Cutaneous Polymeric-Micelles-Based Hydrogel Containing Origanum vulgare L. Essential Oil: In Vitro Release and Permeation, Angiogenesis, and Safety Profile In Ovo
Abstract
1. Introduction
2. Results
2.1. SEM Analysis
2.2. In Vitro Drug Release and Skin Permeation Studies
2.2.1. In Vitro Release of OEO
2.2.2. In Vitro Pig Ear Skin Permeation of OEO
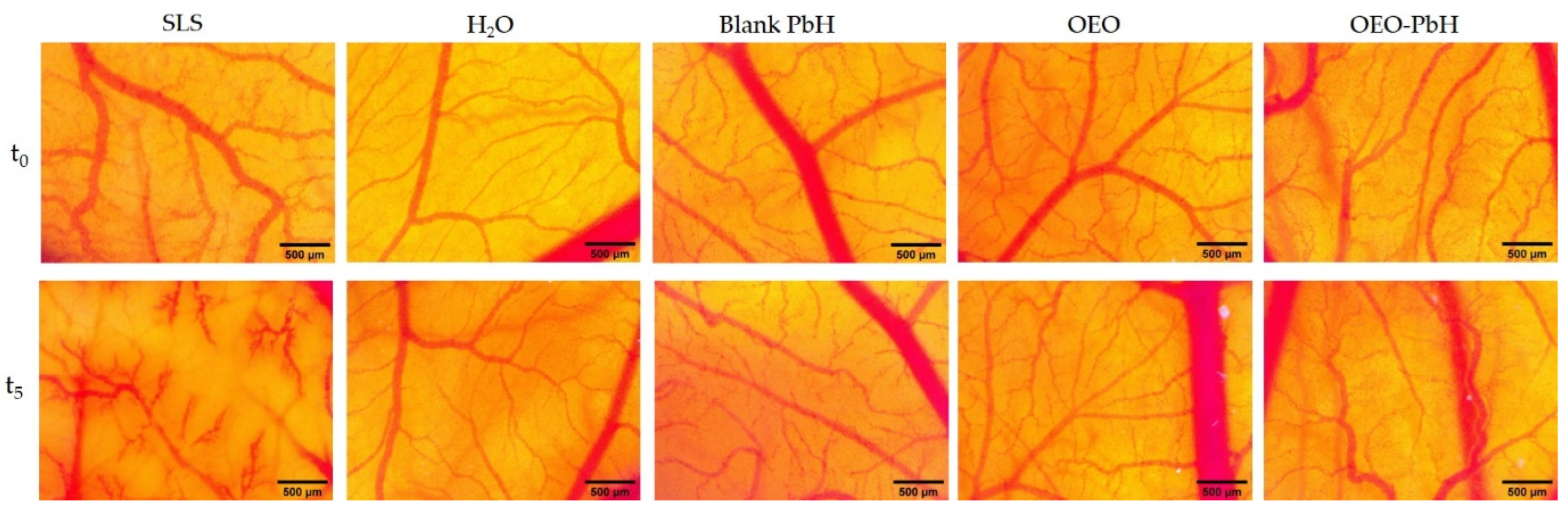
2.3. OEO-PbH Biocompatibility In Vivo on the CAM
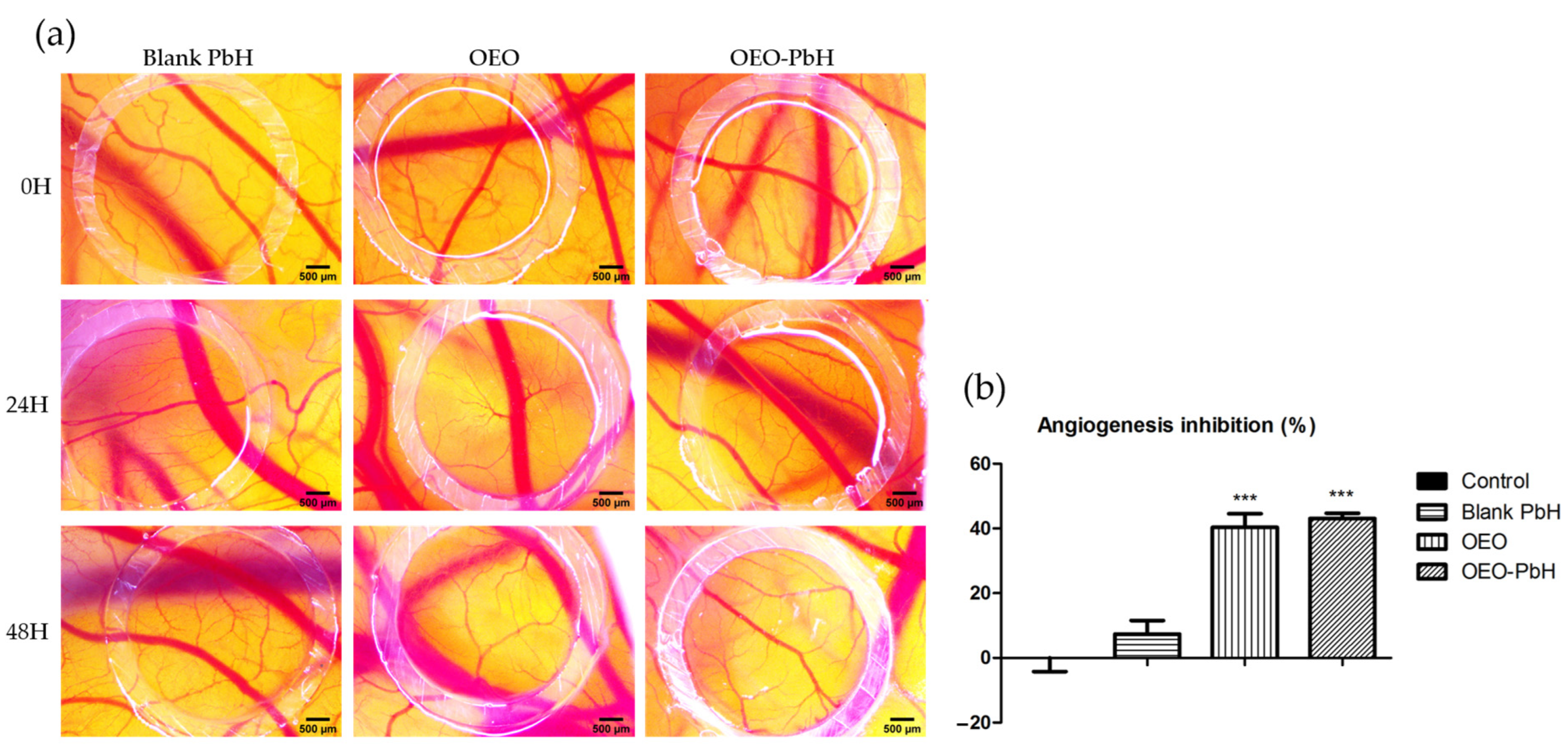
2.4. OEO-PbH Modulates Angiogenesis In Ovo
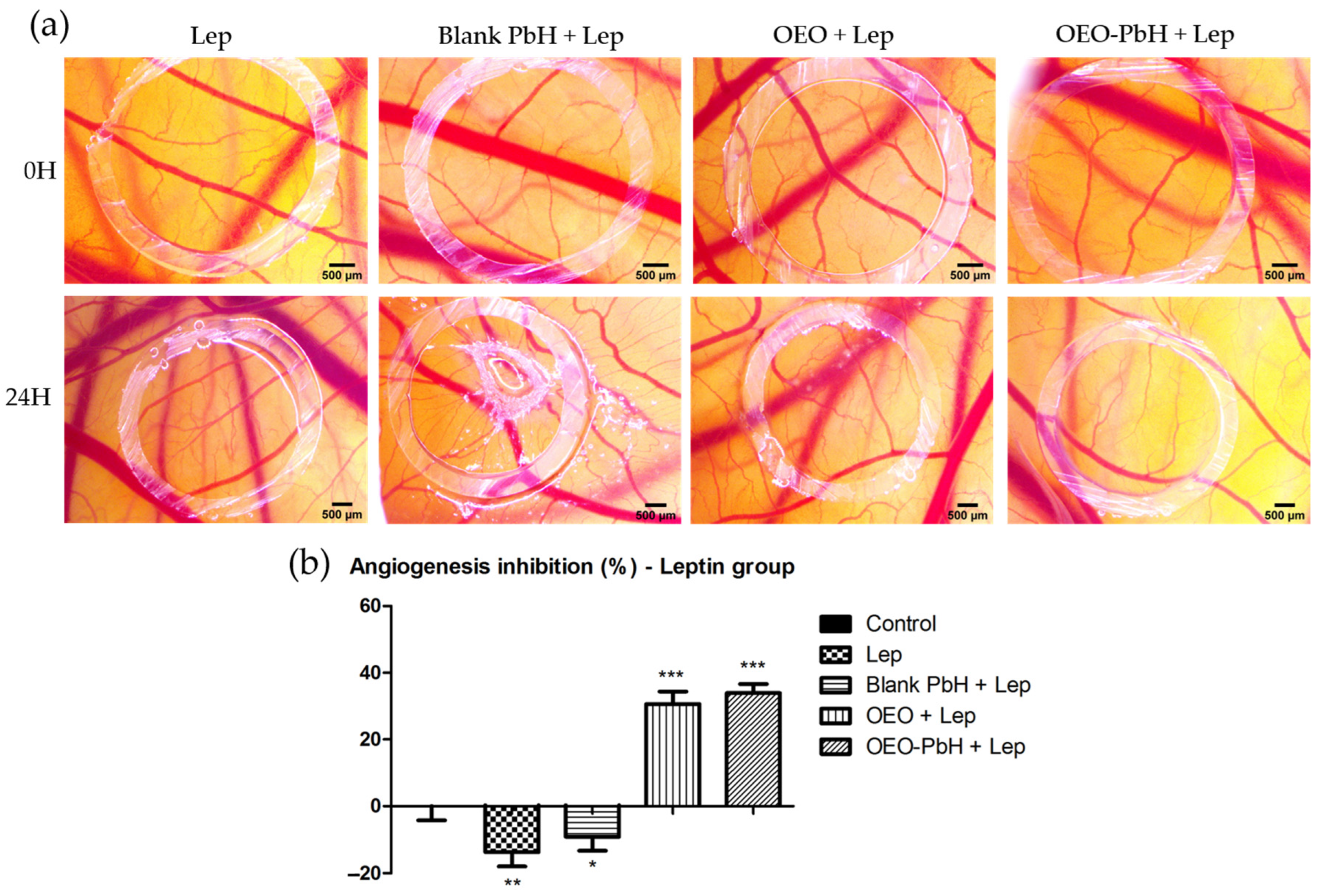
2.5. OEO-PbH Modulates Leptin-Induced Angiogenesis In Ovo on the CAM
2.6. Histological Observations
3. Discussion
4. Materials and Methods
4.1. Materials
4.1.1. Plant Materials
4.1.2. Chemicals
4.2. Preparation of Polymeric-Micelles-Based Hydrogels
4.3. SEM
4.4. OEO In Vitro Release and Skin Permeation Studies
4.4.1. In Vitro OEO Release Studies
4.4.2. In Vitro OEO Skin Permeation Studies
4.4.3. Data Analysis of In Vitro OEO Release and Permeation Studies
4.5. In Ovo CAM Assay
4.5.1. CAM Assay
4.5.2. Irritability Assessment by the HET-CAM Protocol
4.5.3. Angiogenesis Effects Using the CAM Assay
4.6. Histoprocessing and Histology Evaluation
4.6.1. Tissue Collection and Processing
4.6.2. H&E Staining
4.6.3. Microscopic Analysis
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lukas, B.; Schmiderer, C.; Novak, J. Essential Oil Diversity of European Origanum vulgare L. (Lamiaceae). Phytochemistry 2015, 119, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Azizi, A.; Wagner, C.; Honermeier, B.; Friedt, W. Intraspecific Diversity and Relationship between Subspecies of Origanum vulgare Revealed by Comparative AFLP and SAMPL Marker Analysis. Plant Syst. Evol. 2009, 281, 151–160. [Google Scholar] [CrossRef]
- Skoufogianni, E.; Solomou, A.D.; Danalatos, N.G. Ecology, Cultivation and Utilization of the Aromatic Greek Oregano (Origanum vulgare L.): A Review. Not. Bot. Horti Agrobot. Cluj-Napoca 2019, 47, 545–552. [Google Scholar] [CrossRef]
- Hossain, M.B.; Barry-Ryan, C.; Martin-Diana, A.B.; Brunton, N.P. Optimisation of Accelerated Solvent Extraction of Antioxidant Compounds from Rosemary (Rosmarinus officinalis L.), Marjoram (Origanum majorana L.) and Oregano (Origanum vulgare L.) Using Response Surface Methodology. Food Chem. 2011, 126, 339–346. [Google Scholar] [CrossRef]
- Soltani, S.; Shakeri, A.; Iranshahi, M.; Boozari, M. A Review of the Phytochemistry and Antimicrobial Properties of Origanum vulgare L. and Subspecies. Iran. J. Pharm. Res. 2021, 20, 268–285. [Google Scholar] [CrossRef]
- Alekseeva, M.; Zagorcheva, T.; Atanassov, I.; Rusanov, K. Origanum vulgare L.—A Review on Genetic Diversity, Cultivation, Biological Activities and Perspectives for Molecular Breeding. Bulg. J. Agric. Sci. 2020, 26, 1183–1197. [Google Scholar]
- Morshedloo, M.R.; Salami, S.A.; Nazeri, V.; Maggi, F.; Craker, L. Essential Oil Profile of Oregano (Origanum vulgare L.) Populations Grown under Similar Soil and Climate Conditions. Ind. Crops Prod. 2018, 119, 183–190. [Google Scholar] [CrossRef]
- Boskovic, M.; Zdravkovic, N.; Ivanovic, J.; Janjic, J.; Djordjevic, J.; Starcevic, M.; Baltic, M.Z. Antimicrobial Activity of Thyme (Tymus vulgaris) and Oregano (Origanum vulgare) Essential Oils against Some Food-Borne Microorganisms. Procedia Food Sci. 2015, 5, 18–21. [Google Scholar] [CrossRef]
- Sarikurkcu, C.; Zengin, G.; Oskay, M.; Uysal, S.; Ceylan, R.; Aktumsek, A. Composition, Antioxidant, Antimicrobial and Enzyme Inhibition Activities of Two Origanum vulgare Subspecies (Subsp. vulgare and Subsp. hirtum) Essential Oils. Ind. Crops Prod. 2015, 70, 178–184. [Google Scholar] [CrossRef]
- Begnini, K.R.; Nedel, F.; Lund, R.G.; Carvalho, P.H.D.A.; Rodrigues, M.R.A.; Beira, F.T.A.; Del-Pino, F.A.B. Composition and Antiproliferative Effect of Essential Oil of Origanum vulgare against Tumor Cell Lines. J. Med. Food 2014, 17, 1129–1133. [Google Scholar] [CrossRef] [PubMed]
- Ocaña-Fuentes, A.; Arranz-Gutiérrez, E.; Señorans, F.J.; Reglero, G. Supercritical Fluid Extraction of Oregano (Origanum vulgare) Essentials Oils: Anti-Inflammatory Properties Based on Cytokine Response on THP-1 Macrophages. Food Chem. Toxicol. 2010, 48, 1568–1575. [Google Scholar] [CrossRef]
- Béjaoui, A.; Chaabane, H.; Jemli, M.; Boulila, A.; Boussaid, M. Essential Oil Composition and Antibacterial Activity of Origanum vulgare Subsp. Glandulosum Desf. at Different Phenological Stages. J. Med. Food 2013, 16, 1115–1120. [Google Scholar] [CrossRef]
- Koldaş, S.; Demirtas, I.; Ozen, T.; Demirci, M.A.; Behçet, L. Phytochemical Screening, Anticancer and Antioxidant Activities of Origanum vulgare L. ssp. Viride (Boiss.) Hayek, a Plant of Traditional Usage. J. Sci. Food Agric. 2015, 95, 786–798. [Google Scholar] [CrossRef] [PubMed]
- Lombrea, A.; Antal, D.; Ardelean, F.; Avram, S.; Pavel, I.Z.; Vlaia, L.; Mut, A.M.; Diaconeasa, Z.; Dehelean, C.A.; Soica, C.; et al. A Recent Insight Regarding the Phytochemistry and Bioactivity of Origanum vulgare L. Essential Oil. Int. J. Mol. Sci. 2020, 21, 9653. [Google Scholar] [CrossRef] [PubMed]
- Dwinna, R.; Yunilawati, R.; Jati, B.N.; Setiawati, I.; Riyanto, A.; Batubara, I.; Astuti, R.I. Antiaging and Skin Irritation Potential of Four Main Indonesian Essential Oils. Cosmetics 2021, 8, 94. [Google Scholar]
- El Khoury, R.; Michael-Jubeli, R.; Bakar, J.; Dakroub, H.; Rizk, T.; Baillet-Guffroy, A.; Lteif, R.; Tfayli, A. Origanum Essential Oils Reduce the Level of Melanin in B16-F1 Melanocytes. Eur. J. Dermatol. 2019, 29, 596–602. [Google Scholar] [CrossRef]
- Da Silva, S.M.M.; Costa, C.R.R.; Gelfuso, G.M.; Guerra, E.N.S.; De Medeiros Nóbrega, Y.K.; Gomes, S.M.; Pic-Taylor, A.; Fonseca-Bazzo, Y.M.; Silveira, D.; De Oliveira Magalhães, P. Wound Healing Effect of Essential Oil Extracted from Eugenia Dysenterica DC (Myrtaceae) Leaves. Molecules 2019, 24, 2. [Google Scholar] [CrossRef]
- Avola, R.; Granata, G.; Geraci, C.; Napoli, E.; Graziano, A.C.E.; Cardile, V. Oregano (Origanum vulgare L.) Essential Oil Provides Anti-Inflammatory Activity and Facilitates Wound Healing in a Human Keratinocytes Cell Model. Food Chem. Toxicol. 2020, 144, 111586. [Google Scholar] [CrossRef]
- Khan, A.u.R.; Huang, K.; Jinzhong, Z.; Zhu, T.; Morsi, Y.; Aldalbahi, A.; El-Newehy, M.; Yan, X.; Mo, X. PLCL/Silk Fibroin Based Antibacterial Nano Wound Dressing Encapsulating Oregano Essential Oil: Fabrication, Characterization and Biological Evaluation. Colloids Surfaces B Biointerfaces 2020, 196, 111352. [Google Scholar] [CrossRef]
- Taleb, M.H.; Abdeltawab, N.F.; Shamma, R.N.; Abdelgayed, S.S.; Mohamed, S.S.; Farag, M.A.; Ramadan, M.A. Origanum vulgare L. Essential Oil as a Potential Anti-Acne Topical Nanoemulsion—In Vitro and In Vivo Study. Molecules 2018, 23, 2164. [Google Scholar] [CrossRef]
- Mazzarello, V.; Gavini, E.; Rassu, G.; Donadu, M.G.; Usai, D.; Piu, G.; Pomponi, V.; Sucato, F.; Zanetti, S.; Montesu, M.A. Clinical Assessment of New Topical Cream Containing Two Essential Oils Combined with Tretinoin in the Treatment of Acne. Clin. Cosmet. Investig. Dermatol. 2020, 13, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Bora, L.; Avram, S.; Pavel, I.Z.; Muntean, D.; Liga, S.; Buda, V.; Gurgus, D.; Danciu, C. An Up-To-Date Review Regarding Cutaneous Benefits of Origanum vulgare L. Essential Oil. Antibiotics 2022, 11, 549. [Google Scholar] [CrossRef]
- Kashyap, N.; Kumari, A.; Raina, N.; Zakir, F.; Gupta, M. Prospects of Essential Oil Loaded Nanosystems for Skincare. Phytomed. Plus 2022, 2, 100198. [Google Scholar] [CrossRef]
- Chen, J.; Jiang, Q.D.; Wu, Y.M.; Liu, P.; Yao, J.H.; Lu, Q.; Zhang, H.; Duan, J.A. Potential of Essential Oils as Penetration Enhancers for Transdermal Administration of Ibuprofen to Treat Dysmenorrhoea. Molecules 2015, 20, 18219–18236. [Google Scholar] [CrossRef]
- Raina, N.; Rani, R.; Pahwa, R.; Gupta, M. Biopolymers and Treatment Strategies for Wound Healing: An Insight View. Int. J. Polym. Mater. Polym. Biomater. 2022, 71, 359–375. [Google Scholar] [CrossRef]
- Russo, J.; Fiegel, J.; Brogden, N.K. Rheological and Drug Delivery Characteristics of Poloxamer-Based Diclofenac Sodium Formulations for Chronic Wound Site Analgesia. Pharmaceutics 2020, 12, 1214. [Google Scholar] [CrossRef]
- Zarrintaj, P.; Ramsey, J.D.; Samadi, A.; Atoufi, Z.; Yazdi, M.K.; Ganjali, M.R.; Amirabad, L.M.; Zangene, E.; Farokhi, M.; Formela, K.; et al. Poloxamer: A Versatile Tri-Block Copolymer for Biomedical Applications. Acta Biomater. 2020, 110, 37–67. [Google Scholar] [CrossRef]
- European Directorate for the Quality of Medicines & HealthCare of the Council of Europe. Guidance on Essential Oils in Cosmetic Products. Council of Europe, Consumer Health Protection Committee (CD-P-SC). 2016. Available online: https://www.edqm.eu/en/guidance-on-essential-oils-in-cosmetic-products (accessed on 7 May 2023).
- European Commission. European Pharmacopoeia; Maisonneuve: Sainte-Ruffine, France, 1980; Volume 2. [Google Scholar]
- Omeragic, E.; Dedic, M.; Elezovic, A.; Becic, E.; Imamovic, B.; Kladar, N.; Niksic, H. Application of Direct Peptide Reactivity Assay for Assessing the Skin Sensitization Potential of Essential Oils. Sci. Rep. 2022, 12, 7470. [Google Scholar] [CrossRef]
- Fathalla, Z.M.A.; Vangala, A.; Longman, M.; Khaled, K.A.; Hussein, A.K.; El-Garhy, O.H.; Alany, R.G. Poloxamer-Based Thermoresponsive Ketorolac Tromethamine in Situ Gel Preparations: Design, Characterisation, Toxicity and Transcorneal Permeation Studies. Eur. J. Pharm. Biopharm. 2017, 114, 119–134. [Google Scholar] [CrossRef]
- Russo, E.; Villa, C. Poloxamer Hydrogels for Biomedical Applications. Pharmaceutics. 2019, 11, 671. [Google Scholar] [CrossRef]
- Bora, L.; Burkard, T.; Juan, M.H.S.; Radeke, H.H.; Muț, A.M.; Vlaia, L.L.; Magyari-Pavel, I.Z.; Diaconeasa, Z.; Socaci, S.; Borcan, F.; et al. Phytochemical Characterization and Biological Evaluation of Origanum vulgare L. Essential Oil Formulated as Polymeric Micelles Drug Delivery Systems. Pharmaceutics 2022, 14, 2413. [Google Scholar] [CrossRef]
- Bostancioĝlu, R.B.; Kürkçüoĝlu, M.; Başer, K.H.C.; Koparal, A.T. Assessment of Anti-Angiogenic and Anti-Tumoral Potentials of Origanum onites L. Essential Oil. Food Chem. Toxicol. 2012, 50, 2002–2008. [Google Scholar] [CrossRef]
- Khan, I.; Bhardwaj, M.; Shukla, S.; Lee, H.; Oh, M.W.; Bajpai, V.K.; Huh, Y.S.; Kang, S.C. Carvacrol Encapsulated Nanocarrier/ Nanoemulsion Abrogates Angiogenesis by Downregulating COX-2, VEGF and CD31 in Vitro and in Vivo in a Lung Adenocarcinoma Model. Colloids Surf. B Biointerfaces 2019, 181, 612–622. [Google Scholar] [CrossRef] [PubMed]
- Ghiulai, R.; Avram, S.; Stoian, D.; Pavel, I.Z.; Coricovac, D.; Oprean, C.; Vlase, L.; Farcas, C.; Mioc, M.; Minda, D.; et al. Lemon Balm Extracts Prevent Breast Cancer Progression In Vitro and In Ovo on Chorioallantoic Membrane Assay. Evid.-Based Complement. Altern. Med. 2020, 2020, 6489159. [Google Scholar] [CrossRef]
- Rivero, M.N.; Lenze, M.; Izaguirre, M.; Pérez Damonte, S.H.; Aguilar, A.; Wikinski, S.; Gutiérrez, M.L. Comparison between HET-CAM Protocols and a Product Use Clinical Study for Eye Irritation Evaluation of Personal Care Products Including Cosmetics According to Their Surfactant Composition. Food Chem. Toxicol. 2021, 153, 112229. [Google Scholar] [CrossRef] [PubMed]
- Minda, D.; Ghiulai, R.; Banciu, C.D.; Pavel, I.Z.; Danciu, C.; Racoviceanu, R.; Soica, C.; Budu, O.D.; Muntean, D.; Diaconeasa, Z.; et al. Phytochemical Profile, Antioxidant and Wound Healing Potential of Three Artemisia Species: In Vitro and In Ovo Evaluation. Appl. Sci. 2022, 12, 1359. [Google Scholar] [CrossRef]
- Vimalraj, S.; Renugaa, S.; Dhanasekaran, A. Chick Embryo Chorioallantoic Membrane: A Biomaterial Testing Platform for Tissue Engineering Applications. Process Biochem. 2023, 124, 81–91. [Google Scholar] [CrossRef]
- Schneider-Stock, R.; Ribatti, D. The CAM Assay as an Alternative In Vivo Model for Drug Testing. In Organotypic Models in Drug Development. Handbook of Experimental Pharmacology; Schafer-Korting, M., Stuchi Maria-Engler, S., Landsiedel, R., Eds.; Springer: Cham, Switzerland, 2020; Volume 265, pp. 303–323. [Google Scholar] [CrossRef]
- Thakker, K. Summary Report on Workshop on In Vitro Release Test (IVRT) and In Vitro Permeation Test (IVPT) Methods Best Practices and Scientific Considerations for ANDA Submissions. Dissolution Technol. 2021, 28, 50–52. [Google Scholar] [CrossRef]
- Olejnik, A.; Goscianska, J.; Nowak, I. Active Compounds Release from Semisolid Dosage Forms. J. Pharm. Sci. 2012, 101, 4032–4045. [Google Scholar] [CrossRef]
- Brambilla, E.; Locarno, S.; Gallo, S.; Orsini, F.; Pini, C.; Farronato, M.; Thomaz, D.V.; Lenardi, C.; Piazzoni, M.; Tartaglia, G. Poloxamer-Based Hydrogel as Drug Delivery System: How Polymeric Excipients Influence the Chemical-Physical Properties. Polymers 2022, 14, 3624. [Google Scholar] [CrossRef]
- Bodratti, A.M.; Alexandridis, P. Formulation of Poloxamers for Drug Delivery. J. Funct. Biomater. 2018, 9, 11. [Google Scholar] [CrossRef]
- Ruan, X.; Hu, J.; Lu, L.; Wang, Y.; Tang, C.; Liu, F.; Gao, X.; Zhang, L.; Wu, H.; Huang, X.; et al. Poloxamer 407/188 Binary Thermosensitive Gel as a Moxidectin Delivery System: In Vitro Release and In Vivo Evaluation. Molecules 2022, 27, 3063. [Google Scholar] [CrossRef] [PubMed]
- Anderson, B.C.; Pandit, N.K.; Mallapragada, S.K. Understanding Drug Release from Poly(Ethylene Oxide)-b-Poly(Propylene Oxide)-b-Poly(Ethylene Oxide) Gels. J. Control. Release 2001, 70, 157–167. [Google Scholar] [CrossRef] [PubMed]
- European Medicine Agency. Draft Guideline on Quality and Equivalence of Topical Products. Available online: https://www.ema.europa.eu/documents/scientific-guideline/draft-guideline-quality-equivalence-topical-products_en.pdf (accessed on 1 November 2022).
- Chiriac, A.P.; Rusu, A.G.; Nita, L.E.; Chiriac, V.M.; Neamtu, I.; Sandu, A. Polymeric Carriers Designed for Encapsulation of Essential Oils with Biological Activity. Pharmaceutics 2021, 13, 631. [Google Scholar] [CrossRef]
- Kargozar, S.; Baino, F.; Hamzehlou, S.; Hamblin, M.R.; Mozafari, M. Nanotechnology for Angiogenesis: Opportunities and Challenges. Chem. Soc. Rev. 2020, 49, 5008–5057. [Google Scholar] [CrossRef]
- Pontes-Quero, G.M.; Esteban-Rubio, S.; Pérez Cano, J.; Aguilar, M.R.; Vázquez-Lasa, B. Oregano Essential Oil Micro- and Nanoencapsulation With Bioactive Properties for Biotechnological and Biomedical Applications. Front. Bioeng. Biotechnol. 2021, 9, 703684. [Google Scholar] [CrossRef]
- Laothaweerungsawat, N.; Neimkhum, W.; Anuchapreeda, S.; Sirithunyalug, J.; Chaiyana, W. Transdermal Delivery Enhancement of Carvacrol from Origanum vulgare L. Essential Oil by Microemulsion. Int. J. Pharm. 2020, 579, 119052. [Google Scholar] [CrossRef] [PubMed]
- Subbaraj, G.K.; Masoodi, T.; Yasam, S.K.; Chandrashekar, K.; Kulanthaivel, L.; Shaik, N.A.; Hashem, S.; Alshabeeb Akil, A.S.; Bhat, A.A. Anti-Angiogenic Effect of Nano-Formulated Water Soluble Kaempferol and Combretastatin in an In Vivo Chick Chorioallantoic Membrane Model and HUVEC Cells. Biomed. Pharmacother. 2023, 163, 114820. [Google Scholar] [CrossRef]
- Han, X.; Parker, T.L. Anti-Inflammatory, Tissue Remodeling, Immunomodulatory, and Anticancer Activities of Oregano (Origanum vulgare) Essential Oil in a Human Skin Disease Model. Biochim. Open 2017, 4, 73–77. [Google Scholar] [CrossRef]
- Kennedy, D.C.; Coen, B.; Wheatley, A.M.; McCullagh, K.J.A. Microvascular Experimentation in the Chick Chorioallantoic Membrane as a Model for Screening Angiogenic Agents Including from Gene-Modified Cells. Int. J. Mol. Sci. 2022, 23, 452. [Google Scholar] [CrossRef]
- Schmolka, I.R. Artificial Skin I. Preparation and Properties of Pluronic F-127 Gels for Treatment of Burns. J. Biomed. Mater. Res. 1972, 6, 571–582. [Google Scholar] [CrossRef]
- Sahiner, N.; Godbey, W.T.; McPherson, G.L.; John, V.T. Microgel, Nanogel and Hydrogel-Hydrogel Semi-IPN Composites for Biomedical Applications: Synthesis and Characterization. Colloid Polym. Sci. 2006, 284, 1121–1129. [Google Scholar] [CrossRef]
- Akhtar, S.; Asiri, S.M.; Khan, F.A.; Gunday, S.T.; Iqbal, A.; Alrushaid, N.; Labib, O.A.; Deen, G.R.; Henari, F.Z. Formulation of Gold Nanoparticles with Hibiscus and Curcumin Extracts Induced Anti-Cancer Activity. Arab. J. Chem. 2022, 15, 103594. [Google Scholar] [CrossRef]
- United States Pharmacopeial Convention. USP General Chapter <1724> Semisolid Drug Products—Performance Tests. In The United States Pharmacopeia: USP 30; The National formulary: NF 25; United States Pharmacopeial Convention: Rockville, MD, USA, 2006. [Google Scholar]
- Shah, V.P.; Miron, D.S.; Rădulescu, F.Ș.; Cardot, J.M.; Maibach, H.I. In Vitro Release Test (IVRT): Principles and Applications. Int. J. Pharm. 2022, 626, 122159. [Google Scholar] [CrossRef] [PubMed]
- Olejnik, A.; Kapuscinska, A.; Schroeder, G.; Nowak, I. Physico-Chemical Characterization of Formulations Containing Endomorphin-2 Derivatives. Amino Acids 2017, 49, 1719–1731. [Google Scholar] [CrossRef]
- Bruschi, M. Mathematical Models of Drug Release; Woodhead Publishing: London, UK, 2015. [Google Scholar]
- Vitková, Z.; Oremusová, J.; Herdová, P.; Vitko, A.; Ivanková, O. Model Based Approach to Study of Release Kinetics of the Drug Chlorhexidine from Hydrogels. Tenside Surfactants Deterg. 2015, 52, 67–72. [Google Scholar] [CrossRef]
- Ribatti, D. The Chick Embryo Chorioallantoic Membrane in the Study of Tumor Angiogenesis. Rom. J. Morphol. Embryol. 2008, 49, 131–135. [Google Scholar]
- Öztürk, A.A.; Kıyan, H.T. Treatment of Oxidative Stress-Induced Pain and Inflammation with Dexketoprofen Trometamol Loaded Different Molecular Weight Chitosan Nanoparticles: Formulation, Characterization and Anti-Inflammatory Activity by Using in Vivo HET-CAM Assay. Microvasc. Res. 2020, 128, 103961. [Google Scholar] [CrossRef]
- Moreno-Jiménez, I.; Kanczler, J.M.; Hulsart-Billstrom, G.; Inglis, S.; Oreffo, R.O.C. The Chorioallantoic Membrane Assay for Biomaterial Testing in Tissue Engineering: A Short-Term In Vivo Preclinical Model. Tissue Eng.-Part C Methods 2017, 23, 938–952. [Google Scholar] [CrossRef]
- Coricovac, D.; Farcas, C.; Nica, C.; Pinzaru, I.; Simu, S.; Stoian, D.; Soica, C.; Proks, M.; Avram, S.; Navolan, D.; et al. Ethinylestradiol and Levonorgestrel as Active Agents in Normal Skin, and Pathological Conditions Induced by UVB Exposure: In Vitro and in Ovo Assessments. Int. J. Mol. Sci. 2018, 19, 3600. [Google Scholar] [CrossRef]
- Interagency Coordinating Committee on the Validation of Alternative Methods (ICCVAM). ICCVAM Test Method Evaluation Report: Current Validation Status of In Vitro Test Methods Proposed for Identifying Eye Injury Hazard Potential of Chemicals and Products. NIH Publication No. 10-7553. 2010, Research Triangle Park, NC: National Institute of Environmental Health Sciences. Available online: https://ntp.niehs.nih.gov/sites/default/files/iccvam/docs/ocutox_docs/invitro-2010/tmer-vol1.pdf (accessed on 26 May 2023).
- Luepke, N.P. Hen’s Egg Chorioallantoic Membrane Test for Irritation Potential. Food Chem. Toxicol. 1985, 23, 287–291. [Google Scholar] [CrossRef] [PubMed]
- Zhong, L.; Guo, X.N.; Zhang, X.H.; Sun, Q.M.; Tong, L.J.; Wu, Z.X.; Luo, X.M.; Jiang, H.L.; Nan, F.J.; Zhang, X.W.; et al. TKI-31 Inhibits Angiogenesis by Combined Suppression Signaling Pathway of VEGFR2 and PDGFRβ. Cancer Biol. Ther. 2006, 5, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Ghitu, A.; Schwiebs, A.; Radeke, H.H.; Avram, S.; Zupko, I.; Bor, A.; Pavel, I.Z.; Dehelean, C.A.; Oprean, C.; Bojin, F.; et al. A Comprehensive Assessment of Apigenin as an Antiproliferative, Proapoptotic, Antiangiogenic and Immunomodulatory Phytocompound. Nutrients 2019, 11, 858. [Google Scholar] [CrossRef] [PubMed]
- Özcetin, A.; Aigner, A.; Bakowsky, U. A Chorioallantoic Membrane Model for the Determination of Anti-Angiogenic Effects of Imatinib. Eur. J. Pharm. Biopharm. 2013, 85 Pt A, 711–715. [Google Scholar] [CrossRef]



| Membranes Used in In Vitro OEO Release/Permeation Study | JSS (μg/cm2/h) | KP × 10−6 (cm/h) | tL (h) |
|---|---|---|---|
| Synthetic polysulfone membrane | 419.99 ± 8.12 | 83.99 ± 1.32 | 0.44 ± 1.47 |
| Pig ear skin | 155.06 ± 6.57 (2–10 h) | 31.01 ± 4.56 | 1.65 ± 2.38 |
| 49.63 ± 3.19 (10–24 h) | 9.92 ± 3.05 | – |
| Membranes Used in In Vitro OEO Release/ Permeation Study | Zero Order | First Order | Higuchi | Korsmeyer–Peppas | |||||
|---|---|---|---|---|---|---|---|---|---|
| K0 (μg/h) | R2 | K1 (h−1) | R2 | KH (h−0.5) | R2 | KP (h−n) | n | R2 | |
| Synthetic polysulfone membrane | 5.0374 | 0.9962 | 0.0604 | 0.9932 | 10.1080 | 0.8222 | 0.1831 | 1.7605 | 0.9322 |
| Pig ear skin | 1.1015 | 0.9278 | 0.0126 | 0.9425 | 4.6450 | 0.8652 | 0.3346 | 1.4833 | 0.9531 |
| Samples | Irritation Score | Type of Effect |
|---|---|---|
| SLS 1% | 15.89 ± 0.42 | Strong irritant |
| H2O distillate | 0 | Non-irritant |
| Blank-PbH | 0 | Non-irritant |
| OEO | 0 | Non-irritant |
| OEO-PbH | 0 | Non-irritant |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Avram, Ș.; Bora, L.; Vlaia, L.L.; Muț, A.M.; Olteanu, G.-E.; Olariu, I.; Magyari-Pavel, I.Z.; Minda, D.; Diaconeasa, Z.; Sfirloaga, P.; et al. Cutaneous Polymeric-Micelles-Based Hydrogel Containing Origanum vulgare L. Essential Oil: In Vitro Release and Permeation, Angiogenesis, and Safety Profile In Ovo. Pharmaceuticals 2023, 16, 940. https://doi.org/10.3390/ph16070940
Avram Ș, Bora L, Vlaia LL, Muț AM, Olteanu G-E, Olariu I, Magyari-Pavel IZ, Minda D, Diaconeasa Z, Sfirloaga P, et al. Cutaneous Polymeric-Micelles-Based Hydrogel Containing Origanum vulgare L. Essential Oil: In Vitro Release and Permeation, Angiogenesis, and Safety Profile In Ovo. Pharmaceuticals. 2023; 16(7):940. https://doi.org/10.3390/ph16070940
Chicago/Turabian StyleAvram, Ștefana, Larisa Bora, Lavinia Lia Vlaia, Ana Maria Muț, Gheorghe-Emilian Olteanu, Ioana Olariu, Ioana Zinuca Magyari-Pavel, Daliana Minda, Zorița Diaconeasa, Paula Sfirloaga, and et al. 2023. "Cutaneous Polymeric-Micelles-Based Hydrogel Containing Origanum vulgare L. Essential Oil: In Vitro Release and Permeation, Angiogenesis, and Safety Profile In Ovo" Pharmaceuticals 16, no. 7: 940. https://doi.org/10.3390/ph16070940
APA StyleAvram, Ș., Bora, L., Vlaia, L. L., Muț, A. M., Olteanu, G.-E., Olariu, I., Magyari-Pavel, I. Z., Minda, D., Diaconeasa, Z., Sfirloaga, P., Adnan, M., Dehelean, C. A., & Danciu, C. (2023). Cutaneous Polymeric-Micelles-Based Hydrogel Containing Origanum vulgare L. Essential Oil: In Vitro Release and Permeation, Angiogenesis, and Safety Profile In Ovo. Pharmaceuticals, 16(7), 940. https://doi.org/10.3390/ph16070940












