Terpenes and Cannabinoids in Supercritical CO2 Extracts of Industrial Hemp Inflorescences: Optimization of Extraction, Antiradical and Antibacterial Activity
Abstract
:1. Introduction
2. Results and Discussion
2.1. Supercritical CO2 Extraction of Industrial Hemp
2.2. Terpenes in sCO2 Extracts of Industrial Hemp
2.3. Cannabinoids in sCO2 Extracts of Industrial Hemp
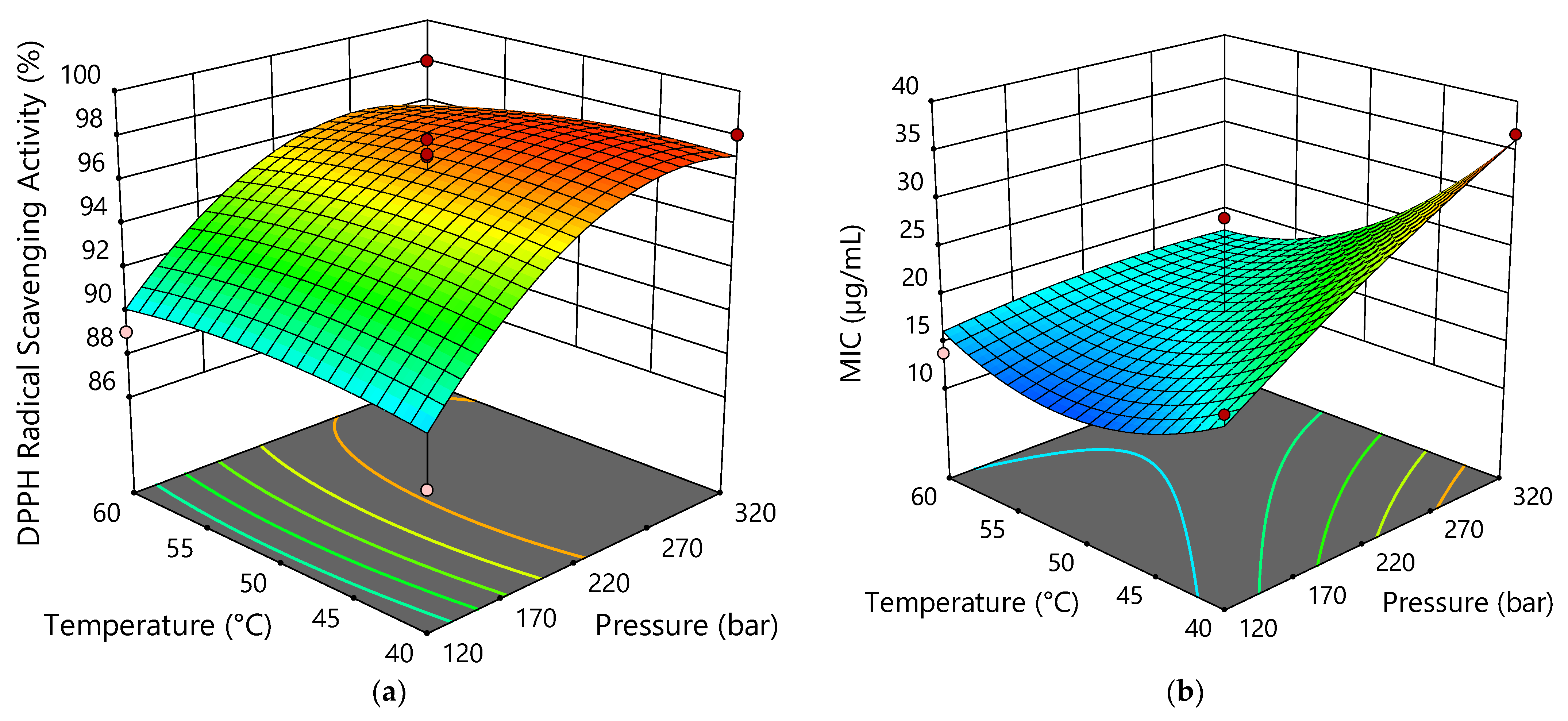
2.4. Antiradical and Antibacterial Activity of Hemp sCO2 Extracts
2.5. Optimization of Extraction Conditions
3. Materials and Methods
3.1. Plant Material
3.2. Chemicals
3.3. Supercritical CO2 Extraction (sCO2)
Statistical Experimental Design
3.4. HPLC Characterization of Cannabinoids
3.5. Gas Chromatography-Mass Spectrometry (GC-MS) Analysis of Volatile Compounds
3.6. Determination of Antiradical Activity of sCO2 Extracts
3.7. Determination of Antibacterial Activity of sCO2 Extracts
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gupta, A.K.; Talukder, M. Cannabinoids for skin diseases and hair regrowth. J. Cosmet. Dermatol. 2021, 20, 2703–2711. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, F.; Lim, M.M.; Kirchhof, M.G. A Survey of topical cannabis use in Canada. J. Cutan. Med. Surg. 2022, 26, 156–161. [Google Scholar] [CrossRef]
- Montero, L.; Meckelmann, S.W.; Kim, H.; Ayala-Cabrera, J.F.; Schmitz, O.J. Differentiation of industrial hemp strains by their cannabinoid and phenolic compounds using LC LC-HRMS. Anal. Bioanal. Chem. 2022, 414, 5445–5459. [Google Scholar] [CrossRef] [PubMed]
- Citti, C.; Braghiroli, D.; Vandelli, M.A.; Cannazza, G. Pharmaceutical and biomedical analysis of cannabinoids: A critical review. J. Pharm. Biomed. Anal. 2018, 147, 565–579. [Google Scholar] [CrossRef]
- Ahmed, A.T.M.F.; Islam, M.Z.; Mahmud, M.S.; Sarker, M.E.; Islam, M.R. Hemp as a potential raw material toward a sustainable world: A review. Heliyon 2022, 8, e08753. [Google Scholar] [CrossRef]
- Jokić, S. Matematičko Modeliranje Ekstrakcije Ulja Iz Zrna Soje Superkritičnim CO2. Ph.D. Thesis, Faculty of Food Technology Osijek, Osijek, Croatia, 2011. [Google Scholar]
- Valizadehderakhshan, M.; Shahbazi, A.; Kazem-Rostami, M.; Todd, M.S.; Bhowmik, A.; Wang, L. Extraction of cannabinoids from Cannabis sativa L. (Hemp)—Review. Agriculture 2021, 11, 384. [Google Scholar] [CrossRef]
- Al Ubeed, H.M.S.; Bhuyan, D.J.; Alsherbiny, M.A.; Basu, A.; Vuong, Q.V. A comprehensive review on the techniques for extraction of bioactive compounds from medicinal cannabis. Molecules 2022, 27, 604. [Google Scholar] [CrossRef]
- Perrotin-Brunel, H.; Kroon, M.C.; van Roosmalen, M.J.E.; van Spronsen, J.; Peters, C.J.; Witkamp, G.J. Solubility of non-psychoactive cannabinoids in supercritical carbon dioxide and comparison with psychoactive cannabinoids. J. Supercrit. Fluids 2010, 55, 603–608. [Google Scholar] [CrossRef]
- Rovetto, L.J.; Aieta, N.V. Supercritical carbon dioxide extraction of cannabinoids from Cannabis sativa L. J. Supercrit. Fluids 2017, 129, 16–27. [Google Scholar] [CrossRef]
- Gallo-Molina, A.C.; Castro-Vargas, H.I.; Garzón-Méndez, W.F.; Ramírez, J.A.M.; Monroy, Z.J.R.; King, J.W.; Parada-Alfonso, F. Extraction, isolation and purification of tetrahydrocannabinol from the Cannabis sativa L. plant using supercritical fluid extraction and solid phase extraction. J. Supercrit. Fluids 2019, 146, 208216. [Google Scholar] [CrossRef]
- Vági, E.; Balázs, M.; Komoczi, A.; Mihalovits, M.; Székely, E. Fractionation of phytocannabinoids from industrial hemp residues with high-pressure technologies. J. Supercrit. Fluids 2020, 164, 104898. [Google Scholar] [CrossRef]
- Qamar, S.; Manrique, Y.J.; Parekh, H.S.; Falconer, J.R. Development and optimization of supercritical fluid extraction setup leading to quantification of 11 cannabinoids derived from medicinal cannabis. Biology 2021, 10, 481. [Google Scholar] [CrossRef] [PubMed]
- Jhawar, N.; Schoenberg, E.; Wang, J.V.; Saedi, N. The growing trend of cannabidiol in skincare products. Clin. Dermatol. 2019, 37, 279–281. [Google Scholar] [CrossRef] [PubMed]
- Atalay, S.; Jarocka-Karpowicz, I.; Skrzydlewska, E. Antioxidative and anti-inflammatory properties of cannabidiol. Antioxidants 2019, 9, 21. [Google Scholar] [CrossRef]
- Marzorati, S.; Friscione, D.; Picchi, E.; Verotta, L. Cannabidiol from inflorescences of Cannabis sativa L.: Green extraction and purification processes. Ind. Crops Prod. 2020, 155, 112816. [Google Scholar] [CrossRef]
- Drinić, Z. Extraction of Industrial Hemp (Cannabis sativa L.). Ph.D. Thesis, Faculty of Technology, University of Novy Sad, Novi Sad, Serbia, 2020. [Google Scholar]
- Aladić, K.; Jarni, K.; Barbir, T.; Vidović, S.; Vladić, J.; Bilić, M.; Jokić, S. Cold pressing and supercritical CO2 extraction of hemp (Cannabis sativa) seed oil. Ind. Crops Prod. 2015, 76, 472–478. [Google Scholar] [CrossRef]
- Kitrytė, V.; Bagdonaitė, D.; Venskutonis, P.R. Biorefining of industrial hemp (Cannabis sativa L.) threshing residues into cannabinoid and antioxidant fractions by supercritical carbon dioxide, pressurized liquid and enzyme-assisted extractions. Food Chem. 2018, 267, 420–429. [Google Scholar] [CrossRef]
- Isidore, E.; Karim, H.; Ioannou, I. Extraction of phenolic compounds and terpenes from Cannabis sativa L. by-products: From conventional to intensified processes. Antioxidants 2021, 10, 942. [Google Scholar] [CrossRef]
- Kovalchuk, I.; Pellino, M.; Rigault, P.O.; van Velzen, R.; Ebersbach, J.; Ashnest, J.R.; Mau, M.; Schranz, M.E.; Alcorn, J.; Laprairie, R.B.; et al. The genomics of cannabis and its close relatives. Ann. Rev. Plant. Biol. 2020, 71, 713–739. [Google Scholar] [CrossRef]
- Allen, K.D.; McKernan, K.; Pauli, C.; Roe, J.; Torres, A.; Gaudino, R. Genomic characterization of the complete terpene synthase gene family from Cannabis sativa. PLoS ONE 2019, 14, e0222363. [Google Scholar]
- Booth, J.K.; Page, J.E.; Bohlmann, J. Terpene synthases from Cannabis sativa. PLoS ONE 2017, 12, e0173911. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hanuš, L.O.; Hod, Y. Terpenes/Terpenoids in Cannabis: Are they important? Med. Cannabis Cannabinoids 2020, 3, 25–60. [Google Scholar] [CrossRef] [PubMed]
- Micalizzi, G.; Vento, F.; Alibrando, F.; Donnarumma, D.; Dugo, P.; Mondello, L. Cannabis Sativa L.: A comprehensive review on the analytical methodologies for cannabinoids and terpenes characterization. J. Chromatogr. A 2021, 1637, 461864. [Google Scholar] [CrossRef]
- Gulluni, N.; Re, T.; Loiacono, I.; Lanzo, G.; Gori, L.; Macchi, C.; Epifani, F.; Bragazzi, N.; Firenzuoli, F. Cannabis essential oil: A preliminary study for the evaluation of the brain effects. Evidence-Based Complement. Alternat. Med. 2018, 2018, 1709182. [Google Scholar] [CrossRef]
- Jansen, C.; Shimoda, L.M.N.; Kawakami, J.K.; Ang, L.; Bacani, A.J.; Baker, J.D.; Badowski, C.; Speck, M.; Stokes, A.J.; Small-Howard, A.L.; et al. Myrcene and terpene regulation of TRPV1. Channels 2019, 13, 344–366. [Google Scholar] [CrossRef] [PubMed]
- Kozioł, A.; Stryjewska, A.; Librowski, T.; Sałat, K.; Gaweł, M.; Moniczewski, A.; Lochyński, S. An overview of the pharmacological properties and potential applications of natural monoterpenes. Mini Rev. Med. Chem. 2014, 14, 1156–1168. [Google Scholar] [CrossRef]
- Salehi, B.; Upadhyay, S.; Orhan, I.E.; Jugran, A.K.; Jayaweera, S.L.D.; Dias, D.A.; Sharopov, F.; Taheri, Y.; Martins, N.; Baghalpour, N.; et al. Therapeutic potential of α- and β-pinene: A miracle gift of nature. Biomolecules 2019, 9, 738. [Google Scholar] [CrossRef]
- Erasto, P.; Viljoen, A.M. Limonene: A review: Biosynthetic, ecological and pharmacological relevance. Nat. Prod. Commun. 2008, 3, 1193–1202. [Google Scholar] [CrossRef]
- Sun, J. D-limonene: Safety and clinical applications. Altern. Med. Rev. 2007, 12, 259–264. [Google Scholar]
- Sharma, C.; Al Kaabi, J.M.; Nurulain, S.M.; Goyal, S.N.; Kamal, M.A.; Ojha, S. Polypharmacological properties and therapeutic potential of β-caryophyllene: A dietary phytocannabinoid of pharmaceutical promise. Curr. Pharm. Des. 2016, 22, 3237–3264. [Google Scholar] [CrossRef]
- Francomano, F.; Caruso, A.; Barbarossa, A.; Fazio, A.; La Torre, C.; Ceramella, J.; Mallamaci, R.; Saturnino, C.; Iacopetta, D.; Sinicropi, M.S. β-Caryophyllene: A sesquiterpene with countless biological properties. Appl. Sci. 2019, 9, 5420. [Google Scholar] [CrossRef]
- Gertsch, J.; Leonti, M.; Raduner, S.; Racz, I.; Jian-Zhong, C.; Xiang-Qun, X.; Altmann, K.H.; Karsak, M.; Zimmer, A. Beta-caryophyllene is a dietary cannabinoid. Proc. Natl. Acad. Sci. USA 2008, 105, 9099–9104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Astani, A.; Reichling, J.; Schnitzler, P. Screening for antiviral activities of isolated compounds from essential oils. Evidence-Based Complement. Altern. Med. 2011, 2011, 253643. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Wu, J.; Luo, Y.; Huang, N.; Zhen, N.; Zhou, Y.; Sun, F.; Li, Z.; Pan, Q.; Li, Y. (–)-Guaiol regulates RAD51 stability via autophagy to induce cell apoptosis in nonsmall cell lung cancer. Oncotarget 2016, 7, 62585–62597. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhu, J.; Wu, J.; Huang, N.; Cui, Z.; Luo, Y.; Sun, F.; Pan, Q.; Li, Y.; Yanga, Q. (–)-Guaiol regulates autophagic cell death depending on mTOR signaling in NSCLC. Cancer Biol. Ther. 2018, 19, 706–714. [Google Scholar] [CrossRef]
- Lu, Y.-C. Studies on the chemical constituents of the essential oil of Rhododendron tsinghaiense Ching. Huaxue Xuebao 1980, 38, 241–249. [Google Scholar]
- Russo, E.B. Taming THC: Potential cannabis synergy and phytocannabinoid-terpenoid entourage effects. Br. J. Pharmacol. 2011, 163, 1344–1364. [Google Scholar] [CrossRef]
- Sexton, M.; Shelton, K.; Haley, P.; West, M. Evaluation of cannabinoid and terpenoid content: Cannabis flower compared to supercritical CO2 concentrate. Planta Med. 2018, 84, 234–241. [Google Scholar] [CrossRef]
- Chandra, S.; Lata, H.; ElSohly, M.A. Cannabis Sativa L.-Botany and Biotechnology; Springer: Cham, Switzerland, 2017. [Google Scholar]
- Zorić, M.; Banožić, M.; Aladić, K.; Vladimir-Knežević, S.; Jokić, S. Supercritical CO2 extracts in cosmetic industry: Current status and future perspectives. Sustain. Chem. Pharm. 2022, 27, 100688. [Google Scholar] [CrossRef]
- Eagleston Lauren, R.M.; Kalani, N.K.; Patel, R.R.; Flaten, H.K.; Dunnick, C.A.; Dellavalle, R.P. Cannabinoids in dermatology: A scoping review. Dermatol. Online J. 2018, 24, 1. [Google Scholar] [CrossRef]
- Cabaleiro, N.; De la Calle, I.; Bendicho, C.; Lavilla, I. Current trends in liquid–liquid and solid–liquid extraction for cosmetic analysis: A review. Anal. Methods 2013, 5, 323–340. [Google Scholar] [CrossRef]
- Vági, E.; Balázs, M.; Komoczi, A.; Kiss, I.; Mihalovits, M.; Székely, E. Cannabinoids enriched extracts from industrial hemp residues. Period. Polytech. Chem. Eng. 2019, 63, 357–363. [Google Scholar] [CrossRef]
- Omar, J.; Olivares, M.; Alzaga, M.; Etxebarria, N. Optimisation and characterisation of marihuana extracts obtained by supercritical fluid extraction and focused ultrasound extraction and retention time locking GC–MS. J. Sep. Sci. 2013, 36, 1397–1404. [Google Scholar] [CrossRef] [PubMed]
- Perrotin-Brunel, H.; van Roosmalen, M.J.E.; Kroon, M.C.; van Spronsen, J.; Witkamp, G.J.; Peters, C.J. Solubility of cannabinol in supercritical carbon dioxide. J. Chem. Eng. Data 2010, 55, 3704–3707. [Google Scholar] [CrossRef]
- Dawidowicz, A.L.; Olszowy-Tomczyk, M.; Typek, R. CBG, CBD, Δ9-THC, CBN, CBGA, CBDA and Δ9-THCA as antioxidant agents and their intervention abilities in antioxidant action. Fitoterapia 2021, 152, 104915. [Google Scholar] [CrossRef]
- McPartland, J.M.; MacDonald, C.; Young, M.; Grant, P.S.; Furkert, D.P.; Glass, M. Affinity and efficacy studies of tetrahydrocannabinolic acid a at cannabinoid receptor types one and two. Cannabis Cannabinoid Res. 2017, 2, 87–95. [Google Scholar] [CrossRef]
- Zhang, G.; Meredith, T.C.; Kahne, D. On the essentiality of lipopolysaccharide to Gram-negative bacteria. Curr. Opin. Microbiol. 2013, 16, 779–785. [Google Scholar] [CrossRef]
- Ibrahim, E.A.; Wang, M.; Radwan, M.M.; Wanas, A.S.; Majumdar, C.G.; Avula, B.; Wang, Y.H.; Khan, I.A.; Chandra, S.; Lata, H.; et al. Analysis of terpenes in Cannabis sativa L. using GC/MS: Method development, validation and application. Planta Med. 2019, 85, 431–438. [Google Scholar] [CrossRef]
- Nissen, L.; Zatta, A.; Stefanini, I.; Grandi, S.; Sgorbati, S.; Biavati, B.; Monti, A. Characterization and antimicrobial activity of essential oils of industrial hemp varieties (Cannabis sativa L.). Fitoterapia 2010, 81, 413–419. [Google Scholar] [CrossRef]
- Leghissa, A.; Hildenbr, Z.L.; Schug, K.A. A review of methods for the chemical characterization of cannabis natural products. J. Sep. Sci. 2018, 41, 398–415. [Google Scholar] [CrossRef]
- Appendino, G.; Gibbons, S.; Giana, A.; Pagani, A.; Grassi, G.; Stavri, M.; Smith, E.; Rahman, M.M. Antibacterial cannabinoids from Cannabis sativa: A structure-activity study. J. Nat. Prod. 2008, 71, 1427–1430. [Google Scholar] [CrossRef] [PubMed]
- Fiorini, D.; Molle, A.; Nabissi, M.; Santini, G.; Benelli, G.; Maggi, G.M. Valorizing industrial hemp (Cannabis sativa L.) by-products: Cannabidiol enrichment in the inflorescence essential oil optimizing sample pre-treatment prior to distillation. Ind. Crops Prod. 2019, 128, 581–589. [Google Scholar] [CrossRef]
- Hazekamp, A.; Fischedick, J.T.; Llano Dìez, M.; Lubbe, A.; Ruhaak, R.L. Chemistry of Cannabis. In Comprehensive Natural Products II Chemistry and Biology, 1st ed.; Mander, L., Liu, H.-W., Eds.; Elsevier: Kidlington, UK, 2010; Volume 3, pp. 1034–1077. [Google Scholar]
- Wang, C.-T.; Wiedinmyer, C.; Ashworth, K.; Harley, P.C.; Ortega, J.; Vizuete, W. Leaf enclosure measurements for determining volatile organic compound nemission capacity from Cannabis spp. Atmos. Environ. 2019, 199, 80–87. [Google Scholar] [CrossRef]
- Blaskovich, M.A.T.; Kavanagh, A.M.; Elliott, A.G.; Zhang, B.; Ramu, S.; Amado, M.; Lowe, G.J.; Hinton, A.O.; Thu Pham, D.M.; Zuegg, J.; et al. The antimicrobial potential of cannabidiol. Commun. Biol. 2021, 4, 7. [Google Scholar] [CrossRef] [PubMed]
- Iseppi, R.; Brighenti, V.; Licata, M.; Lambertini, A.; Sabia, C.; Messi, P.; Pellati, F.; Benvenuti, S. Chemical characterization and evaluation of the antibacterial activity of essential oils from fibre-type Cannabis sativa L. (Hemp). Molecules 2019, 24, 2302. [Google Scholar] [CrossRef]
- Jokić, S.; Molnar, M.; Jakovljević, M.; Aladić, K.; Jerković, I. Optimization of supercritical CO2 extraction of Salvia officinalis L. leaves targeted on oxygenated monoterpenes, α-humulene, viridiflorol and manool. J. Supercrit. Fluid 2018, 133, 253–262. [Google Scholar] [CrossRef]
- Jokić, S.; Horvat, G.; Aladić, K. Design of SFE system using a holistic approach–problems and challenges. In Supercritical Fluid Extraction: Technology, Applications and Limitations; Lindy, J., Ed.; Nova Science Publishers, Inc.: New York, NY, USA, 2015; p. 95. [Google Scholar]
- Bas, D.; Boyaci, I.H. Modeling and optimization I: Usability of response surface methodology. J. Food Eng. 2007, 78, 836–845. [Google Scholar] [CrossRef]
- Chang, S.T.; Wu, J.H.; Wang, S.Y.; Kang, P.L.; Yang, N.S.; Shyur, L.F. Antioxidant activity of extracts from Acacia confusa Bark and Heartwood. J. Agric. Food Chem. 2001, 49, 3420–3424. [Google Scholar] [CrossRef]
- Weinstein, M.P.; Patel, J.B. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically: M07-A11, 11th ed.; Committee for Clinical Laboratory Standards: Wayne, PA, USA, 2018. [Google Scholar]
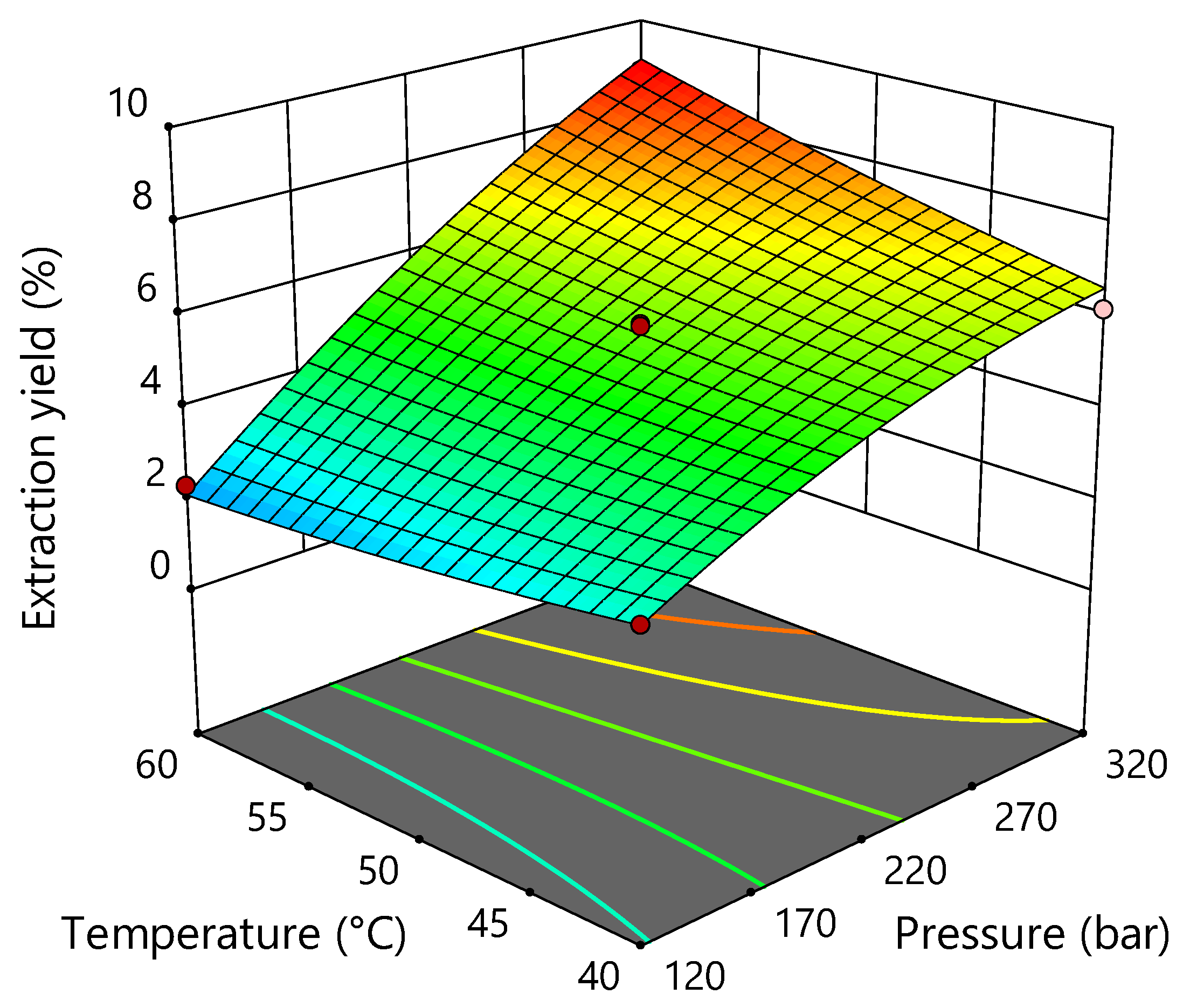
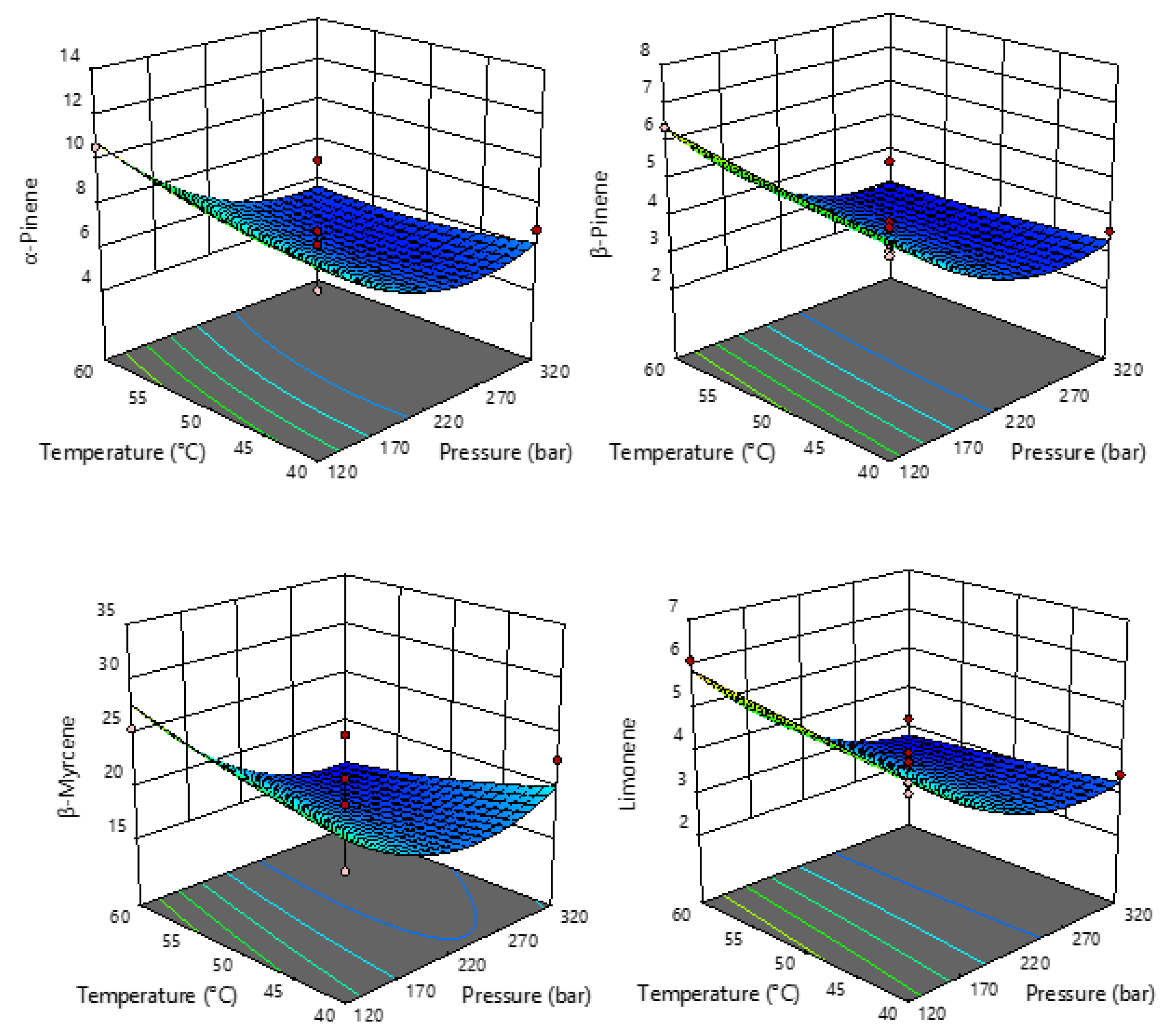
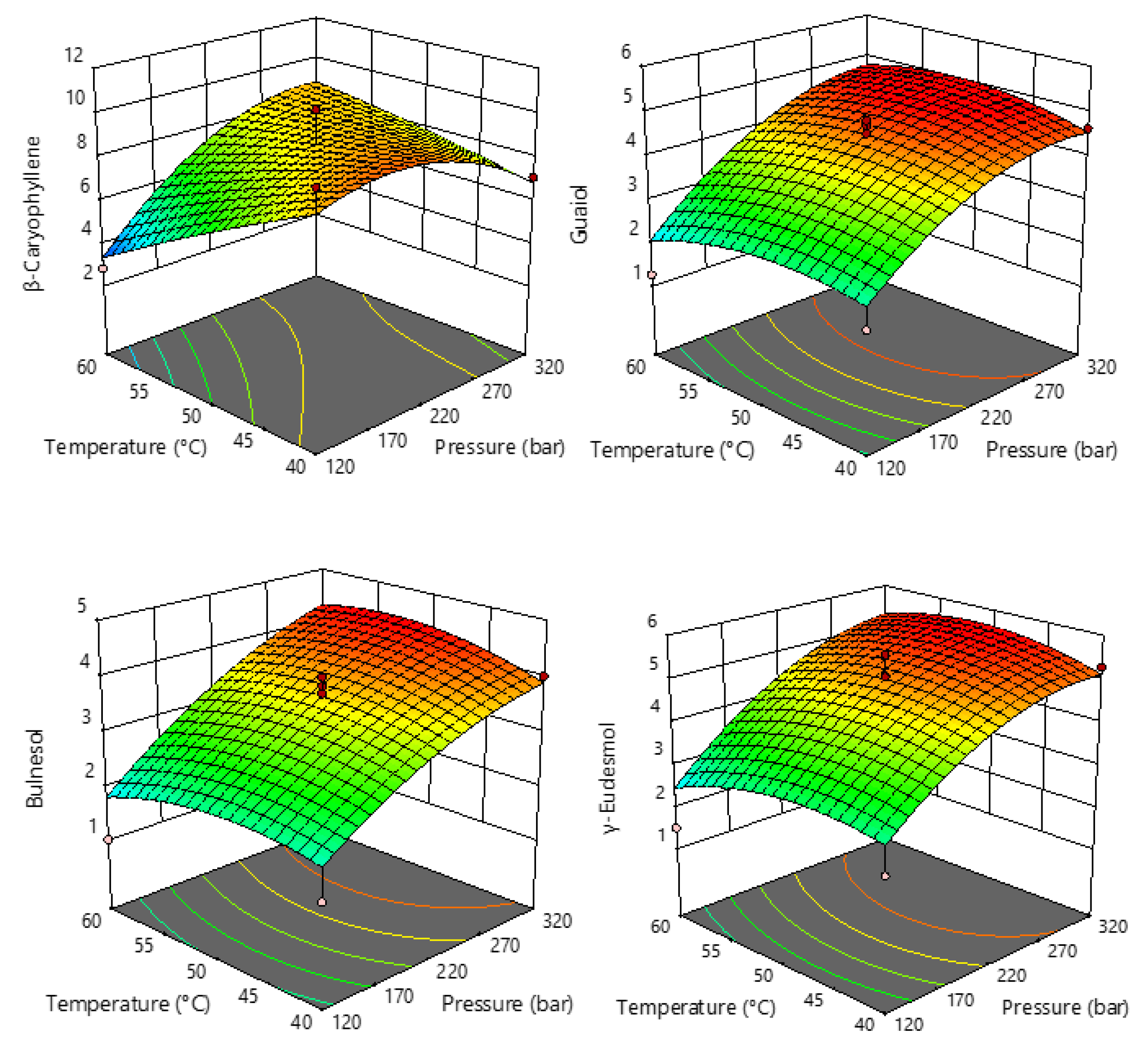
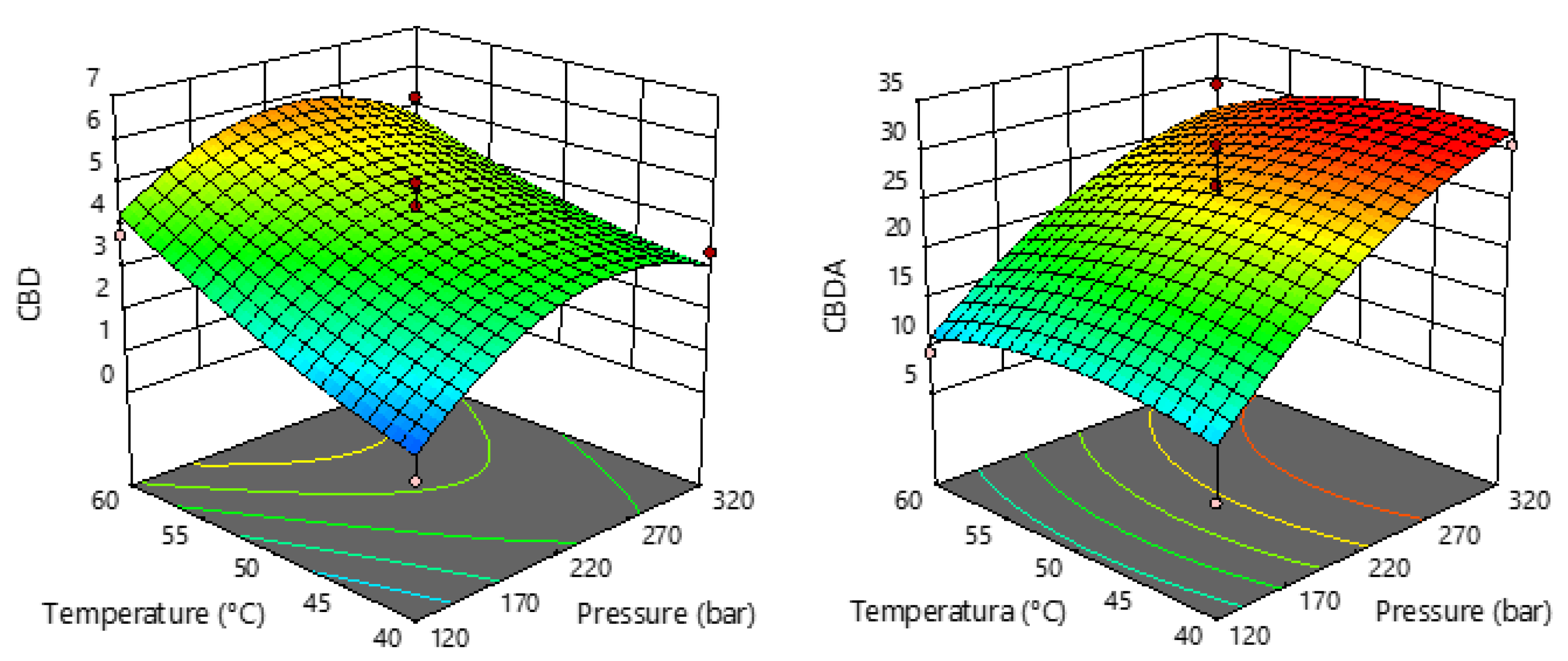
| Run | Pressure (Bar) X1 | Temperature (°C) X2 | Extraction Yield (%) Y |
|---|---|---|---|
| 1 | 320 | 40 | 6.16 |
| 2 | 220 | 35.9 | 5.67 |
| 3 | 220 | 50 | 5.53 |
| 4 | 120 | 40 | 3.20 |
| 5 | 220 | 64.1 | 6.39 |
| 6 | 78.6 | 50 | 0.75 |
| 7 | 220 | 50 | 5.85 |
| 8 | 320 | 60 | 8.83 |
| 9 | 120 | 60 | 2.31 |
| 10 | 361.4 | 50 | 8.79 |
| 11 | 220 | 50 | 5.80 |
| 12 | 220 | 50 | 5.62 |
| 13 | 220 | 50 | 5.39 |
| No. | Compound | RI | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | Inflorescences |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Hexan-1-ol | 875 | 0.06 | 0.06 | 0.08 | 0.09 | 0.10 | 0.06 | 0.09 | 0.08 | 0.11 | 0.09 | 0.09 | 0.08 | 0.09 | 0.06 |
| 2 | Heptanal | 903 | 0.17 | 0.10 | 0.10 | 0.04 | 0.10 | 0.04 | 0.14 | 0.11 | 0.04 | 0.16 | 0.09 | 0.13 | 0.12 | 0.14 |
| 3 | α-Thujene | 934 | - | - | - | - | - | 0.02 | - | - | 0.01 | - | - | - | - | 0.01 |
| 4 | α-Pinene | 941 | 6.82 | 6.46 | 6.03 | 7.89 | 6.41 | 12.88 | 6.77 | 6.94 | 10.58 | 5.14 | 6.12 | 5.13 | 5.55 | 6.81 |
| 5 | Camphene | 956 | 0.15 | 0.15 | 0.16 | 0.11 | 0.16 | 0.40 | 0.17 | 0.17 | 0.32 | 0.12 | 0.16 | 0.13 | 0.15 | 0.16 |
| 6 | Sabinene | 980 | - | - | - | - | - | - | - | - | - | - | - | - | - | 0.03 |
| 7 | β-Pinene | 983 | 3.59 | 3.55 | 3.66 | 5.11 | 3.40 | 7.78 | 3.85 | 3.63 | 6.38 | 2.94 | 3.70 | 3.02 | 3.23 | 3.41 |
| 8 | β-Myrcene | 993 | 22.47 | 18.40 | 18.24 | 19.98 | 18.31 | 33.45 | 20.70 | 18.47 | 25.44 | 15.55 | 17.29 | 15.66 | 15.65 | 16.75 |
| 9 | α-Phellandrene | 1007 | 0.02 | 0.03 | 0.04 | 0.06 | 0.03 | 0.09 | 0.04 | 0.05 | 0.04 | 0.06 | 0.06 | 0.03 | 0.03 | 0.04 |
| 10 | Δ-3-carene | 1014 | 0.03 | 0.02 | 0.03 | 0.05 | 0.03 | 0.06 | 0.03 | 0.04 | 0.04 | 0.04 | 0.05 | 0.02 | 0.02 | 0.03 |
| 11 | α-Terpinene | 1021 | 0.02 | - | 0.03 | 0.04 | 0.03 | 0.05 | 0.03 | 0.04 | 0.03 | 0.05 | 0.05 | 0.02 | 0.02 | 0.03 |
| 12 | Limonene | 1033 | 3.44 | 3.42 | 3.67 | 5.04 | 3.28 | 6.65 | 3.97 | 3.19 | 6.08 | 2.89 | 3.75 | 2.99 | 3.23 | 2.71 |
| 13 | 1,8-Cineole | 1036 | 0.02 | 0.02 | 0.03 | 0.01 | 0.02 | 0.01 | 0.03 | 0.02 | 0.01 | 0.04 | 0.02 | 0.02 | 0.02 | 0.01 |
| 14 | (Z)-β-Ocimene | 1041 | 0.04 | 0.04 | 0.06 | 0.09 | 0.06 | 0.13 | 0.07 | 0.05 | 0.10 | 0.05 | 0.06 | 0.04 | 0.05 | 0.03 |
| 15 | (E)-β-Ocimene | 1052 | 1.24 | 1.28 | 1.38 | 2.08 | 1.34 | 3.10 | 1.61 | 1.11 | 2.36 | 1.11 | 1.51 | 1.20 | 1.21 | 1.04 |
| 16 | γ-Terpinene | 1063 | 0.03 | 0.03 | 0.03 | 0.04 | 0.03 | 0.04 | 0.04 | 0.05 | 0.04 | 0.05 | 0.05 | 0.03 | 0.03 | 0.04 |
| 17 | cis-Sabinenehydrate | 1071 | 0.05 | 0.05 | 0.07 | 0.06 | 0.07 | 0.03 | 0.07 | 0.06 | 0.05 | 0.06 | 0.07 | 0.07 | 0.06 | 0.06 |
| 18 | Octan-1-ol | 1072 | 0.03 | 0.04 | 0.06 | 0.03 | 0.07 | 0.03 | 0.07 | 0.07 | 0.03 | 0.10 | 0.06 | 0.07 | 0.07 | 0.07 |
| 19 | trans-Linalool oxide | 1076 | 0.03 | 0.03 | 0.04 | 0.04 | 0.04 | 0.02 | 0.05 | 0.04 | 0.03 | 0.05 | 0.05 | 0.04 | 0.05 | 0.03 |
| 20 | α-Terpinolene | 1091 | 0.78 | 0.56 | 0.92 | 1.41 | 0.61 | 1.12 | 0.79 | 0.60 | 0.97 | 0.75 | 1.07 | 0.58 | 0.55 | 0.62 |
| 21 | Linalool | 1100 | 0.04 | 0.07 | 0.07 | 0.08 | 0.07 | 0.05 | 0.07 | 0.07 | 0.08 | 0.07 | 0.07 | 0.08 | 0.09 | 0.07 |
| 22 | Nonanal | 1105 | 0.13 | 0.05 | 0.08 | 0.05 | 0.09 | 0.03 | 0.08 | 0.12 | 0.03 | 0.14 | 0.07 | 0.13 | 0.08 | 0.16 |
| 23 | Hexyl propanoate | 1107 | 0.04 | 0.04 | 0.06 | 0.10 | 0.04 | 0.06 | 0.05 | 0.05 | 0.06 | 0.04 | 0.06 | 0.03 | 0.05 | 0.00 |
| 24 | Fenchol | 1115 | 0.52 | 0.61 | 0.64 | 0.65 | 0.66 | 0.33 | 0.68 | 0.57 | 0.55 | 0.62 | 0.68 | 0.66 | 0.65 | 0.54 |
| 25 | trans-Pinocarveol | 1142 | - | 0.02 | - | - | - | - | - | - | - | 0.02 | - | - | - | 0.00 |
| 26 | Borneol | 1169 | 0.17 | 0.18 | 0.21 | 0.17 | 0.21 | 0.10 | 0.24 | 0.20 | 0.12 | 0.22 | 0.23 | 0.22 | 0.23 | 0.18 |
| 27 | Terpinen-4-ol | 1179 | 0.03 | 0.03 | 0.04 | 0.03 | 0.04 | 0.02 | 0.04 | 0.04 | 0.02 | 0.05 | 0.05 | 0.04 | 0.05 | 0.04 |
| 28 | α-Terpineol | 1191 | 0.03 | 0.05 | 0.07 | 0.05 | 0.06 | 0.03 | 0.07 | 0.07 | 0.03 | 0.07 | 0.08 | 0.08 | 0.07 | 0.06 |
| 29 | Hexyl butanoate | 1193 | 0.60 | 0.66 | 0.73 | 0.91 | 0.62 | 0.52 | 0.79 | 0.51 | 0.96 | 0.54 | 0.77 | 0.67 | 0.63 | 0.48 |
| 30 | β-Citronellol | 1230 | 0.04 | 0.04 | 0.06 | 0.06 | 0.07 | 0.02 | 0.07 | 0.06 | 0.04 | 0.07 | 0.08 | 0.07 | 0.07 | 0.07 |
| 31 | 1H-Indole | 1294 | - | - | - | - | - | - | - | - | - | - | - | - | - | 0.03 |
| 32 | α-Ylangene | 1373 | 0.07 | 0.09 | 0.09 | 0.05 | 0.10 | 0.04 | 0.09 | 0.09 | 0.11 | 0.10 | 0.09 | 0.09 | 0.07 | 0.08 |
| 33 | α-Copaene | 1376 | 0.03 | 0.03 | 0.04 | 0.03 | 0.04 | 0.02 | 0.04 | 0.05 | 0.04 | 0.05 | 0.05 | 0.04 | 0.05 | 0.06 |
| 34 | Hexyl hexanoate | 1388 | 0.10 | 0.11 | 0.11 | 0.14 | 0.12 | 0.04 | 0.14 | 0.08 | 0.12 | 0.10 | 0.15 | 0.13 | 0.11 | 0.08 |
| 35 | α-Gurjunene | 1408 | 0.03 | 0.03 | 0.03 | 0.03 | 0.03 | 0.02 | 0.04 | 0.02 | 0.04 | 0.04 | 0.04 | 0.03 | 0.03 | 0.03 |
| 36 | cis-α-Bergamotene | 1415 | 0.07 | 0.09 | 0.09 | 0.11 | 0.09 | 0.05 | 0.09 | 0.07 | 0.12 | 0.09 | 0.09 | 0.08 | 0.11 | 0.08 |
| 37 | β-Caryophyllene | 1419 | 7.09 | 8.53 | 8.45 | 9.88 | 8.34 | 3.94 | 10.16 | 7.49 | 2.83 | 7.56 | 8.31 | 7.99 | 8.15 | 7.18 |
| 38 | γ-Elemene | 1434 | 0.33 | 0.35 | 0.43 | 0.54 | 0.34 | 0.11 | 0.45 | 0.31 | 0.29 | 0.33 | 0.42 | 0.40 | 0.36 | 0.25 |
| 39 | trans-α-Bergamotene | 1437 | 0.53 | 0.61 | 0.60 | 0.75 | 0.58 | 0.29 | 0.62 | 0.50 | 0.78 | 0.52 | 0.59 | 0.58 | 0.66 | 0.48 |
| 40 | α-Guaiene | 1439 | 0.02 | 0.05 | 0.06 | 0.06 | 0.06 | 0.02 | 0.05 | 0.05 | 0.05 | 0.05 | 0.06 | 0.06 | 0.06 | 0.04 |
| 41 | 3,7-Guaiadiene | 1444 | 0.05 | 0.11 | 0.10 | 0.13 | 0.10 | 0.04 | 0.10 | 0.08 | 0.12 | 0.10 | 0.11 | 0.10 | 0.10 | 0.08 |
| 42 | α-Humulene | 1454 | 2.23 | 2.78 | 2.79 | 3.31 | 2.75 | 1.09 | 2.83 | 2.42 | 3.15 | 2.50 | 2.76 | 2.67 | 2.68 | 2.31 |
| 43 | trans-β-Farnesene | 1460 | 1.26 | 1.53 | 1.61 | 1.92 | 1.54 | 0.64 | 1.65 | 1.25 | 1.91 | 1.39 | 1.62 | 1.50 | 1.61 | 1.24 |
| 44 | α-Amorphene | 1466 | 0.02 | 0.04 | 0.04 | 0.05 | 0.06 | 0.04 | 0.05 | 0.05 | 0.05 | 0.05 | 0.05 | 0.04 | 0.06 | 0.06 |
| 45 | γ-Muurolene | 1476 | 0.24 | 0.32 | 0.31 | 0.38 | 0.33 | 0.12 | 0.34 | 0.30 | 0.32 | 0.33 | 0.34 | 0.34 | 0.33 | 0.28 |
| 46 | β-Eudesmene | 1483 | 0.11 | 0.09 | 0.11 | 0.22 | 0.43 | 0.03 | 0.12 | 0.35 | 0.39 | 0.39 | 0.13 | 0.11 | 0.14 | 0.34 |
| 47 | β-Selinene | 1485 | 0.96 | 1.15 | 1.16 | 1.32 | 1.19 | 0.38 | 1.19 | 1.02 | 1.07 | 1.07 | 1.19 | 1.14 | 1.12 | 0.95 |
| 48 | Valencene | 1492 | 0.46 | 0.55 | 0.57 | 0.65 | 0.58 | 0.17 | 0.58 | 0.48 | 0.53 | 0.53 | 0.59 | 0.52 | 0.53 | 0.00 |
| 49 | α-Selinene | 1493 | 0.91 | 1.10 | 1.11 | 1.24 | 1.13 | 0.35 | 1.14 | 0.98 | 1.00 | 1.02 | 1.13 | 1.10 | 1.05 | 1.38 |
| 50 | (E,E)-α-Farnesene | 1509 | 2.10 | 2.55 | 2.67 | 3.28 | 2.49 | 0.79 | 2.71 | 2.09 | 3.02 | 2.34 | 2.64 | 2.47 | 2.44 | 1.98 |
| 51 | γ-Cadinene | 1515 | 0.18 | 0.22 | 0.18 | 0.16 | 0.19 | 0.09 | 0.20 | 0.19 | 0.14 | 0.21 | 0.19 | 0.20 | 0.22 | 0.18 |
| 52 | Bicyclogermacrene | 1517 | 0.67 | 0.83 | 0.83 | 0.92 | 0.84 | 0.67 | 0.85 | 0.72 | 0.70 | 0.75 | 0.85 | 0.82 | 0.79 | 0.68 |
| 53 | β-Cadinene | 1518 | 0.51 | 0.63 | 0.60 | 0.68 | 0.66 | 0.26 | 0.65 | 0.58 | 0.55 | 0.59 | 0.62 | 0.67 | 0.62 | 0.58 |
| 54 | δ-Cadinene | 1524 | 0.46 | 0.60 | 0.57 | 0.67 | 0.61 | 0.18 | 0.62 | 0.54 | 0.52 | 0.59 | 0.62 | 0.61 | 0.61 | 0.00 |
| 55 | γ-Selinene | 1535 | 3.49 | 4.28 | 4.11 | 4.60 | 4.46 | 1.34 | 4.43 | 4.01 | 3.51 | 4.05 | 4.26 | 4.59 | 4.21 | 4.12 |
| 56 | Selina-3,7(11)-diene | 1541 | 4.98 | 6.13 | 5.84 | 6.49 | 6.31 | 1.92 | 1.99 | 5.67 | 4.82 | 5.68 | 6.05 | 6.59 | 5.98 | 5.69 |
| 57 | (E)-α-Bisabolene | 1549 | 0.69 | 0.88 | 0.84 | 0.93 | 0.73 | 0.27 | 0.83 | 0.73 | 0.72 | 0.80 | 0.83 | 0.90 | 0.78 | 0.75 |
| 58 | Germacrene B | 1556 | 1.90 | 1.88 | 2.17 | 2.70 | 1.86 | 0.59 | 2.28 | 1.65 | 1.50 | 1.72 | 2.14 | 2.05 | 1.82 | 1.31 |
| 59 | Nerolidol | 1566 | 0.21 | 0.21 | 0.38 | 0.31 | 0.40 | 0.12 | 0.41 | 0.35 | 0.16 | 0.41 | 0.43 | 0.44 | 0.44 | 0.37 |
| 60 | Caryophyllene oxide | 1581 | 0.60 | 0.72 | 0.76 | 0.67 | 0.77 | 0.31 | 0.77 | 0.69 | 0.33 | 0.73 | 0.80 | 0.78 | 0.80 | 0.75 |
| 61 | Guaiol | 1601 | 4.62 | 3.96 | 3.94 | 2.03 | 4.28 | 2.32 | 4.51 | 4.54 | 1.26 | 4.85 | 4.51 | 4.80 | 4.68 | 4.87 |
| 62 | Ledol | 1599 | 0.13 | 0.12 | 0.11 | 0.13 | 0.12 | 0.08 | 0.12 | 0.12 | 0.04 | 0.14 | 0.12 | 0.12 | 0.11 | 0.13 |
| 63 | γ-Eudesmol | 1623 | 5.27 | 4.48 | 4.43 | 2.36 | 4.66 | 3.03 | 4.92 | 5.14 | 1.50 | 5.35 | 5.05 | 5.55 | 5.07 | 5.53 |
| 64 | 10-epi-γ Eudesmol | 1630 | 0.76 | 0.63 | 0.70 | 0.31 | 0.29 | 0.19 | 0.30 | 0.32 | 0.20 | 0.36 | 0.31 | 0.33 | 0.33 | 0.38 |
| 65 | Dihyro-cis-α-copaene-8-ol ** | 1634 | 0.26 | 0.23 | 0.11 | 0.12 | 0.27 | 0.17 | 0.28 | 0.31 | 0.08 | 0.34 | 0.29 | 0.11 | 0.31 | 0.34 |
| 66 | Hinesol | 1637 | 0.11 | 0.11 | 0.29 | 0.04 | 0.11 | 0.08 | 0.12 | 0.13 | 0.03 | 0.01 | 0.06 | 0.30 | 0.12 | 0.16 |
| 67 | β-Eudesmol | 1649 | 2.21 | 1.77 | 1.92 | 0.85 | 1.89 | 1.26 | 2.00 | 2.21 | 0.55 | 2.37 | 2.05 | 2.17 | 2.07 | 2.53 |
| 68 | α-Eudesmol | 1652 | 2.71 | 2.13 | 2.28 | 1.02 | 2.27 | 1.51 | 2.40 | 2.69 | 0.67 | 2.85 | 2.44 | 2.59 | 2.49 | 2.53 |
| 69 | Bulnesol | 1666 | 4.01 | 3.19 | 3.06 | 1.52 | 3.58 | 2.35 | 3.69 | 4.21 | 1.00 | 4.37 | 3.70 | 3.99 | 3.83 | 4.73 |
| 70 | α-Bisabolol | 1683 | 1.89 | 1.83 | 1.83 | 0.90 | 1.61 | 1.00 | 1.90 | 2.01 | 0.51 | 2.20 | 2.01 | 2.10 | 2.07 | 2.09 |
| 71 | Juniper camphor | 1692 | 0.44 | 0.39 | 0.43 | 0.18 | 0.43 | 0.27 | 0.46 | 0.56 | 0.14 | 0.60 | 0.46 | 0.48 | 0.46 | 0.65 |
| 72 | Hexahydrofarnesyl acetone | 1846 | 0.06 | 0.10 | 0.13 | 0.13 | 0.14 | 0.04 | 0.17 | 0.13 | 0.05 | 0.16 | 0.16 | 0.16 | 0.17 | 0.23 |
| 73 | Heneicosane | 2100 | 0.01 | 0.03 | 0.06 | 0.01 | 0.04 | 0.01 | 0.05 | 0.04 | 0.01 | 0.05 | 0.05 | 0.06 | 0.05 | 0.10 |
| 74 | Phytol | 2116 | 0.02 | 0.01 | 0.22 | 0.05 | 0.01 | 0.01 | 0.24 | 0.20 | 0.03 | 0.37 | 0.29 | 0.01 | 0.34 | 0.23 |
| Run * | CBCA | CBG | CBN | THC | CBDVA | CBD | CBDA | CBGA | THCVA | CBC | THCA-A | Total CBD | Total THC |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 1.98 ± 0.03 | 0.55 ± 0.03 | 0.00 | 0.66 ± 0.03 | 0.20 ± 0.01 | 3.39 ± 0.07 | 30.68 ± 0.21 | 0.23 ± 0.01 | 0.00 | 1.21 ± 0.01 | 1.17 ± 0.03 | 30.21 ± 0.00 | 3.69 ± 0.05 |
| 2 | 0.03 ± 0.00 | 0.27 ± 0.01 | 0.00 | 0.65 ± 0.01 | 0.29 ± 0.00 | 3.45 ± 0.15 | 27.48 ± 0.09 | 0.51 ± 0.01 | 0.00 | 1.12 ± 0.00 | 0.05 ± 0.01 | 27.54 ± 0.00 | 0.69 ± 0.03 |
| 3 | 1.15 ± 0.01 | 0.43 ± 0.00 | 0.00 | 0.65 ± 0.01 | 0.00 | 4.42 ± 0.02 | 21.23 ± 0.03 | 0.11 ± 0.00 | 0.00 | 1.43 ± 0.01 | 1.47 ± 0.01 | 23.04 ± 0.01 | 1.93 ± 0.03 |
| 4 | 0.33 ± 0.00 | 0.29 ± 0.03 | 0.00 | 0.22 ± 0.00 | 0.00 | 0.80 ± 0.05 | 6.32 ± 0.07 | 0.00 | 0.00 | 0.61 ± 0.02 | 1.45 ± 0.02 | 6.34 ± 0.01 | 1.49 ± 0.02 |
| 5 | 0.05 ± 0.00 | 0.69 ± 0.01 | 0.00 | 0.90 ± 0.00 | 0.14 ± 0.00 | 6.58 ± 0.09 | 17.71 ± 0.22 | 0.17 ± 0.00 | 0.00 | 1.23 ± 0.02 | 0.06 ± 0.01 | 22.12 ± 0.00 | 0.95 ± 0.01 |
| 6 | 0.60 ± 0.02 | 0.44 ± 0.05 | 0.00 | 0.44 ± 0.02 | 0.11 ± 0.00 | 1.71 ± 0.02 | 11.08 ± 0.07 | 0.00 | 0.00 | 1.13 ± 0.01 | 0.60 ± 0.04 | 16.44 ± 0.02 | 0.97 ± 0.00 |
| 7 | 1.79 ± 0.03 | 0.68 ± 0.00 | 0.00 | 0.89 ± 0.00 | 0.17 ± 0.00 | 5.01 ± 0.11 | 30.63 ± 0.12 | 0.17 ± 0.00 | 0.00 | 1.42 ± 0.01 | 1.45 ± 0.06 | 31.87 ± 0.01 | 2.43 ± 0.05 |
| 8 | 0.00 | 0.71 ± 0.01 | 0.00 | 1.02 ± 0.01 | 0.29 ± 0.02 | 5.22 ± 0.04 | 29.30 ± 0.09 | 0.45 | 0.00 | 1.29 ± 0.02 | 1.04 ± 0.03 | 30.92 ± 0.01 | 1.93 ± 0.04 |
| 9 | 0.01 ± 0.00 | 0.56 ± 0.01 | 0.00 | 0.41 ± 0.01 | 0.00 | 3.78 ± 0.05 | 9.30 ± 0.26 | 0.00 | 0.00 | 0.75 ± 0.00 | 0.05 ± 0.02 | 11.93 ± 0.00 | 0.46 ± 0.04 |
| 10 | 0.00 | 0.37 ± 0.01 | 0.00 | 0.44 ± 0.01 | 0.00 | 2.01 ± 0.02 | 30.64 ± 0.02 | 0.00 | 0.00 | 0.67 ± 0.02 | 0.55 ± 0.02 | 15.73 ± 0.00 | 0.92 ± 0.03 |
| 11 | 1.42 ± 0.02 | 0.54 ± 0.02 | 0.00 | 0.71 ± 0.02 | 0.14 ± 0.00 | 3.97 ± 0.02 | 24.27 ± 0.05 | 0.14 ± 0.00 | 0.00 | 1.13 ± 0.01 | 1.38 ± 0.05 | 25.25 ± 0.01 | 1.92 ± 0.01 |
| 12 | 1.11 ± 0.00 | 0.52 ± 0.02 | 0.00 | 0.76 ± 0.02 | 0.00 | 4.41 ± 0.06 | 25.58 ± 0.24 | 0.29 ± 0.02 | 0.00 | 1.10 ± 0.00 | 1.27 ± 0.02 | 26.84 ± 0.00 | 1.87 ± 0.06 |
| 13 | 1.37 ± 0.03 | 0.53 ± 0.03 | 0.00 | 0.72 ± 0.03 | 0.00 | 4.47 ± 0.01 | 26.50 ± 0.12 | 0.20 ± 0.02 | 0.00 | 1.42 ± 0.01 | 1.31 ± 0.02 | 27.71 ± 0.00 | 1.87 ± 0.04 |
| Inflorescences | 1.59 ± 0.02 | 0.12 ± 0.00 | 0.19 ± 0.01 | 0.18 ± 0.03 | 0.11 ± 0.00 | 0.59 ± 0.03 | 14.02 ± 0.07 | 0.32 ± 0.01 | 0.00 | 0.28 ± 0.02 | 0.40 ± 0.01 | 30.21 ± 0.03 | 3.69 ± 0.05 |
| Run | DPPH Radical Scavenging Activity (%) |
|---|---|
| 1 | 98.06 ± 0.92 |
| 2 | 96.85 ± 1.37 |
| 3 | 95.58 ± 0.57 |
| 4 | 87.56 ± 0.69 |
| 5 | 94.17 ± 1.12 |
| 6 | 89.00 ± 1.87 |
| 7 | 97.85 ± 0.29 |
| 8 | 97.99 ± 0.92 |
| 9 | 89.07 ± 0.97 |
| 10 | 93.28 ± 0.87 |
| 11 | 97.20 ± 0.39 |
| 12 | 94.93 ± 0.54 |
| 13 | 97.09 ± 1.08 |
| AA | 78.77 ± 2.40 |
| Sample | MIC (µg mL−1) | |||
|---|---|---|---|---|
| B. subtilis | S. aureus | E. coli | P. aeruginosa | |
| 1 | 36.64 | 36.64 | 18.32 | 36.64 |
| 2 | 33.01 | 33.02 | 33.02 | 66.03 |
| 3 | 28.11 | 28.11 | 28.11 | 28.11 |
| 4 | 19.20 | 38.41 | 19.20 | 19.20 |
| 5 | 23.44 | 23.44 | 23.44 | 46.88 |
| 6 | 10.42 | 10.42 | 10.42 | 20.84 |
| 7 | 17.59 | 29.59 | 29.59 | 29.59 |
| 8 | 14.67 | 58.70 | 29.35 | 29.35 |
| 9 | 13.79 | 27.58 | 13.79 | 13.79 |
| 10 | 24.95 | 24.95 | 24.95 | 24.95 |
| 11 | 16.74 | 33.48 | 33.48 | 33.48 |
| 12 | 17.08 | 34.16 | 34.16 | 34.16 |
| 13 | 12.02 | 48.09 | 24.05 | 24.05 |
| C | 0.781 | 0.781 | 0.781 | 1.563 |
| Target | Pressure (Bar) | Temperature (°C) | Desirability |
|---|---|---|---|
| High extraction yield, high CBD, high antioxidative activity, minimum MIC | 284.78 | 60 | 0.817 |
| High extraction yield, high CBD and CBDA, high antioxidative activity, minimum MIC | 294.1 | 58.9 | 0.815 |
| High extraction yield, high monoterpenes, high antioxidative activity, minimum MIC | 131.2 | 60 | 0.521 |
| High extraction yield, high sesquiterpenes, high antioxidative activity, minimum MIC | 319.7 | 58.2 | 0.892 |
| High extraction yield, high terpenes and cannabinoids (most abundant), high antioxidative activity, minimum MIC | 166.9 | 55.7 | 0.503 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jokić, S.; Jerković, I.; Pavić, V.; Aladić, K.; Molnar, M.; Kovač, M.J.; Vladimir-Knežević, S. Terpenes and Cannabinoids in Supercritical CO2 Extracts of Industrial Hemp Inflorescences: Optimization of Extraction, Antiradical and Antibacterial Activity. Pharmaceuticals 2022, 15, 1117. https://doi.org/10.3390/ph15091117
Jokić S, Jerković I, Pavić V, Aladić K, Molnar M, Kovač MJ, Vladimir-Knežević S. Terpenes and Cannabinoids in Supercritical CO2 Extracts of Industrial Hemp Inflorescences: Optimization of Extraction, Antiradical and Antibacterial Activity. Pharmaceuticals. 2022; 15(9):1117. https://doi.org/10.3390/ph15091117
Chicago/Turabian StyleJokić, Stela, Igor Jerković, Valentina Pavić, Krunoslav Aladić, Maja Molnar, Martina Jakovljević Kovač, and Sanda Vladimir-Knežević. 2022. "Terpenes and Cannabinoids in Supercritical CO2 Extracts of Industrial Hemp Inflorescences: Optimization of Extraction, Antiradical and Antibacterial Activity" Pharmaceuticals 15, no. 9: 1117. https://doi.org/10.3390/ph15091117
APA StyleJokić, S., Jerković, I., Pavić, V., Aladić, K., Molnar, M., Kovač, M. J., & Vladimir-Knežević, S. (2022). Terpenes and Cannabinoids in Supercritical CO2 Extracts of Industrial Hemp Inflorescences: Optimization of Extraction, Antiradical and Antibacterial Activity. Pharmaceuticals, 15(9), 1117. https://doi.org/10.3390/ph15091117










