Mānuka Oil—A Review of Antimicrobial and Other Medicinal Properties
Abstract
1. Introduction
2. Search Strategy
2.1. Chemical Composition
2.2. Medicinal Properties of Mānuka Oil
2.3. Antibacterial Activity
2.4. Mechanism of Action
2.5. Gram Positive Bacteria
2.6. Gram Negative Bacteria
2.7. Synergistic Effects
2.8. Antifungal Activity
2.9. Antiparasitic/Insecticidal Activity
2.10. Anti-Inflammatory Effects
2.11. Photo-Protective Effects
2.12. Antiviral Activity
2.13. Spasmolytic Activity
2.14. Safety and Tolerance
3. Future Directions
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ECHA | European Chemicals Agency |
| MIC | Minimum inhibitory concentrations |
| MBC | Minimum Bactericidal Concentration |
| EINECS | European Inventory of Existing Commercial chemical Substances |
| % v/v | volume per volume |
| VRE | vancomycin-resistant Enterococcus faecalis |
| MRSA | methicillin-resistant S. aureus |
| ATCC | American Type Culture Collection |
| EDTA | Ethylenediaminetetraacetic acid |
| LC50 | lethal concentration required to kill 50% of the parasites |
| LD50 | dose required to kill half the members of a tested population after a specified test duration |
| LPS | lipopolysaccharide |
| TNF-α | Tumor necrosis factor alpha |
| IL | Interleukin |
| UV | ultraviolet |
| HSV | Herpes simplex virus |
| IC50 | half maximal inhibitory concentration |
| US | United States of America |
| EU | European Union |
| CTFA | Cosmetic, Toiletry and Fragrance Association |
| HUVEC | human umbilical vein endothelial cells |
| LDM | Living Dermal Matrix |
| 3D | three-dimensional |
| ARTG | Australian Register of Therapeutic Goods. |
Appendix A
| PubMed | Search Strategies |
|---|---|
| #1 | (“leptospermum”[MeSH Terms] OR “leptospermum”[All Fields]) OR “leptospermums scoparium”[All Fields]) OR (“manuka”[All Fields] AND “oil”[All Fields])) OR ((“tea tree oil”[MeSH Terms] OR “tea tree oil”[All Fields] NOT (honey) [All Fields]) |
| #2 | ((“antifungal”[All Fields]) OR ((((((“anti-infective agents” OR “anti-infective agents”[MeSH Terms]) OR “anti-infective agents”[All Fields]) OR “antimicrobial”[All Fields]) OR “anti-microbial”[All Fields]) OR “antimicrobials”[All Fields] OR anti-inflammatory OR anti-inflammatory OR antiviral OR anti-viral OR “anti-viral agents”)) |
| #3 | #1 AND #2 |
| Embase (via Scopus) | |
| #1 | leptospermum OR “leptospermums scoparium” OR “manuka oil” OR “tea tree oil” OR “tea tree” NOT “honey” |
| #2 | “anti-infective agents” OR antibacterial OR “antibacterial agent” OR “anti-infective agents” OR “anti-infective agents” OR “antimicrobial” OR “anti-microbial” OR “antimicrobials” OR anti-inflammatory OR anti-inflammatory OR antiviral OR anti-viral OR “anti-viral agents” |
| #3 | #1 AND #2 |
References
- Wilson, P.G.; O’Brien, M.M.; Gadek, P.A.; Quinn, C.J.; Quinn, P.A.G.J. Myrtaceae revisited: A reassessment of infrafamilial groups. Am. J. Bot. 2001, 88, 2013–2025. [Google Scholar] [CrossRef]
- Maddocks-Jennings, W.; Wilkinson, J.M.; Cavanagh, H.; Shillington, D. Evaluating the effects of the essential oils Leptospermum scoparium (manuka) and Kunzea ericoides (kanuka) on radiotherapy induced mucositis: A randomized, placebo controlled feasibility study. Eur. J. Oncol. Nurs. 2009, 13, 87–93. [Google Scholar] [CrossRef]
- Carson, C.F.; Riley, T.V. Safety, efficacy and provenance of tea tree (Melaleuca alternifolia) oil. Contact Dermat. 2001, 45, 65–67. [Google Scholar] [CrossRef]
- Avonto, C.; Chittiboyina, A.G.; Wang, M.; Vasquez, Y.; Rua, D.; A Khan, I. In Chemico Evaluation of Tea Tree Essential Oils as Skin Sensitizers: Impact of the Chemical Composition on Aging and Generation of Reactive Species. Chem. Res. Toxicol. 2016, 29, 1108–1117. [Google Scholar] [CrossRef] [PubMed]
- Broadsheet, C.F.R. Essential Oil Production from Manuka & Kanuka. Available online: https://tararuacropping.files.wordpress.com/2016/08/essential-oil-manuka-may-2014_kanuka.pdf (accessed on 20 June 2020).
- Melora. Cosmetic Product Safety Report: MELORA™ Mānuka Oil; Melora: Dublin, Ireland, 2017. [Google Scholar]
- Christoph, F.; Kubeczka, K.-H.; Stahl-Biskup, E. The Composition of Commercial Manuka Oils from New Zealand. J. Essent. Oil Res. 1999, 11, 705–710. [Google Scholar] [CrossRef]
- Seyedalikhani, S.; Esperschuetz, J.; Dickinson, N.; Hofmann, R.; Breitmeyer, J.; Horswell, J.; Robinson, B. Biowastes to augment the essential oil production of Leptospermum scoparium and Kunzea robusta in low-fertility soil. Plant Physiol. Biochem. 2019, 137, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Porter, N.G.; Smale, P.E.; Nelson, M.A.; Hay, A.J.; Van Klink, J.W.; Dean, C.M. Variability in essential oil chemistry and plant morphology within aLeptospermum scopariumpopulation. N. Z. J. Bot. 1998, 36, 125–133. [Google Scholar] [CrossRef]
- Porter, N.G.; Wilkins, A.L. Chemical, physical and antimicrobial properties of essential oils of Leptospermum scoparium and Kunzea ericoides. Phytochemistry 1999, 50, 407–415. [Google Scholar] [CrossRef]
- Douglas, M.H.; Van Klink, J.W.; Smallfield, B.M.; Perry, N.B.; Anderson, R.E.; Johnstone, P.; Weavers, R.T. Essential oils from New Zealand manuka: Triketone and other chemotypes of Leptospermum scoparium. Phytochemistry 2004, 65, 1255–1264. [Google Scholar] [CrossRef] [PubMed]
- Christoph, F.; Kaulfers, P.M.; Stahl-Biskup, E. In vitro evaluation of the antibacterial activity of β-triketones admixed to Melaleuca oils. Planta Med. 2001, 67, 768–771. [Google Scholar] [CrossRef] [PubMed]
- Christoph, F.; Stahl-Biskup, E.; Kaulfers, P.M. Death Kinetics of Staphylococcus aureus Exposed to Commercial Tea Tree Oils s.l. J. Essent. Oil Res. 2001, 13, 98–102. [Google Scholar] [CrossRef]
- Fang, F.; Candy, K.; Melloul, E.; Bernigaud, C.; Chai, L.; Darmon, C.; Durand, R.; Botterel, F.; Chosidow, O.; Izri, A.; et al. In vitro activity of ten essential oils against Sarcoptes scabiei. Parasites Vectors 2016, 9, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Killeen, D.P.; Van Klink, J.W.; Smallfield, B.M.; Gordon, K.C.; Perry, N.B. Herbicidal β-triketones are compartmentalized in leaves of Leptospermumspecies: Localization by Raman microscopy and rapid screening. New Phytol. 2015, 205, 339–349. [Google Scholar] [CrossRef] [PubMed]
- Perry, N.B.; Brennan, N.J.; Van Klink, J.W.; Harris, W.; Douglas, M.H.; McGimpsey, J.A.; Smallfield, B.M.; Anderson, R.E. Essential oils from New Zealand manuka and kanuka: Chemotaxonomy of Leptospermum. Phytochemistry 1997, 44, 1485–1494. [Google Scholar] [CrossRef]
- Perry, N.B.; Van Klink, J.W.; Brennan, N.J.; Harris, W.; Anderson, R.E.; Douglas, M.H.; Smallfield, B.M. Essential oils from new zealand manuka and kanuka: Chemotaxonomy of kunzea. Phytochemistry 1997, 45, 1605–1612. [Google Scholar] [CrossRef]
- Dayan, F.E.; Howell, J.L.; Marais, J.P.; Ferreira, D.; Koivunen, M. Manuka Oil, A Natural Herbicide with Preemergence Activity. Weed Sci. 2011, 59, 464–469. [Google Scholar] [CrossRef]
- Jeong, E.Y.; Jeon, J.H.; Kim, H.W.; Kim, M.G.; Lee, H.S. Antimicrobial activity of leptospermone and its derivatives against human intestinal bacteria. Food Chem. 2009, 115, 1401–1404. [Google Scholar] [CrossRef]
- Jeong, E.Y.; Kim, M.G.; Lee, H.S. Acaricidal activity of triketone analogues derived fromLeptospermum scopariumoil against house-dust and stored-food mites. Pest Manag. Sci. 2009, 65, 327–331. [Google Scholar] [CrossRef]
- Muturi, E.J.; Selling, G.W.; Doll, K.M.; Hay, W.T.; Ramirez, J.L. Leptospermum scoparium essential oil is a promising source of mosquito larvicide and its toxicity is enhanced by a biobased emulsifier. PLoS ONE 2020, 15, e229076. [Google Scholar] [CrossRef]
- Govindarajan, M.; Rajeswary, M.; Benelli, G. δ-Cadinene, Calarene and δ-4-Carene from Kadsura heteroclita Essential Oil as Novel Larvicides Against Malaria, Dengue and Filariasis Mosquitoes. Comb. Chem. High Throughput Screen. 2016, 19, 565–571. [Google Scholar] [CrossRef]
- Reichling, J.; Koch, C.; Stahl-Biskup, E.; Sojka, C.; Schnitzler, P. Virucidal Activity of a β-Triketone-Rich Essential Oil ofLeptospermum scoparium (Manuka Oil) Against HSV-1 and HSV-2 in Cell Culture. Planta Med. 2005, 71, 1123–1127. [Google Scholar] [CrossRef] [PubMed]
- Shelly, T.E.; Cowan, A.N.; Edu, J.; Pahio, E. Mating success of male Mediterranean fruit flies following exposure to two sources of alpha-copaene, manuka oil and mango. Fla. Entomol. 2008, 91, 9–15. [Google Scholar] [CrossRef]
- Hanula, J.L.; Mayfield, A.E., III; Reid, L.S.; Horn, S. Influence of trap distance from a source population and multiple traps on captures and attack densities of the redbay ambrosia beetle (Coleoptera: Curculionidae: Scolytinae). J. Econol. Entomol. 2016, 109, 1196–1204. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kong, Q.; Zhang, L.; An, P.; Qi, J.; Yu, X.; Lu, J.; Ren, X. Antifungal mechanisms of α-terpineol and terpene-4-alcohol as the critical components of Melaleuca alternifolia oil in the inhibition of rot disease caused by Aspergillus ochraceus in postharvest grapes. J. Appl. Microbiol. 2019, 126, 1161–1174. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, M.N.M.; Aquino, S.G.; Rossa Junior, C.; Spolidorio, D.M.P. Terpinen-4-ol and alpha-terpineol (tea tree oil components) inhibit the production of IL-1β, IL-6 and IL-10 on human macrophages. Inflamm. Res. 2014, 63, 769–778. [Google Scholar] [CrossRef] [PubMed]
- Lis-Balchin, M.; Hart, S.L.; Deans, S.G. Pharmacological and antimicrobial studies on different tea-tree oils (Melaleuca alternifolia, Leptospermum scoparium or Manuka and Kunzea ericoides or Kanuka), originating in Australia and New Zealand. Phytotherapy Res. 2000, 14, 623–629. [Google Scholar] [CrossRef]
- Manuka Oil Beta Troketones. Manuka Essential Oil. Available online: http://www.mbtk.org.nz/what-msstk/manuka-essential-oil/ (accessed on 23 June 2020).
- Christoph, F.; Kaulfers, P.-M.; Stahl-Biskup, E. A Comparative Study of the in vitro Antimicrobial Activity of Tea Tree Oils s.l. with Special Reference to the Activity of β-Triketones. Planta Med. 2000, 66, 556–560. [Google Scholar] [CrossRef]
- Van Vuuren, S.; Docrat, Y.; Kamatou, G.; Viljoen, A.M. Essential oil composition and antimicrobial interactions of understudied tea tree species. S. Afr. J. Bot. 2014, 92, 7–14. [Google Scholar] [CrossRef]
- Bent, S. Herbal Medicine in the United States: Review of Efficacy, Safety, and Regulation: Grand Rounds at University of California, San Francisco Medical Center. J. Gen. Intern. Med. 2008, 23, 854–859. [Google Scholar] [CrossRef]
- Māori Medicine. Available online: https://www.tepapa.govt.nz/discover-collections/read-watch-play/maori/maori-medicine (accessed on 23 June 2020).
- Plant Use Details: Leptospermum scoparium. Mānuka. Kahikatoa. Available online: https://maoriplantuse.landcareresearch.co.nz/WebForms/PeoplePlantsDetails.aspx?PKey=34969a5f-698d-437d-8a84-c71dd21351a5 (accessed on 23 June 2020).
- Alnaimat, S.; Wainwright, M.; Jaber, S.; Amasha, R. Mechanism of the Antibacterial Action of (Leptospermum scoparium) Oil on Methicillin-resistant Staphylococcus aureus (MRSA)and E. coli). In Proceedings of the 2nd Mediterranean Symposium on Medicinal and Aromatic Plants (MESMAP-2), Antalya, Turkey, 22–25 April 2015. [Google Scholar]
- Fratini, F.; Mancini, S.; Turchi, B.; Friscia, E.; Pistelli, L.; Giusti, G.; Cerri, D. A novel interpretation of the Fractional Inhibitory Concentration Index: The case Origanum vulgare L. and Leptospermum scoparium J.R. et G. Forst essential oils against Staphylococcus aureus strains. Microbiol. Res. 2017, 195, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.C.; Yan, S.H.; Yen, M.Y.; Wu, P.F.; Liao, W.T.; Huang, T.S.; Wen, Z.H.; Wang, H.M.D. Investigations of kanuka and manuka essential oils for in vitro treatment of disease and cellular inflammation caused by infectious microorganisms. J. Microbiol. Immunol. Infect. 2016, 49, 104–111. [Google Scholar] [CrossRef]
- Takarada, K.; Kimizuka, R.; Takahashi, N.; Honma, K.; Okuda, K.; Kato, T. A comparison of the antibacterial efficacies of essential oils against oral pathogens. Oral Microbiol. Immunol. 2004, 19, 61–64. [Google Scholar] [CrossRef] [PubMed]
- Song, C.Y.; Nam, E.H.; Park, S.H.; Hwang, C.Y. In vitroefficacy of the essential oil fromLeptospermum scoparium(manuka) on antimicrobial susceptibility and biofilm formation inStaphylococcus pseudintermediusisolates from dogs. Veter Dermatol. 2013, 24, 404. [Google Scholar] [CrossRef]
- Turchi, B.; Mancini, S.; Pistelli, L.; Najar, B.; Fratini, F. Sub-inhibitory concentration of essential oils induces antibiotic resistance in Staphylococcus aureus. Nat. Prod. Res. 2019, 33, 1509–1513. [Google Scholar] [CrossRef]
- Orchard, A.; Van Vuuren, S.; Viljoen, A.M.; Kamatou, G.P.P. The in vitro antimicrobial evaluation of commercial essential oils and their combinations against acne. Int. J. Cosmet. Sci. 2018, 40, 226–243. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Lee, H.; Lee, J.; Joo, C.; Choe, T. The effects of antimicrobial properties of manuka oil and improvement of acne. J. Korean Soc. Cosmetol. 2011, 17, 245–256. [Google Scholar]
- Wu, Q.; Wu, Q. Antimicrobial Effect of Manuka Honey and Kanuka Honey Alone and in Combination with the Bioactives Against the Growth of Propionibacterium Acnes ATCC 6919. Master’s Thesis, Massey University, Auckland, New Zealand, June 2011. [Google Scholar]
- Prosser, J.; Anderson, C.; Horswell, J.; Speir, T. Can manuka (Leptospermum scoparium) antimicrobial properties be utilised in the remediation of pathogen contaminated land? Soil Biol. Biochem. 2014, 75, 167–174. [Google Scholar] [CrossRef]
- Killeen, D.P.; Larsen, L.; Dayan, F.E.; Gordon, K.C.; Perry, N.B.; Van Klink, J.W. Nortriketones: Antimicrobial Trimethylated Acylphloroglucinols from Mānuka (Leptospermum scoparium). J. Nat. Prod. 2016, 79, 564–569. [Google Scholar] [CrossRef] [PubMed]
- Bergonzelli, G.; Donnicola, D.; Porta, N.; Corthésy-TheulazI, E. Essential Oils as Components of a Diet-Based Approach to Management of Helicobacter Infection. Antimicrob. Agents Chemother. 2003, 47, 3240–3246. [Google Scholar] [CrossRef] [PubMed]
- Schmolz, E.; Doebner, R.; Auste, R.; Daum, R.; Welge, G.; Lamprecht, I. Bioenergetic investigations on tea-tree and related essential oils. Thermochim. Acta 1999, 337, 71–81. [Google Scholar] [CrossRef]
- Prosser, J.; Woods, R.; Horswell, J.; Robinson, B. The potential in-situ antimicrobial ability of Myrtaceae plant species on pathogens in soil. Soil Biol. Biochem. 2016, 96, 1–3. [Google Scholar] [CrossRef]
- Jeong, E.Y.; Lee, M.J.; Lee, H.S. Antimicrobial activities of leptospermone isolated from Leptospermum scoparium seeds and structure-activity relationships of its derivatives against foodborne bacteria. Food Sci. Biotechnol. 2018, 27, 1541–1547. [Google Scholar] [CrossRef] [PubMed]
- Song, S.; Hyun, J.; Kang, J.H.; Hwang, C.Y. In vitro antibacterial activity of the manuka essential oil from Leptospermum scoparium combined with Tris-EDTA against Gram-negative bacterial isolates from dogs with otitis externa. Veter Dermatol. 2020, 31, 81. [Google Scholar] [CrossRef] [PubMed]
- Bensignor, E.; Fabriès, L.; Bailleux, L. A split-body, randomized, blinded study to evaluate the efficacy of a topical spray composed of essential oils and essential fatty acids from plant extracts with antimicrobial properties. Veter Dermatol. 2016, 27, 464. [Google Scholar] [CrossRef] [PubMed]
- Filoche, S.K.; Soma, K.; Sissons, C.H. Antimicrobial effects of essential oils in combination with chlorhexidine digluconate. Oral Microbiol. Immunol. 2005, 20, 221–225. [Google Scholar] [CrossRef]
- Schwiertz, A.; Duttke, C.; Hild, J.; Müller, H.J. In vitro activity of essential oils on microorganisms isolated from vaginal infections. Int. J. Aromatherapy 2006, 16, 169–174. [Google Scholar] [CrossRef]
- Hammer, K.; Carson, C.F.; Riley, T.V. Antifungal effects of Melaleuca alternifolia (tea tree) oil and its components on Candida albicans, Candida glabrata and Saccharomyces cerevisiae. J. Antimicrob. Chemother. 2004, 53, 1081–1085. [Google Scholar] [CrossRef]
- Lee, Y.S.; Kim, J.; Lee, S.G.; Oh, E.; Shin, S.C.; Park, I.K. Effects of plant essential oils and components from Oriental sweetgum (Liquidambar orientalis) on growth and morphogenesis of three phytopathogenic fungi. Pestic. Biochem. Physiol. 2009, 93, 138–143. [Google Scholar] [CrossRef]
- Elisa, B.; Aldo, A.; Ludovica, G.; Viviana, P.; Debora, B.; Massa, N.; Giorgia, N.; Elisa, G. Chemical composition and antimycotic activity of six essential oils (cumin, fennel, manuka, sweet orange, cedar and juniper) against different Candida spp. Nat. Prod. Res. 2019, 11, 6. [Google Scholar] [CrossRef]
- Gonzalez-Audino, P.; Picollo, M.I.; Gallardo, A.; Toloza, A.; Vassena, C.; Cueto, G.M. Comparative toxicity of oxygenated monoterpenoids in experimental hydroalcoholic lotions to permethrin-resistant adult head lice. Arch. Dermatol. Res. 2010, 303, 361–366. [Google Scholar] [CrossRef]
- George, D.R.; Sparagano, O.A.E.; Port, G.; Okello, E.; Shiel, R.S.; Guy, J.H. Environmental interactions with the toxicity of plant essential oils to the poultry red miteDermanyssus gallinae. Med. Veter Entomol. 2010, 24, 1–8. [Google Scholar] [CrossRef] [PubMed]
- George, D.R.; Sparagano, O.A.E.; Port, G.; Okello, E.; Shiel, R.S.; Guy, J.H. Toxicity of plant essential oils to different life stages of the poultry red mite, Dermanyssus gallinae, and non-target invertebrates. Med. Veter Entomol. 2010, 24, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Park, C.G.; Jang, M.; Shin, E.; Kim, J. Myrtaceae Plant Essential Oils and their β-Triketone Components as Insecticides against Drosophila suzukii. Molecules 2017, 22, 1050. [Google Scholar] [CrossRef] [PubMed]
- Muturi, E.J.; Ramirez, J.L.; Doll, K.M.; Bowman, M.J. Combined Toxicity of Three Essential Oils Against Aedes aegypti (Diptera: Culicidae) Larvae. J. Med. Entomol. 2017, 54, 1684–1691. [Google Scholar] [CrossRef] [PubMed]
- George, D.R.; Sparagano, O.A.E.; Port, G.; Okello, E.; Shiel, R.; Guy, J. Repellence of plant essential oils to Dermanyssus gallinae and toxicity to the non-target invertebrate Tenebrio molitor. Veter Parasitol. 2009, 162, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Kendra, P.E.; Montgomery, W.S.; Niogret, J.; Deyrup, M.A.; Guillén, L.; Epsky, N.D. Xyleborus glabratus, X. affinis, and X. ferrugineus (Coleoptera: Curculionidae: Scolytinae): Electroantennogram Responses to Host-Based Attractants and Temporal Patterns in Host-Seeking Flight. Environ. Entomol. 2012, 41, 1597–1605. [Google Scholar] [CrossRef]
- Hanula, J.L.; Sullivan, B.T.; Wakarchuk, D. Variation in Manuka Oil Lure Efficacy for CapturingXyleborus glabratus (Coleoptera: Curculionidae: Scolytinae), and Cubeb Oil as an Alternative Attractant. Environ. Entomol. 2013, 42, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Kwon, O.S.; Jung, S.H.; Yang, B.S. Topical Administration of Manuka Oil Prevents UV-B Irradiation-Induced Cutaneous Photoaging in Mice. Evid. Based Complement. Altern. Med. 2013, 2013, 1–10. [Google Scholar] [CrossRef]
- Park, M.-I.; Lee, B.-J.; Lee, S.W.; Tag, H.; Soonkyu, J. Vitamin C Derivative and Manuka Oil Surface Treated Composite Powder for Skin-Whitening and Wrinkle-Care. Patent KR101639615B1, 15 July 2016. [Google Scholar]
- Looker, K.J.; Garnett, G.P. A systematic review of the epidemiology and interaction of herpes simplex virus types 1 and 2. Sex. Transm. Infect. 2005, 81, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Astani, A.; Reichling, J.; Schnitzler, P. Screening for Antiviral Activities of Isolated Compounds from Essential Oils. Evid. Based Complement. Altern. Med. 2011, 2011, 1–8. [Google Scholar] [CrossRef]
- Schnitzler, P.; Wiesenhofer, K.; Reichling, J. Comparative study on the cytotoxicity of different Myrtaceae essential oils on cultured vero and RC-37 cells. Die Pharm. 2008, 63, 830–835. [Google Scholar]
- Schnitzler, P. Essential Oils for the Treatment of Herpes Simplex Virus Infections. Chemotherapy 2019, 64, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Lis-Balchin, M.; Hart, S.L. An Investigation of the Actions of the Essential oils of Manuka (Leptospermum scoparium) and Kanuka (Kunzea ericoides), Myrtaceae on Guinea-pig Smooth Muscle. J. Pharm. Pharmacol. 1998, 50, 809–811. [Google Scholar] [CrossRef]
- Lis-Balchin, M. Essential oils and ’aromatherapy’: Their modern role in healing. J. R. Soc. Health 1997, 117, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Campo Research PTE Ltd. Manuka Oil Extract Leptospermum Scoparium; Campo Research PTE Ltd.: Singapore, 2015. [Google Scholar]
- Yoo, J.-G.; Han, M.; Hong, N. External Preparation for Antibacterial, Anti-Inflammatory and Skin Protection Containing Manuka Oil as a Main Ingredient. Patent KR19990016741A, 12 May 2003. [Google Scholar]
- Searching the Australian Register of Therapeutic Goods (ARTG). Available online: https://www.tga.gov.au/searching-australian-register-therapeutic-goods-artg (accessed on 1 July 2020).
| Component | Percentage in Commercial Compositions | IUPAC Name | Known Properties | Ref. |
|---|---|---|---|---|
| α-pinene | Up to 21.5% | 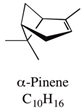 | Reported to have antibiotic resistance modulation, anticoagulant, antitumor, antimicrobial, antimalarial, antioxidant, anti-inflammatory, anti-Leishmania, and analgesic effects in association with other essential oils. | [16,17] |
| Leptospermone | 0.8–19.4% | 6-isovaleryl-2,2,4,4-tetramethyl-1,3,5-cyclohexanetrione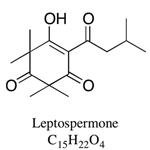 | Herbicidal; antibacterial: treatment with 5–20 mg/disc of concentrate was effective against foodborne bacteria: Listeria monocytogenes, Staphylococcus aureus and Staphylococcus intermedius and three Gram-negative bacteria: Salmonella typhimurium, Shigella flexneri and Shigella sonnei | [18,19,20] |
| Calamenene | 2.5–18.5% | 1,6-Dimethyl-4-isopropyltetralin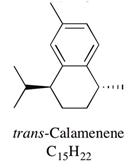 | Major constituent of mānuka oil; contributes to insecticidal, antiseptic, bactericidal, analgesic and anti-inflammatory properties. Antibacterial effect against S. aureus and MRSA was shown. | [12,21] |
| δ-cadinene | 0.9–6.9% | 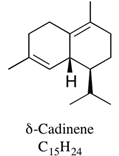 | Pesticidal effects against mosquito have been shown for constituents isolated from Kadsura heteroclita leaf oil. | [22] |
| Cadina-1,4-diene | 0.1–5.9% | 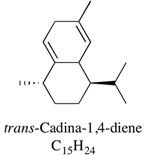 | Not reported | |
| Cadina-3,5-diene | 3.0–10.0% | 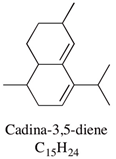 | Not reported | |
| Flavesone | 0.7–5.8% | 2,2,4,4-Tetramethyl-6-(3-methylbutanoyl)cyclohexane-1,3,5-trione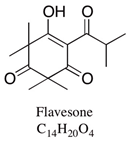 | Antiviral properties. | [23] |
| α-copaene | 4.3–6.5% | (1R,2S,6S,7S,8S)-8-isopropyl-1,3-dimethyltricyclo[4.4.0.02,7]dec-3-ene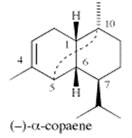 C15H24 | Enhances mating in male Mediterranean fruit flies; Lures for trapping Redbay ambrosia beetle (Xyleborus glabratus). | [24,25] |
| α-selinene | 1.3–5.0% | 7-Epi-alpha-Selinene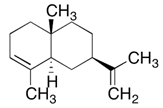 α-selinene C15H24 | Insecticidal: retarding the growth of mosquito larvae. | [21] |
| α-terpineol | 1–2% | 2-(4-methylcyclohex-3-en-1-yl)propan-2-ol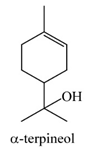 C10H18O | Antifungal effects; preservative for the postharvest storage of grapes and other fruits; has been shown to suppress the production of inflammatory mediators when sourced from Tea tree oil. | [26,27] |
| terpinene-4-ol | 0.8–1.4% | 4-methyl-1-propan-2-ylcyclohex-3-en-1-ol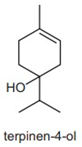 C10H18O | Spasmolytic activity; anti-inflammatory properties have been characterized in constituent isolated from Melaleuca alternifolia. | [28] |
| Organism | Method of Analysis | %(vol/vol) (µg/mL) | Relevance | Ref. | |
|---|---|---|---|---|---|
| # MIC | * MBC | ||||
| Gram positive bacteria | |||||
| Atopobium vaginae | Broth microdilution | 0.001 | 0.001 | Vaginal infections, pre-term birth and neonatal infections | [53] |
| Bacillus subtilis | Broth microdilution | 0.03 | 0.50 | Intestinal bacteria | [35] |
| Bacteroides vulgatus | Broth microdilution | 0.001 | 0.001 | Vaginal infections, pre-term birth and neonatal infections | [53] |
| Lactobacillus plantarum | Broth microdilution | 12.5 | Not reported | Oral probiotic | [52] |
| H2O2-producing lactobacilli and non H2O2-producing lactobacilli | Broth microdilution | 0.075 | 0.075 | Vaginal bacteria | [53] |
| Listeria monocytogenes | Two-fold serial dilution | 0.414 | Not reported | Foodborne pathogen | [49] |
| Gardnerella vaginalis | Broth microdilution | 0.001 | 0.001 | Vaginal infections, pre-term birth and neonatal infections | [53] |
| Propionibacterium acnes ATCC 11827 | Broth microdilution | 0.055 | Not reported | Acne | [41] |
| Propionibacterium acnes | Broth microdilution | 0.211 | 0.25 | Acne | [43] |
| Staphylococcus aureus | Two‘-fold serial dilution | 0.535 | Not reported | Foodborne pathogen | [49] |
| S. aureus strains | Two-fold serial dilution | 0.513 | Not reported | Multiple clinical manifestations in humans | [36] |
| Methicillin-resistant S. aureus | Broth microdilution | 0.03 | 1.0 | Skin infections, pneumonia, sepsis, surgical site infections | [35] |
| Streptococcus agalactiae | Broth microdilution | 0.001 | 0.001 | Meningitis; sepsis | [53] |
| Staphylococcus epidermidis ATCC 2223 | Broth microdilution | 1.40 | Not reported | Acne | [41] |
| S. intermedius | Broth microdilution | 0.0581 | Not reported | Foodborne pathogen | [49] |
| S. sorbinus | Broth microdilution | 0.048 | Not reported | Oral pathogen | [36] |
| S. sorbinus 6715 | 96-well liquid culture microdilution | 0.13 | 0.25 | Oral pathogen | [38] |
| S. sorbinus B13 | 96-well liquid culture microdilution | 0.25 | 0.25 | Oral pathogen | |
| S. mutans JC2 | 96-well liquid culture microdilution | 0.25 | 0.25 | Oral pathogen | |
| S. mutans ATCC 25175 | Two-fold microdilution | 6.2 | Not reported | Oral pathogen; dental caries | [52] |
| Vancomycin-resistant Enterococcus faecalis (VRE) | Broth microdilution | 0.0064 | Not reported | Sepsis; infection of open wounds | [45] |
| Gram negative bacteria | |||||
| Actinobacillus actinomycetemcomitans (now known as Aggregatibacter actinomycetemcomitans) strains Y4, ATCC 29523, 29524, 33384 | 96-well liquid culture microdilution | 0.03 | 0.13 | Oral pathogen | [38] |
| Escherichia coli | Broth microdilution | >4 | >4 | Intestinal bacteria; opportunistic pathogen | [35] |
| E. coli antibiotic and multidrug resistant strains | Two-fold microdilution | 1–4 | 2–4 | Hospital-based infections | [50] |
| Fusobacterium nucleatum ATCC 25586 strains | Broth microdilution | 0.03 | 0.03 | Periodontal disease; dental caries | [50] |
| H. pylori | Broth microdilution | Not reported | 0.4 | Gastritis, gastric ulcers and gastric cancer | [46] |
| Klebsiella pneumoniae spp. antibiotics and multidrug resistant isolates | Microdilution | 2–4 | 2–8 | Hospital-based infections; opportunistic pathogen | [50] |
| Porphyromonas gingivalis ATCC 33277, W50 and Su63 | 96-well liquid culture microdilution | 0.03 | 0.06 | Oral pathogen | [38] |
| P. gingivalis ATCC 53977 | 96-well liquid culture microdilution | 0.03 | 0.03 | Oral pathogen | [38] |
| Pseudomonas aeruginosa antibiotic and multidrug resistant isolates | Two-fold microdilution | ≥8 | ≥8 | Burn wound infections, sepsis | [50] |
| Proteus mirabilis | Two-fold microdilution | 1–4 | 2–8 | Hospital based infections | |
| Salmonella typhimurium | Two-fold serial dilution | 0.00236 | Not reported | Foodborne bacteria | [49] |
| S. flexneri | Two-fold serial dilution | 0.00653 | Foodborne bacteria | [49] | |
| S. sonnei | Two-fold serial dilution | 0.00697 | Foodborne bacteria | [49] | |
| Serratia marcescens | Broth microdilution | ≥4 | ≥4 | Opportunistic pathogen | [35] |
| Organism | * MIC (% v/v) | # MFC (% v/v) | Relevance | Ref. |
|---|---|---|---|---|
| Malassezia furfur | 1.56 | Not reported | Pityriasis versicolor and Pityrosporum folliculitis | [37] |
| Trichosporon mucoides | 1.56 | Not reported | Opportunistic pathogen | |
| Candida albicans | 3.13 | Not reported | Opportunistic pathogen | |
| Candida tropicalis | 3.13 | Not reported | Opportunistic pathogen | |
| Candida albicans | 0.015 | 0.015 | Candida vulvovaginitis infections | [53] |
| Candida glabrata | 0.010 | 0.010 | Vaginal candidiasis |
| Organism | Method | Lethal Effect | Clinical Significance | Ref. |
|---|---|---|---|---|
| Acaricidal activity | ||||
| Dermanyssus gallinae | Contact assay | LC50: 0.02 to 0.03, LC90:0.05 to 0.07 LD99: 0.10 mg/cm2 | Poultry red mite | [58,59] |
| D. farinae | Fabricated disc method | LD50: 0.54 μg/cm2 | House dust mite | [20] |
| D. pteronyssinus | LD50: 0.67 μg/cm2 | House dust mite | ||
| Tyrophagus putrescentiae | LD50: 1.21 μg/cm2 | Stored product mite | ||
| Sarcoptes scabei | Contact assay | LT50: 60 min for 5% solution LT50: 30 min for 10% solution | Human scabies mites | [14] |
| Drosophila suzukii | Contact assay | 0.60 μg/mL for males and 1.10 for females | Fruit fly pest | [60] |
| Aedes aegypti (Linnaeus) larvae | Larvicidal bioassay | LC90: 66.62 | Malaria | [21,61] |
| Repellent effects | ||||
| Dermanyssus gallinae (De Geer) | Fumigant assay | 80–84% | Poultry red mite | [62] |
| Dermanyssus gallinae | Fumigant assay | 80% | Poultry red mite | [8,59] |
| Sarcoptes scabei | Fumigant assay | 80% | Human scabies mites | [14] |
| Cell Line | Assay | Test | * TC50 | Control | Ref. |
|---|---|---|---|---|---|
| RC-37 cells (African green monkey kidney cells) | Neutral red assay after treatment for 72 h | β-pinene | 0.006% | 1% ethanol | [68] |
| RC-37 cells (African green monkey kidney cells) | Neutral red assay after treatment for 96 h | Mānuka oil Leptospermone | 0.0042% 0.04% | 2.6% ethanol | [68] |
| THP-1 (monocyte/macrophage cell line) | XTT cell viability assay after 48 h of treatment | 0.1–10% dissolved in DMSO | No toxicity | DMSO | [37] |
| Vero (African green monkey kidney cells) | Neutral red assay 96 h after treatment | Mānuka oil (0.001% to 1%) Leptospermone | 0.0042% 0.04% | 1% ethanol | [69] |
| HUVEC (Human umbilical vein endothelial cells) | Cell Titre Assay | Mānuka oil | ~0.4% | No treatment | [38] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mathew, C.; Tesfaye, W.; Rasmussen, P.; Peterson, G.M.; Bartholomaeus, A.; Sharma, M.; Thomas, J. Mānuka Oil—A Review of Antimicrobial and Other Medicinal Properties. Pharmaceuticals 2020, 13, 343. https://doi.org/10.3390/ph13110343
Mathew C, Tesfaye W, Rasmussen P, Peterson GM, Bartholomaeus A, Sharma M, Thomas J. Mānuka Oil—A Review of Antimicrobial and Other Medicinal Properties. Pharmaceuticals. 2020; 13(11):343. https://doi.org/10.3390/ph13110343
Chicago/Turabian StyleMathew, Cynthia, Wubshet Tesfaye, Phil Rasmussen, Gregory M Peterson, Andrew Bartholomaeus, Manab Sharma, and Jackson Thomas. 2020. "Mānuka Oil—A Review of Antimicrobial and Other Medicinal Properties" Pharmaceuticals 13, no. 11: 343. https://doi.org/10.3390/ph13110343
APA StyleMathew, C., Tesfaye, W., Rasmussen, P., Peterson, G. M., Bartholomaeus, A., Sharma, M., & Thomas, J. (2020). Mānuka Oil—A Review of Antimicrobial and Other Medicinal Properties. Pharmaceuticals, 13(11), 343. https://doi.org/10.3390/ph13110343






