Abstract
Background: Gemcitabine is a nucleoside analog, widely used either alone or in combination, for the treatment of multiple cancers. However, gemcitabine may also be associated with cardiovascular adverse-drug-reactions (CV-ADR). Methods: First, we searched for all cases of cardiotoxicity associated with gemcitabine, published in MEDLINE on 30 May 2019. Then, we used VigiBase, the World Health Organization’s global database of individual case safety reports, to compare CV-ADR reporting associated with gemcitabine against the full database between inception and 1 April 2019. We used the information component (IC), an indicator value for disproportionate Bayesian reporting. A positive lower end of the 95% credibility interval for the IC (IC025) ≥ 0, is deemed significant. Results: In VigiBase, 46,898 reports were associated with gemcitabine on a total of 18,908,940 in the full database. Gemcitabine was associated with higher reporting for myocardial ischemia (MI, n: 119), pericardial diseases (n: 164), supraventricular arrhythmias (SVA, n: 308) and heart failure (HF, n: 484) versus full database with IC025 ranging between 0.40 and 2.81. CV-ADR were associated with cardiovascular death in up to 17% of cases. Conclusion: Treatment with gemcitabine is associated with potentially lethal CV-ADRs, including MI, pericardial diseases, SVA and HF. These events should be considered in patient care and clinical trial design.
1. Introduction
Gemcitabine is a cytidine analog that incorporates into DNA and terminates chain elongation by inhibiting ribonucleotide reductase and DNA repair [1]. It is still widely used, either alone or in combination to treat various cancers including lung, pancreas, bladder, breast, ovary and bile duct carcinomas, lymphomas and uterine sarcomas [2]. Gemcitabine is generally preferred in elderly or fragile patients due to lower toxicity profile compared to other anticancer drugs. While myelosuppression is the most commonly observed adverse drug reaction (ADR) associated with this molecule, several other ADRs have emerged since gemcitabine was approved by the Food and Drug Administration (FDA) [2], including thrombotic microangiopathy [3], interstitial pneumonitis [4] and capillary leak syndrome (CLS) [5].
According to the FDA label, incidence of cardiovascular ADR (CV-ADR) associated with gemcitabine is low and rarely leads to drug discontinuation [2]. The European Medicines Agency also notifies the particular caution with patients presenting a history of cardiovascular events due to the risk of CV-ADR with gemcitabine [6].
We identified three patients in Saint-Antoine and Pitié-Salpêtrière hospitals (Paris, France) who developed a CV-ADR suspected to be related to gemcitabine. They presented with pancreatic cancer and CV-ADR were pericardial effusion associated with heart failure in two of them, which occurred 6 to 8 months after gemcitabine initiation. Partial or complete recovery was observed after instauration of heart failure therapeutics (see details of in Table 1). One patient presented its pericardial effusion as part of a CLS recovered after glucocorticosteroids administration (Supplementary Figure S1 and Videos S1–S3 present dynamics of his recovery).

Table 1.
Case reports of cardiotoxicity associated with gemcitabine in MEDLINE (thru 30 May 2019).
Herein, we aim to further delineate the overall spectrum of CV-ADR associated with gemcitabine. First, we present three news cases of cardiotoxicity associated with gemcitabine (Table 1) and their management (i), we perform a literature review focusing on the description of CV-ADR reported on gemcitabine in MEDLINE (ii) and (iii) we use VigiBase, the WHO’s international pharmacovigilance database of individual case safety reports (reports, thereafter) to describe the reported CV-ADR cases associated with gemcitabine.
2. Results
2.1. Literature Review
Twenty-three cases reporting gemcitabine-associated CV-ADR were retrieved from literature review including myocardial ischaemia (MI, n: 4), heart failure (HF, n: 10), supraventricular arrhythmias (SVA, n: 6), and pericardial disorders (n: 8), 2 of which were in a context of CLS. Table 1 describes the main characteristics and outcome of these patients. Patients were treated with gemcitabine for pancreatic or lung cancer and lymphoma in respectively, n: 12/23, 52%, n: 5/23, 22%, and n: 5/23, 22%. All patients needed in-hospital management for these CV-ADR and 10/23 (44%) patients did not fully recovered. HF patients presented with altered left ventricular ejection fraction in n: 7/10, 70%; and recovered partially from HF symptoms in n: 6/10, 60%, and completely in n: 4/10, 40% after adequate treatment (i.e., diuretics, betablockers and angiotensin-converting enzyme inhibitors). Three out of 8 patients (38%) with pericardial disorders evolved into constrictive pericarditis. The two patients with pericardial effusion in a context of CLS completely recovered after pericardiocentesis or glucocorticosteroids. Gemcitabine was definitely stopped in 16/23 cases (70%) and 7 patients were rechallenged. Recurrence of CV-ADR after rechallenge occurred in 4/7 cases (57%). Of note, SVA and MI appeared early (i.e., hours/days) after gemcitabine infusion whereas HF occurred later (i.e., weeks/months).
Moreover, among the 106 randomized clinical trials evaluating gemcitabine as monotherapy (overall number of patients = 14015), 17 trials (n = 2386) had reports for at least one CV-ADR in the published work (Supplementary Figure S2 for the flow chart of selected trials). In these latter 17 trials, 33 CV-ADR were reported, leading to an estimate of CV-ADR incidence rate on gemcitabine monotherapy ranging from 0.24% (33/14015) to 1.38% (33/2386), varying with the denominator considered. Quality of CV-ADR reporting in these trials, mostly from decades ago was too low to make sure that all CV-ADR events were effectively captured and reported in the publication. These events were severe in n: 27/33 (82%) (n: 14/33 grade 3/4 and n: 13/33 death), and included 8 MI, 2 pericardial effusions, 7 HF and 1 arrhythmia (Table 2).

Table 2.
Cardiovascular adverse drug reactions in randomized clinical trials evaluating gemcitabine in monotherapy with at least one report for cardiovascular adverse-drug-reactions (CV-ADR).
2.2. Pharmacovigilance Study
The total number of ADR reported with gemcitabine was 46,898 and the total number of ADR in VigiBase was 18,908,940 on 1 April 2019. We identified four broad cardiovascular entities totalizing 973 reports for which reporting was significantly increased with gemcitabine (IC025 > 0) versus full database (Table 3): pericardial diseases (n: 164); MI (n: 119); SVA (n: 308) and HF (n: 484). Sub-classifications and intersections between these CV-ADRs is shown in Supplementary Figure S3. Other CV-ADR including myocarditis, conduction disorders, ventricular arrythmias, long QT, cardiac arrest and valve disorders were not over-reported with gemcitabine (Figure 1). The most overlapping CV-ADRs were HF within pericardial reports (33/164, 20%) vs. HF within SVA and MI reports (36/308, 12%; 5/119, 4%; p < 0.001; respectively). Otherwise, these conditions were moderately overlapping (0–7%, Figure 1, Table 4).

Table 3.
Selected cardiovascular adverse drug reactions (CV-ADRs, detected as signals) reported for gemcitabine versus the full database from VigiBase, on 1 April 2019. A positive IC025 (information component 95% credibility interval lower end) and a reporting odd-ratio (ROR) 95% confidence interval lower-end (95% CI) ≥1 are significant.
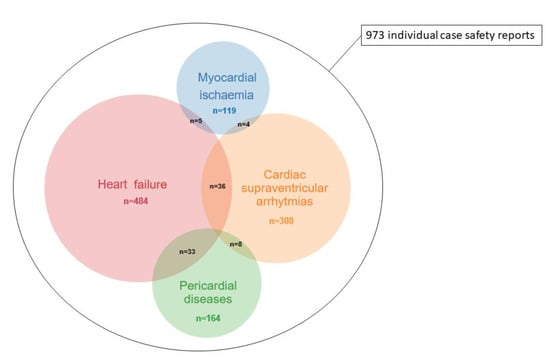
Figure 1.
Individual case safety reports and overlap for gemcitabine-associated cardiovascular adverse drug reactions (CV-ADRs) in VigiBase (accessed on 1 April 2019).

Table 4.
Characteristics of patients with gemcitabine-associated cardiovascular adverse drug reactions (CV-ADRs) in VigiBase on 1 April 2019.
We described the main characteristics of these 973 reports as a function of each type of CV-ADR and concurrent reported conditions in Table 4 and Supplementary Table S1. These cases were declared worldwide mainly from healthcare professionals (90–97%) in post-marketing setting (89–94%), affecting mainly adults (range: 16–89 yo) and increasingly over decades starting in 1997. Indications were mainly for pancreatic (25–39%), and lung (24–36%) cancer. Notably, the third most represented cancer was urothelial for MI (13/93, 14%), bile duct for SVA (21/228, 9%), lymphoma for pericardial diseases (19/97, 20%), and breast cancer for HF reports (36/293, 12%; Table 4, p < 0.0001).
Male were more affected by gemcitabine-associated MI (65/105, 62%), SVA (184/295, 62%), HF (235/451, 52%) while women were over-represented in pericardial reports (95/152, 62%; p < 0.0001). CV-ADR were severe in the majority of cases (76–94%), with cardiovascular related death particularly predominant in MI (20/115, 17%) and HF (83/477, 17%) vs. SVA (17/289, 6%) and pericardial reports (5/144, 3%, p < 0.0001; Supplementary Table S1). Accordingly, when outcome was known, complete recovery was more prevalent in pericardial (48/55, 87%) and SVA (107/132, 81%) vs. MI (35/51, 69%) and HF (115/163, 70%) reports (p:0.02). Median time to onset for HF (75 days, IQR [22, 166]) was longer vs. other CV-ADRs (MI: 29 days, IQR [6, 80]; SVA: 14 days, IQR [4, 52]; pericardial: 55 days, IQR [13, 144], p < 0.0001, Figure 2). Gemcitabine was the only suspected liable drug in patients with pericardial diseases (124/164, 76%) more often than in other CV-ADRs (503/809, 62%; p = 0.001, Table 4). The analysis of co-reported drugs showed that platins were overrepresented in MI reports (55/119, 46%) vs. within other CV-ADRs (209/854, 25%, p = 0.04). Prevalence of other concomitant anticancer drugs intake as a function of type of gemcitabine associated CV-ADRs are presented in Table 4. Combination therapy (gemcitabine and at least one other anticancer drug suspected) was more incriminated than gemcitabine alone in myocardial infarction reports (86/119, 72% vs. 33/119, 28%). A significant association was detected for HER-2 blockers and HF (p = 0.02), taxanes and SVA (p < 0.0001), immune-checkpoint-inhibitors and pericardial disorders (p = 0.001), epidermal growth factor receptor blockers and/or platins and MI (p = 0.001)—see Table 4 for more details.
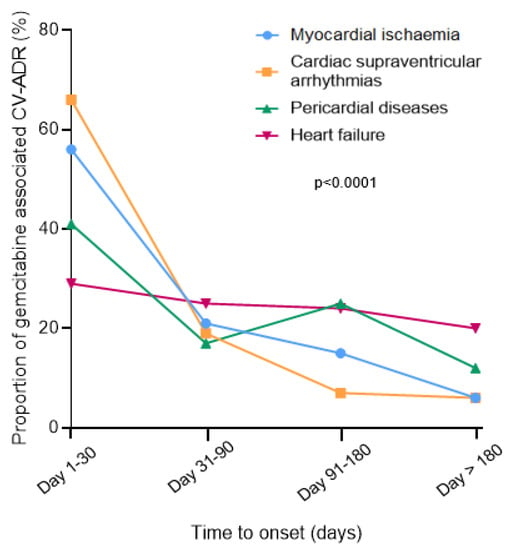
Figure 2.
Time to event onset for gemcitabine-associated cardiovascular adverse drug reactions (CV-ADRs) in VigiBase (accessed on 1 April 2019). The Chi2 test was used to generate the p-value.
Patients with pericardial diseases were younger than those with other CV-ADRs (55.33 ± 15.6 vs. 65.00 ± 10.7 years, p < 0.001). As compared to other CV-ADRs, pericardial diseases were associated with more concurrent CLS (4/164, 2% vs. 5/809, 0.01%; p = 0.03), pleural effusion (56/164, 34% vs. 14/809, 2%; p < 0.0001), ascites (8/164, 5% vs. 7/809, 0.01%; p = 0.001), edema (27/164, 17% vs. 74/809, 9%; p = 0.005) and radiation recall reaction (17/164, 10% vs. 0/809, 0%, p < 0.0001). Lastly, MI patients were associated with more concurrent stroke versus those with other CV-ADRs (10/119, 8% vs. 16/854, 2%; p < 0.0001).
3. Discussion
We report the first large-scale analysis associating specific CV-ADR with gemcitabine. This study of individualized reportable events from the WHO pharmacovigilance database combined with the literature review allowed us to better characterize the CV-ADR associated with gemcitabine, notably the clinical characteristics including time to onset and severity of approximatively 1000 reports with gemcitabine-associated cardiotoxicity, versus few isolated case reports published previously (Table 1).
Gemcitabine was associated with MI, SVA, HF and pericardial diseases. The same cardiotoxicity signals from gemcitabine have also been reported in clinical trials. Aapro et al. pooled 979 patients treated by gemcitabine in 22 phase-2 trials and showed that incidence of MI, HF, arrhythmias and pericarditis were 0.5%, 0.4%, 0.2% and 0.1%, respectively [38]. Our meta-proportion analysis of randomized clinical trials evaluating gemcitabine as monotherapy showed similar incidence estimates of ~1% for overall CV-ADR including MI, SVA, HF and pericardial diseases. We showed that mortality associated with these CV-ADRs ranged from 3% for after pericardial diseases versus 17% for after MI and HF. Severity including death and grade 3/4 events reported in randomized clinical studies (82%) was similar to that of the pharmacovigilance reports (87%). Though, it has to be noted that the quality of CV-ADR adjudication in the trials considered in our analysis was low precluding the possibility of precisely characterizing these CV-ADR. Pericardial diseases had the strongest association with gemcitabine administration (highest IC025, Table 3), and were presented as cardiac tamponade (28/164, 17%) or constrictive pericarditis (6/164, 4%). Noteworthily, pericardial disorders could be part of CLS, a systemic disease determined by vascular protein leakage and diffuse serosa effusions requiring specific management, including glucocorticoids [39,40,41]. Association between gemcitabine and CLS using VigiBase have been described previously [5].
Although CV-ADRs were severe, required hospitalization and often were not fully reversible, cardiac toxicity of gemcitabine is not well known to clinicians. Even in the absence of cardiovascular risk factors, cardiotoxicity related to gemcitabine should be immediately suspected in the case of breathlessness, palpitations, or chest discomfort. Moreover, cardiovascular screening (e.g., echocardiography assessing pericardium and left ventricular ejection fraction, past cardiac history and prior radiation therapy) may identify patients at higher risks of gemcitabine-associated cardiotoxicity but this strategy needs to be evaluated in a dedicated study. Furthermore, in some case-reports (Table 1), gemcitabine re-challenge led to recurrence of the CV-ADR suggesting that decision to restart gemcitabine needs to be weighted between cardiotoxicity and anti-tumor efficacy [42].
Several hypotheses are suggested to explain association of gemcitabine with the various CV-ADRs, but no thorough preclinical mechanistic study is available. Similar to fluoropyrimidines (other antimetabolites), vasospasm has been proposed to be responsible for gemcitabine-associated MI [7,8,9]. Interestingly, MI associated with gemcitabine were often co-reported with strokes in our study, suggesting that shared cardiovascular risk factors and/or a shared pathophysiological mechanisms (i.e., arterial vasospasm) may play a role. For pericarditis, Vogl et al. highlighted for the first time an association between gemcitabine and pericarditis via a radiation recall reaction (acute inflammatory reaction confined to previously irradiated areas triggered when chemotherapy agents are administered) [19]. The recalled inflammation induced by gemcitabine may lead to fluid accumulation in the incompliant pericardial space and ultimately tamponade [43]. Consistently with this hypothesis, we found an association between radiation recall reaction and pericardial diseases. However, prior irradiation was not mandatory for gemcitabine induced pericardial effusion (3 with no prior irradiation among 8 cases with gemcitabine pericarditis, Table 1). For HF, a retrospective study suggested diabetes, coronary artery disease and a total gemcitabine dose >17.000 mg/m2 as risk factors for developing gemcitabine-induced HF but validity of results are limited by a small sample size (7 HF on a total of 156 gemcitabine treated patients) [44]. In VigiBase, we were unable to obtain cumulative doses of gemcitabine but our data showed increased time to onset and increased duration of gemcitabine treatment for HF vs. other CV-ADRs patients, further supporting this cumulative dose effect. Notably, we found that specific concomitant intake of anticancer drugs on top of gemcitabine were more likely to be associated with different type of cardiotoxicities such as platins with MI, HER-2 blockers with HF, and immune checkpoint inhibitors with pericardial disorders. Indeed, these drugs are known to induce these conditions and the hypothesis of toxic synergy or multiple hit mechanisms with these combinations is likely [18,45,46]. Overall, the co-administration of drugs that can induce CV-ADR should strengthen the surveillance of high-risk cardiovascular patients receiving combination of such agents.
We acknowledge several limitations of VigiBase pharmacovigilance analysis, the first of which is under-reporting of suspected CV-ADRs. While the accurate magnitude of underreporting cannot be computed, estimates vary up to 90% of the actual adverse events not being reported [47,48]. However, VigiBase is a worldwide database, gathering data from over 130 countries and with almost 1000 CV-ADR reports related to gemcitabine vs. few case-reports previously described. Sources of reports are non-homogeneous with limited possibility for verification of the clinical, laboratory tests, or radiological findings and re-assessment of the causality of the drug-ADR combination. Thus, in approximately two-third of reports, gemcitabine was the only suspected drug for the reported cardiotoxicity. The exact denominator of patients exposed to gemcitabine cannot be evaluated precluding estimation of incidence of these CV-ADRs using VigiBase. Instead, total number of reports for the studied drug is used as denominator for this kind of analysis [45]. The value of disproportionality analysis for CV-ADR associated with anticancer drugs has already been shown in various settings with confirmation of signals by prospective trials or basic mechanistic studies, such as with myocarditis induced by immune-checkpoint inhibitors, or QT prolongation induced by anti-hormones but nevertheless, there is still a risk that results from pharmacovigilance databases might be misleading [45,49,50]. Yet, these above-described CV-ADRs associated with gemcitabine must warrant caution.
4. Materials and Methods
4.1. Literature Review
We performed a systematic search on PubMed (MEDLINE) using the terms “Cardiotoxicity AND Gemcitabine” with no filter, at the date of 30 May 2019.Then, we screened the bibliography of selected publications. We identified a total of 18 publications including case-reports between 1999 and 2018 focusing on gemcitabine cardiotoxicity. We also performed a systematic search on Pubmed (MEDLINE) for randomized clinical trials using the term “gemcitabine” up to 30 September 2020 and found 958 manuscripts. We then selected articles evaluating gemcitabine monotherapy in at least one of the treatment arms and found 106 studies (Supplementary Figure S2).
4.2. Pharmacovigilance Study
4.2.1. Study Design and Data Sources
This observational, retrospective, pharmacovigilance study is based on ADR reported in deduplicated VigiBase, the WHO’s international database of individual case safety reports (ICSRs, or reports in the text) which includes reports from over 130 countries [51]. These reports originate from different sources, such as healthcare professionals, patients, and pharmaceutical companies, and are generally notified post-marketing. The use of confidential, electronically processed patient data was approved by the French National Commission for Data Protection and Liberties (reference number #1922081). It was not appropriate or possible to involve patients or the public in the design, or conduct, or reporting, or dissemination plans of our research. ClinicalTrials.gov Identifier: NCT03530215
4.2.2. Procedures
This study included all possible CV-ADR according to Preferred Term (PT) levels of the Medical Dictionary for Regulatory Activities (MedDRA; version 21.1, the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH), McLean, VA 22102, USA) within the group query of System Organ Class: cardiac disorders [52] between inception (14 November 1967) through 1 April 2019. Cardiac disorders evaluated included cardiac arrhythmias, neoplasms, valve disorders, congenital disorders, heart failures, coronary artery, myocardial, pericardial and endocardial disorders. CV-ADR specifically assessed in the analysis were those notified as suspected to be induced by gemcitabine. Each report contains general administrative information (country, date, and reporter qualification), patient characteristics (sex and age), drugs (indication, start and end dates), and reactions (onset and end date, seriousness, outcome).
4.2.3. Statistical Analysis
VigiBase allows for disproportionality analysis (also known as case–non-case analysis), which we used to assess whether suspected drug-induced CV-ADR were differentially reported with gemcitabine versus the full database. Disproportionality analysis compares the proportion of selected specific ADR (e.g., pericarditis) reported for a single drug (e.g., gemcitabine) with the proportion of the same ADR for a control group of drugs (e.g., full database). The denominator in these analyses is the total number of ADR reported for each group of drugs. If the proportion of ADR is greater in patients exposed to a specific drug (cases) than in patients not exposed to this drug (non-cases), then an association can be made between the specific drug and the reaction leading to a potential safety concern. Disproportionality in VigiBase is generally calculated using the information component (IC), an indicator value for disproportionate Bayesian reporting when using the full database as comparator [45]. Disproportionality can also be calculated by using a frequentist disproportionality estimate, i.e., the reporting odds ratio (ROR) [49,53].
Calculation of the IC using a Bayesian confidence propagation neural network was developed and validated by the Uppsala Monitoring Centre (organism managing VigiBase) as a flexible, automated indicator value for disproportionate reporting that compares observed and expected drug–ADR associations to find new drug–ADR signals with identification of probability difference from the background data (full database) [54]. Probabilistic reasoning in intelligent systems (information theory) has proved to be effective for the management of large datasets, is robust in handling incomplete data, and can be used with complex variables [54]. Several examples of validation with the IC exist, showing the power of the technique to find signals soon after drug approval by a regulatory agency (e.g., an association between captopril and coughing), and to avoid false positives, whereby an association between a common drug and a common ADR occurs in the database only because the drug is widely used and the ADR is frequently reported (e.g., between digoxin and rash) [54,55].
The statistical formula is as follows:
where
Nexpected is the number of ICSRs expected for the drug–ADR combination. Nobserved is the actual number of ICSRs for the drug–ADR combination. Ndrug is the number of ICSRs for the drug, regardless of ADR. Neffect is the number of ICSRs for the ADR, regardless of drug. Ntotal is the total number of case reports in the database. IC025 is the lower end of a 95% credibility interval for the IC. A positive IC025 value (>0) is the traditional threshold deemed significant [45,54,55]. Characteristics of cases were described in terms of means (standard deviation, SD) or medians (interquartile range, IQR) for quantitative variables, and in terms of numbers and proportion for qualitative ones. Unpaired Student t-tests were performed to compare means of two groups and analysis of variance (ANOVA) or Kruskal–Wallis test was used to compare means or medians of more than two groups, respectively. χ²-tests were performed for comparisons of qualitative variables. p-values < 0.05 were deemed significant.
5. Conclusions
In conclusion, this international global pharmacovigilance study showed that gemcitabine is significantly associated with an over-reporting versus full database of drug-induced myocardial ischemia, pericardial diseases, supraventricular arrhythmias and heart failure with almost one thousand cases and moderate overlap between these conditions (0–20%). These conditions were described rarely in the past literature, with only 20 case-reports. Pericardial effusion associated with gemcitabine were eventually part of a capillary leak syndrome, a systemic condition responsive to glucocorticoids. These cardiovascular adverse events remained rare (incidence ~≤ 1%) and generally occurred within 1–2 months of gemcitabine start, except heart failure adverse events which occurred later and showed less reversibility (30% sequalae or no recovery). Subsequent cardiovascular death occurring in 3–17% of case, emphasizing their seriousness. Hence, cardiotoxicities related to gemcitabine are potentially life-threatening should be investigated in patients care and clinical trials design, particularly in combination with other cardiotoxic drugs.
Supplementary Materials
The following are available online at https://www.mdpi.com/1424-8247/13/10/325/s1, Figure S1: Summary of one of our case-report presenting a capillary leak syndrome associated with gemcitabine reversible after glucocorticoids, Figure S2: Flowchart of selected studies to describe cardiovascular adverse events associated with gemcitabine, Figure S3: UpsetR graph representing overlap between cardiovascular sub-classifications, Table S1: Concurrent adverse events in individual case safety reports of gemcitabine-associated cardiovascular adverse drug reactions (CV-ADRs) in VigiBase, Video S1: Echocardiography at admission, Video S2: Echocardiography at day 5, Video S3: Echocardiography at admission at day 10.
Author Contributions
Conceptualization, J.-E.S., M.H. and S.E.; methodology, J.-E.S. and M.H.; validation, M.H., S.E., X.W., C.F.-B., A.C., A.V., B.L.-V., J.M., L.S.N. and J.-E.S.; formal analysis, M.H.; resources, J.-E.S.; data curation, M.H.; writing—original draft preparation, M.H. and J.-E.S.; writing—review and editing, M.H., L.S.N., J.-E.S., S.E. and J.M.; visualization, M.H. and J.-E.S.; supervision, J.-E.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Acknowledgments
The supplied data from VigiBase come from various sources. The likelihood of a causal relationship is not the same in all reports. The information does not represent the opinion of WHO.
Conflicts of Interest
J.-E.S. has participated to BMS advisory boards. J.M. has Pfizer, Novartis, Bristol-Myers Squibb, Deciphera, Audentes Pharmaceuticals, Nektar, Takeda, Ipsen, Myokardia, AstraZeneca, GlaxoSmithKline, Intrexon, and Regeneron, and is supported by R01 HL141466. Other authors declare no conflicts of interest.
References
- Mini, E.; Nobili, S.; Caciagli, B.; Landini, I.; Mazzei, T. Cellular pharmacology of gemcitabine. Ann. Oncol. 2006, 17 (Suppl. S5), v7–v12. [Google Scholar] [CrossRef]
- Eli Lilly and Company. GEMZAR (Gemcitabine) [package insert]. U.S. Food and Drug Administration Website. Revised May 2014. Available online: www.accessdata.fda.gov/drugsatfda_docs/label/2014/020509s077lbl.pdf (accessed on 28 September 2020).
- Humphreys, B.D.; Sharman, J.P.; Henderson, J.M.; Clark, J.W.; Marks, P.W.; Rennke, H.G.; Zhu, A.X.; Magee, C.C. Gemcitabine-associated thrombotic microangiopathy. Cancer 2004, 100, 2664–2670. [Google Scholar] [CrossRef] [PubMed]
- Roychowdhury, D.F.; Cassidy, C.A.; Peterson, P.; Arning, M. A report on serious pulmonary toxicity associated with gemcitabine-based therapy. Investig. New Drugs 2002, 20, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Mertz, P.; Lebrun-Vignes, B.; Salem, J.E.; Arnaud, L. Characterizing drug-induced capillary leak syndromes using the World Health Organization VigiBase. J. Allergy Clin. Immunol. 2019, 143, 433–436. [Google Scholar] [CrossRef] [PubMed]
- Agency, E.M. Gemzar. Available online: https://www.ema.europa.eu/en/documents/referral/gemzar-article-30-referral-annex-i-ii-iii_en.pdf (accessed on 28 September 2020).
- Ozturk, B.; Tacoy, G.; Coskun, U.; Yaman, E.; Sahin, G.; Buyukberber, S.; Yildiz, R.; Kaya, A.O.; Topal, S.; Ozdemir, M.; et al. Gemcitabine-induced acute coronary syndrome: A case report. Med. Princ. Pract. 2009, 18, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Bdair, F.M.; Graham, S.P.; Smith, P.F.; Javle, M.M. Gemcitabine and acute myocardial infarction—A case report. Angiology 2006, 57, 367–371. [Google Scholar] [CrossRef]
- Kalapura, T.; Krishnamurthy, M.; Reddy, C.V. Acute myocardial infarction following gemcitabine therapy—A case report. Angiology 1999, 50, 1021–1025. [Google Scholar] [CrossRef]
- Santini, D.; Tonini, G.; Abbate, A.; Di Cosimo, S.; Gravante, G.; Vincenzi, B.; Campisi, C.; Patti, G.; Di Sciascio, G. Gemcitabine-induced atrial fibrillation: A hitherto unreported manifestation of drug toxicity. Ann. Oncol. 2000, 11, 479–481. [Google Scholar] [CrossRef]
- Ferrari, D.; Carbone, C.; Codeca, C.; Fumagalli, L.; Gilardi, L.; Marussi, D.; Tartaro, T.; Oldani, S.; Zannier, F.; Foa, P. Gemcitabine and atrial fibrillation: A rare manifestation of chemotherapy toxicity. Anti-Cancer Drugs 2006, 17, 359–361. [Google Scholar] [CrossRef]
- Tavil, Y.; Arslan, U.; Okyay, K.; Sen, N.; Boyaci, B. Atrial fibrillation induced by gemcitabine treatment in a 65-year-old man. Oncol. Res. Treat. 2007, 30, 253–255. [Google Scholar] [CrossRef] [PubMed]
- Ciotti, R.; Belotti, G.; Facchi, E.; Cantu, A.; D’Amico, A.; Gatti, C. Sudden cardio-pulmonary toxicity following a single infusion of gemcitabine. Ann. Oncol. 1999, 10, 997. [Google Scholar] [CrossRef] [PubMed]
- Tayer-Shifman, O.E.; Rottenberg, Y.; Shuvy, M. Gemcitabine-induced supraventricular tachycardia. Tumori J. 2009, 95, 547–549. [Google Scholar] [CrossRef]
- Khan, M.F.; Gottesman, S.; Boyella, R.; Juneman, E. Gemcitabine-induced cardiomyopathy: A case report and review of the literature. J. Med. Case Rep. 2014, 8, 220. [Google Scholar] [CrossRef]
- Yajima, T.; Furukawa, Y.; Ishii, Y.; Hattori, Y.; Matsumoto, N.; Yamamoto, M.; Yamaoka, Y.; Fujihara, M.; Fujita, M.; Kuniki, H. Two cases of advanced pancreatic cancer responding to gemcitabine with long survival of 2 years. Gan Kagaku Ryoho 2004, 31, 953–957. [Google Scholar]
- Alam, S.; Illo, C.; Ma, Y.T.; Punia, P. Gemcitabine-Induced Cardiotoxicity in Patients Receiving Adjuvant Chemotherapy for Pancreatic Cancer: A Case Series. Case Rep. Oncol. 2018, 11, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Mohebali, D.; Matos, J.; Chang, J.D. Gemcitabine induced cardiomyopathy: A case of multiple hit cardiotoxicity. ESC Heart Fail. 2017, 4, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Vogl, D.T.; Glatstein, E.; Carver, J.R.; Schuster, S.J.; Stadtmauer, E.A.; Luger, S.; Nasta, S.D.; Porter, D.L.; Elstrom, R.; Tsai, D.E. Gemcitabine-induced pericardial effusion and tamponade after unblocked cardiac irradiation. Leuk. Lymphoma 2005, 46, 1313–1320. [Google Scholar] [CrossRef]
- Kido, H.; Morizane, C.; Tamura, T.; Hagihara, A.; Kondo, S.; Ueno, H.; Okusaka, T. Gemcitabine-induced pleuropericardial effusion in a patient with pancreatic cancer. Jpn. J. Clin. Oncol. 2012, 42, 845–850. [Google Scholar] [CrossRef][Green Version]
- Konstantinopoulos, P.A.; Cheng, S.C.; Hendrickson, A.E.W.; Penson, R.T.; Schumer, S.T.; Doyle, L.A.; Lee, E.K.; Kohn, E.C.; Duska, L.R.; Crispens, M.A.; et al. Berzosertib plus gemcitabine versus gemcitabine alone in platinum-resistant high-grade serous ovarian cancer: A multicentre, open-label, randomised, phase 2 trial. Lancet Oncol. 2020, 21, 957–968. [Google Scholar] [CrossRef]
- Conroy, T.; Hammel, P.; Hebbar, M.; Ben Abdelghani, M.; Wei, A.C.; Raoul, J.L.; Chone, L.; Francois, E.; Artru, P.; Biagi, J.J.; et al. FOLFIRINOX or Gemcitabine as Adjuvant Therapy for Pancreatic Cancer. N. Engl. J. Med. 2018, 379, 2395–2406. [Google Scholar] [CrossRef]
- Melisi, D.; Garcia-Carbonero, R.; Macarulla, T.; Pezet, D.; Deplanque, G.; Fuchs, M.; Trojan, J.; Oettle, H.; Kozloff, M.; Cleverly, A.; et al. Galunisertib plus gemcitabine vs. gemcitabine for first-line treatment of patients with unresectable pancreatic cancer. Br. J. Cancer 2018, 119, 1208–1214. [Google Scholar] [CrossRef]
- Middleton, G.; Palmer, D.H.; Greenhalf, W.; Ghaneh, P.; Jackson, R.; Cox, T.; Evans, A.; Shaw, V.E.; Wadsley, J.; Valle, J.W.; et al. Vandetanib plus gemcitabine versus placebo plus gemcitabine in locally advanced or metastatic pancreatic carcinoma (ViP): A prospective, randomised, double-blind, multicentre phase 2 trial. Lancet Oncol. 2017, 18, 486–499. [Google Scholar] [CrossRef]
- Neoptolemos, J.P.; Palmer, D.H.; Ghaneh, P.; Psarelli, E.E.; Valle, J.W.; Halloran, C.M.; Faluyi, O.; O’Reilly, D.A.; Cunningham, D.; Wadsley, J.; et al. Comparison of adjuvant gemcitabine and capecitabine with gemcitabine monotherapy in patients with resected pancreatic cancer (ESPAC-4): A multicentre, open-label, randomised, phase 3 trial. Lancet 2017, 389, 1011–1024. [Google Scholar] [CrossRef]
- Evans, T.R.J.; Van Cutsem, E.; Moore, M.J.; Bazin, I.S.; Rosemurgy, A.; Bodoky, G.; Deplanque, G.; Harrison, M.; Melichar, B.; Pezet, D.; et al. Phase 2 placebo-controlled, double-blind trial of dasatinib added to gemcitabine for patients with locally-advanced pancreatic cancer. Ann. Oncol. 2017, 28, 354–361. [Google Scholar] [CrossRef]
- Rougier, P.; Riess, H.; Manges, R.; Karasek, P.; Humblet, Y.; Barone, C.; Santoro, A.; Assadourian, S.; Hatteville, L.; Philip, P.A. Randomised, placebo-controlled, double-blind, parallel-group phase III study evaluating aflibercept in patients receiving first-line treatment with gemcitabine for metastatic pancreatic cancer. Eur. J. Cancer 2013, 49, 2633–2642. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, A.; Gilabert, M.; François, E.; Dahan, L.; Perrier, H.; Lamy, R.; Re, D.; Largillier, R.; Gasmi, M.; Tchiknavorian, X.; et al. BAYPAN study: A double-blind phase III randomized trial comparing gemcitabine plus sorafenib and gemcitabine plus placebo in patients with advanced pancreatic cancer. Ann. Oncol. 2012, 23, 2799–2805. [Google Scholar] [CrossRef]
- Loehrer, P.J., Sr.; Feng, Y.; Cardenes, H.; Wagner, L.; Brell, J.M.; Cella, D.; Flynn, P.; Ramanathan, R.K.; Crane, C.H.; Alberts, S.R.; et al. Gemcitabine alone versus gemcitabine plus radiotherapy in patients with locally advanced pancreatic cancer: An Eastern Cooperative Oncology Group trial. J. Clin. Oncol. 2011, 29, 4105–4112. [Google Scholar] [CrossRef]
- Colucci, G.; Labianca, R.; Di Costanzo, F.; Gebbia, V.; Cartenì, G.; Massidda, B.; Dapretto, E.; Manzione, L.; Piazza, E.; Sannicolò, M.; et al. Randomized phase III trial of gemcitabine plus cisplatin compared with single-agent gemcitabine as first-line treatment of patients with advanced pancreatic cancer: The GIP-1 study. J. Clin. Oncol. 2010, 28, 1645–1651. [Google Scholar] [CrossRef]
- Richards, D.A.; Kuefler, P.R.; Becerra, C.; Wilfong, L.S.; Gersh, R.H.; Boehm, K.A.; Zhan, F.; Asmar, L.; Myrand, S.P.; Hozak, R.R.; et al. Gemcitabine plus enzastaurin or single-agent gemcitabine in locally advanced or metastatic pancreatic cancer: Results of a phase II, randomized, noncomparative study. Investig. New Drugs 2011, 29, 144–153. [Google Scholar] [CrossRef]
- Spano, J.P.; Chodkiewicz, C.; Maurel, J.; Wong, R.; Wasan, H.; Barone, C.; Létourneau, R.; Bajetta, E.; Pithavala, Y.; Bycott, P.; et al. Efficacy of gemcitabine plus axitinib compared with gemcitabine alone in patients with advanced pancreatic cancer: An open-label randomised phase II study. Lancet 2008, 371, 2101–2108. [Google Scholar] [CrossRef]
- Herrmann, R.; Bodoky, G.; Ruhstaller, T.; Glimelius, B.; Bajetta, E.; Schüller, J.; Saletti, P.; Bauer, J.; Figer, A.; Pestalozzi, B.; et al. Gemcitabine plus capecitabine compared with gemcitabine alone in advanced pancreatic cancer: A randomized, multicenter, phase III trial of the Swiss Group for Clinical Cancer Research and the Central European Cooperative Oncology Group. J. Clin. Oncol. 2007, 25, 2212–2217. [Google Scholar] [CrossRef] [PubMed]
- Van Cutsem, E.; van de Velde, H.; Karasek, P.; Oettle, H.; Vervenne, W.L.; Szawlowski, A.; Schoffski, P.; Post, S.; Verslype, C.; Neumann, H.; et al. Phase III trial of gemcitabine plus tipifarnib compared with gemcitabine plus placebo in advanced pancreatic cancer. J. Clin. Oncol. 2004, 22, 1430–1438. [Google Scholar] [CrossRef] [PubMed]
- Sederholm, C. Gemcitabine versus gemcitabine/carboplatin in advanced non-small cell lung cancer: Preliminary findings in a phase III trial of the Swedish Lung Cancer Study Group. Semin. Oncol. 2002, 29, 50–54. [Google Scholar] [CrossRef] [PubMed]
- Cappuzzo, F.; Novello, S.; De Marinis, F.; Selvaggi, G.; Scagliotti, G.V.; Barbieri, F.; Maur, M.; Papi, M.; Pasquini, E.; Bartolini, S.; et al. A randomized phase II trial evaluating standard (50 mg/min) versus low (10 mg/min) infusion duration of gemcitabine as first-line treatment in advanced non-small-cell lung cancer patients who are not eligible for platinum-based chemotherapy. Lung Cancer 2006, 52, 319–325. [Google Scholar] [CrossRef]
- Sederholm, C.; Hillerdal, G.; Lamberg, K.; Kölbeck, K.; Dufmats, M.; Westberg, R.; Gawande, S.R. Phase III trial of gemcitabine plus carboplatin versus single-agent gemcitabine in the treatment of locally advanced or metastatic non-small-cell lung cancer: The Swedish Lung Cancer Study Group. J. Clin. Oncol. 2005, 23, 8380–8388. [Google Scholar] [CrossRef]
- Aapro, M.S.; Martin, C.; Hatty, S. Gemcitabine—A safety review. Anti-Cancer Drugs 1998, 9, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Semb, K.A.; Aamdal, S.; Oian, P. Capillary protein leak syndrome appears to explain fluid retention in cancer patients who receive docetaxel treatment. J. Clin. Oncol. 1998, 16, 3426–3432. [Google Scholar] [CrossRef]
- Piccart, M.J.; Klijn, J.; Paridaens, R.; Nooij, M.; Mauriac, L.; Coleman, R.; Bontenbal, M.; Awada, A.; Selleslags, J.; Van Vreckem, A.; et al. Corticosteroids significantly delay the onset of docetaxel-induced fluid retention: Final results of a randomized study of the European Organization for Research and Treatment of Cancer Investigational Drug Branch for Breast Cancer. J. Clin. Oncol. 1997, 15, 3149–3155. [Google Scholar] [CrossRef]
- Siddall, E.; Khatri, M.; Radhakrishnan, J. Capillary leak syndrome: Etiologies, pathophysiology, and management. Kidney Int. 2017, 92, 37–46. [Google Scholar] [CrossRef]
- Accordino, M.K.; Neugut, A.I.; Hershman, D.L. Cardiac effects of anticancer therapy in the elderly. J. Clin. Oncol. 2014, 32, 2654–2661. [Google Scholar] [CrossRef]
- Burris, H.A., 3rd; Hurtig, J. Radiation recall with anticancer agents. Oncologist 2010, 15, 1227–1237. [Google Scholar] [CrossRef] [PubMed]
- Shrum, K.J.; Gill, S.E.; Thompson, L.K.; Blackhurst, D.W.; Puls, L.E. New-onset congestive heart failure with gemcitabine in ovarian and other solid cancers. Am. J. Clin. Oncol. 2014, 37, 364–368. [Google Scholar] [CrossRef] [PubMed]
- Salem, J.E.; Manouchehri, A.; Moey, M.; Lebrun-Vignes, B.; Bastarache, L.; Pariente, A.; Gobert, A.; Spano, J.P.; Balko, J.M.; Bonaca, M.P.; et al. Cardiovascular toxicities associated with immune checkpoint inhibitors: An observational, retrospective, pharmacovigilance study. Lancet Oncol. 2018, 19, 1579–1589. [Google Scholar] [CrossRef]
- Zamorano, J.L.; Lancellotti, P.; Munoz, D.R.; Aboyans, V.; Asteggiano, R.; Galderisi, M.; Habib, G.; Lenihan, D.J.; Lip, G.Y.H.; Lyon, A.R.; et al. 2016 ESC Position Paper on cancer treatments and cardiovascular toxicity developed under the auspices of the ESC Committee for Practice Guidelines: The Task Force for cancer treatments and cardiovascular toxicity of the European Society of Cardiology (ESC). Eur. Heart J. 2016, 37, 2768–2801. [Google Scholar] [CrossRef] [PubMed]
- Biagi, C.; Montanaro, N.; Buccellato, E.; Roberto, G.; Vaccheri, A.; Motola, D. Underreporting in pharmacovigilance: An intervention for Italian GPs (Emilia-Romagna region). Eur. J. Clin. Pharmacol. 2013, 69, 237–244. [Google Scholar] [CrossRef]
- Tandon, V.R.; Mahajan, V.; Khajuria, V.; Gillani, Z. Under-reporting of adverse drug reactions: A challenge for pharmacovigilance in India. Indian J. Pharmacol. 2015, 47, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Grouthier, V.; Lebrun-Vignes, B.; Glazer, A.M.; Touraine, P.; Funck-Brentano, C.; Pariente, A.; Courtillot, C.; Bachelot, A.; Roden, D.M.; Moslehi, J.J.; et al. Increased long QT and torsade de pointes reporting on tamoxifen compared with aromatase inhibitors. Heart 2018, 104, 1859–1863. [Google Scholar] [CrossRef]
- Salem, J.E.; Yang, T.; Moslehi, J.J.; Waintraub, X.; Gandjbakhch, E.; Bachelot, A.; Hidden-Lucet, F.; Hulot, J.S.; Knollmann, B.C.; Lebrun-Vignes, B.; et al. Androgenic Effects on Ventricular Repolarization: A Translational Study From the International Pharmacovigilance Database to iPSC-Cardiomyocytes. Circulation 2019, 140, 1070–1080. [Google Scholar] [CrossRef]
- Lindquist, M. VigiBase, the WHO Global ICSR Database System: Basic Facts. Drug Inf. J. 2008, 42, 409–419. [Google Scholar] [CrossRef]
- Chang, L.C.; Mahmood, R.; Qureshi, S.; Breder, C.D. Patterns of use and impact of standardised MedDRA query analyses on the safety evaluation and review of new drug and biologics license applications. PLoS ONE 2017, 12, e0178104. [Google Scholar] [CrossRef]
- Rothman, K.J.; Lanes, S.; Sacks, S.T. The reporting odds ratio and its advantages over the proportional reporting ratio. Pharmacoepidemiol. Drug Saf. 2004, 13, 519–523. [Google Scholar] [CrossRef] [PubMed]
- Bate, A.; Lindquist, M.; Edwards, I.R.; Olsson, S.; Orre, R.; Lansner, A.; De Freitas, R.M. A Bayesian neural network method for adverse drug reaction signal generation. Eur. J. Clin. Pharmacol. 1998, 54, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Noren, G.N.; Hopstadius, J.; Bate, A. Shrinkage observed-to-expected ratios for robust and transparent large-scale pattern discovery. Stat. Methods Med. Res. 2013, 22, 57–69. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).