A Comparative Survey of Anti-Melanoma and Anti-Inflammatory Potential of Usnic Acid Enantiomers—A Comprehensive In Vitro Approach
Abstract
:1. Introduction
2. Results and Discussion
2.1. Both Usnic Acid Enantiomers Dose- and Time-Dependently Decreased Melanoma Cells Viability
2.2. Both Usnic Acid Enantiomers Differently Interact with Doxorubicin Towards Melanoma Cells
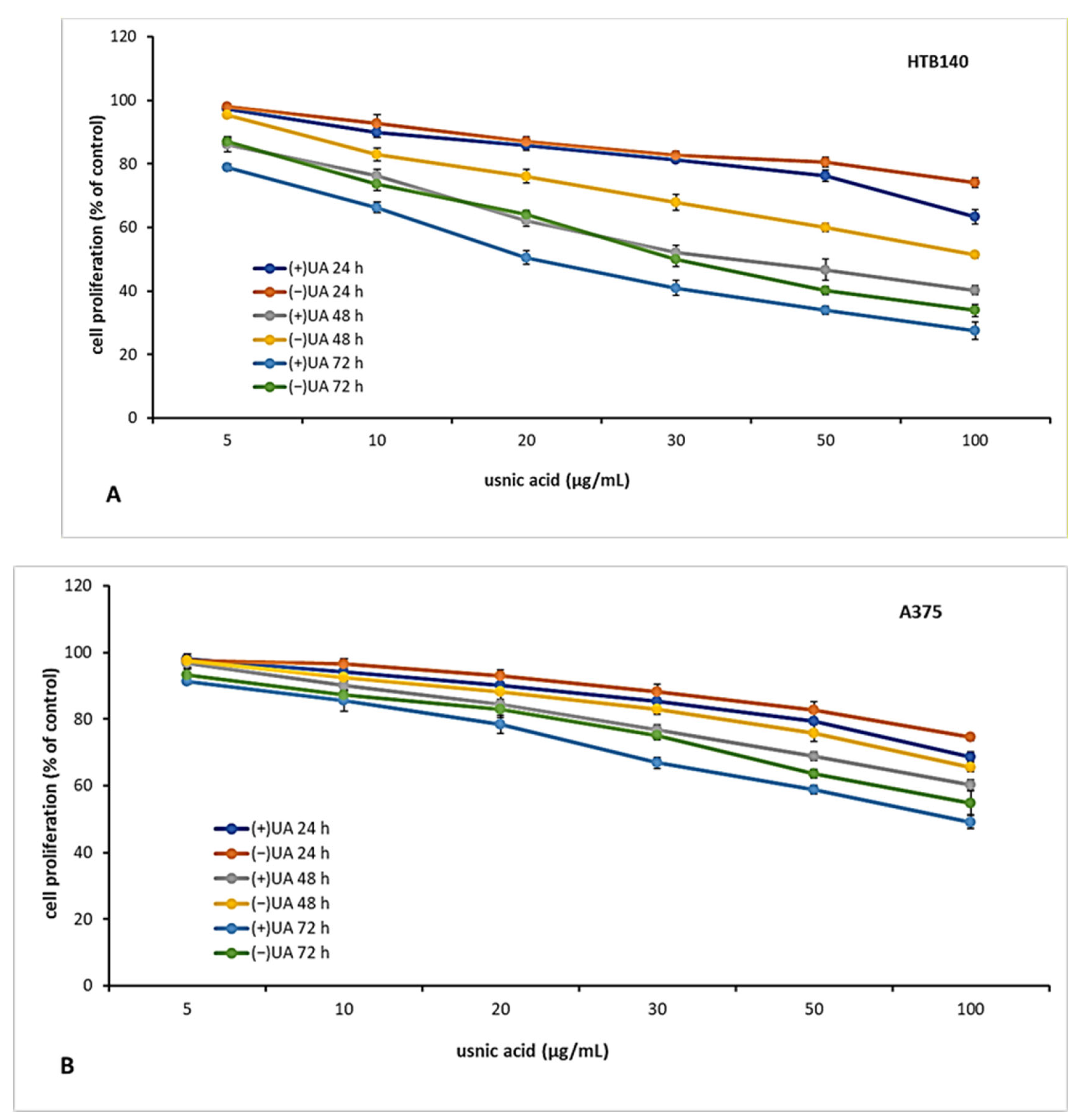
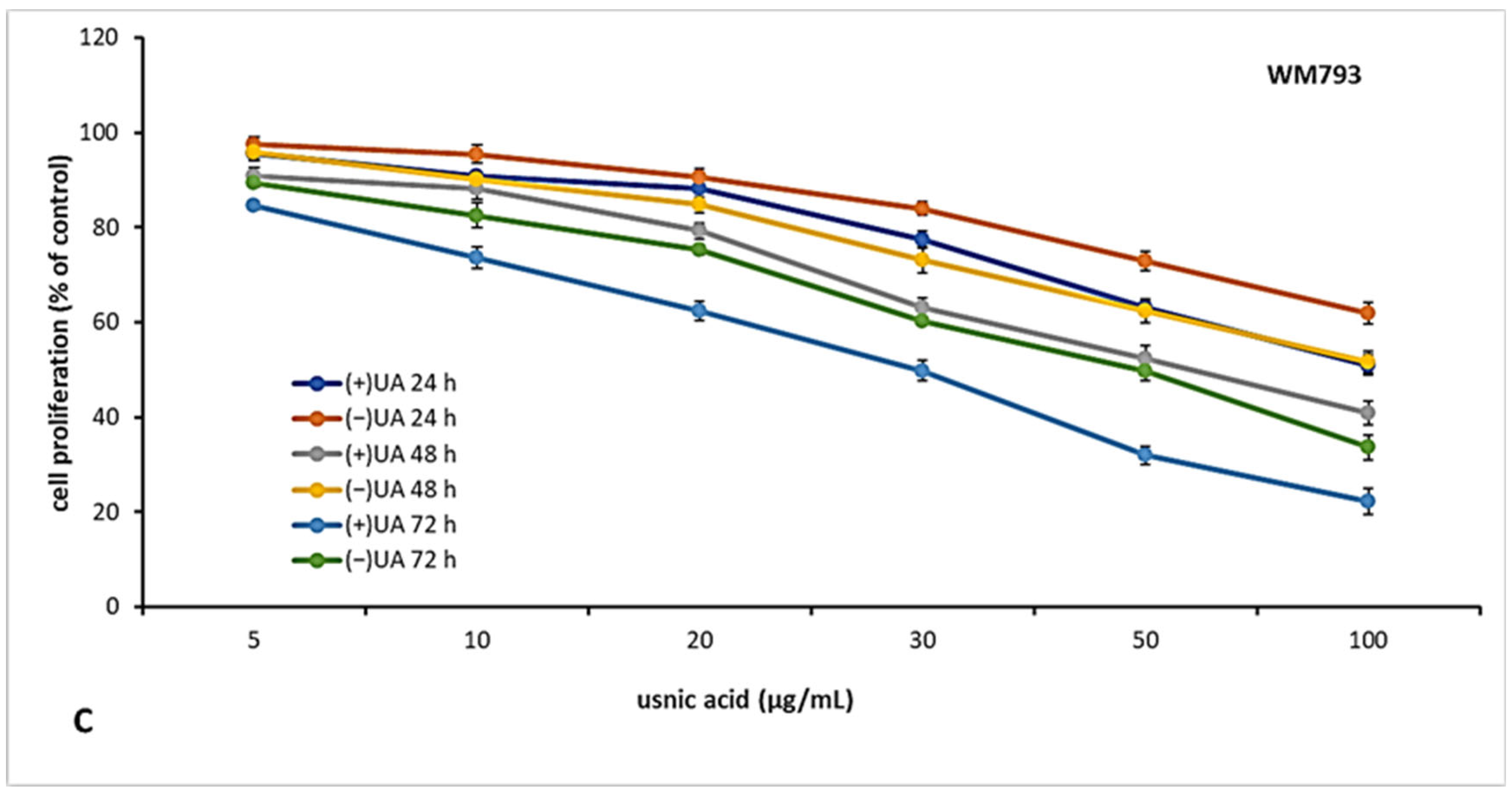
2.3. (+)-Usnic Acid Inhibited Proliferation of Melanoma Cells More Effectively Than (−)-Usnic Acid
2.4. (+)-Usnic Acid at a Sub-Cytotoxic Dose Strongly Inhibited Melanoma Cells Migration
2.5. None of Usnic Acid Enantiomers Effectively Inhibited Tyrosinase Activity
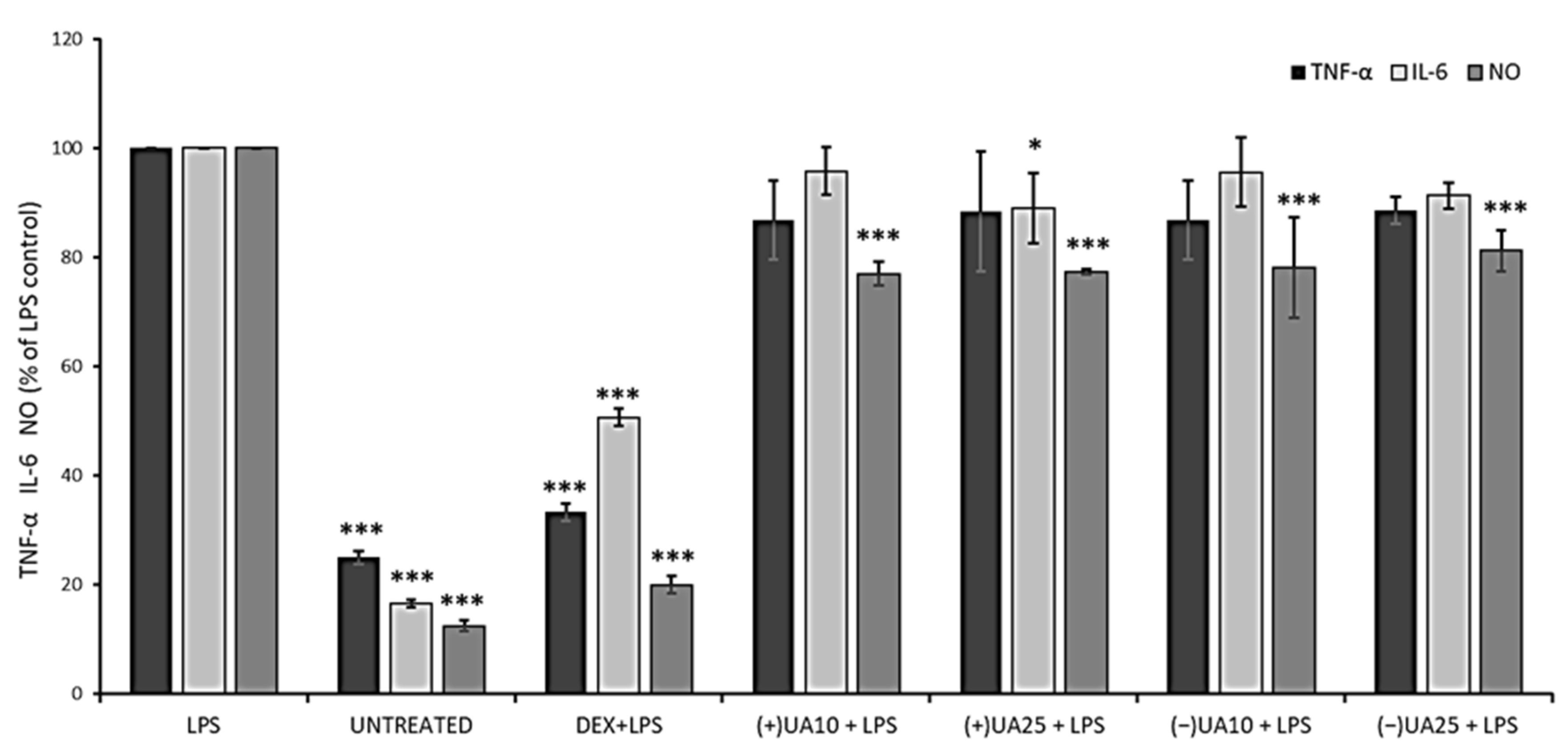
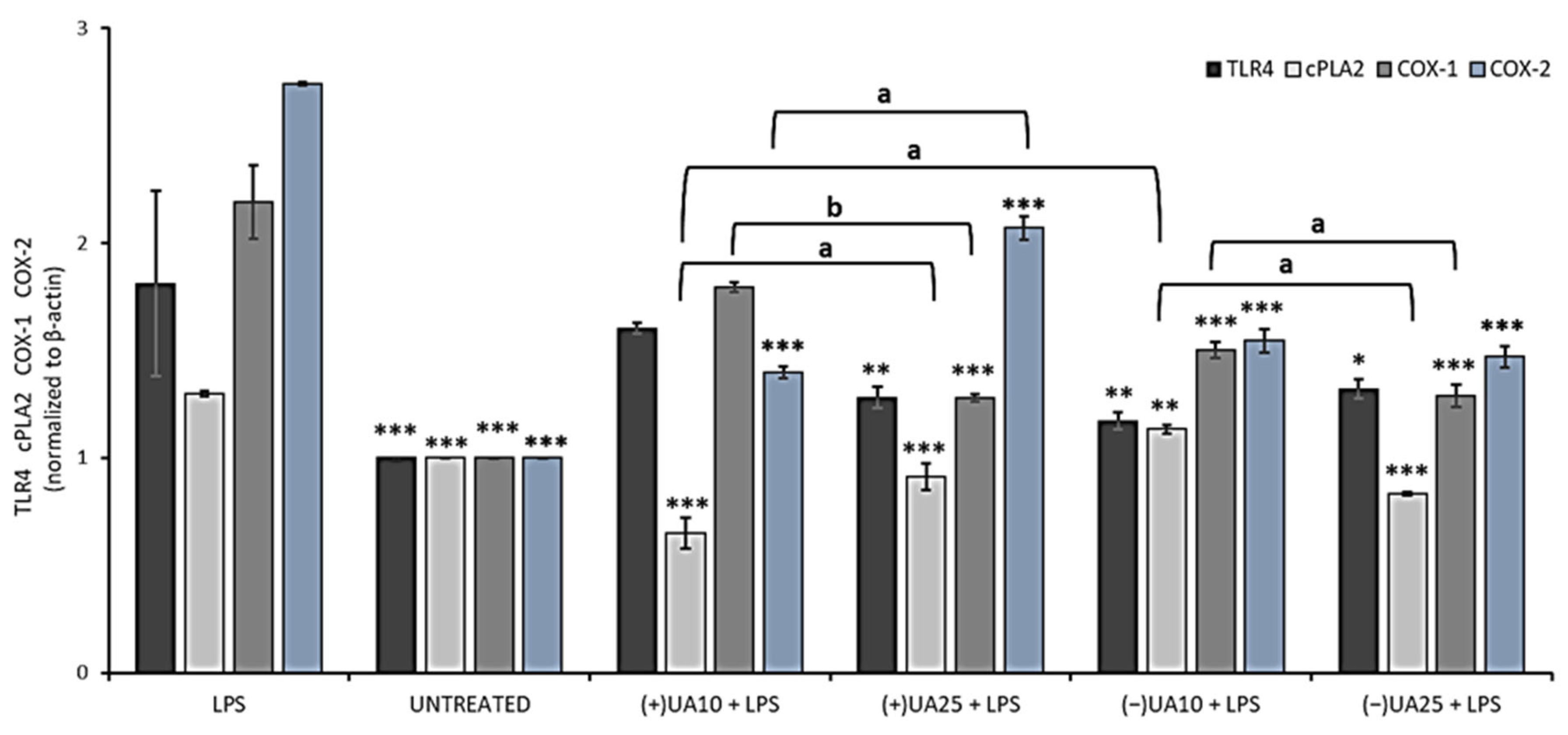
2.6. Both Usnic Acid Enantiomers Decreased Pro-Inflammatory Mediators Release
2.7. (+)-Usnic Acid Strongly Inhibited Hyaluronidase Activity
3. Materials and Methods
3.1. Reagents and Instruments
3.2. Cell Culture Conditions
3.3. Viability Assay
3.4. Isobolographic Analysis
3.5. Proliferation Assay
3.6. Transwell Migration Assay
3.7. Anti-Tyrosinase Assay
3.8. Determination of NO, IL-6 and TNF-Alpha Release in RAW 264.7 Model
3.9. Western Blot Analysis
3.10. Anti-Hyaluronidase Assay
3.11. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Garbe, C.; Eigentler, T.K.; Keilholz, U.; Hauschild, A.; Kirkwood, J.M. Systematic review of medical treatment in melanoma: Current status and future prospects. Oncologist 2011, 16, 5–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Drąg-Zalesińska, M.; Drąg, M.; Poręba, M.; Borska, S.; Kulbacka, J.; Saczko, J. Anticancer properties of ester derivatives of betulin in human metastatic melanoma cells (Me-45). Cancer Cell Int. 2017, 17, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [Green Version]
- Brożyna, A.A.; Van Middlesworth, L.; Slominski, A.T. Inhibition of melanogenesis as a radiation sensitizer for melanoma therapy. Int. J. Cancer 2008, 123, 1448–1456. [Google Scholar] [CrossRef]
- Melnikova, V.O.; Bar-Eli, M. Inflammation and melanoma metastasis. Pigment. Cell Melanoma Res. 2009, 22, 257–267. [Google Scholar] [CrossRef] [PubMed]
- Ingolfsdottir, K. Usnic acid. Phytochemistry 2002, 61, 729–736. [Google Scholar] [CrossRef]
- Araújo, A.A.S.; De Melo, M.G.D.; Rabelo, T.K.; Nunes, P.S.; Santos, S.L.; Serafini, M.R.; Quintans-Junior, L.J.; Gelain, D.P. Review of the biological properties and toxicity of usnic acid. Nat. Prod. Res. 2015, 29, 2167–2180. [Google Scholar] [CrossRef]
- Galanty, A.; Paśko, P.; Podolak, I. Enantioselective activity of usnic acid: A comprehensive review and future perspectives. Phytochem. Rev. 2019, 18, 527–548. [Google Scholar] [CrossRef] [Green Version]
- Galanty, A.; Koczurkiewicz, P.; Wnuk, D.; Paw, M.; Karnas, E.; Podolak, I.; Węgrzyn, M.; Borusiewicz, M.; Madeja, Z.; Czyż, J.; et al. Usnic acid and atranorin exert selective cytostatic and anti-invasive effects on human prostate and melanoma cancer cells. Toxicol. Vitro 2017, 40, 161–169. [Google Scholar] [CrossRef]
- Brandão, L.F.G.; Alcantara, G.B.; de Fátima Cepa Matos, M.; Bogo, D.; dos Santos Freitas, D.; Oyama, N.M.; Honda, N.K. Cytotoxic evaluation of phenolic compounds from lichens against melanoma cells. Chem. Pharm. Bull. 2012, 61, 176–183. [Google Scholar] [CrossRef] [Green Version]
- Ranković, B.; Kosanić, M.; Stanojković, T.; Vasiljević, P.; Manojlović, N. Biological activities of Toninia candida and Usnea barbata together with their norstictic acid and usnic acid constituents. Int. J. Mol Sci. 2012, 13, 14707–14722. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Draut, H.; Rehm, T.; Begemann, G.; Schobert, R. Antiangiogenic and toxic effects of genistein, usnic acid, and their copper complexes in zebrafish embryos at different developmental stages. Chem. Biodivers. 2017, 14, e1600302. [Google Scholar] [CrossRef] [PubMed]
- Piska, K.; Galanty, A.; Koczurkiewicz, P.; Żmudzki, P.; Potaczek, J.; Podolak, I.; Pękala, E. Usnic acid reactive metabolites formation in human, rat, and mice microsomes. Implication for hepatotoxicity. Food Chem. Toxicol. 2018, 120, 112–118. [Google Scholar] [CrossRef]
- Galanty, A.; Popiół, J.; Paczkowska-Walendowska, M.; Studzińska-Sroka, E.; Paśko, P.; Cielecka-Piontek, J.; Pękala, E.; Podolak, I. (+)-Usnic Acid as a Promising Candidate for a Safe and Stable Topical Photoprotective Agent. Molecules 2021, 26, 5224. [Google Scholar] [CrossRef]
- Kılıç, N.; Değerli, E.; Torun, V.; Altaytaş, F.; Cansaran-Duman, D. Investigation of synergistic effect of tamoxifen and usnic acid on breast cancer cell line. JSM Biol. 2016, 1, 1–4. [Google Scholar]
- Güney Eskiler, G.; Eryilmaz, I.E.; Yurdacan, B.; Egeli, U.; Cecener, G.; Tunca, B. Synergistic effects of hormone therapy drugs and usnic acid on hormone receptor-positive breast and prostate cancer cells. J. Biochem. Mol. Toxicol. 2019, 33, e22338. [Google Scholar] [CrossRef]
- Yurdacan, B.; Egeli, U.; Guney Eskiler, G.; Eryilmaz, I.E.; Cecener, G.; Tunca, B. Investigation of new treatment option for hepatocellular carcinoma: A combination of sorafenib with usnic acid. J. Pharm. Pharmacol. 2019, 71, 1119–1132. [Google Scholar] [CrossRef]
- Qi, W.; Lu, C.; Huang, H.; Zhang, W.; Song, S.; Liu, B. (+)-Usnic acid induces ROS-dependent apoptosis via inhibition of mitochondria respiratory chain complexes and Nrf2 expression in lung squamous cell carcinoma. Int. J. Mol. Sci. 2020, 21, 876. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Einarsdóttir, E.; Groeneweg, J.; Björnsdóttir, G.G.; Harðardottir, G.; Omarsdóttir, S.; Ingólfsdóttir, K.; Ögmundsdóttir, H.M. Cellular mechanisms of the anticancer effects of the lichen compound usnic acid. Planta Med. 2010, 76, 969–974. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.T.; Yoon, S.; Yang, Y.; Lee, H.B.; Oh, S.; Jeong, M.H.; Kim, J.J.; Yee, S.T.; Crisan, F.; Moon, C.; et al. Lichen secondary metabolites in Flavocetraria cucullata exhibit anti-cancer effects on human cancer cells through the induction of apoptosis and suppression of tumorigenic potentials. PLoS ONE 2014, 9, e111575. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, Y.; Nguyen, T.T.; Jeong, M.H.; Crişan, F.; Yu, Y.H.; Ha, H.H.; Choi, K.H.; Jeong, H.G.; Jeong, T.C.; Lee, K.Y.; et al. Inhibitory activity of (+)-usnic acid against non-small cell lung cancer cell motility. PLoS ONE 2016, 11, e0146575. [Google Scholar] [CrossRef]
- Wu, W.; Gou, H.; Dong, J.; Yang, X.; Zhao, Y.; Peng, H.; Chen, D.; Geng, R.; Chen, L.; Liu, J. Usnic acid inhibits proliferation and migration through ATM mediated DNA damage response in RKO colorectal cancer cell. Curr. Pharm. Biotechnol. 2021, 22, 1129–1138. [Google Scholar] [CrossRef]
- Kwong, S.P.; Wang, H.; Shi, L.; Huang, Z.; Lu, B.; Cheng, X.; Chou, G.; Ji, L.; Wang, C. Identification of photodegraded derivatives of usnic acid with improved toxicity profile and UVA/UVB protection in normal human L02 hepatocytes and epidermal melanocytes. J. Photochem. Photobiol. B 2020, 205, 111814. [Google Scholar] [CrossRef] [PubMed]
- Dung, P.D.; Huy, D.T.; Van Kieu, N. Synthesis and evaluation of α-glucosidase and tyrosinase inhibitory activities of ester derivatives of usnic acid. Sci. Technol. Devel. J. 2020, 23, 590–597. [Google Scholar] [CrossRef]
- Mantovani, A.; Allavena, P.; Sica, A.; Balkwill, F. Cancer-related inflammation. Nature 2008, 454, 436–444. [Google Scholar] [CrossRef]
- Germano, G.; Allavena, P.; Mantovani, A. Cytokines as a key component of cancer-related inflammation. Cytokine 2008, 43, 374–379. [Google Scholar] [CrossRef]
- Jin, J.Q.; Li, C.Q.; He, L.C. Down-regulatory effect of usnic acid on nuclear factor-κB-dependent tumor necrosis factor-α and inducible nitric oxide synthase expression in lipopolysaccharide-stimulated macrophages RAW 264.7. Phytother. Res. 2008, 22, 1605–1609. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Zheng, G.; Tao, J.; Ruan, J. Anti-inflammatory effects and mechanisms of usnic acid. J. Wuhan Univ. Technol. Mater. Sci. Ed. 2011, 26, 955–959. [Google Scholar] [CrossRef]
- Chen, C.Y.; Kao, C.L.; Liu, C.M. The cancer prevention, anti-inflammatory and anti-oxidation of bioactive phytochemicals targeting the TLR4 signaling pathway. Int. J. Mol. Sci. 2018, 19, 2729. [Google Scholar] [CrossRef] [Green Version]
- Bruno, M.; Trucchi, B.; Burlando, B.; Ranzato, E.; Martinotti, S.; Akkol, E.K.; Süntar, I.; Keleş, H.; Verotta, L. (+)-Usnic acid enamines with remarkable cicatrizing properties. Bioorg. Med. Chem. 2013, 21, 1834–1843. [Google Scholar] [CrossRef]
- Grabowska, K.; Galanty, A.; Koczurkiewicz-Adamczyk, P.; Wróbel-Biedrawa, D.; Żmudzki, P.; Załuski, D.; Wójcik-Pszczoła, K.; Paśko, P.; Pękala, E.; Podolak, I. Multidirectional anti-melanoma effect of galactolipids (MGDG-1 and DGDG-1) from Impatiens parviflora DC. and their synergy with doxorubicin. Toxicol. Vitro 2021, 76, 105231. [Google Scholar] [CrossRef]
- Chou, T.C. Theoretical basis, experimental design, and computerized simulation of synergism and antagonism in drug combination studies. Pharmacol. Rev. 2006, 58, 621–681. [Google Scholar] [CrossRef]
- Koczurkiewicz-Adamczyk, P.; Piska, K.; Gunia-Krzyżak, A.; Bucki, A.; Jamrozik, M.; Lorenc, E.; Ryszawy, D.; Wójcik-Pszczoła, K.; Michalik, M.; Marona, H.; et al. Cinnamic acid derivatives as chemosensitising agents against DOX-treated lung cancer cells–Involvement of carbonyl reductase 1. Eur. J. Pharm. Sci. 2020, 154, 105511. [Google Scholar] [CrossRef]
- Wróbel-Biedrawa, D.; Grabowska, K.; Galanty, A.; Sobolewska, D.; Żmudzki, P.; Podolak, I. Anti-melanoma potential of two benzoquinone homologues embelin and rapanone-a comparative in vitro study. Toxicol. Vitro 2020, 65, 104826. [Google Scholar] [CrossRef] [PubMed]
- Paśko, P.; Galanty, A.; Zagrodzki, P.; Luksirikul, P.; Barasch, D.; Nemirovski, A.; Gorinstein, S. Dragon fruits as a reservoir of natural polyphenolics with chemopreventive properties. Molecules 2021, 26, 2158. [Google Scholar] [CrossRef] [PubMed]
- Gdula-Argasińska, J.; Bystrowska, B. Docosahexaenoic acid attenuates in endocannabinoid synthesis in RAW 264.7 macrophages activated with benzo(a)pyrene and lipopolysaccharide. Toxicol. Lett. 2016, 258, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Grabowska, K.; Wróbel, D.; Żmudzki, P.; Podolak, I. Anti-inflammatory activity of saponins from roots of Impatiens parviflora DC. Nat. Prod. Res. 2020, 34, 1581–1585. [Google Scholar] [CrossRef]


| Treatment | Incubation | IC50 (µg/mL) | ||
|---|---|---|---|---|
| HTB140 | A375 | WM793 | ||
| (+)-UA | 24 h | 16.99 | 15.10 | 45.68 |
| 48 h | 14.72 | 11.84 | 30.05 | |
| (−)-UA | 24 h | 26.24 | 29.90 | 87.95 |
| 48 h | 20.62 | 22.14 | 52.09 | |
| DOX | 24 h | 3.77 | 1.59 | >100 * |
| 48 h | 2.01 | 0.55 | >100 * | |
| (+)UA + DOX | 24 h | 12.74 | 11.80 | 73.45 |
| 48 h | 10.92 | 6.08 | 41.61 | |
| (−)UA + DOX | 24 h | 12.56 | 14.99 | 33.19 |
| 48 h | 9.96 | 8.08 | 26.15 | |
| Treatment | CI after 24 h | CI after 48 h | |||||
|---|---|---|---|---|---|---|---|
| HTB140 | A375 | WM793 | HTB140 | A375 | WM793 | ||
| MIX 1 IC50 | (+)UA + DOX | 3.73 ⬤ | 1.30 ⬤ | 0.75 ⬤ | 2.67 ⬤ | 1.11 ⬤ | 0.36 ⬤ |
| (−)UA + DOX | 0.54 ⬤ | 0.70 ⬤ | 0.003 ⬤ | 0.24 ⬤ | 0.52 ⬤ | 0.05 ⬤ | |
| MIX 2 1/2 IC50 | (+)UA + DOX | 2.54 ⬤ | 0.84 ⬤ | 0.76 ⬤ | 2.04 ⬤ | 0.75 ⬤ | 0.60 ⬤ |
| (−)UA + DOX | 0.88 ⬤ | 0.56 ⬤ | 0.07 ⬤ | 1.01 ⬤ | 0.69 ⬤ | 0.21 ⬤ | |
| MIX 3 1/4 IC50 | (+)UA + DOX | 1.78 ⬤ | 0.52 ⬤ | 1.06 ⬤ | 1.44 ⬤ | 0.57 ⬤ | 0.82 ⬤ |
| (−)UA + DOX | 0.87 ⬤ | 0.46 ⬤ | 0.15 ⬤ | 1.04 ⬤ | 0.64 ⬤ | 0.40 ⬤ | |
| MIX 4 1/6 IC50 | (+)UA + DOX | 1.42 ⬤ | 0.42 ⬤ | 1.37 ⬤ | 1.09 ⬤ | 0.45 ⬤ | 0.87 ⬤ |
| (−)UA + DOX | 0.88 ⬤ | 0.45 ⬤ | 0.24 ⬤ | 1.25 ⬤ | 0.54 ⬤ | 0.52 ⬤ | |
| MIX 5 1/8 IC50 | (+)UA + DOX | 1.39 ⬤ | 0.44 ⬤ | 1.84 ⬤ | 1.00 ⬤ | 0.43 ⬤ | 1.37 ⬤ |
| (−)UA + DOX | 1.06 ⬤ | 0.57 ⬤ | 0.47 ⬤ | 1.28 ⬤ | 0.52 ⬤ | 0.83 ⬤ | |
| Concentration (µg/mL) | % Hyaluronidase Inhibition (Mean ± SD) | ||
|---|---|---|---|
| (+)-Usnic Acid | (−)-Usnic Acid | Quercetin | |
| 50 | 0.00 ± 0.00 | 0.00 ± 1.82 | 1.23 ± 0.99 |
| 100 | 0.62 ± 1.07 a | 3.70 ± 0.42 | 4.39 ± 0.88 |
| 250 | 13.69 ± 4.49 * | 23.33 ± 3.33 | 18.10 ± 1.01 |
| 500 | 37.81 ± 7.51 | 40.80 ± 3.59 | 38.84 ± 1.33 |
| 750 | 44.26 ± 2.84 a | 42.29 ± 4.56 a | 87.27 ± 1.16 |
| 1000 | 88.52 ± 1.64 *** | 72.33 ± 9.31 a | 91.11 ± 1.50 |
| IC50 | 644.45 | 676.27 | 517.33 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Galanty, A.; Zagrodzki, P.; Gdula-Argasińska, J.; Grabowska, K.; Koczurkiewicz-Adamczyk, P.; Wróbel-Biedrawa, D.; Podolak, I.; Pękala, E.; Paśko, P. A Comparative Survey of Anti-Melanoma and Anti-Inflammatory Potential of Usnic Acid Enantiomers—A Comprehensive In Vitro Approach. Pharmaceuticals 2021, 14, 945. https://doi.org/10.3390/ph14090945
Galanty A, Zagrodzki P, Gdula-Argasińska J, Grabowska K, Koczurkiewicz-Adamczyk P, Wróbel-Biedrawa D, Podolak I, Pękala E, Paśko P. A Comparative Survey of Anti-Melanoma and Anti-Inflammatory Potential of Usnic Acid Enantiomers—A Comprehensive In Vitro Approach. Pharmaceuticals. 2021; 14(9):945. https://doi.org/10.3390/ph14090945
Chicago/Turabian StyleGalanty, Agnieszka, Paweł Zagrodzki, Joanna Gdula-Argasińska, Karolina Grabowska, Paulina Koczurkiewicz-Adamczyk, Dagmara Wróbel-Biedrawa, Irma Podolak, Elżbieta Pękala, and Paweł Paśko. 2021. "A Comparative Survey of Anti-Melanoma and Anti-Inflammatory Potential of Usnic Acid Enantiomers—A Comprehensive In Vitro Approach" Pharmaceuticals 14, no. 9: 945. https://doi.org/10.3390/ph14090945
APA StyleGalanty, A., Zagrodzki, P., Gdula-Argasińska, J., Grabowska, K., Koczurkiewicz-Adamczyk, P., Wróbel-Biedrawa, D., Podolak, I., Pękala, E., & Paśko, P. (2021). A Comparative Survey of Anti-Melanoma and Anti-Inflammatory Potential of Usnic Acid Enantiomers—A Comprehensive In Vitro Approach. Pharmaceuticals, 14(9), 945. https://doi.org/10.3390/ph14090945







