From Bush Medicine to Modern Phytopharmaceutical: A Bibliographic Review of Devil’s Claw (Harpagophytum spp.)
Abstract
1. Introduction
2. Materials and Methods
3. Nomenclature
3.1. Taxonomy
- H. procumbens (Burch.) DC. ex Meisn. ssp. procumbens—(1).
- H. procumbens (Burch.) DC. ex Meisn. ssp. transvaalense Ihlenf. & H. Hartm.—(2).
- H. zeyheri Decne. ssp. zeyheri—(3).
- H. zeyheri Decne. ssp. schijffii Ihlenf. & H. Hartm.—(4).
- H. zeyheri Decne. ssp. sublobatum (Engler) Ihlenf. & H. Hartm.—(5).
- H. burchellii Decne. = H. procumbens ssp. procumbens DC. ex Meisn.
- H. zeyheri f. sublobatum Engl. = H. zeyheri ssp. sublobatum (Engl.) Ihlenf. & H. Hartm.
- H. procumbens var. sublobatum (Engl.) Stapf = H. zeyheri ssp. sublobatum (Engl.) Ihlenf. & H. Hartm.
- H. peglerae Stapf = H. zeyheri ssp. zeyheri Decne.
3.2. Vernacular Names
4. Distribution
5. Ethnobotany
6. Economy
6.1. History of Commercialization
6.2. Trade
7. Representation in Pharmacopeias and Authoritative Compendia
8. Biochemistry
| Topic | Year | Reference |
|---|---|---|
| Isolation and characterization of harpagoside | 1960 | [117] |
| Stachyose, raffinose, and a further glucoside in the aqueous phase | 1961 | [118] |
| Characterization of harpagoside | 1961 | [119] |
| Isolation and characterization of harpagoside and harpagide | 1962 | [120] |
| Characterization of harpagoside | 1962 | [121] |
| Characterization of harpagide | 1963 | [122] |
| Isolation of stachyose and a further glucoside | 1963 | [123] |
| Characterization of harpagoside | 1964 | [124] |
| Isolation of procumbide | 1964 | [125] |
| Structural characterization of harpagoside | 1966 | [126] |
| Characterization of procumbide and further constituents | 1967 | [127] |
| Characterization of procumbide | 1968 | [128] |
| Characterization of a chinone and other constituents | 1970 | [185] |
| Characterization of procumbide | 1971 | [186] |
| Further constituents | 1974 | [187] |
| Elucidation of triterpene esters | 1975 | [188] |
| Overview of known mono-, di-, and sesquiterpenoids with pharmacological activity | 1977 | [189] |
| Elucidation of a resin, an essential oil, and a mucilaginous fraction | 1978 | [190] |
| Structural characterization of procumbide | 1979 | [191] |
| Glucose, galactose, fructose, myo-inositol, sucrose, raffinose, and stachyose identified | 1979 | [192] |
| Preparation and structure of harpagogenine | 1981 | [193] |
| Carbohydrates and harpagoside in tissue cultures and roots of devil’s claw | 1982 | [194] |
| New iridoids: 8-O-(p-coumaryl)-harpagide and procumboside | 1983 | [195] |
| Novel iridoid and phenolic compounds | 1987 | [196] |
| Three pyridine monoterpene alkaloids from harpagoside and commercial extract | 1999 | [197] |
| Review of iridoids | 2000 | [198] |
| Review of composition (both species) | 2002 | [199] |
| Two diterpenes, (+)-8,11,13-totaratriene-12,13-diol and ferruginol | 2002 | [200] |
| New iridoid- and phenylethanoid glycosides | 2003 | [201] |
| Acetylated phenolic glycosides | 2003 | [202] |
| Pharmacological characterization of harpagoside | 2004 | [203] |
| Chinane-type tricyclic diterpenes and other minor compounds | 2006 | [204,205] |
| Review of iridoids and other compounds | 2006 | [206,207] |
| Review of chemical constituents | 2007 | [208] |
| Elucidation and characterization of compounds with specific pharmacologic profiles | 2008 | [209,210] |
| New triterpenoid glycoside, harproside, and new iridoid glycoside, pagide | 2010 | [211] |
| Kynurenic acid content | 2013 | [212] |
| New iridoid diglucoside | 2016 | [213] |
9. Analytical Methods and Quality Control
10. Processing, Products, Applications
- Liquid extract (1:1; 30% v/v ethanol)
- Soft extract (2.5–4.0:1; 70% v/v ethanol)
- Dry extract (1.5–2.5:1; water)
- Dry extract (5–10:1; water)
- Dry extract (2.6–4:1; 30% v/v ethanol)
- Dry extract (1.5–2.1:1; 40% v/v ethanol)
- Dry extract (3–5:1; 60% v/v ethanol)
- Dry extract (3–6:1; 80% v/v ethanol)
- Dry extract (6–12:1; 90% v/v ethanol)
- Tincture (1:5), extraction solvent ethanol 25% (v/v)
11. Pre-Clinical Research
11.1. Pharmacology
11.2. Pharmacokinetics
11.3. Toxicology
12. Clinical Research
12.1. Efficacy
| Indication | Trial Type, Size | Results | Year | Reference |
|---|---|---|---|---|
| Chemosis | CR 1 | Initial treatment with multiple preparations that did not lead to improvement, then with 300 mg Harpagophytum extract (not specified) 3 times daily, orally, for 6 months, leading to drastic improvement. | 1983 | Belaiche [489] |
| Familial Mediterranean fever | CR 17 | Harpagophytum extracts characterized as aqueous (DER 1:2.4, 2.5% harpagoside)—this characterization may also apply to previous trials by Belaiche and Dahout (see above)—6–9 g single dose, duration not provided; significantly decreased recurrence in 80% of patients. | 1983 | Belaiche [490] |
| Cancer | CR 2 | Tumor regression after taking Harpagophytum extract (500 mg daily) and/or Essiac respectively, without cytotoxic therapy. | 2009 | Wilson [491] |
| DJD | O ~120 | Harpagophytum D4–D6, IA, and D1 orally; 1–6 months; substantial improvement of symptoms in most cases. | 1971 | Beham [492] |
| CP | O 60 | Harpagophytum D2, IA, plus tea (2–3 tsp per 1 L water) or 3 × 2 tablets orally, duration not provided; dose-dependent response; 60% substantial improvement of symptoms, 20% improvement, 20% no change. | 1972 | Schmidt [43] |
| CP, DJD | O 146 | Harpagophytum D2, IA, duration not provided; improvement in 134 patients. | 1972 | Zimmermann, cited in [130] |
| DJD | O 25 | Harpagophytum D2–D3, IA, and SC, 1–2 mL, pain-free after 6 injections, or tea (1 tsp per 300 mL) daily for 3–6 weeks. | 1972 | Brantner [493] |
| DJD | O 70 | Harpagophytum D2, IA, some + tea, some + indometacin, duration not provided; improvement in 90% of patients. | 1976 | Wilhelmer, cited in [44] |
| CP, DJD | O 21+ | Harpagophytum D1–D3, IA, SC, and i.v., tea, orally, duration not provided; significant improvement in 30% of patients. | 1977 | Zimmermann [494] |
| DJD | O 84 | 250 or 500 mg Harpagophytum extract (not specified) 3 times daily orally for 2–6 months, improvement in 72% of patients. | 1979 | Dahout, cited in [495] |
| CP, DJD | O 600 | Harpagosan tea (2 tea bags in 500 mL water daily) plus D2 SC for up to 6 months. Symptoms disappeared in 200 patients; 400 patients improved after having received additional conventional medication for the first 3–4 weeks. | 1983 | Warning cited in Schmidt [44] |
| Rheumatoid arthritis | O 1 | Improvement after treatment with low-potency Harpagophytum i.v. and orally, duration not provided. | 1987 | Stübler [496,497] |
| DJD | O 553 | Patients treated with 2–6 capsules of 400 mg Harpagophytum extract (1.5–2.5:1) for 8 to 180 days. Outcomes confirmed RCT results in terms of efficacy and safety. | 2000 | Müller et al. [498] |
| DJD | O 255 | Post-marketing surveillance study of biopsychosocial determinants and treatment response. Patients treated with Harpagophytum extract (60 mg harpagoside/day) for 2 months. Outcome parameters were significantly worse in non-responders. | 2009 | Thanner et al. [499] |
| CP, DJD, dyspepsia, hypercholesterolemia, detoxication | O, CR 700+ | Harpagophytum tea, up to 12 weeks, D2, SC, 20 injections, further improvement with additional D2 i.v. and tea. | 1978 | Schmidt [130] |
| Diabetes mellitus with lipometabolic disorder | OT 10 | 4 patients 3 weeks, 6 patients 4 and 3 weeks, over a total of 6 months; Harpagophytum tea, amount not specified; cholesterol, lipid, and blood sugar levels normalized. | 1974 | Hoppe [500] |
| Hypercholesterolemia and hyperuricemia | OT 100 | Harpagophytum tea, 2 tea bags per ½ L water, 3× daily before meals 1/3 of the tea; 20–21 days; lowered cholesterol levels in 80%, normal levels in 45%, 66% improvement in hyperuricemia. | 1978 | Grünewald [405] |
| DJD | OT 13 | Harpagophytum extract (<30 mg harpagoside/day), for 6 weeks, followed up for another six weeks; no overall statistically significant improvements in the conditions. | 1981 | Grahame and Robinson [501] |
| DJD | OT 630 | 42% to 85% of the patients (depending on grouping) showed improvements after 6 months with Harpagophytum extract (>90 mg harpagoside/day). | 1982 | Belaiche [502] |
| DJD | OT 38 | Comparison of Formica rufa D6 with Harpagophytum D4, for 3 months; improvement in pain severity and mobility with both, Formica rufa slightly superior. | 1991 | Kröner [503] |
| Effect on eicosanoid biosynthesis | OT 34 (25/8) healthy volunteers | Harpagophytum, 4 capsules (500 mg powder, 3% of total glucoiridoids) daily for 21 days. No effect vs. control. | 1992 | Moussard et al. [504] |
| MSD | OT 102 (51,51) | Patients treated with Harpagophytum extract (30 mg harpagoside/day) or conventional therapy (mainly oral NSAIDs). Number of pain-free patients and changes in Arhus scores after 4 and 6 weeks of treatment was comparable between the groups. | 1997 | Chrubasik et al. [505] |
| DJD | OT 43 | Harpagophytum powder 3 g daily for 60 days. Reduction of pain intensity in 89%, increased mobility in 83%. | 1997 | Pinget and Lecomte [506] |
| MSD | OT 2053 | Patients treated with Harpagophytum extract (30 mg harpagoside/day) for 6 weeks. Symptoms improved over time. | 1999 | Schwarz et al. [507] |
| DJD | OT 45 | Patients treated with Harpagophytum extract (30 mg harpagoside/day) for two weeks plus NSAID treatment, and devil’s claw alone, for four weeks. No worsening of scores was observed during treatment with devil’s claw alone. | 2000 | Szczepanski et al. [508] |
| MSD | OT 1026 | Patients treated with Harpagophytum extract (30 mg harpagoside/day) for 6 weeks. Symptoms improved. | 2000 | Usbeck [509,510] |
| MSD | OT 130 | Patients treated with Harpagophytum extract (~30 mg harpagoside/day) for 8 weeks. Arhus back pain index decreased significantly during treatment. Other measures also improved significantly. | 2001 | Laudahn et al. [511,512,513] |
| DJD | OT 583 | Patients treated with Harpagophytum extract (~30 mg harpagoside/day) for 8 weeks. Symptoms improved and the dose of co-medication (NSAIDs) could be reduced. | 2001 | Schendel [514] |
| DJD | OT 675 | Patients treated with Harpagophytum extract (~30 mg harpagoside/day) for 8 weeks. Efficacy rated good or very good in 82% of cases. The symptom scores decreased, and co-medication was successfully reduced or even discontinued. | 2001 | Ribbat and Schakau [515] |
| MSD | OT 250 | Patients treated with Harpagophytum extract (60 mg harpagoside/day) for 8 weeks. Both generic and disease-specific outcome measures improved. | 2002 | Chrubasik et al. [516] |
| DJD | OT 614 | Patients treated with Harpagophytum extract (480 mg twice daily) for 8 weeks. Symptoms improved in the majority of patients; treatment was well-tolerated. | 2003 | Kloker and Flammersfeld [517,518] |
| DJD | OT 75 | Patients treated with Harpagophytum extract (50 mg harpagoside/day) for 12 weeks. WOMAC index and 10 cm VAS pain scale improved notably. | 2003 | Wegener and Lüpke [519,520] |
| MSD | OT 99 | Patients treated with Harpagophytum extract (~30 mg harpagoside/day) for 6 weeks. Symptoms improved. | 2005 | Rütten and Kuhn [521] |
| MSD | OT 102 (29/22/51) | Patients treated with Harpagophytum extract (~30 mg harpagoside/day) and/or conventional therapy for 6 weeks. Efficacy was found in all groups, advantages for devil’s claw were not statistically significant. | 2005 | Schmidt et al. [522,523] |
| DJD | OT 65 | Patients treated with combination of Harpagophytum procumbens, Zingiber officinale, and Urtica sp. (ratio not disclosed) for 8 weeks. Improvements in all efficacy parameters were observed. | 2005 | Sohail et al. [524] |
| Endometriosis | OT 6, 12 | Patients treated with Harpagophytum extract (1600 mg daily) for 12 weeks. Reduction of symptoms in 4 (6) patients after 4 weeks, in all patients after 12 weeks. | 2005, 2006 | Arndt et al. [525,526] |
| DJD | OT 259 | Patients treated with Harpagophytum extract (1.5–3:1, 960 mg daily) and NSAIDs for 8 weeks. At the end of the treatment, 44.8% could decrease NSAID dosage. All parameters improved significantly. | 2006 | Suter et al. [527,528] |
| MSD | OT 114 | Patients treated with Harpagophytum extract (60 mg harpagoside/day) for up to 54 weeks. Most outcome scores improved significantly over time. | 2007 | Chrubasik et al. [529] |
| DJD | OT 42 | Patients treated with combination of Harpagophytum (1800 mg), Curcuma longa (1200 mg), and bromelain (900 mg) daily, plus conventional therapies for 2 weeks. Clinically relevant improvement of joint pain scores in all patients. | 2014 | Conrozier et al. [530] |
| DJD | OT 20 | Patients treated with combination of 500 mg glucosamine sulfate, 400 mg chondroitin sulfate, 10 mg collagen type II, and 40 mg Harpagophytum per day for 12 months. Femoral hyaline cartilage thickness significantly improved and radiographic progression of knee osteoarthritis delayed. | 2019 | Vreju et al. [531] |
| MSD | OT 39/40/16 | Otherwise healthy subjects with mild/moderate neck/shoulder pain related to sport; cream containing a combination of ingredients, including H. procumbens root extract + standard treatment, standard treatment, diclofenac patch + standard treatment respectively, for 2 weeks; significant improvement in pain, stiffness, mobility, and working capacity, compared to non-cream groups. | 2021 | Hu et al. [532] |
| DJD | RCT 39 | 400 mg Harpagophytum extract (not specified), and 25 mg diclofenac, or placebo 3× daily for 6 months. Overall confirmation of anti-inflammatory effects without side effects. | ~1980 | Chaouat, cited in [66,67] |
| DJD | RCT 50 (25/25) | Harpagophytum extract (<30 mg harpagoside/day) and phenybutazone (300 mg per day for the first four days, then 200 mg) respectively, for 28 days. Devil’s claw found equally effective to phenybutazone. | 1980 | Schrüffler [533] |
| DJD | RCT 50 (25/25) | Patients treated with Harpagophytum extract (<20 mg harpagoside/day) or placebo for three weeks showed a significant decrease in pain severity vs. placebo. | 1984 | Guyader [534] |
| DJD | RCT 100 (50/50) | Patients treated with Harpagophytum extract (60 mg harpagoside/day) or placebo for 30 days. Only 6 patients in the verum group still experienced moderate pain vs. 32 in the placebo group. | 1990 | Pinget and Lecomte [535] |
| DJD | RCT 89 (45/44) | Patients treated with Harpagophytum extract (60 mg harpagoside/day) or placebo for two months. Significant decrease in severity of pain and significant increase in spinal and cofexomoral mobility vs. placebo. | 1992 | Lecomte and Costa [536] |
| MSD | RCT 118 (59,59) | Patients treated with Harpagophytum extract (50 mg harpagoside/day) or placebo for 4 weeks. Treatment group used less analgesics, had greater improvement in median Arhus scores (20% vs. 8%; p < 0.059), and had more patients pain-free at the end (9/51 vs. 1/54; p = 0.008). | 1996 | Chrubasik et al. [537,538,539] |
| MSD | RCT 109 (54/55) | Patients treated with Harpagophytum extract (50 mg harpagoside/day) or placebo for 4 weeks. Rescue medication: tramadol. Significant improvement in Arhus index and pain index, and co-medication reduced vs. placebo. | 1997 | Chrubasik et al. [540] |
| DJD | RCT 100 (50/50) | Patients treated with Harpagophytum extract (30 mg harpagoside/day) or placebo for 30 days. Favorable effects were evident after 10 days vs. placebo. | 1997 | Schmelz and Hämmerle [541] |
| MSD | RCT 197 (65/66/66) | Patients treated with Harpagophytum extract (50 mg (1), 100 mg (2) harpagoside/day) or placebo (3) for four weeks. 6, 10, and 3 patients were pain-free in groups 1, 2 and 3, respectively. Arhus index score decreased but not statistically significant. Dose-related effect not confirmed. | 1999 | Chrubasik et al. [542] |
| DJD | RCT 122 (62/60) | Patients treated with Harpagophytum extract (57 mg harpagoside/day) or diacerhein at 100 mg daily for four months. Results showed significant improvement in both groups at a similar rate. | 2000 | Chantre et al. [543,544] |
| MSD | RCT 63 (31/32) | Patients treated with Harpagophytum extract (~30 mg harpagoside/day) or placebo for 4 weeks. Significant efficacy for visual analogue scale, pressure algometer test, muscle stiffness test, and muscular ischemia test. No differences to placebo in anti-nociceptive muscular reflexes or electromyogram activity. | 2000 | Göbel et al. [512,513,545,546] |
| DJD | RCT 46 (24/22) | Patients treated with ibuprofen (800 mg) and Harpagophytum extract (~30 mg harpagoside/day) or placebo for 20 weeks. WOMAC scores decreased similarly, but during an ibuprofen-free period, symptoms worsened less than 20% for 71% of devil’s claw patients vs. 41% of placebo patients. | 2001 | Frerick et al. [547] |
| DJD | RCT 78 (39/39) | Patients treated with Harpagophytum extract (~30 mg harpagoside/day) or placebo for 20 weeks. Co-medication ibuprofen. Symptoms improved similarly for both groups. | 2002 | Biller [548] |
| MSD | RCT 88 (44/44) | Patients treated with Harpagophytum extract (60 mg harpagoside/day) for 6 weeks or 12.5 mg/day of rofecoxib. Outcome scores improved similarly for both groups. Follow-up confirmed the results of the pilot study. | 2003 | Chrubasik et al. [538,539,549,550,551,552] |
| MSD | RCT 97 (36/31/30) | Patients treated with Harpagophytum extract (~30 mg harpagoside/day) or NSAID (Voltaren 150 mg or Vioxx 12.5 mg), duration not provided; outcomes show equality of treatment. | 2005 | Lienert et al. [553,554] |
| DJD | RCT 60 (30/30) | Patients treated with combination of Harpagophytum and Apium graveolens extract (cream, 1.5 cm, twice daily) or placebo for 2 weeks. Treatment group showed significant improvement in algometer, flexion, and extension readings. | 2006 | Pillay [555] |
| Sore throat after tracheal intubation | RCT 60 (30/30) | Patients treated with Harpagophytum extract (480 mg one hour before intubation) or placebo plus premedication (fentanyl, midazolam, propofol). No significant difference was observed between groups. | 2016 | Anvari et al. [556] |
| DJD | RCT 92 (46/46) | Patients treated with combination of Rosa canina, Urtica sp., Harpagophytum procumbens, and vitamin D (20.0 g puree and 4.0 g juice concentrate, 160 mg dry extract, 108 mg dry extract, 5 µg, respectively) or placebo for 12 weeks. WOMAC and quality of life scores significantly improved vs. placebo. | 2017 | Moré et al. [557] |
12.2. Safety
12.2.1. Clinical Safety
12.2.2. Interaction Potential
12.2.3. Adverse Event Reports
12.2.4. Side Effects
12.2.5. Pregnancy and Lactation
13. Veterinary Applications
14. Patents
15. Discussion and Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ihlenfeldt, H.-D.; Hartmann, H. Die Gattung Harpagophytum (Burch.) DC. Ex Meissn. (Monographie der afrikanischen Pedaliaceae II). Hambg. Staatsinst. Allg. Bot. Mitt. 1970, 13, 15–69. [Google Scholar]
- Muzila, M.; Werlemark, G.; Ortiz, R.; Sehic, J.; Fatih, M.; Setshogo, M.; Mpoloka, W.; Nybom, H. Assessment of diversity in Harpagophytum with RAPD and ISSR markers provides evidence of introgression. Hereditas 2014, 151, 91–101. [Google Scholar] [CrossRef]
- Brendler, T.; van Wyk, B.E. A historical, scientific and commercial perspective on the medicinal use of Pelargonium sidoides (Geraniaceae). J. Ethnopharmacol. 2008, 119, 420–433. [Google Scholar] [CrossRef]
- Stander, M.A.; Brendler, T.; Redelinghuys, H.; Van Wyk, B.E. The commercial history of Cape herbal teas and the analysis of phenolic compounds in historic teas from a depository of 1933. J. Food Compos. Anal. 2019, 76, 66–73. [Google Scholar] [CrossRef]
- Low, C.H. Different histories of buchu: Euro-American appropriation of San and Khoekhoe knowledge of buchu plants. Environ. Hist. 2007, 13, 333–361. [Google Scholar] [CrossRef]
- Brendler, T.; Cock, I.E. A short history of Cape aloe bitters. S. Afr. J. Bot. 2021. under review. [Google Scholar]
- Helmstädter, A. Xysmalobium undulatum (Uzara) research—How everything began. J. Ethnopharmacol. 2015, 164, 385–387. [Google Scholar] [CrossRef] [PubMed]
- Brendler, T. The rise and fall of Hoodia: A lesson on the art and science of natural product commercialization. In African Natural Plant Products, Volume III: Discoveries and Innovations in Chemistry, Bioactivity, and Applications; ACS Publications: Washington, DC, USA, 2020; pp. 313–324. ISBN 1947-5918. [Google Scholar]
- Van Wyk, B.E. A review of commercially important African medicinal plants. J. Ethnopharmacol. 2015, 176, 118–134. [Google Scholar] [CrossRef]
- CITES. Inclusion of Harpagophytum Procumbens in Appendix II in Accordance with Article II 2(a) and Inclusion of Harpagophytum Zeyheri in Appendix II in Accordance with Article II 2(b) for Reasons of Look-Alike Problems. 2000, pp. 1–9. Available online: https://cites.org/sites/default/files/eng/cop/11/prop/60.pdf (accessed on 12 April 2021).
- Czygan, F.-C.; Krüger, A.; Schier, W.; Volk, O.H. Pharmazeutisch-biologische Untersuchungen der Gattung Harpagophytum (Bruch.) DC ex Meissn. 1. Mitteilung: Phytochemische Standardisierung von Tubera Harpagophyti. Dtsch. Apoth. Ztg. 1977, 117, 1431–1434. [Google Scholar]
- Eich, J.; Schmidt, M.; Betti, G.J.R. HPLC analysis of iridoid compounds of Harpagophytum taxa: Quality control of pharmaceutical drug material. Pharm. Pharmacol. Lett. 1998, 8, 75–78. [Google Scholar]
- Feistel, B.; Gaedcke, F. Analytical identification of Radix Harpagophyti procumbentis and zeyheri. Z. Phytother. 2000, 21, 246–251. [Google Scholar]
- Nott, K. A Survey of the Harvesting and Export of Harpagophytum procumbens and Harpagophytum Zeyheri in SWA/Namibia; Etosha Ecological Institute: Okaukuejo, Namibia, 1986. [Google Scholar]
- EMA. European Union Herbal Monograph on Harpagophytum procumbens DC. and/or Harpagophytum zeyheri Decne., Radix. EMA/HMPC/627057/2015; Committee on Herbal Medicinal Products (HMPC): London, UK, 2016. [Google Scholar]
- European Scientific Cooperative on Phytotherapy. Harpagophyti radix. In ESCOP Monographs, 2nd ed.; Thieme: Stuttgart, Germany, 2003; pp. 233–240. [Google Scholar]
- Menghini, L.; Recinella, L.; Leone, S.; Chiavaroli, A.; Cicala, C.; Brunetti, L.; Vladimir-Knezevic, S.; Orlando, G.; Ferrante, C. Devil’s claw (Harpagophytum procumbens) and chronic inflammatory diseases: A concise overview on preclinical and clinical data. Phytother. Res. 2019, 33, 2152–2162. [Google Scholar] [CrossRef] [PubMed]
- Stewart, K.M.; Cole, D. The commercial harvest of devil’s claw (Harpagophytum spp.) in southern Africa: The devil’s in the details. J. Ethnopharmacol. 2005, 100, 225–236. [Google Scholar] [CrossRef]
- Baghdikian, B.; Lanhers, M.C.; Fleurentin, J.; Ollivier, E.; Maillard, C.; Balansard, G.; Mortier, F. An analytical study, anti-inflammatory and analgesic effects of Harpagophytum procumbens and Harpagophytum zeyheri. Planta Med. 1997, 63, 171–176. [Google Scholar] [CrossRef]
- Anonymous. Harpagophytum procumbens (devil’s claw). Altern. Med. Rev. 2008, 13, 248–252. [Google Scholar]
- Barnes, J. Charms & harms: Devil’s claw. J. Prim. Health Care 2009, 1, 238–239. [Google Scholar] [CrossRef]
- Caprasse, M. Description, identification et usages thérapeutiques de la «griffe du diable»: Harpagophytum procumbens DC. J. Pharm. Belg. 1980, 35, 143–149. [Google Scholar]
- Chrubasik, S. Wirksamkeit pflanzlicher Schmerzmittel am Beispiel des Teufelskrallenwurzelextrakts. Orthopäde 2004, 33, 804–808. [Google Scholar] [CrossRef] [PubMed]
- Czygan, F.-C. Harpago- oder Teufelskrallentee, das Auf und Ab einer Modedroge. Z. Phytother. 1984, 5, 922–925. [Google Scholar]
- Czygan, F.-C. Nochmals Harpagophytum. Z. Phytother. 1984, 5, 972. [Google Scholar]
- Czygan, F.-C. Portrait einer Arzneipflanze: Harpagophytum—Teufelskralle. Z. Phytother. 1987, 8, 17–20. [Google Scholar]
- Dittrich, C. Harpagophytum procumbens DC. Österr. Apoth. Ztg. 1974, 28, 53–54. [Google Scholar]
- Esdorn, I. Afrikanische Reiseeindrücke in pharmazeutischer und kultureller Hinsicht. Dtsch. Apoth. Ztg. 1963, 103, 785–789. [Google Scholar]
- Faivre, C.; Ghedira, K.; Goetz, P.; Lejeune, R. Harpagophytum procumbens (Pedaliaceae). Phytothérapie 2007, 5, 150–153. [Google Scholar] [CrossRef]
- Georgiev, M.; Ivanovska, N.; Alipieva, K.; Dimitrova, P.; Verpoorte, R. Harpagoside: From Kalahari Desert to pharmacy shelf. Phytochemistry 2013, 92, 8–15. [Google Scholar] [CrossRef]
- Hansen, C. Arzneistoff Porträt—Die Afrikanische Teufelskralle—Voodoo oder wirksames Arzneimittel? Dtsch. Apoth. Ztg. 2000, 140, 85–89. [Google Scholar]
- Jaspersen-Schib, R. Harpagophyti radix—Wirklich eine Wunderdroge. Dtsch. Apoth. Ztg. 1990, 130, 71. [Google Scholar]
- Kampffmeyer, H. Teufelskralle—Gibt es eine therapeutische Wirkung? ZFA 1980, 56, 618. [Google Scholar]
- Kannacher, M. Harpagophytum procumbens—Die Teufelskralle. Tubera harpagophyti, die Speicherknollen. Volksheilkunde 1993, 45, 44. [Google Scholar]
- Lis, K. Diabelska moc czarciego pazura. Reumatologia 2010, 48, 128–132. [Google Scholar]
- McGregor, G.; Fiebich, B.; Wartenberg, A.; Brien, S.; Lewith, G.; Wegener, T. Devil’s claw (Harpagophytum procumbens): An anti-inflammatory herb with therapeutic potential. Phytochem. Rev. 2005, 4, 47–53. [Google Scholar] [CrossRef]
- McGregor, G.P. Harpagophytum procumbens—Traditional anti-inflammatory herbal drug with broad therapeutic potential. In Herbal Drugs: Ethnomedicine to Modern Medicine; Springer: New York, NY, USA, 2009; pp. 81–95. [Google Scholar]
- Miraldi, E.; Biagi, M.; Giachetti, D. A comprehensive systematic pharmacological review on Harpagophytum procumbens DC. (Devil’s claw). Biol. Sci. PJSIR 2008, 51, 165–176. [Google Scholar]
- Mncwangi, N.; Chen, W.; Vermaak, I.; Viljoen, A.; Gericke, N. Devil’s claw—A review of the ethnobotany, phytochemistry and biological activity of Harpagophytum procumbens. J. Ethnopharmacol. 2012, 143, 755–771. [Google Scholar] [CrossRef] [PubMed]
- Olivier, D.K. The Ethnobotany and Chemistry of South African Traditional Tonic Plants. Ph.D. Thesis, University of Johannesburg, Johannesburg, South Africa, 2012; p. 481. [Google Scholar]
- Richter, T. Gut beraten mit Teufelskralle? Z. Phytother. 2001, 22, 43. [Google Scholar]
- Schmidt, S. Die antiarthritische Wirkung der Harpagophytum-Wurzel. Österr. Apoth. Ztg. 1971, 25, 829. [Google Scholar]
- Schmidt, S. Rheumatherapie mit Harpagophytum. Therapiewoche 1972, 22, 1072–1074. [Google Scholar]
- Schmidt, S. Teufelskralle und Rheuma. Österr. Apoth. Ztg. 1983, 37, 111–113. [Google Scholar]
- Scholz, H. Die Wurzel aus dem roten Sand. Kosmos 1977, 73, 122–124. [Google Scholar]
- Schwabe, W. Übersicht über neuere Arzneipflanzen, die sich in den letzten 20 Jahren in der Homöopathie und der Phytotherapie bewährt haben. Allg. Homöopath. Ztg. 1980, 225, 217–229. [Google Scholar] [CrossRef]
- Seeger, P.G. Harpagophytum, ein wirksames Phytotherapeutikum. Erfahrungsheilkunde 1973, 8, 255–256. [Google Scholar]
- Seeger, P.G. Harpagophytum—Ein wirksames Phytotherapeutikum. Naturheilpraxis 1973, 10, 488–492. [Google Scholar]
- Sprecher, E. Problems with modern drugs: Ginseng-taiga root—Devil’s claw. Schr. Bundesapothekerkamm. Wiss. Fortbild. Gelbe Reihe 1977, 5, 71–95. [Google Scholar]
- Sticher, O. Die aktuelle Droge: Harpagophytum procumbens. Dtsch. Apoth. Ztg. 1977, 117, 1279–1284. [Google Scholar]
- Vanhaelen, M. La biochimie et l’activite de Harpagophytum procumbens et de Glycyrrhiza glabra. Toxicite de Symphytum consolida. J. Pharm. Belg. 1986, 41, 172–182. [Google Scholar]
- Vanhaelen, M.; Vanhaelen-Fastré, R.; Samaey-Fontaine, J.; Elchamid, A.; Niebes, P.; Matagne, D. Aspects botaniques, constitution chimique et activite pharmacologique d’Harpagophytum procumbens. Phytotherapy 1983, 5, 7–13. [Google Scholar]
- Vogel, A.; Vogel, R. Die Teufelskralle (Harpagophytum). A. Vogel Gesundh. Nachr. 1988, 45, 54–55. [Google Scholar]
- Vogel, A.; Vogel, S. Teufelskralle, Harpago. A. Vogel Gesundh. Nachr. 1973, 30, 102–104. [Google Scholar]
- Vogel, A.; Vogel, S. Harpago, Teufelskralle. A. Vogel Gesundh. Nachr. 1978, 35, 42–43. [Google Scholar]
- Vogel, A.; Vogel, S. Die Teufelskralle. A. Vogel Gesundh. Nachr. 1978, 35, 153–154. [Google Scholar]
- Vogel, G. Wissenschaftliche Erkenntnisse zu Wirksamkeit und Unbedenklichkeit pflanzlicher Arzneimittel. Therapiewoche 1984, 34, 4078–4086. [Google Scholar]
- Volk, O.H. Zur Kenntnis von Harpagophytum procumbens DC. Dtsch. Apoth. Ztg. 1964, 104, 573–576. [Google Scholar]
- Voloshyn, O.I.; Smiyan, S.I.; Voloshyna, L.O.; Horevych, S.S. Испoльзoвание мартинии душистoй (Harpagophytum procumbens) в ревматoлoгии: взгляд сквoзь призму кoмoрбиднoсти (Обзoр литературы). Semejnaâ Med. 2020, 3, 88–97. [Google Scholar] [CrossRef]
- Wegener, T. Die Teufelskralle (Harpagophytum procumbens DC.) in der Therapie rheumatischer Erkrankungen. Z. Phytother. 1998, 19, 284–294. [Google Scholar]
- Wegener, T. Wissenschaftliches Erkenntnismaterial zu Harpagophyti radix (Südafrikanische Teufelskralle) ab 1990—Unter Berücksichtigung Relevanter Früherer Studien; Kooperation Phytopharmaka: Bonn, Germany, 1998; p. 25. [Google Scholar]
- Wegener, T. Devil’s claw: From African traditional remedy to modern analgesic and antiinflammatory. HerbalGram 2000, 50, 47–54. [Google Scholar]
- Wegener, T.; Winterhoff, H. Zubereitungen aus der südafrikanischen Teufelskralle. Dtsch. Apoth. Ztg. 2001, 141, 5613–5621. [Google Scholar]
- Wiss, H.-J. Was Wissen Wir über die Teufelskralle? Library of the Namibia Scientific Society: Windhoek, Namibia, 1974; p. 13. [Google Scholar]
- Graner, G.; Lautenbacher, L. Harpagophytum procumbens DC (Teufelskralle); Kooperation Phytopharmaka: Bonn, Germany; p. 36.
- Brossier, Y. Harpagophytum procumbens DC: Apport Bénéfique de la Phytothérapie dans le Traitement de la Maladie Inflammatoire Chronique. Ph.D. Thesis, Université Paul Sabatier, Toulouse, France, 1986; p. 163. [Google Scholar]
- Mattern, B. L’Harpagophytum procumbens DC: Une Recente Acquisition de la Phytotherapie. Ph.D. Thesis, Universite de Bordeaux II, Bordeaux, France, 1983; p. 76. [Google Scholar]
- Mundy, P.J.; Ncube, S.F. Devil’s claw—A natural substitute for diclofenac? Vulture News 2014, 67, 43–47. [Google Scholar] [CrossRef]
- Anonymous. Teufelskralle als pflanzliche Alternative. Ärztez. Nat. 1999, 40, 500. [Google Scholar]
- Bonnefoy-Cudraz, Q. Le Droguier de la Faculté de Pharmacie de Montpellier (Sauvegarde du Patrimoine et Intérêt d’une des Plantes, l Harpagophytum procumbens Species). Ph.D. Thesis, Université de Montpellier I, Montpellier, France, 2013; p. 145. [Google Scholar]
- Camponovo, F. Mise au Point de Procédés pour l’Analyse Phytochimique et Étude Comparative de Quelques Médicaments à Base de Ginkgo Biloba, Panax Ginseng et Harpagophytum procumbens. Ph.D. Thesis, Université de Lausanne, Lausanne, Switzerland, 1996; p. 289. [Google Scholar]
- Couplan, F.; Danton, P. L’Harpagophytum. Un cadeau d’Afrique à menager. Rev. Monde Végétal 2000, 50, 16–19. [Google Scholar]
- Ferrara, L.; Borrelli, F.; Borbone, N. Harpagophytum procumbens: New scientific evidences. In Proceedings of the 3rd International Symposium on Natural Drugs, Napoli, Italy, 2–4 October 2003. [Google Scholar]
- Fontanel, D. L’Harpagophytum. Lett. Phytothér. Rev. Inf. Pharm. Méd. 2005, 5, 1–6. [Google Scholar]
- Franchi, G.G. Harpagophytum procumbens DC.: Una pianta africana entrata a far parte della medicina europea. Alcune osservazioni a carattere botanico, ecologico e farmacognostico. Piante Med. 2006, 5, 5–10. [Google Scholar]
- Hadolt, H. Harpagophytum procumbens: Teufelskralle, Trampelklette. PhD of Thesis, Universität Wien, Wien, Austria, 1987; p. 57. [Google Scholar]
- Kämpf, R. Harpagophytum procumbens DC, devil’s claw. Schweiz. Apoth. 1976, 114, 337–342. [Google Scholar]
- Quer, J.-C. Harpagophytum procumbens, Aspects Récents. PhD of Thesis, Université Descartes, Paris, France, 2007; p. 84. [Google Scholar]
- Schmidt, T. Harpagophytum procumbens DC. Inf. Biol. Prax. 1972, 8, 21–23. [Google Scholar]
- Smithies, S.J. Harpagophytum procumbens (Burch.) DC. Ex Meisn. Subsp. procumbens and Subsp. Transvaalense Ihlenf. & HEK Hartmann (Pedaliaceae). 2006. Available online: http://opus.sanbi.org/bitstream/20.500.12143/3477/1/Harpagophytumprocumbens_PlantzAfrica.pdf (accessed on 15 April 2021).
- Burchell, W.J. Travels in the Interior of Southern Africa; Printed for Longman, Hurst, Rees, Orme, and Brown: London, UK, 1822; Volume 1, pp. 529–537. [Google Scholar]
- Meisner, C.D.F. Plantarum Vascularium Genera: Secundum Ordines Naturales Digesta Eorumque Differentiae et Affinitates Tabulis Diagnostacis Expositae; Libraria Weidmannia: Leipzig, Germany, 1836–1843; Volume 1,2. [Google Scholar]
- De Candolle, A. Prodromus Systematis Naturalis Regni Vegetabilis, Sive, Enumeratio Contracta Ordinum Generum Specierumque Plantarum Huc Usque Cognitarium, Juxta Methodi Naturalis, Normas Digesta: Pedalineae; Fortin, Masson et Sociorum: Paris, France, 1845; Volume 9, pp. 253–257. [Google Scholar]
- Decaisne, M.J. Revue du groupe des pédalinées: Harpagophytum DC. Ann. Sci. Nat. Bot. 1865, 5, 321–336. [Google Scholar]
- Mncwangi, N.; Vermaak, I.; Viljoen, A. Mid-infrared spectroscopy and short wave infrared hyperspectral imaging—A novel approach in the qualitative assessment of Harpagophytum procumbens and H. zeyheri (Devil’s claw). Phytochem. Lett. 2014, 7, 143–149. [Google Scholar] [CrossRef]
- Muzila, M. Genetic, Morphological and Chemical Variation in the Genus Harpagophytum. Ph.D. Thesis, Swedish University of Agricultural Sciences, Alnarp, Sweden, 2016; p. 76. [Google Scholar]
- Muzila, M.; Setshogo, M.P.; Mpoloka, S.W. Multivariate analysis of Harpagophytum DC. Ex Meisn (Pedaliaceae) based on fruit characters. Int. J. Biodivers. Conserv. 2011, 3, 101–109. [Google Scholar]
- Mncwangi, N.; Viljoen, A.; Vermaak, I.; Chen, W.; Zhang, J.; Khan, I.A. Metabolomic profiling and quality control of Harpagophytum spp. (Devil’s claw). Planta Med. 2014, 80, CL1. [Google Scholar] [CrossRef]
- Mosoabisane, M.F.T. Variation in chemical composition of Harpagophytum species as function of age and locality. Master’s Thesis, University of the Free State, Bloemfontein, South Africa, 2009; p. 181. [Google Scholar]
- Steenkamp, P.A.; Steenkamp, L.H. UPLC-MS profiling, identification of major peaks and comparison of Harpagophytum procumbens extracts from different locations. S. Afr. J. Bot. 2019, 124, 138–143. [Google Scholar] [CrossRef]
- Hargreaves, B.J. The sesame family in Botswana. Botsw. Notes Rec. 1993, 25, 141–159. [Google Scholar]
- Van Wyk, B.E.; Gericke, N. People’s Plants—A Guide to Useful Plants of Southern Africa, 2nd, revised, and expanded ed.; Briza Publications: Queenswood, South Africa, 2018; pp. 174, 224. [Google Scholar]
- Von Koenen, E. Heil-, Gift- und Essbare Pflanzen in Namibia; Klaus Hess Verlag: Göttingen, Germany, 1996; p. 336. ISBN 3980451828. [Google Scholar]
- Smith, C.A. Common Names of South African Plants; Government Printer: Pretoria, South Africa, 1966; p. 642.
- Ihlenfeldt, H.-D. Bemerkungen zur Taxonomie der süDwestafrikanischen Pedaliaceae; Mitteilungen der Botanischen Staatssammlung München Band VI.: München, Germany, 1967; pp. 593–612. [Google Scholar]
- Baum, H. Kunene-Sambesi-Expedition; Otto Warburg, Verlag des Kolonial-Wirtschaftlichen Komitees: Berlin, Germany, 1903; p. 604. [Google Scholar]
- Blank, R.J. Voraussetzungen und Möglichkeiten für Einen Feldmäßigen Anbau der Wildpflanze Harpagophytum procumbens (Teufelskralle). Master Thesis, Universität Hohenhein, Stuttgart-Hohenheim, Germany, 1973; p. 63. [Google Scholar]
- Blank, R.J. Arbeiten und Berichte 19. Versuche zur Vermehrung von Harpagophytum procumbens DC. (Teufelskralle); Universität Hohenheim, Abteilung Pflanzenbau in den Tropen und Subtropen: Stuttgart-Hohenheim, Germany, 1976. [Google Scholar]
- Von Willert, D.J.; Schneider, E. Teufelskralle: Anbau und Wildsammlung—Ein Beitrag zur pharmakognostischen Ökologie. Dtsch. Apoth. Ztg. 2001, 141, 683–688. [Google Scholar]
- Wood, J.G. The Uncivilized Races, or Natural History of Man; American Publishing Company: Hartford, CT, USA, 1870; Volume 1, p. 783. [Google Scholar]
- Cooke, M.C. Freaks and Marvels of Plant Life: Or Curiosities of Vegetation; Society for Promoting Christian Knowledge: London, UK, 1882; p. 463. [Google Scholar]
- Lübbert, A. Aus dem deutsch-südwestafrikanischen Schutzgebiete. Ueber die Heilmethoden und Heilmittel der Eingeborenen in Deutsch-Südwestafrika. Mitth. Forsch. Gelehrt. Dtsch. Schutzgeb. 1901, 14, 77–90. [Google Scholar]
- Hellwig, M. Angaben von Eingeborenen über die Feldkost und die Arzneipflanzen der Herrero und Hottentotten; Reichskolonialamt (Bundesarchiv R 1001/5989, fol. 78–81): Berlin, Germany, 1907. [Google Scholar]
- Dinter, K. Die Vegetabilische Veldkost Deutsch-Südwest-Afrikas; Selbstverlag: Okahandja, Namibia, 1912; p. 24. [Google Scholar]
- Dinter, K. Deutsch-Südwest-Afrika. Flora, Forst- und Landwirtschaftliche Fragmente; Weigel: Leipzig, Germany, 1909; p. 212. [Google Scholar]
- Schön, A. Vom Pfeilgift zur Arznei: Untersuchungen von Arzneidrogen und Giften aus den Ehemaligen Deutschen Kolonien West- und Südwestafrikas, Vornehmlich an Berliner Instituten (1884–1918): Ein Beitrag zur Kolonialpharmazie; Wissenschaftliche Verlagsgesellschaft mbH: Stuttgart, Germany, 2017; p. 593. ISBN 3804737684. [Google Scholar]
- Kroemer, B. Mit Schwert & Pflugschar in Sachsen und Südwestafrika. Anekdoten und Geschichten Eines Südwester Pioniers: Gottreich Hubertus Mehnert; “Glanz & Gloria” Verlag: Windhoek, Namibia, 2007; p. 128. ISBN 9789991668970. [Google Scholar]
- Anderson, S.; Staugard, F. Traditional Midwives (Traditional Medicine in Botswana); Ipelegeng Publishers: Gaborone, Botswana, 1986; p. 264. ISBN 978-9178103973. [Google Scholar]
- Bieg, S. Beiträge Zur Kenntnis Einiger Heilpflanzen aus Deutsch-Südwestafrika mit Einer Liste der Dort Vorkommenden Medizinisch Verwendeten Pflanzen. Ph.D. Thesis, Technische Hochschule, Stuttgart, Germany, 1939; p. 49. [Google Scholar]
- Van Damme, P.; van den Eynden, V.; Vernemmen, P. Plant uses by the Topnaar of the Sesfontein area (Namib desert). Afr. Focus 1992, 8. [Google Scholar] [CrossRef]
- Watt, J.M.; Breyer-Brandwijk, M.G. The Medicinal and Poisonous Plants of Southern and Eastern Africa, 2nd ed.; Livingstone: London, UK, 1962; p. 1457. [Google Scholar]
- Staugard, F. Traditional midwives in Botswana. Botsw. Natl. Health Bull. 1985, 1, 42–60. [Google Scholar] [PubMed]
- Moreki, J.C. Documentation of ethnoveterinary practices used in family poultry in Botswana. Vet. World 2013, 6, 18–21. [Google Scholar] [CrossRef]
- Maas, H. Außenminister Maas zum Abschluss der Verhandlungen mit Namibia. 2021. Available online: https://www.auswaertiges-amt.de/de/newsroom/-/2463396 (accessed on 10 June 2021).
- Kock, R. Erinnerungen an die Internierungszeit (1939–1946) und Zeitgeschichtliche Ergänzungen; Selbstverlag “Andalusia”: Windhoek, Namibia, 1975; p. 209. [Google Scholar]
- Zorn, B. Über die antiarthritische Wirkung der Harpagophytum-Wurzel. Dtsch. Rheumaforsch. 1958, 17, 134–138. [Google Scholar]
- Lux, R.E. Über ein Glukosid der Wurzel von Harpagophytum procumbens. Ph.D. Thesis, Julius-Maximilians-Universität, Würzburg, Germany, 1960; p. 75. [Google Scholar]
- Stierstorfer, N. Ein Beitrag zur Kenntnis der Inhaltsstoffe von Harpagophytum procumbens DC. Ph.D. of Thesis, Julius-Maximilians-Universität, Würzburg, Germany, 1961; p. 54. [Google Scholar]
- Tunmann, P.; Lux, R.E. Zur chemischen Konstitution des Harpagosids. Pharm. Ztg. 1961, 106, 1357. [Google Scholar]
- Tunmann, P.; Lux, R.E. Zur Kenntnis der Inhaltsstoffe aus der Wurzel von Harpagophytum procumbens DC. 1. Mitteilung: Isolierung und Eigenschaften der Glukoside Harpagosid und Harpagid. Dtsch. Apoth. Ztg. 1962, 102, 1274–1275. [Google Scholar]
- Tunmann, P.; Lux, R.E. Zur chemischen Konstitution des Harpagosids. Dtsch. Apoth. Ztg. 1962, 101, 1383. [Google Scholar]
- Fickentscher, K. Beitrag zur Chemischen Konstitution des Harpagids. Ph.D. Thesis, Julius-Maximilians-Universität, Würzburg, Germany, 1963; p. 63. [Google Scholar]
- Tunmann, P.; Stierstorfer, N. Zur Kenntnis der Inhaltsstoffe aus der Wurzel von Harpagophytum procumbens DC. 2. Mitteilung. Dtsch. Apoth. Ztg. 1963, 103, 395–397. [Google Scholar]
- Lichti, H.; von Wartburg, A. Zur Konstitution von Harpagosid. Tetrahedron Lett. 1964, 5, 835–843. [Google Scholar] [CrossRef]
- Tunmann, P.; Stierstorfer, N. Zur Kenntnis der Inhaltsstoffe aus der Wurzel von Harpagophytum procumbens DC. Tetrahedron Lett. 1964, 5, 1697–1699. [Google Scholar] [CrossRef]
- Lichti, H.; von Wartburg, A. Die Struktur des Harpagosids. 2. Mitteilung über Iridoide. Helv. Chim. Acta 1966, 49, 1552–1580. [Google Scholar] [CrossRef]
- Hammer, H.-E. Konstitution des Procumbids und ein Beitrag zur Kenntnis Weiterer Inhaltsstoffe aus der Wurzel von Harpagophytum procumbens DC. Ph.D. Thesis, Julius-Maximilians-Universität, Würzburg, Germany, 1967; p. 65. [Google Scholar]
- Tunmann, P.; Hammer, H.-E. Inhaltsstoffe aus der Wurzel von Harpagophytum procumbens DC., IV. Konstitution des Procumbids. Justus Liebigs Ann. Chem. 1968, 712, 138–145. [Google Scholar] [CrossRef]
- Hagen, E. Correspondence Concerning the Trademarking of “Harpago”; Library of the Namibia Scientific Society: Windhoek, Namibia, 1961–1976; p. 10. [Google Scholar]
- Schmidt, S. Phytotherapie beim rheumatischen Formenkreis. Arch. Arzneither. 1978, 3, 266–271. [Google Scholar]
- Beck, H.; Sand, J.-M.; Kuhlmann, J. Arzneimittelmarkt aktuell. Dtsch. Apoth. Ztg. 1981, 121, 2884–2889. [Google Scholar]
- Marandet, E. Harpagophytum procumbens DC. De l’Utilisation Traditionnelle à la Réglementation Européenne. Ph.D. Thesis, Université de Reims Champagne-Ardenne, Reims, France, 2009; p. 97. [Google Scholar]
- Hagen, E. Harpagophytum. Letter to H.-J. Wiss Concerning Sustainability of Devil’s Claw Exports to Germany; Library of the Namibia Scientific Society: Windhoek, Namibia, 1975; p. 2. [Google Scholar]
- Achtnich, W. Harpagophytum. Letter to the South African Embassy in Germany (Cologne) Concerning Sustainability of Devil’s Claw Exports to Germany; Library of the Namibia Scientific Society: Windhoek, Namibia, 1975; p. 2. [Google Scholar]
- De Bruine, J.R.; Clark, D.L. A Short Revue of the Harpagophytum procumbens Problem; Department of Nature Conservation: Windhoek, Namibia, 1976.
- Kgathi, D.L. The Grapple Plant Project. Seventh Progress Report: Aspects of Grapple Trade; University of Botswana: Gaborone, Botswana, 1987. [Google Scholar]
- Kgathi, D.L. The grapple trade in Botswana. Botsw. Notes Rec. 1988, 20, 119–124. [Google Scholar]
- Taylor, F.W.; Moss, H. Final Report on the Potential for Commercial Utilization of Veld Products. The Resource and its Management; Ministry of Commerce & Industry: Gaborone, Botswana, 1982; Volume 1, p. 205.
- Engels, G.; Brinckmann, J.A. Devil’s claw—Harpagophytum procumbens, H. zeyheri. HerbalGram 2018, 118, 1–14. [Google Scholar]
- Cunningham, A.B. African Medicinal Plants: Setting Priorities at the Interface Between Conservation and Primary Health Care; UNESCO: Paris, France, 1993. [Google Scholar]
- Hachfeld, B. Analysis of the Trade Potential and Possible Over-Exploitation of a Southern African Medicinal Plant: Harpagophytum Procumbens; Bundesamt für Naturschutz: Bonn, Germany, 1999. [Google Scholar]
- CITES. Biological And Trade Status of Harpagophytum. 2002, pp. 1–15. Available online: https://cites.org/sites/default/files/eng/cop/12/doc/E12-46.pdf (accessed on 12 April 2021).
- Grote, K. The Increased Harvest and Trade of Devil’s Claw (Harpagophytum procumbens) and Its Impacts on the Peoples and Environment of Namibia, Botswana and South Africa; Global Facilitation Unit for Underutilized Species: Maccarese, Italy, 2003; p. 30. [Google Scholar]
- Hachfeld, B. Ecology and Utilisation of the Medicinal Plant Harpagophytum procumbens (Burch.) DC. ex Meissn. (Pedaliaceae) in Southern Africa; Bundesamt für Naturschutz: Bonn, Germany, 2004. [Google Scholar]
- Hachfeld, B. Ecology and Utilisation of the Medicinal Plant Harpagophytum procumbens (Burch.) DC. ex Meissn. (Pedaliaceae) in Southern Africa. Ph.D. Thesis, Universität Hamburg, Hamburg, Germany, 2004; p. 305. [Google Scholar]
- Cole, D.; Bennett, B. Trade, Poverty and Natural Products: Lessons Learned from Namibian Organic Devil’S Claw. 2007. Available online: http://searchworks.stanford.edu/view/7838432 (accessed on 12 April 2021).
- Hachfeld, B.; Schippmann, U. Conservation data sheet 2: Exploitation, trade and population status of Harpagophytum procumbens in southern Africa. Med. Plant Conserv. 2000, 6, 4–9. [Google Scholar]
- Kathe, W.; Barsch, F.; Honnef, S. Trade in Devil’s Claw (Harpagophytum spp.) in Germany—Status, Trends and Certification. 2003, pp. 1–40. Available online: http://foris.fao.org/static/pdf/NWFP/Germany_devils_claw.pdf (accessed on 13 April 2021).
- Schippmann, U. Imports of Harpagophytum in Germany. In Proceedings of the First Regional Devil’s Claw Conference, Windhoek, Namibia, 26–28 February 2002; p. 3. [Google Scholar]
- Suckert, B. Successful marketing strategies as a tool for development. In Proceedings of the First Regional Devil’s Claw Conference, Windhoek, Namibia, 26–28 February 2002; p. 2. [Google Scholar]
- Censkowsky, U.; Helberg, U.; Nowack, A.; Steidle, M. Overview of World Production and Marketing of Organic Wild Collected Products; ITC: Geneva, Switzerland, 2007; p. 91. [Google Scholar]
- Nott, K.; Nott, A.; Newton, D. A Critical Assessment of the Economic and Environmental Sustainability of the Namibian Indigenous Forest/Timber Industry with Reference to Zambia and Angola; TRAFFIC: Pretoria, South Africa, 2020; p. 101. [Google Scholar]
- United States Pharmacopeial Convention. Harpagophytum Species Root. Proposed For Development Version 0.1. Herbal Medicines Compendium; USP: Rockville, MD, USA, 2013. [Google Scholar]
- British Herbal Medicine Association. Harpagophytum. In British Herbal Pharmacopoeia, Part Three; BHMA: Cowling, UK, 1981; p. 49. [Google Scholar]
- ANSM. Harpagophyton. Harpagophytum procumbens. In Pharmacopée Française, 10th ed.; Maisonneuve: Sainte-Ruffine, France, 1989; pp. 183–184. [Google Scholar]
- Koch, H.P.; Hadold, H. Harpagophytum procumbens. Teufelskralle, Trampelklette; Kooperation Phytopharmaka: Bonn, Germany, 1988; p. 75. [Google Scholar]
- Kommission, E. Harpagophyti radix (Berichtigung). Bundesanzeiger 1990, 164, 1. [Google Scholar]
- ANSM. Extrait d’Harpagophyton (Sec). Harpagophyti extractum siccum. In Pharmacopée Française, 10th ed.; Maisonneuve: Sainte-Ruffine, France, 1992; pp. 1–4. [Google Scholar]
- Bundesministerium für Gesundheit und Soziale Sicherung. Teufelskrallenwurzel. Harpagophyti radix. In Deutsches Arzneibuch (DAB) 10. 2. Nachtrag; Deutscher Apotheker Verlag: Stuttgart, Germany, 1993. [Google Scholar]
- EDQM. Devil’s claw root, Harpagophyti radix, 1997:1095. In European Pharmacopoeia, 3rd ed.; published June 1996, replaces the 2nd ed on 1 January 1997; Council of Europe: Strasbourg, France, 1997; p. 1821. [Google Scholar]
- EDQM. Devil’s claw root, Harpagophyti radix, 01/2003:1095. In European Pharmacopoeia, Supplement 4.3 to the Fourth Edition, published 20 June 2002; Council of Europe: Strasbourg, France, 2003; p. 359. [Google Scholar]
- ANSM. Devil’s claw root for homoeopathic preparations. Harpagophytum for homoeopathic preparations. Harpagophytum ad praeparationes homoeopathicas. In Pharmacopée Française, 10th ed.; Maisonneuve: Sainte-Ruffine, France, 2007; pp. 1–3. [Google Scholar]
- EDQM. Devil’s claw dry extract, Harpagophyti extractum siccum, 01/2008:1871. In European Pharmacopoeia, 7th ed.; Council of Europe: Strasbourg, France, 2008. [Google Scholar]
- Health Canada. Devil’s Claw—Harpagophytum. Nat. Health Prod. Ingred. Database 2021. Available online: http://webprod.hc-sc.gc.ca/nhpid-bdipsn/ingredReq.do?id=6188&lang=eng (accessed on 12 April 2021).
- EDQM. Devil’s claw root, Harpagophyti radix, 01/2011:1095. In European Pharmacopoeia, 7th ed.; Council of Europe: Strasbourg, France, 2011. [Google Scholar]
- Bacler-Żbikowska, B.; Drobnik, J. Komentarz botaniczny do roślin leczniczych i surowców roślinnych wymienionych w Farmakopei polskiej VIII. Część, I. Ann. Acad Med. Silesiensis 2011, 1–2, 48–60. [Google Scholar]
- EMA. Assessment Report on Harpagophytum procumbens DC. and/or Harpagophytum zeyheri Decne., Radix. EMA/HMPC/627058/2015; Committee on Herbal Medicinal Products (HMPC): London, UK, 2016. [Google Scholar]
- EMA. List of References Supporting the Assessment of Harpagophytum procumbens DC. and/or Harpagophytum zeyheri Decne., radix. EMA/HMPC/627059/2015; Committee on Herbal Medicinal Products (HMPC): London, UK, 2016. [Google Scholar]
- EDQM. Devil’s claw root, Harpagophyti radix, 01/2011:1095 corrected 9.6. In European Pharmacopoeia, 9th ed.; 6th supplement, Council of Europe: Strasbourg, France, 2018. [Google Scholar]
- Kriukova, A.; Vladymyrova, I.; Gubar, S.; Kotov, A.; Kotova, E. Question introduction to the State Pharmacopoeia of Ukraine monograph «Devil’s claw root». Manag. Econ. Qual. Assur. Pharm. 2018, 6–12. [Google Scholar] [CrossRef]
- European Scientific Cooperative on Phytotherapy. Harpagophyti radix. In ESCOP Monographs. Fascicule 2; ESCOP: Exeter, UK, 1996; pp. 1–7. [Google Scholar]
- Bradley, P.R. Addendum to the ESCOP monograph on Harpagophytum procumbens: Reply from ESCOP. Phytomedicine 2004, 11, 696. [Google Scholar]
- Chrubasik, S. Addendum to the ESCOP monograph on Harpagophytum procumbens. Phytomedicine 2004, 11, 696. [Google Scholar] [CrossRef]
- World Health Organization. Radix Harpagophyti. In WHO Monographs on Selected Medicinal Plants; World Health Organization: Geneva, Switzerland, 2007; Volume 3, pp. 182–193. ISBN 9241545372. [Google Scholar]
- Anonymous. Harpagophytum procumbens. In African Herbal Pharmacopoeia; Brendler, T., Eloff, J.N., Gurib-Fakim, A., Phillips, L.D., Eds.; Association for African Medicinal Plants Standards Port Louis: Port Louis, Mauritius, 2010; pp. 127–134. ISBN 9990389098. [Google Scholar]
- Devil’s claw root. In Martindale: The Complete Drug Reference; Alison, B., Ed.; The Pharmaceutical Press: London, UK, 2017; ISBN 978-0857113092. [Google Scholar]
- Kemper, K.J.; Devil’s Claw (Harpagophytum procumbens). Longwood Herb. Task Force Cent. Holist. Pediatr. Educ. Res. 1999, pp. 1–11. Available online: http://ineldea.com.ua/images/devilsclaw.pdf (accessed on 13 April 2021).
- Devil’s claw. In Herbal Medicines; Barnes, J., Baxter, I.A., Smith, M., Veitch, N.C., Eds.; Pharmaceutical Press: London, UK, 2013; pp. 238–245. ISBN 9780857110350. [Google Scholar]
- Edwards, S.E.; da Costa Rocha, I.; Williamson, E.M.; Heinrich, M. Devil’s Claw Harpagophytum procumbens (Burch.) DC. Ex Meissner, H. zeyheri Decne. In Phytopharmacy: An Evidence-Based Guide to Herbal Medicinal Products; Willey Blackwell: Hoboken, NJ, USA, 2015; p. 131. ISBN 9781118543436. [Google Scholar]
- Kooperation Phytopharmaka. Harpagophytum procumbens DC (Teufelskralle). Arzneipflanzenlexikon. 2020. Available online: https://arzneipflanzenlexikon.info/teufelskralle.php (accessed on 12 April 2021).
- Karioti, A.; Fani, E.; Vincieri, F.F.; Bilia, A.R. Analysis and stability of the constituents of Curcuma longa and Harpagophytum procumbens tinctures by HPLC-DAD and HPLC-ESI-MS. J. Pharm. Biomed. Anal. 2011, 55, 479–486. [Google Scholar] [CrossRef]
- Mncwangi, N.; Viljoen, A.; Zhao, J.; Vermaak, I.; Chen, W.; Khan, I.A. What the devil is in your phytomedicine? Exploring species substitution in Harpagophytum through chemometric modeling of 1H-NMR and UHPLC-MS datasets. Phytochemistry 2014, 106, 104–115. [Google Scholar] [CrossRef]
- Mncwangi, N.; Viljoen, A.M.; Vermaak, I.; Chen, W. Variation of the biologically active constituent harpagoside in Harpagophytum procumbens and H. zeyheri. Planta Med. 2013, 79, P58. [Google Scholar] [CrossRef]
- Muzila, M.; Ekholm, A.; Nybom, H.; Widén, C.; Rumpunen, K. Harpagophytum germplasm varies in tuber peel and pulp content of important phenylpropanoids and iridoids. S. Afr. J. Bot. 2018, 115, 153–160. [Google Scholar] [CrossRef]
- Koch, J.-K.-H. Über ein Chinon und Weitere Inhaltsstoffe aus der Wurzel von Harpagophytum procumbens DC. Ph.D. Thesis, Julius-Maximilians-Universität, Würzburg, Germany, 1970; p. 60. [Google Scholar]
- Bianco, A.; Esposito, P.; Guiso, M.; Scarpati, M.L. Iridoids. 10. Procumbide, a diastereoisomer of antirrinoside. Gazz. Chim. Ital. 1971, 101, 764. [Google Scholar]
- Bauersfeld, H.-J. Über Weitere Inhaltsstoffe aus der Wurzel von Harpagophytum procumbens DC. Ph.D. Thesis, Julius-Maximilians-Universität, Würzburg, Germany, 1974; p. 64. [Google Scholar]
- Tunmann, P.; Bauersfeld, H.J. Über weitere Inhaltsstoffe der Wurzel von Harpagophytum procumbens DC. Arch. Pharm. 1975, 308, 655–657. [Google Scholar] [CrossRef] [PubMed]
- Sticher, O. Plant mono-, di- and sesquiterpenoids with pharmacological or therapeutical activity. In New Natural Products and Plant Drugs with Pharmacological, Biological or Therapeutical Activity. In Proceedings of the First International Congress on Medicinal Plant Research, Section A, Munich, Germany, 6–10 September 1976; Wagner, H., Wolff, P., Eds.; Springer: Berlin/Heidelberg, Germany, 1977; pp. 137–176, ISBN 978-3-642-66684-1. [Google Scholar]
- Kwasniewski, V. Beitrag zur Kenntnis der Inhaltsstoffe der Sekundärwurzeln von Harpagophytum procumbens DC. und zur Frage ihres eventuellen Ersatzes durch einheimische Drogen. Dtsch. Apoth. Ztg. 1978, 118, 49–50. [Google Scholar]
- Bendall, M.R.; Ford, C.W.; Thomas, D.M. The structure of procumbide. Aust. J. Chem. 1979, 32, 2085–2091. [Google Scholar] [CrossRef]
- Ziller, K.H.; Franz, G. Analysis of the water soluble fraction from the roots of Harpagophytum procumbens. Planta Med. 1979, 37, 340–348. [Google Scholar] [CrossRef]
- Vanhaelen, M.; Vanhaelen Fastré, R.; Elchami, A.; Fontaine, J. Activité biologique d’Harpagophytum procumbens DC. I. Préparation et structure de l’harpagogénine. J. Pharm. Belg. 1981, 36, 38–42. [Google Scholar]
- Franz, G.; Czygan, F.C.; Abou-Mandour, A.A. Untersuchungen der Gattung Harpagophytum. 4. Mitteilung: Gehalt an freien Zuckern und Harpagosid in Kalluskulturen und genuinen Wurzelgeweben von Harpagophytum procumbens. Planta Med. 1982, 44, 218–220. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, T.; Matsuda, S.; Kubo, Y.; Namba, T. New iridoid glucosides from Harpagophytum procumbens DC. Chem. Pharm. Bull. 1983, 31, 2296–2301. [Google Scholar] [CrossRef]
- Burger, J.F.W.; Brandt, E.V.; Ferreira, D. Iridoid and phenolic glycosides from Harpagophytum procumbens. Phytochemistry 1987, 26, 1453–1457. [Google Scholar] [CrossRef]
- Baghdikian, B.; Ollivier, E.; Faure, R.; Debrauwer, L.; Rathelot, P.; Balansard, G. Two new pyridine monoterpene alkaloids by chemical conversion of a commercial extract of Harpagophytum procumbens. J. Nat. Prod. 1999, 62, 211–213. [Google Scholar] [CrossRef]
- Picavet, S. Iridoides d’Harpagophytum procumbens DC (Plante a Propriete Anti-Rhumatismale). Ph.D. Thesis, Université de Reims Champagne-Ardenne, Reims, France, 2000; p. 31. [Google Scholar]
- Boje, K. Phytochemische und Biopharmazeutische Untersuchungen an Harpagophytum procumbens DC. Ph.D. Thesis, Westfälische Wilhelms-Universität Münster, Münster, Germany, 2002; p. 286. [Google Scholar]
- Clarkson, C.; Campbell, W.E.; Smith, P. In Vitro antiplasmodial activity of abietane and totarane diterpenes isolated from Harpagophytum procumbens (Devil’s claw). Planta Med. 2003, 69, 720–724. [Google Scholar] [CrossRef] [PubMed]
- Boje, K.; Lechtenberg, M.; Nahrstedt, A. New and known iridoid-and phenylethanoid glycosides from Harpagophytum procumbens and their in vitro inhibition of human leukocyte elastase. Planta Med. 2003, 69, 820–825. [Google Scholar] [CrossRef]
- Munkombwe, N.M. Acetylated phenolic glycosides from Harpagophytum procumbens. Phytochemistry 2003, 62, 1231–1234. [Google Scholar] [CrossRef]
- Goroll, K.A. Pharmakologische Charakterisierung von Harpagosid, Einem Wirkstoff der Teufelskralle. Ph.D. Thesis, Universität Ulm, Ulm, Germany, 2004; p. 97. [Google Scholar]
- Clarkson, C.; Staerk, D.; Hansen, S.H.; Smith, P.J.; Jaroszewski, J.W. Discovering new natural products directly from crude extracts by HPLC-SPE-NMR: Chinane diterpenes in Harpagophytum procumbens. J. Nat. Prod. 2006, 69, 527–530. [Google Scholar] [CrossRef]
- Clarkson, C.; Staerk, D.; Hansen, S.H.; Smith, P.J.; Jaroszewski, J.W. Identification of major and minor constituents of Harpagophytum procumbens (Devil’s claw) using HPLC-SPE-NMR and HPLC-ESIMS/APCIMS. J. Nat. Prod. 2006, 69, 1280–1288. [Google Scholar] [CrossRef]
- Qi, J.; Chen, J.J.; Cheng, Z.H.; Zhou, J.H.; Yu, B.Y.; Qiu, S.X. Iridoid glycosides from Harpagophytum procumbens DC (Devil’s claw). Phytochemistry 2006, 67, 1372–1377. [Google Scholar] [CrossRef]
- Qi, J.; Zhou, J.; Chen, L.; Chen, J.J.; Yu, B.; Qiu, S. Study on chemical constituents in tuber of Harpagophytum procumbens DC. Chin. Pharm. J. 2006, 41, 1613–1615. [Google Scholar]
- Qi, J.; Chen, J.; Tu, Y.; Chen, L.; Yu, B. Chemical constituents of African plant Harpagophytum procumbens. Chin. J. Nat. Med. 2007, 5, 105–107. [Google Scholar]
- Wilken, D.; Röhnert, P.; Appel, K. Erschließung und Charakterisierung Therapeutisch Neuroprotektiver and Anti-Inflammatorischer Wirkstoffe aus Harpagophytum procumbens (Teufelskralle)—TheraTek. Abschlußbericht; BioPlanta GmbH: Leipzig, Germany, 2008; p. 41. [Google Scholar]
- Appel, K.; Rose, T.; Fiebich, B.; Röhnert, P.; Claus, D.; Gerth, A.; Wilken, D. Neuroprotective and antiinflammatory effects of extracts from in vitro cultivated Harpagophytum procumbens (Devil’s claw). Z. Phytother. 2009, 30, P03. [Google Scholar] [CrossRef]
- Qi, J.; Li, N.; Zhou, J.H.; Yu, B.Y.; Qiu, S.X. Isolation and anti-inflammatory activity evaluation of triterpenoids and a monoterpenoid glycoside from Harpagophytum procumbens. Planta Med. 2010, 76, 1892–1896. [Google Scholar] [CrossRef] [PubMed]
- Zgrajka, W.; Turska, M.; Rajtar, G.; Majdan, M.; Parada-Turska, J. Kynurenic acid content in anti-rheumatic herbs. Ann. Agric. Environ. Med. 2013, 20, 800–802. [Google Scholar] [PubMed]
- Tomassini, L.; Serafini, M.; Foddai, S.; Ventrone, A.; Nicoletti, M. A new iridoid diglucoside from Harpagophytum procumbens. Nat. Prod. Res. 2016, 30, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, S. “Teufelskralle” vielfach bakteriell bedenklich. Dtsch. Apoth. Ztg. 1978, 118, 1808–1809. [Google Scholar]
- Abramowicz, M. Toxic reactions to plant products sold in health food stores. Med. Lett. Drugs Ther. 1979, 21, 29–31. [Google Scholar]
- Loew, D.; Schuster, O.; Möllerfeld, J. Stabilität und biopharmazeutische Qualität. Voraussetzung für Bioverfügbarkeit und Wirksamkeit von Harpagophytum procumbens. In Phytopharmaka II, Forschung und Klinische Anwendung; Loew, D., Rietbrock, N., Eds.; Steinkopff: Darmstadt, Germany, 1996; pp. 83–93. [Google Scholar]
- Chrubasik, S. Biopharmazeutische Qualität und klinische Wirksamkeit von Zubereitungen aus Harpagophytum Extrakt. In Rheumatherapie mit Phytopharmaka; Chrubasik, S., Wink, M., Eds.; Hippokrates: Stuttgart, Germany, 1997; pp. 77–85. [Google Scholar]
- Schier, W.; Bauersfeld, H.-J. Handelssorten von Harpagophytum procumbens DC. 1. Mitteilung. Dtsch. Apoth. Ztg. 1973, 113, 795–796. [Google Scholar]
- Schier, W. Handelssorten von Harpagophytum procumbens DC. 2. Mitteilung. Dtsch. Apoth. Ztg. 1974, 114, 1800–1801. [Google Scholar]
- Becker, H.; Richter, S. Eine einfache dünnschichtchromatographische Untersuchung von Harpagophytum procumbens für das Apotheken-Labor. Pharm. Ztg. 1975, 120, 441–442. [Google Scholar]
- Czygan, F.-C.; Krüger, A. Pharmazeutisch-biologische Untersuchungen der Gattung Harpagophytum—3. Mitteilung: Zur Verteilung des Iridoid-Glycosids Harpagosid in den einzelnen Organen von Harpagophytum procumbens DC und Harpagophytum zeyheri Decne. Planta Med. 1977, 31, 305–307. [Google Scholar] [CrossRef]
- Haag-Berrurier, M.; Kuballa, B.; Anton, R. Dosage des glucoiridoïdes totaux dans la racine d’Harpagophytum procumbens DC. Plantes Méd. Phytothér. 1978, 12, 197–206. [Google Scholar]
- Sticher, O.; Meier, B. Quantitative Bestimmung von Harpagosid in Wurzeln von Harpagophytum procumbens mit Hochleistungsflüssigkeitschromatographie (HPLC). Dtsch. Apoth. Ztg. 1980, 120, 1592–1594. [Google Scholar]
- Vanhaelen, M.; Vanhaelen-Fastré, R.; Elchami, A.A. Gas-liquid chromatographic determination of the iridoid content in Harpagophytum procumbens DC. J. Chromatogr. A 1981, 209, 476–478. [Google Scholar] [CrossRef]
- Ragusa, S.; Circosta, C.; Galati, E.M.; Tumino, G. A drug used in traditional medicine. Harpagophytum procumbens DC I. Scanning electron microscope observations. J. Ethnopharmacol. 1984, 11, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Pourrat, H.; Texier, O.; Vennat, B.; Pourrat, A.; Galliard, J. Etude de la stabilité des iridoïdes d’Harpagophytum procumbens DC au cours de la préparation de poudres et d’atomisats. Ann. Pharm. Fr. 1985, 43, 601–606. [Google Scholar] [PubMed]
- Ficarra, P.; Ficarra, R.; Tommasini, A.; De Pasquale Costa, R.; Guarniera Fenech, C.; Ragusa, S. Analisi mediante HPLC di una droga della medicina tradizionale: Harpagophytum procumbens DC. Nota I. Boll. Chim. Farm. 1986, 125, 250–253. [Google Scholar] [PubMed]
- Franke, A.; Rimpler, H. GC/MS-Analyse methylierter Iridoidglykoside. Planta Med. 1986, 52, 89–95. [Google Scholar] [CrossRef]
- Guillerault, L.; Ollivier, E.; Elias, R.; Balansard, G. Determination of harpagide, 8-para-coumaroyl harpagide, and harpagoside by high performance liquid chromatography in Harpagophytum procumbens drugs and in a commercial extract. J. Liq. Chromatogr. 1994, 17, 2951–2960. [Google Scholar] [CrossRef]
- Mestdagh, O.; Torck, M. Etude de la qualité de gélules d’Harpagophyton. Ann. Pharm. Fr. 1995, 53, 135–137. [Google Scholar] [PubMed]
- Wolf, J. Mikro-Dünnschichtchromatographie Teufelskrallenwurzel. Pharm. Ztg. 1995, 140, 28. [Google Scholar]
- Chrubasik, S.; Sporer, F.; Wink, M. Zum Harpagosidgehalt verschiedener Trockenextraktpulver aus Harpagophytum procumbens. Complement. Med. Res. 1996, 3, 6–11. [Google Scholar] [CrossRef]
- Chrubasik, S.; Sporer, F.; Wink, M. Zum Wirkstoffgehalt in Teezubereitungen aus Harpagophytum procumbens. Complement. Med. Res. 1996, 3, 116–119. [Google Scholar] [CrossRef]
- Chrubasik, S.; Sporer, F.; Wink, M. Zum Wirkstoffgehalt in Arzneimitteln aus Harpagophytum procumbens. Complement. Med. Res. 1996, 3, 57–63. [Google Scholar] [CrossRef]
- Poukens-Renwart, P.; Tits, M.; Angenot, L. Quantitative densitometric evaluation of harpagoside in the secondary roots of Harpagophytum procumbens DC. J. Planar Chromatogr. 1996, 9, 199–202. [Google Scholar]
- Marotta, M.; Addabbo, I.; Kosasi, S. The quality control and stability testing of homeopathic preparations. Boll. Chim. Farm. 1998, 137, 439–441. [Google Scholar]
- Sporer, F.; Chrubasik, S. Präparate aus der Teufelskralle (Harpagophytum procumbens). Z. Phytother. 1999, 20, 335–336. [Google Scholar]
- Chrubasik, S.; Sporer, F.; Dillmann-Marschner, R.; Friedmann, A.; Wink, M. Physicochemical properties of harpagoside and its in vitro release from Harpagophytum procumbens extract tablets. Phytomedicine 2000, 6, 469–473. [Google Scholar] [CrossRef]
- Schneider, E.; Sanders, J.; Von Willert, D.J. Vermeidung von Verfälschungen der Teufelskralle Harpagophytum procumbens: Ein Beitrag zur pharmakognostischen Ökologie. Drogenreport 2001, 14, 12–16. [Google Scholar]
- Chrubasik, S.; Pollak, S.; Fiebich, B. Harpagophytum extracts. Clin. Pharm. 2002, 71, 104–105. [Google Scholar] [CrossRef] [PubMed]
- Gray, B.R. Retention of the Harpagoside Content in Dried Harpagophytum procumbens (Devil’s Claw) Root Through Controlled Drying and the Application of Near Infrared Spectroscopy (NIRS) as Rapid Method of Determination. Master’s Thesis, University of Stellenbosch, Stellenbosch, South Africa, 2003; p. 138. [Google Scholar]
- Baranska, M.; Schulz, H.; Siuda, R.; Strehle, M.A.; Rosch, P.; Popp, J.; Joubert, E.; Manley, M. Quality control of Harpagophytum procumbens and its related phytopharmaceutical products by means of NIR-FT-Raman spectroscopy. Biopolymers 2005, 77, 1–8. [Google Scholar] [CrossRef]
- Günther, M.; Schmidt, P.C. Comparison between HPLC and HPTLC-densitometry for the determination of harpagoside from Harpagophytum procumbens CO(2)-extracts. J. Pharm. Biomed. Anal. 2005, 37, 817–821. [Google Scholar] [CrossRef]
- Joubert, E.; Manley, M.; Gray, B.R.; Schulz, H. Rapid measurement and evaluation of the effect of drying conditions on harpagoside content in Harpagophytum procumbens (devil’s claw) root. J. Agric. Food Chem. 2005, 53, 3493–3502. [Google Scholar] [CrossRef]
- Schmidt, A.H. Fast HPLC for quality control of Harpagophytum procumbens by using a monolithic silica column: Method transfer from conventional particle-based silica column. J. Chromatogr. A 2005, 1073, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, A.H. Validation of a fast-HPLC method for the separation of iridoid glycosides to distinguish between the Harpagophytum species. J. Liq. Chromatogr. Relat. Technol. 2007, 28, 2339–2347. [Google Scholar] [CrossRef]
- Seger, C.; Godejohann, M.; Tseng, L.H.; Spraul, M.; Girtler, A.; Sturm, S.; Stuppner, H. LC-DAD-MS/SPE-NMR hyphenation. A tool for the analysis of pharmaceutically used plant extracts: Identification of isobaric iridoid glycoside regioisomers from Harpagophytum procumbens. Anal. Chem. 2005, 77, 878–885. [Google Scholar] [CrossRef]
- Zucchi, O.L.; Moreira, S.; de Jesus, E.F.; Neto, H.S.; Salvador, M.J. Characterization of hypoglycemiant plants by total reflection X-ray fluorescence spectrometry. Biol. Trace Elem. Res. 2005, 103, 277–290. [Google Scholar] [CrossRef]
- Arranz, I.; Sizoo, E.; van Egmond, H.; Kroeger, K.; Legarda, T.M.; Burdaspal, P.; Reif, K.; Stroka, J. Determination of aflatoxin B1 in medical herbs: Interlaboratory study. J. AOAC Int. 2006, 89, 595–605. [Google Scholar] [CrossRef] [PubMed]
- Colas, C.; Garcia, P.; Popot, M.A.; Bonnaire, Y.; Bouchonnet, S. Liquid chromatography/electrospray ionization mass spectrometric characterization of Harpagophytum in equine urine and plasma. Rapid Commun. Mass Spectrom. 2006, 20, 3257–3266. [Google Scholar] [CrossRef] [PubMed]
- Colas, C.; Bouchonnet, S.; Rogalewicz-Gilard, F.; Popot, M.A.; Ohanessian, G. Proton and sodium cation affinities of harpagide: A computational study. J. Phys. Chem. A 2006, 110, 7503–7508. [Google Scholar] [CrossRef]
- Schmidt, A.H.; Schiemann, U. Teufelskralle aus dem Drogeriemarkt oder aus der Apotheke? Dtsch. Apoth. Ztg. 2006, 146, 50–55. [Google Scholar]
- Spriano, D.; Krasniqi, B.; Strompen, T.; Tobler, M.; Meier, B. The drug-extract-ratio of aqueous/ethanolic Harpagophyti radix extracts has to be revised. Planta Med. 2006, 72, P_287. [Google Scholar] [CrossRef]
- Street, R.A.; Southway, C.; Stirk, W.A.; Van Staden, J. Determination of mineral elements and heavy metals in indigenous medicinal plants of KwaZulu-Natal. S. Afr. J. Bot. 2007, 73, 315. [Google Scholar] [CrossRef]
- Colas, C.; Popot, M.-A.; Garcia, P.; Bonnaire, Y.; Bouchonnet, S. Analysis of iridoids from Harpagophytum and eleutherosides from Eleutherococcus senticosus in horse urine. Biomed. Chromatogr. 2008, 22, 912–917. [Google Scholar] [CrossRef]
- Colas, C.; Garcia, P.; Popot, M.-A.; Bonnaire, Y.; Bouchonnet, S. Optimization of solid-phase extraction for the liquid chromatography—Mass spectrometry analysis of harpagoside, 8-para-coumaroyl harpagide, and harpagide in equine plasma and urine. J. Chromatogr. Sci. 2008, 46, 174–183. [Google Scholar] [CrossRef][Green Version]
- Wagner, S.; Urena, A.; Reich, E.; Merfort, I. Validated HPTLC methods for the determination of salicin in Salix sp. and of harpagoside in Harpagophytum procumbens. J. Pharm. Biomed. Anal. 2008, 48, 587–591. [Google Scholar] [CrossRef] [PubMed]
- Chigome, S.; Nindi, M.; Munkombwe, N. Quality control of Harpagophytum procumbens products using High Pressure Liquid Chromatography-Diode Array Detection (HPLC-DAD). Niger. J. Nat. Prod. Med. 2009, 13, 26–29. [Google Scholar] [CrossRef]
- Babili, F.E.; Fouraste, I.; Rougaignon, C.; Moulis, C.; Chatelain, C. Anatomical study of secondary tuberized roots of Harpagophytum procumbens DC and quantification of harpagoside by high-performance liquid chromatography method. Pharm. Mag. 2012, 8, 175–180. [Google Scholar] [CrossRef]
- Newmaster, S.G.; Grguric, M.; Shanmughanandhan, D.; Ramalingam, S.; Ragupathy, S. DNA barcoding detects contamination and substitution in North American herbal products. BMC Med. 2013, 11, 222. [Google Scholar] [CrossRef] [PubMed]
- Naidoo, Y.; Heneidak, S.; Bhatt, A.; Kasim, N.; Naidoo, G. Morphology, histochemistry, and ultrastructure of foliar mucilage-producing trichomes of Harpagophytum procumbens (Pedaliaceae). Turk. J. Bot. 2014, 38, 60–67. [Google Scholar] [CrossRef]
- Zhao, J.; Mncwangi, N.; Viljoen, A.; Wang, M.; Khan, I.A. Differentiation of Harpagophytum procumbens and H. zeyheri through NMR-based chemometric approach. Planta Med. 2014, 80, PPL17. [Google Scholar] [CrossRef]
- Kondamudi, N.; Turner, M.W.; McDougal, O.M. Harpagoside content in devil’s claw extracts. Nat. Prod. Commun. 2016, 11, 1215–1216. [Google Scholar] [CrossRef] [PubMed]
- Kriukova, A.; Vladymyrova, I. The definition of numeric indicators for the root of Harpagophytum procumbens. In Proceedings of the Topical Issues of New Drugs Development: XXIII International Scientific and Practical Conference of Young Scientists and Students, Kharkiv, Ukraine, 21 April 2016; p. 188. [Google Scholar]
- Baghdikian, B.; Filly, A.; Fabiano-Tixier, A.-S.; Petitcolas, E.; Mabrouki, F.; Chemat, F.; Ollivier, É. Extraction by solvent using microwave and ultrasound-assisted techniques followed by HPLC analysis of Harpagoside from Harpagophytum procumbens and comparison with conventional solvent extraction methods. C. R. Chim. 2016, 19, 692–698. [Google Scholar] [CrossRef]
- Diuzheva, A.; Carradori, S.; Andruch, V.; Locatelli, M.; De Luca, E.; Tiecco, M.; Germani, R.; Menghini, L.; Nocentini, A.; Gratteri, P.; et al. Use of innovative (micro)extraction techniques to characterise Harpagophytum procumbens root and its commercial food supplements. Phytochem. Anal. 2018, 29, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Kriukova, A.; Vladymyrova, I.; Tishakova, T. Rationale for choosing of extraction solvent for obtaining liquid extract from the roots of Harpagophytum procumbens DC. Scr. Sci. Pharm. 2017, 4, 37. [Google Scholar]
- Kriukova, A.; Vladymyrova, I. The GC-MS determination of chemical constituents from Harpagophytum procumbens DC roots. Technol. Transf. Innov. Solut. Med. 2017, 52–54. [Google Scholar] [CrossRef]
- Pretorius, E.; Van der Bank, M.; Viljoen, A.M. DNA Barcoding detects contamination and substitution in herbal products containing Harpagophytum spp. S. Afr. J. Bot. 2017, 109, 364–365. [Google Scholar] [CrossRef]
- Avato, P.; Argentieri, M.P. Quality assessment of commercial spagyric tinctures of Harpagophytum procumbens and their antioxidant properties. Molecules 2019, 24, 2251. [Google Scholar] [CrossRef] [PubMed]
- Nalluri, B.N.; Sujithkumar, S. Characterization and estimation of harpagoside in dried root extract and oral powder formulations of Harpagophytum procumbens by validated RP-HPLC-PDA method. J. Drug Deliv. Ther. 2019, 9, 38–46. [Google Scholar] [CrossRef]
- De Aragao Tannus, C.; de Souza Dias, F.; Santana, F.B.; Dos Santos, D.; Magalhaes, H.I.F.; de Souza Dias, F.; de Freitas Santos Junior, A. Multielement determination in medicinal plants and herbal medicines containing Cynara scolymus L., Harpagophytum procumbens DC, and Maytenus ilifolia (Mart.) ex Reiss from Brazil using ICP OES. Biol. Trace Elem. Res. 2020. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos Ribeiro, G.; de Assis Carneiro, A.; Martins, D.H.N.; Simeoni, L.A.; Silveira, D.; Magalhães, P.O.; Fonseca-Bazzo, Y.M. Determination of harpagoside in Harpagophytum procumbens DC tablet’s using analytical method by high performance liquid chromatography. Eclét. Quím. J. 2020, 45, 47–55. [Google Scholar] [CrossRef]
- Duband, F. Harpagophytum procumbens DC: Recherche d’une Activité Anti-Inflammatoire Aiguë. Ph.D. Thesis, UFR de Pharmacie, Clermont-Ferrand, France, 1986; p. 76. [Google Scholar]
- Plaizier-Vercammen, J.A.; Bruwier, C. Evaluation of excipients for direct compression of the spray-dried extract of Harpagophytum procumbens. Soc. Fr. Sci. Tech. Pharm. 1986, 2, 525–530. [Google Scholar]
- Günther, M.; Maus, M.; Wagner, K.G.; Schmidt, P.C. Hydrophilic solutes in modified carbon dioxide extraction-prediction of the extractability using molecular dynamic simulation. Eur. J. Pharm. Sci. 2005, 25, 321–329. [Google Scholar] [CrossRef]
- Piechota-Urbanska, M.; Kolodziejska, J.; Berner-Strzelczyk, A. Zastosowanie wyciągu z czarciego pazura w przeciwzapalnych preparatach aplikowanych na skórę, wytworzonych na bazie polimerów kwasu akrylowego. Polim. Med. 2009, 39, 9–15. [Google Scholar]
- Almajdoub, S.S. Polymer Coating of an Optimized Nano Lipid Carrier System of Harpagophytum procumbens Extract for Oral Delivery. Master’s Thesis, University of the Western Cape, Cape Town, South Africa, 2017; p. 182. [Google Scholar]
- Lopes, A.P.; Bagatela, B.S.; Lopes, I.P.; Gregorio, L.E.; Maistro, E.L.; Fonseca, F.L.A.; Perazzo, F.F. Production of gastro-resistant coated tablets prepared from the hydroethanolic standardized roots extract of Harpagophytum procumbens DC. Afr. J. Pharm. Pharmacol. 2017, 11, 491–500. [Google Scholar] [CrossRef]
- Villasenor, I.M. Bioactivities of iridoids. Anti. Inflamm. Anti. Allergy Agents Med. Chem. (Former. Curr. Med. Chem. Anti Inflamm. Anti Allergy Agents) 2007, 6, 307–314. [Google Scholar] [CrossRef]
- Serrano, A.; Ros, G.; Nieto, G. Bioactive compounds and extracts from traditional herbs and their potential anti-inflammatory health effects. Medicines 2018, 5, 76. [Google Scholar] [CrossRef] [PubMed]
- Fontaine, J.; Elchami, A.; Vanhaelen, M.; Vanhaelen Fastré, R. Activité biologique d’Harpagophytum procumbens DC. 2. Analyse pharmacologique des effets de l’harpagoside, l’harpagide et l’harpagogenine sur l’ileon isole de cobaye. J. Pharm. Belg. 1981, 36, 321–324. [Google Scholar]
- Benito, P.B.; Lanza, A.M.D.; Sen, A.M.S.; De Santos Galindez, J.; Matellano, L.F.; Gómez, A.S.; Martínez, M.J.A. Effects of some iridoids from plant origin on arachidonic acid metabolism in cellular systems. Planta Med. 2000, 66, 324–328. [Google Scholar] [CrossRef] [PubMed]
- Fiebich, B.L.; Heinrich, M.; Hiller, K.O.; Kammerer, N. Inhibition of TNF-alpha synthesis in LPS-stimulated primary human monocytes by Harpagophytum extract SteiHap 69. Phytomedicine 2001, 8, 28–30. [Google Scholar] [CrossRef]
- Loew, D.; Mollerfeld, J.; Schrodter, A.; Puttkammer, S.; Kaszkin, M. Investigations on the pharmacokinetic properties of Harpagophytum extracts and their effects on eicosanoid biosynthesis in vitro and ex vivo. Clin. Pharm. 2001, 69, 356–364. [Google Scholar] [CrossRef]
- Wahrendorf, M.S.; Sporer, F.; Wink, M. Anti-inflammation assays on Harpagophytum procumbens. Des Sources du Savoir aux Médicaments du Futur: Actes du 4e Congrès Européen d’Ethnopharmacologie. In Proceedings of the 4th European Congress on Ethnopharmocology, Metz, France, 11–13 May 2000; Fleurentin, J., Pelt, J., Mazars, G., Lejosne, J.C., Cabalion, P., Eds.; IRD: Metz, France, 2002; pp. 399–401, ISBN 2709915049. [Google Scholar]
- Jang, M.H.; Lim, S.; Han, S.M.; Park, H.J.; Shin, I.; Kim, J.W.; Kim, N.J.; Lee, J.S.; Kim, K.A.; Kim, C.J. Harpagophytum procumbens suppresses lipopolysaccharide-stimulated expressions of cyclooxygenase-2 and inducible nitric oxide synthase in fibroblast cell line L929. J. Pharm. Sci. 2003, 93, 367–371. [Google Scholar] [CrossRef] [PubMed]
- Schulze-Tanzil, G.; Hansen, C.; Shakibaei, M. Wirkung des Extraktes aus Harpagophytum procumbens DC auf Matrix-Metalloproteinasen in menschlichen Knorpelzellen in vitro. Arzneimittelforschung 2004, 54, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Chrubasik, J.E. Zur Knorpelprotektion von Harpagophytum procumbens DC: Histologische, Zellbiologische und Molekularbiologische Untersuchungen. Ph.D. Thesis, Justus-Liebig-Universität Gießen, Gießen, Germany, 2006; p. 135. [Google Scholar]
- Chrubasik, J.E.; Neumann, E.; Lindhorst, E.; Chrubasik, S.; Muller-Ladner, U. Evaluation of the chondroprotective effect of Harpagophytum procumbens. Med. Klin. 2006, 101, A90. [Google Scholar]
- Hadzhiyski, H.; Torda, T.; Chrubasik, S.; Lindhorst, E.; Raif, W. Impact of Harpagophytum procumbens on the urinary pyridinoline deoxypyridinoline ratio in experimental osteoarthritis. Focus Altern. Complement. Ther. 2006, 11, 13. [Google Scholar] [CrossRef]
- Günther, M.; Laufer, S.; Schmidt, P.C. High anti-inflammatory activity of harpagoside-enriched extracts obtained from solvent-modified super- and subcritical carbon dioxide extractions of the roots of Harpagophytum procumbens. Phytochem. Anal. 2006, 17, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.H.; Tran, V.H.; Duke, R.K.; Tan, S.; Chrubasik, S.; Roufogalis, B.D.; Duke, C.C. Harpagoside suppresses lipopolysaccharide-induced iNOS and COX-2 expression through inhibition of NF-kappa B activation. J. Ethnopharmacol. 2006, 104, 149–155. [Google Scholar] [CrossRef]
- Balthazar, L.v.; Eggenschwiler, J.; Rohrer, J.; Suter, A. Investigations on the antiinflammatory way of action of a Harpagophytum extract using microarray technology. Planta Med. 2009, 75, PJ158. [Google Scholar] [CrossRef]
- Ebrahim, N.; Uebel, R.A. Direct inhibition of cyclooxygenase-2 enzyme by an extract of Harpagophytum procumbens, harpagoside and harpagide. Afr. J. Pharm. Pharmacol. 2011, 5, 2209–2212. [Google Scholar] [CrossRef]
- Gyurkovska, V.; Alipieva, K.; Maciuk, A.; Dimitrova, P.; Ivanovska, N.; Haas, C.; Bley, T.; Georgiev, M. Anti-inflammatory activity of Devil’s claw in vitro systems and their active constituents. Food Chem. 2011, 125, 171–178. [Google Scholar] [CrossRef]
- Fiebich, B.L.; Munoz, E.; Rose, T.; Weiss, G.; McGregor, G.P. Molecular targets of the antiinflammatory Harpagophytum procumbens (Devil’s claw): Inhibition of TNFalpha and COX-2 gene expression by preventing activation of AP-1. Phytother. Res. 2012, 26, 806–811. [Google Scholar] [CrossRef] [PubMed]
- Ebrahim, N. Development and Characterization of a Transdermal Formula for an Extract of the Medicinal Plant Harpagophytum procumbens. Ph.D. Thesis, University of the Western Cape, Belville, South Africa, 2013; p. 199. [Google Scholar]
- Hostanska, K.; Melzer, J.; Rostock, M.; Suter, A.; Saller, R. Alteration of anti-inflammatory activity of Harpagophytum procumbens (Devil’s claw) extract after external metabolic activation with S9 mix. J. Pharm. Pharmacol. 2014, 66, 1606–1614. [Google Scholar] [CrossRef]
- Haseeb, A.; Leigh, D.; Haqqi, T.M. A small molecule harpagoside inhibits IL-1beta-induced expression of IL-6 by blocking the expression of C-FOS in primary human osteoarthritis chondrocytes. Osteoarthr. Cartil. 2015, 23, A155–A156. [Google Scholar] [CrossRef][Green Version]
- Haseeb, A.; Ansari, M.Y.; Haqqi, T.M. Harpagoside suppresses IL-6 expression in primary human osteoarthritis chondrocytes. J. Orthop. Res. 2017, 35, 311–320. [Google Scholar] [CrossRef]
- Kim, T.K.; Park, K.S. Inhibitory effects of harpagoside on TNF-alpha-induced pro-inflammatory adipokine expression through PPAR-gamma activation in 3T3-L1 adipocytes. Cytokine 2015, 76, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Schopohl, P.; Gruneberg, P.; Melzig, M.F. The influence of harpagoside and harpagide on TNFalpha-secretion and cell adhesion molecule mRNA-expression in IFNgamma/LPS-stimulated THP-1 cells. Fitoterapia 2016, 110, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Mariano, A.; Di Sotto, A.; Leopizzi, M.; Garzoli, S.; Di Maio, V.; Gulli, M.; Dalla Vedova, P.; Ammendola, S.; Scotto d’Abusco, A. Antiarthritic effects of a root extract from Harpagophytum procumbens DC: Novel insights into the molecular mechanisms and possible bioactive phytochemicals. Nutrients 2020, 12, 2545. [Google Scholar] [CrossRef]
- Eichler, O.; Koch, C. Über die antiphlogistische, analgetische und spasmolytische Wirksamkeit von Harpagosid, einem Glykosid aus der Wurzel von Harpagophytum procumbens DC. Drug Res. 1970, 20, 107–109. [Google Scholar]
- Anonymous. Prüfung von “Harpagophytum procumbens DC” auf Antiphlogistische Wirksamkeit am Rattenpfotenödem; International Bio Research, Mitteilung an Fa. Hagen: Freilassing, Germany, 1974. [Google Scholar]
- Anonymous. Subakute Studie an der Ratte zur Prüfung des Einflusses von Harpagosan-Extrakt auf Verschiedene Blutkomponente; International Bio Research, Mitteilung an Fa. Hagen: Freilassing, Germany, 1974. [Google Scholar]
- Erdös, A.; Fontaine, R.; Friehe, H.; Durand, R.; Pöppinghaus, T. Beitrag zur Pharmakologie und Toxikologie verschiedener Extrakte, sowie des Harpagosids aus Harpagophytum procumbens DC. Planta Med. 1978, 34, 97–108. [Google Scholar] [CrossRef]
- McLeod, D.W.; Revell, P.; Robinson, B.V. Investigations of Harpagophytum procumbens (Devil’s claw) in the treatment of experimental inflammation and arthritis in the rat. Br. J. Pharmacol. 1979, 66, 140P–141P. [Google Scholar]
- Manez, S.; Alcaraz, M.; Paya, M.; Rios, J.; Hancke, J. Selected extracts from medicinal plants as anti-inflammatory agents. Planta Med. 1990, 56, 656. [Google Scholar] [CrossRef]
- Lanhers, M.C.; Fleurentin, J.; Mortier, F.; Vinche, A.; Younos, C. Anti-inflammatory and analgesic effects of an aqueous extract of Harpagophytum procumbens. Planta Med. 1992, 58, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Jadot, G.; Lecomte, A. Activité anti-inflammatoire d’Harpagophytum procumbens DC. Lyon Méditerr. Méd. Méd. Sud. Est. 1992, 28, 833–835. [Google Scholar]
- Del Carmen Recio, M.; Giner, R.M.; Máñez, S.; Ríos, J.L. Structural considerations on the iridoids as anti-inflammatory agents. Planta Med. 1994, 60, 232–234. [Google Scholar] [CrossRef] [PubMed]
- Soulimani, R.; Younos, C.; Mortier, F.; Derrieu, C. The role of stomachal digestion on the pharmacological activity of plant extracts, using as an example extracts of Harpagophytum procumbens. Can. J. Physiol. Pharmacol. 1994, 72, 1532–1536. [Google Scholar] [CrossRef] [PubMed]
- Abe, S.; Ishibashi, H.; Masuo, K.; Tanaka, S.-I.; Yamaguchi, H. Suppression of carrageenan-induced edema by oral administration of extracts of Uncaria tomentosa and/or Harpagophytum procumbens. Oyo Yakuri Pharmacomet. 2002, 62, 27–32. [Google Scholar]
- Andersen, M.L.; Santos, E.H.; Seabra Mde, L.; da Silva, A.A.; Tufik, S. Evaluation of acute and chronic treatments with Harpagophytum procumbens on Freund’s adjuvant-induced arthritis in rats. J. Ethnopharmacol. 2004, 91, 325–330. [Google Scholar] [CrossRef]
- Mahomed, I.M.; Ojewole, J.A.O. Analgesic, antiinflammatory and antidiabetic properties of Harpagophytum procumbens DC (Pedaliaceae) secondary root aqueous extract. Phytother. Res. 2004, 18, 982–989. [Google Scholar] [CrossRef]
- Mahomed, I.M. Some Pharmacological Properties of Harpagophytum procumbens DC [Pedaliaceae] Secondary Root Extract. Ph.D. Thesis, University of KwaZulu-Natal, Durban, South Africa, 2004; p. 172. [Google Scholar]
- Ahmed, M.I.; Afifi, M.I.; Younos, I.H. Harpagophytum procumbens (Devil’s claw): A possible natural anti-inflammatory agent (an experimental study). IJPT 2005, 4, 54–63. [Google Scholar]
- Kundu, J.K.; Mossanda, K.S.; Na, H.K.; Surh, Y.J. Inhibitory effects of the extracts of Sutherlandia frutescens (L.) R. Br. and Harpagophytum procumbens DC. on phorbol ester-induced COX-2 expression in mouse skin: AP-1 and CREB as potential upstream targets. Cancer Lett. 2005, 218, 21–31. [Google Scholar] [CrossRef] [PubMed]
- Catelan, S.C.; Belentani, R.M.; Marques, L.C.; Silva, E.R.; Silva, M.A.; Caparroz-Assef, S.M.; Cuman, R.K.; Bersani-Amado, C.A. The role of adrenal corticosteroids in the anti-inflammatory effect of the whole extract of Harpagophytum procumbens in rats. Phytomedicine 2006, 13, 446–451. [Google Scholar] [CrossRef]
- Chrubasik, J.E.; Lindhorst, E.; Neumann, E.; Gerlach, U.; Faller-Marquardt, M.; Torda, T.; Muller-Ladner, U.; Chrubasik, S. Potential molecular basis of the chondroprotective effect of Harpagophytum procumbens. Phytomedicine 2006, 13, 598–600. [Google Scholar] [CrossRef] [PubMed]
- Uchida, S.; Hirai, K.; Hatanaka, J.; Hanato, J.; Umegaki, K.; Yamada, S. Antinociceptive effects of St. John’s wort, Harpagophytum procumbens extract and Grape seed proanthocyanidins extract in mice. Biol. Pharm. Bull. 2008, 31, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Wachsmuth, L.; Lindhorst, E.; Wrubel, S.; Hadzhiyski, H.; Hudelmaier, M.; Eckstein, F.; Chrubasik, S. Micro-morphometrical assessment of the effect of Harpagophytum procumbens extract on articular cartilage in rabbits with experimental osteoarthritis using magnetic resonance imaging. Phytother. Res. 2011, 25, 1133–1140. [Google Scholar] [CrossRef] [PubMed]
- Wrubel, S. Untersuchungen zur Wirkung von Harpagophytum procumbens auf Kniegelenksarthrose im Kaninchen unter Verwendung der Magentresonanztomographie. Ph.D. Thesis, Ludwig-Maximilians-Universität, München, Germany, 2014; p. 65. [Google Scholar]
- Bisinotto, R. Efeito Anti-Inflamatório do Extrato Etanólico da Harpagophytum procumbens Durante a Inflamação Intestinal De Camundongos Infectados Com Salmonella Enteritidis (ATCC13076). Master’s Thesis, Universidade Federal de São Carlos, São Carlos, Brazil, 2014; p. 100. [Google Scholar]
- Lim, D.W.; Kim, J.G.; Han, D.; Kim, Y.T. Analgesic effect of Harpagophytum procumbens on postoperative and neuropathic pain in rats. Molecules 2014, 19, 1060–1068. [Google Scholar] [CrossRef] [PubMed]
- Parenti, C.; Arico, G.; Chiechio, S.; Di Benedetto, G.; Parenti, R.; Scoto, G.M. Involvement of the heme-oxygenase pathway in the antiallodynic and antihyperalgesic activity of Harpagophytum procumbens in Rats. Molecules 2015, 20, 16758–16769. [Google Scholar] [CrossRef]
- Ucuncu, Y.; Celik, N.; Ozturk, C.; Turkoglu, M.; Cetin, N.; Kockara, N.; Sener, E.; Dundar, C.; Arslan, A.; Dogan, H.; et al. Chondroprotective effects of a new glucosamine combination in rats: Gene expression, biochemical and histopathological evaluation. Life Sci. 2015, 130, 31–37. [Google Scholar] [CrossRef]
- Parenti, C.; Arico, G.; Pennisi, M.; Venditti, A.; Scoto, G.M. Harpagophytum procumbens extract potentiates morphine antinociception in neuropathic rats. Nat. Prod. Res. 2016, 30, 1248–1255. [Google Scholar] [CrossRef]
- Radomska-Lesniewska, D.M.; Skopinska-Rozewska, E.; Demkow, U.; Jozwiak, J.; Sobiecka, M.; Balan, B.J. A natural herbal remedy modulates angiogenic activity of bronchoalveolar lavage cells from sarcoidosis patients. Cent. Eur. J. Immunol. 2016, 41, 25–34. [Google Scholar] [CrossRef]
- Tippler, B.; Syrovets, T.; Plaza, N.; Loew, D.; Simmet, T.H. Harpagophytum procumbens DC used in traditional medicine inhibits eicosanoid biosynthesis in human whole blood. Int. J. Tissue React. 1997, 19, 101. [Google Scholar]
- Tippler, B.; Syrovets, T.; Loew, D.; Simmet, T.H. Harpagophytum procumbens: Wirkung von Extrakten auf die Eicosanoidbiosynthese in lonophor A23187-stimuliertem menschlichem Vollblut. In Phytopharmaka II, Forschung und Klinische Anwendung; Loew, D., Rietbrock, N., Eds.; Steinkopff: Darmstadt, Germany, 1996; pp. 95–100. [Google Scholar]
- Anauate, M.C. Efeito Dos Extratos De Harpagohytum procumbens (Garra-do-Diabo) E Suas Frações Na Atividade da COX-1 e COX-2 e Na Produção De NO Em Sangue Total. Ph.D. Thesis, Universidade de São Paulo, São Paulo, Brazil, 2007; p. 98. [Google Scholar]
- Abdelouahab, N.; Heard, C.M. Effect of the major glycosides of Harpagophytum procumbens (Devil’s claw) on epidermal cyclooxygenase-2 (COX-2) in vitro. J. Nat. Prod. 2008, 71, 746–749. [Google Scholar] [CrossRef] [PubMed]
- Abdelouahab, N.; Heard, C.M. Dermal and transcutaneous delivery of the major glycoside constituents of Harpagophytum procumbens (Devil’s claw) in vitro. Planta Med. 2008, 74, 527–531. [Google Scholar] [CrossRef] [PubMed]
- Ouitas, N.A.; Heard, C.M. Topical Delivery and Effects of Harpagophytum procumbens. Ph.D. Thesis, Cardiff University, Cardiff, UK, 2009; p. 298. [Google Scholar]
- Ouitas, N.A.; Heard, C.M. A novel ex vivo skin model for the assessment of the potential transcutaneous anti-inflammatory effect of topically applied Harpagophytum procumbens extract. Int. J. Pharm. 2009, 376, 63–68. [Google Scholar] [CrossRef]
- Ouitas, N.A.; Heard, C.M. Estimation of the relative antiinflammatory efficacies of six commercial preparations of Harpagophytum procumbens (Devil’s claw). Phytother. Res. 2010, 24, 333–338. [Google Scholar] [CrossRef]
- Anauate, M.C.; Torres, L.M.; de Mello, S.B. Effect of isolated fractions of Harpagophytum procumbens DC (Devil’s claw) on COX-1, COX-2 activity and nitric oxide production on whole-blood assay. Phytother. Res. 2010, 24, 1365–1369. [Google Scholar] [CrossRef] [PubMed]
- Whitehouse, L.W.; Znamirowska, M.; Paul, C.J. Devil’s claw (Harpagophytum procumbens): No evidence for anti-inflammatory activity in the treatment of arthritic disease. Can. Med. Assoc. J. 1983, 129, 249–251. [Google Scholar]
- Na, H.K.; Mossanda, K.S.; Lee, J.Y.; Surh, Y.J. Inhibition of phorbol ester-induced COX-2 expression by some edible African plants. Biofactors 2004, 21, 149–153. [Google Scholar] [CrossRef]
- Inaba, K.; Murata, K.; Naruto, S.; Matsuda, H. Inhibitory effects of devil’s claw (secondary root of Harpagophytum procumbens) extract and harpagoside on cytokine production in mouse macrophages. J. Nat. Med. 2010, 64, 219–222. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, A.; Razmkhah, K.; Mehrnia, M.; Mohamadnia, A.; Sahebjamee, H.; Salehi, S.; Asl, E.A.; Tahmasebi, H.; Shandiz, S.A.S.; Davouodbeglou, F.; et al. Molecular docking and binding study of harpagoside and harpagide as novel anti-inflammatory and anti-analgesic compound from Harpagophytum procumbens based on their interactions with COX-2 enzyme. Asian Pac. J. Trop. Dis. 2016, 6, 227–231. [Google Scholar] [CrossRef]
- Locatelli, M.; Ferrante, C.; Carradori, S.; Secci, D.; Leporini, L.; Chiavaroli, A.; Leone, S.; Recinella, L.; Orlando, G.; Martinotti, S.; et al. Optimization of aqueous extraction and biological activity of Harpagophytum procumbens root on ex vivo rat colon inflammatory model. Phytother. Res. 2017, 31, 937–944. [Google Scholar] [CrossRef]
- Leporini, L.; Ferrante, C.; Recinella, L.; Orlando, G.; Chiavaroli, A.; Martinotti, S.; Carradori, S.; Locatelli, M.; Vecchiotti, G.; Menghini, L. Evaluation of protective effect of Harpagophytum procumbens DC. Ex Meisn. Root water extraction by microwave, biological activity on ex vivo rat colon inflammatory model and microscopic investigation. In Proceedings of the 112° Congresso della Società Botanica Italiana, IV International Plant Science Conference (IPSC), Parma, Italy, 20–23 September 2017; p. 1. [Google Scholar]
- Cholet, J.; Decombat, C.; Vareille-Delarbre, M.; Gainche, M.; Berry, A.; Ogéron, C.; Ripoche, I.; Delort, L.; Vermerie, M.; Fraisse, D. Comparison of the anti-inflammatory and immunomodulatory mechanisms of two medicinal herbs: Meadowsweet (Filipendula ulmaria) and Harpagophytum (Harpagophytum procumbens). Int. J. Plant Anim. Environ. Sci. 2019, 9, 145–163. [Google Scholar]
- Recinella, L.; Chiavaroli, A.; Ronci, M.; Menghini, L.; Brunetti, L.; Leone, S.; Tirillini, B.; Angelini, P.; Covino, S.; Venanzoni, R.; et al. Multidirectional pharma-toxicological study on Harpagophytum procumbens DC. Ex Meisn.: An IBD-focused investigation. Antioxidants 2020, 9, 168. [Google Scholar] [CrossRef] [PubMed]
- Grant, L.; McBean, D.E.; Fyfe, L.; Warnock, A.M. The inhibition of free radical generation by preparations of Harpagophytum procumbens in vitro. Phytother. Res. 2009, 23, 104–110. [Google Scholar] [CrossRef]
- Kaszkin, M.; Beck, K.F.; Koch, E.; Erdelmeier, C.; Kusch, S.; Pfeilschifter, J.; Loew, D. Downregulation of iNOS expression in rat mesangial cells by special extracts of Harpagophytum procumbens derives from harpagoside-dependent and independent effects. Phytomedicine 2004, 11, 585–595. [Google Scholar] [CrossRef]
- Bae, Y.H.; Cuong, T.D.; Hung, T.M.; Kim, J.A.; Woo, M.H.; Byeon, J.S.; Choi, J.S.; Min, B.S. Cholinesterase inhibitors from the roots of Harpagophytum procumbens. Arch. Pharm. Res. 2013, 37, 1124–1129. [Google Scholar] [CrossRef]
- Bhattacharya, A.; Bhattcharya, S.K. Anti-oxidant activity of Harpagophytum procumbens (Devil’s claw). Br. J. Phytother. 1998, 5, 68–71. [Google Scholar]
- Langmead, L.; Dawson, C.; Hawkins, C.; Banna, N.; Loo, S.; Rampton, D.S. Antioxidant effects of herbal therapies used by patients with inflammatory bowel disease: An in vitro study. Aliment Pharm. Ther. 2002, 16, 197–205. [Google Scholar] [CrossRef]
- Betancor-Fernandez, A.; Perez-Galvez, A.; Sies, H.; Stahl, W. Screening pharmaceutical preparations containing extracts of turmeric rhizome, artichoke leaf, devil’s claw root and garlic or salmon oil for antioxidant capacity. J. Pharm. Pharmacol. 2003, 55, 981–986. [Google Scholar] [CrossRef] [PubMed]
- Almeida, M.C.; Soares, S.F.; Abreu, P.R.; Jesus, L.M.; Brito, L.C.; Bernardo-Filho, M. Protective effect of an aqueous extract of Harpagophytum procumbens upon Escherichia coli strains submitted to the lethal action of stannous chloride. Cell. Mol. Biol. 2007, 53, OL923-927. [Google Scholar]
- Grant, L. The Putative Anti-Inflammatory and Analgesic Properties of Harpagophytum procumbens. Ph.D. Thesis, Queen Margaret University College, Edinburgh, UK, 2005; p. 350. [Google Scholar]
- Georgiev, M.; Alipieva, K.; Pashova, S.; Denev, P.; Angelova, M.; Kerns, G.; Bley, T. Antioxidant activity of devil’s claw cell biomass and its active constituents. Food Chem. 2010, 121, 967–972. [Google Scholar] [CrossRef]
- Georgiev, M.; Alipieva, K.; Orhan, I.E. Cholinesterases inhibitory and antioxidant activities of Harpagophytum procumbens from in vitro systems. Phytother. Res. 2011, 26, 313–316. [Google Scholar] [CrossRef]
- Schaffer, L.F.; Peroza, L.R.; Boligon, A.A.; Athayde, M.L.; Alves, S.H.; Fachinetto, R.; Wagner, C. Harpagophytum procumbens prevents oxidative stress and loss of cell viability in vitro. Neurochem. Res. 2013, 38, 2256–2267. [Google Scholar] [CrossRef]
- Schaffer, L.F. Efeito Do Harpagophytum procumbens Sobre Parâmetros De Estresse Oxidativo E a Viabilidade Celular In Vitro. Master’s Thesis, Universidade Federal De Santa Maria, Santa Maria, Brazil, 2013; p. 62. [Google Scholar]
- Muzila, M.; Rumpunen, K.; Wright, H.; Roberts, H.; Grant, M.; Nybom, H.; Sehic, J.; Ekholm, A.; Widen, C. Alteration of neutrophil reactive oxygen species production by extracts of Devil’s claw (Harpagophytum). Oxid. Med. Cell. Longev. 2016, 2016, 3841803. [Google Scholar] [CrossRef] [PubMed]
- Schaffer, L.F.; de Freitas, C.M.; Chiapinotto Ceretta, A.P.; Peroza, L.R.; de Moraes Reis, E.; Krum, B.N.; Busanello, A.; Boligon, A.A.; Sudati, J.H.; Fachinetto, R.; et al. Harpagophytum procumbens ethyl acetate fraction reduces fluphenazine-induced vacuous chewing movements and oxidative stress in rat brain. Neurochem. Res. 2016, 41, 1170–1184. [Google Scholar] [CrossRef]
- Mosca, F.; Marruchella, G.; Mariani, F.; Tiscar, P.G. Effetto dell’arpagoside sul burst respiratorio dei neutrofili di suino. In Proceedings of the XLIII Meeting Annuale SIPAS, Reggio Emilia, Italy, 16–17 March 2017; pp. 191–195. [Google Scholar]
- Ungerer, G.; Cui, J.; Ndam, T.; Bekemeier, M.; Song, H.; Li, R.; Siedhoff, H.R.; Yang, B.; Appenteng, M.K.; Greenlief, C.M.; et al. Harpagophytum procumbens extract ameliorates allodynia and modulates oxidative and antioxidant stress pathways in a rat model of spinal cord injury. Neuromol. Med. 2020, 22, 278–292. [Google Scholar] [CrossRef]
- Ncube, S.F.; McGaw, L.J.; Njoya, E.M.; Ndagurwa, H.G.T.; Mundy, P.G.; Sibanda, S. In Vitro antioxidant activity of crude extracts of Harpagophytum zeyheri and their anti-inflammatory and cytotoxicity activity compared with diclofenac. BMC Complement. Altern. Med. 2021, in press. [Google Scholar] [CrossRef]
- Mahomed, I.M.; Nsabimana, A.M.; Ojewole, J.A.O. Pharmacological effects of Harpagophytum procumbens DC [Pedaliaceae] secondary root aqueous extract on isolated gastro-intestinal tract muscles of the chick, guinea-pig and rabbit. Afr. J. Pharm. Pharmacol. 2005, 2, 31–45. [Google Scholar] [CrossRef]
- Guérin, J.C.; Réveillère, H.P. Activité antifongique d’extraits végétaux à usage thérapeutique. II. Étude de 40 extraits sur 9 souches fongiques. Ann. Pharm. Fr. 1985, 43, 77–81. [Google Scholar] [PubMed]
- Weckesser, S.; Engel, K.; Simon-Haarhaus, B.; Wittmer, A.; Pelz, K.; Schempp, C.M. Screening of plant extracts for antimicrobial activity against bacteria and yeasts with dermatological relevance. Phytomedicine 2007, 14, 508–516. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, S.R.P. Avaliação do efeito do extrato etanólico bruto de Harpagophytum procumbens em camundongos infectados com Toxocara canis. Ropositório Inst. UFSCar. 2012. Available online: https://repositorio.ufscar.br/handle/ufscar/7011 (accessed on 13 April 2021).
- Camillo, L.; Oliveira, S.R.P.; Ribeiro, R.D.; Rodolpho, J.M.A.; Caroccia, G.H.G.; Albuquerque, S.; Anibal, F.F. Efeito de bioterápico na eosinofilia durante a SLMV experimental. Rev. Ciênc. Farm. Básica Apl. 2014, 35, 701–708. [Google Scholar]
- Oliveira, S.R.P.; Rodolpho, J.M.A.; Dejani, N.D.; Souza, L.C.; Correia, R.O.; Neris, D.M.; Galvão, A.; de Matos, A.P.K.; Vieira, P.C.; Afonso, A.; et al. Harpagophytum procumbens modulates eosinophilic response during infection by Toxocara canis. Int. J. Recent Sci. Res. 2014, 5, 2008–2013. [Google Scholar]
- Correia, R. Ação Do Tratamento Com Mentha piperita L. e Harpagophytum procumbens Contra Schistosoma mansoni—In Vitro: Análise Proteômica. Ph.D. Thesis, Universidade Federal de São Carlos, São Carlos, Brazil, 2014; p. 122. [Google Scholar]
- Cock, I.E.; Bromley, A. Inhibition of bacterial triggers of selected autoimmune inflammatory diseases by Harpagophytum procumbens Burch. DC. Ex Meisn. fruit extracts. S. Afr. J. Bot. 2017, 100, 329. [Google Scholar] [CrossRef]
- Luigi, M. Etude des Propriétés Antimutagènes de l’Harpagophytum procumbens et de l’Harpagoside: Généralisation aux Plantes Anti-Inflammatoires. Ph.D. Thesis, Ecole Doctorale Sciences de l’Environnement, Aix-Marseille, France, 2014; p. 340. [Google Scholar]
- Luigi, M.; Baghdikian, B.; Orsière, T.; Pompili, J.; Mabrouki, F.; Ollivier, E.; Botta, A. Antimutagenic potential of harpagoside and Harpagophytum procumbens against 1-nitropyrene. Pharmacogn. Mag. 2015, 11, S29–S36. [Google Scholar] [CrossRef]
- Kim, J.Y.; Park, S.H.; Baek, J.M.; Erkhembaatar, M.; Kim, M.S.; Yoon, K.H.; Oh, J.; Lee, M.S. Harpagoside inhibits RANKL-induced osteoclastogenesis via Syk-Btk-PLCgamma2-Ca(2+) signaling pathway and prevents inflammation-mediated bone loss. J. Nat. Prod. 2015, 78, 2167–2174. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.J.; Kyung Kim, W.; Joo Park, H.; Cho, L.; Kim, M.R.; Kim, M.J.; Shin, J.S.; Ho Lee, J.; Ha, I.H.; Kook Lee, S. Anti-osteoporotic activity of harpagide by regulation of bone formation in osteoblast cell culture and ovariectomy-induced bone loss mouse models. J. Ethnopharmacol. 2016, 179, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.J.; Kim, W.K.; Oh, J.; Kim, M.R.; Shin, J.S.; Lee, J.; Ha, I.H.; Lee, S.K. Anti-osteoporotic activity of Harpagoside by upregulation of the BMP2 and Wnt signaling pathways in osteoblasts and suppression of differentiation in osteoclasts. J. Nat. Prod. 2017, 80, 434–442. [Google Scholar] [CrossRef]
- Vollmann, D. Zu Einigen Pharmakologischen Wirkungen von Zubereitungen des Harpagophytum procumbens DC. Ph.D. Thesis, Karl-Marx-Universität, Leipzig, Germany, 1965; p. 34. [Google Scholar]
- Circosta, C.; Occhiuto, F.; Ragusa, S.; Trovato, A.; Tumino, G.; Briguglio, F.; De Pasquale, A. A drug used in traditional medicine: Harpagophytum procumbens DC II. Cardiovascular activity. J. Ethnopharmacol. 1984, 11, 259–274. [Google Scholar] [CrossRef]
- De Pasquale, R.; Circosta, C.; Iauk, L.; Ragusa, S.; Busa, G. Effetti dell’ Harpagophytum procumbens DC e dell’arpagoside sulle aritmie sperimentali “in vitro”. Pharm. Mediterr. 1984, 15, 153. [Google Scholar]
- De Pasquale, R.; Busa, G.; Circosta, C.; Iauk, L.; Ragusa, S.; Ficarra, P.; Occhiuto, F. A drug used in traditional medicine: Harpagophytum procumbens DC. III. Effects on hyperkinetic ventricular arrhythmias by reperfusion. J. Ethnopharmacol. 1985, 13, 193–199. [Google Scholar] [CrossRef]
- Occhiuto, F.; Circosta, C.; Ragusa, S.; Ficarra, P.; Costa De Pasquale, R. A drug used in traditional medicine: Harpagophytum procumbens DC. IV. effects on some isolated muscle preparations. J. Ethnopharmacol. 1985, 13, 201–208. [Google Scholar] [CrossRef]
- Occhiuto, F.; De Pasquale, A. Electrophysiological and haemodynamic effects of some active principles of Harpagophytum procumbens DC. in the dog. Pharm. Res. 1990, 22, 72–73. [Google Scholar] [CrossRef]
- Mahomed, I.M.; Ojewole, J.A.O. Cardiovascular effects of Harpagophytum procumbens DC. Afr. J. Tradit. Complement. Altern. Med. 2004, 1, 30–44. [Google Scholar]
- Mahomed, I.M.; Ojewole, J.A.O. Anticonvulsant activity of Harpagophytum procumbens DC [Pedaliaceae] secondary root aqueous extract in mice. Brain Res. Bull. 2006, 69, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Ferrante, C.; Recinella, L.; Locatelli, M.; Guglielmi, P.; Secci, D.; Leporini, L.; Chiavaroli, A.; Leone, S.; Martinotti, S.; Brunetti, L.; et al. Protective effects induced by microwave-assisted aqueous Harpagophytum extract on rat cortex synaptosomes challenged with amyloid beta-peptide. Phytother. Res. 2017, 31, 1257–1264. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Zhang, H.; Xu, H.; Xue, R.; Zheng, Y.; Wu, T.; Lian, Y. Harpagoside rescues the memory impairments in chronic cerebral hypoperfusion rats by inhibiting PTEN activity. J. Alzheimers Dis. 2018, 63, 445–455. [Google Scholar] [CrossRef] [PubMed]
- Peruru, R.; Usha Rani, R.; Thatiparthi, J.; Sampathi, S.; Dodoala, S.; Prasad, K. Devil’s claw (Harpagophytum procumbens) ameliorates the neurobehavioral changes and neurotoxicity in female rats exposed to arsenic. Heliyon 2020, 6, e03921. [Google Scholar] [CrossRef]
- Prosinska, J.; Sawicka, T.; Drozd, J. Investigation of the thymomimetic activity of a selected phytopharmaceutical preparation-Reumaherb tablets by flow cytometry. Acta Pol. Pharm. 2002, 59, 265–274. [Google Scholar]
- Torres-Fuentes, C.; Theeuwes, W.F.; McMullen, M.K.; McMullen, A.K.; Dinan, T.G.; Cryan, J.F.; Schellekens, H. Devil’s claw to suppress appetite—Ghrelin receptor modulation potential of a Harpagophytum procumbens root extract. PLoS ONE 2014, 9, e103118. [Google Scholar] [CrossRef] [PubMed]
- Saleh, S.; Jahromi, H.K.; Sarikhani, Y.; Jahromi, Z.K.; Dowlatkhah, H. The effects of hydroalcoholic extract of devil’s claw on serum levels of obestatin and body weight in male rats. J. Glob. Pharma Technol. 2016, 12, 40–43. [Google Scholar]
- Anonymous. Der Einfluß von Harpagosan (Harpagophytum procumbens DC) auf die Blei-Einlagerung in Rattenorganen; International Bio Research, Mitteilung an Fa. Hagen: Freilassing, Germany, 1975. [Google Scholar]
- Chrubasik, S.; Wink, M. Zur pharmakologischen Wirkung der Teufelskralle (Harpagophytum procumbens). Complement. Med. Res. 1995, 2, 323–325. [Google Scholar] [CrossRef]
- Loew, D.; Kaszkin, M. Experimentell- und klinisch-pharmakologische Modelle zum Nachweis der analgetischen und antiphlogistischen Wirkungen von Teufelskralle. Ärztez. Nat. 2002, 43, 576–585. [Google Scholar]
- Fleurentin, J.; Mortier, F. Entzündungshemmende und analgetische Wirkungen von Harpagophytum procumbens und H. zeyheri. In Rheumatherapie mit Phytopharmaka; Chrubasik, S., Wink, M., Eds.; Hippokrates: Stuttgart, Germany, 1997; pp. 68–76. [Google Scholar]
- Budzinski, J.W.; Foster, B.C.; Vandenhoek, S.; Arnason, J.T. An in vitro evaluation of human cytochrome P450 3A4 inhibition by selected commercial herbal extracts and tinctures. Phytomedicine 2000, 7, 273–282. [Google Scholar] [CrossRef]
- Puttkammer, S. Untersuchungen zur Pharmakokinetik des Harpagophytum-Spezialextraktes HF 8858 und Einfluß auf Leukotrien C4-Biosynthese Sowie die Thromboxan B2-Freisetzung. Ph.D. Thesis, Johann Wolfgang Goethe-Universität Frankfurt/M., Frankfurt, Germany, 2000; p. 45. [Google Scholar]
- Hilgendorf, C.; Döppenschmitt, S. Popular herbal drugs: Screening of potential CYP inhibition/activation based metabolic interactions. Int. J. Clin. Pharm. 2003, 41, 537. [Google Scholar]
- Unger, M.; Frank, A. Simultaneous determination of the inhibitory potency of herbal extracts on the activity of six major cytochrome P450 enzymes using liquid chromatography/mass spectrometry and automated online extraction. Rapid Commun. Mass Spectrom. 2004, 18, 2273–2281. [Google Scholar] [CrossRef]
- Romiti, N.; Tramonti, G.; Corti, A.; Chieli, E. Effects of Devil’s claw (Harpagophytum procumbens) on the multidrug transporter ABCB1/P-glycoprotein. Phytomedicine 2009, 16, 1095–1100. [Google Scholar] [CrossRef] [PubMed]
- Modarai, M.; Suter, A.; Kortenkamp, A.; Heinrich, M. The interaction potential of herbal medicinal products: A luminescence-based screening platform assessing effects on cytochrome P450 and its use with devil’s claw (Harpagophyti radix) preparations. J. Pharm. Pharmacol. 2011, 63, 429–438. [Google Scholar] [CrossRef]
- Anton, R. Réflexions sur quelques nouvelles acquisitions en phytothérapie. J. Pharm. Belg. 1987, 42, 138–151. [Google Scholar]
- Möse, J.R. Untersuchung der Toxischen Wirkung von Harpago-Tee auf Zellen und Dessen Verhalten in Einem Orientierenden Versuch am Tumor-Tier; Hygiene-Institut, Universität Graz. Mitteilung an Fa. Hagen: Freilassing, Germany, 1969. [Google Scholar]
- Grünewald, K. Erfahrungen mit Harpagosan bei Hypercholesterinämie und Hyperurikämie. Arch. Arzneither. 1978, 1, 73–76. [Google Scholar]
- Anonymous. Akute Toxizitätsprüfung von Harpagophytum D2 nach Intravenöser Applikation an der Ratte; International Bio Research, Mitteilung an Fa. Hagen: Freilassing, Germany, 1974. [Google Scholar]
- Anonymous. Akute Toxizitätsprüfung von Harpagophytum procumbens DC nach Oraler Applikation bei der Ratte; International Bio Research, Mitteilung an Fa. Hagen: Freilassing, Germany, 1975. [Google Scholar]
- Ibrahim, K.E.; Al–Ashban, R.M.; El–Sammani, S.A. Toxicity studies on devil’s claw herbal medicine. Res. J. Pharmacol. 2010, 4, 69–73. [Google Scholar] [CrossRef]
- Al-Harbi, N.O.; Al-Ashban, R.M.; Shah, A.H. Toxicity studies on Harpagophytum procumbens (Devil’s claw) capsules in mice. J. Med. Plants Res. 2013, 7, 3089–3097. [Google Scholar] [CrossRef]
- Allard, T.; Wenner, T.; Greten, H.J.; Efferth, T. Mechanisms of herb-induced nephrotoxicity. Curr. Med. Chem. 2013, 20, 2812–2819. [Google Scholar] [CrossRef] [PubMed]
- Joshi, K.; Parrish, A.; Grunz-Borgmann, E.A.; Gerkovich, M.; Folk, W.R. Toxicology studies of aqueous-alcohol extracts of Harpagophytum procumbens subsp. procumbens (Burch.) DC. Ex Meisn. (Pedaliaceae) in female and male rats. BMC Complement. Altern. Med. 2020, 20, 9. [Google Scholar] [CrossRef]
- Mahomed, I.M.; Ojewole, J.A.O. Oxytocin-like effect of Harpagophytum procumbens DC [Pedaliaceae] secondary root aqueous extract on rat isolated uterus. Afr. J. Tradit. Complement. Altern. Med. 2006, 3, 82–89. [Google Scholar]
- Mahomed, I.M.; Ojewole, J.A. Uterotonic effect of Harpagophytum procumbens DC (Pedaliaceae) secondary root aqueous extract on rat isolated uterine horns. J. Smooth Muscle Res. 2009, 45, 231–239. [Google Scholar] [CrossRef]
- Pearson, W. Reproductive Toxicity Study of an Herbal Product (Nasprin) in Sprague Dawley Rats; Nutraceutical Alliance: Burlington, ON, Canada, 2012. [Google Scholar]
- Davari, S.A. Teratogenic effects of Harpagophytum procumbens ethanolic extract in mice and fetuses. Zahedan J. Res. Med. Sci. 2016, 18, e3481. [Google Scholar] [CrossRef]
- Chrubasik, S.; Ziegler, R. Wirkstoffgehalt in Arzneimitteln aus Harpagophytum procumbens und klinische Wirksamkeit von Harpagophytum-Trockenextrakt. In Phytopharmaka II, Forschung und Klinische Anwendung; Loew, D., Rietbrock, N., Eds.; Steinkopff: Darmstadt, Germany, 1996; pp. 101–114. [Google Scholar]
- Brendler, T.; Grünwald, J.; Ulbricht, C.; Basch, E. Devil’s claw (Harpagophytum procumbens DC): An evidence-based systematic review by the Natural Standard Research Collaboration. J. Herb. Pharm. 2006, 6, 89–126. [Google Scholar] [CrossRef]
- Büechi, S.; Wegener, T. Harpagophyti radix (Teufelskrallenwurzel). Phytotherapie 2002, 4, 28–33. [Google Scholar]
- Chrubasik, S.; Conradt, C. Audiatur et al.tera pars: Teufelskralle in der Diskussion. Z. Phytother. 2002, 23, 84–86. [Google Scholar]
- Chrubasik, S.; Conradt, C.; Black, A. The quality of clinical trials with Harpagophytum procumbens. Phytomedicine 2003, 10, 613–623. [Google Scholar] [CrossRef][Green Version]
- Chrubasik, S.; Conradt, C.; Roufogalis, B.D. Effectiveness of Harpagophytum extracts and clinical efficacy. Phytother. Res. 2004, 18, 187–189. [Google Scholar] [CrossRef] [PubMed]
- Chrubasik, S.; Eisenberg, E. Treatment of rheumatic pain with kampo medicine in Europe. Part 1. Harpagophytum procumbens. Pain Clin. 1999, 11, 171–178. [Google Scholar]
- Chrubasik, S.; Pollak, S. Teufelskrallenwurzelextrakt bei schmerzhafter Arthrose und Rückenschmerzen. Z. Phytother. 2002, 23, 210–215. [Google Scholar]
- Chrubasik, S.; Shvartzman, P. Rheumatic pain treatment with devil’s claw (Harpagophyti radix). 1999, 1/99. Available online: http://www.iaam.nl/coherence/msaima/199-3.html (accessed on 14 April 2021).
- Denner, S.S. A review of the efficacy and safety of devil’s claw for pain associated with degenerative musculoskeletal diseases, rheumatoid, and osteoarthritis. Holist. Nurse Pr. 2007, 21, 203–207. [Google Scholar] [CrossRef]
- Gäbler, H. Harpagophytum procumbens. Homöopath. Mon. 1972, 97, 123–126. [Google Scholar]
- Gagnier, J.J.; Chrubasik, S.; Manheimer, E. Harpgophytum procumbens for osteoarthritis and low back pain: A systematic review. BMC Complement. Altern. Med. 2004, 4, 13. [Google Scholar] [CrossRef] [PubMed]
- Gagnier, J.J.; Chrubasik, S.; Manheimer, E. Correction: Harpagophytum procumbens for osteoarthritis and low back pain: A systematic review. BMC Complement. Altern. Med. 2005, 5, 1. [Google Scholar] [CrossRef]
- Grant, L.; McBean, D.E.; Fyfe, L.; Warnock, A.M. A review of the biological and potential therapeutic actions of Harpagophytum procumbens. Phytother. Res. 2007, 21, 199–209. [Google Scholar] [CrossRef]
- Loew, D. Harpagophytum procumbens DC. Eine Übersicht zur Pharmakologie und WIrksamkeit. Erfahrungsheilkunde 1995, 2, 74–79. [Google Scholar]
- Savustyanenko, A.V. Эффективнoсть экстракта мартинии душистoй (сустамар) при oстеoартритах, пoясничнoй бoли и фибрoмиалгии: Обзoр исследoваний. Pain Jt. Spine 2014, 3, 45–53. [Google Scholar] [CrossRef]
- Tarleton, A. Devil’s claw for low back pain. Herbalgram 1997, 41, 20. [Google Scholar]
- Wegener, T. Degenerative Erkrankungen des Bewegungsapparates—Übersicht zu aktuellen klinischen Studien mit Extrakten aus der Teufelskralle (Harpagophyti radix). Wien. Med. Wochenschr. 2002, 152, 389–392. [Google Scholar] [CrossRef]
- Ameye, L.G.; Chee, W.S. Osteoarthritis and nutrition. From nutraceuticals to functional foods: A systematic review of the scientific evidence. Arthritis Res. 2006, 8, R127. [Google Scholar] [CrossRef]
- Amling, R. Phytotherapeutika in der Neurologie. Z. Phytother. 1991, 12, 9–14. [Google Scholar]
- Arndt, D.; Bobermien, K.; Heyer, H.; Hinken, B. Alternative Therapieoptionen bei Endometriose. Gynäkologe 2007, 40, 553–558. [Google Scholar] [CrossRef]
- Belova, K.Y.; Nazarova, A.V. Стратегия лечения oстеoартрита у мультимoрбидных пациентoв: баланс эффективнoсти и безoпаснoсти при выбoре лекарственнoй терапии. Med. Counc. 2020, 11, 164–176. [Google Scholar] [CrossRef]
- Brien, S.; Lewith, G.T.; McGregor, G. Devil’s claw (Harpagophytum procumbens) as a treatment for osteoarthritis: A review of efficacy and safety. J. Altern. Complement. Med. 2006, 12, 981–993. [Google Scholar] [CrossRef]
- Chrubasik, C.; Black, A.; Muller-Ladner, U.; Chrubasik, S. Impact of herbal medicines on physical impairment. Phytomedicine 2008, 15, 536–539. [Google Scholar] [CrossRef]
- Chrubasik, J.E.; Roufogalis, B.D.; Chrubasik, S. Evidence of effectiveness of herbal antiinflammatory drugs in the treatment of painful osteoarthritis and chronic low back pain. Phytother. Res. 2007, 21, 675–683. [Google Scholar] [CrossRef] [PubMed]
- Chrubasik, S. Osteoarthritis: Pharmacology and clinical indications of selected botanicals. Pain Clin. 2004, 16, 1–16. [Google Scholar] [CrossRef]
- Chrubasik, S.; Neumann, E.; Müller-Ladner, U. Zur antientzündlichen Wirksamkeit von Arzneimitteln aus der Teufelskralle. Z. Phytother. 2009, 30, 222–226. [Google Scholar] [CrossRef]
- Chrubasik, S.; Pollak, S. Schmerzbehandlung mit pflanzlichen Antirheumatika. Wien. Med. Wochenschr. 2002, 152, 198–203. [Google Scholar] [CrossRef]
- Chrubasik, S.; Wink, M. Traditional herbal therapy for the treatment of rheumatic pain: Preparations from devil’s claw and stinging nettle. Pain Dig. 1998, 8, 94–101. [Google Scholar]
- Chrubasik, S.; Wink, M. Treatment of osteoarthritic pain with herbal drugs. In Proceedings of the 8th World Congress: The Pain Clinic, Tenerife, Spain, 6–10 May 1998; pp. 507–514. [Google Scholar]
- Chrubasik-Hausmann, S. Phytotherapie bei Arthrose. Erfahrungsheilkunde 2015, 64, 22–27. [Google Scholar] [CrossRef]
- Corciova, A.; Matei, D.; Ivanescu, B. Medicinal herbs as possible sources of anti-inflammatory products. Balneo Res. J. 2017, 8, 231–241. [Google Scholar] [CrossRef]
- Corp, N.; Pendry, B. The role of Western herbal medicine in the treatment of gout. J. Herb. Med. 2013, 3, 157–170. [Google Scholar] [CrossRef]
- Darshan, S.; Doreswamy, R. Patented antiinflammatory plant drug development from traditional medicine. Phytother. Res. 2004, 18, 343–357. [Google Scholar] [CrossRef] [PubMed]
- Del Grossi Moura, M.; Lopes, L.C.; Biavatti, M.W.; Kennedy, S.A.; de Oliveira, E.S.M.C.; Silva, M.T.; de Cassia Bergamaschi, C. Oral herbal medicines marketed in Brazil for the treatment of osteoarthritis: A systematic review and meta-analysis. Phytother. Res. 2017, 31, 1676–1685. [Google Scholar] [CrossRef]
- Deyo, R.A. Conservative therapy for low back pain. Distinguishing useful from useless therapy. JAMA 1983, 250, 1057–1062. [Google Scholar] [CrossRef] [PubMed]
- Di Lorenzo, C.; dell’Agli, M.; Badea, M.; Dima, L.; Colombo, E.; Sangiovanni, E.; Restani, P.; Bosisio, E. Plant food supplements with anti-inflammatory properties: A systematic review (II). Crit. Rev. Food Sci. Nutr. 2013, 53, 507–516. [Google Scholar] [CrossRef]
- Dragos, D.; Gilca, M.; Gaman, L.; Vlad, A.; Iosif, L.; Stoian, I.; Lupescu, O. Phytomedicine in joint disorders. Nutrients 2017, 9, 70. [Google Scholar] [CrossRef]
- Ernst, E.; Chrubasik, S. Phyto-anti-inflammatories. A systematic review of randomized, placebo-controlled, double-blind trials. Rheum. Dis. Clin. N. Am. 2000, 26, 13–27. [Google Scholar] [CrossRef]
- Gagnier, J.J. Evidence based review of natural health products for non-specific low back pain. Open Pain J. 2010, 3. [Google Scholar] [CrossRef]
- Gagnier, J.J.; van Tulder, M.; Berman, B.; Bombardier, C. Herbal medicine for low back pain. Cochrane Database Syst. Rev. 2006, CD004504. [Google Scholar] [CrossRef]
- Gagnier, J.J.; van Tulder, M.W.; Berman, B.; Bombardier, C. Herbal medicine for low back pain: A Cochrane review. Spine 2007, 32, 82–92. [Google Scholar] [CrossRef] [PubMed]
- Gregory, P.J.; Sperry, M.; Wilson, A.F. Dietary supplements for osteoarthritis. Am. Fam. Physician 2008, 77, 177–184. [Google Scholar] [PubMed]
- Halliwell, B. Drug antioxidant effects. A basis for drug selection? Drugs 1991, 42, 569–605. [Google Scholar] [CrossRef]
- Long, L.; Soeken, K.; Ernst, E. Herbal medicines for the treatment of osteoarthritis: A systematic review. Rheumatology 2001, 40, 779–793. [Google Scholar] [CrossRef]
- Lopez, H.L. Nutritional interventions to prevent and treat osteoarthritis. Part II: Focus on micronutrients and supportive nutraceuticals. PMR 2012, 4, S155–S168. [Google Scholar] [CrossRef]
- Marlowe, D. Complementary and alternative medicine treatments for low back pain. Prim. Care 2012, 39, 533–546. [Google Scholar] [CrossRef]
- Oltean, H.; Robbins, C.; van Tulder, M.W.; Berman, B.M.; Bombardier, C.; Gagnier, J.J. Herbal medicine for low-back pain. Cochrane Database Syst. Rev. 2014, CD004504. [Google Scholar] [CrossRef] [PubMed]
- Park, K.S. A systematic review on anti-inflammatory activity of harpagoside. J. Biochem. Mol. Biol. Res. 2016, 2, 166–169. [Google Scholar] [CrossRef]
- Roth-Utzschneider, S. Erfolgreiche Rheumatherapie mit Teufelskralle und Weidenrinde. Ärztez. Nat. 2006, 47, 488. [Google Scholar]
- Rubinstein, S.M.; van Middelkoop, M.; Kuijpers, T.; Ostelo, R.; Verhagen, A.P.; de Boer, M.R.; Koes, B.W.; van Tulder, M.W. A systematic review on the effectiveness of complementary and alternative medicine for chronic non-specific low-back pain. Eur. Spine J. 2010, 19, 1213–1228. [Google Scholar] [CrossRef] [PubMed]
- Sanders, M.; Grundmann, O. The use of glucosamine, devil’s claw (Harpagophytum procumbens), and acupuncture as complementary and alternative treatments for osteoarthritis. Altern. Med. Rev. 2011, 16, 228. [Google Scholar]
- Schmidt, S. Phytotherapie beim rheumatischen Formenkreis. Heilkunst 1973, 86, 18–20. [Google Scholar]
- Setty, A.R.; Sigal, L.H. Herbal medications commonly used in the practice of rheumatology: Mechanisms of action, efficacy, and side effects. Semin. Arthritis Rheum. 2005, 34, 773–784. [Google Scholar] [CrossRef]
- Soeken, K.L. Selected CAM therapies for arthritis-related pain: The evidence from systematic reviews. Clin. J. Pain. 2004, 20, 13–18. [Google Scholar] [CrossRef]
- Spelman, K.; Burns, J.; Nichols, D.; Winters, N.; Ottersberg, S.; Tenborg, M. Modulation of cytokine expression by traditional medicines: A review of herbal immunomodulators. Altern. Med. Rev. 2006, 11, 128–150. [Google Scholar]
- Teut, M.; Warning, A. Knochenmetastasen bei Mamma-Karzinom. Komplementmed. 2006, 13, 46–48. [Google Scholar] [CrossRef]
- Viljoen, A.; Mncwangi, N.; Vermaak, I. Anti-inflammatory iridoids of botanical origin. Curr. Med. Chem. 2012, 19, 2104–2127. [Google Scholar] [CrossRef]
- Zimmermann, W. Pflanzliche Bitterstoffe in der Gastroenterologie. ZFA 1976, 54, 1178–1184. [Google Scholar]
- Zimmermann, W. Interne Phytotherapie der rheumatischen und arthrotischen Erkrankungen. Herba Pol. 1979, 4, 333–342. [Google Scholar]
- Zimmermann, W. Bitterstoffe. Kassenarzt 1985, 8, 44, 47–48. [Google Scholar]
- Zimmermann, W. Der obere Dünndarm. Eine Phytotherapiestudie. Therapiewoche 1985, 35, 1592–1602. [Google Scholar]
- Gagnier, J.J.; Oltean, H.; van Tulder, M.W.; Berman, B.M.; Bombardier, C.; Robbins, C.B. Herbal medicine for low back pain: A cochrane review. Spine 2016, 41, 116–133. [Google Scholar] [CrossRef] [PubMed]
- Anonymous. Teufelskralle zur Therapie der Osteoarthritis. Dtsch. Apoth. Ztg. 2001, 141, 1–2. [Google Scholar]
- Baillard, O. Intérêt de l’Harpagophytum procumbens dans les Affections Rhumatismales. Ph.D. Thesis, Université François Rabelais, Tours, France, 2009; p. 86. [Google Scholar]
- Beer, A.M. Teufelskralle und Gymnastik für Knie und Schulter. MMW Med. 2014, 156, 24. [Google Scholar]
- Chrubasik, S. Efficacy and safety of Harpagophytum and Salix extract preparations. In Des Sources du Savoir aux Médicaments du Futur; Fleurentin, J., Pelt, J., Mazars, G., Eds.; IRD Éditions: Marseille, France, 2002; ISBN 2709915049. [Google Scholar]
- Chrubasik, S. Weidenrinde und Teufelskralle bei chronischen Gelenk- und Rückenschmerzen: Evidenzlage favorisiert pflanzliche Präparate. Schweiz. Z. Ganzheitsmed. Swiss J. Integr. Med. 2004, 16, 355–359. [Google Scholar]
- Chrubasik, S. Zur Evidenz der Wirksamkeit Pflanzlicher Entzündungshemmer bei Arthrose und Rückenschmerzen. Ph.D. Thesis, Klinikum der Albert-Ludwigs-Universität, Freiburg im Breisgau, Germany, 2013; p. 47. [Google Scholar]
- Schmidt, S. Therapeutische Wirkung der Teufelskralle. ZFA 1983, 56, 1442. [Google Scholar]
- Vanhaelen, M. L’activite de Harpagophytum procumbens et de Glycyrrhiza glabra; Toxicite de drogues contenant des alcaloides derives de la pyrrolizidine. Farm. Tijdschr. Belg. 1986, 63, 31–43. [Google Scholar]
- Wenzel, P.; Wegener, T. Teufelskralle-ein pflanzliches Antirheumatikum. Dtsch. Apoth. Ztg. 1995, 135, 15–27. [Google Scholar]
- Zimmermann, W. Harpagophyturn Ampullen D2 der Fa. Hagen. Klinisches Gutachten; Schriftliche Mitteilung an Fa. Hagen: Freilassing, Germany, 1972. [Google Scholar]
- Belaiche, P. Un cas de chemosis grave traité avec succès par le nébulisat aqueux de radix Harpagophytum procumbens. Phytotherapy 1983, 5, 15–16. [Google Scholar]
- Belaiche, P. Action du nebulisat aqueux de radix Harpagophytum procumbens dans 17 cas de fièvre familiale méditerranéenne (maladie périodique). Phytotherapy 1983, 8, 5–11. [Google Scholar]
- Wilson, K.S. Regression of follicular lymphoma with Devil’s claw: Coincidence or causation? Curr. Oncol. 2009, 16, 67–70. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Beham, A. Die Anwendung der Harpagophytum-Wurzel bei rheumatischen Erkrankungen. Allg. Homöopath. Ztg. 1971, 216, 204–208. [Google Scholar] [CrossRef]
- Brantner, F. Praktische Phytotherapie. Phys. Med. Rehabil. 1972, 13, 87–88. [Google Scholar]
- Zimmermann, W. Erfahrungen mit Harpagophytum. Phys. Med. Rehabil. 1977, 18, 317–319. [Google Scholar]
- Vanhaelen, M. Mise a jour des recherches relatives a harpagophvtum procumbens. Phytotherapy 1985, 16, 19–24. [Google Scholar]
- Stübler, M. Die Behandlung chronischer Gelenkerkrankungen mit Harpagophytum. Allg. Homöopath. Ztg. 1987, 232, 60–62. [Google Scholar] [CrossRef]
- Stübler, M. Harpagophytum procumbens in rheumatoid arthritis. Br. Homoeopath. J. 1987, 76, 161. [Google Scholar] [CrossRef]
- Müller, B.; Deitelhoff, P.; Petrowicz, O. Harpagophytum procumbens ist effizient bei degenerativen Erkrankungen des Bewegungsapparates. Nat. Med. 2000, 21–29. [Google Scholar]
- Thanner, J.; Kohlmann, T.; Kunzel, O.; Chrubasik, S. Retrospective evaluation of biopsychosocial determinants and treatment response in patients receiving devil’s claw extract (doloteffin). Phytother. Res. 2009, 23, 742–744. [Google Scholar] [CrossRef] [PubMed]
- Hoppe, H. Einfluß der Droge Harpagophytum procumbens DC (auch Teufelskralle genannt) auf Diabetes mellitus mit Fettstoffwechselstörungen. Erfahrungsheilkunde 1974, 7, 230–233. [Google Scholar]
- Grahame, R.; Robinson, B.V. Devils’s claw (Harpagophytum procumbens): Pharmacological and clinical studies. Ann. Rheum. Dis. 1981, 40, 632. [Google Scholar] [CrossRef]
- Belaiche, P. Etude clinique de 630 cas d’artrose traites par le nebulisat aqueux d’Harpagophytum procumbens (Radix). Phytotherapy 1982, 1, 22–28. [Google Scholar]
- Kröner, W. Wirksamkeit der Homöopathischen Arzneimittel “Formica rufa” und “Harpagophytum procumbens” bei der Gonarthrose: Eine Kontrollierte Studie bei Niedergelassenen Ärzten. Ph.D. Thesis, Universität Ulm, Ulm, Germany, 1991; p. 82. [Google Scholar]
- Moussard, C.; Alber, D.; Toubin, M.M.; Thevenon, N.; Henry, J.C. A drug used in traditional medicine, Harpagophytum procumbens: No evidence for NSAID-like effect on whole blood eicosanoid production in human. Prostaglandins Leukot. Essent. Fat. Acids 1992, 46, 283–286. [Google Scholar] [CrossRef]
- Chrubasik, S.; Schmidt, A.; Junck, H.; Pfisterer, M. Wirksamkeit und Wirtschaftlichkeit von Teufelskrallenwurzelextrakt bei Rückenschmerzen: Erste Ergebnisse einer therapeutischen Kohortenstudie. Complement. Med. Res. 1997, 4, 332–336. [Google Scholar] [CrossRef]
- Pinget, M.; Lecomte, A. Die Wirkung der “Harpagophytum Arkocaps” bei degenerativem Rheuma. Naturheilpraxis 1997, 50, 267–269. [Google Scholar]
- Schwarz, G.; Hämmerle, H.D.; Benvenuti, E. Teufelskralle hilft sanft bei Rückenschmerzen. Allgemeinarzt 1999, 12, 1036. [Google Scholar]
- Szczepański, L.; Chudzik, D.; Mazurek, M.; Soroka, P. Badania skuteczności i tolerancji preparatu Pagosid (wyciąg z korzenia Harpagophytum procumbens) w leczeniu reumatoidalnego zapalenia stawów i choroby zwyrodnieniowej stawów. Reumatologia 2000, 38, 67–73. [Google Scholar]
- Usbeck, C. Teufelskralle: Behandlung chronischer Schmerzen. Arzneim. Forum 2000, 3, 23–25. [Google Scholar]
- Engel, S. Rivoltan (Li 174) zur Behandlung von Patienten mit degenerativen Erkrankungen des Bewegungsapparates. Dtsch. Apoth. Ztg. 2000, 140, 1369. [Google Scholar]
- Laudahn, D.; Walper, A. Efficacy and tolerance of Harpagophytum extract LI 174 in patients with chronic non-radicular back pain. Phytother. Res. 2001, 15, 621–624. [Google Scholar] [CrossRef]
- Richter, T. Teufelskrallenextrakt bei chronischen Rückenschmerzen. Dtsch. Apoth. Ztg. 2002, 142, 131–133. [Google Scholar]
- Ullmann, M. Therapie chronischer Rückenschmerzen: Teufelskrallen-Extrakt erzielt hochsignifikante Schmerzreduktion. Allgemeinarzt 1999, 11, 1018–1019. [Google Scholar]
- Schendel, U.M. Arthrose-therapie: Verträglich geht es auch. Kassenarzt 2001, 29, 36–39. [Google Scholar]
- Ribbat, J.M.; Schakau, D. Behandlung chronisch aktivierter Schmerzen am Bewegungsapparat. Nat. Med. 2001, 16, 23–32. [Google Scholar]
- Chrubasik, S.; Thanner, J.; Künzel, O.; Conradt, C.; Black, A.; Pollak, S. Comparison of outcome measures during treatment with the proprietary Harpagophytum extract doloteffin in patients with pain in the lower back, knee or hip. Phytomedicine 2002, 9, 181–194. [Google Scholar] [CrossRef]
- Flammersfeld, L.; Weinmann, B. Traitement du rhumatisme par l’extrait sec de racine d’Harpagophytum. Une étude multicentrique en cabinet médical. Phytotherapie 2005, 3, 104–106. [Google Scholar] [CrossRef]
- Kloker, B.; Flammersfeld, L. Rheumatherapie mit Teufelskrallenwurzel-Trockenextrakt. Eine multizentrische Praxisstudie. Ärztez. Nat. 2003, 44, 108–111. [Google Scholar]
- Wegener, T.; Lüpke, N.P. Treatment of patients with arthrosis of hip or knee with an aqueous extract of devil’s claw (Harpagophytum procumbens DC.). Phytother. Res. 2003, 17, 1165–1172. [Google Scholar] [CrossRef]
- Wegener, T. Zur Klinischen Wirksamkeit der Südafrikanischen Teufelskrallenwurzel (Harpagophyti radix) bei Patienten mit Cox-und Gonarthrose: Ergebnisse und Bewertung Einer Klinischen Studie der Phase IV. Ph.D. Thesis, Universität Osnabrück, Osnabrück, Germany, 2005; p. 115. [Google Scholar]
- Rütten, S.; Kuhn, M. Spezialextrakt aus Teufelskrallenwurzel. Klinische Vergleichsstudie bei Patienten mit unspezifischen Schmerzen im Lumbalbereich. Ärztez. Nat. 2005, 46, 114–116. [Google Scholar]
- Schmidt, A.; Berghof, U.; Schmidt, E. Therapie der unspezifischen Lumbalgie mit Teufelskrallenwurzelextrakt—Ergebnisse einer klinischen Studie. Phys. Med. Rehabil. Kurortmed. 2005, 15, 317–321. [Google Scholar] [CrossRef]
- Schmidt, A.P.; Schmidt, E.G. Über die Wirksamkeit von Teufelskrallenwurzelextrakt zur Therapie Unspezifischer Lumbaler Rückenschmerzen; Cuvillier Verlag: Göttingen, Germany, 2005; p. 101. ISBN 3865376118. [Google Scholar]
- Sohail, M.T.; Chaudhry, M.I.; Usman, M.K.; Mian, T.; Ishaq, M.N. Efficacy and tolerance of atrisin in degenerative and inflammatory joint disorders. Phytother. Res. 2005, 19, 365–368. [Google Scholar] [CrossRef] [PubMed]
- Arndt, D.; Bobermien, K.; Heyer, H.; Braun, D.; Köhler, G. Harpagophytum procumbens—Erste Erfahrungen in der Endometriosetherapie. Zent. Gynakol. 2005, 127, A1. [Google Scholar] [CrossRef]
- Arndt, D.; Bobermien, K.; Heyer, H.; Köhler, G. Hormonfreie Endometriosetherapie mit Harpagophytum procumbens—Ein neuer Weg in der Endometriosetherapie. Geburtshilfe Frauenheilkd. 2006, 66, PO_E_03_10. [Google Scholar] [CrossRef]
- Suter, A.; Whittaker, P.; Dickson, S.; McIntyre, L.; Tan, J. Positive influence of a Harpagophytum procumbens preparation on different rheumatic complaints—Results from clinical trial. Planta Med. 2006, 72, P_200. [Google Scholar] [CrossRef]
- Warnock, M.; McBean, D.; Suter, A.; Tan, J.; Whittaker, P. Effectiveness and safety of Devil’s claw tablets in patients with general rheumatic disorders. Phytother. Res. 2007, 21, 1228–1233. [Google Scholar] [CrossRef] [PubMed]
- Chrubasik, S.; Chrubasik, C.; Kunzel, O.; Black, A. Patient-perceived benefit during one year of treatment with Doloteffin. Phytomedicine 2007, 14, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Conrozier, T.; Mathieu, P.; Bonjean, M.; Marc, J.F.; Renevier, J.L.; Balblanc, J.C. A complex of three natural anti-inflammatory agents provides relief of osteoarthritis pain. Altern. Health Med. 2014, 20, 32–37. [Google Scholar]
- Vreju, F.A.; Ciurea, P.L.; Rosu, A.; Chisalau, B.A.; Parvanescu, C.D.; Firulescu, S.C.; Turcu Stiolica, A.; Barbulescu, A.L.; Dinescu, S.C.; Dumitrescu, C.I. The effect of glucosamine, chondroitin and Harpagophytum procumbens on femoral hyaline cartilage thickness in patients with knee osteoarthritis—An MRI versus ultrasonography study. J. Mind Med. Sci. 2019, 6, 162–168. [Google Scholar] [CrossRef]
- Hu, S.; Belcaro, G.; Cesarone, M.R.; Feragalli, B.; Cotellese, R.; Dugall, M.; Scipione, C.; Scipione, V.; Maione, C.; Maramaldi, G.; et al. A sport cream (Harpago-Boswellia-ginger-escin) for localized neck/shoulder pain. Minerva Med. 2021, 112, 255–260. [Google Scholar] [CrossRef] [PubMed]
- Schrüffler, H. Ein Fortschritt in der nichtsteroidalen antirheumatischen Therapie. Med. Publ. 1980, 1, 1–8. [Google Scholar]
- Guyader, M. Les Plantes Anti-Rhumatismales. Etude Historique et Pharmacologique. Et Etude Clinique du Nebulisat d’Harpagophytum procumbens DC chez 50 Patients Arthrosioues Suivis en Service Hospitalier. Ph.D. Thesis, Université Pierre et Marie Curie, Paris, France, 1984; p. 151. [Google Scholar]
- Pinget, M.; Lecomte, A. Etude des effets de l’Harpagophytum en rhumatologie dégénérative. 37°2 Le Mag. 1990, 10, 23–25. [Google Scholar]
- Lecomte, A.; Costa, J.P. Harpagophytum dans l’arthrose: Etude en double insu contre placebo. 37°2 Le Mag. 1992, 15, 27–30. [Google Scholar]
- Chrubasik, S.; Zimpfer, C.; Schütt, U.; Ziegler, R. Effectiveness of Harpagophytum procumbens in treatment of acute low back pain. Phytomedicine 1996, 3, 1–10. [Google Scholar] [CrossRef]
- Chrubasik, S.; Pollak, S.; Black, A. Effectiveness of devil’s claw for osteoarthritis. Rheumatology 2002, 41, 1332–1333, author reply 1333. [Google Scholar] [CrossRef][Green Version]
- Chrubasik, S.; Fiebich, B.; Black, A.; Pollak, S. Treating low back pain with an extract of Harpagophytum that inhibits cytokine release. Eur. J. Anaesthesiol. 2002, 19, 209. [Google Scholar] [CrossRef]
- Chrubasik, S.; Zimpfer, C.; Schütt, U.; Ziegler, R. The arhus low back rating scale: A useful tool to evaluate the success of low back pain treatment. Reg. Anesth. J. Neural Blockade Obstet. Surg. Pain Control 1997, 22, 100. [Google Scholar]
- Schmelz, H.; Hämmerle, H.D.; Springorum, H.W. Analgetische Wirkung eines Teufelskrappenwurzel-Extraktes bei verschiedenen chronisch-degenrativen Gelenkerkrankungen. In Rheumatherapie mit Phytopharmaka; Chrubasik, S., Wink, M., Eds.; Hippokrates: Stuttgart, Germany, 1997; pp. 86–89. [Google Scholar]
- Chrubasik, S.; Junck, H.; Breitschwerdt, H.; Conradt, C.; Zappe, H. Effectiveness of Harpagophytum extract WS 1531 in the treatment of exacerbation of low back pain: A randomized, placebo-controlled, double-blind study. Eur. J. Anaesthesiol. 1999, 16, 118–129. [Google Scholar] [CrossRef] [PubMed]
- Chantre, P.; Cappelaere, A.; Leblan, D.; Guedon, D.; Vandermander, J.; Fournie, B. Efficacy and tolerance of Harpagophytum procumbens versus diacerhein in treatment of osteoarthritis. Phytomedicine 2000, 7, 177–183. [Google Scholar] [CrossRef]
- Leblan, D.; Chantre, P.; Fournié, B. L’harpagophyton dans le traitement de la gonarthrose et de la coxarthrose. Résultats à quatre mois d’une étude prospective multicentrique, contrôlée en double aveugle, versus diacerhéine. Rev. Rhum. 2000, 67, 634–640. [Google Scholar] [CrossRef]
- Göbel, H.; Heinze, A.; Ingwersen, M.; Niederberger, U.; Gerber, D. Wirkmechanismus von Harpagophytum-procumbens-Extrakt L1 174 bei der Behandlung von unspezifischen Rückenschmerzen. In Phytopharmaka VI, Forschung und klinische Anwendung; Rietbrock, N., Ed.; Steinkopff: Darmstadt, Germany, 2000; pp. 99–115. [Google Scholar]
- Göbel, H.; Heinze, A.; Ingwersen, M.; Niederberger, U.; Gerber, D. Harpagophytum-Extrakt LI 174 (Teufelskralle) bei der Behandlung unspezifischer Ruckenschmerzen—Effekte auf die sensible, motorische und vaskulare Muskelreagibilitat. Schmerz 2001, 15, 10–18. [Google Scholar] [CrossRef]
- Frerick, H.; Biller, A.; Schmidt, U. Stufenschema bei Coxarthrose. Kassenarzt 2001, 5, 34–41. [Google Scholar]
- Biller, A. Ergebnisse zweier randomisierter kontrollierter Studien und einer Anwendungsbeobachtung mit Teufelskrallenextrakt. In Phytopharmaka VII, Forschung und klinische Anwendung; Schulz, V., Rietbrock, N., Roots, I., Loew, D., Eds.; Steinkopff: Darmstadt, Germany, 2002; pp. 81–92. ISBN 978-3-642-57528-0. [Google Scholar]
- Chrubasik, S.; Model, A.; Black, A.; Pollak, S. A randomized double-blind pilot study comparing Doloteffin and Vioxx in the treatment of low back pain. Rheumatology 2003, 42, 141–148. [Google Scholar] [CrossRef]
- Chrubasik, S.; Künzel, O.; Thanner, J.; Conradt, C.; Black, A. A 1-year follow-up after a pilot study with Doloteffin for low back pain. Phytomedicine 2005, 12, 1–9. [Google Scholar] [CrossRef]
- Chrubasik, S.; Künzel, O.; Thanner, J.; Conradt, C. A short-term follow-up after a randomised double-blind pilot study comparing Doloteffin® vs. rofecoxib for low back pain. Focus Altern. Complement. Ther. 2003, 8, 133. [Google Scholar] [CrossRef]
- Ullmann, M.; Model, A.; Chrubasik, S. Treatment of acute low back pain with the Harpagophytum extract Doloteffin and the COX-2 inhibitor VIOXX: A pilot study. In Proceedings of the 3rd International Congress on Phytomedicine, Munich, Germany, 11–13 October 2000. [Google Scholar]
- Lienert, A.; Rütten, S.; Kuhn, M.; Wartenberg-Demand, A. A randomised, active-controlled, mono-centric study of the herbal drug, Devil’s claw (Harpagophytum procumbens) (ALLYA® tablets), Voltaren® and Vioxx® indicates equal efficacy in the treatment of patients with unspecific lumbar pain. 54. Jahrestag. Norddtsch. Orthopädenver. Eingetrag. Ver. Hambg. 2005, 16, Doc05novEP26. [Google Scholar]
- Lienert, A.; Rütten, S.; Kuhn, M.; Wartenberg-Demand, A. Efecto analgésico de un extracto estandarizado de harpagofito. Rev. Fitoter. 2007, 7, 15–20. [Google Scholar]
- Pillay, D. A Double-Blinded, Placebo Controlled Clinicaltrial Evaluating the Efficacy of the Harpago and Celery Seed Cream in Mild to Moderate Degenerative Joint Disease of the Knee. Ph.D. Thesis, Durban Institute of Technology, Durban, South Africa, 2006; p. 138. [Google Scholar]
- Anvari, H.M.P.; Panahi, J.R.; Ansari, M.; Iraji, M.; Ghorbanian, N.; Dehghani, A. The effect of single dose of Harpagophytum capsule (teltonal) on post tracheal intubation sore throat after general anesthesia. Arch. Anesthesiol. Crit. Care 2016, 2, 239–242. [Google Scholar]
- Moré, M.; Grünwald, J.; Pohl, U.; Uebelhack, R. A Rosa canina—Urtica dioica—Harpagophytum procumbens/zeyheri combination significantly reduces gonarthritis symptoms in a randomized, placebo-controlled double-blind study. Planta Med. 2017, 83, 1384–1391. [Google Scholar] [CrossRef] [PubMed]
- Anonymous. Devil’s claw root: Ulcers and gastrointestinal bleeding? Prescrire Int. 2013, 22, 296. [Google Scholar]
- Gallo, E.; Lucenteforte, E.; Firenzuoli, F.; Menniti-Ippolito, F.; Maggini, V.; Pugi, A.; Mascherini, V.; Gori, L.; Mugelli, A.; Vannacci, A. Herbalists’ perception of risks involving commonly sold medicinal plants in Italy. Complement. Med. 2014, 22, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Vlachojannis, J.; Roufogalis, B.D.; Chrubasik, S. Systematic review on the safety of Harpagophytum preparations for osteoarthritic and low back pain. Phytother. Res. 2008, 22, 149–152. [Google Scholar] [CrossRef]
- Posadzki, P.; Watson, L.K.; Ernst, E. Adverse effects of herbal medicines: An overview of systematic reviews. Clin. Med. 2013, 13, 7–12. [Google Scholar] [CrossRef]
- Biazi, B.I.; D’Epiro, G.F.; Zanetti, T.A.; de Oliveira, M.T.; Ribeiro, L.R.; Mantovani, M.S. Risk assessment via metabolism and cell growth inhibition in a HepG2/C3A cell line upon treatment with arpadol and its active component harpagoside. Phytother. Res. 2017, 31, 387–394. [Google Scholar] [CrossRef]
- Cordier, W.; Steenkamp, V. Drug interactions in African herbal remedies. Drug Metab. Drug Interact. 2011, 26, 53–63. [Google Scholar] [CrossRef]
- Shaw, D.; Leon, C.; Kolev, S.; Murray, V. Traditional remedies and food supplements. A 5-year toxicological study (1991–1995). Drug Saf. 1997, 17, 342–356. [Google Scholar] [CrossRef] [PubMed]
- Argento, A.; Tiraferri, E.; Marzaloni, M. Anticoagulanti orali e piante medicinali. Una interazione emergente. Ann. Ital. Med. Int. 2000, 15, 139–143. [Google Scholar]
- Heck, A.M.; DeWitt, B.A.; Lukes, A.L. Potential interactions between alternative therapies and warfarin. Am. J. Health Syst. Pharm. 2000, 57, 1221–1227. [Google Scholar] [CrossRef] [PubMed]
- Ramsay, N.A.; Kenny, M.W.; Davies, G.; Patel, J.P. Complimentary and alternative medicine use among patients starting warfarin. Br. J. Haematol. 2005, 130, 777–780. [Google Scholar] [CrossRef]
- Izzo, A.A.; Di Carlo, G.; Borrelli, F.; Ernst, E. Cardiovascular pharmacotherapy and herbal medicines: The risk of drug interaction. Int. J. Cardiol. 2005, 98, 1–14. [Google Scholar] [CrossRef]
- Carvalho, R.R.; Donadel, C.D.; Cortez, A.F.; Valviesse, V.R.; Vianna, P.F.; Correa, B.B. Syndrome of inappropriate antidiuretic hormone secretion induced by the phytotherapy Harpagophytum procumbers: Case report. J. Bras. Nefrol. 2017, 39, 79–81. [Google Scholar] [CrossRef] [PubMed]
- Cuspidi, C.; Sala, C.; Tadic, M.; Grassi, G.; Mancia, G. Systemic Hypertension Induced by Harpagophytum procumbens (Devil’s claw): A case report. J. Clin. Hypertens. 2015, 17, 908–910. [Google Scholar] [CrossRef] [PubMed]
- Douros, A.; Bronder, E.; Andersohn, F.; Klimpel, A.; Thomae, M.; Ockenga, J.; Kreutz, R.; Garbe, E. Drug-induced acute pancreatitis: Results from the hospital-based Berlin case-control surveillance study of 102 cases. Aliment. Pharmacol. Ther. 2013, 38, 825–834. [Google Scholar] [CrossRef]
- Altmeyer, N.; Garnier, R.; Rosenberg, N.; Geerolf, A.M.; Ghaem, A. Conjonctivite, rhinite et asthme rythmes par l’exposition professionnelle a l’Harpagophytum. Arch. Mal. Prof. Méd. Trav. Sécurité Soc. 1991, 53, 289–291. [Google Scholar]
- Rahman, H.; Kim, M.; Leung, G.; Green, J.A.; Katz, S. Drug-herb interactions in the elderly patient with IBD: A growing concern. Curr. Treat. Options Gastroenterol. 2017, 15, 618–636. [Google Scholar] [CrossRef]
- Colas, C. Développement de Méthodes Physico-Chimiques pour le Contrôle de la Médicationpar l’Harpagophytum et l’Eleutherococcus, Principes Actifs Utilisés en Phytothérapie Équine. Ph.D. Thesis, École Polytechnique, Université Paris-Saclay, Paris, France, 2006; p. 313. [Google Scholar]
- Torfs, S.; Delesalle, C.; Vanschandevijl, K.; de Clercq, D.; Van Loon, G.; Nollet, H.; Deprez, P. Anti-inflammatory phytotherapeutics: A valuable alternative to NSAID treatment in horses. Vlaams Diergeneeskd. Tijdschr. 2008, 78, 161–170. [Google Scholar]
- Montavon, S. Efficacité d’une préparation phytothérapique à base d’Harpagophytum procumbens dans les cas d’éparvin chez le cheval adulte. Prat. Vét. Equine 1994, 26, 49–53. [Google Scholar]
- Axmann, S. Studien zur Pharmakokinetik und zur Klinischen Wirksamkeit von Harpagosid aus Einem Extrakt der Teufelskralle (“Harpagophytum procumbens” DC ex Meisn.) bei Pferden. Ph.D. Thesis, Veterinärmedizinische Universität, Wien, Austria, 2019; p. 106. [Google Scholar]
- Axmann, S.; Hummel, K.; Nöbauer, K.; Razzazi-Fazeli, E.; Zitterl-Eglseer, K. Pharmacokinetics of harpagoside in horses after intragastric administration of a Devil’s claw (Harpagophytum procumbens) extract. J. Vet. Pharmacol. Ther. 2019, 42, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Moreau, M.; Lussier, B.; Pelletier, J.P.; Martel-Pelletier, J.; Bedard, C.; Gauvin, D.; Troncy, E. A medicinal herb-based natural health product improves the condition of a canine natural osteoarthritis model: A randomized placebo-controlled trial. Res. Vet. Sci. 2014, 97, 574–581. [Google Scholar] [CrossRef]
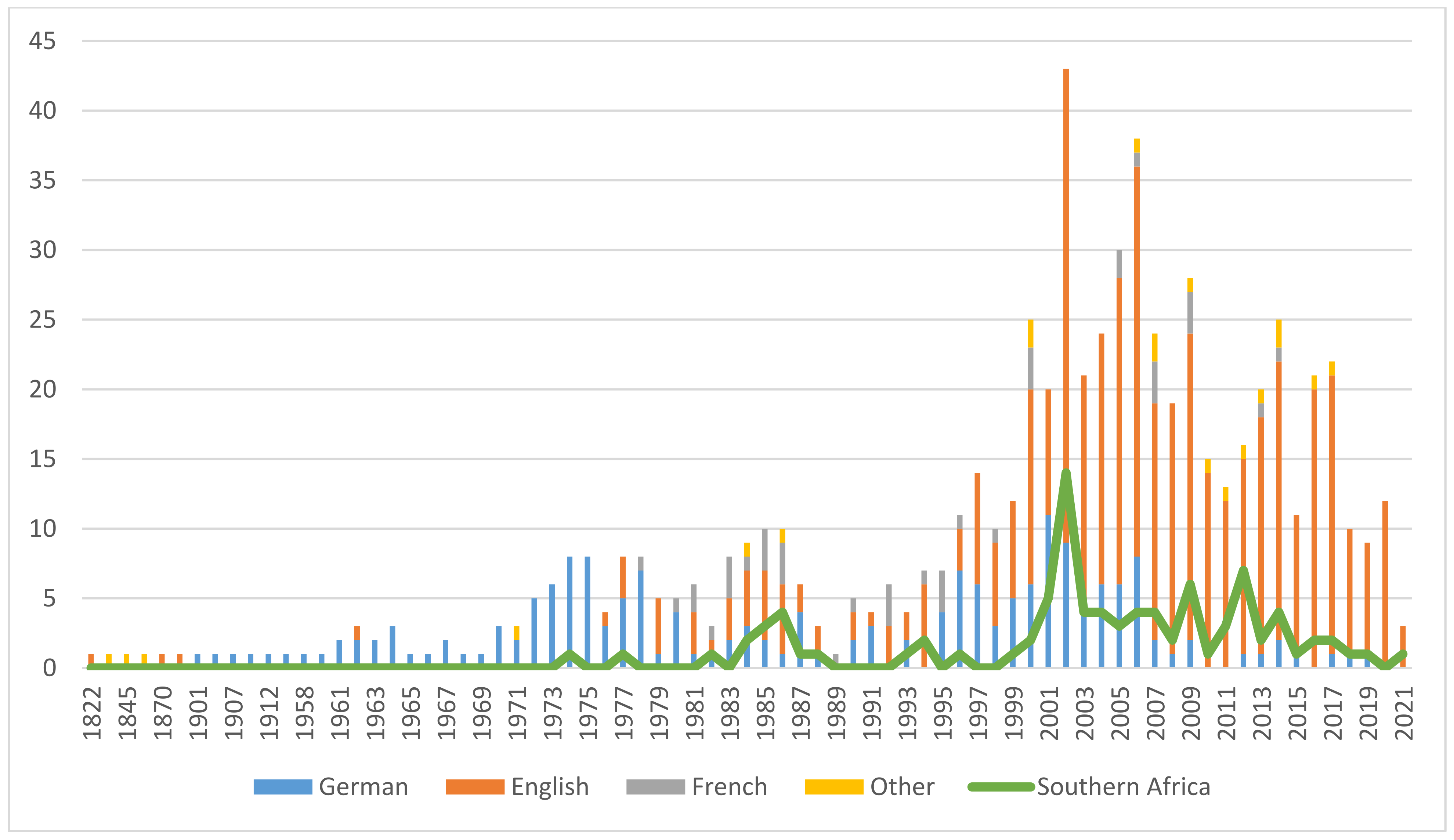
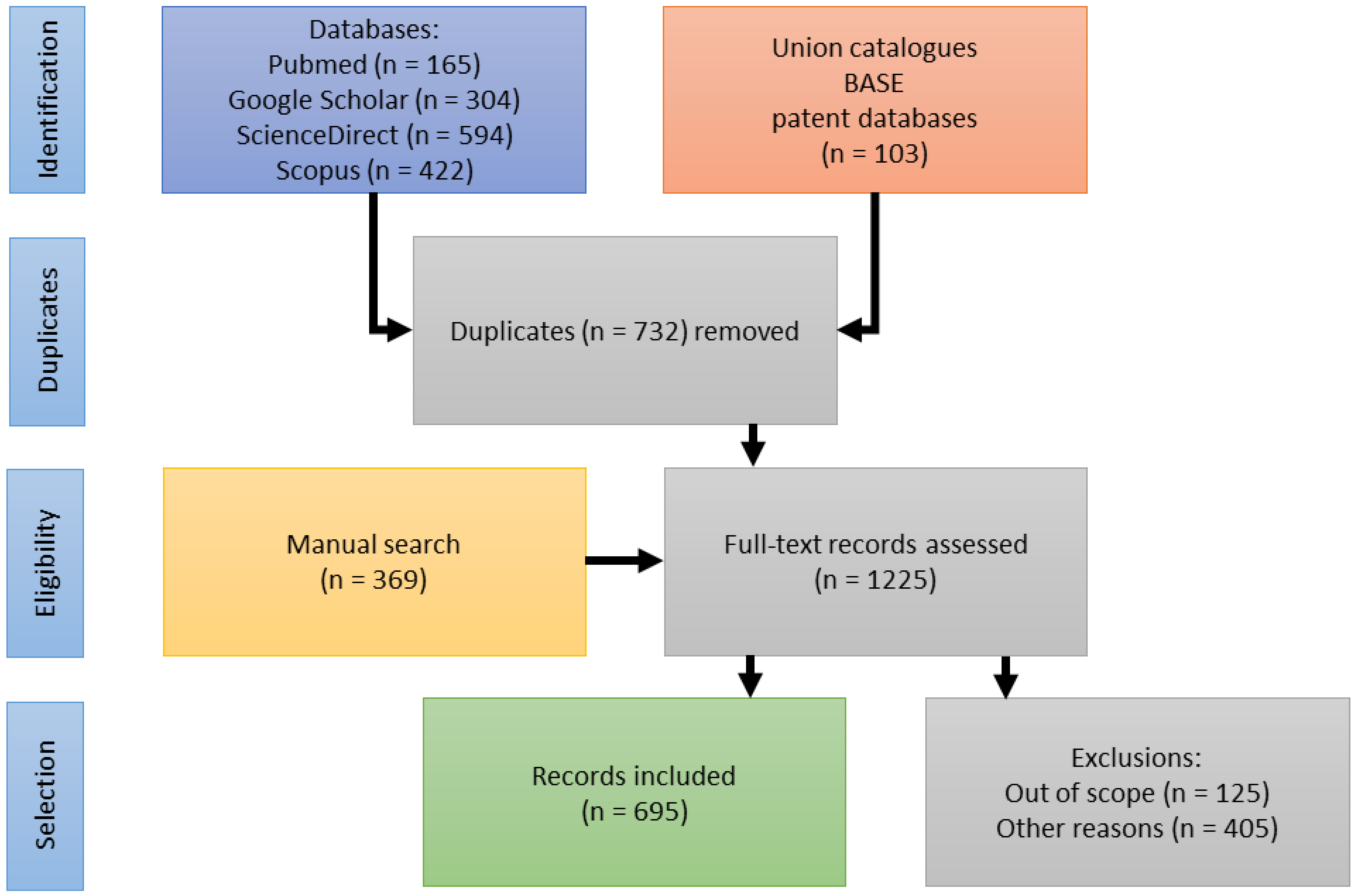
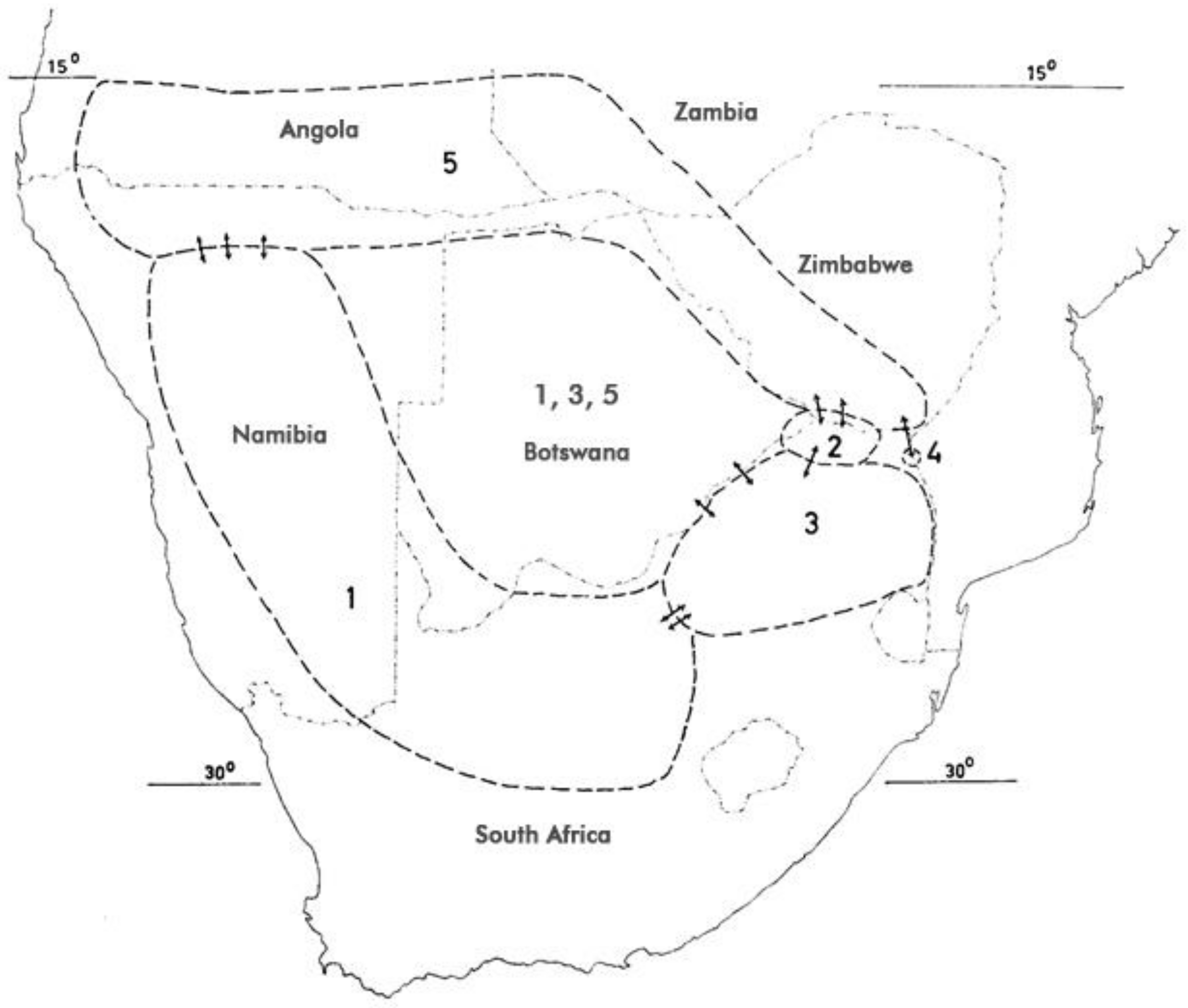
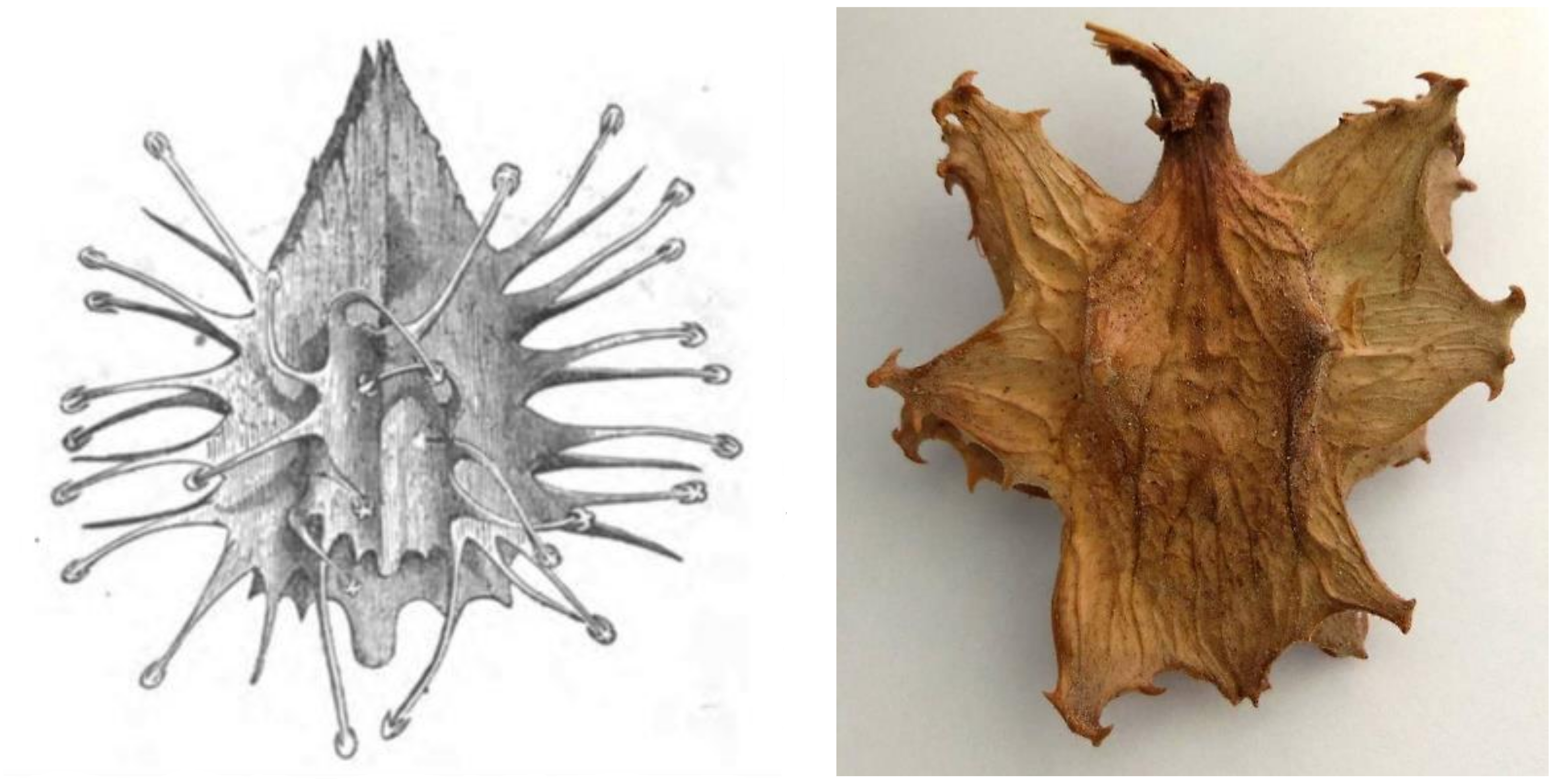

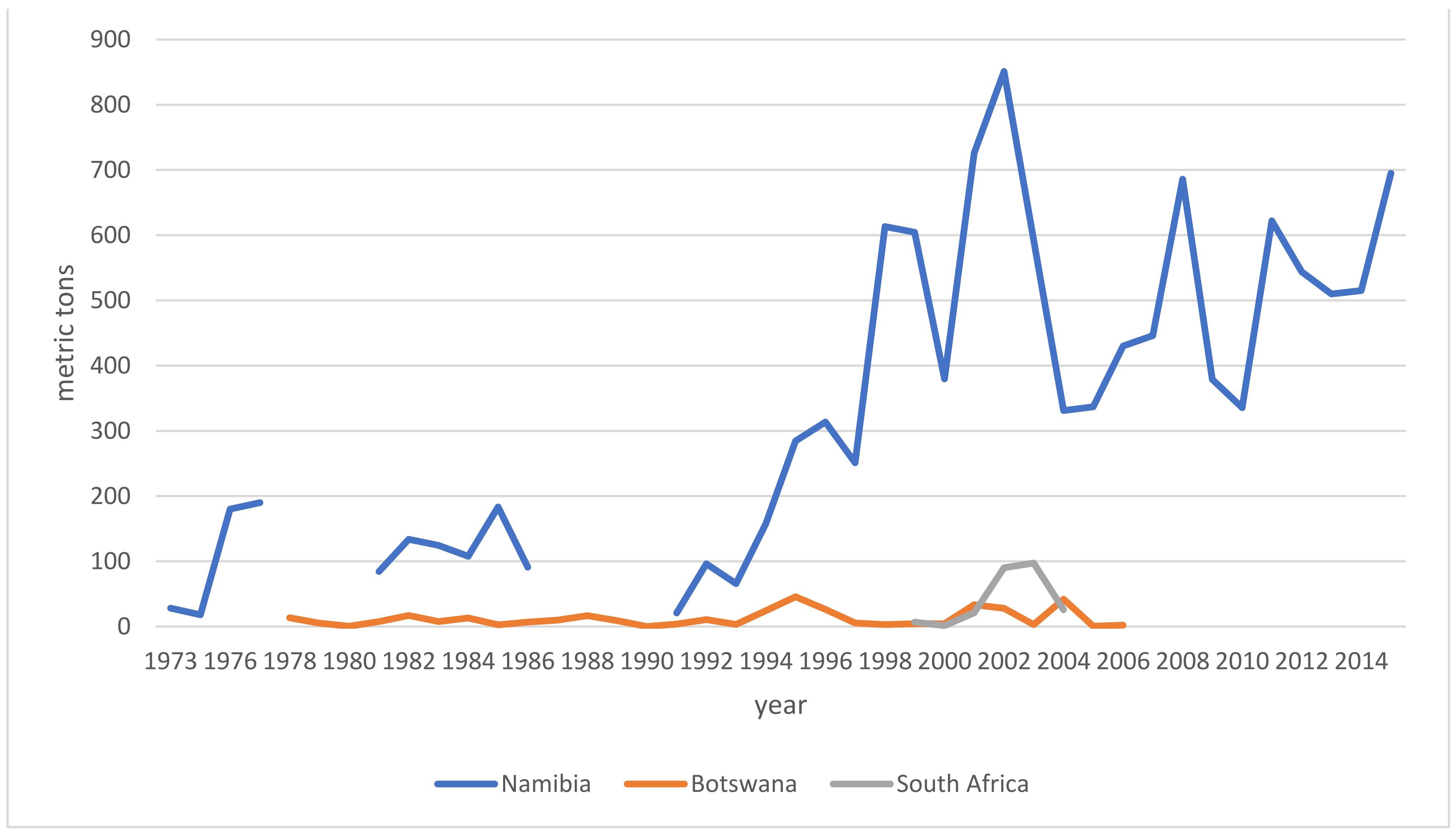
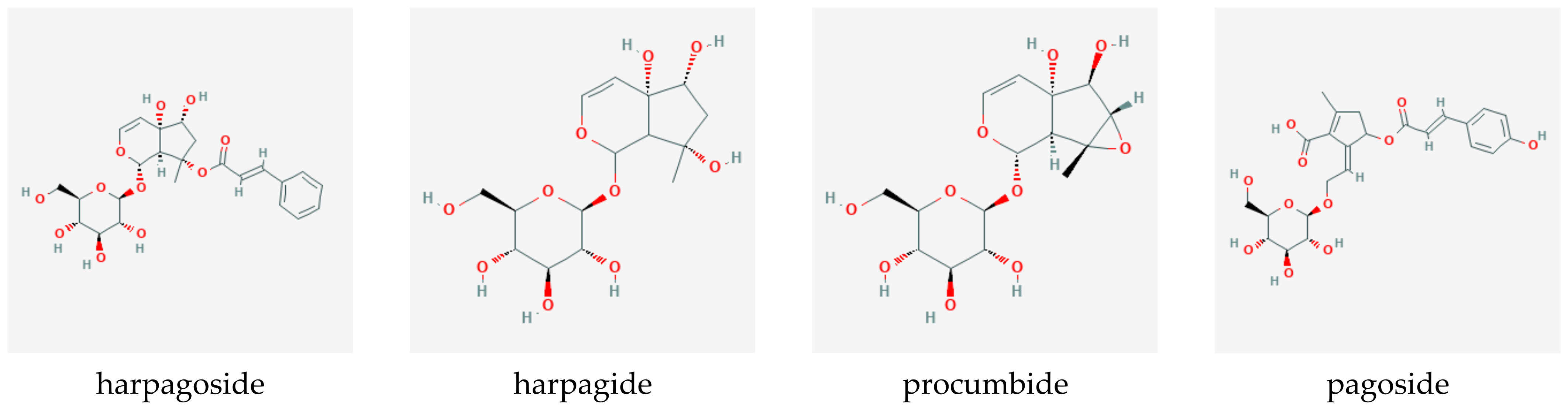

| Source | Species Included | Year | Reference |
|---|---|---|---|
| Official monographs | |||
| British Herbal Pharmacopoeia | H. procumbens | 1981 | [154] |
| Pharmacopée française | H. procumbens | 1989 | [155] |
| Kommission E | H. procumbens (corrected) Monograph was informed by [65,156] | 1990 | [157] |
| Pharmacopée française | H. procumbens dry extract | 1992 | [158] |
| DAB 10 2nd. Supplement | H. procumbens | 1993 | [159] |
| European Pharmacopoeia 3rd ed. | H. procumbens | 1997 | [160] |
| European Pharmacopoeia 4th ed. Suppl. 4.3 | H. procumbens/H. zeyheri (revised) | 2003 | [161] |
| Pharmacopée française | H. procumbens/H. zeyheri (homoeopathic preparations) | 2007 | [162] |
| European Pharmacopoeia 7th ed. | Devil’s claw dry extract | 2008 | [163] |
| Health Canada | H. procumbens | 2008 | [164] |
| European Pharmacopoeia 7th ed. | H. procumbens/H. zeyheri (revised) | 2011 | [165] |
| Polish Pharmacopoeia 8 | H. procumbens/H. zeyheri | 2008 | [166] |
| USP Herbal Medicines Compendium | H. procumbens/H. zeyheri (draft) | 2013 | [153] |
| European Medicines Agency (EMA) | H. procumbens/H. zeyheri (revised from 2008) | 2016 | [15,167,168] |
| European Pharmacopoeia 9.6 | H. procumbens/H. zeyheri (revised) | 2018 | [169] |
| State Pharmacopoeia of Ukraine | H. procumbens/H. zeyheri | 2018 | [170] |
| Health Canada | H. procumbens/H. zeyheri (revised from 2008) | 2018 | [164] |
| Authoritative compendia | |||
| ESCOP | H. procumbens | 1996 | [171] |
| ESCOP | H. procumbens (revised) (omission of H. zeyheri is discussed in [172,173]) | 2003 | [16] |
| World Health Organization | H. procumbens | 2007 | [174] |
| African Herbal Pharmacopoeia | H. procumbens | 2010 | [175] |
| Martindale | H. procumbens (continuously revised from 1997) | 2017 | [176] |
| Other compendia | |||
| Longwood Herbal Task Force | H. procumbens/H. zeyheri | 1999 | [177] |
| Herbal Medicines | H. procumbens/H. zeyheri | 2015 | [178] |
| Phytopharmacy | H. procumbens/H. zeyheri | 2015 | [179] |
| Kooperation Phytopharmaka | H. procumbens | 2020 | [180] |
| Topic | Year | Reference |
|---|---|---|
| Macroscopic and microscopic descriptions | 1964 | [58] |
| Macroscopic, microscopic, and chromatographic differentiation of commercial drug samples | 1973 | [218] |
| Macroscopic, microscopic, and chromatographic differentiation of commercial drug samples | 1974 | [219] |
| Simple TLC with Scrophularia nodosa as a reference standard | 1975 | [220] |
| Distribution of harpagoside within H. procumbens and H. zeyheri | 1977 | [221] |
| Standardization by determination of harpagoside, bitterness value, and dry residue | 1977 | [11] |
| Spectrometric method for the quantitative evaluation of the glycoiridoids | 1978 | [222] |
| Report of falsified, adulterated, and contaminated commercial products | 1978 | [214] |
| Quantitative determination of harpagoside via HPLC | 1980 | [223] |
| GLC method for the determination of harpagide and harpagoside | 1981 | [224] |
| Histological characteristics under scanning electron microscope | 1984 | [225] |
| Stability of iridoids during extraction | 1985 | [226] |
| Determination of harpagoside, luteolin, chlorogenic, caffeic, and cinnamic acid from extracts | 1986 | [227] |
| Analysis of permethylated iridoid glycosides by GC/MS | 1986 | [228] |
| Determination of harpagide, 8-p-coumaroyl harpagide (8-PCHG), and harpagoside by HPLC | 1994 | [229] |
| Analysis of the harpagoside content of commercial samples by HPLC | 1995 | [230] |
| TLC method for determination of harpagoside | 1995 | [231] |
| HPLC/UV for the determination of harpagoside in commercial powdered dry extracts | 1996 | [232] |
| HPLC/UV for the determination of harpagoside in commercial tea products | 1996 | [233] |
| HPLC/UV for the determination of harpagoside in commercial products (multiple dosage forms) | 1996 | [234] |
| HPTLC for quantitative determination of harpagoside | 1996 | [235] |
| HPLC determination of harpagide, 8-PCHG, and harpagoside in H. procumbens and H. zeyheri—ratio of harpagoside/8-PCHG can be used to distinguish species | 1997 | [19] |
| HPLC determination of ratio of harpagoside/8-PCHG, 8-PCHG < 8% proposed for H. procumbens | 1998 | [12] |
| Methods for quality control and stability testing of Harpagophytum homeopathic preparations | 1998 | [236] |
| HPLC/UV for the determination of harpagoside in commercial dry extract products | 1999 | [237] |
| Differentiation of H. procumbens (<9% 8-PCHG), mixtures (10–30% 8-PCHG), and H. zeyheri (>31 8-PCHG) proposed | 2000 | [13] |
| Biopharmaceutical quality, release of active ingredients in vitro, and disintegration tests | 2000 | [238] |
| Methods for detection of adulterations and contaminations | 2001 | [239] |
| Bioequivalence of Harpagophytum products | 2002 | [240] |
| Near infrared spectroscopy (NIRS) determination of harpagoside, 8-PCHG, and their ratio | 2003 | [241] |
| NIR-FT-Raman spectroscopy for identification and quantification of harpagoside | 2005 | [242] |
| Determination of harpagoside from CO2-extracts with HPLC and HPTLC-densitometry | 2005 | [243] |
| NIRS determination of harpagoside, 8-PCHG, and their ratio | 2005 | [244] |
| Fast HPLC determination of harpagoside using a monolithic silica column | 2005 | [245] |
| Validation of a fast-HPLC for separation of iridoid glycosides to distinguish between species | 2005 | [246] |
| LC-DAD-MS/SPE-NMR hyphenation for identification of isobaric iridoid glycoside regioisomers | 2005 | [247] |
| X-ray fluorescence spectrometry (SRTXRF) to determine trace elements | 2005 | [248] |
| Determination of aflatoxin B1 | 2006 | [249] |
| LC/MS determination of harpagoside, 8-PCHG, and their ratio | 2006 | [250] |
| Computational study to estimate the proton and sodium cation affinities of harpagide | 2006 | [251] |
| Quality parameters of finished products in the German market | 2006 | [252] |
| Proposal to revise the drug–extract ratio of aqueous/ethanolic extracts | 2006 | [253] |
| Methods for determination of minerals and heavy metals | 2007 | [254] |
| Analysis of iridoids in horse urine | 2008 | [255] |
| Solid-phase extraction for LC/MS analysis of harpagoside, 8-PCHG, and harpagide in equine plasma | 2008 | [256] |
| Validated HPTLC method for the determination of harpagoside | 2008 | [257] |
| High-Pressure Liquid Chromatography-Diode Array Detection (HPLC-DAD) for harpagoside and isoacteoside contents | 2009 | [258] |
| HPLC-DAD and HPLC–ESI-MS analyses of stability of the constituents | 2011 | [181] |
| Anatomical study of secondary tubers and quantification of harpagoside by HPLC | 2012 | [259] |
| Authenticity and contamination tests by DNA barcoding | 2013 | [260] |
| Exploring species substitution through chemometric modeling of 1H-NMR and UHPLC-MS | 2014 | [182] |
| Mid-infrared spectroscopy and short-wave infrared hyperspectral imaging for qualitative assessment of H. procumbens and H. zeyheri | 2014 | [85,88] |
| Morphology, histochemistry, and ultrastructure of foliar mucilage-producing trichomes | 2014 | [261] |
| NMR-based chemometric approach for species differentiation | 2014 | [262] |
| UPLC Q-TOF ESI determination of harpagosides in H. procumbens, H. zeyheri, and extracts | 2016 | [263] |
| Loss on drying and total ash | 2016 | [264] |
| Comparison of microwave and ultrasound-assisted with conventional solvent extraction methods for harpagoside determination | 2016 | [265] |
| Innovative micro-extraction techniques to determine harpagoside and phenolic patterns in H. procumbens and finished products | 2017 | [266] |
| Determination of suitable extraction solvent | 2017 | [267] |
| GC-MS determination of chemical constituents | 2017 | [268] |
| DNA barcoding to detect contamination and substitution | 2017 | [269] |
| HPLC and MS analyses of spagyric tinctures | 2019 | [270] |
| Validated RP-HPLC-PDA method for quantification of harpagoside in extracts and finished products | 2019 | [271] |
| UPLC–MS profiling of samples from different locations | 2019 | [90] |
| Determination of macro- and micro-elements in finished products using ICP OES | 2020 | [272] |
| HPLC method for harpagoside determination in finished product (tablet) | 2020 | [273] |
| Study | Year | Reference |
|---|---|---|
| Guineapig isolated ileum; harpagoside (40 µg/mL) and harpagogenine (2.5 µg/mL) non-selectively inhibited contractions; harpagide (40 µg/mL) increased the cholinergic response without inhibitory effects. | 1981 | Fontaine et al. [282] |
| Calcium ionophore-stimulated mouse peritoneal macrophages; harpagoside and harpagide inhibited leukotriene C4 (LTC4) and prostaglandin E2 (PGE2) release (not significant) and harpagoside inhibited thromboxane B2 (TXB2) release, similar to ibuprofen. | 2000 | Benito et al. [283] |
| Lipopolysaccharide-stimulated primary human monocytes; Harpagophytum * extract, harpagoside, and harpagide extract prevented synthesis of tumor necrosis factor alpha (TNF-α), isolated substances showed no effect. | 2001 | Fiebich et al. [284] |
| Ionophore A23187 stimulated Cys-LT levels in anticoagulated whole blood; Harpagophytum extract, harpagoside, and extract fractions; inhibitory effect stronger with extract than harpagoside, no effect with fractions without harpagoside, suggesting relation between serum harpagoside and inhibition of leukotriene biosynthesis. | 2001 | Loew et al. [285] |
| Modified Hens-Egg-Test at the Chorion-Allantoin-Membrane (HET-CAM) and lipoxygenase assay; ethanolic extracts of Harpagophytum (60%, 30%, 0%); 30% most potent in HET-CAM, 60% most potent in inhibiting lipoxygenase pathway. | 2002 | Wahrendorf et al. [286] |
| Human neutrophile elastase (HNE); Harpagophytum extract, fractions, and isolates; weak dose-dependent inhibition was observed, with H. procumbens extract twice as strong as H. zeyheri; 6′-O-acetyl-acteoside (not in H. procumbens) the strongest isolate, followed by isoacteoside and pagoside (dominant in H. zeyheri). | 2002, 2003 | Boje [199]; Boje et al. [201] |
| Lipopolysaccharide (LPS)-induced inflammation in mouse fibroblast cell line L929; 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay, reverse transcription-polymerase chain reaction, PGE2 immunoassay, and nitric oxide (NO) detection; aqueous H. procumbens extract; suppression of PGE2 synthesis and NO production. | 2003 | Jang et al. [287] |
| Human chondrocytes stimulated with interleukin (IL)-1β; Harpagophytum dry extract (210, 480 mg); immunofluorescence and Western blot analyses showed dose-dependent suppression of matrix metalloproteinases production via inhibition of cytokine expression. | 2004 | Schulze-Tanzil et al. [288] |
| Bovine and human chondrocytes, stimulated with LPS and IL-1β, respectively; Harpagophytum extracts (100, 33, 1 µg/mL); significant suppression of PGE2 expression and NO synthase in human chondrocytes (bovine experiment was flawed). | 2006 | Chrubasik [289], Chrubasik et al. [290], Hadzhiyski et al. [291] |
| Human whole-blood assay, human polymorph nuclear leucocytes (PMNL) assay; COX-2, 5-lipogenase (LOX) inhibition, respectively; comparison of Harpagophytum aqueous-ethanolic and CO2 extracts (2%, 20%, and 30% harpagoside, respectively); variable but weak PGE2 inhibition for all, superiority of CO2 extracts in 5-LOX inhibition. | 2006 | Günther et al. [292] |
| Human HepG2 hepatocarcinoma and RAW 264.7 macrophage cell lines; harpagoside (0.1–200 µM); LPS-induced mRNA, COX-2 expression, and inducible nitric oxide (iNOS) inhibited, and NF-κB activation suppressed. | 2006 | Huang et al. [293] |
| LPS-stimulated THP-1 cells; incubated with 50 µg/mL H. procumbens dry extract (DER 1.5–3); microarray (gene chip) assay; noted inhibition of several inflammatory targets. | 2009 | Balthazar et al. [294] |
| COX-2 (ovine) enzyme, stimulated by arachidonic acid and TMPD; H. procumbens extract, harpagoside, and harpagide; direct inhibition (68%) of COX-2, harpagoside, and harpagide contributed 1.5% and 13%, respectively. | 2011 | Ebrahim and Uebel [295] |
| Isolated murine macrophages; H. procumbens crude methanolic extract, harpagoside, phenylethanoid-containing fraction, verbascoside; strong inhibitory action related to NO and TNF-α and IL-6 production, and COX-1 and COX-2 expression, comparable to harpagoside. | 2011 | Gyurkovska et al. [296] |
| LPS-stimulated human monocytes and mouse RAW264.7 macrophages; molecular targets; H. procumbens ethanolic extract (2.9% harpagoside); dose-dependent inhibition of TNF-α, IL-6, IL-1β, PGE2, and COX-2, inhibition of activator protein (AP)-1 pathway without affecting NF-κB and mitogen-activated protein (MAP) kinase pathways. | 2012 | Fiebich et al. [297] |
| Pre-transdermal and post-transdermal COX-2 inhibition and permeation studies; H. procumbens extract, harpagoside, harpagide; hydroxypropyl cellulose gels (carrier) with permeation enhancers tested on synthetic membranes, with and without enhancers on human skin, Azone® enhancer chosen, direct COX-2 inhibition maintained (pre-permeation 80%, post-permeation 77% COX-2) | 2013 | Ebrahim [298] |
| LPS-stimulated monocytic THP-1 cells; enzyme-linked immunosorbent assays, WST-1 assay; Harpagophytum extract; dose-dependent suppression of TNF-α, IL-6, IL-8, independent from external metabolic activation. | 2014 | Hostanka et al. [299] |
| Primary human osteoarthritis chondrocytes; harpagoside (600 µM); significant reduction in IL-1β-induced expression of IL-6, no effect on nuclear levels of NF-κB. | 2015 | Haseeb et al. [300,301] |
| Differentiated 3T3-L1 adipocytes; harpagoside; activation of peroxisome proliferator-activated receptor (PPAR)-γ, significant inhibition of TNF-α-induced mRNA synthesis and production of atherogenic adipokines including IL-6, plasminogen activator inhibitor-1, and monocyte chemoattractant protein-1. | 2015 | Kim et al. [302] |
| IFN-γ/LPS-stimulated THP-1 cells; harpagoside and harpagide; decreased TNF-α-secretion in PMA-differentiated THP-1 cells, positive effect on TNF-α and intercellular adhesion molecule-1 mRNA-expression in undifferentiated cells. | 2016 | Schopohl et al. [303] |
| Human synovial membranes from subjects with and without osteoarthritis; H. procumbens extract, multiple solvents; cannabinoid type 2 (CB2) receptor enhanced, phosphatidylinositol-specific phospholipase C β2 downregulated with water and DMSO, fatty acid amide hydrolase (FAAH) activity inhibited with all. | 2020 | Mariano et al. [304] |
| Study | Year | Reference |
|---|---|---|
| Formaldehyde-induced arthritis in rats; Harpagophytum * infusion p.o. and subcutaneous; significant reduction of swelling, subcutaneous application better tolerated. | 1958 | Zorn [116] |
| Albumin-induced paw edema, granuloma-pouch-test, formaldehyde-induced arthritis in rats, rabbit ear-withdrawal test; whole extract and harpagoside, intravenous (i.v.) and i.p.; some (significant) effects shown similar to those of phenylbutazone. | 1970 | Eichler and Koch [305] |
| Rats; blood panel; Harpagophytum aqueous extract 3:1, 30 mg/kg; triglycerides, uric acid, urea, and cholesterol significantly reduced. | 1974 | Int. Bio Research [306] |
| Dextran-induced paw edema; rats; Harpagophytum, aqueous extract 3:1; edema significantly reduced. | 1974 | Int. Bio Research [307] |
| Eight Harpagophytum dry extracts, p.o. and i.v., tested for analgesic and antiphlogistic effects in five animal models; some analgesic and antiphlogistic effects with methanolic, butanolic, and fluid extracts; pure harpagoside superior, semi-chronic models showed better results. | 1978 | Erdös et al. [308] |
| Carrageenan-induced rat paw edema (30) and adjuvant-induced arthritis in rats (40); Harpagophytum 100–1000 mg/kg, single dose and 21 days; no significant effect in the edema model, some effect in the arthritis model at the higher dose. | 1979 | McLeod et al. [309] |
| Carrageenan-induced rat paw edema; aqueous ethanolic crude extract of Harpagophytum and various fractions; only crude extract effective, concludes that harpagoside is likely not the (only) active. | 1986 | Duband [274] |
| Carrageenan-induced rat paw edema; methanolic extract of Harpagophytum; dose-dependent edema inhibition. | 1990 | Mánez et al. [310] |
| Carrageenan-induced rat paw edema; aqueous extract of Harpagophytum (1.8% harpagoside) and harpagoside i.p.; significant reduction of edema with extract, not with harpagoside. | 1992 | Lanhers et al. [311] |
| Adriamycin-induced rat paw edema; Harpagophytum, 37, 370, and 3700 mg/kg; dose-dependent edema inhibition up to 48% after one hour; compared to control (Adriamycin only) effect transient after 5 days. | 1992 | Jadot and Lecomte [312] |
| Carrageenan-induced mouse paw edema and TPA-induced mouse ear edema; harpagoside (p.o. and topically); no notable protective effects. | 1994 | Del Carmen Recio et al. [313] |
| Carrageenan-induced rat paw edema; aqueous extracts of Harpagophytum (400 and 800 mg/kg, 2.72% harpagoside) i.p. pre-treatment, p.o., and intraduodenally; significant inhibition i.p. and intraduodenally, no effect orally. | 1994 | Soulimani et al. [314] |
| Carrageenan-induced mouse paw edema; Harpagophytum and Uncaria tomentosa extracts; no effect on inflammatory response individually, but significant effect combined. | 2002 | Abe et al. [315] |
| Freund’s adjuvant-induced arthritis in rats; acute (25, 50, or 100 mg/kg) or chronic (100 mg/kg) treatments with H. procumbens solution; increased ‘latency of paws’ withdrawal and reduction in paw edema, compared to control. | 2004 | Andersen et al. [316] |
| Fresh egg albumin-induced pedal edema in rats, hot-plate and acetic acid tests in mice; H. procumbens root aqueous extract (50–800 mg/kg i.p.); significant effect against nociceptive pain stimuli and significant, dose-dependent reduction of edema. | 2004 | Mahomed and Ojewole [317], Mahomed [318] |
| Carrageenan-induced back-paw edema, Freund’s adjuvant-induced arthritis, cotton pellet-induced granuloma, and writhing tests in rats and mice; Harpagophytum aqueous extract (800 mg/kg bw), acetyl salicylic acid and indomethacin as controls; significant effects in all models similar to indomethacin and acetyl salicylic acid. | 2005 | Ahmed et al. [319] |
| TPA-induced COX-2 expression in mouse skin; Harpagophytum methanolic extract (200, 400 µg) topically prior to TPA application; significant inhibition of COX-2 expression, COX-1 unchanged, no effect on NF-κB. | 2005 | Kundu et al. [320] |
| Carrageenan-induced back-paw edema in rats; H. procumbens extract (100, 200, 400, or 800 mg/kg) p.o. and i.p.; reduced intensity of inflammatory response when given i.p. | 2006 | Catelan et al. [321] |
| Adult female white New Zealand rabbits, anterior cruciate ligament transected, and medial meniscus removed; Harpagophytum extract (150 mg/day), standard food pellets as control; outcome suggests suppression of metalloproteinase-2 production. | 2006 | Chrubasik et al. [322], Chrubasik [289] |
| Male ICR mice; formalin test; Harpagophytum extract (1.9% harpagoside, 30–300 mg/kg); significant dose-dependent attenuation of licking/biting and spinal nitrites/nitrates. | 2008 | Uchida et al. [323] |
| Rabbits after unilateral meniscectomy and transection of the anterior cruciate ligament; thickness, surface area, and volume of the tibial condylar cartilage per MRI; H. procumbens extract (14% harpagoside); difference in thickness and volume between healthy and operated leg slightly but not significantly smaller with Harpagophytum. | 2011, 2014 | Wachsmuth et al. [324], Wrubel [325] |
| BALB/c mice infected with Salmonella enteritidis; leukocytes, neutrophils, and mononuclear cell counts, TNF-α, IL-4, 10, 12, histopathological analysis of the liver and small intestine; H. procumbens extract (150 µg/day); downregulation of cell counts, TNF-α, IL-10 m 12, IL-4 increased, histopathology of liver unchanged, hypertrophy in the small intestine, reduced with Harpagophytum. | 2014 | Bisinotto [326] |
| Male SD rats; plantar incision and spared nerve injury; mechanical withdrawal threshold (MWT) test and ultrasonic vocalization (USVs); H. procumbens ethanolic extract (300 mg/kg, p.o.); MWT significantly increased, USVs reduced. | 2014 | Lim et al. [327] |
| Rats; carrageenan-induced mechanical allodynia and thermal hyperalgesia, involvement of the hemeoxygenase (HO)-1/carbon monoxide (CO) pathway; H. procumbens extract (300 and 800 mg/kg i.p.); pretreatment with HO inhibiter reduced anti-hyperalgesic effect, pretreatment with hemin- or CO-releasing molecule induced antiallodynic response. | 2015 | Parenti et al. [328] |
| Rats; formalin-induced damage to cartilage tissue; combination of glucosamine hydrochloride, chondroitin sulfate, methylsulfonylmethane, Harpagophytum extract (3% harpagoside), and bromelain extract (500 mg/kg); malondialdehyde, NO, 8-hydroxyguanine, IL-1β, and TNF-α significantly lowered, glutathione significantly increased. | 2015 | Ucuncu et al. [329] |
| Rats, chronic constriction injury (CCI) of left sciatic nerve model; Harpagophytum extract + morphine, each at sub-analgesic dose; significant antiallodynic and anti-hyperalgesic effect suggesting synergistic effect. | 2016 | Parenti et al. [330] |
| Immunological angiogenesis induced by bronchoalveolar lavage (BAL) cells grafted into BALB/c mice skin; ethanolic extract of Harpagophytum, Filipendula ulmaria, and Echinacea purpurea; significant reduction of newly formed blood vessels 1.2 and 0.6 mg daily. | 2016 | Radomska-Lesniewska et al. [331] |
| Study | Year | Reference |
|---|---|---|
| Human whole-blood anticoagulated with heparin; preincubated with Harpagophytum * extract or purified harpagoside; both dose-dependently inhibited cysteinyl-leukotriene and thromboxane B2 release after biotransformation. | 1996, 1997 | Tippler et al. [332,333] |
| Human whole-blood assay (healthy and osteoarthritic) for COX-1 and COX-2 activity and NO production; H. procumbens extract and harpagoside; increased the activity of baseline COX-1 and COX-2 without LPS, crude extract did not alter COX activity; harpagoside inhibited COX-1, COX-2, and NO. | 2007 | Anauate [334] |
| Freshly excised porcine skin; dermal and transcutaneous delivery and effect on COX-2 expression in Western blotting and immunocytochemical assays; Harpagophytum extract in various vehicles, harpagoside, harpagide, 8-coumaroylharpagide, and verbascoside; ratio-dependent inhibition of COX-2 expression, higher penetration of all compounds from ethanol/water. | 2008 | Abdelouahab and Heard [335,336] |
| Freshly excised porcine skin; transcutaneous delivery and effect on COX-2, PGE2, 5-LOX, and inducible NO synthase (iNOS) expression in Western blotting and immunocytochemical assays; commercial Harpagophytum extracts, harpagoside, harpagide, 8-coumaroylharpagide, and verbascoside; ratio-dependent inhibition of COX-2 expression and PGE2, no significant effect on 5-LOX and iNOS, relative proportions of anti- and pro-inflammatory compounds in commercial products varied. | 2009, 2010 | Ouitas and Heard [337,338,339] |
| LPS-stimulated human whole-blood assay (healthy) for COX-1 and COX-2 activity and NO production, incubation of isolated fractions obtained by flash chromatography monitored with HPLC, TLC, and identified by 1HNMR; fractions of H. procumbens extract; highest concentration of harpagoside inhibited COX-1, COX-2, and NO; iridoid pool increased COX-2 while NO and COX-1 activities remained unchanged, fraction containing cinnamic acid reduced NO only. | 2010 | Anaute et al. [340] |
| Study | Type | Year | Reference |
|---|---|---|---|
| Carrageenan-induced rat paw edema and adjuvant-induced arthritis in rats; arachidonic acid and prostaglandin synthetase incubated with various concentrations of indomethacin, acetylsalicylic acid, or Harpagophytum extract (not specified); no effect on edema, anti-inflammatory activity is not mediated by the inhibition of the prostaglandin synthetase. | In Vitro and in vivo | 1983 | Whitehouse et al. [341] |
| Cultured human mammary epithelial cells and female ICR mice; TPA-induced COX expression; Harpagophytum methanolic extract (10, 5, 1 µg/mL, 600, 300, 60 µg, respectively); inhibition of COX-2 expression in both models. | In Vitro and in vivo | 2004 | Na et al. [342] |
| Rat adjuvant-induced chronic arthritis model, LPS-stimulated mouse macrophage cells (RAW 264.7); Harpagophytum ethanolic extract; significant anti-inflammatory effect, and dose-dependent suppression of, IL-6 and TNF-α, respectively. | In Vitro and in vivo | 2010 | Inaba et al. [343] |
| Molecular docking study of harpagoside and harpagide with COX-2; binding energies were −9.13 and −5.53 kcal/mol respectively, finding both harpagoside and harpagide to be highly selective COX-2 inhibitors. | Simulation | 2016 | Rahimi et al. [344] |
| Mouse myoblast C2C12, human colorectal adenocarcinoma HCT116 cell lines, isolated rat colon challenged with LPS; aqueous Harpagophytum extract (1–1000 μg/mL); HCT116 viability reduced, ROS production in both cell lines reduced, PGE2, 8-iso-PGF2α, serotonin, and TNF-α production inhibited. | In Vitro and ex vivo | 2017 | Locatelli et al. [345], Leporini et al. [346] |
| Antioxidant capacity, leukocyte ROS production, COX-2/PGE2 pathway or cytokine secretions; H. procumbens methanolic extract; decreased the secretion of IL-21 and IL-23, increased TNF-α, IL-8, and IFN-γ, immune-stimulant effect. | In Vitro and ex vivo | 2019 | Cholet et al. [347] |
| LPS-stimulated wild-type (C57/BL6) male mice colon and HCT116 cells; experimental model of inflammatory bowel disease; H. procumbens aqueous extract; anti-inflammatory, antioxidative, and antimicrobial effects (against pathogen fungal strains), morphological alterations in the colon tissue indicated. | In Vitro and ex vivo | 2020 | Recinella et al. [348] |
| IC50 | Reference | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Extract/Fraction | Harpagoside (%) | Cys-LT | TXB2 | Enzyme Inhibitors | IL-6 | IL-1β | NF-κB | COX-2 | |
| Special extract WS1531 | 7.3 | 9.2 µM/L | 55.3 µM/L | [332,333] | |||||
| 7.3 | 62 µg/mL | 373 µg/mL | [285] | ||||||
| Aqueous ethanolic H. procumbens extract | 2.1 | 1450 µg/mL | - | [285] | |||||
| 542 μg/mL (HNE) | [199,201] | ||||||||
| 547.69/601.49 µg/mL (MPO) * | [349] | ||||||||
| <100 µg/mL | [297] | ||||||||
| H. procumbens tincture | 915.55/776.49 µg/mL (MPO) * | [349] | |||||||
| Aqueous ethanolic H. zeyheri extract | 1012 µg/mL (HNE) | [199,201] | |||||||
| Aqueous H. procumbens extract | 8.9 | 0.55 µg/mL | [350] | ||||||
| 27 | 0.2 µg/mL | [350] | |||||||
| Ethanolic H. procumbens extract | 65.5 µg/mL (FAAH) | [304] | |||||||
| Ethyl acetate fraction of aqueous ethanolic H. procumbens extract | 19.95 | 391 µg/mL | - | [285] | |||||
| Butanol fraction of aqueous ethanolic H. procumbens extract | 19.5 | 565 µg/mL | 203 µg/mL | [285] | |||||
| Methanolic H. procumbens extract | 1046 µg/mL | [295] | |||||||
| H. procumbens extracts and isolates | ~125 µg/mL | [296] | |||||||
| Isolated compounds | |||||||||
| Harpagoside | 30 µM/L | 48.6 µM/L | [332,333] | ||||||
| 39 μM/L | 49 μM/L | [285] | |||||||
| 1041 µg/mL | [295] | ||||||||
| >600 µg/mL (HNE) | [199,201] | ||||||||
| 92.7 µM (AChE) | [351] | ||||||||
| 95.6 µM (AChE) | [351] | ||||||||
| 96.4 µM | [293] | ||||||||
| 14.04 µM | [302] | ||||||||
| Harpagide | 1186 µg/mL | [295] | |||||||
| 8-PCHG | 179 µg/mL (HNE) | [199,201] | |||||||
| 95.6 µM (AChE) | [351] | ||||||||
| Pagoside | 154 µg/mL (HNE) | [199,201] | |||||||
| Caffeic acid | 86 µg/mL (HNE) | [199,201] | |||||||
| Acetoside | >500 µg/mL (HNE) | [199,201] | |||||||
| 19.9 µM (AChE), 35 µM (BChE) | [351] | ||||||||
| Isoacetocide | 179 µg/mL (HNE) | [199,201] | |||||||
| 21.6 µM (AChE), 29.7 µM (BChE) | [351] | ||||||||
| Decaffeoylverbascoside | 16.1 µM (AChE), 46 µM (BChE) | [351] | |||||||
| 6′-O-Acetylacteosid | 47 µg/mL (HNE) | [199,201] | |||||||
| Effect | Study | Type | Year | Reference |
|---|---|---|---|---|
| Antioxidant | Rats, Harpagophytum * extract, 100 and 200 mg/kg bw or selegiline i.p. for 1, 7, or 14 days; dose-dependent increase of superoxide dismutase, catalase, and glutathione peroxidase activities and reduction of lipid peroxidase similar to selegiline after 7 days. | In Vivo | 1998 | Bhattacharya and Bhattacharya [352] |
| Luminol-enhanced chemiluminescence in a xanthine/xanthine oxidase cell-free system; Harpagophytum root powder; superoxide and peroxyl were scavenged dose-dependently. | In Vitro | 2002 | Langmead et al. [353] | |
| Trolox equivalent antioxidant capacity (TEAC) assay; Harpagophytum aqueous extract (2.6% harpagoside) and harpagoside; extract rich in water-soluble antioxidants, harpagoside showed poor activity. | In Vitro | 2003 | Betancor-Fernandez et al. [354] | |
| Rat renal mesangial cells; IL-1β-induced NO formation and transcriptional regulation of iNOS; H. procumbens extracts with varying harpagoside content and pure harpagoside; dose-dependent and harpagoside-independent inhibition of iNOS expression. | In Vitro | 2004 | Kaszkin et al. [350] | |
| Harpagophytum aqueous extract; protection from DNA-damaging effects of stannous chloride in proficient and deficient E. coli model; possible chelating, scavenger, or oxidant activity postulated. | In Vitro | 2007 | Almeida et al. [355] | |
| Antioxidant characteristics using in vitro test systems, DPPH radical scavenging, stimulated nitrite generation, neutrophil superoxide anion generation, and neutrophil myeloperoxidase (MPO); Harpagophytum extract (1.2% harpagoside), tincture, harpagoside; dose-dependent effect in all models, minimal scavenging activity of harpagoside. | In Vitro | 2005, 2009 | Grant et al. [349], Grant [356] | |
| Antioxidant activities of total methanol extracts, fractions (phenylethanoids, terpenoids, and sugars), and β-OH-verbascoside, verbascoside, and leucosceptoside from cell suspension culture of H. procumbens; DPPH, superoxide anion generation, and oxygen radical absorbance capacity (ORAC) assays; β-OH-verbascoside most active in DPPH and superoxide anion generation, leucosceptoside in ORAC. | In Vitro | 2010 | Georgiev et al. [357] | |
| Ferric-reducing antioxidant power test; H. procumbens crude methanolic extract, phenylethanoid-containing fraction, and verbascoside; strong ferrous ion-chelating capacity. | In Vitro | 2011 | Georgiev et al. [358] | |
| Brain homogenates, catalase activity and thiol levels, brain cortical slices; lipid peroxidation, antioxidant defenses, cell damage, respectively; H. procumbens infusion, crude extract, and fractions; dose-dependent inhibition of lipid peroxidation, ethyl acetate fraction had the highest antioxidant effects. | In Vitro | 2013 | Schaffer et al. [359,360] | |
| Human neutrophils challenged with phorbol myristate acetate (PMA), opsonized Staphylococcus aureus, and Fusobacterium nucleatum; 5 taxa of Harpagophytum, including one hybrid; high variability in suppression of respiratory burst, hybrid with highest antioxidant capacity but proinflammatory effect, three taxa with anti-inflammatory effect. | In Vitro | 2016 | Muzila et al. [361] | |
| Adult male Wistar rats, fluphenazine-induced orofacial dyskinesia (OD); DPPH assay; ethyl acetate fraction of H. procumbens (10, 30, or 100 mg/kg i.p.); inhibition of vacuous chewing movements, decreased locomotion unchanged, protective against change in catalase activity, not against ROS increase. | In Vivo | 2016 | Schaffer et al. [362] | |
| Porcine neutrophils; respiratory burst; harpagoside; significant inhibition of ROS production. | In Vitro | 2017 | Mosca et al. [363] | |
| Male Sprague–Dawley rats, modified rodent contusion model of spinal cord injury, murine BV-2 microglial cells; H. procumbens hydroethanolic extract (5.3% harpagoside, 300 mg/kg); behavioral and neurochemical parameters, improved, some significantly, in cell line, oxidative stress and inflammatory response were suppressed. | In Vitro and in vivo | 2020 | Ungerer et al. [364] | |
| LPS-induced RAW 264.7 mouse and U937 human macrophages; DPPH and 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulphonic acid) (ABTS) assays; aqueous, ethanolic, and ethyl acetate extracts of H. zeyheri; for all extracts, dose-dependent inhibition of IL-10 expression, ethyl acetate fraction with lowest IC50 in both assays, NO and TNF-α inhibition similar to diclofenac. | In Vitro | 2021 | Ncube et al. [365] | |
| Antidiabetic | Streptozotocin-induced diabetes mellitus in rats; H. procumbens root aqueous extract (50–800 mg/kg i.p.); significant reduction in blood glucose levels in normal and diabetic rats. | In Vitro | 2004 | Mahomed and Ojewole [317], Mahomed [318] |
| Anticholinesterase | Chick, guineapig, and rabbit isolated gastro-intestinal smooth muscle preparations; H. procumbens root aqueous extract (10–1000 µg/mL); dose-dependent contractions of gastro-intestinal tract smooth muscles. | In Vitro | 2005 | Mahomed [318], Mahomed et al. [366] |
| Spectrophotometric method using acetylthiocholine and butyrylcholine chloride as substrates; H. procumbens crude methanolic extract, phenylethanoid-containing fraction, and verbascoside; significant cholinesterase inhibitory activity. | In Vitro | 2011 | Georgiev et al. [358] | |
| Spectrophotometric method, acetylcholinesterase (AchE) and butyrylcholinesterase (BchE) inhibition; H. procumbens ethyl acetate extract and fractions; inhibition by verbascosides > 60% | In Vitro | 2013 | Bae et al. [351] | |
| Antimicrobial | Harpagophytum extract (not specified) showed mild antifungal effects against Penicillum digitatum and Botrytis cinerea. | In Vitro | 1985 | Guérin and Réveillère [367] |
| Harpagophytum dry extract (2.6% harpagoside) and harpagoside; inhibition of a panel (all) of aerobic bacteria, C. krusei, and two anaerobic bacteria strains, harpagoside without effect. | In Vitro | 2007 | Weckesser et al. [368] | |
| Chloroquine (CQ)-sensitive and CQ-resistant strains of P. falciparum, and cytotoxicity in CHO and HepG2 cells; extracts of H. procumbens aerial parts and seeds, and petrol ether of the root, (+)-8,11,13-totaratriene-12,13-diol and ferruginol, and CQ diphosphate as control; the two diterpenes showed significant inhibition of both strains without being cytotoxic. | In Vitro | 2003 | Clarkson et al. [200] | |
| Female Balb/c mice, infected with Toxocara canis; Harpagophytum ethanolic extract (100 mg/kg); decrease in eosinophil accumulation, IL-5 and IgE significantly decreased. | In Vivo | 2012, 2014 | Oliveira et al. [369,370,371] | |
| Harpagophytum ethanolic extract showed dose-dependent effect on Schistosoma mansoni, mechanism of action proposed; proteins relevant for cellular homeostasis identified as possible targets. | In Vitro | 2014 | Correia [372] | |
| Bacterial triggers of rheumatoid arthritis, ankylosing spondylitis, multiple sclerosis, and rheumatic fever; powdered Harpagophytum extracts, various solvents; inhibition of Proteus mirabilis, Klebsiella pneumoniae, Acinetobacter baylyi, Pseudomonas aeruginosa, and Streptococcus pyogenes throughout, methanolic extract more potent, no toxicity in Artemia nauplii bioassay. (Note: throughout the publication, the substance of investigation is mislabeled as devil’s claw fruit, while it was, in fact, the root being investigated (pers. comm. Ian Cock, 2021)) | In Vitro | 2017 | Cock and Bromley [373] | |
| Antimutagenic | Cultured human lymphocytes; mutagenic activity of 1-nitropyrene (1-Npy) in cytokinesis-block micronucleus assay; Harpagophytum aqueous-ethanolic extract, harpagoside; genotoxicity significantly reduced for both, only harpagoside significantly reduced the mutagenicity of 1-Npy. | In Vitro | 2014, 2015 | Luigi [374], Luigi et al. [375] |
| Anti-osteoporotic | Male ICR mice, female C57BL/6J mice; receptor activator of nuclear factor κ-Β ligand (RANKL)-induced osteoclastogenesis; harpagoside; inhibition of RANKL, osteoclast formation, and LPS-induced bone loss, but not ovariectomy-mediated bone erosion. | In Vitro | 2015 | Kim et al. [376] |
| Mouse calvaria MC3T3-E1cells; bone formation and resorption, bone-loss in ovariectomized (OVX) mouse model; harpagide; stimulated differentiation and maturation of osteoblast cells and suppressed RANKL-induced osteoclastogenesis, improved bone recovery in OVX model, inhibited markers of bone loss in the serum. | In Vitro and in vivo | 2016 | Chung et al. [377] | |
| Mouse calvaria MC3T3-E1cells; bone formation and resorption, bone-loss in ovariectomized (OVX) mouse model; harpagoside; stimulated differentiation and maturation of osteoblast cells and suppressed RANKL-induced osteoclastogenesis, improved bone recovery in OVX model, inhibited markers of bone loss in the serum. | In Vitro and in vivo | 2017 | Chung et al. [378] | |
| Cardiovascular | Frog and guineapig hearts, cats; cardiac muscle contraction and blood pressure, dose-dependent positive and negative inotropic effects, no effect on blood pressure. | In Vitro and in vivo | 1965 | Vollmann [379] |
| Normotensive rats, rabbit heart; methanolic extract of Harpagophytum, harpagoside, harpagide; decrease in blood pressure and heart rate observed, less with harpagoside; extract mild inotropic at lower and negative inotropic at higher doses, harpagoside more negative chronotropic and positive inotropic, harpagide only slightly negative chronotropic but considerably negative inotropic. | In Vitro and in vivo | 1984 | Circosta et al. [380] | |
| Rat heart; methanolic extract of Harpagophytum (8.5% harpagoside and 10.5% total iridoids) and harpagoside; significant, dose-dependent, protective action toward hyperkinetic ventricular arrhythmias. | In Vitro | 1985 | De Pasquale et al. [381] | |
| Langendorff preparations of rat heart; ischemic perfusion induced hyperkinetic ventricular arrhythmia; H. procumbens, harpagoside; significant, dose-dependent protective action for both. | In Vitro | 1985 | De Pasquale et al. [382] | |
| Guineapig ileum and rabbit jejunum; Harpagophytum extract, harpagoside, harpagide; spasmolytic effect, strongest for harpagoside. | In Vitro | 1985 | Occhiuto et al. [383] | |
| Dogs; harpagoside, harpagide (3.4 mg/kg); decrease of mean aortic pressure with harpagoside. | In Vivo | 1990 | Occhiuto and de Pasquale [384] | |
| Multiple mammalian animal models; H. procumbens root aqueous extract (10–400 mg/kg i.v., 10–1000 µg/mL); dose-dependent, significant hypotensive, cardio-depressant, and vasorelaxant effects. | In Vitro and in vivo | 2004 | Mahomed and Ojewole [385], Mahomed [318] | |
| Neuroprotective | Pentylenetetrazole (PTZ)-, picrotoxin (PCT)-, and bicuculline (BCL)-induced seizures in mice; H. procumbens aqueous extract (100–800 mg/kg i.p.); PZT-induced seizures significantly reduced, PCT and BCL to a lesser extent, CNS depressed. | In Vivo | 2006 | Mahomed and Ojewole [386] |
| Rat hypothalamic (Hypo-E22) cells and rat cortex challenged with amyloid β-peptide; H. procumbens aqueous extract; increased brain-derived neurotrophic factor gene expression and decreased TNF-α gene expression in Hypo-E22 cells, alleviated decreased monoaminergic signaling in cortex presynaptic endings. | In Vitro and ex vivo | 2017 | Ferrante et al. [387] | |
| Male Wistar rats; chronic cerebral hypoperfusion model; harpagoside (15 mg/kg, 60 days); symptoms of vascular dementia spatial and fear memory impairments restored, phosphatase and tensin homolog (PTEN) significantly suppressed. | In Vivo | 2018 | Chen et al. [388] | |
| Female Wistar albino rats, arsenic induced neurotoxicity; Harpagophytum powder (200 and 400 mg/kg, p.o.); behavioral and biochemical parameters improved significantly. | In Vivo | 2020 | Peruru et al. [389] | |
| Immunomodulatory/thymomimetic | Maturation of mice thymocytes in the presence of a glycocorticosteroid, cytotoxicity by microscopy and flow cytometry; ethanolic extract of Harpagophytum, Filipendula ulmaria, and Echinacea purpurea, various dilutions; 17% increase in the number of surviving cells. | In Vitro | 2002 | Prosinska et al. [390] |
| Anorexigenic | Male C57BL/6 mice, calcium mobilization and growth hormone secretagogue receptor (GHS-R1a) internalization; Harpagophytum root powder; significantly increased cellular calcium influx but no induction of GHS-R1a receptor internalization, significant anorexigenic effect. | In Vivo | 2014 | Torres-Fuentes et al. [391] |
| Male Wistar rats; obestatin secretion; Harpagophytum hydroalcoholic extract (150, 300, and 600 mg/kg); significantly increased serum levels of obestatin and reduced body weight at 300 and 600 mg/kg. | In Vivo | 2016 | Saleh et al. [392] | |
| Metal accumulation | Rats, supplemented with lead acetate; Harpagophytum infusion (30 mg/kg); significant reduction of lead deposits | In Vitro | 1975 | Int. Bio Research [393] |
| Title | Date | Number |
|---|---|---|
| Food supplement | 4/3/1984 | US19810287235 |
| Therapeutically active mixture | 11/8/1984 | DE19833316726 |
| Homeopathic remedy for the treatment of rheumatic disorders | 11/19/1987 | DE19863616054 |
| Plant-based medicinal composition for internal use | 4/22/1988 | FR19860014608 |
| Medicinal combination based on plants and trace elements for the treatment of rheumatism and inflammatory states | 11/10/1988 | FR19870006450 |
| Process for the preparation, by extracting, of Harpagophytum | 7/13/1992 | KR19890016112 |
| Anti-pruritic cosmetic composition containing Harpagophytum root extract | 1/27/1993 | EP19920402100 |
| Preparation of concentrated plant extract, particularly from Harpagophytum procumbens | 8/7/1997 | DE1996103788 |
| Harpagosid-angereicherter Extrakt aus Harpagophytum procumbens und Verfahren zu seiner Herstellung [harpagoside- enriched extract of H. procumbens and its manufacture] | 10/2/1997 | DE1996151290 |
| A purified extract from Harpagophytum procumbens and/or Harpagophytum zeyheri, a process for its preparation and its use | 12/18/1997 | |
| Skin care composition contains peroxidized fatty substance, e.g., unsaturated vegetable oils and plant extract | 3/20/1998 | FR19960011438 |
| Natural composition for treating bone or joint inflammation | 11/26/1998 | WO1998US10758 |
| Micro-nutritional compositions having a therapeutic effect containing polyunsaturated fatty acids, trace elements, and vitamins | 7/16/1999 | FR19980000331 |
| A method of producing high anti-inflammatory activity extracts from Harpagophytum procumbens | 10/6/1999 | GB19980006971 |
| Effervescent preparation containing a plant extract | 6/16/1999 | EP0922450A1 |
| Method for producing high activity extracts from Harpagophytum procumbens | 3/6/2001 | US19990280499 |
| Harpagoside-enriched extract from Harpagophytum procumbens and processes for producing the same | 8/28/2001 | US19990155043 |
| Dietary supplement | 12/18/2001 | JP20000172296 |
| Pharmaceutical preparation containing Cibotii rhizoma and Harpagophytum procumbens DC extracts as main ingredients | 6/3/2002 | KR20000071397 |
| Skin care preparation | 6/4/2002 | JP20000402968 |
| Pharmaceutical composition with anti-atherosclerotic activity | 6/5/2002 | EP20010128629 |
| Use of harpagide-related compound as prophylactic and therapeutic agent of osteoporosis, arthritis, and disc and pharmaceutical composition containing compound as effective ingredient | 11/16/2002 | KR20000071497 |
| Composition useful for treating or preventing osteoarthritis, especially in horses, containing extract(s) of Equisetum arvense, Symphytum officinale, and/or Harpagophytum procumbens | 3/27/2003 | DE2001143146 |
| Use of active substance mixtures containing tocopherols and Harpagophytum procumbens extracts for the preparation of a drug against rheumatic arthritis | 12/17/2003 | EP20020012765 |
| Chewing gum composition with vegetal additives | 7/29/2004 | WO2003EP14600 |
| Pain-relieving agent containing extract of Harpagophytum procumbens, Corydalis turtschanovii, and Atractylodes japonica | 2/5/2005 | KR20030052489 |
| Treating or preventing renal diseases, dysfunction, and/or damage, e.g., degenerative and/or inflammatory renal disease, using Harpagophytum extract or harpagoside | 3/10/2005 | DE2003126556 |
| Phyto-composition for the treatment of articular diseases | WO2005092355 | |
| Use of devil’s claw (Harpagophytum procumbens) root extracts for endometriosis treatment | 11/2/2006 | WO2006EP61831 |
| A method for separating harpagide from Harpagophytum procumbens | 2/5/2007 | KR20050102609 |
| Activator of peroxisome proliferator-activated receptor (PPAR) | 5/17/2007 | JP20050317156 |
| Adjuvant composition for physiotherapy | 7/24/2007 | KR20060005183 |
| Maillard reaction inhibitor, skin care preparation containing the same, and food and beverage | 10/4/2007 | JP20060080104 |
| Phyto-composition for the treatment of joint diseases | 12/13/2007 | US20050594439 |
| Natural remedy–dietary supplement combination product | 9/4/2008 | US20060815432 |
| Root extract of Harpagophytum for stimulating hair growth | 5/27/2009 | EP20070802633 |
| Skin care preparation, oral composition, and food and drink | 10/22/2009 | JP20080091677 |
| Novel method for preparing purified extracts of Harpagophytum procumbens | 12/9/2010 | US20080599146 |
| Animal food compositions | 7/21/2011 | WO2010US60804 |
| Compositions comprising plant extracts and use thereof for treating inflammation | 10/27/2011 | US200913120739 |
| Anti-inflammatory composition | 12/21/2011 | EP20110170436 |
| Antirheumatic body cream composition | 12/30/2011 | RO20110000644 |
| Pharmaceutical composition for preventing and treating metabolic bone disease comprising of Harpagophytum | 6/18/2012 | KR20110147135 |
| Phyto-concentrated composition, useful as antispasmodic relaxant, and muscular comfort to, e.g., enhance relaxation of painfully contracted muscle tissue, comprises, e.g., cannabis sativa and an excipient comprising, e.g., castor oil | 10/12/2012 | FR20110001030 |
| Nonabrasive toothpaste containing enzyme papain, Harpagophytum extract d,l-pyrrolidone carboxylate n-cocoyl ethyl arginate, and sodium fluoride | 7/20/2013 | RU20120101119 |
| Cosmetic composition for calming and applying an electric current of skins and manufacturing the same | 12/27/2013 | KR20120065152 |
| Anti-rheumatism medicinal liquor and preparation method thereof | 3/19/2014 | CN20131645408 |
| Composition containing chondroitin sulfate and hyaluronidase | 12/10/2014 | RU20130123301 |
| Mucoadhesive devil’s claw extracts (Harpagophytum procumbens) and uses thereof | 3/11/2015 | EP20140184267 |
| Compositions for alleviating, preventing, or treating pain comprising Harpagophytum procumbens and Acanthopanax senticosus extracts as active ingredients | 6/8/2015 | KR20130146128 |
| Traditional Chinese medicine composite for treating gout | 7/8/2015 | CN20151209743 |
| Cell line cultures from plants belonging to the Harpagophytum genus | 1/4/2018 | WO2017EP65814 |
| Method for preparing purified extracts of Harpagophytum procumbens | 30/10/2018 | US20100311675A1 |
| Oral herbal pain killer formulations | 15/10/2020 | WO2020208395A1 |
| Polyherbal transdermal patch for pain management and its process of preparation | 22/10/2020 | WO2020212820A2 |
| External medicine for inhibiting postoperative venous thrombosis and application thereof | 19/2/2021 | CN109589331B |
| Freedom (nutritional supplement) | 9/2/2021 | US20200060320A1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brendler, T. From Bush Medicine to Modern Phytopharmaceutical: A Bibliographic Review of Devil’s Claw (Harpagophytum spp.). Pharmaceuticals 2021, 14, 726. https://doi.org/10.3390/ph14080726
Brendler T. From Bush Medicine to Modern Phytopharmaceutical: A Bibliographic Review of Devil’s Claw (Harpagophytum spp.). Pharmaceuticals. 2021; 14(8):726. https://doi.org/10.3390/ph14080726
Chicago/Turabian StyleBrendler, Thomas. 2021. "From Bush Medicine to Modern Phytopharmaceutical: A Bibliographic Review of Devil’s Claw (Harpagophytum spp.)" Pharmaceuticals 14, no. 8: 726. https://doi.org/10.3390/ph14080726
APA StyleBrendler, T. (2021). From Bush Medicine to Modern Phytopharmaceutical: A Bibliographic Review of Devil’s Claw (Harpagophytum spp.). Pharmaceuticals, 14(8), 726. https://doi.org/10.3390/ph14080726






