Biological Applications of Thiocarbohydrazones and Their Metal Complexes: A Perspective Review
Abstract
1. Introduction
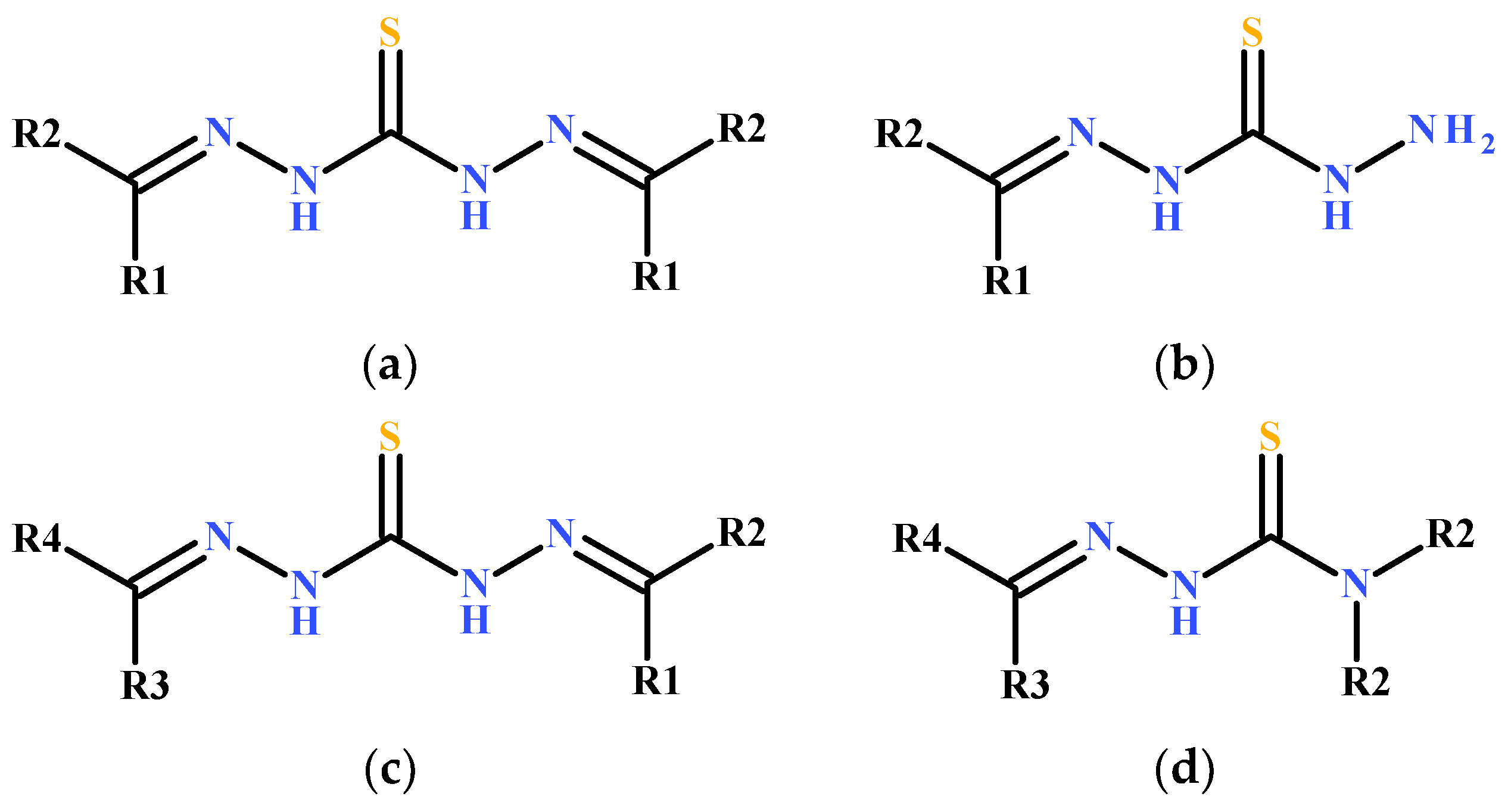
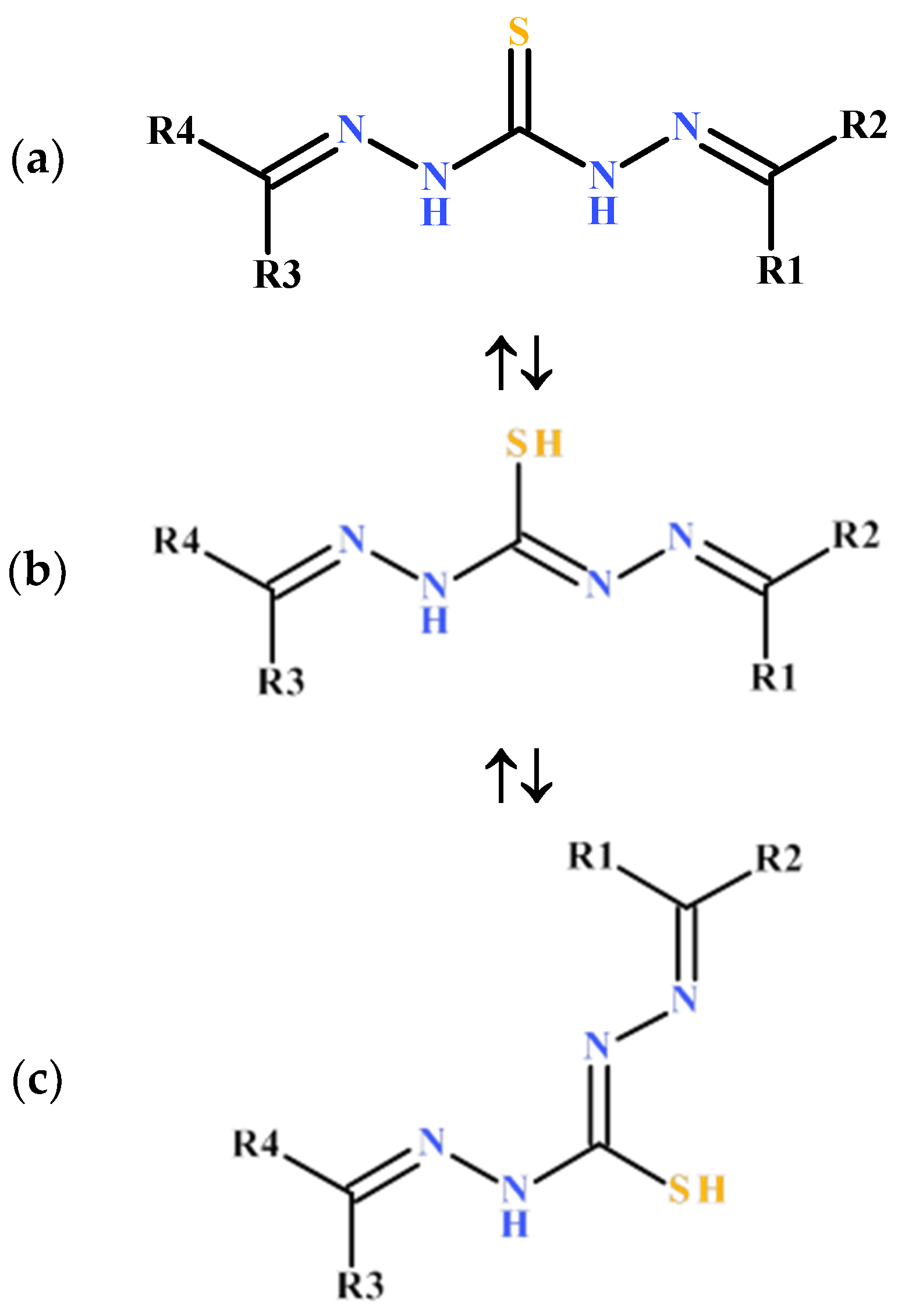
2. Structures of Thiocarbohydrazones
3. Symmetric 1,5-Bisthiocarbohydrazones
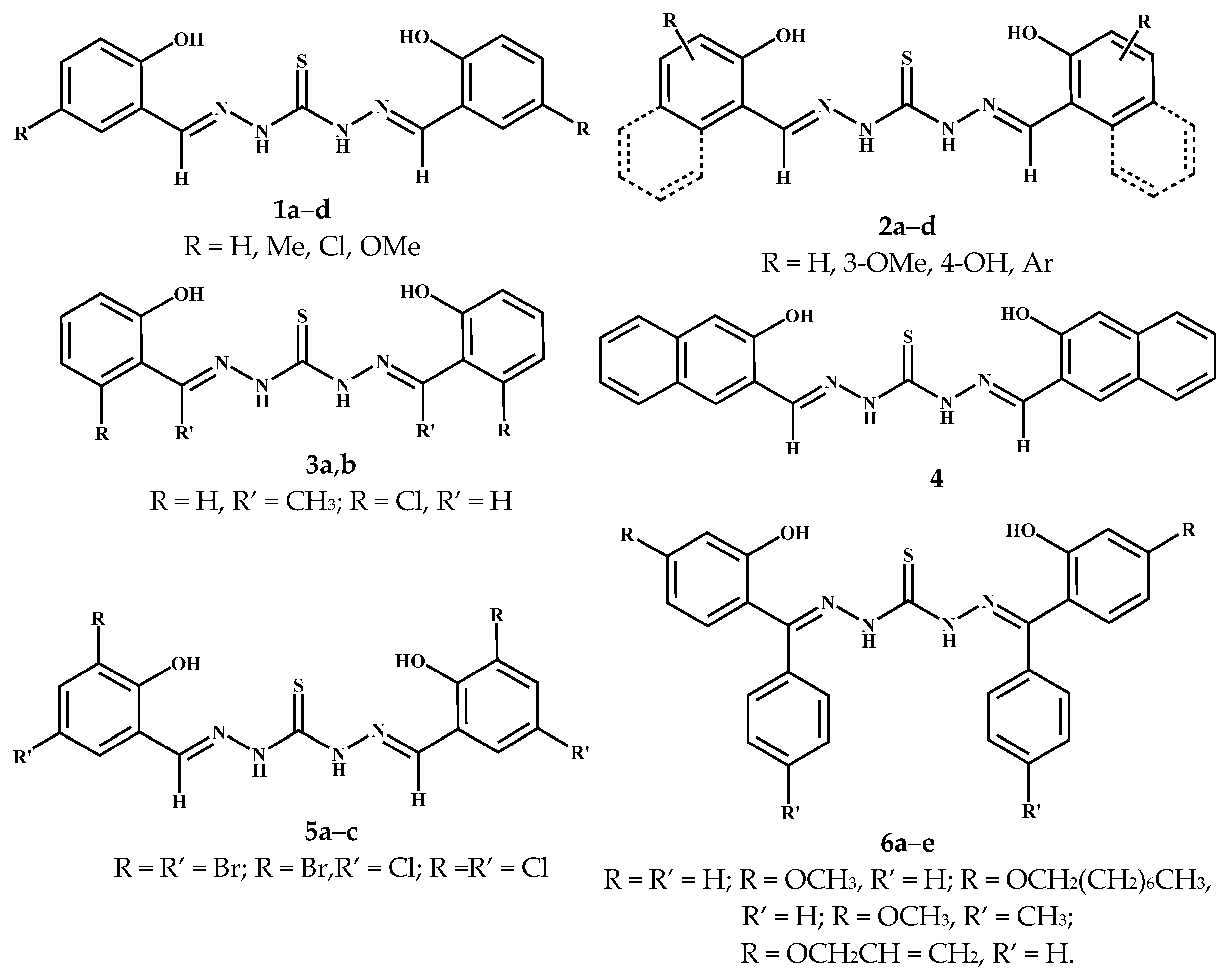
3.1. Symmetric Thiocarbohydrazones Derived from Salycilaldehyde and Related Carbonyl Compounds
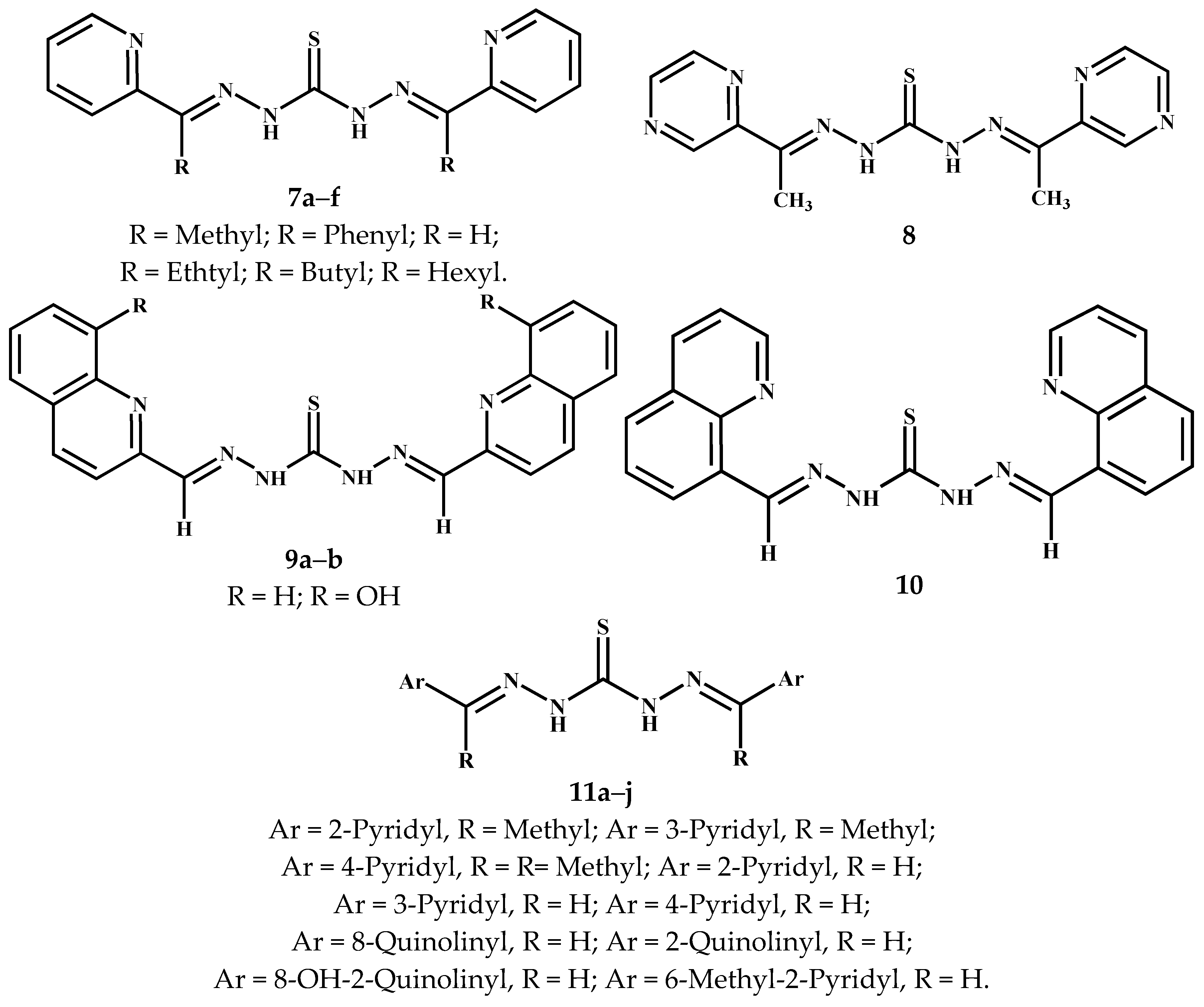
3.2. Symmetric Thiocarbohydrazones Derived from Pyridyl and Related Carbonyl Compounds
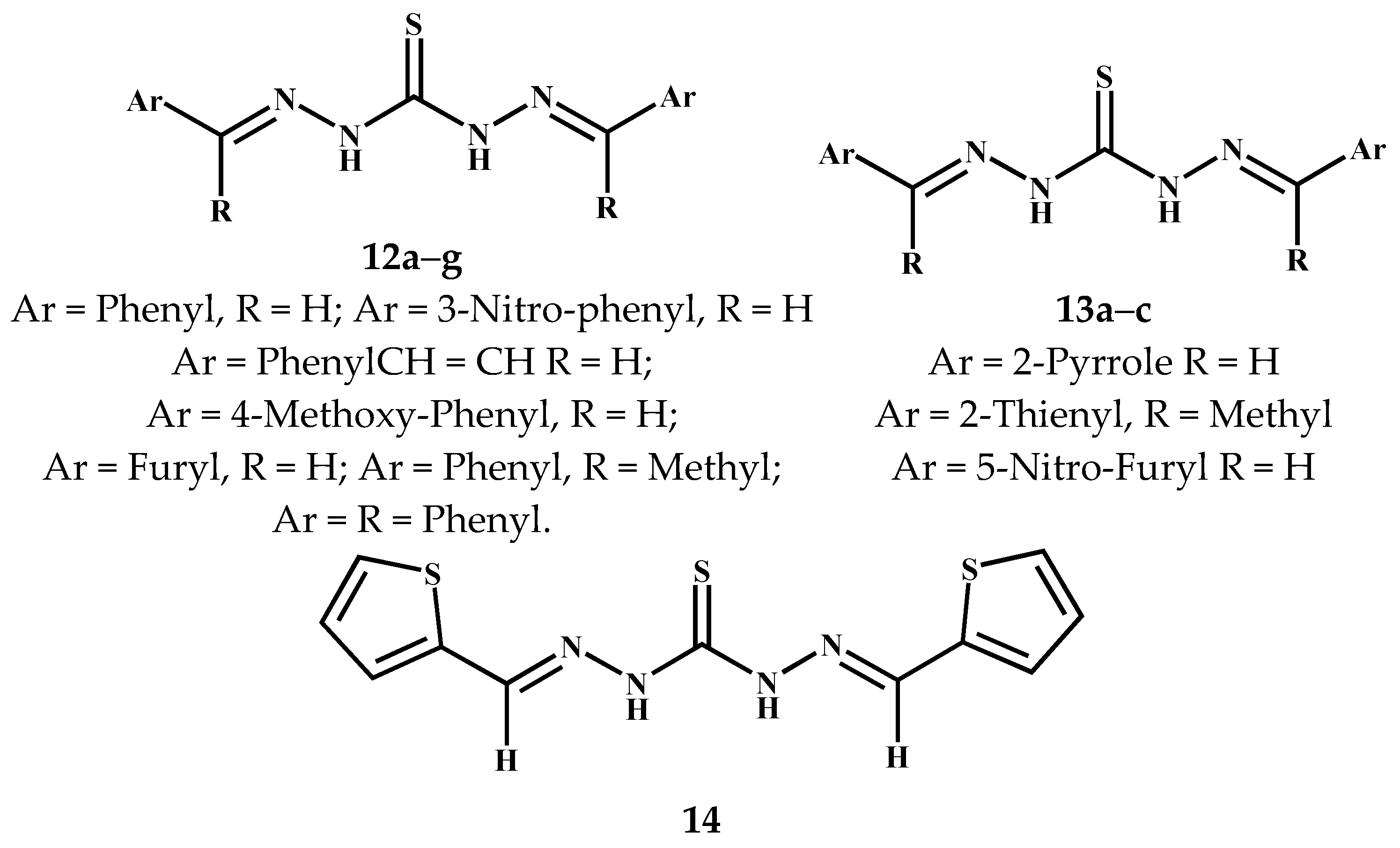
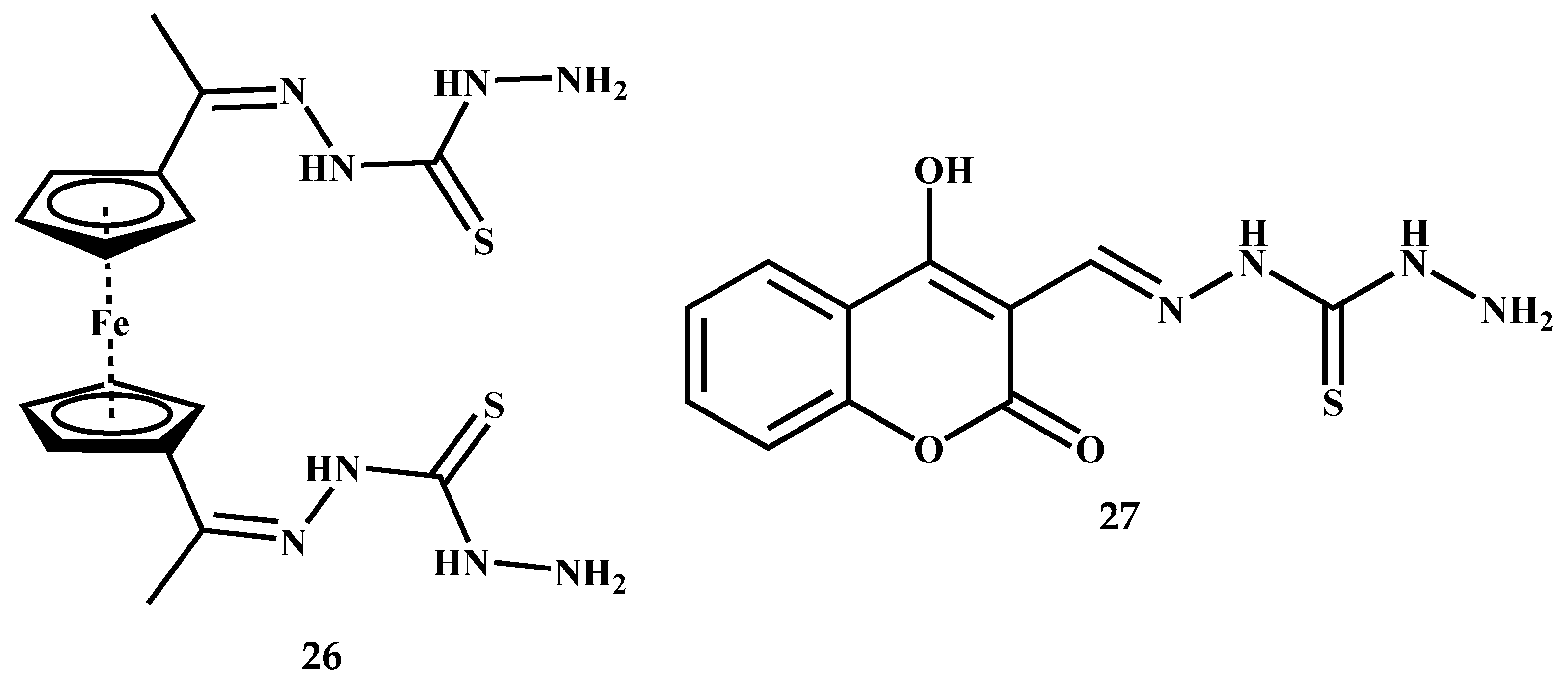
3.3. Symmetric Thiocarbohydrazones Derived from Aromatics/Heteroaromatics and Related Carbonyl Compounds
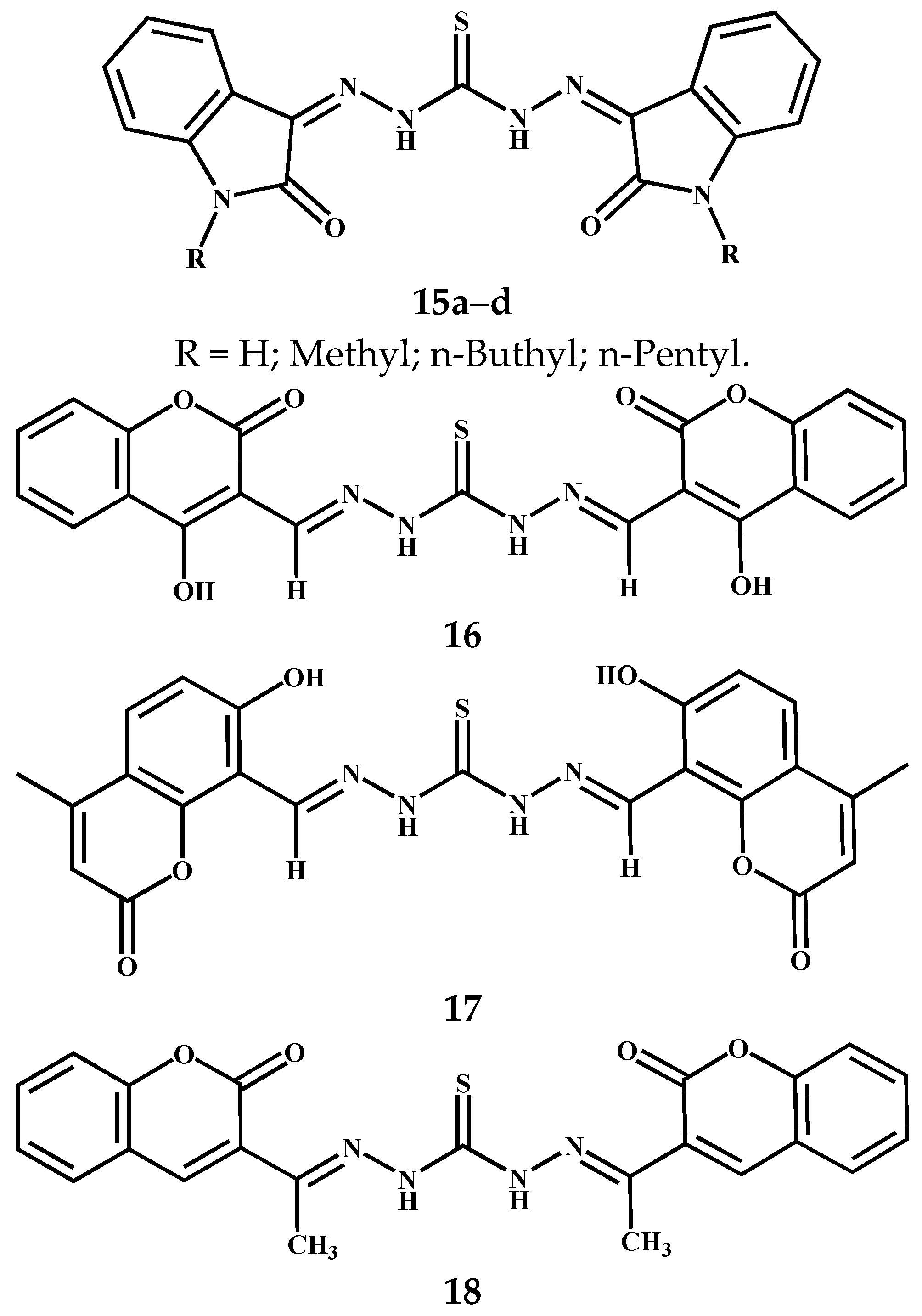
3.4. Symmetric Thiocarbohydrazones Derived from Isatin, Coumarin and Related Compounds
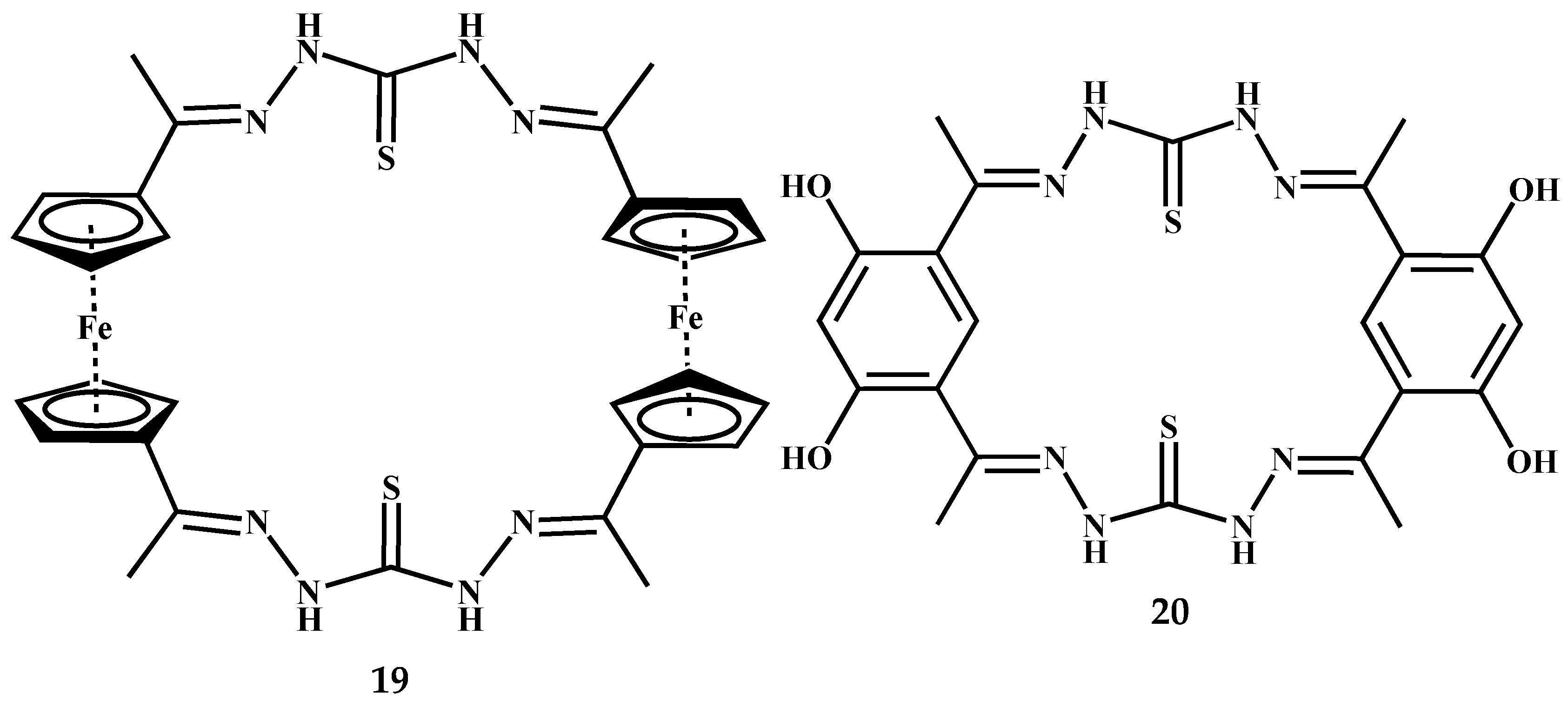
4. Macrocyclic Thiocarbohydrazones
5. Monothiocarbohydrazones and Asymmetric 1,5-Bisthiocarbohydrazones
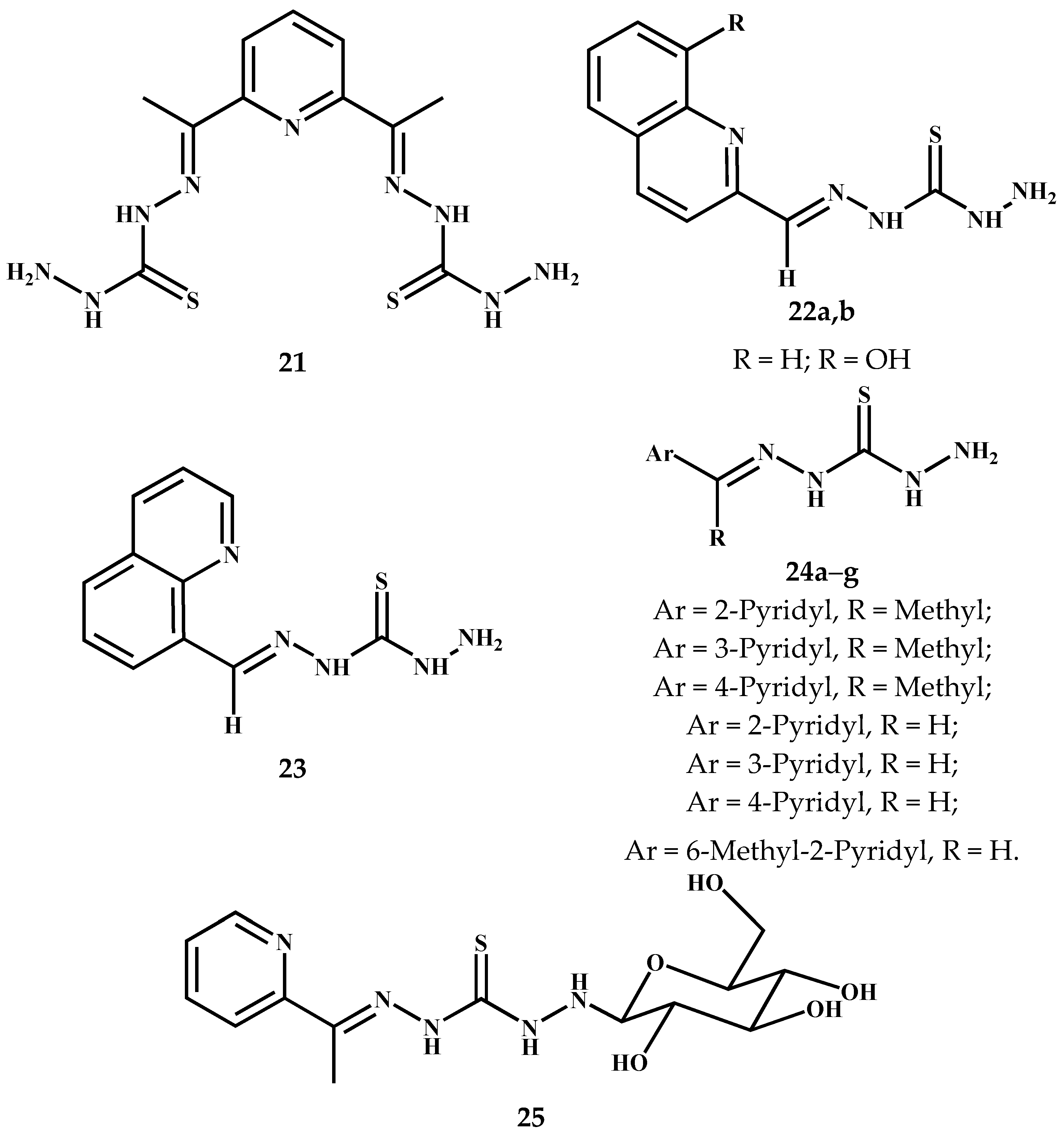
5.1. Asymmetric Thiocarbohydrazones Derived from Pyridyl and Related Compounds
5.2. Asymmetric Thiocarbohydrazones Derived from Aromatics/Heteroaromatics, Isatin, Coumarin and Related Carbonyl Compounds
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Silvestry-Rodriguez, N.; Sicairos-Ruelas, E.E.; Gerba, C.P.; Bright, K.R. Silver as A Disinfectant; Springer: New York, NY, USA, 2007; pp. 23–45. [Google Scholar]
- Pricker, S.P. Medical uses of gold compounds: Past, present and future. Gold Bull. 1996, 29, 53–60. [Google Scholar] [CrossRef]
- Database of Privately and Publicly Funded Clinical Studies Conducted around the World. Available online: https://clinicaltrials.gov/ (accessed on 10 November 2019).
- Marzo, T.; Massai, L.; Pratesi, A.; Stefanini, M.; Cirri, D.; Magherini, F.; Becatti, M.; Landini, I.; Nobili, S.; Mini, E.; et al. Replacement of the Thiosugar of Auranofin with Iodide Enhances the Anticancer Potency in a Mouse Model of Ovarian Cancer. ACS Med. Chem. Lett. 2019, 10, 656–660. [Google Scholar] [CrossRef] [PubMed]
- Marzo, T.; Cirri, D.; Pollini, S.; Prato, M.; Fallani, S.; Cassetta, M.I.; Novelli, A.; Rossolini, G.M.; Messori, L. Auranofin and its Analogues Show Potent Antimicrobial Activity against Multidrug-Resistant Pathogens: Structure-Activity Relationships. CHEMMEDCHEM 2018, 13, 2448–2454. [Google Scholar] [CrossRef] [PubMed]
- List, A.; Beran, M.; DiPersio, J.; Slack, J.; Vey, N.; Rosenfeld, C.S.; Greenberg, P. Opportunities for Trisenox® (arsenic trioxide) in the treatment of myelodysplastic syndromes. Leukemia 2003, 17, 1499–1507. [Google Scholar] [CrossRef]
- Lippard, S.J. Metals in Medicine; University Science Books: Mill Valley, CA, USA, 1994. [Google Scholar]
- Niccoli Asabella, A.; Cascini, G.L.; Altini, C.; Paparella, D.; Notaristefano, A.; Rubini, G. The copper radioisotopes: A systematic review with special interest to 64Cu. BioMed Res. Int. 2014, 2014. [Google Scholar] [CrossRef]
- Rosenberg, B.; Van Camp, L.; Krigas, T. Inhibition of Cell Division in Escherichia coli by Electrolysis Products from a Platinum Electrode. Nature 1965, 205, 698–699. [Google Scholar] [CrossRef]
- Barry, N.P.E.; Sadler, P.J. Exploration of the medical periodic table: Towards new targets. Chem. Commun. 2013, 49, 5106. [Google Scholar] [CrossRef]
- Giorgio, P. Thiosemicarbazone Metal Complexes: From Structure to Activity. Open Crystallogr. J. 2008, 3, 16–28. [Google Scholar]
- Donnelly, P.S.; Caragounis, A.; Du, T.; Laughton, K.M.; Volitakis, I.; Cherny, R.A.; Sharples, R.A.; Hill, A.F.; Li, Q.-X.; Masters, C.L.; et al. Selective Intracellular Release of Copper and Zinc Ions from Bis(thiosemicarbazonato) Complexes Reduces Levels of Alzheimer Disease Amyloid-β Peptide. J. Biol. Chem. 2008, 283, 4568–4577. [Google Scholar] [CrossRef]
- Paterson, B.M.; Donnelly, P.S. Copper complexes of bis(thiosemicarbazones): From chemotherapeutics to diagnostic and therapeutic radiopharmaceuticals. Chem. Soc. Rev. 2011, 40, 3005–3018. [Google Scholar] [CrossRef]
- Jyothi, N.R.; Farook, N.A.M.; Cho, M.; Shim, J. Cytotoxic activities of thiosemicarbazones and their metal complexes. Asian J. Chem. 2013, 25, 5841–5843. [Google Scholar] [CrossRef]
- Shim, J.; Jyothi, N.R.; Farook, N.A.M. Biological applications of thiosemicarbazones and their metal complexes. Asian J. Chem. 2013, 25, 5838–5840. [Google Scholar] [CrossRef]
- Moorthy, N.S.H.N.; Cerqueira, N.M.F.S.A.; Ramos, M.J.; Fernandes, P.A. Aryl- and heteroaryl-thiosemicarbazone derivatives and their metal complexes: A pharmacological template. Recent Pat. Anti-Cancer Drug Discov. 2013, 8, 168–182. [Google Scholar] [CrossRef] [PubMed]
- Jansson, P.J.; Kalinowski, D.S.; Lane, D.J.R.; Kovacevic, Z.; Seebacher, N.A.; Fouani, L.; Sahni, S.; Merlot, A.M.; Richardson, D.R. The renaissance of polypharmacology in the development of anti-cancer therapeutics: Inhibition of the “Triad of Death” in cancer by Di-2-pyridylketone thiosemicarbazones. Pharmacol. Res. 2015, 100, 255–260. [Google Scholar] [CrossRef] [PubMed]
- Silva, B.V.; Silva, B.N.M. Thio- and Semicarbazones: Hope in the Search for Treatment of Leishmaniasis and Chagas Disease. Med. Chem. 2017, 13, 110–126. [Google Scholar] [CrossRef]
- Prajapati, N.P.; Patel, H.D. Novel thiosemicarbazone derivatives and their metal complexes: Recent development. Synth. Commun. 2019, 49, 2767–2804. [Google Scholar] [CrossRef]
- Namiecinska, E.; Sobiesiak, M.; Malecka, M.; Guga, P.; Rozalska, B.; Budzisz, E. Antimicrobial and structural properties of metal ions complexes with thiosemicarbazide motif and related heterocyclic compounds. Curr. Med. Chem. 2019, 26, 664–693. [Google Scholar] [CrossRef]
- Haldys, K.; Latajka, R. Thiosemicarbazones with tyrosinase inhibitory activity. MedChemComm 2019, 10, 378–389. [Google Scholar] [CrossRef]
- Guha, P.C.; Dey, S.C. Hetero-ring formations with thiocarbohydrazide. II. Condensations with diketones and aldehydes. Q. J. Indian Chem. Soc. 1925, 2, 225–239. [Google Scholar]
- Guha, P.K.; Roychoudhury, S.K. Hetero-ring formations with thiocarbohydrazide. IV. Reactions of 1-phenylthiocarbohydrazide. J. Indian Chem. Soc. 1928, 5, 163–174. [Google Scholar]
- Guha, P.C.; Roychoudhury, S.K. Hetero-ring formations with thiocarbohydrazide. III. Reactions of substituted thiocarbohydrazides. J. Indian Chem. Soc. 1928, 5, 149–161. [Google Scholar]
- French, F.A.; Blanz, E.J. The Carcinostatic Activity of Thiosemicarbazones of Formyl Heteroaromatic Compounds. 1 III. Primary Correlation. J. Med. Chem. 1966, 9, 585–589. [Google Scholar] [CrossRef] [PubMed]
- Wiles, D.M.; Suprunchuk, T. Preparation and fungistatic properties of thiocarbohydrazones. J. Med. Chem. 1970, 13, 323–324. [Google Scholar] [CrossRef] [PubMed]
- Cano Pavon, J.M.; Garcia de Torres, A.; Cristofol Alcaraz, E.; Siles Cordero, M.T.; Vereda Alonso, E. Analytical applications of thiocarbohydrazones. A review. Quim. Anal. 1994, 13, 5–10. [Google Scholar]
- Blumenkopf, T.A.; Harrington, J.A.; Koble, C.S.; Bankston, D.D.; Morrison, R.W.; Bigham, E.C.; Styles, V.L.; Spector, T. 2-Acetylpyridine thiocarbonohydrazones. Potent inactivators of herpes simplex virus ribonucleotide reductase. J. Med. Chem. 1992, 35, 2306–2314. [Google Scholar] [CrossRef]
- Marzo, T.; Navas, F.; Cirri, D.; Merlino, A.; Ferraro, G.; Messori, L.; Quiroga, A.G. Reactions of a tetranuclear Pt-thiosemicarbazone complex with model proteins. J. Inorg. Biochem. 2018, 181, 11–17. [Google Scholar] [CrossRef]
- Pahontu, E.; Julea, F.; Rosu, T.; Purcarea, V.; Chumakov, Y.; Petrenco, P.; Gulea, A. Antibacterial, antifungal and in vitro antileukaemia activity of metal complexes with thiosemicarbazones. J. Cell. Mol. Med. 2015, 19, 865–878. [Google Scholar] [CrossRef]
- Sathisha, M.P.; Revankar, V.K.; Pai, K.S.R. Synthesis, structure, electrochemistry, and spectral characterization of bis-isatin thiocarbohydrazone metal complexes and their antitumor activity against ehrlich ascites carcinoma in swiss albino mice. Met.-Based Drugs 2008, 2008. [Google Scholar] [CrossRef][Green Version]
- Abu-Hussen, A.A.A.; Emara, A.A.A. Metal complexes of some thiocarbohydrazone ligands: Synthesis and structure. J. Coord. Chem. 2004, 57, 973–987. [Google Scholar] [CrossRef]
- Buu-Hoi, N.P.; Loc, T.B.; Xuong, N.D. The chemistry of thiocarbohydrazide. Bull. Soc. Chim. Fr. 1955, 694–697. [Google Scholar]
- Patil, S.A.; Kulkarni, V.H. Cobalt(II) and Copper(II) Complexes of Thiocarbohydrazones and Copper(ii) Bimetallic Complexes Of Nickel(II) Thiocarbohydrazones. ACTA Chim. Hung.-Models Chem. 1985, 118, 3–10. [Google Scholar]
- Zabin, S.A.; Jejurkar, C.R. Fluorescence, antibacterial and pigmentation studies of some binuclear Schiff base complexes. Asian J. Chem. 1995, 7, 542–550. [Google Scholar]
- Shebl, M.; Khalil, S.M.E.; Al-Gohani, F.S. Preparation, spectral characterization and antimicrobial activity of binary and ternary Fe(III), Co(II), Ni(II), Cu(II), Zn(II), Ce(III) and UO2(VI) complexes of a thiocarbohydrazone ligand. J. Mol. Struct. 2010, 980, 78–87. [Google Scholar] [CrossRef]
- Chee, D.N.A.; Affan, M.A.; Ahmad, F.B.; Asaruddin, M.R.; Sam, N.; Salam, M.A.; Ismail, A.; Tan, S.H. Synthesis, characterization, and antibacterial activity of organotin(IV) complexes with 2-hydroxyacetophenone thiocarbohydrazone. J. Coord. Chem. 2011, 64, 4191–4200. [Google Scholar] [CrossRef]
- Zafarian, H.; Sedaghat, T.; Motamedi, H.; Amiri Rudbari, H. A multiprotic ditopic thiocarbohydrazone ligand in the formation of mono- and di-nuclear organotin(IV) complexes: Crystal structure, antibacterial activity and DNA cleavage. J. Organomet. Chem. 2016, 825–826, 25–32. [Google Scholar] [CrossRef]
- Kaya, Y.; Ercag, A.; Koca, A. Synthesis, structures, electrochemical studies and antioxidant activities of cis-dioxomolybdenum(VI) complexes of the new bisthiocarbohydrazones. J. Mol. Struct. 2015, 1102, 117–126. [Google Scholar] [CrossRef]
- Kaya, Y.; Erçağ, A.; Kaya, K. Synthesis, characterization and antioxidant activities of dioxomolybdenum(VI) complexes of new Schiff bases derived from substituted benzophenones. J. Coord. Chem. 2018, 71, 3364–3380. [Google Scholar] [CrossRef]
- Siles Cordero, M.T.; Garcia de Torres, A.; Cano Pavon, J.M. Preconcentration of cadmium, cobalt, copper, nickel and zinc by solvent extraction into methyl isobutyl ketone with 1,5-bis[1-(2-pyridyl) ethylidene] thiocarbonohydrazide and 1,5-bis[phenyl-(2-pyridyl) methylene] thiocarbonohydrazide. Talanta 1993, 40, 691–695. [Google Scholar] [CrossRef]
- Bacchi, A.; Bonini, A.; Carcelli, M.; Ferraro, F.; Leporati, E.; Pelizzi, C.; Pelizzi, G. Chelating behaviour of methyl 2-pyridyl ketone carbono- and thiocarbonohydrazones in copper(II) and zinc(II) complexes. J. Chem. Soc. Dalton Trans. 1996, 2699–2704. [Google Scholar] [CrossRef]
- Carcelli, M.; Delledonne, D.; Fochi, A.; Pelizzi, G.; Rodríguez-Argüelles, M.C.; Russo, U. The behaviour of polydentate hydrazonic ligands derived from 2-acetylpyridine towards organotin compounds. Part I: The diorganotin(IV) complexes. J. Organomet. Chem. 1997, 544, 29–35. [Google Scholar] [CrossRef]
- Moubaraki, B.; Murray, K.S.; Ranford, J.D.; Wang, X.; Xu, Y. Structural and magnetic properties of an asymmetric dicopper(II) anticancer drug analog. Chem. Commun. 1998, 353–354. [Google Scholar] [CrossRef]
- Manoj, E.; Kurup, M.R.P.; Fun, H.-K.; Punnoose, A. Self-assembled macrocyclic molecular squares of Ni(II) derived from carbohydrazones and thiocarbohydrazones: Structural and magnetic studies. Polyhedron 2007, 26, 4451–4462. [Google Scholar] [CrossRef]
- Manoj, E.; Kurup, M.R.P.; Suresh, E. Synthesis and Spectral Studies of Bisthiocarbohydrazone and Biscarbohydrazone of Quinoline-2-carbaldehyde: Crystal Structure of Bis(quinoline-2-aldehyde) Thiocarbohydrazone. J. Chem. Crystallogr. 2008, 38, 157–161. [Google Scholar] [CrossRef]
- Manoj, E.; Kurup, M.R.P.; Punnoose, A. Preparation, magnetic and EPR spectral studies of copper(II) complexes of an anticancer drug analogue. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2009, 72A, 474–483. [Google Scholar] [CrossRef] [PubMed]
- Tandon, S.S.; Dul, M.-C.; Lee, J.L.; Dawe, L.N.; Anwar, M.U.; Thompson, L.K. Complexes of ditopic carbo- and thio-carbohydrazone ligands—Mononuclear, 1D chain, dinuclear and tetranuclear examples. Dalton Trans. 2011, 40, 3466–3475. [Google Scholar] [CrossRef]
- Bacchi, A.; Carcelli, M.; Pelagatti, P.; Pelizzi, C.; Pelizzi, G.; Zani, F. Antimicrobial and mutagenic activity of some carbono- and thiocarbonohydrazone ligands and their copper(II), iron(II) and zinc(II) complexes. J. Inorg. Biochem. 1999, 75, 123–133. [Google Scholar] [CrossRef]
- Tiwari, A.D.; Mishra, A.K.; Mishra, S.B.; Mamba, B.B.; Maji, B.; Bhattacharya, S. Synthesis and DNA binding studies of Ni(II), Co(II), Cu(II) and Zn(II) metal complexes of N1,N5-bis[pyridine-2-methylene]-thiocarbohydrazone Schiff-base ligand. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2011, 79, 1050–1056. [Google Scholar] [CrossRef]
- Zhang, N.; Tai, Y.; Li, M.; Ma, P.; Zhao, J.; Niu, J. Main group bismuth(iii), gallium(iii) and diorganotin(iv) complexes derived from bis(2-acetylpyrazine)thiocarbonohydrazone: Synthesis, crystal structures and biological evaluation. Dalton Trans. 2014, 43, 5182–5189. [Google Scholar] [CrossRef]
- Božić, A.; Marinković, A.; Bjelogrlić, S.; Todorović, T.R.; Cvijetić, I.N.; Novaković, I.; Muller, C.D.; Filipović, N.R. Quinoline based mono- and bis-(thio)carbohydrazones: Synthesis, anticancer activity in 2D and 3D cancer and cancer stem cell models. RSC Adv. 2016, 6, 104763–104781. [Google Scholar] [CrossRef]
- Nair, R.S.; Potti, M.E.; Thankappan, R.; Chandrika, S.K.; Kurup, M.R.P.; Srinivas, P. Molecular trail for the anticancer behavior of a novel copper carbohydrazone complex in BRCA1 mutated breast cancer. Mol. Carcinog. 2017, 56, 1501–1514. [Google Scholar] [CrossRef]
- Bozic, A.R.; Bjelogrlic, S.K.; Novakovic, I.T.; Filipovic, N.R.; Petrovic, P.M.; Marinkovic, A.D.; Todorovic, T.R.; Cvijetic, I.N. Antimicrobial Activity of Thiocarbohydrazones: Experimental Studies and Alignment-Independent 3D QSAR Models. ChemistrySelect 2018, 3, 2215–2221. [Google Scholar] [CrossRef]
- Kumar, S.L.A.; Kumar, M.S.; John, R.P.; Muthiah, P.T.; Sreekanth, A. 1,5-Bis(2-hydroxyacetophenone)thiocarbohydrazone: A novel colorimetric and fluorescent dual-mode chemosensor for the recognition of fluoride. Mater. Sci. Eng. C 2013, 33, 2519–2525. [Google Scholar] [CrossRef] [PubMed]
- Rai, A.; Kumari, N.; Srivastava, A.K.; Mishra, L. Synthesis and characterization of bispentafluorophenyl carbohydrazone/thiocarbohydrazone: Selective recognition of F-ions. Tetrahedron Lett. 2013, 54, 7151–7155. [Google Scholar] [CrossRef]
- Biswas, S.; Gangopadhyay, M.; Barman, S.; Sarkar, J.; Singh, N.D.P. Simple and efficient coumarin-based colorimetric and fluorescent chemosensor for F-detection: An ON1-OFF-ON2 fluorescent assay. Sens. Actuators B Chem. 2016, 222, 823–828. [Google Scholar] [CrossRef]
- Avaji, P.G.; Patil, S.A.; Badami, P.S. Synthesis, spectral, thermal, solid state d.c. electrical conductivity and biological studies of lanthanum(III) and thorium(IV) complexes with thiocarbohydrazone. Transit. Met. Chem. 2007, 32, 379–386. [Google Scholar] [CrossRef]
- Bacchi, A.; Carcelli, M.; Pelagatti, P.; Pelizzi, G.; Rodriguez-Arguelles, M.C.; Rogolino, D.; Solinas, C.; Zani, F. Antimicrobial and mutagenic properties of organotin(IV) complexes with isatin and N-alkylisatin bisthiocarbonohydrazones. J. Inorg. Biochem. 2005, 99, 397–408. [Google Scholar] [CrossRef]
- Abou-Melha, K.S.; Faruk, H. Bimetallic complexes of Schiff base bis-[4-hydroxycoumarin-3-yl]-1N,5N-thiocarbohydrazone as a potentially dibasic pentadentate ligand. Synthesis, spectral, and antimicrobial properties. J. Iran. Chem. Soc. 2008, 5, 122–134. [Google Scholar] [CrossRef]
- Bagihalli, G.B.; Avaji, P.G.; Badami, P.S.; Patil, S.A. Synthesis, spectral characterization, electrochemical and biological studies of Co(II), Ni(II) and Cu(II) complexes with thiocarbohydrazone. J. Coord. Chem. 2008, 61, 2793–2806. [Google Scholar] [CrossRef]
- Sathisha, M.P.; Shetti, U.N.; Revankar, V.K.; Pai, K.S.R. Synthesis and antitumor studies on novel Co(II), Ni(II) and Cu(II) metal complexes of bis(3-acetylcoumarin)thiocarbohydrazone. Eur. J. Med. Chem. 2008, 43, 2338–2346. [Google Scholar] [CrossRef]
- Shebl, M.; Seleem, H.S.; El-Shetary, B.A. Ligational behavior of thiosemicarbazone, semicarbazone and thiocarbohydrazone ligands towards VO(IV), Ce(III), Th(IV) and UO2(VI) ions: Synthesis, structural characterization and biological studies. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2010, 75, 428–436. [Google Scholar] [CrossRef]
- Pandey, O.P. Mononuclear and binuclear lanthanum(III) complexes of macrocyclic ligands derived from 2,6-diacetylpyridine. Polyhedron 1987, 6, 1021–1025. [Google Scholar] [CrossRef]
- Chantrapromma, S.; Razak, I.A.; Fun, H.K.; Karalai, C.; Zhang, H.; Xie, F.X.; Tian, Y.P.; Ma, W.; Zhang, Y.H.; Ni, S.S. Bis[2,2′-ethylenedioxydibenzaldehyde bis(thiocarbohydrazide)] tris(pyridine) solvate. Acta Crystallogr. Sect. C Cryst. Struct. Commun. 2001, C57, 289–290. [Google Scholar] [CrossRef] [PubMed]
- Chohan, Z.H.; Khan, K.M.; Supuran, C.T. Synthesis of antibacterial and antifungal cobalt(II), copper(II), nickel(II) and zinc(II) complexes with bis-(1,1′-disubstituted ferrocenyl)thiocarbohydrazone and bis-(1,1′-disubstituted ferrocenyl)carbohydrazone. Appl. Organomet. Chem. 2004, 18, 305–310. [Google Scholar] [CrossRef]
- Abou-Hussein, A.A.A.; Linert, W. Synthesis, spectroscopic and biological activities studies of acyclic and macrocyclic mono and binuclear metal complexes containing a hard-soft Schiff base. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2012, 95, 596–609. [Google Scholar] [CrossRef]
- Sandstrom, J. Cyclizations of thiocarbohydrazide and its monohydrazones. I. Reactions with ortho esters. Acta Chem. Scand. 1960, 14, 1037–1042. [Google Scholar] [CrossRef]
- Brockman, R.W.; Thomson, J.R.; Bell, M.J.; Skipper, H.E. Observations on the antileukemic activity of pyridine-2-carboxaldehyde thiosemicarbazone and thiocarbohydrazone. Cancer Res. 1956, 16, 167–170. [Google Scholar]
- Chandra, S.; Sharma, A.K. Biological and spectral studies of transition metal complexes with a quinquedentate Schiff base, 2,6-diacetylpyridine bis(thiocarbohydrazone). J. Coord. Chem. 2009, 62, 3688–3700. [Google Scholar] [CrossRef]
- Bonaccorso, C.; Grasso, G.; Musso, N.; Barresi, V.; Condorelli, D.F.; La Mendola, D.; Rizzarelli, E. Water soluble glucose derivative of thiocarbohydrazone acts as ionophore with cytotoxic effects on tumor cells. J. Inorg. Biochem. 2018, 182, 92–102. [Google Scholar] [CrossRef]
- Chohan, Z.H.; Pervez, H.; Khan, K.M.; Supuran, C.T. Organometallic-based antibacterial and antifungal compounds: Transition metal complexes of 1,1′-diacetylferrocene-derived thiocarbohydrazone, carbohydrazone, thiosemicarbazone and semicarbazone. J. Enzym. Inhib. Med. Chem. 2005, 20, 81–88. [Google Scholar] [CrossRef]
- Mosa, A.I.; Emara, A.A.A.; Yousef, J.M.; Saddiq, A.A. Novel transition metal complexes of 4-hydroxy-coumarin-3-thiocarbohydrazone: Pharmacodynamic of Co(III) on rats and antimicrobial activity. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2011, 81, 35–43. [Google Scholar] [CrossRef]
- Gangarapu, K.; Manda, S.; Thota, S.; Yerra, R.; Karki, S.S.; Balzarini, J.; De Clercq, E.; Tokuda, H. Microwave assisted synthesis, characterization of some new isatin and thiophene derivatives as cytotoxic and chemopreventive agents. Lett. Drug Des. Discov. 2012, 9, 934–941. [Google Scholar] [CrossRef]
- Gabr, M.T.; El-Gohary, N.S.; El-Bendary, E.R.; El-Kerdawy, M.M.; Ni, N. Isatin-β-thiocarbohydrazones: Microwave-assisted synthesis, antitumor activity and structure-activity relationship. Eur. J. Med. Chem. 2017, 128, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Gabr, M.T.; El-Gohary, N.S.; El-Bendary, E.R.; Ni, N.; Shaaban, M.I.; El-Kerdawy, M.M. Microwave-assisted synthesis, antimicrobial, antiquorum-sensing and cytotoxic activities of a new series of isatin-β-thiocarbohydrazones. Synth. Commun. 2018, 48, 2899–2911. [Google Scholar] [CrossRef]
- Abbas, S.Y.; Farag, A.A.; Ammar, Y.A.; Atrees, A.A.; Mohamed, A.F.; El-Henawy, A.A. Synthesis, characterization, and antiviral activity of novel fluorinated isatin derivatives. Mon. Fuer Chem. 2013, 144, 1725–1733. [Google Scholar] [CrossRef]
- Gangarapu, K.; Manda, S.; Jallapally, A.; Thota, S.; Karki, S.S.; Balzarini, J.; De Erik, C.; Tokuda, H. Synthesis of thiocarbohydrazide and carbohydrazide derivatives as possible biologically active agents. Med. Chem. Res. 2014, 23, 1046–1056. [Google Scholar] [CrossRef]
- Tejasree, C.; Kiran, G.; Rajyalakshmi, G.; Rama Narsimha Reddy, A. Hepatoprotective activity of 1-(4-(Dimethylamino)Benzylidene)-5-(2-Oxoindolin-3-ylidene) Thiocarbohydrazone in rats. Toxicol. Environ. Chem. 2013, 95, 1589–1594. [Google Scholar] [CrossRef]
- Tejasree, C.; Kiran, G.; Rajyalakshmi, G.; Reddy, A.R.N. Antidiabetic activity of 1-(4-(dimethylamino)benzylidene)-5-(2-oxo indolin-3-ylidene) thiocarbohydrazone in rats. Int. J. Pharm. Sci. Res. 2014, 5, 2738–2743. [Google Scholar]
- Srividya, L.; Reddy, A.R.N. Antidiabetic activity of 1-(4-chlorobenzylidene)-5-(2-oxoindolin-3-ylidene) thiocarbohydrazone in chick model. Asian J. Biol. Sci. 2017, 10, 126–130. [Google Scholar]
- La Mendola, D.; Giacomelli, C.; Rizzarelli, E. Intracellular Bioinorganic Chemistry and Cross Talk Among Different-Omics. Curr. Top. Med. Chem. 2016, 16, 3103–3130. [Google Scholar] [CrossRef]
- La Mendola, D. Nerve growth factor catches copper in neuronal inning. Neural Regen. Res. 2020, 15, 665–666. [Google Scholar] [CrossRef]
- Palanimuthu, D.; Poon, R.; Sahni, S.; Anjum, R.; Hibbs, D.; Lin, H.-Y.; Bernhardt, P.V.; Kalinowski, D.S.; Richardson, D.R. A novel class of thiosemicarbazones show multi-functional activity for the treatment of Alzheimer’s disease. Eur. J. Med. Chem. 2017, 139, 612–632. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bonaccorso, C.; Marzo, T.; La Mendola, D. Biological Applications of Thiocarbohydrazones and Their Metal Complexes: A Perspective Review. Pharmaceuticals 2020, 13, 4. https://doi.org/10.3390/ph13010004
Bonaccorso C, Marzo T, La Mendola D. Biological Applications of Thiocarbohydrazones and Their Metal Complexes: A Perspective Review. Pharmaceuticals. 2020; 13(1):4. https://doi.org/10.3390/ph13010004
Chicago/Turabian StyleBonaccorso, Carmela, Tiziano Marzo, and Diego La Mendola. 2020. "Biological Applications of Thiocarbohydrazones and Their Metal Complexes: A Perspective Review" Pharmaceuticals 13, no. 1: 4. https://doi.org/10.3390/ph13010004
APA StyleBonaccorso, C., Marzo, T., & La Mendola, D. (2020). Biological Applications of Thiocarbohydrazones and Their Metal Complexes: A Perspective Review. Pharmaceuticals, 13(1), 4. https://doi.org/10.3390/ph13010004





