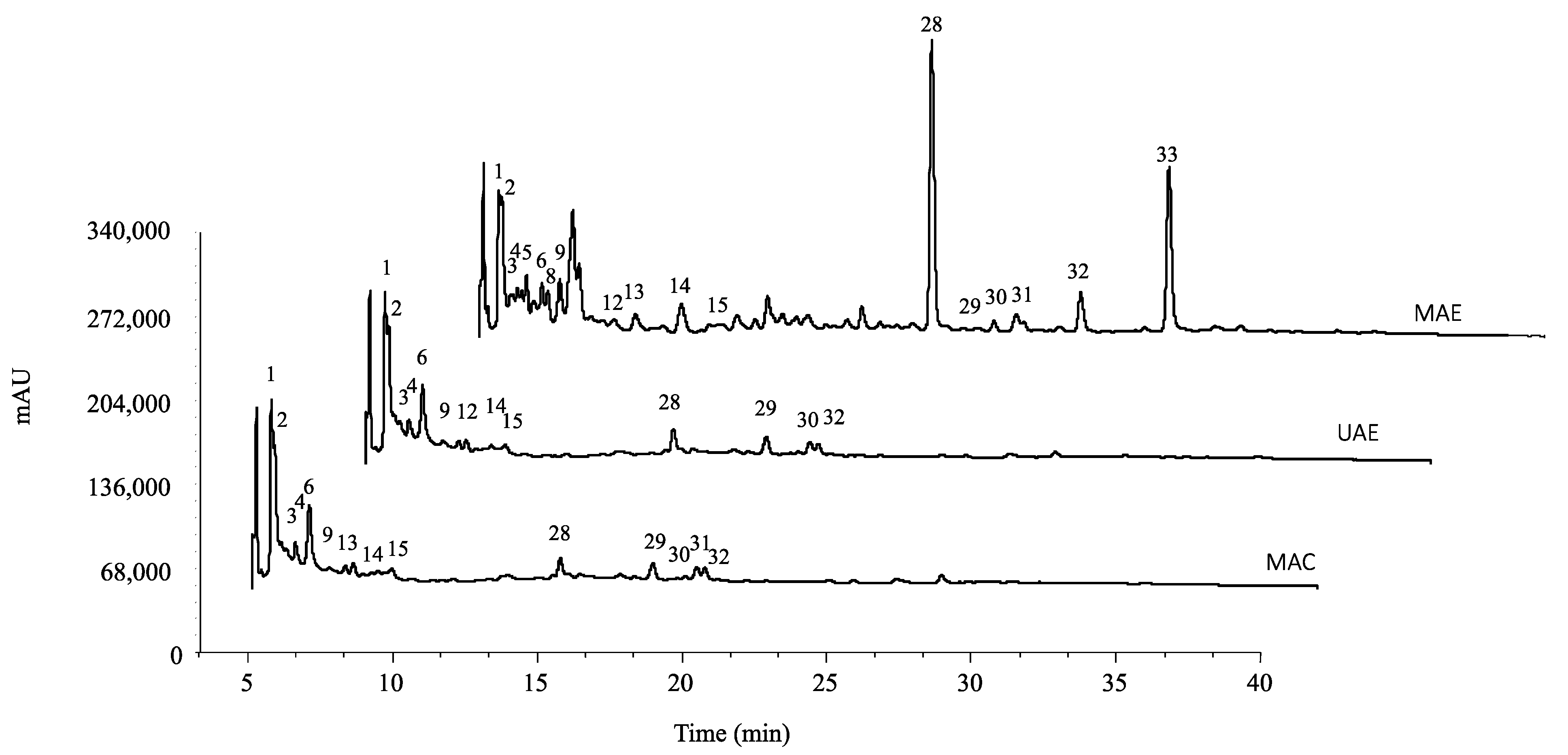
2.1. Phenolic Composition
The tentatively identified compounds and their quantification are presented in
Table 1 and
Table 2, respectively, and the chromatogram is shown in
Figure 1. Thirty-three compounds were tentatively identified, thirteen of which were phenolic acids (peaks 1, 2, 3, 4, 5, 6, 8, 9, 10, 12, 13, 14, 15), fifteen flavonoids (peaks 7, 11, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27), and six tannins (peaks 28, 29, 30, 31, 32, 33).
Peak 1 ([M–H]
− at
m/
z 169) was positively identified as gallic acid based on its retention time, mass, and UV-vis properties in comparison to commercial standards. Peaks 2 and 6 ([M–H]
− at
m/
z 353) were confirmed as
O-caffeoylquinic acid, and the base peak provided deprotonated quinic acid (
m/
z at 191) and another significant ion belonging to the hydroxycinnamic acid residue at
m/
z 179 [caffeic acid-H]
−. Because of the hierarchical fragmentation pattern outlined by Clifford et al. [
35], these assumptions were taken into account. Peak 3 was identified as methyl gallate hexoside ([M–H]
− at
m/
z 345), presenting a λ
max around 278 nm. Díaz-Mula et al. reported a similar compound in their study [
36]. Peaks 5 and 8 ([M–H]
− at
m/
z 355) were identified as glucaric acid (209
m/
z), revealing the loss of a rhamnosyl moiety (- 146 u). Peaks 9 ([M–H]
− at
m/
z 477), 13 ([M–H]
− at
m/
z 609), 14 ([M–H]
− at
m/
z 433), and 15 ([M–H]
− at
m/
z 447) were tentatively identified as methyl ellagic acid hexoside, ellagic acid (
p-coumaroyl) hexoside, ellagic acid pentoside, and methyl ellagic acid pentoside, respectively, all revealing a
λmax around 358 nm and a MS
2 fragment at
m/
z 301, characteristic of ellagic acid. Peak 12 ([M–H]
− at
m/
z 355) revealed, after the loss of a hexoside unit (−162 u), a product ion equivalent to a ferulic acid molecule (193
m/
z) and was tentatively identified as ferulic acid hexoside.
From all the phenolic acids identified, gallic acid was the compound that presented the highest concentrations through UAE and MAC (
Table 2).
Regarding the flavonoids, peaks 7 and 11 ([M–H]
− at
m/
z 289) and peaks 17 ([M–H]
− at
m/
z 609) and 19 ([M–H]
− at
m/
z 463) were identified according to the standard compounds as (+)-catechin, (-)-epicatechin, quercetin-3-
O-rutinoside, and quercetin-3-
O-glucoside, respectively. Peaks 20 ([M–H]
− at
m/
z 477) and 23 ([M–H]
− at
m/
z 505) were assigned as quercetin-
O-glucuronide and quercetin-
O-acetyl-glucoside, respectively. They presented MS
2 fragments, corresponding to the loss of a glucuronide (- 176 u) and acetyl-glucoside (- 42-162 u). Peaks 16 ([M–H]
− at
m/
z 491), 24 ([M–H]
− at
m/
z 447), and 26 ([M–H]
− at
m/
z 461) corresponded to isorhamnetin derivatives (λ
max around 354 nm, and MS
2 fragment at
m/
z 315). Peak 16 presented a pseudomolecular ion [M–H]
− at
m/
z 491, which released fragments at
m/
z 315 ([M–H–176]
−), representing the loss of a glucuronyl moiety. Peak 24 was characterized as isorhamnetin-
O-pentoside, and peak 26 was identified as isorhamnetin-
O-rhamnoside based on its fragmentation pattern. Peaks 18 ([M–H]
− at
m/
z 593), 21 ([M–H]
− at
m/
z 447), and 22 ([M–H]
− at
m/
z 461) were identified as luteolin derivatives. These compounds were tentatively identified as luteolin-7-
O-rutinoside, luteolin-7-
O-glucoside, and luteolin-
O-glucuronide, based on their pseudomolecular ions and MS
2 fragment losses corresponding to rutinosyl (- 308 u), hexosyl (- 162 u), and glucuronyl (- 176 u) moieties, respectively. Peak 25 (kaempferol-3-
O-glucoside) was identified by its retention, mass spectra, and UV-vis characteristics by comparison with commercial standards (max. around 334 nm, and MS
2 fragment at
m/
z 285). Luteolin derivatives have also been identified in the pomegranate’s peel [
37,
38]. Peak 27 was assigned to a flavone, apigenin-
O-glucuronide ([M–H]
− at
m/
z 445), releasing an MS
2 fragment at
m/
z 269 ([M–H–176]
−), apigenin with a loss of a glucuronyl moiety.
Epicatechin (peak 11) was the major flavonoid compound found in MAE and MAC extract, while the UAE revealed quercetin-
O-glucuronide as the main compound (
Table 2).
Finally, peaks 28–33 were assigned as ellagitannins. Peak 29 ([M–H]
− at
m/
z 1083) was identified as punicalagin, which is very characteristic of
P. granatum, and this peak was also described by Fischer et al. [
39] and Lu et al. [
40]. Similarly, peak 28 ([M–H]
− at
m/
z 951) was identified as granatin B (galloyl-HHDP-DHHDP-hexoside), which was previously identified by Canuti et al. [
41]. Peak 30 ([M–H]
− at
m/
z 799) was identified as lagerstannin A, which has also been reported in
P. granatum [
39]. The mass spectral characteristics of peak 31 ([M–H]
− a
m/
z 785, fragments
m/
z 633 and 301) coincide with digalloyl-HHDP-glucose, also described in pomegranate epicarp [
42]. Peaks 32 ([M–H]
− at
m/
z 783) and 33 ([M–H]
− at
m/
z 935) were tentatively identified as pedunculagin (bis-HHDP-glucose) and casuarictin (1-β-
O-galloyl-pedunculagin), respectively, and previously reported by Singh et al. [
43]. In the latter case, the gallic acid would not be bound to punicalagin by the carboxyl group, as denoted by the fragment at
m/
z 783 corresponding to the loss of gallic acid itself (−152 u).
Granatin B (peak 28) was the hydrosoluble tannin found in higher concentrations in MAE and MAC extracts (
Table 2).
MAE extract presented the highest concentration of total phenolic compounds, followed by MAC and then UAE extract (15.9, 10.1, and 6.6 mg/g, respectively). MAE extracts presented higher concentrations of hydrolysable tannins, with granatin B being the one standing out with the highest concentration (3 mg/g). MAE extract also presented the highest concentration of total flavonoids, 6.8 mg/g, with (-)-epicatechin being the one with a higher concentration (2.4 mg/g). In opposition, the extract that presented the highest amount of phenolic acids was the MAC extract. This extract presented a total phenolic acid concentration of 4.45 mg/g, with gallic acid being the phenolic acid compound that presented the highest concentration in all three extracts (MAE—0.95 mg/g; UAE—1.19 mg/g; MAC—1.45 mg/g).
Several authors have already studied this compound and linked these molecules to several health properties of interest, such as anticancer, antidiabetic, antioxidant, and anti-inflammatory [
44,
45,
46]. The extraction of these compounds from pomegranate leaves, which are an economically promising and abundant by-product, could be of interest for the food and pharmaceutic industry.
2.2. Antioxidant Activity
The present work evaluated the antioxidant activity of the three hydroethanolic leaf extracts using the cell-based assays, TBARS and CAA. The obtained results are presented in
Table 3. For the TBARS assay, the results demonstrated that MAC and MAE presented the most potent TBARS inhibitory capacity, exhibiting the lowest IC
50 values (0.83 and 0.86 μg/mL, respectively). On the contrary, UAE extract presented a IC
50 value of 1.70 μg/mL, being the extract with lesser antioxidant potential. Nonetheless, all three extracts exhibited higher antioxidant effects than the positive control trolox (10.7, 10.9, and 5.3 times higher, for MAC, MAE, and UAE, respectively). In addition to the TBARS assay, the pomegranate leaves extracts were also submitted to intracellular ROS inhibition assay induced by dichlorodihydrofluorescein diacetate in RAW 264.7 macrophage cell lines. The results showed no ROS inhibition at the highest tested concentration (2000 µg/mL) for all the studied extracts (
Table 3).
MAE and MAC extracts exhibited lower IC
50 values and, therefore, higher antioxidant potential and are also the extracts that presented higher total phenolic compositions (15.9 and 10.1 mg/g, respectively). Our results are in agreement with the ones obtained by Derakhshan et al. [
20], who studied pomegranate peel, seed, and juice. According to other authors, antioxidant activity is frequently associated with phenolic compounds concentration [
47]. In our investigation, the samples with the highest content of phenolic compounds (MAE and MAC) presented lower IC
50 values in the TBARS production assay when compared to the third extraction methodology (UAE). These data point to a probable positive association between these two measures (phenolic compound concentration and inhibition of TBARS generation). To the best of the author’s knowledge, this is the first study that investigates and analyzes the antioxidant potential of three hydroethanolic pomegranate leaf extracts obtained through two cell-based assays.
2.3. Cytotoxic and Hepatotoxic Activity
The cytotoxic efficacy of several pomegranate leaf extracts was investigated against four human tumor cell lines and a primary pig liver cell line (PLP2).
Table 4 displays the collected qualitative properties. The results are reported as the extract concentration that inhibits cell growth by 50% (GI
50); therefore, a lower GI
50 value indicates more effective cytotoxic action.
All three extracts presented cytotoxic effects against all tested tumor cell lines. Gastric adenocarcinoma (AGS), followed by the colorectal tumor cells (CaCo2), showed a higher sensitivity to the three leaf extracts when compared to the remaining tumor cell lines tested (
Table 4). Both AGS and CaCo
2 cell lines presented the lowest GI
50 values when exposed to the UAE extract (19 and 63 μg/mL, respectively). Overall, the UAE leaf extract presented a higher cytotoxic effect when compared to the other two extracts. It should be noted the existence of toxicity in relation to non-tumor liver cells (PLP2), with values of GI
50 between 54 and 57 μg/mL (
Table 4). Therefore, further studies are required to verify the cytotoxicity of pomegranate leaves against other non-tumor cells and its possible use as an effective and safe chemotherapeutic ingredient. Breast adenocarcinoma cell line (MCF-7) presented similar GI
50 values for the three tested extracts (between 70 and 71 μg/mL). In agreement with our findings, Li et al. [
25] verified that pomegranate leaf extract could inhibit H1299 lung cancer cells proliferation by inhibiting the cell cycle progression and inducing apoptosis. Most of the existent studies that evidence pomegranate as having anti-cancer effects use in general PJ or pomegranate fruit extract [
48,
49]. A previous in vitro study showed that the proliferation and cell growth of the MCF-7 breast cancer cell line were significantly inhibited by PJ [
50]. Nonetheless, the anti-tumor potential of the fruit is not limited to the edible part. Other pomegranate plant parts have also been shown to have antiproliferative and cytotoxic properties against prostate, lung, colon, and skin cancer cell models [
49]. Both Hong et al. [
51] and Seidi et al. [
52] verified that punicalagin and ellagic acid isolated from the pomegranate peel as well as pomegranate leaves extract significantly inhibited the proliferation of A549 and H1299 lung cancer cell lines. In particular, punicalagin, a known hydrolysable tannin present in the three studied extracts, was reported to inhibit cell viability, migration, and invasion in MCF-7 and MDA-MB-231 breast cancer cell models [
53].
2.6. Antimicrobial Activity
In this study, the antibacterial and antifungal properties of the different leaf extracts were accessed. The minimum inhibitory concentration (MIC), minimum bactericidal concentration (MBC), and minimum fungicidal concentration (MFC) values are shown in
Table 6 and
Table 7. All the studied extracts had the capacity to inhibit bacterial growth of the clinical strains screened (0.03125 to 10 mg/mL). The MIC values obtained for both Gram-positive and Gram-negative bacteria were similar.
K. pneumoniae presented the lowest MIC values (0.6 mg/mL), and as such, revealed the highest susceptibility to the three tested pomegranate leaves’ extracts.
Morganella morganii, an opportunistic pathogen that colonizes post-operative wounds and causes urinary tract infections, was also effectively inhibited by MAE extract. Although the antibacterial potential is influenced by the bacteria strain under analysis, the MAE and MAC extracts were the ones with the most promising activity, exhibiting the lowest MIC values for the five Gram-negative strains tested. Regarding the Gram-positive strains, all extracts showed some antibacterial inhibition activity. MRSA was revealed to be the most sensitive bacteria to our different leaf extracts, where the MIC values vary between 0.3 and 0.6 mg/mL. In accordance with other authors, these results could be linked to the fact that phenolic compounds inhibit bacterial proliferation and swimming ability, damaging the integrity and stability of the bacterial cell membrane [
61]. Pisoschi et al. [
62] has explained that polyphenols interact with membrane proteins, disrupting cell membrane functions, and having an impact on nutrient absorption, the electron transfer system, enzyme activity, and protein and nucleic acid synthesis. When the cell membrane’s integrity is compromised, polyphenols, such as punicalagin, enter the cytoplasm and bind to the target gene domain, causing regulatory networks to become disorganized and eventually causing bacterial damage.
As for the food contaminants (
Table 7), as with the clinical strains, the three extracts showed the ability to inhibit bacterial growth and did not cause the bacterial strains’ death. Regarding Gram-negative bacteria, we verified that
Salmonella enterica was the one presenting higher sensibility to the extracts, with the MAE being most effective in inhibiting its growth.
Staphyloccocus aureus was the Gram-positive strain that showed the most significant inhibitory response, when compared with the other two strains. In this case, as with the clinical strains, MAE was the extract showing the best inhibition results.
To the best of our knowledge, no studies have been performed on pomegranate leaf extract and food contaminants’ inhibition growth, but several studies do exist about pomegranate peel extract and the use of pomegranate juice to inhibit bacterial growth. PP is the most studied by-product, and according to Aguilera-Carbo et al. [
63] and Akhtar et al. [
64], pomegranate peel seems to exert antibacterial effects on a wide number of foodborne pathogens and infectious microorganisms (e.g.,
Staphylococcus aureus,
Escherichia coli,
Klebsiella pneumoniae,
Bacillus subtilis). Tayel et al. [
65] and Kharchoufi et al. [
66] explained that the antibacterial activity of pomegranate peel extract could be attributed to the combined effect of its constituents, which include powerful compounds with antibacterial, antifungal, and antioxidant properties, such as kaempferol, castalagin, granatin, gallocatechin, quercetin, and other phytochemical compounds present in minor quantities. Those compounds were also identified in our leaves’ extracts, suggesting a similar antimicrobial potential. Pomegranate peel, as well as the studied hydroethanolic extracts, contain a punicalagin compound, which is said to have powerful antibacterial properties against
Pseudomonas aeruginosa and
Staphyloccocus aureus. According to Nuamsetti et al. [
67], not only does the peel seem to present those properties, pomegranate arils, including seeds, also present antibacterial properties against
Bacillus subtilis,
Staphylococcus aureus,
Salmonella typhimurium, and
Escherichia coli. Okan et al. [
68] also investigated the antifungal properties of pomegranate seed oil (PSO) against five different plant pathogens and discovered that at a concentration of 1000 ppm, PSO inhibited only about 20% mycelial growth.
The antifungal activity was also evaluated (
Table 6), and
A. brasiliensis appeared to be the most sensitive to the tested extracts, with MIC values ranging from 0.6 to 1.25 mg/mL. The MAE extract presented the lowest MIC values and, therefore, has the best antifungal potential, even though the other two tested extracts presented a relative inhibition potential. In the same line of fungal study, Bhinge et al. [
69] studied the alcoholic leaf extract of pomegranate and found that it inhibited the growth of major fungal pathogens that cause dandruff, such as
Candida albicans,
Aspergillus niger, and
Penicillium notatum. As a possible explanation, and according to some authors, the induction of host defense mechanisms and changes in fungal cell membrane permeability could be attributed to the polyphenol content [
70].
Overall, pomegranate leaf extracts constitute a promising natural source of antimicrobial compounds. This knowledge testifies to their capacity to inhibit several multi-resistant bacteria and fungi strains responsible for causing several public health concerns. The obtained results encourage further study, enhancing its bioactive potential and so supporting its sustainable exploitation.