1. Introduction
SF
6 gas, known for its exceptional electrical insulation and arc-quenching properties, has been extensively utilized in high-voltage equipment such as gas-insulated switchgear (GIS), gas-insulated transmission lines (GILs), and high-voltage circuit breakers. However, in recent years, the global power system has undergone significant transformations, with a growing emphasis on the adoption of environmentally friendly materials. Given that SF
6 possesses an extremely high global warming potential (GWP), approximately 23,900 times greater than that of CO
2 [
1,
2], the prevailing trend is to minimize or eliminate its usage. In addition, the tendency of SF
6 to liquefy at low temperatures imposes constraints on its practical application in high-latitude regions. To date, no gas has been identified that can fully substitute for SF
6 in terms of both its insulation and arc-quenching performance. The most effective strategy to address this issue is to reduce its usage within power systems. Employing SF
6-based gas mixtures represents a viable solution [
3,
4]. Commonly, SF
6 is blended with N
2, CO
2, CF
4 or certain rare gases. Notably, CF
4 exhibits a GWP value that is only one-third that of SF
6, with a liquefaction temperature under standard atmospheric pressure as low as −128 °C. Additionally, CF
4 demonstrates strong electronegativity, a superior arc-quenching capability, and favorable thermodynamic properties [
5]. Research indicates that when CF
4 and SF
6 are mixed in a specific ratio, the resulting mixture exhibits an elevated liquefaction temperature while preserving excellent insulation properties [
6]. To date, the SF
6/CF
4 gas mixture has been extensively utilized as an insulating medium for high-voltage circuit breakers in extremely cold regions [
7,
8,
9].
In SF
6-based, high-voltage, sealed electrical equipment, a significant proportion of insulation failures are associated with foreign substances, such as metal particles. These metal particles primarily result from mechanical impacts, vibrations, and friction during the processes of manufacturing, installation, transportation, and operation of the equipment. At accident sites, millimeter-sized metal particles are frequently observed adhering to the equipment surfaces, along with a limited number of micrometer-sized or smaller metal dust particles [
10]. Such particles may either be in direct contact with components or remain in a suspended state, leading to partial discharge phenomena. Partial discharge progressively degrades the insulation of equipment, and under severe conditions, it may evolve into a flashover, ultimately compromising the insulation performance of high-voltage equipment [
11]. Consequently, conducting in-depth research on the partial discharge phenomenon induced by metal particles is crucial for comprehending the progression of partial discharge, refining diagnostic and mitigation strategies, and improving the safety and reliability of power equipment. To achieve a more comprehensive understanding of the occurrence and evolution mechanisms of partial discharge induced by metal particles in SF
6, researchers have carried out a series of systematic experiments. These experiments primarily investigated the behavior of free metal particles under the following conditions: resting on insulating material surfaces, adhering to metal shell surfaces, or existing in a suspended or freely moving state [
12,
13,
14]. When free metal particles make contact with high-voltage electrodes or grounding terminals, they form an equipotential structure, which facilitates the simplification of simulation processes and enhances the stability of the discharge phenomenon. However, when metal particles are in a suspended state, their potential varies in accordance with the spatial electric potential. The metal particles and the surrounding gas medium collectively form a capacitive system. In terms of millimeter-scale metal particles, they typically can only accommodate charge quantities on the order of picocoulombs [
15]. The transient charging and discharging process induced by partial discharge ceases within an extremely short duration due to the minuscule charge transfer and relaxation times in the sub-nanosecond to nanosecond range. This characteristic imparts ultrafast time-response features to the partial discharge signals generated by free metal particles. Conventional detection techniques often struggle to fully capture these transient processes owing to bandwidth constraints. Furthermore, the weak signal amplitudes pose significant challenges in effectively differentiating them from background noise, which has hindered detailed investigations into the partial discharge characteristics and evolution mechanisms of millimeter-scale free metal particles. Additionally, partial discharge arises from the synergistic interactions of multiple physical processes, such as charge generation and transport, charge energy transfer, molecular excitation, and ionization. These processes are interdependent and mutually restrictive; thus, they increase the complexity of the system and render it difficult to characterize using macroscopic parameters obtained experimentally. Consequently, experimental approaches exhibit substantial limitations in elucidating the underlying discharge mechanisms.
Finite element simulation represents an effective approach for investigating the discharge process in SF
6 mixed gas. The fluid model, which is grounded in transport equations, is frequently employed. Based on the local field approximation assumption, this model can accurately capture the microscopic motion dynamics of charged particles with adequate precision [
16,
17,
18]. The reasonable adjustment of the parameters informed by experimental data and Boltzmann equation results establishes a solid foundation for simulation studies utilizing the fluid model [
19,
20,
21,
22]. However, fluid models are incapable of capturing the variations in specific substances and also struggle to adequately represent surface reactions on dielectrics and electrodes. Plasma fluid models that incorporate specific particle reactions [
23] have gained increasing application in gas discharge simulation studies in recent years. These models can capture the dynamics of specific particles within complex gas reactions [
24,
25,
26,
27] and more effectively address surface reactions at boundaries [
28]. Lijun Wang [
29] developed a plasma fluid model for discharge in non-uniform fields using COMSOL Multiphysics. Through numerical simulations, the spatiotemporal evolution of the electron density, positive ion density, negative ion density, and electric field intensity during the negative streamer discharge of SF
6/N
2 mixed gas was investigated. The effects of varying concentration ratios on the streamer development were examined; however, the fundamental mechanisms underlying their influence on the streamer discharge were not fully elucidated. Bin Luo [
25] conducted a simulation of the positive streamer discharge process. By modulating the applied voltage, the spatiotemporal evolution of the streamer propagation and attenuation was systematically analyzed. The accumulation of negative charges in the spatial domain suppressed the ionization of SF
6 and N
2 at the streamer head, consequently influencing the streamer’s propagation characteristics. Zhen Li [
15] also employed the plasma fluid model, with SF
6/N
2 mixed gas as the research medium, to analyze the propagation characteristics of positive streamers under rod–plate electrodes. Additionally, the study investigated the effects of varying the mixing ratios, pressures, and temperatures on the streamer thickness.
Currently, the investigation into the streamer discharge process of SF
6/N
2 mixed gas has reached a relatively mature stage, whereas research on the discharge phenomena of SF
6/CF
4 mixed gas remains limited. Further in-depth studies are urgently required in this field to comprehensively elucidate the associated discharge characteristics and underlying mechanisms. Dengming Xiao et al. [
30,
31,
32] performed a detailed computational analysis of the electronic transport parameters at room temperature. The research team, led by Jia Zhang and Hu Zhao [
33], derived the discharge parameters for 50%/50% SF
6/CF
4 mixed gas at one atmosphere pressure using two approximate methods to solve the Boltzmann equation. All prior studies on SF
6/CF
4 mixed gas have been conducted at the macroscopic level, without considering the impact of chemical reactions on discharge phenomena. In this paper, suspended free metal particles are investigated as the research subject, and the plasma fluid model is employed to explore the streamer phenomenon induced in the discharge space. Additionally, the spatiotemporal evolution characteristics of key parameters, such as the charged particle density and axial electric field strength, are analyzed under different mixing ratios.
3. Result Analysis
The spatial electron density distributions for two distinct concentrations of SF
6/CF
4 mixed gas at 0.6 ns are illustrated in
Figure 3. Metal particles are suspended within the discharge space, and simultaneous discharges occur at both the upper and lower tips, a phenomenon known as the “double-head” discharge mode in certain research studies [
35]. Metal particles can be approximated as equipotential bodies. When placed between a high-voltage electrode and grounded electrode, the particle exhibits a lower potential relative to the high-voltage side and a higher potential relative to the grounded electrode. The discharge phenomenon at the upper end manifests as a negative streamer, whereas that at the lower end takes the form of a positive streamer. In order to clearly demonstrate the discharge process, distinct electron density color scales are employed for the upper and lower regions and are positioned on the corresponding sides. Given that the electron density at the lower end of the discharge region is 1–2 orders of magnitude higher than that at the upper end, the discharge process is significantly more intense. Consequently, this study primarily investigates the underlying mechanisms of the discharge at the lower end of the metal particles.
Figure 4a illustrates the variations in the charging charge and surface floating potential during the discharge process for an SF
6/CF
4 gas mixture ratio of 10%/90%. Specifically, A denotes the charging charge at the upper end, B corresponds to the charging charge at the lower end, and C indicates the surface floating potential. After the discharge process begins, under the influence of the external electric field, positive and negative charges surrounding the metal particles undergo directed movement. This results in the accumulation of positive charges on the upper surface of the particles and negative charges on the lower surface. The charging process is accomplished within an extremely short time frame at the initiation of the discharge. During this period, the potential of the metal particles varies dynamically as a function of the accumulated positive and negative charges. Notably, throughout the charging phase, positive charges dominate the distribution, and the floating potential increases sharply within 0.16 ns. At this time point (0.16 ns), a transition in the dynamic behavior of both the charging process and the floating potential is observed, after which both parameters progressively approach their respective steady-state values.
Figure 4b illustrates the variations in the charging charge and suspended potential for an SF
6/CF
4 gas mixture ratio of 60%/40%. In comparison with the 90% CF
4-10% SF
6 gas mixture, the suspended potential and charging charge reach a certain level and exhibit significant stability. Nevertheless, the overall trends of variation in both the charging charge and suspended potential remain consistent. This study does not explore in depth the specific mechanism by which differences in the mixing ratio influence the charging and discharging of particles, as this involves a complex coupling process across multiple physical fields, which lies beyond the scope of the current investigation.
To investigate the impact of the SF
6/CF
4 gas ratio on the streamer propagation, simulations were conducted for 90% CF
4-10% SF
6 and 40% CF
4-60% SF
6 mixtures.
Figure 5 presents the spatial electron density distributions under these two gas compositions. When the CF
4 concentration was 90%, as depicted in
Figure 5a, the streamer exhibited continuous propagation. After detaching from the tip, the streamer expanded both radially and axially, forming a “water drop”-like shape. The propagation speed of the streamer was estimated to range from 6.7 × 10
5 m/s to 1.6 × 10
6 m/s, based on the distance it extended toward the grounding electrode per unit time, and the streamer reached the ground electrode within 0.9 ns. This phenomenon is referred to as the “continuous propagation” regime in this study. The continuous transmission speed is comparable to the streamer propagation speed of 1 × 10
6 m/s~1.2 × 10
6 m/s observed in the 20% SF
6/80% N
2 gas mixture, as reported in Luo et al. [
25]. However, this value is significantly higher than the propagation speed of pure SF
6 reported in Reference [
20], which ranges from 7 × 10
5 m/s to 8.25 × 10
5 m/s.
When the CF4 concentration was 40%, the streamer took on a “crescent” shape, with a propagation speed ranging from approximately 5 × 104 m/s to 6.4 × 104 m/s. The streamer dissipated within 1.7 ns under these conditions. This phenomenon is referred to as the “attenuation propagation” regime in this study, with the maximum streamer propagation distance being 0.07 mm. Under strictly controlled simulation conditions of constant temperature and pressure, the differences between the two discharge modes can be attributed to variations in the gas mixing ratio. A quantitative analysis of the propagation speed reveals that the peak speed in the continuous propagation regime is approximately two orders of magnitude higher than that in the attenuation regime. These results suggest that the CF4 concentration significantly influences the streamer propagation speed.
Figure 6a illustrates the axial electron density in the continuous propagation scenario, with an order of magnitude of 10
20 m
−3. Starting from the initial state (t = 0 ns), the electron region detaches from the particle tip, forming a localized electron density peak. At 0.2 ns, due to the streamer head being near the particle, there exists a significant distorted electric field, which enhances the ionization rate of the gas, and the peak electron density increases to 3.06 × 10
20 m
−3. When the streamer moves away from the particle tip, the effect brought on by the distorted electric field weakens. As the streamer approaches the grounded electrode, the electric field increases, which causes the peak electron density to increase again. Overall, the peak electron density shows a two-stage evolution characteristic of first decaying and then increasing. The axial electric field distribution depicted in Figure 10a reveals that the applied electric field induces distortion near the tip, leading to the convergence of secondary electron avalanches and driving the electron region toward the grounded electrode. As the streamer moves away from the particle tip, the self-consistent electric field generated by the space charge gradually becomes dominant. This field continuously facilitates the convergence of secondary electron avalanches, sustaining the propagation of the streamer. The evolution of the spatial electron density is illustrated in
Figure 5(a1–a5). During this dynamic process, the driving effect of the applied electric field exhibits a progressive attenuation trend.
Figure 6b illustrates the axial electron density under attenuation conditions. In contrast to continuous propagation, the electron density is on the order of 10
19 m
−3, indicating a relatively weaker discharge state. The electron density initially increases and subsequently decreases, reaching a peak value of 7.26 × 10
19 m
−3 at 0.6 ns before gradually declining to 7.2 × 10
18 m
−3 at 1.7 ns. The spatial distribution of the electron density is presented in
Figure 5(b1–b5).
Figure 7a illustrates the axial distribution of the positive ion density under continuous propagation conditions. Positive ions exhibit an aggregation effect near the particle tip and propagate towards the negative electrode along with the streamer under the influence of the applied electric field. Within the channel, the ion density demonstrates a gradual increase trend, while in the previously propagated regions, the ion density remains uniformly distributed at an order of magnitude of approximately 10
20 m
−3. The propagation speed of positive ions ranges from 6.7 × 10
5 m/s to 1.2 × 10
6 m/s. A comparison with Figure 9a reveals that the peak electron density coincides spatially with the positive ion density, and that their propagation speeds are of the same order of magnitude. This indicates that the streamer’s gradual propagation towards the grounded electrode is primarily dominated by positive ions.
In the attenuated propagation scenario, positive ions fail to form a pronounced peak in ion density, which remains on the order of 1020 m−3, consistent with the continuous propagation case. This suggests that the attenuation of the streamer cannot be ascribed solely to the reduction in the positive ion density. Furthermore, it is noteworthy that, as depicted in the three axial distributions of particles in Figure 9, irrespective of whether the proportion of the mixed gas varies or not, the peak of positive ions consistently precedes both the electron density and the positive ion density at the streamer head, thereby highlighting the critical role of positive ions during propagation.
Figure 8a illustrates the temporal evolution of the axial distribution of negative ions under continuous propagation conditions. As the streamer detaches from the particle tip and propagates towards the grounded electrode, negative ions are generated near the streamer head due to attachment reactions. The primary ions identified include SF
6−, SF
5−, SF
4−, F
2−, and others. Under the influence of the electric field, these negative ions migrate towards the particle tip, forming a peak. Furthermore, their number density exhibits a slight increase as the streamer extends. Over time, in regions far from the particle tip that have been traversed by the streamer, the density of negative ions is approximately one order of magnitude lower than that of positive ions and electrons. This further suggests that during the streamer propagation process, positive space charges play a dominant role in driving the propagation.
In the case of streamer attenuation propagation, the distribution patterns of negative ions and positive ions are consistent, with their number densities being on the same order of magnitude. However, a notable difference is that the concentration of negative ions is the highest among all the charged particles, which may account for the inability of the streamer to propagate continuously. Interestingly, the density of negative ions in the discharge channel remains largely unchanged across both propagation scenarios, suggesting that variations in the mixing ratios do not alter the electronegativity of the gas. Both SF6 and CF4 exhibit strong electronegativity, with comparable electron adsorption capabilities.
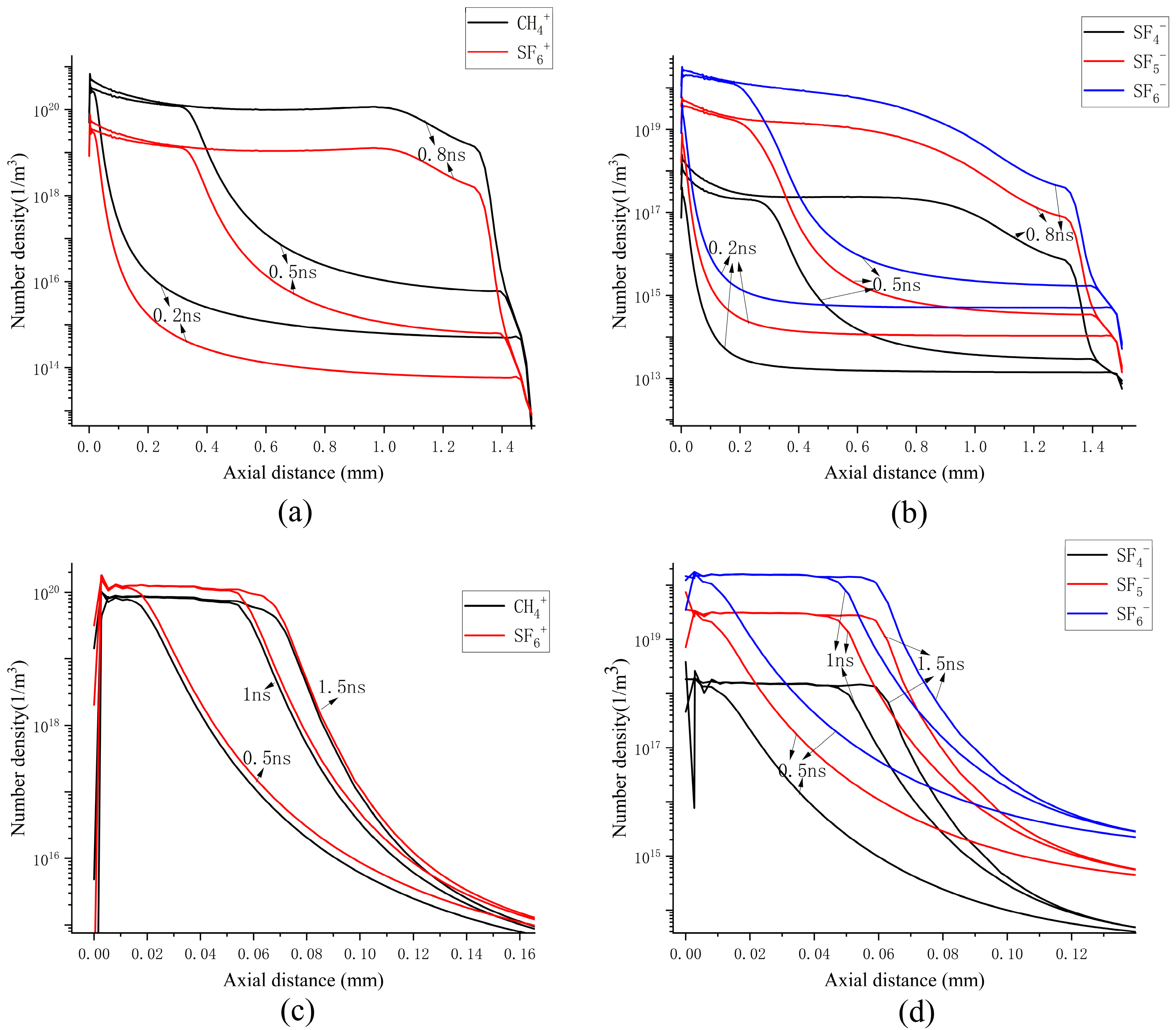
Figure 9 presents the axial distribution of each particle under continuous propagation conditions, providing a more intuitive comparison of the distributions of the three types of particles. The concentration of positive ions is the highest in the already-propagated channel and at the streamer head. At the particle tip, the density of positive ions constitutes a relatively large proportion among the charged particles and is on the same order of magnitude as that of electrons. This suggests that the ionization reaction may be intense throughout the process, particularly at the streamer head, where the densities of positive ions and electrons exceed that of negative ions, thereby controlling electron generation. This is likely to be the primary reason for the sustained development of the streamer.
Figure 9.
Axial particle distributions at 0.8 ns under continuous propagation and attenuation propagation conditions: (a) continuous transmission situation; (b) attenuation propagation situation.
Figure 9.
Axial particle distributions at 0.8 ns under continuous propagation and attenuation propagation conditions: (a) continuous transmission situation; (b) attenuation propagation situation.
Under attenuation conditions, the concentration of negative ions is the highest in the discharge channel. Within the streamer propagation channel, the electron density is one order of magnitude lower than that of both positive and negative ions. At the streamer head, the propagation of positive ions precedes that of negative ions and electrons, while the density of negative ions exceeds that of electrons by one order of magnitude. This suggests that negative ions play a dominant role in controlling the depletion of electrons throughout the entire propagation process.
The axial electric field distributions under both continuous propagation and attenuation propagation conditions are presented in
Figure 10. As shown in the data of
Figure 10a, during continuous propagation, the maximum electric field is located at the streamer head position, followed by a minimum electric field at subsequent spatial positions. Throughout the propagation process, the maximum electric field varies within the range of 1.28 × 10
7 to 8.8 × 10
6 V/m. In the streamer channel, the electric field decreases initially and subsequently increases from the tip to the streamer head, with variations spanning between 1 × 10
6 and 2 × 10
6 V/m.
Figure 10.
Axial electric field distributions under continuous and attenuation propagation conditions: (a) continuous transmission situation; (b) attenuation propagation situation.
Figure 10.
Axial electric field distributions under continuous and attenuation propagation conditions: (a) continuous transmission situation; (b) attenuation propagation situation.
As shown in
Figure 10b, under the streamer attenuation condition, the maximum electric field along the axial direction of the streamer channel is one order of magnitude higher than that in the continuous propagation case and exhibits a gradually increasing trend. According to Morrow’s research [
37,
38], the primary mechanism for streamer attenuation in air or pure SF
6 involves the continuous reduction in the electric field at the streamer head. However, in the SF
6/CF
4 mixed gas (with 40% CF
4 content), an anomalous phenomenon was observed. During the gradual fading of the streamer, the electric field at the streamer head remained at a high level without any significant decrease, thereby excluding the possibility that the streamer attenuation was caused by the decay of the electric field.
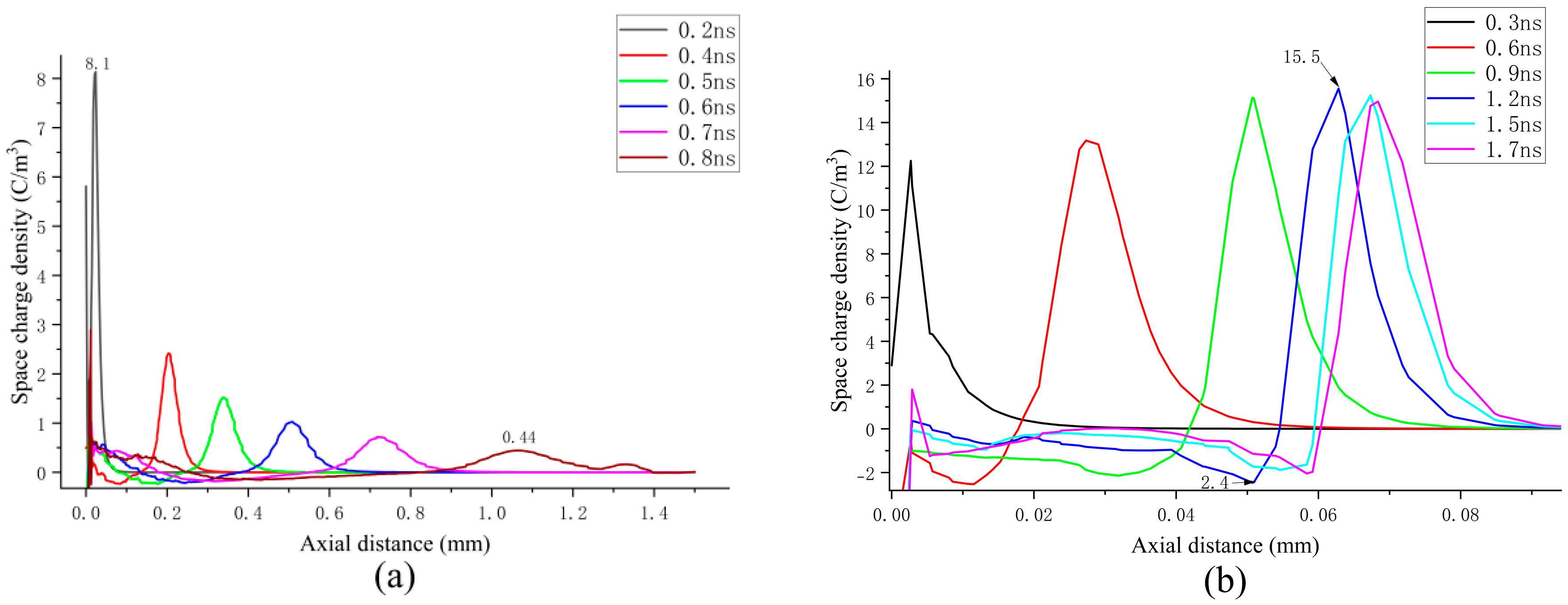
Figure 11a displays the axial spatial net charge density under continuous propagation conditions. It is evident that the highest positive charge density is located at the streamer head. As the streamer propagates toward the grounded electrode, the maximum positive charge density exhibits a decreasing trend, with values ranging from 0.44 to 8.1 C/m
3.
Figure 11b illustrates the axial spatial net charge density under attenuated propagation conditions. The peak of the positive spatial net charge initially increases and subsequently decreases, with the maximum value reaching approximately 15.5C/m
3, which is significantly higher than that observed under continuous propagation conditions.
In the case of streamer attenuation, the variation trend of the positive space charge near the streamer head is in agreement with the trend of the electric field at the streamer head. Within the discharge channel, a peak in the space charge forms at the streamer head. Furthermore, a region of negative space charge is evident in proximity to the streamer head. In the continuous propagation scenario, the negative space charge exhibits minimal magnitude and uniform distribution. For attenuated propagation, the absolute value of the negative space charge density remains within 2.5 C/m
3. When compared to the uniform distribution of the negative space charge during continuous propagation, the “negative charge center” emerges near the “positive charge center” at the streamer head during streamer attenuation at 1.2 ns, where the negative charge density reaches 2.4 C/m
3. During streamer attenuation, as the maximum positive charge near the streamer head increases, the negative charge gradually accumulates at the negative charge center. Referring to
Figure 6b, it can be observed that the decrease in the electron density coincides with the increase in the negative charge, suggesting that a strong attachment reaction may occur in the streamer channel, leading to the generation of negative ions as electrons are consumed. From the weakening effect associated with the accumulated negative charge, it can be inferred that the buildup of negative charge at the negative charge center might be essential in limiting the strong ionization area and facilitating streamer decay.
The distribution of ion species along the axial direction under the two different concentration conditions are presented in
Figure 12. In the positive space charge region at the streamer head, it is composed of CF
4+ and SF
6+. In both cases, the number densities of CF
4+ and SF
6+ at the streamer head range from 10
19 m
−3 to 10
20 m
−3. The difference arises from the varying proportions of positive ions due to the distinct ratios of the mixed gases. When the proportion of CF
4 in the mixed gas is higher, the concentration of CF
4+ is greater; conversely, when the proportion decreases, the concentration of CF
4+ diminishes accordingly. The variation trends of the number densities of CF
4+ and SF
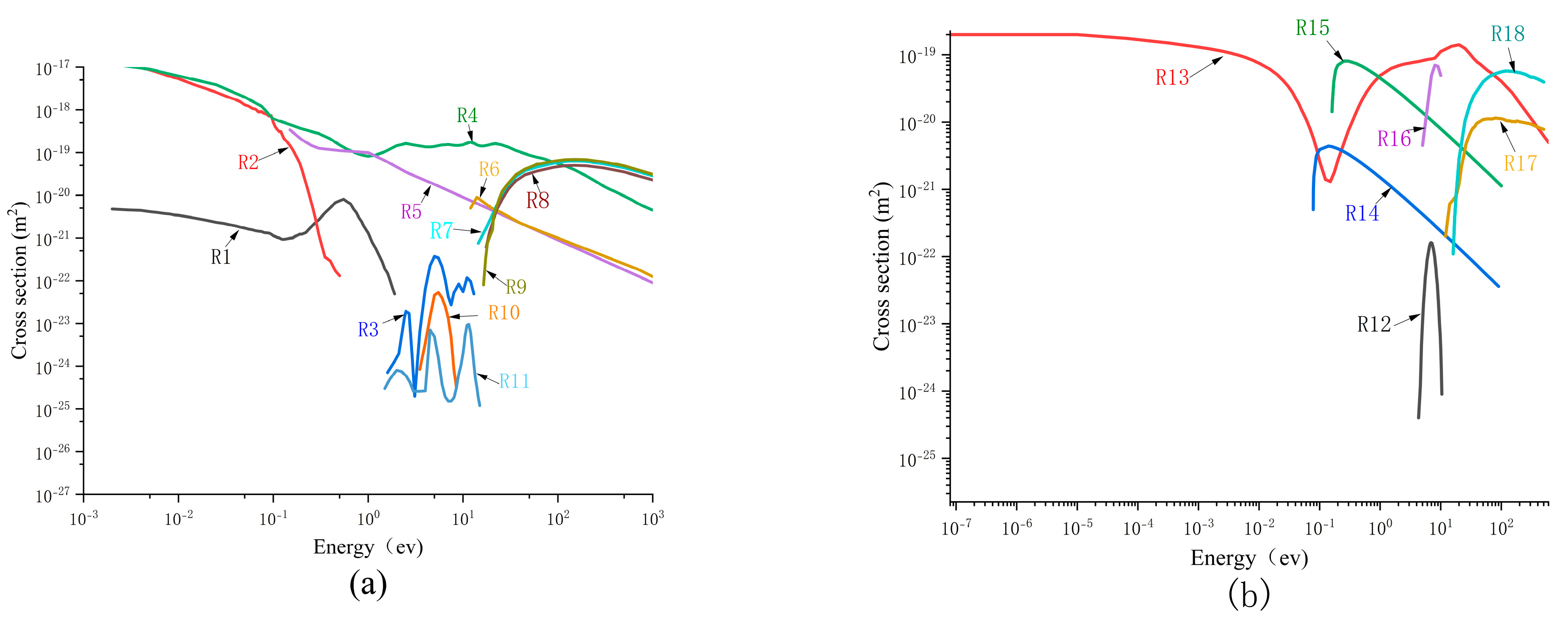
6+ are consistent with the corresponding trends of the spatial positive charge concentration and the electric field at the streamer head, suggesting that positive ions facilitate the propagation of streamers by enhancing the electric field at their heads. The ionization reaction rates of CF
4+ and SF
6+ are presented in
Figure 13a. It can be observed that, when the concentration of CF
4 is 90%, the ionization rate of SF
6 (R9) at the streamer head is one order of magnitude lower than that of CF
4 (R18), suggesting that during continuous streamer propagation, the ionization of CF
4 serves as the primary mechanism for the generation of positive ions. In the case of streamer attenuation, the ionization reaction rates of CF
4 and SF
6 at the streamer head are one order of magnitude lower than those during continuous propagation. As shown in
Figure 13b, the ionization rate of SF
6 (R9) at the streamer head is higher than that of CF
4 (R18), indicating that different mixing ratios alter the primary ionization reaction species at the streamer head, thereby influencing the ionization activity at the streamer head.
Negative ions play a critical role in determining whether propagation is continuous or attenuated. In the streamer channel, regardless of whether the propagation is continuous or attenuated, SF
5− and SF
6− are the predominant negative ions. Their number densities near the streamer head are more than one order of magnitude higher than that of SF
4−, as illustrated in
Figure 12b,d. Electrons are primarily generated at the streamer head, and at the location of the minimum ionization rate, the attachment rates for the formation of SF
6− (R2) and SF
5− (R1) from SF
6 are maximized. Consequently, during their migration towards the anode, the electrons generated at the streamer head attach to SF
6 and SF
5 molecules, thereby forming a region of negative space charge. When the concentration of CF
4 is 90%, the streamer propagates continuously. The attachment rates of SF
6−, SF
5−, and SF
4− from SF
6 are lower than the ionization rates of CF
4 and SF
6, resulting in no significant formation of negative space charge, as illustrated in
Figure 11a. During streamer attenuation, the attachment rate close to the streamer head is considerably greater than the ionization rate. This leads to a substantial buildup of negative ions around the streamer head and facilitates the establishment of a negative space charge center. It can be observed that at 1.2 ns, the charge density of the negative space charge center reaches 2 C/m
3. This leads to a change in the electric field near the streamer head within the streamer channel, enhances the attachment rate, accelerates the depletion of electrons at the streamer head, and thereby suppresses the development of the streamer.
Figure 13a presents the reaction rates of all the reactions under continuous propagation conditions. It is evident that ionization reactions R9 and R18, as well as attachment reactions R1 and R2, are the predominant reactions in the discharge space. Based on the peak positions and the comparison of the reaction types listed in
Table 1, it can be observed that the peaks of both attachment reactions and ionization reactions occur at the same spatial location. In this study, the positions of the two peaks are defined as the “ionization center” and the “attachment center”. The ionization center is spatially ahead, where the ionization reaction rate of CF
4 is the highest, ranging from 4.4 × 10
6 to 1.37 × 10
7 mol/(m
3·s). At the attachment center, the SF
6 attachment reaction R2 exhibits the highest reaction rate, albeit lower than the ionization reaction rate, allowing the streamer to continue developing. When the concentration of CF
4 is 40%, the dominant ionization reaction at the ionization center shifts to SF
6, with an ionization reaction rate ranging from 1.07 × 10
5 to 1.7 × 10
6 mol/(m
3·s), which is one order of magnitude lower than that in the continuous propagation case. This further demonstrates that different mixing ratios significantly influence the ionization activity of the mixed gas. However, the primary reaction at the attachment center remains R2, with a reaction rate of approximately 2.1 × 10
5 to 1.8 × 10
6 mol/(m
3·s). This indicates that variations in the concentration of CF
4 affect the ionization intensity of the mixed gas by altering the main ionization reaction species. In the attenuation scenario, the attachment reaction rate exceeds the ionization reaction rate, leading to a greater disappearance of electrons compared to their generation, thereby causing the streamer to tend toward attenuation.