Commercially Available Enteric Empty Hard Capsules, Production Technology and Application
Abstract
1. Introduction
2. Enterosolvency of Solid Dosage Forms
3. Enteric Polymers
3.1. pH-Dependent Enteric Polymers
3.2. Gel-Forming Polymers for Enteric Coating
4. Related Excipients Used in the Formulation of the Capsule Shell
5. Brief Description of the Production and Filling of Hard Capsules
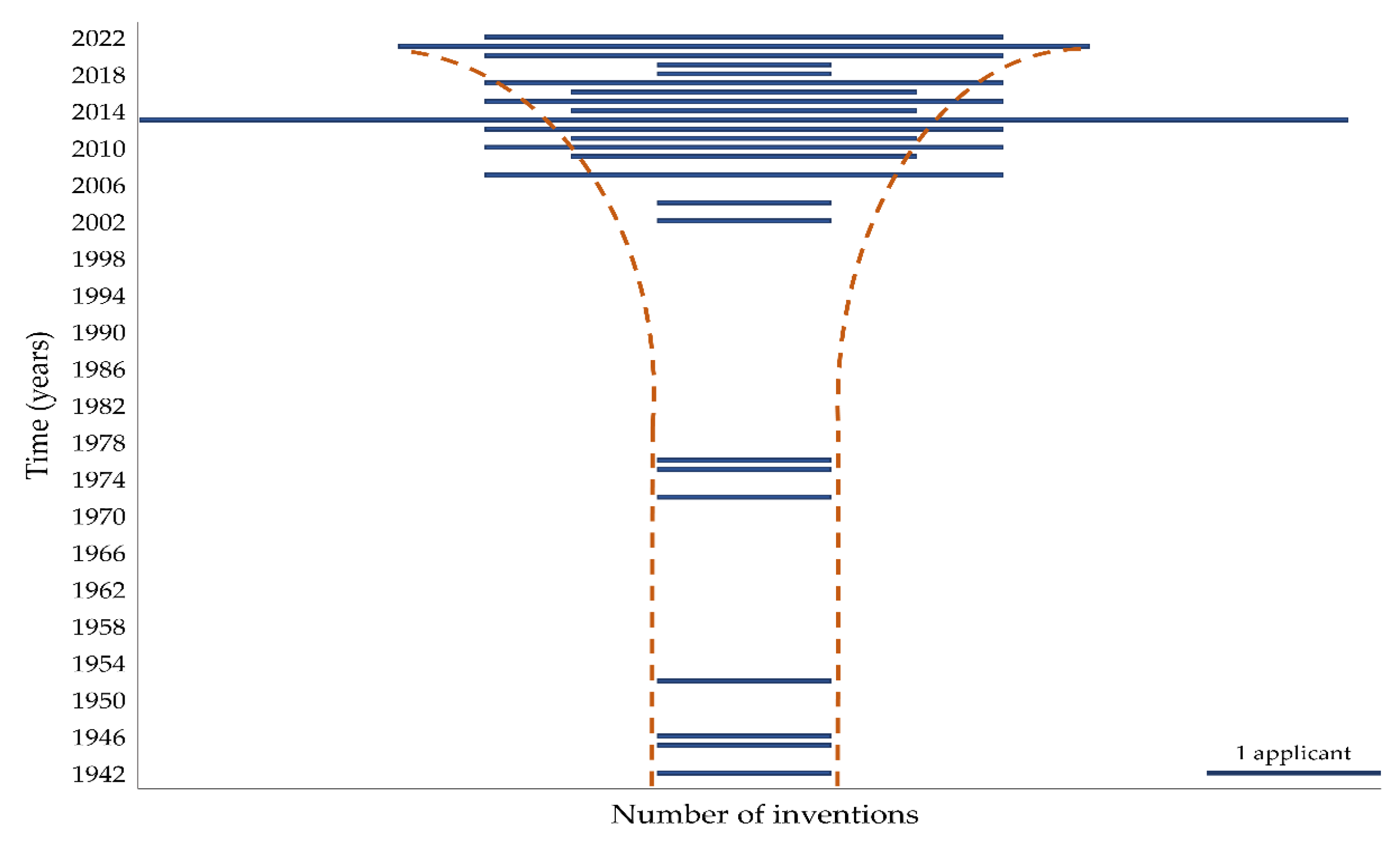
6. Patent Research of Empty Enteric Capsules
7. Commercially Hard Capsules Prepared by ECDDT
7.1. ECDDT Prepared from pH-Dependent Polymers
7.1.1. AR Caps® Capsules
7.1.2. EnTRinsicTM Capsules
7.1.3. Vcaps® Enteric Capsules
7.2. ECDDT Prepared from Gel-Forming Polymers
7.2.1. DRcapsTM Capsules
7.2.2. EMBO CAPS® AP Capsules
7.2.3. BioVXR® Capsules
7.2.4. ACGcapsTM HD Capsules
8. Clinical Studies Performed with Enteric Capsules
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Publication 1 | Applicant | Priority | Composition 2 (Capsule; INNER SHELL of the Capsule; Outer Shell of the Capsule)/Technology |
|---|---|---|---|
| CN115068436 | Jiangsu Zodiac Pharmaceutical Co., Ltd. | 2022-06-23 | Inner shell (HPMC, and carrageenan, gellan gum, agar, and potassium ions); Outer shell (HPMCP, and Tween® 80) |
| CN112546016 | Zhejiang Wanli University | 2022-04-22 | Capsule (HPMCP, agar, potassium chloride, and Tween® 80 in ammonium) |
| CN114376982 | Guangdong Pharmacology University | 2022-01-18 | Inner shell (chitosan); Outer shell (Eudragit® L 100, and HPMC) |
| EP3881834 | Jiangsu Lefan Capsule Co., Ltd. | 2021-09-22 | Capsule (pullulan, carrageenan, glycerol, and SLS) |
| CN215875588 | Zhejiang Xinchang Kangping Capsule Co., Ltd. | 2021-08-30 | Capsule (gelated alginate)/Provided with a special positioning lock ring |
| CN112569204 | Hubei Humanwell Pharmaceutical Excipients Co., Ltd. | 2021-03-30 | Inner shell (HPMS, HPMC, and pullulan, guar gum, carrageenan, xanthan gum, agar, and emulsifier); Outer shell (HPMCP) |
| CN109846852 | Chongqing Hengsheng Pharmaceutical Capsule Co., Ltd. | 2021-02-26 | Capsule (HPMC, potassium chloride, calcium acetate/ carbonate/citrate, sodium alginate) |
| WO2022112422 | Capsugel®, France/ Belgium | 2020-11-25 | Inner shell (gelatin, starch, PVA, and HPMC); Outer shell (HPMCAS, HPMCP, CAP) |
| CN111467318 | Hubei Humanwell Pharmaceutical Excipients Co., Ltd. | 2020-07-31 | Inner shell (tragacanth, gellan gum, carrageenan, konjac gum, xanthan gum, guar gum, agar, and calcium ions, and emulsifier, and coated with chitin, and sodium carbonate) |
| KR102272658 | Suheung Capsule | 2020-05-26 | Capsule (gelatin, and methoxylamide pectin, and buffer, and emulsifier) |
| TW201912150 | Qualicaps® | 2019-04-01 | Capsule (HPMC, MC, HPC, and Eudragit® L, Eudragit® NE, Eudragit® NM, Eudragit® FS 30D and HPMCP, HPMCAS, CAP, and PVA, and plasticizer, and surfactant) |
| CN109528689 | Jiangsu Zodiac Pharmaceutical Co., Ltd. | 2018-12-24 | Inner shell (carrageenan, sesbania gum, maltitol, potassium chloride, PVAP); Outer shell (PVAP, and surfactant) |
| WO2019020134 | Novamed | 2017-07-24 | Capsule (HPMC, CMC, gelatin, and gellan gum, guar gum, tragacanth, agar, alginate) |
| CN106902093 | Huaqiao University | 2017-01-24 | Capsule (alginate, and gellan gum, xanthan gum, carragheen, konjac glucomannan, locust bean gum, guar gum, arabic gum, and potassium ions, and HPMC, HPC, CMC, HPS) |
| CN106924211 | Qingdao Blue Valley Pharmaceutical | 2017-01-18 | Inner shell (HPMC, and carrageenan); Outer shell (low acyl gellan gum, and calcium chloride, acetic acid, calcium lactate) |
| US2021220285 | Capsugel®, Belgium | 2016-07-22 | Capsule (HPMCAS, HPMCP, CAP, PVAP, and HPMC, and ammonia, ethanolamine) |
| KR102335455 | Shinetsu Chemical Co., Ltd. | 2016-07-12 | Capsule (HPMCAS, ammonia) |
| US9452141 | Dah Feng Capsule Co., Ltd. | 2015-05-01 | Outer shell (gellan gum, pectin, pullulan, HPMC, and potassium chloride) |
| CN104434870 | Shaoxing Kangke Capsule Co., Ltd. | 2015-03-25 | Capsule (gelatin, and SLS, and sodium alginate) |
| TWI587880 | Dah Feng Capsule Co., Ltd. | 2015-03-24 | Outer shell (gelatin/ poly triglucose, PVA, modified starch, HPMC, and gellan gum, carrageenan, and monovalent/ divalent salt) |
| CN104288120 | Shaoxing Kangke Capsule Co., Ltd. | 2014-10-16 | Capsule (HPMC, and pectin, and SLS, Tween® 80, and glycerol, PEG, and sodium alginate, agar, carrageenan) |
| CN103919752 | Lou Zhi | 2014-05-07 | Capsule (HPMCP, and PEG, glycerol, castor oil) |
| CN103520134 | Songlin Liu | 2013-10-29 | Capsule (HPMCP, and PPG, glycerol, castor oil) |
| CN103394093 | Chongqing Hengsheng Medicinal Capsules | 2013-08-11 | Capsule (HPMC, and carrageenan, and potassium chloride) |
| CN103394092 | Chongqing Hengsheng Medicinal Capsules | 2013-08-11 | Capsule (HPMC, and gellan gum, and calcium chloride) |
| CN103417511 | Chongqing Hengsheng Medicinal Capsules | 2013-08-11 | Capsule (HPMC, and carrageenan, and calcium chloride) |
| CN103432096 | Chongqing Hengsheng Medicinal Capsules | 2013-08-11 | Capsule (HPMC, and carrageenan and potassium citrate) |
| CN103330695 | Shaoxing Kangke Capsule Co., Ltd. | 2013-06-05 | Capsule (HPMC, and SLS, Tween® 80, glycerol, PEG, and sodium alginate, agar, carrageenan) |
| US10172803 | Capsugel®, Belgium | 2013-03-07 | Capsule (HPMC, and calcium stearate, magnesium stearate, carnauba wax, and guar gum, pectin, carrageenan, xanthan gum, locust bean gum, alginate, alginic acid, tamarind gum, glucomannan, agar, curdlan, gellan gum, collagen) |
| TWI554290 | Samsung Fine Chemicals | 2012-06-05 | Capsule (HPMC, HPMCP, and ammonia, sodium hydroxide, and potassium hydroxide) |
| JP2018058870 | Capsugel®, Belgium | 2012-05-02 | Capsule (Eudragit® L 100, Eudragit® S 100, Eudragit® RS 100, CAP, CAT, HPMCAS, HPMCP, HPMC, HEC, EC, HPC, CMC, MC, and sorbitan monoesters, sorbitan POE esters, glycerol, PEG, castor oil, SLS, TEC, triacetin); Other similar compounds are also listed. |
| US2015080479 | Son Jin Ryul, Park Eun Hee | 2012-03-26 | Capsule (HPMCP, and HPMC, and calcium hydroxide, and ammonia, sodium hydroxide, and potassium hydroxide, and TEC, triacetin, PEG, PPG, and surfactants) |
| EP3566698 | Capsugel®, Belgium | 2011-11-09 | Capsule (CAP, CAT, HPMCAS, CMEC, PVAP, and monovalent/divalent carbonates and hydroxides) |
| KR20120100305 | Samsung Fine Chemicals | 2011-03-03 | Capsule (HPMCP, and HPMC, and calcium hydroxide, and sodium hydroxide, potassium hydroxide, and hydrogenated corn syrup, TEC, triacetin, PEG, PPG) |
| EP3354335 | Capsugel®, Belgium | 2010-10-26 | Capsule (CAP, and HPMC, HPC, EC, MC, CMEC, HPMCAS, HPMCP, and sorbitan monoesters and POE esters, glycerol, PEG, glycol, ricin oleate, SLS, TEC, triacetin, and POE-POP-POE triblock polymers); Other similar compounds are also listed. |
| US9603933 | Son Jin Ryul, Baek Hyon Ho | 2010-10-21 | Capsule (HPMCP, and HPMC, and ammonia, sodium hydroxide, potassium hydroxide, calcium hydroxide, and SLS, sugar ester) |
| US8710105 | Son Jin Ryul, Baek Hyon Ho | 2010-06-11 | Capsule (HPMCP, HPMCAS, HPMC, MC, carrageenan, agar, sodium alginate, gellan gum, pectin, sodium hydroxide, ammonia, potassium hydroxide, calcium hydroxide) |
| US8852631 | Capsugel®, Belgium | 2009-09-24 | Capsule (gellan gum, and HPMC) |
| WO2011030952 | Samsung Fine Chemicals | 2009-09-11 | Capsule (gellan gum, and HPMCP, and HPMCAS, and HPMC, MC, and neutralizing agent) |
| CN101433526 | Shanghai Huiyuan Plant Capsule Co., Ltd. | 2007-11-13 | Outlet shell (HPS, carrageenan, and calcium chloride, sodium carbonate, and CA) |
| WO2009062356 | Shanghai Huiyuan Vegetal Capsule Co., Ltd. | 2007-11-13 | Capsule (PVAP acrylic resin, acetyl pullulan, CAP, 1, 2, 4-benzene tricarboxylic acid acetate, HPMCP, HPMC 1,2,4-benzenetricarboxylate, CAS, succinic acid, acetic acid, HPMC, and monovalent and divalent hydroxides and carbonates, and acetylated gellan gum, carrageenan, agar, pectin, sodium alginate, xanthan gum, scutellaria, paulownia gum, locust bean gum, red algae gum, tamarind gum, tara gum, sclerotium glucan, alginate, carbomers) |
| US2010113620 | Aston University | 2007-03-29 | Capsule (HPMC, and alginate, and gellan gum, sodium, and potassium ions, and PEG) |
| US6752953 | Yung Shin Pharmaceutical Co. | 2004-06-22 | Capsule (cellulose nitrate, CTA, CAP, MC, EC, HPC, HPMC, HPMCP, and Eudragit® L, Eudragit® S, Eudragit® E, Eudragit® RL, Eudragit® RS, and glycerin, PPG, PEG, DEP, DBP, DBS, TEC, acetyl TEC, acetyl TBC, triacyl glycerol, castor oil); Other similar compounds are also listed. |
| WO2004012701 | Ajit Singh, Meena Parashuraman | 2002-08-02 | Capsule (gelatin, and methacrylic acid copolymer, CAP, HPMCP, PVAP, and ammonia, sodium carbonate, sodium bicarbonate, sodium hydroxide, and TEC, DBP, DEP, benzyl phenyl formate, sorbitol, PEG) |
| US4138013 | Parke, Davis & Co. | 1976-08-27 | Capsule (HPMC, and CAP, gelatin, the copolymer of methacrylic acid, and methacrylic acid ester) |
| GB1529901 | Parke, Davis & Co. | 1975-04-17 | Capsule (gelatin and CAP, HPMCP, the copolymer of methacrylic acid with methacrylic acid ester) |
| DE2336807 | Parke, Davis & Co. | 1972-07-20 | Capsule (gelatin, and HPMCP with softener) |
| GB743014 | Kodak Ltd. | 1952-05-01 | Capsule (ethyl ether CP, CAP, both prepared in situ) |
| GB638284 | Parke, Davis & Co. | 1946-03-25 | Capsule (gelatin, and glycerol, and in situ prepared CAP, and sodium carbonate) |
| GB610538 | Gelatin Products Corp. | 1945-11-05 | Capsule (gelatin, and glycerol, in situ prepared CAP, and sodium carbonate) |
| US2390088 | Gelatin Products Corp. | 1942-09-24 | Capsule (gelatin, and glycerol, CAP, and sodium carbonate) |
References
- Stegemann, S.; Tian, W.; Morgen, M.; Brown, S. Hard Capsules in Modern Drug Delivery. In Pharmaceutical Formulation: The Science and Technology of Dosage Forms, 1st ed.; Tovey, G.D., Ed.; Royal Society of Chemistry: London, UK, 2018. [Google Scholar] [CrossRef]
- Overgaard, A.B.A.; Møller-Sonnergaard, J.; Christrup, L.L.; Højsted, J.; Hansen, R. Patients’ Evaluation of Shape, Size and Colour of Solid Dosage Forms. Pharm. World Sci. 2001, 23, 185–188. [Google Scholar] [CrossRef] [PubMed]
- Augsburger, L.L. Pharmaceutical Dosage Forms. In Pharmaceutical Dosage Forms; Augsburger, L.L., Hoag, S.W., Eds.; CRC Press: Boca Raton, FL, USA; London, UK, 2017. [Google Scholar] [CrossRef]
- Franc, A.; Kubová, K.; Elbl, J.; Muselík, J.; Vetchý, D.; Šaloun, J.; Opatřilová, R. Diazepam Filled Hard Capsules Intended for Detoxification of Patients Addicted to Benzodiazepines and Z-Drugs. Eur. J. Hosp. Pharm. 2019, 26, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Hannula, A.M.; Marvola, M.; Kopra, T. Release of Ibuprofen from Hard Gelatin Capsule Formulations: Effect of Various Additives and Filling Method. Acta Pharm. 1989, 98. [Google Scholar]
- Nair, R.; Vemuri, M.; Agrawala, P.; Kim, S. Investigation of Various Factors Affecting Encapsulation on the In-Cap Automatic Capsule-Filling Machine. AAPS PharmSciTech 2004, 5, 46–53. Available online: https://link.springer.com/content/pdf/10.1208/pt050457.pdf (accessed on 19 September 2022). [CrossRef]
- Gullapalli, R.P.; Mazzitelli, C.L. Gelatin and Non-Gelatin Capsule Dosage Forms. J. Pharm. Sci. 2017, 106, 1453–1465. [Google Scholar] [CrossRef] [PubMed]
- Sabadková, D.; Franc, A.; Muselík, J.; Neumann, D.; Vetchý, D. Pulsatile Drug Delivery Systems. Chem. Listy 2015, 109, 353–359. Available online: http://www.chemicke-listy.cz/ojs3/index.php/chemicke-listy/article/view/369 (accessed on 19 September 2022).
- Sivert, A.; Wald, R.; Craig, C.; Benameur, H. Strategies for Modified Release Oral Formulation Development. In Oral Drug Delivery for Modified Release Formulations; Wiley: Hoboken, NJ, USA, 2022; pp. 235–252. Available online: https://onlinelibrary.wiley.com/doi/abs/10.1002/9781119772729.ch13 (accessed on 10 October 2022).
- Bhutiani, N.; Schucht, J.E.; Miller, K.R.; McClave, S.A. Technical Aspects of Fecal Microbial Transplantation (FMT). Curr. Gastroenterol. Rep. 2018, 20, 30. [Google Scholar] [CrossRef]
- Dvořáčková, K.; Franc, A.; Kejdušová, M. Drug Delivery to the Large Intesine. Chem. Listy 2013, 107, 522–529. Available online: http://chemicke-listy.cz/ojs3/index.php/chemicke-listy/article/view/6455 (accessed on 29 September 2022).
- Kaito, S.; Toya, T.; Yoshifuji, K.; Kurosawa, S.; Inamoto, K.; Takeshita, K.; Suda, W.; Kakihana, K.; Honda, K.; Hattori, M.; et al. Fecal Microbiota Transplantation with Frozen Capsules for a Patient with Refractory Acute Gut Graft-versus-Host Disease. Blood Adv. 2018, 2, 3097–3101. [Google Scholar] [CrossRef]
- Ramai, D.; Zakhia, K.; Fields, P.J.; Ofosu, A.; Patel, G.; Shahnazarian, V.; Lai, J.K.; Dhaliwal, A.; Reddy, M.; Chang, S. Fecal Microbiota Transplantation (FMT) with Colonoscopy Is Superior to Enema and Nasogastric Tube While Comparable to Capsule for the Treatment of Recurrent Clostridioides Difficile Infection: A Systematic Review and Meta-Analysis. Dig. Dis. Sci. 2021, 66, 369–380. [Google Scholar] [CrossRef]
- Zheng, L.; Ji, Y.-Y.; Wen, X.-L.; Duan, S.-L. Fecal Microbiota Transplantation in the Metabolic Diseases: Current Status and Perspectives. World J. Gastroenterol. 2022, 28, 2546–2560. [Google Scholar] [CrossRef] [PubMed]
- Huyghebaert, N.; Vermeire, A.; Remon, J.P. Alternative Method for Enteric Coating of HPMC Capsules Resulting in Ready-to-Use Enteric-Coated Capsules. Eur. J. Pharm. Sci. 2004, 21, 617–623. [Google Scholar] [CrossRef] [PubMed]
- Fülöpová, N.; Pavloková, S.; DeBono, I.; Vetchý, D.; Franc, A. Development and Comparison of Various Coated Hard Capsules Suitable for Enteric Administration to Small Patient Cohorts. Pharmaceutics 2022, 14, 1577. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, J.A.C.; Al-Kauraishi, M.M.; Smith, A.M.; Conway, B.R.; Merchant, H.A. Achieving Gastroresistance without Coating: Formulation of Capsule Shells from Enteric Polymers. Eur. J. Pharm. Biopharm. 2019, 144, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Benameur, H. Enteric Capsule Drug Delivery Technology–Achieving Protection without Coating. Drug Dev. Deliv. 2015, 15, 34–37. Available online: https://drug-dev.com/capsule-technology-enteric-capsule-drug-delivery-technology-achieving-protection-without-coating/ (accessed on 15 September 2022).
- Ph. Eur. MMXVII. European Pharmacopoeia, 9th ed.; European Pharmacopoeia Commission: Strasbourg, France, 2017. [Google Scholar]
- Dvořáčková, K.; Rabišková, M.; Muselík, J.; Gajdziok, J.; Bajerová, M. Coated Hard Capsules as the PH-Dependent Drug Transport Systems to Ileo-Colonic Compartment. Drug Dev. Ind. Pharm. 2011, 37, 1131–1140. [Google Scholar] [CrossRef]
- Vraníková, B.; Franc, A.; Gajdziok, J.; Vetchý, D. Biorelevant dissolution media simulating digestive tract conditions. Chem. Listy 2016, 110, 126–132. Available online: http://www.chemicke-listy.cz/ojs3/index.php/chemicke-listy/article/view/233/233 (accessed on 12 October 2022).
- Donauer, N.; Lobenberg, R. A Mini Review of Scientific and Pharmacopeial Requirements for the Disintegration Test. Int. J. Pharm. 2007, 345, 2–8. [Google Scholar] [CrossRef]
- United States Pharmacopeia, 43rd ed.; United States Pharmacopeia Convention Committee of Revision: Rockville, MD, USA, 2021.
- The Japanese Pharmacopeia, 17th ed.; Society of Japanese Pharmacopoeia: Tokyo, Japan, 2016.
- Katona, M.T.; Kakuk, M.; Szabó, R.; Tonka-Nagy, P.; Takács-Novák, K.; Borbás, E. Towards a Better Understanding of the Post-Gastric Behavior of Enteric-Coated Formulations. Pharm. Res. 2022, 39, 201–211. [Google Scholar] [CrossRef]
- Al-Gousous, J.; Langguth, P. European versus United States Pharmacopeia Disintegration Testing Methods for Enteric-Coated Soft Gelatin Capsules. Dissolution Technol. 2015, 22, 6–8. [Google Scholar] [CrossRef]
- Mašková, E.; Kubová, K.; Vetchý, D. Use of (Meth) Acrylate Copolymers in Controlled Release Matrix Tablet Technology. Chem. Listy 2015, 109, 14–20. Available online: http://www.chemicke-listy.cz/ojs3/index.php/chemicke-listy/article/view/408 (accessed on 15 October 2022).
- Maderuelo, C.; Lanao, J.M.; Zarzuelo, A. Enteric Coating of Oral Solid Dosage Forms as a Tool to Improve Drug Bioavailability. Eur. J. Pharm. Sci. 2019, 138, 105019. [Google Scholar] [CrossRef] [PubMed]
- Al-Tabakha, M.M.; Arida, A.I.; Fahelelbom, K.M.S.; Sadek, B.; Abu Jarad, R.A. Performances of New Generation of Delayed Release Capsules. J. Young Pharm. 2014, 7, 36–44. [Google Scholar] [CrossRef]
- Liu, F.; McConnell, E.; Pygall, S. Update on Polymers for Oral Drug Delivery, 1st ed.; Smithers Rapra Technology: Shawburry, UK, 2011. [Google Scholar]
- Edgar, K.J. Cellulose Esters in Drug Delivery. Cellulose 2006, 14, 49–64. [Google Scholar] [CrossRef]
- Barbosa, J. Going Natural: Using Polymers from Nature for Gastroresistant Applications. Br. J. Pharm. 2017, 2, 14–30. [Google Scholar] [CrossRef]
- Sinha, V.R.; Kumria, R. Polysaccharides in Colon-Specific Drug Delivery. Int. J. Pharm. 2001, 224, 19–38. [Google Scholar] [CrossRef]
- Layek, B.; Mandal, S. Natural Polysaccharides for Controlled Delivery of Oral Therapeutics: A Recent Update. Carbohydr. Polym. 2020, 230, 115617. [Google Scholar] [CrossRef]
- Rodríguez, M.T.S.; Urbano, H.C.; Muñoz de los Ríos, D.; Lara-Villoslada, F.; Martínez, M.A.R.; Hernández, M.E.M. Evaluation of Viability of Lactobacillus fermentum CECT 5716 in Gelatin and Gastroresistant Capsules. J. Pharm. Pharmacol. 2016, 4, 413–418. [Google Scholar] [CrossRef][Green Version]
- Saringat, H.B.; Alfadol, K.I.; Khan, G.M. The Influence of Different Plasticizers on Some Physical and Mechanical Properties of Hydroxypropyl Methylcellulose Free Films. Pak. J. Pharm. Sci. 2005, 18, 25–38. [Google Scholar]
- De Oliveira, H.P.; Albuquerque, J.J.F.; Nogueiras, C.; Rieumont, J. Physical Chemistry Behavior of Enteric Polymer in Drug Release Systems. Int. J. Pharm. 2009, 366, 185–189. [Google Scholar] [CrossRef]
- Dudhat, K.R. The Overview of Oral Solid Dosage Forms and Different Excipients Used for Solid Dosage Formulation. Glob. Acad. J. Pharm. Drug Res. 2022, 4, 66–72. [Google Scholar] [CrossRef]
- Coviello, T.; Matricardi, P.; Marianecci, C.; Alhaique, F. Polysaccharide Hydrogels for Modified Release Formulations. J. Control. Release 2007, 119, 5–24. [Google Scholar] [CrossRef] [PubMed]
- Podczeck, F.; Jones Brian, E. Pharmaceutical Capsules; Jones, B.E., Podczeck, F., Eds.; Pharmaceutical Press: London, UK, 2004. [Google Scholar]
- Murachanian, D. Two-piece hard capsules for pharmaceutical formulation. J. GXP Compliance 2010, 14, 31–42. [Google Scholar]
- Zhang, L.; Wang, Y.; Liu, H.; Yu, L.; Liu, X.; Chen, L.; Zhang, N. Developing Hydroxypropyl Methylcellulose/Hydroxypropyl Starch Blends for Use as Capsule Materials. Carbohydr. Polym. 2013, 98, 73–79. [Google Scholar] [CrossRef]
- Al-Tabakha, M.M. HPMC Capsules: Current Status and Future Prospects. J. Pharm. Pharmaceut. Sci. 2010, 13, 428–442. Available online: https://journals.library.ualberta.ca/jpps/index.php/JPPS/article/view/8870/7398 (accessed on 20 August 2022). [CrossRef]
- Hard Gelatin Capsules: Formulation and Manufacturing Considerations. Available online: https://www.pharmappsroach.com/hard-gelatin-capsules-formulation-and-manufacturing-considerations (accessed on 19 September 2022).
- Podczeck, F. The Development of an Instrumented Tamp-Filling Capsule Machine I. Eur. J. Pharm. Sci. 2000, 10, 267–274. [Google Scholar] [CrossRef]
- Kruisz, J.; Faulhammer, E.; Rehrl, J.; Scheibelhofer, O.; Witschnigg, A.; Khinast, J.G. Residence Time Distribution of a Continuously-Operated Capsule Filling Machine: Development of a Measurement Technique and Comparison of Three Volume-Reducing Inserts. Int. J. Pharm. 2018, 550, 180–189. [Google Scholar] [CrossRef]
- Deodhar, C.P. Soft and Empty Hard Gelatine Capsule Technology; Nirali Prakashan: Bombai, India, 2019. [Google Scholar]
- Capsule Sizes. Available online: https://www.saintytec.com/softgel-sizes (accessed on 20 September 2022).
- Sizes_HPMC Capsule. Available online: https://www.saintytec.com/hpmc-capsule (accessed on 20 September 2022).
- Capsule Sizes. Available online: https://www.medisca.com/Files/ReferenceCharts/Capsule%20Size%20Reference%20Chart%20-%20MUS%20&%20MCA.pdf (accessed on 20 September 2022).
- Cohen, W.M.; Goto, A.; Nagata, A.; Nelson, R.R.; Walsh, J.P. R&D Spillovers, Patents and the Incentives to Innovate in Japan and the United States. Res. Policy 2002, 31, 1349–1367. [Google Scholar]
- Varga, A.; Kocsis, B.; Sipos, D.; Kása, P.; Vigvári, S.; Pál, S.; Dembrovszky, F.; Farkas, K.; Péterfi, Z. How to Apply FMT More Effectively, Conveniently and Flexible–A Comparison of FMT Methods. Front. Cell. Infect. Microbiol. 2021, 11, 657320. [Google Scholar] [CrossRef]
- AR CAPS® Enteric Capsules. Available online: https://www.cphi-online.com/ar-caps-enteric-capsules-prod476709.html (accessed on 15 July 2022).
- Gastric acid Resistant Capsules_ARCAPS®. CapsCanada. Available online: https://capscanada.com/products/acid-resistant-capsules (accessed on 15 July 2022).
- Technical Data Sheet AR Caps Acid Resistant Capsules. Available online: https://www.gocaps.com/wp-content/uploads/2021/04/AR-Caps-Technical-Data-SheetPR00373576.pdf (accessed on 15 July 2022).
- Marcial, G.E.; Ford, A.L.; Haller, M.J.; Gezan, S.A.; Harrison, N.A.; Cai, D.; Meyer, J.L.; Perry, D.J.; Atkinson, M.A.; Wasserfall, C.H.; et al. Lactobacillus Johnsonii N6.2 Modulates the Host Immune Responses: A Double-Blind, Randomized Trial in Healthy Adults. Front. Immunol. 2017, 8, 655. [Google Scholar] [CrossRef]
- Capsugel_EnTRinsic Drug Delivery Technology Platform. Available online: https://www.capsugel.com/news/capsugel-launches-breakthrough-entrinsic-drug-delivery-technology-platform (accessed on 6 June 2022).
- EnTRinsic Drug Delivery Technology. Available online: https://pharma.lonza.co.jp/-/media/Lonza/knowledge/Small-Molecules/Tech%20Briefs/Lonza_tech-brief_Targeted-enTRinsic_ODP.pdf (accessed on 10 June 2022).
- Rowe, R.C.; Sheskey, P.J.; Weller, P.J. Handbook of Pharmaceutical Excipients; The Pharmaceutical Press: London, UK, 2003. [Google Scholar]
- Capsugel. New Intrinsically Enteric Capsule Technology for Pharmaceutical Drug Development. 2017. Available online: https://www.youtube.com/watch?v=yKAb_oKMuTY (accessed on 9 September 2022).
- Lee, H.W.; Kang, W.Y.; Jung, W.; Gwon, M.-R.; Cho, K.; Yoon, Y.-R.; Seong, S.J. Pharmacokinetics and Pharmacodynamics of YYD601, a Dual Delayed-Release Formulation of Esomeprazole, Following Single and Multiple Doses in Healthy Adult Volunteers Under Fasting and Fed Conditions. Drug Des. Devel. Ther. 2022, 16, 619–634. [Google Scholar] [CrossRef] [PubMed]
- Sager, M.; Grimm, M.; Aude, P.; Schick, P.; Merdivan, S.; Hasan, M.; Kromrey, M.-L.; Sivert, A.; Benameur, H.; Koziolek, M.; et al. In Vivo Characterization of EnTRinsicTM Drug Delivery Technology Capsule after Intake in Fed State: A Cross-Validation Approach Using Salivary Tracer Technique in Comparison to MRI. J. Control. Release 2019, 313, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Benameur, H. EnTRinsic™ Drug Delivery Technology for Live Biotherapeutics (Microbiomes). Recent Achievements and Further Challenges in Drug Delivery Research. In Proceedings of the 12th France-Japan Drug Delivery Systems Symposium, Abbaye des Vaux de Cernay, France, 9–12 October 2016; Available online: https://www.umr-cnrs8612.universite-paris-saclay.fr/docs/pdf/Prog-Symposium-Japon-2016.pdf#page=19 (accessed on 15 September 2022).
- Jančálková, M. Capsule Coating for Faecal Transplant Transport. Master’s Thesis, Masaryk University, Brno, Czech Republic, 2021. [Google Scholar]
- Vcaps® Enteric Capsules_Capsugel®. Available online: https://www.capsugel.com/biopharmaceutical-products/vcaps-enteric-capsules (accessed on 19 September 2022).
- Capsugel_Vcaps® Enteric. Available online: http://alfresco-static-files.s3.amazonaws.com/alfresco_images/pharma/2018/10/11/0592de88-9026-4c61-b660-f5fd790b7cea/PharmTech_Europe_July2018.pdf#page=38 (accessed on 10 September 2022).
- Monschke, M.; Kayser, K.; Wagner, K.G. Influence of Particle Size and Drug Load on Amorphous Solid Dispersions Containing PH-Dependent Soluble Polymers and the Weak Base Ketoconazole. AAPS PharmSciTech 2021, 22, 44. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Wan, B.; Jog, R.; Costa, A.; Burgess, D.J. Pectin Microparticles for Peptide Delivery: Optimization of Spray Drying Processing. Int. J. Pharm. 2022, 613, 121384. [Google Scholar] [CrossRef]
- Vcaps Enteric. Available online: https://cpsl-web.s3.amazonaws.com/kc/Vcaps-Enteric_Oct-2017.pdf?mtime=20171017051951 (accessed on 10 September 2022).
- Barbosa, J.A.C.; Abdelsadig, M.S.E.; Conway, B.R.; Merchant, H.A. Using Zeta Potential to Study the Ionisation Behaviour of Polymers Employed in Modified-Release Dosage Forms and Estimating Their PKa. Int. J. Pharm. X 2019, 1, 100024. [Google Scholar] [CrossRef] [PubMed]
- Certificate of analysis: Empty DRcapsTM capsules. In Capsugel; Lonza Company: Colmar, France, 2020.
- DRcaps_Capsules. Available online: https://s3.amazonaws.com/cpsl-web/kc/library/c1a-32029_DRCaps-A4_FIN.PDF (accessed on 10 September 2022).
- Tuleu, C.; Khela, M.K.; Evans, D.F.; Jones, B.E.; Nagata, S.; Basit, A.W. A Scintigraphic Investigation of the Disintegration Behaviour of Capsules in Fasting Subjects: A Comparison of Hypromellose Capsules Containing Carrageenan as a Gelling Agent and Standard Gelatin Capsules. Eur. J. Pharm. Sci. 2007, 30, 251–255. [Google Scholar] [CrossRef]
- Amo, R. DRcaps Capsules Achieve Delayed Release Properties for Nutritional Ingredients in Human Clinical Study. Capsugel. 2021. Available online: https://www.pharmaexcipients.com/wp-content/uploads/2021/01/DRcaps%C2%AE-Capsules-Achieve-Delayed-Release-Properties-for-Nutritional-Ingredients-in-Human-Clinical-Study.pdf (accessed on 10 September 2022).
- Grimm, M.; Ball, K.; Scholz, E.; Schneider, F.; Sivert, A.; Benameur, H.; Kromrey, M.L.; Kühn, J.P.; Weitschies, W. Characterization of the Gastrointestinal Transit and Disintegration Behavior of Floating and Sinking Acid-Resistant Capsules Using a Novel MRI Labeling Technique. Eur. J. Pharm. Sci. 2019, 129, 163–172. [Google Scholar] [CrossRef]
- Youngster, I.; Russell, G.H.; Pindar, C.; Ziv-Baran, T.; Sauk, J.; Hohmann, E.L. Oral, Capsulized, Frozen Fecal Microbiota Transplantation for Relapsing Clostridium Difficile Infection. JAMA 2014, 312, 1772–1778. [Google Scholar] [CrossRef]
- Marzorati, M.; Possemiers, S.; Verhelst, A.; Cadé, D.; Madit, N.; Van de Wiele, T. A Novel Hypromellose Capsule, with Acid Resistance Properties, Permits the Targeted Delivery of Acid-Sensitive Products to the Intestine. LWT-Food Sci. Technol. 2015, 60, 544–551. [Google Scholar] [CrossRef]
- Rump, A.; Weiss, F.N.; Schulz, L.; Kromrey, M.L.; Scheuch, E.; Tzvetkov, M.V.; White, T.; Durkee, S.; Judge, K.W.; Jannin, V.; et al. The Effect of Capsule-in-Capsule Combinations on in Vivo Disintegration in Human Volunteers: A Combined Imaging and Salivary Tracer Study. Pharmaceutics 2021, 13, 2002. [Google Scholar] [CrossRef]
- Empty Capsules. Available online: https://www.purecapsules.co.uk/index.php/product/index/mid/76 (accessed on 14 August 2022).
- Embocaps®. Available online: https://www.embocaps.com/sub/capsule-advantage-design.php (accessed on 12 August 2022).
- EMBO CAPS AP_Brochure. Available online: https://www.embocaps.com/sub/brochure/products-ap.pdf (accessed on 15 August 2022).
- Probiotics Swanson®. Available online: https://www.swansonvitamins.com/ncat1/Health+Concerns/ncat2/Digestive+System/ncat3/Probiotics/q (accessed on 13 August 2022).
- Embo caps®_Suheung Capsule Co. Available online: https://www.cphi-online.com/embo-caps-prod047905.html (accessed on 19 July 2022).
- Chang, R.J.; Wu, C.J.; Lin, Y.H. Acid Resistant Capsule Shell Composition, Acid Resistant Capsule Shell and Its Preparing Process. U.S. Patent No. 9,452,141 B1, 27 September 2016. [Google Scholar]
- BioVXR Acid Resistant Capsules. Available online: https://www.dfc.com.tw/archive/product/item/files/BioVXR%20brochure%202022.pdf (accessed on 16 August 2022).
- Acid Resistant Vegetable Capsules Bio-VXR. Available online: https://biocaps.net/portfolio/acid-resistant-vegetable-capsules (accessed on 19 August 2022).
- Sangve, A.V.; Bansode, S.S.; Menon, M.D. Evaluation of Side selective release of Probiotics from an HPMC Delayed release capsule. Disso India-Chandigarh. In Proceedings of the 7th International Annual Symposium, Chandigarh, India, 12–13 September 2019; Available online: https://spds.in/wp-content/uploads/2020/07/Disso-India-Chandhigarh-scientifica-abstract-book-2019-dt-30-8-19.pdf (accessed on 5 October 2022).
- ACG_Formulation: Flexibility with New Era HPMC Capsules. Available online: https://ondrugdelivery.com/wp-content/uploads/2020/07/109_Jul_2020_ACG.pdf (accessed on 10 September 2022).
- ACG Capsule Brochure. Available online: https://issuu.com/acggroup/docs/capsule_20range_20brochure-web_20us (accessed on 10 September 2022).
- ACGcapsTM_ ACG. Available online: https://www.acg-world.com/archives/capsules/hpmc-capsules/acgcapstm-hr (accessed on 12 September 2022).
- ACGCAPS_HD_Capsules. Available online: https://www.justdial.com/jdmart/Mumbai/ACGCAPS-HD-Capsules/pid-2020831557/022P427056 (accessed on 12 September 2022).
- Cold, F.; Baunwall, S.M.D.; Dahlerup, J.F.; Petersen, A.M.; Hvas, C.L.; Hansen, L.H. Systematic Review with Meta-Analysis: Encapsulated Faecal Microbiota Transplantation–Evidence for Clinical Efficacy. Therap. Adv. Gastroenterol. 2021, 14, 175628482110410. [Google Scholar] [CrossRef] [PubMed]
- Walker, E.G.; Lo, K.R.; Pahl, M.C.; Shin, H.S.; Lang, C.; Wohlers, M.W.; Poppitt, S.D.; Sutton, K.H.; Ingram, J.R. An Extract of Hops (Humulus Lupulus L.) Modulates Gut Peptide Hormone Secretion and Reduces Energy Intake in Healthy-Weight Men: A Randomized, Crossover Clinical Trial. Am. J. Clin. Nutr. 2022, 115, 925–940. [Google Scholar] [CrossRef] [PubMed]
- Reid, I.R.; Wen, J.; Mellar, A.; Liu, M.; Jabr, A.; Horne, A.M. Effect of Oral Zoledronate Administration on Bone Turnover in Older Women. Br. J. Clin. Pharmacol. 2022, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Zain, N.M.M.; Ter Linden, D.; Lilley, A.K.; Royall, P.G.; Tsoka, S.; Bruce, K.D.; Mason, A.J.; Hatton, G.B.; Allen, E.; Goldenberg, S.D.; et al. Design and Manufacture of a Lyophilised Faecal Microbiota Capsule Formulation to GMP Standards. J. Control. Release 2022, 350, 324–331. [Google Scholar] [CrossRef]
- Reigadas, E.; Bouza, E.; Olmedo, M.; Vázquez-Cuesta, S.; Villar-Gómara, L.; Alcalá, L.; Marín, M.; Rodríguez-Fernández, S.; Valerio, M.; Muñoz, P. Faecal Microbiota Transplantation for Recurrent Clostridioides Difficile Infection: Experience with Lyophilized Oral Capsules. J. Hosp. Infect. 2020, 105, 319–324. [Google Scholar] [CrossRef]
- Leong, K.S.W.; Jayasinghe, T.N.; Derraik, J.G.B.; Albert, B.B.; Chiavaroli, V.; Svirskis, D.M.; Beck, K.L.; Conlon, C.A.; Jiang, Y.; Schierding, W.; et al. Protocol for the Gut Bugs Trial: A Randomised Double-Blind Placebo-Controlled Trial of Gut Microbiome Transfer for the Treatment of Obesity in Adolescents. BMJ Open 2019, 9, e026174. [Google Scholar] [CrossRef]
- Jiang, Z.-D.; Jenq, R.R.; Ajami, N.J.; Petrosino, J.F.; Alexander, A.A.; Ke, S.; Iqbal, T.; DuPont, A.W.; Muldrew, K.; Shi, Y.; et al. Safety and Preliminary Efficacy of Orally Administered Lyophilized Fecal Microbiota Product Compared with Frozen Product given by Enema for Recurrent Clostridium Difficile Infection: A Randomized Clinical Trial. PLoS ONE 2018, 13, e0205064. [Google Scholar] [CrossRef]
- Chehri, M.; Christensen, A.H.; Halkjær, S.I.; Günther, S.; Petersen, A.M.; Helms, M. Case Series of Successful Treatment with Fecal Microbiota Transplant (FMT) Oral Capsules Mixed from Multiple Donors Even in Patients Previously Treated with FMT Enemas for Recurrent Clostridium Difficile Infection. Medicine 2018, 97, e11706. [Google Scholar] [CrossRef]
- Halkjær, S.I.; Christensen, A.H.; Lo, B.Z.S.; Browne, P.D.; Günther, S.; Hansen, L.H.; Petersen, A.M. Faecal Microbiota Transplantation Alters Gut Microbiota in Patients with Irritable Bowel Syndrome: Results from a Randomised, Double-Blind Placebo-Controlled Study. Gut 2018, 67, 2107–2115. [Google Scholar] [CrossRef]
- Staley, C.; Hamilton, M.J.; Vaughn, B.P.; Graiziger, C.T.; Newman, K.M.; Kabage, A.J.; Sadowsky, M.J.; Khoruts, A. Successful Resolution of Recurrent Clostridium Difficile Infection Using Freeze-Dried, Encapsulated Fecal Microbiota; Pragmatic Cohort Study. Am. J. Gastroenterol. Suppl. 2017, 112, 940–947. [Google Scholar] [CrossRef]
- Khanna, S.; Pardi, D.S.; Kelly, C.R.; Kraft, C.S.; Dhere, T.; Henn, M.R.; Lombardo, M.-J.; Vulic, M.; Ohsumi, T.; Winkler, J.; et al. A Novel Microbiome Therapeutic Increases Gut Microbial Diversity and Prevents Recurrent Clostridium Difficile Infection. J. Infect. Dis. 2016, 214, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, B.E.; Saraiya, N.; Poeth, K.; Schwartz, R.M.; Epstein, M.E.; Honig, G. Effectiveness of Fecal-Derived Microbiota Transfer Using Orally Administered Capsules for Recurrent Clostridium Difficile Infection. BMC Infect. Dis. 2015, 15, 191. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.L.; Martoni, C.J.; Prakash, S. Cholesterol Lowering and Inhibition of Sterol Absorption by Lactobacillus Reuteri NCIMB 30242: A Randomized Controlled Trial. Eur. J. Clin. Nutr. 2012, 66, 1234–1241. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.L.; Martoni, C.J.; di Pietro, E.; Simon, R.R.; Prakash, S. Evaluation of Clinical Safety and Tolerance of a Lactobacillus Reuteri NCIMB 30242 Supplement Capsule: A Randomized Control Trial. Regul. Toxicol. Pharmacol. 2012, 63, 313–320. [Google Scholar] [CrossRef]
- Sonaje, K.; Chen, Y.-J.; Chen, H.-L.; Wey, S.-P.; Juang, J.-H.; Nguyen, H.-N.; Hsu, C.-W.; Lin, K.-J.; Sung, H.-W. Enteric-Coated Capsules Filled with Freeze-Dried Chitosan/Poly(γ-Glutamic Acid) Nanoparticles for Oral Insulin Delivery. Biomaterials 2010, 31, 3384–3394. [Google Scholar] [CrossRef]
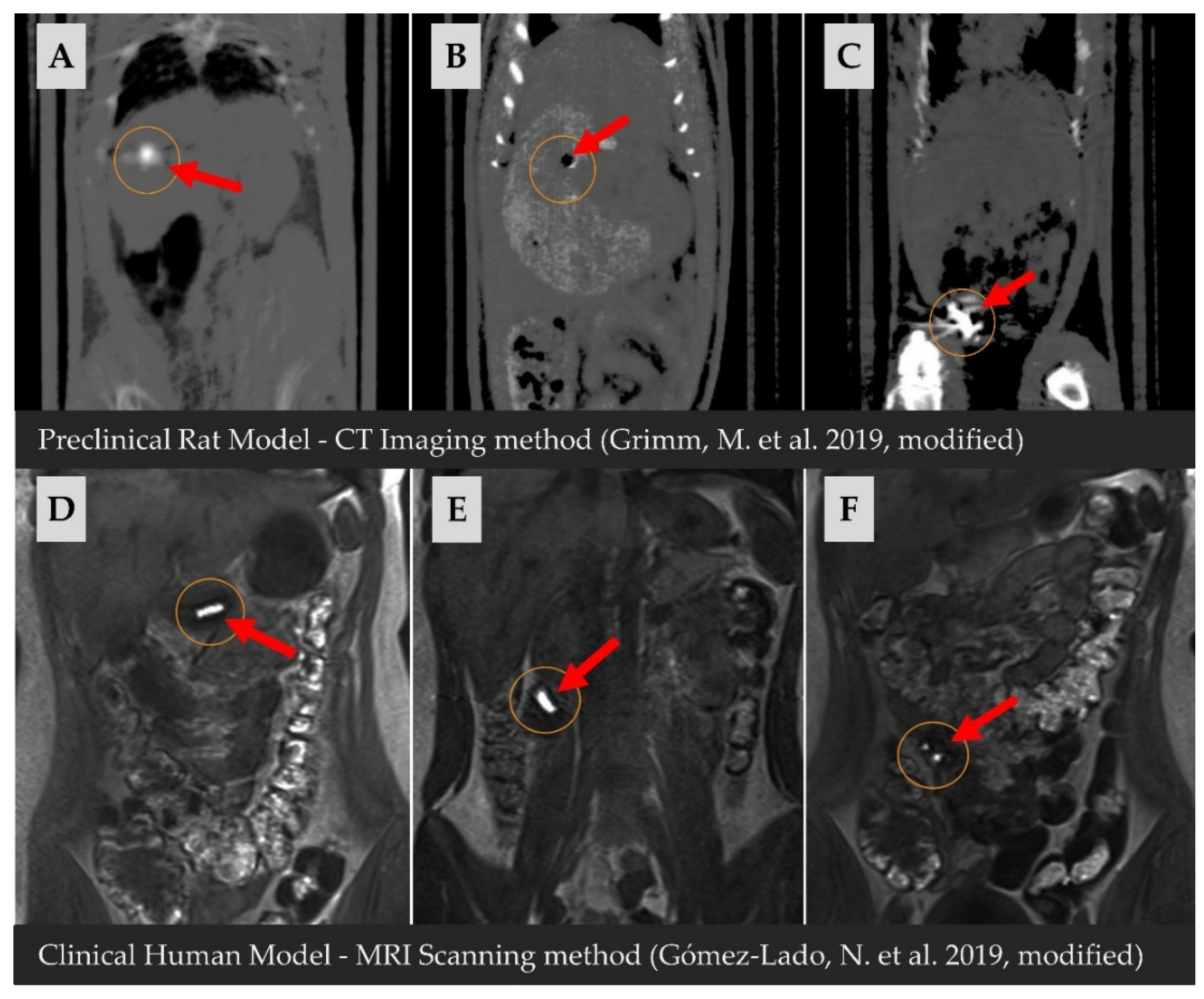
- Saphier, S.; Rosner, A.; Brandeis, R.; Karton, Y. Gastro Intestinal Tracking and Gastric Emptying of Solid Dosage Forms in Rats Using X-ray Imagining. Int. J. Pharm. 2010, 388, 190–195. [Google Scholar] [CrossRef]
- Gómez-Lado, N.; Seoane-Viaño, I.; Matiz, S.; Madla, C.M.; Yadav, V.; Aguiar, P.; Basit, A.W.; Goyanes, A. Gastrointestinal Tracking and Gastric Emptying of Coated Capsules in Rats with or without Sedation Using CT Imaging. Pharmaceutics 2020, 12, 81. [Google Scholar] [CrossRef]
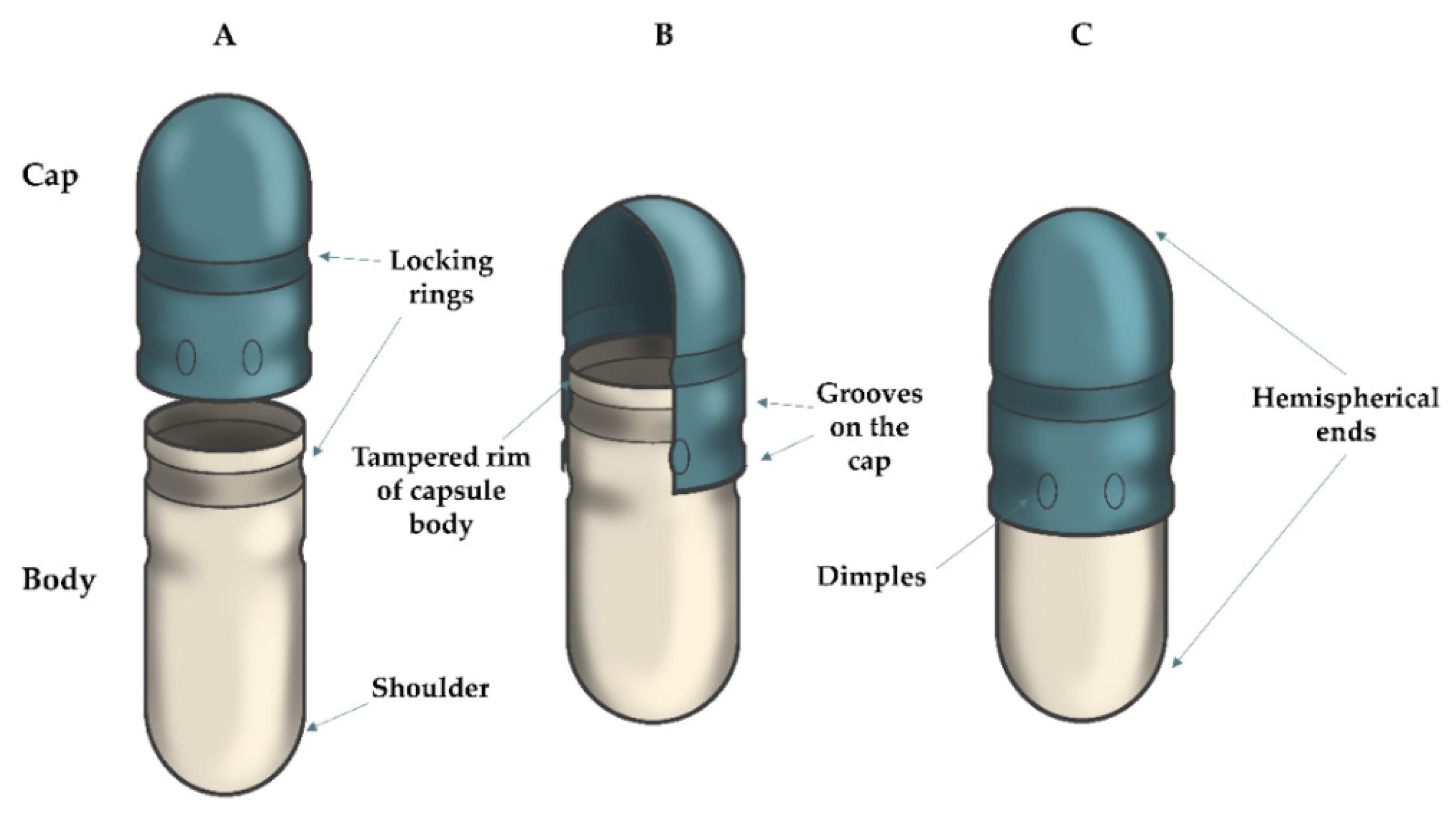
| Pharmacopoeia/Abbreviation | European Pharmacopoeia/Ph. Eur. | United States Pharmacopoeia/USP | Japanese Pharmacopoeia/JP | |||
|---|---|---|---|---|---|---|
| A. Disintegration test | ||||||
| Conditions 1 | 1. | 2. | 1. | 2. | 1. | 2. |
| Time (min) | 120 | 60 | 60 | MON | 120 | 60 |
| Disks | No | Yes | MON | MON | No | Yes |
| Limits (units) | 0/6 | 6/6 | 0/6 | MON | 0/6 | 6/6 |
| B. Dissolution test | ||||||
| Conditions 1 | 1. | 2. | 1. | 2. | 1. | 2. |
| Time (min) | 120 | 45/MON | 120 | 45/MON | 120 | MON |
| Limits (%) | ≤10 | Q + 5 | ≤10 | Q + 5 | ≤10 | QMON + 5 |
| Pharmacopoeia procedure 2 | Method A Continual two-step dissolution | Method A Continual two-step dissolution | If not specified, proceed with both stages of the test separately | |||
| Method B Separated two-step dissolution | Method B Separated two-step dissolution | |||||
| Commercial Name/Abbreviation 1 | Chemical Name | Solubility at pH Values 2 |
|---|---|---|
| Eudragit® FS | A copolymer of methyl acrylate, methyl methacrylate, and methacrylic acid in a ratio of 7:3:1 | 7.0 |
| Eudragit® S | A copolymer of methacrylic acid and methyl methacrylate in a ratio of 1:2 | 7.0 |
| Eudragit® L | A copolymer of methacrylic acid and methyl methacrylate in a ratio of 1:1 | 6.0 |
| Eudragit® FL | A mixture of a copolymer of ethyl methacrylate and methyl methacrylate with a copolymer of methacrylic acid and ethyl methacrylate in a ratio of 1:1 | 5.5 |
| CAS | Cellulose acetate succinate | 5.8–6.2 |
| CAP | Cellulose acetate phthalate | 5.5–6.2 |
| CAT | Cellulose trimellitate | 5.2–5.5 |
| HPMCAS | Hydroxypropyl methylcellulose acetate succinate | 5.5–6.8 |
| HPMCP | Hydroxypropyl methylcellulose phthalate | 5.0–5.5 |
| PVAP | Polyvinyl acetate phthalate | 5.0–5.5 |
| Size | 000 | 00EL | 00 | 0EL | 0 | 1EL | 1 | 2EL | 2 | 3 | 4 | 5 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Body length (mm) | 22.2 | 22.2 | 20.2 | 20.2 | 18.4 | 17.7 | 16.6 | 16.7 | 15.3 | 13.6 | 12.2 | 9.3 |
| Cap length (mm) | 12.9 | 12.9 | 11.7 | 11.7 | 10.7 | 10.5 | 9.8 | 9.7 | 8.9 | 8.1 | 7.2 | 6.2 |
| Capsule length (mm) | 26.1 | 25.3 | 23.3 | 23.1 | 21.7 | 20.4 | 19.4 | 19.3 | 18.0 | 15.9 | 14.3 | 11.1 |
| Weight (mg) 1 | 163 | 130 | 118 | 107 | 96 | 81 | 76 | 66 | 61 | 48 | 38 | 28 |
| Volume (mL) | 1.37 | 1.02 | 0.91 | 0.78 | 0.68 | 0.54 | 0.50 | 0.41 | 0.37 | 0.30 | 0.25 | 0.13 |
| Product Name | Manufacturer/Country of Origin | Composition of Capsule | Pharmacopoeial Acid Resistance |
|---|---|---|---|
| A. Capsules prepared from pH-dependent polymers | |||
| AR Caps® | CapsCanada/Canada | HPMC, HMPCP | No [29] |
| EnTRinsicTM | Capsugel®/USA | CAP | Ph. Eur., USP, JP [60] |
| VCaps® Enteric | Capsugel®/USA | HPMC, HPMCAS | Ph. Eur., USP, JP [66] |
| B. Capsules prepared from gel-forming polymers | |||
| DRcapsTM | Capsugel®/France | HPMC, gellan gum | No [29] |
| EMBO CAPS® AP | Suhueng Capsule/ Republic of Korea | HPMC, pectin | No [75] |
| BioVXR® | BioCaps®/USA | HPMC, gellan gum/pectin 1 | No [75] |
| AGCcapsTM HD | ACG/India | HPMC 2 | Not specified |
| Clinical Study | Type of Capsule | Specification of Dosage Form | Single Dose/A Total Dose of Capsules | Capsule Content | Clinical Indication | Patient Population | Clinical Success Rate |
|---|---|---|---|---|---|---|---|
| Walker, E.G., et al., 2022 [93] | DRcapsTM | - | 2/2 | Bitter extract of Humulus lupulus | Regulating Energy Intake (EI) | 19 | EI ↓ 17.54% |
| Reid I. et al., 2022 [94] | Vcaps® Enteric | One capsule with 20 mg of API | 1; 2; 3/NA | Zoledronic acid (ZA); ZA as microparticles | Safety in bone resorption | 5 | Positive after one week |
| Zain N. et al., 2022 [95] | DRcapsTM | - | 5/5 | Lyophilized bacteria in FMT | rCDI 1 | 7 | 86% |
| Varga A. et al., 2021 [52] | Vcaps® Enteric | - | 5–11/NA | Various parts of fecal suspension | rCDI | 28 | 82.14% |
| Reigadas E. et al., 2020 [96] | DRcapsTM | Cap-in-Cap system | 4–5/NA | Lyophilized bacteria in FMT | rCDI | 32 | 87.5% |
| Leong K. S. W. et al., 2019 [97] | DRcapsTM | Cap-in-Cap system 2 | 14/28 | Frozen bacteria in FMT | Obesity/insulin resistance in adolescent | 80 | NA |
| Jiang Z., et al., 2018 [98] | AR CAPS® | - | NA/27 | Lyophilized bacteria in FMT | rCDI | 31 | 84% |
| Chehri M. et al., 2018 [99] | DRcapsTM | Cap-in-Cap system | 25 (daily)/75 | Frozen bacteria in FMT | rCDI | 9 | 88.9% |
| Halkjær S. et al., 2018 [100] | DRcapsTM | 25 (daily)/ 12 days | Frozen fecal in FMT | Irritable Bowel Syndrome (IBS) | 52 | 36.4 | |
| Staley C. et al., 2017 [101] | DRcapsTM | Cap-in-Cap system | 2–4/NA | Lyophilized bacteria in FMT | rCDI | 49 | 88% |
| Khana S. et al., 2016 [102] | DRcapsTM | - | 15/30 1–12/NA | Various numbers of bacteria | rCDI | 15 15 | 80% 86.7% |
| Hirsch B. et al., 2015 [103] | DRcapsTM | Cap-in-Cap system + gelatin capsule 3 | 6–22/NA | Frozen bacteria in FMT | rCDI | 19 | 89% |
| Youngster I. et al., 2014 [76] | DRcapsTM | Cap-in-Cap system | NA/30 | Frozen bacteria in FMT | rCDI | 20 | 90% |
| Jones M. et al., 2012 [104] | DRcapsTM | - | 2 (daily)/126 | Lyophilized bacteria Lactobacillus reuteri NCIMB 30242 | Hypercholesterolemia | 131 | LDL-C 4 ↓ 11.64% |
| Jones M. et al., 2012 [105] | DRcapsTM | - | 2 (daily)/126 | Lyophilized bacteria Lactobacillus reuteri | Clinical safety of Lactobacillus reuteri | 131 | Positive; Not quantified |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Franc, A.; Vetchý, D.; Fülöpová, N. Commercially Available Enteric Empty Hard Capsules, Production Technology and Application. Pharmaceuticals 2022, 15, 1398. https://doi.org/10.3390/ph15111398
Franc A, Vetchý D, Fülöpová N. Commercially Available Enteric Empty Hard Capsules, Production Technology and Application. Pharmaceuticals. 2022; 15(11):1398. https://doi.org/10.3390/ph15111398
Chicago/Turabian StyleFranc, Aleš, David Vetchý, and Nicole Fülöpová. 2022. "Commercially Available Enteric Empty Hard Capsules, Production Technology and Application" Pharmaceuticals 15, no. 11: 1398. https://doi.org/10.3390/ph15111398
APA StyleFranc, A., Vetchý, D., & Fülöpová, N. (2022). Commercially Available Enteric Empty Hard Capsules, Production Technology and Application. Pharmaceuticals, 15(11), 1398. https://doi.org/10.3390/ph15111398







