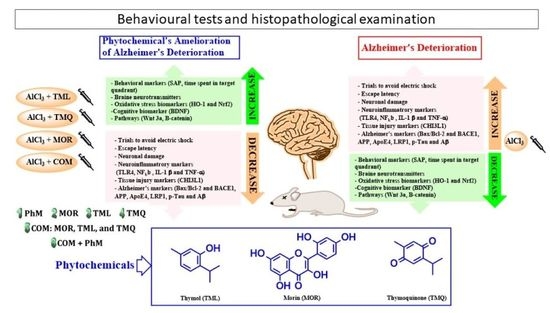
Neuroprotective Effects of Phytochemicals against Aluminum Chloride-Induced Alzheimer’s Disease through ApoE4/LRP1, Wnt3/β-Catenin/GSK3β, and TLR4/NLRP3 Pathways with Physical and Mental Activities in a Rat Model
Abstract
:1. Introduction
2. Results
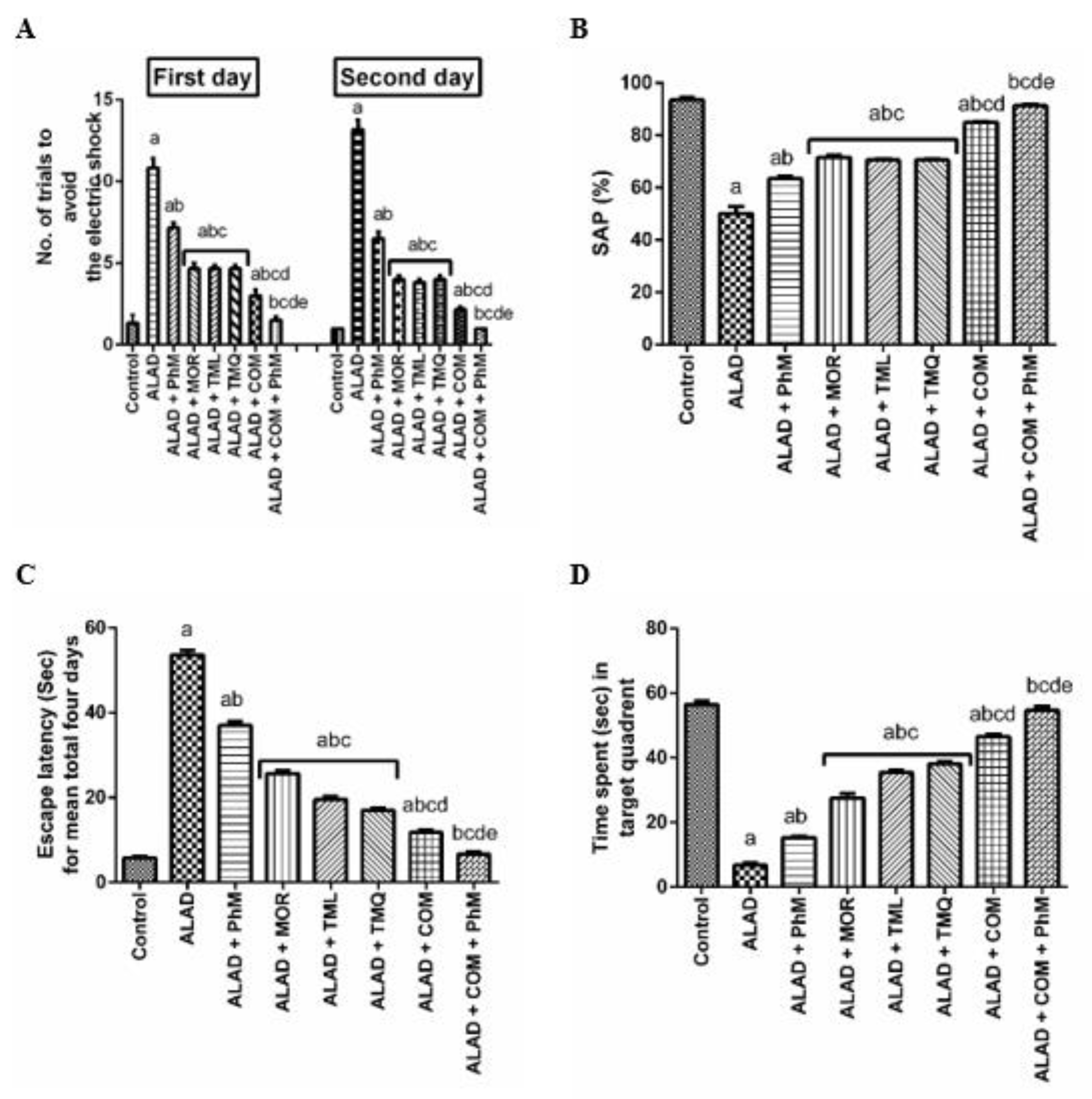
2.1. A Combination of MOR, TML, and TMQ with PhM Ameliorates Learning and Memory Impairment in ALAD Rat Model
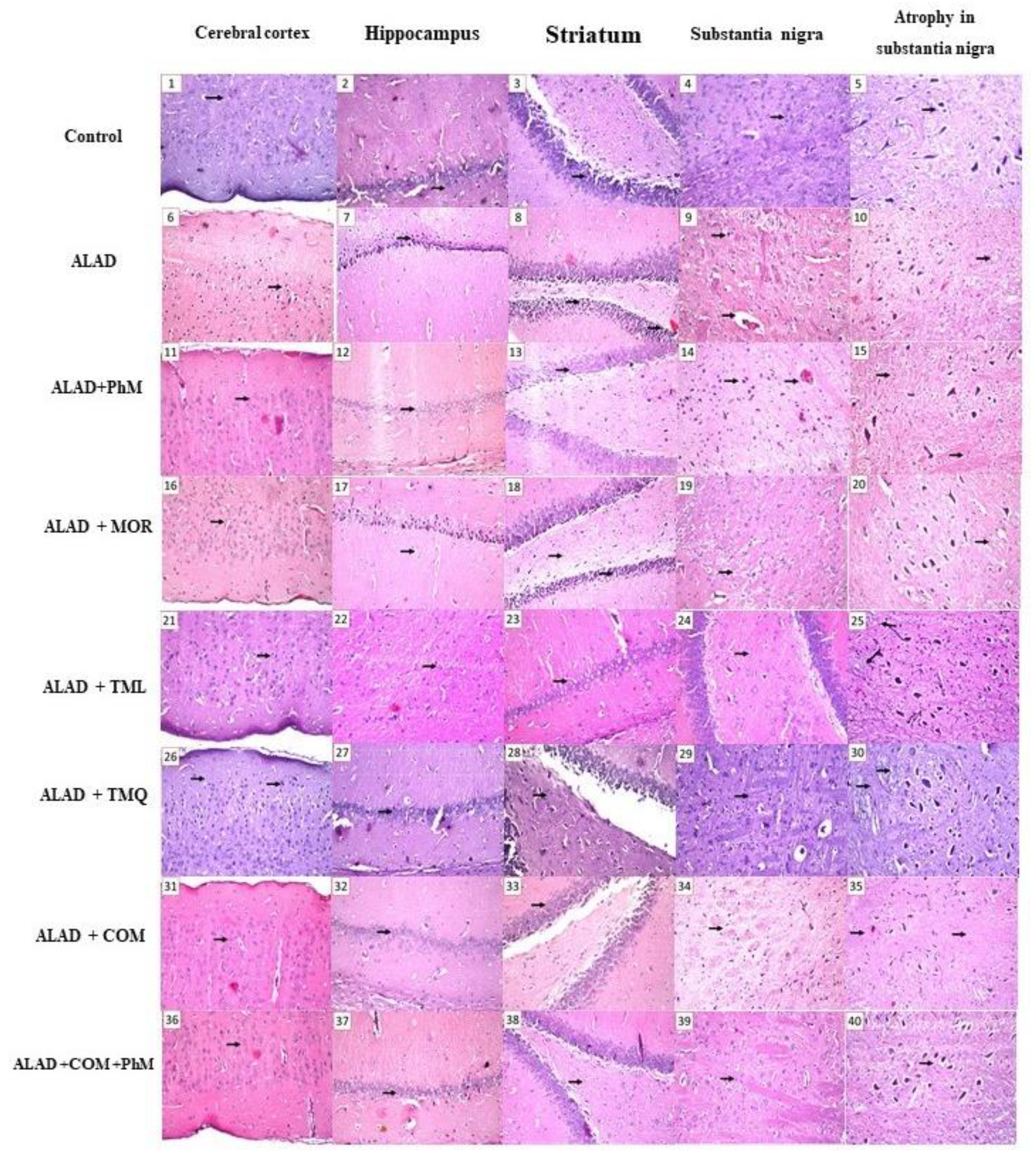
2.2. Histopathological Evaluation of Brain Tissues
2.3. Treatment of Rats with MOR, TML, and TMQ Restores Antioxidant Activities and Neurotransmitters Levels in ALAD
2.4. Treatment of Rats with MOR, TML, and TMQ Stimulates the Gene and Protein Expressions of Oxidative Stress Markers: Nrf2 and HO-1 in the Brain Tissues of ALAD
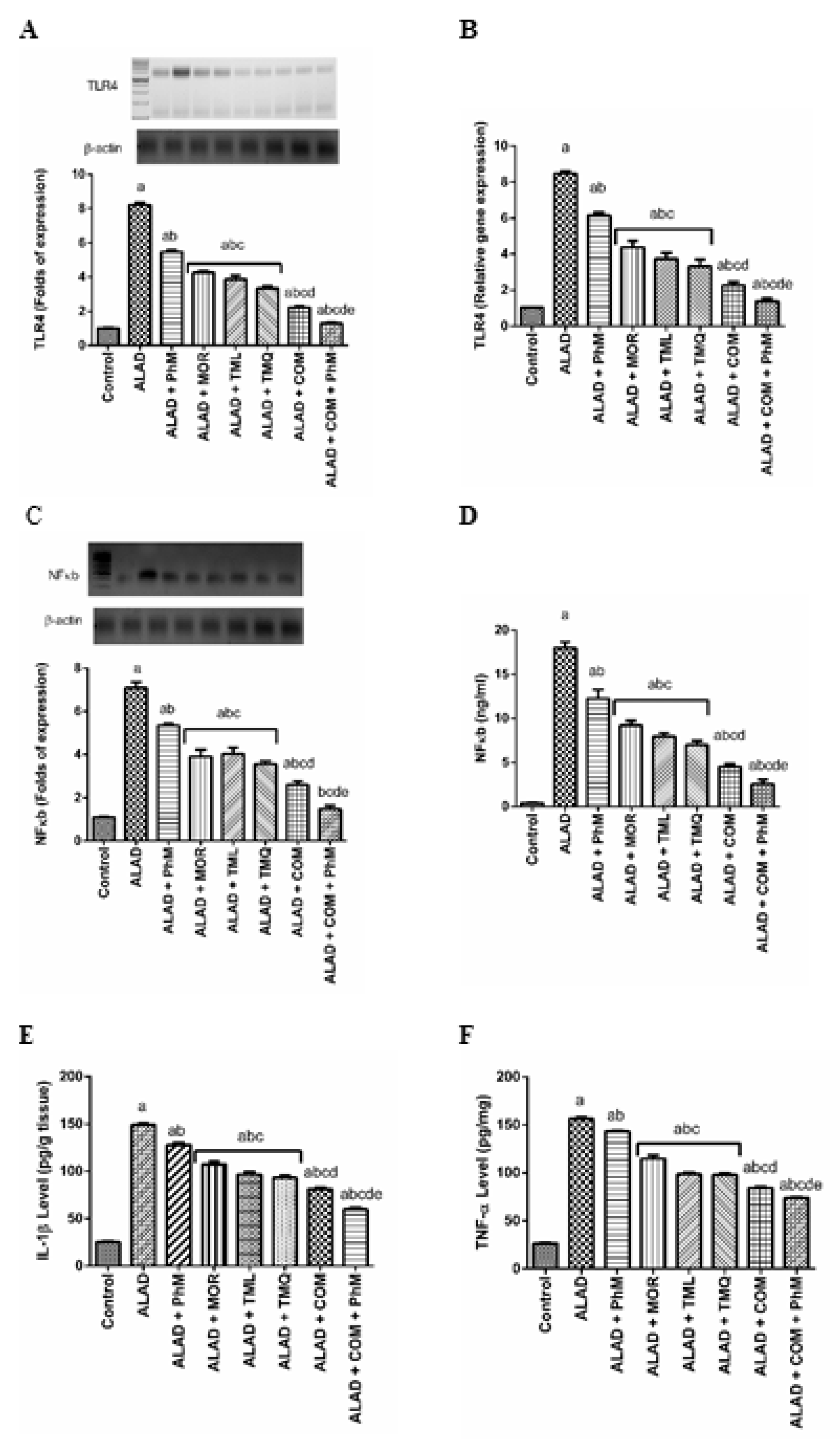
2.5. Phytochemicals with PhM Reduce Neuroinflammatory Biomarkers: TLR4, NF-kB, IL-1β and TNF-α in the Brain Tissues of ALAD
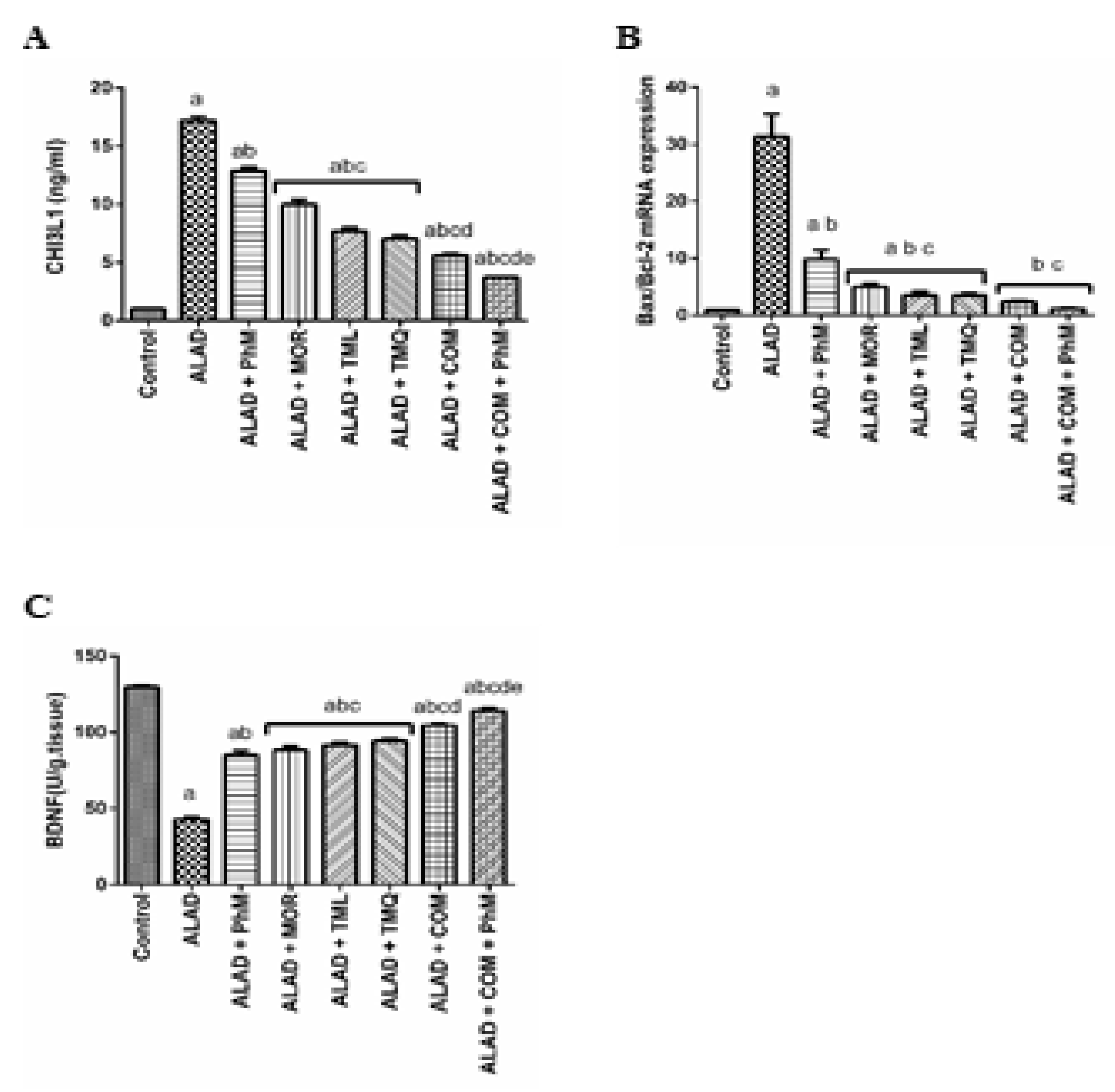
2.6. A Combination of MOR, TML, and TMQ with PhM Reduces Tissue Injury Biomarker; CHI3L1, and Apoptosis Biomarker; Bax/Bcl-2, and Enhances the Cognitive Biomarker; BDNF in Rat Brain Tissues of ALAD
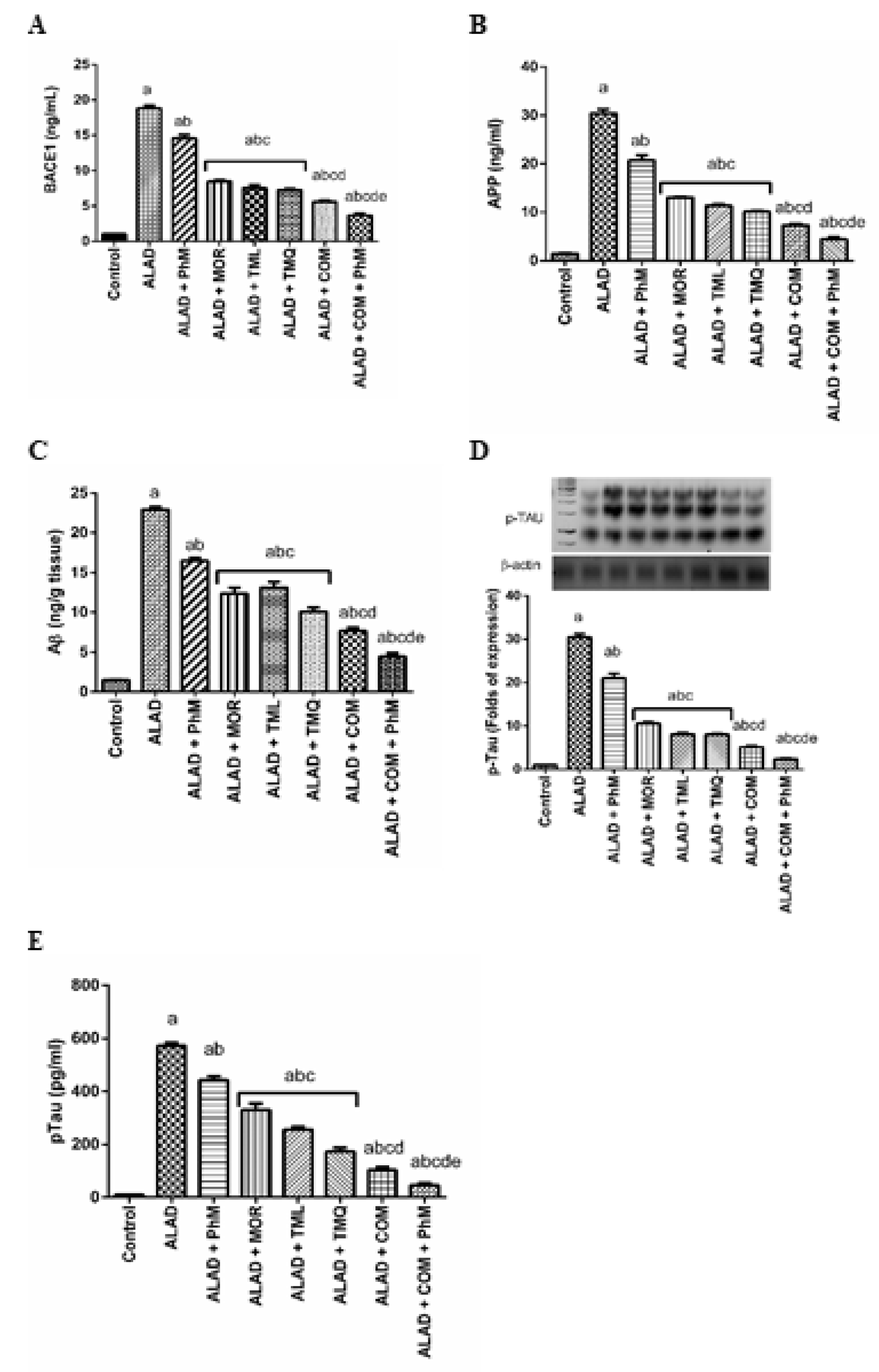
2.7. Treatment of ALAD with Phytochemicals Lowers Potential AD Biomarkers (Aβ Generation, BACE1, APP and p-Tau Levels) in Rat Brain Tissues of ALAD
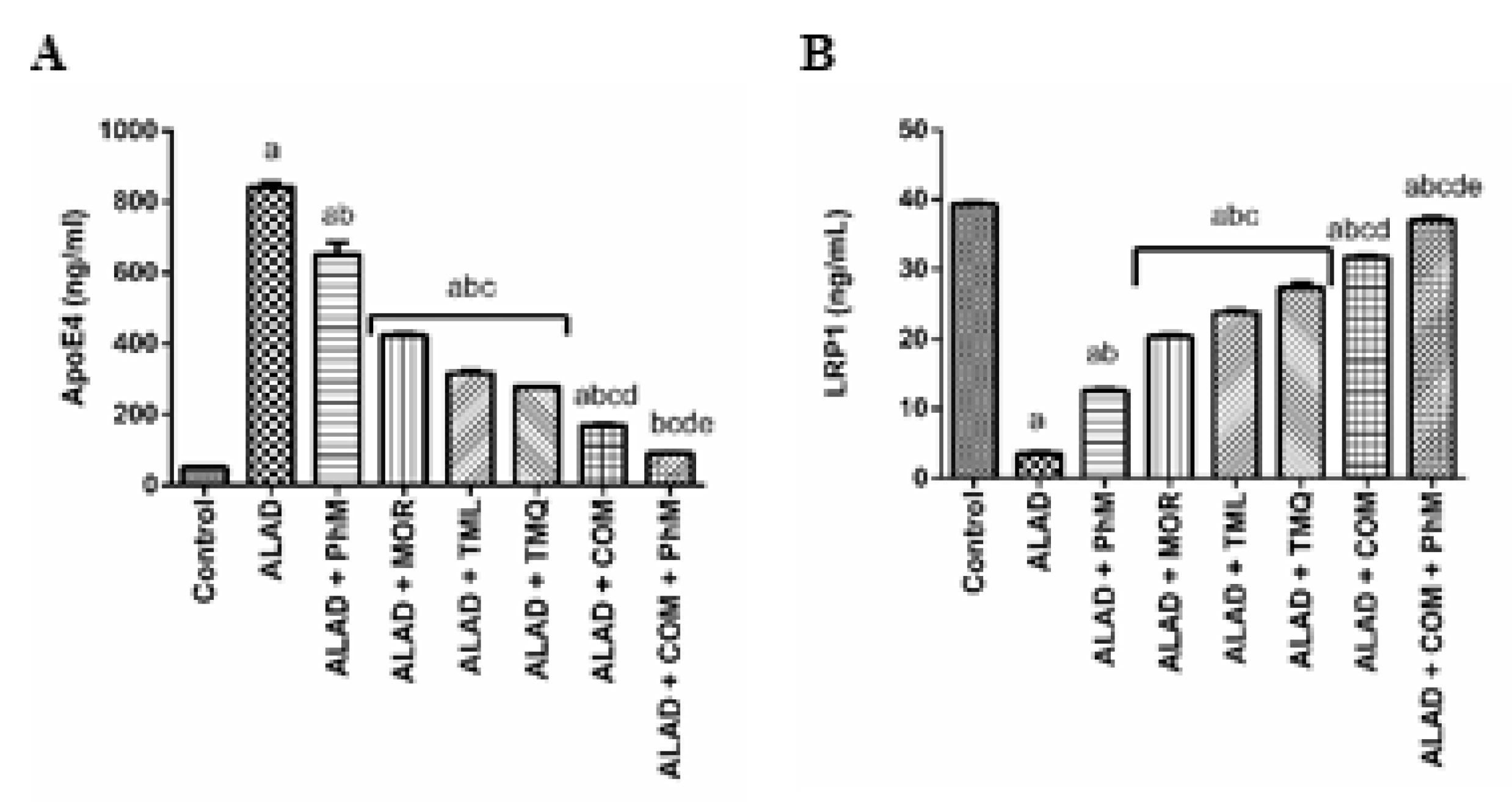
2.8. A Combination of MOR, TML, and TMQ with PhM Modulates AD Pathophysiology Biomarker; ApoE4 and its Neuronal Receptor LRP1 in Brain Tissues of ALAD
2.9. A Combination of MOR, TML, and TMQ with PhM Regulates Wnt3/β-Catenin/GSK3β Signaling Pathway in Brain Tissues of ALAD
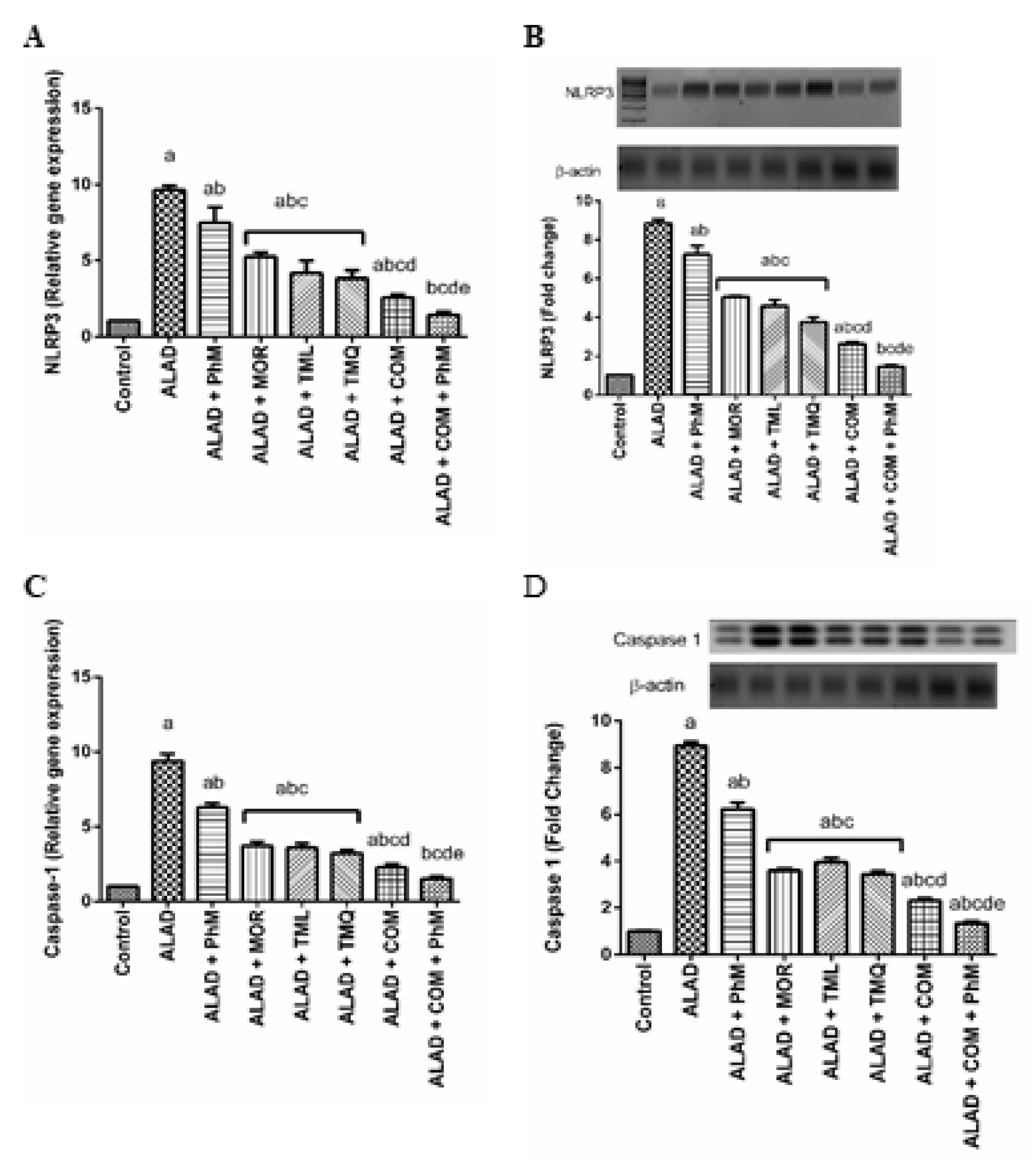
2.10. A Combination of Phytochemicals with PhM Suppreses Inflammasome Activation Biomarkers; NLRP3 and Caspase-1 in Brain Tissues of ALAD
2.11. Molecular Docking of Phytochemicals Used for Treatment of ALAD
3. Discussion
4. Materials and Methods
4.1. Drugs and Chemicals
4.2. Animals and Induction of AD-Like Rat Model
4.3. Experimental Design
4.4. Physical and Mental Activity Study
4.5. Behavioral Tests
4.5.1. The Conditioned Avoidance Response (CAR) Test
4.5.2. Y-Maze Spontaneous Alternation (SAP) Test
4.5.3. Morris Water Maze (MWM) Testing
4.6. Assembling and Preparation of Tissue Samples
4.7. Histopathological Evaluation
4.8. Preparation of Tissue
4.9. Biochemical Analyses
4.9.1. Colorimetric Analysis
4.9.2. Fluorometric Assays
4.9.3. Enzyme-Linked Immunosorbent Assay (ELISA)
4.9.4. Western Blotting Assay
4.9.5. Real-Time Quantitative Polymerase Chain Reaction (RT-qPCR)
4.10. Molecular Docking Study
4.11. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ALAD | Aluminum chloride-induced Alzheimer’s disease |
| AD | Alzheimer’s disease |
| Aβ | Amyloid beta |
| APP | Amyloid precursor protein |
| ApoE4 | Apolipoprotein E variant 4 |
| BACE1 | Beta-site amyloid precursor protein cleaving enzyme 1 |
| BDNF | Brain-derived neurotrophic factor |
| CAR | Conditioned avoidance response |
| DAMPs | Damage-associated molecular patterns |
| i.p | Intraperitoneal |
| Keap-1 | Kelch-like ECH-associated protein 1 |
| LRP1 | Low-density lipoprotein receptor-related protein-1 |
| MOR | Morin |
| NFκB | Nuclear factor kapa B |
| NFTs | Neurofibrillary tangles |
| MWM | Morris water maze testing |
| Nrf2/HO-1 | Nuclear factor erythroid 2-related factor 2/hemoxygenase-1 |
| PhM | Physical and mental activities |
| SAP | Spontaneous alternation percentage |
| THL | Thymol |
| TMQ | Thymoquinone |
| TLR-4 | Toll-like receptor-4 |
References
- Ju, Y.; Tam, K. Pathological mechanisms and therapeutic strategies for Alzheimer’s disease. Neural Regen. Res. 2022, 17, 543–549. [Google Scholar] [PubMed]
- Heilman, K.M.; Nadeau, S.E. Emotional and Neuropsychiatric Disorders Associated with Alzheimer’s Disease. Neurotherapeutics 2022, 19, 99–116. [Google Scholar] [CrossRef] [PubMed]
- Venugopalan, J.; Tong, L.; Hassanzadeh, H.R.; Wang, M.D. Multimodal deep learning models for early detection of Alzheimer’s disease stage. Sci. Rep. 2021, 11, 3254. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, S.; Ahmad, R.; Khare, S.K. Alzheimer’s disease and its treatment by different approaches: A review. Eur. J. Med. Chem. 2021, 216, 113320. [Google Scholar] [CrossRef] [PubMed]
- Graff-Radford, J.; Yong, K.X.X.; Apostolova, L.G.; Bouwman, F.H.; Carrillo, M.; Dickerson, B.C.; Rabinovici, G.D.; Schott, J.M.; Jones, D.T.; Murray, M.E. New insights into atypical Alzheimer’s disease in the era of biomarkers. Lancet. Neurol. 2021, 20, 222–234. [Google Scholar] [CrossRef]
- Abbas, F.; Eladl, M.A.; El-Sherbiny, M.; Abozied, N.; Nabil, A.; Mahmoud, S.M.; Mokhtar, H.I.; Zaitone, S.A.; Ibrahim, D. Celastrol and thymoquinone alleviate aluminum chloride-induced neurotoxicity: Behavioral psychomotor performance, neurotransmitter level, oxidative-inflammatory markers, and BDNF expression in rat brain. Biomed. Pharmacother. 2022, 151, 113072. [Google Scholar] [CrossRef]
- Mehrbeheshti, N.; Esmaili, Z.; Ahmadi, M.; Moosavi, M. A dose response effect of oral aluminum nanoparticle on novel object recognition memory, hippocampal caspase-3 and MAPKs signaling in mice. Behav. Brain Res. 2022, 417, 113615. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.X.; Xiang, J.Y.; Han, J.X.; Yang-Feng; Li, H.Z.; Chen, H.; Xu, M. Essential Oils from Spices Inhibit Cholinesterase Activity and Improve Behavioral Disorder in AlCl 3 Induced Dementia. Chem. Biodivers. 2022, 1, e202100443. [Google Scholar] [CrossRef] [PubMed]
- Aalikhani, M.; Safdari, Y.; Jahanshahi, M.; Alikhani, M.; Khalili, M. Comparison Between Hesperidin, Coumarin, and Deferoxamine Iron Chelation and Antioxidant Activity Against Excessive Iron in the Iron Overloaded Mice. Front. Neurosci. 2022, 15, 811080. [Google Scholar] [CrossRef]
- Elbini-Dhouib, I.; Doghri, R.; Ellefi, A.; Degrach, I.; Srairi-Abid, N.; Gati, A. Curcumin Attenuated Neurotoxicity in Sporadic Animal Model of Alzheimer’s Disease. Molecules 2021, 26, 3011. [Google Scholar] [CrossRef] [PubMed]
- Mesole, S.B.; Alfred, O.O.; Yusuf, U.A.; Lukubi, L.; Ndhlovu, D. Apoptotic Inducement of Neuronal Cells by Aluminium Chloride and the Neuroprotective Effect of Eugenol in Wistar Rats. Oxid. Med. Cell. Longev. 2020, 8425643. [Google Scholar] [CrossRef]
- Skalny, A.V.; Aschner, M.; Jiang, Y.; Gluhcheva, Y.G.; Tizabi, Y.; Lobinski, R.; Tinkov, A.A. Molecular mechanisms of aluminum neurotoxicity: Update on adverse effects and therapeutic strategies. Adv. Neurotoxicology 2021, 5, 1–34. [Google Scholar]
- Hampel, H.; Vassar, R.; De Strooper, B.; Hardy, J.; Willem, M.; Singh, N.; Zhou, J.; Yan, R.; Vanmechelen, E.; De Vos, A.; et al. The β-Secretase BACE1 in Alzheimer’s Disease. Biol. Psychiatry 2021, 89, 745–756. [Google Scholar] [CrossRef]
- Morroni, F.; Sita, G.; Graziosi, A.; Turrini, E.; Fimognari, C.; Tarozzi, A.; Hrelia, P. Neuroprotective effect of caffeic acid phenethyl ester in a mouse model of alzheimer’s disease involves Nrf2/HO-1 pathway. Aging Dis. 2018, 9, 605–622. [Google Scholar] [CrossRef]
- Zhou, Y.; Chen, Y.; Xu, C.; Zhang, H.; Lin, C. TLR4 Targeting as a Promising Therapeutic Strategy for Alzheimer Disease Treatment. Front. Neurosci. 2020, 14, 602508. [Google Scholar] [CrossRef]
- Kabel, A.M.; Arab, H.H.; Atef, A.; Estfanous, R.S. Omarigliptin/galangin combination mitigates lipopolysaccharide-induced neuroinflammation in rats: Involvement of glucagon-like peptide-1, toll-like receptor-4, apoptosis and Akt/GSK-3β signaling. Life Sci. 2022, 295, 120396. [Google Scholar] [CrossRef]
- Liu, Y.; Dai, Y.; Li, Q.; Chen, C.; Chen, H.; Song, Y.; Hua, F.; Zhang, Z. Beta-amyloid activates NLRP3 inflammasome via TLR4 in mouse microglia. Neurosci. Lett. 2020, 736, 135279. [Google Scholar] [CrossRef]
- Abu-Elfotuh, K.; Al-Najjar, A.H.; Mohammed, A.A.; Aboutaleb, A.S.; Badawi, G.A. Fluoxetine ameliorates Alzheimer’s disease progression and prevents the exacerbation of cardiovascular dysfunction of socially isolated depressed rats through activation of Nrf2/HO-1 and hindering TLR4/NLRP3 inflammasome signaling pathway. Int. Immunopharmacol. 2022, 104, 108488. [Google Scholar] [CrossRef]
- He, W.; Long, T.; Pan, Q.; Zhang, S.; Zhang, Y.; Zhang, D.; Qin, G.; Chen, L.; Zhou, J. Microglial NLRP3 inflammasome activation mediates IL-1β release and contributes to central sensitization in a recurrent nitroglycerin-induced migraine model. J. Neuroinflammation 2019, 16, 78. [Google Scholar] [CrossRef]
- Ju, T.; Sun, L.; Fan, Y.; Wang, T.; Liu, Y.; Liu, D.; Liu, T.; Zhao, C.; Wang, W.; Chi, L. Decreased Netrin-1 in Mild Cognitive Impairment and Alzheimer’s Disease Patients. Front. Aging Neurosci. 2022, 13, 762649. [Google Scholar] [CrossRef]
- Fan, M.; Li, Z.; Hu, M.; Zhao, H.; Wang, T.; Jia, Y.; Yang, R.; Wang, S.; Song, J.; Liu, Y.; et al. Formononetin attenuates Aβ 25-35-induced adhesion molecules in HBMECs via Nrf2 activation. Brain Res. Bull. 2022, 183, 162–171. [Google Scholar] [CrossRef]
- Van Gool, B.; Storck, S.E.; Reekmans, S.M.; Lechat, B.; Gordts, P.L.S.M.; Pradier, L.; Pietrzik, C.U.; Roebroek, A.J.M. LRP1 Has a Predominant Role in Production over Clearance of Aβ in a Mouse Model of Alzheimer’s Disease. Mol. Neurobiol. 2019, 56, 7234–7245. [Google Scholar] [CrossRef]
- Tachibana, M.; Holm, M.L.; Liu, C.C.; Shinohara, M.; Aikawa, T.; Oue, H.; Yamazaki, Y.; Martens, Y.A.; Murray, M.E.; Sullivan, P.M.; et al. APOE4-mediated amyloid-β pathology depends on its neuronal receptor LRP1. J. Clin. Invest. 2019, 129, 1272–1277. [Google Scholar] [CrossRef]
- Jha, N.K.; Chen, W.C.; Kumar, S.; Dubey, R.; Tsai, L.W.; Kar, R.; Jha, S.K.; Gupta, P.K.; Sharma, A.; Gundamaraju, R.; et al. Molecular mechanisms of developmental pathways in neurological disorders: A pharmacological and therapeutic review. Open Biol. 2022, 12, 210289. [Google Scholar] [CrossRef]
- Knotek, T.; Janeckova, L.; Kriska, J.; Korinek, V.; Anderova, M. Glia and Neural Stem and Progenitor Cells of the Healthy and Ischemic Brain: The Workplace for the Wnt Signaling Pathway. Genes 2020, 11, 804. [Google Scholar] [CrossRef]
- Yi, S.; Chen, S.; Xiang, J.; Tan, J.; Huang, K.; Zhang, H.; Wang, Y.; Wu, H. Genistein exerts a cell-protective effect via nrf2/ho-1/ /pi3k signaling in ab25-35-induced alzheimer’s disease models in vitro. Folia Histochem. Cytobiol. 2021, 59, 49–56. [Google Scholar] [CrossRef]
- Tayab, M.A.; Islam, M.N.; Chowdhury, K.A.A.; Tasnim, F.M. Targeting neuroinflammation by polyphenols: A promising therapeutic approach against inflammation-associated depression. Biomed. Pharmacother. 2022, 147, 112668. [Google Scholar] [CrossRef]
- Naoi, M.; Maruyama, W.; Shamoto-Nagai, M. Disease-modifying treatment of Parkinson’s disease by phytochemicals: Targeting multiple pathogenic factors. J. Neural Transm. 2021, 129, 737–753. [Google Scholar] [CrossRef]
- Amato, A.; Terzo, S.; Mulè, F. Natural Compounds as Beneficial Antioxidant Agents in Neurodegenerative Disorders: A Focus on Alzheimer’s Disease. Antioxidants 2019, 8, 608. [Google Scholar] [CrossRef]
- Davies, D.A.; Adlimoghaddam, A.; Albensi, B.C. Role of Nrf2 in Synaptic Plasticity and Memory in Alzheimer’s Disease. Cells 2021, 10, 1884. [Google Scholar] [CrossRef]
- Li, H.; Qin, T.; Li, M.; Ma, S. Thymol improves high-fat diet-induced cognitive deficits in mice via ameliorating brain insulin resistance and upregulating NRF2/HO-1 pathway. Metab. Brain Dis. 2017, 32, 385–393. [Google Scholar]
- Javed, H.; Azimullah, S.; Meeran, M.N.; Ansari, S.A.; Ojha, S. Neuroprotective effects of thymol, a dietary monoterpene against dopaminergic neurodegeneration in rotenone-induced rat model of parkinson’s disease. Int. J. Mol. Sci. 2019, 20, 1538. [Google Scholar] [CrossRef]
- Asadbegi, M.; Yaghmaei, P.; Salehi, I.; Komaki, A.; Ebrahim-Habibi, A. Investigation of thymol effect on learning and memory impairment induced by intrahippocampal injection of amyloid beta peptide in high fat diet- fed rats. Metab. Brain Dis. 2017, 32, 827–839. [Google Scholar] [CrossRef]
- Poorgholam, P.; Yaghmaei, P.; Hajebrahimi, Z. Thymoquinone recovers learning function in a rat model of Alzheimer’s disease. Avicenna J. Phytomed. 2018, 8, 188–197. [Google Scholar]
- Caselli, A.; Cirri, P.; Santi, A.; Paoli, P. Morin: A Promising Natural Drug. Curr. Med. Chem. 2016, 23, 774–791. [Google Scholar] [CrossRef]
- Issac, P.K.; Guru, A.; Velayutham, M.; Pachaiappan, R.; Arasu, M.V.; Al-Dhabi, N.A.; Choi, K.C.; Harikrishnan, R.; Arockiaraj, J. Oxidative stress induced antioxidant and neurotoxicity demonstrated in vivo zebrafish embryo or larval model and their normalization due to morin showing therapeutic implications. Life Sci. 2021, 283, 119864. [Google Scholar] [CrossRef]
- Kuzu, M.; Kandemir, F.M.; Yildirim, S.; Kucukler, S.; Caglayan, C.; Turk, E. Morin attenuates doxorubicin-induced heart and brain damage by reducing oxidative stress, inflammation and apoptosis. Biomed. Pharmacother. 2018, 106, 443–453. [Google Scholar] [CrossRef]
- Mohamed, E.A.; Ahmed, H.I.; Zaky, H.S.; Badr, A.M. Sesame oil mitigates memory impairment, oxidative stress, and neurodegeneration in a rat model of Alzheimer’s disease. A pivotal role of NF-κB/p38MAPK/BDNF/PPAR-γ pathways. J. Ethnopharmacol. 2021, 267, 113468. [Google Scholar] [CrossRef]
- Hindam, M.O.; Sayed, R.H.; Skalicka-Woźniak, K.; Budzyńska, B.; EL Sayed, N.S. Xanthotoxin and umbelliferone attenuate cognitive dysfunction in a streptozotocin-induced rat model of sporadic Alzheimer’s disease: The role of JAK2/STAT3 and Nrf2/HO-1 signalling pathway modulation. Phyther. Res. 2020, 34, 2351–2365. [Google Scholar] [CrossRef]
- Saba, K.; Rajnala, N.; Veeraiah, P.; Tiwari, V.; Rana, R.K.; Lakhotia, S.C.; Patel, A.B. Energetics of Excitatory and Inhibitory Neurotransmission in Aluminum Chloride Model of Alzheimer’s Disease: Reversal of Behavioral and Metabolic Deficits by Rasa Sindoor. Front. Mol. Neurosci. 2017, 10, 323. [Google Scholar] [CrossRef]
- Vaiserman, A.; Koliada, A.; Lushchak, O. Neuroinflammation in pathogenesis of Alzheimer’s disease: Phytochemicals as potential therapeutics. Mech. Ageing Dev. 2020, 189, 111259. [Google Scholar] [CrossRef]
- Jukic, M.; Politeo, O.; Maksimovic, M.; Milos, M.; Milos, M. In vitro acetylcholinesterase inhibitory properties of thymol, carvacrol and their derivatives thymoquinone and thymohydroquinone. Phytother. Res. 2007, 21, 259–261. [Google Scholar] [CrossRef]
- Bianchini, A.E.; Garlet, Q.I.; Da Cunha, J.A.; Bandeira Junior, G.; Brusque, I.C.M.; Salbego, J.; Heinzmann, B.M.; Baldisserotto, B. Monoterpenoids (Thymol, carvacrol and S-(+)-linalool) with anesthetic activity in silver catfish (Rhamdia quelen): Evaluation of acetylcholinesterase and GABaergic activity. Braz. J. Med. Biol. Res. 2017, 50, e6346. [Google Scholar] [CrossRef]
- Cascella, M.; Bimonte, S.; Barbieri, A.; Del Vecchio, V.; Muzio, M.R.; Vitale, A.; Benincasa, G.; Ferriello, A.B.; Azzariti, A.; Arra, C.; et al. Dissecting the potential roles of Nigella sativa and its constituent thymoquinone on the prevention and on the progression of Alzheimer’s disease. Front. Aging Neurosci. 2018, 10, 16. [Google Scholar] [CrossRef]
- Hosseinzadeh, H.; Parvardeh, S.; Asl, M.N.; Sadeghnia, H.R.; Ziaee, T. Effect of thymoquinone and Nigella sativa seeds oil on lipid peroxidation level during global cerebral ischemia-reperfusion injury in rat hippocampus. Phytomedicine 2007, 14, 621–627. [Google Scholar] [CrossRef]
- Cobourne-Duval, M.K.; Taka, E.; Mendonca, P.; Soliman, K.F.A. Thymoquinone increases the expression of neuroprotective proteins while decreasing the expression of pro-inflammatory cytokines and the gene expression NFκB pathway signaling targets in LPS/IFNγ -activated BV-2 microglia cells. J. Neuroimmunol. 2018, 320, 87–97. [Google Scholar] [CrossRef]
- Farkhondeh, T.; Samarghandian, S.; Shahri, A.M.P.; Samini, F. The Neuroprotective Effects of Thymoquinone: A Review. Dose. Response. 2018, 16. [Google Scholar] [CrossRef] [PubMed]
- Sharma, D.; Singh, M.; Kumar, P.; Vikram, V.; Mishra, N. Development and characterization of morin hydrate loaded microemulsion for the management of Alzheimer’s disease. Artif. Cells Nanomed. Biotechnol. 2017, 45, 1620–1630. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Thakur, V.; Deshmukh, R.; Sharma, A.; Rathore, M.S.; Kumar, A.; Mishra, N. Development and characterization of morin hydrate-loaded micellar nanocarriers for the effective management of Alzheimer’s disease. J. Microencapsul. 2018, 35, 137–148. [Google Scholar] [CrossRef]
- Zheng, C.; Zhou, X.W.; Wang, J.Z. The dual roles of cytokines in Alzheimer’s disease: Update on interleukins, TNF-α, TGF-β and IFN-γ. Transl. Neurodegener. 2016, 5, 7. [Google Scholar] [CrossRef]
- Ahmad, M.H.; Fatima, M.; Mondal, A.C. Influence of microglia and astrocyte activation in the neuroinflammatory pathogenesis of Alzheimer’s disease: Rational insights for the therapeutic approaches. J. Clin. Neurosci. 2019, 59, 6–11. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Xin, Y.; Chu, T.; Liu, C.; Xu, A. Dexmedetomidine attenuates perioperative neurocognitive disorders by suppressing hippocampal neuroinflammation and HMGB1/RAGE/NF-κB signaling pathway. Biomed. Pharmacother. 2022, 150, 113006. [Google Scholar] [CrossRef] [PubMed]
- Didonna, A. Tau at the interface between neurodegeneration and neuroinflammation. Genes Immun. 2020, 21, 288–300. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Wise, L.; Fukuchi, K.I. TLR4 Cross-Talk With NLRP3 Inflammasome and Complement Signaling Pathways in Alzheimer’s Disease. Front. Immunol. 2020, 11, 724. [Google Scholar] [CrossRef] [PubMed]
- Wree, A.; Eguchi, A.; Mcgeough, M.D.; Pena, C.A.; Johnson, C.D.; Canbay, A.; Hoffman, H.M.; Feldstein, A.E. NLRP3 inflammasome activation results in hepatocyte pyroptosis, liver inflammation, and fibrosis in mice. Hepatology 2014, 59, 898–910. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Wang, H.; Kouadir, M.; Song, H.; Shi, F. Recent advances in the mechanisms of NLRP3 inflammasome activation and its inhibitors. Cell Death Dis. 2019, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Stancu, I.C.; Cremers, N.; Vanrusselt, H.; Couturier, J.; Vanoosthuyse, A.; Kessels, S.; Lodder, C.; Brône, B.; Huaux, F.; Octave, J.N.; et al. Aggregated Tau activates NLRP3-ASC inflammasome exacerbating exogenously seeded and non-exogenously seeded Tau pathology in vivo. Acta Neuropathol. 2019, 137, 599–617. [Google Scholar] [CrossRef] [PubMed]
- Stancu, I.C.; Lodder, C.; Botella Lucena, P.; Vanherle, S.; Gutiérrez de Ravé, M.; Terwel, D.; Bottelbergs, A.; Dewachter, I. The NLRP3 inflammasome modulates tau pathology and neurodegeneration in a tauopathy model. Glia 2022, 70, 1117–1132. [Google Scholar] [CrossRef]
- Tao, L.; Zhang, L.; Gao, R.; Jiang, F.; Cao, J.; Liu, H. Andrographolide Alleviates Acute Brain Injury in a Rat Model of Traumatic Brain Injury: Possible Involvement of Inflammatory Signaling. Front. Neurosci. 2018, 12, 657. [Google Scholar] [CrossRef]
- Qin, Z.Y.; Gu, X.; Chen, Y.L.; Liu, J.B.; Hou, C.X.; Lin, S.Y.; Hao, N.N.; Liang, Y.; Chen, W.; Meng, H.Y. Toll-like receptor 4 activates the NLRP3 inflammasome pathway and periodontal inflammaging by inhibiting Bmi-1 expression. Int. J. Mol. Med. 2021, 47, 137–150. [Google Scholar] [CrossRef]
- Abu-Elfotuh, K.; Hussein, F.H.; Abbas, A.N.; Al-Rekabi, M.D.; Barghash, S.S.; Zaghlool, S.S.; El-Emam, S.Z. Melatonin and zinc supplements with physical and mental activities subside neurodegeneration and hepatorenal injury induced by aluminum chloride in rats: Inclusion of GSK-3β-Wnt/β-catenin signaling pathway. Neurotoxicology 2022, 91, 69–83. [Google Scholar] [CrossRef] [PubMed]
- Cui, W.; Sun, C.; Ma, Y.; Wang, S.; Wang, X.; Zhang, Y. Inhibition of TLR4 Induces M2 Microglial Polarization and Provides Neuroprotection via the NLRP3 Inflammasome in Alzheimer’s Disease. Front. Neurosci. 2020, 14, 444. [Google Scholar] [CrossRef] [PubMed]
- Hung, W.L.; Ho, C.T.; Pan, M.H. Targeting the NLRP3 Inflammasome in Neuroinflammation: Health Promoting Effects of Dietary Phytochemicals in Neurological Disorders. Mol. Nutr. Food Res. 2020, 64, 1900550. [Google Scholar] [CrossRef] [PubMed]
- Jin, X.; Liu, M.Y.; Zhang, D.F.; Zhong, X.; Du, K.; Qian, P.; Yao, W.F.; Gao, H.; Wei, M.J. Baicalin mitigates cognitive impairment and protects neurons from microglia-mediated neuroinflammation via suppressing NLRP3 inflammasomes and TLR4/NF-κB signaling pathway. CNS Neurosci. Ther. 2019, 25, 575. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.Y.; Zhuang, Z.; Lu, Y.; Tao, T.; Zhou, Y.; Liu, G.J.; Wang, H.; Zhang, D.D.; Wu, L.Y.; Dai, H.B.; et al. Curcumin Mitigates Neuro-Inflammation by Modulating Microglia Polarization Through Inhibiting TLR4 Axis Signaling Pathway Following Experimental Subarachnoid Hemorrhage. Front. Neurosci. 2019, 13, 1223. [Google Scholar] [CrossRef] [PubMed]
- Shinohara, M.; Tachibana, M.; Kanekiyo, T.; Bu, G. Role of LRP1 in the pathogenesis of Alzheimer’s disease: Evidence from clinical and preclinical studies: Thematic Review Series: ApoE and Lipid Homeostasis in Alzheimer’s Disease. J. Lipid Res. 2017, 58, 1267–1281. [Google Scholar] [CrossRef]
- Liu, C.C.; Hu, J.; Zhao, N.; Wang, J.; Wang, N.; Cirrito, J.R.; Kanekiyo, T.; Holtzman, D.M.; Bu, G. Astrocytic LRP1 Mediates Brain Aβ Clearance and Impacts Amyloid Deposition. J. Neurosci. 2017, 37, 4023–4031. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, J.; Wan, J.; Liu, A.; Sun, J. Melatonin regulates Aβ production/clearance balance and Aβ neurotoxicity: A potential therapeutic molecule for Alzheimer’s disease. Biomed. Pharmacother. 2020, 132, 110887. [Google Scholar] [CrossRef] [PubMed]
- Cooper, J.M.; Lathuiliere, A.; Migliorini, M.; Arai, A.L.; Wani, M.M.; Dujardin, S.; Muratoglu, S.C.; Hyman, B.T.; Strickland, D.K. Regulation of tau internalization, degradation, and seeding by LRP1 reveals multiple pathways for tau catabolism. J. Biol. Chem. 2021, 296. [Google Scholar] [CrossRef]
- Michaelson, D.M. APOE ε4: The most prevalent yet understudied risk factor for Alzheimer’s disease. Alzheimer’s Dement. 2014, 10, 861–868. [Google Scholar] [CrossRef]
- Safieh, M.; Korczyn, A.D.; Michaelson, D.M. ApoE4: An emerging therapeutic target for Alzheimer’s disease. BMC Med. 2019, 17, 64. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Tian, D.Y.; Wang, Y.J. Peripheral clearance of brain-derived Aβ in Alzheimer’s disease: Pathophysiology and therapeutic perspectives. Transl. Neurodegener. 2020, 9, 113–139. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Wang, S.; Niu, Q. Effect of Aluminum-Maltolate on the Content of Aβ Protein and the Expression of ApoER2, VLDLRs, and LRP1 in PC12-ApoE4 Cells. Neurotox. Res. 2019, 35, 931–944. [Google Scholar] [CrossRef]
- Ismail, N.; Ismail, M.; Azmi, N.H.; Bakar, M.F.A.; Yida, Z.; Abdullah, M.A.; Basri, H. Thymoquinone-rich fraction nanoemulsion (TQRFNE) decreases Aβ40 and Aβ42 levels by modulating APP processing, up-regulating IDE and LRP1, and down-regulating BACE1 and RAGE in response to high fat/cholesterol diet-induced rats. Biomed. Pharmacother. 2017, 95, 780–788. [Google Scholar] [CrossRef] [PubMed]
- Mountaki, C.; Dafnis, I.; Panagopoulou, E.A.; Vasilakopoulou, P.B.; Karvelas, M.; Chiou, A.; Karathanos, V.T.; Chroni, A. Mechanistic insight into the capacity of natural polar phenolic compounds to abolish Alzheimer’s disease-associated pathogenic effects of apoE4 forms. Free Radic. Biol. Med. 2021, 171, 284–301. [Google Scholar] [CrossRef]
- Liu, D.; Chen, L.; Zhao, H.; Vaziri, N.D.; Ma, S.-C.; Zhao, Y.-Y. Small molecules from natural products targeting the Wnt/β-catenin pathway as a therapeutic strategy. Biomed. Pharmacother. 2019, 117, 108990. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Begum, A.N.; Jones, M.R.; Oh, M.S.; Beech, W.K.; Beech, B.H.; Yang, F.; Chen, P.; Ubeda, O.J.; Kim, P.C.; et al. GSK3 inhibitors show benefits in an Alzheimer’s disease (AD) model of neurodegeneration but adverse effects in control animals. Neurobiol. Dis. 2009, 33, 193–206. [Google Scholar] [CrossRef] [PubMed]
- Jia, L.; Piña-Crespo, J.; Li, Y. Restoring Wnt/β-catenin signaling is a promising therapeutic strategy for Alzheimer’s disease. Mol. Brain 2019, 12, 104. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.A.; Abd El-Fattah, A.I.; Abu-Elfotuh, K.; Elariny, H.A. Natural antioxidants enhance the power of physical and mental activities versus risk factors inducing progression of Alzheimer’s disease in rats. Int. Immunopharmacol. 2021, 96, 107729. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, N.; Asle-Rousta, M.; Rahnema, M.; Amini, R. Morin attenuates memory deficits in a rat model of Alzheimer’s disease by ameliorating oxidative stress and neuroinflammation. Eur. J. Pharmacol. 2021, 910, 174506. [Google Scholar] [CrossRef] [PubMed]
- Zaher, M.A.F.; Bendary, M.A.; Aly, A.S. Effect of Thymoquinone against Aluminum Chloride-Induced Alzheimer-Like Model in Rats: A Neurophysiological and Behavioral Study. Med. J. Cairo Univ. 2020, 88, 355–365. [Google Scholar]
- Elariny, H.A.; Abu Elfotuh, K.; Ali, A.A.; Khalil, M.G. The Role of Mental and Physical Activities against Development of Alzheimer ’s Disease in Socialized and Isolated Rats. Brain Disord. Ther. 2017, 06, 1–17. [Google Scholar] [CrossRef]
- Wadenberg, M.-L.G.; Fjällström, A.-K.; Federley, M.; Persson, P.; Stenqvist, P. Effects of adjunct galantamine to risperidone, or haloperidol, in animal models of antipsychotic activity and extrapyramidal side-effect liability: Involvement of the cholinergic muscarinic receptor. Int. J. Neuropsychopharmacol. 2011, 14, 644–654. [Google Scholar] [CrossRef]
- Chou, S.; Davis, C.; Li, M. Maternal immune activation and repeated maternal separation alter offspring conditioned avoidance response learning and antipsychotic response in male rats. Behav. Brain Res. 2021, 403, 113145. [Google Scholar] [CrossRef]
- Hritcu, L.; Cioanca, O.; Hancianu, M. Effects of lavender oil inhalation on improving scopolamine-induced spatial memory impairment in laboratory rats. Phytomedicine 2012, 19, 529–534. [Google Scholar] [CrossRef]
- Cleal, M.; Fontana, B.D.; Parker, M.O. The cognitive and behavioral effects of D-amphetamine and nicotine sensitization in adult zebrafish. Psychopharmacology 2021, 238, 2191–2200. [Google Scholar] [CrossRef] [PubMed]
- Morris, R. Developments of a water-maze procedure for studying spatial learning in the rat. J. Neurosci. Methods 1984, 11, 47–60. [Google Scholar] [CrossRef]
- Ali, F.E.M.; Sayed, A.M.; El-Bahrawy, A.H.; Omar, Z.M.M.; Hassanein, E.H.M. Targeting KEAP1/Nrf2, AKT, and PPAR-γ signals as a potential protective mechanism of diosmin against gentamicin-induced nephrotoxicity. Life Sci. 2021, 275, 119349. [Google Scholar] [CrossRef]
- Greenblatt, H.M.; Kryger, G.; Lewis, T.; Silman, I.; Sussman, J.L. Structure of acetylcholinesterase complexed with (−)-galanthamine at 2.3 Å resolution. FEBS Lett. 1999, 463, 321–326. [Google Scholar] [CrossRef]
- Ahn, J.S.; Radhakrishnan, M.L.; Mapelli, M.; Choi, S.; Tidor, B.; Cuny, G.D.; Musacchio, A.; Yeh, L.-A.; Kosik, K.S. Defining Cdk5 Ligand Chemical Space with Small Molecule Inhibitors of Tau Phosphorylation. Chem. Biol. 2005, 12, 811–823. [Google Scholar] [CrossRef]
- Fujimoto, K.; Yoshida, S.; Tadano, G.; Asada, N.; Fuchino, K.; Suzuki, S.; Matsuoka, E.; Yamamoto, T.; Yamamoto, S.; Ando, S.; et al. Structure-Based Approaches to Improving Selectivity through Utilizing Explicit Water Molecules: Discovery of Selective β-Secretase (BACE1) Inhibitors over BACE2. J. Med. Chem. 2021, 64, 3075–3085. [Google Scholar] [CrossRef] [PubMed]
- Wagner, F.F.; Bishop, J.A.; Gale, J.P.; Shi, X.; Walk, M.; Ketterman, J.; Patnaik, D.; Barker, D.; Walpita, D.; Campbell, A.J.; et al. Inhibitors of Glycogen Synthase Kinase 3 with Exquisite Kinome-Wide Selectivity and Their Functional Effects. ACS Chem. Biol. 2016, 11, 1952–1963. [Google Scholar] [CrossRef] [PubMed]
- Pinto, A.; El Ali, Z.; Moniot, S.; Tamborini, L.; Steegborn, C.; Foresti, R.; de Micheli, C. Effects of 3-Bromo-4,5-dihydroisoxazole Derivatives on Nrf2 Activation and Heme Oxygenase-1 Expression. ChemistryOpen. 2018, 7, 858–864. [Google Scholar] [CrossRef] [PubMed]


| Group | Control | ALAD | ALAD + PhM | ALAD + MOR | ALAD + TML | ALAD + TMQ | ALAD + COM | ALAD + COM + PhM | |
|---|---|---|---|---|---|---|---|---|---|
| Nuclear pyknosis and degeneration | Cerebral cortex | - | +++ | - | - | ++ | + | - | - |
| The subiculum and fascia dentate of the hippocampus | - | +++ | - | ++ | - | - | - | - | |
| Focal eosinophilic plagues in the striatum | - | +++ | +++ | + | + | - | + | - | |
| Atrophy in the neuronal cells of the substantia nigra | - | +++ | +++ | + | - | + | + | - | |
| . | Control | ALAD | ALAD + PhM | ALAD + MOR | ALAD + TML | ALAD + TMQ | ALAD + COM | ALAD + COM + PhM |
|---|---|---|---|---|---|---|---|---|
| TAC(mmol/g) | 45.3 ± 1.2 | 9.7 ± 1.3 a | 14.4 ± 1.0 ab | 24.3 ± 1.4 abc | 25.5 ± 1.7 abc | 25.7 ± 1.3 abc | 30.7 ± 1.0 abcd | 33.7 ± 2.1 abcde |
| SOD (U/g) | 5.9 ± 0.4 | 0.8 ± 0.04 a | 2.5 ± 0.08 ab | 3.1 ± 0.23 abc | 3.3 ± 0.3 abc | 3.8 ± 0.08 abc | 4.4 ± 0.4 abcd | 5.8 ± 0.1 bcde |
| MDA (nmol/g) | 5.5 ± 0.6 | 104.4 ± 7.5 a | 85.4 ± 4.9 ab | 72.7 ± 2.3 abc | 67.0 ± 3.6 abc | 56.7 ± 2.7 abc | 41.4 ± 3.4 abcd | 25.3 ± 3.7 abcde |
| DA (ng/g) | 71.2 ± 1.9 | 13.6 ± 1.9 a | 23.7 ± 2.5 ab | 40.0 ± 0.2 abc | 45.3 ± 0.5 abc | 48.8 ± 0.8 abc | 53.5 ± 1.0 abcd | 58.1 ± 2.4 abcde |
| 5-HT (ng/g) | 11.8 ± 1.0 | 4.2 ± 0.5 a | 6.0 ± 0.4 ab | 7.3 ± 0.5 abc | 7.9 ± 0.6 abc | 8.7 ± 0.4 abc | 10.0 ± 0.4 abcd | 11.2 ± 0.9 bcde |
| NE (nmol/g) | 737.2 ± 4.6 | 176.6 ± 8.2 a | 258.0 ± 8.5 ab | 390.7 ± 14.8 abc | 445.5 ± 7.3 abc | 470.1 ± 12.3 abc | 533.5 ± 26.6 abcd | 594.4 ± 6.4 abcde |
| ACHE (ng/g) | 11.8 ± 1.1 | 66.1 ± 1.9 a | 49.3 ± 3.0 ab | 24.8 ± 0.4 abc | 22.4 ± 1.9 abc | 22.1 ± 1.8 abc | 17.1 ± 1.0 abcd | 13.8 ± 0.1 bcde |
| Gene | The Primer Pair Sequence | Gen ID | Number of Base Pairs |
|---|---|---|---|
| Bax | F: 5′-CACGTCTGCGGGGAGTCA-3’ R: 5’-TAGGAAAGGAGGCCATCCCA-3’ | NM_017059 | 566 bp |
| Bcl-2 | F: 5′-CATCTCATGCCAAGGGGGAA-3’ R: 5′-TATCCCACTCGTAGCCCCTC- 3’ | NM_016993 | 284 bp |
| TLR4 | F: 5′-TCAGCTTTGGTCAGTTGGCT-3’ R: 5′-GTCCTTGACCCACTGCAAGA-3’ | NM_019178 | 692 bp |
| HO-1 | F: 5′-CACCAGCCACACAGCACTAC-3′ R: 5′-CACCCACCCCTCAAAAGACA-3′ | NM_012580 | 1042 bp |
| Nrf2 | F: 5′-CTCTCTGGAGACGGCCATGACT-3′ R: 5′-CTGGGCTGGGGACAGTGGTAGT-3′ | NM_031789 | 145 bp |
| NLRP3 | F: 5′-TGCATGCCGTATCTGGTTGT-3′ R: 5′-ACCTCTTGCGAGGGTCTTTG-3′ | NM_001191642 | 391 bp |
| Caspase-1 | F: 5′-GAACAAAGAAGGTGGCGCAT-3′ R: 5′-GAGGTCAACATCAGCTCCGA-3′ | NM_012762 | 202 bp |
| β-actin | F: 5′-CCGTAAAGACCTCTATGCCA- 3’ R: 5′-AAGAAAGGGTGTAAAACGCA- 3’ | NM_031144 | 299 bp |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hamdan, A.M.E.; Alharthi, F.H.J.; Alanazi, A.H.; El-Emam, S.Z.; Zaghlool, S.S.; Metwally, K.; Albalawi, S.A.; Abdu, Y.S.; Mansour, R.E.-S.; Salem, H.A.; et al. Neuroprotective Effects of Phytochemicals against Aluminum Chloride-Induced Alzheimer’s Disease through ApoE4/LRP1, Wnt3/β-Catenin/GSK3β, and TLR4/NLRP3 Pathways with Physical and Mental Activities in a Rat Model. Pharmaceuticals 2022, 15, 1008. https://doi.org/10.3390/ph15081008
Hamdan AME, Alharthi FHJ, Alanazi AH, El-Emam SZ, Zaghlool SS, Metwally K, Albalawi SA, Abdu YS, Mansour RE-S, Salem HA, et al. Neuroprotective Effects of Phytochemicals against Aluminum Chloride-Induced Alzheimer’s Disease through ApoE4/LRP1, Wnt3/β-Catenin/GSK3β, and TLR4/NLRP3 Pathways with Physical and Mental Activities in a Rat Model. Pharmaceuticals. 2022; 15(8):1008. https://doi.org/10.3390/ph15081008
Chicago/Turabian StyleHamdan, Ahmed Mohsen Elsaid, Fatimah Hussain J. Alharthi, Ahmed Hadi Alanazi, Soad Z. El-Emam, Sameh S. Zaghlool, Kamel Metwally, Sana Abdulaziz Albalawi, Yahia S. Abdu, Reda El-Sayed Mansour, Hoda A. Salem, and et al. 2022. "Neuroprotective Effects of Phytochemicals against Aluminum Chloride-Induced Alzheimer’s Disease through ApoE4/LRP1, Wnt3/β-Catenin/GSK3β, and TLR4/NLRP3 Pathways with Physical and Mental Activities in a Rat Model" Pharmaceuticals 15, no. 8: 1008. https://doi.org/10.3390/ph15081008
APA StyleHamdan, A. M. E., Alharthi, F. H. J., Alanazi, A. H., El-Emam, S. Z., Zaghlool, S. S., Metwally, K., Albalawi, S. A., Abdu, Y. S., Mansour, R. E.-S., Salem, H. A., Abd Elmageed, Z. Y., & Abu-Elfotuh, K. (2022). Neuroprotective Effects of Phytochemicals against Aluminum Chloride-Induced Alzheimer’s Disease through ApoE4/LRP1, Wnt3/β-Catenin/GSK3β, and TLR4/NLRP3 Pathways with Physical and Mental Activities in a Rat Model. Pharmaceuticals, 15(8), 1008. https://doi.org/10.3390/ph15081008