Increased Circulating ADMA in Young Male Rats Caused Cognitive Deficits and Increased Intestinal and Hippocampal NLRP3 Inflammasome Expression and Microbiota Composition Alterations: Effects of Resveratrol
Abstract
1. Introduction
2. Results
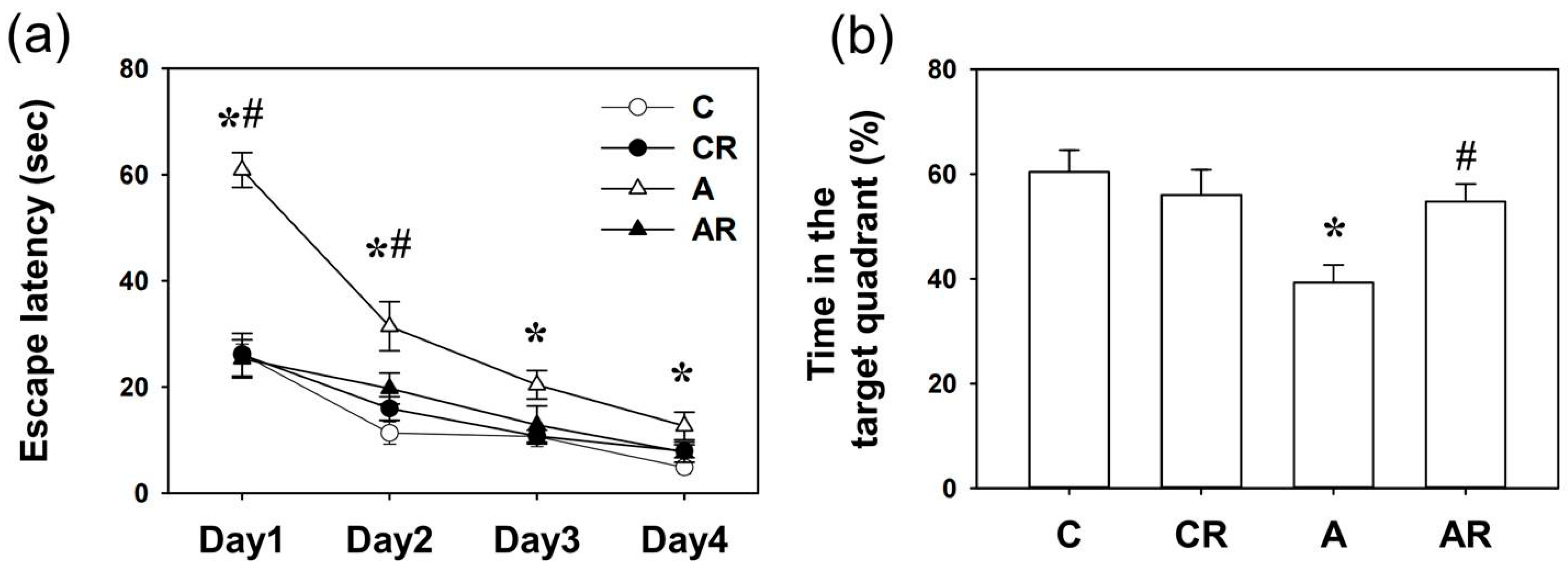
2.1. Morris Water Maze
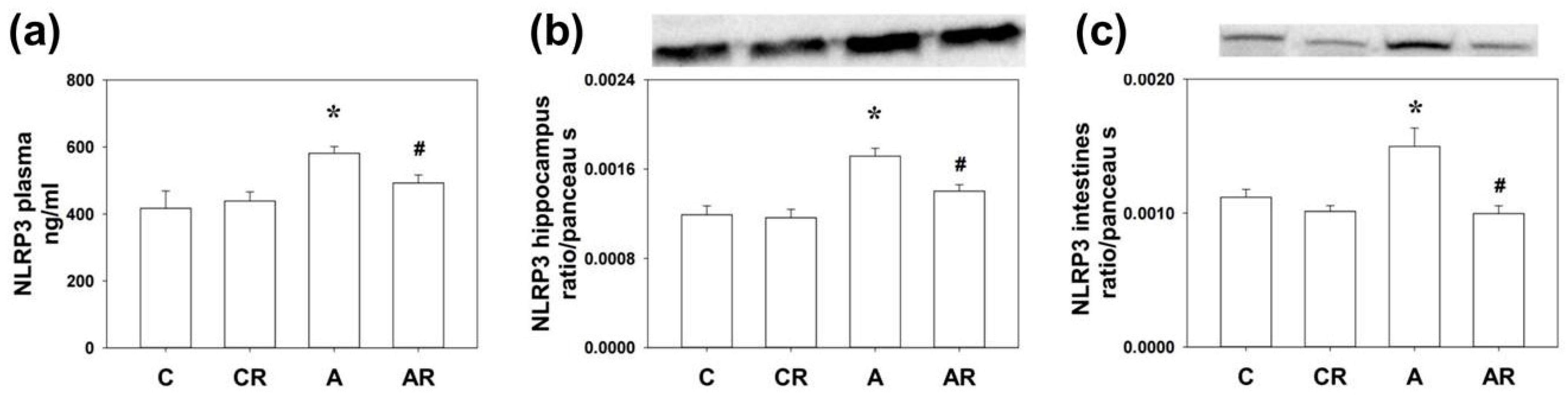
2.2. NLRP3 Expression in the Plasma, Dorsal Hippocampus, and Ileum
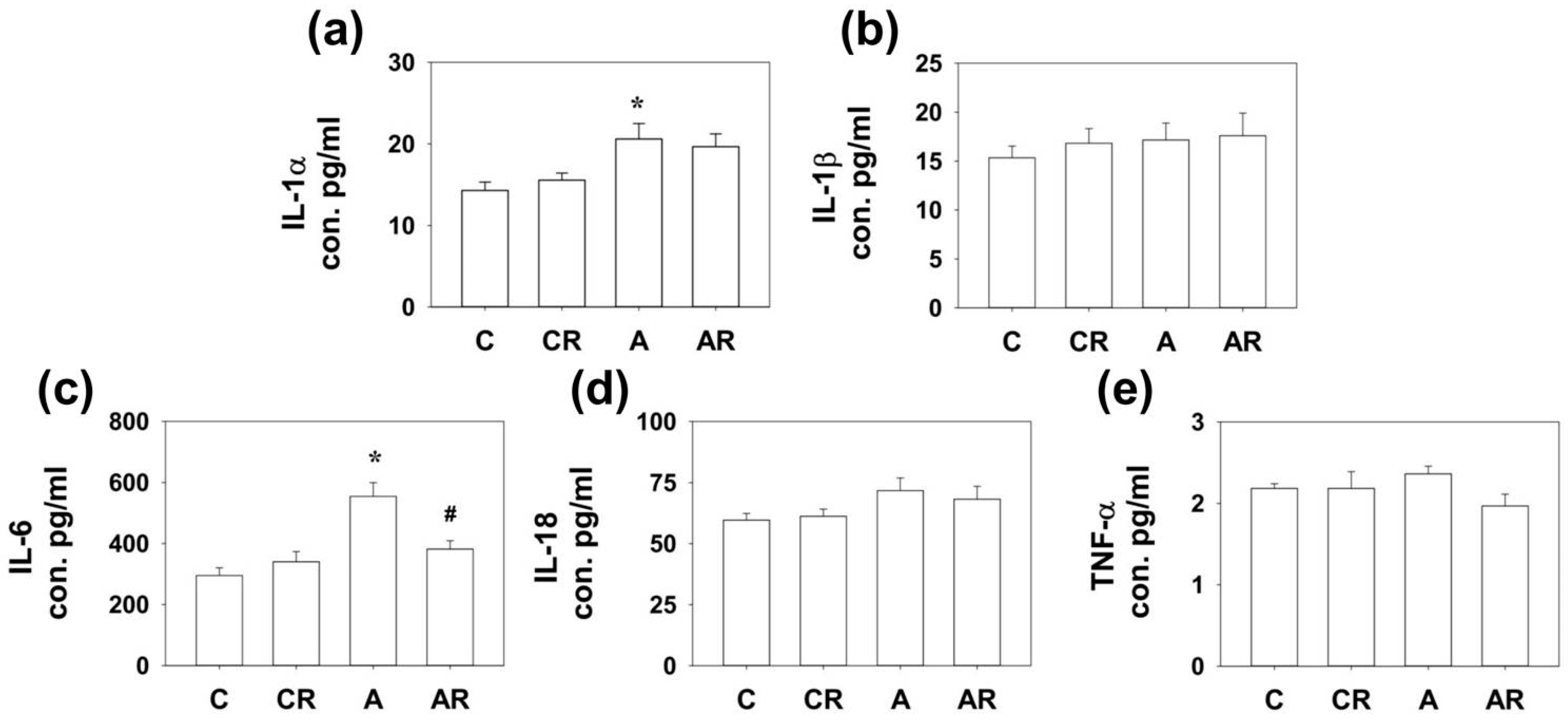
2.3. Expression of Cytokines in Rat Dorsal Hippocampus
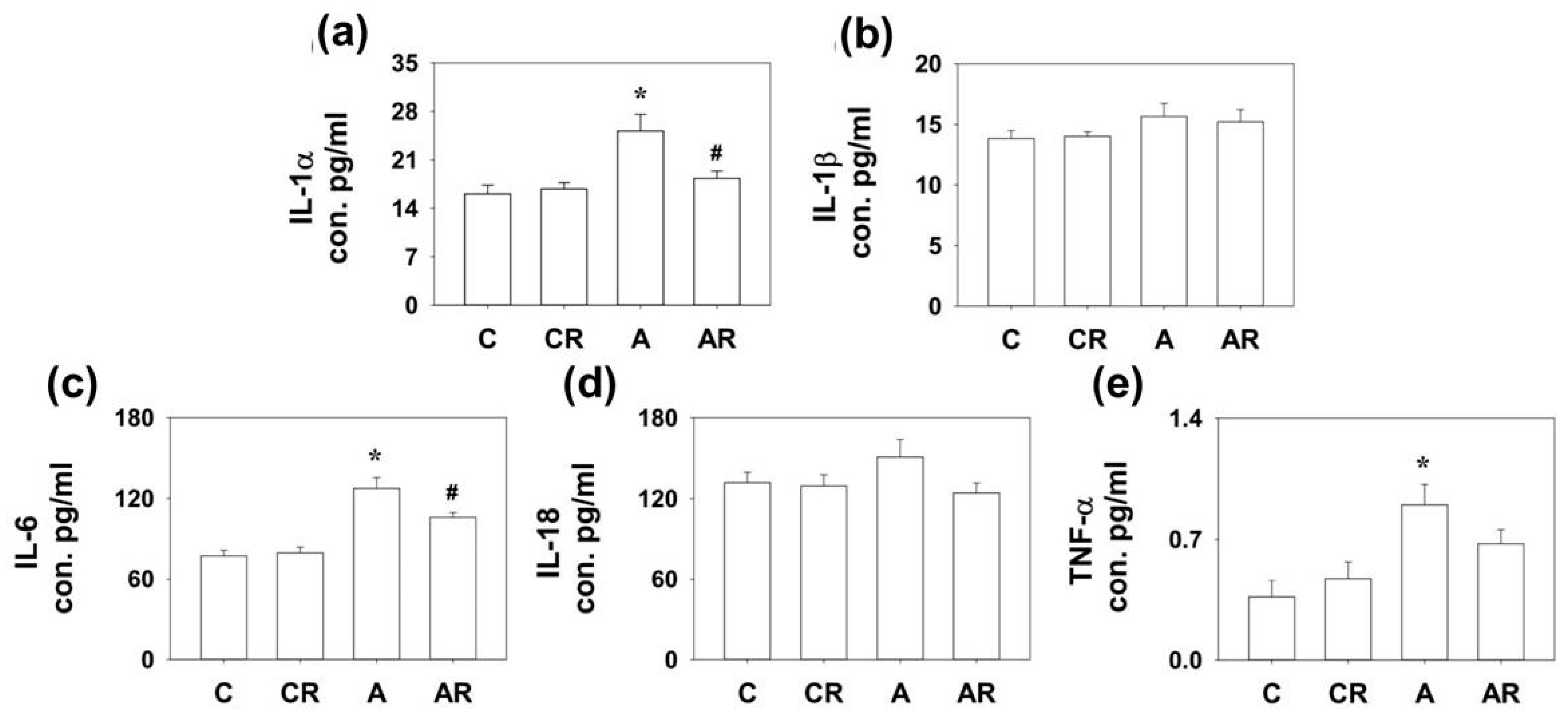
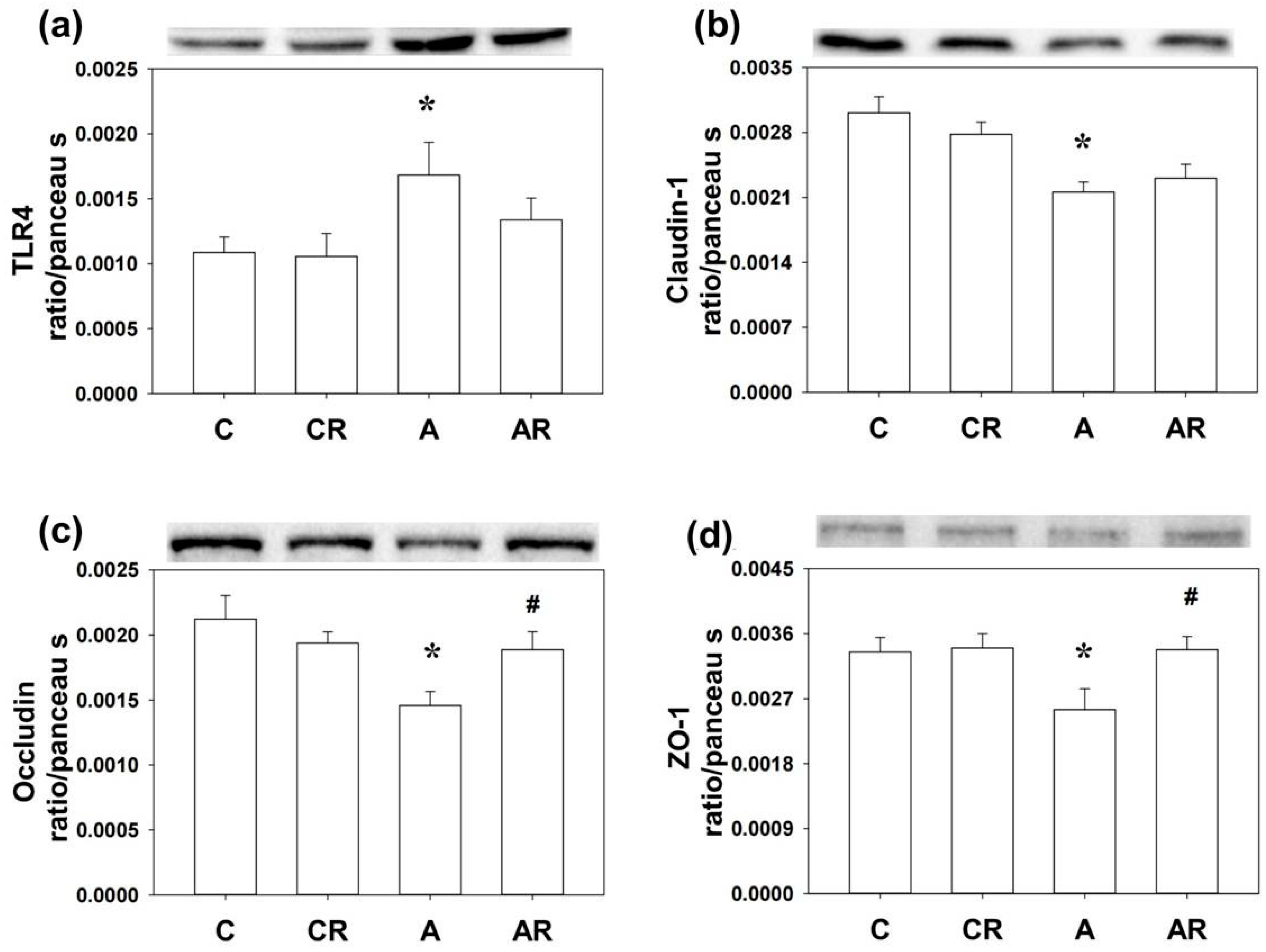
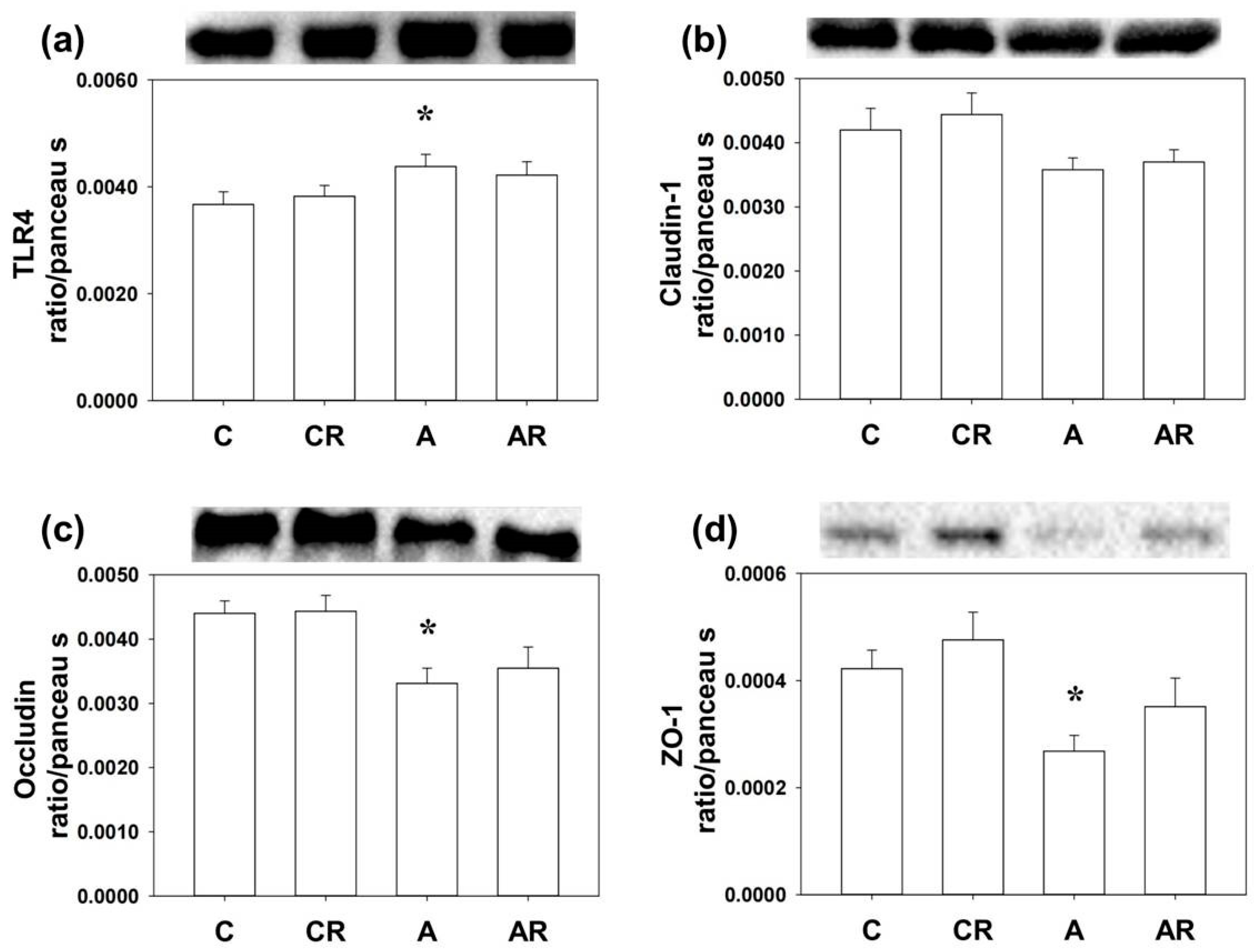
2.4. Inflammation and Tight Junction Protein in Rat Dorsal Hippocampus and Ileum
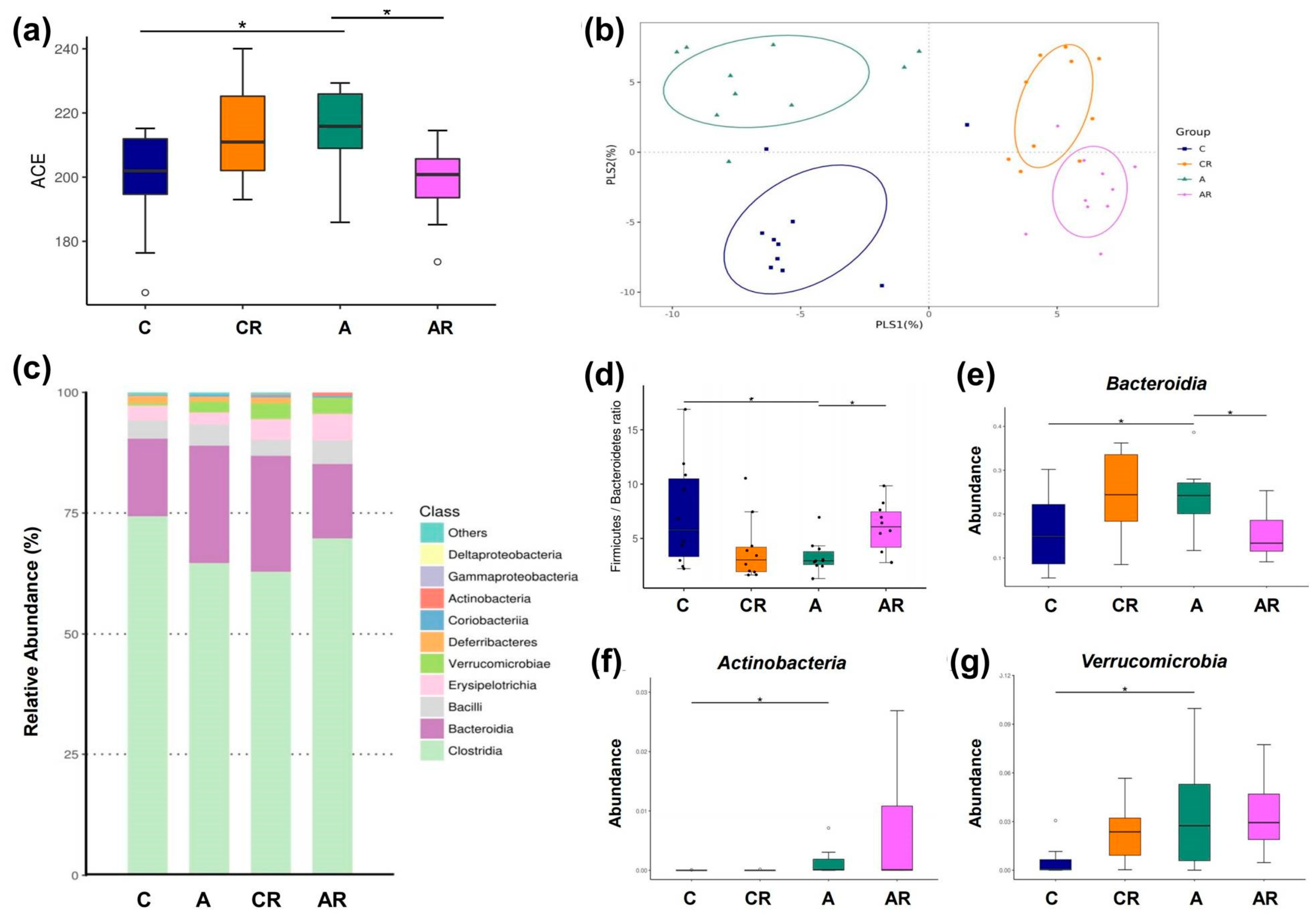
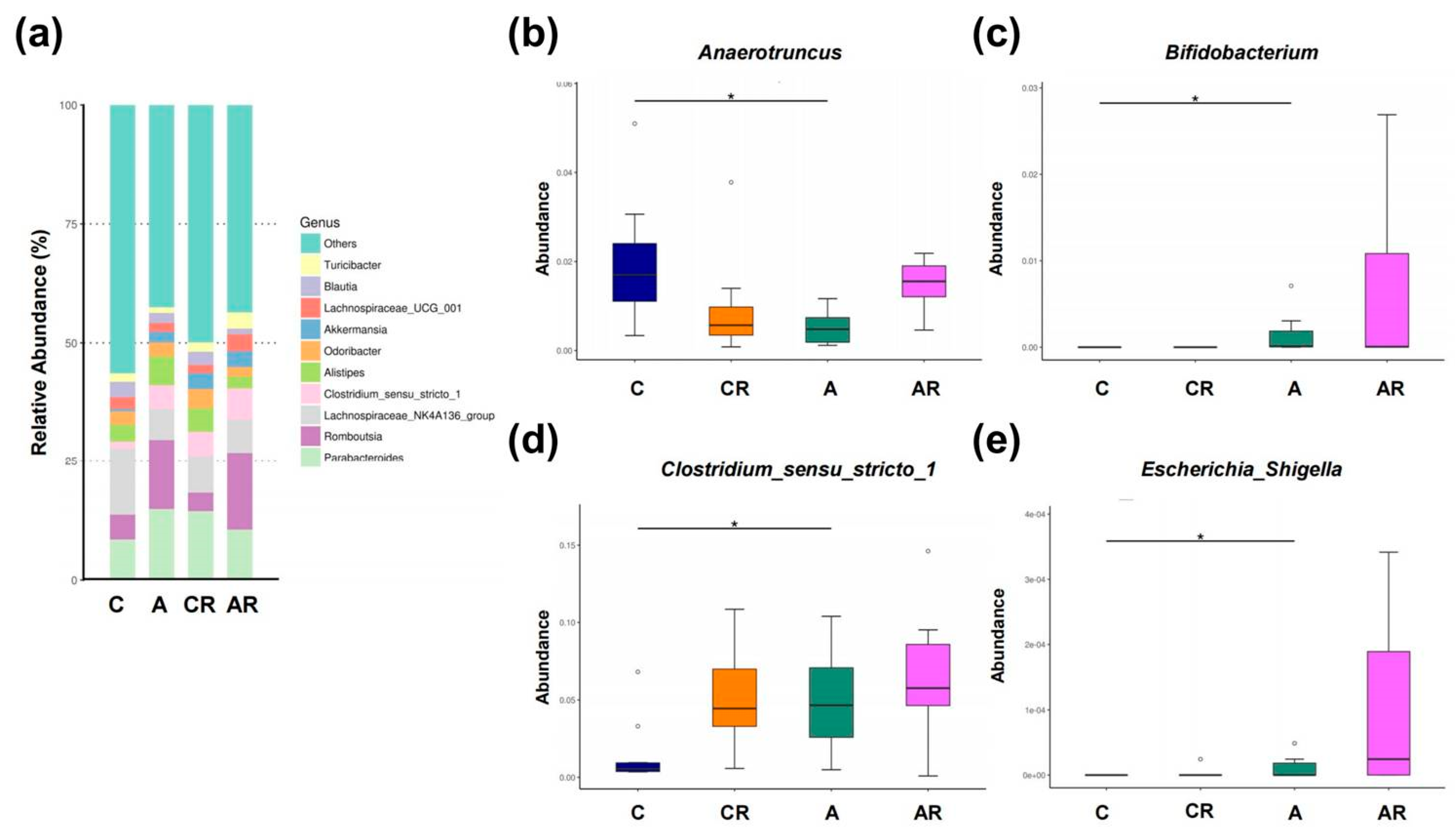
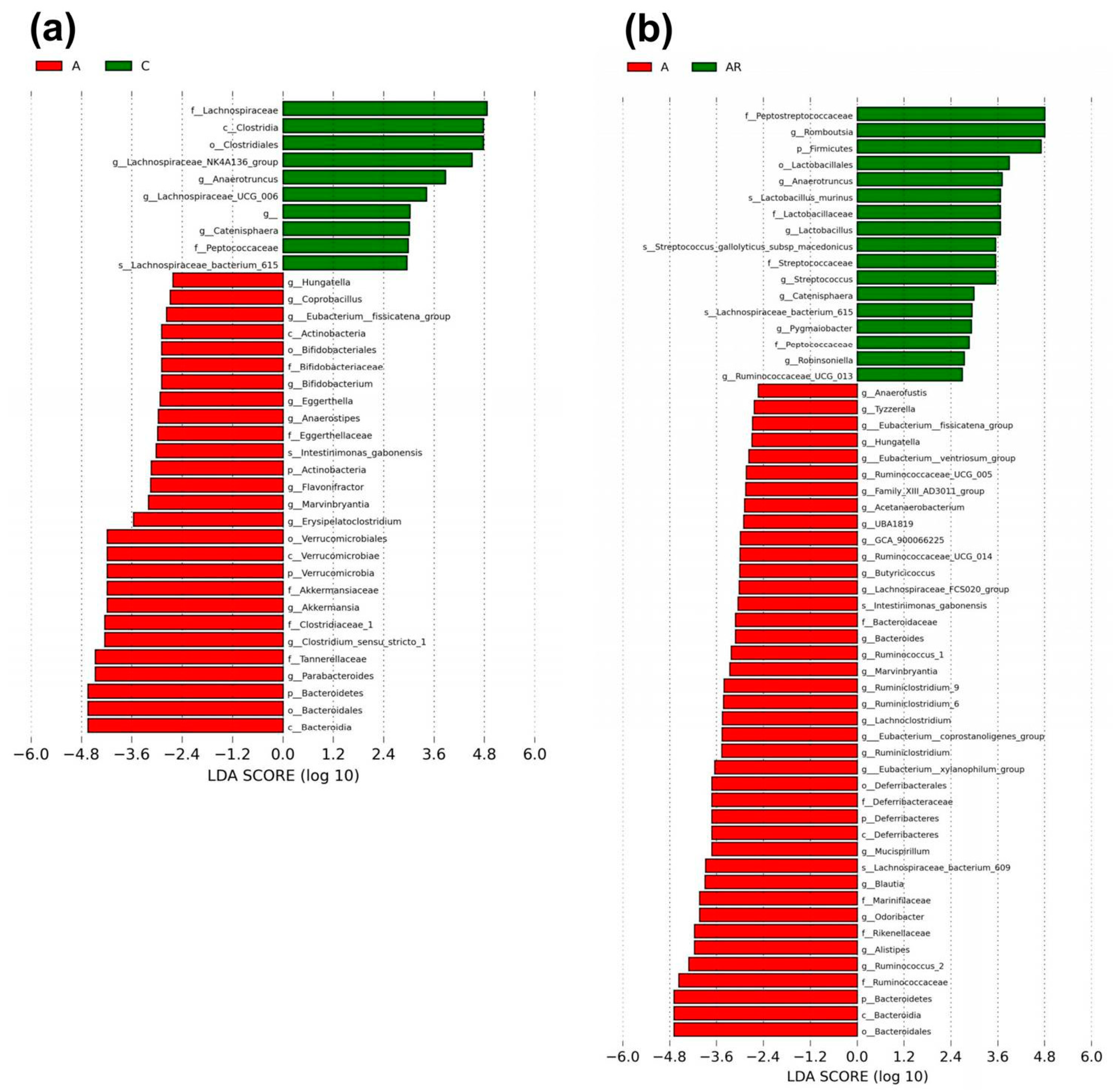
2.5. Gut Microbiota Changes in the Context of ADMA Infusion
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Animals and Grouping
4.3. Tissue Collection
4.4. Morris Water Maze Test
4.5. Enzyme-Linked Immunosorbent Assay (ELISA)
4.6. Western Blot
4.7. Detection of Cytokines in Tissue Protein
4.8. Gut Microbiota Profiling in the Context of ADMA Infusion
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sheen, J.M.; Chen, Y.C.; Hsu, M.H.; Tain, Y.L.; Yu, H.R.; Huang, L.T. Combined Intraperitoneal and Intrathecal Etanercept Reduce Increased Brain Tumor Necrosis Factor-Alpha and Asymmetric Dimethylarginine Levels and Rescues Spatial Deficits in Young Rats after Bile Duct Ligation. Front. Cell. Neurosci. 2016, 10, 167. [Google Scholar] [CrossRef]
- Jayasuriya, R.; Ganesan, K.; Xu, B.; Ramkumar, K.M. Emerging role of long non-coding RNAs in endothelial dysfunction and their molecular mechanisms. Biomed. Pharmacother. Biomed. Pharmacother. 2022, 145, 112421. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, P.; Rengarajan, T.; Thangavel, J.; Nishigaki, Y.; Sakthisekaran, D.; Sethi, G.; Nishigaki, I. The vascular endothelium and human diseases. Int. J. Biol. Sci. 2013, 9, 1057–1069. [Google Scholar] [CrossRef] [PubMed]
- Pellegrini, C.; Antonioli, L.; Calderone, V.; Colucci, R.; Fornai, M.; Blandizzi, C. Microbiota-gut-brain axis in health and disease: Is NLRP3 inflammasome at the crossroads of microbiota-gut-brain communications? Prog. Neurobiol. 2020, 191, 101806. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Pitzer, A.L.; Li, X.; Li, P.L.; Wang, L.; Zhang, Y. Instigation of endothelial Nlrp3 inflammasome by adipokine visfatin promotes inter-endothelial junction disruption: Role of HMGB1. J. Cell. Mol. Med 2015, 19, 2715–2727. [Google Scholar] [CrossRef]
- Falcão Sde, A.; Jaramillo, T.M.; Ferreira, L.G.; Bernardes, D.M.; Santana, J.M.; Favali, C.B. Leishmania infantum and Leishmania braziliensis: Differences and Similarities to Evade the Innate Immune System. Front. Immunol. 2016, 7, 287. [Google Scholar] [CrossRef]
- Li, C.C.; Ye, F.; Xu, C.X.; Chang, Q.; Liu, X.M.; Pan, R.L. Effect of Radix Polygalae extract on the colonic dysfunction in rats induced by chronic restraint stress. J. Ethnopharmacol. 2022, 294, 115349. [Google Scholar] [CrossRef]
- Akbari, E.; Asemi, Z.; Daneshvar Kakhaki, R.; Bahmani, F.; Kouchaki, E.; Tamtaji, O.R.; Hamidi, G.A.; Salami, M. Effect of Probiotic Supplementation on Cognitive Function and Metabolic Status in Alzheimer′s Disease: A Randomized, Double-Blind and Controlled Trial. Front. Aging Neurosci 2016, 8, 256. [Google Scholar] [CrossRef]
- Izquierdo, V.; Palomera-Ávalos, V.; Pallàs, M.; Griñán-Ferré, C. Resveratrol Supplementation Attenuates Cognitive and Molecular Alterations under Maternal High-Fat Diet Intake: Epigenetic Inheritance over Generations. Int. J. Mol. Sci. 2021, 22, 1453. [Google Scholar] [CrossRef]
- Baur, J.A.; Sinclair, D.A. Therapeutic potential of resveratrol: The in vivo evidence. Nat. Rev. Drug Discov. 2006, 5, 493–506. [Google Scholar] [CrossRef]
- Chaplin, A.; Carpéné, C.; Mercader, J. Resveratrol, Metabolic Syndrome, and Gut Microbiota. Nutrients 2018, 10, 1651. [Google Scholar] [CrossRef]
- Huang, Y.C.; Huang, L.T.; Sheen, J.M.; Hou, C.Y.; Yeh, Y.T.; Chiang, C.P.; Lin, I.C.; Tiao, M.M.; Tsai, C.C.; Lin, Y.J.; et al. Resveratrol treatment improves the altered metabolism and related dysbiosis of gut programed by prenatal high-fat diet and postnatal high-fat diet exposure. J. Nutr. Biochem. 2020, 75, 108260. [Google Scholar] [CrossRef]
- Sheen, J.M.; Yu, H.R.; Tain, Y.L.; Chen, Y.C.; Hsu, M.H.; Huang, L.T. Young rats with increased circulatory asymmetric dimethylarginine exhibited spatial deficit and alterations in dorsal hippocampus brain-derived neurotrophic factor and asymmetric dimethylarginine: Effects of melatonin. Int. J. Dev. Neurosci. Off. J. Int. Soc. Dev. Neurosci. 2019, 78, 83–89. [Google Scholar] [CrossRef]
- Hsu, M.H.; Chang, K.A.; Chen, Y.C.; Lin, I.C.; Sheen, J.M.; Huang, L.T. Resveratrol prevented spatial deficits and rescued disarrayed hippocampus asymmetric dimethylarginine and brain-derived neurotrophic factor levels in young rats with increased circulating asymmetric dimethylarginine. Neuroreport 2021, 32, 1091–1099. [Google Scholar] [CrossRef]
- Satish, M.; Agrawal, D.K. Atherothrombosis and the NLRP3 inflammasome—Endogenous mechanisms of inhibition. Transl. Res. 2020, 215, 75–85. [Google Scholar] [CrossRef]
- Zou, P.; Liu, X.; Li, G.; Wang, Y. Resveratrol pretreatment attenuates traumatic brain injury in rats by suppressing NLRP3 inflammasome activation via SIRT1. Mol. Med. Rep. 2018, 17, 3212–3217. [Google Scholar] [CrossRef]
- Jiang, L.; Zhang, L.; Kang, K.; Fei, D.; Gong, R.; Cao, Y.; Pan, S.; Zhao, M.; Zhao, M. Resveratrol ameliorates LPS-induced acute lung injury via NLRP3 inflammasome modulation. Biomed. Pharmacother. Biomed. Pharmacother. 2016, 84, 130–138. [Google Scholar] [CrossRef]
- Dong, W.; Yang, R.; Yang, J.; Yang, J.; Ding, J.; Wu, H.; Zhang, J. Resveratrol pretreatment protects rat hearts from ischemia/reperfusion injury partly via a NALP3 inflammasome pathway. Int. J. Clin. Exp. Pathol. 2015, 8, 8731–8741. [Google Scholar]
- Sandoval, K.E.; Witt, K.A. Blood-brain barrier tight junction permeability and ischemic stroke. Neurobiol. Dis. 2008, 32, 200–219. [Google Scholar] [CrossRef]
- Balzano, T.; Dadsetan, S.; Forteza, J.; Cabrera-Pastor, A.; Taoro-Gonzalez, L.; Malaguarnera, M.; Gil-Perotin, S.; Cubas-Nuñez, L.; Casanova, B.; Castro-Quintas, A.; et al. Chronic hyperammonemia induces peripheral inflammation that leads to cognitive impairment in rats: Reversed by anti-TNF-α treatment. J. Hepatol. 2020, 73, 582–592. [Google Scholar] [CrossRef]
- Keshavarzian, A.; Green, S.J.; Engen, P.A.; Voigt, R.M.; Naqib, A.; Forsyth, C.B.; Mutlu, E.; Shannon, K.M. Colonic bacterial composition in Parkinson′s disease. Mov. Disord. 2015, 30, 1351–1360. [Google Scholar] [CrossRef] [PubMed]
- Fanara, S.; Aprile, M.; Iacono, S.; Schirò, G.; Bianchi, A.; Brighina, F.; Dominguez, L.J.; Ragonese, P.; Salemi, G. The Role of Nutritional Lifestyle and Physical Activity in Multiple Sclerosis Pathogenesis and Management: A Narrative Review. Nutrients 2021, 13, 3774. [Google Scholar] [CrossRef] [PubMed]
- Thye, A.Y.; Law, J.W.; Tan, L.T.; Thurairajasingam, S.; Chan, K.G.; Letchumanan, V.; Lee, L.H. Exploring the Gut Microbiome in Myasthenia Gravis. Nutrients 2022, 14, 1647. [Google Scholar] [CrossRef] [PubMed]
- Vogt, N.M.; Kerby, R.L.; Dill-McFarland, K.A.; Harding, S.J.; Merluzzi, A.P.; Johnson, S.C.; Carlsson, C.M.; Asthana, S.; Zetterberg, H.; Blennow, K.; et al. Gut microbiome alterations in Alzheimer′s disease. Sci. Rep. 2017, 7, 13537. [Google Scholar] [CrossRef] [PubMed]
- Kosuge, A.; Kunisawa, K.; Arai, S.; Sugawara, Y.; Shinohara, K.; Iida, T.; Wulaer, B.; Kawai, T.; Fujigaki, H.; Yamamoto, Y.; et al. Heat-sterilized Bifidobacterium breve prevents depression-like behavior and interleukin-1β expression in mice exposed to chronic social defeat stress. Brain Behav. Immun. 2021, 96, 200–211. [Google Scholar] [CrossRef]
- Shirouchi, B.; Fukuda, A.; Akasaka, T. Unlike Glycerophosphocholine or Choline Chloride, Dietary Phosphatidylcholine Does Not Increase Plasma Trimethylamine-N-Oxide Levels in Sprague-Dawley Rats. Metabolites 2022, 12, 64. [Google Scholar] [CrossRef]
- Wang, Y.; Li, L.; Zhao, X.; Sui, S.; Wang, Q.; Shi, G.; Xu, H.; Zhang, X.; He, Y.; Gu, J. Intestinal Microflora Changes in Patients with Mild Alzheimer’s Disease in a Chinese Cohort. J. Alzheimers Dis. 2022, 88, 563–575. [Google Scholar] [CrossRef]
- Ferlazzo, N.; Currò, M.; Isola, G.; Maggio, S.; Bertuccio, M.P.; Trovato-Salinaro, A.; Matarese, G.; Alibrandi, A.; Caccamo, D.; Ientile, R. Changes in the Biomarkers of Oxidative/Nitrosative Stress and Endothelial Dysfunction Are Associated with Cardiovascular Risk in Periodontitis Patients. Curr. Issues Mol. Biol. 2021, 43, 704–715. [Google Scholar] [CrossRef]
- Wells, S.M.; Holian, A. Asymmetric dimethylarginine induces oxidative and nitrosative stress in murine lung epithelial cells. Am. J. Respir. Cell. Mol. Biol. 2007, 36, 520–528. [Google Scholar] [CrossRef]
- Abderrazak, A.; Syrovets, T.; Couchie, D.; El Hadri, K.; Friguet, B.; Simmet, T.; Rouis, M. NLRP3 inflammasome: From a danger signal sensor to a regulatory node of oxidative stress and inflammatory diseases. Redox Biol. 2015, 4, 296–307. [Google Scholar] [CrossRef]
- Naik, E.; Dixit, V.M. Mitochondrial reactive oxygen species drive proinflammatory cytokine production. J. Exp. Med. 2011, 208, 417–420. [Google Scholar] [CrossRef]
- Qiao, Y.; Sun, J.; Xia, S.; Tang, X.; Shi, Y.; Le, G. Effects of resveratrol on gut microbiota and fat storage in a mouse model with high-fat-induced obesity. Food Funct. 2014, 5, 1241–1249. [Google Scholar] [CrossRef]
- Shen, H.; Guan, Q.; Zhang, X.; Yuan, C.; Tan, Z.; Zhai, L.; Hao, Y.; Gu, Y.; Han, C. New mechanism of neuroinflammation in Alzheimer′s disease: The activation of NLRP3 inflammasome mediated by gut microbiota. Prog. Neuropsychopharmacol. Biol. Psychiatry 2020, 100, 109884. [Google Scholar] [CrossRef]
- Kielstein, H.; Suntharalingam, M.; Perthel, R.; Song, R.; Schneider, S.M.; Martens-Lobenhoffer, J.; Jäger, K.; Bode-Böger, S.M.; Kielstein, J.T. Role of the endogenous nitric oxide inhibitor asymmetric dimethylarginine (ADMA) and brain-derived neurotrophic factor (BDNF) in depression and behavioural changes: Clinical and preclinical data in chronic kidney disease. Nephrol. Dial. Transplant. Off. Publ. Eur. Dial. Transpl. Assoc. Eur. Ren. Assoc. 2015, 30, 1699–1705. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hsu, M.-H.; Huang, Y.-C.; Chen, Y.-C.; Sheen, J.-M.; Huang, L.-T. Increased Circulating ADMA in Young Male Rats Caused Cognitive Deficits and Increased Intestinal and Hippocampal NLRP3 Inflammasome Expression and Microbiota Composition Alterations: Effects of Resveratrol. Pharmaceuticals 2023, 16, 825. https://doi.org/10.3390/ph16060825
Hsu M-H, Huang Y-C, Chen Y-C, Sheen J-M, Huang L-T. Increased Circulating ADMA in Young Male Rats Caused Cognitive Deficits and Increased Intestinal and Hippocampal NLRP3 Inflammasome Expression and Microbiota Composition Alterations: Effects of Resveratrol. Pharmaceuticals. 2023; 16(6):825. https://doi.org/10.3390/ph16060825
Chicago/Turabian StyleHsu, Mei-Hsin, Yi-Chuan Huang, Yu-Chieh Chen, Jiunn-Ming Sheen, and Li-Tung Huang. 2023. "Increased Circulating ADMA in Young Male Rats Caused Cognitive Deficits and Increased Intestinal and Hippocampal NLRP3 Inflammasome Expression and Microbiota Composition Alterations: Effects of Resveratrol" Pharmaceuticals 16, no. 6: 825. https://doi.org/10.3390/ph16060825
APA StyleHsu, M.-H., Huang, Y.-C., Chen, Y.-C., Sheen, J.-M., & Huang, L.-T. (2023). Increased Circulating ADMA in Young Male Rats Caused Cognitive Deficits and Increased Intestinal and Hippocampal NLRP3 Inflammasome Expression and Microbiota Composition Alterations: Effects of Resveratrol. Pharmaceuticals, 16(6), 825. https://doi.org/10.3390/ph16060825






