Optimization of Novel Naproxen-Loaded Chitosan/Carrageenan Nanocarrier-Based Gel for Topical Delivery: Ex Vivo, Histopathological, and In Vivo Evaluation
Abstract
1. Introduction
2. Results and Discussion
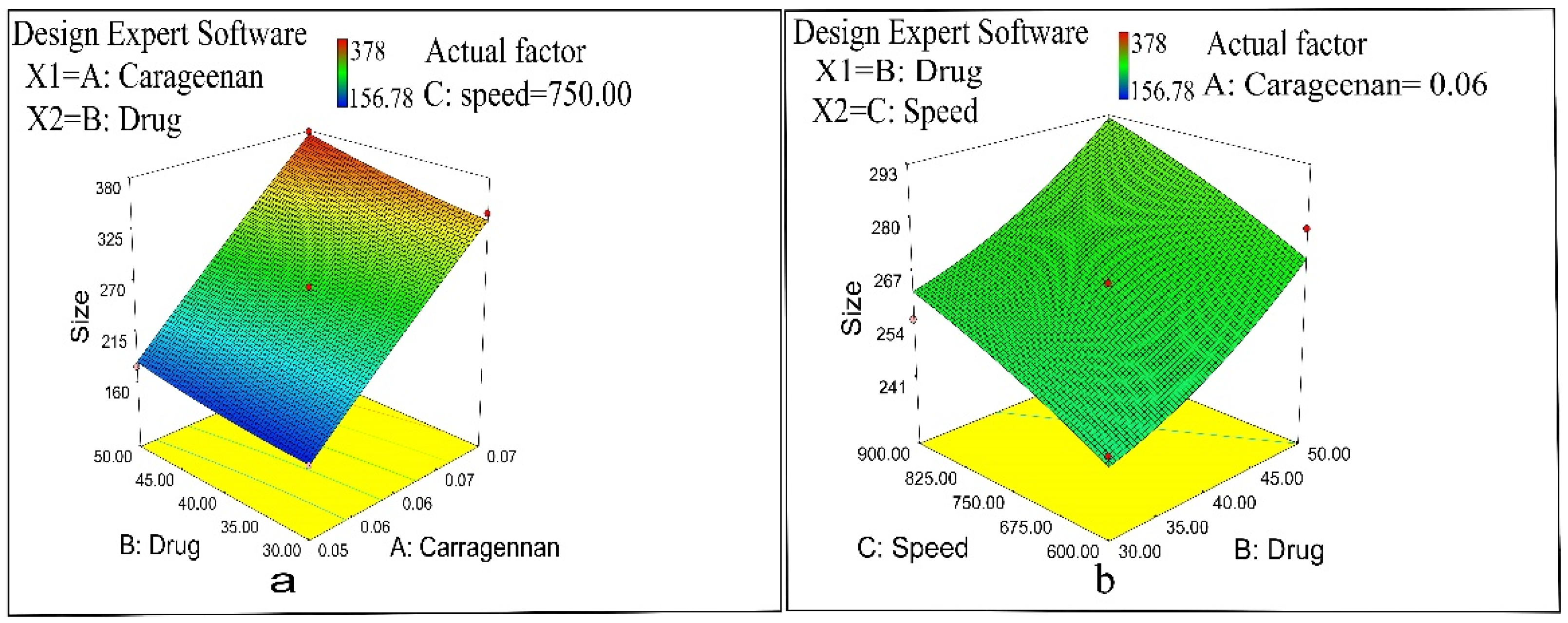
2.1. Preparation and Optimization of Naproxen-Loaded Nanoparticles
2.2. Nanoparticles Characterization
2.2.1. Scanning Electron Microscopic Analysis
2.2.2. FTIR Spectra of Naproxen-Loaded Nanoparticles Formulation
2.2.3. Powdered X-ray Diffraction (pXRD)
2.2.4. Entrapment Efficiency
2.2.5. Zetasizer, Polydispersity Index, and Zeta Potential Determination of NAP-Loaded Nanoparticles
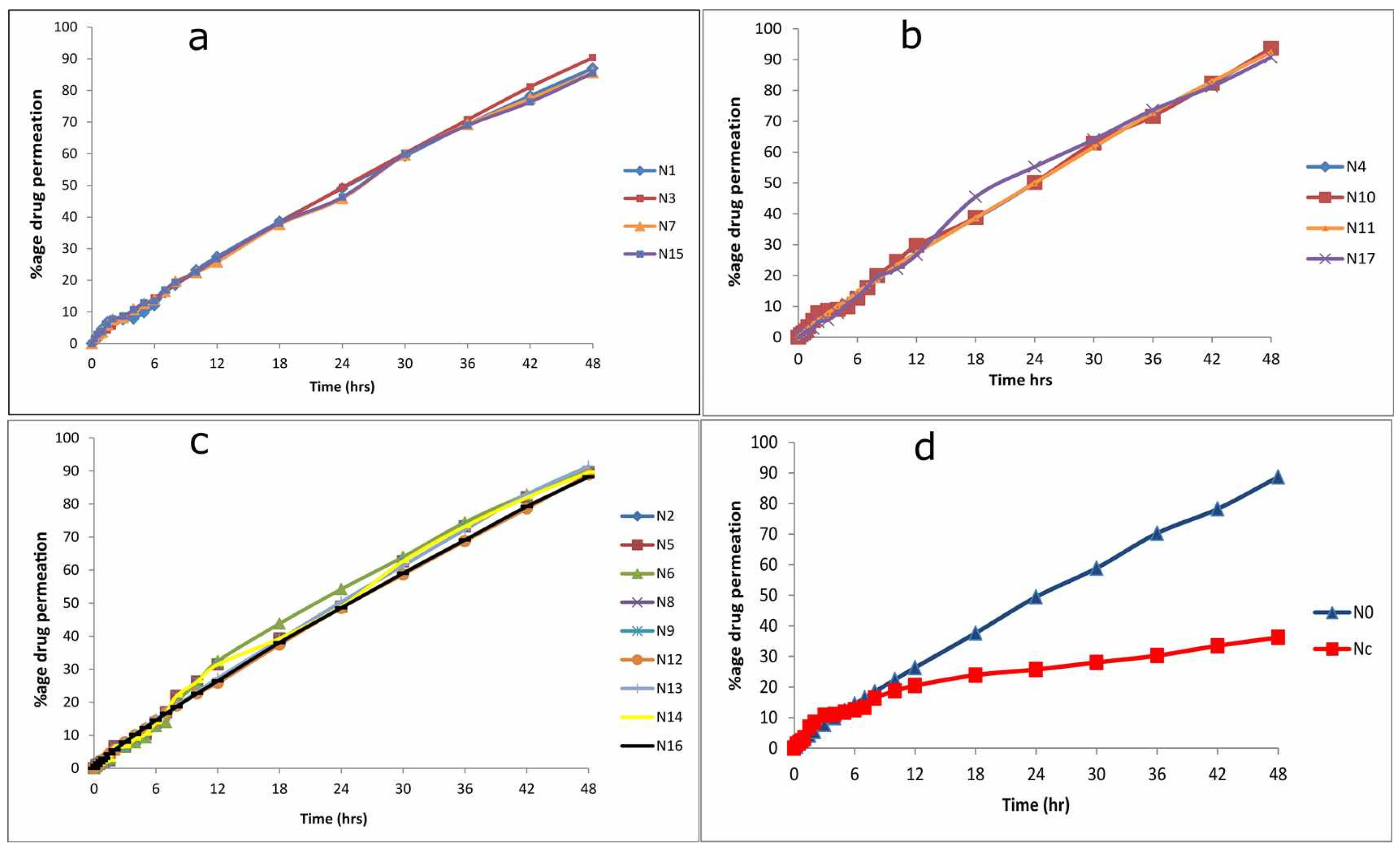
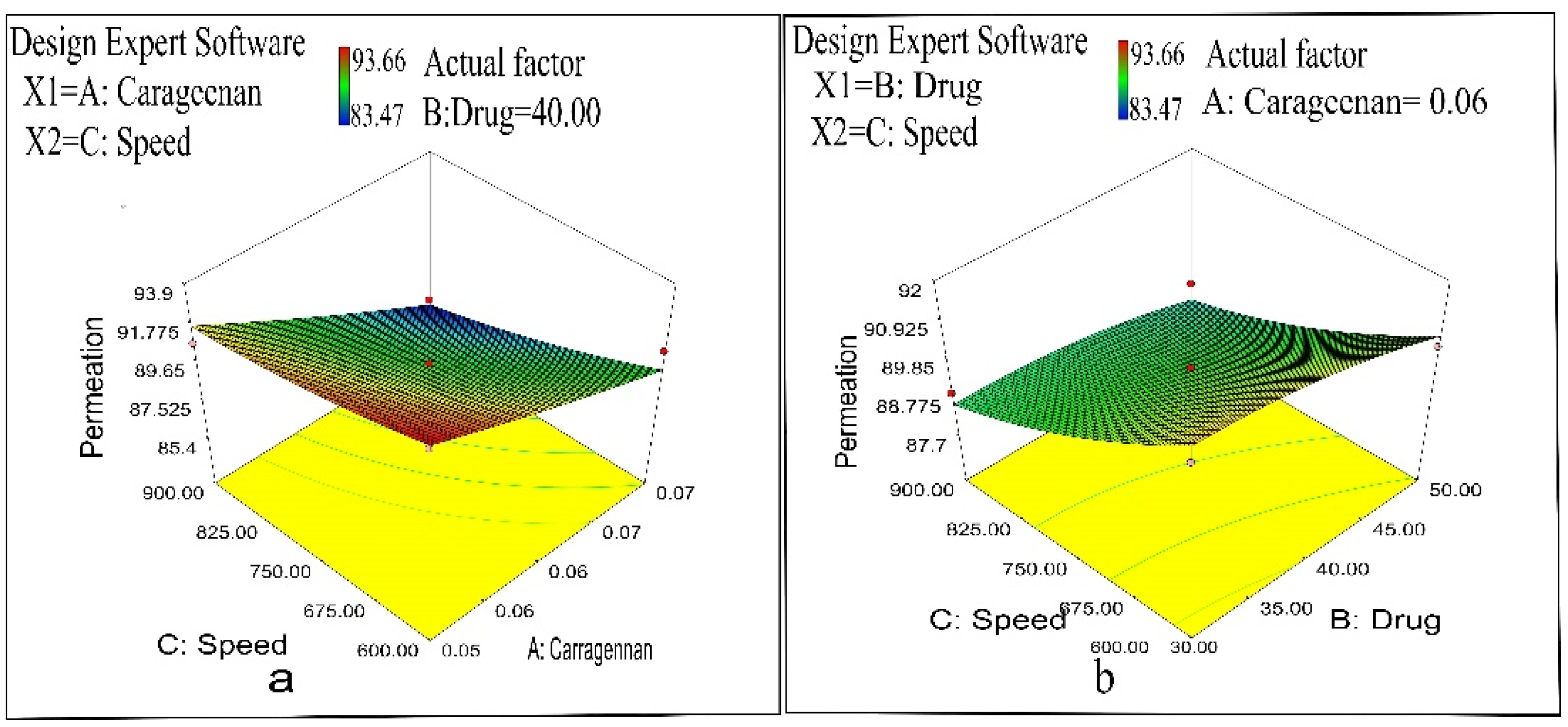
2.3. Ex Vivo Permeation Study
2.4. Characterization of Carbopol 940 Gel
2.4.1. Appearance, Spreadability, pH, Viscosity, and Drug Content
2.4.2. Skin Irritation Studies
2.4.3. Stability Studies for NAP-Loaded Gel
2.5. Kinetics of Drug Permeation
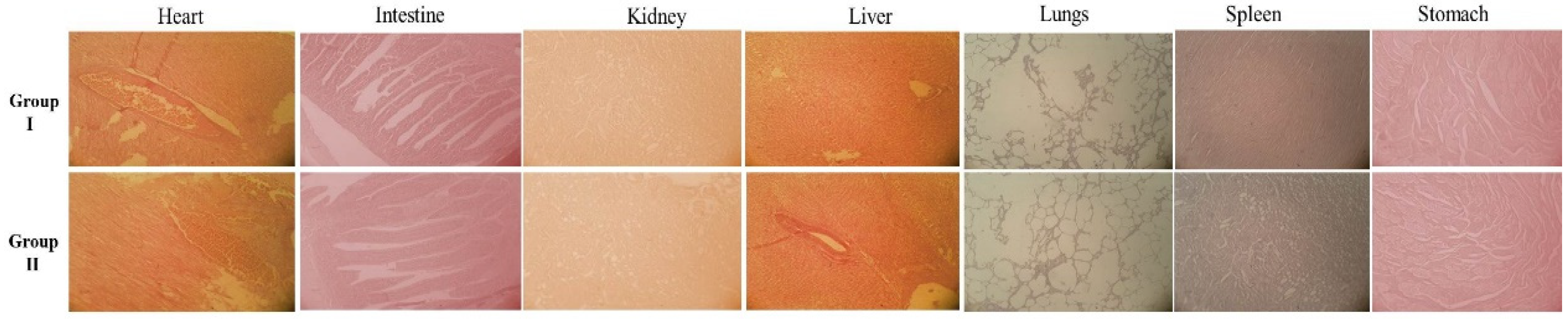
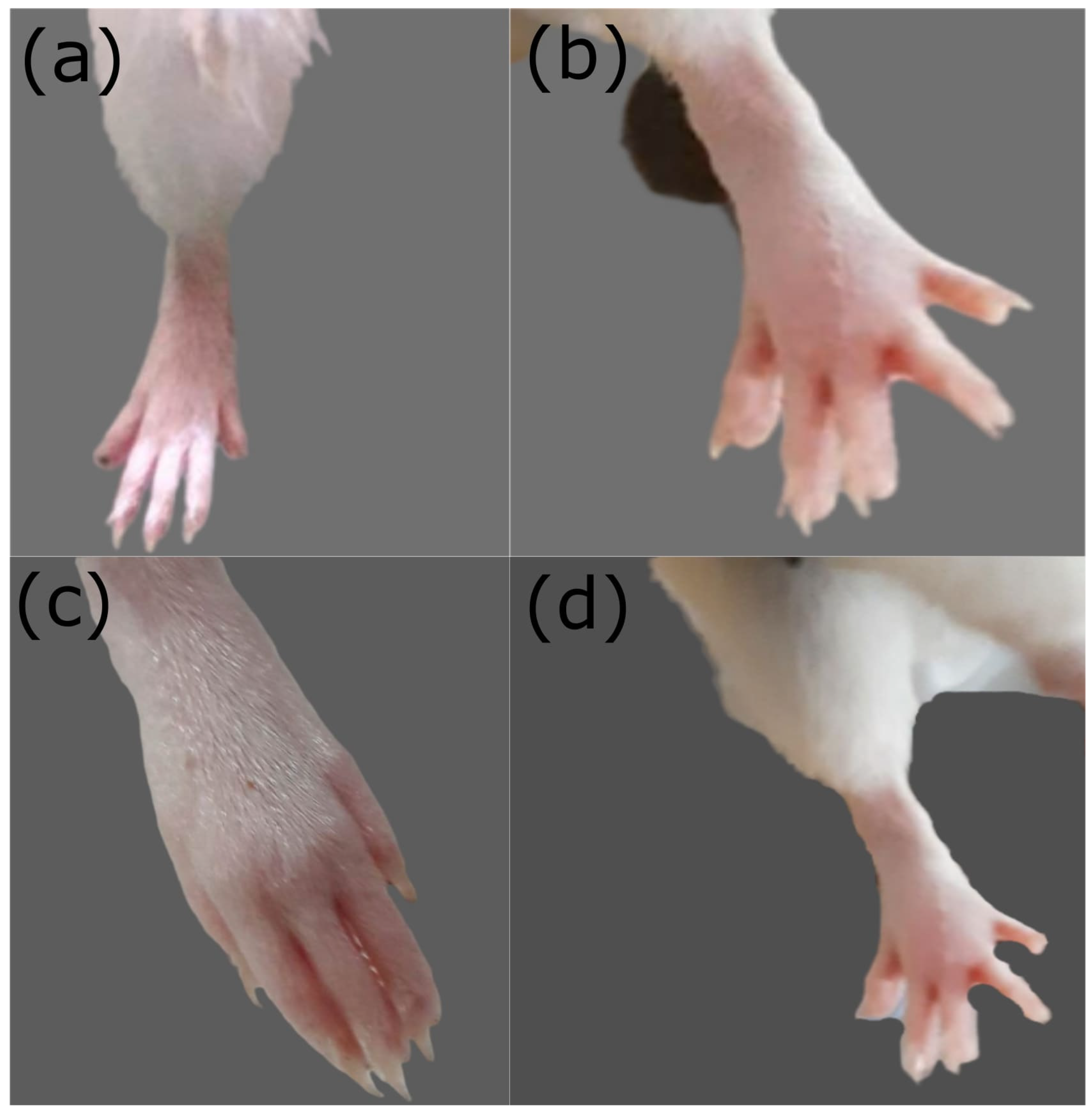
2.6. Toxicity Study in Rabbits
2.7. Optimization
2.7.1. Evaluation of Optimized Formulation of Naproxen-Loaded CS/CRG Nanoparticles
2.7.2. Evaluation of Optimized Formulation of Naproxen-Loaded, CS/CRG-Nanoparticle-Containing Carbopol 940 (Ca-940) Gel
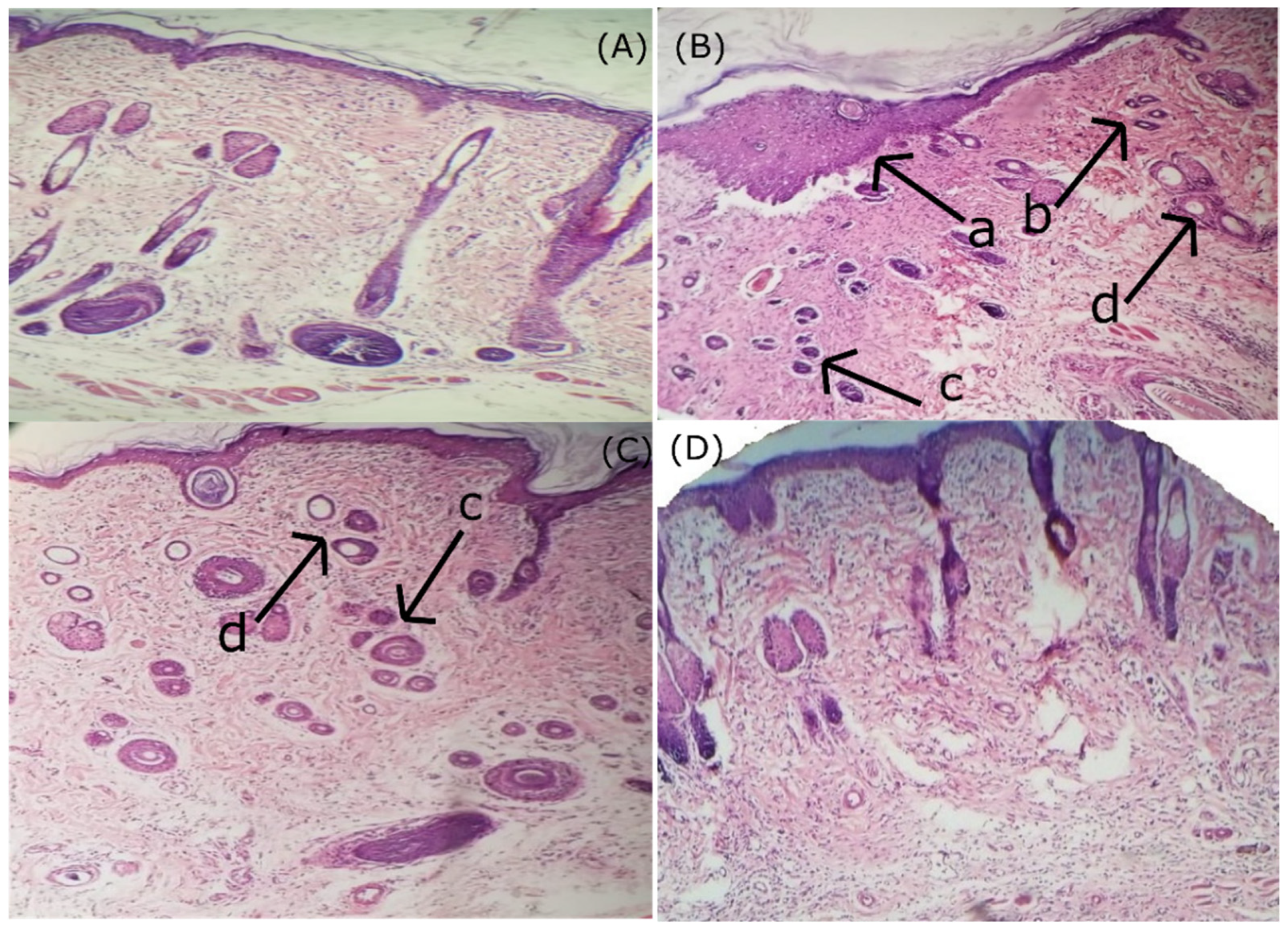
2.8. In Vivo Anti-Inflammatory Studies on Rats
3. Materials and Methods
3.1. Materials
3.2. Methods
3.2.1. Design of Experiment (Box–Behnken Design)
- Y is the dependent variable;
- X1, X2, and X3 are independent variables;
- β1, β2, and β3 are non-linear coefficients;
- β11, β22, and β33 are squares of coefficients;
- β12, β13, and β23 are the interaction coefficients of this non-linear equation.
3.2.2. Experimental Method
- (A.)
- Preparation of NAP-loaded CS/CRG nanoparticles
- (B.)
- Formation of Carbopol 940 gel containing naproxen-loaded CS/CRG nanoparticles
3.3. Characterization of Nanoparticles
3.3.1. Entrapment Efficiency
3.3.2. Particle Size, Polydispersity Index, and Zeta Potential Determination
3.3.3. Scanning Electron Microscopy (SEM)
3.3.4. Fourier Transforms Infrared Spectroscopy (FTIR)
3.3.5. Powdered X-ray Diffraction (pXRD) Analysis
3.3.6. Acute Oral Toxicity Study
3.3.7. Characterization of Naproxen-Loaded, CS/CRG-Nanoparticle-Containing Ca-940 Gel
Appearance, pH, Viscosity, and Spreadability
Drug Content
Skin Irritation Studies for Naproxen-Loaded Gel
Stability Studies
3.4. Evaluation of Ex Vivo Drug Permeation Study
3.4.1. Preparation of Full-Thickness (FT) Rat Skin
3.4.2. Ex Vivo Permeation Studies by Using Franz Diffusion Cells
3.5. In Vivo Anti-Inflammatory Studies in Rats
3.5.1. Animals
3.5.2. Treatment Protocol
3.6. Histopathology Analysis
3.7. Statistical Analysis
3.8. Model Dependent Permeation Kinetic Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lubberts, E.; van den Berg, W.B. Cytokines in the Pathogenesis of Rheumatoid Arthritis and Collagen-Induced Arthritis. In Madame Curie Bioscience Database; Landes Bioscience: Austin, TX, USA, 2013. [Google Scholar]
- Micheli, L.; Bozdag, M.; Akgul, O.; Carta, F.; Guccione, C.; Bergonzi, M.C.; Bilia, A.R.; Cinci, L.; Lucarini, E.; Parisio, C. Pain Relieving Effect of-NSAIDs-CAIs Hybrid Molecules: Systemic and Intra-Articular Treatments against Rheumatoid Arthritis. Int. J. Mol. Sci. 2019, 20, 1923. [Google Scholar] [CrossRef] [PubMed]
- McCarberg, B.; Gibofsky, A. Need to Develop New Nonsteroidal Anti-Inflammatory Drug Formulations. Clin. Ther. 2012, 34, 1954–1963. [Google Scholar] [CrossRef]
- Keyhanian, F.; Alizadeh, N.; Shojaie, A. Spectrophotometric determination of Naproxen as ion-pair with bromophenol blue in bulk, pharmaceutical preparation and human serum samples. Curr. Chem. Lett. 2014, 3, 15–22. [Google Scholar] [CrossRef]
- Adibkia, K.; Javadzadeh, Y.; Dastmalchi, S.; Mohammadi, G.; Niri, F.K.; Alaei-Beirami, M. Naproxen–eudragit® RS100 nanoparticles: Preparation and physicochemical characterization. Colloids Surf. B Biointerfaces 2011, 83, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Guilherme, V.A.; Ribeiro, L.N.M.; Alcântara, A.C.S.; Castro, S.R.; Rodrigues da Silva, G.H.; da Silva, C.G.; Breitkreitz, M.C.; Clemente-Napimoga, J.; Macedo, C.G.; Abdalla, H.B.; et al. Improved efficacy of naproxen-loaded NLC for temporomandibular joint administration. Sci. Rep. 2019, 9, 11160. [Google Scholar] [CrossRef]
- Bjarnason, I.; Scarpignato, C.; Holmgren, E.; Olszewski, M.; Rainsford, K.D.; Lanas, A. Mechanisms of damage to the gastrointestinal tract from nonsteroidal anti-inflammatory drugs. Gastroenterology 2018, 154, 500–514. [Google Scholar] [CrossRef] [PubMed]
- Wallace, J.L. Prostaglandins, NSAIDs, and gastric mucosal protection: Why doesn’t the stomach digest itself? Physiol. Rev. 2008, 88, 1547–1565. [Google Scholar] [CrossRef]
- Cheung, K.; Das, D.B. Microneedles for drug delivery: Trends and progress. Drug Deliv. 2016, 23, 2338–2354. [Google Scholar] [CrossRef] [PubMed]
- Müller, M. (Ed.) Sizing, Shaping and Pharmaceutical Applications of Polyelectrolyte Complex Nanoparticles; Springer: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Qian, F.; Cui, F.; Ding, J.; Tang, C.; Yin, C. Chitosan graft copolymer nanoparticles for oral protein drug delivery: Preparation and characterization. Biomacromolecules 2006, 7, 2722–2727. [Google Scholar] [CrossRef] [PubMed]
- Riva, R.; Ragelle, H.; des Rieux, A.; Duhem, N.; Jérôme, C.; Préat, V. Chitosan and Chitosan Derivatives in Drug Delivery and Tissue Engineering. In Chitosan for Biomaterials II; Springer: Berlin/Heidelberg, Germany, 2011; pp. 19–44. [Google Scholar]
- Singla, A.; Chawla, M. Chitosan: Some pharmaceutical and biological aspects—An update. J. Pharm. Pharm. 2001, 53, 1047–1067. [Google Scholar] [CrossRef] [PubMed]
- Shu, X.; Zhu, K. Controlled drug release properties of ionically cross-linked chitosan beads: The influence of anion structure. Int. J. Pharm. 2002, 233, 217–225. [Google Scholar] [CrossRef]
- Zheng, Z.; Tsai, P.C.; Ramezanli, T.; Michniak-Kohn, B.B. Polymeric nanoparticles-based topical delivery systems for the treatment of dermatological diseases. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2013, 5, 205–218. [Google Scholar] [CrossRef]
- Thein-Han, W.W.; Stevens, W.F. Transdermal delivery controlled by a chitosan membrane. Drug Dev. Ind. Pharm. 2004, 30, 397–404. [Google Scholar] [CrossRef]
- Kählig, H.; Hasanovic, A.; Biruss, B.; Höller, S.; Grim, J.; Valenta, C. Chitosan–glycolic acid: A possible matrix for progesterone delivery into skin. Drug Dev. Ind. Pharm. 2009, 35, 997–1002. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Kong, X.; Zhang, Z.; Nan, K.; Li, L.; Wang, X.; Chen, H. Cytotoxicity and biocompatibility evaluation of N, O-carboxymethyl chitosan/oxidized alginate hydrogel for drug delivery application. Int. J. Biol. Macromol. 2012, 50, 1299–1305. [Google Scholar] [CrossRef] [PubMed]
- Can, A.; Erdal, M.; Güngör, S.; Özsoy, Y. Optimization and characterization of chitosan films for transdermal delivery of ondansetron. Molecules 2013, 18, 5455–5471. [Google Scholar] [CrossRef] [PubMed]
- Bhaskar, K.; Anbu, J.; Ravichandiran, V.; Venkateswarlu, V.; Rao, Y.M. Lipid nanoparticles for transdermal delivery of flurbiprofen: Formulation, in vitro, ex vivo and in vivo studies. Lipids Health Dis. 2009, 8, 6. [Google Scholar] [CrossRef]
- Kyriakou, P.; Mouselimis, D.; Tsarouchas, A.; Rigopoulos, A.; Bakogiannis, C.; Noutsias, M.; Vassilikos, V. Diagnosis of cardiac amyloidosis: A systematic review on the role of imaging and biomarkers. BMC Cardiovasc. Disord. 2018, 18, 221. [Google Scholar] [CrossRef]
- Kumar, N.; Kumbhat, S. Essentials in Nanoscience and Nanotechnology; Wiley: Hoboken, NJ, USA, 2016. [Google Scholar]
- Kalsoom Khan, A.; Saba, A.U.; Nawazish, S.; Akhtar, F.; Rashid, R.; Mir, S.; Nasir, B.; Iqbal, F.; Afzal, S.; Pervaiz, F.; et al. Carrageenan Based Bionanocomposites as Drug Delivery Tool with Special Emphasis on the Influence of Ferromagnetic Nanoparticles. Oxidative Med. Cell. Longev. 2017, 2017, 8158315. [Google Scholar] [CrossRef]
- Deka, C.; Dutta, M.; Deka, D.; Jha, D.K.; Kakati, D.K. Study of olive oil-loaded chitosan/carrageenan coacervate and its antibacterial property. Int. J. Pharm. Sci. 2016, 6, 1524–1533. [Google Scholar]
- Janes, K.; Calvo, P.; Alonso, M. Polysaccharide colloidal particles as delivery systems for macromolecules. Adv. Drug Deliv. Rev. 2001, 47, 83–97. [Google Scholar] [CrossRef]
- Shanmuga, S.; Singhal, M.; Sen, S. Synthesis and Characterization of Carrageenan Coated Magnetic Nanoparticles for Drug Delivery Applications. Transl. Biomed. 2015, 6, 1–3. [Google Scholar]
- Mura, P.; Zerrouk, N.; Mennini, N.; Maestrelli, F.; Chemtob, C. Development and characterization of naproxen–chitosan solid systems with improved drug dissolution properties. Eur. J. Pharm. Sci. 2003, 19, 67–75. [Google Scholar] [CrossRef]
- Chawla, A.; Sharma, P.; Pawar, P. Eudragit S-100 coated sodium alginate microspheres of naproxen sodium: Formulation, optimization and in vitro evaluation/Alginatne mikrosfere naproksen natrija obložene Eudragitom S-100: Priprava, optimizacija i in vitro vrednovanje. Acta Pharm. 2012, 62, 529–545. [Google Scholar] [CrossRef]
- Sharma, P.; Chawla, A.; Pawar, P. Design, development, and optimization of polymeric based-colonic drug delivery system of naproxen. Sci. World J. 2013, 2013, 654829. [Google Scholar] [CrossRef] [PubMed]
- Qi, L.; Xu, Z.; Jiang, X.; Hu, C.; Zou, X. Preparation and antibacterial activity of chitosan nanoparticles. Carbohydr. Res. 2004, 339, 2693–2700. [Google Scholar] [CrossRef]
- Tranquilan-Aranilla, C.; Nagasawa, N.; Bayquen, A.; Rosa, A.D. Synthesis and characterization of carboxymethyl derivatives of kappa-carrageenan. Carbohydr. Polym. 2012, 87, 1810–1816. [Google Scholar] [CrossRef]
- Li, C.; Hein, S.; Wang, K. Chitosan-carrageenan polyelectrolyte complex for the delivery of protein drugs. ISRN Biomater. 2013, 2013, 629807. [Google Scholar] [CrossRef]
- Guo, Z.; Liu, X.-M.; Ma, L.; Li, J.; Zhang, H.; Gao, Y.-P.; Yuan, Y. Effects of particle morphology, pore size and surface coating of mesoporous silica on Naproxen dissolution rate enhancement. Colloids Surf. B Biointerfaces 2013, 101, 228–235. [Google Scholar] [CrossRef]
- Lusiana, R.A.; Protoningtyas, W.P.; Wijaya, A.R.; Siswanta, D.; Santosa, S.J. Chitosan-Tripoly Phosphate (CS-TPP) Synthesis Through Cross-linking Process: The Effect of Concentration Towards Membrane Mechanical Characteristic and Urea Permeation. Orient. J. Chem. 2017, 33, 2913–2919. [Google Scholar] [CrossRef]
- Mary, I.A.; Selvanayagam, S.; Selvasekarapandian, S.; Chitra, R.; Chandra, M.L.; Ponraj, T. Lithium ion conducting biopolymer membrane based on K-carrageenan with LiNO3. Ionics 2020, 26, 4311–4326. [Google Scholar] [CrossRef]
- Shirsat, A.E.; Chitlange, S.S. Application of quality by design approach to optimize process and formulation parameters of rizatriptan loaded chitosan nanoparticles. J. Adv. Pharm. Technol. Res. 2015, 6, 88. [Google Scholar] [CrossRef]
- Jana, S.; Manna, S.; Nayak, A.K.; Sen, K.K.; Basu, S.K. Carbopol gel containing chitosan-egg albumin nanoparticles for transdermal aceclofenac delivery. Colloids Surf. B Biointerfaces 2014, 114, 36–44. [Google Scholar] [CrossRef]
- Cooper, D.L.; Harirforoosh, S. Effect of formulation variables on preparation of celecoxib loaded polylactide-co-glycolide nanoparticles. PLoS ONE 2014, 9, e113558. [Google Scholar] [CrossRef] [PubMed]
- Bayat, A.; Larijani, B.; Ahmadian, S.; Junginger, H.E.; Rafiee-Tehrani, M. Preparation and characterization of insulin nanoparticles using chitosan and its quaternized derivatives. Nanomed. Nanotechnol. 2008, 4, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Bokharaei, M.; Margaritis, A.; Xenocostas, A.; J Freeman, D. Erythropoietin encapsulation in chitosan nanoparticles and kinetics of drug release. Curr. Drug Deliv. 2011, 8, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Grenha, A.; Gomes, M.E.; Rodrigues, M.; Santo, V.E.; Mano, J.F.; Neves, N.M.; Reis, R.L. Development of new chitosan/carrageenan nanoparticles for drug delivery applications. J. Biomed. Mater. Res. A 2010, 92, 1265–1272. [Google Scholar] [CrossRef]
- Kouchak, M.; Azarpanah, A. Preparation and In Vitro Evaluation of Chitosan Nanoparticles Containing Diclofenac Using the Ion-Gelation Method. Jundishapur J. Nat. Pharm. Prod. 2015, 10, 10. [Google Scholar] [CrossRef]
- Shahriari, M.H.; Atai, M.; Zandi, M.; Shokrollahi, P.; Solhi, L. Preparation and characterization of eugenol-loaded oligochitosan nanoparticles through sol–gel and emulsion/sol–gel methods. Polym. Bull. 2018, 75, 3035–3051. [Google Scholar] [CrossRef]
- Li, D.; Kaner, R.B. Shape and aggregation control of nanoparticles: Not shaken, not stirred. J. Am. Chem. Soc. 2006, 128, 968–975. [Google Scholar] [CrossRef]
- Van de Velde, F.; Knutsen, S.; Usov, A.; Rollema, H.; Cerezo, A. 1H and 13C high resolution NMR spectroscopy of carrageenans: Application in research and industry. Trends Food Sci. Technol. 2002, 13, 73–92. [Google Scholar] [CrossRef]
- Bhavin, K.P.; Apurva, R.; Parikh, R.H.; Aboti, P.S. Development of oral sustained release rifampicin loaded chitosan nanoparticles by design of experiment. J. Drug Deliv. 2013, 2013, 370938. [Google Scholar]
- Sharma, N.; Madan, P.; Lin, S. Effect of process and formulation variables on the preparation of parenteral paclitaxel-loaded biodegradable polymeric nanoparticles: A co-surfactant study. Asian J. Pharm. Sci. 2016, 11, 404–416. [Google Scholar] [CrossRef]
- Basha, B.N.; Prakasam, K.; Goli, D. Formulation and evaluation of gel containing fluconazole-antifungal agent. Int. J. Drug Dev. Res. 2011, 3, 109–128. [Google Scholar]
- Kaur, L.P. Topical gel: A recent approach for novel drug delivery. Asian J. Biomed. Pharm. Sci. 2013, 3, 1. [Google Scholar]
- Shilakari Asthana, G.; Asthana, A.; Singh, D.; Sharma, P.K. Etodolac Containing Topical Niosomal Gel: Formulation Development and Evaluation. J. Drug Deliv. 2016, 2016, 9324567. [Google Scholar] [CrossRef]
- Feng, S.-S. Nanoparticles of biodegradable polymers for new-concept chemotherapy. Expert Rev. Med. Devices 2004, 1, 115–125. [Google Scholar] [CrossRef]
- Patel, H.K.; Barot, B.S.; Parejiya, P.B.; Shelat, P.K.; Shukla, A. Topical delivery of clobetasol propionate loaded microemulsion based gel for effective treatment of vitiligo: Ex vivo permeation and skin irritation studies. Colloids Surf. B Biointerfaces 2013, 102, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Shivhare, U.; Jain, K.; Mathur, V.; Bhusari, K.; Roy, A. Formulation development and evaluation of diclofenac sodium gel using water soluble polyacrylamide polymer. Dig. J. Nanomater. Biostruct. 2009, 4, 2. [Google Scholar]
- Senthil, V.; Kumar, R.S.; Nagaraju, C.; Jawahar, N.; Ganesh, G.; Gowthamarajan, K. Design and development of hydrogel nanoparticles for mercaptopurine. J. Adv. Pharm. Technol. Res. 2010, 1, 334. [Google Scholar] [CrossRef] [PubMed]
- Morsi, N.M.; Abdelbary, G.A.; Ahmed, M.A. Silver sulfadiazine based cubosome hydrogels for topical treatment of burns: Development and in vitro/in vivo characterization. Eur. J. Pharm. Biopharm. 2014, 86, 178–189. [Google Scholar] [CrossRef]
- Prajapati, S.T.; Patel, C.G.; Patel, C.N. Formulation and evaluation of transdermal patch of repaglinide. ISRN Pharm. 2011, 2011, 651909. [Google Scholar] [CrossRef]
- Hafner, A.; Lovrić, J.; Pepić, I.; Filipović-Grčić, J. Lecithin/chitosan nanoparticles for transdermal delivery of melatonin. J. Microencapsul. 2011, 28, 807–815. [Google Scholar] [CrossRef]
- Verma, A.; Jain, A.; Hurkat, P.; Jain, S.K. Transfollicular drug delivery: Current perspectives. Res. Rep. Transdermal Drug Deliv. 2016, 5, 1–17. [Google Scholar]
- Cong, H.; Khaziakhmetova, V.; Zigashina, L. Rat paw oedema modeling and NSAIDs: Timing of effects. Int. J. Risk Saf. Med. 2015, 27, S76–S77. [Google Scholar] [CrossRef] [PubMed]
- Valenta, C.; Schultz, K. Influence of carrageenan on the rheology and skin permeation of microemulsion formulations. J. Control. Release 2004, 95, 257–265. [Google Scholar] [CrossRef]
- Rodrigues, S.; da Costa, A.M.R.; Grenha, A. Chitosan/carrageenan nanoparticles: Effect of cross-linking with tripolyphosphate and charge ratios. Carbohydr. Polym. 2012, 89, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Amini, Y.; Amel Jamehdar, S.; Sadri, K.; Zare, S.; Musavi, D.; Tafaghodi, M. Different methods to determine the encapsulation efficiency of protein in PLGA nanoparticles. Biomed. Mater. Eng. 2017, 28, 613–620. [Google Scholar] [CrossRef]
- Rescignano, N.; Tarpani, L.; Tiribuzi, R.; Montesano, S.; Martino, S.; Latterini, L.; Kenny, J.M.; Armentano, I. Protein encapsulation in biodegradable polymeric nanoparticles: Morphology, fluorescence behaviour and stem cell uptake. Macromolecules 2013, 13, 1204–1212. [Google Scholar] [CrossRef] [PubMed]
- Ankanna, S.; Tollamadugu Prasad, N.V.K.V.; Elumalai, E.; Savithramma, N. Production of biogenic silver nanoparticles using Boswellia ovalifoliolata stem bark. Dig. J. Nanomater. Biostruct. 2010, 5, 369–372. [Google Scholar]
- Surassmo, S.; Saengkrit, N.; Ruktanonchai, U.R.; Suktham, K.; Woramongkolchai, N.; Wutikhun, T.; Puttipipatkhachorn, S. Surface modification of PLGA nanoparticles by carbopol to enhance mucoadhesion and cell internalization. Colloids Surf. B Biointerfaces 2015, 130, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.; Sachdeva, R.; Kapoor, G. Comparison of crystalline and amorphous carriers to improve the dissolution profile of water insoluble drug itraconazole. Int. J. Pharm. Biol. Sci. 2013, 4, 934–948. [Google Scholar]
- Sadaquat, H.; Akhtar, M.; Nazir, M.; Ahmad, R.; Alvi, Z.; Akhtar, N. Biodegradable and biocompatible polymeric nanoparticles for enhanced solubility and safe oral delivery of docetaxel: In vivo toxicity evaluation. Int. J. Pharm. 2021, 598, 120363. [Google Scholar] [CrossRef]
- Helal, D.A.; El-Rhman, D.A.; Abdel-Halim, S.A.; El-Nabarawi, M.A. Formulation and evaluation of fluconazole topical gel. Int. J. Pharm. Pharm. Sci. 2012, 4, 176–183. [Google Scholar]
- Sareen, R.; Kumar, S.; Gupta, G.D. Meloxicam carbopol-based gels: Characterization and evaluation. Curr. Drug Deliv. 2011, 8, 407–415. [Google Scholar] [CrossRef]
- Gupta, A.; Mishra, A.; Singh, A.; Gupta, V.; Bansal, P. Formulation and evaluation of topical gel of diclofenac sodium using different polymers. Drug Invent. Today 2010, 2, 250–253. [Google Scholar]
- Sera, U.; Ramana, M. In vitro skin absorption and drug release–a comparison of four commercial hydrophilic gel preparations for topical use. Indian Pharm. 2006, 73, 356–360. [Google Scholar]
- Bachhav, Y.; Patravale, V. Formulation of meloxicam gel for topical application: In vitro and in vivo evaluation. Acta Pharm. 2010, 60, 153–163. [Google Scholar] [CrossRef]
- Bachhav, Y.G.; Patravale, V.B. Microemulsion-based vaginal gel of clotrimazole: Formulation, in vitro evaluation, and stability studies. AAPS PharmSciTech. 2009, 10, 476. [Google Scholar] [CrossRef]
- Aletaha, D.; Neogi, T.; Silman, A.J.; Funovits, J.; Felson, D.T.; Bingham, C.O.; Birnbaum, N.S.; Burmester, G.R.; Bykerk, V.P.; Cohen, M.D. 2010 rheumatoid arthritis classification criteria: An American College of Rheumatology/European League against Rheumatism collaborative initiative. Arthritis Rheumatol. 2010, 62, 2569–2581. [Google Scholar] [CrossRef]
- Fang, J.-Y.; Yu, S.-Y.; Wu, P.-C.; Huang, Y.-B.; Tsai, Y.-H. In vitro skin permeation of estradiol from various proniosome formulations. Int. J. Pharm. 2001, 215, 91–99. [Google Scholar] [CrossRef]
- Renju, G.; Muraleedhara Kurup, G.; Saritha Kumari, C. Anti-inflammatory activity of lycopene isolated from Chlorella marina on Type II Collagen induced arthritis in Sprague Dawley rats. Immunopharmacol. Immunotoxicol. 2013, 35, 282–291. [Google Scholar] [CrossRef] [PubMed]
- Freitas, M.; Marchetti, J. Nimesulide PLA microspheres as a potential sustained release system for the treatment of inflammatory diseases. Int. J. Pharm. 2005, 295, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Chen, Y.; Zhou, Y.; Guo, D.; Fan, Y.; Guo, F.; Zheng, Y.; Chen, W. Preparation of 5-fluorouracil-loaded chitosan nanoparticles and study the sustained release in vitro and in vivo. Asian J. Pharm. Sci. 2017, 12, 418–423. [Google Scholar] [CrossRef]
- Derakhshandeh, K.; Soheili, M.; Dadashzadeh, S.; Saghiri, R. Preparation and in vitro characterization of 9-nitrocamptothecin-loaded long circulating nanoparticles for delivery in cancer patients. Int. J. Nanomed. 2010, 5, 463. [Google Scholar] [CrossRef] [PubMed]
- Dash, S.; Murthy, P.N.; Nath, L.; Chowdhury, P. Kinetic modeling on drug release from controlled drug delivery systems. Acta Pol. Pharm. 2010, 67, 217–223. [Google Scholar] [PubMed]
- Muhamad, I.I.; Selvakumaran, S.; Lazim, N.A.M. Designing polymeric nanoparticles for targeted drug delivery system. Nanomedicine 2014, 287, 287. [Google Scholar]
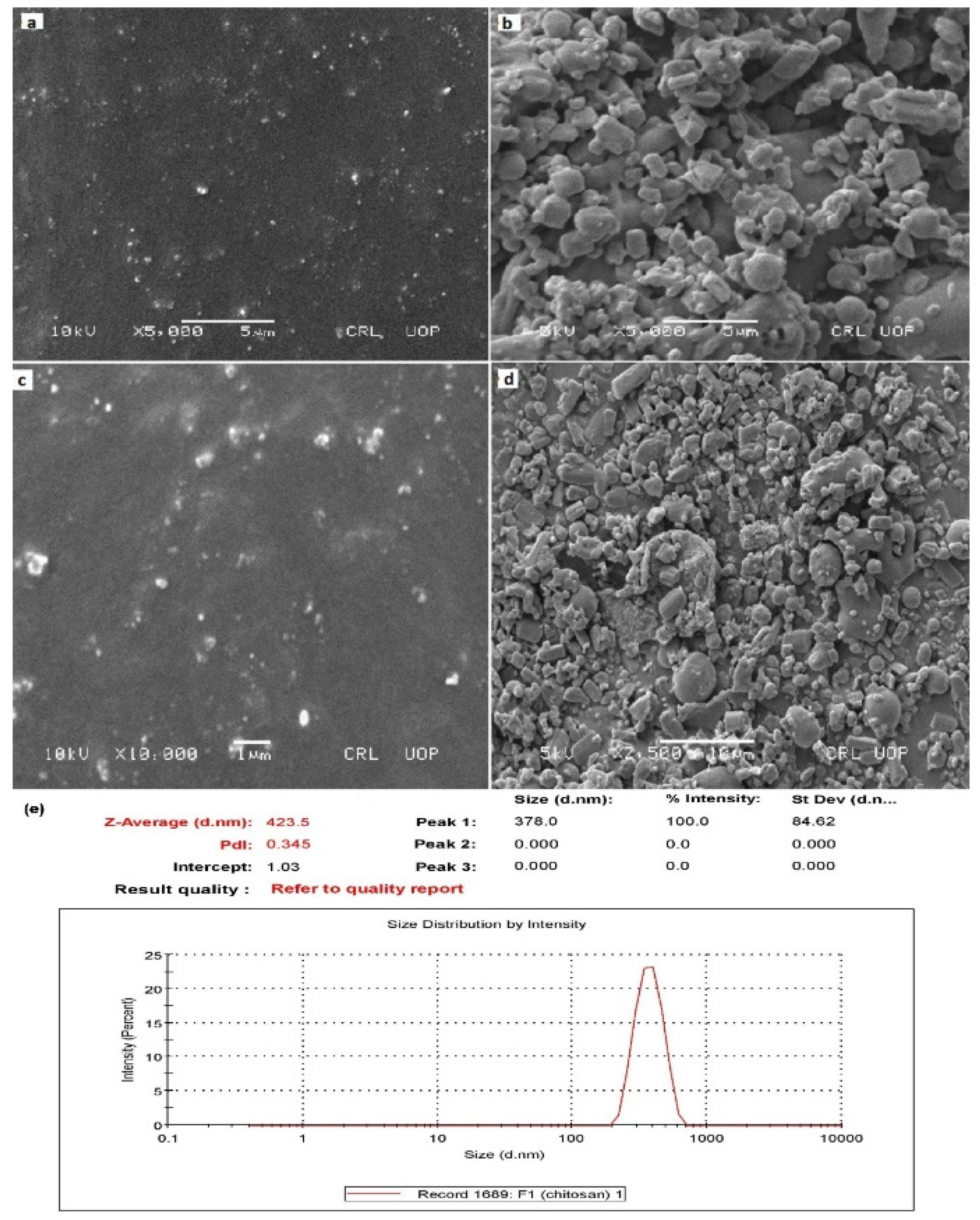
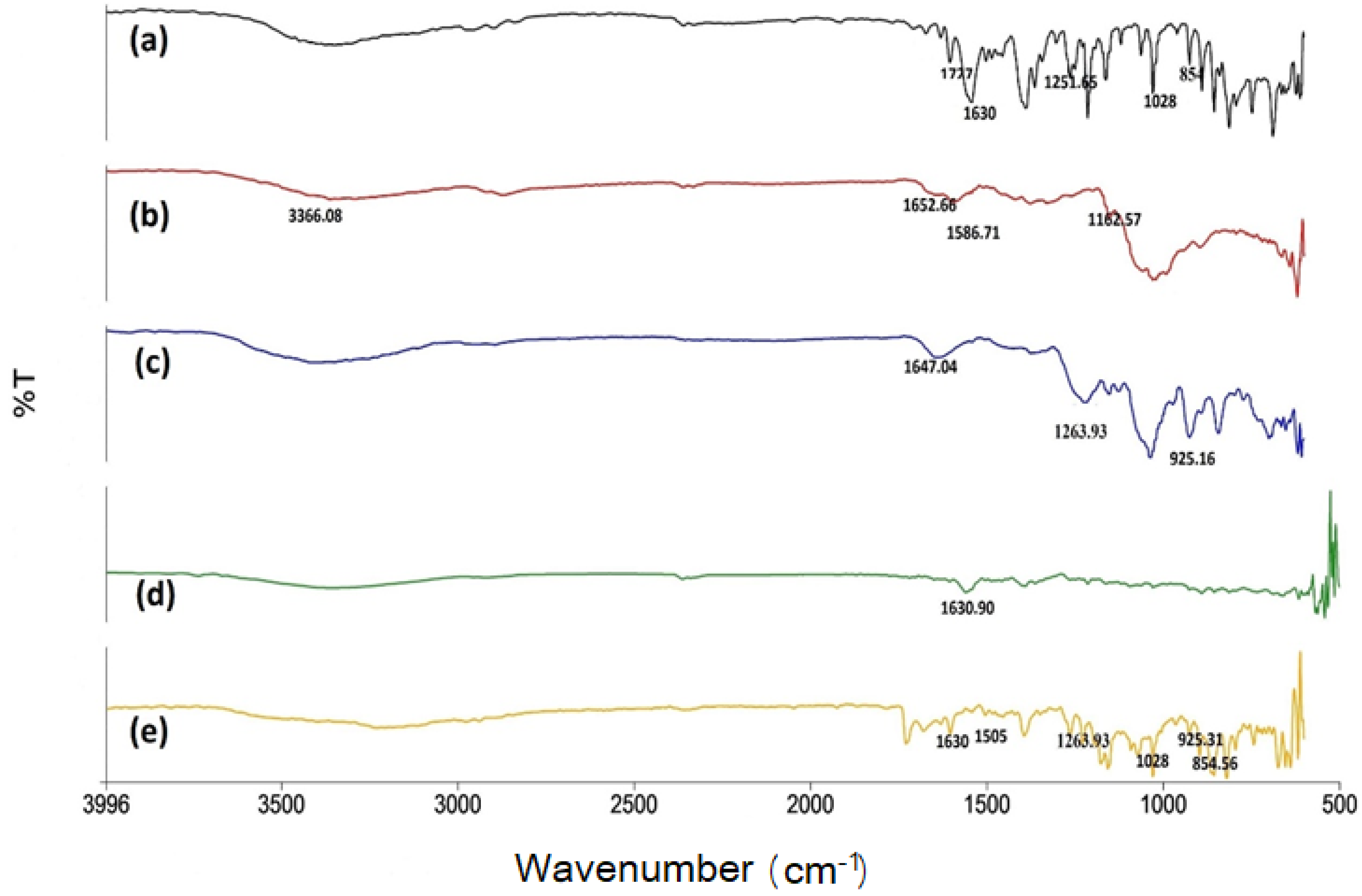
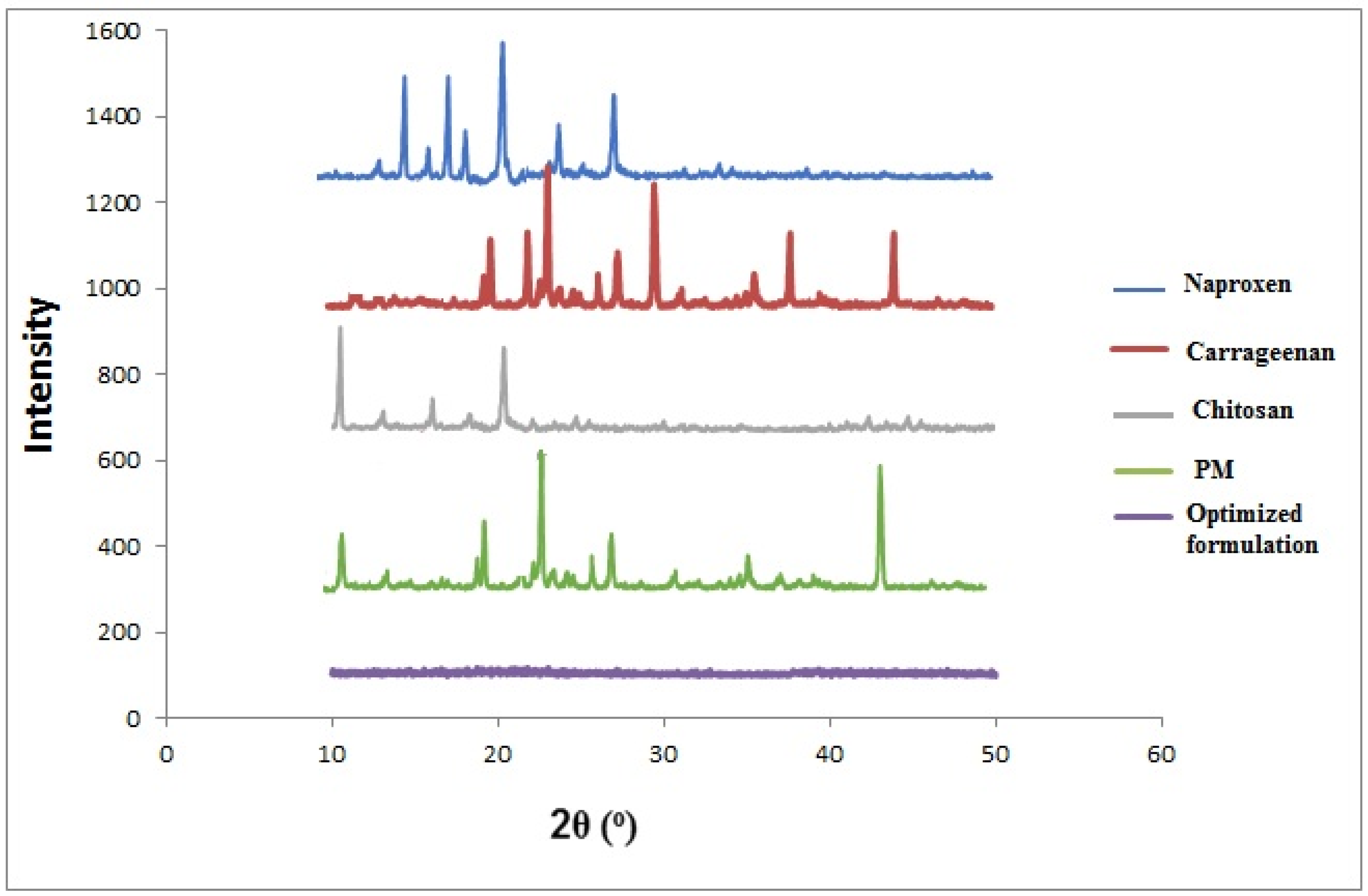
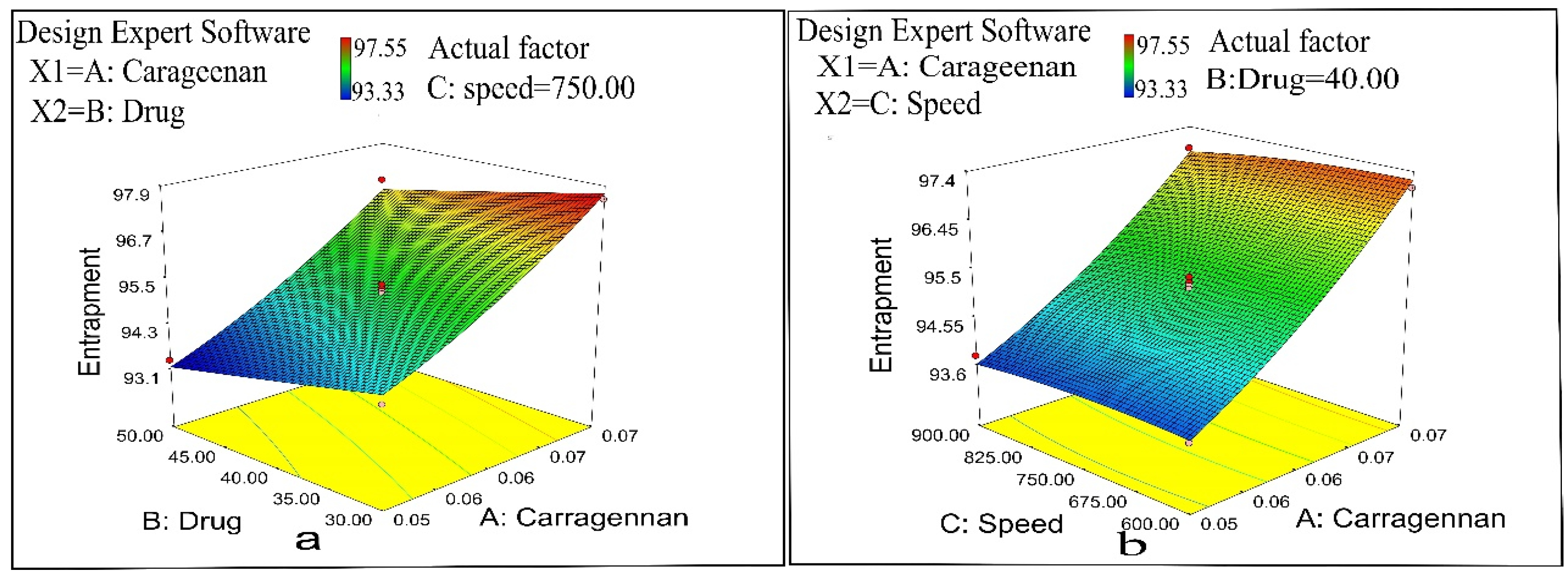
| Formulation Code | Entrapment Efficiency (%) | Particle Size (nm) | PDI | Zeta Potential (mV) | % Cumulative Drug Permeation | % Yield |
|---|---|---|---|---|---|---|
| N1 | 97.55% | 342.45 ± 16.33 | 0.308 | +25 ± 0.9 | 86.98 | 71.76% |
| N2 | 95.12% | 264.33 ± 35.78 | 0.422 | +29 ± 1.3 | 89.66 | 70.46% |
| N3 | 97.08% | 332.29 ± 8.43 | 0.318 | +26 ± 1.9 | 90.34 | 72.48% |
| N4 | 94.12% | 162.75 ± 22.43 | 0.510 | +34 ± 1.1 | 93.33 | 67.01% |
| N5 | 95.25% | 264.33 ± 35.78 | 0.422 | +34 ± 1.6 | 89.66 | 70.54% |
| N6 | 94.66% | 277.54 ± 6.74 | 0.443 | +33 ± 3.1 | 90.25 | 71.61% |
| N7 | 96.94% | 362.43 ± 54.64 | 0.355 | +27 ± 2.6 | 85.66 | 72.72% |
| N8 | 95.36% | 264.33 ± 35.78 | 0.422 | +33 ± 3.1 | 89.66 | 70.22% |
| N9 | 95.08% | 264.33 ± 35.78 | 0.422 | +34 ± 1.1 | 89.66 | 70.72% |
| N10 | 93.66% | 156.78 ± 45.53 | 0.504 | +35 ± 2.1 | 93.66 | 67.26% |
| N11 | 93.33% | 176.68 ± 27.03 | 0.512 | +33 ± 0.8 | 92.34 | 67.96% |
| N12 | 95.77% | 255.38 ± 18.43 | 0.465 | +30 ± 1.3 | 88.93 | 69.92% |
| N13 | 96.03% | 243.22 ± 10.55 | 0.412 | +29 ± 0.6 | 91.45 | 69.72% |
| N14 | 95.18% | 264.33 ± 35.78 | 0.422 | +33 ± 3.1 | 89.66 | 70.60% |
| N15 | 96.86% | 378.0 ± 84.62 | 0.345 | +28 ± 2.3 | 85.47 | 73.96% |
| N16 | 94.01% | 290.08 ± 24.22 | 0.454 | +31 ± 0.4 | 88.23 | 69.53% |
| N17 | 93.78% | 188.32 ± 16.69 | 0.521 | +33 ± 0.8 | 90.76 | 67.46% |
| N18 | 90.22% | 407.20 ± 84.62 | 0.533 | +26 ± 2.1 | 76.80 | 53.2% |
| N19 | 89.34% | 674.20 ± 47.48 | 0.736 | +12 ± 2.2 | 63.34 | 28% |
| No (optimized) | 95.26% | 355.7 ± 79.8 | 0.381 | +25 ± 3.1 | 88.66 | 70.78% |
| Sr. No. | Response Variables | Statistical Term | p-Value |
|---|---|---|---|
| 1. | Entrapment efficiency | Polymer concentration | 0.0001 |
| Drug concentration | 0.0001 | ||
| Stirring speed | 0.2258 | ||
| 2. | Particle size | Polymer concentration | 0.0001 |
| Drug concentration | 0.0005 | ||
| Stirring speed | 0. 0033 | ||
| 3. | % cumulative drug permeation | Polymer concentration | 0.0001 |
| Drug concentration | 0.0810 | ||
| Stirring speed | 0. 0008 |
| Formulation Code | Appearance and Homogeneity | pH | Viscosity (cps) | Spreadability (cm) | Drug Content (%) | Skin Irritation Score |
|---|---|---|---|---|---|---|
| N1 | +++ | 6.8 | 5450 | 3.2 | 92 | 0 |
| N2 | +++ | 6.4 | 5325 | 3.6 | 89 | 1 |
| N3 | +++ | 6.7 | 5455 | 2.8 | 94 | 0 |
| N4 | +++ | 6.8 | 5272 | 4.9 | 87 | 0 |
| N5 | +++ | 6.4 | 5328 | 3.5 | 88 | 1 |
| N6 | +++ | 6.5 | 5335 | 2.4 | 92 | 1 |
| N7 | +++ | 6.8 | 5458 | 2.6 | 93 | 0 |
| N8 | +++ | 6.4 | 5321 | 3.6 | 90 | 1 |
| N9 | +++ | 6.4 | 5332 | 3.7 | 90 | 1 |
| N10 | +++ | 6.2 | 5283 | 4.6 | 87 | 1 |
| N11 | +++ | 6.2 | 5296 | 4.4 | 88 | 1 |
| N12 | +++ | 6.3 | 5340 | 3.3 | 89 | 1 |
| N13 | +++ | 6.4 | 5338 | 3.4 | 90 | 1 |
| N14 | +++ | 6.4 | 5330 | 3.5 | 93 | 1 |
| N15 | +++ | 6.6 | 5466 | 2.9 | 95 | 0 |
| N16 | +++ | 6.3 | 5345 | 3.7 | 92 | 1 |
| N17 | +++ | 6.2 | 5218 | 4.2 | 91 | 1 |
| N18 | +++ | 6.4 | 3200 | 2.6 | 85 | 1 |
| N19 | +++ | 6.3 | 5102 | 2.2 | 87 | 1 |
| No | +++ | 6.5 | 5040 | 2.3 | 93 | 0 |
| Code | Zero-Order | First-Order | Higuchi Model | Korsmeyer–Peppas Model | ||||
|---|---|---|---|---|---|---|---|---|
| R2 | K | R2 | K | R2 | KH | R2 | n | |
| N1 | 0.9899 | 1.922 | 0.9818 | 0.030 | 0.8877 | 10.130 | 0.9976 | 0.869 |
| N2 | 0.9872 | 2.008 | 0.9808 | 0.033 | 0.8890 | 10.594 | 0.9963 | 0.859 |
| N3 | 0.9942 | 1.971 | 0.9782 | 0.031 | 0.8844 | 10.365 | 0.9999 | 0.884 |
| N4 | 0.9921 | 2.020 | 0.9741 | 0.033 | 0.8834 | 10.629 | 0.9980 | 0.883 |
| N5 | 0.9872 | 2.008 | 0.9808 | 0.033 | 0.8890 | 10.594 | 0.9963 | 0.859 |
| N6 | 0.9851 | 2.049 | 0.9800 | 0.034 | 0.8824 | 10.790 | 0.9935 | 0.865 |
| N7 | 0.9877 | 1.901 | 0.9833 | 0.030 | 0.8979 | 10.065 | 0.9986 | 0.846 |
| N8 | 0.9872 | 2.008 | 0.9808 | 0.033 | 0.8890 | 10.594 | 0.9963 | 0.859 |
| N9 | 0.9872 | 2.008 | 0.9808 | 0.033 | 0.8890 | 10.594 | 0.9963 | 0.859 |
| N10 | 0.9922 | 2.025 | 0.9739 | 0.033 | 0.8830 | 10.649 | 0.9980 | 0.884 |
| N11 | 0.9947 | 2.015 | 0.9753 | 0.032 | 0.8825 | 10.586 | 0.9999 | 0.889 |
| N12 | 0.9933 | 1.928 | 0.9809 | 0.031 | 0.8872 | 10.151 | 0.9999 | 0.877 |
| N13 | 0.9938 | 2.008 | 0.9773 | 0.032 | 0.8853 | 10.562 | 0.9999 | 0.881 |
| N14 | 0.9872 | 2.008 | 0.9808 | 0.033 | 0.8890 | 10.594 | 0.9963 | 0.859 |
| N15 | 0.9859 | 1.897 | 0.9853 | 0.030 | 0.9018 | 10.060 | 0.9986 | 0.835 |
| N16 | 0.9923 | 1.933 | 0.9822 | 0.031 | 0.8903 | 10.191 | 0.9999 | 0.868 |
| N17 | 0.9876 | 2.036 | 0.9786 | 0.033 | 0.8774 | 10.693 | 0.9941 | 0.880 |
| N18 | 0.9996 | 1.625 | 0.9779 | 0.023 | 0.8519 | 8.433 | 0.9998 | 0.976 |
| N19 | 0.9981 | 1.366 | 0.9859 | 0.018 | 0.8451 | 7.100 | 0.9983 | 0.977 |
| No | 0.9927 | 1.936 | 0.9818 | 0.031 | 0.8884 | 10.197 | 0.9998 | 0.873 |
| Parameter/Test | Group I (Control) | Group II (Treatment) |
|---|---|---|
| Biochemical Parameters | ||
| AST/SGOT (IU/L) | 144.23 ± 2.0 | 146.02 ± 2.50 |
| Creatinine (mg/dL) | 0.75 ± 0.11 | 0.88 ± 0.09 |
| Triglycerides (mg/dL) | 57 ± 3.11 | 56 ± 2.04 |
| Total cholesterol (mg/dL) | 62.31 ± 3.76 | 60.08 ± 5.10 |
| Serum uric acid (mg/dL) | 3.21 ± 0.02 | 3.43 ± 0.03 |
| Serum urea (mg/dL) | 12.56 ± 3.04 | 14.76 ± 2.32 |
| Hematological Parameters | ||
| Hemoglobin Hb (g/dL) | 13.21 ± 0.32 | 13.48 ± 0.41 |
| Red blood cells (RBCs) × 106/mm3 | 6.12 ± 0.51 | 5.66 ± 0.61 |
| White blood cells (WBCs) × 109/L | 6.62 ± 0.02 | 6.77 ± 0.40 |
| Platelets × 109/L | 4.05 ± 2.05 | 4.16 ± 2.06 |
| Neutrophils (%) | 55.80 ± 4.05 | 57.55 ± 5.11 |
| Lymphocytes (%) | 38.30 ± 1.08 | 39.21 ± 1.02 |
| Monocytes (%) | 3.60 ± 0.11 | 3.65 ± 0.21 |
| Mean corpuscular volume (%) | 83.66 ± 2.10 | 84.84 ± 2.40 |
| Mean corpuscular hemoglobin (pg/cells) | 23 ± 3.05 | 24 ± 2.25 |
| Mean corpuscular hemoglobin concentration (%) | 33.30 ± 2.21 | 33.92 ± 1.41 |
| Rabbit Organ Weights | ||
| Heart | 4.33 ± 0.20 | 4.40 ± 0.16 |
| Kidney | 12.33 ± 0.21 | 13.75 ± 0.81 |
| Liver | 7.22 ± 2.01 | 8.22 ± 2.11 |
| Lungs | 9.22 ± 0.38 | 9.25 ± 0.51 |
| Spleen | 1.12 ± 0.11 | 1.13 ± 0.14 |
| Stomach | 12.22 ± 0.41 | 13.01 ± 0.70 |
| Optimized Formulation (No) | |||||
| Parameters | Entrapment Efficiency (%) | Particle Size (nm) | PDI | Zeta Potential (mV) | Cumulative Drug Permeation (%) |
| Predicted variables by design expert | 97.43 | 345.015 | - | - | 85.7359 |
| Experimental values | 95.26 ± 3.23 | 355.7 | 0.381 | +25 ± 3.1 | 88.66 |
| Stability study of optimized formulation | |||||
| Time | Appearance | pH | Viscosity (cps) | Spreadability (cm) | Drug content (%) |
| Day 0 | ++ | 6.78 | 5483 | 2.82 | 94 |
| Day 30 | ++ | 6.74 | 5496 | 2.87 | 92 |
| Day 60 | ++ | 6.76 | 5509 | 3.20 | 90 |
| Day 90 | ++ | 6.66 | 5483 | 3.27 | 88 |
| Percentage Inhibition of Paw Edema | |||
|---|---|---|---|
| Groups | 1 h | 3 h | 5 h |
| FIE | 16 | 16 | 16 |
| Formalin + NAP NP gel | 36 ± 0.96 | 57 ± 0.79 | 79 ± 0.85 |
| Formalin + NAP control gel | 21 ± 0.76 | 45 ± 1.13 | 59 ± 1.01 |
| Formulation Code | Naproxen (%) | Chitosan (%) | Carrageenan (%) | STPP (%) | Stirring Time (h) | Stirring Speed (rpm) |
|---|---|---|---|---|---|---|
| N1 | 30 | 0.1 | 0.07 | 0.5 | 1 | 750 |
| N2 | 40 | 0.1 | 0.06 | 0.5 | 1 | 750 |
| N3 | 40 | 0.1 | 0.07 | 0.5 | 1 | 600 |
| N4 | 30 | 0.1 | 0.05 | 0.5 | 1 | 750 |
| N5 | 40 | 0.1 | 0.06 | 0.5 | 1 | 750 |
| N6 | 50 | 0.1 | 0.06 | 0.5 | 1 | 600 |
| N7 | 40 | 0.1 | 0.07 | 0.5 | 1 | 900 |
| N8 | 40 | 0.1 | 0.06 | 0.5 | 1 | 750 |
| N9 | 40 | 0.1 | 0.06 | 0.5 | 1 | 750 |
| N10 | 40 | 0.1 | 0.05 | 0.5 | 1 | 600 |
| N11 | 50 | 0.1 | 0.05 | 0.5 | 1 | 750 |
| N12 | 30 | 0.1 | 0.06 | 0.5 | 1 | 900 |
| N13 | 30 | 0.1 | 0.06 | 0.5 | 1 | 600 |
| N14 | 40 | 0.1 | 0.06 | 0.5 | 1 | 750 |
| N15 | 50 | 0.1 | 0.07 | 0.5 | 1 | 750 |
| N16 | 50 | 0.1 | 0.06 | 0.5 | 1 | 900 |
| N17 | 40 | 0.1 | 0.05 | 0.5 | 1 | 900 |
| N18 | 40 | 0.1 | 0 | 0.5 | 1 | 750 |
| N19 | 40 | 0.1 | 0.06 | 0 | 1 | 750 |
| No (optimized) | 30 | 0.1 | 0.07 | 0.5 | 1 | 900 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Noreen, S.; Pervaiz, F.; Ashames, A.; Buabeid, M.; Fahelelbom, K.; Shoukat, H.; Maqbool, I.; Murtaza, G. Optimization of Novel Naproxen-Loaded Chitosan/Carrageenan Nanocarrier-Based Gel for Topical Delivery: Ex Vivo, Histopathological, and In Vivo Evaluation. Pharmaceuticals 2021, 14, 557. https://doi.org/10.3390/ph14060557
Noreen S, Pervaiz F, Ashames A, Buabeid M, Fahelelbom K, Shoukat H, Maqbool I, Murtaza G. Optimization of Novel Naproxen-Loaded Chitosan/Carrageenan Nanocarrier-Based Gel for Topical Delivery: Ex Vivo, Histopathological, and In Vivo Evaluation. Pharmaceuticals. 2021; 14(6):557. https://doi.org/10.3390/ph14060557
Chicago/Turabian StyleNoreen, Sobia, Fahad Pervaiz, Akram Ashames, Manal Buabeid, Khairi Fahelelbom, Hina Shoukat, Irsah Maqbool, and Ghulam Murtaza. 2021. "Optimization of Novel Naproxen-Loaded Chitosan/Carrageenan Nanocarrier-Based Gel for Topical Delivery: Ex Vivo, Histopathological, and In Vivo Evaluation" Pharmaceuticals 14, no. 6: 557. https://doi.org/10.3390/ph14060557
APA StyleNoreen, S., Pervaiz, F., Ashames, A., Buabeid, M., Fahelelbom, K., Shoukat, H., Maqbool, I., & Murtaza, G. (2021). Optimization of Novel Naproxen-Loaded Chitosan/Carrageenan Nanocarrier-Based Gel for Topical Delivery: Ex Vivo, Histopathological, and In Vivo Evaluation. Pharmaceuticals, 14(6), 557. https://doi.org/10.3390/ph14060557








