Automated Radiosynthesis, Preliminary In Vitro/In Vivo Characterization of OncoFAP-Based Radiopharmaceuticals for Cancer Imaging and Therapy
Abstract
:1. Introduction
2. Results
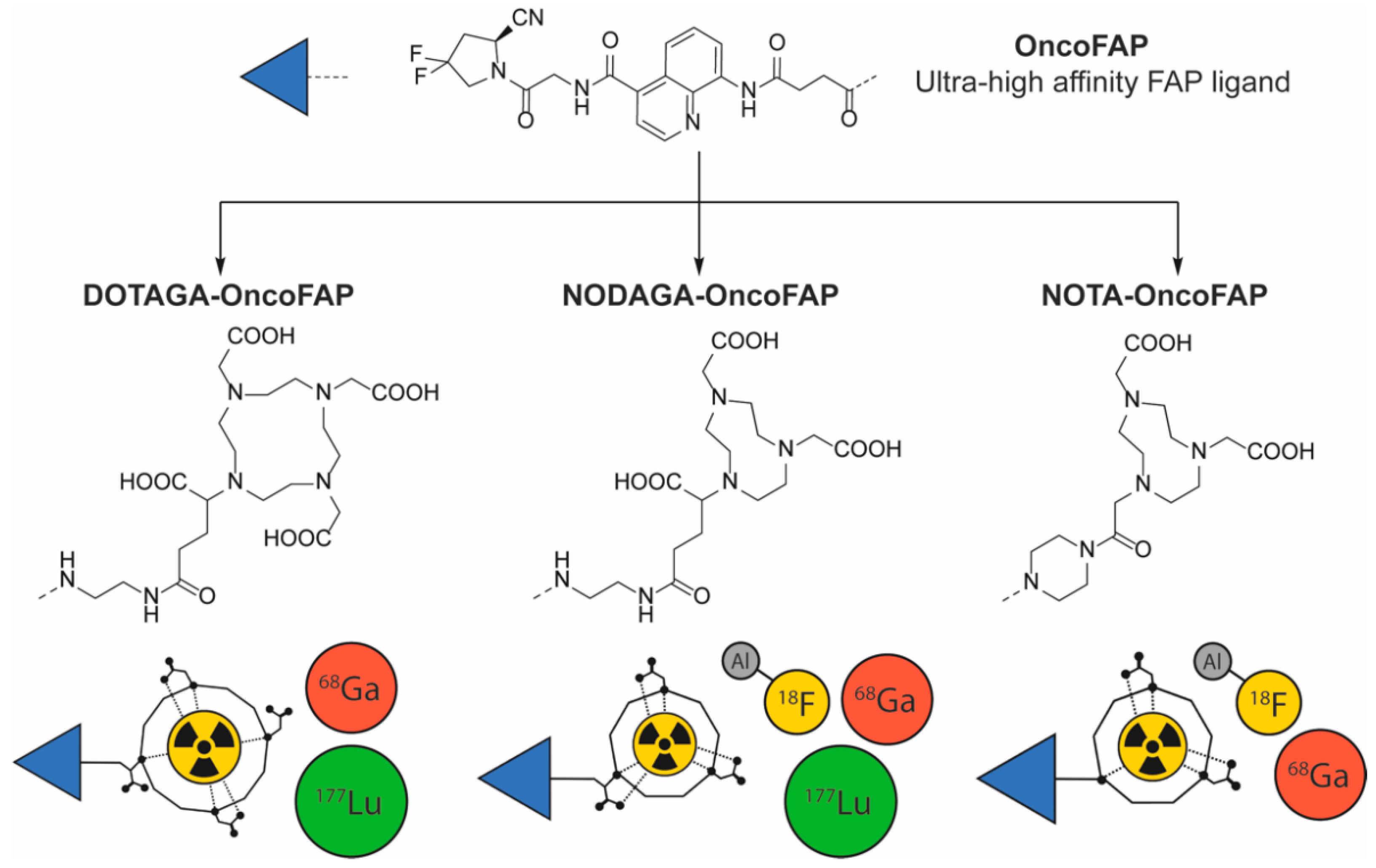
2.1. Radiosynthesis
2.1.1. [68Ga]Ga-OncoFAP-Derivatives
2.1.2. [18F]AlF-NOTA/NODAGA-OncoFAP
2.1.3. [177Lu]Lu-DOTAGA-OncoFAP
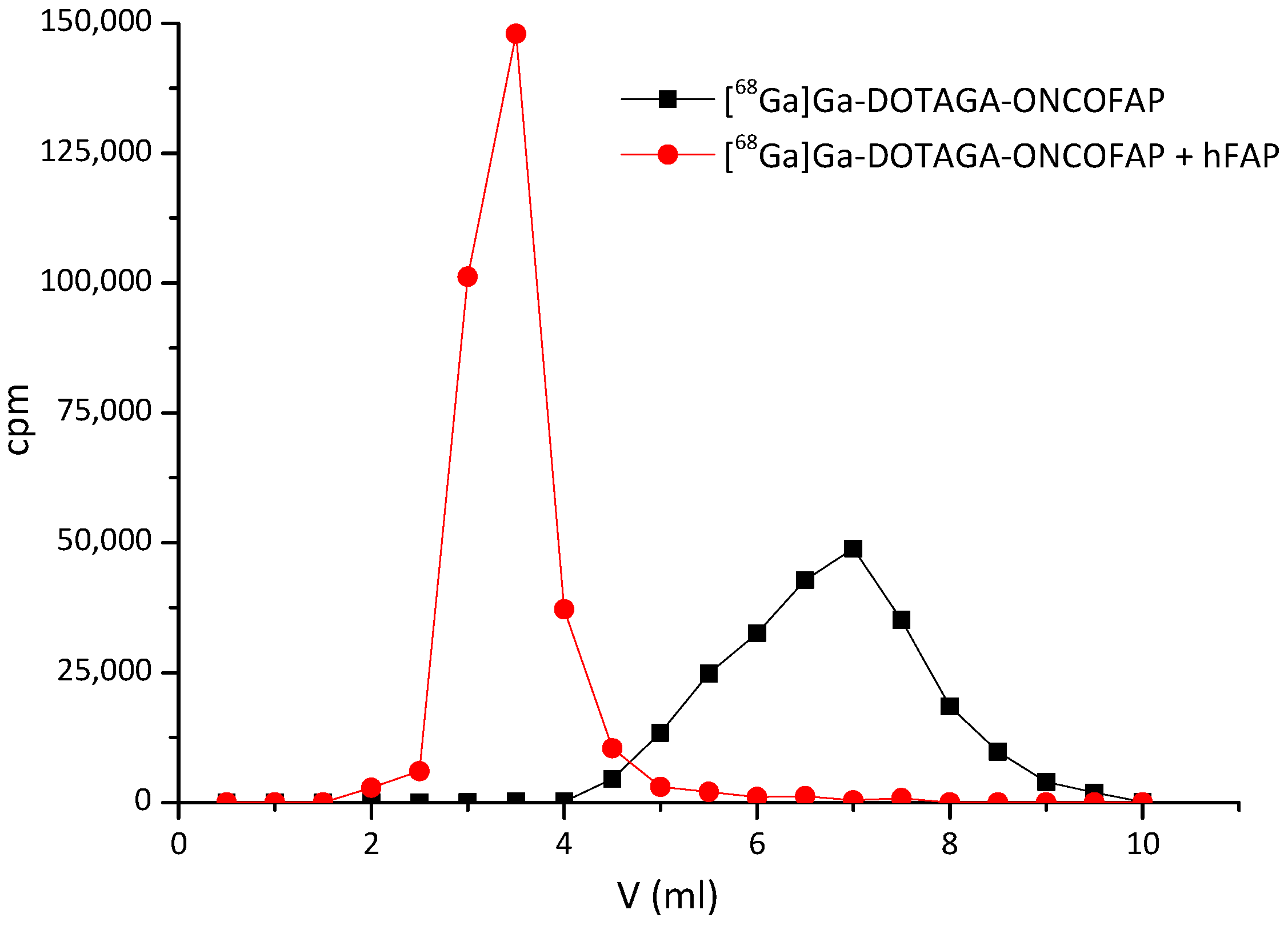
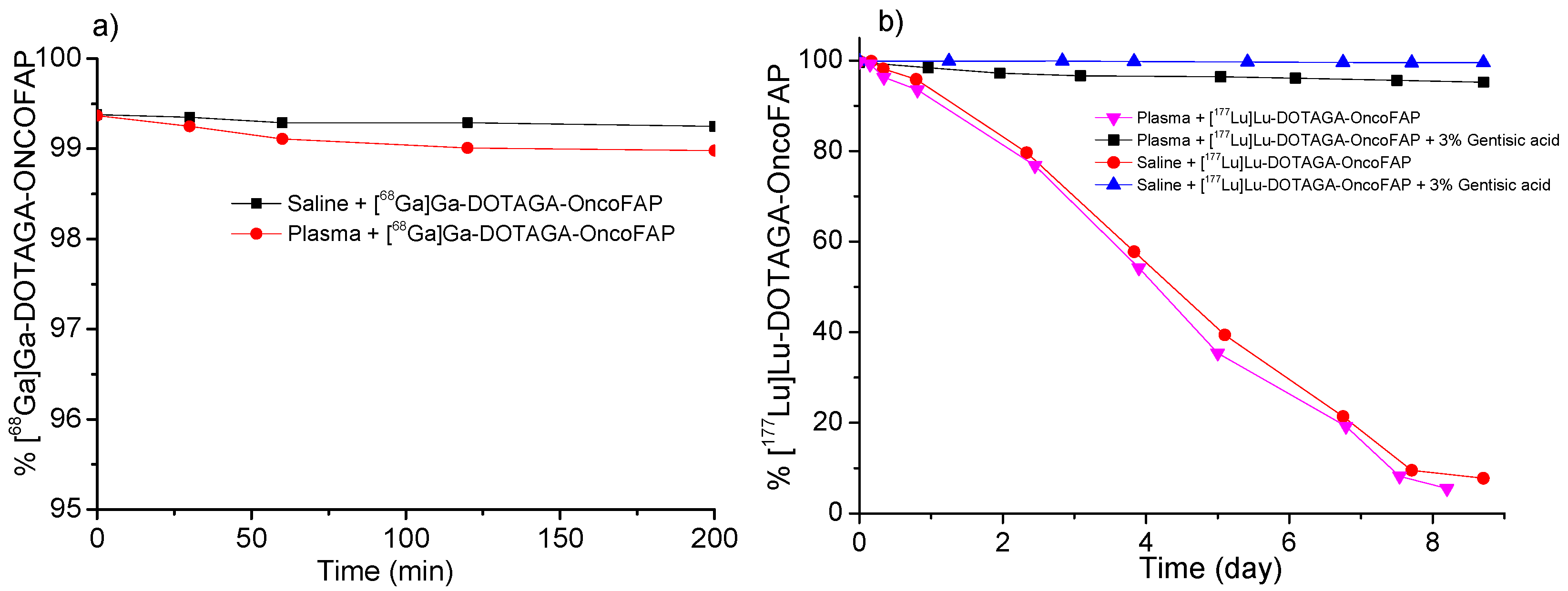
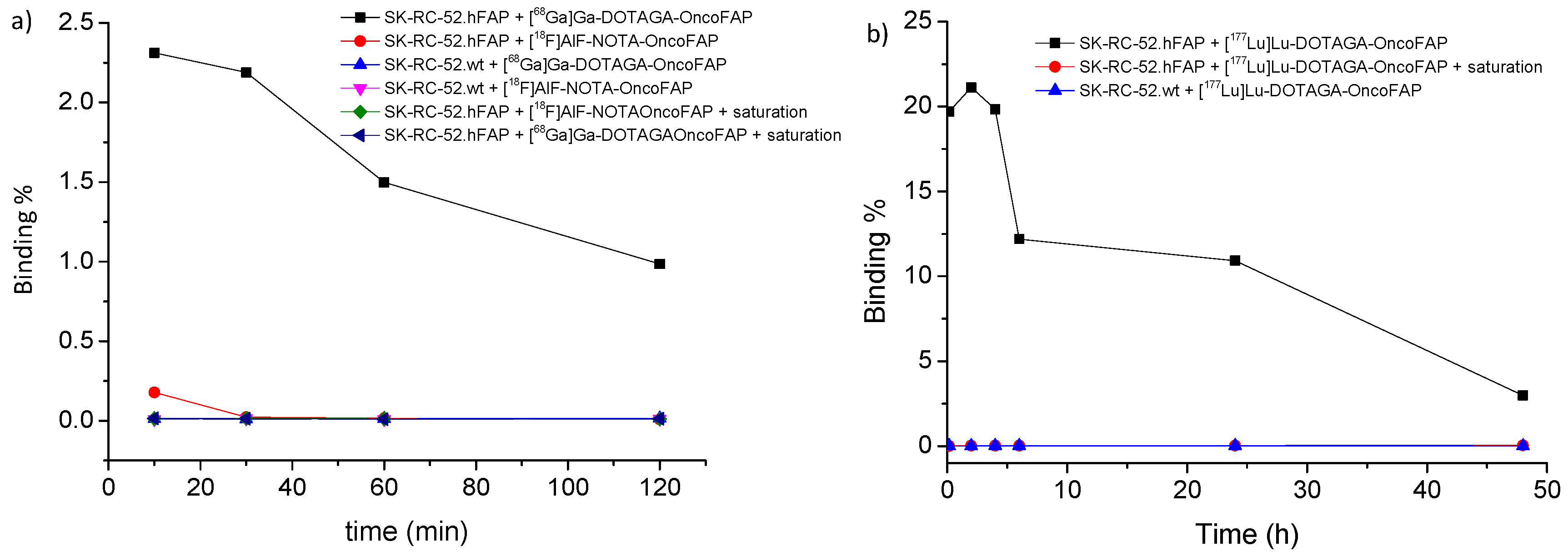
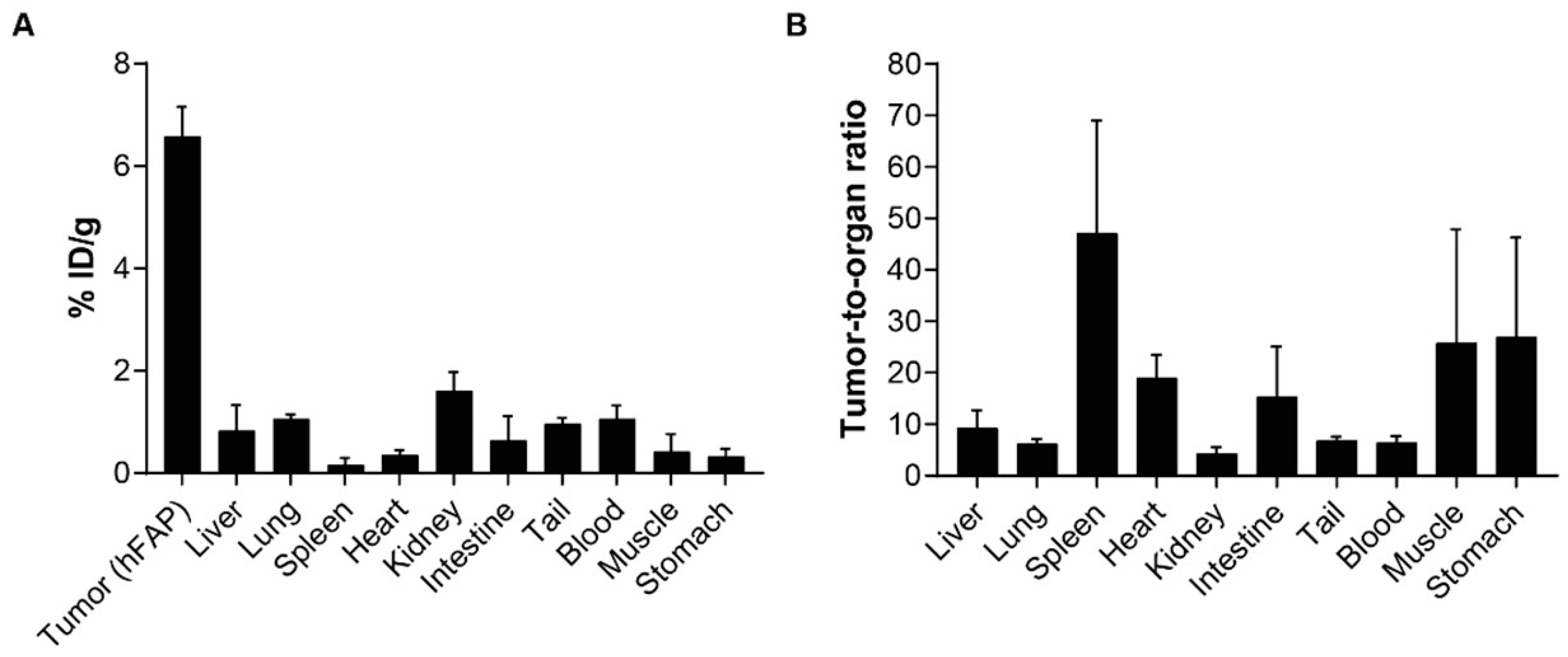
2.2. Preliminary In Vitro/In Vivo Characterization of OncoFA-Derivatives
3. Discussion
4. Materials and Methods
4.1. Chemical Synthesis
4.2. Radiochemistry
4.2.1. [68Ga]GaOncoFAP-Derivatives Synthesis
4.2.2. [18F]AlF-OncoFAP
4.2.3. [177Lu]Lu-OncoFAP
4.2.4. Reproducibility of the Method
4.3. Quality Controls
4.4. In Vitro Stability and Lipophilicity
4.5. Characterization of OncoFAP Binding to hFAP
4.6. Cell Binding
4.7. Animal Studies
4.7.1. Implantation of Subcutaneous Tumors
4.7.2. Biodistribution Studies in Tumor Bearing Mice
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kratochwil, C.; Flechsig, P.; Lindner, T.; Abderrahim, L.; Altmann, A.; Mier, W.; Adeberg, S.; Rathke, H.; Röhrich, M.; Winter, H.; et al. Ga-FAPI PET/CT: Tracer Uptake in 28 Different Kinds of Cancer. J. Nucl. Med. 2019, 60, 801–805. [Google Scholar] [CrossRef] [Green Version]
- Kulterer, O.C.; Pfaff, S.; Wadsak, W.; Garstka, N.; Remzi, M.; Vraka, C.; Nics, L.; Mitterhauser, M.; Bootz, F.; Cazzamalli, S.; et al. A Microdosing Study with 99mTc-PHC-102 for the SPECT/CT Imaging of Primary and Metastatic Lesions in Renal Cell Carcinoma Patients. J. Nucl. Med. 2021, 62, 360–365. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R. The biology and function of fibroblasts in cancer. Nat. Rev. Cancer 2016, 16, 582–598. [Google Scholar] [CrossRef] [PubMed]
- Pang, Y.; Zhao, L.; Shang, Q.; Meng, T.; Feng, L.; Wang, S.; Guo, P.; Wu, X.; Lin, Q.; Wu, H.; et al. Positron emission tomography and computed tomography with [68Ga]Ga-fibroblast activation protein inhibitors improves tumor detection and staging in patients with pancreatic cancer. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 1322–1337. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Pang, Y.; Yao, L.; Zhao, L.; Fan, C.; Ke, J.; Guo, P.; Hao, B.; Fu, H.; Xie, C.; et al. Imaging fibroblast activation protein in liver cancer: A single-center post hoc retrospective analysis to compare [68Ga]Ga-FAPI-04 PET/CT versus MRI and [18F]-FDG PET/CT. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 1604–1617. [Google Scholar] [CrossRef] [PubMed]
- Giesel, F.L.; Kratochwil, C.; Schlittenhardt, J.; Dendl, K.; Eiber, M.; Staudinger, F.; Kessler, L.; Fendler, W.P.; Lindner, T.; Koerber, S.A.; et al. Head-to-head intra-individual comparison of biodistribution and tumor uptake of 68Ga-FAPI and 18F-FDG PET/CT in cancer patients. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 4377–4385. [Google Scholar] [CrossRef]
- Gündoğan, C.; Kömek, H.; Can, C.; Yildirim, Ö.; Kaplan, İ.; Erdur, E.; Poyraz, K.; Güzel, Y.; Oruç, Z.; Çakabay, B. Comparison of 18F-FDG PET/CT and 68Ga-FAPI-04 PET/CT in the staging and restaging of gastric adenocarcinoma. Nucl. Med. Commun. 2022, 43, 64–72. [Google Scholar] [CrossRef]
- Backhaus, P.; Gierse, F.; Burg, M.C.; Büther, F.; Asmus, I.; Dorten, P.; Cufe, J.; Roll, W.; Neri, D.; Cazzamalli, S.; et al. Translational imaging of the fibroblast activation protein (FAP) using the new ligand [68Ga]Ga-OncoFAP-DOTAGA. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 1822–1832. [Google Scholar] [CrossRef]
- Millul, J.; Bassi, G.; Mock, J.; Elsayed, A.; Pellegrino, C.; Zana, A.; Dakhel Plaza, S.; Nadal, L.; Gloger, A.; Schmidt, E.; et al. An ultra-high-affinity small organic ligand of fibroblast activation protein for tumor-targeting applications. Proc. Natl. Acad. Sci. USA 2021, 118, e2101852118. [Google Scholar] [CrossRef]
- Strosberg, J.; El-Haddad, G.; Wolin, E.; Hendifar, A.; Yao, J.; Chasen, B.; Mittra, E.; Kunz, P.L.; Kulke, M.H.; Jacene, H.; et al. Phase 3 Trial of [177Lu]-Dotatate for Midgut Neuroendocrine Tumors. N. Engl. J. Med. 2017, 376, 125–135. [Google Scholar] [CrossRef]
- Sartor, O.; de Bono, J.; Chi, K.N.; Fizazi, K.; Herrmann, K.; Rahbar, K.; Tagawa, S.T.; Nordquist, L.T.; Vaishampayan, N.; El-Haddad, G.; et al. Lutetium-177-PSMA-617 for Metastatic Castration-Resistant Prostate Cancer. N. Engl. J. Med. 2021, 385, 1091–1103. [Google Scholar] [CrossRef] [PubMed]
- Price, E.W.; Orvig, C. Matching chelators to radiometals for radiopharmaceuticals. Chem. Soc. Rev. 2014, 43, 260–290. [Google Scholar] [CrossRef] [PubMed]
- Kostelnik, T.I.; Orvig, C. Radioactive Main Group and Rare Earth Metals for Imaging and Therapy. Chem. Rev. 2019, 119, 902–956. [Google Scholar] [CrossRef]
- Kubícek, V.; Havlícková, J.; Kotek, J.; Tircsó, G.; Hermann, P.; Tóth, E.; Lukes, I. Gallium(III) complexes of DOTA and DOTA-monoamide: Kinetic and thermodynamic studies. Inorg. Chem. 2010, 49, 10960–10969. [Google Scholar] [CrossRef]
- Trindade, V.; Balter, H. Oxidant and Antioxidant Effects of Gentisic Acid in a [177Lu]-Labelled Methionine-Containing Minigastrin Analogue. Curr. Radiopharm. 2020, 13, 107–119. [Google Scholar] [CrossRef] [PubMed]
- Joshi, R.; Gangabhagirathi, R.; Venu, S.; Adhikari, S.; Mukherjee, T. Antioxidant activity and free radical scavenging reactions of gentisic acid: In-vitro and pulse radiolysis studies. Free Radic. Res. 2012, 46, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Baumgarten, J.; Groener, D.; Nguyen Ngoc, C.; Mader, N.; Chaurasia, M.; Davis, K.; Wichert, J.; Chun, F.K.H.; Tselis, N.; Happel, C.; et al. Safety and Efficacy of 177-Lutetium-PSMA-617 Radioligand Therapy Shortly after Failing 223-Radium-Dichloride. Cancers 2022, 14, 557. [Google Scholar] [CrossRef]
- Claringbold, P.G.; Turner, J.H. NeuroEndocrine Tumor Therapy with Lutetium-177-octreotate and Everolimus (NETTLE): A Phase I Study. Cancer Biother. Radiopharm. 2015, 30, 261–269. [Google Scholar] [CrossRef]
- Gentles, A.J.; Newman, A.M.; Liu, C.L.; Bratman, S.V.; Feng, W.; Kim, D.; Nair, V.S.; Xu, Y.; Khuong, A.; Hoang, C.D.; et al. The prognostic landscape of genes and infiltrating immune cells across human cancers. Nat. Med. 2015, 21, 938–945. [Google Scholar] [CrossRef]
- Valkenburg, K.C.; de Groot, A.E.; Pienta, K.J. Targeting the tumour stroma to improve cancer therapy. Nat. Rev. Clin. Oncol. 2018, 15, 366–381. [Google Scholar] [CrossRef]
- Schmidt, A.; Müller, D.; Mersmann, M.; Wüest, T.; Gerlach, E.; Garin-Chesa, P.; Rettig, W.J.; Pfizenmaier, K.; Moosmayer, D. Generation of human high-affinity antibodies specific for the fibroblast activation protein by guided selection. Eur. J. Biochem. 2001, 268, 1730–1738. [Google Scholar] [CrossRef] [PubMed]
- Welt, S.; Divgi, C.R.; Scott, A.M.; Garin-Chesa, P.; Finn, R.D.; Graham, M.; Carswell, E.A.; Cohen, A.; Larson, S.M.; Old, L.J. Antibody targeting in metastatic colon cancer: A phase I study of monoclonal antibody F19 against a cell-surface protein of reactive tumor stromal fibroblasts. J. Clin. Oncol. 1994, 12, 1193–1203. [Google Scholar] [CrossRef] [PubMed]
- Tanswell, P.; Garin-Chesa, P.; Rettig, W.J.; Welt, S.; Divgi, C.R.; Casper, E.S.; Finn, R.D.; Larson, S.M.; Old, L.J.; Scott, A.M. Population pharmacokinetics of antifibroblast activation protein monoclonal antibody F19 in cancer patients. Br. J. Clin. Pharmacol. 2001, 51, 177–180. [Google Scholar] [CrossRef] [PubMed]
- Mersmann, M.; Schmidt, A.; Rippmann, J.F.; Wüest, T.; Brocks, B.; Rettig, W.J.; Garin-Chesa, P.; Pfizenmaier, K.; Moosmayer, D. Human antibody derivatives against the fibroblast activation protein for tumor stroma targeting of carcinomas. Int. J. Cancer 2001, 92, 240–248. [Google Scholar] [CrossRef]
- Galbiati, A.; Zana, A.; Bocci, M.; Millul, J.; Elsayed, A.; Mock, J.; Neri, D.; Cazzamalli, S. A novel dimeric FAP-targeting small molecule-radio conjugate with high and prolonged tumour uptake. J. Nucl. Med. 2022, 63. [Google Scholar] [CrossRef]
- Kolenc Peitl, P.; Rangger, C.; Garnuszek, P.; Mikolajczak, R.; Hubalewska-Dydejczyk, A.; Maina, T.; Erba, P.; Decristoforo, C. Clinical translation of theranostic radiopharmaceuticals: Current regulatory status and recent examples. J. Label. Comp. Radiopharm. 2019, 62, 673–683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khan, I.; Berg, T.C.; Brown, J.; Bhalla, R.; Wilson, A.; Black, A.; McRobbie, G.; Nairne, J.; Olsson, A.; Trigg, W. Development of an automated, GMP compliant FASTlab™ radiosynthesis of [18F]GE-179 for the clinical study of activated NMDA receptors. J. Label. Comp. Radiopharm. 2020, 63, 183–195. [Google Scholar] [CrossRef]
- Edwards, R.; Greenwood, H.E.; McRobbie, G.; Khan, I.; Witney, T.H. Robust and Facile Automated Radiosynthesis of [18F]FSPG on the GE FASTlab. Mol. Imaging Biol. 2021, 23, 854–864. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Jain, M.K.; Cai, H. HPLC-free and cassette-based nucleophilic production of [18F]FDOPA for clinical use. Am. J. Nucl. Med. Mol. Imaging 2021, 11, 290–299. [Google Scholar]
- Illucix Information. Available online: http://illuccixhcp.com/wp-content/uploads/illuccix-prescribing-information.pdf (accessed on 2 February 2022).
- Netspot Information. Available online: https://www.rxlist.com/netspot-drug.htm (accessed on 2 February 2022).
- Somakit-TOC Information. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/somakit-toc (accessed on 2 February 2022).
- Lepareur, N. Cold Kit Labeling: The Future of 68Ga Radiopharmaceuticals? Front. Med. (Lausanne) 2022, 9, 812050. [Google Scholar] [CrossRef]
- Kleynhans, J.; Rubow, S.; le Roux, J.; Marjanovic-Painter, B.; Zeevaart, J.R.; Ebenhan, T. Production of [68Ga]Ga-PSMA: Comparing a manual kit-based method with a module-based automated synthesis approach. J. Label. Comp. Radiopharm. 2020, 63, 553–563. [Google Scholar] [CrossRef]
- Migliari, S.; Sammartano, A.; Scarlattei, M.; Baldari, G.; Janota, B.; Bonadonna, R.C.; Ruffini, L. Feasibility of a Scale-down Production of [68Ga]Ga-NODAGA-Exendin-4 in a Hospital Based Radiopharmacy. Curr. Radiopharm. 2022, 15, 63–75. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.E.; Sladen, H.L.; Biagini, S.C.; Blower, P.J. Inorganic approaches for radiolabelling biomolecules with fluorine-18 for imaging with positron emission tomography. Dalton Trans. 2011, 40, 6196–6205. [Google Scholar] [CrossRef] [PubMed]
- Bruce Martin, R. Ternary complexes of Al3+ and F− with a third ligand. Coord. Chem. Rev. 1996, 149, 23–32. [Google Scholar] [CrossRef]
- Antonny, B.; Chabre, M. Characterization of the aluminum and beryllium fluoride species which activate transducin. Analysis of the binding and dissociation kinetics. J. Biol. Chem. 1992, 267, 6710–6718. [Google Scholar] [CrossRef]
- Li, L. The biochemistry and physiology of metallic fluoride: Action, mechanism, and implications. Crit. Rev. Oral Biol. Med. 2003, 14, 100–114. [Google Scholar] [CrossRef] [Green Version]
- McBride, W.J.; Sharkey, R.M.; Karacay, H.; D’Souza, C.A.; Rossi, E.A.; Laverman, P.; Chang, C.H.; Boerman, O.C.; Goldenberg, D.M. A novel method of 18F radiolabeling for PET. J. Nucl. Med. 2009, 50, 991–998. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Hu, X.; Liu, H.; Bu, L.; Ma, X.; Cheng, K.; Li, J.; Tian, M.; Zhang, H.; Cheng, Z. A comparative study of radiolabeled bombesin analogs for the PET imaging of prostate cancer. J. Nucl. Med. 2013, 54, 2132–2138. [Google Scholar] [CrossRef] [Green Version]
- Chakraborty, S.; Chakravarty, R.; Shetty, P.; Vimalnath, K.V.; Sen, I.B.; Dash, A. Prospects of medium specific activity 177Lu in targeted therapy of prostate cancer using 177Lu-labeled PSMA inhibitor. J. Label. Comp. Radiopharm. 2016, 59, 364–371. [Google Scholar] [CrossRef]
- Luurtsema, G.; Pichler, V.; Bongarzone, S.; Seimbille, Y.; Elsinga, P.; Gee, A.; Vercouillie, J. EANM guideline for harmonisation on molar activity or specific activity of radiopharmaceuticals: Impact on safety and imaging quality. EJNMMI Radiopharm. Chem. 2021, 6, 34. [Google Scholar] [CrossRef]
- Le Bars, D. Fluorine-18 and medical imaging: Radiopharmaceuticals for positron emission tomography. J. Fluor. Chem. 2006, 127, 1488–1493. [Google Scholar] [CrossRef]
- Sanchez-Crespo, A. Comparison of Gallium-68 and Fluorine-18 imaging characteristics in positron emission tomography. Appl. Radiat. Isot. 2013, 76, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Treglia, G.; Muoio, B.; Roustaei, H.; Kiamanesh, Z.; Aryana, K.; Sadeghi, R. Head-to-Head Comparison of Fibroblast Activation Protein Inhibitors (FAPI) Radiotracers versus [18F]F-FDG in Oncology: A Systematic Review. Int. J. Mol. Sci. 2021, 22, 11192. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Chen, S.; Lin, L.; Sun, L.; Wu, H.; Lin, Q.; Chen, H. [68Ga]Ga-DOTA-FAPI-04 improves tumor staging and monitors early response to chemoradiotherapy in a patient with esophageal cancer. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 3188–3189. [Google Scholar] [CrossRef]
- Dendl, K.; Finck, R.; Giesel, F.L.; Kratochwil, C.; Lindner, T.; Mier, W.; Cardinale, J.; Kesch, C.; Röhrich, M.; Rathke, H.; et al. FAP imaging in rare cancer entities-first clinical experience in a broad spectrum of malignancies. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 721–731. [Google Scholar] [CrossRef] [PubMed]
- Giesel, F.L.; Heussel, C.P.; Lindner, T.; Röhrich, M.; Rathke, H.; Kauczor, H.U.; Debus, J.; Haberkorn, U.; Kratochwil, C. FAPI-PET/CT improves staging in a lung cancer patient with cerebral metastasis. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1754–1755. [Google Scholar] [CrossRef]
- Syed, M.; Flechsig, P.; Liermann, J.; Windisch, P.; Staudinger, F.; Akbaba, S.; Koerber, S.A.; Freudlsperger, C.; Plinkert, P.K.; Debus, J.; et al. Fibroblast activation protein inhibitor (FAPI) PET for diagnostics and advanced targeted radiotherapy in head and neck cancers. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 2836–2845. [Google Scholar] [CrossRef]
| Radiosynthesis [68Ga]Ga-NOTA-OncoFAP | |||
|---|---|---|---|
| Condition 1 | Condition 2 | Condition 3 | |
| NOTA-OncoFAP (µg) | 15 | 20 | 25 |
| Reaction time (min) | 10 | 5 | 5 |
| Starting activity (MBq) | 715 ± 115 | 780 ± 150 | 770 ± 135 |
| Final activity (MBq) | 580 ± 110 | 641 ± 135 | 650 ± 116 |
| RCY (%) | 78 ± 3 | 82 ± 2 | 84 ± 1 |
| RCY decay corrected (%) | 89 ± 3 | 88 ± 2 | 90 ± 1 |
| Final molar activity (GBq/µmol) | 39 ± 7 | 32 ± 7 | 26 ± 5 |
| RCP (%) | 99.1 ± 0.1 | 99.1 ± 0.1 | 99.1 ± 0.1 |
| Radiosynthesis [68Ga]Ga-NODAGA-OncoFAP | |||
|---|---|---|---|
| Condition 1 | Condition 2 | Condition 3 | |
| NODAGA-OncoFAP (µg) | 15 | 20 | 25 |
| Reaction time (min) | 10 | 5 | 5 |
| Starting activity (MBq) | 1085 ± 95 | 1000 ± 74 | 1100 ± 60 |
| Final activity (MBq) | 880 ± 70 | 840 ± 56 | 954 ± 28 |
| RCY (%) | 81 ± 2 | 84 ± 2 | 87 ± 2 |
| RCY decay corrected (%) | 93 ± 2 | 90 ± 2 | 93 ± 3 |
| Final molar activity (GBq/µmol) | 59 ± 5 | 42 ± 3 | 38 ± 2 |
| RCP (%) | 99.2 ± 0.1 | 99.2 ± 0.1 | 99.2 ± 0.1 |
| Radiosynthesis [68Ga]Ga-DOTAGA-OncoFAP | |||
|---|---|---|---|
| Condition 1 | Condition 2 | Condition 3 | |
| DOTAGA-OncoFAP (µg) | 15 | 20 | 25 |
| Reaction time (min) | 10 | 5 | 5 |
| Starting activity (MBq) | 1159 ± 93 | 1208 ± 93 | 1159 ± 57 |
| Final activity (MBq) | 924 ± 99 | 942 ± 60 | 915 ± 33 |
| RCY (%) | 75 ± 3 | 78 ± 2 | 79 ± 2 |
| RCY decay corrected (%) | 84.0 ± 3 | 87 ± 2 | 88 ± 2 |
| Final molar activity (GBq/µmol) | 55 ± 6 | 42 ± 3 | 33 ± 1 |
| RCP (%) | 99.3 ± 0.1 | 99.3 ± 0.1 | 99.4 ± 0.1 |
| Parameters | Acceptance Criteria | Average NOTA-OncoFAP | Average NODAGA-OncoFAP | Average DOTAGA-OncoFAP |
|---|---|---|---|---|
| [68Ga]-labeled activity MBq | 200–1300 | 520–580 | 900–970 | 850–920 |
| Volume (mL) | 12 | 12 | 12 | 12 |
| Aspect | Clear colorless solution | Clear colorless solution | Clear colorless solution | Clear colorless solution |
| Half-life (min) | 62–74 | 66.8 | 66.9 | 67.1 |
| - 68Ga3+ - 68Ga colloidal - RCP | ≤2% ≤3% ≥95% | 0.1 ± 0.1% 0.8 ± 0.1% 99.1 ± 0.1% | 0.1 ± 0.1% 0.7 ± 0.1% 99.2 ± 0.1% | 0.1 ± 0.1% 0.5 ± 0.1% 99.4 ± 0.1% |
| Ethanol concentration (v/v) | <10% | 4.9% ± 0.5% | 4.8% ± 0.5% | 4.9% ± 0.5% |
| 68Ge breakthrough | <10−3% | <10−4% | <10−4% | <10−4% |
| pH | 4.0–8.0 | 5.0 ± 0.3 | 5.0 ± 0.3 | 5.0 ± 0.3 |
| Bacterial endotoxins | <175 EU/V | <6 EU/V | <6 EU/V | <6 EU/V |
| Filter integrity (psi) | ≥50 | >50 | >50 | >50 |
| Radiosynthesis [68Ga]Ga-NODAGA-OncoFAP (Simple Preparation) | ||
|---|---|---|
| Formate Buffer | Acetate Buffer | |
| NODAGA-OncoFAP (µg) | 40 | 40 |
| Reaction pH | 3.0 ± 0.1 | 4.4 ± 0.1 |
| Starting activity (GBq) | 1.1 ± 0.2 | 1.1 ± 0.2 |
| Final molar activity (GBq/µmol) | 26 ± 5 | 26 ± 5 |
| - 68Ga3+ content - Colloidal 68Ga content - RCP | 3.4 ± 2.6% 1.5 ± 0.2% 95.1 ± 2.5% | 7.9 ± 1.7% 4.6 ± 1.7% 87.5 ± 2.8% |
| Radiosynthesis [68Ga]Ga-DOTAGA-OncoFAP (Simple Preparation) | ||
|---|---|---|
| Formate Buffer | Acetate Buffer | |
| DOTAGA-OncoFAP (µg) | 40 | 40 |
| Reaction pH | 3.0 ± 0.1 | 4.4 ± 0.1 |
| Starting activity (GBq) | 1.1 ± 0.2 | 1.1 ± 0.2 |
| Final molar activity (GBq/µmol) | 26 ± 5 | 26 ± 5 |
| - 68Ga3+ content - Colloidal 68Ga content - RCP | 3.1 ± 1.1% 2.9 ± 0.3% 94.0 ± 1.4% | 5.6 ± 2.7% 6.4 ± 1.8% 88.0 ± 2.4% |
| Radiosynthesis of [18F]AlF-NOTA-OncoFAP | |||
|---|---|---|---|
| Condition 1 | Condition 2 | Condition 3 | |
| NOTA-OncoFAP (µg) | 200 | 250 | 300 |
| Initial activity (MBq) | 3970 ± 203 | 9250 ± 147 | 16280 ± 976 |
| Product activity (MBq) | 678 ± 33 | 1750 ± 100 | 2967 ± 25 |
| RCY (%) | 16.9 ± 0.5 | 18.9 ± 0.3 | 18.2 ± 1.2 |
| RCY decay corrected (%) | 18.7 ± 0.5 | 20.9 ± 0.3 | 20.2 ± 1.2 |
| Final molar activity (GBq/µmol) | 3.05 ± 0.3 | 6.6 ± 0.8 | 9.3 ± 0.7 |
| RCP (%) | 92.0 ± 0.5 | 89.6 ± 0.8 | 90.5 ± 0.9 |
| Radiosynthesis of [18F]AlF-NODAGA-OncoFAP | |||
|---|---|---|---|
| Condition 1 | Condition 2 | Condition 3 | |
| NODAGA-OncoFAP (µg) | 200 | 250 | 300 |
| Initial activity (MBq) | 3219 ± 150 | 4255 ± 185 | 6660 ± 220 |
| Product activity (MBq) | 78 ± 15 | 96 ± 21 | 137 ± 15 |
| RCY (%) | 2.4 ± 0.5 | 2.3 ± 0.5 | 2.1 ± 0.3 |
| RCY decay corrected (%) | 2.6 ± 0.5 | 2.5 ± 0.5 | 2.3 ± 0.3 |
| Final molar activity (GBq/µmol) | 0.36 ± 0.10 | 0.35 ± 0.10 | 0.42 ± 0.13 |
| RCP (%) | 60.3 ± 5.2 | 68.4 ± 5.8 | 67.5 ± 4.8 |
| Parameters | Acceptance Criteria | Average NOTA-OncoFAP | Average NODAGA-OncoFAP |
|---|---|---|---|
| [18F]-labeled activity | 200–4000 MBq | 2806–3205 MBq | 210–230 MBq |
| Aspect | Clear colorless solution | Clear colorless solution | Clear colorless solution |
| RCY (%) | 95% | 20.2 ± 1.5 | 2.0 ± 0.7 |
| - 18F-content - Other - RCP | ≤2% ≤3% ≥95% | 4.1 ± 0.2% 5.9 ± 0.4% 90.6 ± 0.6% | 12.4 ± 2.5% 27.3 ± 1.4% 60.3 ± 1.3% |
| Ethanol concentration (v/v) | <10% | 5.6 ± 0.5% | 5.4 ± 0.5% |
| pH | 4.0–8.0 | 5.5 ± 0.3 | 5.5 ± 0.3 |
| Bacterial endotoxins | <175 EU/V | <6 EU/V | <6 EU/V |
| Filter integrity (psi) | ≥50 | >50 | >50 |
| Radiosynthesis of [177Lu]Lu-DOTAGA-OncoFAP | ||||
|---|---|---|---|---|
| Condition 1 | Condition 2 | Condition 3 | Condition 4 | |
| DOTAGA-OncoFAP (µg) | 13.3 ± 0.25 | 5.1 ± 0.15 | 8.2 ± 0.50 | 5.3 ± 0.15 |
| Reaction volume (mL) | 2.5 | 2.5 | 2.5 | 1.5 |
| Initial activity (MBq) | 592 ± 9 | 870 ± 6 | 1047 ± 19 | 573 ± 6 |
| Product activity (MBq) | 577 ± 8 | 307 ± 5 | 670 ± 25 | 533 ± 6 |
| RCY % | 97.4 ± 0.4 | 35.3 ± 1.9 | 54.8 ± 1.3 | 92.9 ± 0.75 |
| Gentisic acid (mg) | 0 | 20 | 20 | 20 |
| Final molar activity (GBq/µmol) | 43 ± 2 | 60 ± 5 | 83 ± 3 | 100 ± 3 |
| RCP (%) | > 99.8 | >99.8 | > 99.8 | > 99.8 |
| [177Lu]Lu-DOTAGA-OncoFAP | ||
|---|---|---|
| Parameters | Acceptance Criteria | Average DOTAGA-OncoFAP |
| [177Lu]-labeled activity (GBq) | 5–9 | 7.4–7.7 |
| Aspect | Clear colorless solution | Clear colorless solution |
| - 177LuCl3 - Others - [177Lu]Lu-DOTAGA-OncoFAP- | ≤2% ≤3% ≥95% | 1.9 ± 0.5% - 98.1 ± 0.5% |
| Ethanol concentration (v/v) | <10% | 3.2 ± 0.9% |
| pH | 4.0–8.0 | 5.5 ± 0.3 |
| RCY | 95 ± 2.6% | |
| Bacterial endotoxins | <175 EU/V | <6 EU/V |
| Final molar activity (GBq/µmol) | 105 ± 5 | |
| Filter integrity (psi) | ≥50 | >50 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bartoli, F.; Elsinga, P.; Nazario, L.R.; Zana, A.; Galbiati, A.; Millul, J.; Migliorini, F.; Cazzamalli, S.; Neri, D.; Slart, R.H.J.A.; et al. Automated Radiosynthesis, Preliminary In Vitro/In Vivo Characterization of OncoFAP-Based Radiopharmaceuticals for Cancer Imaging and Therapy. Pharmaceuticals 2022, 15, 958. https://doi.org/10.3390/ph15080958
Bartoli F, Elsinga P, Nazario LR, Zana A, Galbiati A, Millul J, Migliorini F, Cazzamalli S, Neri D, Slart RHJA, et al. Automated Radiosynthesis, Preliminary In Vitro/In Vivo Characterization of OncoFAP-Based Radiopharmaceuticals for Cancer Imaging and Therapy. Pharmaceuticals. 2022; 15(8):958. https://doi.org/10.3390/ph15080958
Chicago/Turabian StyleBartoli, Francesco, Philip Elsinga, Luiza Reali Nazario, Aureliano Zana, Andrea Galbiati, Jacopo Millul, Francesca Migliorini, Samuele Cazzamalli, Dario Neri, Riemer H. J. A. Slart, and et al. 2022. "Automated Radiosynthesis, Preliminary In Vitro/In Vivo Characterization of OncoFAP-Based Radiopharmaceuticals for Cancer Imaging and Therapy" Pharmaceuticals 15, no. 8: 958. https://doi.org/10.3390/ph15080958
APA StyleBartoli, F., Elsinga, P., Nazario, L. R., Zana, A., Galbiati, A., Millul, J., Migliorini, F., Cazzamalli, S., Neri, D., Slart, R. H. J. A., & Erba, P. A. (2022). Automated Radiosynthesis, Preliminary In Vitro/In Vivo Characterization of OncoFAP-Based Radiopharmaceuticals for Cancer Imaging and Therapy. Pharmaceuticals, 15(8), 958. https://doi.org/10.3390/ph15080958







