Evaluation of Antibacterial Effects of Matrix-Induced Silver Ions against Antibiotic-Resistant ESKAPE Pathogens
Abstract
:1. Introduction
2. Results and Discussion
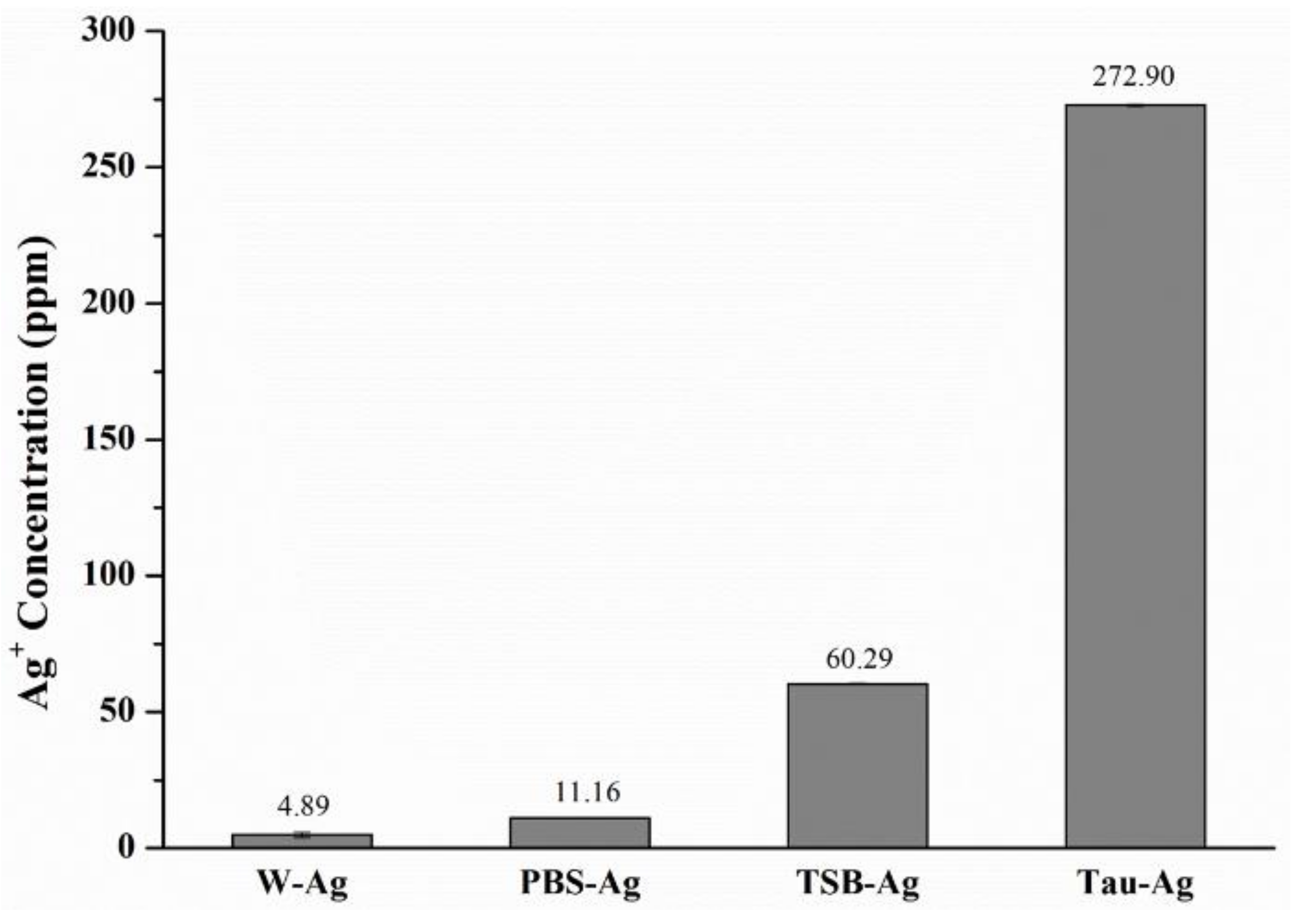
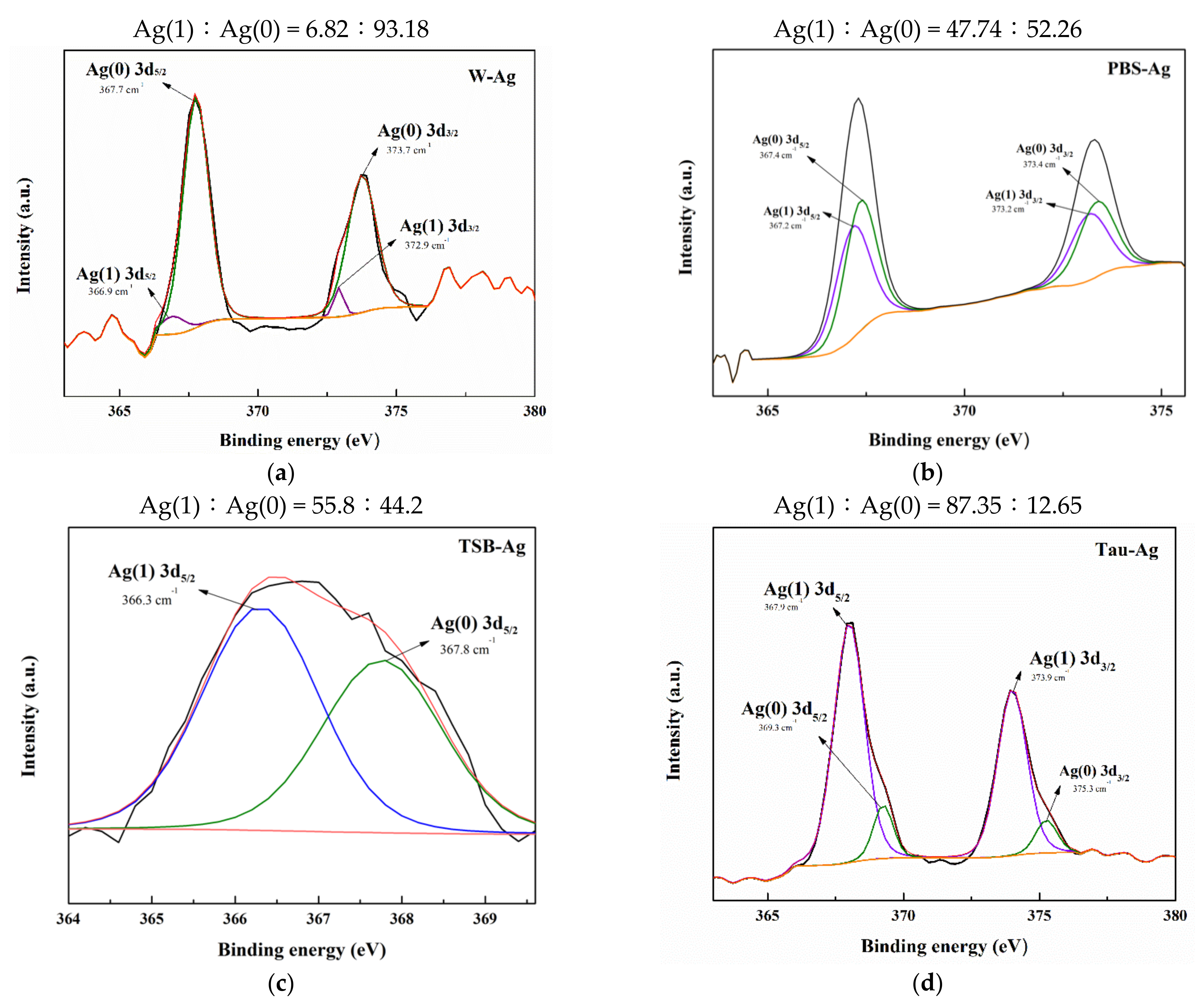
2.1. The Potency of Matrixes to Induce Silver Ions
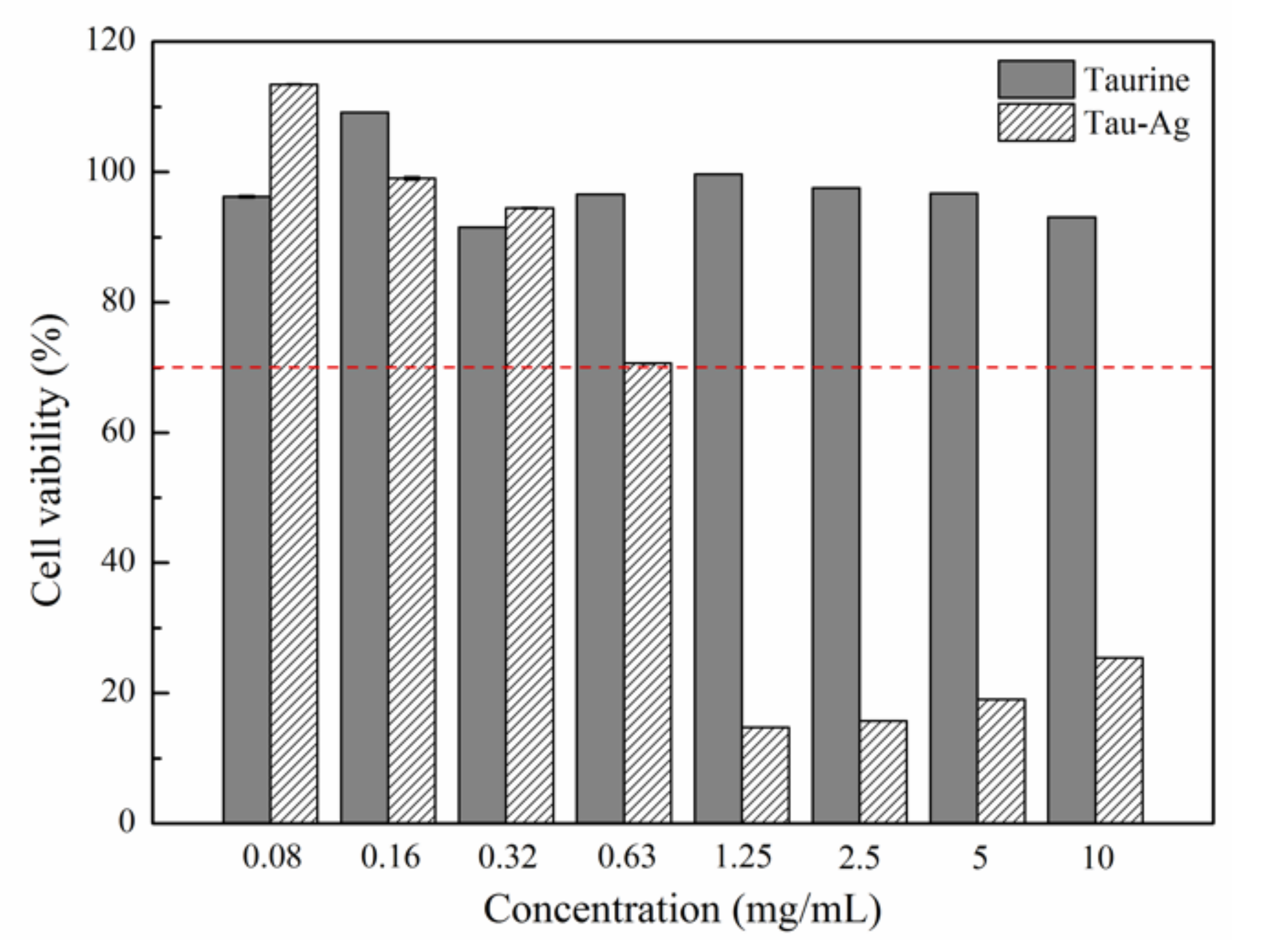
2.2. Cytotoxicity of Tau-Ag
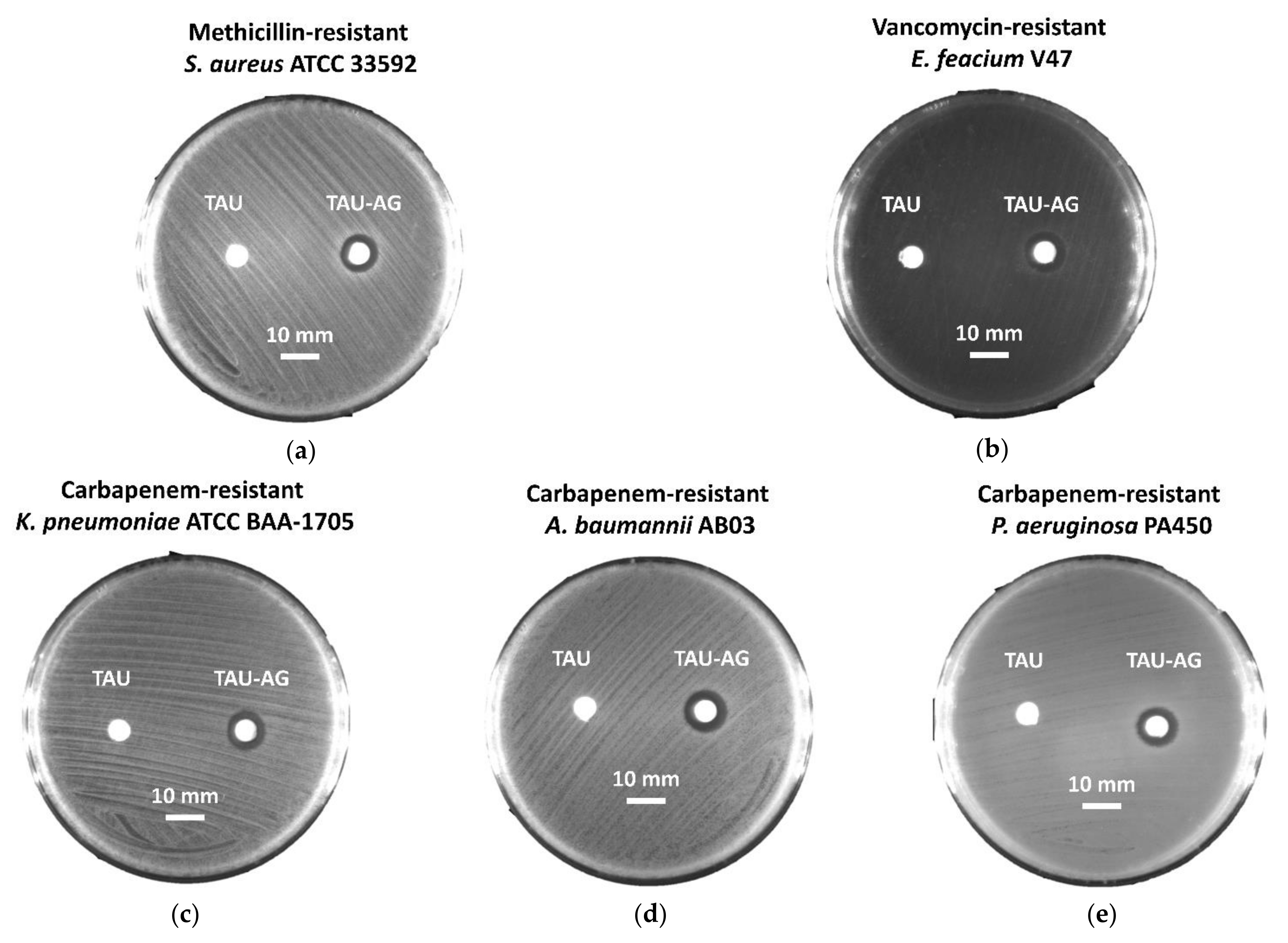
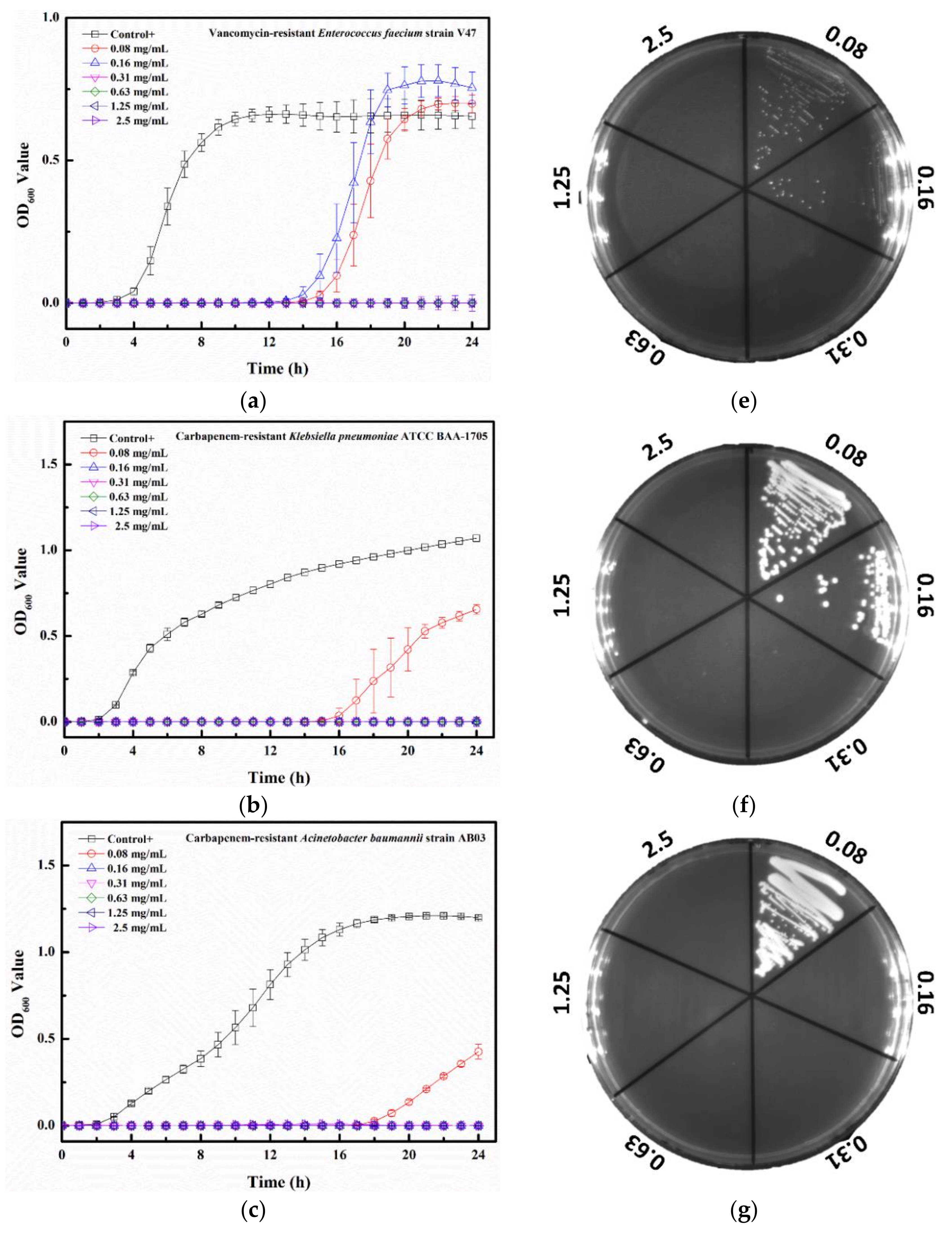
2.3. Tau-Ag Combats Antibiotic-Resistant ESKAPE Pathogens
3. Materials and Methods
3.1. Synthesis of Silver-Containing Mesoporous Bioactive Glass (MBG-Ag)
3.2. Preparation of Matrix-Induced Silver
3.3. Characterization of Matrix-Induced Silver
3.4. Analysis of Tau-Ag Matrix-Induced Silver Ions
3.5. In Vitro Cytotoxicity Testing
3.6. Bacterial Strains
3.7. Disk Diffusion Assay
3.8. Growth Curve Analysis
3.9. Colony-Forming Assay
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pendleton, J.N.; Gorman, S.P.; Gilmore, B.F. Clinical relevance of the ESKAPE pathogens. Expert Rev. Anti-Infect. Ther. 2013, 11, 297–308. [Google Scholar] [CrossRef] [PubMed]
- Santajit, S.; Indrawattana, N. Mechanisms of antimicrobial resistance in ESKAPE pathogens. Biomed Res. Int. 2016, 2016, 2475067. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muhlberg, E.; Umstatter, F.; Domhan, C.; Hertlein, T.; Ohlsen, K.; Krause, A.; Kleist, C.; Beijer, B.; Zimmermann, S.; Haberkorn, U.; et al. Vancomycin-lipopeptide conjugates with high antimicrobial activity on vancomycin-resistant Enterococci. Pharmaceuticals 2020, 13, 110. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-H.; Tseng, S.-P.; Wu, S.-M.; Shih, C.-J. Structure-dependence of anti-methicillin-resistant Staphylococcus aureus (MRSA) activity on ZnO-containing bioglass. J. Alloys Compd. 2020, 848, 156487. [Google Scholar] [CrossRef]
- Codjoe, F.S.; Donkor, E.S. Carbapenem resistance: A Review. Med. Sci. 2017, 6, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, T.-Y.; Kao, H.-Y.; Lu, P.-L.; Chen, P.-Y.; Wang, S.-C.; Wang, L.-C.; Hsieh, Y.-J.; Tseng, S.-P. Evaluation of the organotellurium compound AS101 for treating colistin- and carbapenem-resistant Klebsiella pneumoniae. Pharmaceuticals 2021, 14, 795. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.-Y.; Tseng, S.-P.; Dlamini, H.N.; Lu, P.-L.; Lin, L.; Wang, L.-C.; Hung, W.-C. In vitro and in vivo activity of AS101 against carbapenem-resistant Acinetobacter baumannii. Pharmaceuticals 2021, 14, 823. [Google Scholar] [CrossRef] [PubMed]
- Engeman, E.; Freyberger, H.R.; Corey, B.W.; Ward, A.M.; He, Y.; Nikolich, M.P.; Filippov, A.A.; Tyner, S.D.; Jacobs, A.C. Synergistic killing and re-sensitization of Pseudomonas aeruginosa to antibiotics by phage-antibiotic combination treatment. Pharmaceuticals 2021, 14, 184. [Google Scholar] [CrossRef]
- World Health Organization. WHO Publishes List of Bacteria for Which New Antibiotics Are Urgently Needed. Available online: https://www.who.int/news/item/27-02-2017-who-publishes-list-of-bacteria-for-which-new-antibiotics-are-urgently-needed (accessed on 23 August 2021).
- Marques, C.F.; Olhero, S.; Abrantes, J.; Marote, A.; Ferreira, S.; Vieira, S.I.; Ferreira, J.M. Biocompatibility and antimicrobial activity of biphasic calcium phosphate powders doped with metal ions for regenerative medicine. Ceram. Int. 2017, 43, 15719–15728. [Google Scholar] [CrossRef]
- Burdusel, A.C.; Gherasim, O.; Grumezescu, A.M.; Mogoanta, L.; Ficai, A.; Andronescu, E. Biomedical applications of silver nanoparticles: An Up-to-Date Overview. Nanomaterials 2018, 8, 681. [Google Scholar] [CrossRef] [Green Version]
- Lok, C.N.; Ho, C.M.; Chen, R.; He, Q.Y.; Yu, W.Y.; Sun, H.; Tam, P.K.; Chiu, J.F.; Che, C.M. Silver nanoparticles: Partial oxidation and antibacterial activities. J. Biol. Inorg. Chem. 2007, 12, 527–534. [Google Scholar] [CrossRef]
- Yun’an Qing, L.C.; Li, R.; Liu, G.; Zhang, Y.; Tang, X.; Wang, J.; Liu, H.; Qin, Y. Potential antibacterial mechanism of silver nanoparticles and the optimization of orthopedic implants by advanced modification technologies. Int. J. Nanomed. 2018, 13, 3311. [Google Scholar]
- Zheng, K.; Setyawati, M.I.; Leong, D.T.; Xie, J. Antimicrobial silver nanomaterials. Coord. Chem. Rev. 2018, 357, 1–17. [Google Scholar] [CrossRef]
- Pourshahrestani, S.; Zeimaran, E.; Kadri, N.A.; Gargiulo, N.; Jindal, H.M.; Hasikin, K.; Naveen, S.V.; Sekaran, S.D.; Kamarul, T. Elastomeric biocomposite of silver-containing mesoporous bioactive glass and poly (1, 8-octanediol citrate): Physiochemistry and in vitro antibacterial capacity in tissue engineering applications. Mater. Sci. Eng. C 2019, 98, 1022–1033. [Google Scholar] [CrossRef]
- Rahman, M.S.; Tahir, M.A.; Noreen, S.; Yasir, M.; Khan, M.B.; Mahmood, T.; Bahadur, A.; Shoaib, M. Osteogenic silver oxide doped mesoporous bioactive glass for controlled release of doxorubicin against bone cancer cell line (MG-63): In vitro and in vivo cytotoxicity evaluation. Ceram. Int. 2020, 46, 10765–10770. [Google Scholar] [CrossRef]
- Chen, Y.-H.; Kung, J.-C.; Tseng, S.-P.; Chen, W.-C.; Wu, S.-M.; Shih, C.-J. Effects of AgNPs on the structure and anti-methicillin resistant Staphylococcus aureus (MRSA) properties of SiO2-CaO-P2O5 bioactive glass. J. Non-Cryst. Solids 2021, 553, 120492. [Google Scholar] [CrossRef]
- Letaïef, N.; Lucas-Girot, A.; Oudadesse, H.; Dorbez-Sridi, R.; Boullay, P. Investigation of the surfactant type effect on characteristics and bioactivity of new mesoporous bioactive glass in the ternary system SiO2–CaO–P2O5: Structural, textural and reactivity studies. Microporous Mesoporous Mater. 2014, 195, 102–111. [Google Scholar] [CrossRef]
- Wang, X.; Wang, G.; Zhang, Y. Research on the biological activity and doxorubicin release behavior in vitro of mesoporous bioactive SiO2-CaO-P2O5 glass nanospheres. Appl. Surf. Sci. 2017, 419, 531–539. [Google Scholar] [CrossRef]
- Wu, Z.; Zhou, X.; Zhang, Y.; Luan, J.; Yang, X.; Wu, Z.; Wang, B. Synthesis and characterization of mesoporous bioactive glasses with highly ordered structures and high surface areas by a self-assembly process. J. Non-Cryst. Solids 2019, 517, 1–8. [Google Scholar] [CrossRef]
- Bai, N.; Chen, W.; Luo, L.; Tong, W.; Wen, C.; Zhan, X.; Sa, B. Effect of B2O3 on the structural and in vitro biological assessment of mesoporous bioactive glass nanospheres. J. Am. Ceram. Soc. 2021, 104, 3058–3072. [Google Scholar] [CrossRef]
- Kumar, A.; Mittal, A.; Das, A.; Sen, D.; Mariappan, C. Mesoporous electroactive silver doped calcium borosilicates: Structural, antibacterial and myogenic potential relationship of improved bio-ceramics. Ceram. Int. 2021, 47, 3586–3596. [Google Scholar] [CrossRef]
- Schumacher, M.; Habibovic, P.; van Rijt, S. Mesoporous bioactive glass composition effects on degradation and bioactivity. Bioact. Mater. 2021, 6, 1921–1931. [Google Scholar] [CrossRef]
- Yang, T.Y.; Hsieh, Y.J.; Lu, P.L.; Lin, L.; Wang, L.C.; Wang, H.Y.; Tsai, T.H.; Shih, C.J.; Tseng, S.P. In vitro and in vivo assessments of inspired Ag/80S bioactive nanocomposites against carbapenem-resistant Klebsiella pneumoniae. Mater. Sci. Eng. C 2021, 125, 112093. [Google Scholar] [CrossRef]
- Li, S.; Shen, Y.; Xie, A.; Yu, X.; Qiu, L.; Zhang, L.; Zhang, Q. Green synthesis of silver nanoparticles using Capsicum annuum L. extract. Green Chem. 2007, 9, 852–858. [Google Scholar] [CrossRef]
- Petrova, Y.S.; Neudachina, L. Potentiometric study of complexation between taurine and metal ions. Russ. J. Inorg. Chem. 2013, 58, 617–620. [Google Scholar] [CrossRef] [Green Version]
- Du, Z.-X.; Li, J.-X. Crystal structure of catena-poly {[chloromercury (II)]-μ-[2-(2-pyridylmethylamino) ethanesulfonato-k4N,N′,O:O′]},[Hg (C8H11N2O3S)(Cl)]n. Z. Kristallogr.-New Cryst. Struct. 2008, 223, 211–212. [Google Scholar] [CrossRef]
- Alifkhanova, L.M.; Pestov, A.V.; Mekhaev, A.V.; Marchuk, A.A.; Bosenko, S.N.; Petrova, Y.S.; Neudachina, L.K. Sulfoethylated polyaminostyrene–polymer ligand with high selective interaction with silver ions in multicomponent solutions. J. Environ. Chem. Eng. 2019, 7, 102846. [Google Scholar] [CrossRef]
- Ahmadian, M.; Roshan, V.D.; Aslani, E.; Stannard, S.R. Taurine supplementation has anti-atherogenic and anti-inflammatory effects before and after incremental exercise in heart failure. Ther. Adv. Cardiovasc. Dis. 2017, 11, 185–194. [Google Scholar] [CrossRef]
- Qaradakhi, T.; Gadanec, L.K.; McSweeney, K.R.; Abraham, J.R.; Apostolopoulos, V.; Zulli, A. The anti-inflammatory effect of taurine on cardiovascular disease. Nutrients 2020, 12, 2847. [Google Scholar] [CrossRef]
- Jakaria, M.; Azam, S.; Haque, M.E.; Jo, S.-H.; Uddin, M.S.; Kim, I.-S.; Choi, D.-K. Taurine and its analogs in neurological disorders: Focus on therapeutic potential and molecular mechanisms. Redox Biol. 2019, 24, 101223. [Google Scholar] [CrossRef]
- Stuart, B.; Stan, G.; Popa, A.; Carrington, M.; Zgura, I.; Necsulescu, M.; Grant, D.J.B.M. New solutions for combatting implant bacterial infection based on silver nano-dispersed and gallium incorporated phosphate bioactive glass sputtered films: A preliminary study. Bioact. Mater. 2022, 8, 325–340. [Google Scholar] [CrossRef] [PubMed]
- Vijayarohini, P.; Mercy, A.S.C.; Kavitha, G.; Alwar, S.B.S. Enhanced biological properties of taurine metal complexes via binding active sites. Mater. Today Proc. 2020, 33, 2631–2640. [Google Scholar] [CrossRef]
- Zhang, Z.; Kuang, Y.; Lin, Y.; Wu, D. A closed-loop sustainable scheme for silver recovery from water by reusable thiol-grafted graphene oxide. J. Clean. Prod. 2021, 305, 127146. [Google Scholar] [CrossRef]
- Aglan, H.S.; Safar, M.M.; Ain-Shoka, A.A.M.; Kandil, A.M.; Gebremedhn, S.; Salilew-Wondim, D.; Schellander, K.; Tesfaye, D. Developmental toxicity of lead in rats after gestational exposure and the protective role of taurine. J. Biochem. Mol. Toxicol. 2021, e22816. [Google Scholar] [CrossRef]
- Yan, X.T.; He, B.; Liu, L.H.; Qu, G.B.; Shi, J.B.; Hu, L.G.; Jiang, G.B. Antibacterial mechanism of silver nanoparticles in Pseudomonas aeruginosa: Proteomics approach. Metallomics 2018, 10, 557–564. [Google Scholar] [CrossRef]
- Hamad, A.; Khashan, K.S.; Hadi, A. Silver nanoparticles and silver ions as potential antibacterial agents. J. Inorg. Organomet. Polym. Mater. 2020, 1–18. [Google Scholar] [CrossRef]
- Tian, T.; Dang, B.; Li, F.; Yang, K.; Wei, D.; Zhang, P. Microstructure, Mechanical and antibacterial properties of TiNb-based alloy implanted by silver ions. Coatings 2021, 11, 1213. [Google Scholar] [CrossRef]
- Maiti, N.; Thomas, S.; Debnath, A.; Kapoor, S. Raman and XPS study on the interaction of taurine with silver nanoparticles. RSC Adv. 2016, 6, 56406–56411. [Google Scholar] [CrossRef]
- Salleh, A.; Naomi, R.; Utami, N.D.; Mohammad, A.W.; Mahmoudi, E.; Mustafa, N.; Fauzi, M.B. The potential of silver nanoparticles for antiviral and antibacterial applications: A mechanism of action. Nanomaterials 2020, 10, 1566. [Google Scholar] [CrossRef]
- Dowling, D.; Betts, A.; Pope, C.; McConnell, M.; Eloy, R.; Arnaud, M. Anti-bacterial silver coatings exhibiting enhanced activity through the addition of platinum. Surf. Coat. Technol. 2003, 163, 637–640. [Google Scholar] [CrossRef]
- Li, W.-R.; Sun, T.-L.; Zhou, S.-L.; Ma, Y.-K.; Shi, Q.-S.; Xie, X.-B.; Huang, X.-M. A comparative analysis of antibacterial activity, dynamics, and effects of silver ions and silver nanoparticles against four bacterial strains. Int. Biodeterior. Biodegrad. 2017, 123, 304–310. [Google Scholar] [CrossRef]
- Marcinkiewicz, J.; Kontny, E. Taurine and inflammatory diseases. Amino Acids 2014, 46, 7–20. [Google Scholar] [CrossRef] [Green Version]
- Chiarla, C.; Giovannini, I.; Siegel, J.H.; Boldrini, G.; Castagneto, M. The relationship between plasma taurine and other amino acid levels in human sepsis. J. Nutr. 2000, 130, 2222–2227. [Google Scholar] [CrossRef]
- Koohkan, R.; Hooshmand, T.; Tahriri, M.; Mohebbi-Kalhori, D. Synthesis, characterization and in vitro bioactivity of mesoporous copper silicate bioactive glasses. Ceram. Int. 2018, 44, 2390–2399. [Google Scholar] [CrossRef]
- Chien, C.-S.; Lin, C.-J.; Ko, C.-J.; Tseng, S.-P.; Shih, C.-J. Antibacterial activity of silver nanoparticles (AgNP) confined to mesostructured silica against methicillin-resistant Staphylococcus aureus (MRSA). J. Alloys Compd. 2018, 747, 1–7. [Google Scholar] [CrossRef]
- Bhatia, S.K.; Yetter, A.B. Correlation of visual in vitro cytotoxicity ratings of biomaterials with quantitative in vitro cell viability measurements. Cell Biol. Toxicol. 2008, 24, 315–319. [Google Scholar] [CrossRef]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, Y.-C.; Yang, T.-Y.; Chen, B.-X.; Kung, J.-C.; Shih, C.-J. Evaluation of Antibacterial Effects of Matrix-Induced Silver Ions against Antibiotic-Resistant ESKAPE Pathogens. Pharmaceuticals 2021, 14, 1094. https://doi.org/10.3390/ph14111094
Huang Y-C, Yang T-Y, Chen B-X, Kung J-C, Shih C-J. Evaluation of Antibacterial Effects of Matrix-Induced Silver Ions against Antibiotic-Resistant ESKAPE Pathogens. Pharmaceuticals. 2021; 14(11):1094. https://doi.org/10.3390/ph14111094
Chicago/Turabian StyleHuang, Ya-Chi, Tsung-Ying Yang, Bo-Xuan Chen, Jung-Chang Kung, and Chi-Jen Shih. 2021. "Evaluation of Antibacterial Effects of Matrix-Induced Silver Ions against Antibiotic-Resistant ESKAPE Pathogens" Pharmaceuticals 14, no. 11: 1094. https://doi.org/10.3390/ph14111094
APA StyleHuang, Y.-C., Yang, T.-Y., Chen, B.-X., Kung, J.-C., & Shih, C.-J. (2021). Evaluation of Antibacterial Effects of Matrix-Induced Silver Ions against Antibiotic-Resistant ESKAPE Pathogens. Pharmaceuticals, 14(11), 1094. https://doi.org/10.3390/ph14111094







