Production and Characterization of Chitosan–Polyanion Nanoparticles by Polyelectrolyte Complexation Assisted by High-Intensity Sonication for the Modified Release of Methotrexate
Abstract
1. Introduction
2. Experimental
2.1. Materials
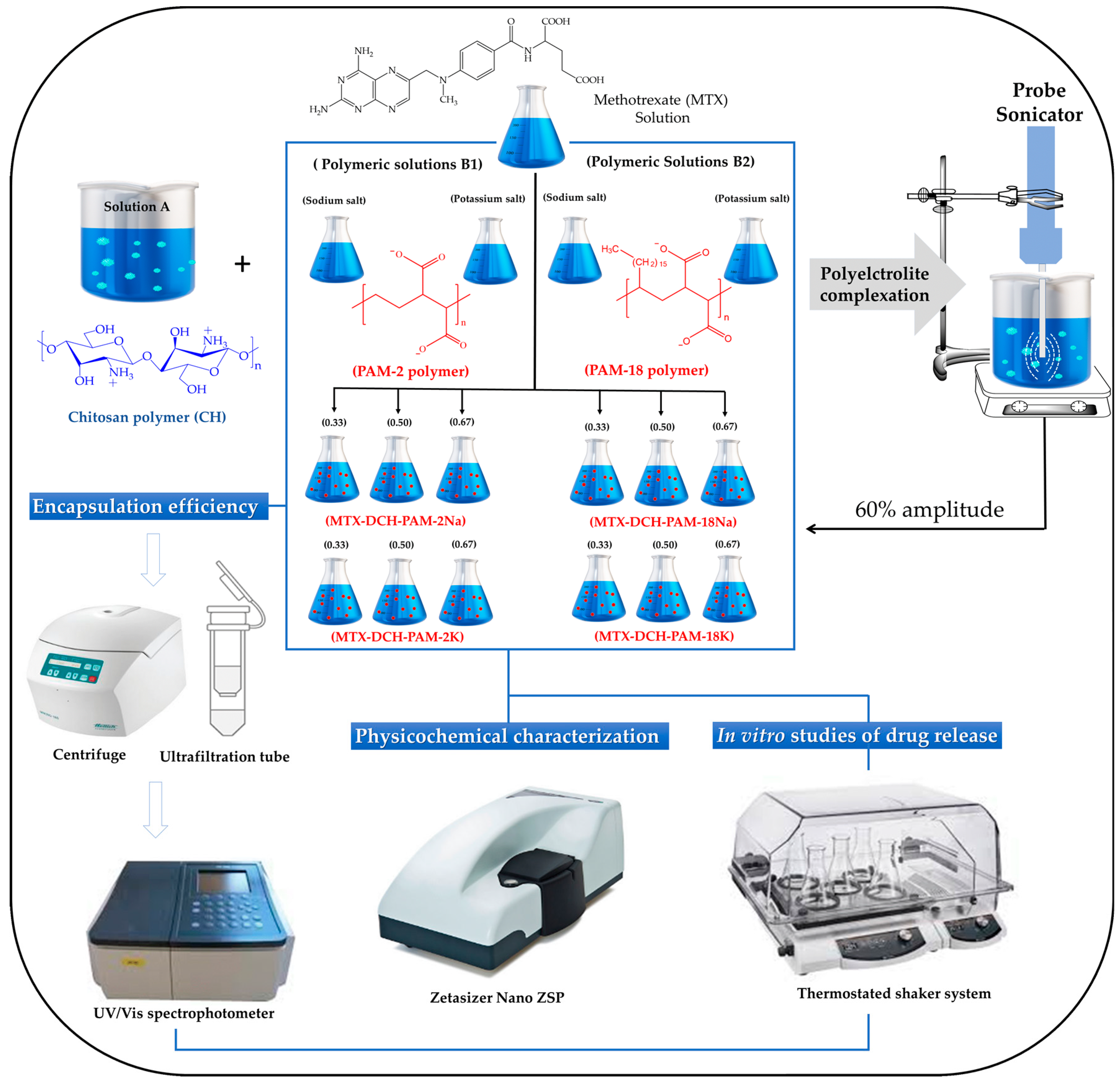
2.2. Preparation of NP Systems
2.3. Physicochemical Characterization of NPs
2.3.1. Particle Size, Polydispersity Index (PDI) and Zeta Potential Analyses
2.3.2. Encapsulation Efficiency (EE)
2.4. Physical Stability of the Nanoparticulate Systems
2.5. In Vitro Release Studies of Methotrexate
2.6. Statistical Analysis
3. Results and Discussion
3.1. Production and Characterization of Nanoparticulate Systems
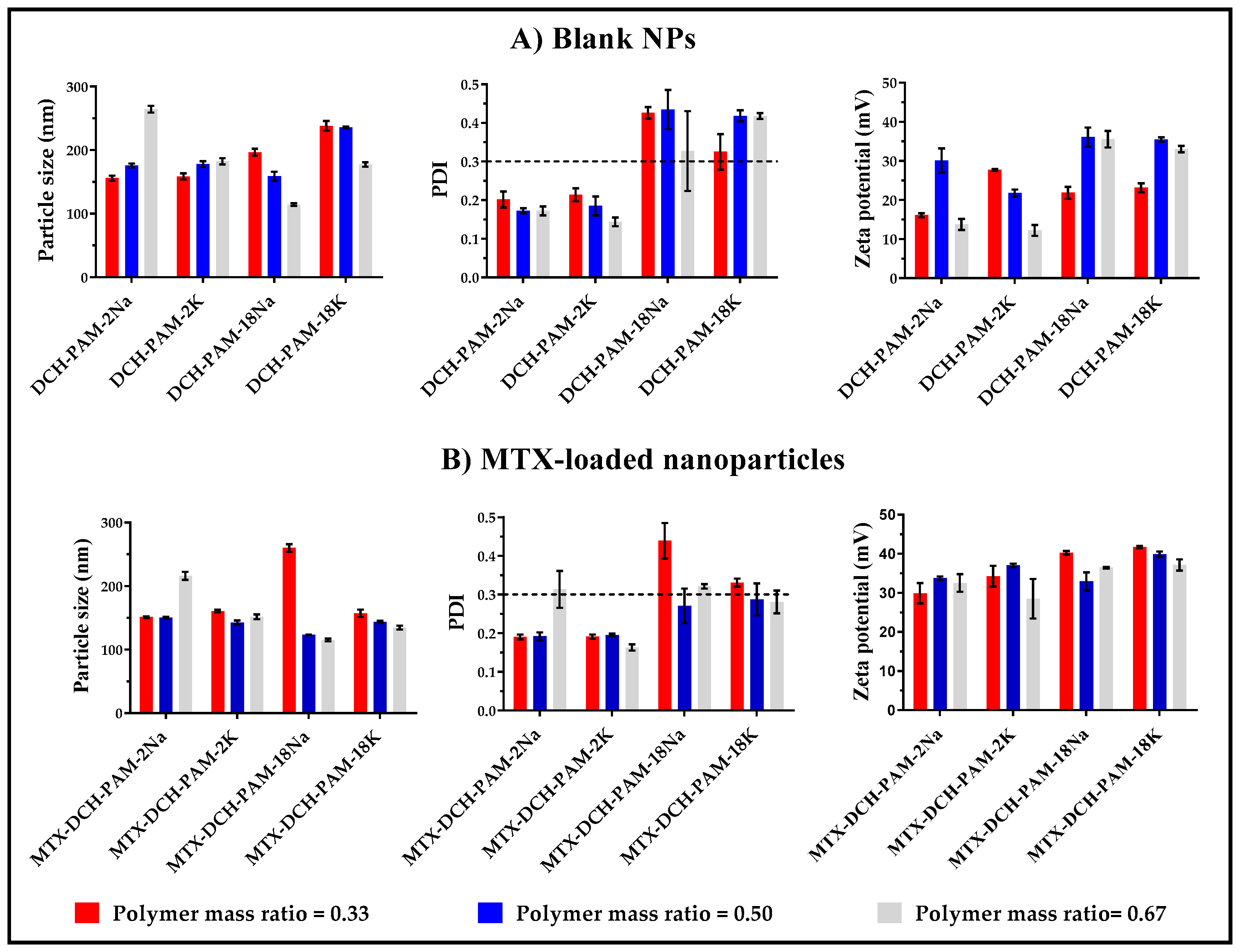
3.1.1. Particle Size
3.1.2. Polydispersity
3.1.3. Zeta Potential
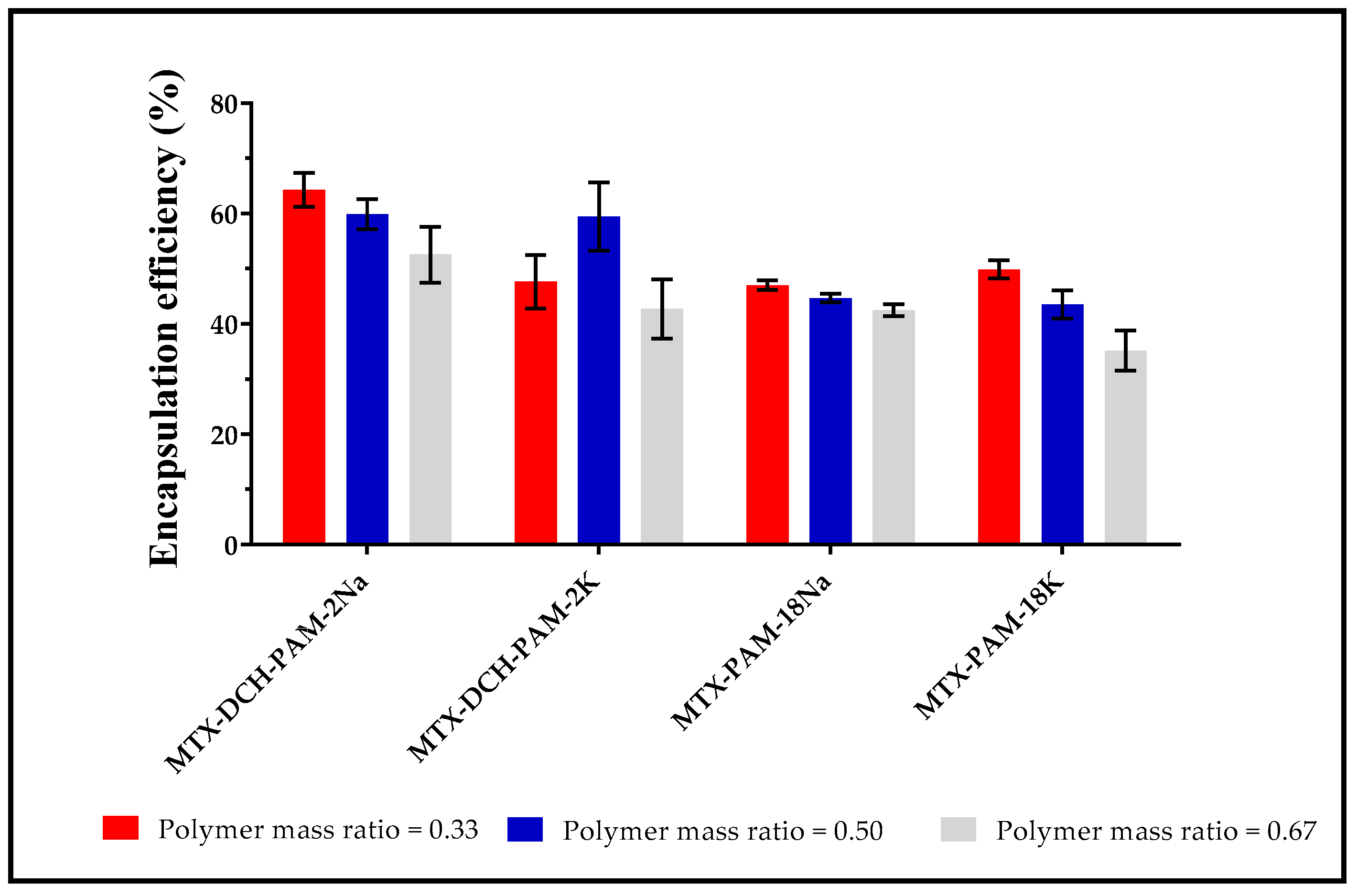
3.1.4. Encapsulation Efficiency
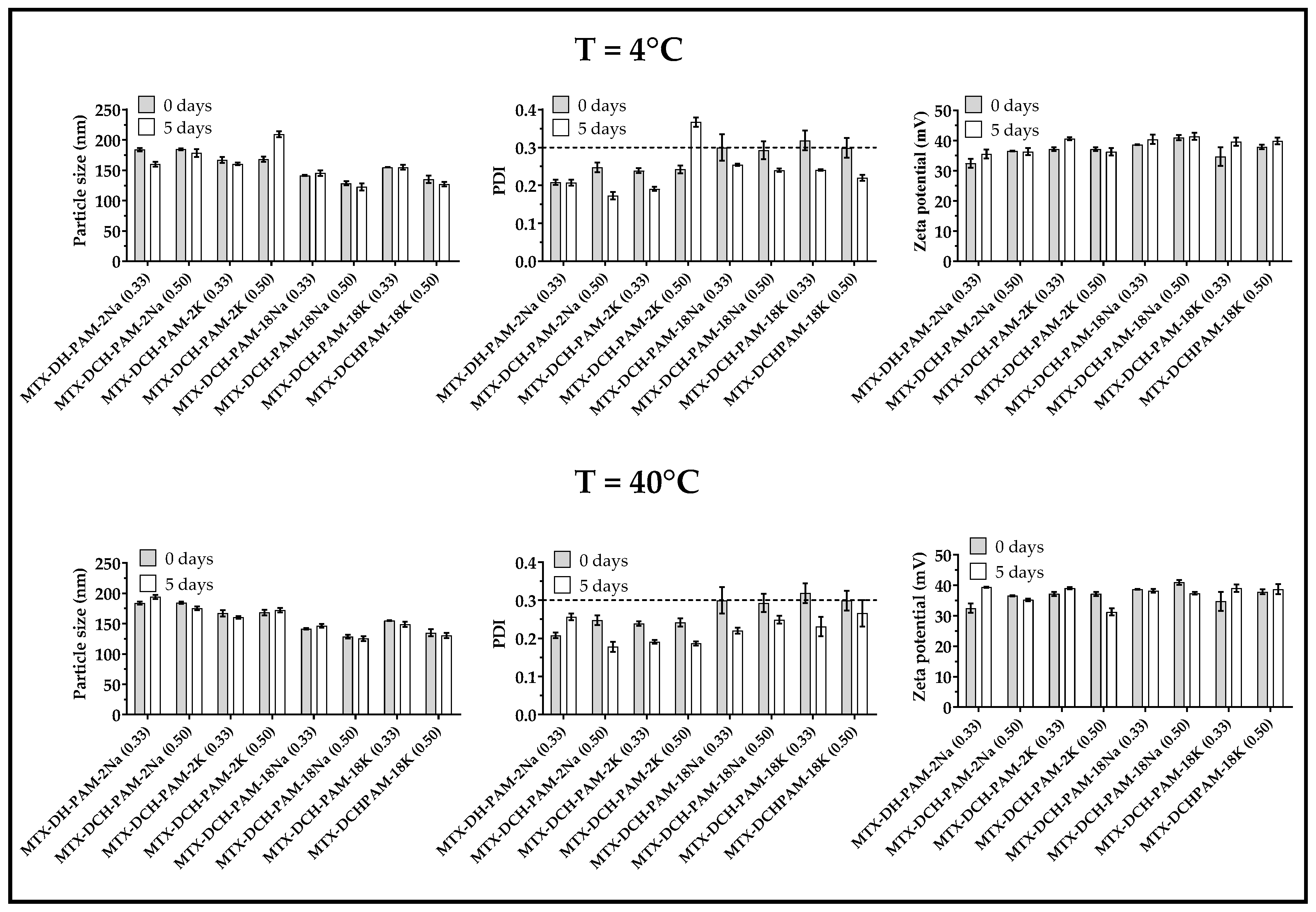
3.2. Physical Stability of the Nanoparticulate Systems
3.3. In Vitro Release Studies of Methotrexate
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| MTX | Methotrexate |
| DCH | Deacetylated chitosan polymer |
| CH | Chitosan polymer |
| PAM-2Na | Sodium salt of poly(maleic acid-alt-ethylene) |
| PAM-2K | Potassium salt of poly(maleic acid-alt-ethylene) |
| PAM-18Na | Sodium salt of poly(maleic acid-alt-octadecene) |
| PAM-18K | Potassium poly(maleic acid-alt-octadecene) |
| NPs | Nanoparticles |
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef]
- World Health Organization. Cancer. 2018. Available online: https://www.who.int/health-topics/cancer#tab=tab_1 (accessed on 4 November 2018).
- Rojas, J.; Ciro, Y.; Salamanca, C. Thiolated chitosan: A promising strategy for improving the effectiveness of anticancer drugs. In Analytical and Pharmaceutical Chemistry; SMGroup: Dover, DE, USA, 2017; pp. 1–17. [Google Scholar]
- Saboktakin, M.R.; Tabatabaie, R.M.; Maharramov, A.; Ramazanv, M.A. Synthesis and characterization of pH-dependent glycol chitosan and dextran sulfate nanoparticles for effective brain cancer treatment. Int. J. Biol. Macromol. 2011, 49, 747–751. [Google Scholar] [CrossRef]
- Beidokhti, H.R.; Ghaffarzadegan, R.; Mirzakhanlouei, S.; Ghazizadeh, L.; Dorkoosh, F.A. Preparation, Characterization, and Optimization of Folic Acid-Chitosan-Methotrexate Core-Shell Nanoparticles by Box-Behnken Design for Tumor-Targeted Drug Delivery. AAPS PharmSciTech 2017, 18, 115–129. [Google Scholar] [CrossRef]
- Sun, Y.; Cui, F.; Shi, K.; Wang, J.; Niu, M.; Ma, R. The effect of chitosan molecular weight on the characteristic of spray-dried methotrexate-loaded microspheres for nasal administration. Drug Dev. Ind. Pharm. 2009, 35, 379–386. [Google Scholar] [CrossRef]
- Zhao, Y.; Guo, Y.; Li, R.; Wang, T.; Han, M.; Zhu, C.; Wan, X. Methotrexate nanoparticles prepared with codendrimer from polyamidoamine (PAMAM) and oligoethylene glycols (OEG) dendrons: Antitumor efficacy in vitro and in vivo. Sci. Rep. 2016, 6, 28983. [Google Scholar] [CrossRef] [PubMed]
- Yhee, J.Y.; Koo, H.; Lee, D.E.; Choi, K.; Kwon, I.C.; Kim, k. Multifunctional chitosan nanoparticles for tumor imaging and therapy. Adv. Polym. Sci. 2011, 243, 139–162. [Google Scholar]
- Jia, M.; Li, Y.; yang, X.; Huang, Y.; Wu, H.; Huang, Y.; Lin, J.; Li, Y.; Hou, Z.; Zhang, Q. Development of both methotrexate and mitomycin C loaded pegylated chitosan nanoparticles for targeted drug Co-delivery and synergistic anticancer effect. ACS Appl. Mater. Interfaces 2014, 6, 11413–11423. [Google Scholar] [CrossRef] [PubMed]
- Wimardhani, Y.S.; Suniarti, D.F.; Freisleben, H.J.; Wanandi, S.I.; Siregar, N.C.; Ikeda, M.A. Chitosan exerts anticancer activity through induction of apoptosis and cell cycle arrest in oral cancer cells. J. Oral Sci. 2014, 56, 119–126. [Google Scholar] [CrossRef]
- Park, J.K.; Chung, M.J.; Choi, H.N.; Park, Y. Effects of the molecular weight and the degree of deacetylation of chitosano ligosaccharides on antitumor activity. Int. J. Mol. Sci. 2011, 12, 266–277. [Google Scholar] [CrossRef]
- Adhikari, H.S.; Yadav, P.N. Anticancer activity of chitosan, chitosan derivatives, and their mechanism of action. Int. J. Biomater. 2018, 2018, 1–29. [Google Scholar] [CrossRef]
- Nur, S.G.; Buket, O.; Sezgi, K.; Cagdas, A.; Cansu, A.; Guliz, A.; Habibe, Y.; Cigir, B.A.; Yavuz, D.; Senay, H.S. Synthesis of Methotrexate Loaded Chitosan Nanoparticles and in vitro Evaluation of the Potential in Treatment of Prostate Cancer. Anticancer Agents Med. Chem. 2016, 16, 1038–1042. [Google Scholar] [PubMed]
- Kumari, S.D.C. Formulation and Evaluation of Drug Loaded Nanoparticles for Rheumatoid Arthritis. Ph.D. Thesis, Madras Medical College, Chennai, India, 2014. [Google Scholar]
- Nematpour, N.; Azandaryani, A.H.; Arkan, E. Preparation and In Vitro Characterization of pH-Sensitive Methotrexate Conjugated Chitosan Nanoparticles. Iran. J. Pharmceut. Sci. 2017, 13, 159. [Google Scholar]
- Chen, J.; Huang, L.; Lai, H.; Lu, C.; Fang, M.; Zhang, Q.; Luo, X. Methotrexate-loaded PEGylated chitosan nanoparticles: Synthesis, characterization, and in vitro and in vivo antitumoral activity. Mol. Pharm. 2014, 11, 2213–2223. [Google Scholar] [CrossRef] [PubMed]
- Vrignaud, S.; Benoit, J.P.; Saulnier, P. Strategies for the nanoencapsulation of hydrophilic molecules in polymer-based nanoparticles. Biomaterials 2011, 32, 8593–8604. [Google Scholar] [CrossRef]
- Dong, W.; Bodmeier, R. Encapsulation of lipophilic drugs within enteric microparticles by a novel coacervation method. Int. J. Pharm. 2006, 326, 128–138. [Google Scholar] [CrossRef] [PubMed]
- Teja, S.P.S.; Damodharan, N. 23 Full Factorial Model for Particle Size Optimization of Methotrexate Loaded Chitosan Nanocarriers: A Design of Experiments (DoE) Approach. Biomed. Res. Int. 2018, 2018. [Google Scholar] [CrossRef]
- Salem, D.S.; Shouman, S.A.; Badr, Y. Laser-triggered release of drug encapsulated in chitosan nanoparticles for therapy of hepatocellular carcinoma. In Proceedings of the Colloidal Nanoparticles for Biomedical Applications XIV, San Francisco, CA, USA, 7 March 2019; p. 108920Z. [Google Scholar]
- Qi, L.; Xu, Z.; Jiang, X.; Hu, C.; Zou, X. Preparation and antibacterial activity of chitosan nanoparticles. Carbohydr. Res. 2004, 339, 2693–2700. [Google Scholar] [CrossRef]
- Bugnicourt, L.; Ladavière, C. Interests of chitosan nanoparticles ionically cross-linked with tripolyphosphate for biomedical applications. Prog. Polym. Sci. 2016, 60, 1–17. [Google Scholar] [CrossRef]
- Rodrigues, S.; da Costa, A.M.; Grenha, A. Chitosan/carrageenan nanoparticles: Effect of cross-linking with tripolyphosphate and charge ratios. Carbohydr. Polym. 2012, 89, 282–289. [Google Scholar] [CrossRef]
- Saïed, N.; Aider, M. Zeta Potential and Turbidimetry Analyzes for the Evaluation of Chitosan/Phytic Acid Complex Formation. J. Food Res. 2014, 3, 71. [Google Scholar] [CrossRef]
- Yang, C.Y.; Hsu, C.H.; Tsai, M.L. Effect of crosslinked condition on characteristics of chitosan/ tripolyphosphate/genipin beads and their application in the selective adsorption of phytic acid from soybean whey. Carbohydr. Polym. 2011, 86, 659–665. [Google Scholar] [CrossRef]
- Zheng, C.H.; Gao, J.Q.; Zhang, Y.P.; Liang, W.Q. A protein delivery system: Biodegradable alginate-chitosan-poly(lactic-co-glycolic acid) composite microspheres. Biochem. Biophys. Res. Commun. 2004, 323, 1321–1327. [Google Scholar] [CrossRef] [PubMed]
- Gazori, T.; Khoshayand, M.R.; Azizi, E.; Yazdizade, P.; Nomani, A.; Haririan, I. Evaluation of Alginate/Chitosan nanoparticles as antisense delivery vector: Formulation, optimization and in vitro characterization. Carbohydr. Polym. 2009, 77, 599–606. [Google Scholar] [CrossRef]
- González-Rodríguez, M.L.; Holgado, M.A.; Sánchez-Lafuente, C.; Rabasco, A.M.; Fini, A. Alginate/chitosan particulate systems for sodium diclofenac release. Int. J. Pharm. 2002, 232, 225–234. [Google Scholar] [CrossRef]
- De, S.; Robinson, D. Polymer relationships during preparation of chitosan-alginate and poly-l-lysine-alginate nanospheres. J. Control. Release 2003, 89, 101–112. [Google Scholar] [CrossRef]
- Tan, C.; Xie, J.; Zhang, X.; Cai, J.; Xia, S. Polysaccharide-based nanoparticles by chitosan and gum arabic polyelectrolyte complexation as carriers for curcumin. Food Hydrocoll. 2016, 57, 236–245. [Google Scholar] [CrossRef]
- Avadi, M.R.; Sadeghi, A.M.M.; Mohammadpour, N.; Abedin, S.; Atyabi, F.; Dinarvand, R.; Rafiee-Tehrani, M. Preparation and characterization of insulin nanoparticles using chitosan and Arabic gum with ionic gelation method. Nanomed. Nanotechnol. Biol. Med. 2010, 6, 58–63. [Google Scholar] [CrossRef]
- Luo, Y.; Wang, Q. Recent development of chitosan-based polyelectrolyte complexes with natural polysaccharides for drug delivery. Int. J. Biol. Macromol. 2014, 64, 353–367. [Google Scholar] [CrossRef]
- Butstraen, C.; Salaün, F. Preparation of microcapsules by complex coacervation of gum Arabic and chitosan. Carbohydr. Polym. 2014, 99, 608–616. [Google Scholar] [CrossRef]
- Razak, S.I.A.; Sharif, N.F.A.; Rahman, W.A.A. Biodegradable Polymers and their Bone Applications: A Review. Int. J. Eng. Sci. 2012, 12, 31–49. [Google Scholar]
- Popescu, I.; Suflet, D.M.; Pelin, I.M.; Chiţanu, G.C. Biomedical applications of maleic anhydride copolymers. Rev. Roum. Chim. 2011, 56, 173–188. [Google Scholar]
- Atta, A.M.; El-Hamouly, S.H.; Alsabagh, A.M.; Gabr, M.M. Crosslinked poly(octadecene-alt-maleic anhydride) copolymers as crude oil sorbers. J. Appl. Polym. Sci. 2007, 105, 2112–2120. [Google Scholar] [CrossRef]
- Carneiro, J.; Tedim, J.; Fernandes, S.C.M.; Freire, C.S.R.; Gandini, A.; Ferreira, M.G.S.; Zheludkevich, M.L. Functionalized chitosan-based coatings for active corrosion protection. Surf. Coat. Technol. 2013, 226, 51–59. [Google Scholar] [CrossRef]
- Di Corato, R.; Quarta, A.; Piacenza, P.; Ragusa, A.; Figuerola, A.; Buonsanti, R.; Cingolani, R.; Manna, L.; Pellegrino, T. Water solubilization of hydrophobic nanocrystals by means of poly(maleic anhydride-alt-1-octadecene). J. Mater. Chem. 2008, 18, 1991–1996. [Google Scholar] [CrossRef]
- Salamanca, C.H.; Yarce, C.J.; Roman, Y.; Davalos, A.F.; Rivera, G.R. Application of nanoparticle technology to reduce the anti-microbial resistance through B-lactam antibiotic polymer inclusion nano-complex. Pharmaceuticals 2018, 11, 19. [Google Scholar] [CrossRef] [PubMed]
- Yarce, C.J.C.; Echeverri, J.J.D.; Palacio, M.M.A.; Rivera, C.C.A.; Salamanca, C.H.C. Relationship between Surface Properties and In Vitro Drug Release from Compressed Matrix Containing Polymeric Materials with Different Hydrophobicity Degrees. Pharmaceuticals 2017, 10, 15. [Google Scholar] [CrossRef]
- Ciro, Y.; Rojas, J.; Oñate-Garzon, J.; Salamanca, C.H. Synthesis, Characterisation and Biological Evaluation of Ampicillin-Chitosan-Polyanion Nanoparticles Produced by Ionic Gelation and Polyelectrolyte Complexation Assisted by High-Intensity Sonication. Polymers 2019, 11, 1758. [Google Scholar] [CrossRef]
- Ciro, Y.; Rojas, J.; Yarce, C.J.; Salamanca, C.H. Preparation, characterization and rheological behavior of glutathione-chitosan conjugates in aqueous media. Appl. Rheol. 2019, 29, 1–12. [Google Scholar] [CrossRef]
- Podczeck, F. Comparison of in vitro dissolution profiles by calculating mean dissolution time (MDT) or mean residence time (MRT). Int. J. Pharm. 1993, 97, 93–100. [Google Scholar] [CrossRef]
- Siepmann, J.; Siepmann, F. Mathematical modeling of drug dissolution. Int J Pharm 2013, 453, 12–24. [Google Scholar] [CrossRef]
- Mathematical models of drug release. In Strategies to Modify the Drug Release from Pharmaceutical Systems; Bruschi, M.L., Ed.; Woodhead Publishing: Cambridge, UK, 2015; pp. 63–86. [Google Scholar]
- Ritger, P.L.; Peppas, N.A. A simple equation for description of solute release II. Fickian and anomalous release from swellable devices. J. Control. Release 1987, 5, 37–42. [Google Scholar] [CrossRef]
- Ritger, P.L.; Peppas, N.A. A simple Fickian equation for description of solute release. I. Fickian and non-Fickian release from non-swellable devices in the form of slabs, spheres, cylinders or discs. J. Control. Release 1987, 5, 23–26. [Google Scholar] [CrossRef]
- Peppas, N.A.; Sahlin, J.J. A simple equation for description of solute release. II. Fickian and anomalous release from swellable devices. Int. J. Pharm. 1989, 57, 169–172. [Google Scholar] [CrossRef]
- Higuchi, W.I. Diffusional models useful in biopharmaceutics drug releaserate processes. J. Pharm. Sci. 1967, 56, 315–324. [Google Scholar] [CrossRef]
- Diaz, D.A.; Colgan, S.T.; Langer, C.S.; Bandi, N.T.; Likar, M.D.; Alstine, L.V. Dissolution similarity requirements: How similar or dissimilar are the global regulatory expectations? AAPS J. 2016, 18, 15–22. [Google Scholar] [CrossRef]
- Usmiati, S.; Richana, N.; Mangunwidjaja, D.; Noor, E.; Prangdimurti, E. The Using of Ionic Gelation Method Based on Polysaccharides for Encapsulating the Macromolecules—A Review. In Proceedings of the International Conference on Food Security and Nutrition, Singapore, April 2014; pp. 80–84. [Google Scholar]
- Tang, E.S.K.; Huang, M.; Lim, L.Y. Ultrasonication of chitosan and chitosan nanoparticles. Int. J. Pharm. 2003, 265, 103–114. [Google Scholar] [CrossRef]
- Mishra, B.; Mishra, M.; Yadav, S.K. Antibacterial loaded spray dried chitosan polyelectrolyte complexes as dry powder aerosol for the treatment of lung infections. Iran. J. Pharm. Res. 2017, 16, 74–92. [Google Scholar]
- Patil, P.; Chavanke, D.; Wagh, M. A review on ionotropic gelation method: Novel approach for controlled gastroretentive gelispheres. Int. J. Pharm. Pharm. Sci. 2012, 4, 27–32. [Google Scholar]
- Moustafine, R.I.; Margulis, E.B.; Sibgatullina, L.F.; Kemenova, V.A.; Van der Mooter, G. Comparative evaluation of interpolyelectrolyte complexes of chitosan with Eudragit® L100 and Eudragit® L100-55 as potential carriers for oral controlled drug delivery. Eur. J. Pharm. Biopharm. 2008, 70, 215–225. [Google Scholar] [CrossRef]
- Patil, J.S.; Marapur, S.C.; Gurav, P.B.; Banagar, A.V. Ionotropic gelation and polyelectrolyte complexation technique: Novel approach to drug encapsulation. In Handbook of Encapsulation and Controlled Release; Taylor & Francis Group: Boca Raton, FL, USA, 2016; pp. 273–296. [Google Scholar]
- Badawy, M.E.I.; Taktak, N.E.M.; Awad, O.M.; Elfiki, S.A.; Abou El-Ela, N.E. Preparation and Characterization of Biopolymers Chitosan/Alginate/Gelatin Gel Spheres Crosslinked by Glutaraldehyde. J. Macromol. Sci. Part B 2017, 56, 359–372. [Google Scholar] [CrossRef]
- Lima, A.; Sousa, H.; Monteiro, J.; Azevedo, R.; Medeiros, R.; Seabra, V. Genetic polymorphisms in low-dose methotrexate transporters: Current relevance as methotrexate therapeutic outcome biomarkers. Pharmacogenomics 2014, 15, 1611–1635. [Google Scholar] [CrossRef] [PubMed]
- van der Vegt, N.F.; Nayar, D. The hydrophobic effect and the role of cosolvents. J. Phys. Chem. 2017, 121, 9986–9998. [Google Scholar] [CrossRef]
- Akagi, T.; Baba, M.; Akashi, M. Preparation of nanoparticles by the self-organization of polymers consisting of hydrophobic and hydrophilic segments: Potential applications. Polymer 2007, 48, 6729–6747. [Google Scholar] [CrossRef]
- Smith, R.A.; Walker, R.C.; Levit, S.L.; Tang, C. Single-Step Self-Assembly and Physical Crosslinking of PEGylated Chitosan Nanoparticles by Tannic Acid. Polymer 2019, 11, 749. [Google Scholar] [CrossRef] [PubMed]
- Rampino, A.; Borgogna, M.; Blasi, P.; Bellich, B.; Cesáro, A. Chitosan nanoparticles: Preparation, size evolution and stability. Int. J. Pharm. 2013, 455, 219–228. [Google Scholar] [CrossRef]
- Kaloti, M.; Bohidar, H.B. Kinetics of coacervation transition versus nanoparticle formation in chitosan–sodium tripolyphosphate solutions. Colloids Surf. B Biointerfaces 2010, 81, 165–173. [Google Scholar] [CrossRef]
- Barbosa, A.I.; Costa Lima, S.A.; Reis, S. Development of methotrexate loaded fucoidan/chitosan nanoparticles with anti-inflammatory potential and enhanced skin permeation. Int. J. Biol. Macromol. 2019, 124, 1115–1122. [Google Scholar] [CrossRef]
- Jonassen, H.; Kjoniksen, A.L.; Hiorth, M. Stability of chitosan nanoparticles cross-linked with tripolyphosphate. Biomacromolecules 2012, 13, 3747–3756. [Google Scholar] [CrossRef]
- Costa, P.; Sousa, L. Modeling and comparison of dissolution profiles. Eur. J. Pharm. Sci. 2001, 13, 123–133. [Google Scholar] [CrossRef]
- Korsmeyer, R.W.; Gurny, R.; Doelker, E.; Buri, P.; Peppas, N.A. Mechanisms of solute release from porous hydrophilic polymer. Int. J. Pharm. 1983, 15, 25–35. [Google Scholar] [CrossRef]
- Hancoc, K.B.; Zografi, G. The relationship between the glass transition temperature and the water content of amorphous pharmaceutical solids. Pharm. Res. 1994, 11, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Peppas, N.A.; Sahlin, J.J. A simple equation for the description of solute release. III. Coupling of diffusion and relaxation. Int. J. Pharm. 1989, 57, 169–172. [Google Scholar] [CrossRef]
- Unagolla, J.M.; Jayasuriya, A.C. Drug transport mechanisms and in vitro release kinetics of vancomycin encapsulated chitosan-alginate polyelectrolyte microparticles as a controlled drug delivery system. Eur. J. Pharm. Sci. 2018, 114, 199–209. [Google Scholar] [CrossRef] [PubMed]
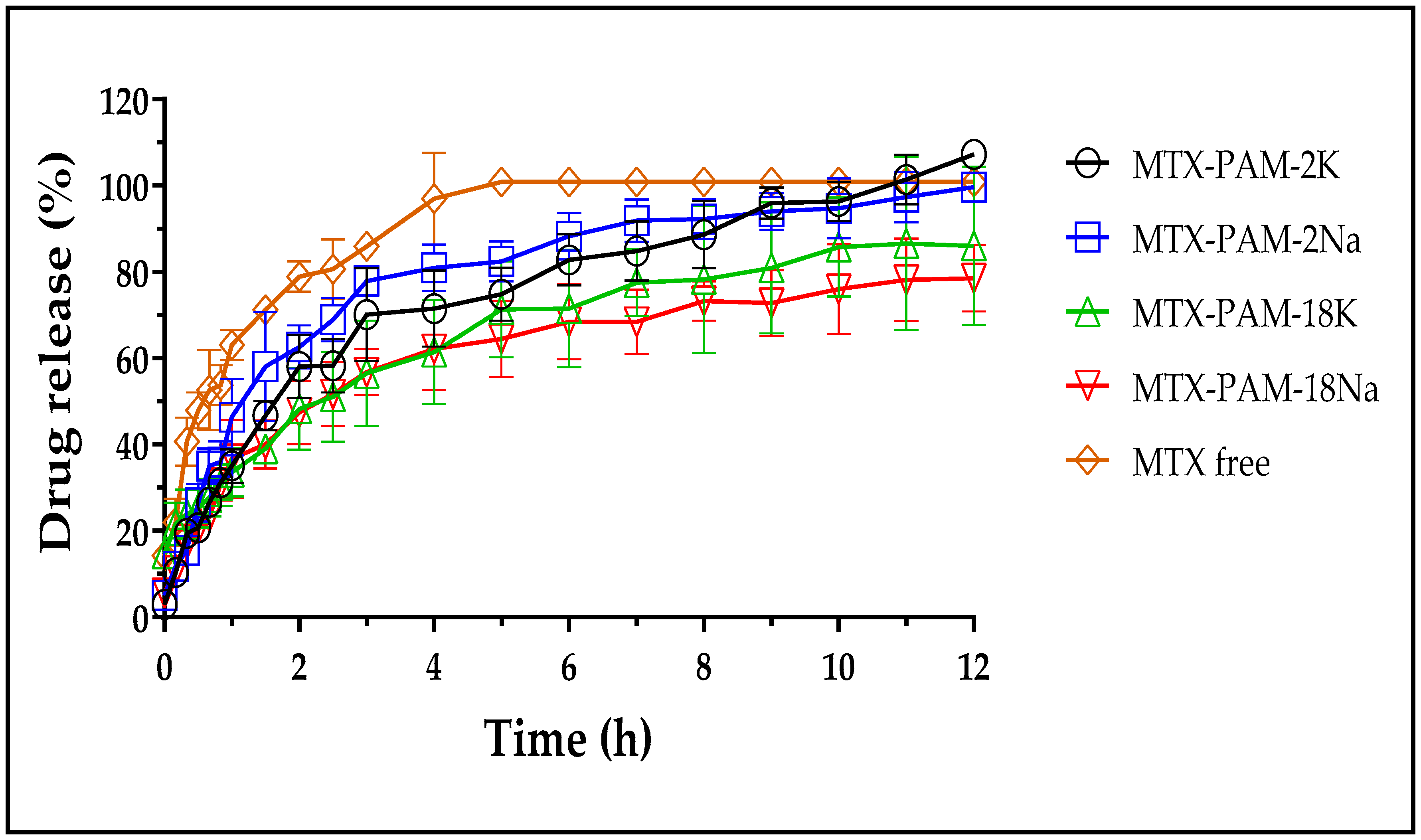
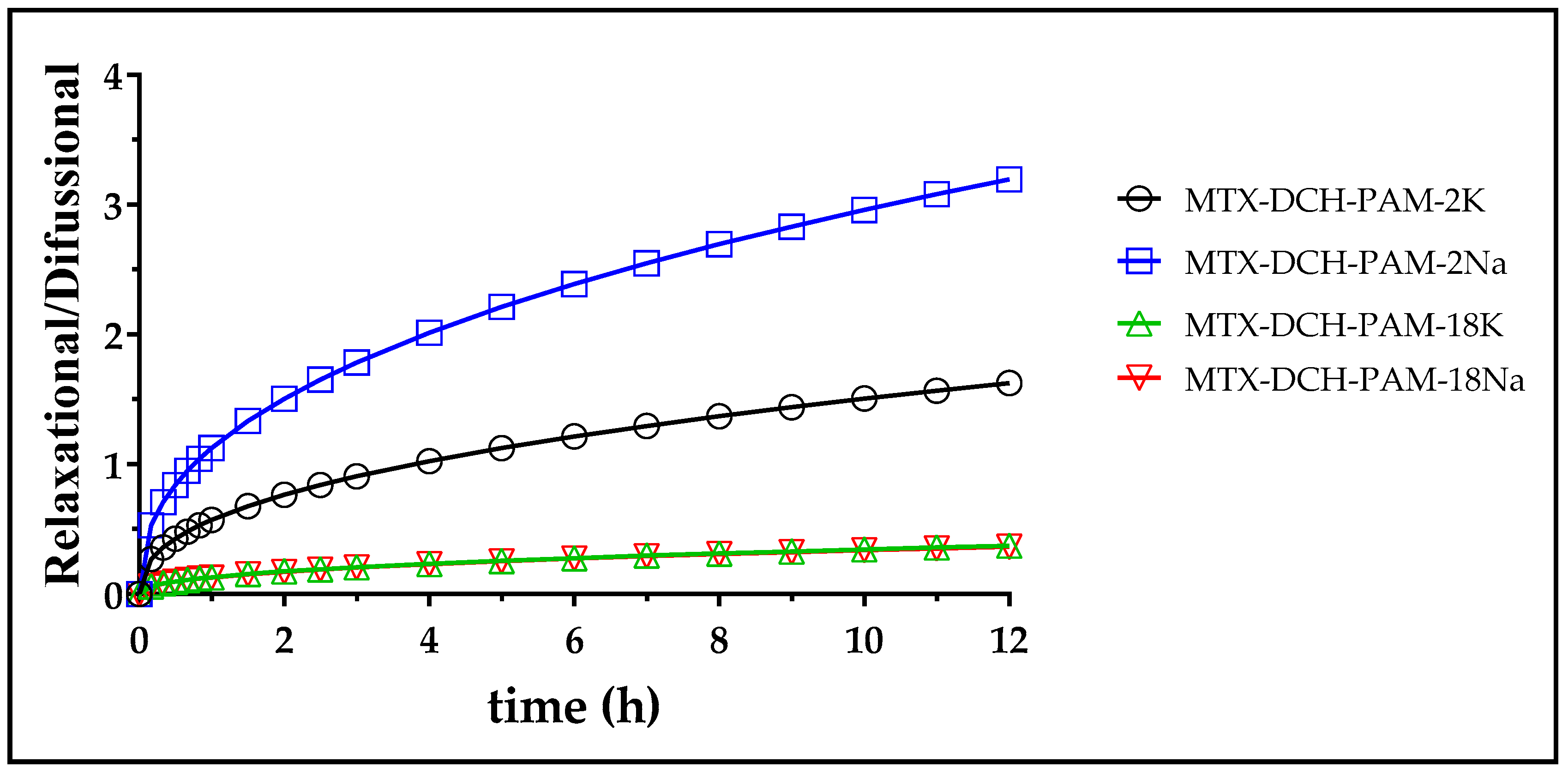
| Polymer | Free MTX | MTX-PAM-2K | MTX-PAM-2Na | MTX-PAM-18K | MTX-PAM-18Na |
|---|---|---|---|---|---|
| MRT | 0.8 | 1.2 | 1.1 | 1.3 | 1.3 |
| Release Profiles | f2 Factor |
|---|---|
| MTX-PAM-2K-MTX-PAM-2Na | 43.3 |
| MTX-PAM-18K- MTX-PAM-18Na | 55.1 |
| MTX-PAM-2Na- MTX-PAM-18K | 47.9 |
| MTX-PAM-2Na- MTX-PAM-18Na | 65.5 |
| MTX-PAM-2K- MTX-PAM-18K | 47.2 |
| MTX-PAM-2K- MTX-PAM-18Na | 46.6 |
| Polymer | Order One | Higuchi | Korshmeyer-Peppas with Burst | Peppas-Sahlin | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| k | r2 | k | r2 | K | n | b | r2 | kd | kr | r2 | |
| Free MTX | 0.72 | 0.9479 | 40.8 | 0.9688 | - | - | - | - | - | - | - |
| MTX-PAM-2K | 0.30 | 0.9669 | 30.5 | 0.9748 | 0.33 | 0.66 | 0.02 | 0.9818 | 0.19 | 0.16 | 0.9803 |
| MTX-PAM-2Na | 0.35 | 0.9136 | 28.5 | 0.9171 | 0.40 | 0.85 | 0.04 | 0.9745 | 0.08 | 0.35 | 0.9687 |
| MTX-PAM-18K | 0.16 | 0.9821 | 23.3 | 0.9824 | 0.18 | 0.76 | 0.15 | 0.9916 | 0.36 | 0.00 | 0.8344 |
| MTX-PAM-18Na | 0.12 | 0.9262 | 22.3 | 0.9482 | 0.28 | 0.61 | 0.01 | 0.9576 | 0.24 | 0.08 | 0.9704 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ciro, Y.; Rojas, J.; Alhajj, M.J.; Carabali, G.A.; Salamanca, C.H. Production and Characterization of Chitosan–Polyanion Nanoparticles by Polyelectrolyte Complexation Assisted by High-Intensity Sonication for the Modified Release of Methotrexate. Pharmaceuticals 2020, 13, 11. https://doi.org/10.3390/ph13010011
Ciro Y, Rojas J, Alhajj MJ, Carabali GA, Salamanca CH. Production and Characterization of Chitosan–Polyanion Nanoparticles by Polyelectrolyte Complexation Assisted by High-Intensity Sonication for the Modified Release of Methotrexate. Pharmaceuticals. 2020; 13(1):11. https://doi.org/10.3390/ph13010011
Chicago/Turabian StyleCiro, Yhors, John Rojas, Maria J. Alhajj, Gustavo A. Carabali, and Constain H. Salamanca. 2020. "Production and Characterization of Chitosan–Polyanion Nanoparticles by Polyelectrolyte Complexation Assisted by High-Intensity Sonication for the Modified Release of Methotrexate" Pharmaceuticals 13, no. 1: 11. https://doi.org/10.3390/ph13010011
APA StyleCiro, Y., Rojas, J., Alhajj, M. J., Carabali, G. A., & Salamanca, C. H. (2020). Production and Characterization of Chitosan–Polyanion Nanoparticles by Polyelectrolyte Complexation Assisted by High-Intensity Sonication for the Modified Release of Methotrexate. Pharmaceuticals, 13(1), 11. https://doi.org/10.3390/ph13010011







