Bioactivity of the Genus Turnera: A Review of the Last 10 Years
Abstract
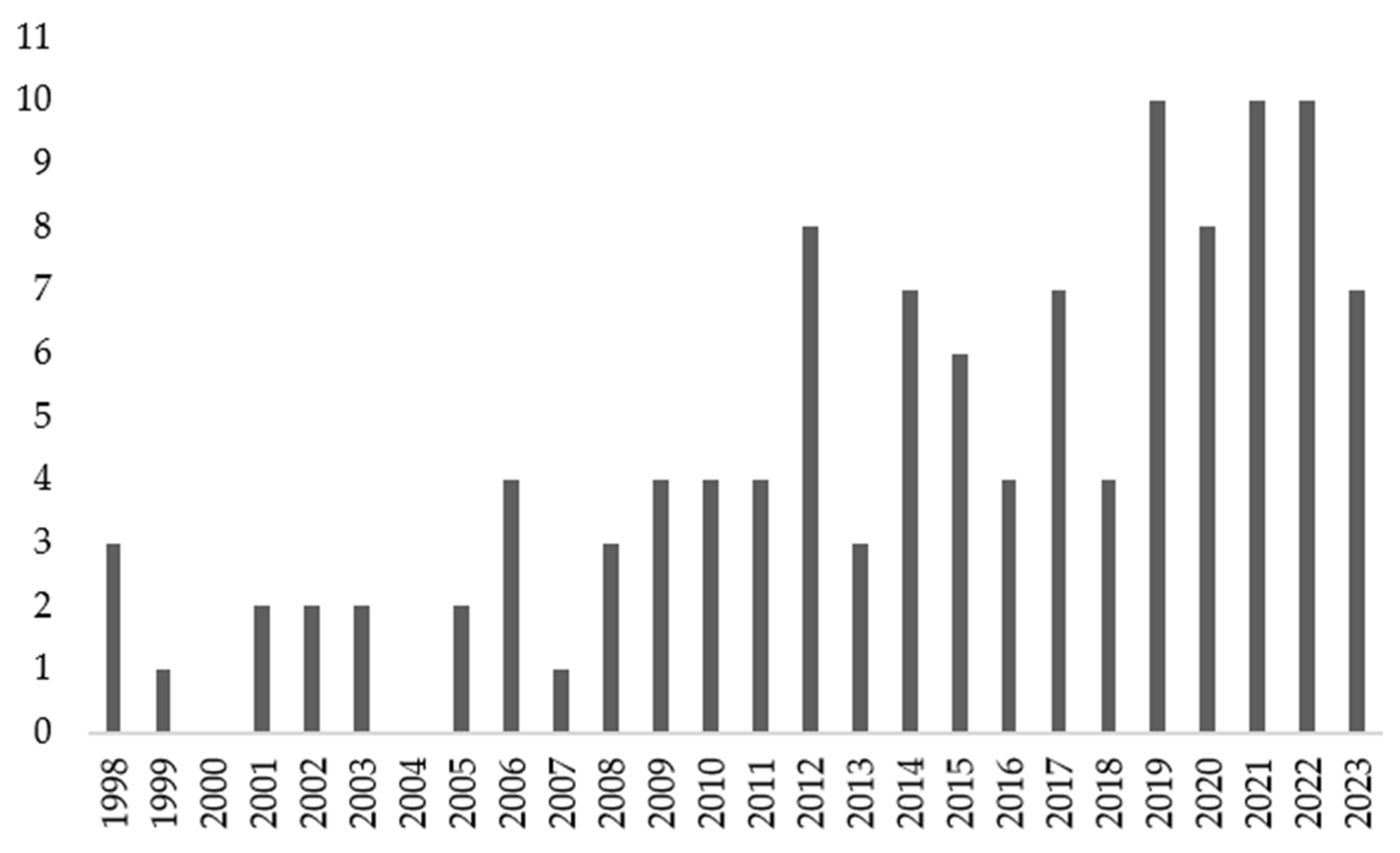
1. Introduction
2. Search Strategy
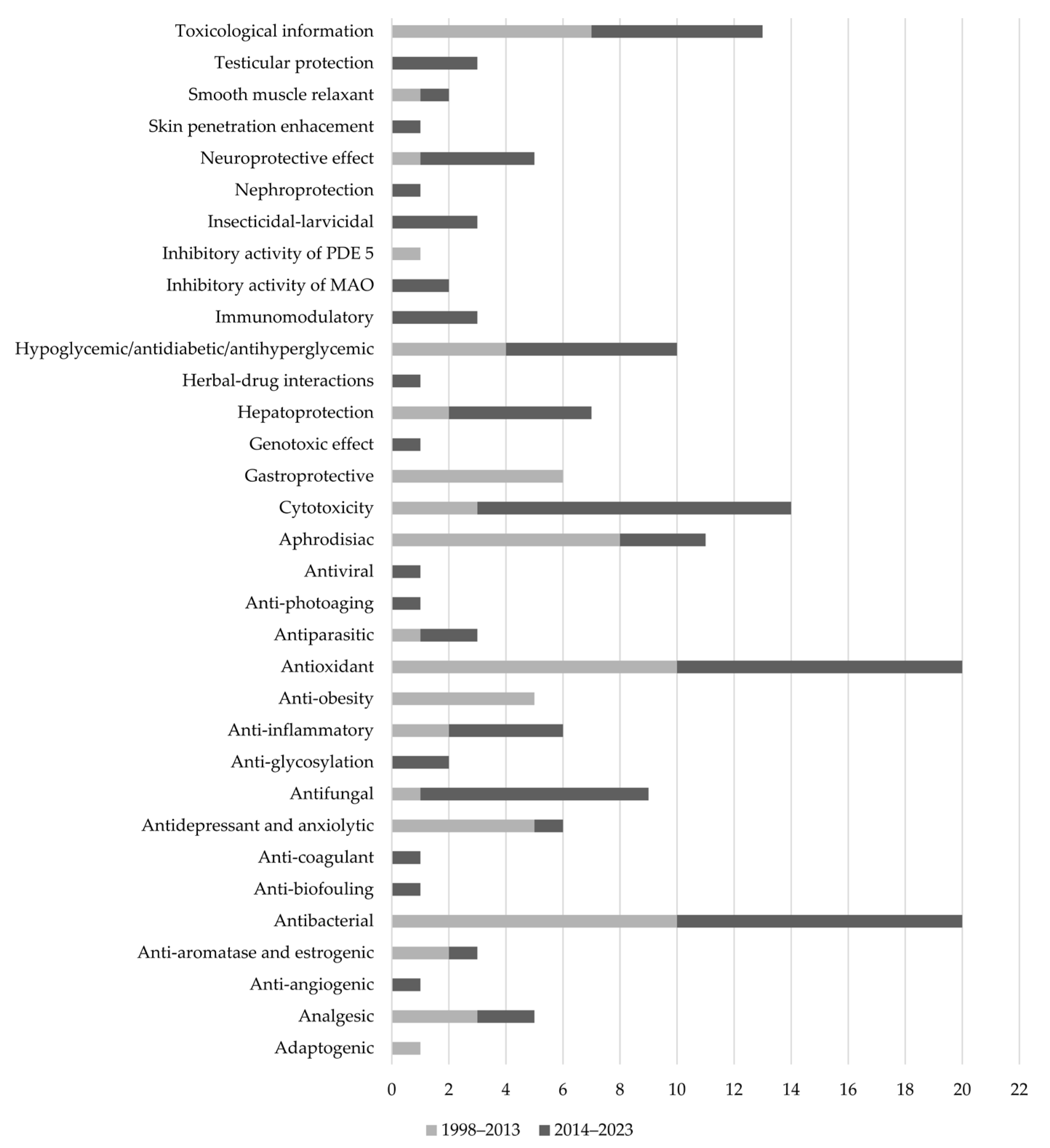
3. Results
3.1. Antioxidant Activity
3.2. Anti-Inflammatory Activity
3.3. Immunomodulatory Activity
3.4. Hepatoprotection
3.5. Nephroprotection
3.6. Testicular Protection
3.7. Neuroprotective Effect
3.8. Hypoglycemic/Antidiabetic/Antihyperglycemic Activity
3.9. Anti-Obesity Activity
3.10. Analgesic Activity
3.11. Antidepressant and Anxiolytic Activity
3.12. Aphrodisiac Activity
3.13. Herbal–Drug Interactions
3.14. Anti-Glycosylation Activity
3.15. Inhibitory Activity of Monoamine Oxidases
3.16. Inhibitory Activity of Phosphodiesterase 5 (PDE-5)
3.17. Anti-Aromatase and Estrogenic Activity
3.18. Anti-Photoaging Activity
3.19. Skin Penetration Enhancement Activity
3.20. Anti-Angiogenic Activity
3.21. Anti-Coagulant Activity
3.22. Smooth Muscle Relaxant Activity
3.23. Antiparasitic Activity
3.24. Antiviral Activity
3.25. Antifungal Activity
3.26. Antibacterial Activity
3.27. Anti-Biofouling Activity
3.28. Insecticidal–Larvicidal Activity
3.29. Cytotoxicity
3.30. Toxicological Information
3.31. Genotoxic Effect
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rocha, L.; Ribeiro, P.L.; Rapini, A. A lineage-based infrageneric classification of Turnera (Turneraceae). Taxon 2020, 69, 290–306. [Google Scholar] [CrossRef]
- Soriano-Melgar, L.d.A.A.; Alcaraz-Meléndez, L.; Méndez-Rodríguez, L.C.; Puente, M.E.; Rivera-Cabrera, F.; Zenteno-Savín, T. Antioxidant and trace element content of damiana (Turnera diffusa Willd) under wild and cultivated conditions in semi-arid zones. Ind. Crops Prod. 2012, 37, 321–327. [Google Scholar] [CrossRef]
- Ankli, A.; Sticher, O.; Heinrich, M. Medical Ethnobotany of the Yucatec Maya: Healers’ Consensus as a Quantitative Criterion. Econ. Bot. 1999, 53, 144–160. [Google Scholar] [CrossRef]
- Szewczyk, K.; Zidorn, C. Ethnobotany, phytochemistry, and bioactivity of the genus Turnera (Passifloraceae) with a focus on damiana—Turnera diffusa. J. Ethnopharmacol. 2014, 152, 424–443. [Google Scholar] [CrossRef]
- POWO. Plants of the World Online. 2023. Available online: http://www.plantsoftheworldonline.org/ (accessed on 11 October 2023).
- Forman, H.J.; Zhang, H. Targeting oxidative stress in disease: Promise and limitations of antioxidant therapy. Nat. Rev. Drug Discov. 2021, 20, 689–709. [Google Scholar] [CrossRef] [PubMed]
- Wong-Paz, J.E.; Muñiz-Márquez, D.B.; Aguilar-Zárate, P.; Rodríguez-Herrera, R.; Aguilar, C.N. Microplate Quantification of Total Phenolic Content from Plant Extracts Obtained by Conventional and Ultrasound Methods. Phytochem. Anal. 2014, 25, 439–444. [Google Scholar] [CrossRef]
- Wong-Paz, J.E.; Contreras-Esquivel, J.C.; Muñiz-Marquez, D.; Belmares, R.; Rodriguez, R.; Flores, P.; Aguilar, C.N. Microwave-assisted extraction of phenolic antioxidants from semiarid plants. Am. J. Agric. Biol. Sci. 2014, 9, 299–310. [Google Scholar] [CrossRef]
- Soriano-Melgar, L.d.A.A.; Alcaraz-Meléndez, L.; Méndez-Rodríguez, L.C.; Puente, M.E.; Rivera-Cabrera, F.; Zenteno-Savín, T. Antioxidant responses of damiana (Turnera diffusa Willd) to exposure to artificial ultraviolet (UV) radiation in an in vitro model; part I; UV-C radiation. Nutr. Hosp. 2014, 29, 1109–1115. [Google Scholar] [CrossRef]
- Soriano-Melgar, L.d.A.A.; Alcaraz-Meléndez, L.; Méndez-Rodríguez, L.C.; Puente, M.E.; Rivera-Cabrera, F.; Zenteno-Savín, T. Antioxidant responses of damiana (Turnera diffusa Willd) to exposure to artificial ultraviolet (UV) radiation in an in vitro model; part II.; UV-B radiation. Nutr. Hosp. 2014, 29, 1116–1122. [Google Scholar] [CrossRef]
- Wong-Paz, J.E.; Contreras-Esquivel, J.C.; Rodríguez-Herrera, R.; Carrillo-Inungaray, M.L.; López, L.I.; Nevárez-Moorillón, G.V.; Aguilar, C.N. Total phenolic content, in vitro antioxidant activity and chemical composition of plant extracts from semiarid Mexican region. Asian Pac. J. Trop. Med. 2015, 8, 104–111. [Google Scholar] [CrossRef]
- Wong Paz, J.E.; Muñiz Márquez, D.B.; Martínez Ávila, G.C.; Belmares Cerda, R.E.; Aguilar, C.N. Ultrasound-assisted extraction of polyphenols from native plants in the Mexican desert. Ultrason. Sonochem. 2015, 22, 474–481. [Google Scholar] [CrossRef] [PubMed]
- Stashenko, E.; Cordoba, Y.; Martinez, J. Oxygen radical absorption capacity of Lippia origanoides (thymol and phellandrene chemotypes) and Turnera diffusa essential oils and extracts. Planta Med. 2015, 81, PW_199. [Google Scholar] [CrossRef]
- Kalimuthu, K.; Chinnadurai, V.; Prabakaran, R.; Subramaniam, P.; Juliet, Y.S. Antimicrobial, Antioxidant and Anticancer Activities of Turnera ulmifolia (Yellow Alder) Callus. J. Pharm. Res. Int. 2016, 9, 1–12. [Google Scholar] [CrossRef]
- Urbizu-González, A.L.; Castillo-Ruiz, O.; Martínez-Ávila, G.C.; Torres-Castillo, J.A. Natural variability of essential oil and antioxidants in the medicinal plant Turnera diffusa. Asian Pac. J. Trop. Med. 2017, 10, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Esquivel-Gutiérrez, E.R.; Alcaraz-Meléndez, L.; Salgado-Garciglia, R.; Saavedra-Molina, A. Antioxidant effects of damiana (Turnera diffusa Willd. ex Schult.) in kidney mitochondria from streptozotocin-diabetic rats. Nat. Prod. Res. 2018, 32, 2840–2843. [Google Scholar] [CrossRef]
- Pío-León, J.F.; Nieto-Garibay, A.; León-de la Luz, J.L.; Delgado-Vargas, F.; Vega-Aviña, R.; Ortega Rubio, A. Wild plants consumed as recreational teas by ranchers in Baja California Sur, Mexico. Acta Bot. Mex. 2018, 123, 7–19. [Google Scholar] [CrossRef]
- Tsaltaki, C.; Katsouli, M.; Kekes, T.; Chanioti, S.; Tzia, C. Comparison study for the recovery of bioactive compounds from Tribulus terrestris, Panax ginseng, Gingko biloba, Lepidium meyenii, Turnera diffusa and Withania somnifera by using microwave-assisted, ultrasound-assisted and conventional extraction methods. Ind. Crops Prod. 2019, 142, 111875:1–111875:9. [Google Scholar] [CrossRef]
- Aguirre Crespo, F.J.; Cerino Pérez, E.; Valdovinos Estrella, J.; Maldonado Velazquez, M.; Ortega Morales, B.; Zamora Crecencio, P.; Hernández Nuñez, E.; Estrada Soto, S. Vasorelaxant and antioxidant activity of some medicinal plants from Campeche, Mexico. Pharmacogn. Mag. 2021, 17, 23–30. [Google Scholar] [CrossRef]
- Kim, M.; Ha, L.K.; Oh, S.; Fang, M.; Zheng, S.; Bellere, A.D.; Jeong, J.; Yi, T.H. Antiphotoaging Effects of Damiana (Turnera diffusa) Leaves Extract via Regulation AP-1 and Nrf2/ARE Signaling Pathways. Plants 2022, 11, 1486. [Google Scholar] [CrossRef]
- Saravanan, M.; Senthilkumar, P.; Chinnadurai, V.; Murugesan Sakthivel, K.; Rajeshkumar, R.; Pugazhendhi, A. Antiangiogenic, anti-inflammatory and their antioxidant activities of Turnera subulata Sm. (Turneraceae). Process Biochem. 2020, 89, 71–80. [Google Scholar] [CrossRef]
- Delgado-Montemayor, C.; Cordero-Pérez, P.; Torres-González, L.; Salazar-Cavazos, M.D.L.L.; Saucedo, A.L.; Paniagua-Vega, D.; Waksman-Minsky, N.H. Development of a Hepatoprotective Herbal Drug from Turnera diffusa. Evid.-Based Complement. Altern. Med. 2022, 2022, 5114948:1–5114948:10. [Google Scholar] [CrossRef] [PubMed]
- Velez-Huerta, J.; Ramirez-Cabrera, M.A.; Gonzalez-Santiago, O.; Favela-Hernandez, J.; Arredondo-Espinoza, E.; Balderas-Renteria, I. Neuroprotective Activity of Datura inoxia and Turnera diffusa Extracts in an in vitro Model of Neurotoxicity. Pharmacogn. Mag. 2022, 18, 470–475. [Google Scholar]
- Rebouças, E.d.L.; da Silva, A.W.; Coutinho Rodrigues, M.; Amâncio Ferreira, M.K.; Silva Mendes, F.R.; Machado Marinho, M.; Machado Marinho, E.; Ramos Pereira, L.; Feitosa de Araújo, J.I.; Gomes da Silva, J.Y.; et al. Antinociceptive, anti-inflammatory and hypoglycemic activities of the ethanolic Turnera subulata Sm. flower extract in adult zebrafish (Danio rerio). J. Biomol. Struct. Dyn. 2022, 40, 13062–13074. [Google Scholar] [CrossRef] [PubMed]
- Viel, A.M.; Figueiredo, C.C.M.; Granero, F.O.; Silva, L.P.; Ximenes, V.F.; Godoy, T.M.; Quintas, L.E.M.; Gonçalves da Silva, R.M. Antiglycation, antioxidant and cytotoxicity activities of crude extract of Turnera ulmifolia L. before and after microencapsulation process. J. Pharm. Biomed. Anal. 2022, 219, 114975:1–114975:8. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Becerril, M.; Ginera, P.; Silva-Jara, J.; Macias, A.; Velazquez-Carriles, C.; Alcaraz-Meléndez, L.; Angulo, C. Assessment of chemical, biological and immunological properties of “Damiana de California” Turnera diffusa Willd extracts in Longfin yellowtail (Seriola rivoliana) leukocytes. Fish Shellfish Immunol. 2020, 100, 418–426. [Google Scholar] [CrossRef]
- Bernardo, J.; Malheiro, I.; Videira, R.A.; Valentão, P.; Santos, A.C.; Veiga, F.; Andrade, P.B. Trichilia catigua and Turnera diffusa extracts: In vitro inhibition of tyrosinase, antiglycation activity and effects on enzymes and pathways engaged in the neuroinflammatory process. J. Ethnopharmacol. 2021, 271, 113865:1–113865:8. [Google Scholar] [CrossRef]
- Bathula, N.K.; Chowdhury, B. Hepatoprotective activity of ethanolic and aqueous extract of Turnera aphrodisiaca leaves against CCl4-induced liver injury in rats. Int. J. Basic Clin. Pharmacol. 2019, 8, 1758–1765. [Google Scholar] [CrossRef]
- El-Demerdash, F.M.; Jebur, A.B.; Nasr, H.M.; Hamid, H.M. Modulatory effect of Turnera diffusa against testicular toxicity induced by fenitrothion and/or hexavalent chromium in rats. Environ. Toxicol. 2019, 34, 330–339. [Google Scholar] [CrossRef]
- Tousson, E.; Hafez, E.; Zaki, S.; Gad, A.; Elgharabawy, R.M. Evaluation of the testicular protection conferred by damiana (Turnera diffusa Willd.) against amitriptyline-induced testicular toxicity, DNA damage and apoptosis in rats. Biomed. Pharmacother. 2020, 132, 110819:1–110819:8. [Google Scholar] [CrossRef]
- Miles, E.A.; Calder, P.C. Effects of Citrus Fruit Juices and Their Bioactive Components on Inflammation and Immunity: A Narrative Review. Front. Immunol. 2021, 12, 712608:1–712608:18. [Google Scholar] [CrossRef]
- Lawal, O.A.; Avoseh, O.N.; Tinubu, O.J.; Ogunwande, I.A.; Ascrizzi, R.; Flamini, G.; Mtunzi, F.M. Anti-nociceptive Property, Anti-inflammatory Activity and Constituents of Essential Oils from the Leaves and Stem Bark of Turnera diffusa Wild (Passifloraceae) Growing in Nigeria. J. Biol. Act. Prod. Nat. 2020, 10, 473–483. [Google Scholar] [CrossRef]
- Cabral Souza, N.; Medeiros de Oliveira, J.; da Silva Morrone, M.; D´Olveira Albanus, R.; do Socorro Medeiros Amarante, M.; da Silva Camillo, C.; Zucolotto Langassner, S.M.; Pens Gelain, D.; Fonseca Moreira, J.C.; Siqueira Dalmolin, R.J.; et al. Turnera subulata Anti-Inflammatory Properties in Lipopolysaccharide-Stimulated RAW 264.7 Macrophages. J. Med. Food 2016, 19, 922–930. [Google Scholar] [CrossRef] [PubMed]
- Harun, N.H.; Septama, A.W.; Ahmad, W.A.N.W.; Suppian, R. Immunomodulatory effects and structure-activity relationship of botanical pentacyclic triterpenes: A review. Chin. Herb. Med. 2020, 12, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Soromou, L.W.; Jiang, L.; Wei, M.; Chen, N.; Huo, M.; Chu, X.; Zhong, W.; Wu, Q.; Baldé, A.; Deng, X.; et al. Protection of mice against lipopolysaccharide-induced endotoxic shock by pinocembrin is correlated with regulation of cytokine secretion. J. Immunotoxicol. 2014, 11, 56–61. [Google Scholar] [CrossRef]
- Duarte da Luz, J.R.; Barbosa, E.A.; Silva do Nascimento, T.E.; Augusto de Rezende, A.; Galvão Ururahy, M.A.; da Silva Brito, A.; Araujo-Silva, G.; López, J.A.; das Graças Almeida, M. Chemical Characterization of Flowers and Leaf Extracts Obtained from Turnera subulata and Their Immunomodulatory Effect on LPS-Activated RAW 264.7 Macrophages. Molecules 2022, 27, 1084. [Google Scholar] [CrossRef]
- Arman, M.; Chowdhury, K.A.A.; Bari, M.S.; Khan, M.F.; Huq, M.M.A.; Haque, M.A.; Capasso, R. Hepatoprotective potential of selected medicinally important herbs: Evidence from ethnomedicinal, toxicological and pharmacological evaluations. Phytochem. Rev. 2022, 21, 1863–1886. [Google Scholar] [CrossRef]
- Brito, N.J.; López, J.A.; do Nascimento, M.A.; Macêdo, J.B.; Silva, G.A.; Oliveira, C.N.; de Rezende, A.A.; Brandão-Neto, J.; Schwarz, A.; Almeida, M. Antioxidant activity and protective effect of Turnera ulmifolia Linn. var. elegans against carbon tetrachloride-induced oxidative damage in rats. Food Chem. Toxicol. 2012, 50, 4340–4347. [Google Scholar] [CrossRef]
- Bathula, N.K.; Chowdhury, B. Pharmacological Evaluation of Turnera aphrodisiaca Leaves for Hepatoprotective Activity in Albino Rats. Nat. Volatiles Essent. Oils 2021, 8, 5275–5283. [Google Scholar]
- Hasan, A.F.; Mutar, T.F.; Tousson, E.M.; Felemban, S.G. Therapeutic Effects of Turnera diffusa Extract Against Amitriptyline-Induced Toxic Hepatic Inflammation. Online J. Biol. Sci. 2021, 21, 395–408. [Google Scholar] [CrossRef]
- Rodriguez-Rodriguez, D.R.; Lozano-Sepulveda, S.A.; Delgado-Montemayor, C.; Waksman, N.; Cordero-Perez, P.; Rivas-Estilla, A.M. Turnera diffusa extract attenuates profibrotic, extracellular matrix and mitochondrial markers in activated human hepatic stellate cells (HSC). Ann. Hepatol. 2021, 22, 100281:1–100281:9. [Google Scholar] [CrossRef]
- Delgado-Montemayor, C.; Perez-Meseguer, J.; Salazar-Aranda, R.; Cordero-Perez, P.; Waksman, N. Hepatodamianol as hepatoprotective constituent of Turnera diffusa. Pak. J. Pharm. Sci 2023, 36, 1553–1559. [Google Scholar]
- Mody, H.; Ramakrishnan, V.; Chaar, M.; Lezeau, J.; Rump, A.; Taha, K.; Lesko, L.; Ait-Oudhia, S. A Review on Drug-Induced Nephrotoxicity: Pathophysiological Mechanisms, Drug Classes, Clinical Management, and Recent Advances in Mathematical Modeling and Simulation Approaches. Clin. Pharmacol. Drug Dev. 2020, 9, 896–909. [Google Scholar] [CrossRef] [PubMed]
- Hasan, A.F.; Hameed, H.M.; Tousson, E.; Massoud, A.; Atta, F.; Youssef, H.; Hussein, Y. Role of Oral Supplementation of Damiana (Turnera diffusa) Reduces the Renal Toxicity, Apoptosis and DNA Damage Associated with Amitriptyline Administration in Rats. Biomed. Pharmacol. J. 2022, 15, 1245–1253. [Google Scholar] [CrossRef]
- Matzkin, M.E.; Calandra, R.S.; Rossi, S.P.; Bartke, A.; Frungieri, M.B. Hallmarks of Testicular Aging: The challenge of Anti-Inflammatory and Antioxidant Therapies Using Natural and/or Pharmacological Compounds to Improve the Physiopathological Status of the Aged Male Gonad. Cells 2021, 10, 3114. [Google Scholar] [CrossRef] [PubMed]
- Manfo, F.; Nantia, E.; Mathur, P. Effect of Environmental Contaminants on Mammalian Testis. Curr. Mol. Pharmacol. 2014, 7, 119–135. [Google Scholar] [CrossRef]
- Kumar, G.G.; Kilari, E.K.; Nelli, G.; Bin Salleh, N. Oral administration of Turnera diffusa Willd. ex Schult. extract ameliorates steroidogenesis and spermatogenesis impairment in the testes of rats with type-2 diabetes mellitus. J. Ethnopharmacol. 2023, 314, 116638:1–116638:16. [Google Scholar] [CrossRef]
- Huang, X.; Li, N.; Pu, Y.; Zhang, T.; Wang, B. Neuroprotective Effects of Ginseng Phytochemicals: Recent Perspectives. Molecules 2019, 24, 2939. [Google Scholar] [CrossRef]
- Gomes Bezerra, A.; Soubhi Smaili, S.; Silva Lopes, G.; Araújo Carlini, E. Effects of Panax ginseng, Turnera diffusa and Heteropterys tomentosa extracts on hippocampal apoptosis of aged rats. Einstein 2013, 11, 163–167. [Google Scholar] [CrossRef]
- Bernardo, J.; Ferreres, F.; Gil-Izquierdo, Á.; Valentão, P.; Andrade, P.B. Medicinal species as MTDLs: Turnera diffusa Willd. Ex Schult inhibits CNS enzymes and delays glutamate excitotoxicity in SH-SY5Y cells via oxidative damage. Food Chem. Toxicol. 2017, 106, 466–476. [Google Scholar] [CrossRef]
- Domínguez-Avila, J.A.; Salazar-López, N.J.; Montiel-Herrera, M.; Martínez-Martínez, A.; Villegas-Ochoa, M.A.; González-Aguilar, G.A. Phenolic compounds can induce systemic and central immunomodulation, which result in a neuroprotective effect. J. Food Biochem. 2022, 46, e14260:1–e14260:12. [Google Scholar] [CrossRef]
- Chen, S.; Zou, H. Lipoxygenase Metabolism: Critical Pathways in Microglia-mediated Neuroinflammation and Neurodevelopmental Disorders. Neurochem. Res. 2022, 47, 3213–3220. [Google Scholar] [CrossRef]
- Bernardo, J.; Santos, A.C.; Videira, R.A.; Valentão, P.; Veiga, F.; Andrade, P. Trichilia catigua and Turnera diffusa phyto-phospholipid nanostructures: Physicochemical characterization and bioactivity in cellular models of induced neuroinflammation and neurotoxicity. Int. J. Pharm. 2022, 620, 121774:1–121774:11. [Google Scholar] [CrossRef]
- Cole, J.B.; Florez, J.C. Genetics of diabetes mellitus and diabetes complications. Nat. Rev. Nephrol. 2020, 16, 377–390. [Google Scholar] [CrossRef] [PubMed]
- Parra-Naranjo, A.; Delgado-Montemayor, C.; Fraga-López, A.; Castañeda-Corral, G.; Salazar-Aranda, R.; Acevedo-Fernández, J.J.; Waksman, N. Acute Hypoglycemic and Antidiabetic Effect of Teuhetenone A Isolated from Turnera diffusa. Molecules 2017, 22, 599. [Google Scholar] [CrossRef]
- Esquivel-Gutiérrez, E.R.; Alcaraz-Meléndez, L.; Hernández-Herrera, R.; Torres, A.; Rodríguez-Jaramillo, C. Effects of damiana (Turnera diffusa; var. diffusa and var. aphrodisiaca) on diabetic rats. Acta Univ. 2018, 28, 84–92. [Google Scholar] [CrossRef]
- Adame-Miranda, S.J.; Granados-Guzmán, G.; Silva-Mares, D.A.; Acevedo-Fernández, J.J.; Waksman-Minsky, N.; Salazar-Aranda, R. Evaluation of antihyperglycemic activity of plants in northeast Mexico. Cell. Mol. Biol. 2021, 67, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Hernández Bautista, R.J.; Mahmoud, A.M.; Königsberg, M.; López Díaz Guerrero, N.E. Obesity: Pathophysiology, monosodium glutamate-induced model and anti-obesity medicinal plants. Biomed. Pharmacother. 2019, 111, 503–516. [Google Scholar] [CrossRef]
- Ruxton, C.H.S.; Kirkwood, L.; Mcmillan, B.; St John, D.; Evans, C.E.L. Effectiveness of a herbal supplement (ZotrimTM) for weight management. Br. Food J. 2007, 109, 416–428. [Google Scholar] [CrossRef]
- Dorantes-Barron, A.M.; Vigueras Villasenor, R.M.; Mayagoitia-Novales, L.; Martinez-Mota, L.; Gutiérrez-Perez, O.; Estrada-Reyes, R. Neurobehavioral and toxicological effects of an aqueous extract of Turnera diffusa Willd (Turneraceae) in mice. J. Ethnopharmacol. 2019, 236, 50–62. [Google Scholar] [CrossRef]
- Yam, M.F.; Loh, Y.C.; Tan, C.S.; Adam, S.K.; Manan, N.A.; Basir, R. General Pathways of Pain Sensation and the Major Neurotransmitters Involved in Pain Regulation. Int. J. Mol. Sci. 2018, 19, 2164. [Google Scholar] [CrossRef]
- Antônio, M.A.; Souza Brito, A.R. Oral anti-inflammatory and anti-ulcerogenic activities of a hydroalcoholic extract and partitioned fractions of Turnera ulmifolia (Turneraceae). J. Ethnopharmacol. 1998, 61, 215–228. [Google Scholar] [CrossRef]
- Kumar, S.; Madaan, R.; Sharma, A. Pharmacological evaluation of bioactive principle of Turnera aphrodisiaca. Indian J. Pharm. Sci. 2008, 70, 740–744. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Depression and Other Common Mental Disorders. Global Health Estimates. Available online: https://apps.who.int/iris/handle/10665/254610 (accessed on 18 August 2023).
- Sandroni, P. Aphrodisiacs past and present: A historical review. Clin. Auton. Res. 2001, 11, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Shah, G.R.; Chaudhari, M.V.; Patankar, S.B.; Pensalwar, S.V.; Sabale, V.P.; Sonawane, N.A. Evaluation of a multi-herb supplement for erectile dysfunction: A randomized double-blind, placebo-controlled study. BMC Complement. Altern. Med. 2012, 12, 155:1–155:9. [Google Scholar] [CrossRef] [PubMed]
- Palacios, S.; Soler, E.; Ramírez, M.; Lilue, M.; Khorsandi, D.; Losa, F. Effect of a multi-ingredient based food supplement on sexual function in women with low sexual desire. BMC Womens. Health 2019, 19, 58:1–58:7. [Google Scholar] [CrossRef] [PubMed]
- Kuchernig, J. Sexuelle Unlust—Wirkmechanismen des natürlichen Aphrodisiakums Damiana (Turnera diffusa). J. Gynäkologische Endokrinol. 2021, 24, 120–127. [Google Scholar] [CrossRef]
- Rotmann, A.-R.; Mordhorst, N.; Klein, T.; Kassen, A. Nicht-Interventionelle Studie zum Einfluss eines Damiana-Präparates auf die Verbesserung der Symptomatik des Mangels oder Verlusts von sexuellem Verlangen bei Frauen. Z. Phyther. 2021, 42, 241–248. [Google Scholar] [CrossRef]
- Gurley, B.J. Pharmacokinetic herb-drug interactions (part 1): Origins, mechanisms, and the impact of botanical dietary supplements. Planta Med. 2012, 78, 1478–1489. [Google Scholar] [CrossRef]
- Husain, I.; Dale, O.R.; Martin, K.; Gurley, B.J.; Adams, S.J.; Avula, B.; Chittiboyina, A.G.; Khan, I.A.; Khan, S.I. Screening of medicinal plants for possible herb-drug interactions through modulating nuclear receptors, drug-metabolizing enzymes and transporters. J. Ethnopharmacol. 2023, 301, 115822:1–115822:14. [Google Scholar] [CrossRef]
- Twarda-Clapa, A.; Olczak, A.; Białkowska, A.M.; Koziołkiewicz, M. Advanced Glycation End-Products (AGEs): Formation, Chemistry, Classification, Receptors, and Diseases Related to AGEs. Cells 2022, 11, 1312. [Google Scholar] [CrossRef]
- Dariya, B.; Nagaraju, G.P. Advanced glycation end products in diabetes, cancer and phytochemical therapy. Drug Discov. Today 2020, 25, 1614–1623. [Google Scholar] [CrossRef]
- Duarte, P.; Cuadrado, A.; León, R. Monoamine Oxidase Inhibitors: From Classic to New Clinical Approaches. In Reactive Oxygen Species. Handbook of Experimental Pharmacology; Schmidt, H.H.H.W., Ghezzi, P., Cuadrado, A., Eds.; Springer: Cham, Switzerland, 2021; Volume 264, pp. 229–259. ISBN 978-3-030-68510-2. [Google Scholar]
- Chaurasiya, N.D.; Zhao, J.; Pandey, P.; Doerksen, R.J.; Muhammad, I.; Tekwani, B.L. Selective Inhibition of Human Monoamine Oxidase B by Acacetin 7-Methyl Ether Isolated from Turnera diffusa (Damiana). Molecules 2019, 24, 810. [Google Scholar] [CrossRef] [PubMed]
- Anand Ganapathy, A.; Hari Priya, V.M.; Kumaran, A. Medicinal plants as a potential source of Phosphodiesterase-5 inhibitors: A review. J. Ethnopharmacol. 2021, 267, 113536:1–113536:36. [Google Scholar] [CrossRef]
- Feistel, B.; Walbroel, B.; Benedek, B. Damiana (Turnera diffusa Willd.)—A traditionally used aphrodisiac as modern PDE-5 inhibitor. Planta Med. 2010, 76, P002. [Google Scholar] [CrossRef]
- Kharb, R.; Haider, K.; Neha, K.; Yar, M.S. Aromatase inhibitors: Role in postmenopausal breast cancer. Arch. Pharm. 2020, 353, e2000081:1–e2000081:20. [Google Scholar] [CrossRef] [PubMed]
- Korani, M. Aromatase inhibitors in male: A literature review. Med. Clin. Pract. 2023, 6, 100356:1–100356:5. [Google Scholar] [CrossRef]
- Zava, D.T.; Dollbaum, C.M.; Blen, M. Estrogen and Progestin Bioactivity of Foods, Herbs, and Spices. Proc. Soc. Exp. Biol. Med. 1998, 217, 369–378. [Google Scholar] [CrossRef]
- Powers, C.N.; Setzer, W.N. A molecular docking study of phytochemical estrogen mimics from dietary herbal supplements. In Silico Pharmacol. 2015, 3, 4:1–4:63. [Google Scholar] [CrossRef]
- Salminen, A.; Kaarniranta, K.; Kauppinen, A. Photoaging: UV radiation-induced inflammation and immunosuppression accelerate the aging process in the skin. Inflamm. Res. 2022, 71, 817–831. [Google Scholar] [CrossRef]
- Kováčik, A.; Kopečnáand, M.; Vávrová, K. Permeation enhancers in transdermal drug delivery: Benefits and limitations. Expert Opin. Drug Deliv. 2020, 17, 145–155. [Google Scholar] [CrossRef]
- Carreño, H.; Stashenko, E.E.; Escobar, P. Essential Oils Distilled from Colombian Aromatic Plants and Their Constituents as Penetration Enhancers for Transdermal Drug Delivery. Molecules 2023, 28, 2872. [Google Scholar] [CrossRef]
- Olejarz, W.; Kubiak-Tomaszewska, G.; Chrzanowska, A.; Lorenc, T. Exosomes in Angiogenesis and Anti-angiogenic Therapy in Cancers. Int. J. Mol. Sci. 2020, 21, 5840. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Bai, R.; Liu, Y.; Zhang, X.; Kan, J.; Jin, C. Isolation, structural characterization and bioactivities of naturally occurring polysaccharide–polyphenolic conjugates from medicinal plants—A reivew. Int. J. Biol. Macromol. 2018, 107, 2242–2250. [Google Scholar] [CrossRef] [PubMed]
- Duarte da Luz, J.R.; Silva do Nascimento, T.E.; Fernandes de Morais, L.V.; Menezes da Cruz, A.K.; Augusto de Rezende, A.; Brandão Neto, J.; Galvão Ururahy, M.A.; Ducati Luchessi, A.; López, J.A.; Oliveira Rocha, H.A.; et al. Thrombin Inhibition: Preliminary Assessment of the Anticoagulant Potential of Turnera subulata (Passifloraceae). J. Med. Food 2019, 22, 384–392. [Google Scholar] [CrossRef]
- Hnatyszyn, O.; Moscatelli, V.; Garcia, J.; Rondina, R.; Costa, M.; Arranz, C.; Balaszczuk, A.; Ferraro, G.; Coussio, J.D. Argentinian plant extracts with relaxant effect on the smooth muscle of the corpus cavernosum of Guinea pig. Phytomedicine 2003, 10, 669–674. [Google Scholar] [CrossRef][Green Version]
- Brenner, D.M.; Lacy, B.E. Antispasmodics for Chronic Abdominal Pain: Analysis of North American Treatment Options. Am. J. Gastroenterol. 2021, 116, 1587–1600. [Google Scholar] [CrossRef]
- Garcia, M.; Monzote, L.; Montalvo, A.M.; Scull, R. Screening of medicinal plants against Leishmania amazonensis. Pharm. Biol. 2010, 48, 1053–1058. [Google Scholar] [CrossRef]
- Santos, K.K.A.; Matias, E.F.F.; Sobral-Souza, C.E.; Tintino, S.R.; Morais-Braga, M.F.B.; Guedes, G.M.M.; Rolón, M.; Vega, C.; de Arias, A.R.; Costa, J.G.M.; et al. Evaluation of the anti-Trypanosoma and anti-Leishmania activity of Mentha arvensis and Turnera ulmifolia. Bol. Latinoam. Caribe Plantas Med. Aromat. 2012, 11, 147–153. [Google Scholar]
- Oliveira, A.F.; Costa Junior, L.M.; Lima, A.S.; Silva, C.R.; Ribeiro, M.N.S.; Mesquista, J.W.C.; Rocha, C.Q.; Tangerina, M.M.P.; Vilegas, W. Anthelmintic activity of plant extracts from Brazilian savanna. Vet. Parasitol. 2017, 236, 121–127. [Google Scholar] [CrossRef]
- Silva-Trujillo, L.; Quintero-Rueda, E.; Stashenko, E.E.; Conde-Ocazionez, S.; Rondón-Villarreal, P.; Ocazionez, R.E. Essential Oils from Colombian Plants: Antiviral Potential against Dengue Virus Based on Chemical Composition, In Vitro and In Silico Analyses. Molecules 2022, 27, 6844. [Google Scholar] [CrossRef]
- Correa-Royero, J.; Tangarife, V.; Duran, C.; Stashenko, E.; Mesa-Arango, A. In vitro antifungal activity and cytotoxic effect of essential oils and extracts of medicinal and aromatic plants against Candida krusei and Aspergillus fumigatus. Rev. Bras. Farmacogn. 2010, 20, 734–741. [Google Scholar] [CrossRef]
- Baez-Parra, K.M.; Soto-Beltran, M.; Lopez-Cuevas, O.; Basilio Heredia, J.; Alcaraz-Melendez, L.; Angulo-Escalante, M.A. In Vitro Antimicrobial Activity of Methanolic and Hexanic Extracts of Turnera diffusa Against Common Urinary Pathogens. Rev. Bio. Cienc. 2019, 6, e670:1–e670:17. [Google Scholar] [CrossRef]
- Ong, C.; Chan, Y.S.; Chan, S.M.; Chan, M.W.; The, E.L.; Soh, C.L.D.; Khoo, K.S.; Ong, H.C.; Sit, N.W. Antifungal, antibacterial and cytotoxic activities of non-indigenous medicinal plants naturalised in Malaysia. Farmacia 2020, 68, 687–696. [Google Scholar] [CrossRef]
- Saravanan, M.; Senthilkumar, P.; Kalimuthu, K.; Chinnadurai, V.; Vasantharaj, S.; Pugazhendhi, A. Phytochemical and pharmacological profiling of Turnera subulata Sm., a vital medicinal herb. Ind. Crops Prod. 2018, 124, 822–833. [Google Scholar] [CrossRef]
- Tapia-Quirós, P.; Martínez-Téllez, M.Á.; Ávila-Quezada, G.D.; Vargas-Arispuro, I. Inhibition of fungal endo-1,3-B-glucanase by phenols isolated from Turnera diffusa: An alternative for conventional antifungals. Rev. Mex. Fitopatol. 2020, 38, 160–169. [Google Scholar] [CrossRef]
- Sri, P.S.B.J.K.; Kumar, M.P.; Padmavathy, S. Antitumor Activity of Turnera subulata Sm. (Turneraceae) in Hep G2 Cancer Cell Line. J. Pharm. Res. Int. 2021, 33, 191–199. [Google Scholar] [CrossRef]
- Dilkin, E.R.S.; Matias, R.; Oliveira, A.K.M.; Corrêa, B.O. Fungitoxic effect and phytochemical characteristics of Brazilian Cerrado weeds against Rhizoctonia solani and Macrophomina phaseolina fungi. Braz. J. Biol. 2022, 84, e263114:1–e263114:8. [Google Scholar] [CrossRef]
- Domfeh, S.A.; Kyeremeh, G.; Belifini, M. Evaluation of Anti- Candida albicans Activities of Herbal Preparations Sold at the Kumasi Central Market in the Ashanti Region of Ghana. Evid.-Based Complement. Altern. Med. 2023, 2023, 6162532:1–6162532:7. [Google Scholar] [CrossRef]
- Andrade-Pinheiro, J.C.; Sobral de Souza, C.E.; Ribeiro, D.A.; Silva, A.; da Silva, V.B.; dos Santos, A.T.L.; Juno Alencar Fonseca, V.; de Macêdo, D.G.; da Cruz, R.P.; Almeida-Bezerra, J.W.; et al. LC-MS Analysis and Antifungal Activity of Turnera subulata Sm. Plants 2023, 12, 415. [Google Scholar] [CrossRef]
- Silva, A.C.O.; Santana, E.F.; Saraiva, A.M.; Coutinho, F.N.; Castro, R.H.A.; Pisciottano, M.N.C.; Amorim, E.L.C.; Albuquerque, U.P. Which Approach Is More Effective in the Selection of Plants with Antimicrobial Activity? Evid.-Based Complement. Altern. Med. 2013, 2013, 308980. [Google Scholar] [CrossRef]
- Freitas, C.L.A.; Santos, F.F.P.; Dantas-Junior, O.M.; Inacio, V.V.; Matias, E.F.F.; Quintans-Junior, L.J.; Aguiar, J.J.S.; Coutinho, H.D.M. Enhancement of antibiotic activity by phytocompounds of Turnera subulata. Nat. Prod. Res. 2020, 34, 2384–2388. [Google Scholar] [CrossRef]
- Snowden, R.; Harrington, H.; Morrill, K.; Jeane, L.; Garrity, J.; Orian, M.; Lopez, E.; Rezaie, S.; Hassberger, K.; Familoni, D.; et al. A Comparison of the Anti-Staphylococcus aureus Activity of Extracts from Commonly Used Medicinal Plants. J. Altern. Complement. Med. 2014, 20, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Barrios Martínez, S.X.; Stashenko, E.E.; Ocazionez Jimenez, R.E.; Fuentes Lorenzo, J.L. Antibacterial activity of essential oils of plants growing in Colombia and its effect on the activity of β-lactam antibiotics. Rev. Cuba. Farm. 2021, 54, e627:1–e627:15. [Google Scholar]
- Fernandes, M.G.; Gomes, R.A.; Brito-Filho, S.G.; Silva-Filho, R.N.; Agra, M.F.; Falcão-Silva, V.S.; Siqueira-Junior, J.P.; Vieira, M.A.R.; Marques, M.O.M. Characterization and anti-staphylococcal activity of the essential oil from Turnera subulata Sm. Rev. Bras. Plantas Med. 2014, 16, 534–538. [Google Scholar] [CrossRef]
- Arantes, V.P.; Sato, D.N.; Vilegas, W.; Santos, L.C.; Leite, C.Q.F. Brazilian cerrado plants active against Mycobacterium fortuitum. J. Basic Appl. Pharm. Sci. 2005, 26, 195–198. [Google Scholar]
- Coutinho, H.D.M.; Costa, J.G.M.; Lima, E.O.; Falcão-Silva, V.S.; Siqueira-Júnior, J.P. Increasing of the aminoglicosyde antibiotic activity against a multidrug-resistant E. coli by Turnera ulmifolia L. and chlorpromazine. Biol. Res. Nurs. 2010, 11, 332–335. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.N.; Trentin, D.d.S.; Zimmer, K.R.; Treter, J.; Brandelli, C.L.; Frasson, A.P.; Tasca, T.; Da Silva, A.G.; Da Silva, M.V.; Macedo, A.J. Anti-infective effects of Brazilian Caatinga plants against pathogenic bacterial biofilm formation. Pharm. Biol. 2015, 53, 464–468. [Google Scholar] [CrossRef][Green Version]
- Qian, P.Y.; Cheng, A.; Wang, R.; Zhang, R. Marine biofilms: Diversity, interactions and biofouling. Nat. Rev. Microbiol. 2022, 20, 671–684. [Google Scholar] [CrossRef]
- Agostini, V.O.; Macedo, A.J.; Muxagata, E.; da Silva, M.V.; Pinho, G.L.L. Non-toxic antifouling potential of Caatinga plant extracts: Effective inhibition of marine initial biofouling. Hydrobiologia 2020, 847, 45–60. [Google Scholar] [CrossRef]
- Hari, I.; Mathew, N. Larvicidal activity of selected plant extracts and their combination against the mosquito vectors Culex quinquefasciatus and Aedes aegypti. Environ. Sci. Pollut. Res. 2018, 25, 9176–9185. [Google Scholar] [CrossRef]
- de Andrade Porto, K.R.; Motti, P.R.; Yano, M.; Roel, A.R.; Cardoso, C.A.L.; Matias, R. Screening of plant extracts and fractions on Aedes aegypti larvae found in the state of Mato Grosso do Sul (linnaeus, 1762) (culicidae). An. Acad. Bras. Cienc. 2017, 89, 895–906. [Google Scholar] [CrossRef] [PubMed]
- Ríos, N.; Stashenko, E.E.; Duque, J.E. Evaluation of the insecticidal activity of essential oils and their mixtures against Aedes aegypti (Diptera: Culicidae). Rev. Bras. Entomol. 2017, 61, 307–311. [Google Scholar] [CrossRef]
- Duque, J.E.; Urbina, D.L.; Vesga, L.C.; Ortiz-Rodríguez, L.A.; Vanegas, T.S.; Stashenko, E.E.; Mendez-Sanchez, S.C. Insecticidal activity of essential oils from American native plants against Aedes aegypti (Diptera: Culicidae): An introduction to their possible mechanism of action. Sci. Rep. 2023, 13, 2989:1–2989:15. [Google Scholar] [CrossRef] [PubMed]
- Gavanji, S.; Bakhtari, A.; Famurewa, A.C.; Othman, E.M. Cytotoxic Activity of Herbal Medicines as Assessed in Vitro: A Review. Chem. Biodivers. 2023, 20, e202201098:1–e202201098:27. [Google Scholar] [CrossRef] [PubMed]
- Velandia, S.A.; Flechas, M.; Stashenko, E.; Ocazionez, R.E. Proposal to select essential oils from Colombian plants for research based on its cytoxicity. Vitae 2016, 23, 18–29. [Google Scholar] [CrossRef]
- Avelino-Flores, M.d.C.; Cruz-Lopez, M.d.C.; Jimenez-Montejo, F.E.; Reyes-Leyva, J. Cytotoxic Activity of the Methanolic Extract of Turnera diffusa Willd on Breast Cancer Cells. J. Med. Food 2015, 18, 299–305. [Google Scholar] [CrossRef]
- Bueno, J.; Escobar, P.; Martínez, J.R.; Leal, S.M.; Stashenko, E.E. Composition of three essential oils, and their mammalian cell toxicity and antimycobacterial activity against drug resistant-tuberculosis and nontuberculous mycobacteria strains. Nat. Prod. Commun. 2011, 6, 1743–1748. [Google Scholar] [CrossRef]
- Gomes Bezerra, A.; Negri, G.; Duarte-Almeida, J.M.J.M.; Soubhi Smaili, S.; Araújo Carlini, E.; Bezerra, A.G.; Negri, G.; Duarte-Almeida, J.M.J.M.; Smaili, S.S.; Carlini, E.A. Phytochemical analysis of hydroethanolic extract of Turnera diffusa Willd and evaluation of its effects on astrocyte cell death. Einstein 2016, 14, 56–63. [Google Scholar] [CrossRef]
- Willer, J.; Jöhrer, K.; Greil, R.; Zidorn, C.; Çiçek, S.S.S.S. Cytotoxic Properties of Damiana (Turnera diffusa) Extracts and Constituents and A Validated Quantitative UHPLC-DAD Assay. Molecules 2019, 24, 855. [Google Scholar] [CrossRef]
- Cragg, G.M.; Boyd, M.R.; Cardellina, J.H., 2nd; Newman, D.J.; Snader, K.M.; McCloud, T.G. Ethnobotany and drug discovery: The experience of the US National Cancer Institute. Ciba Found Symp. 1994, 185, 178–190. [Google Scholar] [CrossRef]
- Boik, J. Background for Parts I and II. In Natural Compounds in Cancer Therapy. Promising Nontoxic Antitumor Agents from Plants & Other Natural Sources; Oregon Medical Press, LLC: Princeton, MN, USA, 2001; pp. 1–10. [Google Scholar]
- Bezerra, A.G.; Mendes, F.R.; Tabach, R.; Carlini, E.A. Effects of a hydroalcoholic extract of Turnera diffusa in tests for adaptogenic activity. Rev. Bras. Farmacogn. 2011, 21, 121–127. [Google Scholar] [CrossRef]
- Gracioso, J.d.S.; Vilegas, W.; Hiruma-Lima, C.A.; Souza Brito, A.R. Effects of tea from Turnera ulmifolia L. on mouse gastric mucosa support the Turneraceae as a new source of antiulcerogenic drugs. Biol. Pharm. Bull. 2002, 25, 487–491. [Google Scholar] [CrossRef] [PubMed]
- Bathula, N.K.; Chowdhury, B. Assessment of acute and subacute oral toxicity elicited by ethanolic extract of Turnera aphrodisiaca leaves in albino rats. Asian J. Pharm. Clin. Res. 2020, 13, 144–148. [Google Scholar] [CrossRef]
- Coe, F.G.; Parikh, D.M.; Johnson, C.A.; Anderson, G.J. The good and the bad: Alkaloid screening and brineshrimp bioassays of aqueous extracts of 31 medicinal plants of eastern Nicaragua. Pharm. Biol. 2012, 50, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Lopes Da Costa, L.J.; Ribeiro Dantas, V.C.; Pereira De Souza, T.; Lira Soares, L.A.; das Graças Almeida, M.; Brandão Neto, J.; Schwarz, A.; Dantas, V.C.R.; De Souza, T.P.; Soares, L.A.L.; et al. Investigation of Turnera ulmifolia effects in pregnant rats and offspring. Pharm. Biol. 2009, 47, 1071–1077. [Google Scholar] [CrossRef]
- Bardoloi, A.; Soren, A.D. Genotoxicity induced by medicinal plants. Bull. Natl. Res. Cent. 2022, 46, 119:1–119:11. [Google Scholar] [CrossRef]
- Senes-Lopes, T.F.; Lopez, J.A.; Souza Do Amaral, V.; Brandao-Neto, J.; Augusto de Rezende, A.; Duarte da Luz, J.R.; da Rosa Guterres, Z.; das Gracas Almeida, M. Genotoxicity of Turnera subulata and Spondias mombin × Spondias tuberosa Extracts from Brazilian Caatinga Biome. J. Med. Food 2018, 21, 372–379. [Google Scholar] [CrossRef]

| Method | Species | Part of the Plant | Extract/Compound | % Inh. | EC50 (µg/mL) | Ref. |
|---|---|---|---|---|---|---|
| ABTS | T. diffusa | H | HA | - | 381.2 | [20] |
| H, T | AQ | 60.8–72.3 | - | [15] | ||
| H, T | HA | 30 | - | [11] | ||
| T. subulata | C | AE | - | 250 | [21] | |
| C | CH | - | 415 | [21] | ||
| C | E | - | 250 | [21] | ||
| C | HX | - | 93.7 | [21] | ||
| C | M | - | 500 | [21] | ||
| C | PE | - | 450 | [21] | ||
| DPPH | T. diffusa | A | Fr. AE:M | - | 26.9–90.3 | [22] |
| A | Hepatodamianol | - | 4.7 | [22] | ||
| H | E | 36.2 | - | [23] | ||
| H | HA | - | 305.4 | [20] | ||
| H | M | - | 28.9 | [19] | ||
| H, T | A | 77.9–78.1 | - | [7] | ||
| H, T | HA | 78.6–77.8 | - | [7] | ||
| H, T | HA | 62 | - | [12] | ||
| H, T | HA | 76 | - | [11] | ||
| T. ulmifolia | C | AE | - | 847.3 | [21] | |
| C | CH | - | 45.6 | [21] | ||
| C | E | - | 10.2 | [21] | ||
| C | HX | - | 991.6 | [21] | ||
| C | M | - | 10.4 | [21] | ||
| C | PE | - | 358 | [21] | ||
| CL | E | 12.5–53.2 | - | [14] | ||
| CL | M | 15.2–56.2 | - | [14] | ||
| F | E | - | 25.4 | [24] | ||
| H, R, F | HA | 70.6 | - | [25] | ||
| H2O2 | T. subulata | C | AE | - | 120 | [21] |
| C | CH | - | 91.2 | [21] | ||
| C | E | - | 106.2 | [21] | ||
| C | HX | - | 102 | [21] | ||
| C | M | - | 25 | [21] | ||
| C | PE | - | 425 | [21] | ||
| NO | T. subulata | C | AE | - | 31.9 | [21] |
| C | CH | - | 24.6 | [21] | ||
| C | E | - | 36.1 | [21] | ||
| C | HX | - | 111 | [21] | ||
| C | M | - | 25.1 | [21] | ||
| C | PE | - | 338 | [21] | ||
| OH• | T. subulata | C | AE | - | 24.4 | [21] |
| C | CH | - | 23.7 | [21] | ||
| C | E | - | 21.8 | [21] | ||
| C | HX | - | 30.8 | [21] | ||
| C | M | - | 120 | [21] | ||
| C | PE | - | 42.5 | [21] | ||
| β-carotene bleaching assay | T. diffusa | H | M | - | 233.1 | [19] |
| Method | Species | Part of the Plant | Extract/Compound | AC | Notes | Ref. |
|---|---|---|---|---|---|---|
| ABTS | T. diffusa var. aphrodisiaca | C | AQ | 666.5 | µmol ET/g | [17] |
| ABTS | T. diffusa var. diffusa | C | AQ | 446.6 | µmol ET/g | [17] |
| DPPH | T. diffusa | H | HA | 145–377 | mg Trolox/dry weight | [18] |
| NO | T. ulmifolia | H, R, F | HA | 34.1 | µM/mL nitrite formed | [25] |
| ORAC | T. diffusa | H, T | EO | 813 | µmol Trolox/g | [13] |
| FRAP | T. diffusa | H, T | AQ | 18.5–21.3 | mg GAE/g | [15] |
| FRAP | T. ulmifolia | H, R, F | HA | 628.4 | µM TE/g | [25] |
| Species | Part of the Plant | Extract/Compound | Total Phenolics | Ref. |
|---|---|---|---|---|
| T. diffusa | H | AQ | 9.2 | [26] |
| E | 395.3 | [18] | ||
| HA | 101.5 | [20] | ||
| M | 0.4 | [26] | ||
| M | 0.3 | [19] | ||
| T. diffusa | H, T | AQ | 23.9–33.8 | [15] |
| HA | 4.7–5.4 | [8] | ||
| HA | 2.5–4.7 | [11] | ||
| T. diffusa var. aphrodisiaca | C | AQ | 20.8 | [17] |
| T. diffusa var. diffusa | C | AQ | 8.3 | [17] |
| T. subulata | C | AE | 18 | [21] |
| CH | 24.1 | [21] | ||
| E | 21.2 | [21] | ||
| HX | 12 | [21] | ||
| M | 17.1 | [21] | ||
| PE | 8 | [21] | ||
| T. subulata | F | E | 1007.4 | [24] |
| T. ulmifolia | H, R, F | HA | 604.8 | [25] |
| Species | Part of the Plant | Extract/Compound | EC50 µg/mL | Inh.% | Ref. |
|---|---|---|---|---|---|
| T. diffusa | B | AQ | 283.39 | - | [27] |
| T. diffusa | H, T | HA | - | 23 | [12] |
| Ref. | Species | Part of the Plant | Extract/ Compound | Dosage (mg/kg p.o.) | Agent of Damage (Dose) | Tissue Analyzed | Parameter | Levels | Notes |
|---|---|---|---|---|---|---|---|---|---|
| [28] | T. aphrodisiaca | H | AQ | 200–400 | CCl4 (0.7 mL/kg i.p.) | Kidney | CAT | 2.7–2.9 | U/mg protein |
| SOD | 5.9–7.1 | U/mg protein | |||||||
| [28] | T. aphrodisiaca | H | E | 200–400 | CCl4 (0.7 mL/kg i.p.) | Kidney | CAT | 2.4–2.5 | U/mg protein |
| SOD | 6.9–7–6 | U/mg protein | |||||||
| [16] | T. diffusa | A | HA | 150 | STZ (65 mg/kg i.p.) | Liver mitochondria | MDA | 3.3 | nmol/mg protein |
| NO | 0.4 | µmol/mg protein | |||||||
| [29] | T. diffusa | H | - | 50 | Fenitrothion, K2Cr2O7 (68 mg/kg, 2 mg/kg) | Testicle | CAT | 6 | U/mg protein |
| H2O2 | 78.7 | µmol/g tissue | |||||||
| GPx | 1.7 | U/mg | |||||||
| GR | 15.9 | U/mg | |||||||
| GSH | 1.6 | mmol/mg protein | |||||||
| GST | 0.4 | µmol/h/mg protein | |||||||
| SOD | 51.3 | U/mg protein | |||||||
| TBARS | 24 | nmol/g tissue | |||||||
| [30] | T. diffusa | H | AQ | 80 | Amitriptyline (70 mg/kg i.p.) | Testicle | GPx | 6.8 | U/mg |
| GR | 16 | U/mg | |||||||
| GSH | 2.6 | mmol | |||||||
| GST | 0.5 | µmol/h/mg protein | |||||||
| SOD | 67.9 | U/mg protein | |||||||
| TBARS | 22.5 | nmol/g tissue |
| Ref. | Fungal Species | Plant Species | Part of the Plant | Extract/ Compound | MIC (µg/mL) | MFC (µg/mL) |
|---|---|---|---|---|---|---|
| [94] | C. krusei ATCC 6258 | T. diffusa | A, R | EO | 90 | - |
| [94] | Aspergillus fumigatus ATCC 204305 | T. diffusa | A, R | EO | 500 | - |
| [95] | C. albicans | T. diffusa var. aphrodisiaca | H | HX | 50 | 50 |
| M | 50 | 50 | ||||
| [95] | C. albicans | T. diffusa var. diffusa | H | HX | 50 | 50 |
| M | 50 | 50 | ||||
| [96] | C. albicans ATCC 90028 | T. subulata | A | AE | 310 | 2500 |
| AQ | 1250 | NA | ||||
| CH | 630 | NA | ||||
| E | 1250 | NA | ||||
| HX | 310 | 2500 | ||||
| M | 1250 | NA | ||||
| [96] | C. krusei ATCC 6258 | T. subulata | A | AE | 160 | 310 |
| AQ | 630 | 2500 | ||||
| CH | 310 | 630 | ||||
| E | 630 | 2500 | ||||
| HX | 40 | 160 | ||||
| M | 630 | 2500 | ||||
| [96] | C. parapsilosis ATCC 22019 | T. subulata | A | AE | 310 | 1250 |
| AQ | 2500 | NA | ||||
| CH | 630 | 630 | ||||
| E | 1250 | NA | ||||
| HX | 160 | 630 | ||||
| M | 1250 | NA | ||||
| [96] | C. neoformans ATCC 90112 | T. subulata | A | AE | 80 | 310 |
| AQ | 160 | NA | ||||
| CH | 40 | 160 | ||||
| E | 160 | NA | ||||
| HX | 20 | 160 | ||||
| M | 160 | NA | ||||
| [96] | Trichophyton interdigitale ATCC 9533 | T. subulata | A | CH | 630 | NA |
| HX | 310 | NA |
| Ref. | Bacteria Species | Plant Species | Part of the Plant | Extract/ Compound | MIC (µg/mL) | MBC (µg/mL) |
|---|---|---|---|---|---|---|
| [96] | Acinetobacter baumannii ATCC 19606 | T. subulata | A | E | 1250 | 2500 |
| M | 1250 | NA | ||||
| AE | 2500 | NA | ||||
| [96] | Bacillus cereus ATCC 11778 | T. subulata | A | M | 2500 | NA |
| HX | 630 | NA | ||||
| AE | 630 | NA | ||||
| CH | 630 | NA | ||||
| [95] | E. coli ATCC 25922 | T. diffusa var. aphrodisiaca | H | M | 50 | 100 |
| HX | 50 | 100 | ||||
| T. diffusa var. diffusa | H | M | 100 | 100 | ||
| HX | 50 | 100 | ||||
| [104] | E. coli MR 27 | T. subulata | H | M | 512 | - |
| [104] | E. coli ATCC 25922 | T. subulata | H | M | 406 | - |
| [96] | E. coli ATCC 35218 | T. subulata | A | M | 2500 | 2500 |
| HX | 310 | 310 | ||||
| AE | 630 | 630 | ||||
| CH | 630 | 630 | ||||
| E | 2500 | 2500 | ||||
| [95] | E. faecalis ATCC 29212 | T. diffusa var. aphrodisiaca | H | HX | 1500 | NA |
| M | 3000 | 3000 | ||||
| T. diffusa var. diffusa | H | HX | 1500 | NA | ||
| M | 3000 | 3000 | ||||
| [95] | K. pneumoniae ATCC 13883 | T. diffusa var. aphrodisiaca | H | HX | 50 | 50 |
| M | 50 | 100 | ||||
| T. diffusa var. diffusa | H | HX | 50 | 50 | ||
| M | 50 | 100 | ||||
| [96] | K. pneumoniae ATCC 13883 | T. subulata | A | HX | 630 | 630 |
| CH | 630 | 630 | ||||
| AE | 630 | 630 | ||||
| E | 2500 | 2500 | ||||
| M | 2500 | 2500 | ||||
| [96] | P. aeruginosa ATCC 27853 | T. subulata | A | CH | 630 | NA |
| AE | 630 | NA | ||||
| E | 630 | NA | ||||
| M | 630 | NA | ||||
| HX | 1250 | NA | ||||
| AQ | 1250 | NA | ||||
| [104] | P. aeruginosa ATCC 27853 | T. subulata | H | M | 512 | - |
| [104] | P. aeruginosa MR 31 | T. subulata | H | M | 512 | - |
| [105] | S. aureus ATCC 11632 | T. diffusa | H | E | 300 | - |
| [106] | S. aureus ATCC 25923 | T. diffusa | - | AE | 780–3120 | - |
| [106] | S. aureus ATCC 33592 | T. diffusa | - | AE | 780–3120 | - |
| [95] | S. aureus ATCC 25923 | T. diffusa var. aphrodisiaca | H | HX | 500 | 3000 |
| M | 2000 | 2000 | ||||
| T. diffusa var. diffusa | H | HX | 500 | 3000 | ||
| M | 3000 | 3000 | ||||
| [104] | S. aureus MR 35 | T. subulata | H | M | 512 | - |
| [104] | S. aureus ATCC 25923 | T. subulata | H | M | 512 | - |
| [96] | S. aureus ATCC 6538 | T. subulata | A | HX | 630 | 630 |
| CH | 1250 | 1250 | ||||
| AE | 1250 | 2500 | ||||
| [107] | S. aureus ATCC 25923 | T. subulata | A | EO | 200 | - |
| [107] | S. aureus MRSA | T. subulata | A | EO | 25–1600 | - |
| [107] | S. aureus effluxing strain | T. subulata | A | EO | 800–1600 | - |
| [108] | M. fortuitum ATCC 6841 | T. ulmifolia | F | CH | 125 | - |
| H | DM | 125 | - |
| Method | Ref. | Species | Part of the Plant | Extract/Compound | Cell Line | CC50 (µg/mL) |
|---|---|---|---|---|---|---|
| MTT | [118] | T. diffusa | - | EO | B16F10 | 83.9 * |
| B16F10 | 161.3 ** | |||||
| HEK293 | 143.2 ** | |||||
| HEK293 | 152.5 * | |||||
| HELA | 109.5 ** | |||||
| HepG2 | 186.5 * | |||||
| HepG2 | 199.3 ** | |||||
| MCF7 | 139.9 ** | |||||
| Vero | 90.5 * | |||||
| Vero | 135 ** | |||||
| [94] | T. diffusa | A, R | EO | Vero | 52.2 | |
| [50] | T. diffusa | H | AQ | SH-SY5Y | >500 | |
| [119] | T. diffusa | H, T | M | C-33 A | 45.1 | |
| Fibroblasts | 63.24 | |||||
| HepG2 | 43.87 | |||||
| MDA-MB-231 | 30.67 | |||||
| SiHa | 50.14 | |||||
| T-47D | 54.02 | |||||
| [120] | T. diffusa | H, T | EO | THP-1 | >100 | |
| Vero | 88.1 | |||||
| [14] | T. subulata | CL | E | MCF 7 | >300 | |
| M | MCF 7 | >300 | ||||
| [87] | T. subulata | H | HA | 3T3 | >1000 | |
| HEK293 | >1000 | |||||
| Fr. AE | 3T3 | >1000 | ||||
| HEK293 | >1000 | |||||
| [20] | T. subulata | H | HA | HaCat | 100 | |
| HDF | 100 | |||||
| [25] | T. ulmifolia | T, H, F | HA | HFF1 | >1000 | |
| MTT and Alamar blue | [36] | T. subulata | F | AQ | RAW 264.7 | >500 |
| HA | RAW 264.7 | >500 | ||||
| H | AQ | RAW 264.7 | >500 | |||
| HA | RAW 264.7 | >500 | ||||
| Neutral red uptake | [96] | T. subulata | A | AE | Vero | 245.4 |
| AQ | Vero | nd | ||||
| CH | Vero | 281.4 | ||||
| E | Vero | nd | ||||
| HX | Vero | 471.3 | ||||
| M | Vero | nd | ||||
| Total macromolecular content | [62] | T. ulmifolia | - | Fr. E | V79 | 465,000 |
| Assays | Experimental Design | Species | Part of the Plant | Extract/ Compound | Dosage | LD50 | Notes | Ref. |
|---|---|---|---|---|---|---|---|---|
| Acute Toxicity | Mice | T. diffusa | A | HA | 2 g/kg i.p | - | Death at day 3 | [125] |
| A | HA | 5 g/kg p.o. | - | No signs of toxicity over 14 days | [125] | |||
| H | AQ | 10–1000 mg/kg | - | No signs of toxicity or animal death, no S.D. was observed in renal and liver function parameters vs. control. | [60] | |||
| T. ulmifolia | A | AQ | 500–5000 mg/kg p.o. | - | There were no signs of toxicity or death. No S:D. in organ weight, feeding or feces vs. control. | [126] | ||
| C | HA | 5000 mg/kg p.o. | 7.82 g/kg | - | [62] | |||
| Acute Toxicity | Rats | T. subulata | H | HA | 500 and 2000 mg/kg p.o. | - | No changes in behavior or death of animals were observed. No S.D. was observed in hematological parameters or weight vs. control. | [87] |
| Fr. EA | 500 and 2000 mg/kg p.o. | - | No changes in behavior or death of animals were observed. No S.D. was observed in hematological parameters or weight vs. control. | [87] | ||||
| T. aphrodisiaca | H | E | 5000 mg/kg p.o. | >5000 mg/kg | No signs of toxicity or changes in biochemical and hematological parameters were observed. | [127] | ||
| Subacute Toxicity | Rattus norvegicus | T. aphrodisiaca | H | E | 250–1000 mg/kg p.o | >5000 mg/kg | No signs of toxicity or changes in biochemical and hematological parameters were observed. | [127] |
| Lethality assay | Artemia salina | T. ulmifolia | H | AQ | 500–10,000 µg/mL | 6650 µg/mL | - | [128] |
| T. hermannioides | H | AQ | 1 mg/mL | - | No changes in swimming pattern. | [112] | ||
| T. subulata | F | E | 1000 µg/mL | >1000 µg/mL | - | [24] | ||
| Chaetoceros calcintrans | T. hermannioides | H | AQ | 1 mg/mL | - | No significant growth inhibition. | [112] | |
| Danio rerio | T. subulata | F | E | 40 mg/kg p.o. | >40 mg/mL | - | [24] | |
| Nitokra sp. | T. hermannioides | H | AQ | 1 mg/mL | - | No decrease in survival rate. | [112] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Parra-Naranjo, A.; Delgado-Montemayor, C.; Salazar-Aranda, R.; Waksman-Minsky, N. Bioactivity of the Genus Turnera: A Review of the Last 10 Years. Pharmaceuticals 2023, 16, 1573. https://doi.org/10.3390/ph16111573
Parra-Naranjo A, Delgado-Montemayor C, Salazar-Aranda R, Waksman-Minsky N. Bioactivity of the Genus Turnera: A Review of the Last 10 Years. Pharmaceuticals. 2023; 16(11):1573. https://doi.org/10.3390/ph16111573
Chicago/Turabian StyleParra-Naranjo, Aída, Cecilia Delgado-Montemayor, Ricardo Salazar-Aranda, and Noemí Waksman-Minsky. 2023. "Bioactivity of the Genus Turnera: A Review of the Last 10 Years" Pharmaceuticals 16, no. 11: 1573. https://doi.org/10.3390/ph16111573
APA StyleParra-Naranjo, A., Delgado-Montemayor, C., Salazar-Aranda, R., & Waksman-Minsky, N. (2023). Bioactivity of the Genus Turnera: A Review of the Last 10 Years. Pharmaceuticals, 16(11), 1573. https://doi.org/10.3390/ph16111573







