How Should the Worldwide Knowledge of Traditional Cancer Healing Be Integrated with Herbs and Mushrooms into Modern Molecular Pharmacology?
Abstract
1. Introduction
2. Cancer Features Affected by Natural Drugs
3. Plants from Different Continents Used in Ethnomedicine for the Treatment of Malignancies
3.1. Africa
3.2. South America
3.3. Asia
3.3.1. Traditional China Herbal Medicine
3.3.2. Ayurvedic Medicine
3.4. Europe
3.5. North America
3.6. Australia
4. Mushrooms
5. Why Should Medical Plants and Mushrooms Be Used Today?
6. Limitations of Using Plants and Mushrooms as Medicine
6.1. Bioavailability
6.2. Safety
6.3. Standardization
7. Overcoming Limitations to Integrate Folk and Modern Medicine
8. Conclusions and Future Perspectives
- −
- To date, a lot of information about a number of plants and mushrooms, and their individual bioactive compounds with well-documented antitumor properties, has been accumulated. Their respective full-scale multi-level studies should be top priorities.
- −
- Despite there being a lot of investigations on the anticancer properties of a certain plant using tumor cell models, only a limited number of studies have been carried out with implication of control non-tumor cell models and subsequent animal studies. As the next step, comprehensive studies on their effectiveness, toxicity to non-cancer cells, and animal tissues in various doses are required to authorize natural-derived extracts and individual compounds into the next pre-clinical or clinical investigation.
- −
- Progress in standardization is highly required to transform anecdotal folk herbal medicine into modern molecular pharmacology with clear mechanisms of action. This process includes investments into big programs regarding investigations, monitoring, and certifications of manufacturing the final product.
- −
- On the examples of etoposide, irino- and topotecan, vinorelbine, docetaxel, and omacetaxine, the development of semi-synthetic derivates of newly identified natural compounds with significant anticancer properties may improve their characteristics and lead to new antineoplastic drugs.
- −
- The study of a synergistic interaction of isolated natural compounds and crude plant- and mushroom-derived extracts with widely used anticancer therapeutics should help define the right dosage and compatibility between the natural and synthetic therapeutics.
- −
- Natural compounds may sensitize tumors for modern therapeutics and be effective in adjuvant and neoadjuvant therapy.
- −
- There is a variety of standardized dietary supplements from plants and mushrooms with presumable antineoplastic properties produced by large world-class companies. The important approach is to test their antitumor potential using animal models, especially in combination with relevant modern therapeutics.
- −
- The pharmacological effects of active compounds are much higher in herbal extracts than in pure compounds due to co-existing constituents which may provide the pharmacokinetic synergy during intestinal absorption and the ‘’first-path’’ metabolism.
- −
- Folk medicine may point to certain plants or mushrooms with highly potent anti-cancer properties and bioactive compounds. Herewith, the cooperation between cancer researchers and ethnobotanists or ethnomedicine specialists can benefit the development of new therapeutics.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- WHO. CureAll Framework: WHO Global Initiative for Childhood Cancer: Increasing Access, Advancing Quality, Saving Lives; WHO: Geneva, Switzerland, 2021. [Google Scholar]
- Garodia, P.; Ichikawa, H.; Malani, N.; Sethi, G.; Aggarwal, B.B. From ancient medicine to modern medicine: Ayurvedic concepts of health and their role in inflammation and cancer. J. Soc. Integr. Oncol. 2007, 5, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Mann, M.; Pathak, S.R. Ayurveda: A new dimension in the era of modern medicine. In Synthesis of Medicinal Agents from Plants; Elsevier: Amsterdam, The Netherlands, 2018; pp. 283–303. [Google Scholar]
- Vaghora, B.; Shukla, V. Impact of different phytochemical classes and Ayurvedic plants in battle against cancer. Skin 2016, 13, 14. [Google Scholar]
- Wu, M.; Lu, P.; Shi, L.; Li, S. Traditional Chinese patent medicines for cancer treatment in China: A nationwide medical insurance data analysis. Oncotarget 2015, 6, 38283. [Google Scholar] [CrossRef]
- Zhang, N.; Shi, N.; Li, S.; Liu, G.; Han, Y.; Liu, L.; Zhang, X.; Kong, X.; Zhang, B.; Yuan, W. A Retrospective Study on the Use of Chinese Patent Medicine in 24 Medical Institutions for COVID-19 in China. Front. Pharmacol. 2020, 11, 574562. [Google Scholar] [CrossRef] [PubMed]
- Ovais, M.; Khalil, A.T.; Jan, S.A.; Ayaz, M.; Ullah, I.; Shinwari, W.; Shinwari, Z.K. Traditional Chinese Medicine Going Global: Opportunities for Belt and Road Countries: TCM Importance in the Context of Belt Road Initiative. Proc. Pak. Acad. Sci. B Life Environ. Sci. 2019, 56, 17–26. [Google Scholar]
- Cyranoski, D. Why Chinese medicine is heading for clinics around the world. Nature 2018, 561, 448. [Google Scholar] [CrossRef]
- Katayama, K.; Yoshino, T.; Munakata, K.; Yamaguchi, R.; Imoto, S.; Miyano, S.; Watanabe, K. Prescription of kampo drugs in the Japanese health care insurance program. Evid. Based Complement. Altern. Med. 2013, 2013, 576973. [Google Scholar] [CrossRef]
- Motoo, Y.; Cameron, S. Kampo medicines for supportive care of patients with cancer: A brief review. Integr. Med. Res. 2022, 11, 100839. [Google Scholar] [CrossRef]
- Shimizu, M.; Takayama, S.; Kikuchi, A.; Arita, R.; Ono, R.; Ishizawa, K.; Ishii, T. Kampo medicine treatment for advanced pancreatic cancer: A case series. Front. Nutr. 2021, 8, 513. [Google Scholar] [CrossRef]
- Takayama, S.; Tomita, N.; Arita, R.; Ono, R.; Kikuchi, A.; Ishii, T. Kampo Medicine for various aging-related symptoms: A review of geriatric syndrome. Front. Nutr. 2020, 7, 86. [Google Scholar] [CrossRef]
- Yamakawa, J.-i.; Motoo, Y.; Moriya, J.; Ogawa, M.; Uenishi, H.; Akazawa, S.; Sasagawa, T.; Nishio, M.; Kobayashi, J. Role of Kampo medicine in integrative cancer therapy. Evid. Based Complement. Altern. Med. 2013, 2013, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, K.; Matsuura, K.; Gao, P.; Hottenbacher, L.; Tokunaga, H.; Nishimura, K.; Imazu, Y.; Reissenweber, H.; Witt, C.M. Traditional Japanese Kampo medicine: Clinical research between modernity and traditional medicine—The state of research and methodological suggestions for the future. Evid. Based Complement. Altern. Med. 2011, 2011, 513842. [Google Scholar] [CrossRef] [PubMed]
- Ito, A.; Munakata, K.; Imazu, Y.; Watanabe, K. First nationwide attitude survey of Japanese physicians on the use of traditional Japanese medicine (Kampo) in cancer treatment. Evid. Based Complement. Altern. Med. 2012, 2012, 957082. [Google Scholar] [CrossRef] [PubMed]
- Aoyama, T.; Tamagawa, H. The clinical effect of Kampo medicine in multimodal treatment for Gastrointestinal Cancer in Japan. J. Cancer 2020, 11, 5390. [Google Scholar] [CrossRef] [PubMed]
- Rondilla, N.A.; Rocha, I.C.N.; Roque, S.J.; Lu, R.M.; Apolinar, N.L.B.; Solaiman-Balt, A.A.; Abion, T.J.; Banatin, P.B.; Javier, C.V. Folk Medicine in the Philippines: A Phenomenological Study of Health-Seeking Individuals. Int. J. Med. Stud. 2021, 9, 25–32. [Google Scholar] [CrossRef]
- Arnason, J.; Cal, V.; Pesek, T.; Awad, R.; Bourbonnais-Spear, N.; Collins, S.; Otarola-Rojas, M.; Walshe-Roussel, B.; Audet, P.; Ta, C.A. A review of ethnobotany and ethnopharmacology of traditional medicines used by Q’eqchi’Maya Healers of Xna’ajeb’aj Ralch’o’och’, Belize. Botany 2022, 100, 219–230. [Google Scholar] [CrossRef]
- Jorim, R.Y.; Korape, S.; Legu, W.; Koch, M.; Barrows, L.R.; Matainaho, T.K.; Rai, P.P. An ethnobotanical survey of medicinal plants used in the eastern highlands of Papua New Guinea. J. Ethnobiol. Ethnomed. 2012, 8, 47. [Google Scholar] [CrossRef]
- Neergheen, V.S.; Kam, A.H.; Pem, Y.; Ramsaha, S.; Bahorun, T. Regulation of cancer cell signaling pathways as key events for therapeutic relevance of edible and medicinal mushrooms. Proc. Semin. Cancer Biol. 2020, 80, 145–156. [Google Scholar] [CrossRef]
- Shoaib, A.; Tabish, M.; Ali, S.; Arafah, A.; Wahab, S.; Almarshad, F.M.; Rashid, S.; Rehman, M.U. Dietary phytochemicals in cancer signalling pathways: Role of miRNA targeting. Curr. Med. Chem. 2021, 28, 8036–8067. [Google Scholar] [CrossRef]
- Oyenihi, O.R.; Oyenihi, A.B.; Erhabor, J.O.; Matsabisa, M.G.; Oguntibeju, O.O. Unravelling the anticancer mechanisms of traditional herbal medicines with metabolomics. Molecules 2021, 26, 6541. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Kontomanolis, E.N.; Koutras, A.; Syllaios, A.; Schizas, D.; Mastoraki, A.; Garmpis, N.; Diakosavvas, M.; Angelou, K.; Tsatsaris, G.; Pagkalos, A. Role of oncogenes and tumor-suppressor genes in carcinogenesis: A review. Anticancer Res. 2020, 40, 6009–6015. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Vega, F.; Mina, M.; Armenia, J.; Chatila, W.K.; Luna, A.; La, K.C.; Dimitriadoy, S.; Liu, D.L.; Kantheti, H.S.; Saghafinia, S. Oncogenic signaling pathways in the cancer genome atlas. Cell 2018, 173, 321–337.e310. [Google Scholar] [CrossRef]
- Witsch, E.; Sela, M.; Yarden, Y. Roles for growth factors in cancer progression. Physiology 2010, 25, 85–101. [Google Scholar] [CrossRef] [PubMed]
- Patranabis, S. Recent Advances in the Therapeutic Development of Receptor Tyrosine Kinases (RTK) against Different Types of Cancer. In Protein Kinases—Promising Targets for Anticancer Drug Research; InTechOpen: London, UK, 2021; p. 103. [Google Scholar]
- Zou, Z.; Tao, T.; Li, H.; Zhu, X. mTOR signaling pathway and mTOR inhibitors in cancer: Progress and challenges. Cell Biosci. 2020, 10, 31. [Google Scholar] [CrossRef] [PubMed]
- Yun, C.W.; Jeon, J.; Go, G.; Lee, J.H.; Lee, S.H. The dual role of autophagy in cancer development and a therapeutic strategy for cancer by targeting autophagy. Int. J. Mol. Sci. 2020, 22, 179. [Google Scholar] [CrossRef]
- Al-Masri, M.; Paliotti, K.; Tran, R.; Halaoui, R.; Lelarge, V.; Chatterjee, S.; Wang, L.-T.; Moraes, C.; McCaffrey, L. Architectural control of metabolic plasticity in epithelial cancer cells. Commun. Biol. 2021, 4, 371. [Google Scholar] [CrossRef]
- Shuvalov, O.; Petukhov, A.; Daks, A.; Fedorova, O.; Vasileva, E.; Barlev, N.A. One-carbon metabolism and nucleotide biosynthesis as attractive targets for anticancer therapy. Oncotarget 2017, 8, 23955. [Google Scholar] [CrossRef]
- Shuvalov, O.; Daks, A.; Fedorova, O.; Petukhov, A.; Barlev, N. Linking metabolic reprogramming, plasticity and tumor progression. Cancers 2021, 13, 762. [Google Scholar] [CrossRef]
- Daks, A.; Shuvalov, O.; Fedorova, O.; Petukhov, A.; Lezina, L.; Zharova, A.; Baidyuk, E.; Khudiakov, A.; Barlev, N.A. p53-Independent Effects of Set7/9 Lysine Methyltransferase on Metabolism of Non-Small Cell Lung Cancer Cells. Front. Oncol. 2021, 11, 706668. [Google Scholar] [CrossRef]
- Munshi, P.N.; Lubin, M.; Bertino, J.R. 6-thioguanine: A drug with unrealized potential for cancer therapy. Oncologist 2014, 19, 760–765. [Google Scholar] [CrossRef] [PubMed]
- Morgunkova, A.; Barlev, N.A. Lysine methylation goes global. Cell Cycle 2006, 5, 1308–1312. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Yan, Q. Cancer epigenetics, tumor immunity, and immunotherapy. Trends Cancer 2020, 6, 580–592. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; He, C.; Wang, M.; Ma, X.; Mo, F.; Yang, S.; Han, J.; Wei, X. Targeting epigenetic regulators for cancer therapy: Mechanisms and advances in clinical trials. Signal Transduct. Target. Ther. 2019, 4, 62. [Google Scholar] [CrossRef] [PubMed]
- Daks, A.; Vasileva, E.; Fedorova, O.; Shuvalov, O.; Barlev, N.A. The Role of Lysine Methyltransferase SET7/9 in Proliferation and Cell Stress Response. Life 2022, 12, 362. [Google Scholar] [CrossRef]
- Vasileva, E.; Shuvalov, O.; Petukhov, A.; Fedorova, O.; Daks, A.; Nader, R.; Barlev, N. KMT Set7/9 is a new regulator of Sam68 STAR-protein. Biochem. Biophys. Res. Commun. 2020, 525, 1018–1024. [Google Scholar] [CrossRef]
- Pottier, C.; Fresnais, M.; Gilon, M.; Jérusalem, G.; Longuespée, R.; Sounni, N.E. Tyrosine kinase inhibitors in cancer: Breakthrough and challenges of targeted therapy. Cancers 2020, 12, 731. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, L.; Hei, R.; Li, X.; Cai, H.; Wu, X.; Zheng, Q.; Cai, C. CDK inhibitors in cancer therapy, an overview of recent development. Am. J. Cancer Res. 2021, 11, 1913. [Google Scholar]
- Mukherjee, N.; Amato, C.M.; Skees, J.; Todd, K.J.; Lambert, K.A.; Robinson, W.A.; Van Gulick, R.; Weight, R.M.; Dart, C.R.; Tobin, R.P. Simultaneously inhibiting BCL2 and MCL1 is a therapeutic option for patients with advanced melanoma. Cancers 2020, 12, 2182. [Google Scholar] [CrossRef]
- He, P.; Qiu, K.; Jia, Y. Modeling of mesenchymal hybrid epithelial state and phenotypic transitions in EMT and MET processes of cancer cells. Sci. Rep. 2018, 8, 14323. [Google Scholar] [CrossRef]
- Bhatia, S.; Wang, P.; Toh, A.; Thompson, E.W. New insights into the role of phenotypic plasticity and EMT in driving cancer progression. Front. Mol. Biosci. 2020, 7, 71. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Pestell, T.G.; Lisanti, M.P.; Pestell, R.G. Cancer stem cells. Int. J. Biochem. Cell Biol. 2012, 44, 2144–2151. [Google Scholar] [CrossRef] [PubMed]
- Peitzsch, C.; Tyutyunnykova, A.; Pantel, K.; Dubrovska, A. Cancer stem cells: The root of tumor recurrence and metastases. Proc. Semin. Cancer Biol. 2017, 44, 10–24. [Google Scholar] [CrossRef]
- Ermakov, A.; Daks, A.; Fedorova, O.; Shuvalov, O.; Barlev, N.A. Ca2+-depended signaling pathways regulate self-renewal and pluripotency of stem cells. Cell Biol. Int. 2018, 42, 1086–1096. [Google Scholar] [CrossRef] [PubMed]
- Waldman, A.D.; Fritz, J.M.; Lenardo, M.J. A guide to cancer immunotherapy: From T cell basic science to clinical practice. Nature Rev. Immunol. 2020, 20, 651–668. [Google Scholar] [CrossRef] [PubMed]
- Smirnov, S.; Petukhov, A.; Levchuk, K.; Kulemzin, S.; Staliarova, A.; Lepik, K.; Shuvalov, O.; Zaritskey, A.; Daks, A.; Fedorova, O. Strategies to Circumvent the Side-Effects of Immunotherapy Using Allogeneic CAR-T Cells and Boost Its Efficacy: Results of Recent Clinical Trials. Front. Immunol. 2021, 12, 780145. [Google Scholar] [CrossRef]
- Alves-Silva, J.M.; Romane, A.; Efferth, T.; Salgueiro, L. North African medicinal plants traditionally used in cancer therapy. Front. Pharmacol. 2017, 8, 383. [Google Scholar] [CrossRef]
- Mbaveng, A.T.; Kuete, V.; Efferth, T. Potential of Central, Eastern and Western Africa medicinal plants for cancer therapy: Spotlight on resistant cells and molecular targets. Front. Pharmacol. 2017, 8, 343. [Google Scholar] [CrossRef]
- Klopper, R.R.; Gautier, L.; Chatelain, C.; Smith, G.F.; Spichiger, R. Floristics of the angiosperm flora of Sub-Saharan Africa: An analysis of the African Plant Checklist and Database. Taxon 2007, 56, 201–208. [Google Scholar]
- Linder, H.P. The evolution of African plant diversity. Front. Ecol. Evolut. 2014, 2, 38. [Google Scholar] [CrossRef]
- Rather, L.J.; Mohammad, F. Acacia nilotica (L.): A review of its traditional uses, phytochemistry, and pharmacology. Sustain. Chem. Pharm. 2015, 2, 12–30. [Google Scholar] [CrossRef]
- Malami, I.; Jagaba, N.M.; Abubakar, I.B.; Muhammad, A.; Alhassan, A.M.; Waziri, P.M.; Yahaya, I.Z.Y.; Mshelia, H.E.; Mathias, S.N. Integration of medicinal plants into the traditional system of medicine for the treatment of cancer in Sokoto State, Nigeria. Heliyon 2020, 6, e04830. [Google Scholar] [CrossRef] [PubMed]
- Barapatre, A.; Meena, A.S.; Mekala, S.; Das, A.; Jha, H. In vitro evaluation of antioxidant and cytotoxic activities of lignin fractions extracted from Acacia nilotica. Int. J. Biol. Macromol. 2016, 86, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Zheleva-Dimitrova, D.; Sinan, K.I.; Etienne, O.K.; Ak, G.; Sharmeen, J.B.; Dervisoglu, G.; Ozdemir, F.A.; Mahomoodally, M.F.; Zengin, G. Comprehensive chemical characterization and biological evaluation of two Acacia species: A. nilotica and A. ataxacantha. Food Chem. Toxicol. 2021, 156, 112446. [Google Scholar] [CrossRef]
- Revathi, S.; Hakkim, F.L.; Kumar, N.R.; Bakshi, H.A.; Rashan, L.; Al-Buloshi, M.; Hasson, S.S.; Krishnan, M.; Javid, F.; Nagarajan, K. Induction of HT-29 colon cancer cells apoptosis by Pyrogallol with growth inhibiting efficacy against drug-resistant Helicobacter pylori. Anticancer Agents Med. Chem. 2018, 18, 1875–1884. [Google Scholar] [CrossRef]
- Sakthive, K.; Kannan, N.; Angeline, A.; Guruvayoorappan, C. Anticancer activity of Acacia nilotica (L.) Wild. Ex. Delile subsp. indica against Dalton’s ascitic lymphoma induced solid and ascitic tumor model. Asian Pac. J. Cancer Prev. 2012, 13, 3989–3995. [Google Scholar] [CrossRef]
- Revathi, S.; Hakkim, F.L.; Kumar, N.R.; Bakshi, H.A.; Sangilimuthu, A.Y.; Tambuwala, M.M.; Changez, M.; Nasef, M.M.; Krishnan, M.; Kayalvizhi, N. In vivo anti cancer potential of pyrogallol in murine model of colon cancer. Asian Pac. J. Cancer Prev. 2019, 20, 2645. [Google Scholar] [CrossRef]
- Al-Nour, M.Y.; Ibrahim, M.M.; Elsaman, T. Ellagic acid, Kaempferol, and Quercetin from Acacia nilotica: Promising combined drug with multiple mechanisms of action. Curr. Pharmacol. Rep. 2019, 5, 255–280. [Google Scholar] [CrossRef]
- Thiagarajan, K.; Mohan, S.; Roy, T.K.; Chandrasekaran, R. Antiproliferative effect of Acacia nilotica (L.) leaf extract rich in ethyl gallate against human carcinoma cell line KB. Indian J. Pharmacol. 2020, 52, 488. [Google Scholar]
- Dirar, A.I.; Devkota, H.P. Ethnopharmacological uses, phytochemistry and pharmacological activities of Guiera senegalensis JF Gmel. (Combretaceae). J. Ethnopharmacol. 2021, 267, 113433. [Google Scholar] [CrossRef]
- Adebayo, I.A.; Gagman, H.A.; Balogun, W.G.; Adam, M.A.A.; Abas, R.; Hakeem, K.R.; Nik Him, N.A.I.I.B.; Samian, M.R.B.; Arsad, H. Detarium microcarpum, Guiera senegalensis, and Cassia siamea induce apoptosis and cell cycle arrest and inhibit metastasis on MCF7 breast cancer cells. Evid. Based Complement. Altern. Med. 2019, 2019, 6104574. [Google Scholar]
- Kuete, V.; Eichhorn, T.; Wiench, B.; Krusche, B.; Efferth, T. Cytotoxicity, anti-angiogenic, apoptotic effects and transcript profiling of a naturally occurring naphthyl butenone, guieranone A. Cell Div. 2012, 7, 16. [Google Scholar] [CrossRef] [PubMed]
- Bello, B.A.; Khan, S.A.; Khan, J.A.; Syed, F.Q.; Anwar, Y.; Khan, S.B. Antiproliferation and antibacterial effect of biosynthesized AgNps from leaves extract of Guiera senegalensis and its catalytic reduction on some persistent organic pollutants. J. Photochem. Photobiol. B Biol. 2017, 175, 99–108. [Google Scholar] [CrossRef]
- Singh, R.; Kaur, H. Advances in synthetic approaches for the preparation of combretastatin-based anti-cancer agents. Synthesis 2009, 2009, 2471–2491. [Google Scholar] [CrossRef]
- Karatoprak, G.Ş.; Küpeli Akkol, E.; Genç, Y.; Bardakcı, H.; Yücel, Ç.; Sobarzo-Sánchez, E. Combretastatins: An overview of structure, probable mechanisms of action and potential applications. Molecules 2020, 25, 2560. [Google Scholar] [CrossRef] [PubMed]
- Nwankwo, J. Anticancer potentials of phytochemicals from some indigenous food and medicinal plants of West Africa. Adv. Cancer Prev. 2017, 2, 124–130. [Google Scholar] [CrossRef]
- Twilley, D.; Rademan, S.; Lall, N. A review on traditionally used South African medicinal plants, their secondary metabolites and their potential development into anticancer agents. J. Ethnopharmacol. 2020, 261, 113101. [Google Scholar] [CrossRef]
- Sagbo, I.J.; Otang-Mbeng, W. Plants used for the traditional management of cancer in the eastern cape province of south africa: A review of ethnobotanical surveys, ethnopharmacological studies and active phytochemicals. Molecules 2021, 26, 4639. [Google Scholar] [CrossRef]
- Khorombi, T.; Fouché, G.; Kolesnikova, N.; Maharaj, V.; Nthambeleni, R.; Van der Merwe, M.R. Investigation of South African plants for anti cancer properties. Pharmacology 2006, 3, 494–500. [Google Scholar]
- Matowa, P.R.; Gundidza, M.; Gwanzura, L.; Nhachi, C.F. A survey of ethnomedicinal plants used to treat cancer by traditional medicine practitioners in Zimbabwe. BMC Complement. Med. Ther. 2020, 20, 278. [Google Scholar] [CrossRef]
- Akindele, A.J.; Wani, Z.; Mahajan, G.; Sharma, S.; Aigbe, F.R.; Satti, N.; Adeyemi, O.O.; Mondhe, D.M. Anticancer activity of Aristolochia ringens Vahl. (Aristolochiaceae). J. Tradit. Complement. Med. 2015, 5, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, J.; Ajani, E.; Sabiu, S. Chemical group profiling, in vitro and in silico evaluation of Aristolochia ringens on α-amylase and α-glucosidase activity. Evid. Based Complement. Altern. Med. 2021, 2021, 6679185. [Google Scholar] [CrossRef] [PubMed]
- Kuete, V.; Tankeo, S.B.; Saeed, M.E.; Wiench, B.; Tane, P.; Efferth, T. Cytotoxicity and modes of action of five Cameroonian medicinal plants against multi-factorial drug resistance of tumor cells. J. Ethnopharmacol. 2014, 153, 207–219. [Google Scholar] [CrossRef] [PubMed]
- Somaida, A.; Tariq, I.; Ambreen, G.; Abdelsalam, A.M.; Ayoub, A.M.; Wojcik, M.; Dzoyem, J.P.; Bakowsky, U. Potent cytotoxicity of four cameroonian plant extracts on different cancer cell lines. Pharmaceuticals 2020, 13, 357. [Google Scholar] [CrossRef]
- Pieme, C.A.; Guru, S.K.; Ambassa, P.; Kumar, S.; Ngameni, B.; Ngogang, J.Y.; Bhushan, S.; Saxena, A.K. Induction of mitochondrial dependent apoptosis and cell cycle arrest in human promyelocytic leukemia HL-60 cells by an extract from Dorstenia psilurus: A spice from Cameroon. BMC Complement. Altern. Med. 2013, 13, 223. [Google Scholar] [CrossRef]
- Kuete, V.; Sandjo, L.P.; Wiench, B.; Efferth, T. Cytotoxicity and modes of action of four Cameroonian dietary spices ethno-medically used to treat cancers: Echinops giganteus, Xylopia aethiopica, Imperata cylindrica and Piper capense. J. EthnoPharmacol. 2013, 149, 245–253. [Google Scholar] [CrossRef]
- Sandjo, L.P.; Kuete, V.; Siwe, X.N.; Poumale, H.M.; Efferth, T. Cytotoxicity of an unprecedented brominated oleanolide and a new furoceramide from the Cameroonian spice, Echinops giganteus. Nat. Product Res. 2016, 30, 2529–2537. [Google Scholar] [CrossRef]
- Jung, Y.-K.; Shin, D. Imperata cylindrica: A review of phytochemistry, pharmacology, and industrial applications. Molecules 2021, 26, 1454. [Google Scholar] [CrossRef]
- Wamba, B.E.; Ghosh, P.; Mbaveng, A.T.; Bhattacharya, S.; Debarpan, M.; Depanwita, S.; Saunak, M.M.; Kuete, V.; Murmu, N. Botanical from Piper capense Fruit Can Help to Combat the Melanoma as Demonstrated by In Vitro and In Vivo Studies. Evid. Based Complement. Altern. Med. 2021, 2021, 8810368. [Google Scholar] [CrossRef]
- Ulloa Ulloa, C.; Acevedo-Rodríguez, P.; Beck, S.; Belgrano, M.J.; Bernal, R.; Berry, P.E.; Brako, L.; Celis, M.; Davidse, G.; Forzza, R.C. An integrated assessment of the vascular plant species of the Americas. Science 2017, 358, 1614–1617. [Google Scholar] [CrossRef]
- Zhang, J.; Hunto, S.T.; Yang, Y.; Lee, J.; Cho, J.Y. Tabebuia impetiginosa: A comprehensive review on Tradit. uses, phytochemistry, and immunopharmacological properties. Molecules 2020, 25, 4294. [Google Scholar] [CrossRef] [PubMed]
- Warashina, T.; Nagatani, Y.; Noro, T. Constituents from the bark of Tabebuia impetiginosa. PhytoChem. 2004, 65, 2003–2011. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, B.; Telang, N.; Wong, G. Growth inhibition of estrogen receptor positive human breast cancer cells by Taheebo from the inner bark of Tabebuia avellandae tree. Int. J. Mol. Med. 2009, 24, 253–260. [Google Scholar] [PubMed]
- Kee, J.-Y.; Han, Y.-H.; Park, J.; Kim, D.-S.; Mun, J.-G.; Ahn, K.S.; Kim, H.-J.; Um, J.-Y.; Hong, S.-H. β-Lapachone inhibits lung metastasis of colorectal cancer by inducing apoptosis of CT26 cells. Integr. Cancer Ther. 2017, 16, 585–596. [Google Scholar] [CrossRef]
- Woo, H.J.; Park, K.-Y.; Rhu, C.-H.; Lee, W.H.; Choi, B.T.; Kim, G.Y.; Park, Y.-M.; Choi, Y.H. β-lapachone, a quinone isolated from Tabebuia avellanedae, induces apoptosis in HepG2 hepatoma cell line through induction of Bax and activation of caspase. J. Med. Food 2006, 9, 161–168. [Google Scholar] [CrossRef]
- Pires, T.C.; Dias, M.I.; Calhelha, R.C.; Carvalho, A.M.; Queiroz, M.-J.R.; Barros, L.; Ferreira, I.C. Bioactive properties of Tabebuia impetiginosa-based phytopreparations and phytoformulations: A comparison between extracts and dietary supplements. Molecules 2015, 20, 22863–22871. [Google Scholar] [CrossRef]
- De Melo, J.G.; Santos, A.G.; de Amorim, E.L.C.; de Nascimento, S.C.; de Albuquerque, U.P. Medicinal plants used as antitumor agents in Brazil: An ethnobotanical approach. Evid. Based Complement. Altern. Med. 2011, 2011, 365359. [Google Scholar] [CrossRef]
- Shankar Babu, M.; Mahanta, S.; Lakhter, A.J.; Hato, T.; Paul, S.; Naidu, S.R. Lapachol inhibits glycolysis in cancer cells by targeting pyruvate kinase M2. PLoS ONE 2018, 13, e0191419. [Google Scholar] [CrossRef]
- Zahra, K.; Dey, T.; Mishra, S.P.; Pandey, U. Pyruvate kinase M2 and cancer: The role of PKM2 in promoting tumorigenesis. Front. Oncol. 2020, 10, 159. [Google Scholar] [CrossRef]
- Shuvalov, O.; Kizenko, A.; Petukhov, A.; Fedorova, O.; Daks, A.; Bottrill, A.; Snezhkina, A.V.; Kudryavtseva, A.V.; Barlev, N. SEMG1/2 augment energy metabolism of tumor cells. Cell Death Dis. 2020, 11, 1047. [Google Scholar] [CrossRef]
- Zhao, W.; Jiang, L.; Fang, T.; Fang, F.; Liu, Y.; Zhao, Y.; You, Y.; Zhou, H.; Su, X.; Wang, J. β-Lapachone Selectively Kills Hepatocellular Carcinoma Cells by Targeting NQO1 to Induce Extensive DNA Damage and PARP1 Hyperactivation. Front. Oncol. 2021, 11, 747282. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.-L.; Lu, Y.-F.; Chen, H.; Shen, Z.-Y.; Liu, J. Liver expression of Nrf2-related genes in different liver diseases. Hepatobiliary Pancreat. Dis. Int. 2015, 14, 485–491. [Google Scholar] [CrossRef]
- Yang, Y.; Zheng, J.; Wang, M.; Zhang, J.; Tian, T.; Wang, Z.; Yuan, S.; Liu, L.; Zhu, P.; Gu, F. NQO1 promotes an aggressive phenotype in hepatocellular carcinoma via amplifying ERK-NRF2 signaling. Cancer Sci. 2021, 112, 641–654. [Google Scholar] [CrossRef] [PubMed]
- Hussain, H.; Green, I.R. Lapachol and lapachone analogs: A journey of two decades of patent research (1997–2016). Exp. Opin. Ther. Pat. 2017, 27, 1111–1121. [Google Scholar] [CrossRef]
- Rone, A.; Oliveira, K.M.; Guedes, A.P.; Dos Santos, P.W.; Aissa, A.F.; Batista, A.A.; Pavan, F.R. A Novel Ruthenium (II) Complex with Lapachol Induces G2/M Phase Arrest through Aurora-B Kinase Down-Regulation and ROS-Mediated Apoptosis in Human Prostate Adenocarcinoma Cells. Front. Oncol. 2021, 11, 2148. [Google Scholar]
- Chen, Q.; Bai, L.; Zhou, X.; Xu, P.; Li, X.; Xu, H.; Zheng, Y.; Zhao, Y.; Lu, S.; Xue, M. Development of long-circulating lapachol nanoparticles: Formation, characterization, pharmacokinetics, distribution and cytotoxicity. RSC Adv. 2020, 10, 30025–30034. [Google Scholar] [CrossRef]
- Beg, M.; Boothman, D.; Khosama, L.; Arriaga, Y.; Verma, U.; Sanjeeviaiah, A.; Kazmi, S.; Fattah, F.; Pilarski, S.; Rodriguez, M. A phase I/Ib, multi-center trial of ARQ-761 (Beta-Lapachone) with gemcitabine/nab-paclitaxel in patients with advanced pancreatic cancer. Ann. Oncol. 2019, 30, v271. [Google Scholar] [CrossRef]
- Tahara, T.; Watanabe, A.; Yutani, M.; Yamano, Y.; Sagara, M.; Nagai, S.; Saito, K.; Yamashita, M.; Ihara, M.; Iida, A. STAT3 inhibitory activity of naphthoquinones isolated from Tabebuia avellanedae. Bioorg. Med. Chem. 2020, 28, 115347. [Google Scholar] [CrossRef]
- Rauwald, H.W.; Maucher, R.; Dannhardt, G.; Kuchta, K. Dihydroisocoumarins, Naphthalenes, and Further Polyketides from Aloe vera and A. plicatilis: Isolation, Identification and Their 5-LOX/COX-1 Inhibiting Potency. Molecules 2021, 26, 4223. [Google Scholar] [CrossRef]
- Huang, P.-H.; Huang, C.-Y.; Chen, M.-C.; Lee, Y.-T.; Yue, C.-H.; Wang, H.-Y.; Lin, H. Emodin and aloe-emodin suppress breast cancer cell proliferation through ERα inhibition. Evid. Based Complement. Altern. Med. 2013, 2013, 376123. [Google Scholar] [CrossRef]
- Wang, Z.; Tang, T.; Wang, S.; Cai, T.; Tao, H.; Zhang, Q.; Qi, S.; Qi, Z. Aloin inhibits the proliferation and migration of gastric cancer cells by regulating NOX2–ROS-mediated pro-survival signal pathways. Drug Des. Dev. Ther. 2020, 14, 145. [Google Scholar] [CrossRef] [PubMed]
- Sun, R.; Zhai, R.; Ma, C.; Miao, W. Combination of aloin and metformin enhances the antitumor effect by inhibiting the growth and invasion and inducing apoptosis and autophagy in hepatocellular carcinoma through PI3K/AKT/mTOR pathway. Cancer Med. 2020, 9, 1141–1151. [Google Scholar] [CrossRef] [PubMed]
- Pan, Q.; Pan, H.; Lou, H.; Xu, Y.; Tian, L. Inhibition of the angiogenesis and growth of Aloin in human colorectal cancer in vitro and in vivo. Cancer Cell Int. 2013, 13, 69. [Google Scholar] [CrossRef] [PubMed]
- Sanders, B.; Ray, A.M.; Goldberg, S.; Clark, T.; McDaniel, H.R.; Atlas, S.E.; Farooqi, A.; Konefal, J.; Lages, L.C.; Lopez, J. Anti-cancer effects of aloe-emodin: A systematic review. J. Clin. Transl. Res. 2018, 3, 283. [Google Scholar] [PubMed]
- Manirakiza, A.; Irakoze, L.; Manirakiza, S. Aloe and its Effects on Cancer: A Narrative Literature Review. East Afr. Health Res. J. 2021, 5, 1. [Google Scholar] [CrossRef]
- Dong, X.; Zeng, Y.; Liu, Y.; You, L.; Yin, X.; Fu, J.; Ni, J. Aloe-emodin: A review of its pharmacology, toxicity, and pharmacokinetics. Phytother. Res. 2020, 34, 270–281. [Google Scholar] [CrossRef]
- Cheng, C.; Dong, W. Aloe-emodin induces endoplasmic reticulum stress-dependent apoptosis in colorectal cancer cells. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2018, 24, 6331. [Google Scholar] [CrossRef]
- Shen, F.; Ge, C.; Yuan, P. Aloe-emodin induces autophagy and apoptotic cell death in non-small cell lung cancer cells via Akt/mTOR and MAPK signaling. Eur. J. Pharmacol. 2020, 886, 173550. [Google Scholar] [CrossRef]
- Wang, S.; Yan, W.-W.; He, M.; Wei, D.; Long, Z.-J.; Tao, Y.-M. Aloe emodin inhibits telomerase activity in breast cancer cells: Transcriptional and enzymological mechanism. Pharmacol. Rep. 2020, 72, 1383–1396. [Google Scholar] [CrossRef]
- Kosiol, N.; Juranek, S.; Brossart, P.; Heine, A.; Paeschke, K. G-quadruplexes: A promising target for cancer therapy. Mol. Cancer 2021, 20, 40. [Google Scholar] [CrossRef]
- Das, A.; Dutta, S. Binding Studies of Aloe-Active Compounds with G-Quadruplex Sequences. ACS Omega 2021, 6, 18344–18351. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Ren, K.; Liang, Y.; Liu, J.; Liang, Z.; Zhang, Y. Aloin promotes cell apoptosis by targeting HMGB1-TLR4-ERK axis in human melanoma cells. EXCLI J. 2020, 19, 641. [Google Scholar]
- Tao, H.; Tang, T.; Wang, S.; Wang, Z.; Ma, Y.; Cai, T.; Cheng, X.; Qi, S.; Zhang, Y.; Qi, Z. The molecular mechanisms of Aloin induce gastric cancer cells apoptosis by targeting High Mobility Group Box 1. Drug Des. Dev. Ther. 2019, 13, 1221. [Google Scholar] [CrossRef]
- Birari, L.; Wagh, S.; Patil, K.R.; Mahajan, U.B.; Unger, B.; Belemkar, S.; Goyal, S.N.; Ojha, S.; Patil, C.R. Aloin alleviates doxorubicin-induced cardiotoxicity in rats by abrogating oxidative stress and pro-inflammatory cytokines. Cancer Chemother. Pharmacol. 2020, 86, 419–426. [Google Scholar] [CrossRef] [PubMed]
- Lissoni, P.; Rovelli, F.; Brivio, F.; Zago, R.; Colciago, M.; Messina, G.; Mora, A.; Porro, G. A randomized study of chemotherapy versus biochemotherapy with chemotherapy plus Aloe arborescens in patients with metastatic cancer. In Vivo 2009, 23, 171–175. [Google Scholar] [PubMed]
- Lissoni, P.; Giani, L.; Zerbini, S.; Trabattoni, P.; Rovelli, F. Biotherapy with the pineal immunomodulating hormone melatonin versus melatonin plus Aloe vera in untreatable advanced solid neoplasms. Nat. Immun. 1998, 16, 27–33. [Google Scholar] [CrossRef]
- Damani, M.R.; Shah, A.R.; Karp, C.L.; Orlin, S.E. Treatment of ocular surface squamous neoplasia with topical Aloe vera drops. Cornea 2015, 34, 87–89. [Google Scholar] [CrossRef]
- Peng, C.; Zhang, W.; Dai, C.; Li, W.; Shen, X.; Yuan, Y.; Yan, L.; Zhang, W.; Yao, M. Study of the aqueous extract of Aloe vera and its two active components on the Wnt/β-catenin and Notch signaling pathways in colorectal cancer cells. J. Ethnopharmacol. 2019, 243, 112092. [Google Scholar] [CrossRef]
- Arul, B.; Kothai, R. Anticancer effect of capsaicin and its analogues. In Capsicum; IntechOpen: London, UK, 2020. [Google Scholar]
- Chapa-Oliver, A.M.; Mejía-Teniente, L. Capsaicin: From plants to a cancer-suppressing agent. Molecules 2016, 21, 931. [Google Scholar] [CrossRef]
- Chakraborty, S.; Adhikary, A.; Mazumdar, M.; Mukherjee, S.; Bhattacharjee, P.; Guha, D.; Choudhuri, T.; Chattopadhyay, S.; Sa, G.; Sen, A. Capsaicin-induced activation of p53-SMAR1 auto-regulatory loop down-regulates VEGF in non-small cell lung cancer to restrain angiogenesis. PLoS ONE 2014, 9, e99743. [Google Scholar] [CrossRef]
- Dai, N.; Ye, R.; He, Q.; Guo, P.; Chen, H.; Zhang, Q. Capsaicin and sorafenib combination treatment exerts synergistic anti-hepatocellular carcinoma activity by suppressing EGFR and PI3K/Akt/mTOR signaling. Oncol. Rep. 2018, 40, 3235–3248. [Google Scholar] [PubMed]
- Lin, Y.-T.; Wang, H.-C.; Hsu, Y.-C.; Cho, C.-L.; Yang, M.-Y.; Chien, C.-Y. Capsaicin induces autophagy and apoptosis in human nasopharyngeal carcinoma cells by downregulating the PI3K/AKT/mTOR pathway. Int. J. Mol. Sci. 2017, 18, 1343. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.-F.; Islam, A.; Liu, P.-F.; Zhan, J.-H.; Chueh, P.J. Capsaicin acts through tNOX (ENOX2) to induce autophagic apoptosis in p53-mutated HSC-3 cells but autophagy in p53-functional SAS oral cancer cells. Am. J. Cancer Res. 2020, 10, 3230. [Google Scholar] [PubMed]
- Friedman, J.R.; Richbart, S.D.; Merritt, J.C.; Brown, K.C.; Denning, K.L.; Tirona, M.T.; Valentovic, M.A.; Miles, S.L.; Dasgupta, P. Capsaicinoids: Multiple effects on angiogenesis, invasion and metastasis in human cancers. Biomed. Pharmacother. 2019, 118, 109317. [Google Scholar] [CrossRef] [PubMed]
- Amantini, C.; Morelli, M.B.; Nabissi, M.; Cardinali, C.; Santoni, M.; Gismondi, A.; Santoni, G. Capsaicin triggers autophagic cell survival which drives epithelial mesenchymal transition and chemoresistance in bladder cancer cells in an Hedgehog-dependent manner. Oncotarget 2016, 7, 50180. [Google Scholar] [CrossRef] [PubMed]
- Fedorova, O.; Daks, A.; Parfenyev, S.; Shuvalov, O.; Netsvetay, S.; Vasileva, J.; Gudovich, A.; Golotin, V.; Semenov, O.; Petukhov, A. Zeb1-mediated autophagy enhances resistance of breast cancer cells to genotoxic drugs. Biochem. Biophys. Res. Commun. 2022, 589, 29–34. [Google Scholar] [CrossRef]
- de Souza, P.O.; Bianchi, S.E.; Figueiró, F.; Heimfarth, L.; Moresco, K.S.; Gonçalves, R.M.; Hoppe, J.B.; Klein, C.P.; Salbego, C.G.; Gelain, D.P. Anticancer activity of flavonoids isolated from Achyrocline satureioides in gliomas cell lines. Toxicol. In Vitro 2018, 51, 23–33. [Google Scholar] [CrossRef]
- Soares Machado, M.; Palma, A.; Panelo, L.C.; Paz, L.A.; Rosa, F.; Lira, M.C.; Azurmendi, P.; Rubio, M.F.; Lenz, G.; Urtreger, A.J. Extract from Aloysia polystachya induces the cell death of colorectal cancer stem cells. Nutr. Cancer 2020, 72, 1004–1017. [Google Scholar] [CrossRef]
- Lamorte, D.; Faraone, I.; Laurenzana, I.; Trino, S.; Russo, D.; Rai, D.K.; Armentano, M.F.; Musto, P.; Sgambato, A.; De Luca, L. Advances in Azorella glabra Wedd. Extract research: In vitro antioxidant activity, antiproliferative effects on acute myeloid leukemia cells and bioactive compound characterization. Molecules 2020, 25, 4890. [Google Scholar] [CrossRef]
- Mellado, M.; Soto, M.; Madrid, A.; Montenegro, I.; Jara-Gutiérrez, C.; Villena, J.; Werner, E.; Godoy, P.; Aguilar, L.F. In vitro antioxidant and antiproliferative effect of the extracts of Ephedra chilensis K Presl aerial parts. BMC Complement. Altern. Med. 2019, 19, 53. [Google Scholar] [CrossRef]
- Alonso-Castro, A.J.; Ortiz-Sánchez, E.; Domínguez, F.; López-Toledo, G.; Chávez, M.; de Jesús Ortiz-Tello, A.; García-Carrancá, A. Antitumor effect of Croton lechleri mull. arg.(euphorbiaceae). J. Ethnopharmacol. 2012, 140, 438–442. [Google Scholar] [CrossRef] [PubMed]
- Montopoli, M.; Bertin, R.; Chen, Z.; Bolcato, J.; Caparrotta, L.; Froldi, G. Croton lechleri sap. and isolated alkaloid taspine exhibit inhibition against human melanoma SK23 and colon cancer HT29 cell lines. J. Ethnopharmacol. 2012, 144, 747–753. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, A.; Saito, Y.; Fukuyoshi, S.; Goto, M.; Miyake, K.; Newman, D.J.; O’Keefe, B.R.; Lee, K.-H.; Nakagawa-Goto, K. Corymbulosins D–H, 2-Hydroxy-and 2-Oxo-clerodane Diterpenes from the Bark of Laetia corymbulosa. J. Nat. Prod. 2017, 80, 1065–1072. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.; Wei, J.; Gao, Y.; Chen, R. Antioxidant and antitumoral activities of isolated macamide and macaene fractions from Lepidium meyenii (Maca). Talanta 2021, 221, 121635. [Google Scholar] [CrossRef] [PubMed]
- Bosio, C.; Tomasoni, G.; Martínez, R.; Olea, A.F.; Carrasco, H.; Villena, J. Cytotoxic and apoptotic effects of leptocarpin, a plant-derived sesquiterpene lactone, on human cancer cell lines. Chem. Biol. Interact. 2015, 242, 415–421. [Google Scholar] [CrossRef]
- Amaral, R.G.; Gomes, S.V.; Andrade, L.N.; Dos Santos, S.A.; Severino, P.; de Albuquerque Júnior, R.L.; Souto, E.B.; Brandão, G.C.; Santos, S.L.; David, J.M. Cytotoxic, antitumor and toxicological profile of Passiflora alata leaf extract. Molecules 2020, 25, 4814. [Google Scholar] [CrossRef]
- Ramos-Silva, A.; Tavares-Carreón, F.; Figueroa, M.; la Torre-Zavala, D.; Gastelum-Arellanez, A.; Rodríguez-García, A.; Galán-Wong, L.J.; Avilés-Arnaut, H. Anticancer potential of Thevetia peruviana fruit methanolic extract. BMC Complement. Altern. Med. 2017, 17, 241. [Google Scholar] [CrossRef]
- Kantarjian, H.M.; Talpaz, M.; Santini, V.; Murgo, A.; Cheson, B.; O’Brien, S.M. Homoharringtonine: History, current research, and future directions. Cancer 2001, 92, 1591–1605. [Google Scholar] [CrossRef]
- Kantarjian, H.M.; O’Brien, S.; Cortes, J. Homoharringtonine/omacetaxine mepesuccinate: The long and winding road to food and drug administration approval. Clin. Lymphoma Myeloma Leuk. 2013, 13, 530–533. [Google Scholar] [CrossRef]
- Xie, M.; Jiang, Q.; Li, L.; Zhu, J.; Zhu, L.; Zhou, D.; Zheng, Y.; Yang, X.; Zhu, M.; Sun, J. HAG (homoharringtonine, cytarabine, G-CSF) regimen for the treatment of acute myeloid leukemia and myelodysplastic syndrome: A meta-analysis with 2,314 participants. PLoS ONE 2016, 11, e0164238. [Google Scholar] [CrossRef]
- Gu, L.-F.; Zhang, W.-G.; Wang, F.-X.; Cao, X.-M.; Chen, Y.-X.; He, A.-L.; Liu, J.; Ma, X.-R. Low dose of homoharringtonine and cytarabine combined with granulocyte colony-stimulating factor priming on the outcome of relapsed or refractory acute myeloid leukemia. J. Cancer Res. Clin. Oncol. 2011, 137, 997–1003. [Google Scholar] [CrossRef] [PubMed]
- Winer, E.S.; DeAngelo, D.J. A review of omacetaxine: A chronic myeloid leukemia treatment resurrected. Oncol. Ther. 2018, 6, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.-J.; Zhang, W.-N.; Chen, B.; Xi, W.-D.; Lu, Y.; Huang, J.-Y.; Wang, Y.-Y.; Long, J.; Wu, S.-F.; Zhang, Y.-X. Homoharringtonine deregulates MYC transcriptional expression by directly binding NF-κB repressing factor. Proc. Nat. Acad. Sci. USA 2019, 116, 2220–2225. [Google Scholar] [CrossRef] [PubMed]
- Tong, H.; Ren, Y.; Zhang, F.; Jin, J. Homoharringtonine affects the JAK2-STAT5 signal pathway through alteration of protein tyrosine kinase phosphorylation in acute myeloid leukemia cells. Eur. J. Haematol. 2008, 81, 259–266. [Google Scholar] [CrossRef]
- Han, X.; Zhang, X.; Wang, Q.; Wang, L.; Yu, S. Antitumor potential of Hedyotis diffusa Willd: A systematic review of bioactive constituents and underlying molecular mechanisms. Biomed. Pharmacother. 2020, 130, 110735. [Google Scholar] [CrossRef]
- Lee, Y.K.; Bae, K.; Yoo, H.-S.; Cho, S.-H. Benefit of adjuvant Tradit. herbal medicine with chemotherapy for resectable gastric cancer. Integr. Cancer Ther. 2018, 17, 619–627. [Google Scholar] [CrossRef]
- Yeh, Y.-C.; Chen, H.-Y.; Yang, S.-H.; Lin, Y.-H.; Chiu, J.-H.; Lin, Y.-H.; Chen, J.-L. Hedyotis diffusa combined with Scutellaria barbata are the core treatment of Chinese herbal medicine used for breast cancer patients: A population-based study. Evid. Based Complement. Altern. Med. 2014, 2014, 202378. [Google Scholar] [CrossRef]
- Yang, B.; Wang, N.; Wang, S.; Li, X.; Zheng, Y.; Li, M.; Song, J.; Zhang, F.; Mei, W.; Lin, Y. Network-pharmacology-based identification of caveolin-1 as a key target of Oldenlandia diffusa to suppress breast cancer metastasis. Biomed. Pharmacother. 2019, 112, 108607. [Google Scholar] [CrossRef]
- Chung, T.-W.; Choi, H.; Lee, J.-M.; Ha, S.-H.; Kwak, C.-H.; Abekura, F.; Park, J.-Y.; Chang, Y.-C.; Ha, K.-T.; Cho, S.-H. Oldenlandia diffusa suppresses metastatic potential through inhibiting matrix metalloproteinase-9 and intercellular adhesion molecule-1 expression via p38 and ERK1/2 MAPK pathways and induces apoptosis in human breast cancer MCF-7 cells. J. Ethnopharmacol. 2017, 195, 309–317. [Google Scholar] [CrossRef]
- Lu, P.-H.; Chen, M.-B.; Ji, C.; Li, W.-T.; Wei, M.-X.; Wu, M.-H. Aqueous Oldenlandia diffusa extracts inhibits colorectal cancer cells via activating AMP-activated protein kinase signalings. Oncotarget 2016, 7, 45889. [Google Scholar] [CrossRef]
- Chen, Q.; Rahman, K.; Wang, S.-J.; Zhou, S.; Zhang, H. Scutellaria barbata: A review on chemical constituents, pharmacological activities and clinical applications. Curr. Pharm. Des. 2020, 26, 160–175. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.M.; Su, X.L. Anticancer effect of ursolic acid via mitochondria-dependent pathways. Oncol. Lett. 2019, 17, 4761–4767. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Xu, B.; Wang, X.; Zheng, B.; Du, J.; Liu, S. The analysis of the anti-tumor mechanism of ursolic acid using connectively map approach in breast cancer cells line MCF-7. Cancer Manag. Res. 2020, 12, 3469. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Ma, H.; Shi, W.; Duan, J.; Wang, Y.; Zhang, C.; Li, C.; Lin, J.; Li, S.; Lv, J. Inhibition of STAT3 signaling pathway by ursolic acid suppresses growth of hepatocellular carcinoma. Int. J. Oncol. 2017, 51, 555–562. [Google Scholar] [CrossRef]
- Wang, S.; Chang, X.; Zhang, J.; Li, J.; Wang, N.; Yang, B.; Pan, B.; Zheng, Y.; Wang, X.; Ou, H. Ursolic Acid Inhibits Breast Cancer Metastasis by Suppressing Glycolytic Metabolism via Activating SP1/Caveolin-1 Signaling. Front. Oncol. 2021, 11, 745584. [Google Scholar] [CrossRef]
- Lewinska, A.; Adamczyk-Grochala, J.; Kwasniewicz, E.; Deregowska, A.; Wnuk, M. Ursolic acid-mediated changes in glycolytic pathway promote cytotoxic autophagy and apoptosis in phenotypically different breast cancer cells. Apoptosis 2017, 22, 800–815. [Google Scholar] [CrossRef]
- Wang, J.; Jiang, Z.; Xiang, L.; Li, Y.; Ou, M.; Yang, X.; Shao, J.; Lu, Y.; Lin, L.; Chen, J. Synergism of ursolic acid derivative US597 with 2-deoxy-D-glucose to preferentially induce tumor cell death by dual-targeting of apoptosis and glycolysis. Sci. Rep. 2014, 4, 1–12. [Google Scholar] [CrossRef]
- Tang, Z.-Y.; Li, Y.; Tang, Y.-T.; Ma, X.-D.; Tang, Z.-Y. Anticancer activity of oleanolic acid and its derivatives: Recent advances in evidence, target profiling and mechanisms of action. Biomed. Pharmacother. 2022, 145, 112397. [Google Scholar] [CrossRef]
- Li, Y.; Xu, Q.; Yang, W.; Wu, T.; Lu, X. Oleanolic acid reduces aerobic glycolysis-associated proliferation by inhibiting yes-associated protein in gastric cancer cells. Gene 2019, 712, 143956. [Google Scholar] [CrossRef]
- Lee, J.-H.; Yoo, E.-S.; Han, S.-H.; Jung, G.-H.; Han, E.-J.; Jung, S.-H.; Kim, B.S.; Cho, S.-D.; Nam, J.-S.; Choi, C. Oleanolic acid induces apoptosis and autophagy via the PI3K/AKT/mTOR pathway in AGS human gastric cancer cells. J. Funct. Foods 2021, 87, 104854. [Google Scholar] [CrossRef]
- Nie, H.; Wang, Y.; Qin, Y.; Gong, X.G. Oleanolic acid induces autophagic death in human gastric cancer cells in vitro and in vivo. Cell Biol. Int. 2016, 40, 770–778. [Google Scholar] [CrossRef] [PubMed]
- Xiaofei, J.; Mingqing, S.; Miao, S.; Yizhen, Y.; Shuang, Z.; Qinhua, X.; Kai, Z. Oleanolic acid inhibits cervical cancer Hela cell proliferation through modulation of the ACSL4 ferroptosis signaling pathway. Biochem. Biophys. Res. Commun. 2021, 545, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Wang, S.; Niu, M. Scutellarin inhibits the growth and EMT of gastric cancer cells through regulating PTEN/PI3K pathway. Biol. Pharm. Bull. 2021, 44, 780–788. [Google Scholar] [CrossRef] [PubMed]
- Niu, G.; Sun, L.; Pei, Y.; Wang, D. Oleanolic acid inhibits colorectal cancer angiogenesis by blocking the VEGFR2 signaling pathway. Anti Cancer Agents Med. Chem. Formerly Curr. Med. Chem. Anti-Cancer Agents 2018, 18, 583–590. [Google Scholar] [CrossRef]
- Ma, T.-T.; Zhang, G.-L.; Dai, C.-F.; Zhang, B.-R.; Cao, K.-X.; Wang, C.-G.; Yang, G.-W.; Wang, X.-M. Scutellaria barbata and Hedyotis diffusa herb pair for breast cancer treatment: Potential mechanism based on network pharmacology. J. Ethnopharmacol. 2020, 259, 112929. [Google Scholar] [CrossRef]
- Gao, J.; Yin, W.; Corcoran, O. From Scutellaria barbata to BZL101 in cancer patients: Phytochemistry, pharmacology, and clinical evidence. Nat. Prod. Commun. 2019, 14, 1934578X19880645. [Google Scholar] [CrossRef]
- Chen, V.; Staub, R.E.; Fong, S.; Tagliaferri, M.; Cohen, I.; Shtivelman, E. Bezielle selectively targets mitochondria of cancer cells to inhibit glycolysis and OXPHOS. PLoS ONE 2012, 7, e30300. [Google Scholar] [CrossRef]
- Lang, X.; Chen, Z.; Yang, X.; Yan, Q.; Xu, M.; Liu, W.; He, Q.; Zhang, Y.; Cheng, W.; Zhao, W. Scutellarein induces apoptosis and inhibits proliferation, migration, and invasion in ovarian cancer via inhibition of EZH2/FOXO1 signaling. J. Biochem. Mol. Toxicol. 2021, 35, e22870. [Google Scholar] [CrossRef]
- Ha, S.E.; Kim, S.M.; Vetrivel, P.; Kim, H.H.; Bhosale, P.B.; Heo, J.D.; Lee, H.J.; Kim, G.S. Inhibition of cell proliferation and metastasis by scutellarein regulating PI3K/Akt/NF-κB signaling through PTEN activation in hepatocellular carcinoma. Int. J. Mol. Sci. 2021, 22, 8841. [Google Scholar] [CrossRef]
- Zeng, S.; Chen, L.; Sun, Q.; Zhao, H.; Yang, H.; Ren, S.; Liu, M.; Meng, X.; Xu, H. Scutellarin ameliorates colitis-associated colorectal cancer by suppressing Wnt/β-catenin signaling cascade. Eur. J. Pharmacol. 2021, 906, 174253. [Google Scholar] [CrossRef]
- Jiang, Q.; Li, Q.; Chen, H.; Shen, A.; Cai, Q.; Lin, J.; Peng, J. Scutellaria barbata D. Don inhibits growth and induces apoptosis by suppressing IL-6-inducible STAT3 pathway activation in human colorectal cancer cells. Exp. Ther. Med. 2015, 10, 1602–1608. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wei, L.-H.; Lin, J.-M.; Chu, J.-F.; Chen, H.-W.; Li, Q.-Y.; Peng, J. Scutellaria barbata D. Don inhibits colorectal cancer growth via suppression of Wnt/β-catenin signaling pathway. Chin. J. Integr. Med. 2017, 23, 858–863. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Feng, J.; Yang, H.; Yan, Z.; Li, Q.; Wei, L.; Lai, Z.; Jin, Y.; Peng, J. Scutellaria barbata D. Don inhibits 5-fluorouracil resistance in colorectal cancer by regulating PI3K/AKT pathway. Oncol. Rep. 2017, 38, 2293–2300. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Zhong, Z.; Tan, H.Y.; Guo, W.; Zhang, C.; Tan, C.-W.; Li, S.; Wang, N.; Feng, Y. Uncovering the anticancer mechanisms of Chin. herbal medicine formulas: Ther. alternatives for liver cancer. Front. Pharmacol. 2020, 11, 293. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Ma, Z.; Zhao, L.; Wang, W.; Gao, M.; Jia, X.; Ouyang, H.; He, J. Anti-tumor activities and mechanisms of Traditional Chinese medicines formulas: A review. Biomed. Pharmacother. 2020, 132, 110820. [Google Scholar] [CrossRef]
- Wang, K.; Chen, Q.; Shao, Y.; Yin, S.; Liu, C.; Liu, Y.; Wang, R.; Wang, T.; Qiu, Y.; Yu, H. Anticancer activities of TCM and their active components against tumor metastasis. Biomed. Pharmacother. 2021, 133, 111044. [Google Scholar] [CrossRef]
- Liu, S.-H.; Chen, P.-S.; Huang, C.-C.; Hung, Y.-T.; Lee, M.-Y.; Lin, W.-H.; Lin, Y.-C.; Lee, A.Y.-L. Unlocking the Mystery of the Therapeutic Effects of Chinese Medicine on Cancer. Front. Pharmacol. 2021, 2013, 601785. [Google Scholar] [CrossRef]
- Lan, H.-Y.; An, P.; Liu, Q.-P.; Chen, Y.-Y.; Yu, Y.-Y.; Luan, X.; Tang, J.-Y.; Zhang, H. Aidi injection induces apoptosis of hepatocellular carcinoma cells through the mitochondrial pathway. J. Ethnopharmacol. 2021, 274, 114073. [Google Scholar] [CrossRef]
- Shi, Q.; Diao, Y.; Jin, F.; Ding, Z. Anti-metastatic effects of Aidi on human esophageal squamous cell carcinoma by inhibiting epithelial-mesenchymal transition and angiogenesis. Mol. Med. Rep. 2018, 18, 131–138. [Google Scholar] [CrossRef]
- Yang, M.; Shen, C.; Zhu, S.-j.; Zhang, Y.; Jiang, H.-l.; Bao, Y.-d.; Yang, G.-y.; Liu, J.-p. Chin. patent medicine Aidi injection for cancer care: An overview of systematic reviews and meta-analyses. J. Ethnopharmacol. 2022, 282, 114656. [Google Scholar] [CrossRef]
- Zhao, Z.; Fan, H.; Higgins, T.; Qi, J.; Haines, D.; Trivett, A.; Oppenheim, J.J.; Wei, H.; Li, J.; Lin, H. Fufang Kushen injection inhibits sarcoma growth and tumor-induced hyperalgesia via TRPV1 signaling pathways. Cancer Lett. 2014, 355, 232–241. [Google Scholar] [CrossRef] [PubMed]
- Zhu, A.; Wang, F.; Fan, Q.; He, W.; Wang, L.; Zhao, P. Apoptosis and growth arrest of human esophageal squamous cell carcinoma cell EC9706 induced by Fufangkushen injection. Zhonghua Yi Xue Za Zhi 2011, 91, 2797–2800. [Google Scholar] [PubMed]
- Lu, Y.; Li, C.-S.; Dong, Q. Chin. herb related molecules of cancer-cell-apoptosis: A minireview of progress between Kanglaite injection and related genes. J. Exp. Clin. Cancer Res. 2008, 27, 31. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, W.; Wang, X.-J.; Liu, S. Antitumor effect of Kanglaite® injection in human pancreatic cancer xenografts. BMC Complement. Altern. Med. 2014, 14, 228. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Sun, M.; Yao, W.; Wang, F.; Li, X.; Wang, W.; Li, J.; Gao, Z.; Qiu, L.; You, R. Compound Kushen injection relieves tumor-associated macrophage-mediated immunosuppression through TNFR1 and sensitizes hepatocellular carcinoma to sorafenib. J. Immunother. Cancer 2020, 8, e000317. [Google Scholar] [CrossRef] [PubMed]
- Qi-Yue, Y.; Ting, Z.; Ya-Nan, H.; Sheng-Jie, H.; Xuan, D.; Li, H.; Chun-Guang, X. From natural dye to herbal medicine: A systematic review of chemical constituents, pharmacological effects and clinical applications of indigo naturalis. Chin. Med. 2020, 15, 127. [Google Scholar] [CrossRef]
- Yao, C.-J.; Chow, J.-M.; Yang, C.-M.; Kuo, H.-C.; Chang, C.-L.; Lee, H.-L.; Lai, I.; Chuang, S.-E.; Lai, G.-M. Chinese herbal mixture, Tien-Hsien liquid, induces G2/M cycle arrest and radiosensitivity in MCF-7 human breast cancer cells through mechanisms involving DNMT1 and Rad51 downregulation. Evid. Based Complement. Altern. Med. 2016, 2016, 3251046. [Google Scholar] [CrossRef]
- Yao, C.-J.; Yang, C.-M.; Chuang, S.-E.; Yan, J.-L.; Liu, C.-Y.; Chen, S.-W.; Yan, K.-H.; Lai, T.-Y.; Lai, G.-M. Targeting PML-RARα and oncogenic signaling pathways by Chin. herbal mixture Tien-Hsien liquid in acute promyelocytic leukemia NB4 cells. Evid. Based Complement. Altern. Med. 2011, 2011, 984154. [Google Scholar] [CrossRef]
- Yang, P.-M.; Du, J.-L.; Wang, G.N.-K.; Chia, J.-S.; Hsu, W.-B.; Pu, P.-C.; Sun, A.; Chiang, C.-P.; Wang, W.-B. The Chin. Herbal Mixture Tien-Hsien Liquid Augments the Anticancer Immunity in Tumor Cell–Vaccinated Mice. Integr. Cancer Ther. 2017, 16, 319–328. [Google Scholar] [CrossRef]
- Yin, T.; Yang, G.; Ma, Y.; Xu, B.; Hu, M.; You, M.; Gao, S. Developing an activity and absorption-based quality control platform for Chin. Tradit. medicine: Application to Zeng-Sheng-Ping (Antitumor B). J. Ethnopharmacol. 2015, 172, 195–201. [Google Scholar] [CrossRef]
- Wang, Y.; Yao, R.; Gao, S.; Wen, W.; Du, Y.; Szabo, E.; Hu, M.; Lubet, R.A.; You, M. Chemopreventive effect of a mixture of Chin. Herbs (antitumor B) on chemically induced oral carcinogenesis. Mol. Carcinogen. 2013, 52, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Lim, K.J.; Rajan, K.; Eberhart, C.G. Effects of Zeng Sheng Ping/ACAPHA on malignant brain tumor growth and notch signaling. Anticancer Res. 2012, 32, 2689–2696. [Google Scholar] [PubMed]
- Kuruppu, A.I.; Paranagama, P.; Goonasekara, C.L. Medicinal plants commonly used against cancer in Tradit. medicine formulae in Sri Lanka. Saudi Pharm. J. 2019, 27, 565–573. [Google Scholar]
- Sujatha, V. The Universal and the Global: Contextualising European Ayurvedic Practices. Soc. Cult. South Asia 2020, 6, 52–73. [Google Scholar] [CrossRef]
- Rosenberg, M. The European academy of Ayurveda: 20 years of Ayurvedic education in Germany. Anc. Sci. Life 2012, 32, 63. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, N. Is ayurveda the key to universal healthcare in India? BMJ 2015, 350, h2879. [Google Scholar] [CrossRef]
- Kumar, S.; Jawaid, T.; Dubey, S.D. Ther. plants of Ayurveda; a review on anticancer. Pharmacogn. J. 2011, 3, 1–11. [Google Scholar] [CrossRef]
- Palliyaguru, D.L.; Singh, S.V.; Kensler, T.W. Withania somnifera: From Prev. to treatment of cancer. Mol. Nutr. Food Res. 2016, 60, 1342–1353. [Google Scholar] [CrossRef]
- Singh, N.; Bhalla, M.; de Jager, P.; Gilca, M. An overview on ashwagandha: A Rasayana (rejuvenator) of Ayurveda. Afr. J. Tradit. Complement. Altern. Med. 2011, 8, 208–213. [Google Scholar] [CrossRef]
- Widodo, N.; Kaur, K.; Shrestha, B.G.; Takagi, Y.; Ishii, T.; Wadhwa, R.; Kaul, S.C. Selective killing of cancer cells by leaf extract of Ashwagandha: Identification of a tumor-inhibitory factor and the first molecular insights to its effect. Clin. Cancer Res. 2007, 13, 2298–2306. [Google Scholar] [CrossRef]
- Wadhwa, R.; Singh, R.; Gao, R.; Shah, N.; Widodo, N.; Nakamoto, T.; Ishida, Y.; Terao, K.; Kaul, S.C. Water extract of Ashwagandha leaves has anticancer activity: Identification of an active component and its mechanism of action. PLoS ONE 2013, 8, e77189. [Google Scholar] [CrossRef]
- Mehta, V.; Chander, H.; Munshi, A. Mechanisms of anti-tumor activity of Withania somnifera (Ashwagandha). Nutr. Cancer 2021, 73, 914–926. [Google Scholar] [CrossRef] [PubMed]
- Kataria, H.; Kumar, S.; Chaudhary, H.; Kaur, G. Withania somnifera suppresses tumor growth of intracranial allograft of glioma cells. Mol. Neurobiol. 2016, 53, 4143–4158. [Google Scholar] [CrossRef] [PubMed]
- Moselhy, J.; Suman, S.; Alghamdi, M.; Chandarasekharan, B.; Das, T.P.; Houda, A.; Ankem, M.; Damodaran, C. Withaferin A inhibits prostate carcinogenesis in a PTEN-deficient mouse model of prostate cancer. Neoplasia 2017, 19, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.-C.; Choi, B.Y. Withaferin-A—A natural anticancer agent with pleitropic mechanisms of action. Int. J. Mol. Sci. 2016, 17, 290. [Google Scholar] [CrossRef] [PubMed]
- Sultana, T.; Okla, M.K.; Ahmed, M.; Akhtar, N.; Al-Hashimi, A.; Abdelgawad, H.; Haq, I.-U. Withaferin A: From Ancient Remedy to Potential Drug Candidate. Molecules 2021, 26, 7696. [Google Scholar] [CrossRef]
- Kyakulaga, A.H.; Aqil, F.; Munagala, R.; Gupta, R.C. Withaferin a inhibits epithelial to mesenchymal transition in non-small cell lung cancer cells. Sci. Rep. 2018, 8, 15737. [Google Scholar] [CrossRef]
- Hahm, E.-R.; Lee, J.; Kim, S.-H.; Sehrawat, A.; Arlotti, J.A.; Shiva, S.S.; Bhargava, R.; Singh, S.V. Metabolic alterations in mammary cancer Prev. by withaferin A in a clinically relevant mouse model. J. Nat. Cancer Inst. 2013, 105, 1111–1122. [Google Scholar] [CrossRef]
- Kakar, S.S.; Parte, S.; Kelsey Carter, I.G.J.; Worth, C.; Rameshwar, P.; Ratajczak, M.Z. Withaferin A (WFA) inhibits tumor growth and metastasis by targeting ovarian cancer stem cells. Oncotarget 2017, 8, 74494. [Google Scholar] [CrossRef]
- Kim, S.-H.; Singh, S.V. Mammary cancer chemoPrev. by withaferin A is accompanied by in vivo suppression of self-renewal of cancer stem cells. Cancer Prev. Res. 2014, 7, 738–747. [Google Scholar] [CrossRef]
- Nelson, K.M.; Dahlin, J.L.; Bisson, J.; Graham, J.; Pauli, G.F.; Walters, M.A. The essential medicinal chemistry of curcumin: Miniperspective. J. Med. Chem. 2017, 60, 1620–1637. [Google Scholar] [CrossRef] [PubMed]
- Dosoky, N.S.; Setzer, W.N. Chemical composition and biological activities of essential oils of Curcuma species. Nutrients 2018, 10, 1196. [Google Scholar] [CrossRef] [PubMed]
- Sandur, S.K.; Pandey, M.K.; Sung, B.; Ahn, K.S.; Murakami, A.; Sethi, G.; Limtrakul, P.; Badmaev, V.; Aggarwal, B.B. Curcumin, demethoxycurcumin, bisdemethoxycurcumin, tetrahydrocurcumin and turmerones differentially regulate anti-inflammatory and anti-proliferative responses through a ROS-independent mechanism. Carcinogenesis 2007, 28, 1765–1773. [Google Scholar] [CrossRef] [PubMed]
- Giordano, A.; Tommonaro, G. Curcumin and cancer. Nutrients 2019, 11, 2376. [Google Scholar] [CrossRef] [PubMed]
- Sultana, S.; Munir, N.; Mahmood, Z.; Riaz, M.; Akram, M.; Rebezov, M.; Kuderinova, N.; Moldabayeva, Z.; Shariati, M.A.; Rauf, A. Molecular targets for the Manag. of cancer using Curcuma longa Linn. phytoconstituents: A Review. Biomed. Pharmacother. 2021, 135, 111078. [Google Scholar] [CrossRef]
- Lev-Ari, S.; Starr, A.; Vexler, A.; Karaush, V.; Loew, V.; Greif, J.; Fenig, E.; Aderka, D.; Ben-Yosef, R. Inhibition of pancreatic and lung adenocarcinoma cell survival by curcumin is associated with increased apoptosis, down-regulation of COX-2 and EGFR and inhibition of Erk1/2 activity. Anticancer Res. 2006, 26, 4423–4430. [Google Scholar]
- Zhao, G.; Han, X.; Zheng, S.; Li, Z.; Sha, Y.; Ni, J.; Sun, Z.; Qiao, S.; Song, Z. Curcumin induces autophagy, inhibits proliferation and invasion by downregulating AKT/mTOR signaling pathway in human melanoma cells. Oncol. Rep. 2016, 35, 1065–1074. [Google Scholar] [CrossRef]
- Li, W.; Zhou, Y.; Yang, J.; Li, H.; Zhang, H.; Zheng, P. Curcumin induces apoptotic cell death and protective autophagy in human gastric cancer cells. Oncol. Rep. 2017, 37, 3459–3466. [Google Scholar] [CrossRef]
- Zhuang, W.; Long, L.; Zheng, B.; Ji, W.; Yang, N.; Zhang, Q.; Liang, Z. Curcumin promotes differentiation of glioma-initiating cells by inducing autophagy. Cancer Sci. 2012, 103, 684–690. [Google Scholar] [CrossRef]
- Almanaa, T.N.; Geusz, M.E.; Jamasbi, R.J. Effects of curcumin on stem-like cells in human esophageal squamous carcinoma cell lines. BMC Complement. Altern. Med. 2012, 12, 195. [Google Scholar] [CrossRef]
- Wang, J.; Wang, C.; Bu, G. Curcumin inhibits the growth of liver cancer stem cells through the phosphatidylinositol 3-kinase/protein kinase B/mammalian target of rapamycin signaling pathway. Exp. Ther. Med. 2018, 15, 3650–3658. [Google Scholar] [CrossRef] [PubMed]
- Yallapu, M.M.; Maher, D.M.; Sundram, V.; Bell, M.C.; Jaggi, M.; Chauhan, S.C. Curcumin induces chemo/radio-sensitization in ovarian cancer cells and curcumin nanoparticles inhibit ovarian cancer cell growth. J. Ovar. Res. 2010, 3, 11. [Google Scholar] [CrossRef]
- Zhang, P.; Lai, Z.-L.; Chen, H.-F.; Zhang, M.; Wang, A.; Jia, T.; Sun, W.-Q.; Zhu, X.-M.; Chen, X.-F.; Zhao, Z. Curcumin synergizes with 5-fluorouracil by impairing AMPK/ULK1-dependent autophagy, AKT activity and enhancing apoptosis in colon cancer cells with tumor growth inhibition in xenograft mice. J. Exp. Clin. Cancer Res. 2017, 36, 190. [Google Scholar] [CrossRef] [PubMed]
- Kong, W.-Y.; Ngai, S.C.; Goh, B.-H.; Lee, L.-H.; Htar, T.-T.; Chuah, L.-H. Is curcumin the answer to future chemotherapy cocktail? Molecules 2021, 26, 4329. [Google Scholar] [CrossRef]
- Farghadani, R.; Naidu, R. Curcumin as an Enhancer of Ther. Efficiency of Chemotherapy Drugs in Breast Cancer. Int. J. Mol. Sci. 2022, 23, 2144. [Google Scholar] [CrossRef]
- Hsu, F.-T.; Liu, Y.-C.; Liu, T.-T.; Hwang, J.-J. Curcumin sensitizes hepatocellular carcinoma cells to radiation via suppression of radiation-induced NF-κB activity. Biomed Res. Int. 2015, 2015, 363671. [Google Scholar] [CrossRef]
- Zoi, V.; Galani, V.; Tsekeris, P.; Kyritsis, A.P.; Alexiou, G.A. Radiosensitization and Radioprotection by Curcumin in Glioblastoma and Other Cancers. Biomedicines 2022, 10, 312. [Google Scholar] [CrossRef]
- Prasad, S.; Tyagi, A.K. Ginger and its constituents: Role in Prev. and treatment of gastrointestinal cancer. Gastroenterol. Res. Pract. 2015. [Google Scholar] [CrossRef]
- Mao, Q.-Q.; Xu, X.-Y.; Cao, S.-Y.; Gan, R.-Y.; Corke, H.; Beta, T.; Li, H.-B. Bioactive compounds and bioactivities of ginger (Zingiber officinale Roscoe). Foods 2019, 8, 185. [Google Scholar] [CrossRef]
- de Lima, R.M.T.; Dos Reis, A.C.; de Menezes, A.A.P.M.; Santos, J.V.d.O.; Filho, J.W.G.d.O.; Ferreira, J.R.d.O.; de Alencar, M.V.O.B.; da Mata, A.M.O.F.; Khan, I.N.; Islam, A. Protective and therapeutic potential of ginger (Zingiber officinale) extract and [6]-gingerol in cancer: A comprehensive review. Phytother. Res. 2018, 32, 1885–1907. [Google Scholar] [CrossRef]
- Habib, S.H.M.; Makpol, S.; Hamid, N.A.A.; Das, S.; Ngah, W.Z.W.; Yusof, Y.A.M. Ginger extract (Zingiber officinale) has anti-cancer and anti-inflammatory effects on ethionine-induced hepatoma rats. Clinics 2008, 63, 807–813. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.M.; Kim, C.; Bae, H.; Lee, J.H.; Baek, S.H.; Nam, D.; Chung, W.S.; Shim, B.S.; Lee, S.G.; Kim, S.H. 6-Shogaol exerts anti-proliferative and pro-apoptotic effects through the modulation of STAT3 and MAPKs signaling pathways. Mol. Carcinog. 2015, 54, 1132–1146. [Google Scholar] [CrossRef] [PubMed]
- Saha, A.; Blando, J.; Silver, E.; Beltran, L.; Sessler, J.; DiGiovanni, J. 6-Shogaol from dried ginger inhibits growth of prostate cancer cells both in vitro and in vivo through inhibition of STAT3 and NF-κB signaling. Cancer Prev. Res. 2014, 7, 627–638. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-J.; Jeon, Y.; Kim, T.; Lim, W.-C.; Ham, J.; Park, Y.N.; Kim, T.-J.; Ko, H. Combined treatment with zingerone and its novel derivative synergistically inhibits TGF-β1 induced epithelial-mesenchymal transition, migration and invasion of human hepatocellular carcinoma cells. Bioorg. Med. Chem. Lett. 2017, 27, 1081–1088. [Google Scholar] [CrossRef]
- Ray, A.; Vasudevan, S.; Sengupta, S. 6-Shogaol inhibits breast cancer cells and stem cell-like spheroids by modulation of Notch signaling pathway and induction of autophagic cell death. PLoS ONE 2015, 10, e0137614. [Google Scholar] [CrossRef]
- El-Ashmawy, N.E.; Khedr, N.F.; El-Bahrawy, H.A.; Abo Mansour, H.E. Ginger extract adjuvant to doxorubicin in mammary carcinoma: Study of some molecular mechanisms. Eur. J. Nutr. 2018, 57, 981–989. [Google Scholar] [CrossRef]
- Rahimi Babasheikhali, S.; Rahgozar, S.; Mohammadi, M. Ginger extract has anti-leukemia and anti-drug resistant effects on malignant cells. J. Cancer Res. Clin. Oncol. 2019, 145, 1987–1998. [Google Scholar] [CrossRef]
- Liu, C.-M.; Kao, C.-L.; Tseng, Y.-T.; Lo, Y.-C.; Chen, C.-Y. Ginger phytochemicals inhibit cell growth and modulate drug resistance factors in docetaxel resistant prostate cancer cell. Molecules 2017, 22, 1477. [Google Scholar] [CrossRef]
- Almatroudi, A.; Alsahli, M.A.; Alrumaihi, F.; Allemailem, K.S.; Rahmani, A.H. Ginger: A novel strategy to battle cancer through modulating cell signalling pathways: A review. Curr. Pharm. Biotechnol. 2019, 20, 5–16. [Google Scholar] [CrossRef]
- Zadorozhna, M.; Mangieri, D. Mechanisms of chemopreventive and Ther. proprieties of ginger extracts in cancer. Int. J. Mol. Sci. 2021, 22, 6599. [Google Scholar] [CrossRef]
- Iram, F.; Khan, S.A.; Husain, A. Phytochemistry and potential Ther. actions of Boswellic acids: A mini-review. Asian Pac. J. Trop. Biomed. 2017, 7, 513–523. [Google Scholar] [CrossRef]
- Roy, N.K.; Parama, D.; Banik, K.; Bordoloi, D.; Devi, A.K.; Thakur, K.K.; Padmavathi, G.; Shakibaei, M.; Fan, L.; Sethi, G. An update on pharmacological potential of boswellic acids against chronic diseases. Int. J. Mol. Sci. 2019, 20, 4101. [Google Scholar] [CrossRef]
- Gupta, M.; Rout, P.; Misra, L.; Gupta, P.; Singh, N.; Darokar, M.; Saikia, D.; Singh, S.; Bhakuni, R. Chemical composition and bioactivity of Boswellia serrata Roxb. essential oil in relation to geographical variation. Plant Biosyst. Int. J. All Aspects Plant Biol. 2017, 151, 623–629. [Google Scholar]
- Ayub, M.A.; Hanif, M.A.; Blanchfield, J.; Zubair, M.; Abid, M.A.; Saleh, M.T. Chemical composition and antimicrobial activity of Boswellia serrata oleo-gum-resin essential oil extracted by superheated steam. Nat. Prod. Res. 2022; in press. [Google Scholar]
- Khajehdehi, M.; Khalaj-Kondori, M.; Baradaran, B. Molecular evidences on anti-inflammatory, anticancer, and memory-boosting effects of frankincense. Phytother. Res. 2022, 36, 1194–1215. [Google Scholar] [CrossRef]
- Ahmed, H.H.; Abd-Rabou, A.A.; Hassan, A.Z.; Kotob, S.E. Phytochemical analysis and anti-cancer investigation of Boswellia serrata bioactive constituents in vitro. Asian Pac. J. Cancer Prev. 2015, 16, 7179–7188. [Google Scholar] [CrossRef]
- Hakkim, F.L.; Bakshi, H.A.; Khan, S.; Nasef, M.; Farzand, R.; Sam, S.; Rashan, L.; Al-Baloshi, M.S.; Hasson, S.S.A.A.; Al Jabri, A. Frankincense essential oil suppresses melanoma cancer through down regulation of Bcl-2/Bax cascade signaling and ameliorates heptotoxicity via phase I and II drug metabolizing enzymes. Oncotarget 2019, 10, 3472. [Google Scholar] [CrossRef] [PubMed]
- Takada, Y.; Ichikawa, H.; Badmaev, V.; Aggarwal, B.B. Acetyl-11-keto-β-boswellic acid potentiates apoptosis, inhibits invasion, and abolishes osteoclastogenesis by suppressing NF-κB and NF-κB-regulated gene expression. J. Immunol. 2006, 176, 3127–3140. [Google Scholar] [CrossRef]
- Kunnumakkara, A.B.; Nair, A.S.; Sung, B.; Pandey, M.K.; Aggarwal, B.B. Boswellic acid blocks STAT3 Signaling, proliferation, and survival of multiple myeloma via the protein tyrosine phosphatase SHP-1. Mol. Cancer Res. MCR 2009, 7, 118. [Google Scholar] [CrossRef]
- Shen, Y.; Takahashi, M.; Byun, H.-M.; Link, A.; Sharma, N.; Balaguer, F.; Leung, H.-C.; Boland, C.R.; Goel, A. Boswellic acid induces epigenetic alterations by modulating DNA methylation in colorectal cancer cells. Cancer Biol. Ther. 2012, 13, 542–552. [Google Scholar] [CrossRef]
- Mazzio, E.A.; Lewis, C.A.; Soliman, K.F. Transcriptomic Profiling of MDA-MB-231 cells exposed to Boswellia Serrata and 3-O-Acetyl-B-Boswellic Acid; ER/UPR mediated programmed cell death. Cancer Genom. Proteom. 2017, 14, 409–425. [Google Scholar]
- Hussain, H.; Ali, I.; Wang, D.; Hakkim, F.L.; Westermann, B.; Rashan, L.; Ahmed, I.; Green, I.R. Boswellic acids: Privileged structures to develop lead compounds for anticancer drug discovery. Expert Opin. Drug Discov. 2021, 16, 851–867. [Google Scholar] [CrossRef] [PubMed]
- Hussain, H.; Al-Harrasi, A.; Csuk, R.; Shamraiz, U.; Green, I.R.; Ahmed, I.; Khan, I.A.; Ali, Z. Therapeutic potential of boswellic acids: A patent review (1990–2015). Expert Opin. Ther. Pat. 2017, 27, 81–90. [Google Scholar] [CrossRef]
- Takeda, S.; Noguchi, M.; Matsuo, K.; Yamaguchi, Y.; Kudo, T.; Nishimura, H.; Okamoto, Y.; Amamoto, T.; Shindo, M.; Omiecinski, C.J. (−)-Xanthatin up-regulation of the GADD45γ tumor suppressor gene in MDA-MB-231 breast cancer cells: Role of topoisomerase IIα inhibition and reactive oxygen species. Toxicology 2013, 305, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Yu, J.; Pei, C.G.; Li, Y.Y.; Tu, P.; Gao, G.P.; Shao, Y. Xanthatin, a novel potent inhibitor of VEGFR2 signaling, inhibits angiogenesis and tumor growth in breast cancer cells. Int. J. Clin. Exp. Pathol. 2015, 8, 10355. [Google Scholar] [PubMed]
- Zhang, L.; Ruan, J.; Yan, L.; Li, W.; Wu, Y.; Tao, L.; Zhang, F.; Zheng, S.; Wang, A.; Lu, Y. Xanthatin induces cell cycle arrest at G2/M checkpoint and apoptosis via disrupting NF-κB pathway in A549 non-small-cell lung cancer cells. Molecules 2012, 17, 3736–3750. [Google Scholar] [CrossRef]
- Tao, L.; Fan, F.; Liu, Y.; Li, W.; Zhang, L.; Ruan, J.; Shen, C.; Sheng, X.; Zhu, Z.; Wang, A. Concerted suppression of STAT3 and GSK3β is involved in growth inhibition of non-small cell lung cancer by xanthatin. PLoS ONE 2013, 8, e81945. [Google Scholar] [CrossRef]
- Tao, L.; Sheng, X.; Zhang, L.; Li, W.; Wei, Z.; Zhu, P.; Zhang, F.; Wang, A.; Woodgett, J.R.; Lu, Y. Xanthatin anti-tumor cytotoxicity is mediated via glycogen synthase kinase-3β and β-catenin. Biochem. Pharmacol. 2016, 115, 18–27. [Google Scholar] [CrossRef]
- Yang, J.; Li, Y.; Zong, C.; Zhang, Q.; Ge, S.; Ma, L.; Fan, J.; Zhang, J.; Jia, R. Xanthatin Selectively Targets Retinoblastoma by Inhibiting the PLK1-Mediated Cell Cycle. Investig. Ophthalmol. Visual Sci. 2021, 62, 11. [Google Scholar] [CrossRef]
- Geng, Y.-d.; Zhang, L.; Wang, G.-Y.; Feng, X.-J.; Chen, Z.-L.; Jiang, L.; Shen, A.-Z. Xanthatin mediates G2/M cell cycle arrest, autophagy and apoptosis via ROS/XIAP signaling in human colon cancer cells. Nat. Prod. Res. 2020, 34, 2616–2620. [Google Scholar] [CrossRef]
- Li, L.; Liu, P.; Xie, Y.; Liu, Y.; Chen, Z.; Geng, Y.; Zhang, L. Xanthatin inhibits human colon cancer cells progression via mTOR signaling mediated energy metabolism alteration. Drug Dev. Res. 2022, 83, 119–130. [Google Scholar] [CrossRef]
- Ma, Y.-Y.; Di, Z.-M.; Cao, Q.; Xu, W.-S.; Bi, S.-X.; Yu, J.-S.; Shen, Y.-J.; Yu, Y.-Q.; Shen, Y.-X.; Feng, L.-J. Xanthatin induces glioma cell apoptosis and inhibits tumor growth via activating endoplasmic reticulum stress-dependent CHOP pathway. Acta Pharmacol. Sin. 2020, 41, 404–414. [Google Scholar] [CrossRef]
- Hashim, Y.; Latimer, C.; Ternan, N.; Abbas, P. Studies of Malaysian plants in prevention and treatment of colorectal cancer. In Colorectal Cancer; InTechOpen: London, UK, 2016; Volume 18, pp. 377–394. [Google Scholar]
- Mazumder, K.; Biswas, B.; Raja, I.M.; Fukase, K. A review of cytotoxic plants of the Indian subcontinent and a broad-spectrum analysis of their bioactive compounds. Molecules 2020, 25, 1904. [Google Scholar] [CrossRef] [PubMed]
- Meiyanto, E.; Larasati, Y.A. The chemopreventive activity of Indonesia medicinal plants targeting on hallmarks of cancer. Adv. Pharm. Bull. 2019, 9, 219. [Google Scholar] [CrossRef] [PubMed]
- Sithisarn, P.; Rojsanga, P. Anticancer Effects of Some Medicinal Thai Plants. In Natural Products and Cancer Drug Discovery; IntechOpen: London, UK, 2017. [Google Scholar]
- Nguyen, N.H.; Ta, Q.T.H.; Pham, Q.T.; Luong, T.N.H.; Phung, V.T.; Duong, T.-H.; Vo, V.G. Anticancer activity of novel plant extracts and compounds from Adenosma bracteosum (Bonati) in human lung and liver cancer cells. Molecules 2020, 25, 2912. [Google Scholar] [CrossRef] [PubMed]
- Shuvalov, O.; Fedorova, O.; Tananykina, E.; Gnennaya, Y.; Daks, A.; Petukhov, A.; Barlev, N. An arthropod hormone, ecdysterone, inhibits the growth of breast cancer cells via different mechanisms. Front. Pharmacol. 2020, 11, 561537. [Google Scholar] [CrossRef]
- Zirak, N.; Shafiee, M.; Soltani, G.; Mirzaei, M.; Sahebkar, A. Hypericum perforatum in the treatment of psychiatric and neurodegenerative disorders: Current evidence and potential mechanisms of action. J. Cell. Physiol. 2019, 234, 8496–8508. [Google Scholar] [CrossRef]
- Menegazzi, M.; Masiello, P.; Novelli, M. Anti-tumor activity of Hypericum perforatum L. and hyperforin through modulation of inflammatory signaling, ROS generation and proton dynamics. Antioxidants 2020, 10, 18. [Google Scholar] [CrossRef]
- Napoli, E.; Siracusa, L.; Ruberto, G.; Carrubba, A.; Lazzara, S.; Speciale, A.; Cimino, F.; Saija, A.; Cristani, M. Phytochemical profiles, phototoxic and antioxidant properties of eleven Hypericum species–A comparative study. Phytochem. 2018, 152, 162–173. [Google Scholar] [CrossRef]
- Alper, M.; Güneş, H. The anticancer and anti-inflammatory effects of Centaurea solstitialis extract on human cancer cell lines. Turk. J. Pharm. Sci. 2019, 16, 273. [Google Scholar] [CrossRef]
- Koeberle, A.; Rossi, A.; Bauer, J.; Dehm, F.; Verotta, L.; Northoff, H.; Sautebin, L.; Werz, O. Hyperforin, an anti-inflammatory constituent from St. John’s wort, inhibits microsomal prostaglandin E2 synthase-1 and suppresses prostaglandin E2 formation in vivo. Front. Pharmacol. 2011, 2, 7. [Google Scholar] [CrossRef]
- Benedí, J.; Arroyo, R.; Romero, C.; Martín-Aragón, S.; Villar, A.M. Antioxidant properties and protective effects of a standardized extract of Hypericum perforatum on hydrogen peroxide-induced oxidative damage in PC12 cells. Life Sci. 2004, 75, 1263–1276. [Google Scholar] [CrossRef] [PubMed]
- Imreova, P.; Feruszova, J.; Kyzek, S.; Bodnarova, K.; Zduriencikova, M.; Kozics, K.; Mucaji, P.; Galova, E.; Sevcovicova, A.; Miadokova, E. Hyperforin exhibits antigenotoxic activity on human and bacterial cells. Molecules 2017, 22, 167. [Google Scholar] [CrossRef] [PubMed]
- Hsu, F.T.; Chen, W.T.; Wu, C.T.; Chung, J.G. Hyperforin induces apoptosis through extrinsic/intrinsic pathways and inhibits EGFR/ERK/NF-κB-mediated anti-apoptotic potential in glioblastoma. Environ. Toxicol. 2020, 35, 1058–1069. [Google Scholar] [CrossRef] [PubMed]
- Merhi, F.; Tang, R.; Piedfer, M.; Mathieu, J.; Bombarda, I.; Zaher, M.; Kolb, J.-P.; Billard, C.; Bauvois, B. Hyperforin inhibits Akt1 kinase activity and promotes caspase-mediated apoptosis involving Bad and Noxa activation in human myeloid tumor cells. PLoS ONE 2011, 6, e25963. [Google Scholar] [CrossRef]
- Quiney, C.; Billard, C.; Faussat, A.; Salanoubat, C.; Ensaf, A.; Nait-Si, Y.; Fourneron, J.; Kolb, J. Pro-apoptotic properties of hyperforin in leukemic cells from patients with B-cell chronic lymphocytic leukemia. Leukemia 2006, 20, 491–497. [Google Scholar] [CrossRef]
- Lorusso, G.; Vannini, N.; Sogno, I.; Generoso, L.; Garbisa, S.; Noonan, D.M.; Albini, A. Mechanisms of Hyperforin as an anti-angiogenic angioPrev. agent. Eur. J. Cancer 2009, 45, 1474–1484. [Google Scholar] [CrossRef]
- Donà, M.; Dell’Aica, I.; Pezzato, E.; Sartor, L.; Calabrese, F.; Della Barbera, M.; Donella-Deana, A.; Appendino, G.; Borsarini, A.; Caniato, R. Hyperforin inhibits cancer invasion and metastasis. Cancer Res. 2004, 64, 6225–6232. [Google Scholar] [CrossRef]
- Sell, T.S.; Belkacemi, T.; Flockerzi, V.; Beck, A. Protonophore properties of hyperforin are essential for its pharmacological activity. Sci. Rep. 2014, 4, 7500. [Google Scholar] [CrossRef]
- Scotti, F.; Löbel, K.; Booker, A.; Heinrich, M. St. John’s Wort (Hypericum perforatum) products–How variable is the primary material? Front. Plant Sci. 2019, 9, 1973. [Google Scholar] [CrossRef]
- Fulda, S. Betulinic acid for cancer treatment and prevention. Int. J. Mol. Sci. 2008, 9, 1096–1107. [Google Scholar] [CrossRef]
- Zhang, X.; Hu, J.; Chen, Y. Betulinic acid and the pharmacological effects of tumor suppression. Mol. Med. Rep. 2016, 14, 4489–4495. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.; Yu, R.; Pezzuto, J.M. Betulinic acid-induced programmed cell death in human melanoma cells involves mitogen-activated protein kinase activation. Clin. Cancer Res. 2003, 9, 2866–2875. [Google Scholar] [PubMed]
- Guo, Y.; Zhu, H.; Weng, M.; Wang, C.; Sun, L. Chemopreventive effect of Betulinic acid via mTOR-Caspases/Bcl2/Bax apoptotic signaling in pancreatic cancer. BMC Complement. Med. Ther. 2020, 20, 178. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Wang, K.; Zhang, C.; Zhang, W.; Xu, Q.; Wang, Y.; Zhang, Y.; Li, Y.; Zhang, Y.; Zhu, H. Overaccumulation of p53-mediated autophagy protects against betulinic acid-induced apoptotic cell death in colorectal cancer cells. Cell Death Dis. 2017, 8, e3087. [Google Scholar] [CrossRef]
- Potze, L.; Mullauer, F.; Colak, S.; Kessler, J.; Medema, J. Betulinic acid-induced mitochondria-dependent cell death is counterbalanced by an autophagic salvage response. Cell Death Dis. 2014, 5, e1169. [Google Scholar] [CrossRef]
- Zheng, Y.; Liu, P.; Wang, N.; Wang, S.; Yang, B.; Li, M.; Chen, J.; Situ, H.; Xie, M.; Lin, Y. Betulinic acid suppresses breast cancer metastasis by targeting GRP78-mediated glycolysis and ER stress apoptotic pathway. Oxid. Med. Cell. Longev. 2019, 2019, 8781690. [Google Scholar] [CrossRef]
- Saeed, M.E.; Mahmoud, N.; Sugimoto, Y.; Efferth, T.; Abdel-Aziz, H. Betulinic acid exerts cytotoxic activity against multidrug-resistant tumor cells via targeting autocrine motility factor receptor (AMFR). Front. Pharmacol. 2018, 9, 481. [Google Scholar] [CrossRef]
- Nedopekina, D.A.; Gubaidullin, R.R.; Odinokov, V.N.; Maximchik, P.V.; Zhivotovsky, B.; Bel’skii, Y.P.; Khazanov, V.A.; Manuylova, A.V.; Gogvadze, V.; Spivak, A.Y. Mitochondria-targeted betulinic and ursolic acid derivatives: Synthesis and anticancer activity. MedChemComm 2017, 8, 1934–1945. [Google Scholar] [CrossRef]
- Sharma, V.; Katiyar, A.; Agrawal, R. Glycyrrhiza glabra: Chemistry and pharmacological activity. Sweeteners 2018, 87–100. [Google Scholar]
- Wang, K.-L.; Yu, Y.-C.; Hsia, S.-M. Perspectives on the Role of Isoliquiritigenin in Cancer. Cancers 2021, 13, 115. [Google Scholar] [CrossRef]
- Tian, T.; Sun, J.; Wang, J.; Liu, Y.; Liu, H. Isoliquiritigenin inhibits cell proliferation and migration through the PI3K/AKT signaling pathway in A549 lung cancer cells. Oncol. Lett. 2018, 16, 6133–6139. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Shenoy, A.K.; Padia, R.; Fang, D.; Jing, Q.; Yang, P.; Su, S.-B.; Huang, S. Suppression of lung cancer progression by isoliquiritigenin through its metabolite 2, 4, 2’, 4’-Tetrahydroxychalcone. J. Exp. Clin. Cancer Res. 2018, 37, 243. [Google Scholar] [CrossRef] [PubMed]
- Bolós, V.; Gasent, J.M.; López-Tarruella, S.; Grande, E. The dual kinase complex FAK-Src as a promising therapeutic target in cancer. Onco Targets Ther. 2010, 3, 83. [Google Scholar] [CrossRef] [PubMed]
- Nag, S.; Qin, J.; Srivenugopal, K.S.; Wang, M.; Zhang, R. The MDM2-p53 pathway revisited. J. Biomed. Res. 2013, 27, 254. [Google Scholar]
- Bohlman, S.; Manfredi, J.J. p53-independent effects of Mdm2. Mutant p53 and MDM2 in Cancer. Subcell Biochem. 2014, 85, 235–246. [Google Scholar]
- Shuvalov, O.; Kizenko, A.; Shakirova, A.; Fedorova, O.; Petukhov, A.; Aksenov, N.; Vasileva, E.; Daks, A.; Barlev, N. Nutlin sensitizes lung carcinoma cells to interferon-alpha treatment in MDM2-dependent but p53-independent manner. Biochem. Biophys. Res. Commun. 2018, 495, 1233–1239. [Google Scholar] [CrossRef]
- Xiang, S.; Chen, H.; Luo, X.; An, B.; Wu, W.; Cao, S.; Ruan, S.; Wang, Z.; Weng, L.; Zhu, H. Isoliquiritigenin suppresses human melanoma growth by targeting miR-301b/LRIG1 signaling. J. Exp. Clin. Cancer Res. 2018, 37, 184. [Google Scholar] [CrossRef]
- Kroll, D.J.; Shaw, H.S.; Oberlies, N.H. Milk thistle nomenclature: Why it matters in cancer research and pharmacokinetic studies. Integr. Cancer Ther. 2007, 6, 110–119. [Google Scholar] [CrossRef]
- Karimi, G.; Vahabzadeh, M.; Lari, P.; Rashedinia, M.; Moshiri, M. “Silymarin”, a promising pharmacological agent for treatment of diseases. Iran. J. Basic Med. Sci. 2011, 14, 308. [Google Scholar]
- Won, D.-H.; Kim, L.-H.; Jang, B.; Yang, I.-H.; Kwon, H.-J.; Jin, B.; Oh, S.H.; Kang, J.-H.; Hong, S.-D.; Shin, J.-A. In vitro and in vivo anti-cancer activity of silymarin on oral cancer. Tumor Biol. 2018, 40, 1010428318776170. [Google Scholar] [CrossRef]
- Eo, H.J.; Park, G.H.; Jeong, J.B. Inhibition of Wnt signaling by silymarin in human colorectal cancer cells. Biomol. Ther. 2016, 24, 380. [Google Scholar] [CrossRef] [PubMed]
- Delmas, D.; Xiao, J.; Vejux, A.; Aires, V. Silymarin and Cancer: A Dual Strategy in Both in Chemoprevention and Chemosensitivity. Molecules 2020, 25, 2009. [Google Scholar] [CrossRef] [PubMed]
- Ramasamy, K.; Agarwal, R. Multitargeted therapy of cancer by silymarin. Cancer Lett. 2008, 269, 352–362. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Choo, G.S.; Yoo, E.S.; Woo, J.S.; Han, S.H.; Lee, J.H.; Jung, J.Y. Silymarin induces inhibition of growth and apoptosis through modulation of the MAPK signaling pathway in AGS human gastric cancer cells. Oncol. Rep. 2019, 42, 1904–1914. [Google Scholar] [CrossRef] [PubMed]
- Mao, J.; Yang, H.; Cui, T.; Pan, P.; Kabir, N.; Chen, D.; Ma, J.; Chen, X.; Chen, Y.; Yang, Y. Combined treatment with sorafenib and silibinin synergistically targets both HCC cells and cancer stem cells by enhanced inhibition of the phosphorylation of STAT3/ERK/AKT. Eur. J. Pharmacol. 2018, 832, 39–49. [Google Scholar] [CrossRef]
- Soleimani, V.; Delghandi, P.S.; Moallem, S.A.; Karimi, G. Safety and toxicity of silymarin, the major constituent of milk thistle extract: An updated review. Phytother. Res. 2019, 33, 1627–1638. [Google Scholar] [CrossRef]
- Shang, A.; Cao, S.-Y.; Xu, X.-Y.; Gan, R.-Y.; Tang, G.-Y.; Corke, H.; Mavumengwana, V.; Li, H.-B. Bioactive compounds and biological functions of garlic (Allium sativum L.). Foods 2019, 8, 246. [Google Scholar] [CrossRef]
- Taleb Agha, M.; Baharetha, H.M.; Al-Mansoub, M.A.; Tabana, Y.M.; Kaz Abdul Aziz, N.H.; Yam, M.F.; Abdul Majid, A.M.S. Proapoptotic and antiangiogenic activities of Arctium lappa L. on breast cancer cell lines. Scientifica 2020, 2020. [Google Scholar] [CrossRef]
- Li, X.; Lin, Y.-Y.; Tan, J.-Y.; Liu, K.-L.; Shen, X.-L.; Hu, Y.-J.; Yang, R.-Y. Lappaol F, an anticancer agent, inhibits YAP via transcriptional and post-translational regulation. Pharm. Biol. 2021, 59, 619–628. [Google Scholar] [CrossRef]
- He, Y.; Fan, Q.; Cai, T.; Huang, W.; Xie, X.; Wen, Y.; Shi, Z. Molecular mechanisms of the action of Arctigenin in cancer. Biomed. Pharmacother. 2018, 108, 403–407. [Google Scholar] [CrossRef]
- Erenler, R.; Sen, O.; Sahin Yaglioglu, A.; Demirtas, I. Bioactivity-guided isolation of antiproliferative sesquiterpene lactones from Centaurea solstitialis L. ssp. solstitialis. Comb. Chem. High Throughput Screen. 2016, 19, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Simsek, E.; Imir, N.; Aydemir, E.A.; Gokturk, R.S.; Yesilada, E.; Fiskin, K. Caspase-mediated apoptotic effects of Ebenus boissieri barbey extracts on human cervical cancer cell line hela. Pharmacogn. Mag. 2017, 13, 254. [Google Scholar] [CrossRef] [PubMed]
- Aydemir, E.A.; Simsek, E.; Imir, N.; Göktürk, R.S.; Yesilada, E.; Fiskin, K. Cytotoxic and apoptotic effects of Ebenus boissieri Barbey on human lung cancer cell line A549. Pharmacogn. Mag. 2015, 11, S37. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Imir, N.; Aydemir, E.; Şimşek, E.; Göktürk, R.; Yesilada, E.; Fişkin, K. Cytotoxic and immunomodulatory effects of Ebenus boissieri Barbey on breast cancer cells. Genet. Mol. Res. 2016, 15. [Google Scholar] [CrossRef]
- Allegra, A.; Tonacci, A.; Pioggia, G.; Musolino, C.; Gangemi, S. Anticancer activity of Rosmarinus officinalis L.: Mechanisms of Action and Therapeutic Potentials. Nutrients 2020, 12, 1739. [Google Scholar] [CrossRef]
- Kowalczyk, T.; Sitarek, P.; Skała, E.; Toma, M.; Wielanek, M.; Pytel, D.; Wieczfińska, J.; Szemraj, J.; Śliwiński, T. Induction of apoptosis by in vitro and in vivo plant extracts derived from Menyanthes trifoliata L. in human cancer cells. Cytotechnology 2019, 71, 165–180. [Google Scholar] [CrossRef]
- Vervandier-Fasseur, D.; Latruffe, N. The potential use of resveratrol for cancer prevention. Molecules 2019, 24, 4506. [Google Scholar] [CrossRef]
- Aja, I.; Ruiz-Larrea, M.B.; Courtois, A.; Krisa, S.; Richard, T.; Ruiz-Sanz, J.-I. Screening of natural stilbene oligomers from Vitis vinifera for anticancer activity on human hepatocellular carcinoma cells. Antioxidants 2020, 9, 469. [Google Scholar] [CrossRef]
- Marvibaigi, M.; Supriyanto, E.; Amini, N.; Abdul Majid, F.A.; Jaganathan, S.K. Preclinical and clinical effects of mistletoe against breast cancer. Biomed Res. Int. 2014, 2014, 785479. [Google Scholar] [CrossRef]
- de Oliveira Melo, M.N.; Oliveira, A.P.; Wiecikowski, A.F.; Carvalho, R.S.; de Lima Castro, J.; de Oliveira, F.A.G.; Pereira, H.M.G.; da Veiga, V.F.; Capella, M.M.A.; Rocha, L. Phenolic compounds from Viscum album tinctures enhanced antitumor activity in melanoma murine cancer cells. Saudi Pharm. J. 2018, 26, 311–322. [Google Scholar] [CrossRef]
- Chen, T.; Li, B.; Qiu, Y.; Qiu, Z.; Qu, P. Funct. mechanism of Ginsenosides on tumor growth and metastasis. Saudi J. Biol. Sci. 2018, 25, 917–922. [Google Scholar] [CrossRef] [PubMed]
- Jia, L.; Zhao, Y. Current evaluation of the millennium phytomedicine-ginseng (I): Etymology, pharmacognosy, phytochemistry, market and regulations. Curr. Med. Chem. 2009, 16, 2475–2484. [Google Scholar] [CrossRef] [PubMed]
- Hong, H.; Baatar, D.; Hwang, S.G. Anticancer activities of ginsenosides, the main active components of ginseng. Evid. Based Complement. Altern. Med. 2021, 2021, 8858006. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.-L.; Lv, G.-Y.; He, B.-C.; Zhang, B.-Q.; Zhang, H.; Wang, N.; Wang, C.-Z.; Du, W.; Yuan, C.-S.; He, T.-C. Ginseng saponin metabolite 20 (S)-protopanaxadiol inhibits tumor growth by targeting multiple cancer signaling pathways. Oncol. Rep. 2013, 30, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Han, L.; Li, P.; Zhang, S.; Zhang, M.; Li, X.; Chu, J.; Wang, L.; Tu, P.; Zhang, Y. Region-Specific Biomarkers and Their Mechanisms in the Treatment of Lung Adenocarcinoma: A Study of Panax quinquefolius from Wendeng, China. Molecules 2021, 26, 6829. [Google Scholar] [CrossRef] [PubMed]
- Ham, S.W.; Kim, J.-K.; Jeon, H.-Y.; Kim, E.-J.; Jin, X.; Eun, K.; Park, C.G.; Lee, S.Y.; Seo, S.; Kim, J.Y. Korean Red ginseng extract inhibits glioblastoma propagation by blocking the Wnt signaling pathway. J. Ethnopharmacol. 2019, 236, 393–400. [Google Scholar] [CrossRef]
- Guo, J.-Q.; Zheng, Q.-H.; Chen, H.; Chen, L.; Xu, J.-B.; Chen, M.-Y.; Lu, D.; Wang, Z.-H.; Tong, H.-F.; Lin, S. Ginsenoside Rg3 inhibition of vasculogenic mimicry in pancreatic cancer through downregulation of VE-cadherin/EphA2/MMP9/MMP2 expression. Int. J. Oncol. 2014, 45, 1065–1072. [Google Scholar] [CrossRef]
- Nag, S.A.; Qin, J.; Wang, W.; Wang, M.-H.; Wang, H.; Zhang, R. Ginsenosides as anticancer agents: In vitro and in vivo activities, structure-activity relationships, and molecular mechanisms of action. Front. Pharmacol. 2012, 3, 25. [Google Scholar] [CrossRef]
- Lerma-Herrera, M.A.; Beiza-Granados, L.; Ochoa-Zarzosa, A.; López-Meza, J.E.; Hernández-Hernández, J.D.; Aviña-Verduzco, J.; García-Gutiérrez, H.A. In vitro cytotoxic potential of extracts from Aristolochia foetida Kunth against MCF-7 and bMECs cell lines. Saudi J. Biol. Sci. 2021, 28, 7082–7089. [Google Scholar] [CrossRef]
- Nam, J.-S.; Park, S.-Y.; Lee, S.-O.; Lee, H.-J.; Jang, H.-L.; Rhee, Y.H. The growth-inhibitory effects of pawpaw (Asimina triloba [L.] Dunal) roots, twigs, leaves, and fruit against human gastric (AGS) and cervical (HeLa) cancer cells and their anti-inflammatory activities. Mol. Biology Rep. 2021, 48, 2173–2181. [Google Scholar] [CrossRef]
- Wisintainer, G.; Scola, G.; Moura, S.; Lemos, T.; Pessoa, C.; de Moraes, M.; Souza, L.; Roesch-Ely, M.; Henriques, J. O-naphthoquinone isolated from Capraria biflora L. induces selective cytotoxicity in tumor cell lines. Genet. Mol. Res. 2015, 14, 17472–17481. [Google Scholar] [CrossRef] [PubMed]
- Barbosa-Jobim, G.S.; Costa-Lira, E.; Ralph, A.C.L.; Gregorio, L.; Lemos, T.L.; Burbano, R.R.; Calcagno, D.Q.; Smith, M.A.; Montenegro, R.C.; Vasconcellos, M.C. Biflorin inhibits the proliferation of gastric cancer cells by decreasing MYC expression. Toxicol. In Vitro 2020, 63, 104735. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.; Gong, L.; Qiu, K.; Zhang, Z.; Wan, L. Echinacoside inhibits breast cancer cells by suppressing the Wnt/β-catenin signaling pathway. Biochem. Biophys. Res. Commun. 2020, 526, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Hosami, F.; Manayi, A.; Salimi, V.; Khodakhah, F.; Nourbakhsh, M.; Nakstad, B.; Tavakoli-Yaraki, M. The pro-apoptosis effects of Echinacea purpurea and Cannabis sativa extracts in human lung cancer cells through caspase-dependent pathway. BMC Complement. Med. Ther. 2021, 21, 37. [Google Scholar] [CrossRef] [PubMed]
- Kuttikrishnan, S.; Siveen, K.S.; Prabhu, K.S.; Khan, A.Q.; Akhtar, S.; Mateo, J.M.; Merhi, M.; Taha, R.; Omri, H.E.; Mraiche, F. Sanguinarine suppresses growth and induces apoptosis in childhood acute lymphoblastic leukemia. Leuk. Lymphoma 2019, 60, 782–794. [Google Scholar] [CrossRef] [PubMed]
- Tuzimski, T.; Petruczynik, A.; Plech, T.; Kaproń, B.; Makuch-Kocka, A.; Szultka-Młyńska, M.; Misiurek, J.; Buszewski, B. Determination of Cytotoxic Activity of Sanguinaria canadensis Extracts against Human Melanoma Cells and Comparison of Their Cytotoxicity with Cytotoxicity of Some Anticancer Drugs. Molecules 2021, 26, 1738. [Google Scholar] [CrossRef]
- Croaker, A.; King, G.J.; Pyne, J.H.; Anoopkumar-Dukie, S.; Liu, L. Sanguinaria canadensis: Traditional medicine, phytochemical composition, biological activities and current uses. Int. J. Mol. Sci. 2016, 17, 1414. [Google Scholar] [CrossRef]
- Singab, A.N.; Youssef, F.S.; Ashour, M.L.; Wink, M. The genus Eremophila (Scrophulariaceae): An ethnobotanical, biological and phytochemical review. J. Pharm. Pharmacol. 2013, 65, 1239–1279. [Google Scholar] [CrossRef]
- Petersen, M.J.; Lund, X.L.; Semple, S.J.; Buirchell, B.; Franzyk, H.; Gajhede, M.; Kongstad, K.T.; Stenvang, J.; Staerk, D. Reversal of ABCG2/BCRP-Mediated Multidrug Resistance by 5, 3′, 5′-Trihydroxy-3, 6, 7, 4′-Tetramethoxyflavone Isolated from the Australian Desert Plant Eremophila galeata Chinnock. Biomolecules 2021, 11, 1534. [Google Scholar] [CrossRef]
- An, G.; Morris, M.E. Efflux transporters in cancer resistance: Molecular and Funct. characterization of breast cancer resistance protein. In Drug Efflux Pumps in Cancer Resistance Pathways: From Molecular Recognition and Characterization to Possible Inhibition Strategies in Chemotherapy; Elsevier: Amsterdam, The Netherlands, 2020; pp. 67–96. [Google Scholar]
- Shalom, J.; Cock, I.E. Terminalia ferdinandiana Exell. fruit and leaf extracts inhibit proliferation and induce apoptosis in selected human cancer cell lines. Nutr. Cancer 2018, 70, 579–593. [Google Scholar] [CrossRef]
- Mohanty, S.; Cock, I.E. The chemoTher. potential of Terminalia ferdinandiana: Phytochemistry and bioactivity. Pharmacogn. Rev. 2012, 6, 29. [Google Scholar] [PubMed]
- Sakulnarmrat, K.; Fenech, M.; Thomas, P.; Konczak, I. Cytoprotective and pro-apoptotic activities of native Australian herbs polyphenolic-rich extracts. Food Chem. 2013, 136, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Chuen, T.L.; Vuong, Q.V.; Hirun, S.; Bowyer, M.C.; Predebon, M.J.; Goldsmith, C.D.; Sakoff, J.A.; Scarlett, C.J. Antioxidant and anti-proliferative properties of Davidson’s plum (Davidsonia pruriens F. Muell) phenolic-enriched extracts as affected by different extraction solvents. J. Herbal Med. 2016, 6, 187–192. [Google Scholar] [CrossRef]
- Jamieson, N.; Sirdaarta, J.; Cock, I. The Anti-Proliferative Properties of Australian Plants with High Antioxidant Capacities Against Cancer Cell Lines. Pharmacogn. Commun. 2014, 4, 71–82. [Google Scholar]
- Bäcker, C.; Jenett-Siems, K.; Siems, K.; Wurster, M.; Bodtke, A.; Lindequist, U. Cytotoxic saponins from the seeds of Pittosporum angustifolium. Z. Nat. C 2014, 69, 191–198. [Google Scholar] [CrossRef]
- Hawksworth, D.L.; Lücking, R. Fungal diversity revisited: 2.2 to 3.8 million species. Microbiol. Spectr. 2017, 5, 10. [Google Scholar] [CrossRef]
- Nowakowski, P.; Markiewicz-Żukowska, R.; Bielecka, J.; Mielcarek, K.; Grabia, M.; Socha, K. Treasures from the forest: Evaluation of mushroom extracts as anti-cancer agents. Biomed. Pharmacother. 2021, 143, 112106. [Google Scholar] [CrossRef] [PubMed]
- Al-Obaidi, J.R.; Jambari, N.N.; Ahmad-Kamil, E. Mycopharmaceuticals and Nutraceuticals: Promising Agents to Improve Human Well-Being and Life Quality. J. Fungi 2021, 7, 503. [Google Scholar] [CrossRef]
- Keller, N.P. Fungal secondary metabolism: Regulation, function and drug discovery. Nat. Rev. Microbiol. 2019, 17, 167–180. [Google Scholar] [CrossRef]
- Avalos, J.; Limón, M.C. Fungal Secondary Metabolism. Encyclopedia 2021, 2, 1–13. [Google Scholar] [CrossRef]
- Szychowski, K.A.; Skóra, B.; Pomianek, T.; Gmiński, J. Inonotus obliquus–from folk medicine to clinical use. J. Tradit. Complement. Med. 2021, 11, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Yukawa, H.; Ishikawa, S.; Kawanishi, T.; Tamesada, M.; Tomi, H. Direct cytotoxicity of Lentinula edodes mycelia extract on human hepatocellular carcinoma cell line. Biol. Pharm. Bull. 2012, 35, 1014–1021. [Google Scholar] [CrossRef] [PubMed]
- Israilides, C.; Kletsas, D.; Arapoglou, D.; Philippoussis, A.; Pratsinis, H.; Ebringerová, A.; Hříbalová, V.; Harding, S. In vitro cytostatic and immunomodulatory properties of the medicinal mushroom Lentinula edodes. Phytomedicine 2008, 15, 512–519. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Gu, J.; Qi, J.; Zeng, X.N.; Ji, J.; Chen, Z.Z.; Sun, X.L. Lentinan exerts synergistic apoptotic effects with paclitaxel in A549 cells via activating ROS-TXNIP-NLRP 3 inflammasome. J. Cell. Mol. Med. 2015, 19, 1949–1955. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Zhao, W.; Xie, Q.; Zhan, Y.; Wu, B. Lentinan reduces tumor progression by enhancing gemcitabine chemotherapy in urothelial bladder cancer. Surg. Oncol. 2015, 24, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Xiao, Y.; Xiao, N. Effect of lentinan combined with docetaxel and cisplatin on the proliferation and apoptosis of BGC823 cells. Tumor Biol. 2013, 34, 1531–1536. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, Q.; Wang, J.; Cheng, F.; Huang, X.; Cheng, Y.; Wang, K. Polysaccharide from Lentinus edodes combined with oxaliplatin possesses the synergy and attenuation effect in hepatocellular carcinoma. Cancer Lett. 2016, 377, 117–125. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, J.; Han, Q.; Luo, Q.; Zhang, H.; Wang, Y. Construction of doxorubicin-conjugated lentinan nanoparticles for enhancing the cytotoxocity effects against breast cancer cells. Coll. Surf. A Physicochem. Eng. Asp. 2019, 579, 123657. [Google Scholar] [CrossRef]
- Wang, J.; Li, W.; Huang, X.; Liu, Y.; Li, Q.; Zheng, Z.; Wang, K. A polysaccharide from Lentinus edodes inhibits human colon cancer cell proliferation and suppresses tumor growth in athymic nude mice. Oncotarget 2017, 8, 610. [Google Scholar] [CrossRef]
- Ina, K.; Kataoka, T.; Ando, T. The use of lentinan for treating gastric cancer. Anticancer Agents Med. Chem. 2013, 13, 681–688. [Google Scholar] [CrossRef]
- Ina, H.; Yoneda, M.; Kanda, M.; Kodera, Y.; Kabeya, M.; Yuasa, S. Lentinan, a shiitake mushroom beta-glucan, stimulates tumor-specific adaptive immunity through PD-L1 down-regulation in gastric cancer cells. Med. Chem 2016, 6, 710–714. [Google Scholar] [CrossRef]
- Antonelli, M.; Donelli, D.; Firenzuoli, F. Lentinan for Integrative Cancer Treatment: An Umbrella Review. Multidiscip. Digit. Publ. Inst. Proc. 2020, 79, 1. [Google Scholar]
- Zhang, M.; Zhang, Y.; Zhang, L.; Tian, Q. Mushroom polysaccharide lentinan for treating different types of cancers: A review of 12 years clinical studies in China. Progress Mol. Biol. Translat. Sci. 2019, 163, 297–328. [Google Scholar]
- Zhao, H.; Wu, L.; Yan, G.; Chen, Y.; Zhou, M.; Wu, Y.; Li, Y. Inflammation and tumor progression: Signaling pathways and targeted intervention. Signal Transduct. Target. Ther. 2021, 6, 263. [Google Scholar] [CrossRef]
- Basu, A.; Ramamoorthi, G.; Albert, G.; Gallen, C.; Beyer, A.; Snyder, C.; Koski, G.; Disis, M.L.; Czerniecki, B.J.; Kodumudi, K. Differentiation and regulation of TH cells: A balancing act for cancer immunotherapy. Front. Immunol. 2021, 12, 669474. [Google Scholar] [CrossRef] [PubMed]
- Ina, K.; Furuta, R.; Kataoka, T.; Kayukawa, S.; Yoshida, T.; Miwa, T.; Yamamura, Y.; Takeuchi, Y. Lentinan prolonged survival in patients with gastric cancer receiving S-1-based chemotherapy. World J. Clin. Oncol. 2011, 2, 339. [Google Scholar] [CrossRef]
- Howard, R.; Kanetsky, P.A.; Egan, K.M. Exploring the prognostic value of the neutrophil-to-lymphocyte ratio in cancer. Sci. Rep. 2019, 9, 19673. [Google Scholar] [CrossRef]
- Szor, D.J.; Dias, A.R.; Pereira, M.A.; Ramos, M.F.K.P.; Zilberstein, B.; Cecconello, I.; Ribeiro-Júnior, U. Prognostic role of neutrophil/lymphocyte ratio in resected gastric cancer: A systematic review and meta-analysis. Clinics 2018, 73, e360. [Google Scholar] [CrossRef]
- Tavakkoli, M.; Wilkins, C.R.; Mones, J.V.; Mauro, M.J. A novel paradigm between leukocytosis, G-CSF secretion, neutrophil-to-lymphocyte ratio, myeloid-derived suppressor cells, and prognosis in non-small cell lung cancer. Front. Oncol. 2019, 9, 295. [Google Scholar] [CrossRef]
- Matsuoka, H.; Seo, Y.; Wakasugi, H.; Saito, T.; Tomoda, H. Lentinan potentiates immunity and prolongs the survival time of some patients. Anticancer Res. 1997, 17, 2751–2755. [Google Scholar]
- Wang, X.-E.; Wang, Y.-H.; Zhou, Q.; Peng, M.; Zhang, J.; Chen, M.; Ma, L.-J.; Xie, G.-M. Immunomodulatory effect of lentinan on aberrant T subsets and cytokines profile in non-small cell lung cancer patients. Pathol. Oncol. Res. 2020, 26, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Oba, K.; Kobayashi, M.; Matsui, T.; Kodera, Y.; Sakamoto, J. Individual patient based meta-analysis of lentinan for unresectable/recurrent gastric cancer. Anticancer Res. 2009, 29, 2739–2745. [Google Scholar] [PubMed]
- Isoda, N.; Eguchi, Y.; Nukaya, H.; Hosho, K.; Suga, Y.; Suga, T.; Nakazawa, S.; Sugano, K. Clinical efficacy of superfine dispersed lentinan (beta-1, 3-glucan) in patients with hepatocellular carcinoma. Hepato Gastroenterol. 2009, 56, 437–441. [Google Scholar]
- Shimizu, K.; Watanabe, S.; Matsuda, K.; Suga, T.; Nakazawa, S.; Shiratori, K. Efficacy of oral administered superfine dispersed lentinan for advanced pancreatic cancer. Hepato Gastroenterol. 2009, 56, 240–244. [Google Scholar]
- Wang, H.; Cai, Y.; Zheng, Y.; Bai, Q.; Xie, D.; Yu, J. Efficacy of biological response modifier lentinan with chemotherapy for advanced cancer: A meta-analysis. Cancer Med. 2017, 6, 2222–2233. [Google Scholar] [CrossRef]
- Babu, P.D.; Subhasree, R. The sacred mushroom “Reishi”—A review. Am. Eurasian J. Bot. 2008, 1, 107–110. [Google Scholar]
- Suarez-Arroyo, I.J.; Rosario-Acevedo, R.; Aguilar-Perez, A.; Clemente, P.L.; Cubano, L.A.; Serrano, J.; Schneider, R.J.; Martínez-Montemayor, M.M. Anti-tumor effects of Ganoderma lucidum (reishi) in inflammatory breast cancer in in vivo and in vitro models. PLoS ONE 2013, 8, e57431. [Google Scholar] [CrossRef]
- Martínez-Montemayor, M.M.; Acevedo, R.R.; Otero-Franqui, E.; Cubano, L.A.; Dharmawardhane, S.F. Ganoderma lucidum (Reishi) inhibits cancer cell growth and expression of key molecules in inflammatory breast cancer. Nutr. Cancer 2011, 63, 1085–1094. [Google Scholar] [CrossRef]
- Acevedo-Díaz, A.; Ortiz-Soto, G.; Suárez-Arroyo, I.J.; Zayas-Santiago, A.; Martínez Montemayor, M.M. Ganoderma lucidum extract reduces the motility of breast cancer cells mediated by the RAC–lamellipodin Axis. Nutrients 2019, 11, 1116. [Google Scholar] [CrossRef]
- Liang, C.; Tian, D.; Liu, Y.; Li, H.; Zhu, J.; Li, M.; Xin, M.; Xia, J. Review of the molecular mechanisms of Ganoderma lucidum triterpenoids: Ganoderic acids A, C2, D, F, DM, X and Y. Eur. J. Med. Chem. 2019, 174, 130–141. [Google Scholar] [CrossRef]
- Sohretoglu, D.; Huang, S. Ganoderma lucidum polysaccharides as an anti-cancer agent. Anti-Cancer Agents Med. Chem. 2018, 18, 667–674. [Google Scholar] [CrossRef] [PubMed]
- Radwan, F.F.; Perez, J.M.; Haque, A. Apoptotic and immune restoration effects of ganoderic acids define a new prospective for complementary treatment of cancer. J. Clin. Cell. Immunol. 2011, 11, 4. [Google Scholar] [CrossRef] [PubMed]
- Gill, B.S.; Kumar, S. Ganoderic acid targeting multiple receptors in cancer: In silico and in vitro study. Tumor Biol. 2016, 37, 14271–14290. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Dai, L.; Wang, L.; Zhu, J. Ganoderic acid DM induces autophagic apoptosis in non-small cell lung cancer cells by inhibiting the PI3K/Akt/mTOR activity. Chem. Biol. Interact. 2020, 316, 108932. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.-S.; Lu, J.-J.; Guo, J.-J.; Li, Y.-B.; Tan, W.; Dang, Y.-Y.; Zhong, Z.-F.; Xu, Z.-T.; Chen, X.-P.; Wang, Y.-T. Ganoderic acid DM, a natural triterpenoid, induces DNA damage, G1 cell cycle arrest and apoptosis in human breast cancer cells. Fitoterapia 2012, 83, 408–414. [Google Scholar] [CrossRef]
- Chen, N.-H.; Liu, J.-W.; Zhong, J.-J. Ganoderic acid Me inhibits tumor invasion through down-regulating matrix metalloproteinases 2/9 gene expression. J. Pharmacol. Sci. 2008, 108, 212–216. [Google Scholar] [CrossRef]
- Xu, J.; Chen, F.; Wang, G.; Liu, B.; Song, H.; Ma, T. The Versatile Functions of G. Lucidum Polysaccharides and G. Lucidum Triterpenes in Cancer Radiotherapy and Chemotherapy. Cancer Manag. Res. 2021, 13, 6507. [Google Scholar] [CrossRef]
- Pan, H.; Wang, Y.; Wang, Y.; Li, M.; Li, Z.; Xu, J.; Wang, X. Ganoderma lucidum polysaccharides induce cytotoxicity in colorectal cancer cells through inducing autophagosome accumulation and inhibiting autophagic flux. FASEB J. 2019, 33, lb395. [Google Scholar] [CrossRef]
- Zhong, J.; Fang, L.; Chen, R.; Xu, J.; Guo, D.; Guo, C.; Guo, C.; Chen, J.; Chen, C.; Wang, X. Polysaccharides from sporoderm-removed spores of Ganoderma lucidum induce apoptosis in human gastric cancer cells via disruption of autophagic flux. Oncol. Lett. 2021, 21, 425. [Google Scholar] [CrossRef]
- Su, J.; Li, D.; Chen, Q.; Li, M.; Su, L.; Luo, T.; Liang, D.; Lai, G.; Shuai, O.; Jiao, C. Anti-breast cancer enhancement of a polysaccharide from spore of Ganoderma lucidum with paclitaxel: Suppression on tumor metabolism with gut microbiota reshaping. Front. Microbiol. 2018, 9, 3099. [Google Scholar] [CrossRef]
- Jin, H.; Song, C.; Zhao, Z.; Zhou, G. Ganoderma Lucidum Polysaccharide, an Extract from Ganoderma Lucidum, Exerts Suppressive Effect on Cervical Cancer Cell Malignancy through Mitigating Epithelial-Mesenchymal and JAK/STAT5 Signaling Pathway. Pharmacology 2020, 105, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Hsu, W.-H.; Hua, W.-J.; Qiu, W.-L.; Tseng, A.-J.; Cheng, H.-C.; Lin, T.-Y. WSG, a glucose-enriched polysaccharide from Ganoderma lucidum, suppresses tongue cancer cells via inhibition of EGFR-mediated signaling and potentiates cisplatin-induced apoptosis. Int. J. Biol. Macromol. 2021, 193, 1201–1208. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Xu, X.; Liu, S.; Huang, L.; Gu, J. Ganoderma: A cancer immunotherapy review. Front. Pharmacol. 2018, 9, 1217. [Google Scholar] [CrossRef]
- Zhao, R.; Chen, Q.; He, Y.-m. The effect of Ganoderma lucidum extract on immunological function and identify its anti-tumor immunostimulatory activity based on the biological network. Sci. Rep. 2018, 8, 12680. [Google Scholar] [CrossRef]
- Chien, C.M.; Cheng, J.-L.; Chang, W.-T.; Tien, M.-H.; Tsao, C.-M.; Chang, Y.-H.; Chang, H.-Y.; Hsieh, J.-F.; Wong, C.-H.; Chen, S.-T. Polysaccharides of Ganoderma lucidum alter cell immunophenotypic expression and enhance CD56+ NK-cell cytotoxicity in cord blood. Bioorg. Med. Chem. 2004, 12, 5603–5609. [Google Scholar] [CrossRef]
- Chen, H.-S.; Tsai, Y.-F.; Lin, S.; Lin, C.-C.; Khoo, K.-H.; Lin, C.-H.; Wong, C.-H. Studies on the immuno-modulating and anti-tumor activities of Ganoderma lucidum (Reishi) polysaccharides. Bioorg. Med. Chem. 2004, 12, 5595–5601. [Google Scholar] [CrossRef]
- Zhang, S.; Pang, G.; Chen, C.; Qin, J.; Yu, H.; Liu, Y.; Zhang, X.; Song, Z.; Zhao, J.; Wang, F. Effective cancer Immunother. by Ganoderma lucidum polysaccharide-gold nanocomposites through dendritic cell activation and memory T cell response. Carbohydr. Polym. 2019, 205, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Guo, D.; Fang, L.; Sang, T.; Wu, J.; Guo, C.; Wang, Y.; Wang, Y.; Chen, C.; Chen, J. Ganoderma lucidum polysaccharide modulates gut microbiota and immune cell function to inhibit inflammation and tumorigenesis in colon. Carbohydr. Polym. 2021, 267, 118231. [Google Scholar] [CrossRef]
- Chiu, H.-F.; Fu, H.-Y.; Lu, Y.-Y.; Han, Y.-C.; Shen, Y.-C.; Venkatakrishnan, K.; Golovinskaia, O.; Wang, C.-K. Triterpenoids and polysaccharide peptides-enriched Ganoderma lucidum: A randomized, double-blind placebo-controlled crossover study of its antioxidation and hepatoprotective efficacy in healthy volunteers. Pharm. Biol. 2017, 55, 1041–1046. [Google Scholar] [CrossRef]
- Deng, Y.; Ma, J.; Tang, D.; Zhang, Q. Dynamic biomarkers indicate the immunological benefits provided by Ganoderma spore powder in post-operative breast and lung cancer patients. Clin. Translat. Oncol. 2021, 23, 1481–1490. [Google Scholar] [CrossRef]
- Wu, J.-Y.; Siu, K.-C.; Geng, P. Bioactive ingredients and medicinal values of Grifola frondosa (Maitake). Foods 2021, 10, 95. [Google Scholar] [CrossRef] [PubMed]
- De Silva, D.D.; Rapior, S.; Fons, F.; Bahkali, A.H.; Hyde, K.D. Medicinal mushrooms in supportive cancer therapies: An approach to anti-cancer effects and putative mechanisms of action. Fungal Divers. 2012, 55, 1–35. [Google Scholar] [CrossRef]
- Alonso, E.N.; Orozco, M.; Nieto, A.E.; Balogh, G.A. Genes related to suppression of malignant phenotype induced by Maitake D-Fraction in breast cancer cells. J. Med. Food 2013, 16, 602–617. [Google Scholar] [CrossRef] [PubMed]
- Alonso, E.N.; Ferronato, M.J.; Fermento, M.E.; Gandini, N.A.; Romero, A.L.; Guevara, J.A.; Facchinetti, M.M.; Curino, A.C. Antitumoral and antimetastatic activity of Maitake D-Fraction in triple-negative breast cancer cells. Oncotarget 2018, 9, 23396. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-H.; Chang, C.-Y.; Lee, K.-R.; Lin, H.-J.; Lin, W.-C.; Chen, T.-H.; Wan, L. Cold-water extracts of Grifola frondosa and its purified active fraction inhibit hepatocellular carcinoma in vitro and in vivo. Exp. Biol. Med. 2016, 241, 1374–1385. [Google Scholar] [CrossRef] [PubMed]
- Fullerton, S.; Samadi, A.; Tortorelis, D.; Choudhury, M.; Mallouh, C.; Tazaki, H.; Konno, S. Induction of apoptosis in human prostatic cancer cells with beta-glucan (Maitake mushroom polysaccharide). Mol. Urology 2000, 4, 7–13. [Google Scholar]
- Alexander, B.; Fishman, A.I.; Eshghi, M.; Choudhury, M.; Konno, S. Induction of cell death in renal cell carcinoma with combination of D-fraction and vitamin C. Integr. Cancer Ther. 2013, 12, 442–448. [Google Scholar] [CrossRef]
- Roldan-Deamicis, A.; Alonso, E.; Brie, B.; Braico, D.A.; Balogh, G.A. Maitake Pro4X has anti-cancer activity and prevents oncogenesis in BALB c mice. Cancer Med. 2016, 5, 2427–2441. [Google Scholar] [CrossRef]
- Kodama, N.; Komuta, K.; Sakai, N.; Nanba, H. Effects of D-Fraction, a polysaccharide from Grifola frondosa on tumor growth involve activation of NK cells. Biol. Pharm. Bull. 2002, 25, 1647–1650. [Google Scholar] [CrossRef]
- Zhao, F.; Guo, Z.; Zhang, Y.; Song, L.; Ma, L.; Zhao, J. Anti-tumor and immunomodulatory effects of Grifola frondosa polysaccharide combined with vitamin C on Heps-bearing mice: Based on inducing apoptosis and autophagy. J. Funct. Foods 2021, 87, 104797. [Google Scholar] [CrossRef]
- Masuda, Y.; Nakayama, Y.; Tanaka, A.; Naito, K.; Konishi, M. Antitumor activity of orally administered maitake α-glucan by stimulating antitumor immune response in murine tumor. PLoS ONE 2017, 12, e0173621. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Guo, Z.; Ma, Z.-R.; Ma, L.-L.; Zhao, J. Antitumor activities of Grifola frondosa (Maitake) polysaccharide: A meta-analysis based on preclinical evidence and quality assessment. J. Ethnopharmacol. 2021, 280, 114395. [Google Scholar] [CrossRef] [PubMed]
- Kodama, N.; Komuta, K.; Nanba, H. Can maitake MD-fraction aid cancer patients? Altern. Med. Rev. 2002, 7, 236–239. [Google Scholar] [PubMed]
- Kodama, N.; Komuta, K.; Nanba, H. Effect of Maitake (Grifola frondosa) D-Fraction on the activation of NK cells in cancer patients. J. Med. Food 2003, 6, 371–377. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Zhang, L.; Wang, H. The biological activities of the antitumor drug Grifola frondosa polysaccharide. Prog. Mol. Biol. Translat. Sci. 2019, 163, 221–261. [Google Scholar]
- Zhou, X.; Gong, Z.; Su, Y.; Lin, J.; Tang, K. Cordyceps fungi: Natural products, pharmacological functions and developmental products. J. Pharm. Pharmacol. 2009, 61, 279–291. [Google Scholar] [CrossRef]
- Yue, K.; Ye, M.; Zhou, Z.; Sun, W.; Lin, X. The genus Cordyceps: A chemical and pharmacological review. J. Pharm. Pharmacol. 2013, 65, 474–493. [Google Scholar] [CrossRef]
- Jędrejko, K.J.; Lazur, J.; Muszyńska, B. Cordyceps militaris: An Overview of Its Chemical Constituents in Relation to Biological Activity. Foods 2021, 10, 2634. [Google Scholar] [CrossRef]
- Li, J.; Cai, H.; Sun, H.; Qu, J.; Zhao, B.; Hu, X.; Li, W.; Qian, Z.; Yu, X.; Kang, F. Extracts of Cordyceps sinensis inhibit breast cancer growth through promoting M1 macrophage polarization via NF-κB pathway activation. J. Ethnopharmacol. 2020, 260, 112969. [Google Scholar] [CrossRef]
- Seo, H.; Song, J.; Kim, M.; Han, D.-W.; Park, H.-J.; Song, M. Cordyceps militaris grown on germinated soybean suppresses KRAS-driven colorectal cancer by inhibiting the RAS/ERK pathway. Nutrients 2018, 11, 20. [Google Scholar] [CrossRef]
- Jeong, M.-K.; Yoo, H.-S.; Kang, I.-C. The extract of Cordyceps militaris inhibited the proliferation of cisplatin-resistant a549 lung cancer cells by downregulation of H-Ras. J. Med. Food 2019, 22, 823–832. [Google Scholar] [CrossRef] [PubMed]
- Jo, E.; Jang, H.-J.; Shen, L.; Yang, K.E.; Jang, M.S.; Huh, Y.H.; Yoo, H.-S.; Park, J.; Jang, I.S.; Park, S.J. Cordyceps militaris Exerts Anticancer Effect on Non–Small Cell Lung Cancer by Inhibiting Hedgehog Signaling via Suppression of TCTN3. Integr. Cancer Ther. 2020, 19, 1534735420923756. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.Y.; Park, S.J.; Park, Y.J. The anticancer properties of cordycepin and their underlying mechanisms. Int. J. Mol. Sci. 2018, 19, 3027. [Google Scholar] [CrossRef] [PubMed]
- Fishman, P.; Bar-Yehuda, S.; Synowitz, M.; Powell, J.; Klotz, K.; Gessi, S.; Borea, P. Adenosine receptors and cancer. Adenosine Recept. Health Dis. 2009, 193, 399–441. [Google Scholar]
- Cao, H.-L.; Liu, Z.-J.; Chang, Z. Cordycepin induces apoptosis in human bladder cancer cells via activation of A3 adenosine receptors. Tumor Biol. 2017, 39, 1010428317706915. [Google Scholar] [CrossRef]
- Nakamura, K.; Yoshikawa, N.; Yamaguchi, Y.; Kagota, S.; Shinozuka, K.; Kunitomo, M. Antitumor effect of cordycepin (3′-deoxyadenosine) on mouse melanoma and lung carcinoma cells involves adenosine A3 receptor stimulation. Anticancer Res. 2006, 26, 43–47. [Google Scholar]
- Yoshikawa, N.; Yamada, S.; Takeuchi, C.; Kagota, S.; Shinozuka, K.; Kunitomo, M.; Nakamura, K. Cordycepin (3′-deoxyadenosine) inhibits the growth of B16-BL6 mouse melanoma cells through the stimulation of adenosine A3 receptor followed by glycogen synthase kinase-3β activation and cyclin D1 suppression. Naunyn Schmiedeberg’s Arch. Pharmacol. 2008, 377, 591–595. [Google Scholar] [CrossRef]
- Zhao, X.; Guan, J.-L. Focal adhesion kinase and its signaling pathways in cell migration and angiogenesis. Adv. Drug Deliv. Rev. 2011, 63, 610–615. [Google Scholar] [CrossRef]
- Yao, W.-L.; Ko, B.-S.; Liu, T.-A.; Liang, S.-M.; Liu, C.-C.; Lu, Y.-J.; Tzean, S.-S.; Shen, T.-L.; Liou, J.-Y. Cordycepin suppresses integrin/FAK signaling and epithelial-mesenchymal transition in hepatocellular carcinoma. Anti Cancer Agents Med. Chem. 2014, 14, 29–34. [Google Scholar] [CrossRef]
- Jin, Y.; Meng, X.; Qiu, Z.; Su, Y.; Yu, P.; Qu, P. Anti-tumor and anti-metastatic roles of cordycepin, one bioactive compound of Cordyceps militaris. Saudi J. Biol. Sci. 2018, 25, 991–995. [Google Scholar] [CrossRef]
- Lee, S.Y.; Debnath, T.; Kim, S.-K.; Lim, B.O. Anti-cancer effect and apoptosis induction of cordycepin through DR3 pathway in the human colonic cancer cell HT-29. Food Chem. Toxicol. 2013, 60, 439–447. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.H.; Kim, S.O.; Kim, G.-Y.; Moon, S.-K.; Kim, W.-J.; Jeong, Y.K.; Yoo, Y.H.; Choi, Y.H. Involvement of autophagy in cordycepin-induced apoptosis in human prostate carcinoma LNCaP cells. Environ. Toxicol. Pharmacol. 2014, 38, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.-M.; Hong, S.-Y.; Yang, S.-H.; Wu, C.-C.; Wang, C.-Y.; Huang, B.-M. Anti-cancer effect of cordycepin on FGF9-induced testicular tumorigenesis. Int. J. Mol. Sci. 2020, 21, 8336. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chen, H.; Li, W.; Shan, L. Cordyceps acid alleviates lung cancer in nude mice. J. Biochem. Mol. Toxicol. 2021, 35, e22670. [Google Scholar] [CrossRef]
- Ka Wai Lee, S.; Kwok Wong, C.; Kai Kong, S.; Nam Leung, K.; Wai Kei Lam, C. Immunomodulatory activities of HERBSnSENSES™ Cordyceps—in vitro and in vivo studies. Immunopharmacol. Immunotoxicol. 2006, 28, 341–360. [Google Scholar] [CrossRef]
- Wang, M.; Meng, X.Y.; Le Yang, R.; Qin, T.; Wang, X.Y.; Zhang, K.Y.; Fei, C.Z.; Li, Y.; liang Hu, Y.; Xue, F.Q. Cordyceps militaris polysaccharides can enhance the immunity and antioxidation activity in immunosuppressed mice. Carbohydr. Polym. 2012, 89, 461–466. [Google Scholar] [CrossRef]
- Das, G.; Shin, H.-S.; Leyva-Gómez, G.; Prado-Audelo, M.L.D.; Cortes, H.; Singh, Y.D.; Panda, M.K.; Mishra, A.P.; Nigam, M.; Saklani, S. Cordyceps spp.: A review on its immune-stimulatory and other biological potentials. Front. Pharmacol. 2021, 11, 602364. [Google Scholar] [CrossRef]
- Qi, W.; Zhou, X.; Wang, J.; Zhang, K.; Zhou, Y.; Chen, S.; Nie, S.; Xie, M. Cordyceps sinensis polysaccharide inhibits colon cancer cells growth by inducing apoptosis and autophagy flux blockage via mTOR signaling. Carbohydr. Polym. 2020, 237, 116113. [Google Scholar] [CrossRef]
- Borodina, I.; Kenny, L.C.; McCarthy, C.M.; Paramasivan, K.; Pretorius, E.; Roberts, T.J.; van der Hoek, S.A.; Kell, D.B. The biology of ergothioneine, an antioxidant nutraceutical. Nutr. Res. Rev. 2020, 33, 190–217. [Google Scholar] [CrossRef]
- D’Onofrio, N.; Martino, E.; Balestrieri, A.; Mele, L.; Cautela, D.; Castaldo, D.; Balestrieri, M.L. Diet-derived ergothioneine induces necroptosis in colorectal cancer cells by activating the SIRT3/MLKL pathway. FEBS Lett. 2022. [Google Scholar] [CrossRef]
- Yoshida, S.; Shime, H.; Matsumoto, M.; Kasahara, M.; Seya, T. Anti-oxidative amino acid L-ergothioneine modulates the tumor microenvironment to facilitate adjuvant vaccine immunotherapy. Front. Immunol. 2019, 10, 671. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.; Ottosson, F.; Hellstrand, S.; Ericson, U.; Orho-Melander, M.; Fernandez, C.; Melander, O. Ergothioneine is associated with reduced mortality and decreased risk of cardiovascular disease. Heart 2020, 106, 691–697. [Google Scholar] [CrossRef] [PubMed]
- Winkels, R.M.; Van Brakel, L.; Van Baar, H.; Beelman, R.B.; Van Duijnhoven, F.J.; Geijsen, A.; Van Halteren, H.K.; Hansson, B.M.; Richie, J.P.; Sun, D. Are Ergothioneine Levels in Blood Associated with Chronic Peripheral Neuropathy in Colorectal Cancer Patients Who Underwent Chemotherapy? Nutr. Cancer 2020, 72, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Zheng, W. Deciphering the antitumoral potential of the bioactive metabolites from medicinal mushroom Inonotus obliquus. J. Ethnopharmacol. 2021, 265, 113321. [Google Scholar] [CrossRef]
- Arata, S.; Watanabe, J.; Maeda, M.; Yamamoto, M.; Matsuhashi, H.; Mochizuki, M.; Kagami, N.; Honda, K.; Inagaki, M. Continuous intake of the Chaga mushroom (Inonotus obliquus) aqueous extract suppresses cancer progression and maintains body temperature in mice. Heliyon 2016, 2, e00111. [Google Scholar] [CrossRef]
- Lee, M.-G.; Kwon, Y.-S.; Nam, K.-S.; Kim, S.Y.; Hwang, I.H.; Kim, S.; Jang, H. Chaga mushroom extract induces autophagy via the AMPK-mTOR signaling pathway in breast cancer cells. J. Ethnopharmacol. 2021, 274, 114081. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, J.; He, H.; Liu, H.; Yan, X.; Zou, K. Trametenolic Acid B Reverses Multidrug Resistance in Breast Cancer Cells Through Regulating the Expression Level of P-Glycoprotein. Phytother. Res. 2014, 28, 1037–1044. [Google Scholar] [CrossRef]
- Lu, Y.; Jia, Y.; Xue, Z.; Li, N.; Liu, J.; Chen, H. Recent developments in Inonotus obliquus (Chaga mushroom) polysaccharides: Isolation, structural characteristics, biological activities and application. Polymers 2021, 13, 1441. [Google Scholar] [CrossRef]
- Kim, Y.O.; Park, H.W.; Kim, J.H.; Lee, J.Y.; Moon, S.H.; Shin, C.S. Anti-cancer effect and structural characterization of endo-polysaccharide from cultivated mycelia of Inonotus obliquus. Life Sci. 2006, 79, 72–80. [Google Scholar] [CrossRef]
- Jiang, S.; Shi, F.; Lin, H.; Ying, Y.; Luo, L.; Huang, D.; Luo, Z. Inonotus obliquus polysaccharides induces apoptosis of lung cancer cells and alters energy metabolism via the LKB1/AMPK axis. Int. J. Biol. Macromol. 2020, 151, 1277–1286. [Google Scholar] [CrossRef]
- Pei, J.; Velu, P.; Zareian, M.; Feng, Z.; Vijayalakshmi, A. Effects of Syringic Acid on Apoptosis, Inflammation, and AKT/mTOR Signaling Pathway in Gastric Cancer Cells. Front. Nutr. 2021, 8, 788929. [Google Scholar] [CrossRef] [PubMed]
- Sung, B.; Pandey, M.K.; Nakajima, Y.; Nishida, H.; Konishi, T.; Chaturvedi, M.M.; Aggarwal, B.B. Identification of a novel blocker of IκBα kinase activation that enhances apoptosis and inhibits proliferation and invasion by suppressing nuclear factor-κB. Mol. Cancer Ther. 2008, 7, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Bao, C.; Zhang, J. Inotodiol suppresses proliferation of breast cancer in rat model of type 2 diabetes mellitus via downregulation of β-catenin signaling. Biomed. Pharmacother. 2018, 99, 142–150. [Google Scholar] [CrossRef]
- Zhang, S.-D.; Yu, L.; Wang, P.; Kou, P.; Li, J.; Wang, L.-T.; Wang, W.; Yao, L.-P.; Zhao, X.-H.; Fu, Y.-J. Inotodiol inhibits cells migration and invasion and induces apoptosis via p53-dependent pathway in HeLa cells. Phytomedicine 2019, 60, 152957. [Google Scholar] [CrossRef]
- Sarfraz, A.; Rasul, A.; Sarfraz, I.; Shah, M.A.; Hussain, G.; Shafiq, N.; Masood, M.; Adem, Ş.; Sarker, S.D.; Li, X. Hispolon: A natural polyphenol and emerging cancer killer by multiple cellular signaling pathways. Environ. Res. 2020, 190, 110017. [Google Scholar] [CrossRef] [PubMed]
- Masood, M.; Rasul, A.; Sarfraz, I.; Jabeen, F.; Liu, S.; Liu, X.; Wei, W.; Li, J.; Li, X. Hispolon induces apoptosis against prostate DU145 cancer cells via modulation of mitochondrial and STAT3 pathways. Pak. J. Pharm. Sci 2019, 32, 2237–2243. [Google Scholar] [PubMed]
- Al Saqr, A.; Majrashi, M.; Alrbyawi, H.; Govindarajulu, M.; Fujihashi, A.; Gottumukkala, S.; Poudel, I.; Arnold, R.D.; Babu, R.J.; Dhanasekaran, M. Elucidating the anti-melanoma effect and mechanisms of Hispolon. Life Sci. 2020, 256, 117702. [Google Scholar] [CrossRef]
- Hsin, M.-C.; Hsieh, Y.-H.; Wang, P.-H.; Ko, J.-L.; Hsin, I.-L.; Yang, S.-F. Hispolon suppresses metastasis via autophagic degradation of cathepsin S in cervical cancer cells. Cell Death Dis. 2017, 8, e3089. [Google Scholar] [CrossRef]
- Palkina, K.A.; Ipatova, D.A.; Shakhova, E.S.; Balakireva, A.V.; Markina, N.M. Ther. Potential of Hispidin—Fungal and Plant Polyketide. J. Fungi 2021, 7, 323. [Google Scholar] [CrossRef]
- Lv, L.-X.; Zhou, Z.-X.; Zhou, Z.; Zhang, L.-J.; Yan, R.; Zhao, Z.; Yang, L.-Y.; Bian, X.-Y.; Jiang, H.-Y.; Li, Y.-D. Hispidin induces autophagic and necrotic death in SGC-7901 gastric cancer cells through lysosomal membrane permeabilization by inhibiting tubulin polymerization. Oncotarget 2017, 8, 26992. [Google Scholar] [CrossRef]
- Chandimali, N.; JIN, W.Y.; KWON, T. Combination effects of hispidin and gemcitabine via inhibition of stemness in pancreatic cancer stem cells. Anticancer Res. 2018, 38, 3967–3975. [Google Scholar] [CrossRef] [PubMed]
- Ahn, W.-S.; Kim, D.-J.; Chae, G.-T.; Lee, J.-M.; Bae, S.-M.; Sin, J.-I.; Kim, Y.-W.; Namkoong, S.-E.; Lee, I. Natural killer cell activity and quality of life were improved by consumption of a mushroom extract, Agaricus blazei Murill Kyowa, in gynecological cancer patients undergoing chemotherapy. Int. J. Gynecol. Cancer 2004, 14, 589–594. [Google Scholar] [CrossRef] [PubMed]
- Hetland, G.; Tangen, J.-M.; Mahmood, F.; Mirlashari, M.R.; Nissen-Meyer, L.S.H.; Nentwich, I.; Therkelsen, S.P.; Tjønnfjord, G.E.; Johnson, E. Antitumor, anti-inflammatory and antiallergic effects of Agaricus blazei mushroom extract and the related medicinal Basidiomycetes mushrooms, Hericium erinaceus and Grifola frondosa: A review of preclinical and clinical studies. Nutrients 2020, 12, 1339. [Google Scholar] [CrossRef] [PubMed]
- Tangen, J.-M.; Tierens, A.; Caers, J.; Binsfeld, M.; Olstad, O.K.; Trøseid, A.-M.S.; Wang, J.; Tjønnfjord, G.E.; Hetland, G. Immunomodulatory effects of the Agaricus blazei Murrill-based mushroom extract AndoSan in patients with multiple myeloma undergoing high dose chemotherapy and autologous stem cell transplantation: A randomized, double blinded clinical study. Biomed Res. Int. 2015, 2015, 718539. [Google Scholar] [CrossRef]
- Lee, S.H.; Hwang, H.K.; Kang, C.M.; Lee, W.J. Potential impact of Phellinus linteus on adherence to adjuvant treatment after curative resection of pancreatic ductal adenocarcinoma: Outcomes of a propensity score–matched analysis. Integr. Cancer Ther. 2019, 18, 1534735418816825. [Google Scholar] [CrossRef]
- Chen, W.; Tan, H.; Liu, Q.; Zheng, X.; Zhang, H.; Liu, Y.; Xu, L. A review: The bioactivities and pharmacological applications of Phellinus linteus. Molecules 2019, 24, 1888. [Google Scholar] [CrossRef]
- Zhang, C.-C.; Cao, C.-Y.; Kubo, M.; Harada, K.; Yan, X.-T.; Fukuyama, Y.; Gao, J.-M. Chemical constituents from Hericium erinaceus promote neuronal survival and potentiate neurite outgrowth via the TrkA/Erk1/2 pathway. Int. J. Mol. Sci. 2017, 18, 1659. [Google Scholar] [CrossRef]
- Tung, S.-Y.; Lee, K.-C.; Lee, K.-F.; Yang, Y.-L.; Huang, W.-S.; Lee, L.-Y.; Chen, W.-P.; Chen, C.-C.; Teng, C.-C.; Shen, C.-H. Apoptotic mechanisms of gastric cancer cells induced by isolated erinacine S through epigenetic histone H3 methylation of FasL and TRAIL. Food Funct. 2021, 12, 3455–3468. [Google Scholar] [CrossRef]
- Kuo, H.-C.; Kuo, Y.-R.; Lee, K.-F.; Hsieh, M.-C.; Huang, C.-Y.; Hsieh, Y.-Y.; Lee, K.-C.; Kuo, H.-L.; Lee, L.-Y.; Chen, W.-P. A comparative proteomic analysis of Erinacine A’s inhibition of gastric cancer cell viability and invasiveness. Cell. Physiol. Biochem. 2017, 43, 195–208. [Google Scholar] [CrossRef]
- Lee, K.-C.; Lee, K.-F.; Tung, S.-Y.; Huang, W.-S.; Lee, L.-Y.; Chen, W.-P.; Chen, C.-C.; Teng, C.-C.; Shen, C.-H.; Hsieh, M.-C. Induction apoptosis of erinacine a in human colorectal cancer cells involving the expression of TNFR, fas, and fas ligand via the JNK/p300/p50 signaling pathway with histone acetylation. Front. Pharmacol. 2019, 10, 1174. [Google Scholar] [CrossRef]
- TSANG, K.W.; Lam, C.; Yan, C.; Mak, J.; Ooi, G.; Ho, J.; Lam, B.; Man, R.; Sham, J.; Lam, W. Coriolus versicolor polysaccharide peptide slows progression of advanced non-small cell lung cancer. Resp. Med. 2003, 97, 618–624. [Google Scholar] [CrossRef] [PubMed]
- Ohwada, S.; Ikeya, T.; Yokomori, T.; Kusaba, T.; Roppongi, T.; Takahashi, T.; Nakamura, S.; Kakinuma, S.; Iwazaki, S.; Ishikawa, H. Adjuvant immunochemotherapy with oral Tegafur/Uracil plus PSK in patients with stage II or III colorectal cancer: A randomised controlled study. Br. J. Cancer 2004, 90, 1003–1010. [Google Scholar] [CrossRef] [PubMed]
- Li, J.W.-H.; Vederas, J.C. Drug discovery and natural products: End of an era or an endless frontier? Science 2009, 325, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Luan, X.; Zheng, M.; Tian, X.-H.; Zhao, J.; Zhang, W.-D.; Ma, B.-L. Synergistic mechanisms of constituents in herbal extracts during intestinal absorption: Focus on natural occurring nanoparticles. Pharmaceutics 2020, 12, 128. [Google Scholar] [CrossRef]
- Jürgenliemk, G.; Nahrstedt, A. Dissolution, solubility and cooperativity of phenolic compounds from Hypericum perforatum L. in aqueous systems. Int. J. Pharm. Sci. 2003, 58, 200–203. [Google Scholar]
- Cai, T.-Y.; Zhang, Y.-R.; Ji, J.-B.; Xing, J. Investigation of the component in Artemisia annua L. leading to enhanced antiplasmodial potency of artemisinin via regulation of its metabolism. J. Ethnopharmacol. 2017, 207, 86–91. [Google Scholar] [CrossRef]
- Gröning, R.; Breitkreutz, J.; Müller, R. Physico-chemical interactions between extracts of Hypericum perforatum L. and drugs. Eur. J. Pharm. Biopharm. 2003, 56, 231–236. [Google Scholar] [CrossRef]
- Zhuang, Y.; Yan, J.; Zhu, W.; Chen, L.; Liang, D.; Xu, X. Can the aggregation be a new approach for understanding the mechanism of Tradit. Chin. Medicine? J. Ethnopharmacol. 2008, 117, 378–384. [Google Scholar] [CrossRef]
- Mukherjee, P.K.; Harwansh, R.K.; Bhattacharyya, S. Bioavailability of herbal products: Approach toward improved pharmacokinetics. In Evidence-Based Validation of Herbal Medicine; Elsevier: Amsterdam, The Netherlands, 2015; pp. 217–245. [Google Scholar]
- Phansalkar, P.S.; Zhang, Z.; Verenich, S.; Gerk, P.M. Pharmacokinetics and bioavailability enhancement of natural products. In Natural Products for Cancer Chemoprevention; Springer: Berlin/Heidelberg, Germany, 2020; pp. 109–141. [Google Scholar]
- Khedekar, K.; Mittal, S. Self emulsifying drug delivery system: A review. Int. J. Pharm. Sci. Res. 2013, 4, 4494. [Google Scholar]
- Singh, N.; Rai, S.; Bhattacharya, S. A Conceptual Analysis of solid Self-emulsifying drug Delivery System and its Associate Patents for the Treatment of Cancer. Recent Pat. Nanotechnol 2021, 15, 92–104. [Google Scholar] [CrossRef]
- Murugesan, M.P.; Ratnam, M.V.; Mengitsu, Y.; Kandasamy, K. Evaluation of anti-cancer activity of phytosomes formulated from Aloe vera extract. Mat. Today Proc. 2021, 42, 631–636. [Google Scholar] [CrossRef]
- Flaig, T.W.; Gustafson, D.L.; Su, L.-J.; Zirrolli, J.A.; Crighton, F.; Harrison, G.S.; Pierson, A.S.; Agarwal, R.; Glodé, L.M. A phase I and pharmacokinetic study of silybin-phytosome in prostate cancer patients. Investig. New Drugs 2007, 25, 139–146. [Google Scholar] [CrossRef]
- Flaig, T.W.; Glodé, M.; Gustafson, D.; van Bokhoven, A.; Tao, Y.; Wilson, S.; Su, L.J.; Li, Y.; Harrison, G.; Agarwal, R. A study of high-dose oral silybin-phytosome followed by prostatectomy in patients with localized prostate cancer. Prostate 2010, 70, 848–855. [Google Scholar] [CrossRef] [PubMed]
- Alhakamy, N.A.; Fahmy, U.A.; Eldin, S.M.B.; Ahmed, O.A.; Aldawsari, H.M.; Okbazghi, S.Z.; Alfaleh, M.A.; Abdulaal, W.H.; Alamoudi, A.J.; Mady, F.M. Scorpion Venom-Functionalized Quercetin Phytosomes for Breast Cancer Management: In Vitro Response Surface Optimization and Anticancer Activity against MCF-7 Cells. Polymers 2021, 14, 93. [Google Scholar] [CrossRef] [PubMed]
- Alhakamy, N.A.; Badr-Eldin, S.M.; Fahmy, U.A.; Alruwaili, N.K.; Awan, Z.A.; Caruso, G.; Alfaleh, M.A.; Alaofi, A.L.; Arif, F.O.; Ahmed, O.A. Thymoquinone-loaded soy-phospholipid-based phytosomes exhibit anticancer potential against human lung cancer cells. Pharmaceutics 2020, 12, 761. [Google Scholar] [CrossRef]
- Muhamad, N.; Plengsuriyakarn, T.; Na-Bangchang, K. Application of active targeting nanoparticle delivery system for chemoTher. drugs and traditional/herbal medicines in cancer therapy: A systematic review. Int. J. Nanomed. 2018, 13, 3921. [Google Scholar] [CrossRef]
- Rehman, M.U.; Khan, A.; Imtiyaz, Z.; Ali, S.; Makeen, H.A.; Rashid, S.; Arafah, A. Current Nano-Therapeutic Approaches Ameliorating Inflammation in Cancer Progression. Semin. Cancer Biol. 2022; Online ahead of print. [Google Scholar]
- Salunkhe, R.; Gadgoli, C.; Naik, A.; Patil, N. Pharmacokinetic Profile and Oral Bioavailability of Diosgenin, Charantin, and Hydroxychalcone from a Polyherbal Formulation. Front. Pharmacol. 2021, 12, 629272. [Google Scholar] [CrossRef]
- Peterson, B.; Weyers, M.; Steenekamp, J.H.; Steyn, J.D.; Gouws, C.; Hamman, J.H. Drug bioavailability enhancing agents of natural origin (bioenhancers) that modulate drug membrane permeation and pre-systemic metabolism. Pharmaceutics 2019, 11, 33. [Google Scholar] [CrossRef]
- Apolone, G.; Joppi, R.; Bertele, V.; Garattini, S. Ten years of marketing approvals of anticancer drugs in Europe: Regulatory policy and guidance documents need to find a balance between different pressures. Br. J. Cancer 2005, 93, 504–509. [Google Scholar] [CrossRef][Green Version]
- Zhang, J.; Onakpoya, I.J.; Posadzki, P.; Eddouks, M. The safety of herbal medicine: From prejudice to evidence. Evid. Based Complement. Alternat. Med. 2015, 2015, 316706. [Google Scholar] [CrossRef]
- Mazzanti, G.; Menniti-Ippolito, F.; Moro, P.A.; Cassetti, F.; Raschetti, R.; Santuccio, C.; Mastrangelo, S. Hepatotoxicity from green tea: A review of the literature and two unpublished cases. Eur. J. Clin. Pharmacol. 2009, 65, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Webster, D.; Cao, J.; Shao, A. The safety of green tea and green tea extract consumption in adults–results of a systematic review. Regul. Toxicol. Pharmacol. 2018, 95, 412–433. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Schmitz, H.-J.; Baumgart, A.; Guedon, D.; Netsch, M.; Kreuter, M.-H.; Schmidlin, C.; Schrenk, D. Toxicity of green tea extracts and their constituents in rat hepatocytes in primary culture. Food Chem. Toxicol. 2005, 43, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Additives, E.P.o.F.; Food, N.S.a.t.; Younes, M.; Aggett, P.; Aguilar, F.; Crebelli, R.; Dusemund, B.; Filipič, M.; Frutos, M.J.; Galtier, P.; et al. Scientific opinion on the safety of green tea catechins. EFSA J. 2018, 16, e05239. [Google Scholar]
- Fasinu, P.S.; Bouic, P.J.; Rosenkranz, B. An overview of the evidence and mechanisms of herb–drug interactions. Front. Pharmacol. 2012, 3, 69. [Google Scholar] [CrossRef]
- Yadav, P.; Mahour, K.; Kumar, A. Standardization and evaluation of herbal drug formulations. J. Adv. Lab. Res. Biol. 2011, 2, 161–166. [Google Scholar]
- Bijauliya, R.K.; Alok, S.; Chanchal, D.K.; Kumar, M. A comprehensive review on standardization of herbal drugs. Int. J. Pharm. Sci. Res. 2017, 8, 3663–3677. [Google Scholar]
- Govindaraghavan, S.; Sucher, N.J. Quality assessment of medicinal herbs and their extracts: Criteria and prerequisites for consistent safety and efficacy of herbal medicines. Epilepsy Behav. 2015, 52, 363–371. [Google Scholar] [CrossRef]
- Wang, J.; Guo, Y.; Li, G.L. Current Status of Standardization of Traditional Chinese Medicine in China. Evid. Based Complement. Altern. Med. 2016, 2016, 9123103. [Google Scholar] [CrossRef]
- Downer, S.; Berkowitz, S.A.; Harlan, T.S.; Olstad, D.L.; Mozaffarian, D. Food is medicine: Actions to integrate food and nutrition into healthcare. BMJ 2020, 369, m2482. [Google Scholar] [CrossRef]
- Chen, X.; Yue, W.; Tian, L.; Li, N.; Chen, Y.; Zhang, L.; Chen, J. A plant-based medicinal food inhibits the growth of human gastric carcinoma by reversing epithelial–mesenchymal transition via the canonical Wnt/β-catenin signaling pathway. BMC Complement. Med. Ther. 2021, 21, 137. [Google Scholar] [CrossRef] [PubMed]
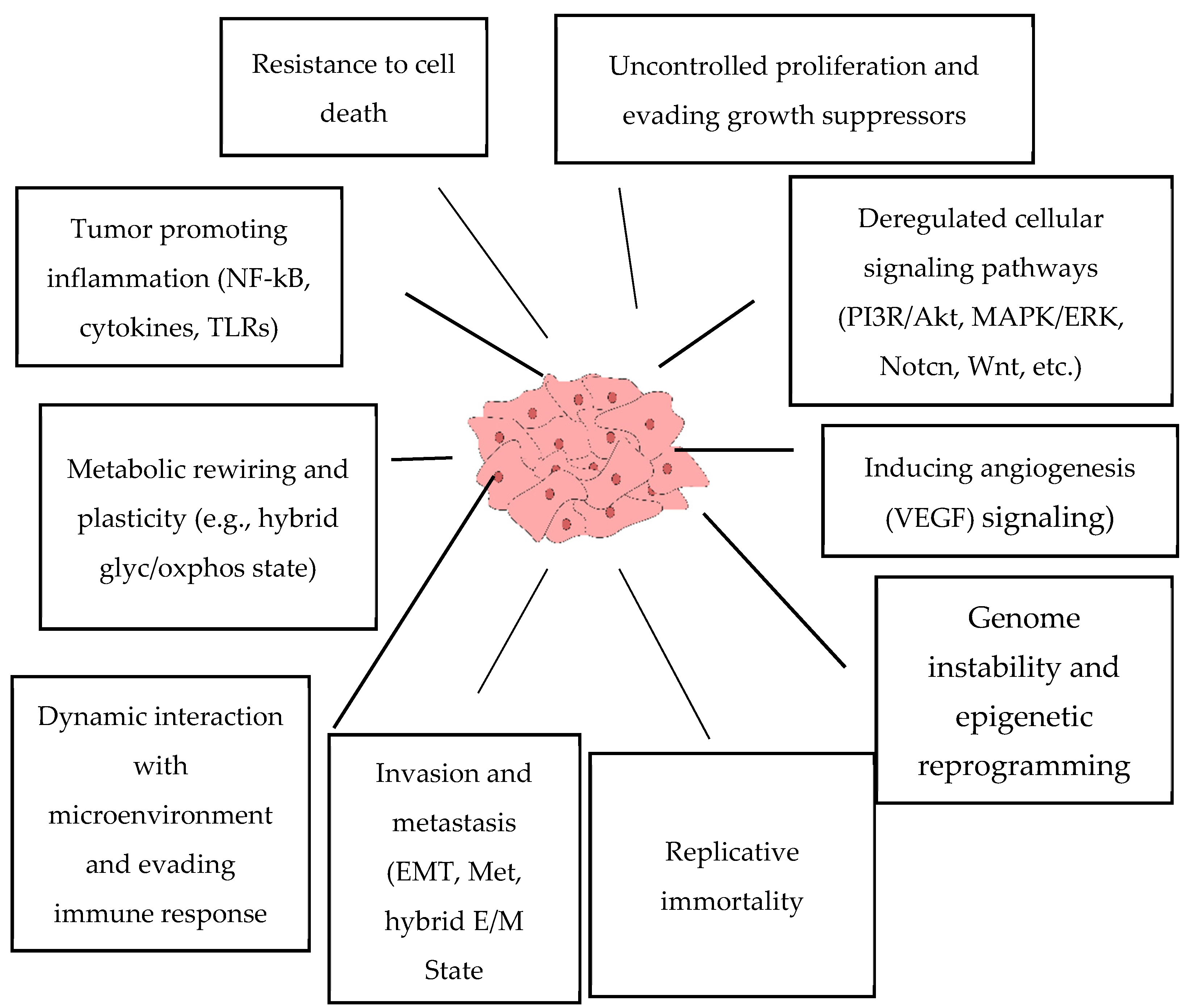
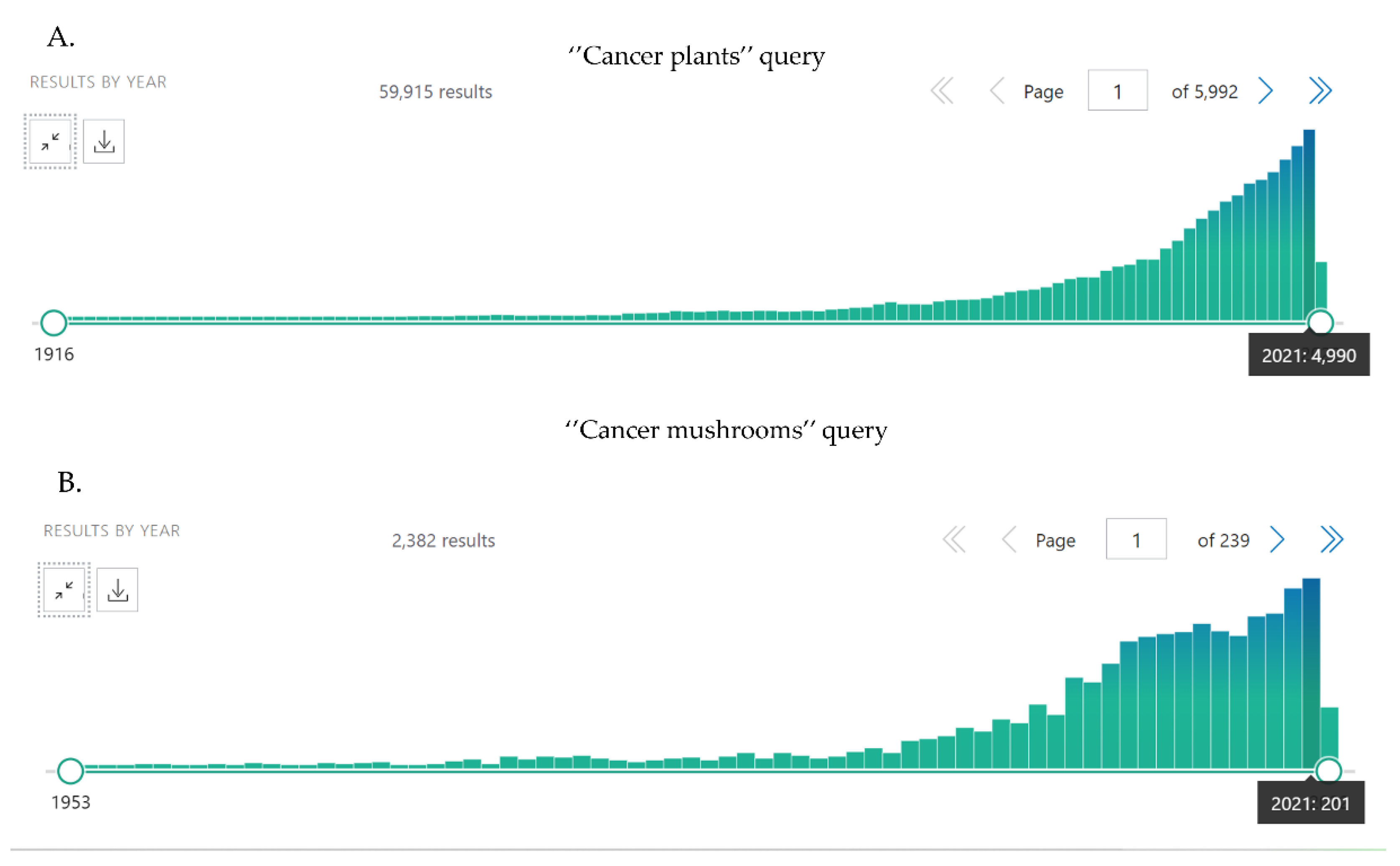
| Plant | Active Compounds | Effects | References |
|---|---|---|---|
| Aristolochia ringens | Triterpenes | Cytotoxic effects of root extract against several cancer cell lines, solid tumors, and leukemia models in vivo. | [74,75] |
| Beilschmiedia acuta | Flavonoids, saponins, alkaloids | Bark-derived extract inhibited proliferation and induced apoptosis in human leukemia CCRF-CEM cells, but was less toxic to human normal hepatocytes AML12 cells. | [76] |
| Dorstenia psilurus | Alkaloids, phenolic compounds, flavonoids | Cytotoxic and anti-proliferative effects in HeLa cancer cells; caspase 3/7 up-regulation and induction of apoptosis in HL-60 cells. | [77,78] |
| Echinops giganteus | Brominated oleanolide | Root extract inhibited proliferation and induced apoptosis in leukemia CCRF-CEM cells. | [79,80] |
| Imperata cylindrica | Saponins, flavonoids Antineoplastic activity: arundoin, daucosterol | Cytotoxicity against the panel of cancer cells. Root extracts induced apoptosis, increased caspase 3/7 activity, and significant down-regulated MMPs. | [77,81] |
| Piper capense | Alkaloids, polyphenols, saponins | Seeds extracts were cytotoxic for a number of cancer cell lines. Fruits extracts induced the shrinkage of tumor size in animal models by inhibiting the development of VM tubes and microvessel density. | [79,82] |
| Polyscia fulva | Anthocyanins, flavonoids, triterpenes, saponins | Roots extracted inhibited proliferation and induced apoptosis in leukemia CCRF-CEM cells via the enhanced production of ROS. It was significantly less toxic for human normal hepatocytes AML12 cells. | [76] |
| Plant | Active Compounds | Effects | References |
|---|---|---|---|
| Achyrocline satureioides | Achyrobichalcone, 3-O-methylquercetin, and other flavonoids | In vitro cytotoxicity and apoptosis in human breast cancer cells; inhibition of c-MYC and ERK/JNK in glioma cell lines. | [82,131] |
| Aloysia polystachya | Flavonoids | In vitro apoptosis in human colorectal cancer cells, and a decrease in the percentage of cancer stem cells (CSCs). In vivo inhibition of tumor growth in non-toxic doses. | [132] |
| Azorella glabra | Mulinic acid, azorellane terpenoids | G0/G1 cell cycle arrest and apoptosis in AML cell lines. A slight decrease in the survival of non-tumor cells | [133] |
| Ephedra chilensis | Terpens and fatty acids | IC50 of non-polar extracts for one breast cancer and three colon cell lines was at the level of doxorubicin; in vitro cytotoxicity for normal colon epithelium cells was less than doxorubicin. | [134] |
| Croton lechleri | Taspine | Leaf extracts exhibited cytotoxic antiproliferative effects on HeLa and SK23 cells in vitro, and antitumor effects in mice in vivo; moderate toxicity to mice. | [135,136] |
| Laetia corymbulosa | Corymbulosins B, C, D, E, F, G | Bark extract exhibited cytotoxicity to panel of cancer cells. | [137] |
| Lepidium meyenii | macamide and macaene fractions | Macadamine displayed anticancer activities against multiple cancer cell lines. | [138] |
| Leptocarpha rivularis | Leptocarpin | Cytotoxic effects against several cancer cell lines. | [139] |
| Passiflora alata | Flavonoids and saponins | In vitro cytotoxicity against the set of cancer cell lines, and in vivo antitumor activity against sarcoma S180-bearing mice with low general toxicity. | [140] |
| Thevetia peruviana | Thevetiaflavone, and individual cardiac glycosides | Fruit extract exhibited cytostatic and cytotoxic effects in cancer cell lines with moderate toxicity to non-tumor cells. | [141] |
| Clinical Formulation | Composition | Type of Cancer | Effects | References |
|---|---|---|---|---|
| Aidi Injection | Mylabris Phalerata, Astragalus Membranaceus, Panax Ginseng, Acanthopanax Senticosus | Different solid tumors, gynecologic tumors | Suppression of proliferation, migration, invasion, angiogenesis, and metastasis. Decreased p-PI3K and Bcl-xL in liver cancer cells. Induction of apoptosis. In Clinic: improved overall survival, the quality of life, and the effectiveness of chemotherapy. | [182,183,184] |
| Fufangkushen Injection | Sophora flavescens, Heterosmilacis Japonica | Different solid tumors | Reduced proliferation, tumor growth, and TRPV1-ERK phosphorylation; decreased IFN-γ, IL-6, and KC levels in S-180 sarcoma. Induced apoptosis via up-regulation of caspase-3 and Fas in esophageal carcinoma. In Clinic: improved the quality of life and the effectiveness of chemotherapy. | [185,186] |
| Kanglaite injection | Coix lacryma-jobi | Non-small cell lung cancer, colorectal cancer, pancreatic cancer | In vitro suppressed cell growth and induced apoptosis via up-regulation of p53, Fas, and caspase-3. In vivo down-regulation of the PI3K/Akt/mTOR signaling pathway, and tumor growth suppression. | [187,188] |
| Kushen injection | Sophorae Flavescentis, Radix, Smilacis Glabrae Rhixoma | Different solid tumors | Immunomodulatory activity via regulation of macrophages and CD8+ T cells, TNFR1, NF-κB p65, and MAPK p38. | [189] |
| Qing-Dai | Baphicacanthus cusia, Polygonum tinctorium, Isatis indigotica | Acute promyelocytic leukemia | Down-regulation of NF-κB, Iap1, Iap2, Bcl-2, BCL-xL, cyclin D1, and c-Myc; inhibition of angiogenesis by reducing JAK/STAT3, VEGFR2, ERK 1/2, Ang-1, PDGFB, and MMP2. Immunomodulatory activity through impact on CD4+ CD25+ Treg cells. | [190] |
| Tien-Hsien | Cordyceps sinensis, Oldenlandia diffusa, Indigo Pulverata Levis, Polyporus umbellatus, Radix Astragali, Panax ginseng, Solanum nigrum, Pogostemon cablin, Atractylodis Macrocephalae Rhizoma, Trichosanthes Radix, Clematis Radix, Margarite, Ligustrum lucidum, Glycyrrhizae Radix | Acute promyelocytic leukemia, breast cancer | Down-regulation of DNMT1, cyclin A, cyclin B1, p-AKT, Bcl-2, Akt/mTOR, Stat3, and ERK; induction of p21, p15, and apoptosis. Suppression of angiogenesis, metastasis, and tumor growth. Radiosensitization and immunomodulatory activity. | [191,192,193] |
| Zeng-Sheng-Ping | Sophora tonkinensis, Polygonum bistorta, Prunella vulgaris, Sonchus brachyotus, Dictamnus dasycarpus, Dioscorea bulbifera | Alimentary tract cancer; oral, lung, and bladder cancer | Inhibition of EGFR and Notch signaling. In Clinic: slowed down tumor growth and increased overall survival. | [194,195,196] |
| Plant | Biologically Active Compounds | Effects | References |
|---|---|---|---|
| Allium sativum | Allicin, alliin, diallyl disulfide, diallyl trisulfide, Z-ajoene, S-allyl cysteine, S-propargyl-l-cysteine, S-allyl cysteine | Multiple anticancer effects and known molecular mechanisms of both crude extracts and individual compounds. Suppression of angiogenesis and migration in vivo. | [314] |
| Arctium lappa | Arctigenin, lappaol F, stigmasterol ß-sitosterol | Suppression of growth, invasion, and migration of cancer cell lines. Inhibition of hippo-signaling pathway. Reduction in tumor growth in vivo. | [315,316,317] |
| Centaurea solstitialis | Solstitialin A | Cytotoxic and cytostatic effects in a panel of cancer cell models | [276,318] |
| Ebenus boissieri | Antiproliferative and cytotoxic effects in human breast, cervical, and lung cancer cell lines. Induction of TNF-α expression. | [319,320,321] | |
| Rosmarinus officinalis | Carnosol, carnosic acid, sageone, rosmarinic acid | Multiple antineoplastic effects in vitro and in vivo with known molecular mechanisms, including epigenetic regulation. | [322] |
| Menyanthes trifoliata | Betulinic acid, syringic acid, ellagic acid, rutin, chlorogenic acid | Cell cycle G2/M arrest and apoptosis in grade IV glioma. No toxicity to normal human astrocytes. | [323] |
| Vitis vinifera | Viniferin, resveratrol | Multiple anticancirogenic and antineoplastic effects with known molecular mechanisms. | [324,325] |
| Viscum album | Iscodor, helixor A, lectins (ML-I, ML-II, and ML-III), viscotoxins, polysaccharides, phenolic compounds | Cytostatic and cytotoxic effects in vitro and in vivo. Immunomodulatory activity and reduction in cancer-related fatigue in clinical studies. Helixor A and Iscador are used in Europe as adjuvants in cancer therapy. | [326,327] |
| Plant | Biologically Active Compounds | Effects | References |
|---|---|---|---|
| Aristolochia foetida | β-sitosterol, stigmasterol, and other compounds | Apoptosis in MCF-7 cancer cells with less toxicity to non-tumor cells. | [336] |
| Asimina triloba | Acetogenins | Extracts from different parts of plant suppressed proliferation; induced apoptosis in AGS and HeLa cells; inhibited inflammatory makers NO, TNF-α, IL-6, and iNOS (inducible nitric oxide synthase). | [337] |
| Capraria biflora | Biflorin | Different anticancer effects in a number of tumor cell lines. Inhibition of c-MYC expression. | [338,339] |
| Echinacea purpurea | Echinacoside, alkylamides | Reduced proliferation, increased level of ROS, caspase 3 activity, and apoptosis in human lung cells. Inhibition of Wnt/β-catenin pathway. Immunomodulatory activity in vivo. | [340,341] |
| Sanguinaria canadensis | Sanguinarine, chelerythrine, berberine | Cytotoxic and antiproliferative effects in melanoma and child ALL cell lines. Induction of apoptosis by cIAP1, cIAP2, and XIAP suppression in pre-ALL cell lines. Sanguinarine and berberine binds G-quadruplex in oncogenes and telomeres. | [342,343,344] |
| Plant | Active Compounds | Effects | References |
|---|---|---|---|
| Terminalia ferdinandiana | Tannins, flavonoids: gallic acid, ellagic acid, lutein, hesperitin, kaempferol, luteolin, and quercetin | Antiproliferative and proapoptotic activity in cancer cell lines. No toxicity to human dermal fibroblasts and shrimp Artemia franciscana bioassay. | [348,349] |
| Tasmannia lanceolata | Phenolic acids: coumaric acid, chlorogenic acid. Flavonoids: quercetin, quercetin 3-rutinoside, and anthocyanin (cyanidin 3-rutinoside) | Cytotoxicity to different cancer cell models with non-significant effects on normal colon, stomach, and intestine cells. | [350] |
| Davidsonia pruriens | Anthocyanin compounds, flavanoids | Significant cytotoxicity to a panel of cancer cell models and low toxicity in the Artemia nauplii bioassay. | [351,352] |
| Elaeocarpus angustifolius | Not identified | Significant cytotoxicity to Hela and Caco-2 cell models and low toxicity in the Artemia nauplii bioassay. | [352] |
| Pittosporum angustifolium | Alcaloids, saponins | Antiproliferative effects of 7 saponins with IC50 values in a range of 1.74–34.1 µM for MCF7, HaCaT, LN18, and 5637 cancer cells. | [353] |
| Mushroom | Active Compounds | Effects | References |
|---|---|---|---|
| Agaricus subrufescens (“mushroom of the sun”) | β-glucans (β-(1–3) linked backbone with (1–6) linked side branches); ergosterol | Pre-clinic: various immune stimulatory response. Clinic: increased activity of natural killer (NK) cells, and improved quality of life; increased number of plasmacytoid dendritic cells (DCs), Tregs, IL-5, and IL-7 in the blood. | [470,471,472] |
| Phellinus linteus | Polysaccharides, hispolon, phellinulin A, atractylenolide I, phellifuropyranone, meshimakobnol A, and meshimakobnol B | Pre-clinic: down-regulation of PI3K/AKT, ERK1/2, NF-kB Snail and Twist, cyclin D1 and -E, MMP-2 and -9, TGF-α; increased p53, p21, p27, and Bax; suppression of pancreatic CSCs. In clinic: Disease-free and overall survival of pancreatic cancer patients after tumor resection. | [469,473,474] |
| Hericium erinaceus (Lion’s mane) | 4-chloro-3,5-dimethoxybenzoic methyl ester, erinacine A, herierin III, herierin IV, and erinacerin G | The epigenetic regulation of FasL and TRAIL; sustained phosphorylation of FAK/AKT/p70S6K and the PAK1 pathways; generation of ROS; apoptosis via activation of JNK, p300, and NFκB p50; increased expression of TNFR, Fas, and FasL. | [475,476,477,478] |
| Trametes versicolor (Turkey tail) | Protein polysaccharide beta-glucan β-1,4 main chain with β-1,3 and β-1,6 side chains (PSKs) | In clinic: PSK, including adjuvant immunochemotherapy, significantly prolonged 5-year survival and disease-free rate for patients with gastric and colorectal cancer; slows progression of advanced non-small cell lung cancer. | [479,480] |
| High Priority | ||
| Plants | Mushrooms | Individual Compounds |
| Cephalotaxus harringtonia, Oldenlandia diffusa, Scutellaria barbata, Curcuma longa, Xanthium ssp., Zingiber officinalis, Hypericum perforatum, Glycyrrhiza glabra, Silybum marianum, Panax americanus and P. ginseng, Aloe vera and A. arborescence, Tabebuia impetiginosa, Viscum album, Allium sativum, Vitis vinifera, Rosmarinus officinalis, Echinacea purpurea, Sanguinaria canadensis | Lentinula edodes (Shiitake), Ganoderma lucidum (Reishi), Grifola frondosa (Maitake), Cordyceps sinensis and C. militaris, Agaricus blazei, Trametes versicolor, Phellinus linteus | Quercetin, kaempferol, ginsenosides (especially Rg3), silibinin, isoliquiritigenin, (−)- epicatechin, oleanolic acid, ursolic acid, hyperforin, hypericin, xanthatin, curcumin, withaferin A, withanone, scutellarein, scutellarin, homoharringtonine and its semi-synthetic derivates, chlorogenic acid, caffeic acid, carnosol, rosmarinic acid, resveratrol, iscodor, helixor A, shogaol, boswellic acids, hispolon, lentinan, cordycepin, echinacoside, and myricetin |
| Secondary priority | ||
| Betula pubescens, Eremophila galeata, Combretum caffrum, Acacia nilotica, Guera senegalis, Tasmannia lanceolata, Davidsonia pruriens, Elaeocarpus angustifolius, Pittosporum angustifolium, Terminalia ferdinandiana, Aristolochia ringens, Beilschmiedia acuta, Dorstenia psilurus, Aristolochia ringens, Beilschmiedia acuta, Dorstenia psilurus, Echinops giganteus, Imperata cylindrica, Piper capense, Polyscia fulva, Achyrocline satureioides, Aloysia polystachya, Azorella glabra, Ephedra chilensis, Croton lechleri, Laetia corymbulosa, Lepidium meyenii, Leptocarpha rivularis, Passiflora alata, Thevetia peruviana, Menyanthes trifoliata, Ebenus boissieri, Centaurea solstitialis, Arctium lappa, Capraria biflora, Asimina triloba, Aristolochia foetida | Hericium erinaceus (Lion’s mane), Inonotus obliquus (Chaga) | Gallic acid, combrestastatins, pyrogallol, betulinic acid, guieranone B, harringtonine, isoharringtonine, and doxyharringtonine, aloe-emodin, aloins, leptocarpin, macamide and macaene, corymbulosins, taspine, mulinic acid, achyrobichalcone, 3-O-methylquercetin, arctigenin, lappaol F, solstitialin A, sageone, biflorin, acetogenins, β-sitosterol, stigmasterol, sanguinarine, gandoderic acid, grifolin, illudin-S, lapachol and β-lapachone, carthamidin, carnosic acid, hispidin, inotodiol, syringic acid, p-coumaric acid, caffeoyl quinic acids, viniferin, lectins (ML-I, ML-II, and ML-III) from Viscum album |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kirdeeva, Y.; Fedorova, O.; Daks, A.; Barlev, N.; Shuvalov, O. How Should the Worldwide Knowledge of Traditional Cancer Healing Be Integrated with Herbs and Mushrooms into Modern Molecular Pharmacology? Pharmaceuticals 2022, 15, 868. https://doi.org/10.3390/ph15070868
Kirdeeva Y, Fedorova O, Daks A, Barlev N, Shuvalov O. How Should the Worldwide Knowledge of Traditional Cancer Healing Be Integrated with Herbs and Mushrooms into Modern Molecular Pharmacology? Pharmaceuticals. 2022; 15(7):868. https://doi.org/10.3390/ph15070868
Chicago/Turabian StyleKirdeeva, Yulia, Olga Fedorova, Alexandra Daks, Nikolai Barlev, and Oleg Shuvalov. 2022. "How Should the Worldwide Knowledge of Traditional Cancer Healing Be Integrated with Herbs and Mushrooms into Modern Molecular Pharmacology?" Pharmaceuticals 15, no. 7: 868. https://doi.org/10.3390/ph15070868
APA StyleKirdeeva, Y., Fedorova, O., Daks, A., Barlev, N., & Shuvalov, O. (2022). How Should the Worldwide Knowledge of Traditional Cancer Healing Be Integrated with Herbs and Mushrooms into Modern Molecular Pharmacology? Pharmaceuticals, 15(7), 868. https://doi.org/10.3390/ph15070868







