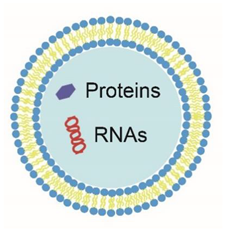
Extracellular Vesicles in Human Milk
Abstract
:1. Introduction
2. Physical Properties and Cellular Origin of Extracellular Vesicles in Milk
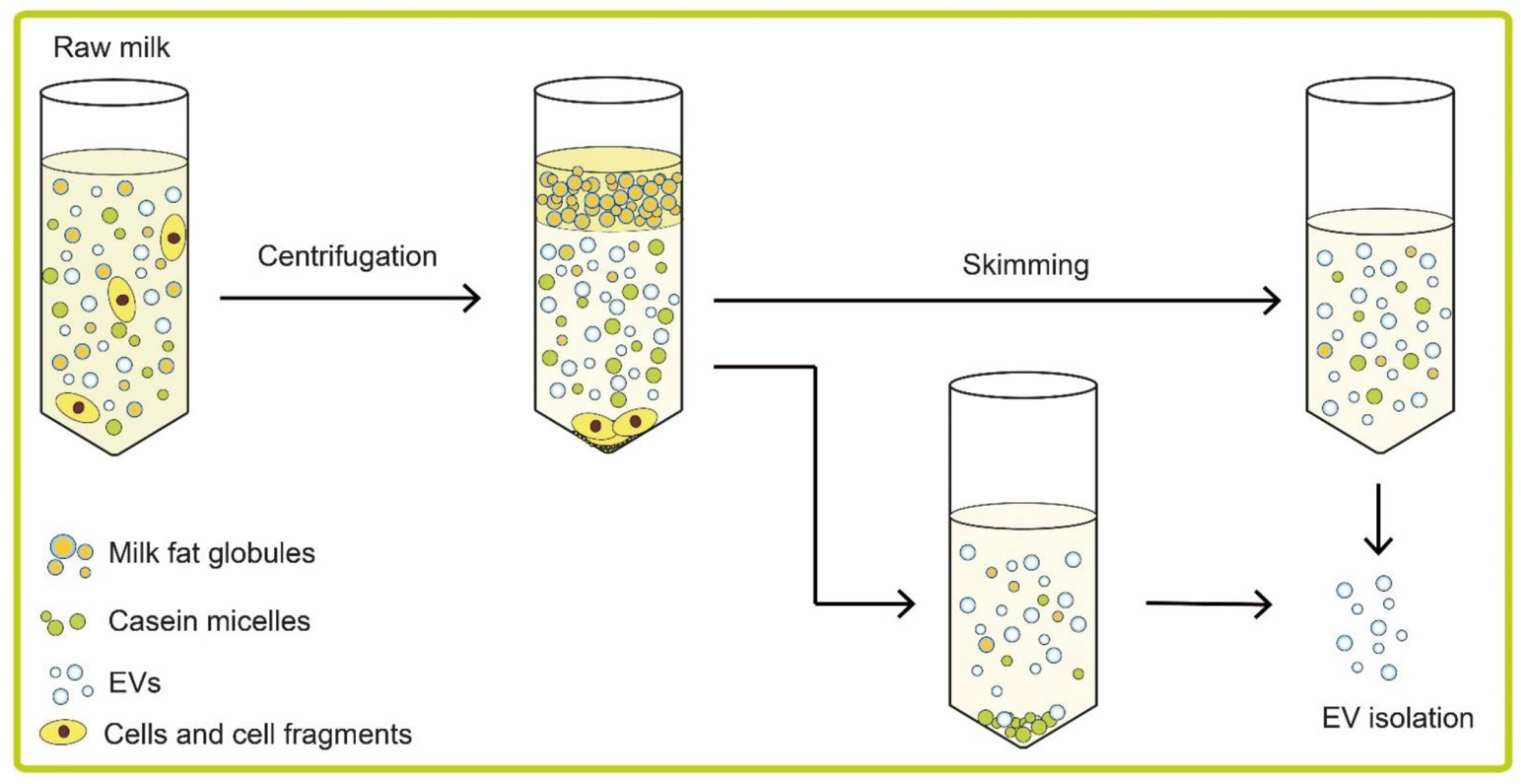
3. Isolation of Extracellular Vesicles from Milk
3.1. Removal of Cells and Cell Fragments
3.2. Removal of Milk Fat Globules
3.3. Removal of Casein Micelles
4. The Biochemical Composition of Extracellular Vesicles: Proteins and RNA
4.1. Proteins
| Isolation Procedure | Proteins | Detection | Study |
|---|---|---|---|
| Ultracentrifugation | CXCL5, SOD1, PRSS8, EPHA2, MIA, FR-alpha, MET, CD69, PDGF subunit B, CCL19 | PEA | [56] |
| Ultracentrifugation | MHC class II, CD81, MUC1, HSPs, MFG-E8 | FCM, WB, MS | [21] |
| Ultracentrifugation, size exclusive chromatography | TF, CD9, CD63 | WB | [33] |
| Density gradient centrifugation | Colostrum: LCP1, IGHG1, IGKV3-20, LAMP1, PRDX1 Mature milk: SLC1A5, RPL10, Tetraspanin, GMPPA | LC-MS/MS | [57] |
| Density gradient centrifugation | CD9, CD63, MHC-class II, FLOT-1 | LC-MS/MS, WB | [58] |
| Density gradient centrifugation | CD36, CD63, MUC1 | FCM | [59] |
| Milk source | Isolation Procedure | Protein | Detection | Study |
|---|---|---|---|---|
| Bovine | Ultracentrifugation | CD63, HSP70, CD9, CD81 | WB | [60] |
| Bovine | Ultracentrifugation | CD9, CD81, CD63, CD82, CD47, MHC class I, syndecan, NT5E, CD59 | LC-MS/MS | [48] |
| Bovine | Ultracentrifugation | FASN, XDH, BTN1A1, HSPA8, PLIN2, MFG-E8, IDH1, GDI2 | LC-MS/MS | [61] |
| Bovine | Ultracentrifugation | CD63, CD81, TSG101, CD9 | WB | [62] |
| Bovine | Ultracentrifugation | LGB, PAEP, CSN1S1 | LC-MS/MS | [63] |
| Bovine (yak) | Ultracentrifugation | CD63, HSP70, TSG101 | WB | [64] |
| Bovine | Density gradient centrifugation | CD63, HSP70, MFG-E8, BTN1A1 | WB | [65] |
| Bovine | Density gradient centrifugation | Colostrum: B2M, Clusterin, PDGFC, CCN1 Mature milk: LTF, ANG1, LPO, QSOX1 | LC-MS/MS | [57] |
| Horse | Ultracentrifugation | CD81, CD63, LGB, MFG-E8 | MALDI-MS, MS/MS | [66] |
4.2. RNA
| Isolation Procedure | RNA | Detection | Study |
|---|---|---|---|
| Ultracentrifugation | hsa-miR-30d-5p, hsa-let-7b-5p, hsa-let-7a-5p, hsa-miR-125a-5p, hsa-miR-21–5p, hsa-miR-423–5p, hsa-let-7g-5p, hsa-let-7f-5p, hsa-miR-30a-5p, hsa-miR-146b-5p | RT-qPCR | [18] |
| Ultracentrifugation | Milk from mother who delivered pre-term infant: hsa-miR-22-3p, hsa-miR-148a-3p, hsa-miR-141-3p, hsa-miR-181a-5p, hsa-miR-320a, hsa-miR-378a-3p, hsa-miR-30d-5p, hsa-miR-30a-5p, hsa-miR-26a-5p, hsa-miR-191-5p; Milk from mother who delivered term infant: hsa-miR-22a-3p, hsa-miR-181a-5p, hsa-miR-148a-3p, hsa-miR-141a-3p, hsa-miR-30a-5p, hsa-miR-99b-5p, hsa-miR-191-5p, hsa-miR-378a-3p, hsa-miR-146b-5p, hsa-miR-30d-5p | UHTS | [68] |
| Ultracentrifugation | hsa-let-7c, hsa-miR-21, hsa-miR-34a, hsa-miR-146b, and hsa-miR-200b. | HTS | [69] |
| Density gradient centrifugation | hsa-miR-30d-5p, hsa-miR-148a-3p, hsa-miR-200a-3p, hsa-miR-200c-3p, hsa-let-7a-5p, hsa-miR-21-5p, hsa-let-7b-5p, hsa-let-7f-5p, hsa-miR-30a-5p, hsa-let-7g-5p | UHTS, RT-qPCR | [70] |
| ExoQuick-TC precipitation solution | hsa-miR-148a-3p, hsa-miR-22-3p, hsa-miR-30d-5p, hsa-let-7b-5p, hsa-miR-200a-3p | HTS | [71] |
| ExoQuick precipitation solution | hsa-miR-148a-3p, hsa-miR-30b-5p, hsa-let-7f-1-5p & -2-5p, hsa-miR-146b-5p, hsa-miR-29a-3p, hsa-let-7a-2-5p & -3-5p, hsa-miR-141-3p, hsa-miR-182-5p, hsa-miR-200a-3p, hsa-miR-378-3p | qPCR | [72] |
| ExoQuick-TC precipitation solution | hsa-miR-22-3p, hsa-miR-30d-5p, hsa-miR-148a-3p, hsa-miR-181a-5p, hsa-miR-141-3p, hsa-miR-30b-5p, hsa-miR-26a-5p, hsa-miR-92a-3p, hsa-miR-375, hsa-miR-30a-5p | UHTS | [73] |
| ExoQuick-TC precipitation solution | hsa-miR-148a-3p, hsa-miR-6073, hsa-miR-200c, hsa-miR-200b, hsa-miR-99a-5p, hsa-miR-30a-5p, hsa-miR-30d, hsa-miR-320-3p, hsa-let-7a-5p, hsa-miR-26a-5p | WGS | [74] |
| Total exosomes isolation reagent | miRNA from HIV-infected breast milk vs. uninfected; upregulated: hsa-miR-320e; hsa-miR-630; hsa-miR-148a-3p; hsa-miR-23a-3p; hsa-miR-378g; hsa-miR-30a-5p; hsa-miR-93-5p; hsa-miR-497-5p; hsa-miR-200b-3p; hsa-miR-200a-3p | NGS | [75] |
| ExoEasy maxi kit | hsa-miR-99b-3p, hsa-miR -96-5p, hsa-miR-550a-5p, hsa-miR-616-5p, hsa-miR-155-5p, hsa-miR-604 | qPCR | [76] |
| Milk Source | Isolation Procedure | RNA | Detection | Study |
|---|---|---|---|---|
| Bovine | Ultracentrifugation | bta-miR-223, bta-miR125b | RT-qPCR | [61] |
| Bovine | Ultracentrifugation | Colostrum: bta-miR-24, bta-miR-30d, bta-miR-93, bta-miR-106a, bta-miR-181a, bta-miR-200a, bta-miR451 | RT-qPCR | [77] |
| Bovine | Ultracentrifugation | bta-miR-10b, bta-miR-143, bta-miR-10a, bta-miR-26a, bta-let-7a, bta-miR-21, bta-let-7f, bta-miR-222, bta-miR-99b, bta-let-7i | RT-qPCR | [78] |
| Bovine | Ultracentrifugation | bta-let-7a-5p, bta-let-7b, bta-let-7c, bta-let-7e and bta-let-7f, bta-miR-30a-5p, bta-miR-30d and bta-miR-30e-5p, bta-miR-148a, bta-miR-26a | HTS | [79] |
| Bovine | Ultracentrifugation, Density gradient centrifugation | bta-miR-223, bta-miR125b | RT-qPCR | [25] |
| Bovine | Total exosome isolation kit | bta-miR-26a, bta-miR-191, bta-miR-423-5p, bta-let-7f, bta-miR-30d, bta-let-7a-5p, bta-miR-27b, bta-let-7b, bta-miR-92a, bta-miR-125a | WGS | [80] |
| Bovine | ExoQuick reagent | - bta-miR-21, bta-miR-30a, bta-miR-92a, bta-miR-99a, bta-miR-223 | qPCR | [81] |
| Porcine | Ultracentrifugation | ssc-miR-193a-3p, ssc-miR-423-5p, ssc-miR-320, ssc-miR-181a, ssc-miR-30a3p, ssc-miR-378, ssc-miR-191, ssc-let-7a, ssc-let-7f, ssc-let-7c. | HTS | [82] |
| Porcine | Density gradient centrifugation | ssc-let-7a-5p, ssc-miR-30a-5p, ssc-miR-191-5p, ssc-miR-21-5p, ssc-miR-30d-5p, ssc-let-7f-5p, ssc-let-7c, ssc-miR-200c-3p, ssc-let-7g-5p, ssc-miR-320a-3p | HTS | [70] |
| Porcine | ExoQuick exosome precipitation solution | ssc-miR-148a-3p, ssc-miR-182-5p, ssc-miR-200c-3p, ssc-miR-25-3p, ssc-miR-30a-5p, ssc-miR-30d-5p, ssc-miR-574-3p | HTS | [83] |
| Caprinae (sheep) | Total exosome isolation kit | oar-miR-26a, oar-miR-191, oar-let-7f, oar-let-7b, oar-miR-10b, oar-miR-148a, oar-let-7b, oar-let-7a, oar-miR-21, oar-let-7c | HTS, PCR | [84] |
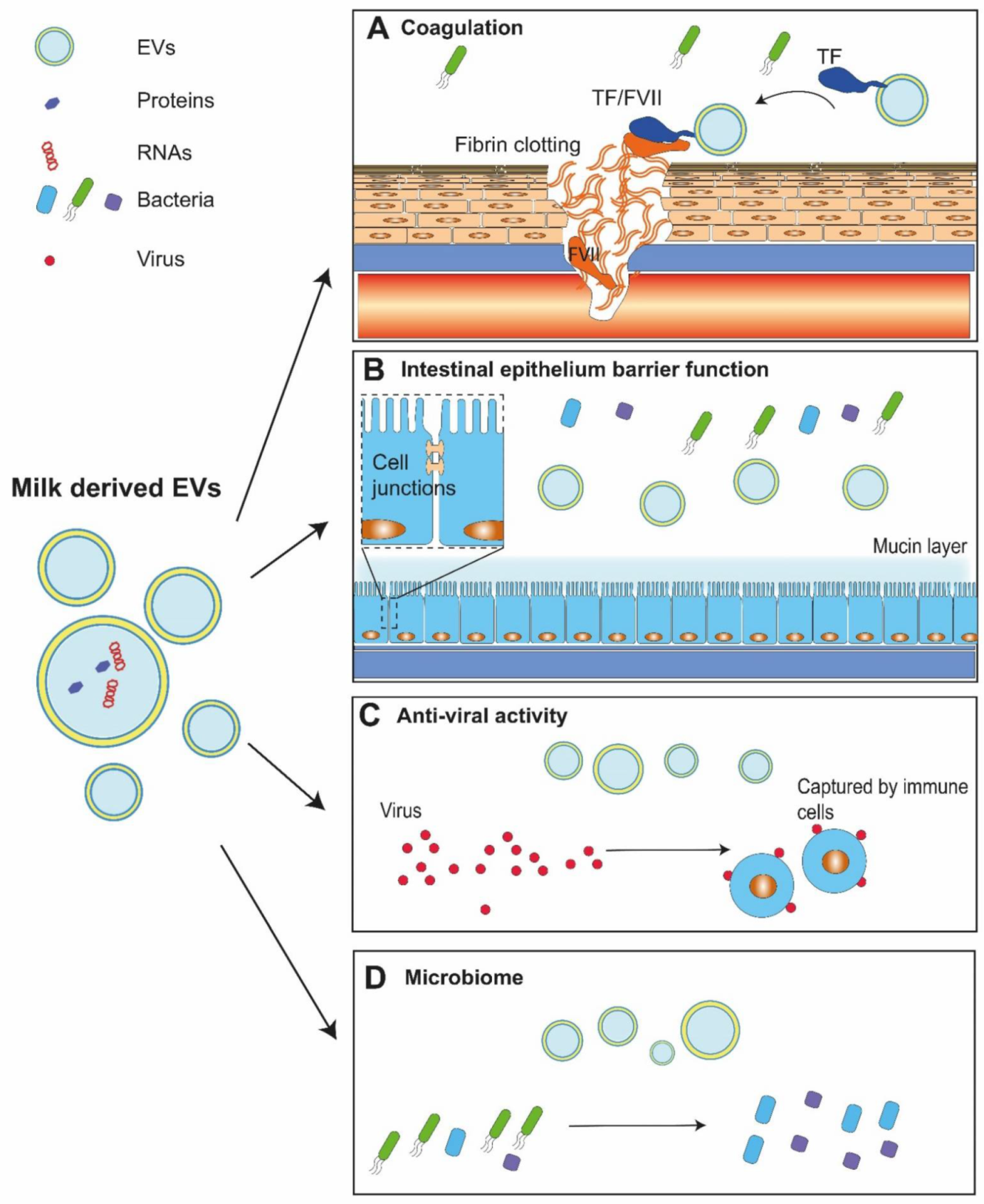
5. Functions of Extracellular Vesicles in Milk
5.1. Coagulation
5.2. Intestinal Epithelium Barrier Function
5.3. Anti-Viral Activity
5.4. Microbiome
6. Therapeutic Application of Milk-Derived Extracellular Vesicles
6.1. Osteoporosis
6.2. Arthritis
6.3. Cancer
6.4. Drug Delivery
7. Survival of Extracellular Vesicles from Mothers’ Milk in the Infants’ Digestive Tract
8. Possible Uptake of Milk-Derived Extracellular Vesicles
9. Summary
10. Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| hsa | Homo sapiens |
| bta | Bos taurus |
| ssc | Sus scrofa |
| oar | Ovis aries |
| ANG1 | Angiogenin-1 |
| B2M | Beta-2-microglobulin |
| BTN1A1 | Butyrophilin, subfamily 1, member A1 |
| CCL19 | C-C motif chemokine ligand 19 |
| CCN1 | Cellular communication network factor 1 |
| CD | Cluster of differentiation |
| CSN1S1 | Casein alpha S1 |
| CXCL5 | C-X-C motif chemokine 5 |
| EVs | Extracellular vesicles |
| EPHA2 | EPH receptor A2 |
| FASN | Fatty acid synthase |
| FCM | Flow cytometry |
| FLOT-1 | Flotillin-1 |
| FR-alpha | Folate receptor 1 |
| GDI2 | Rab GDP dissociation inhibitor beta |
| GMPPA | Mannose-1-phosphate guanyltransferase alpha |
| HSP | Heat shock protein |
| HTS | High throughput sequencing |
| IDH1 | Isocitrate dehydrogenase |
| IGHG1 | Ig gamma-1 chain C region |
| IGKV3-20 | Ig kappa chain V-III region |
| LAMP1 | Lysosome-associated membrane glycoprotein 1 |
| LC-MS/MS | Liquid chromatography-tandem mass spectrometry analysis |
| LCP1 | Lymphocyte cytosolic protein 1 |
| LGB | Beta-lactoglobulin |
| LPO | Lactoperoxidase |
| LTF | Lactoferrin |
| MALDI | Matrix-assisted laser desorption/ionization |
| MET | Hepatocyte growth factor receptor |
| MFG-E8 | Milk fat globule-EGF factor 8 protein (lactadherin) |
| MHC | Major histocompatibility complex |
| MIA | Melanoma inhibitory activity |
| MS | Mass spectrometry |
| MS/MS | Tandem mass spectrometry |
| MUC1 | Mucin 1 |
| NGS | Next-generation sequencing |
| NT5E | 5′-nucleotidase |
| PAEP | Progestogen-associated endometrial protein |
| PEA | Proximity extension assay |
| PDGF | Plate-derived growth factor |
| PLIN2 | Perilipin 2 |
| PRDX1 | Peroxiredoxin-1 |
| PRSS8 | Serine protease 8 |
| qPCR | Quantitative polymerase chain reaction |
| QSOX1 | Quiescin sulfhydryl oxidase 1 |
| RPL10 | Ribosomal protein L10 |
| RT-qPCR | Quantitative reverse transcription polymerase chain reaction |
| SLC1A5 | Solute carrier family 1 member 5 |
| SOD1 | Superoxide dismutase 1 |
| TF | Tissue factor |
| TSG101 | Tumor susceptibility gene 101 |
| UHTS | Ultra-high throughput sequencing |
| WB | Western blot |
| WGS | Whole-genome sequencing |
| XDH | Xanthine dehydrogenase/oxidase |
References
- Corpeleijn, W.E.; Kouwenhoven, S.M.; Paap, M.C.; van Vliet, I.; Scheerder, I.; Muizer, Y.; Helder, O.K.; van Goudoever, J.B.; Vermeulen, M.J. Intake of own mother’s milk during the first days of life is associated with decreased morbidity and mortality in very low birth weight infants during the first 60 days of life. Neonatology 2012, 102, 276–281. [Google Scholar] [CrossRef] [Green Version]
- Sosa-Castillo, E.; Rodriguez-Cruz, M.; Molto-Puigmarti, C. Genomics of lactation: Role of nutrigenomics and nutrigenetics in the fatty acid composition of human milk. Br. J. Nutr. 2017, 118, 161–168. [Google Scholar] [CrossRef] [Green Version]
- Pereira, P.C. Milk nutritional composition and its role in human health. Nutrition 2014, 30, 619–627. [Google Scholar] [CrossRef]
- De la Torre Gomez, C.; Goreham, R.V.; Bech Serra, J.J.; Nann, T.; Kussmann, M. “Exosomics”-A Review of Biophysics, Biology and Biochemistry of Exosomes with a Focus on Human Breast Milk. Front. Genet. 2018, 9, 92. [Google Scholar] [CrossRef] [Green Version]
- Park, Y.W.; Juárez, M.; Ramos, M.; Haenlein, G.F.W. Physico-chemical characteristics of goat and sheep milk. Small Rumin. Res. 2007, 68, 88–113. [Google Scholar] [CrossRef] [Green Version]
- Sullivan, S.; Schanler, R.J.; Kim, J.H.; Patel, A.L.; Trawoger, R.; Kiechl-Kohlendorfer, U.; Chan, G.M.; Blanco, C.L.; Abrams, S.; Cotten, C.M.; et al. An exclusively human milk-based diet is associated with a lower rate of necrotizing enterocolitis than a diet of human milk and bovine milk-based products. J. Pediatr. 2010, 156, 562–567.e561. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sole, A. Die Muttermilch als Blutstillungsmittel. Wien. Klin. Wochenschr. 1935, 38, 1354–1359. [Google Scholar] [CrossRef]
- Glanzmann, E. Einführung in die Kinderheilkunde; Springer: Vienna, Austria, 1934. [Google Scholar]
- Wolf, P. The nature and significance of platelet products in human plasma. Br. J. Haematol. 1967, 13, 269–288. [Google Scholar] [CrossRef]
- Hell, L.; Wisgrill, L.; Ay, C.; Spittler, A.; Schwameis, M.; Jilma, B.; Pabinger, I.; Altevogt, P.; Thaler, J. Procoagulant extracellular vesicles in amniotic fluid. Transl. Res. 2017, 184, 12–20.e11. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Gool, E.; Berckmans, R.J.; Coumans, F.A.W.; Barendrecht, A.D.; Maas, C.; van der Wel, N.N.; Altevogt, P.; Sturk, A.; Nieuwland, R. Extracellular vesicles from human saliva promote hemostasis by delivering coagulant tissue factor to activated platelets. J. Thromb. Haemost. 2018, 16, 1153–1163. [Google Scholar] [CrossRef] [Green Version]
- Rikkert, L.G.; de Rond, L.; van Dam, A.; van Leeuwen, T.G.; Coumans, F.A.W.; de Reijke, T.M.; Terstappen, L.; Nieuwland, R. Detection of extracellular vesicles in plasma and urine of prostate cancer patients by flow cytometry and surface plasmon resonance imaging. PLoS ONE 2020, 15, e0233443. [Google Scholar] [CrossRef]
- Brooker, B.E. The epithelial cells and cell fragments in human milk. Cell Tissue Res. 1980, 210, 321–332. [Google Scholar] [CrossRef]
- Baddela, V.S.; Nayan, V.; Rani, P.; Onteru, S.K.; Singh, D. Physicochemical Biomolecular Insights into Buffalo Milk-Derived Nanovesicles. Appl. Biochem. Biotechnol. 2016, 178, 544–557. [Google Scholar] [CrossRef]
- Mecocci, S.; Gevi, F.; Pietrucci, D.; Cavinato, L.; Luly, F.R.; Pascucci, L.; Petrini, S.; Ascenzioni, F.; Zolla, L.; Chillemi, G.; et al. Anti-Inflammatory Potential of Cow, Donkey and Goat Milk Extracellular Vesicles as Revealed by Metabolomic Profile. Nutrients 2020, 12, 2908. [Google Scholar] [CrossRef]
- Ibrahim, H.M.; Mohammed-Geba, K.; Tawfic, A.A.; El-Magd, M.A. Camel milk exosomes modulate cyclophosphamide-induced oxidative stress and immuno-toxicity in rats. Food Funct. 2019, 10, 7523–7532. [Google Scholar] [CrossRef] [PubMed]
- Xie, M.Y.; Hou, L.J.; Sun, J.J.; Zeng, B.; Xi, Q.Y.; Luo, J.Y.; Chen, T.; Zhang, Y.L. Porcine Milk Exosome MiRNAs Attenuate LPS-Induced Apoptosis through Inhibiting TLR4/NF-κB and p53 Pathways in Intestinal Epithelial Cells. J. Agric. Food Chem. 2019, 67, 9477–9491. [Google Scholar] [CrossRef] [PubMed]
- Leiferman, A.; Shu, J.; Upadhyaya, B.; Cui, J.; Zempleni, J. Storage of Extracellular Vesicles in Human Milk, and MicroRNA Profiles in Human Milk Exosomes and Infant Formulas. J. Pediatr. Gastroenterol. Nutr. 2019, 69, 235–238. [Google Scholar] [CrossRef] [PubMed]
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): A position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 2018, 7, 1535750. [Google Scholar] [CrossRef] [Green Version]
- Royo, F.; Théry, C.; Falcón-Pérez, J.M.; Nieuwland, R.; Witwer, K.W. Methods for Separation and Characterization of Extracellular Vesicles: Results of a Worldwide Survey Performed by the ISEV Rigor and Standardization Subcommittee. Cells 2020, 9, 1955. [Google Scholar] [CrossRef]
- Admyre, C.; Johansson, S.M.; Qazi, K.R.; Filen, J.J.; Lahesmaa, R.; Norman, M.; Neve, E.P.; Scheynius, A.; Gabrielsson, S. Exosomes with immune modulatory features are present in human breast milk. J. Immunol. 2007, 179, 1969–1978. [Google Scholar] [CrossRef]
- Hock, A.; Miyake, H.; Li, B.; Lee, C.; Ermini, L.; Koike, Y.; Chen, Y.; Maattanen, P.; Zani, A.; Pierro, A. Breast milk-derived exosomes promote intestinal epithelial cell growth. J. Pediatr. Surg. 2017, 52, 755–759. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yan, X.; Zhang, L.; Cai, J.; Zhou, Y.; Liu, H.; Hu, Y.; Chen, W.; Xu, S.; Liu, P.; et al. Identification and Peptidomic Profiling of Exosomes in Preterm Human Milk: Insights into Necrotizing Enterocolitis Prevention. Mol. Nutr. Food Res. 2019, 63, e1801247. [Google Scholar] [CrossRef]
- Yu, S.; Zhao, Z.; Xu, X.; Li, M.; Li, P. Characterization of three different types of extracellular vesicles and their impact on bacterial growth. Food Chem. 2019, 272, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Benmoussa, A.; Lee, C.H.; Laffont, B.; Savard, P.; Laugier, J.; Boilard, E.; Gilbert, C.; Fliss, I.; Provost, P. Commercial Dairy Cow Milk microRNAs Resist Digestion under Simulated Gastrointestinal Tract Conditions. J. Nutr. 2016, 146, 2206–2215. [Google Scholar] [CrossRef]
- Sims, B.; Farrow, A.L.; Williams, S.D.; Bansal, A.; Krendelchtchikov, A.; Gu, L.; Matthews, Q.L. Role of TIM-4 in exosome-dependent entry of HIV-1 into human immune cells. Int. J. Nanomed. 2017, 12, 4823–4833. [Google Scholar] [CrossRef] [Green Version]
- Blans, K.; Hansen, M.S.; Sorensen, L.V.; Hvam, M.L.; Howard, K.A.; Moller, A.; Wiking, L.; Larsen, L.B.; Rasmussen, J.T. Pellet-free isolation of human and bovine milk extracellular vesicles by size-exclusion chromatography. J. Extracell. Vesicles 2017, 6, 1294340. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, M.; Shimizu, K.; Rahman, M.; Ishikawa, H.; Takase, H.; Ugawa, S.; Okada, A.; Inoshima, Y. Efficient method for isolation of exosomes from raw bovine milk. Drug Dev. Ind. Pharm. 2019, 45, 359–364. [Google Scholar] [CrossRef]
- Kunz, C.; Lönnerdal, B. Casein micelles and casein subunits in human milk. In Protein and Non-Protein Nitrogen in Human Milk; CRC Press: Boca Raton, FL, USA, 1989; pp. 9–27. [Google Scholar]
- Veenstra, C.; Lenferink, A.; Petersen, W.; Steenbergen, W.; Bosschaart, N. Optical properties of human milk. Biomed. Opt. Express 2019, 10, 4059–4074. [Google Scholar] [CrossRef]
- Kamigaki, T.; Ito, Y.; Nishino, Y.; Miyazawa, A. Microstructural observation of casein micelles in milk by cryo-electron microscopy of vitreous sections (CEMOVIS). Microscopy 2018, 67, 164–170. [Google Scholar] [CrossRef]
- Michalski, M.C.; Briard, V.; Michel, F.; Tasson, F.; Poulain, P. Size distribution of fat globules in human colostrum, breast milk, and infant formula. J. Dairy Sci. 2005, 88, 1927–1940. [Google Scholar] [CrossRef] [Green Version]
- Hu, Y.; Hell, L.; Kendlbacher, R.A.; Hajji, N.; Hau, C.; van Dam, A.; Berckmans, R.J.; Wisgrill, L.; Ay, C.; Pabinger, I.; et al. Human milk triggers coagulation via tissue factor-exposing extracellular vesicles. Blood Adv. 2020, 4, 6274–6282. [Google Scholar] [CrossRef]
- Zhang, M.; Ma, Z.; Li, R.; Guo, S.; Qiu, Y.; Gao, X. Proteomic Analysis Reveals Proteins and Pathways Associated with Lactation in Bovine Mammary Epithelial Cell-Derived Exosomes. J. Proteome Res. 2020, 19, 3211–3219. [Google Scholar] [CrossRef]
- Li, S.; Zhang, L.; Zhou, Q.; Jiang, S.; Yang, Y.; Cao, Y. Characterization of Stem Cells and Immune Cells in Preterm and Term Mother’s Milk. J. Hum. Lact. 2019, 35, 528–534. [Google Scholar] [CrossRef]
- Chen, P.W.; Lin, Y.L.; Huang, M.S. Profiles of commensal and opportunistic bacteria in human milk from healthy donors in Taiwan. J. Food Drug Anal. 2018, 26, 1235–1244. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jost, T.; Lacroix, C.; Braegger, C.; Chassard, C. Impact of human milk bacteria and oligosaccharides on neonatal gut microbiota establishment and gut health. Nutr. Rev. 2015, 73, 426–437. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Yi, D.Y. Analysis of the human breast milk microbiome and bacterial extracellular vesicles in healthy mothers. Exp. Mol. Med. 2020, 52, 1288–1297. [Google Scholar] [CrossRef] [PubMed]
- Alex, A.; Bhandary, E.; McGuire, K.P. Anatomy and Physiology of the Breast during Pregnancy and Lactation. Adv. Exp. Med. Biol. 2020, 1252, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Honvo-Houéto, E.; Truchet, S. Indirect Immunofluorescence on Frozen Sections of Mouse Mammary Gland. J. Vis. Exp. 2015. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ballard, O.; Morrow, A.L. Human milk composition: Nutrients and bioactive factors. Pediatr. Clin. North. Am. 2013, 60, 49–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, R.; Zhang, R.; Qian, T.; Peng, X.; He, W.; Zheng, S.; Cao, Y.; Pierro, A.; Shen, C. A comparison of exosomes derived from different periods breast milk on protecting against intestinal organoid injury. Pediatr. Surg. Int. 2019, 35, 1363–1368. [Google Scholar] [CrossRef] [PubMed]
- Samuel, M.; Chisanga, D.; Liem, M.; Keerthikumar, S.; Anand, S.; Ang, C.S.; Adda, C.G.; Versteegen, E.; Jois, M.; Mathivanan, S. Bovine milk-derived exosomes from colostrum are enriched with proteins implicated in immune response and growth. Sci. Rep. 2017, 7, 5933. [Google Scholar] [CrossRef]
- Somiya, M.; Yoshioka, Y.; Ochiya, T. Biocompatibility of highly purified bovine milk-derived extracellular vesicles. J. Extracell. Vesicles 2018, 7, 1440132. [Google Scholar] [CrossRef] [Green Version]
- Hernell, O. Human milk vs. cow’s milk and the evolution of infant formulas. Nestle Nutr. Workshop Ser. Pediatr. Program. 2011, 67, 17–28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sood, S.M.; Herbert, P.J.; Slattery, C.W. Structural studies on casein micelles of human milk: Dissociation of beta-casein of different phosphorylation levels induced by cooling and ethylenediaminetetraacetate. J. Dairy Sci. 1997, 80, 628–633. [Google Scholar] [CrossRef]
- Stewart, P.S.; Puppione, D.L.; Patton, S. The presence of microvilli and other membrane fragments in the non-fat phase of bovine milk. Z Zellforsch. Mikrosk. Anat. 1972, 123, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Tong, L.; Hao, H.; Zhang, X.; Zhang, Z.; Lv, Y.; Zhang, L.; Yi, H. Oral Administration of Bovine Milk-Derived Extracellular Vesicles Alters the Gut Microbiota and Enhances Intestinal Immunity in Mice. Mol. Nutr. Food Res. 2020, 64, e1901251. [Google Scholar] [CrossRef]
- Rahman, M.M.; Shimizu, K.; Yamauchi, M.; Takase, H.; Ugawa, S.; Okada, A.; Inoshima, Y. Acidification effects on isolation of extracellular vesicles from bovine milk. PLoS ONE 2019, 14, e0222613. [Google Scholar] [CrossRef] [Green Version]
- Griffin, M.C.; Lyster, R.L.; Price, J.C. The disaggregation of calcium-depleted casein micelles. Eur. J. Biochem. 1988, 174, 339–343. [Google Scholar] [CrossRef]
- Udabage, P.; McKinnon, I.R.; Augustin, M.A. Mineral and casein equilibria in milk: Effects of added salts and calcium-chelating agents. J. Dairy Res. 2000, 67, 361–370. [Google Scholar] [CrossRef]
- Yáñez-Mó, M.; Siljander, P.R.; Andreu, Z.; Zavec, A.B.; Borràs, F.E.; Buzas, E.I.; Buzas, K.; Casal, E.; Cappello, F.; Carvalho, J.; et al. Biological properties of extracellular vesicles and their physiological functions. J. Extracell. Vesicles 2015, 4, 27066. [Google Scholar] [CrossRef] [Green Version]
- Gohar, M.; Gilois, N.; Graveline, R.; Garreau, C.; Sanchis, V.; Lereclus, D. A comparative study of Bacillus cereus, Bacillus thuringiensis and Bacillus anthracis extracellular proteomes. Proteomics 2005, 5, 3696–3711. [Google Scholar] [CrossRef] [PubMed]
- Prasad, R.; Sen, P. Structural modulation of factor VIIa by full-length tissue factor (TF(1-263)): Implication of novel interactions between EGF2 domain and TF. J. Biomol. Struct. Dyn. 2018, 36, 621–633. [Google Scholar] [CrossRef] [PubMed]
- Palviainen, M.; Saraswat, M.; Varga, Z.; Kitka, D.; Neuvonen, M.; Puhka, M.; Joenväärä, S.; Renkonen, R.; Nieuwland, R.; Takatalo, M.; et al. Extracellular vesicles from human plasma and serum are carriers of extravesicular cargo-Implications for biomarker discovery. PLoS ONE 2020, 15, e0236439. [Google Scholar] [CrossRef] [PubMed]
- Larssen, P.; Wik, L.; Czarnewski, P.; Eldh, M.; Lof, L.; Ronquist, K.G.; Dubois, L.; Freyhult, E.; Gallant, C.J.; Oelrich, J.; et al. Tracing Cellular Origin of Human Exosomes Using Multiplex Proximity Extension Assays. Mol. Cell Proteom. 2017, 16, 502–511. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, M.; Song, D.; Cao, X.; Wu, R.; Liu, B.; Ye, W.; Wu, J.; Yue, X. Comparative proteomic analysis of milk-derived exosomes in human and bovine colostrum and mature milk samples by iTRAQ-coupled LC-MS/MS. Food Res. Int. 2017, 92, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Van Herwijnen, M.J.; Zonneveld, M.I.; Goerdayal, S.; Nolte-‘t Hoen, E.N.; Garssen, J.; Stahl, B.; Maarten Altelaar, A.F.; Redegeld, F.A.; Wauben, M.H. Comprehensive Proteomic Analysis of Human Milk-derived Extracellular Vesicles Unveils a Novel Functional Proteome Distinct from Other Milk Components. Mol. Cell Proteom. 2016, 15, 3412–3423. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Torregrosa Paredes, P.; Gutzeit, C.; Johansson, S.; Admyre, C.; Stenius, F.; Alm, J.; Scheynius, A.; Gabrielsson, S. Differences in exosome populations in human breast milk in relation to allergic sensitization and lifestyle. Allergy 2014, 69, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Hock, A.; Wu, R.Y.; Minich, A.; Botts, S.R.; Lee, C.; Antounians, L.; Miyake, H.; Koike, Y.; Chen, Y.; et al. Bovine milk-derived exosomes enhance goblet cell activity and prevent the development of experimental necrotizing enterocolitis. PLoS ONE 2019, 14, e0211431. [Google Scholar] [CrossRef]
- Benmoussa, A.; Ly, S.; Shan, S.T.; Laugier, J.; Boilard, E.; Gilbert, C.; Provost, P. A subset of extracellular vesicles carries the bulk of microRNAs in commercial dairy cow’s milk. J. Extracell. Vesicles 2017, 6, 1401897. [Google Scholar] [CrossRef] [Green Version]
- Ma, S.; Niu, M.; Hao, Z.; Liu, M.; Tong, C.; Zhao, X. Selective packaged circular RNAs in milk extracellular vesicles during Staphylococcus aureus infection may have potential against bacterial infection. RNA Biol. 2021, 18, 818–831. [Google Scholar] [CrossRef]
- Benmoussa, A.; Gotti, C.; Bourassa, S.; Gilbert, C.; Provost, P. Identification of protein markers for extracellular vesicle (EV) subsets in cow’s milk. J. Proteom. 2019, 192, 78–88. [Google Scholar] [CrossRef]
- Gao, H.N.; Guo, H.Y.; Zhang, H.; Xie, X.L.; Wen, P.C.; Ren, F.Z. Yak-milk-derived exosomes promote proliferation of intestinal epithelial cells in an hypoxic environment. J. Dairy Sci. 2019, 102, 985–996. [Google Scholar] [CrossRef] [Green Version]
- Kirchner, B.; Buschmann, D.; Paul, V.; Pfaffl, M.W. Postprandial transfer of colostral extracellular vesicles and their protein and miRNA cargo in neonatal calves. PLoS ONE 2020, 15, e0229606. [Google Scholar] [CrossRef]
- Sedykh, S.E.; Purvinish, L.V.; Monogarov, A.S.; Burkova, E.E.; Grigor’eva, A.E.; Bulgakov, D.V.; Dmitrenok, P.S.; Vlassov, V.V.; Ryabchikova, E.I.; Nevinsky, G.A. Purified horse milk exosomes contain an unpredictable small number of major proteins. Biochim. Open 2017, 4, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Albanese, M.; Chen, Y.-F.A.; Hüls, C.; Gärtner, K.; Tagawa, T.; Mejias-Perez, E.; Keppler, O.T.; Göbel, C.; Zeidler, R.; Shein, M.; et al. Micro RNAs are minor constituents of extracellular vesicles and are hardly delivered to target cells. bioRxiv 2020, 2020.05.20.106393. [Google Scholar] [CrossRef]
- Kahn, S.; Liao, Y.; Du, X.; Xu, W.; Li, J.; Lonnerdal, B. Exosomal MicroRNAs in Milk from Mothers Delivering Preterm Infants Survive in Vitro Digestion and Are Taken Up by Human Intestinal Cells. Mol. Nutr. Food Res. 2018, 62, e1701050. [Google Scholar] [CrossRef] [PubMed]
- Mirza, A.H.; Kaur, S.; Nielsen, L.B.; Størling, J.; Yarani, R.; Roursgaard, M.; Mathiesen, E.R.; Damm, P.; Svare, J.; Mortensen, H.B.; et al. Breast Milk-Derived Extracellular Vesicles Enriched in Exosomes from Mothers with Type 1 Diabetes Contain Aberrant Levels of microRNAs. Front. Immunol. 2019, 10, 2543. [Google Scholar] [CrossRef]
- Van Herwijnen, M.J.C.; Driedonks, T.A.P.; Snoek, B.L.; Kroon, A.M.T.; Kleinjan, M.; Jorritsma, R.; Pieterse, C.M.J.; Hoen, E.; Wauben, M.H.M. Abundantly Present miRNAs in Milk-Derived Extracellular Vesicles Are Conserved Between Mammals. Front. Nutr. 2018, 5, 81. [Google Scholar] [CrossRef] [PubMed]
- Simpson, M.R.; Brede, G.; Johansen, J.; Johnsen, R.; Storrø, O.; Sætrom, P.; Øien, T. Human Breast Milk miRNA, Maternal Probiotic Supplementation and Atopic Dermatitis in Offspring. PLoS ONE 2015, 10, e0143496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, Q.; Li, M.; Wang, X.; Li, Q.; Wang, T.; Zhu, Q.; Zhou, X.; Wang, X.; Gao, X.; Li, X. Immune-related microRNAs are abundant in breast milk exosomes. Int. J. Biol. Sci. 2012, 8, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.; Du, X.; Li, J.; Lonnerdal, B. Human milk exosomes and their microRNAs survive digestion in vitro and are taken up by human intestinal cells. Mol. Nutr. Food Res. 2017, 61, 1700082. [Google Scholar] [CrossRef]
- Golan-Gerstl, R.; Elbaum Shiff, Y.; Moshayoff, V.; Schecter, D.; Leshkowitz, D.; Reif, S. Characterization and biological function of milk-derived miRNAs. Mol. Nutr. Food Res. 2017, 61, 1700009. [Google Scholar] [CrossRef]
- Zahoor, M.A.; Yao, X.D.; Henrick, B.M.; Verschoor, C.P.; Abimiku, A.; Osawe, S.; Rosenthal, K.L. Expression profiling of human milk derived exosomal microRNAs and their targets in HIV-1 infected mothers. Sci. Rep. 2020, 10, 12931. [Google Scholar] [CrossRef]
- Bozack, A.K.; Colicino, E.; Rodosthenous, R.; Bloomquist, T.R.; Baccarelli, A.A.; Wright, R.O.; Wright, R.J.; Lee, A.G. Associations between maternal lifetime stressors and negative events in pregnancy and breast milk-derived extracellular vesicle microRNAs in the programming of intergenerational stress mechanisms (PRISM) pregnancy cohort. Epigenetics 2021, 16, 389–404. [Google Scholar] [CrossRef]
- Sun, Q.; Chen, X.; Yu, J.; Zen, K.; Zhang, C.Y.; Li, L. Immune modulatory function of abundant immune-related microRNAs in microvesicles from bovine colostrum. Protein Cell 2013, 4, 197–210. [Google Scholar] [CrossRef] [Green Version]
- Benmoussa, A.; Laugier, J.; Beauparlant, C.J.; Lambert, M.; Droit, A.; Provost, P. Complexity of the microRNA transcriptome of cow milk and milk-derived extracellular vesicles isolated via differential ultracentrifugation. J. Dairy Sci. 2020, 103, 16–29. [Google Scholar] [CrossRef]
- Chen, Z.; Xie, Y.; Luo, J.; Chen, T.; Xi, Q.; Zhang, Y.; Sun, J. Milk exosome-derived miRNAs from water buffalo are implicated in immune response and metabolism process. BMC Vet. Res. 2020, 16, 123. [Google Scholar] [CrossRef] [PubMed]
- Quan, S.Y.; Nan, X.M.; Wang, K.; Zhao, Y.G.; Jiang, L.S.; Yao, J.H.; Xiong, B.H. Replacement of forage fiber with non-forage fiber sources in dairy cow diets changes milk extracellular vesicle-miRNA expression. Food Funct. 2020, 11, 2154–2162. [Google Scholar] [CrossRef] [PubMed]
- Pieters, B.C.; Arntz, O.J.; Bennink, M.B.; Broeren, M.G.; van Caam, A.P.; Koenders, M.I.; van Lent, P.L.; van den Berg, W.B.; de Vries, M.; van der Kraan, P.M.; et al. Commercial cow milk contains physically stable extracellular vesicles expressing immunoregulatory TGF-beta. PLoS ONE 2015, 10, e0121123. [Google Scholar] [CrossRef]
- Chen, T.; Xi, Q.Y.; Ye, R.S.; Cheng, X.; Qi, Q.E.; Wang, S.B.; Shu, G.; Wang, L.N.; Zhu, X.T.; Jiang, Q.Y.; et al. Exploration of microRNAs in porcine milk exosomes. BMC Genom. 2014, 15, 100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gu, Y.; Li, M.; Wang, T.; Liang, Y.; Zhong, Z.; Wang, X.; Zhou, Q.; Chen, L.; Lang, Q.; He, Z.; et al. Lactation-related microRNA expression profiles of porcine breast milk exosomes. PLoS ONE 2012, 7, e43691. [Google Scholar] [CrossRef]
- Quan, S.; Nan, X.; Wang, K.; Jiang, L.; Yao, J.; Xiong, B. Characterization of Sheep Milk Extracellular Vesicle-miRNA by Sequencing and Comparison with Cow Milk. Animals 2020, 10, 331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tripisciano, C.; Weiss, R.; Eichhorn, T.; Spittler, A.; Heuser, T.; Fischer, M.B.; Weber, V. Different Potential of Extracellular Vesicles to Support Thrombin Generation: Contributions of Phosphatidylserine, Tissue Factor, and Cellular Origin. Sci. Rep. 2017, 7, 6522. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M.; Asaka, Y.; Ogawara, T.; Yorozu, Y. Nipple Skin Trauma in Breastfeeding Women During Postpartum Week One. Breastfeed. Med. 2018, 13, 479–484. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Foitzik, T.; Kruschewski, M.; Kroesen, A.J.; Hotz, H.G.; Eibl, G.; Buhr, H.J. Does glutamine reduce bacterial translocation? A study in two animal models with impaired gut barrier. Int. J. Colorectal Dis. 1999, 14, 143–149. [Google Scholar] [CrossRef]
- Nolan, L.S.; Parks, O.B.; Good, M. A Review of the Immunomodulating Components of Maternal Breast Milk and Protection Against Necrotizing Enterocolitis. Nutrients 2019, 12, 14. [Google Scholar] [CrossRef] [Green Version]
- Chen, T.; Xie, M.Y.; Sun, J.J.; Ye, R.S.; Cheng, X.; Sun, R.P.; Wei, L.M.; Li, M.; Lin, D.L.; Jiang, Q.Y.; et al. Porcine milk-derived exosomes promote proliferation of intestinal epithelial cells. Sci. Rep. 2016, 6, 33862. [Google Scholar] [CrossRef]
- Yu, S.; Zhao, Z.; Sun, L.; Li, P. Fermentation Results in Quantitative Changes in Milk-Derived Exosomes and Different Effects on Cell Growth and Survival. J. Agric. Food Chem. 2017, 65, 1220–1228. [Google Scholar] [CrossRef]
- Pisano, C.; Galley, J.; Elbahrawy, M.; Wang, Y.; Farrell, A.; Brigstock, D.; Besner, G.E. Human Breast Milk-Derived Extracellular Vesicles in the Protection Against Experimental Necrotizing Enterocolitis. J. Pediatr. Surg. 2020, 55, 54–58. [Google Scholar] [CrossRef] [Green Version]
- Reif, S.; Elbaum Shiff, Y.; Golan-Gerstl, R. Milk-derived exosomes (MDEs) have a different biological effect on normal fetal colon epithelial cells compared to colon tumor cells in a miRNA-dependent manner. J. Transl. Med. 2019, 17, 325. [Google Scholar] [CrossRef]
- Miyake, H.; Lee, C.; Chusilp, S.; Bhalla, M.; Li, B.; Pitino, M.; Seo, S.; O’Connor, D.L.; Pierro, A. Human breast milk exosomes attenuate intestinal damage. Pediatr. Surg. Int. 2020, 36, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Reif, S.; Elbaum-Shiff, Y.; Koroukhov, N.; Shilo, I.; Musseri, M.; Golan-Gerstl, R. Cow and Human Milk-Derived Exosomes Ameliorate Colitis in DSS Murine Model. Nutrients 2020, 12, 2589. [Google Scholar] [CrossRef] [PubMed]
- Xie, M.Y.; Chen, T.; Xi, Q.Y.; Hou, L.J.; Luo, J.Y.; Zeng, B.; Li, M.; Sun, J.J.; Zhang, Y.L. Porcine milk exosome miRNAs protect intestinal epithelial cells against deoxynivalenol-induced damage. Biochem. Pharmacol. 2020, 175, 113898. [Google Scholar] [CrossRef]
- Wu, D.; Kittana, H.; Shu, J.; Kachman, S.D.; Cui, J.; Ramer-Tait, A.E.; Zempleni, J. Dietary Depletion of Milk Exosomes and Their MicroRNA Cargos Elicits a Depletion of miR-200a-3p and Elevated Intestinal Inflammation and Chemokine (C-X-C Motif) Ligand 9 Expression in Mdr1a(−/−) Mice. Curr. Dev. Nutr. 2019, 3, nzz122. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Moon, K.M.; Kim, C.Y. Tight Junction in the Intestinal Epithelium: Its Association with Diseases and Regulation by Phytochemicals. J. Immunol. Res. 2018, 2018, 2645465. [Google Scholar] [CrossRef] [Green Version]
- Stremmel, W.; Weiskirchen, R.; Melnik, B.C. Milk Exosomes Prevent Intestinal Inflammation in a Genetic Mouse Model of Ulcerative Colitis: A Pilot Experiment. Inflamm. Intest. Dis. 2020, 5, 117–123. [Google Scholar] [CrossRef]
- Wu, R.Y.; Li, B.; Koike, Y.; Maattanen, P.; Miyake, H.; Cadete, M.; Johnson-Henry, K.C.; Botts, S.R.; Lee, C.; Abrahamsson, T.R.; et al. Human Milk Oligosaccharides Increase Mucin Expression in Experimental Necrotizing Enterocolitis. Mol. Nutr. Food Res. 2019, 63, e1800658. [Google Scholar] [CrossRef]
- Komine-Aizawa, S.; Ito, S.; Aizawa, S.; Namiki, T.; Hayakawa, S. Cow milk exosomes activate NK cells and γδT cells in human PBMCs in vitro. Immunol. Med. 2020, 43, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Naslund, T.I.; Paquin-Proulx, D.; Paredes, P.T.; Vallhov, H.; Sandberg, J.K.; Gabrielsson, S. Exosomes from breast milk inhibit HIV-1 infection of dendritic cells and subsequent viral transfer to CD4+ T cells. Aids 2014, 28, 171–180. [Google Scholar] [CrossRef] [Green Version]
- Donalisio, M.; Cirrincione, S.; Rittà, M.; Lamberti, C.; Civra, A.; Francese, R.; Tonetto, P.; Sottemano, S.; Manfredi, M.; Lorenzato, A.; et al. Extracellular Vesicles in Human Preterm Colostrum Inhibit Infection by Human Cytomegalovirus In Vitro. Microorganisms 2020, 8, 1087. [Google Scholar] [CrossRef]
- Shukla, P.K.; Gangwar, R.; Manda, B.; Meena, A.S.; Yadav, N.; Szabo, E.; Balogh, A.; Lee, S.C.; Tigyi, G.; Rao, R. Rapid disruption of intestinal epithelial tight junction and barrier dysfunction by ionizing radiation in mouse colon in vivo: Protection by N-acetyl-l-cysteine. Am. J. Physiol. Gastrointest. Liver Physiol. 2016, 310, G705–G715. [Google Scholar] [CrossRef] [Green Version]
- Yu, M.; Yang, S.; Qiu, Y.; Chen, G.; Wang, W.; Xu, C.; Cai, W.; Sun, L.; Xiao, W.; Yang, H. Par-3 modulates intestinal epithelial barrier function through regulating intracellular trafficking of occludin and myosin light chain phosphorylation. J. Gastroenterol. 2015, 50, 1103–1113. [Google Scholar] [CrossRef]
- Baumgart, M.; Dogan, B.; Rishniw, M.; Weitzman, G.; Bosworth, B.; Yantiss, R.; Orsi, R.H.; Wiedmann, M.; McDonough, P.; Kim, S.G.; et al. Culture independent analysis of ileal mucosa reveals a selective increase in invasive Escherichia coli of novel phylogeny relative to depletion of Clostridiales in Crohn’s disease involving the ileum. ISME J. 2007, 1, 403–418. [Google Scholar] [CrossRef] [Green Version]
- Frank, D.N.; St Amand, A.L.; Feldman, R.A.; Boedeker, E.C.; Harpaz, N.; Pace, N.R. Molecular-phylogenetic characterization of microbial community imbalances in human inflammatory bowel diseases. Proc. Natl. Acad. Sci. USA 2007, 104, 13780–13785. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, F.; Paz, H.A.; Sadri, M.; Cui, J.; Kachman, S.D.; Fernando, S.C.; Zempleni, J. Dietary bovine milk exosomes elicit changes in bacterial communities in C57BL/6 mice. Am. J. Physiol. Gastrointest. Liver Physiol. 2019, 317, G618–g624. [Google Scholar] [CrossRef]
- Oliveira, M.C.; Di Ceglie, I.; Arntz, O.J.; van den Berg, W.B.; van den Hoogen, F.H.; Ferreira, A.V.; van Lent, P.L.; van de Loo, F.A. Milk-Derived Nanoparticle Fraction Promotes the Formation of Small Osteoclasts but Reduces Bone Resorption. J. Cell Physiol. 2017, 232, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.C.; Arntz, O.J.; Blaney Davidson, E.N.; van Lent, P.L.; Koenders, M.I.; van der Kraan, P.M.; van den Berg, W.B.; Ferreira, A.V.; van de Loo, F.A. Milk extracellular vesicles accelerate osteoblastogenesis but impair bone matrix formation. J. Nutr. Biochem. 2016, 30, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.C.; Pieters, B.C.H.; Guimarães, P.B.; Duffles, L.F.; Heredia, J.E.; Silveira, A.L.M.; Oliveira, A.C.C.; Teixeira, M.M.; Ferreira, A.V.M.; Silva, T.A.; et al. Bovine Milk Extracellular Vesicles Are Osteoprotective by Increasing Osteocyte Numbers and Targeting RANKL/OPG System in Experimental Models of Bone Loss. Front. Bioeng. Biotechnol. 2020, 8, 891. [Google Scholar] [CrossRef] [PubMed]
- Yun, B.; Maburutse, B.E.; Kang, M.; Park, M.R.; Park, D.J.; Kim, Y.; Oh, S. Short communication: Dietary bovine milk-derived exosomes improve bone health in an osteoporosis-induced mouse model. J. Dairy Sci. 2020. [Google Scholar] [CrossRef]
- Arntz, O.J.; Pieters, B.C.; Oliveira, M.C.; Broeren, M.G.; Bennink, M.B.; de Vries, M.; van Lent, P.L.; Koenders, M.I.; van den Berg, W.B.; van der Kraan, P.M.; et al. Oral administration of bovine milk derived extracellular vesicles attenuates arthritis in two mouse models. Mol. Nutr. Food Res. 2015, 59, 1701–1712. [Google Scholar] [CrossRef]
- Guerreiro, C.S.; Calado, Â.; Sousa, J.; Fonseca, J.E. Diet, Microbiota, and Gut Permeability-The Unknown Triad in Rheumatoid Arthritis. Front. Med. 2018, 5, 349. [Google Scholar] [CrossRef] [Green Version]
- Aarts, J.; Boleij, A.; Pieters, B.C.H.; Feitsma, A.L.; van Neerven, R.J.J.; Ten Klooster, J.P.; M’Rabet, L.; Arntz, O.J.; Koenders, M.I.; van de Loo, F.A.J. Flood Control: How Milk-Derived Extracellular Vesicles Can Help to Improve the Intestinal Barrier Function and Break the Gut-Joint Axis in Rheumatoid Arthritis. Front. Immunol. 2021, 12, 703277. [Google Scholar] [CrossRef] [PubMed]
- Badawy, A.A.; El-Magd, M.A.; AlSadrah, S.A. Therapeutic Effect of Camel Milk and Its Exosomes on MCF7 Cells In Vitro and In Vivo. Integr. Cancer Ther. 2018, 17, 1235–1246. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qin, W.; Tsukasaki, Y.; Dasgupta, S.; Mukhopadhyay, N.; Ikebe, M.; Sauter, E.R. Exosomes in Human Breast Milk Promote EMT. Clin. Cancer Res. 2016, 22, 4517–4524. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsuda, A.; Patel, T. Milk-derived Extracellular Vesicles for Therapeutic Delivery of Small Interfering RNAs. Methods Mol. Biol. 2018, 1740, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, A.; Moirangthem, A.; Angom, R.S.; Ishiguro, K.; Driscoll, J.; Yan, I.K.; Mukhopadhyay, D.; Patel, T. Safety of bovine milk derived extracellular vesicles used for delivery of RNA therapeutics in zebrafish and mice. J. Appl. Toxicol. 2019, 40, 706–718. [Google Scholar] [CrossRef]
- Munagala, R.; Aqil, F.; Jeyabalan, J.; Gupta, R.C. Bovine milk-derived exosomes for drug delivery. Cancer Lett. 2016, 371, 48–61. [Google Scholar] [CrossRef] [Green Version]
- Aqil, F.; Munagala, R.; Jeyabalan, J.; Agrawal, A.K.; Gupta, R. Exosomes for the Enhanced Tissue Bioavailability and Efficacy of Curcumin. AAPS J. 2017, 19, 1691–1702. [Google Scholar] [CrossRef]
- Li, D.; Yao, S.; Zhou, Z.; Shi, J.; Huang, Z.; Wu, Z. Hyaluronan decoration of milk exosomes directs tumor-specific delivery of doxorubicin. Carbohydr. Res. 2020, 493, 108032. [Google Scholar] [CrossRef]
- Agrawal, A.K.; Aqil, F.; Jeyabalan, J.; Spencer, W.A.; Beck, J.; Gachuki, B.W.; Alhakeem, S.S.; Oben, K.; Munagala, R.; Bondada, S.; et al. Milk-derived exosomes for oral delivery of paclitaxel. Nanomedicine 2017, 13, 1627–1636. [Google Scholar] [CrossRef]
- Kunihiro, A.G.; Brickey, J.A.; Frye, J.B.; Luis, P.B.; Schneider, C.; Funk, J.L. Curcumin, but not curcumin-glucuronide, inhibits Smad signaling in TGFβ-dependent bone metastatic breast cancer cells and is enriched in bone compared to other tissues. J. Nutr. Biochem. 2019, 63, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Vashisht, M.; Rani, P.; Onteru, S.K.; Singh, D. Curcumin Encapsulated in Milk Exosomes Resists Human Digestion and Possesses Enhanced Intestinal Permeability in Vitro. Appl. Biochem. Biotechnol. 2017, 183, 993–1007. [Google Scholar] [CrossRef] [PubMed]
- Carobolante, G.; Mantaj, J.; Ferrari, E.; Vllasaliu, D. Cow Milk and Intestinal Epithelial Cell-derived Extracellular Vesicles as Systems for Enhancing Oral Drug Delivery. Pharmaceutics 2020, 12, 226. [Google Scholar] [CrossRef] [Green Version]
- Aqil, F.; Munagala, R.; Jeyabalan, J.; Agrawal, A.K.; Kyakulaga, A.H.; Wilcher, S.A.; Gupta, R.C. Milk exosomes—Natural nanoparticles for siRNA delivery. Cancer Lett. 2019, 449, 186–195. [Google Scholar] [CrossRef] [PubMed]
- Laso-García, F.; Ramos-Cejudo, J.; Carrillo-Salinas, F.J.; Otero-Ortega, L.; Feliú, A.; Gómez-de Frutos, M.; Mecha, M.; Díez-Tejedor, E.; Guaza, C.; Gutiérrez-Fernández, M. Therapeutic potential of extracellular vesicles derived from human mesenchymal stem cells in a model of progressive multiple sclerosis. PLoS ONE 2018, 13, e0202590. [Google Scholar] [CrossRef] [Green Version]
- Nassar, W.; El-Ansary, M.; Sabry, D.; Mostafa, M.A.; Fayad, T.; Kotb, E.; Temraz, M.; Saad, A.N.; Essa, W.; Adel, H. Umbilical cord mesenchymal stem cells derived extracellular vesicles can safely ameliorate the progression of chronic kidney diseases. Biomater. Res. 2016, 20, 21. [Google Scholar] [CrossRef] [Green Version]
- Mason, S. Some aspects of gastric function in the newborn. Arch. Dis. Child. 1962, 37, 387–391. [Google Scholar] [CrossRef] [Green Version]
- Kamstrup, D.; Berthelsen, R.; Sassene, P.J.; Selen, A.; Mullertz, A. In Vitro Model Simulating Gastro-Intestinal Digestion in the Pediatric Population (Neonates and Young Infants). AAPS PharmSciTech 2017, 18, 317–329. [Google Scholar] [CrossRef] [PubMed]
- Zoppi, G.; Andreotti, G.; Pajno-Ferrara, F.; Njai, D.M.; Gaburro, D. Exocrine pancreas function in premature and full term neonates. Pediatr. Res. 1972, 6, 880–886. [Google Scholar] [CrossRef] [Green Version]
- Liu, W.; Ye, A.; Liu, W.; Liu, C.; Singh, H. Stability during in vitro digestion of lactoferrin-loaded liposomes prepared from milk fat globule membrane-derived phospholipids. J. Dairy Sci. 2013, 96, 2061–2070. [Google Scholar] [CrossRef] [Green Version]
- Hata, T.; Murakami, K.; Nakatani, H.; Yamamoto, Y.; Matsuda, T.; Aoki, N. Isolation of bovine milk-derived microvesicles carrying mRNAs and microRNAs. Biochem. Biophys. Res. Commun. 2010, 396, 528–533. [Google Scholar] [CrossRef]
- Wolf, T.; Baier, S.R.; Zempleni, J. The Intestinal Transport of Bovine Milk Exosomes Is Mediated by Endocytosis in Human Colon Carcinoma Caco-2 Cells and Rat Small Intestinal IEC-6 Cells. J. Nutr. 2015, 145, 2201–2206. [Google Scholar] [CrossRef] [Green Version]
- Kusuma, R.J.; Manca, S.; Friemel, T.; Sukreet, S.; Nguyen, C.; Zempleni, J. Human vascular endothelial cells transport foreign exosomes from cow’s milk by endocytosis. Am. J. Physiol. Cell Physiol. 2016, 310, C800–C807. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Betker, J.L.; Angle, B.M.; Graner, M.W.; Anchordoquy, T.J. The Potential of Exosomes from Cow Milk for Oral Delivery. J. Pharm. Sci. 2019, 108, 1496–1505. [Google Scholar] [CrossRef] [PubMed]
- De Rond, L.; van der Pol, E.; Hau, C.M.; Varga, Z.; Sturk, A.; van Leeuwen, T.G.; Nieuwland, R.; Coumans, F.A.W. Comparison of Generic Fluorescent Markers for Detection of Extracellular Vesicles by Flow Cytometry. Clin. Chem. 2018, 64, 680–689. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bagci Bosi, A.T.; Eriksen, K.G.; Sobko, T.; Wijnhoven, T.M.; Breda, J. Breastfeeding practices and policies in WHO European Region Member States. Public Health Nutr. 2016, 19, 753–764. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, Y.; Thaler, J.; Nieuwland, R. Extracellular Vesicles in Human Milk. Pharmaceuticals 2021, 14, 1050. https://doi.org/10.3390/ph14101050
Hu Y, Thaler J, Nieuwland R. Extracellular Vesicles in Human Milk. Pharmaceuticals. 2021; 14(10):1050. https://doi.org/10.3390/ph14101050
Chicago/Turabian StyleHu, Yong, Johannes Thaler, and Rienk Nieuwland. 2021. "Extracellular Vesicles in Human Milk" Pharmaceuticals 14, no. 10: 1050. https://doi.org/10.3390/ph14101050
APA StyleHu, Y., Thaler, J., & Nieuwland, R. (2021). Extracellular Vesicles in Human Milk. Pharmaceuticals, 14(10), 1050. https://doi.org/10.3390/ph14101050