Paeonol Protects against Methotrexate Hepatotoxicity by Repressing Oxidative Stress, Inflammation, and Apoptosis—The Role of Drug Efflux Transporters
Abstract
1. Introduction
2. Results
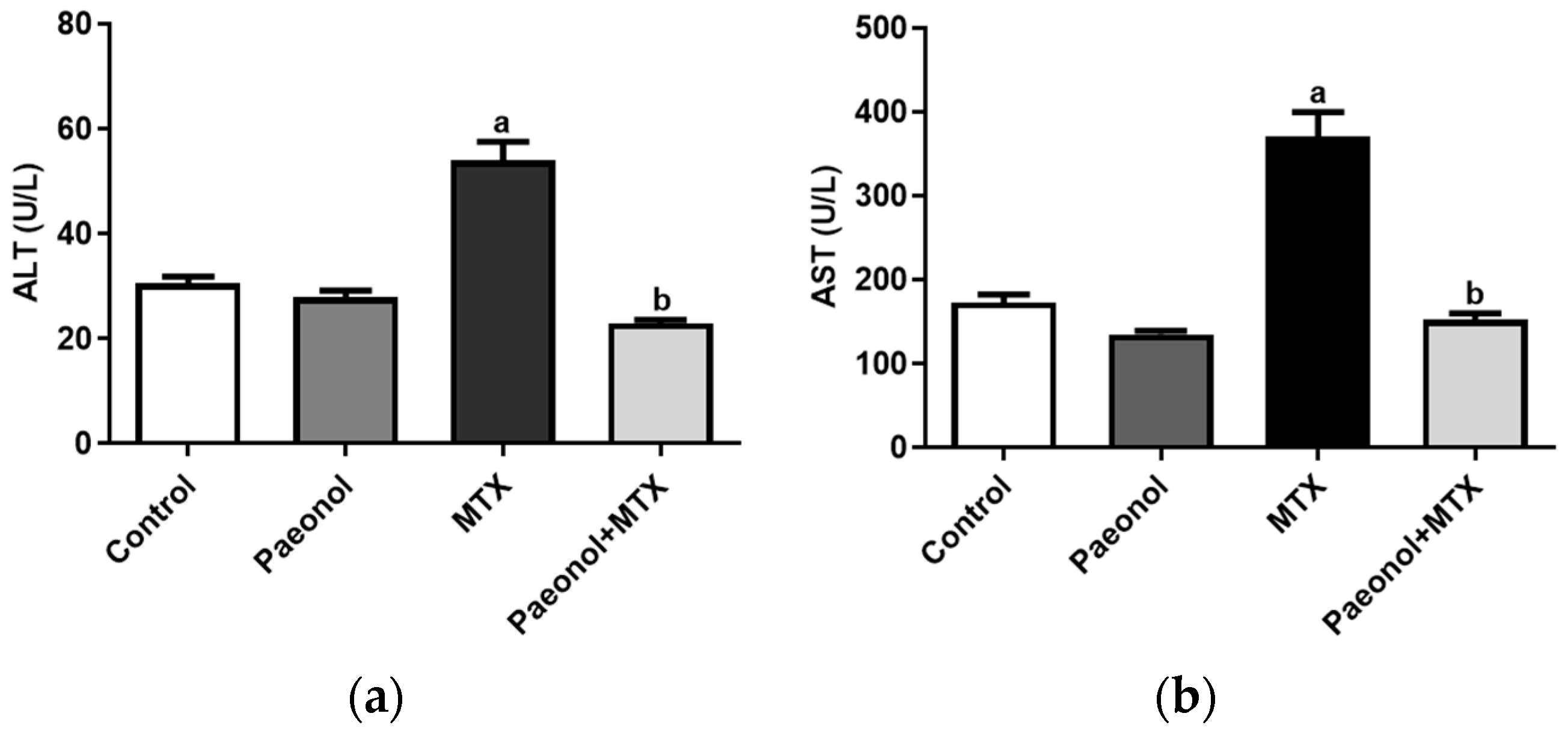
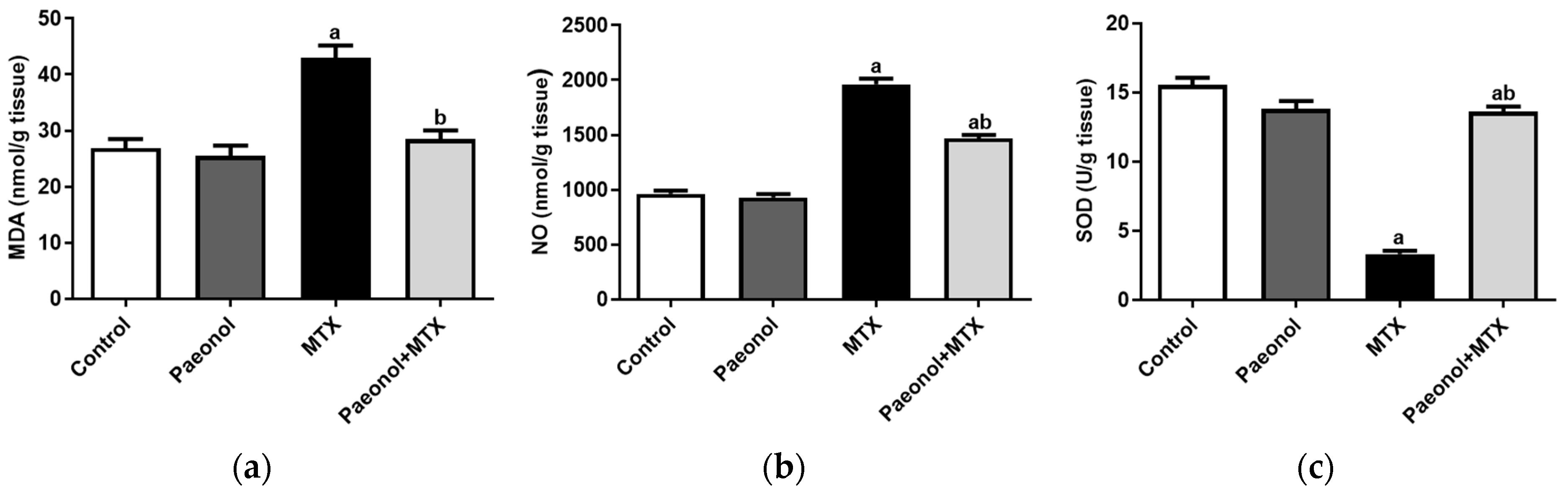
2.1. Effect on Liver Function and Hepatic Oxidative Status
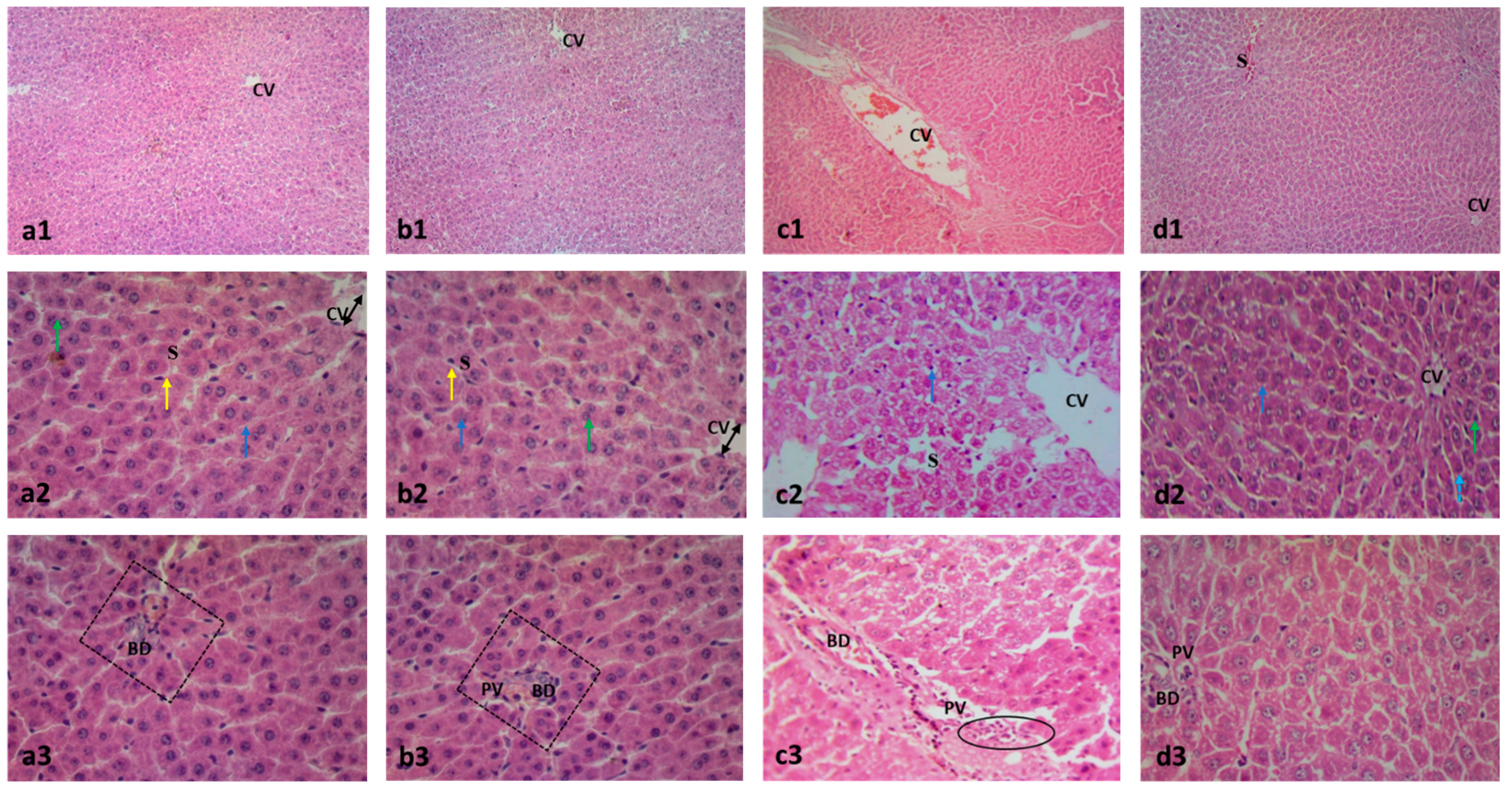
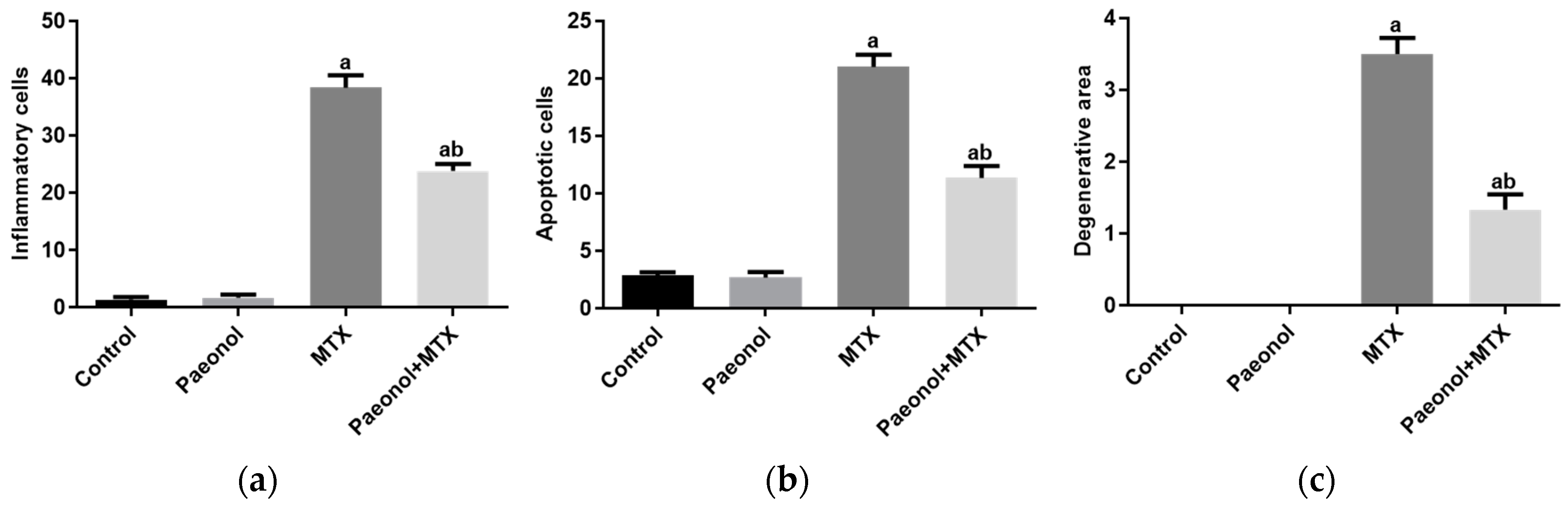
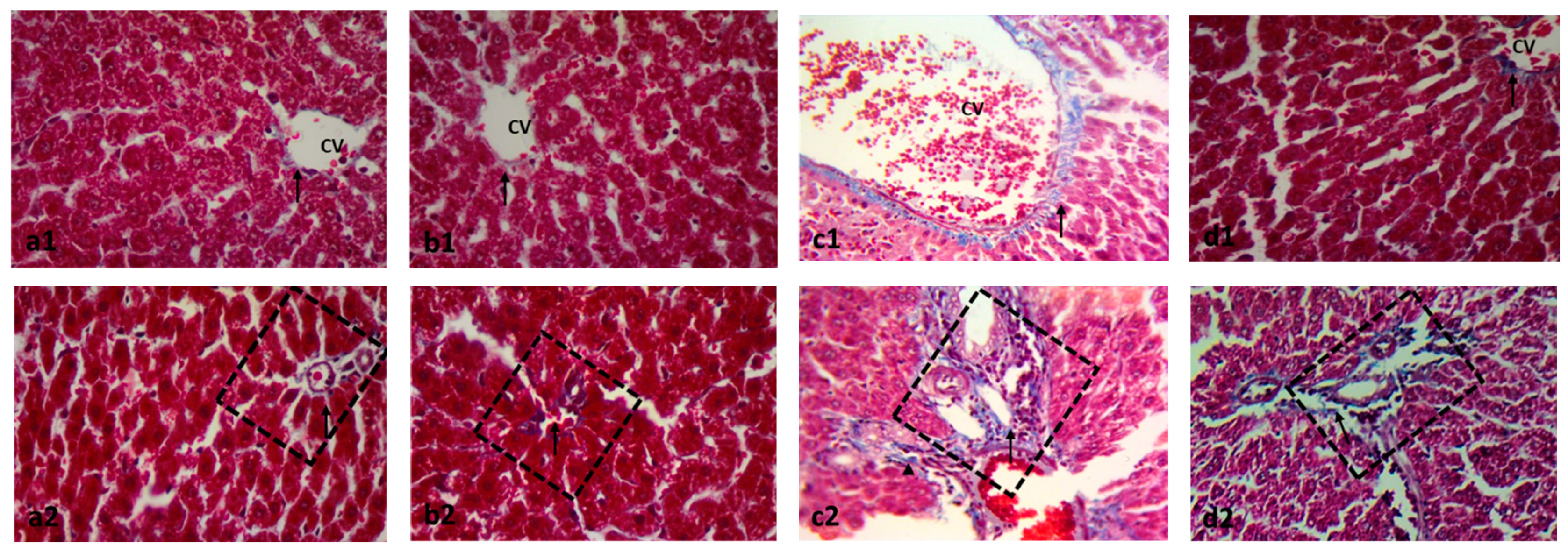
2.2. Effect on Histopathology Examination
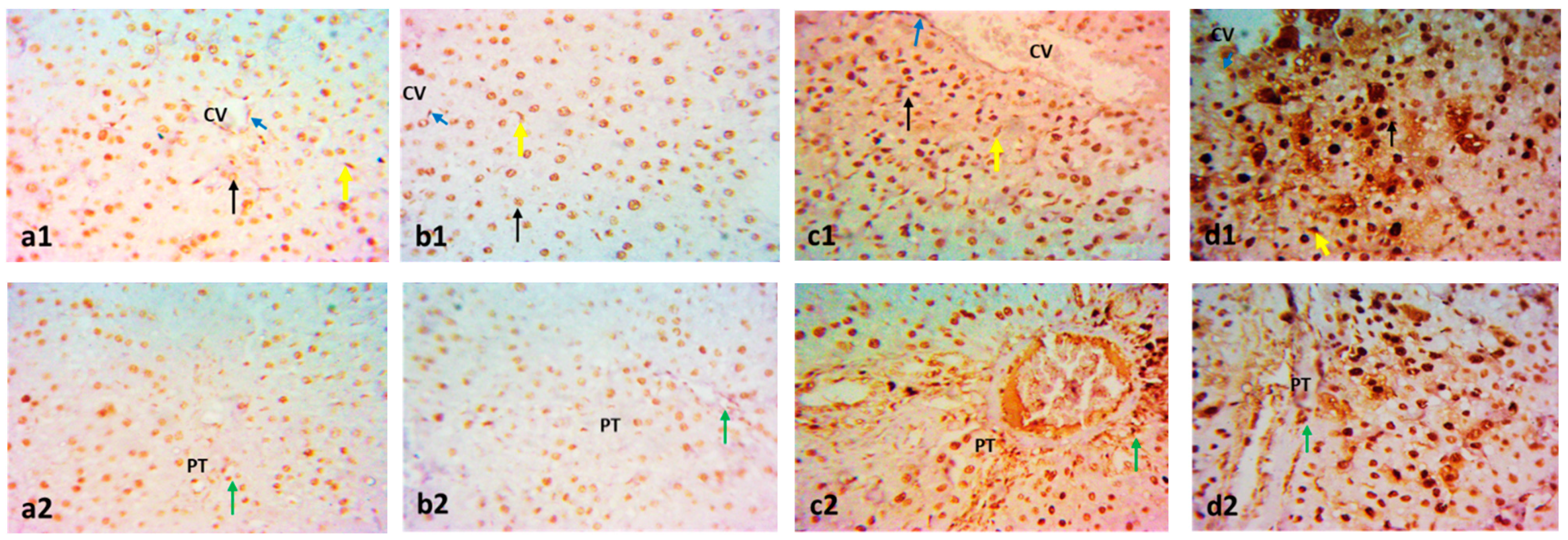
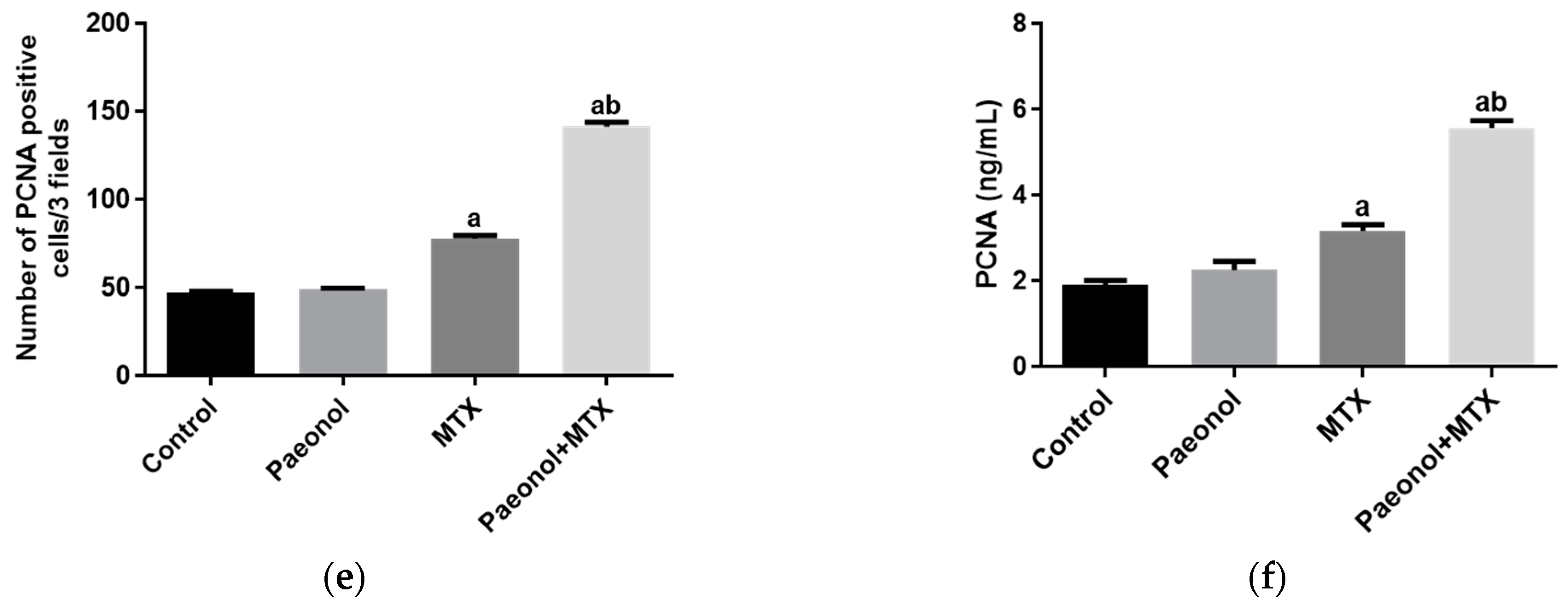
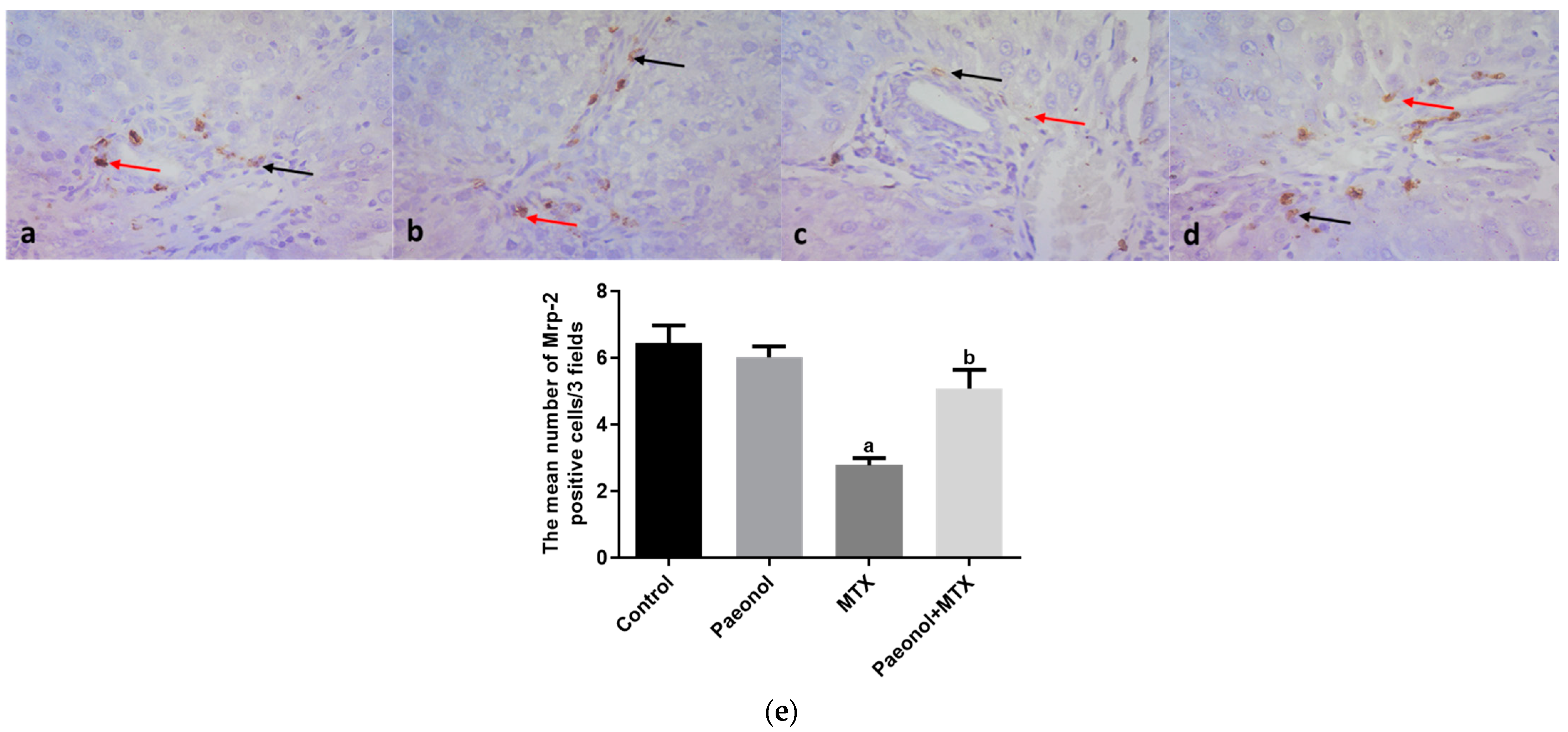
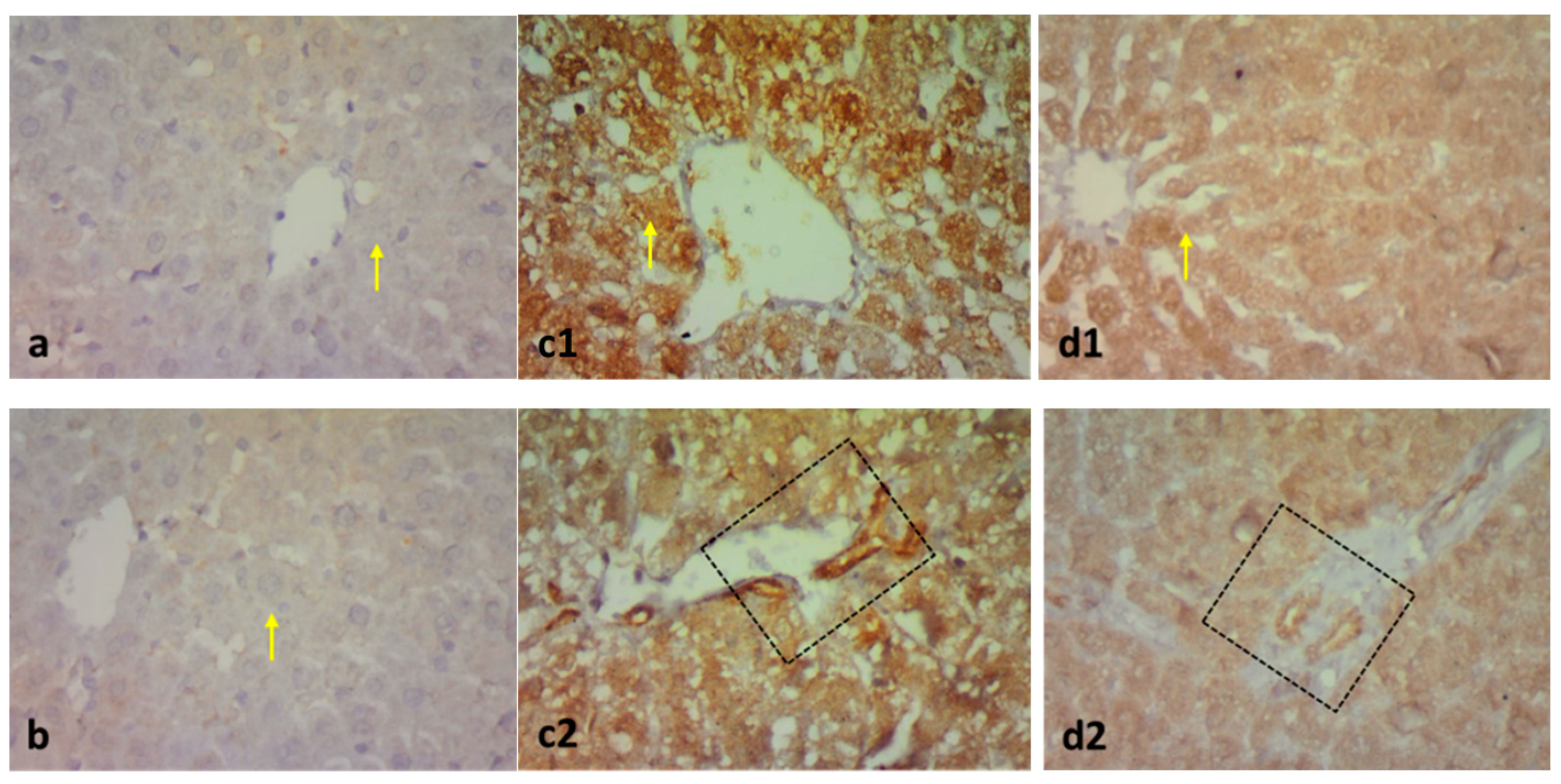
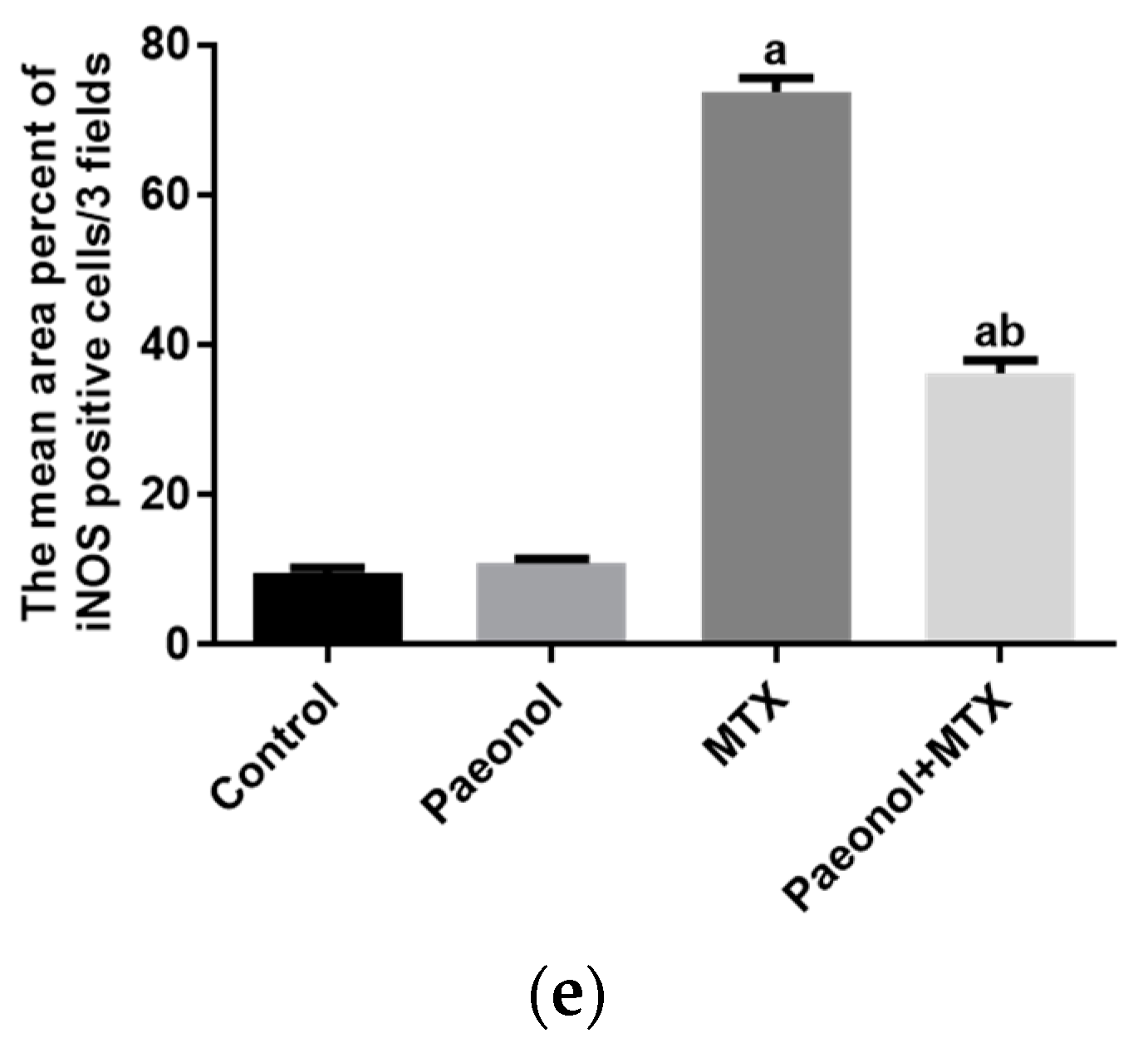
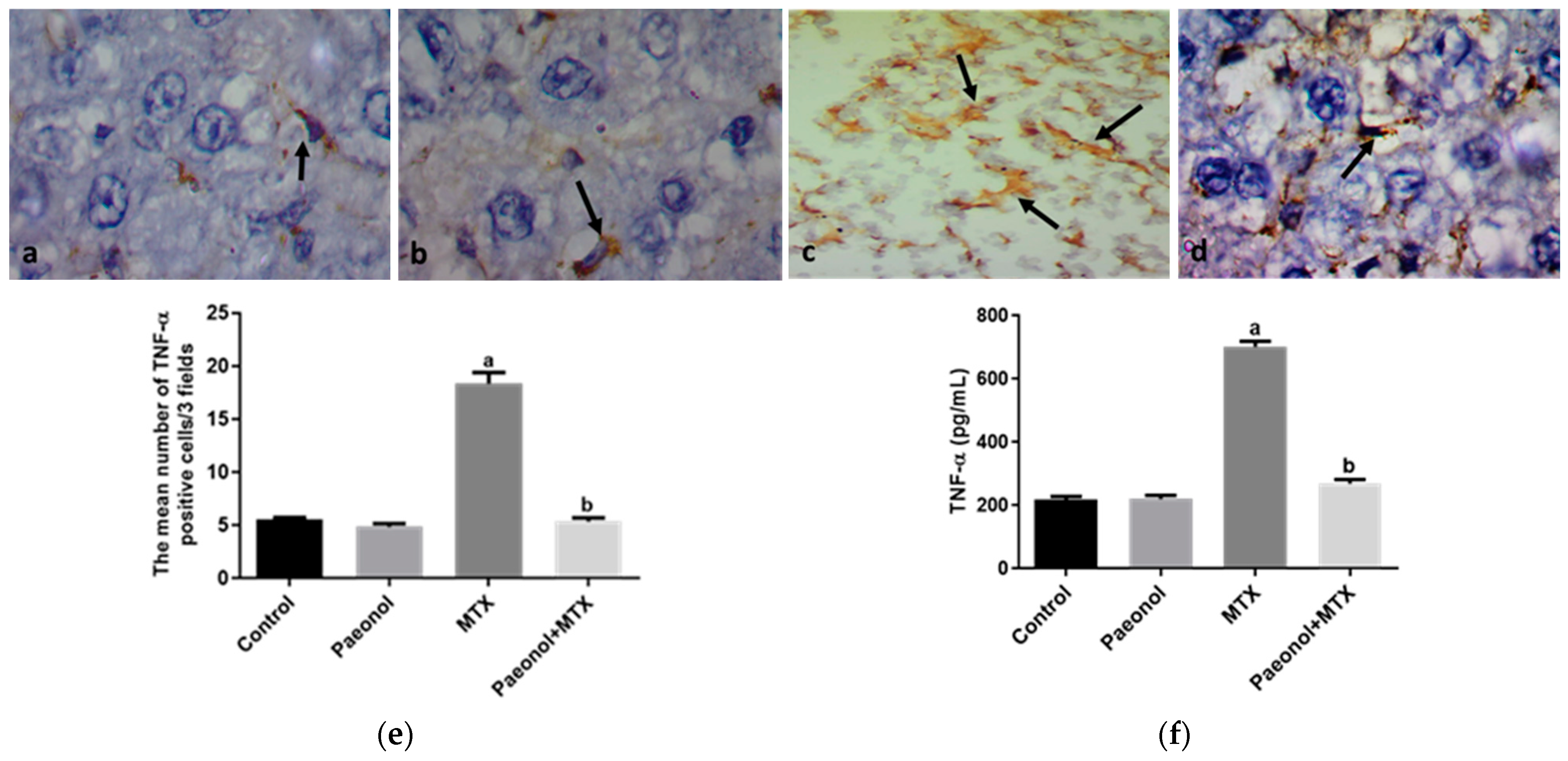
2.3. Effect on Immunohistochemistry and ELISA
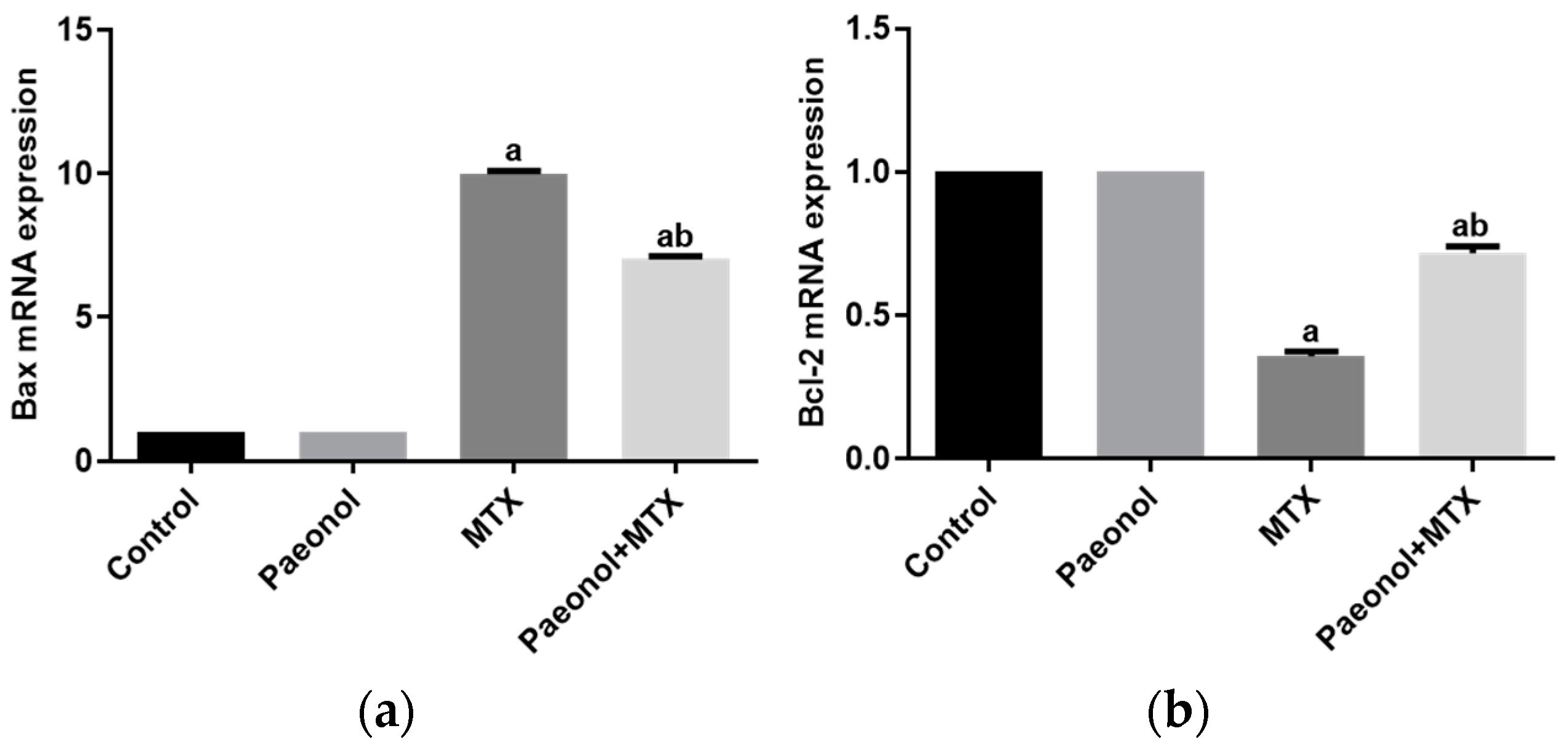
2.4. Effect on Hepatic Apoptosis
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Experimental Design
4.3. Sample Preparation
4.4. Biochemical Analysis
4.4.1. Determination of Liver Function Tests
4.4.2. Determination of Hepatic Oxidative Stress Biomarkers
4.5. Histological Evaluation
4.6. Immunohistochemical Examination
4.7. ELISA Analysis
4.8. Morphometric Analysis
4.9. Real-Time PCR Determination of Hepatic Bax and Bcl-2
4.10. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, W.; Zhou, H.; Liu, L. Side effects of methotrexate therapy for rheumatoid arthritis: A systematic review. Eur. J. Med. Chem. 2018, 158, 502–516. [Google Scholar] [CrossRef]
- Koźmiński, P.; Halik, P.K.; Chesori, R.; Gniazdowska, E. Overview of dual-acting drug methotrexate in different neurological diseases, autoimmune pathologies and cancers. Int. J. Mol. Sci. 2020, 21, 3483. [Google Scholar] [CrossRef]
- Bath, R.K.; Brar, N.K.; Forouhar, F.A.; Wu, G.Y. A review of methotrexate-associated hepatotoxicity. J. Dig. Dis. 2014, 15, 517–524. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Zeng, J.; Xing, L.; Li, C. Extra-and intra-cellular mechanisms of hepatic stellate cell activation. Biomedicines 2021, 9, 1014. [Google Scholar] [CrossRef] [PubMed]
- Conway, R.; Carey, J.J. Risk of liver disease in methotrexate treated patients. World J. Hepatol. 2017, 9, 1092. [Google Scholar] [CrossRef]
- Ezhilarasan, D. Hepatotoxic potentials of methotrexate: Understanding the possible toxicological molecular mechanisms. Toxicology 2021, 458, 152840. [Google Scholar] [CrossRef] [PubMed]
- Goudarzi, M.; Kalantar, M.; Sadeghi, E.; Karamallah, M.H.; Kalantar, H. Protective effects of apigenin on altered lipid peroxidation, inflammation, and antioxidant factors in methotrexate-induced hepatotoxicity. Naunyn-Schmiedeberg Arch. Pharmacol. 2021, 394, 523–531. [Google Scholar] [CrossRef]
- Cure, E.; Kirbas, A.; Tumkaya, L.; Cure, M.C.; Kalkan, Y.; Yilmaz, A.; Yuce, S. Protective effect of infliximab on methotrexate-induced liver injury in rats: Unexpected drug interaction. J. Cancer Res. Ther. 2015, 11, 164. [Google Scholar] [CrossRef] [PubMed]
- Ezhilarasan, D. Oxidative stress is bane in chronic liver diseases: Clinical and experimental perspective. Arab J. Gastroenterol. 2018, 19, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Chan, E.S.; Montesinos, M.C.; Fernandez, P.; Desai, A.; Delano, D.L.; Yee, H.; Reiss, A.B.; Pillinger, M.H.; Chen, J.-F.; Schwarzschild, M.A. Adenosine A2A receptors play a role in the pathogenesis of hepatic cirrhosis. Br. J. Pharmacol. 2006, 148, 1144. [Google Scholar] [CrossRef] [PubMed]
- Chu, X.; Korzekwa, K.; Elsby, R.; Fenner, K.; Galetin, A.; Lai, Y.; Matsson, P.; Moss, A.; Nagar, S.; Rosania, G. Intracellular drug concentrations and transporters: Measurement, modeling, and implications for the liver. Clin. Pharmacol. Ther. 2013, 94, 126–141. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kwon, M.; Choi, M.-K.; Song, I.-S. Effects of red ginseng extract on the pharmacokinetics and elimination of methotrexate via Mrp2 regulation. Molecules 2018, 23, 2948. [Google Scholar] [CrossRef] [PubMed]
- Al-Taher, A.Y.; Morsy, M.A.; Rifaai, R.A.; Zenhom, N.M.; Abdel-Gaber, S.A. Paeonol attenuates methotrexate-induced cardiac toxicity in rats by inhibiting oxidative stress and suppressing TLR4-induced NF-κB inflammatory pathway. Mediat. Inflamm. 2020, 2020, 8641026. [Google Scholar] [CrossRef]
- Mohamed, M.Z.; Morsy, M.A.; Mohamed, H.H.; Hafez, H.M. Paeonol protects against testicular ischaemia-reperfusion injury in rats through inhibition of oxidative stress and inflammation. Andrologia 2020, 52, e13599. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Tan, S.-Y.; Wang, X.-F. Paeonol exerts an anticancer effect on human colorectal cancer cells through inhibition of PGE2 synthesis and COX-2 expression. Oncol. Rep. 2014, 32, 2845–2853. [Google Scholar] [CrossRef]
- Hafez, H.; Morsy, M.; Mohamed, M.; Zenhom, N. Mechanisms underlying gastroprotective effect of paeonol against indomethacin-induced ulcer in rats. Hum. Exp. Toxicol. 2019, 38, 510–518. [Google Scholar] [CrossRef]
- Liang, Y.; Liu, Z.-Y.; Wang, P.-Y.; Li, Y.-J.; Wang, R.-R.; Xie, S.-Y. Nanoplatform-based natural products co-delivery system to surmount cancer multidrug-resistant. J. Control. Release 2021, 336, 396–409. [Google Scholar] [CrossRef]
- Chung, S.Y.; Sung, M.K.; Kim, N.H.; Jang, J.O.; Go, E.J.; Lee, H.J. Inhibition of P-glycoprotein by natural products in human breast cancer cells. Arch. Pharmacal Res. 2005, 28, 823–828. [Google Scholar] [CrossRef]
- Fouad, A.; Hafez, H.; Hamouda, A. Hydrogen sulfide modulates IL-6/STAT3 pathway and inhibits oxidative stress, inflammation, and apoptosis in rat model of methotrexate hepatotoxicity. Hum. Exp. Toxicol. 2020, 39, 77–85. [Google Scholar] [CrossRef]
- Jahovic, N.; Cevik, H.; Şehirli, A.Ö.; Yeğen, B.Ç.; Şener, G. Melatonin prevents methotrexate-induced hepatorenal oxidative injury in rats. J. Pineal Res. 2003, 34, 282–287. [Google Scholar] [CrossRef]
- Çetin, A.; Kaynar, L.; Kocyigit, I.; Hacioglu, S.K.; Saraymen, R.; Ozturk, A.; Sari, I.; Sagdic, O. Role of grape seed extract on methotrexate induced oxidative stress in rat liver. Am. J. Chin. Med. 2008, 36, 861–872. [Google Scholar] [CrossRef]
- McKim, S.E.; Gäbele, E.; Isayama, F.; Lambert, J.C.; Tucker, L.M.; Wheeler, M.D.; Connor, H.D.; Mason, R.P.; Doll, M.A.; Hein, D.W. Inducible nitric oxide synthase is required in alcohol-induced liver injury: Studies with knockout mice. Gastroenterology 2003, 125, 1834–1844. [Google Scholar] [CrossRef] [PubMed]
- Koppelmann, T.; Pollak, Y.; Mogilner, J.; Bejar, J.; Coran, A.G.; Sukhotnik, I. Dietary L-arginine supplementation reduces Methotrexate-induced intestinal mucosal injury in rat. BMC Gastroenterol. 2012, 12, 41. [Google Scholar] [CrossRef] [PubMed]
- Morsy, M.A.; El-Sheikh, A.A.; Abdel-Hafez, S.M.N.; Kandeel, M.; Abdel-Gaber, S.A. Paeonol Protects Against Methotrexate-Induced Nephrotoxicity via Upregulation of P-gp Expression and Inhibition of TLR4/NF-κB Pathway. Front. Pharmacol. 2022, 13, 774387. [Google Scholar] [CrossRef] [PubMed]
- Morsy, M.A.; Abdel-Aziz, A.M.; Abdel-Hafez, S.; Venugopala, K.N.; Nair, A.B.; Abdel-Gaber, S.A. The possible contribution of P-glycoprotein in the protective effect of paeonol against methotrexate-induced testicular injury in rats. Pharmaceuticals 2020, 13, 223. [Google Scholar] [CrossRef] [PubMed]
- González-Magaña, A.; Blanco, F.J. Human PCNA structure, function, and interactions. Biomolecules 2020, 10, 570. [Google Scholar] [CrossRef]
- Banlunara, W.; Bintvihok, A.; Kumagai, S. Immunohistochemical study of proliferating cell nuclear antigen (PCNA) in duckling liver fed with aflatoxin B1 and esterified glucomannan. Toxicon 2005, 46, 954–957. [Google Scholar] [CrossRef]
- Antonescu, C.R.; Leung, D.H.; Dudas, M.; Ladanyi, M.; Brennan, M.; Woodruff, J.M.; Cordon-Cardo, C. Alterations of cell cycle regulators in localized synovial sarcoma: A multifactorial study with prognostic implications. Am. J. Pathol. 2000, 156, 977–983. [Google Scholar] [CrossRef]
- Ahmed, Z.S.O.; Hussein, S.; Ghandour, R.A.; Azouz, A.A.; El-Sakhawy, M.A. Evaluation of the effect of methotrexate on the hippocampus, cerebellum, liver, and kidneys of adult male albino rat: Histopathological, immunohistochemical and biochemical studies. Acta Histochem. 2021, 123, 151682. [Google Scholar] [CrossRef]
- Felemban, S.G.; Aldubayan, M.A.; Alhowail, A.H.; Almami, I.S. Vitamin B17 ameliorates methotrexate-induced reproductive toxicity, oxidative stress, and testicular injury in male rats. Oxidative Med. Cell. Longev. 2020, 2020, 4372719. [Google Scholar] [CrossRef]
- Yazici, A.C.; Tursen, U.; Apa, D.D.; Ikizoglu, G.; Api, H.; Baz, K.; Tasdelen, B. The changes in expression of ICAM-3, Ki-67, PCNA, and CD31 in psoriatic lesions before and after methotrexate treatment. Arch. Dermatol. Res. 2005, 297, 249–255. [Google Scholar] [CrossRef]
- Ahmed, A.A.; Selim, M.A.; El-Sayed, N.M. α-Lipoic acid ameliorates oral mucositis and oxidative stress induced by methotrexate in rats. Histological and immunohistochemical study. Life Sci. 2017, 171, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Zeng, H. Selenium as an essential micronutrient: Roles in cell cycle and apoptosis. Molecules 2009, 14, 1263–1278. [Google Scholar] [CrossRef] [PubMed]
- Fayez, A.M.; Zakaria, S.; Moustafa, D. Alpha lipoic acid exerts antioxidant effect via Nrf2/HO-1 pathway activation and suppresses hepatic stellate cells activation induced by methotrexate in rats. Biomed. Pharmacother. 2018, 105, 428–433. [Google Scholar] [CrossRef] [PubMed]
- Shu, Y.; Yang, Y.; Zhao, Y.; Ma, L.; Fu, P.; Wei, T.; Zhang, L. Melittin inducing the apoptosis of renal tubule epithelial cells through upregulation of Bax/Bcl-2 expression and activation of TNF-α signaling pathway. BioMed Res. Int. 2019, 2019, 9450368. [Google Scholar] [CrossRef] [PubMed]
- Jafarinejad-Farsangi, S.; Farazmand, A.; Mahmoudi, M.; Gharibdoost, F.; Karimizadeh, E.; Noorbakhsh, F.; Faridani, H.; Jamshidi, A.R. MicroRNA-29a induces apoptosis via increasing the Bax: Bcl-2 ratio in dermal fibroblasts of patients with systemic sclerosis. Autoimmunity 2015, 48, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Shibayama, Y.; Takeda, Y.; Yamada, K. Effect of methotrexate treatment on expression levels of organic anion transporter polypeptide 2, P-glycoprotein and bile salt export pump in rats. Biol. Pharm. Bull. 2009, 32, 493–496. [Google Scholar] [CrossRef]
- Mohamed, M.; El Sheikh, A.K.; Mohammed, H.H. Modulation of Liver P-Glycoprotien Expression May Contribute to Gossypin Protection against Methotrexate-Induced Hepatotoxicity. Indian J. Pharmacol. 2021, 53, 25. [Google Scholar] [CrossRef]
- Liu, J.; Wang, S.; Feng, L.; Ma, D.; Fu, Q.; Song, Y.; Jia, X.; Ma, S. Hypoglycemic and antioxidant activities of paeonol and its beneficial effect on diabetic encephalopathy in streptozotocin-induced diabetic rats. J. Med. Food 2013, 16, 577–586. [Google Scholar] [CrossRef]
- El-Sheikh, A.A.; Morsy, M.A.; Al-Taher, A.Y. Protective mechanisms of resveratrol against methotrexate-induced renal damage may involve BCRP/ABCG2. Fundam. Clin. Pharmacol. 2016, 30, 406–418. [Google Scholar] [CrossRef]
- Buege, J.A.; Aust, S.D. Microsomal lipid peroxidation. Methods Enzymol. 1978, 52, 302–310. [Google Scholar] [PubMed]
- Sastry, K.; Moudgal, R.; Mohan, J.; Tyagi, J.; Rao, G. Spectrophotometric determination of serum nitrite and nitrate by copper–cadmium alloy. Anal. Biochem. 2002, 306, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Marklund, S.; Marklund, G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem. 1974, 47, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Hofman, F.M.; Taylor, C.R. Immunohistochemistry. Curr. Protoc. Immunol. 2013, 103, 21.4.1–21.4.26. [Google Scholar] [CrossRef]
- Ahmed, A.-S.F.; Bayoumi, A.; Eltahir, H.M.; Abdel Hafez, S.; Abouzied, M.M. Amelioration of Sepsis-induced liver and lung injury by a superoxide dismutase mimetic; role of TNF-α and Caspase-3. J. Adv. Biomed. Pharm. Sci. 2020, 3, 31–39. [Google Scholar] [CrossRef]
- Horiuchi, M.; Itoh, A.; Pleasure, D.; Itoh, T. MEK-ERK Signaling Is Involved in Interferon-γ-induced Death of Oligodendroglial Progenitor Cells. J. Biol. Chem. 2006, 281, 20095–20106. [Google Scholar] [CrossRef]
- Refaie, M.M.; El-Hussieny, M.; Zenhom, N.M. Protective role of nebivolol in cadmium-induced hepatotoxicity via downregulation of oxidative stress, apoptosis and inflammatory pathways. Environ. Toxicol. Pharmacol. 2018, 58, 212–219. [Google Scholar] [CrossRef]
- VanGuilder, H.D.; Vrana, K.E.; Freeman, W.M. Twenty-five years of quantitative PCR for gene expression analysis. Biotechniques 2008, 44, 619–626. [Google Scholar] [CrossRef]


| Group | Treatment Regimen | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Day 1 | Day 2 | Day 3 | Day 4 | Day 5 | Day 6 | Day 7 | Day 8 | Day 9 | Day 10 | |
| Control | Vehicle 1 mL, p.o. | Vehicle 1 mL, p.o. | Vehicle 1 mL, p.o. | Vehicle 1 mL, p.o. | Vehicle 1 mL, p.o. | Vehicle 1 mL, p.o. | Vehicle 1 mL, p.o. | Vehicle 1 mL, p.o. | Vehicle 1 mL, p.o. | Vehicle 1 mL, p.o. |
| Paeonol | 100 mg/kg, p.o. | 100 mg/kg, p.o. | 100 mg/kg, p.o. | 100 mg/kg, p.o. | 100 mg/kg, p.o. | 100 mg/kg, p.o. | 100 mg/kg, p.o. | 100 mg/kg, p.o. | 100 mg/kg, p.o. | 100 mg/kg, p.o. |
| MTX | 20 mg/kg, i.p. | |||||||||
| Paeonol + MTX | 100 mg/kg, p.o. | 100 mg/kg, p.o. | 100 mg/kg, p.o. | 100 mg/kg, p.o. | 100 mg/kg, p.o. + 20 mg/kg, i.p. | 100 mg/kg, p.o. | 100 mg/kg, p.o. | 100 mg/kg, p.o. | 100 mg/kg, p.o. | 100 mg/kg, p.o. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morsy, M.A.; Abdel-Latif, R.; Hafez, S.M.N.A.; Kandeel, M.; Abdel-Gaber, S.A. Paeonol Protects against Methotrexate Hepatotoxicity by Repressing Oxidative Stress, Inflammation, and Apoptosis—The Role of Drug Efflux Transporters. Pharmaceuticals 2022, 15, 1296. https://doi.org/10.3390/ph15101296
Morsy MA, Abdel-Latif R, Hafez SMNA, Kandeel M, Abdel-Gaber SA. Paeonol Protects against Methotrexate Hepatotoxicity by Repressing Oxidative Stress, Inflammation, and Apoptosis—The Role of Drug Efflux Transporters. Pharmaceuticals. 2022; 15(10):1296. https://doi.org/10.3390/ph15101296
Chicago/Turabian StyleMorsy, Mohamed A., Rania Abdel-Latif, Sara Mohamed Naguib Abdel Hafez, Mahmoud Kandeel, and Seham A. Abdel-Gaber. 2022. "Paeonol Protects against Methotrexate Hepatotoxicity by Repressing Oxidative Stress, Inflammation, and Apoptosis—The Role of Drug Efflux Transporters" Pharmaceuticals 15, no. 10: 1296. https://doi.org/10.3390/ph15101296
APA StyleMorsy, M. A., Abdel-Latif, R., Hafez, S. M. N. A., Kandeel, M., & Abdel-Gaber, S. A. (2022). Paeonol Protects against Methotrexate Hepatotoxicity by Repressing Oxidative Stress, Inflammation, and Apoptosis—The Role of Drug Efflux Transporters. Pharmaceuticals, 15(10), 1296. https://doi.org/10.3390/ph15101296









