Liposomes for the Treatment of Brain Cancer—A Review
Abstract
1. Introduction
1.1. Conventional Treatment Methods
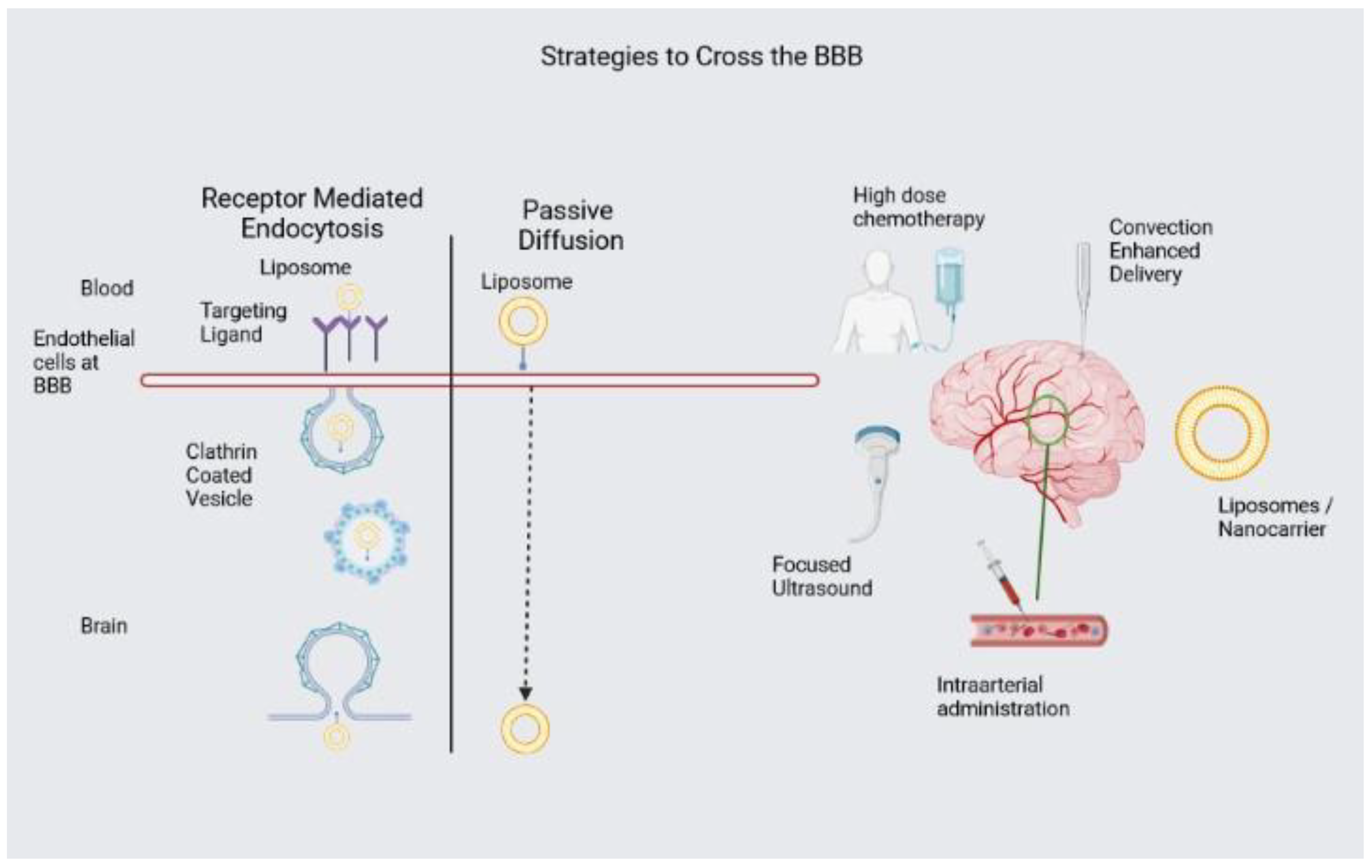
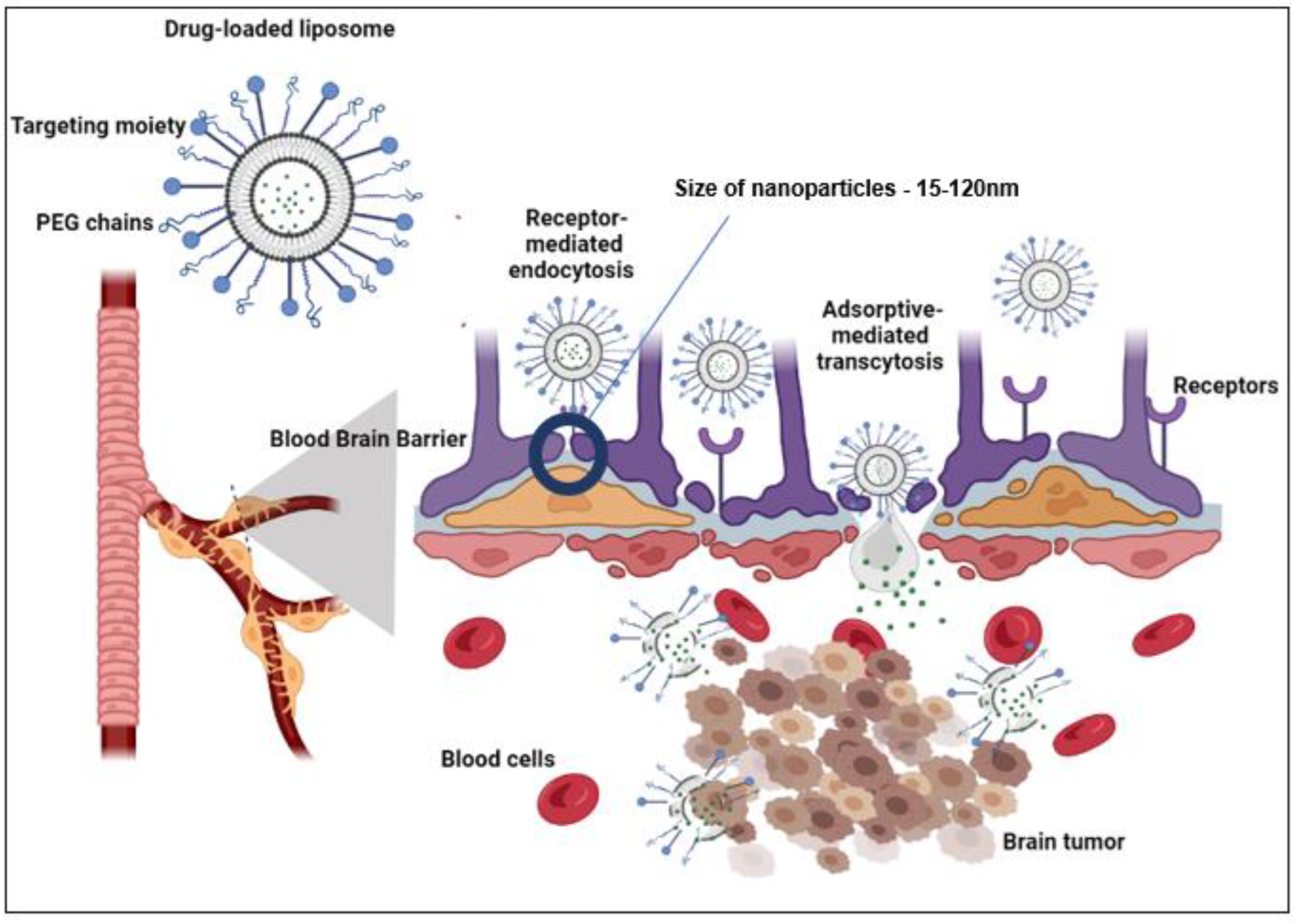
1.2. Drug Delivery across the BBB
| Internalization Pathway | Ligand | Remarks | Reference |
|---|---|---|---|
| Receptor-Mediated Transcytosis | Transferrin (Tf) | -PEGylated liposomes covalently bonded to poly-L-arginine peptide were administered to rats in vivo -β-galactosidase activity was reported to be two-fold higher than the control | [60] |
| Tripeptide glutathione (GSH) | -In vivo study conducted of the administration of ribavirin GSH-PEG liposomes at 50 mg/kg ribavirin -Three-fold increase in ribavirin concentration in the brain in mice treated with GSH-PEG liposomes compared to control | [64] | |
| Low-density lipoprotein receptor (LDLR) | -The study involved in vivo intravenous administration of melanotransferrin (P94) -P94 was accumulated in the mouse brain at an amount over 14 times higher than that of the control group (holo-transferrin) | [65] | |
| Receptor-Mediated Endocytosis | Anti-transferrin receptor IgG | -Gold nanoparticles were conjugated with IgG, and its uptake by brain endothelial cells was studied in vivo -Higher accumulation of IgG was observed in mice when compared to the control | [54] |
| Adsorptive Mediated Transcytosis | Cationized human serum albumin (CHSA) | -CBSA proteins were covalently conjugated to PEGylated liposomes -In vitro studies on porcine brain models exhibited higher uptake in brain capillary endothelial cells (BCEC) when compared to control liposomes | [66] |
| Adsorptive Mediated Endocytosis | Immunoglobulin γ (IgG) | -Fluorophores conjugated with IgG were administered in vitro to human induced pluripotent stem-cell-derived BECs (iBECs) -Fluorescence was noted to be increased in cells treated with IgG-conjugated fluorophores due to endocytosis of the IgG across the BBB | [67] |
1.3. Drug Delivery Challenges across the BBB
1.4. Drug Delivery Platforms to cross the BBB
2. Nanoparticle-Based Drug Delivery Systems
2.1. Liposomes
2.1.1. Preparation and Functionalization
- Direct dissolution: In this method, lipids are directly dissolved in an aqueous medium, forming liposomes.
- Thin film hydration: A thin lipid film, formed by evaporating lipids dissolved in an organic solvent, is deposited on a glass substrate. The film is then hydrated using an aqueous solution, leading to the formation of liposomes.
- Electroformation: This method involves applying an external electric field during the hydration step of the thin lipid film. The electric field enhances water influx between the bilayer sheets of the thin film, resulting in the formation of giant unilamellar vesicles (GUVs).
2.1.2. Functionalization Approaches
2.1.3. Advantages of Liposomes over Other Nanocarriers
3. Liposomal Treatments for Brain Tumors
3.1. Preclinical Studies
3.2. Clinical Trials
4. Concluding Remarks
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviation
| BBB | Blood–Brain Barrier |
| CNS | Central Nervous System |
| Nanoparticle DDs | Smart Drug Delivery Systems |
| CDDs | Conventional Drug Delivery Systems |
| MOA | Mechanism Of Action |
| TAA | Tumor-Associated Antigen |
| TSA | Tumor-Specific Antigen |
| DC | Dendritic Cell |
| TTP | Time To Progression |
| OS | Overall Survival |
| ACT | Adoptive Cell Transfer |
| IL7 | Interleukin-7 |
| IL15 | Interleukin-15 |
| CAR | Chimeric Antigen Receptor |
| GBM | Glioblastoma Multiforme |
| IL13Rα2 | Interleukin-13 Receptor A2 |
| mAb/moAb | Monoclonal Antibody |
| ADCC | Antibody-Dependent Cellular Cytotoxicity |
| CMC | Complement-Mediated Cytotoxicity |
| EGFR | Epidermal Growth Factor Receptor |
| CTLA4 | Cytotoxic T Lymphocyte-Associated Antigen 4 |
| VEGF | Vascular Endothelial Growth Factor |
| BEV | Bevacizumab |
| KPS | Karnofsky Performance Status Karnofsky Performance Status |
| TGF | Transforming Growth Factor |
| TJ | Tight Junction |
| AJ | Adherens Junction |
| ABC | Atp-Binding Cassette |
| Pgp | P-Glycoprotein |
| MRP4 | Multidrug Resistance Proteins 4 |
| BCRP | Breast Cancer Resistance Protein |
| DOX | Doxorubicin |
| BTB | Blood–Tumor Barrier |
| TMT | Transporter-Mediated Transcytosis |
| RME | Receptor-Mediated Endocytosis |
| AMT | Adsorptive-Mediated Transcytosis |
| RMT | Receptor-Mediated Transcytosis |
| AMT | Adsorptive- Mediated Transcytosis |
| ADR. | Adriamycin |
| LRP | Low-Density Lipoprotein Receptor-Related Proteins |
| PEG | Polyethylene Glycol |
| DSPC | Distearoylphosphatidylcholine |
| HSPC | Hydrogenated soybean phosphatidylcholine |
| DSPE-PEG2000 | 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[carboxy(polyethylene glycol)-1000] |
| HIR | Human Insulin Receptor |
| RES | Reticuloendothelial System |
| MOF | Metal–Organic Framework |
| QD | Quantum Dots |
| NP | Polymeric Nanoparticles |
| SUV | Small Unilamellar Vesicles |
| LUV | Large Unilamellar Vesicles |
| MLV | Multilamellar Vesicles |
| FUS | Focused Ultrasound |
| CB | Carboplatin |
| CPP | Cell-Penetrating Peptide |
| Tf | Transferrin |
| TMZ | Temozolomide |
| PAP | Pro-Apoptotic Peptide |
| ELE | Elemene |
| CTX | Cabazitaxel |
| TPGS | Alpha-Tocopheryl Polyethylene Glycol 1000 Succinate |
| TML | Thermosensitive Magnetic Liposome |
| TRAIL | Tumor Necrosis Factor-Related Apoptosis-Inducing Ligand |
| MAN | P-Aminophenyl-A-D-Manno-Pyranoside |
| WGA | Wheat Germ Agglutinin |
References
- Dunn, B. Cancer: Solving an Age-Old Problem. Nature 2012, 483, S2–S6. [Google Scholar] [CrossRef]
- Hill, B.T. Etiology of Cancer. Clin. Ophthalmic Oncol. 2019, 11–17. [Google Scholar]
- Jones, P.A.; Baylin, S.B. The Epigenomics of Cancer. Cell 2007, 128, 683. [Google Scholar] [CrossRef] [PubMed]
- Feinberg, A.P. The Epigenetics of Cancer Etiology. Semin. Cancer Biol. 2004, 14, 427–432. [Google Scholar] [CrossRef]
- Brown, G. Oncogenes, Proto-Oncogenes, and Lineage Restriction of Cancer Stem Cells. Int. J. Mol. Sci. 2021, 22, 9367. [Google Scholar] [CrossRef] [PubMed]
- Seemayer, T.A.; Cavenee, W.K. Molecular Mechanisms of Oncogenesis. Pathol. Rev. 1990, 1, 155–169. [Google Scholar]
- Chen, L.; Liu, S.; Tao, Y. Regulating Tumor Suppressor Genes: Post-Translational Modifications. Signal Transduct. Target. Ther. 2020, 5, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Deo, S.V.S.; Sharma, J.; Kumar, S. GLOBOCAN 2020 Report on Global Cancer Burden: Challenges and Opportunities for Surgical Oncologists. Ann. Surg. Oncol. 2022, 29, 6494–6500. [Google Scholar] [CrossRef]
- Barnholtz-Sloan, J.S.; Ostrom, Q.T.; Cote, D. Epidemiology of Brain Tumors. Neurol. Clin. 2018, 36, 392–419. [Google Scholar] [CrossRef]
- Louis, D.N.; Perry, A.; Reifenberger, G.; von Deimling, A.; Figarella-Branger, D.; Cavenee, W.K.; Ohgaki, H.; Wiestler, O.D.; Kleihues, P.; Ellison, D.W. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: A Summary. Acta Neuropathol. 2016, 131, 803–820. [Google Scholar] [CrossRef]
- Bertolini, F.; Spallanzani, A.; Fontana, A.; Depenni, R.; Luppi, G. Brain Metastases: An Overview. CNS Oncol. 2015, 4, 37–46. [Google Scholar] [CrossRef]
- Sharma, P.; Gupta, R. Meningioma. PET/MR Imaging A Case-Based Approach 2022, 45, 293–294. [Google Scholar]
- Kapoor, M.; Gupta, V. Astrocytoma. Tumors Cancers Cent. Peripher. Nerv. Syst. 2022, 212. [Google Scholar]
- Davis, M.E. Glioblastoma: Overview of Disease and Treatment. Clin. J. Oncol. Nurs. 2016, 20, S2–S8. [Google Scholar] [CrossRef]
- Taylor, O.G.; Brzozowski, J.S.; Skelding, K.A. Glioblastoma Multiforme: An Overview of Emerging Therapeutic Targets. Front. Oncol. 2019, 9, 933. [Google Scholar] [CrossRef]
- Park, J.H.; de Lomana, A.L.G.; Marzese, D.M.; Juarez, T.; Feroze, A.; Hothi, P.; Cobbs, C.; Patel, A.P.; Kesari, S.; Huang, S.; et al. A Systems Approach to Brain Tumor Treatment. Cancers 2021, 13, 3152. [Google Scholar] [CrossRef]
- Hossen, S.; Hossain, M.K.; Basher, M.K.; Mia, M.N.H.; Rahman, M.T.; Uddin, M.J. Smart Nanocarrier-Based Drug Delivery Systems for Cancer Therapy and Toxicity Studies: A Review. J. Adv. Res. 2019, 15, 1–18. [Google Scholar] [CrossRef]
- Huda, S.; Alam, M.A.; Sharma, P.K. Smart Nanocarriers-Based Drug Delivery for Cancer Therapy: An Innovative and Developing Strategy. J. Drug Deliv. Sci. Technol. 2020, 60, 102018. [Google Scholar] [CrossRef]
- Li, X.Y.; Zhao, Y.; Sun, M.G.; Shi, J.F.; Ju, R.J.; Zhang, C.X.; Li, X.T.; Zhao, W.Y.; Mu, L.M.; Zeng, F.; et al. Multifunctional Liposomes Loaded with Paclitaxel and Artemether for Treatment of Invasive Brain Glioma. Biomaterials 2014, 35, 5591–5604. [Google Scholar] [CrossRef]
- Chabner, B.A.; Roberts, T.G. Chemotherapy and the War on Cancer. Nat. Rev. Cancer 2005, 5, 65–72. [Google Scholar] [CrossRef]
- Nurgali, K.; Jagoe, R.T.; Abalo, R. Editorial: Adverse Effects of Cancer Chemotherapy: Anything New to Improve Tolerance and Reduce Sequelae? Front Pharmacol. 2018, 9, 245. [Google Scholar] [CrossRef]
- Monsuez, J.J.; Charniot, J.C.; Vignat, N.; Artigou, J.Y. Cardiac Side-Effects of Cancer Chemotherapy. Int. J. Cardiol. 2010, 144, 3–15. [Google Scholar] [CrossRef]
- Waldman, A.D.; Fritz, J.M.; Lenardo, M.J. A Guide to Cancer Immunotherapy: From T Cell Basic Science to Clinical Practice. Nat. Rev. Immunol. 2020, 20, 651–668. [Google Scholar] [CrossRef]
- Saxena, M.; van der Burg, S.H.; Melief, C.J.M.; Bhardwaj, N. Therapeutic Cancer Vaccines. Nat. Rev. Cancer 2021, 21, 360–378. [Google Scholar] [CrossRef]
- Lin, M.J.; Svensson-Arvelund, J.; Lubitz, G.S.; Marabelle, A.; Melero, I.; Brown, B.D.; Brody, J.D. Cancer Vaccines: The next Immunotherapy Frontier. Nat. Cancer 2022, 3, 911–926. [Google Scholar] [CrossRef]
- Platten, M.; Bunse, L.; Wick, A.; Bunse, T.; Le Cornet, L.; Harting, I.; Sahm, F.; Sanghvi, K.; Tan, C.L.; Poschke, I.; et al. A Vaccine Targeting Mutant IDH1 in Newly Diagnosed Glioma. Nature 2021, 592, 463–468. [Google Scholar] [CrossRef]
- Yu, J.S.; Liu, G.; Ying, H.; Yong, W.H.; Black, K.L.; Wheeler, C.J. Vaccination with Tumor Lysate-Pulsed Dendritic Cells Elicits Antigen-Specific, Cytotoxic T-Cells in Patients with Malignant Glioma. Cancer Res. 2004, 64, 4943–4949. [Google Scholar] [CrossRef]
- Srivastava, S.; Salim, N.; Robertson, M. Interleukin-18: Biology and Role in the Immunotherapy of Cancer. Curr. Med. Chem. 2010, 17, 3353–3357. [Google Scholar] [CrossRef]
- Dudley, M.E.; Rosenberg, S.A. Adoptive-Cell-Transfer Therapy for the Treatment of Patients with Cancer. Nat. Rev. Cancer 2003, 3, 666–675. [Google Scholar] [CrossRef]
- Rosenberg, S.A.; Restifo, N.P.; Yang, J.C.; Morgan, R.A.; Dudley, M.E. Adoptive Cell Transfer: A Clinical Path to Effective Cancer Immunotherapy. Nat. Rev. Cancer 2008, 8, 299–308. [Google Scholar] [CrossRef]
- Brown, C.E.; Badie, B.; Barish, M.E.; Weng, L.; Ostberg, J.R.; Chang, W.C.; Naranjo, A.; Starr, R.; Wagner, J.; Wright, C.; et al. Bioactivity and Safety of IL13Rα2-Redirected Chimeric Antigen Receptor CD8+ T Cells in Patients with Recurrent Glioblastoma. Clin. Cancer Res. 2015, 21, 4062–4072. [Google Scholar] [CrossRef]
- Boskovitz, A.; Wikstrand, C.J.; Kuan, C.T.; Zalutsky, M.R.; Reardon, D.A.; Bigner, D.D. Monoclonal Antibodies for Brain Tumour Treatment. Expert Opin. Biol. Ther. 2005, 4, 1453–1471. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Kumar, N.K.; Dwiwedi, P.; Charan, J.; Kaur, R.; Sidhu, P.; Chugh, V.K. Monoclonal Antibodies: A Review. Curr. Clin. Pharmacol. 2018, 13, 651–654. [Google Scholar] [CrossRef]
- Quinteros, D.A.; Bermúdez, J.M.; Ravetti, S.; Cid, A.; Allemandi, D.A.; Palma, S.D. Therapeutic Use of Monoclonal Antibodies: General Aspects and Challenges for Drug Delivery. Nanostruct. Drug Deliv. 2017, 10, 807–833. [Google Scholar]
- Lu, R.M.; Hwang, Y.C.; Liu, I.J.; Lee, C.C.; Tsai, H.Z.; Li, H.J.; Wu, H.C. Development of Therapeutic Antibodies for the Treatment of Diseases. J. Biomed. Sci. 2020, 27, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Redman, J.M.; Hill, E.M.; AlDeghaither, D.; Weiner, L.M. Mechanisms of Action of Therapeutic Antibodies for Cancer. Mol. Immunol. 2015, 67, 28–45. [Google Scholar] [CrossRef]
- Scott, A.M.; Allison, J.P.; Wolchok, J.D. Monoclonal Antibodies in Cancer Therapy. Cancer Immun. 2012, 12, 14. [Google Scholar]
- Lai, A.; Tran, A.; Nghiemphu, P.L.; Pope, W.B.; Solis, O.E.; Selch, M.; Filka, E.; Yong, W.H.; Mischel, P.S.; Liau, L.M.; et al. Phase II Study of Bevacizumab plus Temozolomide during and after Radiation Therapy for Patients with Newly Diagnosed Glioblastoma Multiforme. J. Clin. Oncol. 2011, 29, 142–148. [Google Scholar] [CrossRef]
- Bartels, U.; Wolff, J.; Gore, L.; Dunkel, I.; Gilheeney, S.; Allen, J.; Goldman, S.; Yalon, M.; Packer, R.J.; Korones, D.N.; et al. Phase 2 Study of Safety and Efficacy of Nimotuzumab in Pediatric Patients with Progressive Diffuse Intrinsic Pontine Glioma. Neuro Oncol. 2014, 16, 1554. [Google Scholar] [CrossRef] [PubMed]
- Crombet Ramos, T.; Mestre Fernández, B.; Mazorra Herrera, Z.; Iznaga Escobar, N.E. Nimotuzumab for Patients With Inoperable Cancer of the Head and Neck. Front Oncol. 2020, 10, 817. [Google Scholar] [CrossRef]
- Daneman, R.; Prat, A. The Blood-Brain Barrier. Cold Spring Harb. Perspect. Biol. 2015, 7, 20–42. [Google Scholar] [CrossRef]
- Abbott, N.J.; Patabendige, A.A.K.; Dolman, D.E.M.; Yusof, S.R.; Begley, D.J. Structure and Function of the Blood-Brain Barrier. Neurobiol. Dis. 2010, 37, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Yhee, J.Y.; Son, S.; Son, S.; Joo, M.K.; Kwon, I.C. The EPR Effect in Cancer Therapy. Cancer Target. Drug Deliv. Elus. Dream 2013, 1, 621–632. [Google Scholar]
- Maeda, H. Tumor-Selective Delivery of Macromolecular Drugs via the EPR Effect: Background and Future Prospects. Bioconjugate Chem. 2010, 21, 794–802. [Google Scholar] [CrossRef] [PubMed]
- Maruyama, K. Intracellular Targeting Delivery of Liposomal Drugs to Solid Tumors Based on EPR Effects. Adv. Drug Deliv. Rev. 2011, 63, 161–169. [Google Scholar] [CrossRef]
- Arvanitis, C.D.; Ferraro, G.B.; Jain, R.K. The Blood–Brain Barrier and Blood–Tumour Barrier in Brain Tumours and Metastases. Nat. Rev. Cancer 2019, 20, 26–41. [Google Scholar] [CrossRef]
- Hardwick, J.P.; Eckman, K.; Lee, Y.K.; Abdelmegeed, M.A.; Esterle, A.; Chilian, W.M.; Chiang, J.Y.; Song, B.J. Eicosanoids in Metabolic Syndrome. Adv. Pharmacol. 2013, 66, 157–266. [Google Scholar]
- Pardridge, W.M. Drug Transport across the Blood-Brain Barrier. J. Cereb. Blood Flow Metab. 2012, 32, 1929–1942. [Google Scholar] [CrossRef] [PubMed]
- Belykh, E.; Shaffer, K.V.; Lin, C.; Byvaltsev, V.A.; Preul, M.C.; Chen, L. Blood-Brain Barrier, Blood-Brain Tumor Barrier, and Fluorescence-Guided Neurosurgical Oncology: Delivering Optical Labels to Brain Tumors. Front. Oncol. 2020, 10, 739. [Google Scholar] [CrossRef]
- Steeg, P.S. The Blood-Tumour Barrier in Cancer Biology and Therapy. Nat. Rev. Clin. Oncol. 2021, 18, 693–714. [Google Scholar] [CrossRef]
- Ohtsuki, S.; Terasaki, T. Contribution of Carrier-Mediated Transport Systems to the Blood-Brain Barrier as a Supporting and Protecting Interface for the Brain; Importance for CNS Drug Discovery and Development. Pharm. Res. 2007, 24, 1745–1758. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.U.; Miao, T.; Ju, X.; Guo, Q.; Han, L. Carrier-Mediated Transportation through BBB. Brain Target. Drug Deliv. Syst. A Focus Nanotechnol. Nanopart. 2019, 32, 129–158. [Google Scholar]
- Zhang, W.; Liu, Q.Y.; Haqqani, A.S.; Leclerc, S.; Liu, Z.; Fauteux, F.; Baumann, E.; Delaney, C.E.; Ly, D.; Star, A.T.; et al. Differential Expression of Receptors Mediating Receptor-Mediated Transcytosis (RMT) in Brain Microvessels, Brain Parenchyma and Peripheral Tissues of the Mouse and the Human. Fluids Barriers CNS 2020, 17, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Johnsen, K.B.; Bak, M.; Kempen, P.J.; Melander, F.; Burkhart, A.; Thomsen, M.S.; Nielsen, M.S.; Moos, T.; Andresen, T.L. Antibody Affinity and Valency Impact Brain Uptake of Transferrin Receptor-Targeted Gold Nanoparticles. Theranostics 2018, 8, 3416–3436. [Google Scholar] [CrossRef]
- Pulgar, V.M. Transcytosis to Cross the Blood Brain Barrier, New Advancements and Challenges. Front Neurosci. 2019, 13, 1019. [Google Scholar] [CrossRef]
- Simonneau, C.; Duschmalé, M.; Gavrilov, A.; Brandenberg, N.; Hoehnel, S.; Ceroni, C.; Lassalle, E.; Kassianidou, E.; Knoetgen, H.; Niewoehner, J.; et al. Investigating Receptor-Mediated Antibody Transcytosis Using Blood–Brain Barrier Organoid Arrays. Fluids Barriers CNS 2021, 18, 1–17. [Google Scholar] [CrossRef]
- Gabathuler, R.; Arthur, G.; Kennard, M.; Chen, Q.; Tsai, S.; Yang, J.; Schoorl, W.; Vitalis, T.Z.; Jefferies, W.A. Development of a Potential Protein Vector (NeuroTrans) to Deliver Drugs across the Blood–Brain Barrier. Int. Congr. Ser. 2005, 1277, 171–184. [Google Scholar] [CrossRef]
- Miyajima, Y.; Nakamura, H.; Kuwata, Y.; Lee, J.D.; Masunaga, S.; Ono, K.; Maruyama, K. Transferrin-Loaded Nido-Carborane Liposomes: Tumor-Targeting Boron Delivery System for Neutron Capture Therapy. Bioconjugate Chem. 2006, 17, 1314–1320. [Google Scholar] [CrossRef]
- Zhang, Y.; Schlachetzki, F.; Pardridge, W.M. Global Non-Viral Gene Transfer to the Primate Brain Following Intravenous Administration. Mol. Ther. 2003, 7, 11–18. [Google Scholar] [CrossRef]
- Lu, W. Adsorptive-Mediated Brain Delivery Systems. Curr. Pharm. Biotechnol. 2012, 13, 2340–2348. [Google Scholar] [CrossRef]
- Hervé, F.; Ghinea, N.; Scherrmann, J.M. CNS Delivery via Adsorptive Transcytosis. AAPS J. 2008, 10, 455–472. [Google Scholar] [CrossRef] [PubMed]
- Vieira, D.B.; Gamarra, L.F. Getting into the Brain: Liposome-Based Strategies for Effective Drug Delivery across the Blood-Brain Barrier. Int. J. Nanomed. 2016, 11, 5381–5414. [Google Scholar] [CrossRef]
- Mahringer, A.; Puris, E.; Fricker, G. Crossing the Blood-Brain Barrier: A Review on Drug Delivery Strategies Using Colloidal Carrier Systems. Neurochem. Int. 2021, 147, 105–117. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Gaillard, P.J.; de Lange, E.C.M.; Hammarlund-Udenaes, M. Targeted Brain Delivery of Methotrexate by Glutathione PEGylated Liposomes: How Can the Formulation Make a Difference? Eur. J. Pharm. Biopharm. 2019, 139, 194–204. [Google Scholar] [CrossRef] [PubMed]
- Demeule, M.; Poirier, J.; Jodoin, J.; Bertrand, Y.; Desrosiers, R.R.; Dagenais, C.; Nguyen, T.; Lanthier, J.; Gabathuler, R.; Kennard, M.; et al. High Transcytosis of Melanotransferrin (P94) across the Blood-Brain Barrier. J. Neurochem. 2002, 83, 924–933. [Google Scholar] [CrossRef]
- Thöle, M.; Nobmann, S.; Huwyler, J.; Bartmann, A.; Fricker, G. Uptake of Cationzied Albumin Coupled Liposomes by Cultured Porcine Brain Microvessel Endothelial Cells and Intact Brain Capillaries. J. Drug Target. 2002, 10, 337–344. [Google Scholar] [CrossRef]
- Ruano-Salguero, J.S.; Lee, K.H. Antibody Transcytosis across Brain Endothelial-like Cells Occurs Nonspecifically and Independent of FcRn. Sci. Rep. 2020, 10, 36–85. [Google Scholar] [CrossRef]
- Achar, A.; Myers, R.; Ghosh, C. Drug Delivery Challenges in Brain Disorders across the Blood–Brain Barrier: Novel Methods and Future Considerations for Improved Therapy. Biomedicines 2021, 9, 18–34. [Google Scholar] [CrossRef]
- Bors, L.A.; Erdö, F. Overcoming the Blood–Brain Barrier. Challenges and Tricks for CNS Drug Delivery. Sci. Pharm. 2019, 87, 6. [Google Scholar] [CrossRef]
- Wu, D.; Chen, Q.; Chen, X.; Han, F.; Chen, Z.; Wang, Y. The Blood–Brain Barrier: Structure, Regulation, and Drug Delivery. Signal Transduct. Target. Ther. 2023, 8, 217. [Google Scholar] [CrossRef]
- Bellettato, C.M.; Scarpa, M. Possible Strategies to Cross the Blood-Brain Barrier. Ital. J. Pediatr. 2018, 44, 127–133. [Google Scholar] [CrossRef]
- Banks, W.A. Characteristics of Compounds That Cross the Blood-Brain Barrier. BMC Neurol. 2009, 9, S3. [Google Scholar] [CrossRef] [PubMed]
- Khawli, L.A.; Prabhu, S. Drug Delivery across the Blood–Brain Barrier. Mol. Pharm. 2013, 10, 1471–1472. [Google Scholar] [CrossRef] [PubMed]
- Ceña, V.; Játiva, P. Nanoparticle Crossing of Blood–Brain Barrier: A Road to New Therapeutic Approaches to Central Nervous System Diseases. Nanomedicine 2018, 13, 1513–1516. [Google Scholar] [CrossRef]
- Hersh, A.M.; Alomari, S.; Tyler, B.M. Crossing the Blood-Brain Barrier: Advances in Nanoparticle Technology for Drug Delivery in Neuro-Oncology. Int. J. Mol. Sci. 2022, 23, 4153. [Google Scholar] [CrossRef]
- Terstappen, G.C.; Meyer, A.H.; Bell, R.D.; Zhang, W. Strategies for Delivering Therapeutics across the Blood–Brain Barrier. Nat. Rev. Drug Discov. 2021, 20, 362–383. [Google Scholar] [CrossRef]
- Liu, D.; Yang, F.; Xiong, F.; Gu, N. The Smart Drug Delivery System and Its Clinical Potential. Theranostics 2016, 6, 1306. [Google Scholar] [CrossRef]
- Pinheiro, R.G.R.; Coutinho, A.J.; Pinheiro, M.; Neves, A.R. Nanoparticles for Targeted Brain Drug Delivery: What Do We Know? Int. J. Mol. Sci. 2021, 22, 11654. [Google Scholar] [CrossRef]
- Ghezzi, M.; Pescina, S.; Padula, C.; Santi, P.; Del Favero, E.; Cantù, L.; Nicoli, S. Polymeric Micelles in Drug Delivery: An Insight of the Techniques for Their Characterization and Assessment in Biorelevant Conditions. J. Control. Release 2021, 332, 312–336. [Google Scholar] [CrossRef] [PubMed]
- Ravi Kiran, A.V.V.V.; Kusuma Kumari, G.; Krishnamurthy, P.T. Carbon Nanotubes in Drug Delivery: Focus on Anticancer Therapies. J. Drug Deliv. Sci. 2020, 59, 101892. [Google Scholar] [CrossRef]
- Dadfar, S.M.; Roemhild, K.; Drude, N.I.; von Stillfried, S.; Knüchel, R.; Kiessling, F.; Lammers, T. Iron Oxide Nanoparticles: Diagnostic, Therapeutic and Theranostic Applications. Adv. Drug Deliv. Rev. 2019, 138, 302–325. [Google Scholar] [CrossRef]
- Sun, Y.; Zheng, L.; Yang, Y.; Qian, X.; Fu, T.; Li, X.; Yang, Z.; Yan, H.; Cui, C.; Tan, W. Metal–Organic Framework Nanocarriers for Drug Delivery in Biomedical Applications. Nano-Micro Lett. 2020, 12, 1–29. [Google Scholar] [CrossRef]
- Badıllı, U.; Mollarasouli, F.; Bakirhan, N.K.; Ozkan, Y.; Ozkan, S.A. Role of Quantum Dots in Pharmaceutical and Biomedical Analysis, and Its Application in Drug Delivery. TrAC Trends Anal. Chem. 2020, 131, 116013. [Google Scholar] [CrossRef]
- Zielinska, A.; Carreiró, F.; Oliveira, A.M.; Neves, A.; Pires, B.; Nagasamy Venkatesh, D.; Durazzo, A.; Lucarini, M.; Eder, P.; Silva, A.M.; et al. Polymeric Nanoparticles: Production, Characterization, Toxicology and Ecotoxicology. Molecules 2020, 25, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Svenson, S.; Tomalia, D.A. Dendrimers in Biomedical Applications—Reflections on the Field. Adv. Drug Deliv. Rev. 2012, 64, 102–115. [Google Scholar] [CrossRef]
- Hu, Y.-J.; Ju, R.-J.; Zeng, F.; Qi, X.-R.; Lu, W.-L. Liposomes in Drug Delivery: Status and Advances; Springer: Berlin/Heidelberg, Germany, 2021; Volume 8, pp. 3–24. [Google Scholar]
- Sercombe, L.; Veerati, T.; Moheimani, F.; Wu, S.Y.; Sood, A.K.; Hua, S. Advances and Challenges of Liposome Assisted Drug Delivery. Front. Pharmacol. 2015, 6, 286. [Google Scholar] [CrossRef] [PubMed]
- Kalaydina, R.-V.; Bajwa, K.; Qorri, B.; Decarlo, A.; Szewczuk, M.R. Recent Advances in “Smart” Delivery Systems for Extended Drug Release in Cancer Therapy. Int. J. Nanomed. Dovepress 2018, 13, 13–47. [Google Scholar] [CrossRef] [PubMed]
- Castañeda-Reyes, E.D.; Perea-Flores, M.D.J.; Davila-Ortiz, G.; Lee, Y.; de Mejia, E.G. Development, Characterization and Use of Liposomes as Amphipathic Transporters of Bioactive Compounds for Melanoma Treatment and Reduction of Skin Inflammation: A Review. Int. J. Nanomed. 2020, 15, 7627–7650. [Google Scholar] [CrossRef]
- Zalba, S.; ten Hagen, T.L.M.; Burgui, C.; Garrido, M.J. Stealth Nanoparticles in Oncology: Facing the PEG Dilemma. J. Control. Release 2022, 351, 22–36. [Google Scholar] [CrossRef]
- Briuglia, M.-L.; Rotella, C.; McFarlane, A.; Lamprou, D.A. Influence of Cholesterol on Liposome Stability and on in vitro Drug Release. Drug Deliv. Transl. Res. 2015, 5, 231–242. [Google Scholar] [CrossRef]
- Weder, H.G.; Zumbuehl, O. The Preparation of Variably Sized Homogeneous Liposomes for Laboratory, Clinical, and Industrial Use by Controlled Detergent Dialysis. Liposome Technol. 2019, 79–107. [Google Scholar]
- Guimarães, D.; Cavaco-Paulo, A.; Nogueira, E. Design of Liposomes as Drug Delivery System for Therapeutic Applications. Int. J. Pharm. 2021, 601, 20–47. [Google Scholar] [CrossRef] [PubMed]
- Samad, A.; Sultana, Y.; Aqil, M. Liposomal Drug Delivery Systems: An Update Review. Curr. Drug Deliv. 2007, 4, 294–305. [Google Scholar] [CrossRef] [PubMed]
- Alotaibi, B.S.; Buabeid, M.; Ibrahim, N.A.; Kharaba, Z.J.; Ijaz, M.; Noreen, S.; Murtaza, G. Potential of Nanocarrier-Based Drug Delivery Systems for Brain Targeting: A Current Review of Literature. Int. J. Nanomed. 2021, 16, 7517–7533. [Google Scholar] [CrossRef] [PubMed]
- Batool, S.; Sohail, S.; ud Din, F.; Alamri, A.H.; Alqahtani, A.S.; Alshahrani, M.A.; Alshehri, M.A.; Choi, H.G. A Detailed Insight of the Tumor Targeting Using Nanocarrier Drug Delivery System. Drug Deliv. 2023, 30, 2183815. [Google Scholar] [CrossRef]
- Karaz, S.; Senses, E. Liposomes under Shear: Structure, Dynamics, and Drug Delivery Applications. Adv. NanoBiomed Res. 2023, 5, 2200101. [Google Scholar] [CrossRef]
- Nel, J.; Elkhoury, K.; Velot, É.; Bianchi, A.; Acherar, S.; Francius, G.; Tamayol, A.; Grandemange, S.; Arab-Tehrany, E. Functionalized Liposomes for Targeted Breast Cancer Drug Delivery. Bioact. Mater. 2023, 24, 401–437. [Google Scholar] [CrossRef] [PubMed]
- Large, D.E.; Abdelmessih, R.G.; Fink, E.A.; Auguste, D.T. Liposome Composition in Drug Delivery Design, Synthesis, Characterization, and Clinical Application. Adv. Drug Deliv. Rev. 2021, 176, 38–51. [Google Scholar] [CrossRef]
- Tomatsu, I.; Marsden, H.R.; Rabe, M.; Versluis, F.; Zheng, T.; Zope, H.; Kros, A. Influence of Pegylation on Peptide-Mediated Liposome Fusion. J. Mater. Chem. 2011, 21, 18927–18933. [Google Scholar] [CrossRef]
- Filipczak, N.; Pan, J.; Yalamarty, S.S.K.; Torchilin, V.P. Recent Advancements in Liposome Technology. Adv. Drug Deliv. Rev. 2020, 156, 4–22. [Google Scholar] [CrossRef]
- Lombardo, D.; Kiselev, M.A.; Caccamo, M.T. Smart Nanoparticles for Drug Delivery Application: Development of Versatile Nanocarrier Platforms in Biotechnology and Nanomedicine. J. Nanomater. 2019, 2019, 70–95. [Google Scholar] [CrossRef]
- Zhang, H. Thin-Film Hydration Followed by Extrusion Method for Liposome Preparation. Methods Mol. Biol. 2017, 1522, 17–22. [Google Scholar] [PubMed]
- Has, C.; Sunthar, P. A Comprehensive Review on Recent Preparation Techniques of Liposomes. J. Liposome Res. 2020, 30, 336–365. [Google Scholar] [CrossRef]
- Winterhalter, M.; Lasic, D.D. Liposome Stability and Formation: Experimental Parameters and Theories on the Size Distribution. Chem. Phys. Lipids 1993, 64, 35–43. [Google Scholar] [CrossRef]
- Sundar, S.K.; Tirumkudulu, M.S. Synthesis of Sub-100-Nm Liposomes via Hydration in a Packed Bed of Colloidal Particles. Ind. Eng. Chem. Res. 2013, 53, 198–205. [Google Scholar] [CrossRef]
- Skalko-Basnet, N.; Pavelic, Z.; Becirevic-Lacan, M. Liposomes Containing Drug and Cyclodextrin Prepared by the One-Step Spray-Drying Method. Drug Dev. Ind. Pharm. 2000, 26, 1279–1284. [Google Scholar] [CrossRef]
- Li, C.; Deng, Y. A Novel Method for the Preparation of Liposomes: Freeze Drying of Monophase Solutions. J. Pharm. Sci. 2004, 93, 1403–1414. [Google Scholar] [CrossRef]
- De Leo, V.; Milano, F.; Agostiano, A.; Catucci, L. Recent Advancements in Polymer/Liposome Assembly for Drug Delivery: From Surface Modifications to Hybrid Vesicles. Polymers 2021, 13, 1027. [Google Scholar] [CrossRef]
- Lin, Y.C.; Huang, K.S.; Chiang, J.T.; Yang, C.H.; Lai, T.H. Manipulating Self-Assembled Phospholipid Microtubes Using Microfluidic Technology. Sens. Actuators B Chem. 2006, 117, 464–471. [Google Scholar] [CrossRef]
- Ota, S.; Yoshizawa, S.; Takeuchi, S. Microfluidic Formation of Monodisperse, Cell-Sized, and Unilamellar Vesicles. Angew. Chem. Int. Ed. 2009, 48, 6533–6537. [Google Scholar] [CrossRef]
- Jahn, A.; Vreeland, W.N.; Gaitan, M.; Locascio, L.E. Controlled Vesicle Self-Assembly in Microfluidic Channels with Hydrodynamic Focusing. J. Am. Chem. Soc. 2004, 126, 2674–2675. [Google Scholar] [CrossRef]
- Kastner, E.; Kaur, R.; Lowry, D.; Moghaddam, B.; Wilkinson, A.; Perrie, Y. High-Throughput Manufacturing of Size-Tuned Liposomes by a New Microfluidics Method Using Enhanced Statistical Tools for Characterization. Int. J. Pharm. 2014, 477, 361–368. [Google Scholar] [CrossRef]
- Moku, G.; Gopalsamuthiram, V.R.; Hoye, T.R.; Panyam, J. Surface Modification of Nanoparticles: Methods and Applications. Surf. Modif. Polym. Methods Appl. 2019, 46, 317–346. [Google Scholar]
- Du, Y.; He, W.; Xia, Q.; Zhou, W.; Yao, C.; Li, X. Thioether Phosphatidylcholine Liposomes: A Novel ROS-Responsive Platform for Drug Delivery. ACS Appl. Mater. Interfaces 2019, 11, 37411–37420. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Liang, N.; Yang, H.; Liu, H.; Li, S. Temozolomide Encapsulated and Folic Acid Decorated Chitosan Nanoparticles for Lung Tumor Targeting: Improving Therapeutic Efficacy Both In Vitro and In Vivo. Oncotarget 2017, 8, 111318. [Google Scholar] [CrossRef]
- Weecharangsan, W.; Yu, B.; Liu, S.; Pang, J.X.; Lee, L.J.; Marcucci, G.; Lee, R.J. Disulfide-Linked Liposomes: Effective Delivery Vehicle for Bcl-2 Antisense Oligodeoxyribonucleotide G3139. Anticancer. Res. 2010, 30, 31. [Google Scholar] [PubMed]
- Kanamala, M.; Palmer, B.D.; Jamieson, S.M.; Wilson, W.R.; Wu, Z. Dual PH-Sensitive Liposomes with Low PH-Triggered Sheddable PEG for Enhanced Tumor-Targeted Drug Delivery. Nanomedicine 2019, 14, 1971–1989. [Google Scholar] [CrossRef] [PubMed]
- Doane, T.; Burda, C. Nanoparticle Mediated Non-Covalent Drug Delivery. Adv. Drug Deliv. Rev. 2013, 65, 607. [Google Scholar] [CrossRef]
- Gentili, D.; Ori, G. Reversible Assembly of Nanoparticles: Theory, Strategies and Computational Simulations. Nanoscale 2022, 14, 14385–14432. [Google Scholar] [CrossRef]
- Awad, N.S.; Paul, V.; Alsawaftah, N.M.; Ter Haar, G.; Allen, T.M.; Pitt, W.G.; Husseini, G.A. Ultrasound-Responsive Nanocarriers in Cancer Treatment: A Review. ACS Pharmacol. Transl. Sci. 2021, 4, 589. [Google Scholar] [CrossRef]
- Yuba, E.; Osaki, T.; Ono, M.; Park, S.; Harada, A.; Yamashita, M.; Azuma, K.; Tsuka, T.; Ito, N.; Imagawa, T.; et al. Bleomycin-Loaded PH-Sensitive Polymer–Lipid-Incorporated Liposomes for Cancer Chemotherapy. Polymers 2018, 10, 74. [Google Scholar] [CrossRef]
- Lajunen, T.; Nurmi, R.; Wilbie, D.; Ruoslahti, T.; Johansson, N.G.; Korhonen, O.; Rog, T.; Bunker, A.; Ruponen, M.; Urtti, A. The Effect of Light Sensitizer Localization on the Stability of Indocyanine Green Liposomes. J. Control. Release 2018, 284, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Zeng, C.; Yu, F.; Yang, Y.; Cheng, X.; Liu, Y.; Zhang, H.; Zhao, S.; Yang, Z.; Li, M.; Li, Z.; et al. Preparation and Evaluation of Oxaliplatin Thermosensitive Liposomes with Rapid Release and High Stability. PLoS ONE 2016, 11, e0158517. [Google Scholar] [CrossRef] [PubMed]
- Hoosain, F.G.; Choonara, Y.E.; Tomar, L.K.; Kumar, P.; Tyagi, C.; Du Toit, L.C.; Pillay, V. Bypassing P-Glycoprotein Drug Efflux Mechanisms: Possible Applications in Pharmacoresistant Schizophrenia Therapy. BioMed Res. Int. 2015, 2015, 484933. [Google Scholar] [CrossRef]
- Löscher, W.; Potschka, H. Drug Resistance in Brain Diseases and the Role of Drug Efflux Transporters. Nat. Rev. Neurosci. 2005, 6, 591–602. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Si, L.; Pan, H.; Rabba, A.K.; Yan, F.; Qiu, J.; Li, G. Excipients Enhance Intestinal Absorption of Ganciclovir by P-Gp Inhibition: Assessed in Vitro by Everted Gut Sac and in Situ by Improved Intestinal Perfusion. Int. J. Pharm. 2011, 403, 37–45. [Google Scholar] [CrossRef]
- Simonis, B.; Vignone, D.; Gonzalez Paz, O.; Donati, E.; Falchetti, M.L.; Bombelli, C.; Cellucci, A.; Auciello, G.; Fini, I.; Galantini, L.; et al. Transport of Cationic Liposomes in a Human Blood Brain Barrier Model: Role of the Stereochemistry of the Gemini Amphiphile on Liposome Biological Features. J. Colloid Interface Sci. 2022, 627, 283–298. [Google Scholar] [CrossRef] [PubMed]
- Lin, Q.; Mao, K.L.; Tian, F.R.; Yang, J.J.; Chen, P.P.; Xu, J.; Fan, Z.L.; Zhao, Y.P.; Li, W.F.; Zheng, L.; et al. Brain Tumor-Targeted Delivery and Therapy by Focused Ultrasound Introduced Doxorubicin-Loaded Cationic Liposomes. Cancer Chemother. Pharmacol. 2016, 77, 269–280. [Google Scholar] [CrossRef]
- Aryal, M.; Vykhodtseva, N.; Zhang, Y.Z.; Park, J.; McDannold, N. Multiple Treatments with Liposomal Doxorubicin and Ultrasound-Induced Disruption of Blood-Tumor and Blood-Brain Barriers Improve Outcomes in a Rat Glioma Model. J. Control. Release 2013, 169, 103–111. [Google Scholar] [CrossRef]
- Li, J.; Zeng, H.; You, Y.; Wang, R.; Tan, T.; Wang, W.; Yin, L.; Zeng, Z.; Zeng, Y.; Xie, T. Active Targeting of Orthotopic Glioma Using Biomimetic Liposomes Co-Loaded Elemene and Cabazitaxel Modified by Transferritin. J. Nanobiotechnol. 2021, 19, 1–19. [Google Scholar] [CrossRef]
- Sonali; Singh, R.P.; Sharma, G.; Kumari, L.; Koch, B.; Singh, S.; Bharti, S.; Rajinikanth, P.S.; Pandey, B.L.; Muthu, M.S. RGD-TPGS Decorated Theranostic Liposomes for Brain Targeted Delivery. Colloids Surf. B Biointerfaces 2016, 147, 129–141. [Google Scholar] [CrossRef]
- Lu, Y.J.; Chuang, E.Y.; Cheng, Y.H.; Anilkumar, T.S.; Chen, H.A.; Chen, J.P. Thermosensitive Magnetic Liposomes for Alternating Magnetic Field-Inducible Drug Delivery in Dual Targeted Brain Tumor Chemotherapy. Chem. Eng. J. 2019, 373, 720–733. [Google Scholar] [CrossRef]
- Duong, V.-A.; Nguyen, T.-T.-L.; Maeng, H.-J. Recent Advances in Intranasal Liposomes for Drug, Gene, and Vaccine Delivery. Pharmaceutics 2023, 15, 207. [Google Scholar] [CrossRef]
- Gao, J.Q.; Lv, Q.; Li, L.M.; Tang, X.J.; Li, F.Z.; Hu, Y.L.; Han, M. Glioma Targeting and Blood-Brain Barrier Penetration by Dual-Targeting Doxorubincin Liposomes. Biomaterials 2013, 34, 5628–5639. [Google Scholar] [CrossRef]
- Ghaferi, M.; Raza, A.; Koohi, M.; Zahra, W.; Akbarzadeh, A.; Ebrahimi Shahmabadi, H.; Alavi, S.E. Impact of PEGylated Liposomal Doxorubicin and Carboplatin Combination on Glioblastoma. Pharmaceutics 2022, 14, 2183. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.M.; Filipczak, N.; Torchilin, V.P. Cell Penetrating Peptides: A Versatile Vector for Co-Delivery of Drug and Genes in Cancer. J. Control. Release 2021, 330, 1220–1228. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Duan, W.; Zhang, S.; Chen, D.; Feng, J.; Qi, N. Muscone/RI7217 Co-Modified Upward Messenger DTX Liposomes Enhanced Permeability of Blood-Brain Barrier and Targeting Glioma. Theranostics 2020, 10, 4308–4322. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Fan, L.; Pang, Z.; Ren, J.; Ren, Y.; Li, J.; Chen, J.; Wen, Z.; Jiang, X. TRAIL and Doxorubicin Combination Enhances Anti-Glioblastoma Effect Based on Passive Tumor Targeting of Liposomes. J. Control. Release 2011, 154, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Fortin, D.; Paquette, B.; Sanche, L. Convection-Enhancement Delivery of Liposomal Formulation of Oxaliplatin Shows Less Toxicity than Oxaliplatin yet Maintains a Similar Median Survival Time in F98 Glioma-Bearing Rat Model. Investig. New Drugs 2016, 34, 269–276. [Google Scholar] [CrossRef]
- Kong, D.; Hong, W.; Yu, M.; Li, Y.; Zheng, Y.; Ying, X. Multifunctional Targeting Liposomes of Epirubicin Plus Resveratrol Improved Therapeutic Effect on Brain Gliomas. Int. J. Nanomed. 2022, 17, 1087. [Google Scholar] [CrossRef]
- Zhao, G.; Huang, Q.; Wang, F.; Zhang, X.; Hu, J.; Tan, Y.; Huang, N.; Wang, Z.; Wang, Z.; Cheng, Y. Targeted ShRNA-Loaded Liposome Complex Combined with Focused Ultrasound for Blood Brain Barrier Disruption and Suppressing Glioma Growth. Cancer Lett. 2018, 418, 147–158. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Huang, X.; Wang, J.; Cai, F.; Zhao, P.; Yan, F. Targeted Delivery of Liposomal Temozolomide Enhanced Anti-Glioblastoma Efficacy through Ultrasound-Mediated Blood-Brain Barrier Opening. Pharmaceutics 2021, 13, 1270. [Google Scholar] [CrossRef] [PubMed]
- Lakkadwala, S.; Singh, J. Dual Functionalized 5-Fluorouracil Liposomes as Highly Efficient Nanomedicine for Glioblastoma Treatment as Assessed in an In Vitro Brain Tumor Model. J. Pharm. Sci. 2018, 107, 2902–2913. [Google Scholar] [CrossRef]
- Liu, Y.; Mei, L.; Xu, C.; Yu, Q.; Shi, K.; Zhang, L.; Wang, Y.; Zhang, Q.; Gao, H.; Zhang, Z.; et al. Dual Receptor Recognizing Cell Penetrating Peptide for Selective Targeting, Efficient Intratumoral Diffusion and Synthesized Anti-Glioma Therapy. Theranostics 2016, 6, 177–191. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Sun, D.; Wang, G.L.; Yang, H.G.; Xu, H.F.; Chen, J.H.; Xie, Y.; Wang, Z.Q. An Efficient PEGylated Liposomal Nanocarrier Containing Cell-Penetrating Peptide and PH-Sensitive Hydrazone Bond for Enhancing Tumor-Targeted Drug Delivery. Int. J. Nanomed. 2015, 10, 6199–6214. [Google Scholar] [CrossRef]
- Birngruber, T.; Raml, R.; Gladdines, W.; Gatschelhofer, C.; Gander, E.; Ghosh, A.; Kroath, T.; Gaillard, P.J.; Pieber, T.R.; Sinner, F. Enhanced Doxorubicin Delivery to the Brain Administered through Glutathione PEGylated Liposomal Doxorubicin (2B3-101) as Compared with Generic Caelyx,(®)/Doxil(®)--a Cerebral Open Flow Microperfusion Pilot Study. J. Pharm. Sci. 2014, 103, 1945–1948. [Google Scholar] [CrossRef]
- Zhang, Y.; Qu, H.; Xue, X. Blood–Brain Barrier Penetrating Liposomes with Synergistic Chemotherapy for Glioblastoma Treatment. Biomater. Sci. 2022, 10, 423–434. [Google Scholar] [CrossRef]
- Lippens, R.J.J. Liposomal Daunorubicin (DaunoXome) in Children with Recurrent or Progressive Brain Tumors. Pediatr. Hematol. Oncol. 1999, 16, 131–139. [Google Scholar] [CrossRef]
- Wagner, S.; Peters, O.; Fels, C.; Janssen, G.; Liebeskind, A.K.; Sauerbrey, A.; Suttorp, M.; Hau, P.; Wolff, J.E.A. Pegylated-Liposomal Doxorubicin and Oral Topotecan in Eight Children with Relapsed High-Grade Malignant Brain Tumors. J. Neuro-Oncol. 2008, 86, 175–181. [Google Scholar] [CrossRef]
- Marina, N.; Cochrane, D.; Harney, E.; Zomorodi, K.; Blaney, S.; Winick, N.; Bernstein, M.; Link, M. Dose Escalation and Pharmacokinetics of Pegylated Liposomal Doxorubicin (Doxil) in Children with Solid Tumors: A Pediatric Oncology Group Study. Clin. Cancer Res. 2002, 8, 413. [Google Scholar]
- Phillips, W.; Bao, A.; Floyd, J.; Awasthi, V.; Patel, T.; Hedrick, M.; LaFrance, N.; Rice, C.; Michalek, J.; Weinberg, J.; et al. Rhenium-186-NanoLiposome (186RNL) in the Treatment of Relapse/Recurrent Glioblastoma (RGBM): A Novel Approach for Cancer Therapy. J. Nucl. Med. 2022, 63, 2488. [Google Scholar]
- Ananda, S.; Nowak, A.K.; Cher, L.; Dowling, A.; Brown, C.; Simes, J.; Rosenthal, M.A. Phase 2 Trial of Temozolomide and Pegylated Liposomal Doxorubicin in the Treatment of Patients with Glioblastoma Multiforme Following Concurrent Radiotherapy and Chemotherapy. J. Clin. Neurosci. 2011, 18, 1444–1448. [Google Scholar] [CrossRef] [PubMed]

| Nanocarrier | Description | Characteristics | Advantages | Disadvantages | Refs. |
|---|---|---|---|---|---|
Carbon nanotubes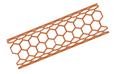 | Nanoparticles with a high surface-to-volume ratio, high tensile strength and stiffness, and high drug-loading capacity. Encapsulation of drugs is achieved through the utilization of the van der Waals force. While carbon nanotubes are generally not biocompatible, they can be rendered biocompatible by coating them with molecules such as proteins and organic polymers. | -Repeating hexagonal units of sp2 hybridized carbon atoms -Can be single- or double-walled -Passive and endocytosis-independent cellular uptake | -Protects entrapped drug and allows prolonged release -Bioactive moieties can be conjugated -Large internal surface area allows for high encapsulation efficiencies | -Low pK value -Water-insoluble -Biocompatible if functionalized with molecules such as proteins (antibodies, DNA, RNA) or organic polymers | [80] |
Gold nanoparticles | Metal and metal oxide nanoparticles can adopt various shapes, including nanoparticles, nanorods, nanocapsules, nanocuboids, and nanowires. In the context of targeted drug delivery, chemotherapeutic drugs can be chemically attached to the surface of metal nanoparticles or physically loaded into hollow gold or silver nanoparticles. Additionally, their surfaces can be easily functionalized to target specific ligands, further enhancing their potential in drug delivery applications. | -Colloidal gold in aqueous media -Size ranges from 1 to 100 nm | -Facile synthesis protocols -Allows for dual/multiple functionalizations with moieties -Controllable size distribution | -Non-biodegradable -Expensive | [81,82] |
QDs | QDs are nanosized semiconductors with unique optical and electrical characteristics. They exhibit bright and narrow-band fluorescence, and their emission can be tailored based on their size and chemistry. Unlike conventional organic label dyes, which have limitations in emitting in the near-infrared spectrum (>650 nm). They can be encapsulated within layers of polymeric or therapeutic material. For instance, a polymer layer can be used to encapsulate hydrophobic drugs, while the surface of the polymer shell can be functionalized with targeting ligands. | -Exhibit distinct optical and electrical features, such as intense and brilliant fluorescence -Can be excited by UV light; when excited electrons return to lower energy states, the energy difference is released as light | -Imaging characteristics -Theranostic applications -Controllable size distribution | -Toxicity/instability limitations -Sensitivity to air -Tendency to form aggregates | [83] |
Polymeric nanoparticles | Polymeric nanoparticles are solid colloidal systems in which a pharmacological drug is dissolved, entrapped, encapsulated, or adsorbed onto the polymer matrix. The resulting polymeric nanoparticles can have various structures, ranging from nanospheres to nanocapsules, depending on the method of nanoparticle synthesis. Nanocapsules consist of an oily core where the drug is typically dissolved, surrounded by a polymeric shell that controls the release of the drug from the core. On the other hand, nanospheres are composed of a continuous polymeric network that either dissolves drugs within it or allows drugs to be adsorbed onto its surface. These nanoparticles can be designed to respond to stimuli such as temperature, pH, and redox reactions. | -Particle size ranges from 1 to 1000 nm -May be loaded with active drugs that are either entrapped inside or surface-adsorbed onto the polymeric core -Large NPs used for near-infrared fluorescent in vivo imaging | -Tailored drug release profiles -Flexibility in delivery methods -Tendency to accumulate in tumors by EPR -Excellent stability | -Prone to agglomeration -Toxicity in the case of non-degradable polymer usage | [84] |
Dendrimers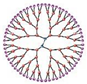 | Dendrimers are nanosized, radially symmetric, often polymeric, monodisperse molecules. These molecules branch out from a central molecule, resulting in a highly structured architecture. A first-generation dendrimer forms when the core molecule reacts with monomer molecules having one reactive and two inactive groups. The newly formed periphery of the molecule is then activated, allowing for repetitive branching growth in subsequent layers. | -Nanosized dendrimers have a core, an inner shell, and an outer shell that is symmetrical around the center -Structure is well-defined, homogenous, and monodisperse | -High physiochemical stability due to water solubility, specific molecular weight, and polyvalency -High biocompatibility -Low toxicity | -Batch-to-batch fluctuation in solubilized drug concentration | [85] |
| Entrance Mechanism | Liposome Type | Payload | Liposome Size | Drug Encapsulation Efficiency | Overview | Reference |
|---|---|---|---|---|---|---|
| Not mentioned | Liposomal Doxorubicin | Doxorubicin | Not mentioned | Not mentioned | In 9 L rat glioma tumors, three weekly FUS and DOX treatments were evaluated. FUS + DOX (N = 8) substantially enhanced median survival time (p 0.001) compared to just DOX (N = 6), FUS solo (N = 8), or no therapy (N = 7). FUS + DOX doubled median survival compared to untreated controls, but DOX alone barely doubled it. FUS-only animals did not improve. | [144] |
| Liposomal temozolomide formulation (TMZ-lipo) | Temozolomide | 148.13 ± 2.66 nm | 53% | TMZ-liposomes showed greater C6 tumor-cell-killing efficacy in vivo when paired with ultrasonic (US) irradiation, compared to free TMZ as a control Survival time increased from 40 days to 120 days in mice administered with TMZ-lipo + US | [146] | |
| Receptor-Mediated Endocytosis | PEGylated Liposomes | Doxorubicin and Carboplatin (CB) | 212 nm ± 10 nm | 83.9% | PEG-Lip nanoparticles containing Doxorubicin (DOX) and carboplatin were studied in vivo for brain cancer cells Animal survival was 23.1% higher with PEG-Lip-DOX/CB than with DOX + CB. | [136] |
| Liposomes | Cell-penetrating peptides (CPPs) and Transferrin (Tf) | 155 nm | 87.4 ± 3.85% | In vivo studies showed 7.7% higher accumulation of brain tumors than the control of liposomes in brain tumors when compared to control. | [145] | |
| RI7217 (mouse transferrin) and Muscone-Conjugated Liposomes | Docetaxel (DTX) | 159.1 ± 4.4 nm | 65.37 ± 0.78% | Muscone and RI7217 co-modified DTX liposomes boosted absorption in hCMEC/D3 and U87-MG cells in vitro Increased tumor spheroid penetration improved brain targeting in vivo. The median survival period of the group given R17217-Muscone-DTX liposome was 24 days, or 1.6 times that of the group given saline. | [139] | |
| RGD-TPGS-theranostic liposomes | Docetaxel and quantum dots (QDs) | 175.6 ± 3.2 nm | 68.41 ± 3.56% | RGD-TPGS-theranostic liposomes proved to be 6.47- and 6.98-fold more efficacious than DocelTM in vivo. RGD-TPGS liposomes successfully decreased ROS production and showed no evidence of brain injury or edema. | [132] | |
| Thermosensitive magnetic liposomes (TML) | Camptosar (CPT-11)) and DOX coated with magnetic Fe3O4 nanoparticles and conjugated with Cetuximab (CET) | 193.7 ± 2.3 nm | 87.9 ± 1.4% | Enhanced cellular uptake in vivo. High biocompatibility and significant tumor shrinkage in vivo when compared to control. Survival time increased from 22 days to 30 days in mice administered with TML-CPT-11 liposomes and in conjunction with magnetic guidance | [133] | |
| PEGylated liposomes (Lipoxal) | Oxaliplatin | 118.5 nm | 54% | In vivo studies in F98 murine models showed a higher accumulation of Lipoxal in tumor cells than the free drug administration. Survival time increased from 30 days to 38.5 days in mice administered with Oxaliplatin + LipoxalA, and notable increases in median survival times were observed. | [140] | |
| Receptor-Mediated Transcytosis | Glutathione doxorubicin-PEGylated liposome (2B3-101) | Doxil®/Caelyx® | 95 nm, P.D < 0.1% | >90% | In vitro studies showed complete tumor regression in 3 out of 9 rats. Ten-fold DOX accumulation was noted in animals treated with 2B3-101 when compared to free Doxil®/Caelyx®. | [146] |
| Glutathione doxorubicin-PEGylated liposome (2B3-101) | Doxil®/Caelyx® | 97 nm | Not mentioned | In vitro studies showed that four-fold DOX accumulation was noted in animals treated with 2B3-101 when compared to free Doxil®/Caelyx®. | [147] | |
| Glucose-functionalized liposomes (gLTP) | Temozolomide (TMZ) and pro-apoptotic peptide (PAP) | 133 nm | 79.32% | In vivo studies showed higher susceptibility of GBM cells than controls. Median survival time increased from 10 days to 30 days. | [148] |
| Compound | Lipid Composition | Payload | Trial Phase | Indications | Ref. |
|---|---|---|---|---|---|
| DaunoXome | DSPC, Cholesterol | Daunorubicin | I | Pediatric Gliomas | [149] |
| Doxil®/Caelyx | HSPC, Cholesterol, DSPE-PEG2000 | DOX | II | GBM, Pediatric Gliomas | [153] |
| Doxil® | HSPC, Cholesterol, DSPE-PEG2000 | DOX | I | Pediatric Glioma | [150] |
| Doxil® | HSPC, Cholesterol, DSPE-PEG2000 | DOX | I | Refractory brain tumors | [151] |
| 186RNL | Not mentioned | Rhenium-186 Nanoliposome | I | Recurrent GBM | [152] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raju, R.; Abuwatfa, W.H.; Pitt, W.G.; Husseini, G.A. Liposomes for the Treatment of Brain Cancer—A Review. Pharmaceuticals 2023, 16, 1056. https://doi.org/10.3390/ph16081056
Raju R, Abuwatfa WH, Pitt WG, Husseini GA. Liposomes for the Treatment of Brain Cancer—A Review. Pharmaceuticals. 2023; 16(8):1056. https://doi.org/10.3390/ph16081056
Chicago/Turabian StyleRaju, Richu, Waad H. Abuwatfa, William G. Pitt, and Ghaleb A. Husseini. 2023. "Liposomes for the Treatment of Brain Cancer—A Review" Pharmaceuticals 16, no. 8: 1056. https://doi.org/10.3390/ph16081056
APA StyleRaju, R., Abuwatfa, W. H., Pitt, W. G., & Husseini, G. A. (2023). Liposomes for the Treatment of Brain Cancer—A Review. Pharmaceuticals, 16(8), 1056. https://doi.org/10.3390/ph16081056







