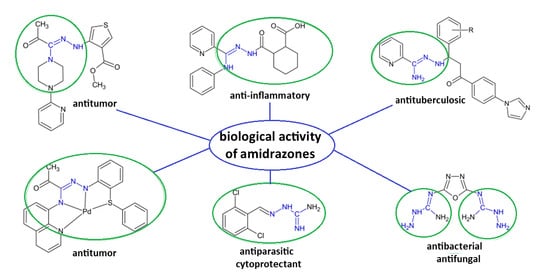
A Review of the Biological Activity of Amidrazone Derivatives
Abstract
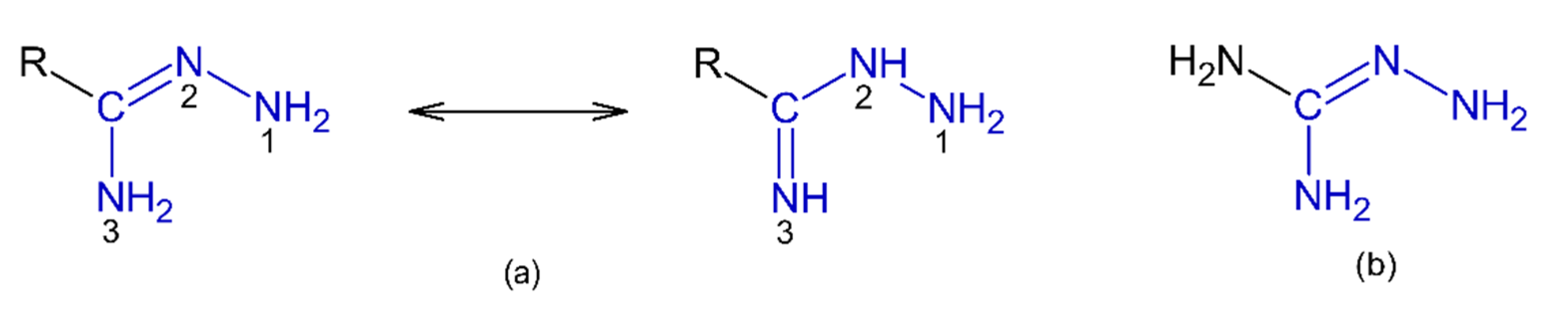
1. Introduction
2. Results
2.1. Antimicrobial Activity
2.1.1. Tuberculostatic Activity
2.1.2. Antibacterial Activity
2.1.3. Antifungal Activity
2.2. Antiparasitic Activity
2.3. Antiviral Activity
2.4. Anti-Inflammatory Activity
2.5. Cytoprotective Activity
2.6. Antitumor Activity
| Comp. | IC50 MCF-7 | IC50 K562 | Ref. |
|---|---|---|---|
| 69 | 2.50 µM | 3.10 µM | [69] |
| 70 | 2.70 µM | 3.50 µM | [69] |
| 71 | 7.26 µM | 9.91 µM | [70] |
| 72 | >50 µM | 1.02 µM | [71] |
| 73 | 5.18 µM | 2.89 µM | [72] |
| 74 | 5.91 µM | 5.02 µM | [73] |
| 75 | 20.20 µM | 9.30 µM | [74] |
| 76 | 4.50 µM | 1.10 µM | [75] |
| 79 | 4.30 µM | 3.00 µM | [77] |
| 81 | 0.09 µM | - | [78] |
2.7. Furin Inhibition
2.8. Acetylocholinesterase Inhibition
3. Summary
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AChE | acetylcholinesterase |
| BChE | butyrylocholinesterase |
| CNS | central nervous system |
| COX | cyclooxygenase |
| EC50 | half maximal effective concentration |
| HIV-RT | HIV reverse transcriptase |
| IC50 | half-maximal inhibitory concentration |
| IL-6 | interleukin-6 |
| LD50 | dose which causes the death of 50% of a group of test animals |
| LPS | lipopolysaccharide |
| MBC | minimal bactericidal concentration |
| MIC | minimal inhibitory concentration |
| MRSA | methicillin-resistant Staphylococcus aureus |
| MSSA | methicillin-susceptible Staphylococcus aureus |
| PBMC | peripheral blood mononuclear cell |
| TNF-α | tumor necrosis factor |
References
- Aly, A.; El-Din, A.M.N. Functionality of amidines and amidrazones. Arkivoc 2008, 1, 153–194. [Google Scholar] [CrossRef]
- Neilson, D.G.; Roger, R.; Heatlie, J.W.M.; Newlands, L.R. Chemistry of amidrazones. Chem. Rev. 1970, 70, 151–170. [Google Scholar] [CrossRef] [PubMed]
- Ben Salem, A.; Ben Salah, B.; Mhalla, D.; Trigui, M.; Mourer, M.; Regnouf-de-Vains, J.-B.; Kossentini, M. Synthesis, crystal structure and biological studies of novel amidrazones, triazoles, Thiatriazole and Triazine compounds. J. Mol. Struct. 2020, 1214, 128209. [Google Scholar] [CrossRef]
- Stepanov, A.I.; Sannikov, V.S.; Dashko, D.V.; Roslyakov, A.G.; Astrat’Ev, A.A.; Stepanova, E.V. A new preparative method and some chemical properties of 4-R-furazan-3-carboxylic acid amidrazones. Chem. Heterocycl. Compd. 2015, 51, 350–360. [Google Scholar] [CrossRef]
- Nomenclature of Organic Chemistry. IUPAC Recommendations and Preferred Names 2013. Available online: https://iupac.qmul.ac.uk/BlueBook/PDF/2013BlueBook.pdf (accessed on 18 February 2022).
- Abdel-Aziz, H.A.; Mekawey, A.A. Stereoselective synthesis and antimicrobial activity of benzofuran-based (1E)-1-(piperidin-1-yl)-N2-arylamidrazones. Eur. J. Med. Chem. 2009, 44, 4985–4997. [Google Scholar] [CrossRef]
- Modzelewska-Banachiewicz, B.; Matysiak, J.; Niewiadomy, A. Synthesis and mycological activity of the compounds obtained in the reaction of N3-substituted amidrazones with sulphinyl-bis-2,4-dihydroxybenzenethioyl. Eur. J. Med. Chem. 2001, 36, 75–80. [Google Scholar] [CrossRef]
- Gokhale, N.H.; Padhye, S.B.; Billington, D.C.; Rathbone, D.L.; Croft, S.L.; Kendrick, H.D.; Anson, C.E.; Powell, A.K. Synthesis and characterization of copper(II) complexes of pyridine-2-carboxamidrazones as potent antimalarial agents. Inorg. Chim. Acta 2003, 349, 23–29. [Google Scholar] [CrossRef]
- Modzelewska-Banachiewicz, B.; Michalec, B.; Kamińska, T.; Mazur, L.; Kozioł, A.E.; Banachiewicz, J.; Ucherek, M.; Kandefer-Szerszeń, M. Synthesis and biological activity of (Z) and (E) isomers of 3-(3,4-diaryl-1,2,4-triazole-5-yl)prop-2-enoic acid. Monatsh. Chem. 2009, 140, 439–444. [Google Scholar] [CrossRef]
- Hassan, H.Y.; El-Shorbagi, A.-N.; El-Koussi, N.A.; Abdel-Zaher, A.O. Design and synthesis of’ some new 1H-1,2,4-triazoles of potential anti-inflammatory and analgesic activities. Bull. Pharm. Sci. Assiut 1994, 17, 27–39. [Google Scholar] [CrossRef]
- Modzelewska-Banachiewicz, B.; Banachiewicz, J.; Chodkowska, A.; Jagiełło-Wójtowicz, E.; Mazur, L. Synthesis and biological activity of new derivatives of 3-(3,4-diaryl-1,2,4-triazole-5-yl)propenoic acid. Eur. J. Med. Chem. 2004, 39, 873–877. [Google Scholar] [CrossRef]
- Cocco, M.T.; Onnis, V.; Ponticelli, G.; Meier, B.; Rehder, D.; Garribba, E.; Micera, G. Synthesis, characterisation and insulin-mimetic activity of oxovanadium(IV) complexes with amidrazone derivatives. J. Inorg. Biochem. 2007, 101, 19–29. [Google Scholar] [CrossRef]
- Lee, K.; Jung, W.-H.; Park, C.W.; Park, H.D.; Lee, S.H.; Kwon, O.H. Noncovalent tripeptidic thrombin inhibitors incorporating amidrazone, amine and amidine functions at P1. Bioorg. Med. Chem. Lett. 2002, 12, 1017–1022. [Google Scholar] [CrossRef]
- Sączewski, F.; Balewski, Ł. Biological activities of guanidine compounds. Expert Opin. Ther. Patents 2009, 19, 1417–1448. [Google Scholar] [CrossRef]
- Cho, Y.L.; Jang, J. Development of Delpazolid for the Treatment of Tuberculosis. Appl. Sci. 2020, 10, 2211. [Google Scholar] [CrossRef]
- ClinicalTrials. Available online: http://www.clinicaltrials.gov/ct2/show/NCT04550832 (accessed on 22 May 2021).
- Zampieri, D.; Mamolo, M.G.; Vio, L.; Romano, M.; Skoko, N.; Baralle, M.; Pau, V.; De Logu, A. Antimycobacterial activity of new N1-[1-[1-aryl-3-[4-(1H-imidazol-1-yl)phenyl]-3-oxo]propyl]-pyridine-2-carboxamidrazone derivatives. Bioorg. Med. Chem. Lett. 2016, 26, 3287–3290. [Google Scholar] [CrossRef]
- Krause, M.; Foks, H.; Ziembicka, D.; Augustynowicz-Kopeć, E.; Głogowska, A.; Korona-Głowniak, I.; Bojanowski, K.; Siluk, D.; Gobis, K. 4-Substituted picolinohydrazonamides as a new class of potential antitubercular agents. Eur. J. Med. Chem. 2020, 190, 112106. [Google Scholar] [CrossRef]
- Tapera, M.; Kekeçmuhammed, H.; Sahin, K.; Krishna, V.S.; Lherbet, C.; Homberset, H.; Chebaiki, M.; Tønjum, T.; Mourey, L.; Zorlu, Y.; et al. Synthesis, characterization, anti-tuberculosis activity and molecular modeling studies of thiourea derivatives bearing aminoguanidine moiety. J. Mol. Struct. 2022, 1270, 133899. [Google Scholar] [CrossRef]
- Foks, H.; Balewski, L.; Gobis, K.; Dabrowska-Szponar, M.; Wisniewska, K. Studies on pyrazine derivatives LII: Antibacterial and antifungal activity of nitrogen heterocyclic compounds obtained by pyrazinamidrazone usage. Heteroat. Chem. 2011, 23, 49–58. [Google Scholar] [CrossRef]
- Senina, A.S.; Evdokimov, A.A.; Moskvin, A.V.; Fedorova, E.V. Synthesis, characterization and antimicrobial activity of amidrazone derivatives. J. Adv. Chem. Sci. 2016, 2, 183–187. Available online: https://www.jacsdirectory.com/journal-of-advanced-chemical-sciences/articleview.php?id=69 (accessed on 21 September 2022).
- Abdullah, A.H.; Zahra, J.A.; El-Abadelah, M.M.; Sabri, S.S.; Khanfar, M.A.; Matar, S.A.; Voelter, W. Synthesis and antibacterial activity of N1-(carbazol-3-yl)amidrazones incorporating piperazines and related congeners. Z. Naturforsch. 2016, 71, 857–867. [Google Scholar] [CrossRef]
- Saadeh, H.A.; Al-Qaoud, K.M.; Abu-Qatouseh, L.F.; Shihab, P.A.; Kaur, H.; Goyal, K.; Sehgal, R.; Mubarak, M.S. Synthesis and biological activity of novel amidrazones incorporating 5-nitroimidazole, ciprofloxacin, and 7-chloro-4-piperazinylquinoline. Med. Chem. Res. 2015, 24, 2247–2256. [Google Scholar] [CrossRef]
- Hkiri, S.; Hafidh, A.; Cavalier, J.-F.; Touil, S.; Samarat, A. Design, synthesis, antimicrobial evaluation, and molecular docking studies of novel symmetrical 2,5-difunctionalized 1,3,4-oxadiazoles. J. Heterocycl. Chem. 2019, 57, 1044–1054. [Google Scholar] [CrossRef]
- Wei, Z.-Y.; Chi, K.-Q.; Yu, Z.-K.; Liu, H.-Y.; Sun, L.-P.; Zheng, C.-J.; Piao, H.-R. Synthesis and biological evaluation of chalcone derivatives containing aminoguanidine or acylhydrazone moieties. Bioorg. Med. Chem. Lett. 2016, 26, 5920–5925. [Google Scholar] [CrossRef]
- Li, Y.-R.; Li, C.; Liu, J.-C.; Guo, M.; Zhang, T.-Y.; Sun, L.-P.; Zheng, C.-J.; Piao, H.-R. Synthesis and biological evaluation of 1,3-diaryl pyrazole derivatives as potential antibacterial and anti-inflammatory agents. Bioorg. Med. Chem. Lett. 2015, 25, 5052–5057. [Google Scholar] [CrossRef]
- Bai, X.; Zhao, L.; Liu, Z.; Li, Y.; Zhang, T.; Liu, X. Synthesis and antibacterial activity evaluation of aminoguanidine or dihydrotriazine derivatives. Indian J. Biochem. Biophys. 2019, 56, 301–308. Available online: http://op.niscair.res.in/index.php/IJBB/article/view/27359/0 (accessed on 15 September 2022).
- Song, M.; Wang, S.; Wang, Z.; Fu, Z.; Zhou, S.; Cheng, H.; Liang, Z.; Deng, X. Synthesis, antimicrobial and cytotoxic activities, and molecular docking studies of N-arylsulfonylindoles containing an aminoguanidine, a semicarbazide, and a thiosemicarbazide moiety. Eur. J. Med. Chem. 2019, 166, 108–118. [Google Scholar] [CrossRef]
- Yao, X.; Hu, H.; Wang, S.; Zhao, W.; Song, M.; Zhou, Q. Synthesis, Antimicrobial Activity, and Molecular Docking Studies of Aminoguanidine Derivatives Containing an Acylhydrazone Moiety. Iran. J. Pharm. Res. 2021, 20, 536–545. [Google Scholar] [CrossRef]
- Elsebaei, M.; Mohammad, H.; Abouf, M.; Abutaleb, N.; Hegazy, Y.A.; Ghiaty, A.; Chen, L.; Zhang, J.; Malwal, S.R.; Oldfield, E.; et al. Alkynyl-containing phenylthiazoles: Systemically active antibacterial agents effective against methicillin-resistant Staphylococcus aureus (MRSA). Eur. J. Med. Chem. 2018, 148, 195–209. [Google Scholar] [CrossRef]
- Paprocka, R.; Modzelewska-Banachiewicz, B.; Pazderski, L.; Mazur, L.; Kutkowska, J.; Niedzielska, D.; Psurski, M.; Wietrzyk, J.; Sączewski, J. Synthesis, crystal structure, 1H, 13C and 15N NMR studies, and biological evaluation of a new amidrazone-derived Au(III) complex. J. Mol. Struct. 2019, 1176, 357–365. [Google Scholar] [CrossRef]
- Senina, A.S.; Gurina, S.V.; Moskvin, A.V. Antimicrobial activity of amidrazone hydrohalogenides. Farmacija 2017, 66, 41–44. [Google Scholar]
- Kumar, N.S.; Pradeep, T.; Jani, G.; Silpa, D.; Kumar, B.V. Design, synthesis, and antimicrobial screening of novel pyridyl-2-amidrazone incorporated isatin mannich bases. J. Adv. Pharm. Technol. Res. 2012, 3, 57–61. Available online: https://www.japtr.org/article.asp?issn=2231-4040;year=2012;volume=3;issue=1;spage=57;epage=61;aulast=Kumar (accessed on 21 September 2022). [PubMed]
- Ribeiro, A.I.; Gabriel, C.; Cerqueira, F.; Maia, M.; Pinto, E.; Sousa, J.C.; Medeiros, R.; Proença, M.F.; Dias, A.M. Synthesis and antimicrobial activity of novel 5-aminoimidazole-4-carboxamidrazones. Bioorg. Med. Chem. Lett. 2014, 24, 4699–4702. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, C.; Grenho, L.; Cerqueira, F.; Medeiros, R.; Dias, A.M.; Ribeiro, A.I.; Proença, M.F.; Fernandes, M.H.; Sousa, J.C.; Monteiro, F.J.; et al. Inhibitory Effect of 5-Aminoimidazole-4-Carbohydrazonamides Derivatives against Candida spp. Biofilm on Nanohydroxyapatite Substrate. Mycopathologia 2019, 184, 775–786. [Google Scholar] [CrossRef] [PubMed]
- Cerqueira, F.; Maia, M.; Gabriel, C.; Medeiros, R.; Cravo, S.; Ribeiro, A.; Dantas, D.; Dias, A.; Saraiva, L.; Raimundo, L.; et al. Mechanism of Antifungal Activity by 5-Aminoimidazole-4-Carbohydrazonamide Derivatives against Candida albicans and Candida krusei. Antibiotics 2021, 10, 183. [Google Scholar] [CrossRef]
- Douadi, K.; Chafaa, S.; Douadi, T.; Al-Noaimi, M.; Kaabi, I. Azoimine quinoline derivatives: Synthesis, classical and electrochemical evaluation of antioxidant, anti-inflammatory, antimicrobial activities and the DNA/BSA binding. J. Mol. Struct. 2020, 1217, 128305. [Google Scholar] [CrossRef]
- Babahan, I.; Çoban, E.P.; Özmen, A.; Biyik, H.; Isman, B. Synthesis, characterization and biological activity of vic-dioxime derivatives containing benzaldehydehydrazone groups and their metal complexes. Afr. J. Microbiol. Res. 2011, 5, 271–283. [Google Scholar] [CrossRef]
- Biyik, H.; Babahan, I.; Coban, E.P. Synthesis, characterisation and antimicrobial activities of vic-dioxime derivatives containing heteroaromatic hydrazone groups and their metal complexes. Maejo Int. J. Sci. Technol. 2013, 7, 26–41. [Google Scholar] [CrossRef]
- Ajdačić, V.; Senerovic, L.; Vranić, M.; Pekmezovic, M.; Arsic-Arsnijevic, V.; Veselinovic, A.; Veselinovic, J.; Šolaja, B.A.; Nikodinovic-Runic, J.; Opsenica, I.M. Synthesis and evaluation of thiophene-based guanylhydrazones (iminoguanidines) efficient against panel of voriconazole-resistant fungal isolates. Bioorg. Med. Chem. 2016, 24, 1277–1291. [Google Scholar] [CrossRef]
- Do Nascimento, M.S.; Câmara, V.R.; Da Costa, J.S.; Barbosa, J.M.; Lins, A.S.; Salomão, K.; De Castro, S.L.; Carvalho, S.A.; Da Silva, E.F.; Fraga, C.A. Identification of Novel Functionalized Carbohydrazonamides Designed as Chagas Disease Drug Candidates. Med. Chem. 2020, 16, 774–783. [Google Scholar] [CrossRef]
- Abraham, R.J.; Abraham, S.; Stevens, A.J.; Page, S.W.; McCluskey, A.; Trott, D.J.; O’Handley, R.M. Aminoguanidines: New leads for treatment of Giardia duodenalis infection. Int. J. Parasitol. Drugs Drug Resist. 2019, 10, 38–44. [Google Scholar] [CrossRef]
- Benmerzouga, I.; Checkley, L.A.; Ferdig, M.T.; Arrizabalaga, G.; Wek, R.C.; Sullivan, W.J., Jr. Guanabenz Repurposed as an Antiparasitic with Activity against Acute and Latent Toxoplasmosis. Antimicrob. Agents Chemother. 2015, 59, 6939–6945. [Google Scholar] [CrossRef]
- Konrad, C.; Queener, S.F.; Wek, R.C.; Sullivan, W.J. Inhibitors of eIF2α Dephosphorylation Slow Replication and Stabilize Latency in Toxoplasma gondii. Antimicrob. Agents Chemother. 2013, 57, 1815–1822. [Google Scholar] [CrossRef]
- De Aquino, T.M.; França, P.H.B.; Rodrigues, E.E.S.; Nascimento, I.J.; Santos-Júnior, P.F.S.; Aquino, P.G.V.; Santos, M.S.; Queiroz, A.C.; Araújo, M.V.; Alexandre-Moreira, M.S.; et al. Synthesis, Antileishmanial Activity and in silico Studies of Aminoguanidine Hydrazones (AGH) and Thiosemicarbazones (TSC) Against Leishmania chagasi Amastigotes. Med. Chem. 2022, 18, 151–169. [Google Scholar] [CrossRef]
- Paprocka, R.; Kołodziej, P.; Wiese-Szadkowska, M.; Helmin-Basa, A.; Bogucka-Kocka, A. Evaluation of Anthelmintic and Anti-Inflammatory Activity of 1,2,4-Triazole Derivatives. Molecules 2022, 27, 4488. [Google Scholar] [CrossRef]
- Abdel-Aziza, H.A.; Abdel-Wahab, B.F.; Badria, F.A. Stereoselective Synthesis and Antiviral Activity of (1E,2Z,3E)-1-(Piperidin-1-yl)-1-(arylhydrazono)-2-[(benzoyl/benzothiazol- 2-oyl)hydrazono]-4-(aryl1)but-3-enes. Arch. Pharm. 2010, 343, 152–159. [Google Scholar] [CrossRef]
- Gomha, S.M.; Badrey, M.G.; Abdalla, M.M.; Arafa, R.K. Novel anti-HIV-1 NNRTIs based on a pyrazolo[4,3-d]isoxazole backbone scaffold: Design, synthesis and insights into the molecular basis of action. MedChemComm 2014, 5, 1685–1692. [Google Scholar] [CrossRef]
- Mazur, L.; Modzelewska-Banachiewicz, B.; Paprocka, R.; Zimecki, M.; Wawrzyniak, U.E.; Kutkowska, J.; Ziółkowska, G. Synthesis, crystal structure and biological activities of a novel amidrazone derivative and its copper(II) complex—A potential antitumor drug. J. Inorg. Biochem. 2012, 114, 55–64. [Google Scholar] [CrossRef]
- Modzelewska-Banachiewicz, B.; Ucherek, M.; Zimecki, M.; Kutkowska, J.; Kaminska, T.; Morak-Młodawska, B.; Paprocka, R.; Szulc, M.; Lewandowski, G.; Marciniak, J.; et al. Reactions of N3-Substituted Amidrazones with cis-1,2-Cyclohexanedicarboxylic Anhydride and Biological Activities of the Products. Arch. Pharm. 2012, 345, 486–494. [Google Scholar] [CrossRef]
- Paprocka, R.; Modzelewska-Banachiewicz, B.; Wiese, M.; Eljaszewicz, A.; Michałkiewicz, J. Synthesis and anti-inflammatory activity of hydrazide derivatives of 2-methylidene-1,4-dicarboxybutanoic acid. Acta Pol. Pharm. 2012, 69, 1390–1394. Available online: https://www.ptfarm.pl/pub/File/Acta_Poloniae/2012/6/1390.pdf (accessed on 22 November 2012).
- Paprocka, R.; Wiese-Szadkowska, M.; Helmin-Basa, A.; Mazur, L.; Kutkowska, J.; Michałkiewicz, J.; Modzelewska-Banachiewicz, B.; Pazderski, L. Synthesis and evaluation of new amidrazone-derived hydrazides as a potential anti-inflammatory agents. Monatsh. Chem. 2018, 149, 1493–1500. [Google Scholar] [CrossRef]
- Paprocka, R.; Wiese, M.; Eljaszewicz, A.; Helmin-Basa, A.; Gzella, A.; Modzelewska-Banachiewicz, B.; Michalkiewicz, J. Synthesis and anti-inflammatory activity of new 1,2,4-triazole derivatives. Bioorg. Med. Chem. Lett. 2015, 25, 2664–2667. [Google Scholar] [CrossRef]
- Paprocka, R.; Pazderski, L.; Mazur, L.; Wiese-Szadkowska, M.; Kutkowska, J.; Nowak, M.; Helmin-Basa, A. Synthesis and Structural Study of Amidrazone Derived Pyrrole-2,5-Dione Derivatives: Potential Anti-Inflammatory Agents. Molecules 2022, 27, 2891. [Google Scholar] [CrossRef]
- El-Din, N.; Barseem, A. Synthesis, bioactivity and Docking Study of Some New Indole-hydrazone Derivatives. J. Appl. Pharm. Sci. 2016, 6, 075–083. [Google Scholar] [CrossRef][Green Version]
- Zhou, H.; Wang, Z.S.; Liu, X.H.; Chen, F.H. Novel amidrazone derivatives: Design, synthesis and activity evaluation. Bioorg. Med. Chem. 2018, 26, 3158–3165. [Google Scholar] [CrossRef]
- Pasten, C.; Lozano, M.; Rocco, J.; Carrión, F.; Alvarado, C.; Liberona, J.; Michea, L.; Irarrázabal, C.E. Aminoguanidine Prevents the Oxidative Stress, Inhibiting Elements of Inflammation, Endothelial Activation, Mesenchymal Markers, and Confers a Renoprotective Effect in Renal Ischemia and Reperfusion Injury. Antioxidants 2021, 10, 1724. [Google Scholar] [CrossRef]
- Heimfarth, L.; Carvalho, A.M.S.; Quintans, J.D.S.S.; Pereira, E.W.M.; Teles Lima, N.; Carvalho, M.T.B.; Barreto, R.D.S.S.; Moreira, J.C.F.; Da Silva-Júnior, E.F.; Schmitt, M.; et al. Indole-3-guanylhydrazone hydrochloride mitigates long-term cognitive impairment in a neonatal sepsis model with involvement of MAPK and NFκB pathways. Neurochem. Int. 2020, 134, 104647. [Google Scholar] [CrossRef]
- Luh, L.M.; Bertolotti, A. Potential benefit of manipulating protein quality control systems in neurodegenerative diseases. Curr. Opin. Neurobiol. 2020, 61, 125–132. [Google Scholar] [CrossRef]
- ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT02423083 (accessed on 21 February 2022).
- Bella, E.D.; Bersano, E.; Antonini, G.; Borghero, G.; Capasso, M.; Caponnetto, C.; Chiò, A.; Corbo, M.; Filosto, M.; Giannini, F.; et al. The unfolded protein response in amyotrophic later sclerosis: Results of a phase 2 trial. Brain 2021, 144, 2635–2647. [Google Scholar] [CrossRef]
- Wang, L.; Popko, B.; Tixier, E.; Roos, R.P. Guanabenz, which enhances the unfolded protein response, ameliorates mutant SOD1-induced amyotrophic lateral sclerosis. Neurobiol. Dis. 2014, 71, 317–324. [Google Scholar] [CrossRef]
- Martynowicz, J.; Augusto, L.; Wek, R.C.; Boehm, S.L.; Sullivan, W.J. Guanabenz Reverses a Key Behavioral Change Caused by Latent Toxoplasmosis in Mice by Reducing Neuroinflammation. mBio 2019, 10, e00381-19. [Google Scholar] [CrossRef]
- ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT03124459 (accessed on 21 February 2022).
- Chen, Y.; Podojil, J.R.; Kunjamma, R.B.; Jones, J.; Weiner, M.; Lin, W.; Miller, S.D.; Popko, B. Sephin1, which prolongs the integrated stress response, is a promising therapeutic for multiple sclerosis. Brain 2019, 142, 344–361. [Google Scholar] [CrossRef] [PubMed]
- Krzyzosiak, A.; Sigurdardottir, A.; Luh, L.; Carrara, M.; Das, I.; Schneider, K.; Bertolotti, A. Target-Based Discovery of an Inhibitor of the Regulatory Phosphatase PPP1R15B. Cell 2018, 174, 1216–1228.e19. [Google Scholar] [CrossRef] [PubMed]
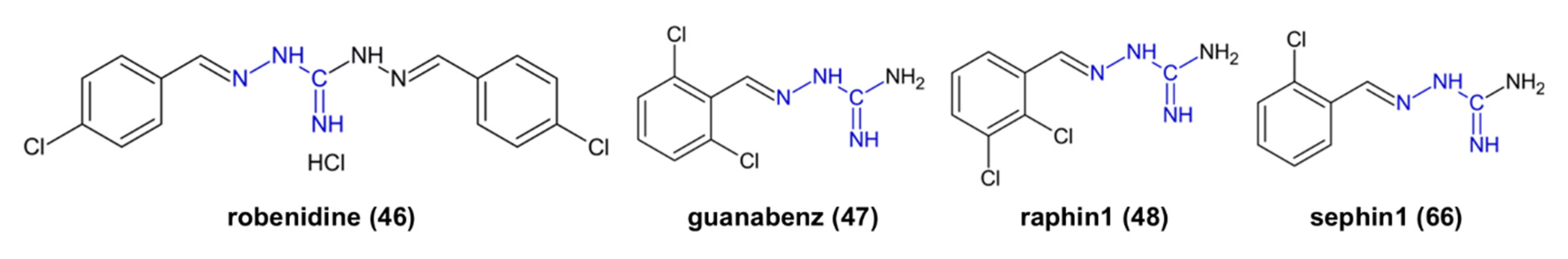
- Claes, Z.; Jonkhout, M.; Crespillo-Casado, A.; Bollen, M. The antibiotic robenidine exhibits guanabenz-like cytoprotective properties by a mechanism independent of protein phosphatase PP1:PPP1R15A. J. Biol. Chem. 2019, 294, 13478–13486. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Jalil, R.J.; El Momani, E.Q.; Hamad, M.; Voelter, W.; Mubarak, M.S.; Smith, B.H.; Peters, D.G. Synthesis, antitumor activity, and electrochemical behavior of some piperazinyl amidrazones. Monatsh. Chem. 2010, 141, 251–258. [Google Scholar] [CrossRef]
- Habashneh, A.Y.; El-Abadelah, M.M.; Bardaweel, S.K.; Taha, M. Synthesis and Structure-Activity Relationship; Exploration of some Potent Anti-Cancer Phenyl Amidrazone Derivatives. Med. Chem. 2018, 14, 468–477. [Google Scholar] [CrossRef]
- Abadleh, M.M.; El-Abadelah, M.M.; Sabri, S.S.; Mohammed, H.H.; Zihlif, M.A.; Voelter, W. Synthesis and Antitumor Activity of Some N2-(Thien-3-yl)amidrazones. Z. Naturforsch. 2014, 69, 811–816. [Google Scholar] [CrossRef]
- Almansour, A.M.; Zahra, J.A.; Sabri, S.S.; El-Abadelah, M.M.; Zihlif, M.A.; Taha, M.O. Synthesis and Anticancer Properties of Methyl N1-(thien-4-yl)amidrazone- 3-carboxylates. Lett. Drug Des. Discov. 2018, 15, 1268–1275. [Google Scholar] [CrossRef]
- Habashneh, A.Y.; Zihlif, M.A.; Imraish, A.; Taha, M.O.; El-Abadelah, M.M. Synthesis and Antitumor Activities of Some New N1-(Flavon-6-yl)amidrazone Derivatives. Arch. Pharm. 2014, 347, 415–422. [Google Scholar] [CrossRef]
- Abu-Aisheh, M.N.; Mustafa, M.S.; El-Abadelah, M.M.; Naffa, R.G.; Ismail, S.I.; Zihlif, M.A.; Taha, M.O.; Mubarak, M.S. Synthesis and biological activity assays of some new N1-(flavon-7-yl)amidrazone derivatives and related congeners. Eur. J. Med. Chem. 2012, 54, 65–74. [Google Scholar] [CrossRef]
- Mustafa, M.S.; El-Abadelah, M.M.; Zihlif, M.A.; Naffa, R.; Mubarak, M.S. Synthesis, and Antitumor Activity of Some N1-(Coumarin-7-yl) Amidrazones and Related Congeners. Molecules 2011, 16, 4305–4317. [Google Scholar] [CrossRef]
- Sweidan, K.; Zalloum, H.; Sabbah, D.A.; Idris, G.; Abudosh, K.; Mubarak, M.S. Synthesis, characterization, and anticancer evaluation of some new N1-(anthraquinon-2-yl) amidrazone derivatives. Can. J. Chem. 2018, 96, 1123–1128. [Google Scholar] [CrossRef]
- Sabbah, D.A.; Hajjo, R.; Sweidan, K.; Zhong, H.A. An Integrative Informatics Approach to Explain the Mechanism of Action of N1-(Anthraquinon-2-yl) Amidrazones as BCR/ABL Inhibitors. Curr. Comput. Aided-Drug Des. 2020, 17, 817–830. [Google Scholar] [CrossRef]
- Al-Qtaitat, M.A.; El-Abadelah, M.M.; Sabbah, D.A.; Bardaweel, S.; Sweidan, K.; Sabri, S.S.; Mubarak, M.S. Synthesis, characterization, and bioactivity of new bisamidrazone derivatives as possible anticancer agents. Med. Chem. Res. 2018, 27, 1419–1431. [Google Scholar] [CrossRef]
- Qian, Y.; Zhang, H.-J.; Lv, P.-C.; Zhu, H.-L. Synthesis, molecular modeling and biological evaluation of guanidine derivatives as novel antitubulin agents. Bioorg. Med. Chem. 2010, 18, 8218–8225. [Google Scholar] [CrossRef]
- Andreani, A.; Granaiola, M.; Leoni, A.; Locatelli, A.; Morigi, R.; Rambaldi, M.; Varoli, L.; Lannigan, D.; Smith, J.; Scudiero, D.; et al. Imidazo[2,1-b]thiazole guanylhydrazones as RSK2 inhibitors. Eur. J. Med. Chem. 2011, 46, 4311–4323. [Google Scholar] [CrossRef]
- Basu, A.; Sinha, B.N.; Saiko, P.; Graser, G.; Szekeres, T. N-Hydroxy-N′-aminoguanidines as anti-cancer lead molecule: QSAR, synthesis and biological evaluation. Bioorg. Med. Chem. Lett. 2011, 21, 3324–3328. [Google Scholar] [CrossRef]
- Silva, F.P.L.; Dantas, B.B.; Martins, G.V.F.; De Araújo, D.A.M.; Vasconcellos, M.L.A.D.A. Synthesis and Anticancer Activities of Novel Guanylhydrazone and Aminoguanidine Tetrahydropyran Derivatives. Molecules 2016, 21, 671. [Google Scholar] [CrossRef]
- Liu, D.C.; Gao, M.J.; Huo, Q.; Ma, T.; Wang, Y.; Wu, C.Z. Design, synthesis, and apoptosis-promoting effect evaluation of novel pyrazole with benzo[d]thiazole derivatives containing aminoguanidine units. J. Enzym. Inhib. Med. Chem. 2019, 34, 829–837. [Google Scholar] [CrossRef]
- Al-Noaimi, M.; Awwadi, F.F.; Mansi, I.A.; Sawwan, M.; Abu-Irmaileh, B.; Dege, N. Polymorphism, spectroscopic, DFT and anticancer activity of a palladium(II) complex with a thiophenyl azoimine-quinoline SNN’N” ligand. Polyhedron 2022, 211, 115541. [Google Scholar] [CrossRef]
- Al-Noaimi, M.; Awwadi, F.F.; Talib, W.; Atia, S.; Hammud, H.H. Cis and trans- palladium (II) complexes derived from SNN amidrazone pincer ligand: Synthesis, crystal structures and biological evaluation. J. Mol. Struct. 2019, 1197, 282–291. [Google Scholar] [CrossRef]
- Lapasam, A.; Pinder, E.; Phillips, R.M.; Kaminsky, W.; Kollipara, M.R. Synthesis, structure and bonding modes of pyrazine based ligands of Cp*Rh and Cp*Ir complexes: The study of in-vitro cytotoxicity against human cell lines. J. Organomet. Chem. 2019, 899, 120887. [Google Scholar] [CrossRef]
- Dömötör, O.; May, N.V.; Gál, G.T.; Spengler, G.; Dobrova, A.; Arion, V.B.; Enyedy, É.A. Solution Equilibrium Studies on Salicylidene Aminoguanidine Schiff Base Metal Complexes: Impact of the Hybridization with L-Proline on Stability, Redox Activity and Cytotoxicity. Molecules 2022, 27, 2044. [Google Scholar] [CrossRef]
- Climova, A.; Pivovarova, E.; Rogalewicz, B.; Raducka, A.; Szczesio, M.; Korona-Głowniak, I.; Korga-Plewko, A.; Iwan, M.; Gobis, K.; Czylkowska, A. New Coordination Compounds Based on a Pyrazine Derivative: Design, Characterization, and Biological Study. Molecules 2022, 27, 3467. [Google Scholar] [CrossRef]
- Czylkowska, A.; Rogalewicz, B.; Szczesio, M.; Raducka, A.; Gobis, K.; Szymański, P.; Czarnecka, K.; Camargo, B.C.; Szczytko, J.; Babich, A.; et al. Antitumor Activity against A549 Cancer Cells of Three Novel Complexes Supported by Coating with Silver Nanoparticles. Int. J. Mol. Sci. 2022, 23, 2980. [Google Scholar] [CrossRef]
- Sielaff, F.; Than, M.E.; Bevec, D.; Lindberg, I.; Steinmetzer, T. New furin inhibitors based on weakly basic amidinohydrazones. Bioorg. Med. Chem. Lett. 2011, 21, 836–840. [Google Scholar] [CrossRef][Green Version]
- Cheng, Y.-W.; Chao, T.-L.; Li, C.-L.; Chiu, M.-F.; Kao, H.-C.; Wang, S.-H.; Pang, Y.-H.; Lin, C.-H.; Tsai, Y.-M.; Lee, W.-H.; et al. Furin Inhibitors Block SARS-CoV-2 Spike Protein Cleavage to Suppress Virus Production and Cytopathic Effects. Cell Rep. 2020, 33, 108254. [Google Scholar] [CrossRef]
- Abu-Aisheh, M.N.; Al-Aboudi, A.; Mustafa, M.S.; El-Abadelah, M.M.; Ali, S.Y.; Ul-Haq, Z.; Mubarak, M.S. Coumarin derivatives as acetyl- and butyrylcholinestrase inhibitors: An in vitro, molecular docking, and molecular dynamics simulations study. Heliyon 2019, 5, e01552. [Google Scholar] [CrossRef]
- Krátký, M.; Štěpánková, Š.; Konečná, K.; Svrčková, K.; Maixnerová, J.; Švarcová, M.; Janďourek, O.; Trejtnar, F.; Vinšová, J. Novel Aminoguanidine Hydrazone Analogues: From Potential Antimicrobial Agents to Potent Cholinesterase Inhibitors. Pharmaceuticals 2021, 14, 1229. [Google Scholar] [CrossRef]








| Comp. | IC50 AChE [µM] | IC50 BChE [µM] | Ref. |
|---|---|---|---|
| 75 | 24.25 ± 2.97 | 0.002 ± 0.0014 | [91] |
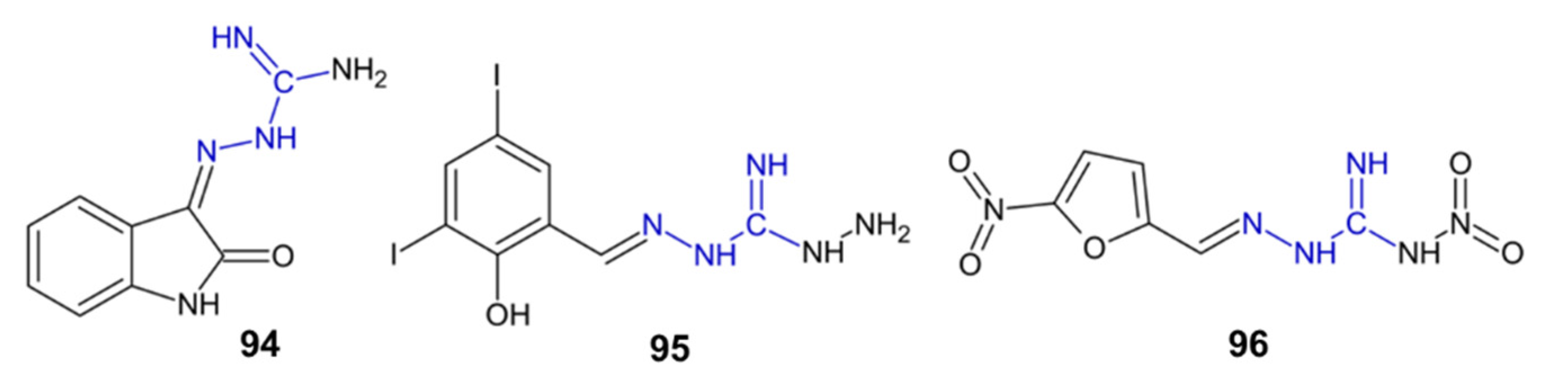
| 94 | 17.95 ± 0.90 | 17.51 ± 0.21 | [92] |
| 95 | 28.16 ± 0.98 | 1.69 ± 0.17 | [92] |
| 96 | 24.75 ± 0.17 | >500 | [92] |
| tacrine | 0.124 ± 0.02 | 7.8 ± 0.06 | [91] |
| rivastigmine | 56.10 ± 1.41 | 38.40 ± 1.97 | [92] |
| Comp. | Activity | Animal Model | Dose | Effect | Reference Drug | Ref. |
|---|---|---|---|---|---|---|
| 23 | anti-inflammatory | xylene-induced ear edema test in mice | 100 mg/kg | 92.45% edema reduction | indomethacin 89.38% reduction, ibuprofen 87.36% reduction | [25] |
| 25 | anti-inflammatory | xylene-induced ear edema test in mice | 50 mg/kg | 93.56% edema reduction | indomethacin 45.23% reduction, ibuprofen 29.56% reduction | [26] |
| 26 | 50 mg/kg | 81.65% edema reduction | ||||
| 30 | antibacterial | MRSA-infected C. elegans | 20 mg/mL | reduction in the MRSA burden by ~90% | vancomycin ~90% reduction | [30] |
| MRSA murine skin infection | 2% suspension | 73% reduction in MRSA burden | fusidic acid 78% reduction | |||
| MRSA-infected mice | 20 mg/kg | 77% reduction in MRSA burden | vancomycin 66% reduction | |||
| 31 | MRSA-infected C. elegans | 20 mg/mL | reduction in the MRSA burden by ~90% | vancomycin ~90% reduction | ||
| MRSA murine skin infection | 2% suspension | 71% reduction in MRSA burden | fusidic acid 78% reduction | |||
| 55 | anti-inflammatory | carrageenan-induced rat hind paw edema | 21 mg/kg | 65–73% edema reduction (0.5–2 h) | diclofenac 50–58% edema reduction (0.5–8 h) | [50] |
| 42 mg/kg | 38–60% edema reduction (0.5–2 h) | |||||
| antinociceptive | hot-plate test in mice | 21 mg/kg | analgesic effect (0.5–2 h) | morphine analgesic effect(0.5–1 h) | ||
| 42 mg/kg | analgesic effect (0.5–2 h) | |||||
| 60 | anti-inflammatory | carrageenan-induced rat hind paw edema | 65 mg/kg | 89.3% edema reduction | indomethacin 46% edema reduction | [55] |
| 61 | 65 mg/kg | 87.7% edema reduction | ||||
| 62 | 61 mg/kg | 80.7% edema reduction | ||||
| 63 | 61 mg/kg | 79.5% edema reduction | ||||
| 65 | anti-inflammatory neonatal sepsis treatment | LPS-induced sepsis in neonatal mice | 50 mg/kg | reduction in anxiety-like behavior and cognitive disorders in adult life | - | [58] |
| Comp. | Activity | Mechanism | Ref. |
|---|---|---|---|
| AG | anti-inflammatory | suppression of oxidative stress, inhibition of IL-1β, IL-6, and Foxp3 mRNA upregulation | [57] |
| 1 | antituberculosic | inhibiting protein synthesis via direct binding to the bacterial ribosomal subunit | [15] |
| 8 | antibacterial | inhA inhibition | [19] |
| 27 | antibacterial | inhibition of DHFR protein | [27] |
| 28 | antibacterial | interaction with E. coli FabH-CoA receptor. | [28] |
| 29 | antibacterial | interaction with β-ketoacyl-ACP synthase III (FabH) | [29] |
| 30 | antibacterial | inhibitor of undecaprenyl diphosphate phosphatase and undecaprenyl diphosphate | [30] |
| 38–39 | antifungal | interaction with DNA (intercalation) | [37] |
| 43 | antifungal | inhibition of 14-α-demethylase (CYP51) | [40] |
| 46 | antigiardial | inhibition of adherence of trophozoides | [42] |
| 47 | cytoprotective | inhibition of R15A, inhibition of dephosphorylation of enzyme eIF2α | [59] |
| 48 | cytoprotective | inhibition of R15B, inhibition of dephosphorylation of enzyme eIF2α | [66] |
| 48–50 | antiparasitic | binding trypanothione reductase enzyme | [45] |
| 53 | antiviral | inhibition of HIV-RT | [48] |
| 54 | anti-inflammatory | decreasing production of TNF-α | [49] |
| 55 | anti-inflammatory | decreasing production of IL-6 | [50] |
| 56 | anti-inflammatory | decreasing production of TNF-α | [51] |
| 57 | anti-inflammatory | G1 phase arrest | [52] |
| 58 | anti-inflammatory | decreasing production of IL-6 | [54] |
| 60–63 | anti-inflammatory | inhibition of COX-1 and COX-2 | [55] |
| 64 | antarthritic | inhibition expression of ASIC1a protein | [56] |
| 65 | anti-inflammatory | inhibition of NFκB activation | [58] |
| 66 | cytoprotective | inhibition of R15A, inhibition of dephosphorylation of enzyme eIF2α | [59] |
| 72 | antitumor | tyrosine kinase brc-abl inhibitor | [71] |
| 73 | antitumor | tyrosine kinase brc-abl inhibitor | [72] |
| 74 | antitumor | tyrosine kinase brc-abl inhibitor | [73] |
| 75 | antitumor | tyrosine kinase brc-abl inhibitor | [74] |
| 76 | antitumor | tyrosine kinase brc-abl inhibitor | [75] |
| 79 | antitumor | phosphatidylinositol 3-kinase inhibitor | [77] |
| 81 | antitumor | inhibition of tubulin polymerization, colchicine binding | [78] |
| 82 | antitumor | inhibition of ribosomal kinase RSK2 | [79] |
| 92 | enzyme inhibition | furin inhibitor, trypsin inhibitor | [89] |
| 93 | enzyme inhibition | furin inhibitor, thrombin inhibitor | [89] |
| 75 | enzyme inhibition | BChE inhibitor | [91] |
| 94 | enzyme inhibition | AChE and BChE inhibitor | [92] |
| 95–96 | enzyme inhibition | BChE inhibitor | [92] |
| Comp. | Animal Model | Time | Toxicity | Ref. |
| 18 | brine shrimp | 24 h | IC50 > 50 µg/mL | [23] |
| 19 | brine shrimp | 24 h | IC50 > 50 µg/mL | [23] |
| 20 | brine shrimp | 24 h | IC50 > 12.5 µg/mL | [23] |
| 21 | brine shrimp | 24 h | IC50 > 12.5 µg/mL | [23] |
| 43 | zebrafish embryos | 96 h | LC50 = 8.2 µg/mL | [40] |
| 55 | Swiss mice | - | LD50 = 417 mg/kg | [50] |
| 78 | brine shrimp | 24 h | IC50 > 50 µg/mL | [23] |
| Comp. | Studied cells | Origin | Toxicity | Ref. |
| 2 | Vero | monkey | IC50 = 28.7 µM | [17] |
| 3 | Vero | monkey | IC50 = 23.1 µM | [17] |
| 4 | Vero | monkey | IC50 = 27.8 µM | [17] |
| 5 | Vero | monkey | IC50 = 298 µM | [17] |
| 6 | fibroblasts | human | IC50 = 10.39 µg/mL | [18] |
| 7 | fibroblasts | human | IC50 = 3.29 µg/mL | [18] |
| 28 | HEK 293T | human | IC50 = 56.39 µmol/L | [28] |
| 32 | fibroblasts | mice | IC50 = 41.8 µg/mL | [31] |
| 43 | MRC-5 | human | IC50 = 2.5 µg/mL | [40] |
| 23 | LO2 | human | IC50 = 18.1 µg/mL | [25] |
| 30–31 | HRT-18 | human | IC50 > 32 µg/mL | [30] |
| 44 | macrophages | mice | IC50 = 79.59 µM | [41] |
| 45 | macrophages | mice | IC50 = 423.33 µM | [41] |
| 46 | RAW264.7 | mice | IC50 = 17.1 µM | [42] |
| 48–50 | J774.A1 | mice | IC50 > 10 μM | [45] |
| 51 | PBMC | human | IC50 > 100 µg/mL | [46] |
| 54 | PBMC | human | IC50 > 100 µg/mL | [49] |
| 56 | PBMC | human | IC50 > 10 µg/mL | [51] |
| 57 | PBMC | human | IC50 > 50 µg/mL | [52] |
| 58–59 | PBMC | human | IC50 > 100 µg/mL | [54] |
| 64 | chondrocytes | rat | IC50 > 25 µM | [56] |
| 72 | fibroblasts | human | IC50 > 50 µM | [71] |
| 76 | fibroblasts | human | IC50 = 15 µM | [75] |
| 83 | Vero | monkey | IC50 > 100 µM | [80] |
| 87 | Vero | monkey | IC50 > 611.09 µM | [84] |
| 88 | ARPE-19 | human | IC50 = 38.82 µM | [85] |
| 89 | ARPE-19 | human | IC50 = 41.23 µM | [85] |
| 90 | MRC-5 | human | IC50 = 58.9 µM | [86] |
| 91 | PBMC | human | IC50 > 25 µg/mL | [49] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paprocka, R.; Wiese-Szadkowska, M.; Kosmalski, T.; Frisch, D.; Ratajczak, M.; Modzelewska-Banachiewicz, B.; Studzińska, R. A Review of the Biological Activity of Amidrazone Derivatives. Pharmaceuticals 2022, 15, 1219. https://doi.org/10.3390/ph15101219
Paprocka R, Wiese-Szadkowska M, Kosmalski T, Frisch D, Ratajczak M, Modzelewska-Banachiewicz B, Studzińska R. A Review of the Biological Activity of Amidrazone Derivatives. Pharmaceuticals. 2022; 15(10):1219. https://doi.org/10.3390/ph15101219
Chicago/Turabian StylePaprocka, Renata, Małgorzata Wiese-Szadkowska, Tomasz Kosmalski, Daria Frisch, Magdalena Ratajczak, Bożena Modzelewska-Banachiewicz, and Renata Studzińska. 2022. "A Review of the Biological Activity of Amidrazone Derivatives" Pharmaceuticals 15, no. 10: 1219. https://doi.org/10.3390/ph15101219
APA StylePaprocka, R., Wiese-Szadkowska, M., Kosmalski, T., Frisch, D., Ratajczak, M., Modzelewska-Banachiewicz, B., & Studzińska, R. (2022). A Review of the Biological Activity of Amidrazone Derivatives. Pharmaceuticals, 15(10), 1219. https://doi.org/10.3390/ph15101219