Smart Biomimetic Nanozymes for Precise Molecular Imaging: Application and Challenges
Abstract
1. Introduction
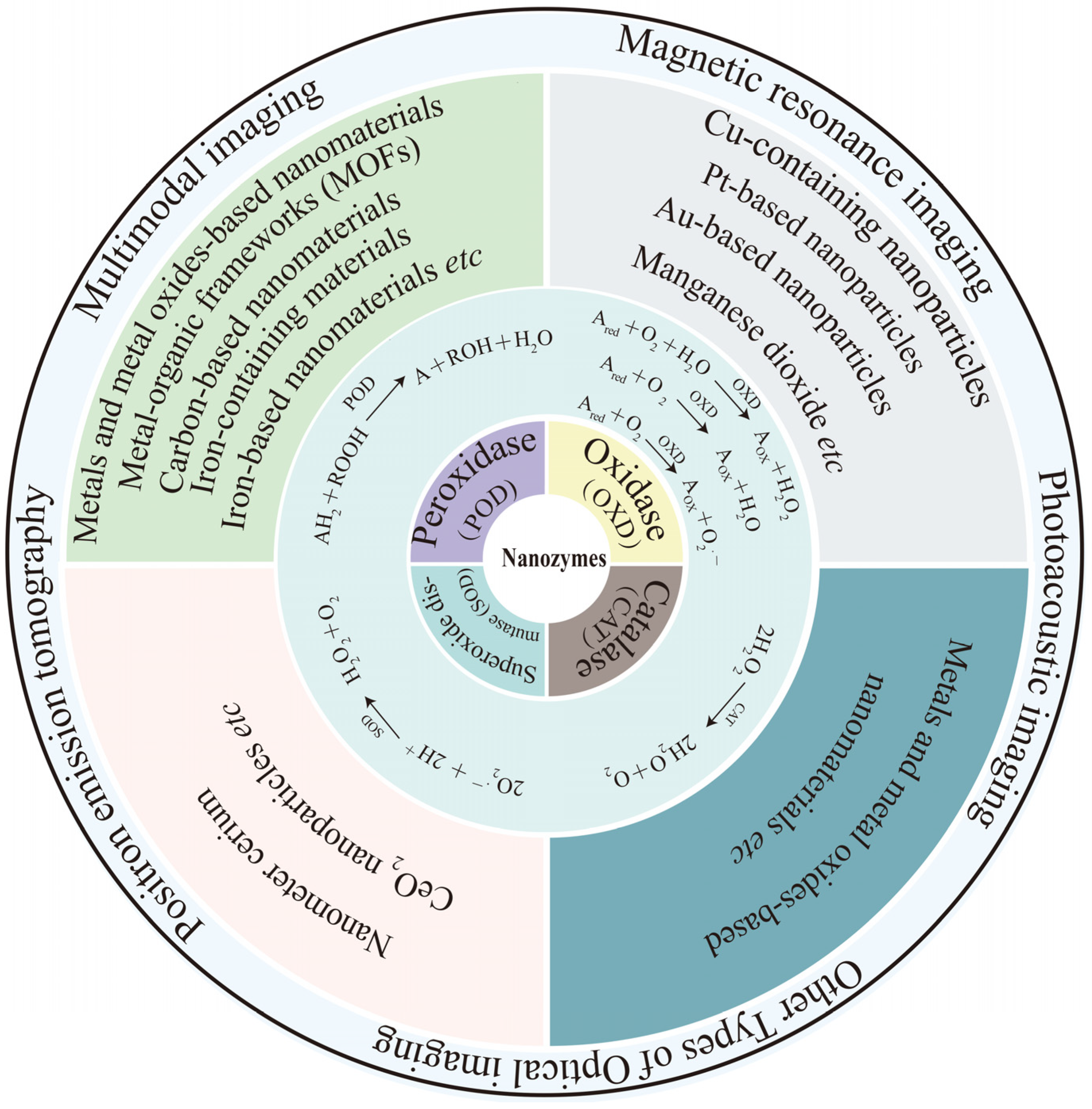
2. Classification of Nanozymes
2.1. POD-like Nanozymes
2.2. OXD-like Nanozymes
2.3. CAT-like Nanozymes
2.4. SOD-like Nanozymes
3. Nanozymes for Molecular Imaging
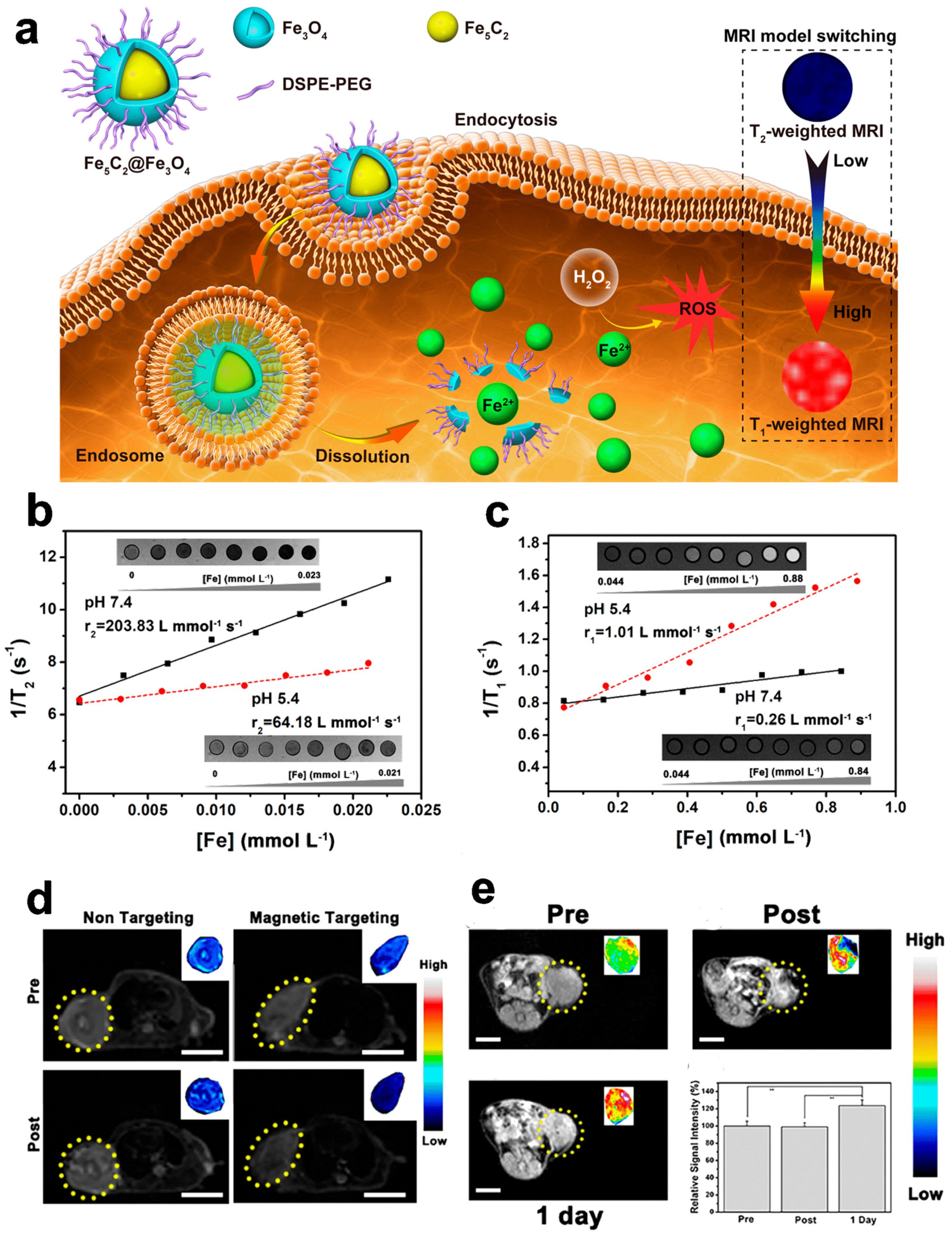
3.1. Magnetic Resonance Imaging
3.2. Photoacoustic Imaging
3.3. Other Types of Optical Imaging
3.4. Positron Emission Tomography
3.5. Multimodal Imaging
4. Conclusions and Outlook
- Poor dispersion, uncontrollable precipitation after surface modification, limited types of catalytic activity, poor substrate selectivity, and potential nanotoxicity due to the drawbacks of current preparation technologies and methods.
- Systemic toxicity derived from the overaccumulation of metal elements; for instance, the direct toxic reaction caused by gold metal overaccumulation after long-term application and the indirect toxic response, such as ion-induced oxidative stress, DNA damage, and normal tissue apoptosis. Reducing potential adverse side effects and improving the accuracy and efficacy of diagnosis and treatment are significant problems that must be solved for the viable and sustainable development of metal nanozymes.
- There is a lack of a comprehensive monitoring and evaluation system for nanozymes after administration in living systems, such as histocompatibility, blood compatibility, immunogenicity, biological distribution, cytotoxicity, in vivo uptake, and metabolism. These factors affect the effective clinical conversion of nanozymes.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Massoud, T.F.; Gambhir, S.S. Molecular Imaging in Living Subjects: Seeing Fundamental Biological Processes in a New Light. Genes Dev. 2003, 17, 545–580. [Google Scholar] [CrossRef]
- James, M.L.; Gambhir, S.S. A Molecular Imaging Primer: Modalities, Imaging Agents, and Applications. Physiol. Rev. 2012, 92, 897–965. [Google Scholar] [CrossRef] [PubMed]
- Böhmer, V.I.; Szymanski, W.; Feringa, B.L.; Elsinga, P.H. Multivalent Probes in Molecular Imaging: Reality or Future? Trends Mol. Med. 2021, 27, 379–393. [Google Scholar] [CrossRef] [PubMed]
- van Duijnhoven, S.M.J.; Robillard, M.S.; Langereis, S.; Grüll, H. Bioresponsive Probes for Molecular Imaging: Concepts and in Vivo Applications: Bioresponsive Molecular Imaging Probes. Contrast Media Mol. Imaging 2015, 10, 282–308. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, F.; Kelly, K.A. Techniques for Molecular Imaging Probe Design. Mol. Imaging 2011, 10, 407–419. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Chen, X. Design and Development of Molecular Imaging Probes. CTMC 2010, 10, 1227–1236. [Google Scholar] [CrossRef]
- Nerella, S.G.; Singh, P.; Sanam, T.; Digwal, C.S. PET Molecular Imaging in Drug Development: The Imaging and Chemistry. Front. Med. 2022, 9, 812270. [Google Scholar] [CrossRef]
- Bouleau, A.; Lebon, V.; Truillet, C. PET Imaging of Immune Checkpoint Proteins in Oncology. Pharmacol. Ther. 2021, 222, 107786. [Google Scholar] [CrossRef]
- Massoud, T.F.; Gambhir, S.S. Integrating Noninvasive Molecular Imaging into Molecular Medicine: An Evolving Paradigm. Trends Mol. Med. 2007, 13, 183–191. [Google Scholar] [CrossRef]
- Hussain, T.; Nguyen, Q.T. Molecular Imaging for Cancer Diagnosis and Surgery. Adv. Drug Deliv. Rev. 2014, 66, 90–100. [Google Scholar] [CrossRef]
- Lee, S.; Xie, J.; Chen, X. Peptides and Peptide Hormones for Molecular Imaging and Disease Diagnosis. Chem. Rev. 2010, 110, 3087–3111. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Hu, Z. Targeting Peptide-Based Probes for Molecular Imaging and Diagnosis. Adv. Mater. 2019, 31, 1804827. [Google Scholar] [CrossRef]
- He, H.; Zhang, X.; Du, L.; Ye, M.; Lu, Y.; Xue, J.; Wu, J.; Shuai, X. Molecular Imaging Nanoprobes for Theranostic Applications. Adv. Drug Deliv. Rev. 2022, 186, 114320. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, G.; Zeng, Z.; Pu, K. Activatable Molecular Probes for Fluorescence-Guided Surgery, Endoscopy and Tissue Biopsy. Chem. Soc. Rev. 2022, 51, 566–593. [Google Scholar] [CrossRef]
- Li, K.C.P.; Pandit, S.D.; Guccione, S.; Bednarski, M.D. Molecular Imaging Applications in Nanomedicine. Biomed. Microdevices 2004, 6, 113–116. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Swierczewska, M.; Lee, S.; Chen, X. Functional Nanoparticles for Molecular Imaging Guided Gene Delivery. Nano Today 2010, 5, 524–539. [Google Scholar] [CrossRef] [PubMed]
- Toy, R.; Bauer, L.; Hoimes, C.; Ghaghada, K.B.; Karathanasis, E. Targeted Nanotechnology for Cancer Imaging. Adv. Drug Deliv. Rev. 2014, 76, 79–97. [Google Scholar] [CrossRef] [PubMed]
- Thakor, A.S.; Gambhir, S.S. Nanooncology: The Future of Cancer Diagnosis and Therapy. CA A Cancer J. Clin. 2013, 63, 395–418. [Google Scholar] [CrossRef]
- Cai, W.; Chen, X. Nanoplatforms for Targeted Molecular Imaging in Living Subjects. Small 2007, 3, 1840–1854. [Google Scholar] [CrossRef]
- Golchin, J.; Golchin, K.; Alidadian, N.; Ghaderi, S.; Eslamkhah, S.; Eslamkhah, M.; Akbarzadeh, A. Nanozyme Applications in Biology and Medicine: An Overview. Artif. Cells Nanomed. Biotechnol. 2017, 45, 1069–1076. [Google Scholar] [CrossRef]
- Estelrich, J.; Busquets, M.A. Prussian Blue: A Nanozyme with Versatile Catalytic Properties. IJMS 2021, 22, 5993. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Wang, E. Nanomaterials with Enzyme-like Characteristics (Nanozymes): Next-Generation Artificial Enzymes. Chem. Soc. Rev. 2013, 42, 6060. [Google Scholar] [CrossRef]
- Wu, J.; Wang, X.; Wang, Q.; Lou, Z.; Li, S.; Zhu, Y.; Qin, L.; Wei, H. Nanomaterials with Enzyme-like Characteristics (Nanozymes): Next-Generation Artificial Enzymes (II). Chem. Soc. Rev. 2019, 48, 1004–1076. [Google Scholar] [CrossRef]
- Meng, X.; Fan, K.; Yan, X. Nanozymes: An Emerging Field Bridging Nanotechnology and Enzymology. Sci. China Life Sci. 2019, 62, 1543–1546. [Google Scholar] [CrossRef] [PubMed]
- Sindhu, R.K.; Najda, A.; Kaur, P.; Shah, M.; Singh, H.; Kaur, P.; Cavalu, S.; Jaroszuk-Sierocińska, M.; Rahman, M.H. Potentiality of Nanoenzymes for Cancer Treatment and Other Diseases: Current Status and Future Challenges. Materials 2021, 14, 5965. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Wang, T.; Hong, J.; Yan, X.; Liang, M. Nanozymes: A New Disease Imaging Strategy. Front. Bioeng. Biotechnol. 2020, 8, 15. [Google Scholar] [CrossRef]
- Vallabani, N.V.S.; Singh, S.; Karakoti, A.S. Magnetic Nanoparticles: Current Trends and Future Aspects in Diagnostics and Nanomedicine. CDM 2019, 20, 457–472. [Google Scholar] [CrossRef]
- Fan, W.; Tang, W.; Lau, J.; Shen, Z.; Xie, J.; Shi, J.; Chen, X. Breaking the Depth Dependence by Nanotechnology-Enhanced X-Ray-Excited Deep Cancer Theranostics. Adv. Mater. 2019, 31, 1806381. [Google Scholar] [CrossRef] [PubMed]
- Darabdhara, G.; Das, M.R.; Singh, S.P.; Rengan, A.K.; Szunerits, S.; Boukherroub, R. Ag and Au Nanoparticles/Reduced Graphene Oxide Composite Materials: Synthesis and Application in Diagnostics and Therapeutics. Adv. Colloid Interface Sci. 2019, 271, 101991. [Google Scholar] [CrossRef]
- Sarbadhikary, P.; George, B.P.; Abrahamse, H. Recent Advances in Photosensitizers as Multifunctional Theranostic Agents for Imaging-Guided Photodynamic Therapy of Cancer. Theranostics 2021, 11, 9054–9088. [Google Scholar] [CrossRef]
- Yun, S.-W.; Kang, N.-Y.; Park, S.-J.; Ha, H.-H.; Kim, Y.K.; Lee, J.-S.; Chang, Y.-T. Diversity Oriented Fluorescence Library Approach (DOFLA) for Live Cell Imaging Probe Development. Accounts Chem. Res. 2014, 47, 1277–1286. [Google Scholar] [CrossRef] [PubMed]
- Sharkey, R.M.; Goldenberg, D.M. Targeted Therapy of Cancer: New Prospects for Antibodies and Immunoconjugates. CA A Cancer J. Clin. 2006, 56, 226–243. [Google Scholar] [CrossRef]
- Jiang, D.; Ni, D.; Rosenkrans, Z.T.; Huang, P.; Yan, X.; Cai, W. Nanozyme: New Horizons for Responsive Biomedical Applications. Chem. Soc. Rev. 2019, 48, 3683–3704. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Ren, J.; Qu, X. Nanozymes: Classification, Catalytic Mechanisms, Activity Regulation, and Applications. Chem. Rev. 2019, 119, 4357–4412. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wan, K.; Shi, X. Recent Advances in Nanozyme Research. Adv. Mater. 2019, 31, 1805368. [Google Scholar] [CrossRef]
- Robert, A.; Meunier, B. How to Define a Nanozyme. ACS Nano 2022, 16, 6956–6959. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, X.; Zhao, Y. Nanozymes: Versatile Platforms for Cancer Diagnosis and Therapy. Nano-Micro Lett. 2022, 14, 95. [Google Scholar] [CrossRef]
- Liang, M.; Yan, X. Nanozymes: From New Concepts, Mechanisms, and Standards to Applications. Accounts Chem. Res. 2019, 52, 2190–2200. [Google Scholar] [CrossRef]
- Cao, C.; Zhang, T.; Yang, N.; Niu, X.; Zhou, Z.; Wang, J.; Yang, D.; Chen, P.; Zhong, L.; Dong, X.; et al. POD Nanozyme Optimized by Charge Separation Engineering for Light/PH Activated Bacteria Catalytic/Photodynamic Therapy. Signal Transduct. Target Ther. 2022, 7, 86. [Google Scholar] [CrossRef]
- Chen, G.-Q.; Benthani, F.A.; Wu, J.; Liang, D.; Bian, Z.-X.; Jiang, X. Artemisinin Compounds Sensitize Cancer Cells to Ferroptosis by Regulating Iron Homeostasis. Cell Death Differ. 2020, 27, 242–254. [Google Scholar] [CrossRef]
- Shi, C.; Li, Y.; Gu, N. Iron-Based Nanozymes in Disease Diagnosis and Treatment. ChemBioChem 2020, 21, 2722–2732. [Google Scholar] [CrossRef]
- Gao, L.; Zhuang, J.; Nie, L.; Zhang, J.; Zhang, Y.; Gu, N.; Wang, T.; Feng, J.; Yang, D.; Perrett, S.; et al. Intrinsic Peroxidase-like Activity of Ferromagnetic Nanoparticles. Nat. Nanotechnol. 2007, 2, 577–583. [Google Scholar] [CrossRef]
- Chen, J.; Wang, Q.; Huang, L.; Zhang, H.; Rong, K.; Zhang, H.; Dong, S. Prussian Blue with Intrinsic Heme-like Structure as Peroxidase Mimic. Nano Res. 2018, 11, 4905–4913. [Google Scholar] [CrossRef]
- Gautam, M.; Poudel, K.; Yong, C.S.; Kim, J.O. Prussian Blue Nanoparticles: Synthesis, Surface Modification, and Application in Cancer Treatment. Int. J. Pharm. 2018, 549, 31–49. [Google Scholar] [CrossRef]
- Cheng, N.; Li, J.; Liu, D.; Lin, Y.; Du, D. Single-Atom Nanozyme Based on Nanoengineered Fe–N–C Catalyst with Superior Peroxidase-Like Activity for Ultrasensitive Bioassays. Small 2019, 15, 1901485. [Google Scholar] [CrossRef]
- Wang, X.; Wang, H.; Zhou, S. Progress and Perspective on Carbon-Based Nanozymes for Peroxidase-like Applications. J. Phys. Chem. Lett. 2021, 12, 11751–11760. [Google Scholar] [CrossRef]
- Pan, T.; Chen, H.; Gao, X.; Wu, Z.; Ye, Y.; Shen, Y. Engineering Efficient Artificial Nanozyme Based on Chitosan Grafted Fe-Doped-Carbon Dots for Bacteria Biofilm Eradication. J. Hazard. Mater. 2022, 435, 128996. [Google Scholar] [CrossRef]
- Lu, C.; Tang, L.; Gao, F.; Li, Y.; Liu, J.; Zheng, J. DNA-Encoded Bimetallic Au-Pt Dumbbell Nanozyme for High-Performance Detection and Eradication of Escherichia Coli O157:H7. Biosens. Bioelectron. 2021, 187, 113327. [Google Scholar] [CrossRef]
- Wu, S.-C.; Tsai, T.-T.; Li, T.-H.; Tung, C.-Y.; Chiu, P.-Y.; Lin, J.-H.; Chen, C.-F. Palladium-Platinum Bimetallic Nanomaterials and Their Application in Staphylococcus Aureus Detection on Paper-Based Devices. Biosens. Bioelectron. 2022, 216, 114669. [Google Scholar] [CrossRef] [PubMed]
- Dadigala, R.; Bandi, R.; Alle, M.; Park, C.-W.; Han, S.-Y.; Kwon, G.-J.; Lee, S.-H. Effective Fabrication of Cellulose Nanofibrils Supported Pd Nanoparticles as a Novel Nanozyme with Peroxidase and Oxidase-like Activities for Efficient Dye Degradation. J. Hazard. Mater. 2022, 436, 129165. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Wang, H.; Chen, J.; Zhang, Y.; Mo, Q.; Zhang, P.; Wang, M.; Liu, H.; Bao, X.; Sun, Y.; et al. Silk-Based Hydrogel Incorporated with Metal-Organic Framework Nanozymes for Enhanced Osteochondral Regeneration. Bioact. Mater. 2023, 20, 221–242. [Google Scholar] [CrossRef] [PubMed]
- Weng, Q.; Hu, X.; Zheng, J.; Xia, F.; Wang, N.; Liao, H.; Liu, Y.; Kim, D.; Liu, J.; Li, F.; et al. Toxicological Risk Assessments of Iron Oxide Nanocluster- and Gadolinium-Based T1MRI Contrast Agents in Renal Failure Rats. ACS Nano 2019, 13, 6801–6812. [Google Scholar] [CrossRef]
- Li, J.; Liu, W.; Wu, X.; Gao, X. Mechanism of PH-Switchable Peroxidase and Catalase-like Activities of Gold, Silver, Platinum and Palladium. Biomaterials 2015, 48, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Sahu, A.; Kwon, I.; Tae, G. Improving Cancer Therapy through the Nanomaterials-Assisted Alleviation of Hypoxia. Biomaterials 2020, 228, 119578. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Jin, Y.; Cui, H.; Yan, X.; Fan, K. Nanozyme-Based Catalytic Theranostics. RSC Adv. 2019, 10, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Liu, B.; Sun, Q.; Feng, L.; He, F.; Yang, P.; Gai, S.; Quan, Z.; Lin, J. Upconverted Metal–Organic Framework Janus Architecture for Near-Infrared and Ultrasound Co-Enhanced High Performance Tumor Therapy. ACS Nano 2021, 15, 12342–12357. [Google Scholar] [CrossRef]
- Sang, Y.; Cao, F.; Li, W.; Zhang, L.; You, Y.; Deng, Q.; Dong, K.; Ren, J.; Qu, X. Bioinspired Construction of a Nanozyme-Based H2O2 Homeostasis Disruptor for Intensive Chemodynamic Therapy. J. Am. Chem. Soc. 2020, 142, 5177–5183. [Google Scholar] [CrossRef]
- Xu, Z.; Chen, J.; Li, Y.; Hu, T.; Fan, L.; Xi, J.; Han, J.; Guo, R. Yolk-Shell Fe3O4@Carbon@Platinum-Chlorin E6 Nanozyme for MRI-Assisted Synergistic Catalytic-Photodynamic-Photothermal Tumor Therapy. J. Colloid Interface Sci. 2022, 628, 1033–1043. [Google Scholar] [CrossRef]
- Wang, M.; Wang, D.; Chen, Q.; Li, C.; Li, Z.; Lin, J. Recent Advances in Glucose-Oxidase-Based Nanocomposites for Tumor Therapy. Small 2019, 15, 1903895. [Google Scholar] [CrossRef]
- Nagvenkar, A.P.; Gedanken, A. Cu 0.89 Zn 0.11 O, A New Peroxidase-Mimicking Nanozyme with High Sensitivity for Glucose and Antioxidant Detection. ACS Appl. Mater. Interfaces 2016, 8, 22301–22308. [Google Scholar] [CrossRef]
- Comotti, M.; Della Pina, C.; Matarrese, R.; Rossi, M. The Catalytic Activity of “Naked” Gold Particles. Angew. Chem. Int. Ed. 2004, 43, 5812–5815. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-Y.; Hsu, Y.-H.; Chen, Y.; Yang, L.-C.; Tseng, S.-C.; Chen, W.-R.; Huang, C.-C.; Wan, D. Robust O2 Supplementation from a Trimetallic Nanozyme-Based Self-Sufficient Complementary System Synergistically Enhances the Starvation/Photothermal Therapy against Hypoxic Tumors. ACS Appl. Mater. Interfaces 2021, 13, 38090–38104. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Lin, H.; Zhang, H.; Yao, H.; Chen, Y.; Shi, J. Nanocatalytic Tumor Therapy by Biomimetic Dual Inorganic Nanozyme-Catalyzed Cascade Reaction. Adv. Sci. 2019, 6, 1801733. [Google Scholar] [CrossRef]
- Yuan, Z.; Liu, X.; Ling, J.; Huang, G.; Huang, J.; Zhu, X.; He, L.; Chen, T. In Situ-Transition Nanozyme Triggered by Tumor Microenvironment Boosts Synergistic Cancer Radio-/Chemotherapy through Disrupting Redox Homeostasis. Biomaterials 2022, 287, 121620. [Google Scholar] [CrossRef]
- Xu, D.; Wu, L.; Yao, H.; Zhao, L. Catalase-Like Nanozymes: Classification, Catalytic Mechanisms, and Their Applications. Small 2022, 18, 2203400. [Google Scholar] [CrossRef]
- Yuan, B.; Chou, H.-L.; Peng, Y.-K. Disclosing the Origin of Transition Metal Oxides as Peroxidase (and Catalase) Mimetics. ACS Appl. Mater. Interfaces 2022, 14, 22728–22736. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Zare, I.; Yan, X.; Fan, K. Protein-protected Metal Nanoclusters: An Emerging Ultra-small Nanozyme. WIREs Nanomed. Nanobiotechnol. 2020, 12, e1602. [Google Scholar] [CrossRef]
- Zhang, R.; Xue, B.; Tao, Y.; Zhao, H.; Zhang, Z.; Wang, X.; Zhou, X.; Jiang, B.; Yang, Z.; Yan, X.; et al. Edge-Site Engineering of Defective Fe–N 4 Nanozymes with Boosted Catalase-Like Performance for Retinal Vasculopathies. Adv. Mater. 2022, 34, 2205324. [Google Scholar] [CrossRef]
- Li, Y.; Yang, L.; Xu, J.; Zhou, H.; Gao, Z.; Song, Y. Pt Nanoparticle-Coupled WO2.72 Nanoplates as Multi-Enzyme Mimetics for Colorimetric Detection and Radical Elimination. Anal. Bioanal. Chem. 2020, 412, 521–530. [Google Scholar] [CrossRef]
- Chen, F.; Song, T. AuPt Bimetallic Nanozymes for Enhanced Glucose Catalytic Oxidase. Front. Chem. 2022, 10, 854516. [Google Scholar] [CrossRef]
- Dong, P.; Wang, W.; Pan, M.; Yu, W.; Liu, Y.; Shi, T.; Hu, J.; Zhou, Y.; Yu, S.; Wang, F.; et al. Cascaded Amplifier Nanoreactor for Efficient Photodynamic Therapy. ACS Appl. Mater. Interfaces 2021, 13, 16075–16083. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Pang, X.; Cheng, Y.; Ming, J.; Xiang, S.; Zhang, C.; Lv, P.; Chu, C.; Chen, X.; Liu, G.; et al. Ultrasound-Switchable Nanozyme Augments Sonodynamic Therapy against Multidrug-Resistant Bacterial Infection. ACS Nano 2020, 14, 2063–2076. [Google Scholar] [CrossRef]
- Singh, N.; NaveenKumar, S.K.; Geethika, M.; Mugesh, G. A Cerium Vanadate Nanozyme with Specific Superoxide Dismutase Activity Regulates Mitochondrial Function and ATP Synthesis in Neuronal Cells. Angew. Chem. Int. Ed. 2021, 60, 3121–3130. [Google Scholar] [CrossRef]
- Korschelt, K.; Ragg, R.; Metzger, C.S.; Kluenker, M.; Oster, M.; Barton, B.; Panthöfer, M.; Strand, D.; Kolb, U.; Mondeshki, M.; et al. Glycine-Functionalized Copper(ii) Hydroxide Nanoparticles with High Intrinsic Superoxide Dismutase Activity. Nanoscale 2017, 9, 3952–3960. [Google Scholar] [CrossRef]
- Korsvik, C.; Patil, S.; Seal, S.; Self, W.T. Superoxide Dismutase Mimetic Properties Exhibited by Vacancy Engineered Ceria Nanoparticles. Chem. Commun. 2007, 1056–1058. [Google Scholar] [CrossRef]
- Landa, P. Positive Effects of Metallic Nanoparticles on Plants: Overview of Involved Mechanisms. Plant Physiol. Biochem. 2021, 161, 12–24. [Google Scholar] [CrossRef] [PubMed]
- Dong, S.; Dong, Y.; Liu, B.; Liu, J.; Liu, S.; Zhao, Z.; Li, W.; Tian, B.; Zhao, R.; He, F.; et al. Guiding Transition Metal-Doped Hollow Cerium Tandem Nanozymes with Elaborately Regulated Multi-Enzymatic Activities for Intensive Chemodynamic Therapy. Adv. Mater. 2022, 34, 2107054. [Google Scholar] [CrossRef] [PubMed]
- Rowe, S.P.; Pomper, M.G. Molecular Imaging in Oncology: Current Impact and Future Directions. CA A Cancer J. Clin. 2022, 72, 333–352. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Bhatnagar, S.; Deschenes, E.; Thurber, G.M. Mechanistic and Quantitative Insight into Cell Surface Targeted Molecular Imaging Agent Design. Sci. Rep. 2016, 6, 25424. [Google Scholar] [CrossRef]
- Zhu, X.; Xu, N.; Zhang, L.; Wang, D.; Zhang, P. Novel Design of Multifunctional Nanozymes Based on Tumor Microenvironment for Diagnosis and Therapy. Eur. J. Med. Chem. 2022, 238, 114456. [Google Scholar] [CrossRef]
- Khramtsov, P.; Kropaneva, M.; Minin, A.; Bochkova, M.; Timganova, V.; Maximov, A.; Puzik, A.; Zamorina, S.; Rayev, M. Prussian Blue Nanozymes with Enhanced Catalytic Activity: Size Tuning and Application in ELISA-like Immunoassay. Nanomaterials 2022, 12, 1630. [Google Scholar] [CrossRef]
- Chang, Y.; Gao, S.; Liu, M.; Liu, J. Designing Signal-on Sensors by Regulating Nanozyme Activity. Anal. Methods 2020, 12, 4708–4723. [Google Scholar] [CrossRef]
- Li, S.; Zhao, X.; Gang, R.; Cao, B.; Wang, H. Doping Nitrogen into Q-Graphene by Plasma Treatment toward Peroxidase Mimics with Enhanced Catalysis. Anal. Chem. 2020, 92, 5152–5157. [Google Scholar] [CrossRef] [PubMed]
- Meng, Y.; Chen, Y.; Zhu, J.; Qi, Y.; Ding, J.; Zhou, W. Polarity Control of DNA Adsorption Enabling the Surface Functionalization of CuO Nanozymes for Targeted Tumor Therapy. Mater. Horiz. 2021, 8, 972–986. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, M.A.; Arora, S.; Prakasam, G.; Calin, G.A.; Syed, M.A. MicroRNA in Lung Cancer: Role, Mechanisms, Pathways and Therapeutic Relevance. Mol. Asp. Med. 2019, 70, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Hu, P.; Wang, R.; Zhou, L.; Chen, L.; Wu, Q.; Han, Y.; El-Toni, A.M.; Zhao, D.; Zhang, F. Near Infrared-Activated Upconversion Nanoprobes for Sensitive Endogenous Zn2+ Detection and Selective On-Demand Photodynamic Therapy. Anal. Chem. 2017, 89, 34923500. [Google Scholar] [CrossRef]
- Hori, S.S.; Tummers, W.S.; Gambhir, S.S. On-Target Probes for Early Detection. Nat. Biomed. Eng. 2017, 1, 0062. [Google Scholar] [CrossRef]
- Ren, X.; Chen, D.; Wang, Y.; Li, H.; Zhang, Y.; Chen, H.; Li, X.; Huo, M. Nanozymes-Recent Development and Biomedical Applications. J. Nanobiotechnol. 2022, 20, 92. [Google Scholar] [CrossRef]
- Ma, Q.; Liu, Y.; Zhu, H.; Zhang, L.; Liao, X. Nanozymes in Tumor Theranostics. Front. Oncol. 2021, 11, 666017. [Google Scholar] [CrossRef]
- Wang, P.; Min, D.; Chen, G.; Li, M.; Tong, L.; Cao, Y. Inorganic Nanozymes: Prospects for Disease Treatments and Detection Applications. Front. Chem. 2021, 9, 773285. [Google Scholar] [CrossRef]
- Wang, Z.; Li, Z.; Sun, Z.; Wang, S.; Ali, Z.; Zhu, S.; Liu, S.; Ren, Q.; Sheng, F.; Wang, B.; et al. Visualization Nanozyme Based on Tum–or Microenvironment “Unlocking” for Intensive Combination Therapy of Breast Cancer. Sci. Adv. 2020, 6, eabc8733. [Google Scholar] [CrossRef]
- Yang, F.; Hu, S.; Zhang, Y.; Cai, X.; Huang, Y.; Wang, F.; Wen, S.; Teng, G.; Gu, N. A Hydrogen Peroxide-Responsive O2 Nanogenerator for Ultrasound and Magnetic-Resonance Dual Modality Imaging. Adv. Mater. 2012, 24, 5205–5211. [Google Scholar] [CrossRef]
- Huang, Y.; Hsu, J.C.; Koo, H.; Cormode, D.P. Repurposing Ferumoxytol: Diagnostic and Therapeutic Applications of an FDA-Approved Nanoparticle. Theranostics 2022, 12, 796–816. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Lan, N.; Zhang, Y.; Zeng, W.; He, H.; Liu, X. Benzobisthiadiazole and Its Derivative-Based Semiconducting Polymer Nanoparticles for Second Near-Infrared Photoacoustic Imaging. Front. Chem. 2022, 10, 842712. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; Fan, K.; Zhang, R.; Zhang, D.; Zhang, J.; Gao, Y.; Zhang, T.; Li, W.; Li, J.; Yan, X.; et al. Precise Visual Distinction of Brain Glioma from Normal Tissues via Targeted Photoacoustic and Fluorescence Navigation. Nanomed. Nanotechnol. Biol. Med. 2020, 27, 102204. [Google Scholar] [CrossRef]
- Khan, S.; Sharifi, M.; Bloukh, S.H.; Edis, Z.; Siddique, R.; Falahati, M. In Vivo Guiding Inorganic Nanozymes for Biosensing and Therapeutic Potential in Cancer, Inflammation and Microbial Infections. Talanta 2021, 224, 121805. [Google Scholar] [CrossRef]
- Que, E.L.; Chang, C.J. Responsive Magnetic Resonance Imaging Contrast Agents as Chemical Sensors for Metals in Biology and Medicine. Chem. Soc. Rev. 2010, 39, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Toth, G.B.; Varallyay, C.G.; Horvath, A.; Bashir, M.R.; Choyke, P.L.; Daldrup-Link, H.E.; Dosa, E.; Finn, J.P.; Gahramanov, S.; Harisinghani, M.; et al. Current and Potential Imaging Applications of Ferumoxytol for Magnetic Resonance Imaging. Kidney Int. 2017, 92, 47–66. [Google Scholar] [CrossRef] [PubMed]
- Angelovski, G.; Tóth, É. Strategies for Sensing Neurotransmitters with Responsive MRI Contrast Agents. Chem. Soc. Rev. 2017, 46, 324–336. [Google Scholar] [CrossRef] [PubMed]
- Dadfar, S.M.; Roemhild, K.; Drude, N.I.; von Stillfried, S.; Knüchel, R.; Kiessling, F.; Lammers, T. Iron Oxide Nanoparticles: Diagnostic, Therapeutic and Theranostic Applications. Adv. Drug Deliv. Rev. 2019, 138, 302–325. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhao, X.; Liu, X.; Cheng, K.; Han, X.; Zhang, Y.; Min, H.; Liu, G.; Xu, J.; Shi, J.; et al. A Bioinspired Nanoprobe with Multilevel Responsive T1—Weighted MR Signal-Amplification Illuminates Ultrasmall Metastases. Adv. Mater. 2020, 32, 1906799. [Google Scholar] [CrossRef]
- Yue, H.; Yuan, L.; Zhang, W.; Zhang, S.; Wei, W.; Ma, G. Macrophage Responses to the Physical Burden of Cell-Sized Particles. J. Mater. Chem. B 2018, 6, 393–400. [Google Scholar] [CrossRef]
- Zhou, C.; Zhang, L.; Sun, T.; Zhang, Y.; Liu, Y.; Gong, M.; Xu, Z.; Du, M.; Liu, Y.; Liu, G.; et al. Activatable NIR-II Plasmonic Nanotheranostics for Efficient Photoacoustic Imaging and Photothermal Cancer Therapy. Adv. Mater. 2021, 33, 2006532. [Google Scholar] [CrossRef]
- Chen, Y.-S.; Zhao, Y.; Yoon, S.J.; Gambhir, S.S.; Emelianov, S. Miniature Gold Nanorods for Photoacoustic Molecular Imaging in the Second Near-Infrared Optical Window. Nat. Nanotechnol. 2019, 14, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Knox, H.J.; Chan, J. Acoustogenic Probes: A New Frontier in Photoacoustic Imaging. Accounts Chem. Res. 2018, 51, 2897–2905. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Bhattarai, P.; Dai, Z.; Chen, X. Photothermal Therapy and Photoacoustic Imaging via Nanotheranostics in Fighting Cancer. Chem. Soc. Rev. 2019, 48, 2053–2108. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Stein, E.W.; Ashkenazi, S.; Wang, L.V. Nanoparticles for Photoacoustic Imaging. WIREs Nanomed. Nanobiotechnol. 2009, 1, 360–368. [Google Scholar] [CrossRef]
- Biffi, S.; Voltan, R.; Rampazzo, E.; Prodi, L.; Zauli, G.; Secchiero, P. Applications of Nanoparticles in Cancer Medicine and beyond: Optical and Multimodal in Vivo Imaging, Tissue Targeting and Drug Delivery. Expert Opin. Drug Deliv. 2015, 12, 1837–1849. [Google Scholar] [CrossRef]
- Serkova, N.J.; Glunde, K.; Haney, C.R.; Farhoud, M.; De Lille, A.; Redente, E.F.; Simberg, D.; Westerly, D.C.; Griffin, L.; Mason, R.P. Preclinical Applications of Multi-Platform Imaging in Animal Models of Cancer. Cancer Res. 2021, 81, 1189–1200. [Google Scholar] [CrossRef]
- Leary, J.; Key, J. Nanoparticles for Multimodal in Vivo Imaging in Nanomedicine. IJN 2014, 711. [Google Scholar] [CrossRef]
- Ametamey, S.M.; Honer, M.; Schubiger, P.A. Molecular Imaging with PET. Chem. Rev. 2008, 108, 1501–1516. [Google Scholar] [CrossRef]
- Ge, J.; Zhang, Q.; Zeng, J.; Gu, Z.; Gao, M. Radiolabeling Nanomaterials for Multimodality Imaging: New Insights into Nuclear Medicine and Cancer Diagnosis. Biomaterials 2020, 228, 119553. [Google Scholar] [CrossRef]
- You, Q.; Zhang, K.; Liu, J.; Liu, C.; Wang, H.; Wang, M.; Ye, S.; Gao, H.; Lv, L.; Wang, C.; et al. Persistent Regulation of Tumor Hypoxia Microenvironment via a Bioinspired Pt-Based Oxygen Nanogenerator for Multimodal Imaging-Guided Synergistic Phototherapy. Adv. Sci. 2020, 7, 1903341. [Google Scholar] [CrossRef]
- Wu, M.; Shu, J. Multimodal Molecular Imaging: Current Status and Future Directions. Contrast Media Mol. Imaging 2018, 2018, 1382183. [Google Scholar] [CrossRef] [PubMed]
- Vermeulen, I.; Isin, E.M.; Barton, P.; Cillero-Pastor, B.; Heeren, R.M.A. Multimodal Molecular Imaging in Drug Discovery and Development. Drug Discov. Today 2022, 27, 2086–2099. [Google Scholar] [CrossRef]
- Li, X.; Lovell, J.F.; Yoon, J.; Chen, X. Clinical Development and Potential of Photothermal and Photodynamic Therapies for Cancer. Nat. Rev. Clin. Oncol. 2020, 17, 657–674. [Google Scholar] [CrossRef] [PubMed]
- Shin, T.-H.; Choi, Y.; Kim, S.; Cheon, J. Recent Advances in Magnetic Nanoparticle-Based Multi-Modal Imaging. Chem. Soc. Rev. 2015, 44, 4501–4516. [Google Scholar] [CrossRef]
- Qin, R.; Feng, Y.; Ding, D.; Chen, L.; Li, S.; Deng, H.; Chen, S.; Han, Z.; Sun, W.; Chen, H. Fe-Coordinated Carbon Nanozyme Dots as Peroxidase-Like Nanozymes and Magnetic Resonance Imaging Contrast Agents. ACS Appl. Bio Mater. 2021, 4, 5520–5528. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Zhao, F.; Gao, W.; Yang, X.; Ju, Y.; Zhao, L.; Guo, W.; Xie, J.; Liang, X.; Tao, X.; et al. Magnetic Reactive Oxygen Species Nanoreactor for Switchable Magnetic Resonance Imaging Guided Cancer Therapy Based on PH-Sensitive Fe5C2@Fe3O4 Nanoparticles. ACS Nano 2019, 13, 10002–10014. [Google Scholar] [CrossRef]
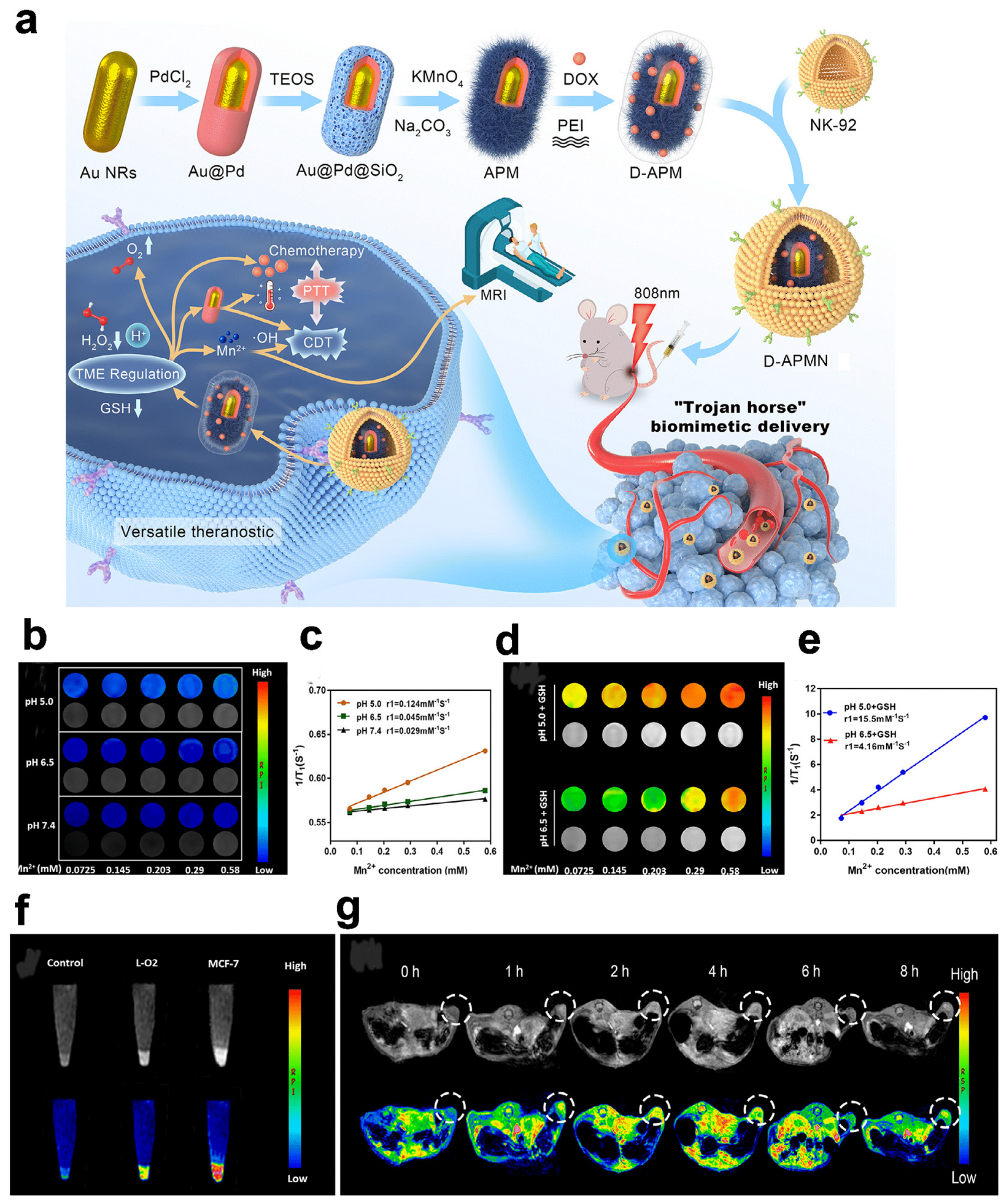
- Huang, Q.; Pan, Y.; Wang, M.; Liu, Z.; Chen, H.; Wang, J.; Zhao, Z.; Zhang, Y. Tumor Microenvironment–Responsive Versatile “Trojan Horse” Theranostic Nanoplatform for Magnetic Resonance Imaging–Guided Multimodal Synergistic Antitumor Treatment. Acta Biomater. 2022, 147, 270–286. [Google Scholar] [CrossRef]
- Zhang, D.-Y.; Liu, H.; Zhu, K.S.; He, T.; Younis, M.R.; Yang, C.; Lei, S.; Wu, J.; Lin, J.; Qu, J.; et al. Prussian Blue-Based Theranostics for Ameliorating Acute Kidney Injury. J. Nanobiotechnol. 2021, 19, 266. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Xia, F.; Zhang, L.; Fang, C.; Lee, J.; Gong, L.; Gao, J.; Ling, D.; Li, F. A ROS-Sensitive Nanozyme-Augmented Photoacoustic Nanoprobe for Early Diagnosis and Therapy of Acute Liver Failure. Adv. Mater. 2022, 34, 2108348. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Chang, M.; Chen, Q.; Wang, D.; Li, C.; Hou, Z.; Lin, J.; Jin, D.; Xing, B. Au2Pt-PEG-Ce6 Nanoformulation with Dual Nanozyme Activities for Synergistic Chemodynamic Therapy/Phototherapy. Biomaterials 2020, 252, 120093. [Google Scholar] [CrossRef]
- Liang, S.; Deng, X.; Chang, Y.; Sun, C.; Shao, S.; Xie, Z.; Xiao, X.; Ma, P.; Zhang, H.; Cheng, Z.; et al. Intelligent Hollow Pt-CuS Janus Architecture for Synergistic Catalysis-Enhanced Sonodynamic and Photothermal Cancer Therapy. Nano Lett. 2019, 19, 4134–4145. [Google Scholar] [CrossRef]
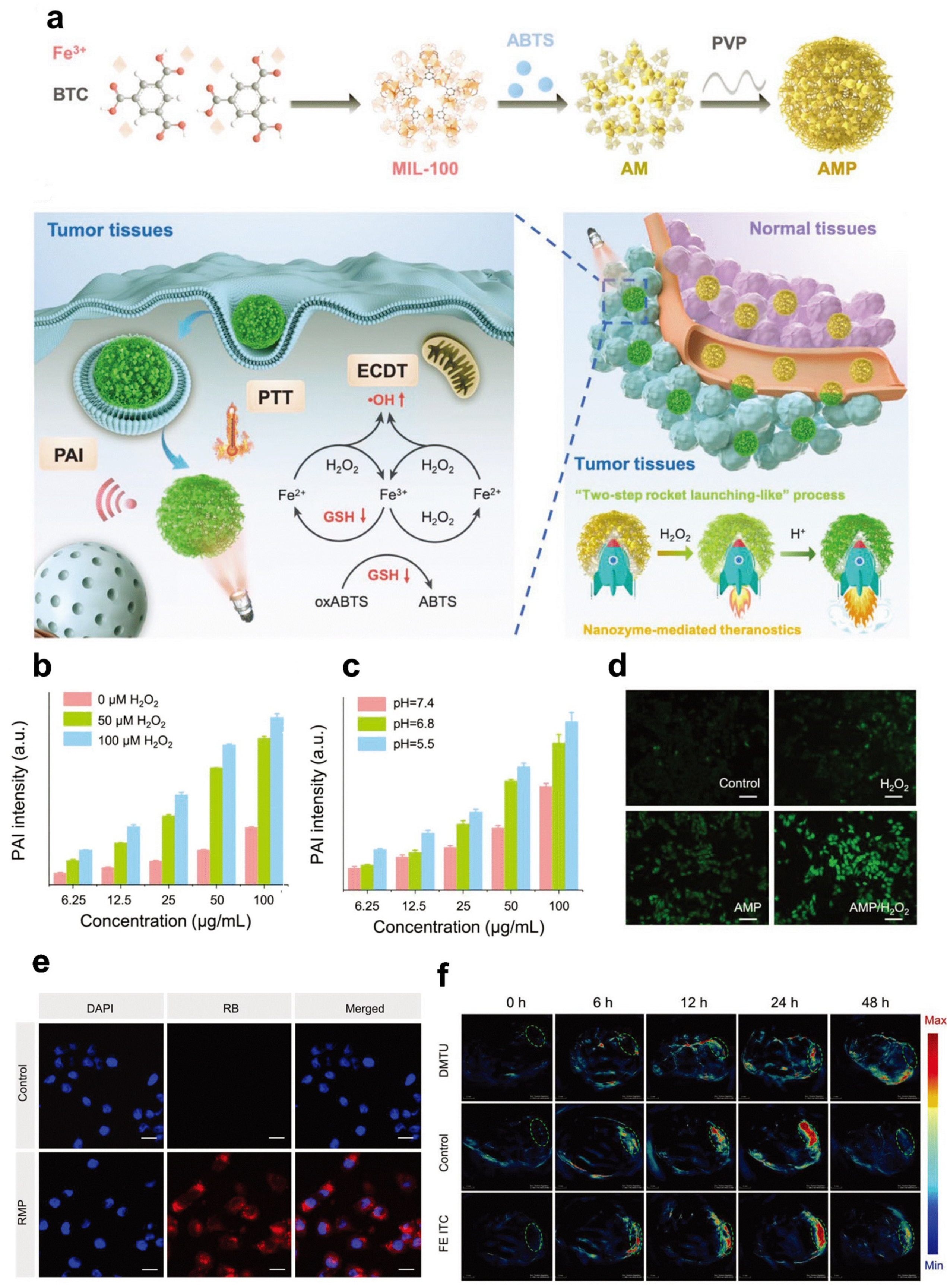
- Ding, H.; Cai, Y.; Gao, L.; Liang, M.; Miao, B.; Wu, H.; Liu, Y.; Xie, N.; Tang, A.; Fan, K.; et al. Exosome-like Nanozyme Vesicles for H2O2-Responsive Catalytic Photoacoustic Imaging of Xenograft Nasopharyngeal Carcinoma. Nano Lett. 2019, 19, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Lin, L.; Zhang, Y.; Wang, Y.; Sheng, S.; Xu, C.; Tian, H.; Chen, X. A Tumor-Microenvironment-Activated Nanozyme-Mediated Theranostic Nanoreactor for Imaging-Guided Combined Tumor Therapy. Adv. Mater. 2019, 31, 1902885. [Google Scholar] [CrossRef]
- Zheng, Z.; Jia, Z.; Qin, Y.; Dai, R.; Chen, X.; Ma, Y.; Xie, X.; Zhang, R. All-in-One Zeolite–Carbon-Based Nanotheranostics with Adjustable NIR-II Window Photoacoustic/Fluorescence Imaging Performance for Precise NIR-II Photothermal-Synergized Catalytic Antitumor Therapy. Small 2021, 17, 2103252. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, K.; Xing, G.; Dong, X.; Zhu, D.; Yang, W.; Mei, L.; Lv, F. Nanozyme-Laden Intelligent Macrophage EXPRESS Amplifying Cancer Photothermal-Starvation Therapy by Responsive Stimulation. Mater. Today Bio 2022, 16, 100421. [Google Scholar] [CrossRef]
- Zhang, A.; Pan, S.; Zhang, Y.; Chang, J.; Cheng, J.; Huang, Z.; Li, T.; Zhang, C.; de la Fuentea, J.M.; Zhang, Q.; et al. Carbon-Gold Hybrid Nanoprobes for Real-Time Imaging, Photothermal/Photodynamic and Nanozyme Oxidative Therapy. Theranostics 2019, 9, 3443–3458. [Google Scholar] [CrossRef]
- Dan, Q.; Yuan, Z.; Zheng, S.; Ma, H.; Luo, W.; Zhang, L.; Su, N.; Hu, D.; Sheng, Z.; Li, Y. Gold Nanoclusters-Based NIR-II Photosensitizers with Catalase-like Activity for Boosted Photodynamic Therapy. Pharmaceutics 2022, 14, 1645. [Google Scholar] [CrossRef]
- Li, L.; Yang, Z.; Fan, W.; He, L.; Cui, C.; Zou, J.; Tang, W.; Jacobson, O.; Wang, Z.; Niu, G.; et al. In Situ Polymerized Hollow Mesoporous Organosilica Biocatalysis Nanoreactor for Enhancing ROS-Mediated Anticancer Therapy. Adv. Funct. Mater. 2020, 30, 1907716. [Google Scholar] [CrossRef]
- Yuan, H.; Wilks, M.Q.; Normandin, M.D.; El Fakhri, G.; Kaittanis, C.; Josephson, L. Heat-Induced Radiolabeling and Fluorescence Labeling of Feraheme Nanoparticles for PET/SPECT Imaging and Flow Cytometry. Nat. Protoc. 2018, 13, 392–412. [Google Scholar] [CrossRef]
- Jia, T.; Li, D.; Du, J.; Fang, X.; Gerasimov, V.; Ågren, H.; Chen, G. A Bimodal Type of AgPd Plasmonic Blackbody Nanozyme with Boosted Catalytic Efficacy and Synergized Photothermal Therapy for Efficacious Tumor Treatment in the Second Biological Window. J. Nanobiotechnol. 2022, 20, 424. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Zhang, W.; Chen, Q.; Hou, J.; Wang, J.; Zhong, Y.; Wang, X.; Jiang, W.; Ran, H.; Guo, D. Multifunctional Nanozyme for Multimodal Imaging-Guided Enhanced Sonodynamic Therapy by Regulating the Tumor Microenvironment. Nanoscale 2021, 13, 14049–14066. [Google Scholar] [CrossRef] [PubMed]
- Gong, F.; Yang, N.; Wang, Y.; Zhuo, M.; Zhao, Q.; Wang, S.; Li, Y.; Liu, Z.; Chen, Q.; Cheng, L. Oxygen-Deficient Bimetallic Oxide FeWO X Nanosheets as Peroxidase-Like Nanozyme for Sensing Cancer via Photoacoustic Imaging. Small 2020, 16, 2003496. [Google Scholar] [CrossRef] [PubMed]
- Cao, C.; Zou, H.; Yang, N.; Li, H.; Cai, Y.; Song, X.; Shao, J.; Chen, P.; Mou, X.; Wang, W.; et al. Fe3O4/Ag/Bi2MoO6 Photoactivatable Nanozyme for Self-Replenishing and Sustainable Cascaded Nanocatalytic Cancer Therapy. Adv. Mater. 2021, 33, 2106996. [Google Scholar] [CrossRef] [PubMed]
- Zhou, I.Y.; Catalano, O.A.; Caravan, P. Advances in Functional and Molecular MRI Technologies in Chronic Liver Diseases. J. Hepatol. 2020, 73, 1241–1254. [Google Scholar] [CrossRef]
- Zijlstra, L.E.; Trompet, S.; Jukema, J.W.; Kroft, L.J.M.; de Bresser, J.; van Osch, M.J.P.; Hammer, S.; Witjes, M.-N.; van Buren, M.; Mooijaart, S.P. Association of Cardiovascular Structure and Function with Cerebrovascular Changes and Cognitive Function in Older Patients with End-Stage Renal Disease. Aging 2020, 12, 1496–1511. [Google Scholar] [CrossRef]
- Datta, A.; Raymond, K.N. Gd-Hydroxypyridinone (HOPO)-Based High-Relaxivity Magnetic Resonance Imaging (MRI) Contrast Agents. Accounts Chem. Res. 2009, 42, 938–947. [Google Scholar] [CrossRef]
- Sukstanskii, A.L.; Yablonskiy, D.A. Microscopic Theory of Spin–Spin and Spin–Lattice Relaxation of Bound Protons in Cellular and Myelin Membranes—A Lateral Diffusion Model (LDM). Magn. Reson. Med. 2023, 89, 370–383. [Google Scholar] [CrossRef]
- Aime, S.; Castelli, D.D.; Crich, S.G.; Gianolio, E.; Terreno, E. Pushing the Sensitivity Envelope of Lanthanide-Based Magnetic Resonance Imaging (MRI) Contrast Agents for Molecular Imaging Applications. Accounts Chem. Res. 2009, 42, 822–831. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Dong, H.; Tao, Q.; Ye, C.; Yu, M.; Li, J.; Zhou, H.; Yang, S.; Ding, G.; Xie, X. Enhancing the Magnetic Relaxivity of MRI Contrast Agents via the Localized Superacid Microenvironment of Graphene Quantum Dots. Biomaterials 2020, 250, 120056. [Google Scholar] [CrossRef] [PubMed]
- Yoo, D.; Lee, J.-H.; Shin, T.-H.; Cheon, J. Theranostic Magnetic Nanoparticles. Accounts Chem. Res. 2011, 44, 863–874. [Google Scholar] [CrossRef]
- Wahsner, J.; Gale, E.M.; Rodríguez-Rodríguez, A.; Caravan, P. Chemistry of MRI Contrast Agents: Current Challenges and New Frontiers. Chem. Rev. 2019, 119, 957–1057. [Google Scholar] [CrossRef] [PubMed]
- Levine, D.; McDonald, R.J.; Kressel, H.Y. Gadolinium Retention After Contrast-Enhanced MRI. JAMA 2018, 320, 1853. [Google Scholar] [CrossRef]
- Verwilst, P.; Park, S.; Yoon, B.; Kim, J.S. Recent Advances in Gd-Chelate Based Bimodal Optical/MRI Contrast Agents. Chem. Soc. Rev. 2015, 44, 1791–1806. [Google Scholar] [CrossRef]
- Sieber, M.A.; Pietsch, H.; Walter, J.; Haider, W.; Frenzel, T.; Weinmann, H.-J. A Preclinical Study to Investigate the Development of Nephrogenic Systemic Fibrosis: A Possible Role for Gadolinium-Based Contrast Media. Investig. Radiol. 2008, 43, 65–75. [Google Scholar] [CrossRef]
- Mitka, M. MRI Contrast Agents May Pose Risk for Patients With Kidney Disease. JAMA 2007, 297, 249. [Google Scholar] [CrossRef]
- Choi, S.H.; Kim, S.Y.; Lim, Y.-S. Selection of MRI Contrast Agent and Diagnostic Criteria for HCC to Maximize the Advantages of Contrast Agents. J. Hepatol. 2020, 73, 714–715. [Google Scholar] [CrossRef]
- Gale, E.M.; Atanasova, I.P.; Blasi, F.; Ay, I.; Caravan, P. A Manganese Alternative to Gadolinium for MRI Contrast. J. Am. Chem. Soc. 2015, 137, 15548–15557. [Google Scholar] [CrossRef]
- Lu, Z.-R.; Laney, V.; Li, Y. Targeted Contrast Agents for Magnetic Resonance Molecular Imaging of Cancer. Accounts Chem. Res. 2022, 55, 2833–2847. [Google Scholar] [CrossRef]
- Shen, Z.; Chen, T.; Ma, X.; Ren, W.; Zhou, Z.; Zhu, G.; Zhang, A.; Liu, Y.; Song, J.; Li, Z.; et al. Multifunctional Theranostic Nanoparticles Based on Exceedingly Small Magnetic Iron Oxide Nanoparticles for T1—Weighted Magnetic Resonance Imaging and Chemotherapy. ACS Nano 2017, 11, 10992–11004. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, H. Fe3O4-Based Nanotheranostics for Magnetic Resonance Imaging-Synergized Multifunctional Cancer Management. Nanomedicine 2019, 14, 1493–1512. [Google Scholar] [CrossRef] [PubMed]
- Peters, J.A. Relaxivity of Manganese Ferrite Nanoparticles. Prog. Nucl. Magn. Reson. Spectrosc. 2020, 120–121, 72–94. [Google Scholar] [CrossRef] [PubMed]
- Petralia, G.; Summers, P.E.; Agostini, A.; Ambrosini, R.; Cianci, R.; Cristel, G.; Calistri, L.; Colagrande, S. Dynamic Contrast-Enhanced MRI in Oncology: How We Do It. Radiol. Med. 2020, 125, 1288–1300. [Google Scholar] [CrossRef]
- Lee, N.; Yoo, D.; Ling, D.; Cho, M.H.; Hyeon, T.; Cheon, J. Iron Oxide Based Nanoparticles for Multimodal Imaging and Magnetoresponsive Therapy. Chem. Rev. 2015, 115, 10637–10689. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Yu, X.; Qian, Y.; Chen, W.; Shen, J. Multifunctional Magnetic Iron Oxide Nanoparticles: An Advanced Platform for Cancer Theranostics. Theranostics 2020, 10, 6278–6309. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, X.; Li, X.; Qiao, S.; Huang, G.; Hermann, D.M.; Doeppner, T.R.; Zeng, M.; Liu, W.; Xu, G.; et al. A Co-Doped Fe3O4 Nanozyme Shows Enhanced Reactive Oxygen and Nitrogen Species Scavenging Activity and Ameliorates the Deleterious Effects of Ischemic Stroke. ACS Appl. Mater. Interfaces 2021, 13, 46213–46224. [Google Scholar] [CrossRef]
- Jin, R.; Lin, B.; Li, D.; Ai, H. Superparamagnetic Iron Oxide Nanoparticles for MR Imaging and Therapy: Design Considerations and Clinical Applications. Curr. Opin. Pharmacol. 2014, 18, 18–27. [Google Scholar] [CrossRef]
- Russell, E.; Dunne, V.; Russell, B.; Mohamud, H.; Ghita, M.; McMahon, S.J.; Butterworth, K.T.; Schettino, G.; McGarry, C.K.; Prise, K.M. Impact of Superparamagnetic Iron Oxide Nanoparticles on in Vitro and in Vivo Radiosensitisation of Cancer Cells. Radiat. Oncol. 2021, 16, 104. [Google Scholar] [CrossRef]
- Gale, E.M.; Wey, H.-Y.; Ramsay, I.; Yen, Y.-F.; Sosnovik, D.E.; Caravan, P. A Manganese-Based Alternative to Gadolinium: Contrast-Enhanced MR Angiography, Excretion, Pharmacokinetics, and Metabolism. Radiology 2018, 286, 865–872. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Zhu, Q.; Zeng, Y.; Zeng, Q.; Chen, X.; Zhan, Y. Manganese Oxide Nanoparticles As MRI Contrast Agents In Tumor Multimodal Imaging And Therapy. IJN 2019, 14, 8321–8344. [Google Scholar] [CrossRef]
- Ragg, R.; Schilmann, A.M.; Korschelt, K.; Wieseotte, C.; Kluenker, M.; Viel, M.; Völker, L.; Preiß, S.; Herzberger, J.; Frey, H.; et al. Intrinsic Superoxide Dismutase Activity of MnO Nanoparticles Enhances the Magnetic Resonance Imaging Contrast. J. Mater. Chem. B 2016, 4, 7423–7428. [Google Scholar] [CrossRef] [PubMed]
- Hsu, B.Y.W.; Kirby, G.; Tan, A.; Seifalian, A.M.; Li, X.; Wang, J. Relaxivity and Toxicological Properties of Manganese Oxide Nanoparticles for MRI Applications. RSC Adv. 2016, 6, 45462–45474. [Google Scholar] [CrossRef]
- Sobańska, Z.; Roszak, J.; Kowalczyk, K.; Stępnik, M. Applications and Biological Activity of Nanoparticles of Manganese and Manganese Oxides in In Vitro and In Vivo Models. Nanomaterials 2021, 11, 1084. [Google Scholar] [CrossRef]
- Zhu, D.; Zhu, X.-H.; Ren, S.-Z.; Lu, Y.-D.; Zhu, H.-L. Manganese Dioxide (MnO2) Based Nanomaterials for Cancer Therapies and Theranostics. J. Drug Target. 2021, 29, 911–924. [Google Scholar] [CrossRef]
- Yang, G.; Ji, J.; Liu, Z. Multifunctional MnO2 Nanoparticles for Tumor Microenvironment Modulation and Cancer Therapy. WIREs Nanomed. Nanobiotechnol. 2021, 13, e1720. [Google Scholar] [CrossRef] [PubMed]
- He, T.; Jiang, C.; He, J.; Zhang, Y.; He, G.; Wu, J.; Lin, J.; Zhou, X.; Huang, P. Manganese-Dioxide-Coating-Instructed Plasmonic Modulation of Gold Nanorods for Activatable Duplex-Imaging-Guided NIR-II Photothermal-Chemodynamic Therapy. Adv. Mater. 2021, 33, 2008540. [Google Scholar] [CrossRef]
- Fan, W.; Bu, W.; Shen, B.; He, Q.; Cui, Z.; Liu, Y.; Zheng, X.; Zhao, K.; Shi, J. Intelligent MnO2 Nanosheets Anchored with Upconversion Nanoprobes for Concurrent PH-/H2O2 -Responsive UCL Imaging and Oxygen-Elevated Synergetic Therapy. Adv. Mater. 2015, 27, 4155–4161. [Google Scholar] [CrossRef]
- Zhao, M.; Xie, M.; Guo, J.; Feng, W.; Xu, Y.; Liu, X.; Liu, S.; Zhao, Q. Facile Phototherapeutic Nanoplatform by Integrating a Multifunctional Polymer and MnO2 for Enhancing Tumor Synergistic Therapy. Adv. Healthc. Mater. 2019, 8, 1900414. [Google Scholar] [CrossRef]
- Lan, G.; Ni, K.; Lin, W. Nanoscale Metal–Organic Frameworks for Phototherapy of Cancer. Coord. Chem. Rev. 2019, 379, 65–81. [Google Scholar] [CrossRef]
- Chen, Y.; Li, Z.; Pan, P.; Hu, J.; Cheng, S.; Zhang, X. Tumor-Microenvironment-Triggered Ion Exchange of a Metal–Organic Framework Hybrid for Multimodal Imaging and Synergistic Therapy of Tumors. Adv. Mater. 2020, 32, 2001452. [Google Scholar] [CrossRef] [PubMed]
- Kale, S.; Burga, R.; Sweeney, E.; Zun, Z.; Sze, R.; Tuesca, A.; Subramony, A.; Fernandes, R. Composite Iron Oxide—Prussian Blue Nanoparticles for Magnetically Guided T1-Weighted Magnetic Resonance Imaging and Photothermal Therapy of Tumors. IJN 2017, 12, 6413–6424. [Google Scholar] [CrossRef]
- Qin, Z.; Chen, B.; Huang, X.; Mao, Y.; Li, Y.; Yang, F.; Gu, N. Magnetic Internal Heating-Induced High Performance Prussian Blue Nanoparticle Preparation and Excellent Catalytic Activity. Dalton Trans. 2019, 48, 17169–17173. [Google Scholar] [CrossRef]
- Ma, X.; Hao, J.; Wu, J.; Li, Y.; Cai, X.; Zheng, Y. Prussian Blue Nanozyme as a Pyroptosis Inhibitor Alleviates Neurodegeneration. Adv. Mater. 2022, 34, 2106723. [Google Scholar] [CrossRef]
- Fan, S.; Jiang, X.; Yang, M.; Wang, X. Sensitive Colorimetric Assay for the Determination of Alkaline Phosphatase Activity Utilizing Nanozyme Based on Copper Nanoparticle-Modified Prussian Blue. Anal. Bioanal. Chem. 2021, 413, 3955–3963. [Google Scholar] [CrossRef]
- Upputuri, P.K.; Pramanik, M. Recent Advances in Photoacoustic Contrast Agents for in Vivo Imaging. WIREs Nanomed. Nanobiotechnol. 2020, 12, e1618. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Teng, L.; Yin, B.; Meng, H.; Yin, X.; Huan, S.; Song, G.; Zhang, X.-B. Chemical Design of Activatable Photoacoustic Probes for Precise Biomedical Applications. Chem. Rev. 2022, 122, 6850–6918. [Google Scholar] [CrossRef]
- Fu, Q.; Zhu, R.; Song, J.; Yang, H.; Chen, X. Photoacoustic Imaging: Contrast Agents and Their Biomedical Applications. Adv. Mater. 2018, 31, 1805875. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Pu, K. Development of Organic Semiconducting Materials for Deep-Tissue Optical Imaging, Phototherapy and Photoactivation. Chem. Soc. Rev. 2019, 48, 38–71. [Google Scholar] [CrossRef]
- Jiang, Y.; Pu, K. Advanced Photoacoustic Imaging Applications of Near-Infrared Absorbing Organic Nanoparticles. Small 2017, 13, 1700710. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Swartchick, C.B.; Chan, J. Targeted Contrast Agents and Activatable Probes for Photoacoustic Imaging of Cancer. Chem. Soc. Rev. 2022, 51, 829–868. [Google Scholar] [CrossRef] [PubMed]
- Ju, Y.; Zhang, H.; Yu, J.; Tong, S.; Tian, N.; Wang, Z.; Wang, X.; Su, X.; Chu, X.; Lin, J.; et al. Monodisperse Au–Fe2C Janus Nanoparticles: An Attractive Multifunctional Material for Triple-Modal Imaging-Guided Tumor Photothermal Therapy. ACS Nano 2017, 11, 9239–9248. [Google Scholar] [CrossRef] [PubMed]
- Mu, X.; Tang, Y.; Wu, F.; Ma, H.; Huang, S.; Liang, M.; Yang, J.; Lu, Y.; Zhou, X.; Li, Z. A Simple Small Molecule with Synergistic Passive and Active Dual-Targeting Effects for Imaging-Guided Photothermal Cancer Therapy. ACS Appl. Mater. Interfaces 2021, 13, 36958–36966. [Google Scholar] [CrossRef]
- Wu, Z.; Zhou, J.; Nkanga, C.I.; Jin, Z.; He, T.; Borum, R.M.; Yim, W.; Zhou, J.; Cheng, Y.; Xu, M.; et al. One-Step Supramolecular Multifunctional Coating on Plant Virus Nanoparticles for Bioimaging and Therapeutic Applications. ACS Appl. Mater. Interfaces 2022, 14, 13692–13702. [Google Scholar] [CrossRef]
- Li, C.; Cheng, Y.; Li, D.; An, Q.; Zhang, W.; Zhang, Y.; Fu, Y. Antitumor Applications of Photothermal Agents and Photothermal Synergistic Therapies. IJMS 2022, 23, 7909. [Google Scholar] [CrossRef] [PubMed]
- Duan, Q.; Wang, J.; Zhang, B.; Wang, X.; Xue, J.; Zhang, W.; Sang, S. Polydopamine Coated Au-Pt Nanorods: Enhanced Photothermal Properties and Efficient Reactive Oxygen Scavengers. Colloids Surf. B Biointerfaces 2022, 210, 112247. [Google Scholar] [CrossRef]
- Xia, Y.; Li, W.; Cobley, C.M.; Chen, J.; Xia, X.; Zhang, Q.; Yang, M.; Cho, E.C.; Brown, P.K. Gold Nanocages: From Synthesis to Theranostic Applications. Accounts Chem. Res. 2011, 44, 914–924. [Google Scholar] [CrossRef]
- Jiang, T.; Song, J.; Zhang, W.; Wang, H.; Li, X.; Xia, R.; Zhu, L.; Xu, X. Au–Ag@Au Hollow Nanostructure with Enhanced Chemical Stability and Improved Photothermal Transduction Efficiency for Cancer Treatment. ACS Appl. Mater. Interfaces 2015, 7, 21985–21994. [Google Scholar] [CrossRef]
- Li, J.; Zhang, C.; Gong, S.; Li, X.; Yu, M.; Qian, C.; Qiao, H.; Sun, M. A Nanoscale Photothermal Agent Based on a Metal-Organic Coordination Polymer as a Drug-Loading Framework for Effective Combination Therapy. Acta Biomater. 2019, 94, 435–446. [Google Scholar] [CrossRef]
- Luo, Y.; Qiao, B.; Zhang, P.; Yang, C.; Cao, J.; Yuan, X.; Ran, H.; Wang, Z.; Hao, L.; Cao, Y.; et al. TME-Activatable Theranostic Nanoplatform with ATP Burning Capability for Tumor Sensitization and Synergistic Therapy. Theranostics 2020, 10, 6987–7001. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.; Zhang, Y.; Lin, J.; Huang, P. Nanomaterials for Photoacoustic Imaging in the Second Near-Infrared Window. Biomater. Sci. 2019, 7, 472–479. [Google Scholar] [CrossRef] [PubMed]
- Pirovano, G.; Roberts, S.; Kossatz, S.; Reiner, T. Optical Imaging Modalities: Principles and Applications in Preclinical Research and Clinical Settings. J. Nucl. Med. 2020, 61, 1419–1427. [Google Scholar] [CrossRef]
- Qi, M.; Pan, H.; Shen, H.; Xia, X.; Wu, C.; Han, X.; He, X.; Tong, W.; Wang, X.; Wang, Q. Nanogel Multienzyme Mimics Synthesized by Biocatalytic ATRP and Metal Coordination for Bioresponsive Fluorescence Imaging. Angew. Chem. Int. Ed. 2020, 59, 11748–11753. [Google Scholar] [CrossRef] [PubMed]
- Zhen, X.; Jiang, X. Polymer-based Activatable Optical Probes for Tumor Fluorescence and Photoacoustic Imaging. WIREs Nanomed. Nanobiotechnol. 2020, 12, e1593. [Google Scholar] [CrossRef]
- Douma, K.; Megens, R.T.A.; van Zandvoort, M.A.M.J. Optical Molecular Imaging of Atherosclerosis Using Nanoparticles: Shedding New Light on the Darkness. WIREs Nanomed. Nanobiotechnol. 2011, 3, 376–388. [Google Scholar] [CrossRef]
- Li, B.; Zhao, M.; Lin, J.; Huang, P.; Chen, X. Management of Fluorescent Organic/Inorganic Nanohybrids for Biomedical Applications in the NIR-II Region. Chem. Soc. Rev. 2022, 51, 7692–7714. [Google Scholar] [CrossRef]
- Wu, L.; Zou, H.; Wang, H.; Zhang, S.; Liu, S.; Jiang, Y.; Chen, J.; Li, Y.; Shao, M.; Zhang, R.; et al. Update on the Development of Molecular Imaging and Nanomedicine in China: Optical Imaging. WIREs Nanomed. Nanobiotechnol. 2021, 13, e1660. [Google Scholar] [CrossRef] [PubMed]
- Son, J.; Yi, G.; Yoo, J.; Park, C.; Koo, H.; Choi, H.S. Light-Responsive Nanomedicine for Biophotonic Imaging and Targeted Therapy. Adv. Drug Deliv. Rev. 2019, 138, 133–147. [Google Scholar] [CrossRef]
- Yuan, H.; Jiang, A.; Fang, H.; Chen, Y.; Guo, Z. Optical Properties of Natural Small Molecules and Their Applications in Imaging and Nanomedicine. Adv. Drug Deliv. Rev. 2021, 179, 113917. [Google Scholar] [CrossRef]
- Ni, D.; Ehlerding, E.B.; Cai, W. Multimodality Imaging Agents with PET as the Fundamental Pillar. Angew. Chem. Int. Ed. 2019, 58, 2570–2579. [Google Scholar] [CrossRef] [PubMed]
- Pellico, J.; Gawne, P.J.; de Rosales, R.T.M. Radiolabelling of Nanomaterials for Medical Imaging and Therapy. Chem. Soc. Rev. 2021, 50, 3355–3423. [Google Scholar] [CrossRef]
- Goel, S.; England, C.G.; Chen, F.; Cai, W. Positron Emission Tomography and Nanotechnology: A Dynamic Duo for Cancer Theranostics. Adv. Drug Deliv. Rev. 2017, 113, 157–176. [Google Scholar] [CrossRef]
- Gawne, P.J.; Man, F.; Blower, P.J.; de Rosales, R.T.M. Direct Cell Radiolabeling for in Vivo Cell Tracking with PET and SPECT Imaging. Chem. Rev. 2022, 122, 10266–10318. [Google Scholar] [CrossRef]
- Chen, D.; Yang, D.; Dougherty, C.A.; Lu, W.; Wu, H.; He, X.; Cai, T.; Van Dort, M.E.; Ross, B.D.; Hong, H. In Vivo Targeting and Positron Emission Tomography Imaging of Tumor with Intrinsically Radioactive Metal–Organic Frameworks Nanomaterials. ACS Nano 2017, 11, 4315–4327. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Cai, W.; Chen, X. Positron Emission Tomography Imaging Using Radiolabeled Inorganic Nanomaterials. Accounts Chem. Res. 2015, 48, 286–294. [Google Scholar] [CrossRef]
- Carregal-Romero, S.; Miguel-Coello, A.B.; Martínez-Parra, L.; Martí-Mateo, Y.; Hernansanz-Agustín, P.; Fernández-Afonso, Y.; Plaza-García, S.; Gutiérrez, L.; del Mar Muñoz-Hernández, M.; Carrillo-Romero, J.; et al. Ultrasmall Manganese Ferrites for In Vivo Catalase Mimicking Activity and Multimodal Bioimaging. Small 2022, 18, 2106570. [Google Scholar] [CrossRef]
- Tang, W.; Fan, W.; Zhang, W.; Yang, Z.; Li, L.; Wang, Z.; Chiang, Y.; Liu, Y.; Deng, L.; He, L.; et al. Wet/Sono-Chemical Synthesis of Enzymatic Two-Dimensional MnO2 Nanosheets for Synergistic Catalysis-Enhanced Phototheranostics. Adv. Mater. 2019, 31, 1900401. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Kim, J.; Heo, I.; Kim, S.J.; Jang, S.; Lee, H.-Y.; Jang, K.-S.; Yang, C.-S.; Lee, Y.; Yoo, W.C.; et al. One-Pot Bifunctionalization of Silica Nanoparticles Conjugated with Bioorthogonal Linkers: Application in Dual-Modal Imaging. Biomater. Sci. 2022, 10, 3540–3546. [Google Scholar] [CrossRef]
- Boehm, K.M.; Khosravi, P.; Vanguri, R.; Gao, J.; Shah, S.P. Harnessing Multimodal Data Integration to Advance Precision Oncology. Nat. Rev. Cancer 2022, 22, 114–126. [Google Scholar] [CrossRef]
- Wu, Y.; Zeng, F.; Zhao, Y.; Wu, S. Emerging Contrast Agents for Multispectral Optoacoustic Imaging and Their Biomedical Applications. Chem. Soc. Rev. 2021, 50, 7924–7940. [Google Scholar] [CrossRef] [PubMed]
- Bax, J.J.; Di Carli, M.; Narula, J.; Delgado, V. Multimodality Imaging in Ischaemic Heart Failure. Lancet 2019, 393, 1056–1070. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.R.; Gambhir, S.S. Nanomaterials for In Vivo Imaging. Chem. Rev. 2017, 117, 901–986. [Google Scholar] [CrossRef] [PubMed]
- Fan, W.; Yung, B.; Huang, P.; Chen, X. Nanotechnology for Multimodal Synergistic Cancer Therapy. Chem. Rev. 2017, 117, 13566–13638. [Google Scholar] [CrossRef] [PubMed]
- Lo, P.-C.; Rodríguez-Morgade, M.S.; Pandey, R.K.; Ng, D.K.P.; Torres, T.; Dumoulin, F. The Unique Features and Promises of Phthalocyanines as Advanced Photosensitisers for Photodynamic Therapy of Cancer. Chem. Soc. Rev. 2020, 49, 1041–1056. [Google Scholar] [CrossRef]
- Cheng, Y.; Kong, X.; Chang, Y.; Feng, Y.; Zheng, R.; Wu, X.; Xu, K.; Gao, X.; Zhang, H. Spatiotemporally Synchronous Oxygen Self-Supply and Reactive Oxygen Species Production on Z-Scheme Heterostructures for Hypoxic Tumor Therapy. Adv. Mater. 2020, 32, 1908109. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Xu, B.; Li, S.; Wu, Q.; Lu, M.; Han, A.; Liu, H. A Photoresponsive Nanozyme for Synergistic Catalytic Therapy and Dual Phototherapy. Small 2021, 17, 2007090. [Google Scholar] [CrossRef]


| Enzyme-mimicking Activity | Imaging Modalities | Disease | Refs | |
|---|---|---|---|---|
| Fe3O4 | POD | MRI | Inflammatory cardiovascular disease | [97] |
| Fe-CDs | POD | MRI | Hepatocarcinoma, Glioblastoma | [98] |
| Fe5C2@Fe3O4 | POD, CAT | MRI | Breast cancer | [99] |
| APMN NPs | POD, CAT | MRI/PAI | Breast cancer | [100] |
| PBNZs | POD, CAT, SOD | MRI/PAI | Acute renal injury | [101] |
| RSPN | SOD | PAI | Acute liver failure | [102] |
| Au2Pt-PEG-Ce6 | POD, CAT | PAI/CT | Cervical cancer | [103] |
| Pt-CuS Janus | CAT | PAI | Colon cancer | [104] |
| GQDzyme | POD | PAI | Nasopharyngeal carcinoma | [105] |
| AMP NRs | POD | PAI | Breast cancer | [106] |
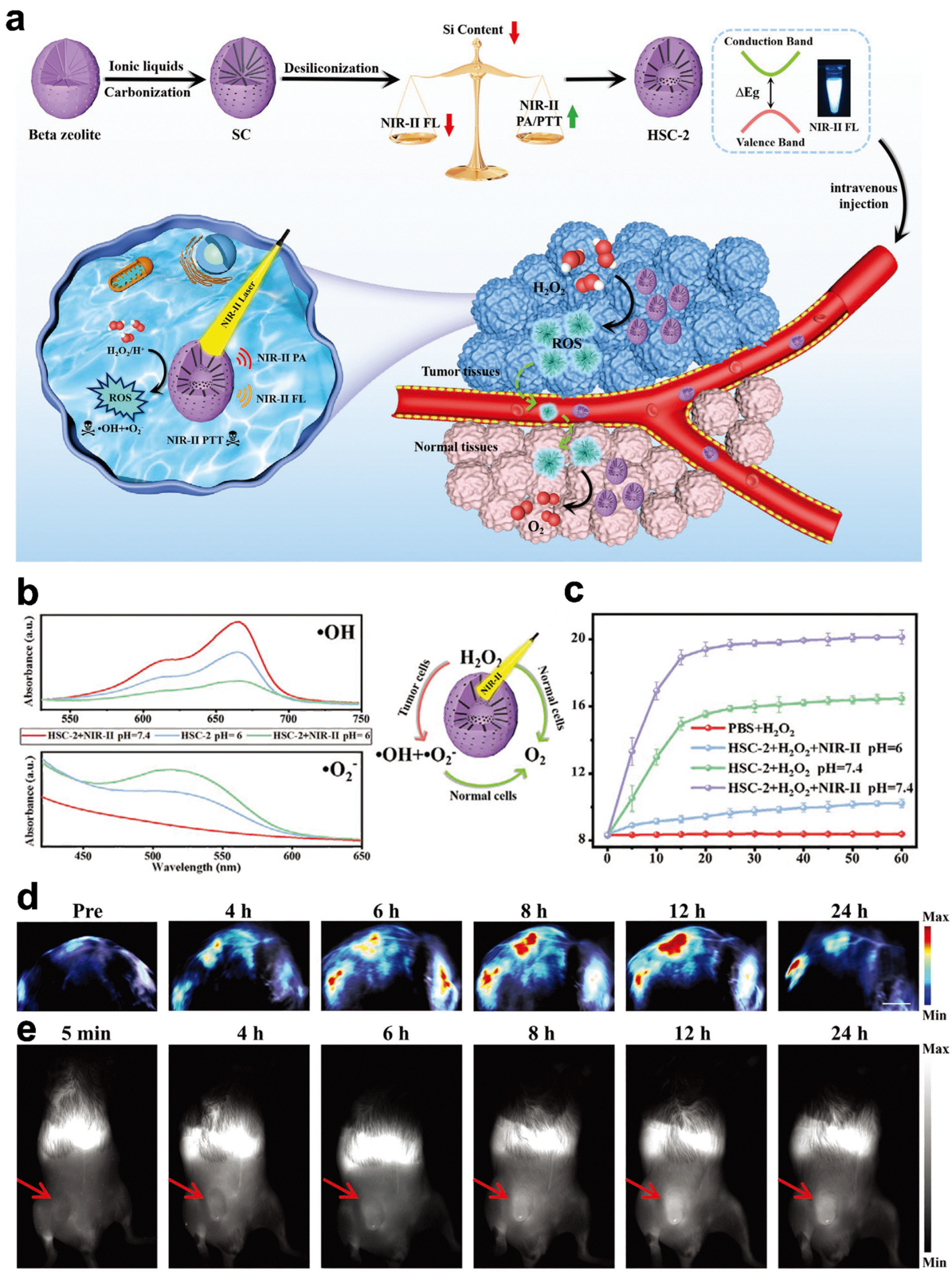
| HSC-2 | POD, CAT | PAI/FLI | Breast cancer | [107] |
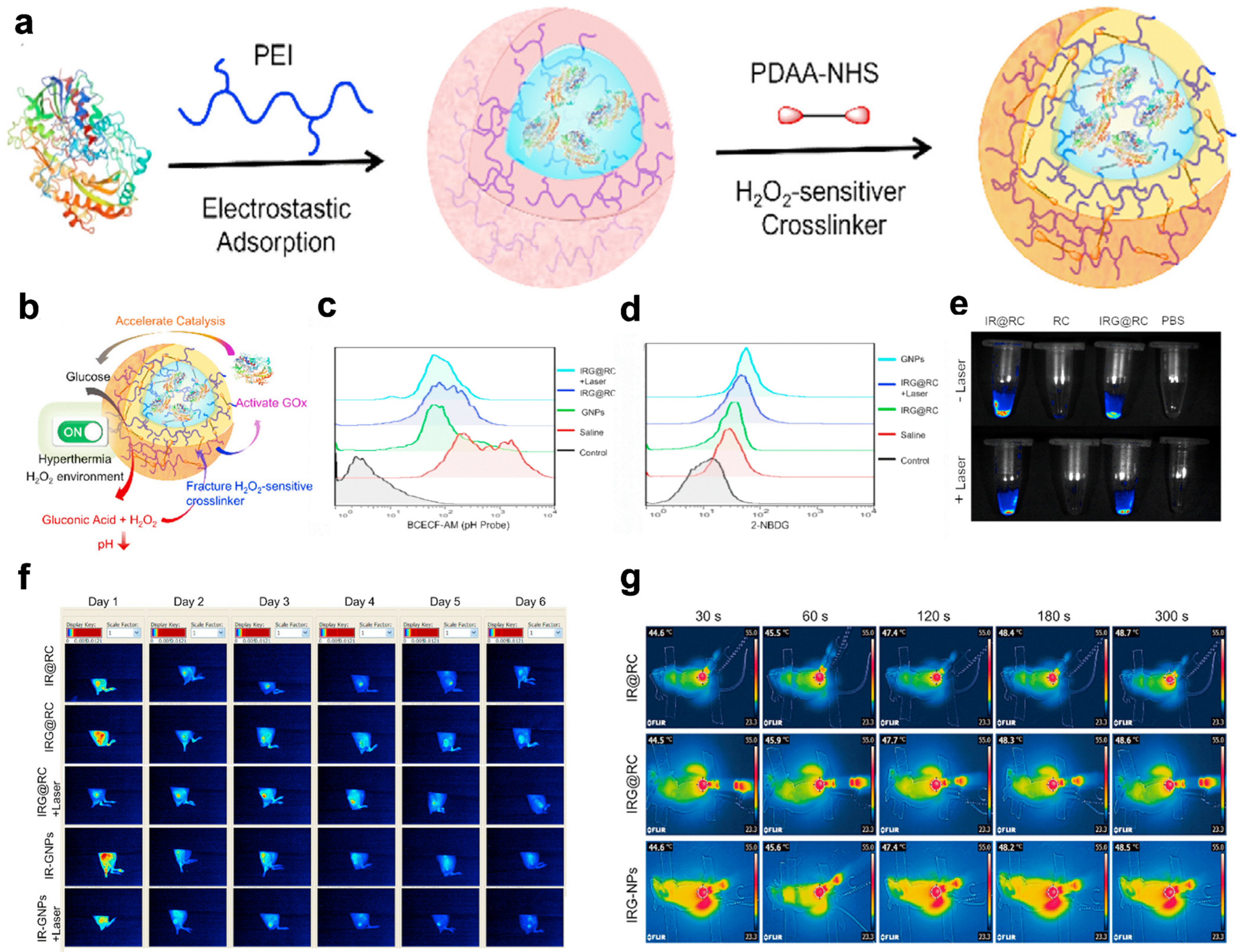
| GNPs | POD, CAT | FLI/PAI | Breast cancer | [108] |
| OMCAPs@rBSA-FA@IR780 | CAT | FLI | Gastric carcinoma | [109] |
| BSA@Au | CAT | FLI | Breast cancer | [110] |
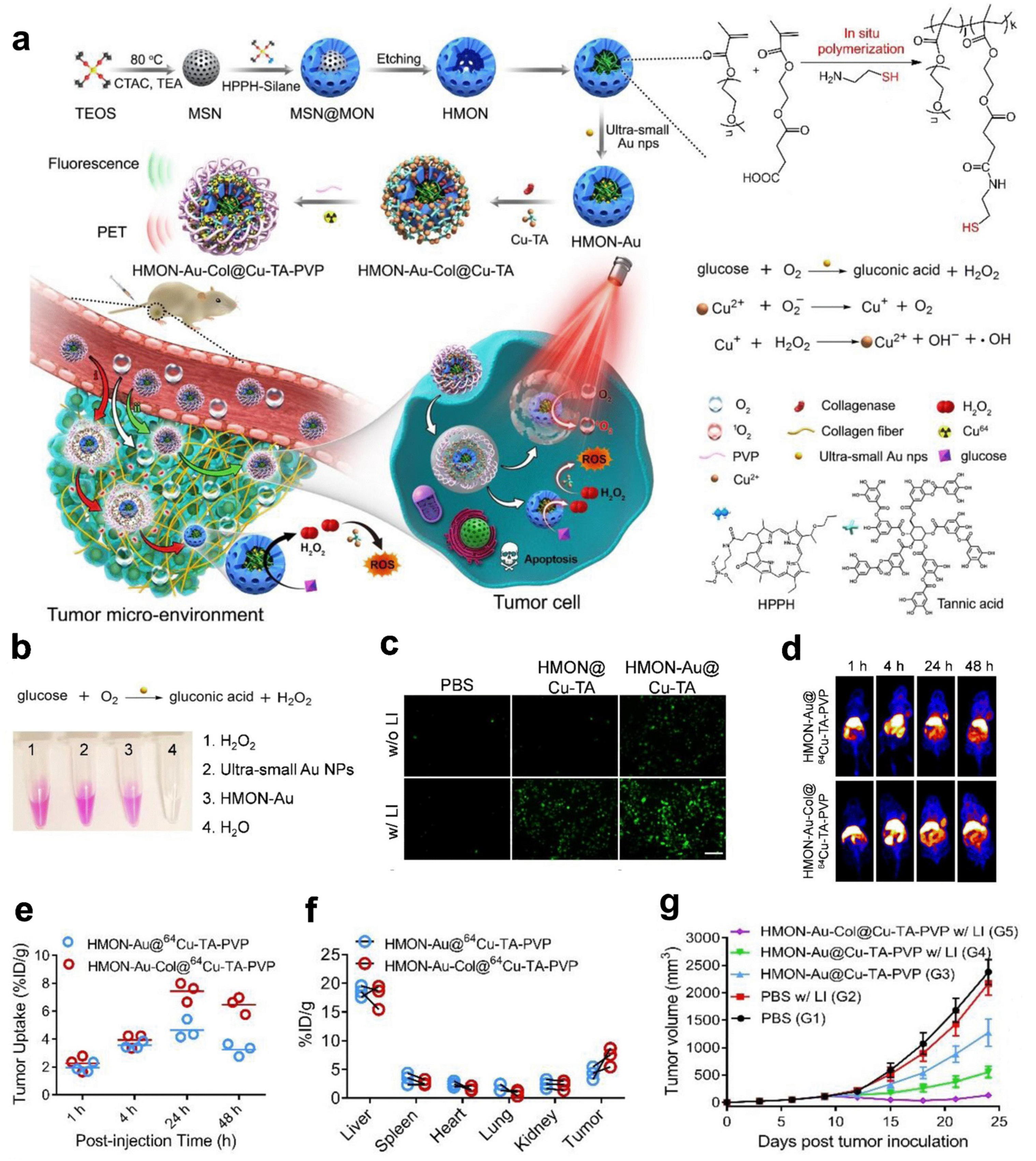
| HMON-Au-col@Cu-TA | XOD | PET/FLI | Pancreatic ductal adenocarcinoma | [111] |
| FHNPs | POD, CAT | PET/SPECT | Inflammation | [112] |
| AgPd PB | CAT | PAI/FLI/CT | Cervical cancer | [113] |
| Ang-IR780-MnO2-PLGA | POD, CAT | PAI/FLI/MRI | Glioblastoma | [114] |
| FeWOX NSs | POD | PAI/MRI/CT | Breast cancer | [115] |
| Fe3O4/Ag/Bi2MoO6 NP | POD, CAT, SOD | PAI/MRI/PTI | Breast cancer | [116] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luo, Q.; Shao, N.; Zhang, A.-C.; Chen, C.-F.; Wang, D.; Luo, L.-P.; Xiao, Z.-Y. Smart Biomimetic Nanozymes for Precise Molecular Imaging: Application and Challenges. Pharmaceuticals 2023, 16, 249. https://doi.org/10.3390/ph16020249
Luo Q, Shao N, Zhang A-C, Chen C-F, Wang D, Luo L-P, Xiao Z-Y. Smart Biomimetic Nanozymes for Precise Molecular Imaging: Application and Challenges. Pharmaceuticals. 2023; 16(2):249. https://doi.org/10.3390/ph16020249
Chicago/Turabian StyleLuo, Qiao, Ni Shao, Ai-Chen Zhang, Chun-Fang Chen, Duo Wang, Liang-Ping Luo, and Ze-Yu Xiao. 2023. "Smart Biomimetic Nanozymes for Precise Molecular Imaging: Application and Challenges" Pharmaceuticals 16, no. 2: 249. https://doi.org/10.3390/ph16020249
APA StyleLuo, Q., Shao, N., Zhang, A.-C., Chen, C.-F., Wang, D., Luo, L.-P., & Xiao, Z.-Y. (2023). Smart Biomimetic Nanozymes for Precise Molecular Imaging: Application and Challenges. Pharmaceuticals, 16(2), 249. https://doi.org/10.3390/ph16020249






