Polydeoxyribonucleotide: A Promising Biological Platform to Accelerate Impaired Skin Wound Healing
Abstract
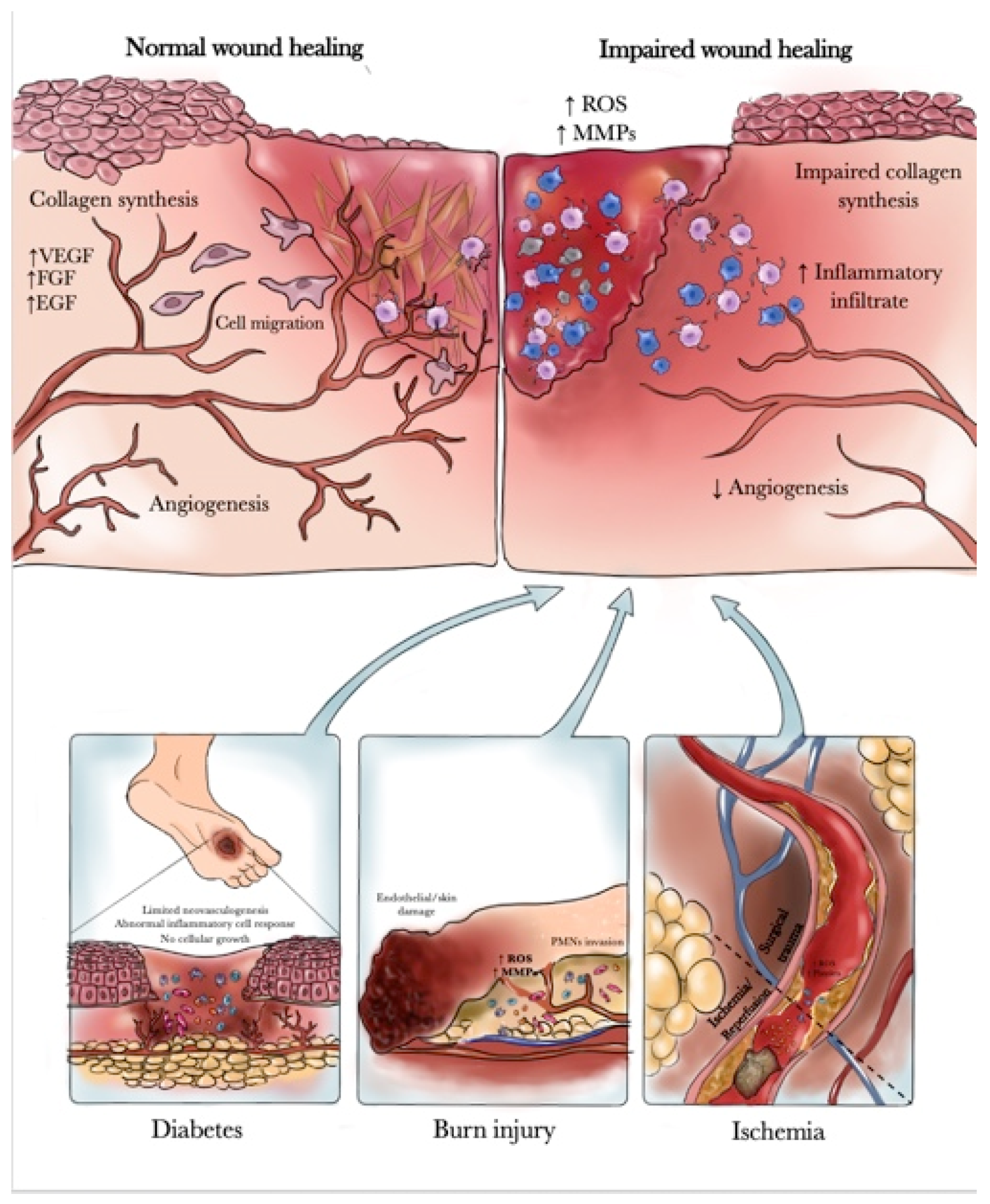
:1. Introduction
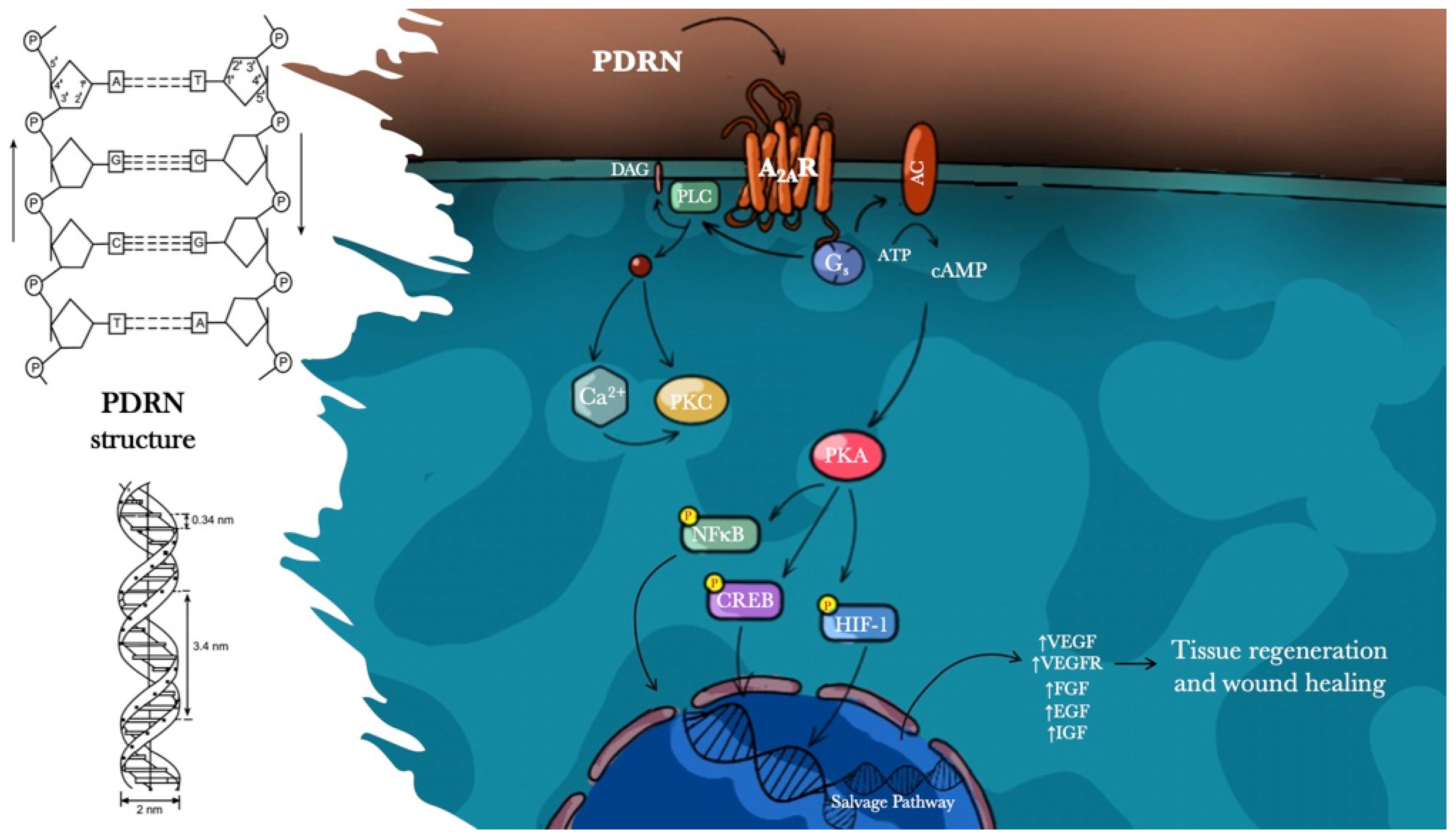
Polydeoxyribonucleotide
2. Methodology
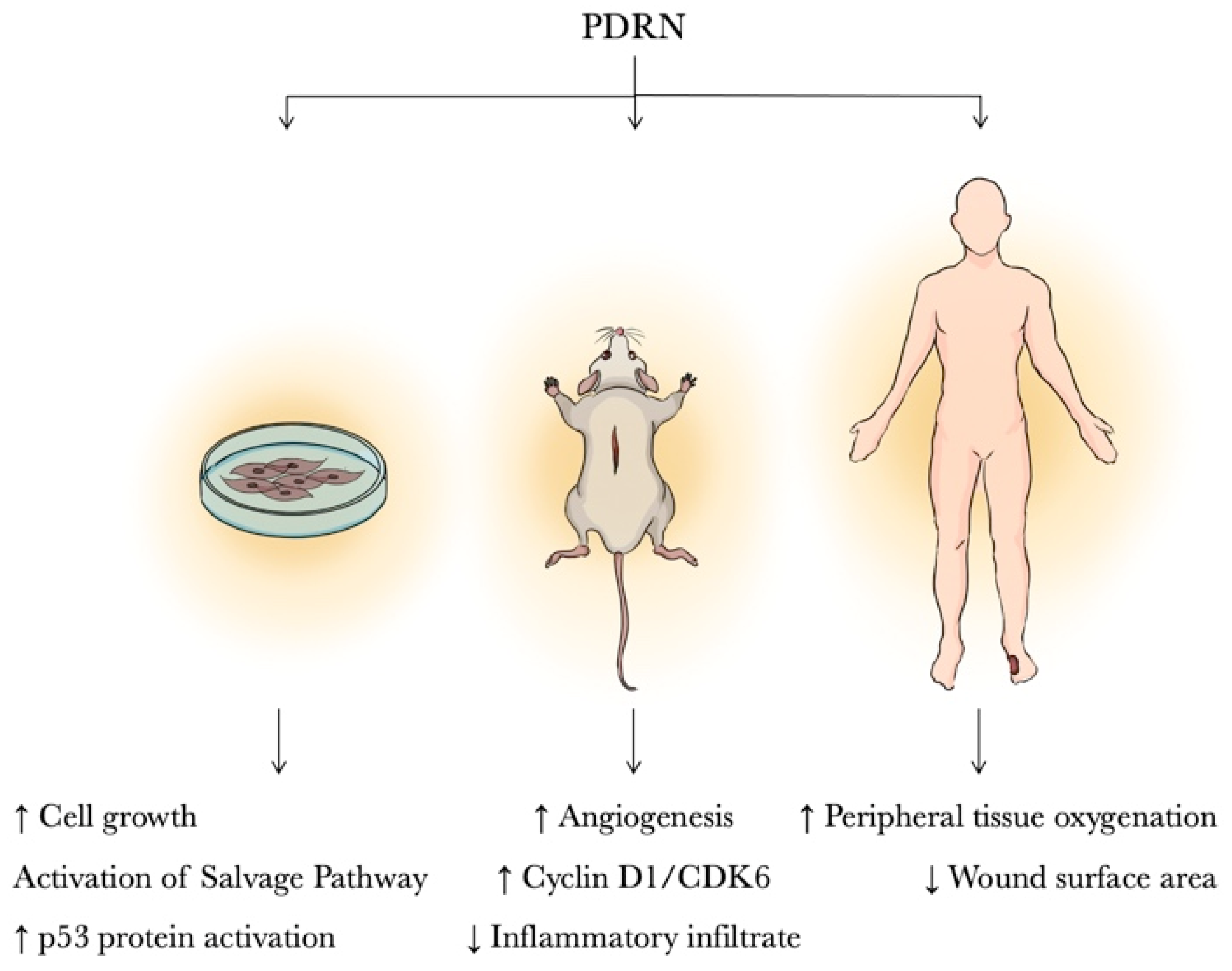
3. Results
3.1. In Vitro Studies
3.2. Animal Studies
3.3. Clinical Studies
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Singer, A.J.; Clark, R.A.F. Cutaneous wound healing. N. Engl. J. Med. 1999, 341, 738–746. [Google Scholar] [CrossRef]
- Gurtner, G.C.; Werner, S.; Barrandon, Y.; Longaker, M.T. Wound repair and regeneration. Nature 2008, 453, 314–321. [Google Scholar] [CrossRef]
- Menke, N.B.; Ward, K.R.; Witten, T.M.; Bonchev, D.G.; Diegelmann, R.F. Impaired wound healing. Clin. Dermatol. 2007, 25, 19–25. [Google Scholar] [CrossRef]
- Greenhalgh, D.G.; Sprugel, K.H.; Murray, M.J.; Ross, R. PDGF and FGF stimulate wound healing in the genetically diabetic mouse. Am. J. Pathol. 1990, 136, 1235–1246. [Google Scholar]
- Falanga, V. Wound healing and its impairment in the diabetic foot. Lancet 2005, 366, 1736–1743. [Google Scholar] [CrossRef]
- Blakytny, R.; Jude, E. The molecular biology of chronic wounds and delayed healing in diabetes. Diabet. Med. 2006, 23, 594–608. [Google Scholar] [CrossRef]
- Delamaire, M.; Maugendre, D.; Moreno, M.; Le Goff, M.C.; Allannic, H.; Genetet, B. Impaired leucocyte functions in diabetic patients. Diabet. Med. 1997, 14, 29–34. [Google Scholar] [CrossRef]
- Altavilla, D.; Saitta, A.; Cucinotta, D.; Galeano, M.; Deodato, B.; Colonna, M.; Torre, V.; Russo, G.; Sardella, A.; Urna, G.; et al. Inhibition of lipid peroxidation restores impaired vascular endothelial growth factor expression and stimulates wound healing and angiogenesis in the genetically diabetic mouse. Diabetes 2001, 50, 667–674. [Google Scholar] [CrossRef] [Green Version]
- Ferguson, M.W.; Herrick, S.E.; Spencer, M.J.; Shaw, J.E.; Boulton, A.J.; Sloan, P. The histology of diabetic foot ulcers. Diabet. Med. 1996, 13, SS30–SS33. [Google Scholar] [CrossRef]
- Ipaktchi, K.; Vogt, P.M. Immunology and sepsis syndrome in burn trauma. Unfallchirurg 2009, 112, 472–478. [Google Scholar] [CrossRef]
- Evers, L.H.; Bhavsar, D.; Mailänder, P. The biology of burn injury. Exp. Dermatol. 2010, 19, 777–783. [Google Scholar] [CrossRef] [PubMed]
- Arturson, G. Forty years in burns research—The postburn inflammatory response. Burns 2000, 26, 599–604. [Google Scholar] [CrossRef]
- Rawlingson, A. Nitric oxide, inflammation and acute burn injury. Burns 2003, 29, 631–640. [Google Scholar] [CrossRef]
- Lund, T.; Onarheim, H.; Reed, R.K. Pathogenesis of edema formation in burn injuries. World J. Surg. 1992, 16, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.P.; Most, D.; Efron, D.T.; Tantry, U.; Fischel, M.H.; Barbul, A. The role of iNOS in wound healing. Surgery 2001, 130, 225–229. [Google Scholar] [CrossRef]
- Witte, M.B.; Barbul, A. Role of nitric oxide in wound repair. Am. J. Surg. 2002, 183, 406–412. [Google Scholar] [CrossRef]
- Lindblom, L.; Cassuto, J.; Yregård, L.; Mattsson, U.; Tarnow, P.; Sinclair, R. Importance of nitric oxide in the regulation of burn oedema, proteinuria and urine output. Burns 2000, 26, 13–17. [Google Scholar] [CrossRef]
- Sloan, G.M.; Reinsch, J.F. Flap physiology and the prediction of flap viability. Hand Clin. 1985, 1, 609–619. [Google Scholar] [CrossRef]
- Khomullo, G.V.; Lotova, V.I.; Cherniaev, A.N.; Vinogradov, I.N. Effect of hypoxia on DNA synthesis and collagen levels in regenerating skin. Kosm. Biol. Aviakosm. Med. 1986, 20, 57–61. [Google Scholar]
- Kathawala, M.H.; Ng, W.L.; Liu, D.; Naing, M.W.; Yeong, W.Y.; Spiller, K.L.; Van Dyke, M.; Ng, K.W. Healing of Chronic Wounds: An Update of Recent Developments and Future Possibilities. Tissue Eng. Part B Rev. 2019, 25, 429–444. [Google Scholar] [CrossRef]
- Kosaric, N.; Kiwanuka, H.; Gurtner, G.C. Stem cell therapies for wound healing. Expert Opin. Biol. Ther. 2019, 19, 575–585. [Google Scholar] [CrossRef]
- Ng, W.L.; Wang, S.; Yeong, W.Y.; Naing, M.W. Skin Bioprinting: Impending Reality or Fantasy? Trends Biotechnol. 2016, 34, 689–699. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.S.; Cho, R.K.; In, Y. The efficacy and safety of polydeoxyribonucleotide for the treatment of knee osteoarthritis: Systematic review and meta-analysis of randomized controlled trials. Medicine 2019, 98, e17386. [Google Scholar] [CrossRef]
- Tonello, G.; Daglio, M.; Zaccarelli, N.; Sottofattori, E.; Mazzei, M.; Balbi, A. Characterization and quantitation of the active polynucleotide fraction (PDRN) from human placenta, a tissue repair stimulating agent. J. Pharm. Biomed. Anal. 1996, 14, 1555–1560. [Google Scholar] [CrossRef]
- Chavan, A.J.; Haley, B.E.; Volkin, D.B.; Marfia, K.E.; Verticelli, A.M.; Bruner, M.W.; Draper, J.P.; Burke, C.J.; Middaugh, C.R. Interaction of nucleotides with acidic fibroblast growth factor (FGF-1). Biochemestry 1994, 33, 7193–7202. [Google Scholar] [CrossRef] [PubMed]
- Middlemiss, P.J.; Gysbers, J.W.; Rathbone, M.P. Extracellular guanosine and guanosine-5’-trisphoshate increase NGF synthesis and release from cultured mouse neopallial astrocytes. Brain Res. 1995, 677, 152–156. [Google Scholar] [CrossRef]
- Irrera, N.; D’Ascola, A.; Pallio, G.; Bitto, A.; Mannino, F.; Arcoraci, V.; Rottura, M.; Ieni, A.; Minutoli, L.; Metro, D.; et al. β-Caryophyllene Inhibits Cell Proliferation through a Direct Modulation of CB2 Receptors in Glioblastoma Cells. Cancers 2020, 12, 1038. [Google Scholar] [CrossRef] [Green Version]
- Thellung, S.; Florio, T.; Maragliano, A.; Cattarini, G.; Schettini, G. Polydeoxyribonucleotides enhance the proliferation of human skin fibroblasts: Involvement of A2 purinergic receptor subtypes. Life Sci. 1999, 64, 1661–1674. [Google Scholar] [CrossRef]
- Muratore, O.; Schito, A.P.; Cattarini, G.; Tonoli, E.L.; Gianoglio, S.; Schiappacasse, S.; Felli, L.; Picchetta, F.; Schito, G.C. Evaluation of the trophic effect of human placental polydeoxyribonucleotide on human knee skin fibroblasts in primary culture. Cell Mol. Life Sci. 1997, 53, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Gennero, L.; Denysenko, T.; Calisti, G.F.; Vercelli, A.; Vercelli, C.M.; Amedeo, S.; Mioletti, S.; Parino, E.; Montanaro, M.; Melcarne, A.; et al. Protective effects of polydeoxyribonucleotides on cartilage degradation in experimental cultures. Cell Biochem. Funct. 2013, 31, 214–227. [Google Scholar] [CrossRef]
- Raposio, E.; Guida, C.; Coradeghini, R.; Scanarotti, C.; Parodi, A.; Baldelli, I.; Fiocca, R.; Santi, P.L. In vitro polydeoxyribonucleotide effects on human pre-adipocytes. Cell Prolif. 2008, 41, 739–754. [Google Scholar] [CrossRef]
- Guizzardi, S.; Galli, C.; Govoni, P.; Boratto, R.; Cattarini, G.; Martini, D.; Belletti, S.; Scandroglio, R. Polydeoxyribonucleotide (PDRN) promotes human osteoblast proliferation: A new proposal for bone tissue repair. Life Sci. 2003, 29, 1973–1983. [Google Scholar] [CrossRef]
- Guizzardi, S.; Martini, D.; Bacchelli, B.; Valdatta, L.; Thione, A.; Scamoni, S.; Uggeri, J.; Ruggeri, A. Effect of heat deproteinate bone and polynucleotides on bone regeneration: An experimental study on rat. Micron 2007, 38, 722–728. [Google Scholar] [CrossRef] [PubMed]
- Sini, P.; Denti, A.; Cattarini, G.; Daglio, M.; Tira, M.E.; Balduini, C. Effect of polydeoxyribonucleotides on human fibroblasts in primary culture. Cell Biochem. Funct. 1999, 17, 107–114. [Google Scholar] [CrossRef]
- Belletti, S.; Uggeri, J.; Gatti, R.; Govoni, P.; Guizzardi, S. Polydeoxyribonucleotide promotes cyclobutane pyrimidine dimer repair in UVB-exposed dermal fibroblasts. Photodermatol. Photoimmunol. Photomed. 2007, 23, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Muratore, O.; Cattarini, G.; Gianoglio, S.; Tonoli, E.L.; Saccà, S.C.; Ghiglione, D.; Venzano, D.; Ciurlo, C.; Lantieri, P.B.; Schito, G.C. A human placental polydeoxyribonucleotide (PDRN) may promote the growth of human corneal fibroblasts and iris pigment epithelial cells in primary culture. New Microbiol. 2003, 26, 13–26. [Google Scholar]
- Galeano, M.; Bitto, A.; Altavilla, D.; Minutoli, L.; Polito, F.; Calò, M.; Lo Cascio, P.; d’Alcontres, F.S.; Squadrito, F. Polydeoxyribonucleotide stimulates angiogenesis and wound healing in the genetically diabetic mouse. Wound Repair Regen. 2008, 16, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Kwon, T.R.; Han, S.W.; Kim, J.H.; Lee, B.C.; Kim, J.M.; Hong, J.Y.; Kim, B.J. Polydeoxyribonucleotides Improve Diabetic Wound Healing in Mouse Animal Model for Experimental Validation. Ann. Dermatol. 2019, 31, 403–413. [Google Scholar] [CrossRef]
- Altavilla, D.; Squadrito, F.; Polito, F.; Irrera, N.; Calò, M.; Lo Cascio, P.; Galeano, M.; La Cava, L.; Minutoli, L.; Marini, H.; et al. Activation of adenosine A2A receptors restores the altered cell-cycle machinery during impaired wound healing in genetically diabetic mice. Surgery 2011, 149, 253–261. [Google Scholar] [CrossRef]
- Jeong, W.; Yang, C.E.; Roh, T.S.; Kim, J.H.; Lee, J.H.; Lee, W.J. Scar Prevention and Enhanced Wound Healing Induced by Polydeoxyribonucleotide in a Rat Incisional Wound-Healing Model. Int. J. Mol. Sci. 2017, 18, 1698. [Google Scholar] [CrossRef]
- Shin, D.Y.; Park, J.U.; Choi, M.H.; Kim, S.; Kim, H.E.; Jeong, S.H. Polydeoxyribonucleotide-delivering therapeutic hydrogel for diabetic wound healing. Sci. Rep. 2020, 10, 16811. [Google Scholar] [CrossRef]
- Bitto, A.; Galeano, M.; Squadrito, F.; Minutoli, L.; Polito, F.; Dye, J.F.; Clayton, E.A.; Calò, M.; Venuti, F.S.; Vaccaro, M.; et al. Polydeoxyribonucleotide improves angiogenesis and wound healing in experimental thermal injury. Crit. Care Med. 2008, 36, 1594–1602. [Google Scholar] [CrossRef]
- Polito, F.; Bitto, A.; Galeano, M.; Irrera, N.; Marini, H.; Calò, M.; Squadrito, F.; Altavilla, D. Polydeoxyribonucleotide restores blood flow in an experimental model of ischemic skin flaps. J. Vasc. Surg. 2012, 55, 479–488. [Google Scholar] [CrossRef] [Green Version]
- Chung, K.I.; Kim, H.K.; Kim, W.S.; Bae, T.H. The effects of polydeoxyribonucleotide on the survival of random pattern skin flaps in rats. Arch. Plast. Surg. 2013, 40, 181–186. [Google Scholar] [CrossRef] [Green Version]
- Lee, D.W.; Hong, H.J.; Roh, H.; Lee, W.J. The Effect of Polydeoxyribonucleotide on Ischemic Rat Skin Flap Survival. Ann. Plast. Surg. 2015, 75, 84–90. [Google Scholar] [CrossRef]
- Rubegni, P.; De Aloe, G.; Mazzatenta, C.; Cattarini, L.; Fimiani, M. Clinical evaluation of the trophic effect of polydeoxyribonucleotide (PDRN) in patients undergoing skin explants. A Pilot Study. Curr. Med. Res. Opin. 2001, 17, 128–131. [Google Scholar] [CrossRef] [PubMed]
- Valdatta, L.; Thione, A.; Mortarino, C.; Buoro, M.; Tuinder, S. Evaluation of the efficacy of polydeoxyribonucleotides in the healing process of autologous skin graft donor sites: A pilot study. Curr. Med. Res. Opin. 2004, 20, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Squadrito, F.; Bitto, A.; Altavilla, D.; Arcoraci, V.; De Caridi, G.; De Feo, M.E.; Corrao, S.; Pallio, G.; Sterrantino, C.; Minutoli, L.; et al. The effect of PDRN, An adenosine receptor A2A agonist, On the healing of chronic diabetic foot ulcers: Results of a clinical trial. J. Clin. Endocrinol. Metab. 2014, 99, E746–E753. [Google Scholar]
- Kim, S.; Kim, J.; Choi, J.; Jeong, W.; Kwon, S. Polydeoxyribonucleotide Improves Peripheral Tissue Oxygenation and Accelerates Angiogenesis in Diabetic Foot Ulcers. Arch. Plast. Surg. 2017, 44, 482–489. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.Y.; Pak, C.S.; Park, J.H.; Jeong, J.H.; Heo, C.Y. Effects of polydeoxyribonucleotide in the treatment of pressure ulcers. J. Korean Med. Sci. 2014, 29, S222–S227. [Google Scholar] [CrossRef] [Green Version]
- Haskó, G.; Linden, J.; Cronstein, B.; Pacher, P. Adenosine receptors: Therapeutic aspects for inflammatory and immune diseases. Nat. Rev. Drug Discov. 2008, 7, 759–770. [Google Scholar] [CrossRef] [PubMed]
- Burnstock, G.; Knight, G.E.; Greig, A.V. Purinergic signaling in healthy and diseased skin. J. Investig. Dermatol. 2012, 132, 526–546. [Google Scholar] [CrossRef] [Green Version]
- Ernens, I.; Léonard, F.; Vausort, M.; Rolland-Turner, M.; Devaux, Y.; Wagner, D.R. Adenosine up-regulates vascular endothelial growth factor in human macrophages. Biochem. Biophys. Res. Commun. 2010, 12, 351–356. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.H.; Jacobson, K.A.; Kim, J.; Heppel, L.A. Presence of both A1 and A2a adenosine receptors in human cells and their interaction. Biochem. Biophys. Res. Commun. 1995, 17, 871–878. [Google Scholar] [CrossRef] [Green Version]
- Olanrewaju, H.A.; Qin, W.; Feoktistov, I.; Scemama, J.L.; Mustafa, S.J. Adenosine A2A and A2B receptors in cultured human and porcine coronary artery endothelial cells. Am. J. Physiol. Heart Circ. Physiol. 2000, 279, H650–H656. [Google Scholar] [CrossRef] [PubMed]
- Valls, M.D.; Cronstein, B.N.; Montesinos, M.C. Adenosine receptor agonists for promotion of dermal wound healing. Biochem. Pharm. 2009, 77, 1117–1124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Altavilla, D.; Bitto, A.; Polito, F.; Marini, H.; Minutoli, L.; Di Stefano, V.; Irrera, N.; Cattarini, G.; Squadrito, F. Polydeoxyribonucleotide (PDRN): A safe approach to induce therapeutic angiogenesis in peripheral artery occlusive disease and in diabetic foot ulcers. Cardiovasc. Hematol. Agents Med. Chem. 2009, 7, 313–321. [Google Scholar] [CrossRef]
| Study | Experimental Model | PDRN DOSES | PDRN Effects | References |
|---|---|---|---|---|
| Thellung et al., 1999 | In vitro study Human Skin fibroblasts | 10 mg/mL | ↑ Cell growth | [28] |
| Raposio et al., 2008 | In vitro study Human Pre-adipocytes | 80–100 mg/mL | ↑ Cell proliferation | [31] |
| Sini et al., 1999 | In vitro study Human Skin fibroblasts | 80 mg/mL | ↑ Cell growth | [34] |
| Belletti et al., 2007 | In vitro study Human skin fibroblasts | 100 mg/mL | ↑ DNA repair | [35] |
| Muratori et al., 2003 | In vitro study Human Corneal fibroblasts | 100 mg/mL | ↑ Cell growth | [36] |
| Galeano et al., 2008 | Animal study C57BL/ksJ-m+/+Leptdb (db+/db+) and (db+/+m) | 8 mg/kg | ↑ Wound-breaking strength ↑ VEGF expression | [37] |
| Kwon et al., 2019 | Animal study BKS.Cg-+Leprdb/+Leprdb and m+/Leprdb | 0.09 mg/mice | ↑ Wound-breaking strength ↑ VEGF expression | [38] |
| Altavilla et al., 2011 | Animal study C57BL/ksJ-m+/+Leptdb (db+/db+) and (db+/+m) | 8 mg/kg | ↑ Cyclin D1/CDK6, ↑ Cyclin E/CDK2 ↓ p15 and p27 | [39] |
| Jeong et al., 2017 | Animal study Sprague-Dawley | 8 mg/kg | ↓ Inflammatory cell infiltrate ↑ Collagen deposition | [40] |
| Shin et al., 2020 | Animal study C57BLKS/J -db/db | 100 mg/mL | ↑ Collagen density ↑ VEGF and α-SMA ↓ Number of inflammatory cells | [41] |
| Bitto et al., 2008 | Animal study C57BL/6 | 8 mg/kg | ↓ TNF-α wound ↑ VEGF, eNOS, iNOS | [42] |
| Polito et al., 2012 | Animal study Sprague-Dawley | 8 mg/kg | ↑ VEGF and iNOS ↓ HIF-1α | [43] |
| Chung et al., 2013 | Animal study Sprague-Dawley | 8 mg/kg | ↑ VEGF, PECAM, CD31 | [44] |
| Lee et al., 2015 | Animal study Sprague-Dawley | 8 mg/kg | ↑ VEGF and CD31 | [45] |
| Rubegni et al., 2001 | Clinical study | 5.625 mg/die | ↑ Re-epithelialisation | [46] |
| Valdatta et al., 2004 | Clinical study | 5.625 mg/die | ↑ Re-epithelialisation ↓ Time to complete wound healing | [47] |
| Squadrito et al., 2014 | Clinical study | 5.625 mg/die | ↑ Re-epithelialisation ↓ Time to complete wound healing | [48] |
| Kim et al., 2017 | Clinical study | 5.625 mg/die | ↑ Tissue oxygenation ↓ Inflammatory infiltrate | [49] |
| Kim et al., 2014 | Clinical study | 5.625 mg/die | ↓ Wound size ↓ PUSH score | [50] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Galeano, M.; Pallio, G.; Irrera, N.; Mannino, F.; Bitto, A.; Altavilla, D.; Vaccaro, M.; Squadrito, G.; Arcoraci, V.; Colonna, M.R.; et al. Polydeoxyribonucleotide: A Promising Biological Platform to Accelerate Impaired Skin Wound Healing. Pharmaceuticals 2021, 14, 1103. https://doi.org/10.3390/ph14111103
Galeano M, Pallio G, Irrera N, Mannino F, Bitto A, Altavilla D, Vaccaro M, Squadrito G, Arcoraci V, Colonna MR, et al. Polydeoxyribonucleotide: A Promising Biological Platform to Accelerate Impaired Skin Wound Healing. Pharmaceuticals. 2021; 14(11):1103. https://doi.org/10.3390/ph14111103
Chicago/Turabian StyleGaleano, Mariarosaria, Giovanni Pallio, Natasha Irrera, Federica Mannino, Alessandra Bitto, Domenica Altavilla, Mario Vaccaro, Giovanni Squadrito, Vincenzo Arcoraci, Michele Rosario Colonna, and et al. 2021. "Polydeoxyribonucleotide: A Promising Biological Platform to Accelerate Impaired Skin Wound Healing" Pharmaceuticals 14, no. 11: 1103. https://doi.org/10.3390/ph14111103
APA StyleGaleano, M., Pallio, G., Irrera, N., Mannino, F., Bitto, A., Altavilla, D., Vaccaro, M., Squadrito, G., Arcoraci, V., Colonna, M. R., Lauro, R., & Squadrito, F. (2021). Polydeoxyribonucleotide: A Promising Biological Platform to Accelerate Impaired Skin Wound Healing. Pharmaceuticals, 14(11), 1103. https://doi.org/10.3390/ph14111103









