Scholar]
- Wu, Z.; Shao, P.; Zhang, S.; Bai, M. Targeted Zwitterionic near Infrared Fluorescent Probe for Improved Imaging of Type 2 Cannabinoid Receptors. J. Biomed. Opt. 2014, 19, 36006. [Google Scholar] [CrossRef]
- Zhang, S.; Jia, N.; Shao, P.; Tong, Q.; Xie, X.-Q.; Bai, M. Target-Selective Phototherapy Using a Ligand-Based Photosensitizer for Type 2 Cannabinoid Receptor. Chem. Biol. 2014, 21, 338–344. [Google Scholar] [PubMed]
- Jia, N.; Zhang, S.; Shao, P.; Bagia, C.; Janjic, J.M.; Ding, Y.; Bai, M. Cannabinoid CB2 Receptor as a New Phototherapy Target for the Inhibition of Tumor Growth. Mol. Pharm. 2014, 11, 1919–1929. [Google Scholar] [CrossRef] [PubMed]
- Spinelli, F.; Capparelli, E.; Abate, C.; Colabufo, N.A.; Contino, M. Perspectives of Cannabinoid Type 2 Receptor (CB2R) Ligands in Neurodegenerative Disorders: Structure-Affinity Relationship (SAfiR) and Structure-Activity Relationship (SAR) Studies. J. Med. Chem. 2017, 60, 9913–9931. [Google Scholar]
- Aghazadeh Tabrizi, M.; Baraldi, P.G.; Saponaro, G.; Moorman, A.R.; Romagnoli, R.; Preti, D.; Baraldi, S.; Ruggiero, E.; Tintori, C.; Tuccinardi, T.; et al. Discovery of 7-Oxopyrazolo[1,5-a]Pyrimidine-6-Carboxamides as Potent and Selective CB(2) Cannabinoid Receptor Inverse Agonists. J. Med. Chem. 2013, 56, 4482–4496. [Google Scholar] [CrossRef] [PubMed]
- Ling, X.; Zhang, S.; Shao, P.; Li, W.; Yang, L.; Ding, Y.; Xu, C.; Stella, N.; Bai, M. A Novel Near-Infrared Fluorescence Imaging Probe That Preferentially Binds to Cannabinoid Receptors CB2R over CB1R. Biomaterials 2015, 57, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Ling, X.; Du, F.; Wang, Q.; Huang, W.; Wang, Z.; Ding, X.; Bai, M.; Wu, Z. Molecular Imaging of Pancreatic Duct Adenocarcinoma Using a Type 2 Cannabinoid Receptor-Targeted Near-Infrared Fluorescent Probe. Transl. Oncol. 2018, 11, 1065–1073. [Google Scholar] [PubMed]
- Wu, Z.; Shao, P.; Zhang, S.; Ling, X.; Bai, M. Molecular Imaging of Human Tumor Cells That Naturally Overexpress Type 2 Cannabinoid Receptors Using a Quinolone-Based near-Infrared Fluorescent Probe. J. Biomed. Opt. 2014, 19, 76016. [Google Scholar] [CrossRef] [PubMed]
- Cooper, A.G.; Oyagawa, C.R.M.; Manning, J.J.; Singh, S.; Hook, S.; Grimsey, N.L.; Glass, M.; Tyndall, J.D.A.; Vernall, A.J. Development of Selective, Fluorescent Cannabinoid Type 2 Receptor Ligands Based on a 1,8-Naphthyridin-2-(1H)-One-3-Carboxamide Scaffold. Medchemcomm 2018, 9, 2055–2067. [Google Scholar]
- Spinelli, F.; Giampietro, R.; Stefanachi, A.; Riganti, C.; Kopecka, J.; Abatematteo, F.S.; Leonetti, F.; Colabufo, N.A.; Mangiatordi, G.F.; Nicolotti, O.; et al. Design and Synthesis of Fluorescent Ligands for the Detection of Cannabinoid Type 2 Receptor (CB2R). Eur. J. Med. Chem. 2020, 188, 112037. [Google Scholar]
- Haider, A.; Kretz, J.; Gobbi, L.; Ahmed, H.; Atz, K.; Bürkler, M.; Bartelmus, C.; Fingerle, J.; Guba, W.; Ullmer, C.; et al. Structure-Activity Relationship Studies of Pyridine-Based Ligands and Identification of a Fluorinated Derivative for Positron Emission Tomography Imaging of Cannabinoid Type 2 Receptors. J. Med. Chem. 2019, 62, 11165–11181. [Google Scholar] [CrossRef]
- Haider, A.; Gobbi, L.; Kretz, J.; Ullmer, C.; Brink, A.; Honer, M.; Woltering, T.J.; Muri, D.; Iding, H.; Bürkler, M.; et al. Identification and Preclinical Development of a 2,5,6-Trisubstituted Fluorinated Pyridine Derivative as a Radioligand for the Positron Emission Tomography Imaging of Cannabinoid Type 2 Receptors. J. Med. Chem. 2020, 63, 10287–10306. [Google Scholar] [CrossRef]
- Kosar, M.; Sykes, D.A.; Viray, A.E.G.; Vitale, R.M.; Sarott, R.C.; Ganzoni, R.L.; Onion, D.; Tobias, J.M.; Leippe, P.; Ullmer, C.; et al. Platform Reagents Enable Synthesis of Ligand-Directed Covalent Probes: Study of Cannabinoid Receptor 2 in Live Cells. J. Am. Chem. Soc. 2023, 145, 15094–15108. [Google Scholar] [CrossRef]
- Khan, M.I.; Sobocińska, A.A.; Czarnecka, A.M.; Król, M.; Botta, B.; Szczylik, C. The Therapeutic Aspects of the Endocannabinoid System (ECS) for Cancer and Their Development: From Nature to Laboratory. Curr. Pharm. Des. 2016, 22, 1756–1766. [Google Scholar] [CrossRef]
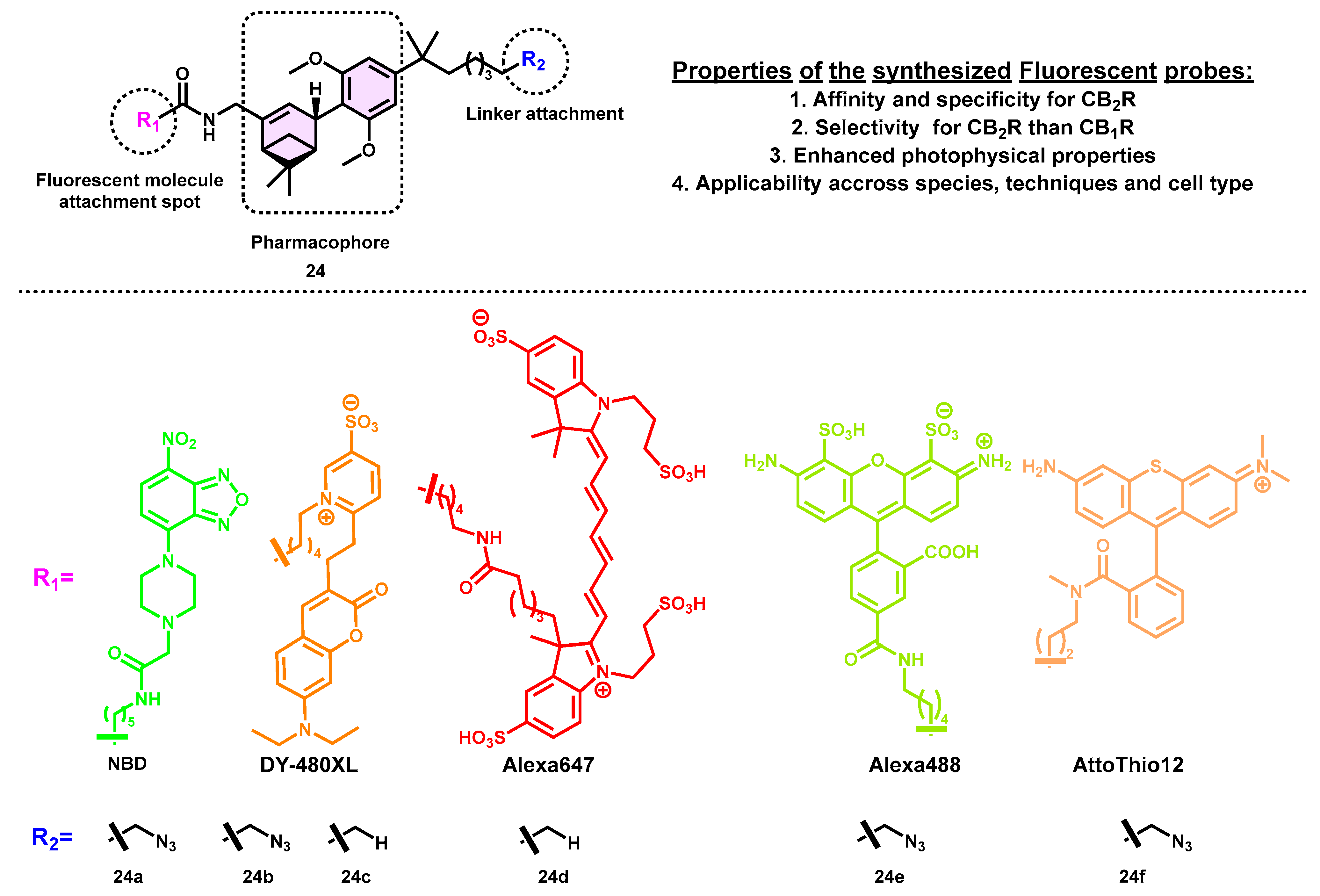

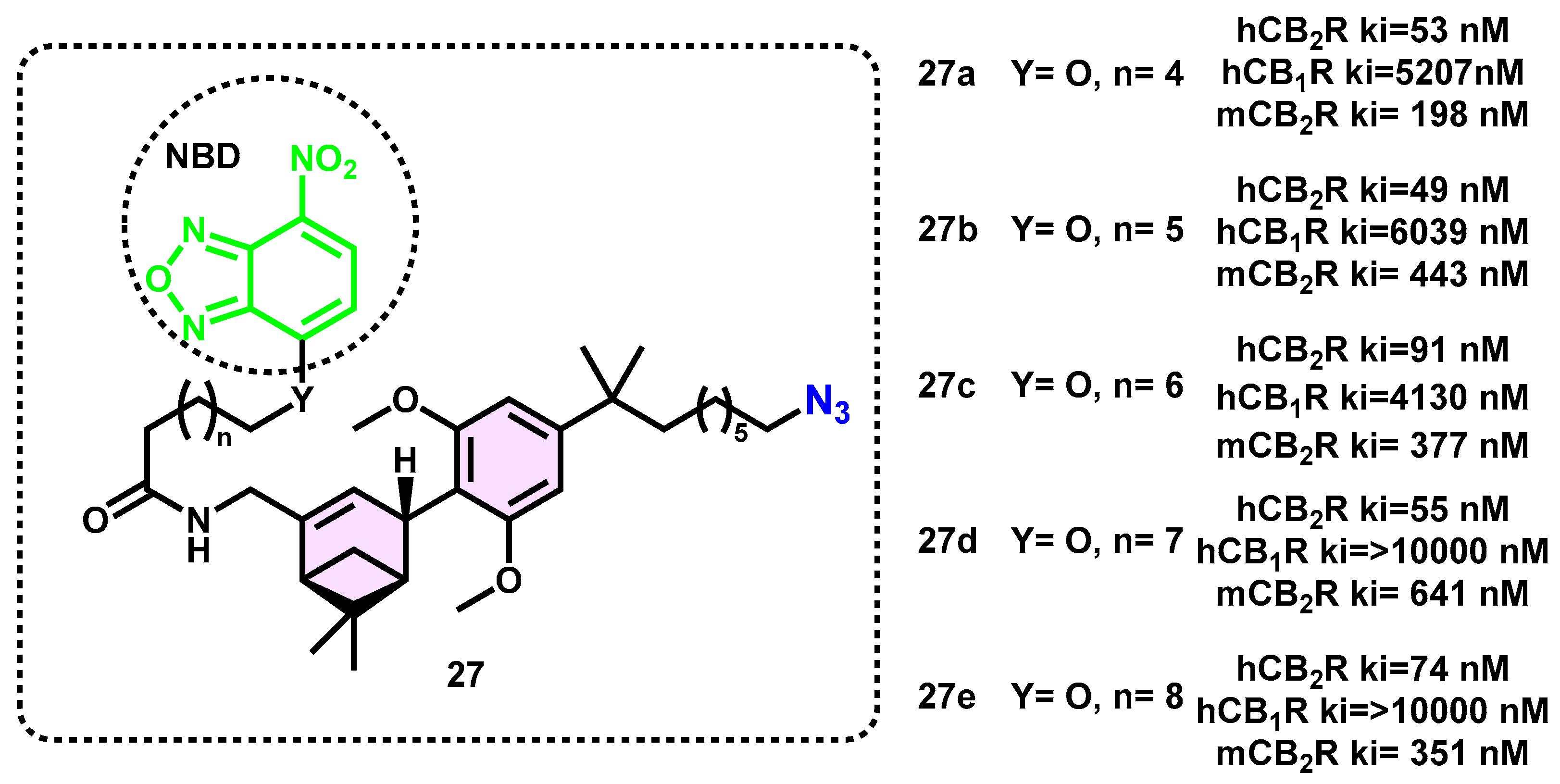
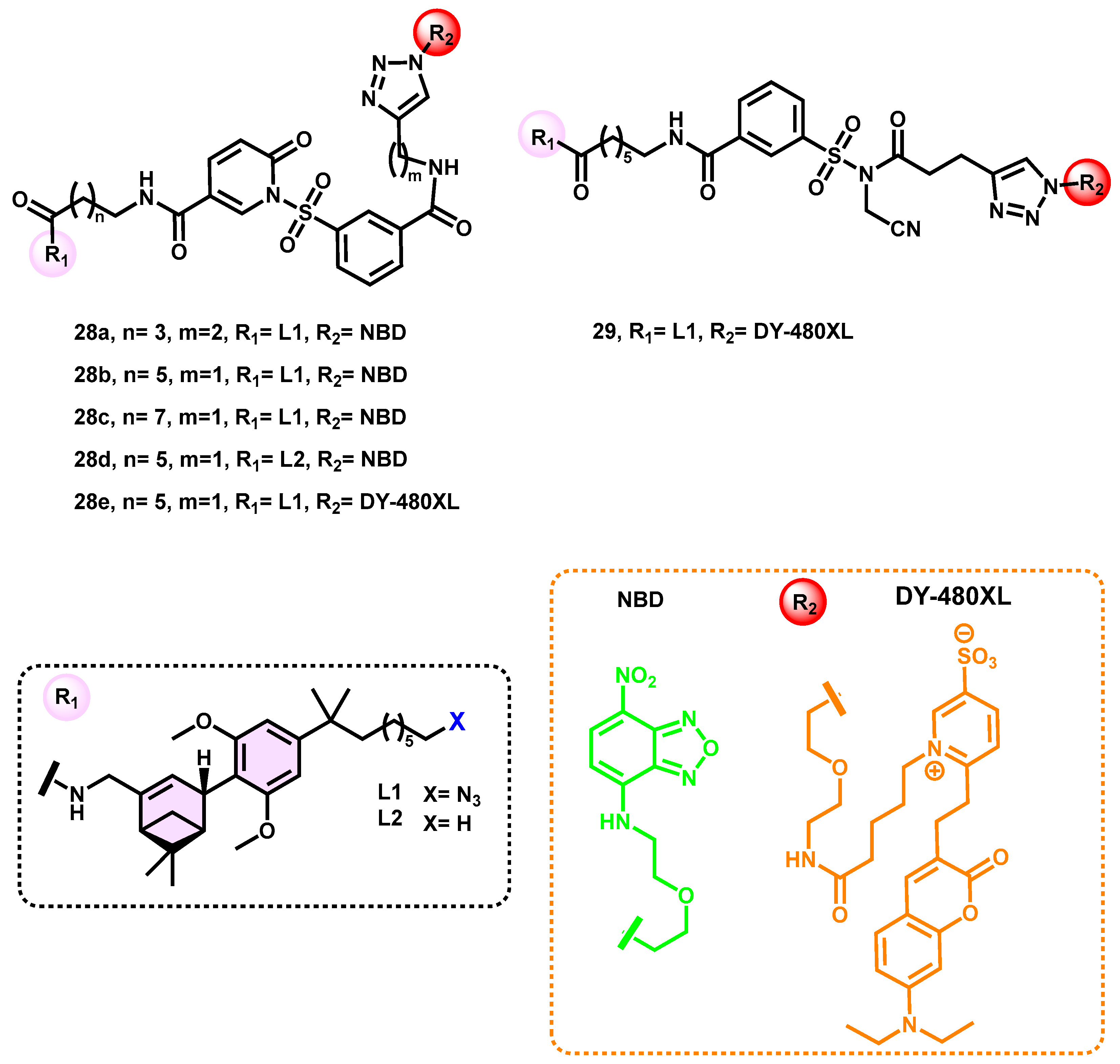
| Ki (nM) | EC50 (nM) | ||||||
|---|---|---|---|---|---|---|---|
| Compound | Dye | hCB2R | hCB1R | mCB2R | hCB2R | hCB1R | mCB2R |
| 24a | NBD | 4.2 | >10,000 | n.d. | n.d. | >10,000 | n.d. |
| 24b | DY-480XL | 99 | 4031 | 1986 | >10,000 | >10,000 | >10,000 |
| 24c | DY-480XL | 21 | 2378 | 1459 | 171 | 2152 | 118 |
| 24d | Alexa647 | 2565 | >10,000 | >10,000 | 25 | 2565 | 370 |
| 24e | Alexa488 | 268 | >10,000 | 1204 | n.d | n.d. | n.d. |
| 24f | Atto Thio12 | 4.7 | 1075 | 1.1 | 5.6 | >10,000 | 17 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bhattacharjee, P.; Iyer, M.R. Rational Design, Synthesis, and Evaluation of Fluorescent CB2 Receptor Ligands for Live-Cell Imaging: A Comprehensive Review. Pharmaceuticals 2023, 16, 1235. https://doi.org/10.3390/ph16091235
Bhattacharjee P, Iyer MR. Rational Design, Synthesis, and Evaluation of Fluorescent CB2 Receptor Ligands for Live-Cell Imaging: A Comprehensive Review. Pharmaceuticals. 2023; 16(9):1235. https://doi.org/10.3390/ph16091235
Chicago/Turabian StyleBhattacharjee, Pinaki, and Malliga R. Iyer. 2023. "Rational Design, Synthesis, and Evaluation of Fluorescent CB2 Receptor Ligands for Live-Cell Imaging: A Comprehensive Review" Pharmaceuticals 16, no. 9: 1235. https://doi.org/10.3390/ph16091235
APA StyleBhattacharjee, P., & Iyer, M. R. (2023). Rational Design, Synthesis, and Evaluation of Fluorescent CB2 Receptor Ligands for Live-Cell Imaging: A Comprehensive Review. Pharmaceuticals, 16(9), 1235. https://doi.org/10.3390/ph16091235







