Novel Anthranilic Acid Hybrids—An Alternative Weapon against Inflammatory Diseases
Abstract
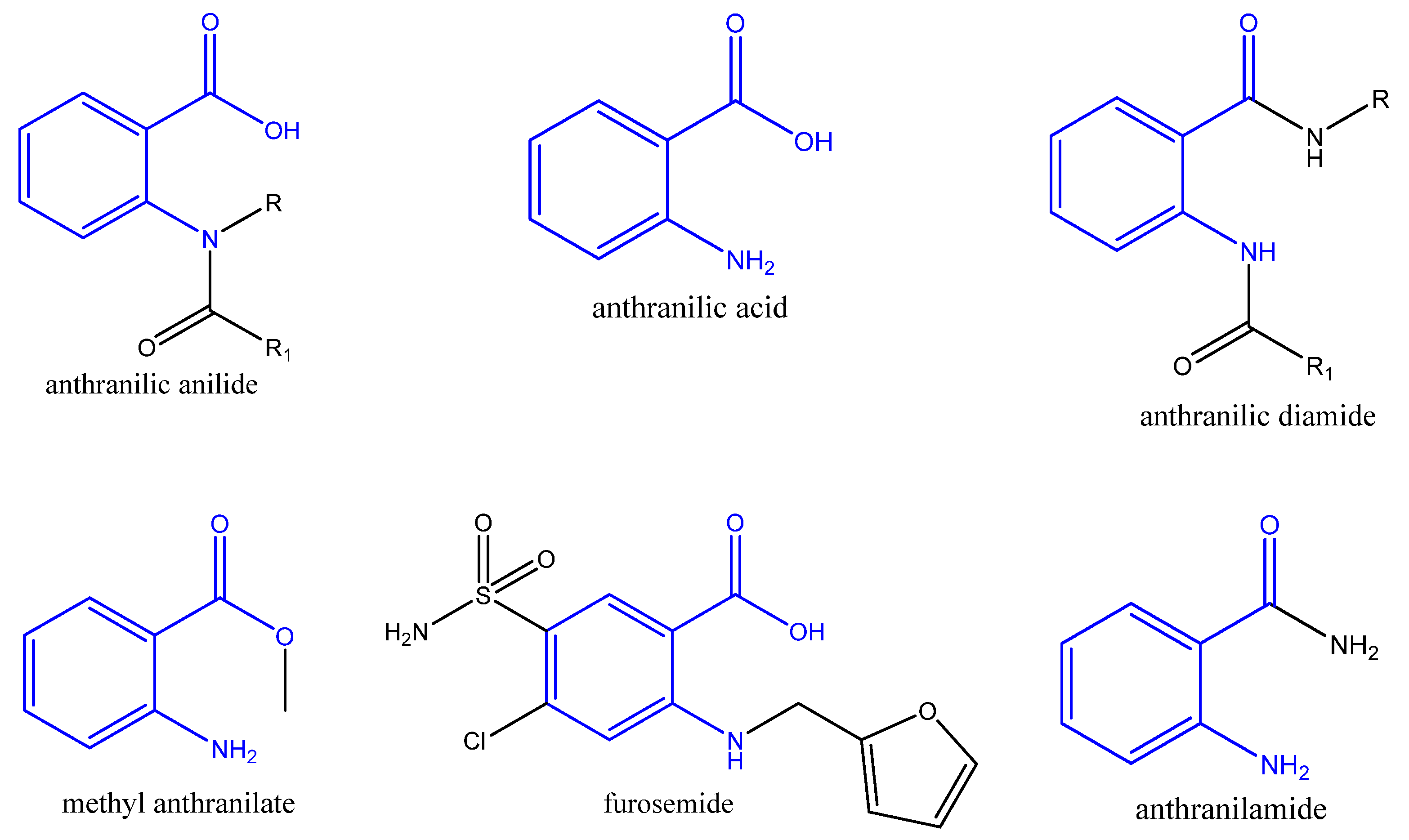
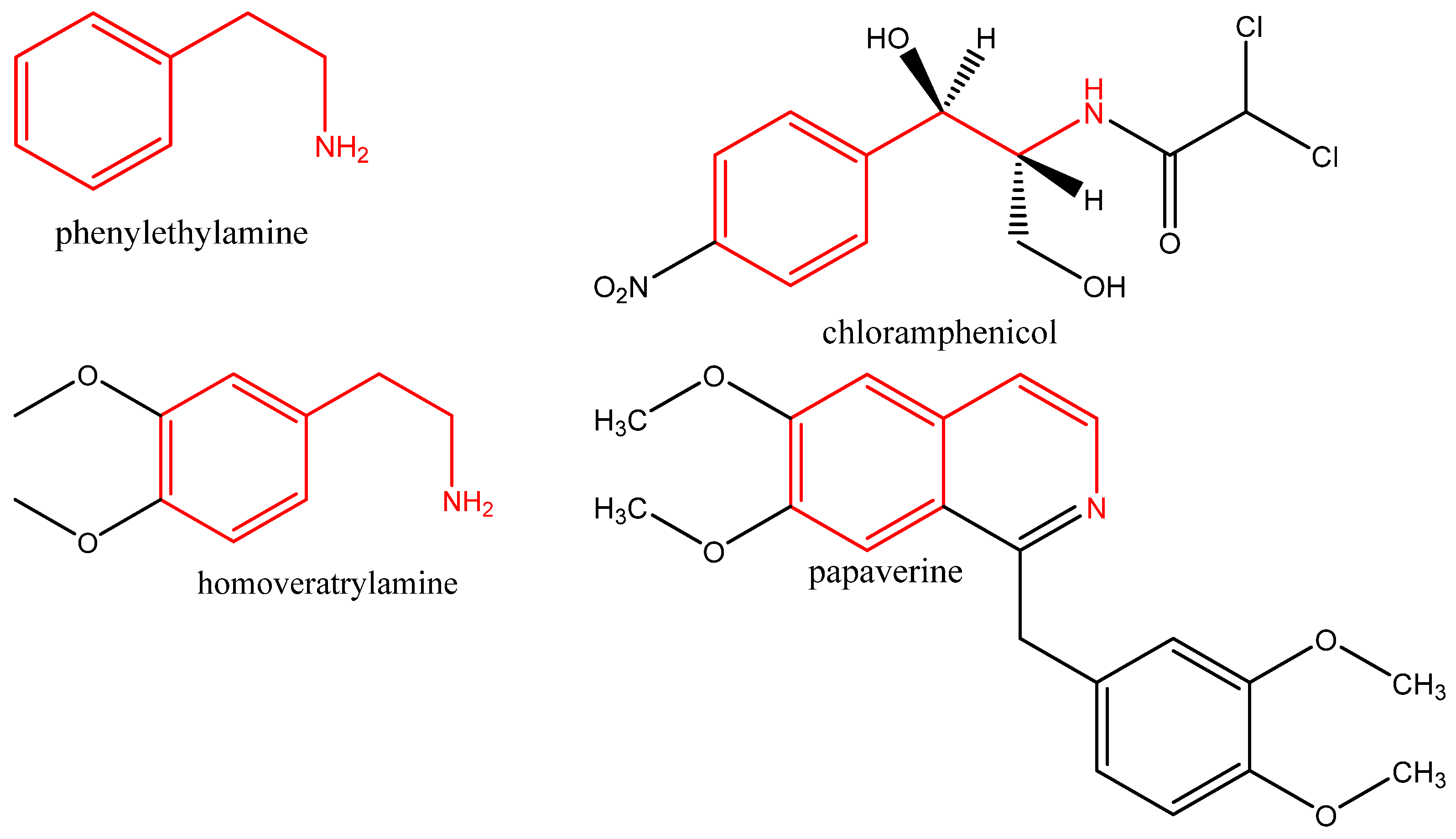
1. Introduction
2. Results and Discussion
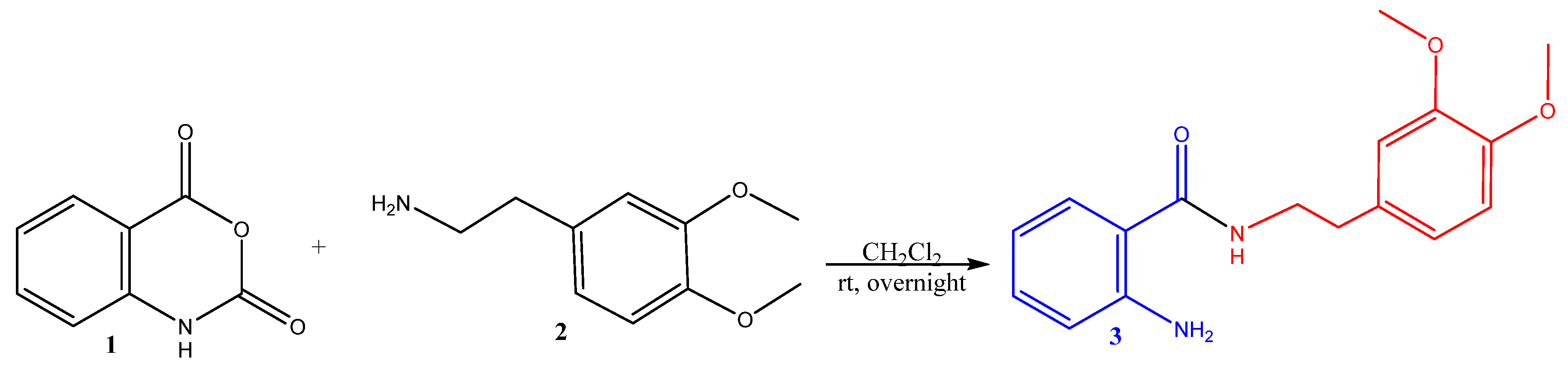
2.1. Synthesis of Hybrid Molecules
2.2. In Silico Predictions
2.3. Biological Evaluation of the Hybrid Molecules
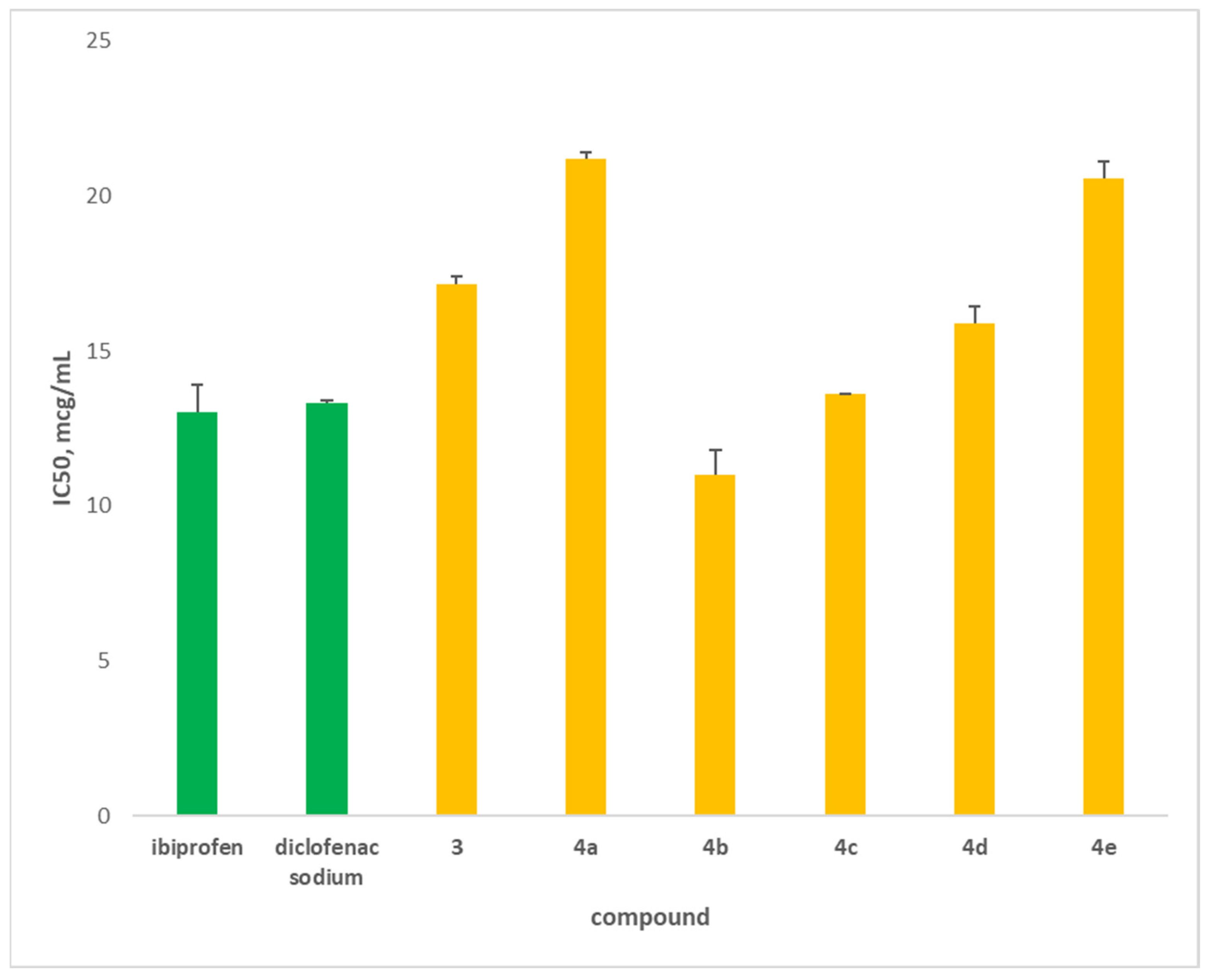
2.3.1. Anti-Inflammatory Activity
In Vitro Inhibition of Albumin Denaturation
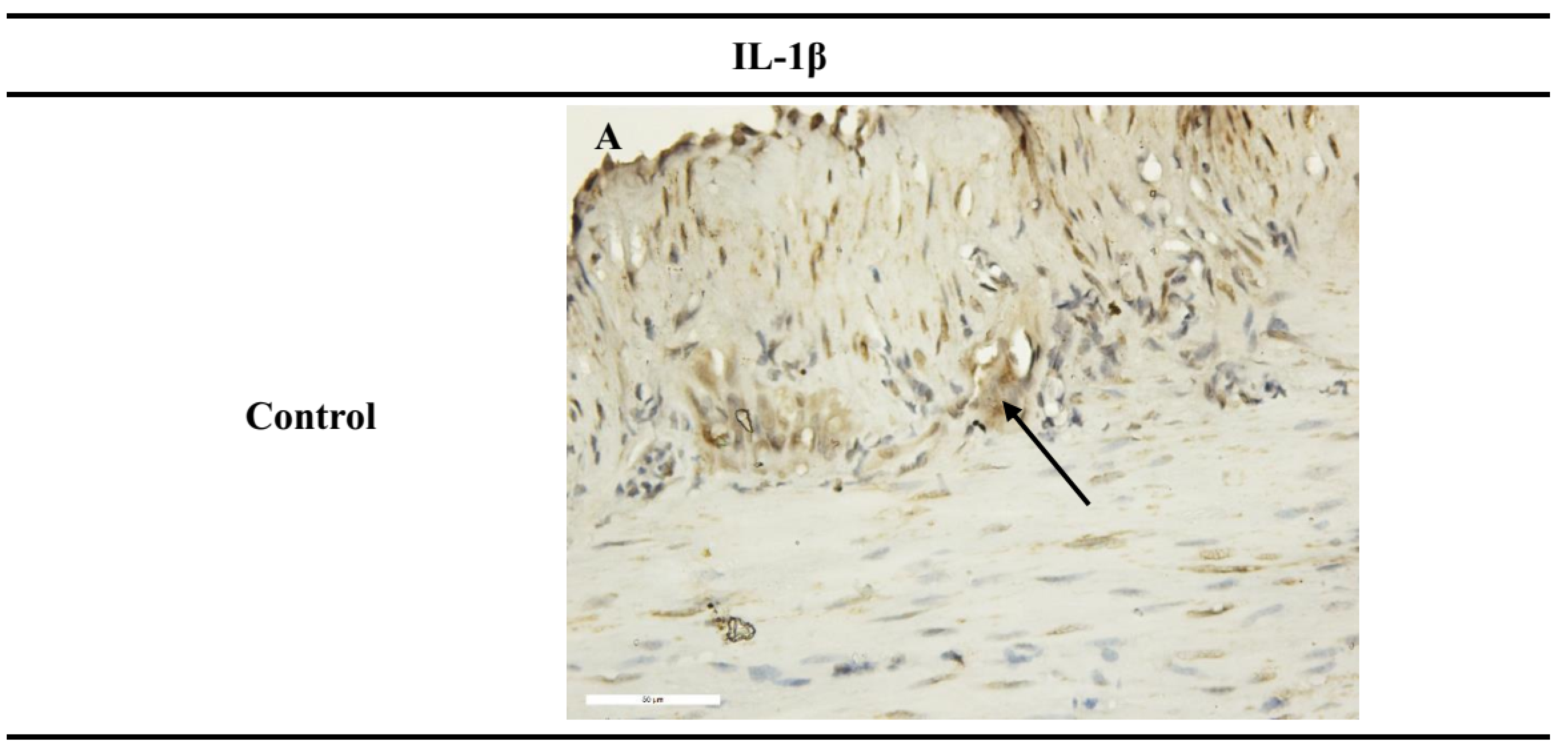
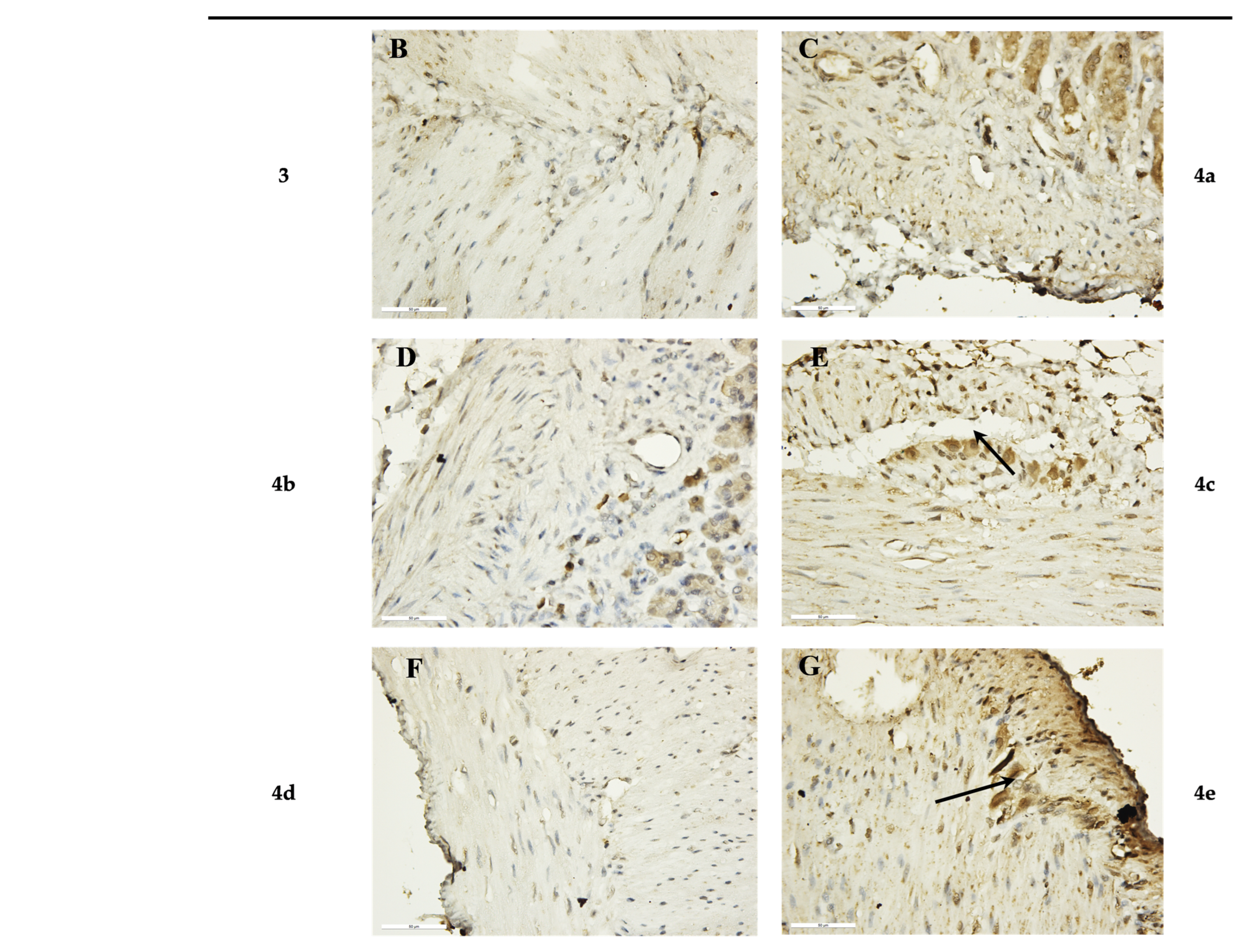
Ex Vivo Immunohistochemical Analysis
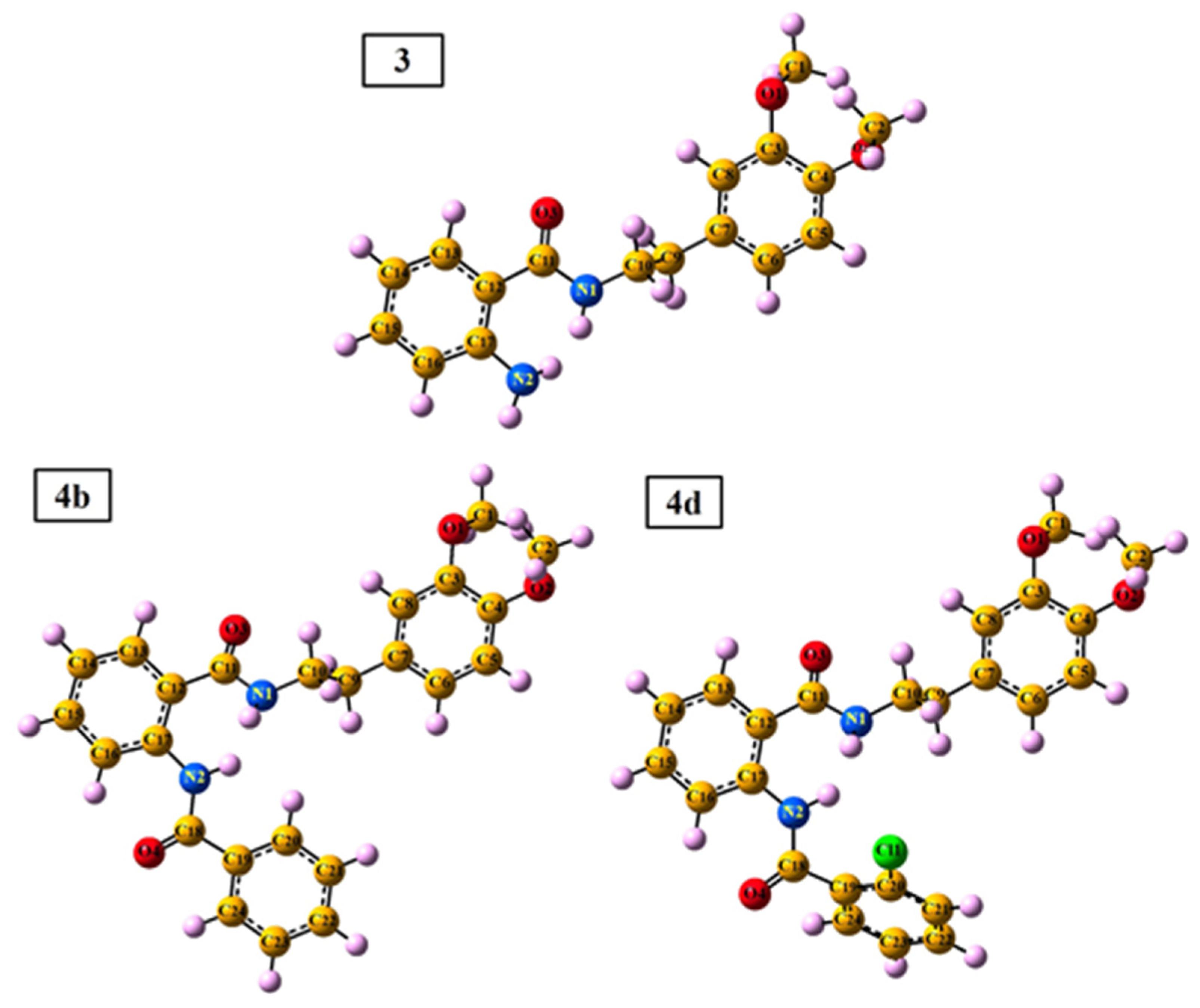
2.3.2. DFT (Density Functional Theory) Calculation
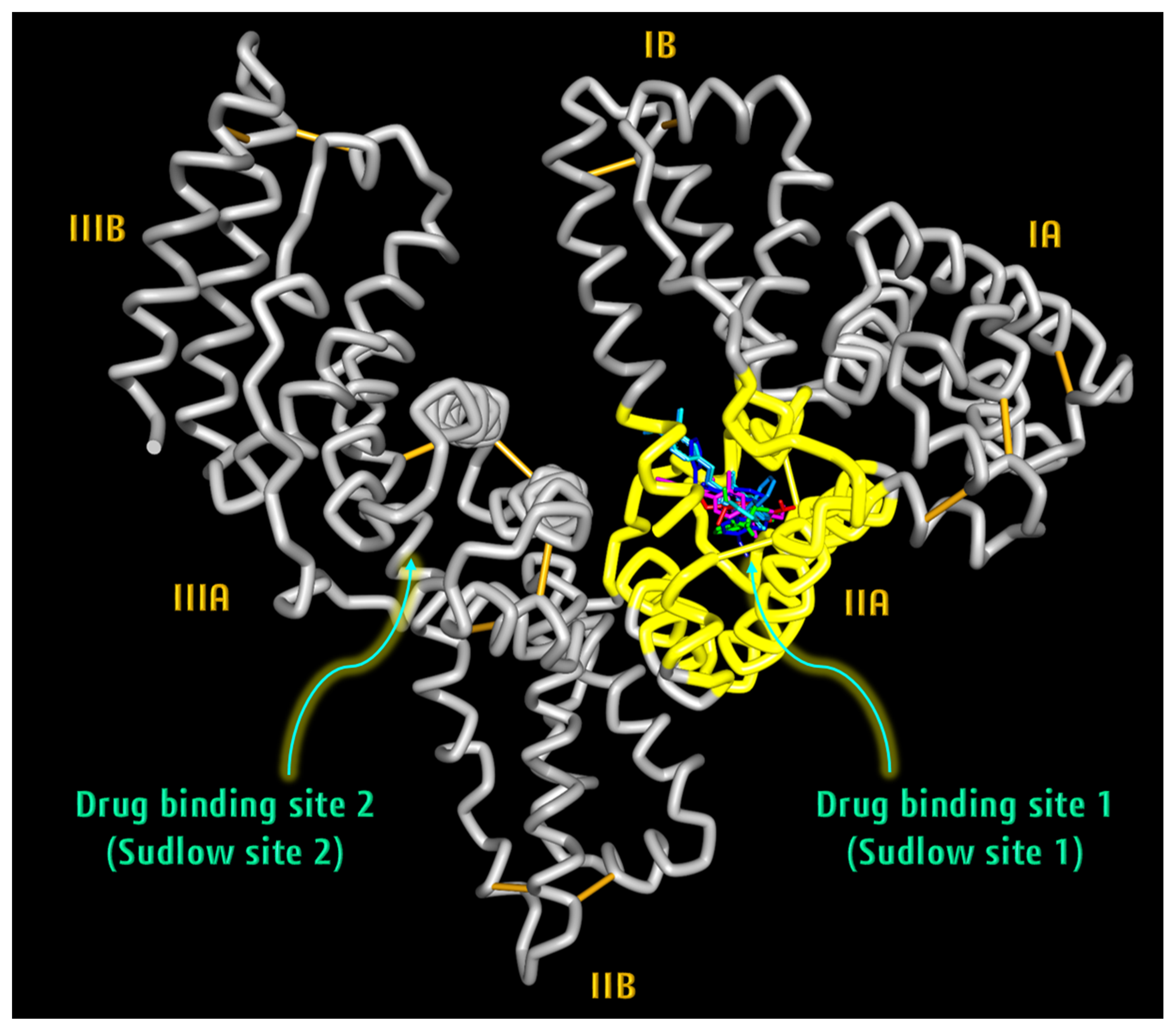
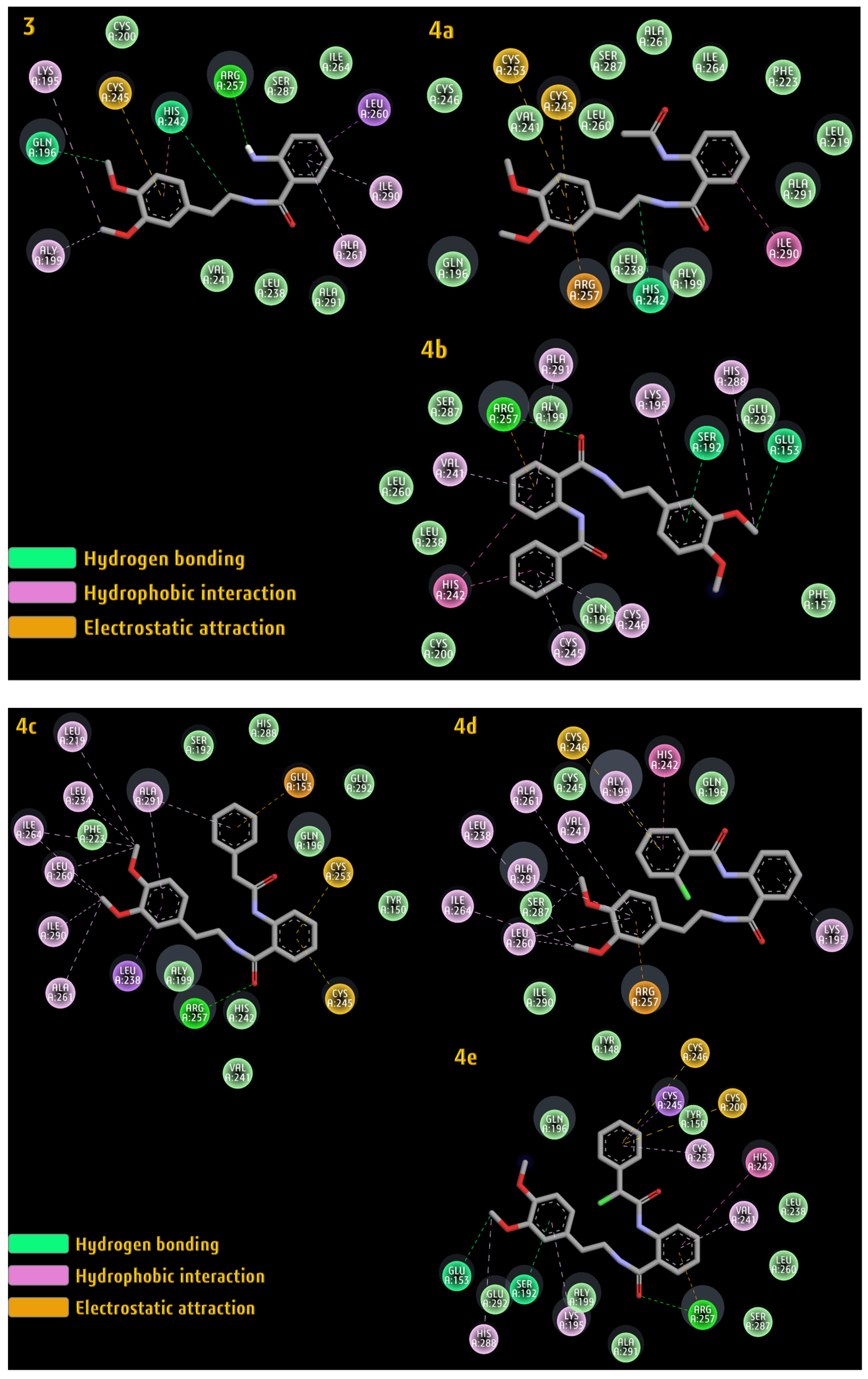
2.3.3. Albumin Simulation
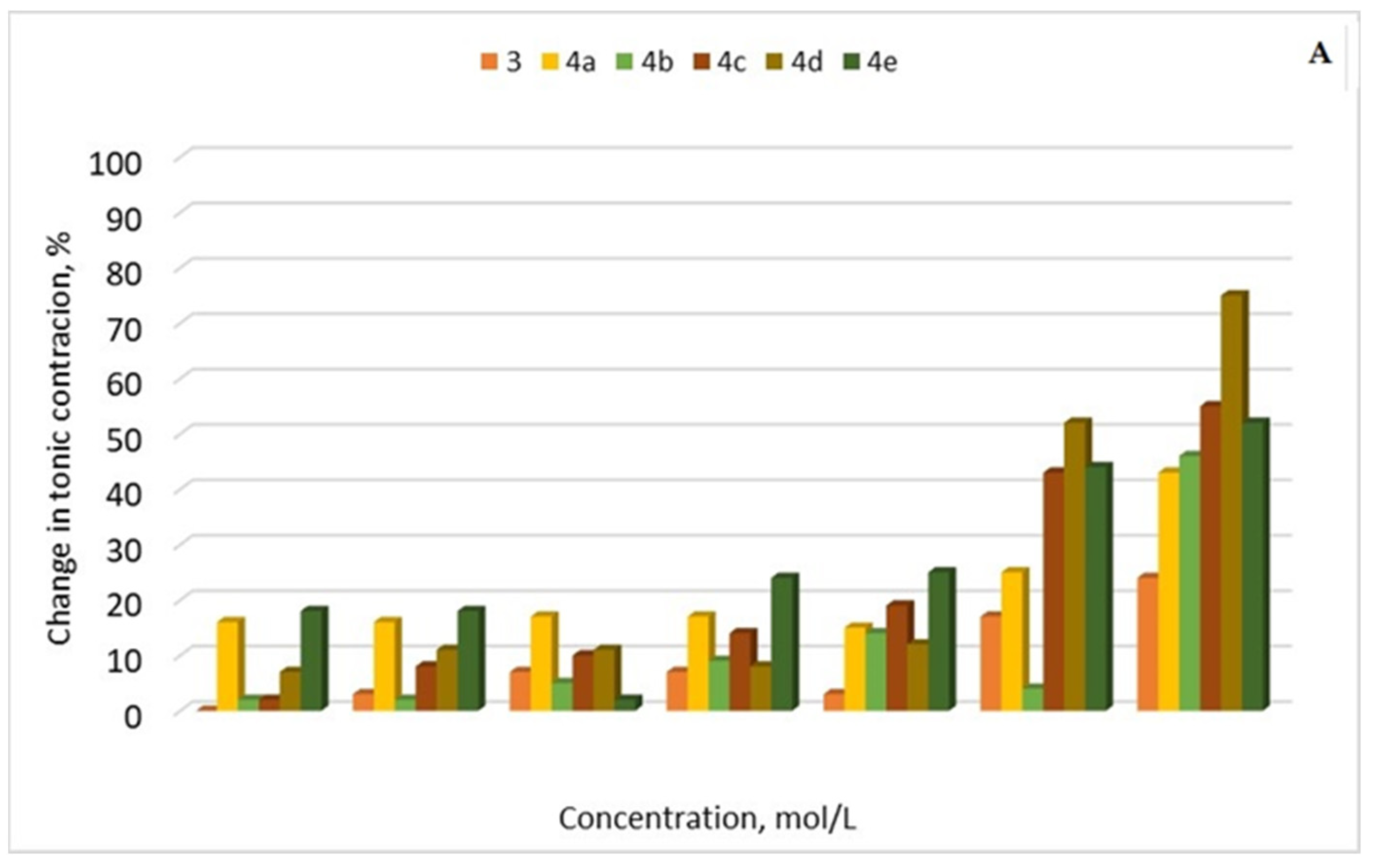
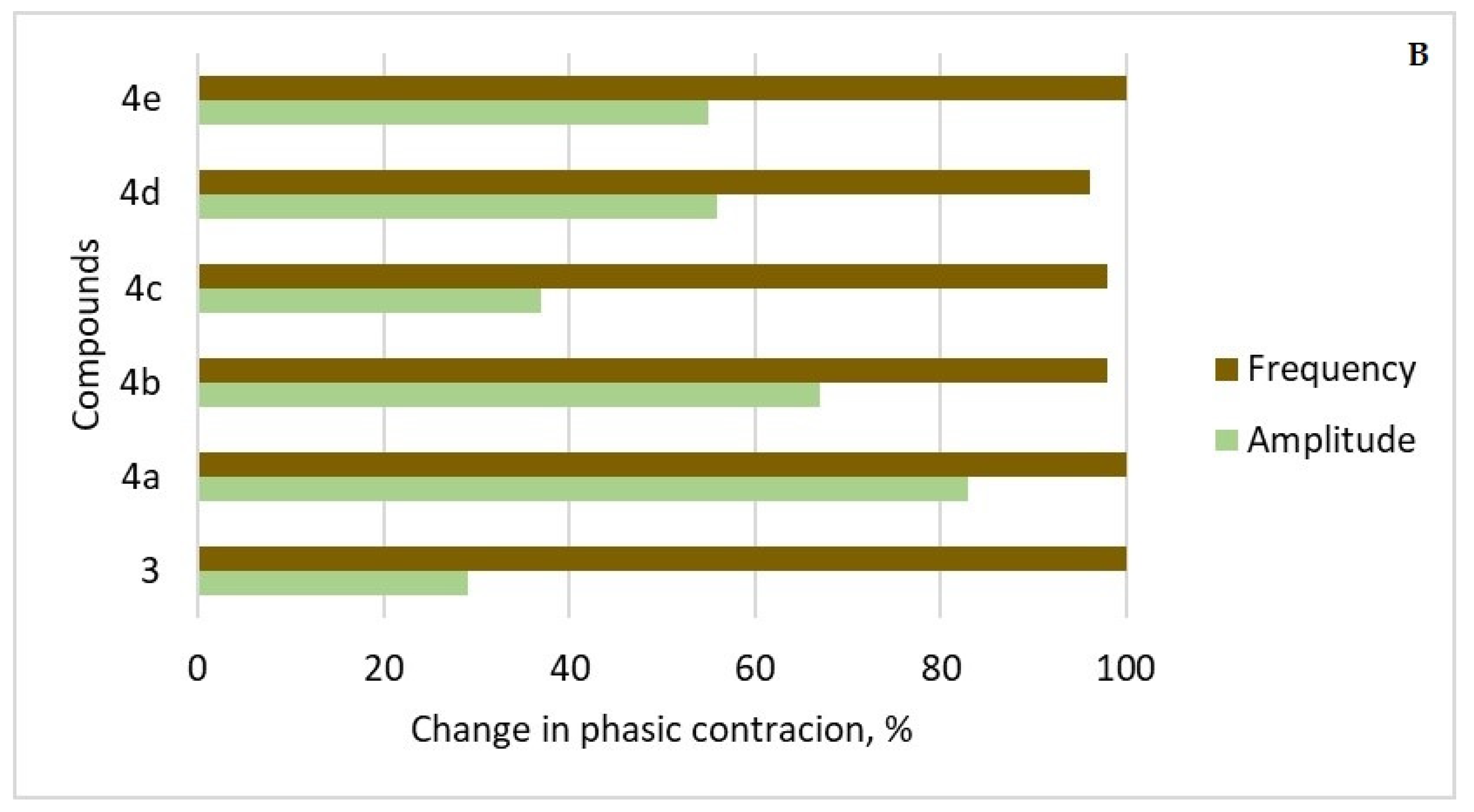
2.3.4. Ex Vivo Smooth Muscle-Relaxant Activity
2.3.5. Antimicrobial Activity
3. Materials and Methods
3.1. Chemicals
3.2. Synthetic Methods Experimental Protocols and Spectral Data
3.2.1. Synthesis of Hybrid Molecule 2-Amino-N-(3-chlorophenethyl)benzamide 3
3.2.2. Diamides Synthesis 4a–e; Typical Procedure
3.3. In Silico Predictions
3.3.1. PASS Online Predictions
3.3.2. Theoretical Prediction of Pharmacokinetic Parameters (ADME)
3.3.3. Theoretical Prediction of Toxicity
3.4. DFT Calculations
3.5. Molecular Docking Simulation
3.6. Microbiological Tests
3.6.1. Tested Microorganisms
3.6.2. Culture Media
Malt Extract Agar (MEA)
3.6.3. Antimicrobial Activity Assay
3.7. Inhibition of Albumin Denaturation
3.8. Immunohistochemical Methods
3.8.1. Histology
3.8.2. Immunohistochemistry
3.8.3. Analysis of Immunohistochemical Reactions
3.9. Smooth Muscle Activity
3.9.1. Animals, Tissues, and Preparations
3.9.2. Measuring Smooth Muscle Tension
3.9.3. Studying the Mechanical Activity of Isolated SMPs
3.10. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ibrahim, B.; Sowemimo, A.; van Rooyen, A.; Van de Venter, M. Antiinflammatory, Analgesic and Antioxidant Activities of Cyathula Prostrata (Linn.) Blume (Amaranthaceae). J. Ethnopharmacol. 2012, 141, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Ejaz Ahmed, M.; Khan, M.M.; Javed, H.; Vaibhav, K.; Khan, A.; Tabassum, R.; Ashafaq, M.; Islam, F.; Safhi, M.M.; Islam, F. Amelioration of Cognitive Impairment and Neurodegeneration by Catechin Hydrate in Rat Model of Streptozotocin-Induced Experimental Dementia of Alzheimer’s Type. Neurochem. Int. 2013, 62, 492–501. [Google Scholar] [CrossRef]
- Kodydkova, J.; Vavrova, L.; Stankova, B.; Macasek, J.; Krechler, T.; Zak, A. Antioxidant Status and Oxidative Stress Markers in Pancreatic Cancer and Chronic Pancreatitis. Pancreas 2013, 42, 614–621. [Google Scholar] [CrossRef]
- Kinne, R.W.; Bräuer, R.; Stuhlmüller, B.; Palombo-Kinne, E.; Burmester, G.-R. Macrophages in Rheumatoid Arthritis. Arthritis Res. 2000, 2, 189. [Google Scholar] [CrossRef]
- Yu, T.; Lee, J.; Lee, Y.G.; Byeon, S.E.; Kim, M.H.; Sohn, E.-H.; Lee, Y.J.; Lee, S.G.; Cho, J.Y. In Vitro and in Vivo Anti-Inflammatory Effects of Ethanol Extract from Acer Tegmentosum. J. Ethnopharmacol. 2010, 128, 139–147. [Google Scholar] [CrossRef]
- White, M. Mediators of Inflammation and the Inflammatory Process. J. Allergy Clin. Immunol. 1999, 103, S378–S381. [Google Scholar] [CrossRef]
- Obreja, O.; Rathee, P.K.; Lips, K.S.; Distler, C.; Kress, M. IL-1J Potentiates Heat-Activated Currents in Rat Sensory Neurons: Involvement of IL-1RI, Tyrosine Kinase, and Protein Kinase C. FASEB J. 2002, 16, 1497–1503. [Google Scholar] [CrossRef]
- Khan, A.; Baki, M.; Al-Bari, M.; Hasan, S.; Mosaddik, M.; Rahman, M.; Haque, M. Antipyretic Activity of Roots of Laportea Crenulata Gaud in Rabbit. Res. J. Med. Med. Sci. 2007, 2, 58–61. [Google Scholar]
- Rang, H.P.; Dale, M.; Ritter, J. Pharmacology, 4th ed.; Churchill Livingstone: New York, NY, USA, 2001. [Google Scholar]
- Dewanjee, S.; Maiti, A.; Sahu, R.; Dua, T.K.; Mandal, S.C. Study of Anti-Inflammatory and Antinociceptive Activity of Hydroalcoholic Extract of Schima Wallichii Bark. Pharm. Biol. 2009, 47, 402–407. [Google Scholar] [CrossRef]
- Sengar, N.; Joshi, A.; Prasad, S.K.; Hemalatha, S. Anti-Inflammatory, Analgesic and Anti-Pyretic Activities of Standardized Root Extract of Jasminum Sambac. J. Ethnopharmacol. 2015, 160, 140–148. [Google Scholar] [CrossRef] [PubMed]
- Abdulredha, F.H.; Mahdi, M.F.; Khan, A.K. In Silico Molecular Docking, ADME Study and Synthesis of New 1,3-Diazetidin-2-One Derivatives with High Anti-Proliferative Activity. F1000Research 2023, 12, 1090. [Google Scholar] [CrossRef]
- Valdes, A.M.; Walter, J.; Segal, E.; Spector, T.D. Role of the Gut Microbiota in Nutrition and Health. BMJ 2018, 361, k2179. [Google Scholar] [CrossRef] [PubMed]
- Mitropoulou, G.; Stavropoulou, E.; Vaou, N.; Tsakris, Z.; Voidarou, C.; Tsiotsias, A.; Tsigalou, C.; Taban, B.M.; Kourkoutas, Y.; Bezirtzoglou, E. Insights into Antimicrobial and Anti-Inflammatory Applications of Plant Bioactive Compounds. Microorganisms 2023, 11, 1156. [Google Scholar] [CrossRef] [PubMed]
- Bürger, M.; Lange, K.; Stallmach, A. Intestinales Mikrobiom Und Chronisch-Entzündliche Darmerkrankungen: Feindschaft Oder Freundschaft? Der Gastroenterol. 2015, 10, 87–101. [Google Scholar] [CrossRef]
- Burke, A.; Smyth, E.; Fitzgerald, G. Analgesic-Antipyretic Agents: Pharmacotherapy of Gout. In Goodman and Gilmans the Pharmacological Basis of Therapeutic; Brunton, L., Lazo JS, J., Parker, K., Eds.; McGraw Hill: New York, NY, USA, 2006. [Google Scholar]
- Shah, A.S.; Alagawadi, K.R. Anti-Inflammatory, Analgesic and Antipyretic Properties of Thespesia Populnea Soland Ex. Correa Seed Extracts and Its Fractions in Animal Models. J. Ethnopharmacol. 2011, 137, 1504–1509. [Google Scholar] [CrossRef] [PubMed]
- Maroon, J.C.; Bost, J.W.; Maroon, A. Natural anti-inflammatory agents for pain relief. Surg. Neurol. Int. 2010, 1, 80. [Google Scholar] [CrossRef]
- Eissa, A.A.H.M.; Soliman, G.A.E.H.; Khataibeh, M.H. Design, synthesis and anti-inflammatory activity of structurally simple anthranilic acid congeners devoid of ulcerogenic side effects. Chem. Pharm. Bull. 2012, 60, 1290–1300. [Google Scholar] [CrossRef]
- Milusheva, M.; Gledacheva, V.; Stefanova, I.; Feizi-Dehnayebi, M.; Mihaylova, R.; Nedialkov, P.; Cherneva, E.; Tumbarski, Y.; Tsoneva, S.; Todorova, M.; et al. Synthesis, Molecular Docking, and Biological Evaluation of Novel Anthranilic Acid Hybrid and Its Diamides as Antispasmodics. Int. J. Mol. Sci. 2023, 24, 13855. [Google Scholar] [CrossRef]
- Oxenkrug, G.; van der Hart, M.; Roeser, J.; Summergrad, P. Anthranilic Acid: A Potential Biomarker and Treatment Target for Schizophrenia. Ann. Psychiatry Ment. Health 2016, 4, 1059. [Google Scholar]
- Prasher, P.; Sharma, M. “Azole” as Privileged Heterocycle for Targeting the Inducible Cyclooxygenase Enzyme. Drug Dev. Res. 2020, 82, 167–197. [Google Scholar] [CrossRef]
- Prasher, P.; Sharma, M.; Zacconi, F.; Gupta, G.; Aljabali, A.A.A.; Mishra, V.; Tambuwala, M.M.; Kapoor, D.N.; Negi, P.; Andreoli Pinto, T.d.J.; et al. Synthesis and Anticancer Properties of “Azole” Based Chemotherapeutics as Emerging Chemical Moieties: A Comprehensive Review. Curr. Org. Chem. 2021, 25, 654–668. [Google Scholar]
- Varnavas, A.; Lassiani, L.; Valenta, V.; Mennuni, L.; Makovec, F.; Hadjipavlou-Litina, D. Anthranilic Acid Based CCK1 Receptor Antagonists: Preliminary Investigation on Their Second “Touch Point”. Eur. J. Med. Chem. 2005, 40, 563–581. [Google Scholar] [CrossRef]
- Kim, D.-I.; Kim, H.-Y.; Kwon, L.-S.; Park, S.-D.; Jeon, G.-H.; Jung, K.-Y.; Min, J.-K.; Nam, W.-H.; Lee, K.; Chung, Y.-S.; et al. Synthesis and biological activity of KCB-328 and its analogues: Novel class III antiarrhythmic agents with little reverse frequency dependence. Bioorg. Med. Chem. Lett. 1999, 9, 85–90. [Google Scholar] [CrossRef]
- Bentley, K.W. Beta-phenylethylamines and the isoquinoline alkaloids. Nat. Prod. Rep. 2001, 18, 148–170. [Google Scholar] [CrossRef]
- Tigabu, A.; Jember, A.; Nega, T.; Wubishet, G.; Misganaw, H.; Goshu, T.; Negash, M. Bacterial Meningitis Among Adult Patients at University of Gondar Comprehensive Specialized Referral Hospital. Infect. Drug Resist. 2021, 14, 565–574. [Google Scholar] [CrossRef]
- Mirecka, A. Etiological agents of bacterial meningitis in adults and antibiotic susceptibility of Streptococcus pneumoniae isolated between 2009-2016 from patients of Regional Specialist Hospital of Dr Wł. Biegański in Lódź. Prz. Epidemiol. 2018, 72, 313–324. [Google Scholar] [CrossRef]
- Roughley, S.D.; Jordan, A.M. The Medicinal Chemist’s Toolbox: An Analysis of Reactions Used in the Pursuit of Drug Candidates. J. Med. Chem. 2011, 54, 3451–3479. [Google Scholar] [CrossRef]
- Seavill, P.W.; Wilden, J.D. The Preparation and Applications of Amides Using Electrosynthesis. Green Chem. 2020, 22, 7737–7759. [Google Scholar] [CrossRef]
- Wang, X. Challenges and Outlook for Catalytic Direct Amidation Reactions. Nat. Catal. 2019, 2, 98–102. [Google Scholar] [CrossRef]
- Bray, B.L. Large-Scale Manufacture of Peptide Therapeutics by Chemical Synthesis. Nat. Rev. Drug Discov. 2003, 2, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Milusheva, M.; Gledacheva, V.; Batmazyan, M.; Nikolova, S.; Stefanova, I.; Dimitrova, D.; Saracheva, K.; Tomov, D.; Chaova-Gizdakova, V. Ex Vivo and in Vivo Study of Some Isoquinoline Precursors. Sci. Pharm. 2022, 90, 37. [Google Scholar] [CrossRef]
- Daina, A.; Michielin, O.; Zoete, V. SwissADME: A Free Web Tool to Evaluate Pharmacokinetics, Drug-Likeness and Medicinal Chemistry Friendliness of Small Molecules. Sci. Rep. 2017, 7, 42717. [Google Scholar] [CrossRef]
- Anzali, S.; Barnickel, G.; Cezanne, B.; Krug, M.; Filimonov, D.; Poroikov, V. Discriminating between Drugs and Nondrugs by Prediction of Activity Spectra for Substances (PASS). J. Med. Chem. 2001, 44, 2432–2437. [Google Scholar] [CrossRef]
- Mathew, B.; Suresh, J.; Anbazhagan, S. Synthesis and PASS-Assisted in Silico Approach of Some Novel 2-Substituted Benzimidazole Bearing a Pyrimidine-2, 4, 6(Trione) System as Mucomembranous Protector. J. Pharm. Bioallied Sci. 2013, 5, 39–43. [Google Scholar] [CrossRef]
- Ekins, S.; Olechno, J.; Williams, A.J. Dispensing Processes Impact Apparent Biological Activity as Determined by Computational and Statistical Analyses. PLoS ONE 2013, 8, e62325. [Google Scholar] [CrossRef]
- Zoete, V.; Daina, A.; Bovigny, C.; Michielin, O. SwissSimilarity: A Web Tool for Low to Ultra High Throughput Ligand-Based Virtual Screening. J. Chem. Inf. Model. 2016, 56, 1399–1404. [Google Scholar] [CrossRef]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 2001, 46, 3–26. [Google Scholar] [CrossRef]
- Martin, Y.C. A bioavailability score. J. Med. Chem. 2005, 48, 3164–3170. [Google Scholar] [CrossRef] [PubMed]
- Shaaban, S.; Abdou, A.; Alhamzani, A.G.; Abou-Krisha, M.M.; Al-Qudah, M.A.; Alaasar, M.; Youssef, I.; Yousef, T.A. Synthesis and in Silico Investigation of Organoselenium-Clubbed Schiff Bases as Potential Mpro Inhibitors for the SARS-CoV-2 Replication. Life 2023, 13, 912. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, N.; Kurata, M.; Yamamoto, T.; Morikawa, S.; Masumoto, J. The role of interleukin-1 in general pathology. Inflamm. Regen. 2019, 39, 12. [Google Scholar] [CrossRef]
- Mantovani, A.; Allavena, P.; Sica, A.; Balkwill, F. Cancer-related inflammation. Nature 2008, 454, 436–444. [Google Scholar] [CrossRef]
- Sell, H.; Habich, C.; Eckel, J. Adaptive Immunity in Obesity and Insulin Resistance. Nat. Rev. Endocrinol. 2012, 8, 709–716. [Google Scholar] [CrossRef] [PubMed]
- Boulangé, C.L.; Neves, A.L.; Chilloux, J.; Nicholson, J.K.; Dumas, M.-E. Impact of the Gut Microbiota on Inflammation, Obesity, and Metabolic Disease. Genome Med. 2016, 8, 42. [Google Scholar] [CrossRef]
- Acharya, A.; Thakur, S.; Muddapur, M. Evaluation of Serum Interleukin-10 Levels as a Predictor of Glycemic Alteration in Chronic Periodontitis and Type 2 Diabetes Mellitus. J. Indian Soc. Periodontol. 2015, 19, 388. [Google Scholar] [CrossRef] [PubMed]
- Al Qudah, M.; Alfaqih, M.; Al-Shboul, O.; Saadeh, R.; Al-Dwairi, A. Effect of Cytokine Treatment on the Expression and Secretion of Brain Derived Neurotrophic Factor in the Smooth Muscle of the Rat Colon. Biomed. Rep. 2020, 13, 55–60. [Google Scholar] [CrossRef]
- Xia, Y.; Hu, H.; Liu, S.; Ren, J.; Zafirov, D.H.; Wood, J.D. IL-1β and IL-6 Excite Neurons and Suppress Nicotinic and Noradrenergic Neurotransmission in Guinea Pig Enteric Nervous System. J. Clin. Investig. 1999, 103, 1309–1316. [Google Scholar] [CrossRef] [PubMed]
- Hagar, M.; Ahmed, H.A.; Aljohani, G.; Alhaddad, O.A. Investigation of Some Antiviral N-Heterocycles as COVID 19 Drug: Molecular Docking and DFT Calculations. Int. J. Mol. Sci. 2020, 21, 3922. [Google Scholar] [CrossRef]
- Abraham, C.S.; Muthu, S.; Prasana, J.C.; Fathima Rizwana, B.; Armaković, S.; Armaković, S.J. Vibrational and Electronic Absorption Spectroscopic Profiling, Natural Hybrid Orbital, Charge Transfer, Electron Localization Function and Molecular Docking Analysis on 3-Amino-3-(2-Nitrophenyl) Propionic Acid. J. Mol. Struct. 2018, 1171, 733–746. [Google Scholar] [CrossRef]
- Nikolova, S.; Milusheva, M.; Gledacheva, V.; Feizi-Dehnayebi, M.; Kaynarova, L.; Georgieva, D.; Delchev, V.B.; Stefanova, I.; Tumbarski, Y.; Mihaylova, R.; et al. Drug-Delivery Silver Nanoparticles: A New Perspective for Phenindione as an Anticoagulant. Biomedicines 2023, 11, 2201. [Google Scholar] [CrossRef]
- Zinatloo-Ajabshir, S.; Rakhshani, S.; Mehrabadi, Z.; Farsadrooh, M.; Feizi-Dehnayebi, M.; Rakhshani, S.; Dušek, M.; Eigner, V.; Rtimi, S.; Aminabhavi, T.M. Novel rod-like [Cu (phen) 2 (OAc)]·PF6 complex for high-performance visible-light-driven photocatalytic degradation of hazardous organic dyes: DFT approach, Hirshfeld and fingerprint plot analysis. J. Environ. Manag. 2024, 350, 119545. [Google Scholar] [CrossRef]
- Feizi-Dehnayebi, M.; Dehghanian, E.; Mansouri-Torshizi, H. Biological Activity of Bis-(Morpholineacetato)Palladium(II) Complex: Preparation, Structural Elucidation, Cytotoxicity, DNA-/Serum Albumin-Interaction, Density Functional Theory, In-Silico Prediction and Molecular Modeling. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2022, 281, 121543. [Google Scholar] [CrossRef]
- Ismael, M.; Abdel-Mawgoud, A.-M.M.; Rabia, M.K.; Abdou, A. Ni(II) Mixed-Ligand Chelates Based on 2-Hydroxy-1-Naphthaldehyde as Antimicrobial Agents: Synthesis, Characterization, and Molecular Modeling. J. Mol. Liquids 2021, 330, 115611. [Google Scholar] [CrossRef]
- Dheivamalar, S.; Bansura Banu, K. A DFT Study on Functionalization of Acrolein on Ni-Doped (ZnO)6 Nanocluster in Dye-Sensitized Solar Cells. Heliyon 2019, 5, e02903. [Google Scholar] [CrossRef] [PubMed]
- İnaç, H.; Ashfaq, M.; Dege, N.; Feizi-Dehnayebi, M.; Munawar, K.S.; Yağcı, N.K.; Çınar, E.B.; Tahir, M.N. Synthesis, Spectroscopic Characterizations, Single Crystal XRD, Supramolecular Assembly Inspection via Hirshfeld Surface Analysis, and DFT Study of a Hydroxy Functionalized Schiff Base Cu(II) Complex. J. Mol. Struct. 2023, 1295, 136751. [Google Scholar] [CrossRef]
- Ricci, C.G.; Netz, P.A. Docking Studies on DNA-Ligand Interactions: Building and Application of a Protocol to Identify the Binding Mode. J. Chem. Inf. Model. 2009, 49, 1925–1935. [Google Scholar] [CrossRef]
- Lee, P.; Wu, X. Review: Modifications of Human Serum Albumin and Their Binding Effect. Curr. Pharm. Des. 2015, 21, 1862–1865. [Google Scholar] [CrossRef] [PubMed]
- Ndoye Foe, F.M.-C.; Tchinang, T.F.K.; Nyegue, A.M.; Abdou, J.-P.; Yaya, A.J.G.; Tchinda, A.T.; Essame, J.-L.O.; Etoa, F.-X. Chemical Composition, in Vitro Antioxidant and Anti-Inflammatory Properties of Essential Oils of Four Dietary and Medicinal Plants from Cameroon. BMC Complement. Altern. Med. 2016, 16, 117. [Google Scholar] [CrossRef]
- Osman, N.; Sidik, N.; Awal, A.; Adam, N.; Rezali, N. In Vitro Xanthine Oxidase (XO) and Albumin Denaturation Inhibition Assay of Barringtonia Racemosa L. And Total Phenolic Content Analysis for Potential Anti-Inflammatory Use in Gouty Arthritis. J. Intercult. Ethnopharmacol. 2016, 5, 343. [Google Scholar] [CrossRef]
- Zheng, Y.-B.; Shi, L.; Zhu, X.-M.; Bao, Y.-P.; Bai, L.-J.; Li, J.-Q.; Liu, J.-J.; Han, Y.; Shi, J.; Lu, L. Anticholinergic Drugs and the Risk of Dementia: A Systematic Review and Meta-Analysis. Neurosci. Biobehav. Rev. 2021, 127, 296–306. [Google Scholar] [CrossRef]
- Brenner, D.M.; Lacy, B.E. Antispasmodics for Chronic Abdominal Pain: Analysis of North American Treatment Options. Am. J. Gastroenterol. 2021, 116, 1587–1600. [Google Scholar] [CrossRef]
- Karaki, H.; Ozaki, H.; Hori, M.; Mitsui-Saito, M.; Amano, K.; Harada, K.; Miyamoto, S.; Nakazawa, H.; Won, K.J.; Sato, K. Calcium Movements, Distribution, and Functions in Smooth Muscle. Pharmacol. Rev. 1997, 49, 157–230. [Google Scholar]
- Unno, T.; Matsuyama, H.; Sakamoto, T.; Uchiyama, M.; Izumi, Y.; Okamoto, H.; Yamada, M.; Wess, J.; Komori, S. M2 and M3 Muscarinic Receptor-Mediated Contractions in Longitudinal Smooth Muscle of the Ileum Studied with Receptor Knockout Mice. Br. J. Pharmacol. 2005, 146, 98–108. [Google Scholar] [CrossRef]
- Kullmann, F.A.; Daugherty, S.L.; de Groat, W.C.; Birder, L.A. Bladder Smooth Muscle Strip Contractility as a Method to Evaluate Lower Urinary Tract Pharmacology. J. Vis. Exp. 2014, 90, e51807. [Google Scholar] [CrossRef]
- Kelber, O.; Bauer, R.; Kubelka, W. Phytotherapy in Functional Gastrointestinal Disorders. Dig. Dis. 2017, 35, 36–42. [Google Scholar] [CrossRef]
- Jayanthi, M.; Rajakumar, P. Synthesis and Antimicrobial Activity of Unsymmetrical Dendrimers with Indazole, Salicylates and Anthranilates as Surface Units. J. Heterocycl. Chem. 2017, 54, 1963–1973. [Google Scholar] [CrossRef]
- Wang, L.; Linares-Otoya, V.; Liu, Y.; Mettal, U.; Marner, M.; Armas-Mantilla, L.; Willbold, S.; Kurtán, T.; Linares-Otoya, L.; Schäberle, T.F. Discovery and Biosynthesis of Antimicrobial Phenethylamine Alkaloids from the Marine Flavobacterium Tenacibaculum discolor Sv11. J. Nat. Prod. 2022, 85, 1039–1051. [Google Scholar] [CrossRef] [PubMed]
- Muchaamba, F.; Stephan, R.; Tasara, T. β-Phenylethylamine as a Natural Food Additive Shows Antimicrobial Activity against Listeria Monocytogenes on Ready-To-Eat Foods. Foods 2020, 9, 1363. [Google Scholar] [CrossRef]
- Pontiki, E.; Hadjipavlou-Litina, D. Synthesis and Pharmacochemical Evaluation of Novel Aryl-Acetic Acid Inhibitors of Lipoxygenase, Antioxidants, and Anti-Inflammatory Agents. Bioorg. Med. Chem. 2007, 15, 5819–5827. [Google Scholar] [CrossRef] [PubMed]
- Ottaviani, G.; Gosling, D.J.; Patissier, C.; Rodde, S.; Zhou, L.; Faller, B. What Is Modulating Solubility in Simulated Intestinal Fluids? Eur. J. Pharm. Sci. 2010, 41, 452–457. [Google Scholar] [CrossRef] [PubMed]
- Hollenberg, P.F. Characteristics and Common Properties of Inhibitors, Inducers, and Activators of CYP Enzymes. Drug Metab. Rev. 2002, 34, 17–35. [Google Scholar] [CrossRef] [PubMed]
- Frisch, M.; Trucks, G.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G. Gaussian 09, Revision D.01; Gaussian, Inc.: Wallingford, CT, USA, 2009; p. 201. [Google Scholar]
- Morris, G.M.; Huey, R.; Lindstrom, W.; Sanner, M.F.; Belew, R.K.; Goodsell, D.S.; Olson, A.J. AutoDock4 and AutoDockTools4: Automated Docking with Selective Receptor Flexibility. J. Comput. Chem. 2009, 30, 2785–2791. [Google Scholar] [CrossRef]
- MGL Tools. 1.5. 6 (ADT)/MGL Tools 1. 6.; The Scripps Research Institute: La Jolla, CA, USA, 2016. [Google Scholar]
- Yang, F.; Bian, C.; Zhu, L.; Zhao, G.; Huang, Z.; Huang, M. Effect of Human Serum Albumin on Drug Metabolism: Structural Evidence of Esterase Activity of Human Serum Albumin. J. Struct. Biol. 2007, 157, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Tumbarski, Y.; Deseva, I.; Mihaylova, D.; Stoyanova, M.; Krastev, L.; Nikolova, R.; Yanakieva, V.; Ivanov, I. Isolation, Characterization and Amino Acid Composition of a Bacteriocin Produced by Bacillus Methylotrophicus Strain BM47. Food Technol. Biotechnol. 2018, 56, 546. [Google Scholar] [CrossRef] [PubMed]
- Kumari, C.S.; Yasmin, N.; Hussain, M.R.; Babuselvam, M. Invitro Anti-Inflammatory and Anti-Artheritic Property of Rhizopora Mucronata Leaves. Int. J. Pharm. Sci. Res. 2015, 6, 482–485. [Google Scholar]
- Batista-Lima, F.J.; Gadelha, K.K.L.; Oliveira, D.M.; Vasconcelos, T.B.; Brito, T.S.; Magalhães, P.J.C. A Simple Laboratory Exercise with Rat Isolated Esophagus and Stomach Fundus to Reveal Functional Differences between Striated and Smooth Muscle Cells. Adv. Physiol. Educ. 2017, 41, 291–297. [Google Scholar] [CrossRef]
- Bailey, L.E.; Ong, S.D. Krebs-Henseleit Solution as a Physiological Buffer in Perfused and Superfused Preparations. J. Pharmacol. Methods 1978, 1, 171–175. [Google Scholar] [CrossRef]
- Jespersen, B.; Tykocki, N.R.; Watts, S.W.; Cobbett, P.J. Measurement of Smooth Muscle Function in the Isolated Tissue Bath-Applications to Pharmacology Research. J. Vis. Exp. 2015, 95, e52324. [Google Scholar] [CrossRef]
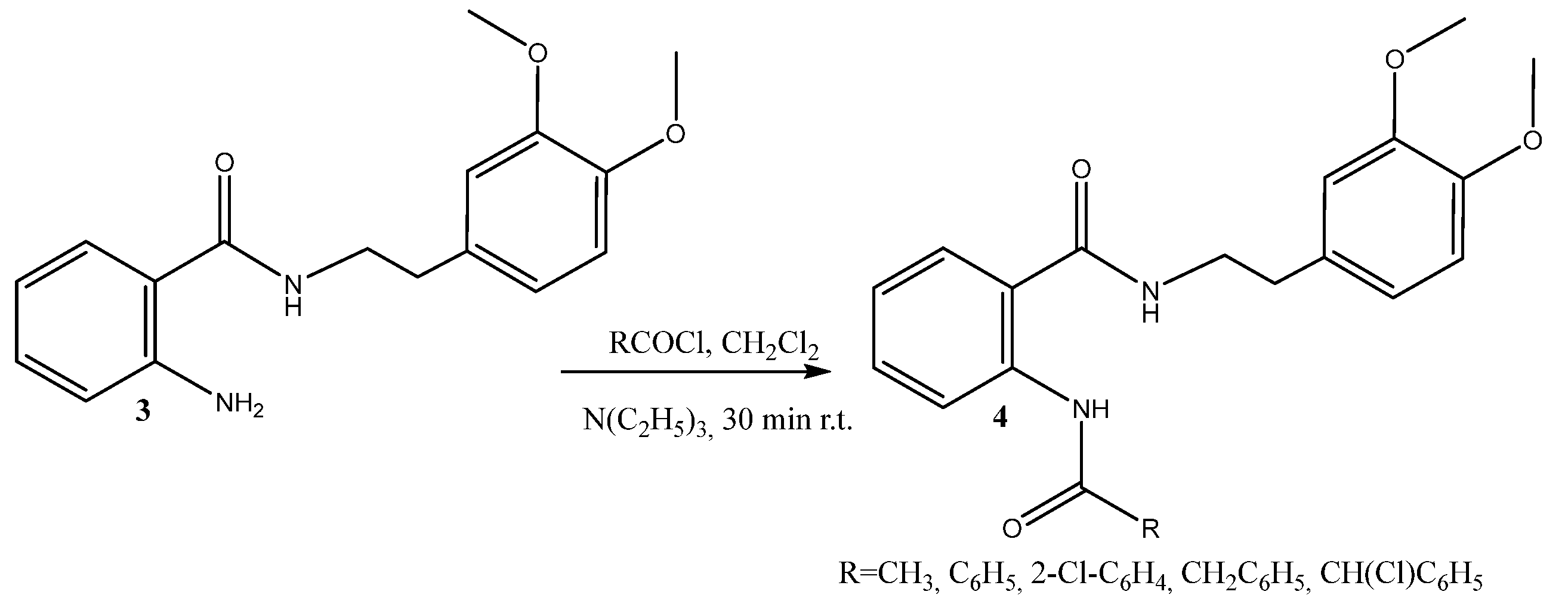
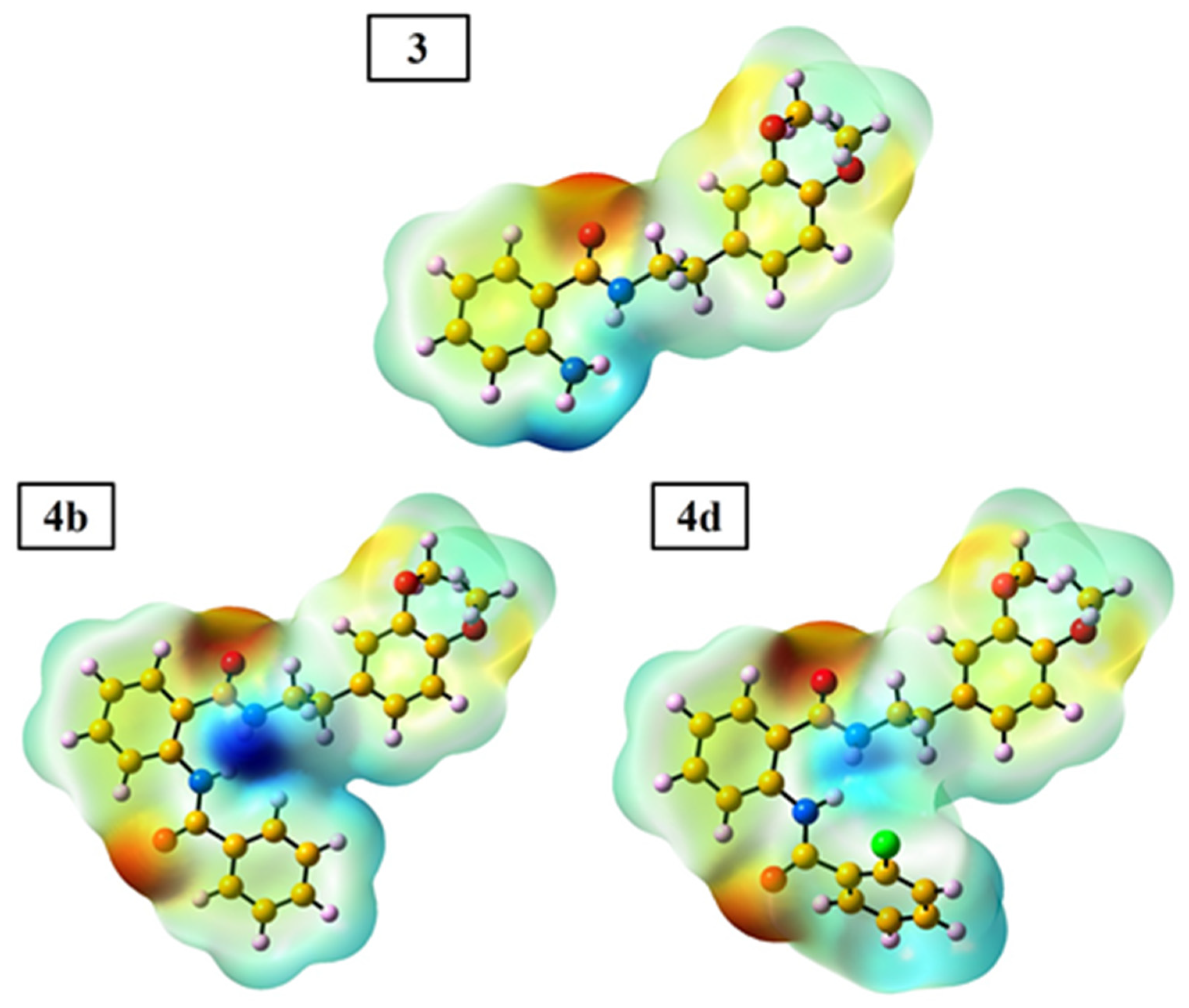

| 4 | R | Yield, % | mp, °C |
|---|---|---|---|
| a | CH3 | 80 | 95–97 |
| b | C6H5 | 78 | 121–124 |
| c | CH2-C6H5 | 79 | 92–93 |
| d | 2-Cl-C6H4 | 81 | 94–95 |
| e | CH(Cl)C6H5 | 82 | 102–103 |
| Compound | MW, g/mol | XLOGP3 | TPSA, Å2 | ESOL Log S | Fraction Csp3 | RB | BA Score | SA Score | Lipinski’s Violation |
|---|---|---|---|---|---|---|---|---|---|
| 3 | 300.35 | 2.97 | 73.58 | −3.51 | 0.24 | 7 | 0.55 | 2 | 0 |
| 4a | 342.39 | 2.83 | 76.66 | −3.51 | 0.26 | 9 | 0.55 | 2.42 | 0 |
| 4b | 404.46 | 4.48 | 76.66 | −4.95 | 0.17 | 10 | 0.55 | 2.73 | 0 |
| 4c | 418.48 | 4.42 | 76.66 | −4.82 | 0.20 | 11 | 0.55 | 2.90 | 0 |
| 4d | 438.93 | 5.11 | 76.66 | −5.55 | 0.17 | 10 | 0.55 | 2.81 | 1 |
| 4e | 452.93 | 5.14 | 76.66 | −5.58 | 0.20 | 11 | 0.55 | 3.48 | 0 |
| Compound | Control | Response to Compound |
|---|---|---|
| IL-1β | ++ | |
| 3 | − | |
| 4a | ++ | |
| 4b | − | |
| 4c | +++ | |
| 4d | − | |
| 4e | +++ |
| Compound | EHOMO | ELUMO | ΔE | χ | Pi | η | σ | ω |
|---|---|---|---|---|---|---|---|---|
| 3 | −6.16 | −1.00 | 5.16 | 3.58 | −3.58 | 2.58 | 0.39 | 2.48 |
| 4b | −6.37 | −1.69 | 4.68 | 4.03 | −4.03 | 2.34 | 0.43 | 3.47 |
| 4d | −6.34 | −1.57 | 4.77 | 3.95 | −3.95 | 2.38 | 0.42 | 3.27 |
| Type of Interaction with Residue | ||||
|---|---|---|---|---|
| Compound | ΔG (kcal/mol) | Hydrogen Bonding | Hydrophobic Interaction | Electrostatic Attraction |
| 3 | −5.36 | ARG257, HIS242, GLN196 | LEU260, ILE290, ALA261, HIS242, LYS195, ALY199 | CYS245 |
| 4a | −5.22 | HIS242 | ILE290 | CYS245, CYS253, ARG257 |
| 4b | −6.11 | ARG257, SER192, GLU153 | HIS288, LYS195, ALA291, VAL241, HIS242, CYS245, CYS246 | ARG257 |
| 4c | −4.87 | ARG257 | LEU238, ALA261, ILE290, LEU260, ILE264, LEU234, ALA291, LEU219 | CYS245, CYS253, GLU153 |
| 4d | −6.21 | GLN196 | LYS195, HIS242, ALY199, VAL241, ALA261, ALA291, LEU238, ILE264, LEU260 | CYS246, ARG257 |
| 4e | −5.28 | ARG257, SER192, GLU153 | CYS245, CYS253, HIS242, VAL241, LYS195, HIS288 | CYS246, CYS200 |
| Inhibition Zones, mm/Anthranilic Acid Hybrid and Its Diamides | |||
|---|---|---|---|
| 3 | 4b | 4d | |
| Bacillus subtilis ATCC 6633 | - | - | 10 |
| Bacillus amyloliquefaciens 4BCL-YT | - | - | 10 |
| Micrococcus luteus 2YC-YT | 10 | - | 15 |
| Salmonella enteritidis ATCC 13076 | 10 | - | - |
| Salmonella typhimurium NBIMCC 1672 | 8 | 10 | 8 |
| Klebsiella pneumoniae ATCC 13883 | - | 15 | - |
| Escherichia coli ATCC 25922 | 8 | 10 | 10 |
| Pseudomonas aeruginosa ATCC 9027 | 8 | 8 | 10 |
| Aspergillus niger ATCC 1015 | 12 | 10 | 8 |
| Penicillium chrysogenum | 10 | 9 | 8 |
| Rhizopus sp. | 9 | - | 8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Milusheva, M.; Todorova, M.; Gledacheva, V.; Stefanova, I.; Feizi-Dehnayebi, M.; Pencheva, M.; Nedialkov, P.; Tumbarski, Y.; Yanakieva, V.; Tsoneva, S.; et al. Novel Anthranilic Acid Hybrids—An Alternative Weapon against Inflammatory Diseases. Pharmaceuticals 2023, 16, 1660. https://doi.org/10.3390/ph16121660
Milusheva M, Todorova M, Gledacheva V, Stefanova I, Feizi-Dehnayebi M, Pencheva M, Nedialkov P, Tumbarski Y, Yanakieva V, Tsoneva S, et al. Novel Anthranilic Acid Hybrids—An Alternative Weapon against Inflammatory Diseases. Pharmaceuticals. 2023; 16(12):1660. https://doi.org/10.3390/ph16121660
Chicago/Turabian StyleMilusheva, Miglena, Mina Todorova, Vera Gledacheva, Iliyana Stefanova, Mehran Feizi-Dehnayebi, Mina Pencheva, Paraskev Nedialkov, Yulian Tumbarski, Velichka Yanakieva, Slava Tsoneva, and et al. 2023. "Novel Anthranilic Acid Hybrids—An Alternative Weapon against Inflammatory Diseases" Pharmaceuticals 16, no. 12: 1660. https://doi.org/10.3390/ph16121660
APA StyleMilusheva, M., Todorova, M., Gledacheva, V., Stefanova, I., Feizi-Dehnayebi, M., Pencheva, M., Nedialkov, P., Tumbarski, Y., Yanakieva, V., Tsoneva, S., & Nikolova, S. (2023). Novel Anthranilic Acid Hybrids—An Alternative Weapon against Inflammatory Diseases. Pharmaceuticals, 16(12), 1660. https://doi.org/10.3390/ph16121660











