Involvement of PI3K/HIF-1α/c-MYC/iNOS Pathway in the Anticancer Effect of Suaeda vermiculata in Rats
Abstract
1. Introduction
2. Results
2.1. Effect of SV, in the Presence or Absence of NDEA, on AFP, CEA, and PI3K Serum Levels
2.2. Effect of SV, in the Presence or Absence of NDEA, on the Liver Tissue Levels of MDA, CAT, SOD, and GSH
2.3. Effect of SV on EGFR and VEGFR Serum Level and Gene Expression of c-MYC and HIF-1α in the Presence or Absence of NDEA
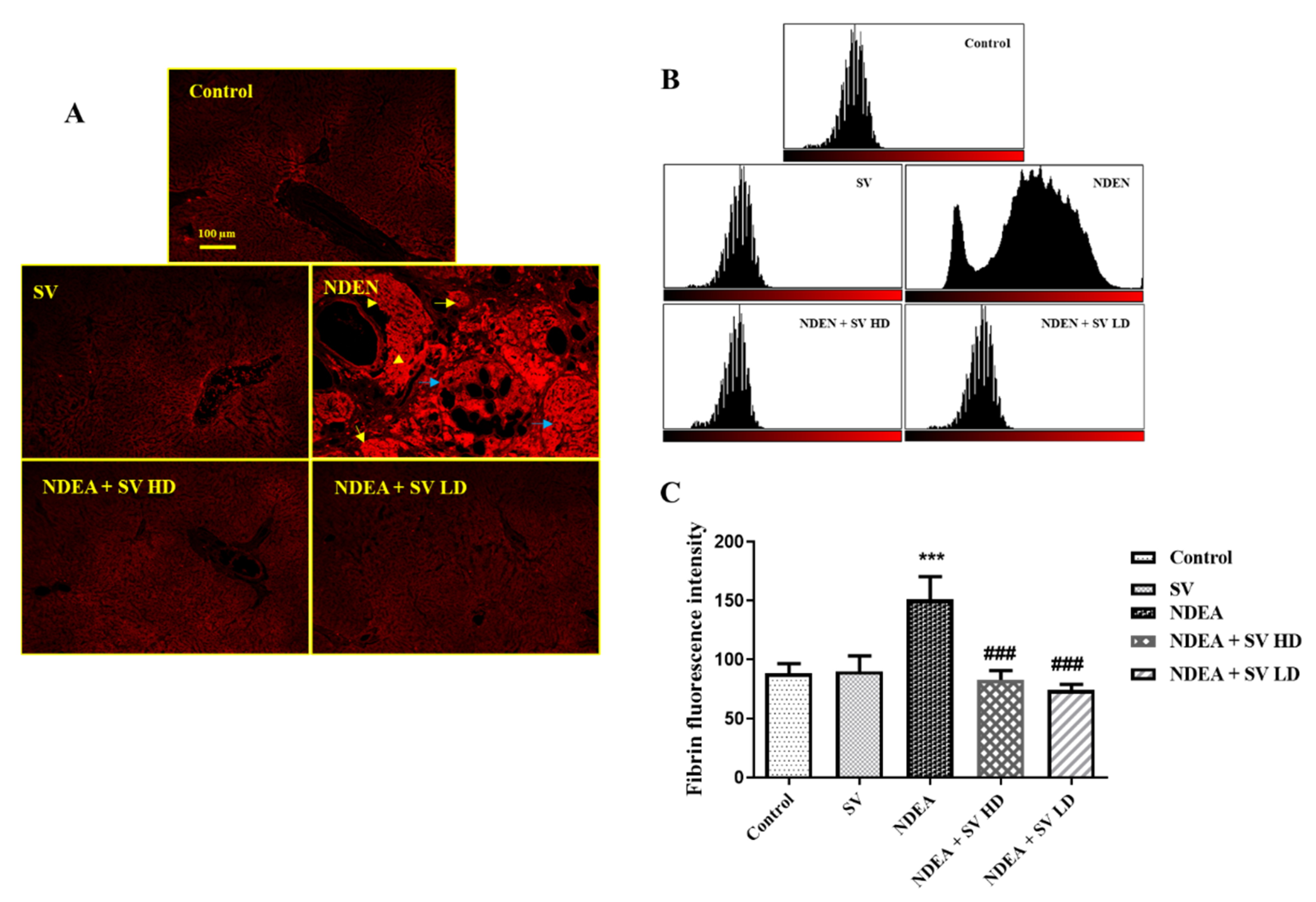
2.4. Effect of NDEA with or without SV on Fibrinogen Protein Expression
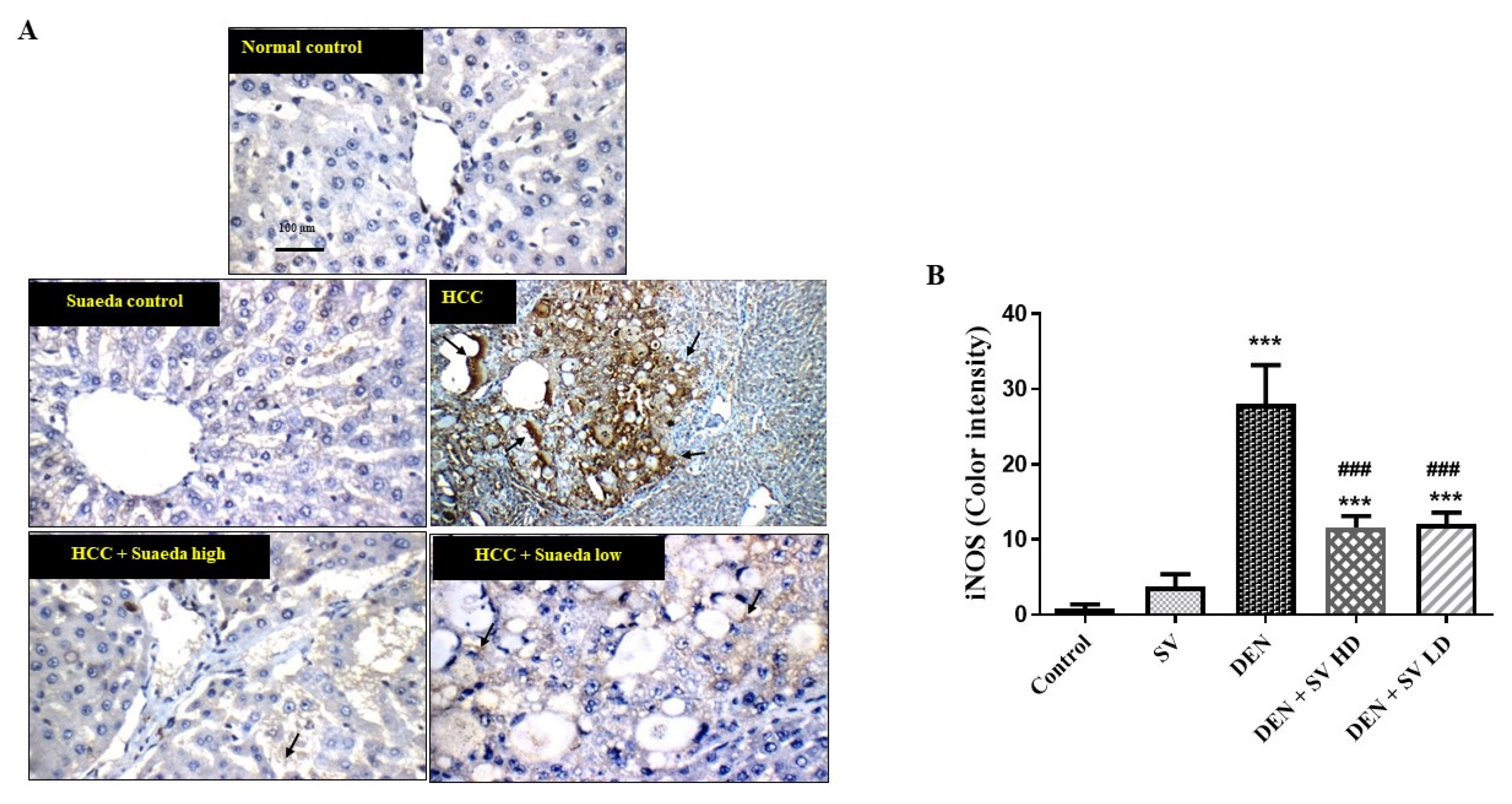
2.5. Effect of SV on iNOS Proteins Expression in Liver Tissues with or without NDEA
3. Discussion
4. Materials and Methods
4.1. Chemicals, Kits, and Antibodies
4.2. Plant Material
4.3. Animals
4.4. Experimental Study Protocol
4.5. Assessment of Serum Tumor Biomarkers
4.6. Assessment of the Products of Polyunsaturated Fatty Acids Peroxidation, MDA, and Antioxidant Biomarkers
4.7. Assessment of EGFR, PI3K, and VEGFR Levels
4.8. RNA Extraction for Real Time-PCR
4.9. Fluorescence Microscopic Examination
4.10. Immunohistochemical Analysis
4.11. Statistical Investigation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ozougwu, J.C. Physiology of the Liver. Int. J. Res. Pharm. Biosci. 2017, 4, 13–24. [Google Scholar]
- Capelletti, M.M.; Manceau, H.; Puy, H.; Peoc’h, K. Ferroptosis in Liver Diseases: An Overview. Int. J. Mol. Sci. 2020, 21, 4908. [Google Scholar] [CrossRef]
- Cichoż-Lach, H. Oxidative Stress as a Crucial Factor in Liver Diseases. World J. Gastroenterol. 2014, 20, 8082. [Google Scholar] [CrossRef]
- Akbari, B.; Baghaei-Yazdi, N.; Bahmaie, M.; Mahdavi Abhari, F. The Role of Plant-derived Natural Antioxidants in Reduction of Oxidative Stress. BioFactors 2022, 48, 611–633. [Google Scholar] [CrossRef]
- Del Campo, J.A.; Gallego, P.; Grande, L. Role of Inflammatory Response in Liver Diseases: Therapeutic Strategies. World J. Hepatol. 2018, 10, 1. [Google Scholar] [CrossRef]
- Viktorova, J.; Stranska-Zachariasova, M.; Fenclova, M.; Vitek, L.; Hajslova, J.; Kren, V.; Ruml, T. Complex Evaluation of Antioxidant Capacity of Milk Thistle Dietary Supplements. Antioxidants 2019, 8, 317. [Google Scholar] [CrossRef]
- Das, R.; Mitra, S.; Tareq, A.M.; Emran, T.B.; Hossain, M.J.; Alqahtani, A.M.; Alghazwani, Y.; Dhama, K.; Simal-Gandara, J. Medicinal Plants Used against Hepatic Disorders in Bangladesh: A Comprehensive Review. J. Ethnopharmacol. 2022, 282, 114588. [Google Scholar] [CrossRef]
- Ali, S.A.; Sharief, N.H.; Mohamed, Y.S. Hepatoprotective Activity of Some Medicinal Plants in Sudan. Evid.-Based Complement. Altern. Med. 2019, 2019, 2196315. [Google Scholar] [CrossRef]
- Majeed, A.; Muhammad, Z.; Ahmad, H.; Hayat, S.S.S.; Inayat, N.; Siyyar, S. Nigella sativa L.: Uses in Traditional and Contemporary Medicines—An Overview. Acta Ecol. Sin. 2021, 41, 253–258. [Google Scholar] [CrossRef]
- Rolnik, A.; Olas, B. The Plants of the Asteraceae Family as Agents in the Protection of Human Health. Int. J. Mol. Sci. 2021, 22, 3009. [Google Scholar] [CrossRef]
- Venmathi Maran, B.A.; Iqbal, M.; Gangadaran, P.; Ahn, B.-C.; Rao, P.V.; Shah, M.D. Hepatoprotective Potential of Malaysian Medicinal Plants: A Review on Phytochemicals, Oxidative Stress, and Antioxidant Mechanisms. Molecules 2022, 27, 1533. [Google Scholar] [CrossRef]
- Lu, Q.; Tan, S.; Gu, W.; Li, F.; Hua, W.; Zhang, S.; Chen, F.; Tang, L. Phytochemical Composition, Isolation and Hepatoprotective Activity of Active Fraction from Veronica Ciliata against Acetaminophen-Induced Acute Liver Injury via P62-Keap1-Nrf2 Signaling Pathway. J. Ethnopharmacol. 2019, 243, 112089. [Google Scholar] [CrossRef]
- Tafere, G.G.; Tuem, K.B.; Gebre, A.K.; Balasubramaniam, R. In Vitro Antioxidant and in Vivo Hepatoprotective Activities of Root Bark Extract and Solvent Fractions of Croton Macrostachyus Hochst. Ex Del.(Euphorbiaceae) on Paracetamol-Induced Liver Damage in Mice. J. Exp. Pharmacol. 2020, 12, 301–311. [Google Scholar]
- Mohammed, H.A. The Valuable Impacts of Halophytic Genus Suaeda Mathsemicolon Nutritional, Chemical, and Biological Values. Med. Chem. 2020, 16, 1044–1057. [Google Scholar] [CrossRef]
- Al-Omar, M.S.; Sajid, M.S.M.; Alnasyan, N.S.; Almansour, B.S.; Alruthaya, R.M.; Khan, R.A.; Mohammed, S.A.A.; Al-Damigh, A.M.; Mohammed, H.A. The Halophytic Plant, Suaeda vermiculata Forssk Extracts Reduce the Inflamed Paw Edema and Exert Potential Antimicrobial Activity. Pak. J. Bot. 2021, 53, 351–356. [Google Scholar] [CrossRef]
- Mohammed, H.A.; Almahmoud, S.A.; Arfeen, M.; Srivastava, A.; El-Readi, M.Z.; Ragab, E.A.; Shehata, S.M.; Mohammed, S.A.A.; Mostafa, E.M.; El-khawaga, H.A.; et al. Phytochemical Profiling, Molecular Docking, and in Vitro Anti-Hepatocellular Carcinoid Bioactivity of Suaeda vermiculata Extracts. Arab. J. Chem. 2022, 15, 103950. [Google Scholar] [CrossRef]
- Mohammed, S.A.A.; Eldeeb, H.M.; Mohammed, H.A.; Al-Omar, M.S.; Almahmoud, S.A.; El-Readi, M.Z.; Ragab, E.A.; Sulaiman, G.M.; Aly, M.S.A.; Khan, R.A. Roles of Suaeda vermiculata Aqueous-Ethanolic Extract, Its Subsequent Fractions, and the Isolated Compounds in Hepatoprotection against Paracetamol-Induced Toxicity as Compared to Silymarin. Oxid. Med. Cell. Longev. 2021, 2021, 174897. [Google Scholar] [CrossRef]
- Mohammed, S.A.A.; Khan, R.A.; El-readi, M.Z.; Emwas, A.H.; Sioud, S.; Poulson, B.G.; Jaremko, M.; Eldeeb, H.M.; Al-omar, M.S.; Mohammed, H.A. Suaeda vermiculata Aqueous-ethanolic Extract-based Mitigation of Ccl4-induced Hepatotoxicity in Rats, and Hepg-2 and Hepg-2/Adr Cell-lines-based Cytotoxicity Evaluations. Plants 2020, 9, 1291. [Google Scholar] [CrossRef]
- Udoh, U.-A.; Sanabria, J.D.; Rajan, P.K.; Banerjee, M.; Schade, M.; Sanabria, J.A.; Smith, G.; Udoh, G.; Sodhi, K.; Pierre, S. Non-Alcoholic Fatty Liver Disease Progression to Non-Alcoholic Steatohepatitis-Related Primary Liver Cancer. Exon Publ. 2021, 3, 55–76. [Google Scholar]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Montella, M.; Crispo, A.; Giudice, A. HCC, Diet and Metabolic Factors: Diet and HCC. Hepat. Mon. 2011, 11, 159. [Google Scholar] [PubMed]
- Idelchik, M.d.P.S.; Begley, U.; Begley, T.J.; Melendez, J.A. Mitochondrial ROS Control of Cancer. Semin. Cancer Biol. 2017, 47, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Moloney, J.N.; Cotter, T.G. ROS Signalling in the Biology of Cancer. Semin. Cell Dev. Biol. 2018, 80, 50–64. [Google Scholar] [CrossRef] [PubMed]
- Marra, M.; Sordelli, I.M.; Lombardi, A.; Lamberti, M.; Tarantino, L.; Giudice, A.; Stiuso, P.; Abbruzzese, A.; Sperlongano, R.; Accardo, M. Molecular Targets and Oxidative Stress Biomarkers in Hepatocellular Carcinoma: An Overview. J. Transl. Med. 2011, 9, 171. [Google Scholar] [CrossRef]
- Anoopraj, R.; Hemalatha, S.; Balachandran, C. A Preliminary Study on Serum Liver Function Indices of Diethylnitrosamine Induced Hepatocarcinogenesis and Chemoprotective Potential of Eclipta Alba in Male Wistar Rats. Vet. World 2014, 7, 439–442. [Google Scholar] [CrossRef][Green Version]
- Kumar, V.; Bhatt, P.C.; Rahman, M.; Kaithwas, G.; Choudhry, H.; Al-Abbasi, F.A.; Anwar, F.; Verma, A. Fabrication, Optimization, and Characterization of Umbelliferone β-D-Galactopyranoside-Loaded PLGA Nanoparticles in Treatment of Hepatocellular Carcinoma: In Vitro and in Vivo Studies. Int. J. Nanomed. 2017, 12, 6747. [Google Scholar] [CrossRef]
- Matsuzawa-Nagata, N.; Takamura, T.; Ando, H.; Nakamura, S.; Kurita, S.; Misu, H.; Ota, T.; Yokoyama, M.; Honda, M.; Miyamoto, K. Increased Oxidative Stress Precedes the Onset of High-Fat Diet–Induced Insulin Resistance and Obesity. Metabolism 2008, 57, 1071–1077. [Google Scholar] [CrossRef]
- Rahman, M.; Al-Ghamdi, S.A.; Alharbi, K.S.; Beg, S.; Sharma, K.; Anwar, F.; Al-Abbasi, F.A.; Kumar, V. Ganoderic Acid Loaded Nano-Lipidic Carriers Improvise Treatment of Hepatocellular Carcinoma. Drug Deliv. 2019, 26, 782–793. [Google Scholar] [CrossRef]
- Unsal, V.; Cicek, M.; Sabancilar, İ. Toxicity of Carbon Tetrachloride, Free Radicals and Role of Antioxidants. Rev. Environ. Health 2021, 36, 279–295. [Google Scholar] [CrossRef]
- Morgis, R.A.; Haan, K.; Schrey, J.M.; Zimmerman, R.M.; Hersperger, A.R. The Epidermal Growth Factor Ortholog of Ectromelia Virus Activates EGFR/ErbB1 and Demonstrates Mitogenic Function in Vitro. Virology 2021, 564, 1–12. [Google Scholar] [CrossRef]
- Chung, Y.; Kim, J.A.; Song, B.; Lee, G.C.; Koh, M.S.; Lee, Y.S.; Lee, S.G.; Suh, D.J. Expression of Transforming Growth Factor-α MRNA in Livers of Patients with Chronic Viral Hepatitis and Hepatocellular Carcinoma. Cancer Interdiscip. Int. J. Am. Cancer Soc. 2000, 89, 977–982. [Google Scholar] [CrossRef]
- Schiffer, E.; Housset, C.; Cacheux, W.; Wendum, D.; Desbois-Mouthon, C.; Rey, C.; Clergue, F.; Poupon, R.; Barbu, V.; Rosmorduc, O. Gefitinib, an EGFR Inhibitor, Prevents Hepatocellular Carcinoma Development in the Rat Liver with Cirrhosis. Hepatology 2005, 41, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, J.-N.; Tang, J.-M.; Kong, X.; Yang, J.-Y.; Zheng, F.; Guo, L.-Y.; Huang, Y.-Z.; Zhang, L.; Tian, L. VEGF Is Essential for the Growth and Migration of Human Hepatocellular Carcinoma Cells. Mol. Biol. Rep. 2012, 39, 5085–5093. [Google Scholar] [CrossRef] [PubMed]
- Dakowicz, D.; Zajkowska, M.; Mroczko, B. Relationship between VEGF Family Members, Their Receptors and Cell Death in the Neoplastic Transformation of Colorectal Cancer. Int. J. Mol. Sci. 2022, 23, 3375. [Google Scholar] [CrossRef] [PubMed]
- Peng, S.; Wang, Y.; Peng, H.; Chen, D.; Shen, S.; Peng, B.; Chen, M.; Lencioni, R.; Kuang, M. Autocrine Vascular Endothelial Growth Factor Signaling Promotes Cell Proliferation and Modulates Sorafenib Treatment Efficacy in Hepatocellular Carcinoma. Hepatology 2014, 60, 1264–1277. [Google Scholar] [CrossRef]
- Bruix, J.; Reig, M.; Sherman, M. Evidence-Based Diagnosis, Staging, and Treatment of Patients with Hepatocellular Carcinoma. Gastroenterology 2016, 150, 835–853. [Google Scholar] [CrossRef]
- Villanueva, A.; Chiang, D.Y.; Newell, P.; Peix, J.; Thung, S.; Alsinet, C.; Tovar, V.; Roayaie, S.; Minguez, B.; Sole, M. Pivotal Role of MTOR Signaling in Hepatocellular Carcinoma. Gastroenterology 2008, 135, 1972–1983. [Google Scholar] [CrossRef]
- Zhu, H.; Yin, J.; Chen, D.; He, S.; Chen, H. Agmatinase Promotes the Lung Adenocarcinoma Tumorigenesis by Activating the NO-MAPKs-PI3K/Akt Pathway. Cell Death Dis. 2019, 10, 854. [Google Scholar] [CrossRef]
- Bao, H.; Ge, Y.; Wang, Z.; Zhuang, S.; Dworkin, L.; Peng, A.; Gong, R. Delayed Administration of a Single Dose of Lithium Promotes Recovery from AKI. J. Am. Soc. Nephrol. 2014, 25, 488–500. [Google Scholar] [CrossRef]
- Kashfi, K. The Dichotomous Role of H2S in Cancer Cell Biology? Deja vu All over Again. Biochem. Pharmacol. 2018, 149, 205–223. [Google Scholar] [CrossRef]
- Granados-Principal, S.; Liu, Y.; Guevara, M.L.; Blanco, E.; Choi, D.S.; Qian, W.; Patel, T.; Rodriguez, A.A.; Cusimano, J.; Weiss, H.L. Inhibition of INOS as a Novel Effective Targeted Therapy against Triple-Negative Breast Cancer. Breast Cancer Res. 2015, 17, 25. [Google Scholar] [CrossRef]
- Yang, D.; Yin, J.; Mishra, S.; Mishra, R.A.J.; Hsu, C.Y. NO-mediated Chemoresistance in C6 Glioma Cells. Ann. N. Y. Acad. Sci. 2002, 962, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K.; Lee, K.R.; Yang, J.H.; Yoo, S.J.; Lee, S.W.; Jang, H.-J.; Park, S.J.; Moon, Y.-S.; Park, J.-W.; Kim, C.-M. Plasma Levels of D-Dimer and Soluble Fibrin Polymer in Patients with Hepatocellular Carcinoma: A Possible Predictor of Tumor Thrombosis. Thromb. Res. 2003, 109, 125–129. [Google Scholar] [CrossRef]
- Huang, G.; Jiang, H.; Lin, Y.; Wu, Y.; Cai, W.; Shi, B.; Luo, Y.; Jian, Z.; Zhou, X. Prognostic Value of Plasma Fibrinogen in Hepatocellular Carcinoma: A Meta-Analysis. Cancer Manag. Res. 2018, 10, 5027. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Guo, H.; Gao, F.; Shan, Q.; Li, J.; Xie, H.; Zhou, L.; Xu, X.; Zheng, S. Fibrinogen and D-dimer Levels Elevate in Advanced Hepatocellular Carcinoma: High Pretreatment Fibrinogen Levels Predict Poor Outcomes. Hepatol. Res. 2017, 47, 1108–1117. [Google Scholar] [CrossRef] [PubMed]
- Fathy, A.H.; Bashandy, M.A.; Bashandy, S.A.E.; Mansour, A.M.; Elsadek, B. Sequential Analysis and Staging of a Diethylnitrosamine-Induced Hepatocellular Carcinoma in Male Wistar Albino Rat Model. Can. J. Physiol. Pharmacol. 2017, 95, 1462–1472. [Google Scholar] [CrossRef]
- Abdel-Bakky, M.S.; Alqasoumi, A.; Altowayan, W.M.; Amin, E.; Darwish, M.A. Simvastatin Mitigates Streptozotocin-Induced Type 1 Diabetes in Mice through Downregulation of ADAM10 and ADAM17. Life Sci. 2022, 289, 120224. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohammed, H.A.; Ewees, M.G.; Mahmoud, N.I.; Ali, H.M.; Amin, E.; Abdel-Bakky, M.S. Involvement of PI3K/HIF-1α/c-MYC/iNOS Pathway in the Anticancer Effect of Suaeda vermiculata in Rats. Pharmaceuticals 2023, 16, 1470. https://doi.org/10.3390/ph16101470
Mohammed HA, Ewees MG, Mahmoud NI, Ali HM, Amin E, Abdel-Bakky MS. Involvement of PI3K/HIF-1α/c-MYC/iNOS Pathway in the Anticancer Effect of Suaeda vermiculata in Rats. Pharmaceuticals. 2023; 16(10):1470. https://doi.org/10.3390/ph16101470
Chicago/Turabian StyleMohammed, Hamdoon A., Mohamed G. Ewees, Nesreen I. Mahmoud, Hussein M. Ali, Elham Amin, and Mohamed S. Abdel-Bakky. 2023. "Involvement of PI3K/HIF-1α/c-MYC/iNOS Pathway in the Anticancer Effect of Suaeda vermiculata in Rats" Pharmaceuticals 16, no. 10: 1470. https://doi.org/10.3390/ph16101470
APA StyleMohammed, H. A., Ewees, M. G., Mahmoud, N. I., Ali, H. M., Amin, E., & Abdel-Bakky, M. S. (2023). Involvement of PI3K/HIF-1α/c-MYC/iNOS Pathway in the Anticancer Effect of Suaeda vermiculata in Rats. Pharmaceuticals, 16(10), 1470. https://doi.org/10.3390/ph16101470







