Reversal of Ovarian Cancer Cell Lines Multidrug Resistance Phenotype by the Association of Apiole with Chemotherapies
Abstract
1. Introduction
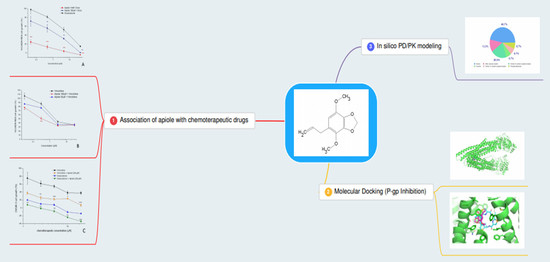
2. Results
2.1. Apiole Identification
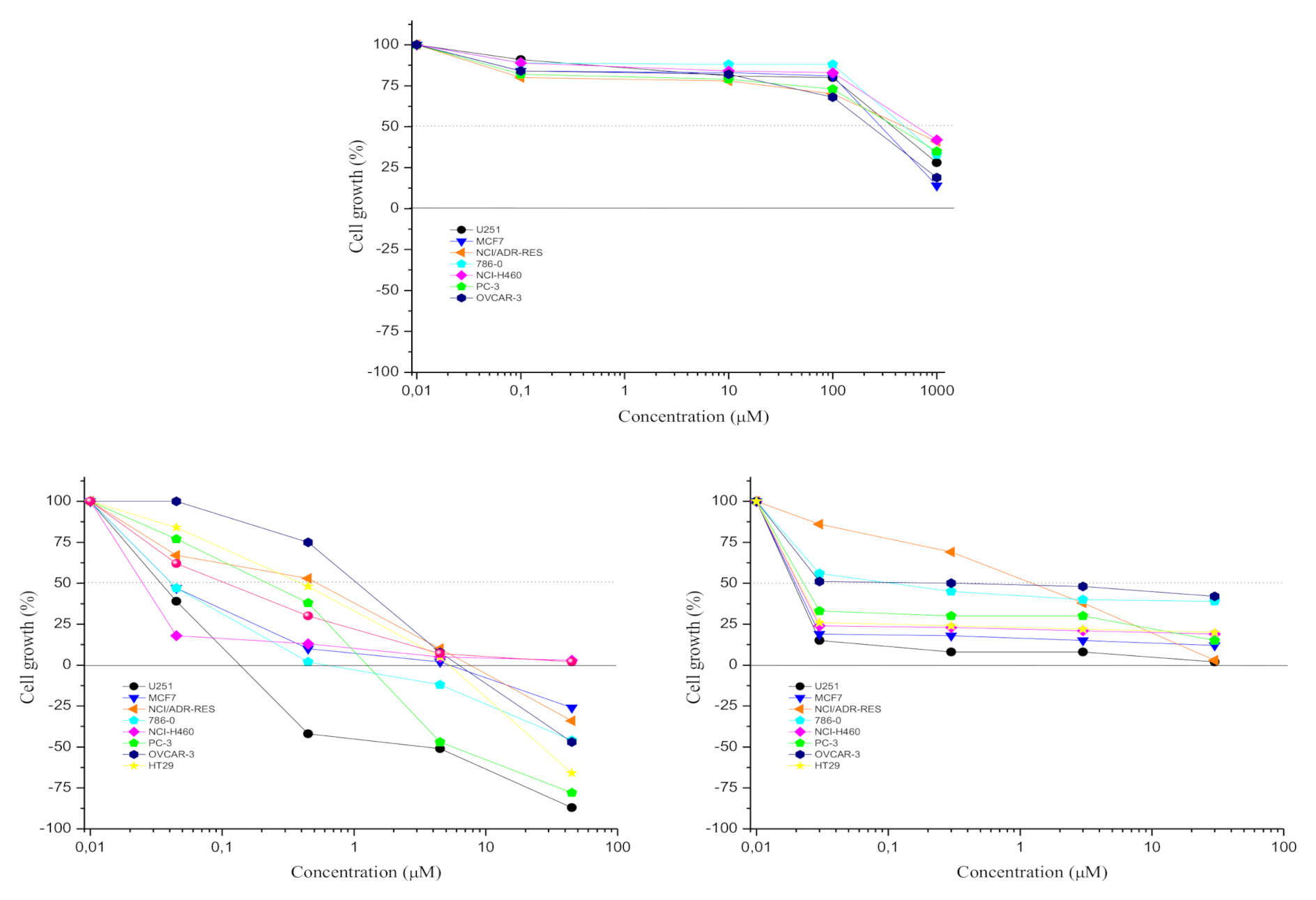
2.2. Antiproliferative Activity of Apiole, Doxorubicin and Vincristine
2.3. Association of Apiole with Doxorubicin on NCI/ADR-RES Cell Line
2.4. Association of Apiole with Vincristine on NCI/ADR-RES Cell Line
2.5. Association of Apiole-Doxorubicin with Apiole-Vincristine on OVCAR-3 Cell Line
2.6. Molecular Docking
2.7. In Silico PK
3. Discussion
4. Material and Methods
4.1. Isolation of Apiole Obtained from Piper Regnellii (Miq.) C. DC. var. Regnellii Leaves
4.2. Cell Culture
4.3. Sample Preparation
4.4. Antiproliferative Activity Evaluation
4.5. Influence of Apiole on the Antiproliferative Activity of Doxorubicin and Vincristine in Human Ovarian Tumor Lines (NCI/ADR-RES and OVCAR-3)
4.6. Molecular Docking
4.7. In Silico Pharmacodynamics
4.8. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Longley, D.B.; Johnston, P.G. Molecular mechanisms of drug resistance. J. Pathol. 2005, 205, 275–292. [Google Scholar] [CrossRef]
- Vert, A.; Castro, J.; Ribó, M.; Vilanova, M.; Benito, A. Transcriptional profiling of NCI/ADR-RES cells unveils a complex network of signaling pathways and molecular mechanisms of drug resistance. Onco Targets Ther. 2018, 11, 221–237. [Google Scholar] [CrossRef]
- Li, W.; Zhang, H.; Assaraf, Y.G.; Zhao, K.; Xu, X.; Xie, J.; Yang, D.H.; Chen, Z.S. Overcoming ABC transporter-mediated multidrug resistance: Molecular mechanisms and novel therapeutic drug strategies. Drug Resist. Updat. 2016, 27, 14–29. [Google Scholar] [CrossRef]
- Dong, J.; Qin, Z.; Zhang, W.D.; Cheng, G.; Yehuda, A.G.; Ashby, C.R.; Chen, Z.S.; Cheng, X.D.; Qin, J.J. Medicinal chemistry strategies to discover P-glycoprotein inhibitors: An update. Drug Resist. Updat. 2020, 49, 100681. [Google Scholar] [CrossRef]
- Robey, R.W.; Pluchino, K.M.; Hall, M.D.; Fojo, A.T.; Bates, S.E.; Gottesman, M.M. Revisiting the role of ABC transporters in multidrug-resistant cancer. Nat. Rev. Cancer 2018, 18, 452–464. [Google Scholar] [CrossRef] [PubMed]
- Sodani, K.; Patel, A.; Kathawala, R.J.; Chen, Z.S. Multidrug resistance associated proteins in multindrung resistance. Chin. J. Cancer 2012, 31, 58–72. [Google Scholar] [CrossRef]
- Callaghan, R.; Luk, F.; Bebawy, M. Special Section on Transporters in Toxicity and Disease—Commentary The Role of Transporters in Toxicity and Disease. Drug Metab. Dispos. 2014, 42, 623–631. [Google Scholar] [CrossRef]
- da Silva, E.M.J. Morfologia e ontogênese das estruturas secretoras em Piper regnellii (Miq.) C. DC. var. regnellii: Piperaceae. Acta Bot. Brasilica 1997, 11, 293. [Google Scholar] [CrossRef][Green Version]
- Prinsloo, G.; Nogemane, N.; Street, R. The use of plants containing genotoxic carcinogens as foods and medicine. Food Chem. Toxicol. 2018, 116, 27–39. [Google Scholar] [CrossRef]
- Lai, Y.Y.; Lien, H.M.; Kuo, P.T.; Huang, C.L.; Kao, J.Y.; Lin, H.; Yang, D.Y. Study of the anti-proliferative activity of 5-substituted 4,7-dimethoxy-1,3-benzodioxole derivatives of sy-1 from Antrodia camphorata on human COLO 205 colon cancer cells. Evidence -Based Complement. Altern. Med. 2011, 2011, 1–8. [Google Scholar]
- Wei, P.L.; Tu, S.H.; Lien, H.M.; Chen, L.C.; Chen, C.S.; Wu, C.H.; Huang, C.-S.; Chang, H.-W.; Chang, C.-H.; Ho, Y.-S.; et al. The in vivo antitumor effects on human COLO 205 cancer cells of the 4,7-dimethoxy-5-(2-propen-1-yl)-1,3-benzodioxole (apiole) derivative of 5-substituted 4,7-dimethoxy-5-methyl-l,3-benzodioxole (SY-1) isolated from the fruiting body of Antrodia camphorate. J. Cancer Res. Ther. 2012, 8, 532–536. [Google Scholar] [PubMed]
- Wu, K.H.; Lee, W.J.; Cheng, T.C.; Chang, H.W.; Chen, L.C.; Chen, C.C.; Lien, H.-M.; Lin, T.-N.; Ho, Y.-S. Study of the antitumor mechanisms of apiole derivatives (AP-02) from Petroselinum crispum through induction of G0/G1 phase cell cycle arrest in human COLO 205 cancer cells. BMC Complement. Altern. Med. 2019, 19, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Neuhaus-Carlisle, K.; Vierling, W.; Wagner, H. Calcium-channel blocking activity of essential oils from Petroselinum crisp., Api graveolens and isolated phenylpropane constituents. Pharm. Pharmacol. Lett. 1993, 3, 77–79. [Google Scholar]
- Neuhaus-Carlisle, K.; Vierling, W.; Wagner, H. Screening of plant extracts and plant constituents for calcium-channel blocking activity. Phytomedicine 1997, 4, 67–71. [Google Scholar] [CrossRef]
- Micucci, M.; Viale, M.; Chiarini, A.; Spinelli, D.; Frosini, M.; Tavani, C.; Maccagno, M.; Bianchi, L.; Gangemi, R.; Budriesi, R. 3-Aryl-4-nitrobenzothiochromans S,S-dioxide: From calcium-channel modulators properties to multidrug-resistance reverting activity. Molecules 2020, 25, 1056. [Google Scholar] [CrossRef]
- Benevides, P.J.C.; Sartorelli, P.; Kato, M.J. Phenylpropanoids and neolignans from Piper regnellii. Phytochemistry 1999, 52, 339–343. [Google Scholar] [CrossRef]
- Kim, S.; Chen, J.; Cheng, T.; Gindulyte, A.; He, J.; He, S.; Li, Q.; Shoemaker, B.A.; Thiessen, P.A.; Yu, B.; et al. PubChem 2019 update: Improved access to chemical data. Nucleic Acids Res. 2019, 47, D1102–D1109. [Google Scholar] [CrossRef]
- Palmeira, A.; Sousa, E.; Vasconcelos, M.H.; Pinto, M.M. Three Decades of P-gp Inhibitors: Skimming Through Several Generations and Scaffolds. Curr. Med. Chem. 2012, 19, 1946–2025. [Google Scholar] [CrossRef] [PubMed]
- Jagodinsky, J.C.; Akgun, U. Characterizing the binding interactions between P-glycoprotein and eight known cardiovascular transport substrates. Pharmacol. Res. Perspect. 2015, 3, 1–11. [Google Scholar] [CrossRef]
- David, M.A.; Orlowski, S.; Prichard, R.K.; Hashem, S.; André, F.; Lespine, A. In silico analysis of the binding of anthelmintics to Caenorhabditis elegans P-glycoprotein 1. Int. J. Parasitol. Drugs Drug Resist. 2016, 6, 299–313. [Google Scholar] [CrossRef]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 2012, 64, 4–17. [Google Scholar] [CrossRef]
- Lepeltier, E.; Rijo, P.; Rizzolio, F.; Popovtzer, R.; Petrikaite, V.; Assaraf, Y.G.; Passirani, C. Nanomedicine to target multidrug resistant tumors. Drug Resist. Updat. 2020, 52, 100704. [Google Scholar] [CrossRef]
- Huber, P.C.; Maruiama, C.H.; Almeida, W.P. Glicoproteína-P, resistência a múltiplas drogas (MDR) e relação estrutura-atividade de moduladores. Quim. Nova 2010, 33, 2148–2154. [Google Scholar] [CrossRef]
- Adovelande, J.; Delèze, J.; Schrével, J. Synergy between two calcium channel blockers, verapamil and fantofarone (SR33557), in reversing chloroquine resistance in Plasmodium falciparum. Biochem. Pharmacol. 1998, 55, 433–440. [Google Scholar] [CrossRef]
- Oneschuk, D.; Younus, J. Natural health products and cancer chemotherapy and radiation therapy. Oncol. Rev. 2008, 1, 233–242. [Google Scholar] [CrossRef]
- Liscovitch, M.; Ravid, D. A case study in misidentification of cancer cell lines: MCF-7/AdrR cells (re-designated NCI/ADR-RES) are derived from OVCAR-8 human ovarian carcinoma cells. Cancer Lett. 2007, 245, 350–352. [Google Scholar] [CrossRef]
- Hansch, C.; Selassie, C. Quantitative Structure-activity relationship—A historical perspective and the future. Compr. Med. Chem. II 2007, 4, 43–63. [Google Scholar]
- Menichetti, R.; Kanekal, K.H.; Bereau, T. Drug-Membrane Permeability across Chemical Space. ACS Cent. Sci. 2019, 5, 290–298. [Google Scholar] [CrossRef]
- Zhou, G.-D.; Moorthy, B.; Bi, J.; Donnelly, K.C.; Randerath, K. DNA Adducts From Alkoxyallylbenzene Herb and Spice Constituents in Cultured Human (HepG2) Cells. Environ. Mol. Mutagen. 2007, 405, 391–405. [Google Scholar] [CrossRef]
- Samet, A.V.; Shevchenko, O.G.; Rusak, V.V.; Chartov, E.M.; Myshlyavtsev, A.B.; Rusanov, D.A.; Semenova, M.N.; Semenov, V.V. Antioxidant Activity of Natural Allylpolyalkoxybenzene Plant Essential Oil Constituents. J. Nat. Prod. 2019, 82, 1451–1458. [Google Scholar] [CrossRef]
- Callaghan, R.; Luk, F.; Bebawy, M. Inhibition of the multidrug resistance P-glycoprotein: Time for a change of strategy? Drug Metab. Dispos. 2014, 42, 623–631. [Google Scholar] [CrossRef]
- Liu, Y.; Lu, Z.; Fan, P.; Duan, Q.; Li, Y.; Tong, S.; Hu, B.; Lv, R.; Hu, L.; Zhuang, J. Clinical Efficacy of Chemotherapy Combined with Verapamil in Metastatic Colorectal Patients. Cell Biochem. Biophys 2011, 61, 393–398. [Google Scholar] [CrossRef]
- Gadhe, C.G.; Cho, S.J. Modulation of Multidrug Resistance in Cancer by P-Glycoprotein Modulation of Multidrug Resistance in Cancer by P-Glycoprotein. J. Chosun. Nat. Sci. 2011, 4, 23–30. [Google Scholar]
- Banzato, T.P.; Gubiani, J.R.; Bernardi, D.I.; Nogueira, C.R.; Monteiro, A.F.; Juliano, F.F.; De Alencar, S.M.; Pilli, R.A.; De Lima, C.A.D.; Longato, G.B.; et al. Antiproliferative Flavanoid Dimers Isolated from Brazilian Red Propolis. J. Nat. Prod. 2020, 83, 1784–1793. [Google Scholar] [CrossRef]
- Franco, Y.E.M.; Okubo, M.Y.; Torre, A.D.; Paiva, P.P.; Rosa, M.N.; Silva, V.A.O.; Reis, R.M.; Ruiz, A.L.T.G.; Imamura, P.M.; de Carvalho, J.E.; et al. Coronarin D induces apoptotic cell death and cell cycle arrest in human glioblastoma cell line. Molecules 2019, 24, 4498. [Google Scholar] [CrossRef]
- Chou, T.C. Drug combination studies and their synergy quantification using the chou-talalay method. Cancer Res. 2010, 70, 440–446. [Google Scholar] [CrossRef]
- ChemInfo. Available online: http://www.cheminfo.org/ (accessed on 10 August 2020).
- Daina, A.; Michielin, O.; Zoete, V. SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef]
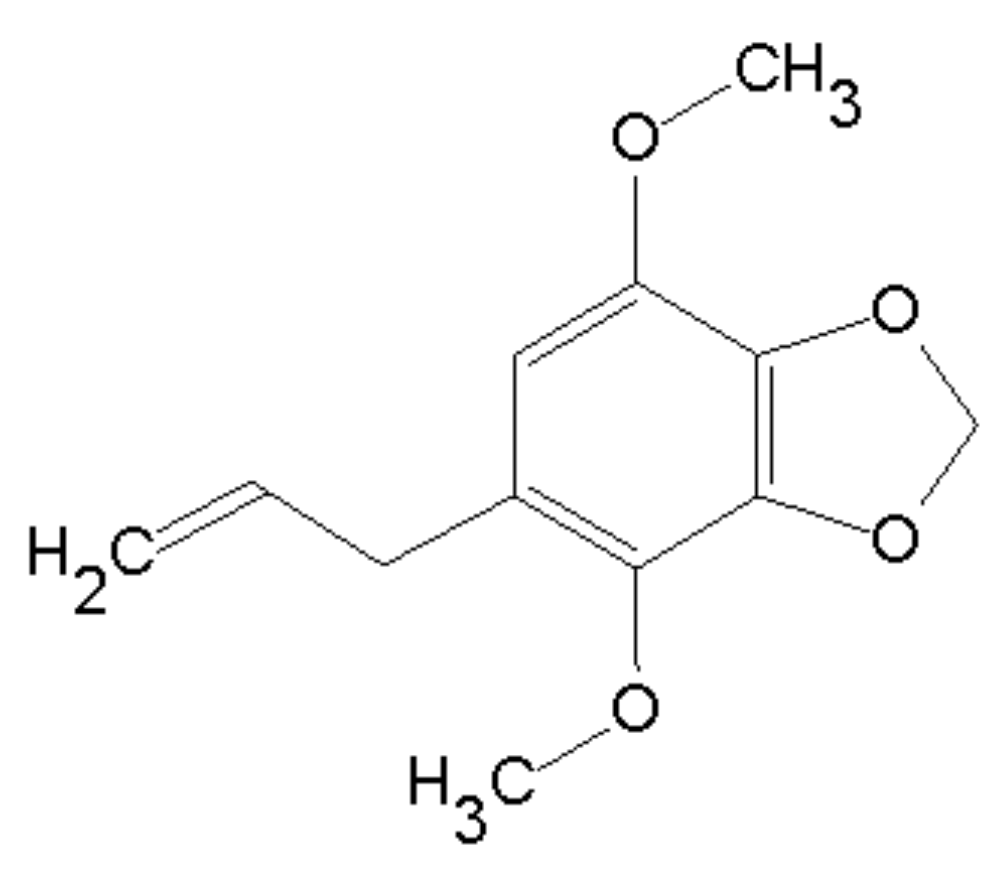



| Carbon | 1H | 13C | HMBC | |
|---|---|---|---|---|
| 1 | CH2 | 3.32 | 34.01 | 5, 4, 6, 7 |
| 2 | CH3 | 3.84 | 56.78 | 3, 10 |
| 3 | CH3 | 3.87 | 60.03 | 2, 10 |
| 4 | CH2 | 5.94 | 101.43 | 5 |
| 5 | CH | 6.30 | 108.17 | 4 |
| 6 | CH2 | 5.01 | 115.27 | |
| 7 | CO | -- | 125.68 | |
| 8 | CO | -- | 135.06 | |
| 9 | CO | -- | 136.19 | |
| 10 | CH | 5.94 | 137.28 | |
| 11 | CO | -- | 138.66 | |
| 12 | CO | -- | 139.00 | |
| Cell Lines | Doxorubicin | Vincristine | Apiole |
|---|---|---|---|
| Central nervous system, glioblastoma (U251) | <0.045 | <0.03 | 357.63 ± 0.25 |
| Breast, adenocarcinoma (MCF7) | <0.045 | <0.03 | 271.69 ± 0.53 |
| Multidrug-resistant ovarian adenocarcinoma (NCI/ADR-RES) | 4.63 ± 0.27 | 4.71 ± 0.27 | 687.63 ± 0.09 |
| Renal adenocarcinoma (786-0) | <0.045 | 0.59 ± 0.10 | 529.87 ± 0.34 |
| Lung, large cell carcinoma (NCI-H460) | 0.04 ± 0.01 | <0.03 | 711.04 ± 0.20 |
| Prostate, adenocarcinoma (PC-3) | 0.20 ± 0.10 | 0.04 ± 0.05 | 423.26 ± 0.14 |
| Ovarian adenocarcinoma (OVCAR-3) | 0.70 ± 0.20 | 1.22 ± 0.10 | 190.62 ± 0.24 |
| Colon, adenocarcinoma (HT29) | 0.37 ± 0.09 | <0.03 | n.t. |
| Apiole | Doxorubicin (Doxo) | Apiole 1 mM + (Doxo) | Apiole 100 µM + (Doxo) | Apiole 10 µM + (Doxo) | |
|---|---|---|---|---|---|
| GI50 b | 436.52 ± 27.38 *** | 4.63 ± 0.61 | <0.045 *** | 0.55 ± 0.28 ** | 5.85 ± 1.60 |
| CRI c | n.a. | n.a. | >102.89 | 8.42 | 0.79 |
| Apiole | Vincristine | Apiole 100 µM + Vincristine | Apiole 50 µM + Vincristine | Apiole 25 µM + Vincristine | |
|---|---|---|---|---|---|
| GI50 b | >100 *** | 4.71 ± 1.14 | 0.75 ± 0.08 ** | 2.70 ± 0.21 * | 4.90 ± 0.77 |
| CRI c | n.a. | n.a. | 6.28 | 1.74 | 0.96 |
| Doxorubicin | Apiole 100 µM + Doxorubicin | Vincristine | Apiole 100 µM + Vincristine | |
|---|---|---|---|---|
| GI50 b | 0.70 ± 0.10 | 0.32 ± 0.11 ** | 1.22 ± 0.19 | 0.43 ± 0.12 ** |
| CRI c | n.a. | 2.2 | n.a. | 2.8 |
| Parameter | Criteria | Apiole |
|---|---|---|
| Molecular weight | <500 g/mol | 222.24 g/mol |
| Hydrogen-bond acceptors (HBA) | <10 | 4 |
| Hydrogen-bond donors (HBD) | <5 | 0 |
| LogP | 2 a 5 | 2.85 |
| Topological polar surface area (TPSA) | 40 a 100 A2 | 36.92 A2 |
| Gastrointestinal absorption | Yes | Yes (high) |
| Blood-brain barrier permeant | No | Yes |
| Metabolism enzymes inhibitors | <2 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Afonso de Lima, C.; de Souza Bueno, I.L.; Nunes Siqueira Vasconcelos, S.; Sciani, J.M.; Ruiz, A.L.T.G.; Foglio, M.A.; Carvalho, J.E.d.; Barbarini Longato, G. Reversal of Ovarian Cancer Cell Lines Multidrug Resistance Phenotype by the Association of Apiole with Chemotherapies. Pharmaceuticals 2020, 13, 327. https://doi.org/10.3390/ph13100327
Afonso de Lima C, de Souza Bueno IL, Nunes Siqueira Vasconcelos S, Sciani JM, Ruiz ALTG, Foglio MA, Carvalho JEd, Barbarini Longato G. Reversal of Ovarian Cancer Cell Lines Multidrug Resistance Phenotype by the Association of Apiole with Chemotherapies. Pharmaceuticals. 2020; 13(10):327. https://doi.org/10.3390/ph13100327
Chicago/Turabian StyleAfonso de Lima, Carolina, Ian Lucas de Souza Bueno, Stanley Nunes Siqueira Vasconcelos, Juliana Mozer Sciani, Ana Lúcia Tasca Gois Ruiz, Mary Ann Foglio, João Ernesto de Carvalho, and Giovanna Barbarini Longato. 2020. "Reversal of Ovarian Cancer Cell Lines Multidrug Resistance Phenotype by the Association of Apiole with Chemotherapies" Pharmaceuticals 13, no. 10: 327. https://doi.org/10.3390/ph13100327
APA StyleAfonso de Lima, C., de Souza Bueno, I. L., Nunes Siqueira Vasconcelos, S., Sciani, J. M., Ruiz, A. L. T. G., Foglio, M. A., Carvalho, J. E. d., & Barbarini Longato, G. (2020). Reversal of Ovarian Cancer Cell Lines Multidrug Resistance Phenotype by the Association of Apiole with Chemotherapies. Pharmaceuticals, 13(10), 327. https://doi.org/10.3390/ph13100327