3. Processing EMG Signal Features
3.1. Feature Extraction
In the initial phase of the analysis, electromyographic signals acquired from the bionic hand were subjected to a feature extraction process. The purpose of this step was to identify salient signal characteristics that could be utilized as input features for subsequent processing and analysis.
Depending on the computational methodology, the extracted features can be categorized into three distinct groups: features obtained directly from the raw signal, features derived from the signal envelope, and features extracted through signal decomposition techniques (
Table 2).
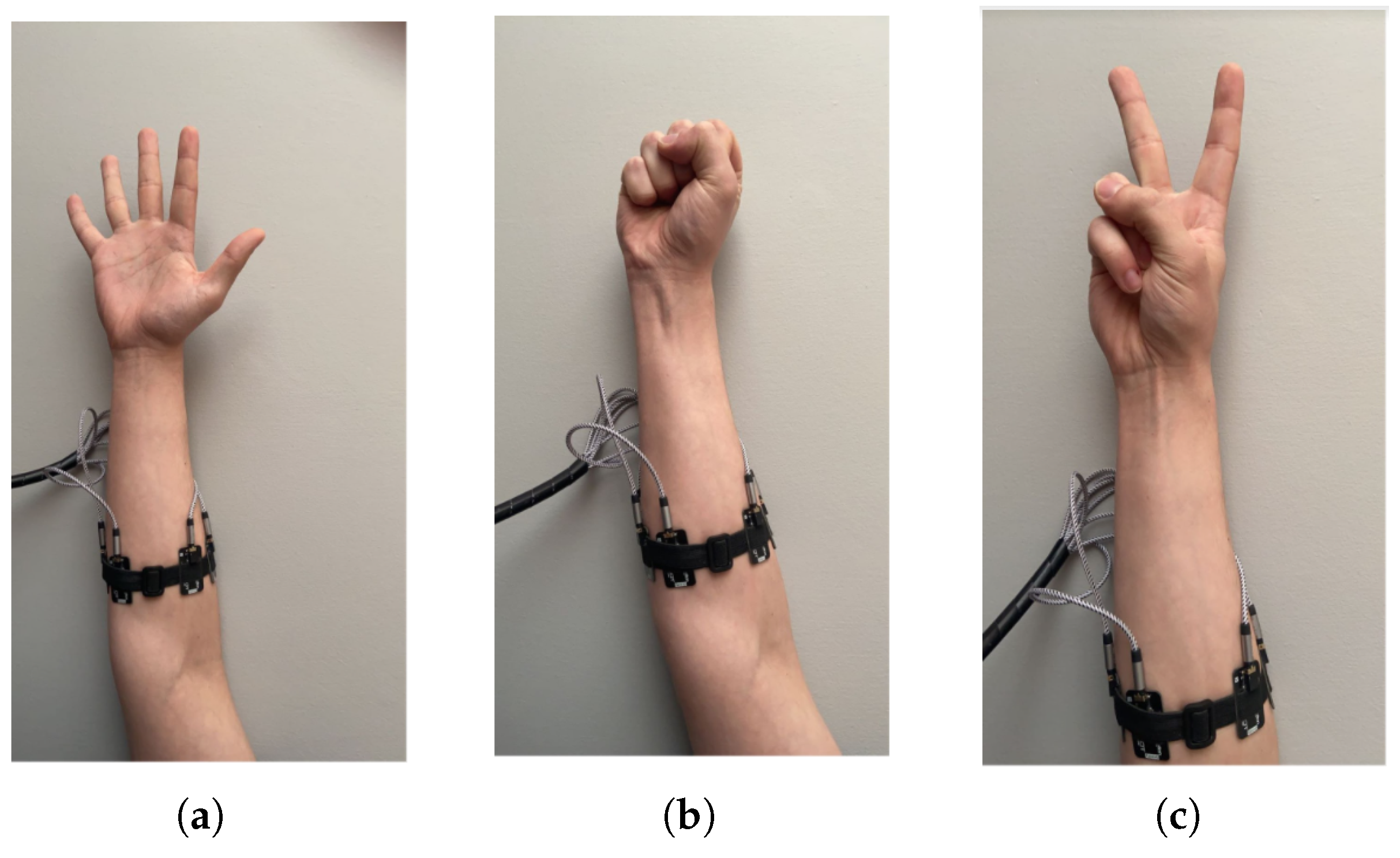
In this study, EMG signals were acquired using an eight-channel measurement system. Data acquisition was performed for 18 defined factors, each representing different states or tasks. Consequently, eight data channels were obtained for each of the 18 factors, resulting in a data set comprising 144 scalar features derived from unprocessed EMG signals.
Additionally, 16 additionalnal scalar features were incorporated into the set of features. These additional features represent the mean and standard deviation calculated from the upper-range envelope of the signal for each of the eight individual channels.
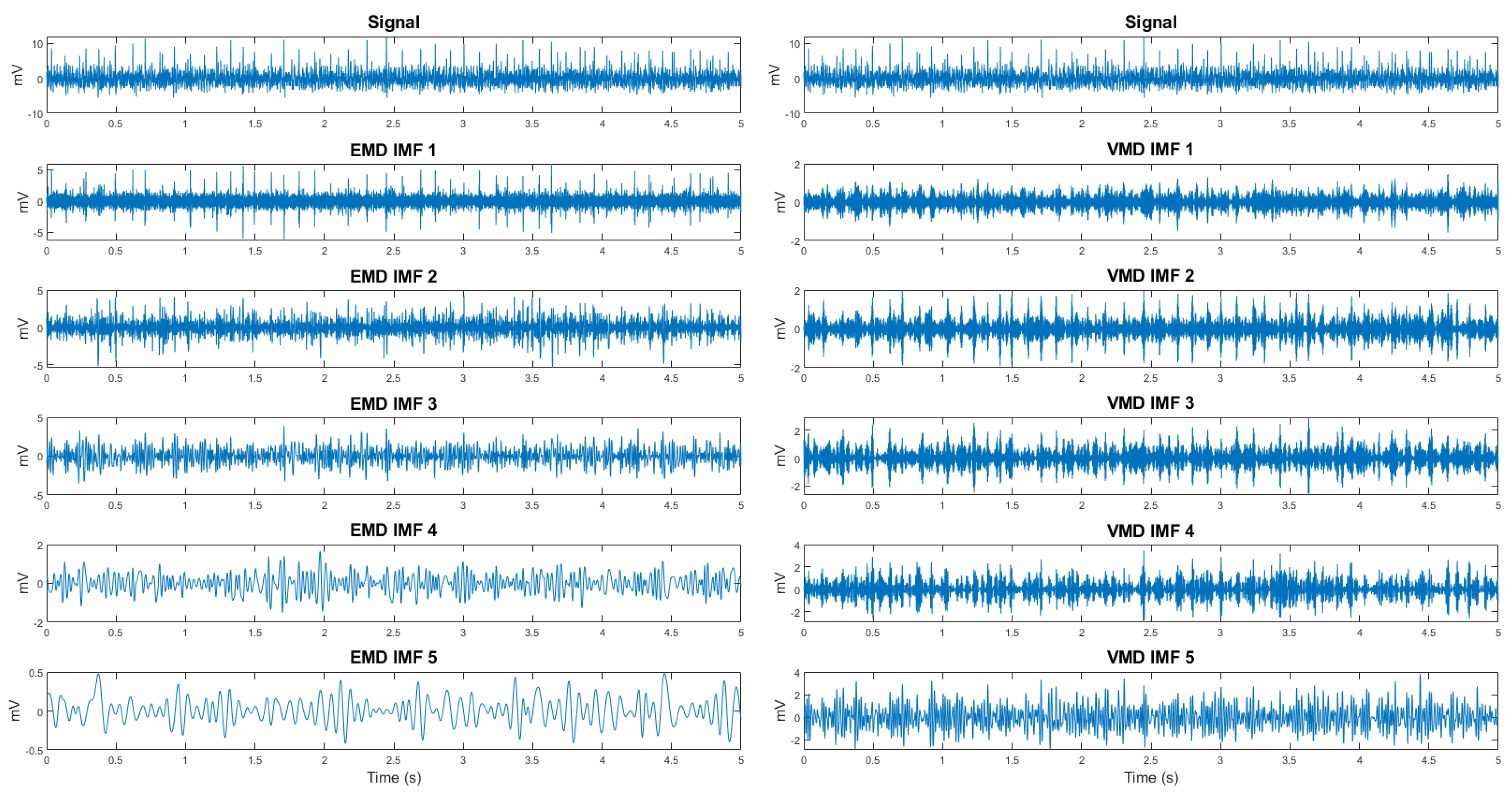
An additional set of features was derived from the decomposition (
Figure 7) of the signal using Empirical Mode Decomposition (EMD) [
18] and Variational Mode Decomposition (VMD) [
19]. From the EMD decomposition, the seven fundamental intrinsic mode functions (IMFs) were selected, while five modes were chosen from the VMD decomposition for each of the eight electromyographic (EMG) signal channels. This resulted in a total of 96 components. Subsequently, the energy and approximate entropy were calculated for each of these components, resulting in a final set of 192 novel features.
The comprehensive feature set consisted of 352 distinct scalar features, encompassing a diverse range of information extracted from EMG signals. This set included 144 features derived from unprocessed EMG data, 16 features representing the mean and standard deviation of the upper-range signal envelope, and 192 features obtained through EMD and VMD analysis. These features collectively aimed to capture the multifaceted characteristics of muscle activity, providing a robust foundation for subsequent analytical and modeling procedures.
3.2. Data Preprocessing
The data set, containing 362 instances, was partitioned into training, validation, and test subsets with a distribution of 78% for training, 11% for validation, and 11% for testing. The subsets were randomly selected, maintaining the representation of all five gesture classes within each subset.
Due to the significant class imbalance observed within the gesture dataset, the Receiver Operating Characteristic Area Under the Curve (ROC AUC) was used as a primary evaluation metric for predictive models. This metric was selected to provide a robust assessment of model performance, where it is particularly useful in scenarios where traditional accuracy measures may be misleading due to uneven class distributions. The ROC AUC assesses the model’s ability to discriminate between classes across various threshold settings.
3.3. Building Models
Research has consistently shown that advanced machine learning algorithms can achieve high accuracy in classifying electromyographic (EMG) signals for the identification of specific hand movements [
20]. Building on this, investigations were conducted to evaluate various machine learning models, specifically emphasizing regularized and dimensionality-reduced logistic regression models, alongside neural network classifiers. While the classification performance of the selected methods was largely comparable, neural network classifiers demonstrated a marginal superiority. Nevertheless, logistic regression models present distinct advantages, including notable resistance to overfitting, simplicity of implementation, computational efficiency, and enhanced interpretability. Collectively, these attributes render logistic regression a highly appropriate solution for controlling bionic prostheses.
Among the logistic regression models, regularized variants employing L1 (LASSO—Least Absolute Shrinkage and Selection Operator [
21]), Ridge Regression (L2) [
22] and Elastic Net [
23] penalties were evaluated. These regularization techniques were implemented to mitigate the risk of overfitting, particularly in scenarios with a high number of input features relative to the sample size. By adding a penalty term to the loss function, these methods constrain the magnitude of the coefficient estimates. This helps in reducing the impact of multicollinearity.
The LASSO method modifies the standard cost function (typically the sum of squared residuals for linear regression or negative log-likelihood for logistic regression) by adding an L1 penalty (L1 norm) to the sum of the absolute values of the regression coefficients.
where
is the observed value of the dependent variable for the i-th observation;
is the vector of independent variables for the i-th observation;
is the intercept term;
is the vector of regression coefficients for the predictors;
n is the number of observations;
p is the number of predictors;
(lambda) is the regularization parameter (penalty strength), controlling the degree of coefficient shrinkage, where a larger indicates a stronger penalty;
is the L1 penalty, representing the sum of the absolute values of the coefficients.
This formula can be simplified to minimize a cost function (also known as a loss function). This cost function combines the prediction error with a regularization term as defined below:
For the LASSO regularization, a penalty fraction (
) of 0.001 was applied.
Elastic Net combines the L1 penalty of LASSO with an L2 penalty (sum of squared coefficients,
), which is characteristic of Ridge Regression. Elastic Net offers a balance between feature selection and coefficient shrinkage, potentially outperforming either L1 or L2 alone in situations with groups of correlated variables.
where
, , , , n, and p have the same meaning as in LASSO;
(lambda) is the regularization parameter, controlling the overall penalty strength;
(alpha) is the mixing parameter, in the range of ;
If , Elastic Net becomes LASSO (L1 penalty only);
If , Elastic Net becomes Ridge (L2 penalty only);
For , Elastic Net is a combination of both penalties;
is the L1 penalty;
is the L2 penalty (sum of squared coefficients).
Consistent with other regularization techniques, Elastic Net regularization also seeks to minimize a cost function, whose formulation can likewise be simplified as follows:
Elastic Net regularization was performed with an
value of 0.99 and a penalty fraction (
) of 0.001.
The performance of these regularized logistic regression models was compared to assess their suitability for the specific classification task, considering factors such as predictive accuracy, model complexity, and interpretability.
In addition to the regularization methods, a feature selection technique employing Recursive Feature Elimination (RFE [
24]) was also implemented. RFE is a wrapper-type feature selection algorithm that operates by iteratively fitting the model and removing the least important features based on the model’s coefficient magnitudes (in the case of linear models like logistic regression). This process is repeated until the desired number of features is reached. The rationale behind employing RFE was to identify the most informative subset of features for the classification task, potentially improving model parsimony, reducing computational cost, and enhancing generalization performance by removing noisy or redundant variables. A modification to the RFE method was introduced, wherein the selected feature subset maximized the minimum F1-score obtained for any category. Specifically, the objective was to find the subset of features that maximized the F1 score as follows:
where F1c represents the F1-score for category c.
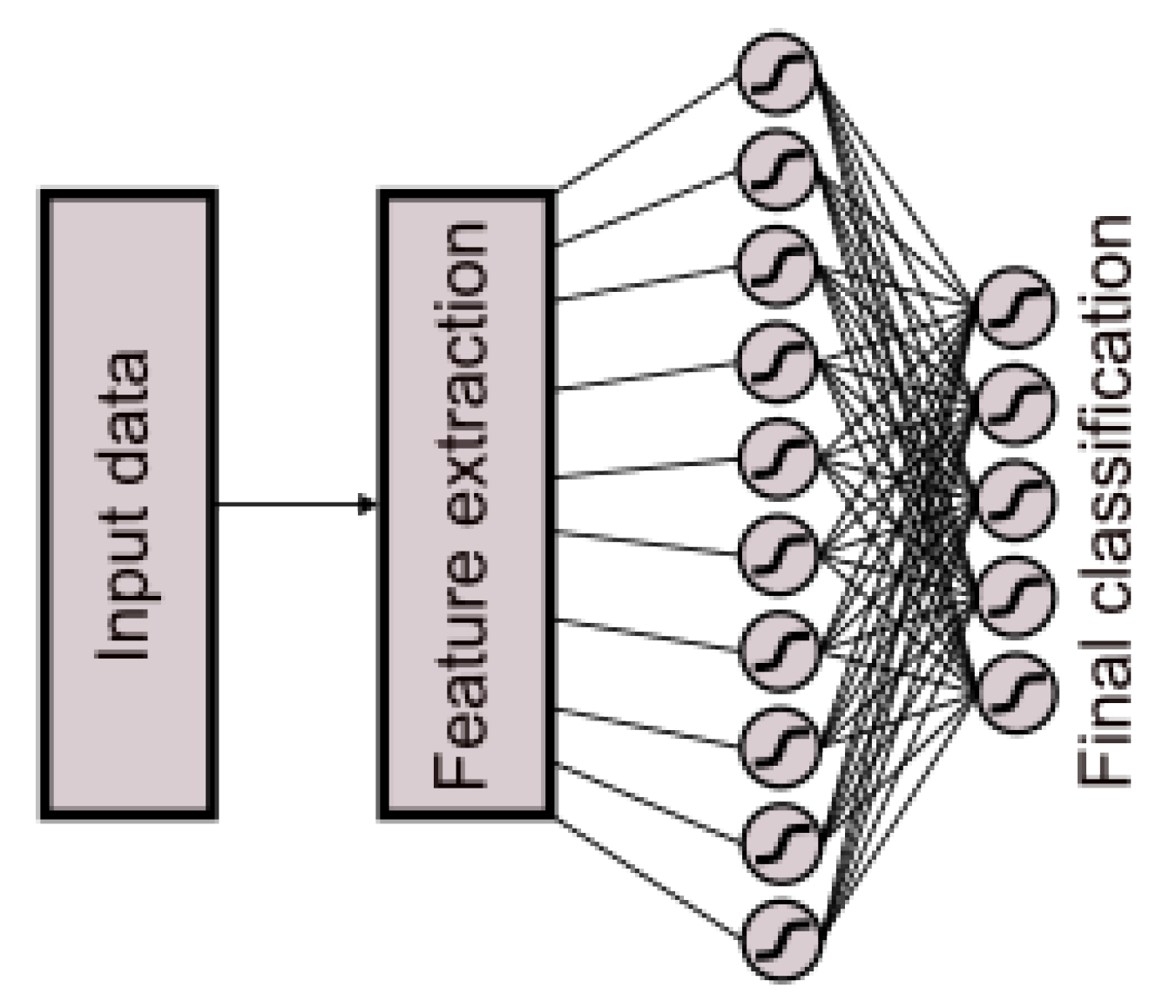
Within the experimental framework, neural network classifier models, in addition to regression models, exhibited significant performance. Therefore, a neural network classifier model was implemented, comprising 10 neurons employing the tanh activation function in the hidden layer and 5 neurons using the tanh activation function in the output layer (
Figure 8).
In JMP, hyperparameters were primarily determined through internal validation mechanisms within the platform, such as cross-validation or validation set performance, which JMP automatically optimizes. This approach allows the software to iteratively search for and select the parameter values that yield the best model performance based on the chosen validation metric. JMP, like many advanced statistical and analytical software packages, typically does not disclose the specific fixed name of the optimization algorithm used “under the hood” for its artificial neural networks (ANN). Instead, it focuses on delivering optimized results and offers the user certain configuration options that influence the optimization process.
Although direct access to neural network hyperparameters was unavailable, a significant benefit emerged from the unified testing environment provided by JMP. This enabled a more robust and comparable evaluation of the machine learning algorithms.
Furthermore, an exploration of signal classification capabilities was conducted employing GRU and LSTM architectures applied to unprocessed signal data. However, the insufficient sample size within the data set limited the ability to establish the effectiveness of these network models.
4. Implementation Simple Bionic Hand
Model of Bionic Hand—Hardware
The bionic hand model (
Figure 9 and
Figure 10) was made using a 3D printer (Prusa i3 MK3S+, Prague, Czech Republic) equipped with the Multi Material 2 upgrade. All components of the model were designed by Gael Langevin [
25], who made them publicly available on the Thingiverse platform under a Creative Commons license.
Individual parts of the prototype were printed using Spectrum PETG filament (1.75 mm diameter, Silver Star color). This material was selected due to its favorable mechanical properties, including high durability, flexibility, and resistance to moisture-induced degradation. The assembled components were joined using appropriate fasteners and adhesive to ensure structural integrity.
The bionic hand is actuated by six Tower Pro MG996R servo motors, each with an angular range of 180°, and controlled via a PCA9685 PWM driver board (Adafruit 815) connected to the main controller. The first servo is responsible for wrist articulation, while the remaining five control the individual fingers by tensioning or releasing braided fishing lines.
To ensure reliable actuation and minimize elastic deformation, high-strength braided fishing line (PowerPro Spectra, 0.76 mm diameter, tensile strength 90 kg) was used. Each line was routed through dedicated guide holes to prevent tangling. In order to maintain consistent tension in the tendons of each finger, return springs were incorporated into the tensioning mechanism.
The servo control system was integrated with the same LPC1347 microcontroller board that was used during the initial stages for surface electromyography (sEMG) signal acquisition, enabling a compact and unified hardware architecture.
Each servo motor was individually calibrated to define precise initial and terminal positions for each finger. Finger flexion is achieved by varying the servo positions, resulting in proportional joint articulation. When all servo motors are set to their zero-angle positions, the hand assumes a fully open posture, which serves as the system’s reference configuration.
5. Results and Discussion
The experiment assessed the effectiveness of recognizing five distinct hand gestures using both regularized logistic regression models and artificial neural networks (ANNs). These two methods demonstrated superior classification performance compared to other initially evaluated algorithms, including k-Nearest Neighbors (k-NN), Support Vector Machines (SVMs), Random Forests, and Decision Trees.
The analysis was based on complete 5-second signal sequences, from which scalar features were subsequently extracted. The use of full-length sequences allowed for improved model fitting to representative patterns in the EMG signals, including potential artifacts that could arise during the real-world operation of the bionic hand.
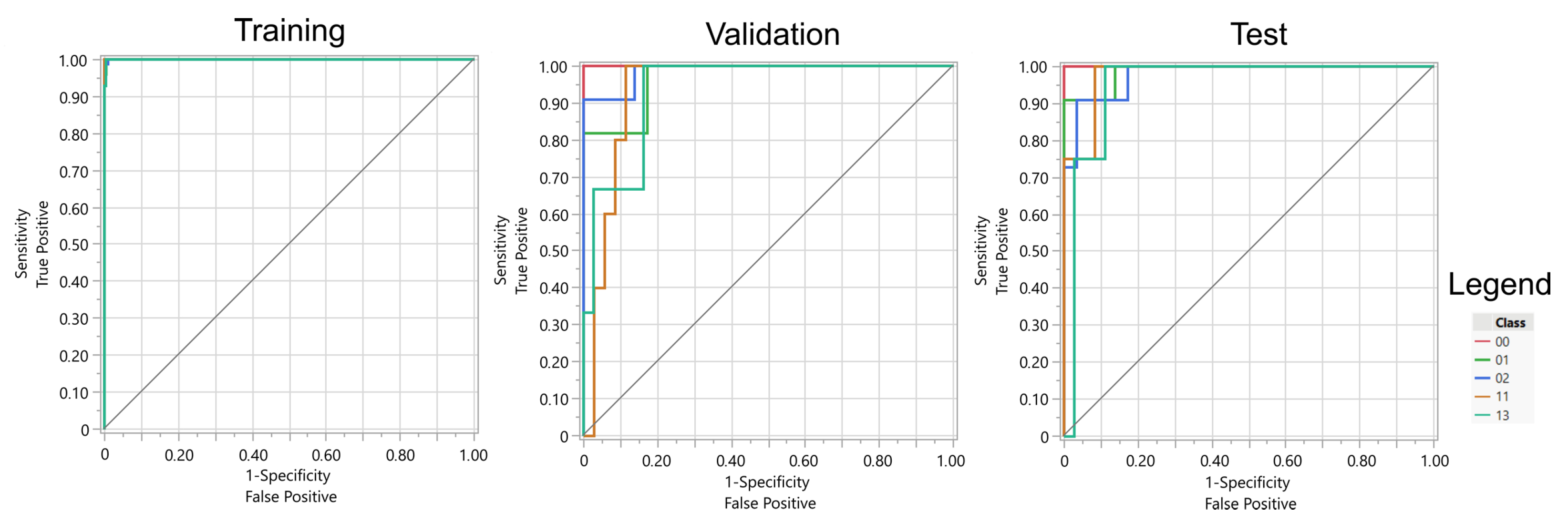
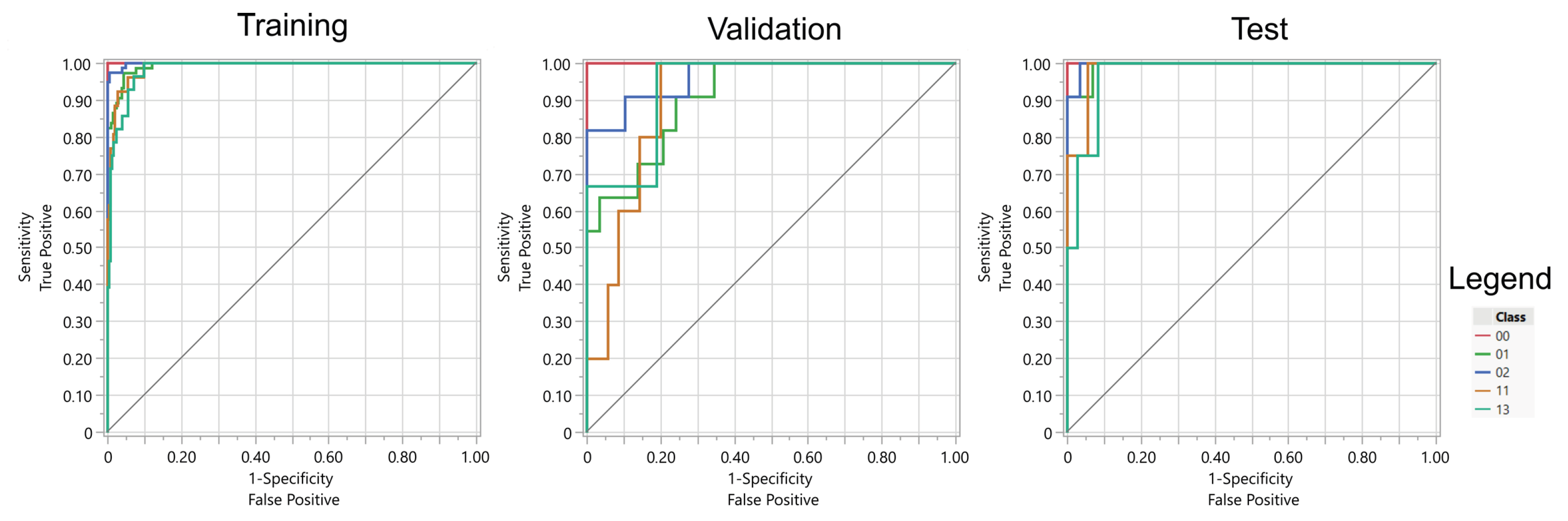
The results indicate that both artificial neural networks (ANNs) employing sigmoidal activation functions and regularized logistic regression models achieved comparable performance (see
Table 3 and
Table 4). Neural networks exhibited a slightly greater tendency to overfit the training data. Additionally, the ANNs utilized the full set of available features, whereas the regularized models selected a subset—103 features in the case of LASSO regression and 135 features with Elastic Net regularization.
Overall, the most favorable results were achieved through the application of logistic regression with Elastic Net regularization. Considering the lowest classification accuracies across gestures, LASSO and Elastic Net yielded the highest values. It is also pertinent to note that the performance of the ANNs varied depending on the local minimum of the loss function identified by the optimization algorithm. The table presents the optimal outcome attained across multiple experimental runs, which consequently positions the results obtained for the logistic regression methods in a more advantageous perspective.
The absence of a significant advantage of ANNs over regression methods on the test data suggests that Elastic Net or LASSO are the recommended models for practical application (see
Figure 11 and
Figure 12). Furthermore, the high generalization capabilities inherent in regularized regression models, stemming from their propensity to avoid overfitting the data, further support their adoption. Moreover, regression models offer transparency, ease of interpretation, and straightforward implementation on the bionic hand’s control system. These models also possess a significantly smaller computational footprint, thereby enabling a more rapid response of the bionic hand to EMG signals.
A primary limitation of employing neural networks in this study lies in their substantial demand for diverse and extensive datasets, the acquisition of which necessitates considerable time and financial resources without providing an absolute guarantee of successful implementation.
The ElasticNet model, as demonstrated in the provided example, incorporated nearly all features enumerated in
Table 1 during its construction. Notably, the features Kurtosis, Approximate Entropy, and Energy were entirely excluded from the model training process. All other features participated to a greater or lesser extent in the training process of the ElasticNet model.
This study employed numerous features for constructing the Elastic Net regression model, from which several statistically significant ones were identified. The assessment of statistical significance was based on rejecting the null hypothesis of a zero regression coefficient (with a p-value below 0.05).
The most significant predictors, exhibiting non-zero coefficients in the model, include the following:
Signal skewness in channel 3.
Mean signal value in channel 1.
Entropy of the first intrinsic mode function (IMF) from VMD decomposition in channel 8.
Mean signal value in channel 4.
Mean signal frequency in channel 6.
Energy of the fifth IMF from EMD decomposition in channel 4.
Correlation Dimension in channel 3.
Total Harmonic Distortion (THD) in channel 6.
These findings indicate that the most influential factors in the model were derived from data originating in channels 1, 3, 4, 6, and 8 (
Figure 1). A detailed description of all utilized features is beyond the scope of this section. The diverse nature of the identified features—ranging from statistical moments (skewness, mean) to spectral (frequency, THD) and decomposition-based (IMF entropy, energy) as well as non-linear dynamics (Correlation Dimension) metrics—underscores the complexity of the underlying process and the necessity of a multi-faceted approach to feature extraction.
In the LASSO regression model, twelve predictors demonstrated statistical significance, with their p-values falling below the 0.05 threshold. The list of these significant predictors is as follows (those also identified in the Elastic Net model are italicized):
Mean upper envelope of the signal in channel 5.
Correlation Dimension in channel 2.
Signal skewness in channel 3.
Entropy of the first intrinsic mode function (IMF) from VMD decomposition in channel 8.
Mean signal value in channel 1.
Mean signal frequency in channel 6.
Shape Factor in channel 1.
Correlation Dimension in channel 3.
Mean signal value in channel 4.
Energy of the fifth IMF from EMD decomposition in channel 4.
Energy of the first IMF from VMD decomposition in channel 6.
Total Harmonic Distortion (THD) in channel 6.
The identification of these statistically significant predictors in the LASSO model highlights the most influential features contributing to the model’s predictive power. The substantial overlap with features selected by the Elastic Net model (8 out of 12) suggests a robust set of key indicators across both regularization techniques. This consistency underscores the reliability of these particular features in capturing the underlying patterns within the data. Furthermore, the inclusion of additional unique features by the LASSO model, such as the mean upper envelope in channel 5 and the form factor in channel 1, indicates that while core predictive elements are shared, each regularization method may prioritize slightly different aspects of the data—potentially due to their distinct penalty functions (L1 for LASSO’s sparsity vs. combined L1/L2 for Elastic Net’s grouping effect).
6. Conclusions
In summary, although artificial neural networks (ANNs) exhibited comparable or, in certain cases, marginally superior performance in gesture classification, several considerations advocate for the use of regularized logistic regression models—specifically Elastic Net and LASSO—in the practical implementation of bionic hand control based on EMG signals. The absence of a definitive advantage of ANNs on the test dataset, combined with their intrinsic dependence on large and diverse training datasets—whose acquisition is both resource-intensive and does not ensure optimal performance—constitutes a significant practical limitation.
Conversely, regularized regression models constitute a compelling alternative due to their superior generalization capabilities, which reduce the risk of overfitting—an issue more prominently observed in the evaluated artificial neural networks. Moreover, the intrinsic transparency, interpretability, and ease of implementation of these models within the bionic hand control system represent significant advantages. Their lower computational demands enable more rapid processing, thereby facilitating a potentially critical real-time response of the prosthetic limb to the user’s intended gestures—an essential requirement for practical applications.
Conclusions drawn from a review of advancements in robotic upper-limb prostheses [
26] indicate that a major limitation of machine learning methods is their black-box nature coupled with high computational demands. The application of effective models based on regularized logistic regression addresses these fundamental challenges inherent to conventional ML approaches.
Considering these factors—particularly the trade-offs between performance, data requirements, interpretability, implementability, and real-time responsiveness—regularized logistic regression methods, notably Elastic Net and LASSO, emerge as a more pragmatic and potentially efficacious choice for controlling bionic hands in this context.
The classification accuracy of gestures was substantially improved compared to the initial study [
11], owing to an expanded feature set employed to characterize EMG signals. An additional advantage of this increased feature dimensionality was the feasibility of applying efficient logistic regression techniques that demonstrated competitive performance relative to artificial neural networks (ANNs). Furthermore, the evaluation methodology for gesture classification was revised from accuracy-based metrics [
11] to the Receiver Operating Characteristic (ROC) curve and Area Under the Curve (AUC). This shift to more robust model quality indicators provides a better representation of classification performance, especially when addressing imbalanced datasets.
A detailed analysis of feature extraction methods contributing to the development of controllable prosthetic hands has been conducted [
27]. Beyond applications in computer or mobile device gesture control, research in this domain holds potential for prostheses addressing hand hemimelia. Such prosthetic systems could be further enhanced by incorporating modules supported by Inertial Measurement Units (IMUs) to detect hand rotation.
All experiments were performed using MATLAB 2024b [
16], JMP Student Edition 18.2.0 [
17], and Python 3.13 software, with JMP Student Edition 18.2.0 additionally employed for ROC AUC visualization.