Rediscovering Psilocybin as an Antidepressive Treatment Strategy
Abstract
1. Introduction
2. Materials and Methods
3. Discussion
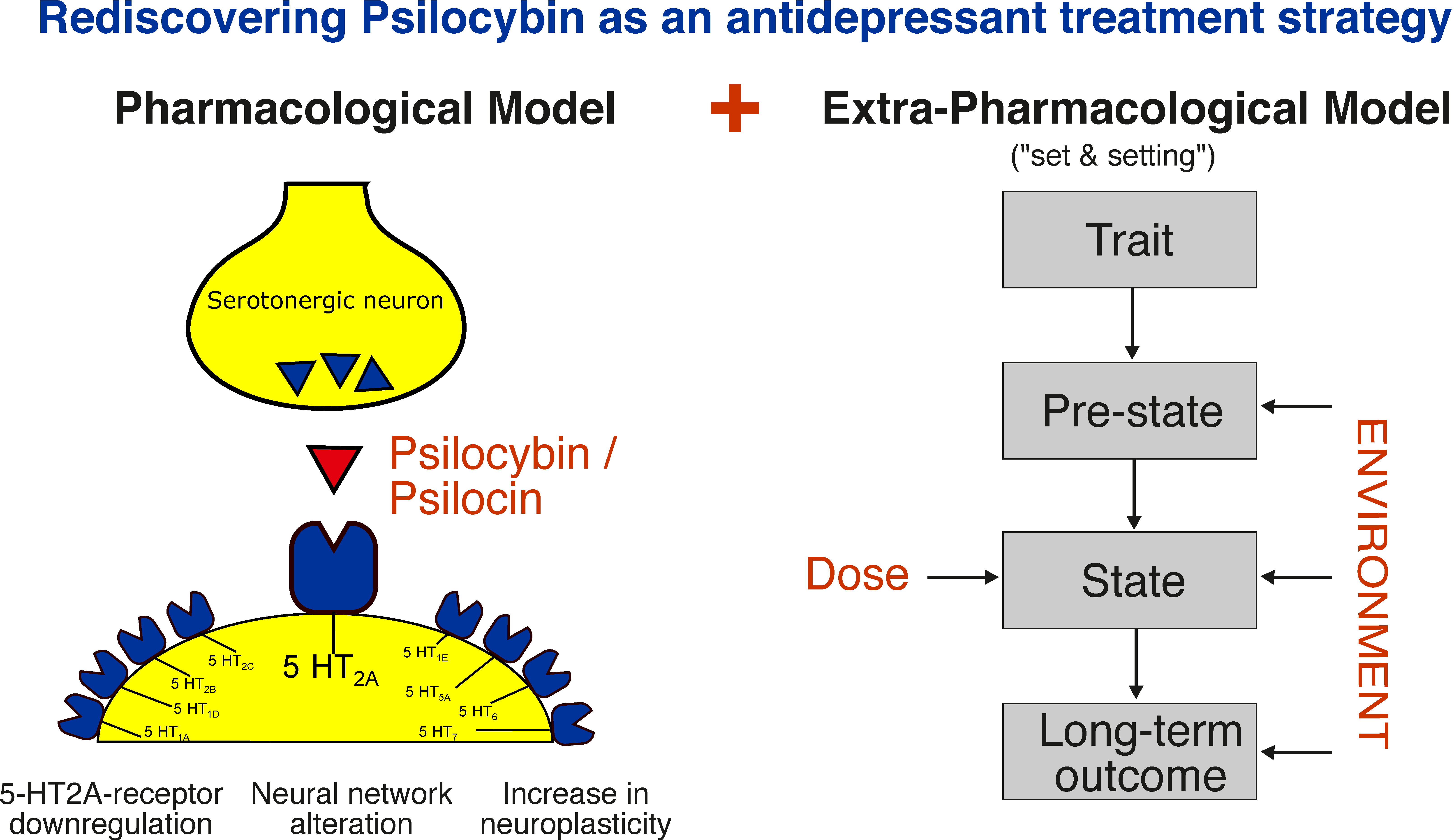
3.1. Mechanism of Action
3.1.1. Pharmacological, Neurobiological and Neuroimaging Findings
3.1.2. Set and Setting: Nondrug Parameters of Psychopharmacology and the Role of the Psychedelic Experience
3.2. Psilocybin in Antidepressive Therapy
3.2.1. Antidepressive Effects
3.2.2. Systematic Reviews and Meta-Analysis
3.3. Adverse Drug Reactions and Harm Potential
3.3.1. Physical and Mental Harm Potential
3.3.2. Adverse Drug Reactions in Therapeutic Settings
3.4. Limitations of Current Research, Challenges and an Outlook
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Abbafati, C.; Machado, D.B.; Cislaghi, B.; Salman, O.M.; Karanikolos, M.; McKee, M.; Abbas, K.M.; Brady, O.J.; Larson, H.J.; Trias-Llimós, S.; et al. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1204–1222. [Google Scholar] [CrossRef]
- Geschwind, D.H.; Flint, J. Genetics and genomics of psychiatric disease. Science 2015, 349, 1489–1494. [Google Scholar] [CrossRef] [PubMed]
- Otte, C.; Gold, S.M.; Penninx, B.W.; Pariante, C.M.; Etkin, A.; Fava, M.; Mohr, D.C.; Schatzberg, A.F. Major depressive disorder. Nat. Rev. Dis. Prim. 2016, 2, 16065. [Google Scholar] [CrossRef] [PubMed]
- Koesters, M.; Ostuzzi, G.; Guaiana, G.; Breilmann, J.; Barbui, C. Vortioxetine for Depression in Adults. Cochrane Database Syst. Rev. 2017, 7, 1–113. [Google Scholar] [CrossRef] [PubMed]
- Rizvi, S.J.; Grima, E.; Tan, M.; Rotzinger, S.; Lin, P.; McIntyre, R.S.; Kennedy, S.H. Treatment-resistant depression in primary care across Canada. Can. J. Psychiatry 2014, 59, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Bennabi, D.; Charpeaud, T.; Yrondi, A.; Genty, J.-B.; Destouches, S.; Lancrenon, S.; Alaïli, N.; Bellivier, F.; Bougerol, T.; Camus, V.; et al. Clinical guidelines for the management of treatment-resistant depression: French recommendations from experts, the French Association for Biological Psychiatry and Neuropsychopharmacology and the fondation FondaMental. BMC Psychiatry 2019, 19, 262. [Google Scholar] [CrossRef]
- Berg, R.C.; Høie, B. Effectiveness of psychotherapy for adults with depression: A systematic review of the best available evidence. Procedia Soc. Behav. Sci. 2010, 5, 2194–2200. [Google Scholar] [CrossRef][Green Version]
- Nieuwsma, J.A.; Trivedi, R.B.; McDuffie, J.; Kronish, I.; Benjamin, D.; Williams, J.W. Brief psychotherapy for depression: A systematic review and meta-analysis. Int. J. Psychiatry Med. 2012, 43, 129–151. [Google Scholar] [CrossRef]
- National Institute for Heath and Care Excellence National Institute for Health and Care Excellence Depression in Adults: Treatment and Management. 2018. Available online: https://www.nice.org.uk/guidance/gid-cgwave0725/documents/fullguideline-updated (accessed on 30 May 2021).
- Fava, M.; Davidson, K.G. Definition and epidemiology of treatment-resistant depression. Psychiatr. Clin. N. Am. 1996, 19, 179–200. [Google Scholar] [CrossRef]
- Nichols, D.E. Psychedelics. Pharm. Rev. 2016, 68, 264–355. [Google Scholar] [CrossRef]
- Tullis, P. The rise of psychedelic psychiatry. Nature 2021, 589, 506–509. [Google Scholar] [CrossRef] [PubMed]
- Tullis, P. How ecstasy and psilocybin are shaking up psychiatry. Nature 2021, 589, 506–509. [Google Scholar] [CrossRef] [PubMed]
- Nutt, D.; Erritzoe, D.; Carhart-Harris, R. Psychedelic Psychiatry’s Brave New World. Cell 2020, 181, 24–28. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, J.M.; Bogenschutz, M.; Lilienstein, A.; Harrison, C.; Kleiman, S.; Parker-Guilbert, K.; G., M.O.; Garas, W.; Paleos, C.; Gorman, I.; et al. MDMA-assisted therapy for severe PTSD: A randomized, double-blind, placebo-controlled phase 3 study. Nat. Med. 2021, 27, 1025–1033. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, A.; Heim, R.; Brack, A.; Kobel, H.; Frey, A.; Ott, H.; Petrzilka, T.; Troxler, F. Psilocybin und Psilocin, zwei psychotrope Wirkstoffe aus mexikanischen Rauschpilzen. Helv. Chim. Acta 1959, 42, 1557–1572. [Google Scholar] [CrossRef]
- Brenan, J.P.M.; Schultes, R.E.; Hofmann, A. Plants of the Gods: Origins of Hallucinogenic Use. Kew Bull. 1980, 35, 708. [Google Scholar] [CrossRef][Green Version]
- Schultes, R.E. Hallucinogens of plant origin. Science 1969, 163, 245–254. [Google Scholar] [CrossRef]
- Nichols, D.E. Psilocybin: From ancient magic to modern medicine. J. Antibiot 2020, 73, 679–686. [Google Scholar] [CrossRef]
- Wasson, G.R. Seeking the Magic Mushroom. LIFE Magazine, 15 May 1957; 109–120. [Google Scholar]
- Vollenweider, F.X.; Preller, K.H. Psychedelic drugs: Neurobiology and potential for treatment of psychiatric disorders. Nat. Rev. Neurosci. 2020, 21, 611–624. [Google Scholar] [CrossRef]
- Wark, C.; Galliher, J.F. Timothy Leary, Richard Alpert (Ram Dass) and the changing definition of psilocybin. Int. J. Drug Policy 2010, 21, 234–239. [Google Scholar] [CrossRef]
- UN General Assembly, 1971 Convention on Psychotropic Substances, 9 December 1975, A/RES/3443. Available online: https://www.refworld.org/docid/3b00f1ad4b.html (accessed on 27 September 2021).
- Abbas, A.I.; Jeanne, T.; Knox, R.; Korthuis, P.T.; Hamade, A.; Christopher, S.; Uehling, J. Oregon Psilocybin Advisory Board Rapid Evidence Review and Recommendations; Oregon Psilocybin Advisory Board: Salem, OR, USA, 2021. [Google Scholar]
- Hallucinogenic Mushrooms Drug Profile. Available online: https://www.emcdda.europa.eu/publications/drug-profiles/hallucinogenic-mushrooms_en (accessed on 17 September 2021).
- Johnson, M.W.; Garcia-Romeu, A.; Cosimano, M.P.; Griffiths, R.R. Pilot study of the 5-HT2AR agonist psilocybin in the treatment of tobacco addiction. J. Psychopharmacol. 2014, 28, 983–992. [Google Scholar] [CrossRef] [PubMed]
- Bogenschutz, M.P.; Forcehimes, A.A.; Pommy, J.A.; Wilcox, C.E.; Barbosa, P.; Strassman, R.J. Psilocybin-assisted treatment for alcohol dependence: A proof-of-concept study. J. Psychopharmacol. 2015, 29, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Grob, C.S.; Danforth, A.L.; Chopra, G.S.; Hagerty, M.; McKay, C.R.; Halberstad, A.L.; Greer, G.R. Pilot study of psilocybin treatment for anxiety in patients with advanced-stage cancer. Arch. Gen. Psychiatry 2011, 68, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Ross, S.; Bossis, A.; Guss, J.; Agin-Liebes, G.; Malone, T.; Cohen, B.; Mennenga, S.E.; Belser, A.; Kalliontzi, K.; Babb, J.; et al. Rapid and sustained symptom reduction following psilocybin treatment for anxiety and depression in patients with life-threatening cancer: A randomized controlled trial. J. Psychopharmacol. 2016, 30, 1165–1180. [Google Scholar] [CrossRef] [PubMed]
- Moreno, F.A.; Wiegand, C.B.; Taitano, E.K.; Delgado, P.L. Safety, tolerability, and efficacy of psilocybin in 9 patients with obsessive-compulsive disorder. J. Clin. Psychiatry 2006, 67, 1735–1740. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, S.B.; Pace, B.T.; Nicholas, C.R.; Raison, C.L.; Hutson, P.R. The experimental effects of psilocybin on symptoms of anxiety and depression: A meta-analysis. Psychiatry Res. 2020, 284, 112749. [Google Scholar] [CrossRef]
- FDA grants Breakthrough Therapy Designation to Usona Institute’s Psilocybin Program for Major Depressive Disorder|Business Wire. Available online: https://www.businesswire.com/news/home/20191122005452/en/FDA-grants-Breakthrough-Therapy-Designation-Usona-Institutes (accessed on 15 June 2021).
- De Gregorio, D.; Aguilar-Valles, A.; Preller, K.H.; Heifets, B.D.; Hibicke, M.; Mitchell, J.; Gobbi, G. Hallucinogens in Mental Health: Preclinical and Clinical Studies on LSD, Psilocybin, MDMA, and Ketamine. J. Neurosci. 2021, 41, 891–900. [Google Scholar] [CrossRef]
- Sard, H.; Kumaran, G.; Morency, C.; Roth, B.L.; Toth, B.A.; He, P.; Shuster, L. SAR of psilocybin analogs: Discovery of a selective 5-HT2C agonist. Bioorg. Med. Chem. Lett. 2005, 15, 4555–4559. [Google Scholar] [CrossRef]
- Blair, J.B.; Kurrasch-Orbaugh, D.; Marona-Lewicka, D.; Gumbay, M.G.; Watts, V.J.; Barker, E.L.; Nichols, D.E. Effect of ring fluorination on the pharmacology of hallucinogenic tryptamines. J. Med. Chem. 2000, 43, 4701–4710. [Google Scholar] [CrossRef]
- Halberstadt, A.L.; Geyer, M.A. Multiple receptors contribute to the behavioral effects of indoleamine hallucinogens. Neuropharmacology 2011, 61, 364–381. [Google Scholar] [CrossRef]
- Kuypers, K.P.C.; Ng, L.; Erritzoe, D.; Knudsen, G.M.; Nichols, C.D.; Nichols, D.E.; Pani, L.; Soula, A.; Nutt, D. Microdosing psychedelics: More questions than answers? An overview and suggestions for future research. J. Psychopharmacol. 2019, 33, 1039–1057. [Google Scholar] [CrossRef] [PubMed]
- Roth, B.L.; Lopez, E.; Patel, S.; Kroeze, W.K. The Multiplicity of Serotonin Receptors: Uselessly Diverse Molecules or an Embarrassment of Riches? Neurosci. 2000, 6, 252–262. [Google Scholar] [CrossRef]
- McKenna, D.J.; Peroutka, S.J. Differentiation of 5-hydroxytryptamine2 receptor subtypes using 125I-R-(-)2,5-dimethoxy-4-iodo-phenylisopropylamine and 3H-ketanserin. J. Neurosci. 1989, 9, 3482–3490. [Google Scholar] [CrossRef] [PubMed]
- Pandey, G.N.; Dwivedi, Y.; Rizavi, H.S.; Ren, X.; Pandey, S.C.; Pesold, C.; Roberts, R.C.; Conley, R.R.; Tamminga, C.A. Higher expression of serotonin 5-HT2A receptors in the postmortem brains of teenage suicide victims. Am. J. Psychiatry 2002, 159, 419–429. [Google Scholar] [CrossRef]
- Underwood, M.D.; Kassir, S.A.; Bakalian, M.J.; Galfalvy, H.; Dwork, A.J.; Mann, J.J.; Arango, V. Serotonin receptors and suicide, major depression, alcohol use disorder and reported early life adversity. Transl. Psychiatry 2018, 8, 279. [Google Scholar] [CrossRef]
- Carhart-Harris, R.L.; Nutt, D.J. Serotonin and brain function: A tale of two receptors. J. Psychopharmacol. 2017, 31, 1091–1120. [Google Scholar] [CrossRef]
- Vollenweider, F.X.; Vollenweider-Scherpenhuyzen, M.F.I.; Bäbler, A.; Vogel, H.; Hell, D. Psilocybin induces schizophrenia-like psychosis in humans via a serotonin-2 agonist action. Neuroreport 1998, 9, 3897–3902. [Google Scholar] [CrossRef]
- Kometer, M.; Schmidt, A.; Bachmann, R.; Studerus, E.; Seifritz, E.; Vollenweider, F.X. Psilocybin biases facial recognition, goal-directed behavior, and mood state toward positive relative to negative emotions through different serotonergic subreceptors. Biol. Psychiatry 2012, 72, 898–906. [Google Scholar] [CrossRef]
- Madsen, M.K.; Fisher, P.M.; Burmester, D.; Dyssegaard, A.; Stenbæk, D.S.; Kristiansen, S.; Johansen, S.S.; Lehel, S.; Linnet, K.; Svarer, C.; et al. Psychedelic effects of psilocybin correlate with serotonin 2A receptor occupancy and plasma psilocin levels. Neuropsychopharmacology 2019, 44, 1328. [Google Scholar] [CrossRef]
- Buckholtz, N.S.; Zhou, D.; Freedman, D.X. Serotonin2 agonist administration down-regulates rat brain serotonin2 receptors. Life Sci. 1988, 42, 2439–2445. [Google Scholar] [CrossRef]
- Madsen, M.K.; Fisher, P.M.D.; Stenbæk, D.S.; Kristiansen, S.; Burmester, D.; Lehel, S.; Páleníček, T.; Kuchař, M.; Svarer, C.; Ozenne, B.; et al. A single psilocybin dose is associated with long-term increased mindfulness, preceded by a proportional change in neocortical 5-HT2A receptor binding. Eur. Neuropsychopharmacol. 2020, 33, 71–80. [Google Scholar] [CrossRef]
- Raval, N.R.; Johansen, A.; Donovan, L.L.; Ros, N.F.; Ozenne, B.; Hansen, H.D.; Knudsen, G.M. A Single Dose of Psilocybin Increases Synaptic Density and Decreases 5-HT2A Receptor Density in the Pig Brain. Int. J. Mol. Sci. 2021, 22, 835. [Google Scholar] [CrossRef]
- Pittenger, C.; Duman, R.S. Stress, depression, and neuroplasticity: A convergence of mechanisms. Neuropsychopharmacology 2008, 33, 88–109. [Google Scholar] [CrossRef]
- Hyman, S.E.; Nestler, E.J. Initiation and adaptation: A paradigm for understanding psychotropic drug action. Am. J. Psychiatry 1996, 153, 151–162. [Google Scholar] [CrossRef]
- Ly, C.; Greb, A.C.; Cameron, L.P.; Wong, J.M.; Barragan, E.V.; Wilson, P.C.; Burbach, K.F.; Zarandi, S.S.; Sood, A.; Paddy, M.R.; et al. Psychedelics Promote Structural and Functional Neural Plasticity. Cell Rep. 2018, 23, 3170–3182. [Google Scholar] [CrossRef]
- Hesselgrave, N.; Troppoli, T.A.; Wulff, A.B.; Cole, A.B.; Thompson, S.M. Harnessing psilocybin: Antidepressant-like behavioral and synaptic actions of psilocybin are independent of 5-HT2R activation in mice. Proc. Natl. Acad. Sci. USA 2021, 118, e2022489118. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Liu, L.; Friston, K.J.; Shen, H.; Wang, L.; Zeng, L.L.; Hu, D. A treatment-resistant default mode subnetwork in major depression. Biol. Psychiatry 2013, 74, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Carhart-Harris, R.L.; Erritzoe, D.; Williams, T.; Stone, J.M.; Reed, L.J.; Colasanti, A.; Tyacke, R.J.; Leech, R.; Malizia, A.L.; Murphy, K.; et al. Neural correlates of the psychedelic state as determined by fMRI studies with psilocybin. Proc. Natl. Acad. Sci. USA 2012, 109, 2138–2143. [Google Scholar] [CrossRef] [PubMed]
- Carhart-Harris, R.L.; Roseman, L.; Bolstridge, M.; Demetriou, L.; Pannekoek, J.N.; Wall, M.B.; Tanner, M.; Kaelen, M.; McGonigle, J.; Murphy, K.; et al. Psilocybin for treatment-resistant depression: FMRI-measured brain mechanisms. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef]
- Daws, R.; Timmerman, C.; Giribaldi, B.; Sexton, J.; Wall, M.; Erritzoe, D.; Roseman, L.; Nutt, D.; Carhart-Harris, R. Decreased Brain Modularity after Psilocybin Therapy for Depression. Research Square 2021. Research Square: rs.3.rs-513323/v1. Available online: https://doi.org/10.21203/rs.3.rs-513323/v1 (accessed on 30 May 2021).
- Barrett, F.S.; Krimmel, S.R.; Griffiths, R.; Seminowicz, D.A.; Mathur, B.N. Psilocybin acutely alters the functional connectivity of the claustrum with brain networks that support perception, memory, and attention. Neuroimage 2020, 218, 116980. [Google Scholar] [CrossRef] [PubMed]
- Roseman, L.; Demetriou, L.; Wall, M.B.; Nutt, D.J.; Carhart-Harris, R.L. Increased amygdala responses to emotional faces after psilocybin for treatment-resistant depression. Neuropharmacology 2018, 142, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Li, B.; Wang, C.; Zhang, W.; Rao, Y.; Han, S. Allelic variation in 5-HTTLPR and the effects of citalopram on the emotional neural network. Br. J. Psychiatry 2015, 206, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Godlewska, B.R.; Norbury, R.; Selvaraj, S.; Cowen, P.J.; Harmer, C.J. Short-term SSRI treatment normalises amygdala hyperactivity in depressed patients. Psychol. Med. 2012, 42, 2609–2617. [Google Scholar] [CrossRef] [PubMed]
- Mertens, L.J.; Wall, M.B.; Roseman, L.; Demetriou, L.; Nutt, D.J.; Carhart-Harris, R.L. Therapeutic mechanisms of psilocybin: Changes in amygdala and prefrontal functional connectivity during emotional processing after psilocybin for treatment-resistant depression. J. Psychopharmacol. 2020, 34, 167–180. [Google Scholar] [CrossRef] [PubMed]
- McCulloch, D.E.-W.; Madsen, M.K.; Stenbæk, D.S.; Kristiansen, S.; Ozenne, B.; Jensen, P.S.; Knudsen, G.M.; Fisher, P.M. Lasting effects of a single psilocybin dose on resting-state functional connectivity in healthy individuals. J. Psychopharmacol. 2021. [Google Scholar] [CrossRef]
- Leary, T.; Litwin, G.H.; Metzner, R. Reactions to psilocybin administered in a supportive environment. J. Nerv. Ment. Dis. 1963, 137, 561–573. [Google Scholar] [CrossRef]
- Hartogsohn, I. Constructing drug effects: A history of set and setting. Drug Sci. Policy Law 2017, 3, 205032451668332. [Google Scholar] [CrossRef]
- Hartogsohn, I. Set and setting, psychedelics and the placebo response: An extra-pharmacological perspective on psychopharmacology. J. Psychopharmacol. 2016, 30, 1259–1267. [Google Scholar] [CrossRef]
- Roseman, L.; Nutt, D.J.; Carhart-Harris, R.L. Quality of acute psychedelic experience predicts therapeutic efficacy of psilocybin for treatment-resistant depression. Front. Pharmacol. 2018, 8, 974. [Google Scholar] [CrossRef]
- Studerus, E.; Gamma, A.; Kometer, M.; Vollenweider, F.X. Prediction of psilocybin response in healthy volunteers. PLoS ONE 2012, 7, e30800. [Google Scholar] [CrossRef]
- Haijen, E.C.H.M.; Kaelen, M.; Roseman, L.; Timmermann, C.; Kettner, H.; Russ, S.; Nutt, D.; Daws, R.E.; Hampshire, A.D.G.; Lorenz, R.; et al. Predicting Responses to Psychedelics: A Prospective Study. Front. Pharmacol. 2018, 9, 897. [Google Scholar] [CrossRef]
- Olson, D.E. The Subjective Effects of Psychedelics May Not Be Necessary for Their Enduring Therapeutic Effects. ACS Pharmacol. Transl. Sci. 2021, 4, 563–567. [Google Scholar] [CrossRef] [PubMed]
- Dong, C.; Ly, C.; Dunlap, L.E.; Vargas, M.V.; Sun, J.; Hwang, I.W.; Azinfar, A.; Oh, W.C.; Wetsel, W.C.; Olson, D.E.; et al. Psychedelic-inspired drug discovery using an engineered biosensor. Cell 2021, 184, 2779–2792.e18. [Google Scholar] [CrossRef] [PubMed]
- Cameron, L.P.; Tombari, R.J.; Lu, J.; Pell, A.J.; Hurley, Z.Q.; Ehinger, Y.; Vargas, M.V.; McCarroll, M.N.; Taylor, J.C.; Myers-Turnbull, D.; et al. A non-hallucinogenic psychedelic analogue with therapeutic potential. Nature 2021, 589, 474–479. [Google Scholar] [CrossRef] [PubMed]
- Yaden, D.B.; Griffiths, R.R. The Subjective Effects of Psychedelics Are Necessary for Their Enduring Therapeutic Effects. ACS Pharmacol. Transl. Sci. 2021, 4, 568–572. [Google Scholar] [CrossRef] [PubMed]
- Rucker, J.J.H.; Jelen, L.A.; Flynn, S.; Frowde, K.D.; Young, A.H. Psychedelics in the treatment of unipolar mood disorders: A systematic review. J. Psychopharmacol. 2016, 30, 1220–1229. [Google Scholar] [CrossRef] [PubMed]
- Andersen, K.A.A.; Carhart-Harris, R.; Nutt, D.J.; Erritzoe, D. Therapeutic effects of classic serotonergic psychedelics: A systematic review of modern-era clinical studies. Acta Psychiatr. Scand. 2021, 143, 101–118. [Google Scholar] [CrossRef] [PubMed]
- Upton, J. Beck Depression Inventory (BDI). In Encyclopedia of Behavioral Medicine; Gellman, M.D., Turner, J.R., Eds.; Springer: New York, NY, USA, 2013; pp. 178–179. ISBN 978-1-4419-1005-9. [Google Scholar]
- Agin-Liebes, G.I.; Malone, T.; Yalch, M.M.; Mennenga, S.E.; Ponté, K.L.; Guss, J.; Bossis, A.P.; Grigsby, J.; Fischer, S.; Ross, S. Long-term follow-up of psilocybin-assisted psychotherapy for psychiatric and existential distress in patients with life-threatening cancer. J. Psychopharmacol. 2020, 34, 155–166. [Google Scholar] [CrossRef]
- Griffiths, R.R.; Johnson, M.W.; Carducci, M.A.; Umbricht, A.; Richards, W.A.; Richards, B.D.; Cosimano, M.P.; Klinedinst, M.A. Psilocybin produces substantial and sustained decreases in depression and anxiety in patients with life-threatening cancer: A randomized double-blind trial. J. Psychopharmacol. 2016, 30, 1181–1197. [Google Scholar] [CrossRef]
- Carhart-Harris, R.L.; Bolstridge, M.; Rucker, J.; Day, C.M.J.; Erritzoe, D.; Kaelen, M.; Bloomfield, M.; Rickard, J.A.; Forbes, B.; Feilding, A.; et al. Psilocybin with psychological support for treatment-resistant depression: An open-label feasibility study. Lancet Psychiatry 2016, 3, 619–627. [Google Scholar] [CrossRef]
- Carhart-Harris, R.L.; Bolstridge, M.; Day, C.M.J.; Rucker, J.; Watts, R.; Erritzoe, D.E.; Kaelen, M.; Giribaldi, B.; Bloomfield, M.; Pilling, S.; et al. Psilocybin with psychological support for treatment-resistant depression: Six-month follow-up. Psychopharmacolog 2018, 235, 399–408. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.K.; Barrett, F.S.; May, D.G.; Cosimano, M.P.; Sepeda, N.D.; Johnson, M.W.; Finan, P.H.; Griffiths, R.R. Effects of Psilocybin-Assisted Therapy on Major Depressive Disorder: A Randomized Clinical Trial. JAMA Psychiatry 2021, 78, 481–489. [Google Scholar] [CrossRef] [PubMed]
- Carhart-Harris, R.; Giribaldi, B.; Watts, R.; Baker-Jones, M.; Murphy-Beiner, A.; Murphy, R.; Martell, J.; Blemings, A.; Erritzoe, D.; Nutt, D.J. Trial of Psilocybin versus Escitalopram for Depression. N. Engl. J. Med. 2021, 384, 1402–1411. [Google Scholar] [CrossRef] [PubMed]
- Aday, J.S.; Mitzkovitz, C.M.; Bloesch, E.K.; Davoli, C.C.; Davis, A.K. Long-term effects of psychedelic drugs: A systematic review. Neurosci. Biobehav. Rev. 2020, 113, 179–189. [Google Scholar] [CrossRef]
- Muttoni, S.; Ardissino, M.; John, C. Classical psychedelics for the treatment of depression and anxiety: A systematic review. J. Affect. Disord. 2019, 258, 11–24. [Google Scholar] [CrossRef]
- Galvão-Coelho, N.L.; Marx, W.; Gonzalez, M.; Sinclair, J.; de Manincor, M.; Perkins, D.; Sarris, J. Classic serotonergic psychedelics for mood and depressive symptoms: A meta-analysis of mood disorder patients and healthy participants. Psychopharmacology 2021, 238, 341–354. [Google Scholar] [CrossRef]
- Van Amsterdam, J.; Opperhuizen, A.; van den Brink, W. Harm potential of magic mushroom use: A review. Regul. Toxicol. Pharmacol. 2011, 59, 423–429. [Google Scholar] [CrossRef]
- Johnson, M.W.; Griffiths, R.R.; Hendricks, P.S.; Henningfield, J.E. The abuse potential of medical psilocybin according to the 8 factors of the Controlled Substances Act. Neuropharmacology 2018, 142, 143–166. [Google Scholar] [CrossRef]
- Espiard, M.L.; Lecardeur, L.; Abadie, P.; Halbecq, I.; Dollfus, S. Hallucinogen persisting perception disorder after psilocybin consumption: A case study. Eur. Psychiatry 2005, 20, 458–460. [Google Scholar] [CrossRef] [PubMed]
- Johansen, P.O.; Krebs, T.S. Psychedelics not linked to mental health problems or suicidal behavior: A population study. J. Psychopharmacol. 2015, 29, 270–279. [Google Scholar] [CrossRef] [PubMed]
- Hendricks, P.S.; Thorne, C.B.; Clark, C.B.; Coombs, D.W.; Johnson, M.W. Classic psychedelic use is associated with reduced psychological distress and suicidality in the United States adult population. J. Psychopharmacol. 2015, 29, 280–288. [Google Scholar] [CrossRef]
- Blacha, C.; Schmid, M.M.; Gahr, M.; Freudenmann, R.W.; Plener, P.L.; Finter, F.; Connemann, B.J.; Schönfeldt-Lecuona, C. Self-inflicted testicular amputation in first lysergic acid diethylamide use. J. Addict. Med. 2013, 7, 83–84. [Google Scholar] [CrossRef] [PubMed]
- Gahr, M.; Plener, P.L.; Kölle, M.A.; Freudenmann, R.W.; Schönfeldt-Lecuona, C. Self-mutilation induced by psychotropic substances: A systematic review. Psychiatry Res 2012, 200, 977–983. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, R.G.; Bouso, J.C.; Hallak, J.E.C. Ayahuasca, dimethyltryptamine, and psychosis: A systematic review of human studies. Ther. Adv. Psychopharmacol. 2017, 7, 141–157. [Google Scholar] [CrossRef] [PubMed]
- Carbonaro, T.M.; Bradstreet, M.P.; Barrett, F.S.; MacLean, K.A.; Jesse, R.; Johnson, M.W.; Griffiths, R.R. Survey study of challenging experiences after ingesting psilocybin mushrooms: Acute and enduring positive and negative consequences. J. Psychopharmacol. 2016, 30, 1268–1278. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.; Richards, W.; Griffths, R. Human Hallucinogen Research. J. Psychopharmacol. 2008, 22, 603–620. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.B.; Popp, D.; Kobak, K.A.; Detke, M.J. P-640—The power of expectation bias. Eur. Psychiatry 2012, 27, 1. [Google Scholar] [CrossRef]
- Cameron, L.P.; Benson, C.J.; DeFelice, B.C.; Fiehn, O.; Olson, D.E. Chronic, Intermittent Microdoses of the Psychedelic N,N-Dimethyltryptamine (DMT) Produce Positive Effects on Mood and Anxiety in Rodents. ACS Chem. Neurosci. 2019, 10, 3261–3270. [Google Scholar] [CrossRef] [PubMed]
- Martin, D.J.; Garske, J.P.; Katherine Davis, M. Relation of the therapeutic alliance with outcome and other variables: A meta-analytic review. J. Consult. Clin. Psychol. 2000, 68, 438–450. [Google Scholar] [CrossRef]
- Sellers, E.M.; Romach, M.K.; Leiderman, D.B. Studies with psychedelic drugs in human volunteers. Neuropharmacology 2018, 142, 116–134. [Google Scholar] [CrossRef]
- Usona Institute A Study of Psilocybin for Major Depressive Disorder (MDD). Available online: https://clinicaltrials.gov/ct2/show/NCT03866174 (accessed on 3 July 2021).
- COMPASS Trial. Available online: https://www.clinicaltrialsregister.eu/ctr-search/trial/2017-003288-36/NL (accessed on 3 July 2021).
- EPIsoDE Trial. Available online: https://www.clinicaltrialsregister.eu/ctr-search/trial/2019-003984-24/DE (accessed on 3 July 2021).

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zeiss, R.; Gahr, M.; Graf, H. Rediscovering Psilocybin as an Antidepressive Treatment Strategy. Pharmaceuticals 2021, 14, 985. https://doi.org/10.3390/ph14100985
Zeiss R, Gahr M, Graf H. Rediscovering Psilocybin as an Antidepressive Treatment Strategy. Pharmaceuticals. 2021; 14(10):985. https://doi.org/10.3390/ph14100985
Chicago/Turabian StyleZeiss, Rene, Maximilian Gahr, and Heiko Graf. 2021. "Rediscovering Psilocybin as an Antidepressive Treatment Strategy" Pharmaceuticals 14, no. 10: 985. https://doi.org/10.3390/ph14100985
APA StyleZeiss, R., Gahr, M., & Graf, H. (2021). Rediscovering Psilocybin as an Antidepressive Treatment Strategy. Pharmaceuticals, 14(10), 985. https://doi.org/10.3390/ph14100985




