Source-Based EEG Neurofeedback for Sustained Motor Imagery of a Single Leg
Abstract
1. Introduction
- To investigate the activation of brain structures due to single-leg MI with neurofeedback at the source level and how these compare to motor imagination and motor execution without feedback.
- To examine how lateralization in the sensory-motor cortex is affected by neurofeedback and compare it with the laterality during imagined and executed movement of a single leg without feedback.
2. Materials and Methods
2.1. Participants
2.2. Experimental Setup
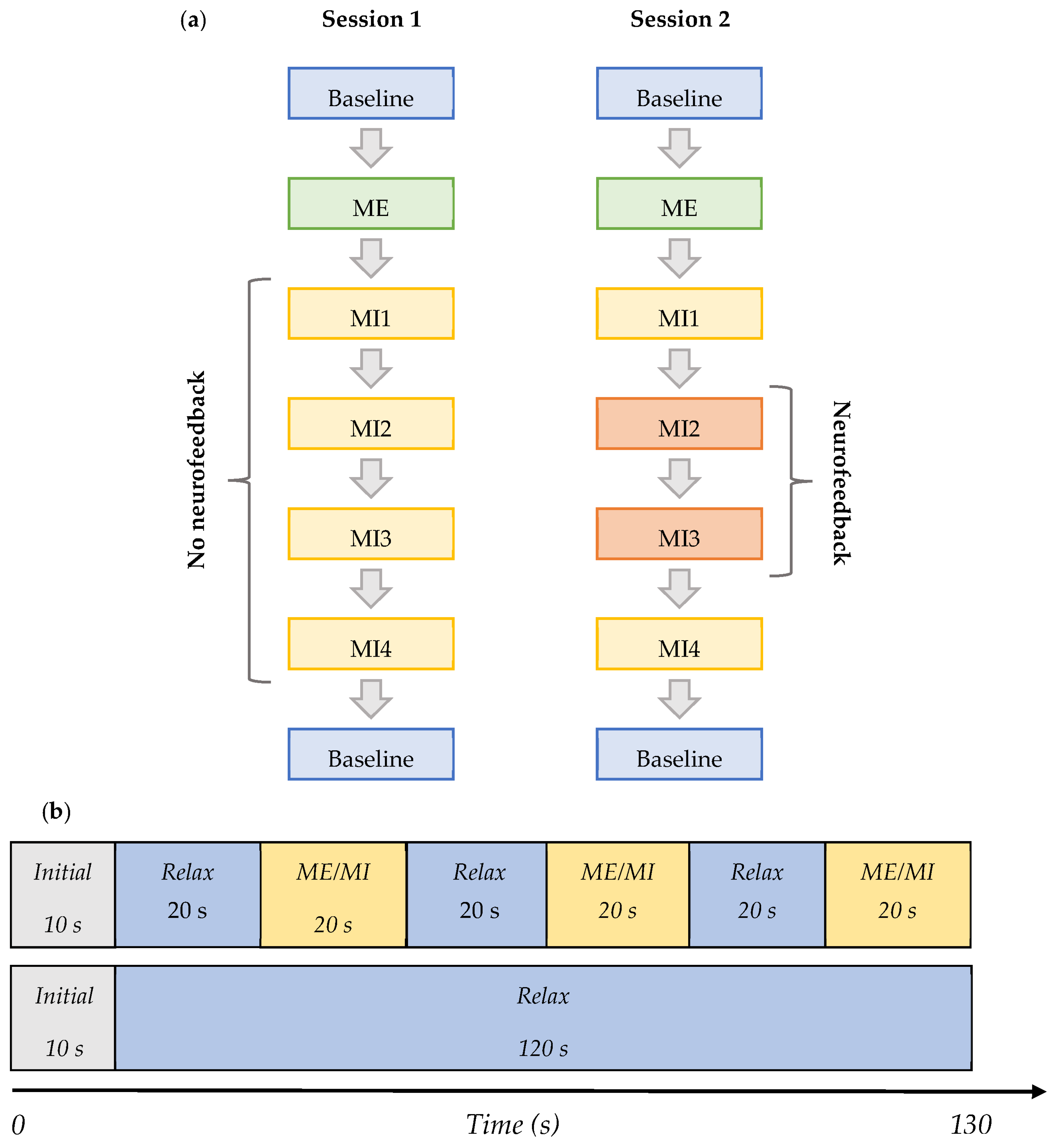
2.3. Experimental Protocol
- Baseline (BL)—Initial baseline data were acquired while a participant was sitting relaxed: two trials of 130 s with eyes open (EO) focusing on a fixation cross, and one of the same length with eyes closed (EC) in the following order—EO, EC, and EO. This task was the same in both Session 1 and 2, and it is shown in row 2 in Figure 2b.
- ME—The initial baseline was followed by the motor execution task where a subject was instructed to move the right leg by alternating the plantar flexion and dorsiflexion of the ankle when cued for movement: four trials of 130 s with alternating 20 s blocks of ME and resting, as shown in row 1 of Figure 2b. This task was the same in both Sessions 1 and 2.
- MI1—Following motor execution, the participant was asked to perform kinaesthetic MI by imagining the feeling of moving their right leg in the same manner as during the motor execution in the previous trials: four trials of 130 s with alternating 20 s blocks of MI and resting, as shown in row 1 of Figure 2b. This task was the same in both Sessions 1 and 2.
- MI2 and MI3—In Session 1, this task did not differ from MI1. However, in Session 2, a participant performed the task as in MI1 but additionally received visual neurofeedback. It comprised ten trials of 130 s with alternating 20 s blocks of MI and resting.
- MI4—A participant was free to choose the type of right-leg kinaesthetic MI to perform without neurofeedback: four trials of 130 s with alternating 20 s blocks of MI and the resting state are shown in row 1 of Figure 2b. This task was the same in both Session 1 and 2.
- Baseline (final)—The final baseline task was similar to the initial baseline task. Three trials of 130 s were acquired. The task was the same in both Session 1 and 2, and it is shown in row 2 of Figure 2b.
2.4. Offline Data Analysis
2.4.1. EEG Pre-Processing
2.4.2. Source Localization
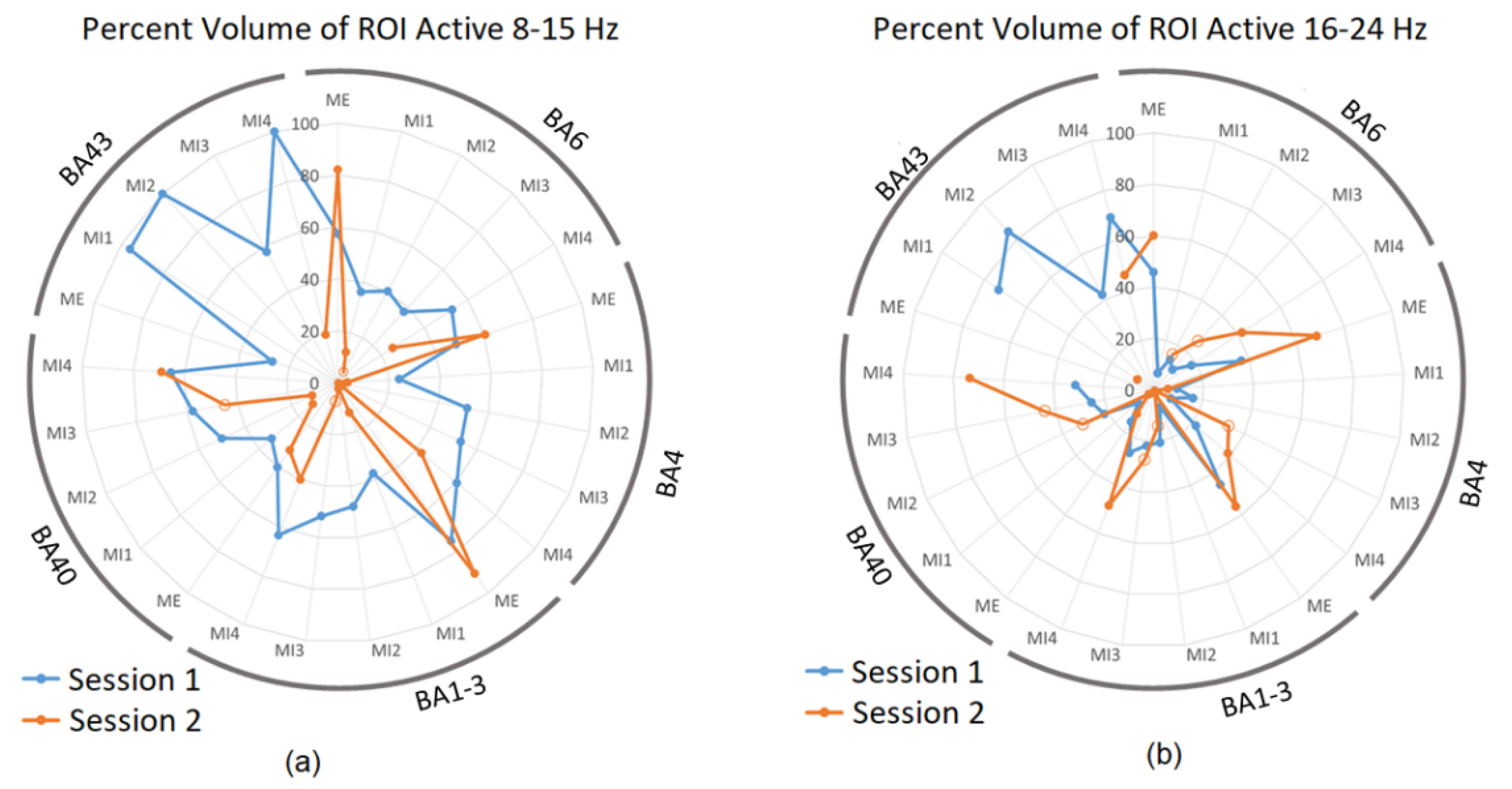
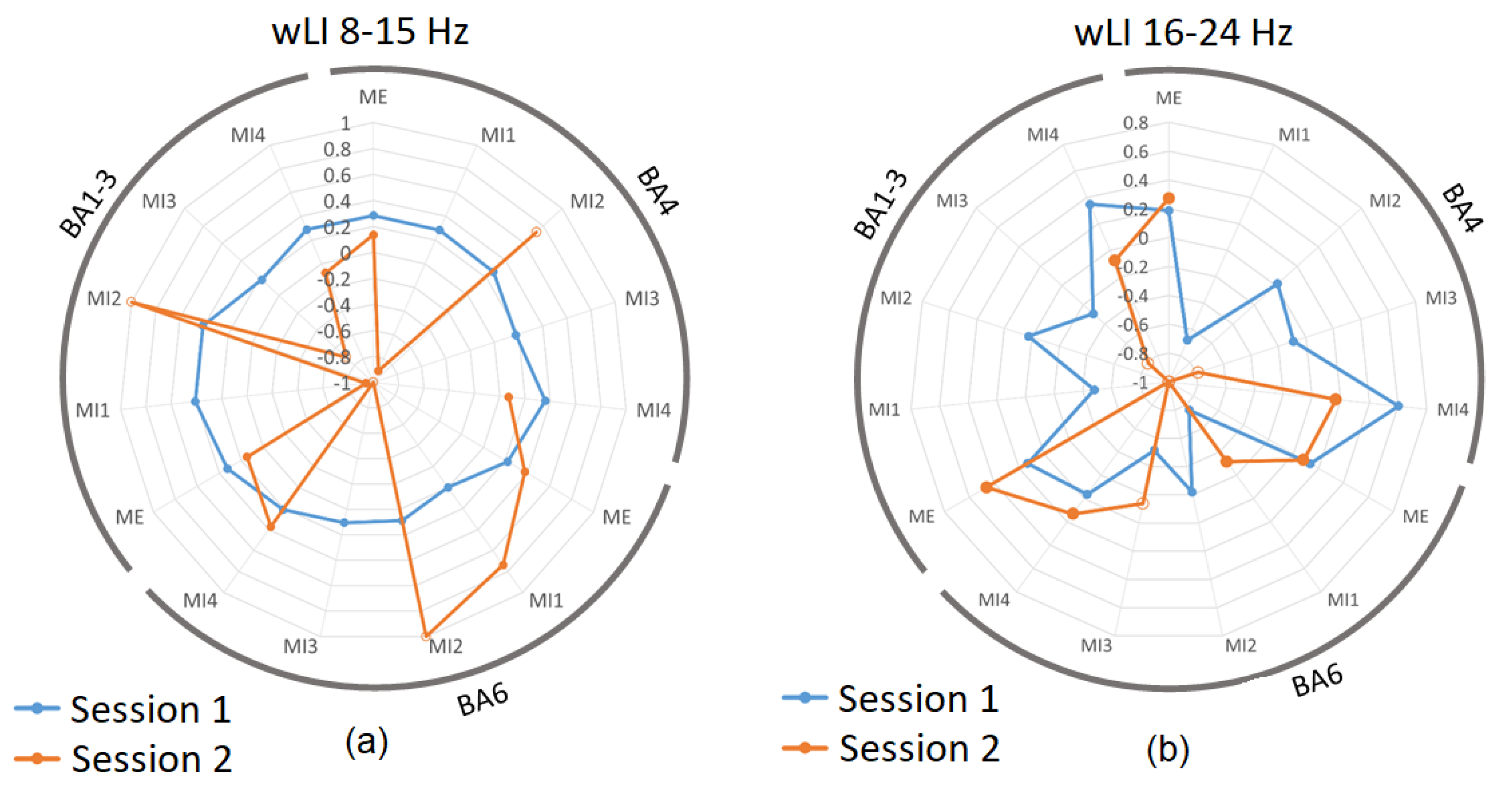
2.4.3. Lateralization Index and Percentage of Active Volumes
2.4.4. Statistical Analysis
3. Results
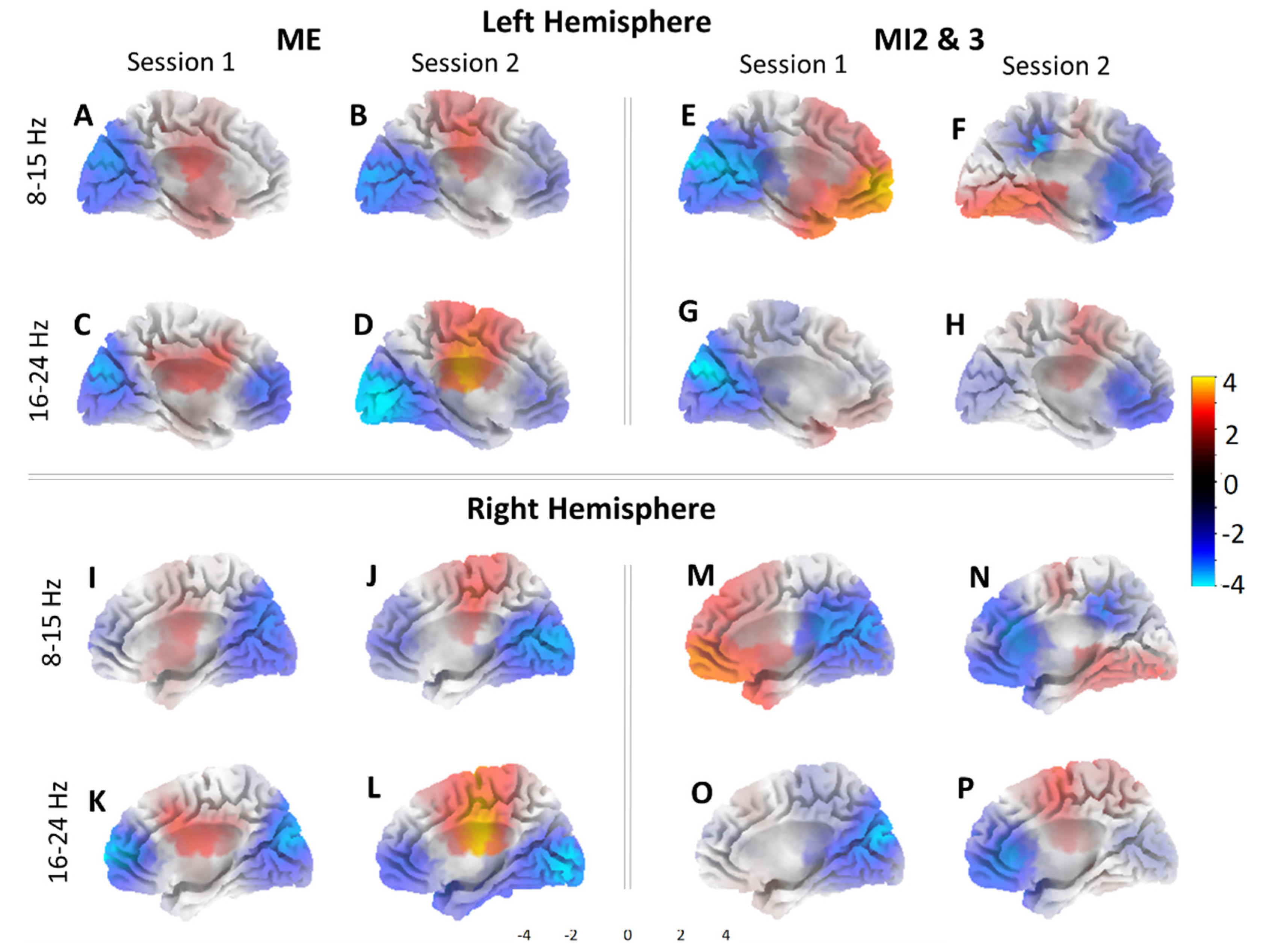
3.1. Kinesthetic and Visual Motor Imagery
3.2. sLORETA Volume Analysis
3.3. Laterality Index during Real and Imagined Movements
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khan, M.A.; Das, R.; Iversen, H.K.; Puthusserypady, S. Review on motor imagery based BCI systems for upper limb post-stroke neurorehabilitation: From designing to application. Comput. Biol. Med. 2020, 123, 103843. [Google Scholar] [CrossRef] [PubMed]
- Camargo-Vargas, D.; Callejas-Cuervo, M.; Mazzoleni, S. Brain-Computer Interfaces Systems for Upper and Lower Limb Rehabilitation: A Systematic Review. Sensors 2021, 21, 4312. [Google Scholar] [CrossRef] [PubMed]
- Asanza, V.; Peláez, E.; Loayza, F.; Lorente-Leyva, L.L.; Peluffo-Ordóñez, D.H. Identification of Lower-Limb Motor Tasks via Brain–Computer Interfaces: A Topical Overview. Sensors 2022, 22, 2028. [Google Scholar] [CrossRef]
- Selves, C.; Stoquart, G.; Lejeune, T. Gait rehabilitation after stroke: Review of the evidence of predictors, clinical outcomes and timing for interventions. Acta Neurol. Belg. 2020, 120, 783–790. [Google Scholar] [CrossRef]
- Kwakkel, G.; Veerbeek, J.M.; Van Wegen, E.E.H.; Wolf, S.L. Constraint-induced movement therapy after stroke. Lancet Neurol. 2015, 14, 224–234. [Google Scholar] [CrossRef]
- Biasiucci, A.; Leeb, R.; Iturrate, I.; Perdikis, S.; Al-Khodairy, A.; Corbet, T.; Schnider, A.; Schmidlin, T.; Zhang, H.; Bassolino, M.; et al. Brain-actuated functional electrical stimulation elicits lasting arm motor recovery after stroke. Nat. Commun. 2018, 9, 2421. [Google Scholar] [CrossRef] [PubMed]
- Saladin, K.S. Anatomy & Physiology: The Unity of Form and Function, 4th ed.; McGraw-Hill: New York, NY, USA, 2007. [Google Scholar]
- de Almeida, P.M.D.; Vieira, A.I.C.M.D.F.; Canário, N.I.S.; Castelo-Branco, M.; Caldas, A.L.D.C. Brain Activity during Lower-Limb Movement with Manual Facilitation: An fMRI Study. Neurol. Res. Int. 2015, 2015, 701452. [Google Scholar] [CrossRef]
- Nakata, H.; Domoto, R.; Mizuguchi, N.; Sakamoto, K.; Kanosue, K. Negative BOLD responses during hand and foot movements: An fMRI study. PLoS ONE 2019, 14, e0215736. [Google Scholar] [CrossRef]
- Kline, A.; Ghiroaga, C.G.; Pittman, D.; Goodyear, B.; Ronsky, J. EEG differentiates left and right imagined Lower Limb movement. Gait Posture 2020, 84, 148–154. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Ushiba, J. EEG-based classification of imaginary left and right foot movements using beta rebound. Clin. Neurophysiol. 2013, 124, 2153–2160. [Google Scholar] [CrossRef]
- Lee, Y.S.; Bae, S.H.; Lee, S.H.; Kim, K.Y. Neurofeedback training improves the dual-task performance ability in stroke patients. Tohoku J. Exp. Med. 2015, 236, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Mihara, M.; Fujimoto, H.; Hattori, N.; Otomune, H.; Kajiyama, Y.; Konaka, K.; Watanabe, Y.; Hiramatsu, Y.; Sunada, Y.; Miyai, I.; et al. Effect of Neurofeedback Facilitation on Poststroke Gait and Balance Recovery: A Randomized Controlled Trial. Neurology 2021, 96, e2587–e2598. [Google Scholar] [CrossRef] [PubMed]
- Sitaram, R.; Caria, A.; Veit, R.; Gaber, T.; Rota, G.; Kuebler, A.; Birbaumer, N. fMRI Brain-Computer Interface: A Tool for Neuroscientific Research and Treatment. Comput. Intell. Neurosci. 2007, 2007, 25487. [Google Scholar] [CrossRef]
- Congedo, M.; Lubar, J.; Joffe, D. Low-resolution electromagnetic tomography neurofeedback. IEEE Trans. Neural Syst. Rehabil. Eng. 2004, 12, 387–397. [Google Scholar] [CrossRef]
- Steinberg, B.; Blum, K.; McLaughlin, T.; Lubar, J.; Febo, M.; Braverman, E.R.; Badgaiyan, R.D. Low-Resolution Electromagnetic Tomography (LORETA) of changed Brain Function Provoked by Pro-Dopamine Regulator (KB220z) in one Adult ADHD case. Open J. Clin. Med. Case Rep. 2016, 2, 1121. [Google Scholar] [PubMed]
- Prinsloo, S.; Rosenthal, D.I.; Lyle, R.; Garcia, S.M.; Gabel-Zepeda, S.; Cannon, R.; Bruera, E.; Cohen, L. Exploratory Study of Low Resolution Electromagnetic Tomography (LORETA) Real-Time Z-Score Feedback in the Treatment of Pain in Patients with Head and Neck Cancer. Brain Topogr. 2019, 32, 283–285. [Google Scholar] [CrossRef] [PubMed]
- Lin, I.-M.; Yu, H.-E.; Yeh, Y.-C.; Huang, M.-F.; Wu, K.-T.; Ke, C.-L.K.; Lin, P.-Y.; Yen, C.-F. Prefrontal Lobe and Posterior Cingulate Cortex Activations in Patients with Major Depressive Disorder by Using Standardized Weighted Low-Resolution Electromagnetic Tomography. J. Pers. Med. 2021, 11, 1054. [Google Scholar] [CrossRef]
- White, D.J.; Congedo, M.; Ciorciari, J. Source-based neurofeedback methods using EEG recordings: Training altered brain activity in a functional brain source derived from blind source separation. Front. Behav. Neurosci. 2014, 8, 373. [Google Scholar] [CrossRef]
- Gu, L.; Yu, Z.; Ma, T.; Wang, H.; Li, Z.; Fan, H. EEG-based Classification of Lower Limb Motor Imagery with Brain Network Analysis. Neuroscience 2020, 436, 93–109. [Google Scholar] [CrossRef]
- Abeln, V.; Harig, A.; Knicker, A.; Vogt, T.; Schneider, S. Brain-imaging during an isometric leg extension task at graded intensities. Front. Physiol. 2013, 4, 296. [Google Scholar] [CrossRef]
- Jurcak, V.; Tsuzuki, D.; Dan, I. 10/20, 10/10, and 10/5 systems revisited: Their validity as relative head-surface-based positioning systems. NeuroImage 2007, 34, 1600–1611. [Google Scholar] [CrossRef] [PubMed]
- Malouin, F.; Richards, C.L.; Jackson, P.; LaFleur, M.F.; Durand, A.; Doyon, J. The Kinesthetic and Visual Imagery Questionnaire (KVIQ) for Assessing Motor Imagery in Persons with Physical Disabilities: A Reliability and Construct Validity Study. J. Neurol. Phys. Ther. 2007, 31, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Pfurtscheller, G.; Lopes, F.H. Event-related EEG/MEG synchronization and desynchronization: Basic principles. Clin. Neurophysiol. 1999, 110, 1842–1857. [Google Scholar] [CrossRef] [PubMed]
- Delorme, A.; Makeig, S. EEGLAB: An Open Source Toolbox for Analysis of Single-Trial EEG Dynamics Including Independent Component Analysis. J. Neurosci. Methods 2004, 134, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Ben-Shabat, E.; Matyas, T.A.; Pell, G.S.; Brodtmann, A.; Carey, L.M. The Right Supramarginal Gyrus Is Important for Proprioception in Healthy and Stroke-Affected Participants: A Functional MRI Study. Front. Neurol. 2015, 6, 248. [Google Scholar] [CrossRef]
- Pascual-Marqui, R.D. Standardized low-resolution brain electromagnetic tomography (sLORETA): Technical details. Methods Find. Exp. Clin. Pharmacol. 2002, 24 (Suppl. D), 5–12. [Google Scholar]
- Oberhuber, M.; Hope, T.M.H.; Seghier, M.L.; Jones, O.P.; Prejawa, S.; Green, D.W.; Price, C.J. Four Functionally Distinct Regions in the Left Supramarginal Gyrus Support Word Processing. Cereb. Cortex 2016, 26, 4212–4226. [Google Scholar] [CrossRef]
- Caspers, S.; Schleicher, A.; Bacha-Trams, M.; Palomero-Gallagher, N.; Amunts, K.; Zilles, K. Organization of the Human Inferior Parietal Lobule Based on Receptor Architectonics. Cereb. Cortex 2013, 23, 615–628. [Google Scholar] [CrossRef]
- Rolls, E.T. The cingulate cortex and limbic systems for emotion, action, and memory. Brain Struct. Funct. 2019, 224, 3001–3018. [Google Scholar] [CrossRef]
- Sebastian-Romagosa, M.; Ortner, R.; Udina-Bonet, E.; Dinares-Ferran, J.; Mayr, K.; Cao, F.; Guger, C. Laterality Coefficient: An EEG parameter related with the functional improvement in stroke patients. In Proceedings of the 2019 IEEE EMBS International Conference on Biomedical & Health Informatics (BHI), Chicago, IL, USA, 19–22 May 2019. [Google Scholar] [CrossRef]
- Seghier, M.L. Laterality index in functional MRI: Methodological issues. Magn. Reason. Imaging 2008, 26, 594–601. [Google Scholar] [CrossRef]
- Li, G.; Huang, S.; Xu, W.; Jiao, W.; Jiang, Y.; Gao, Z.; Zhang, J. The impact of mental fatigue on brain activity: A comparative study both in resting state and task state using EEG. BMC Neurosci. 2020, 21, 20. [Google Scholar] [CrossRef] [PubMed]
- Talukdar, U.; Hazarika, S.M.; Gan, J.Q. Motor imagery and mental fatigue: Inter-relationship and EEG based estimation. J. Comput. Neurosci. 2019, 46, 55–76. [Google Scholar] [CrossRef] [PubMed]
- Ritter, P.; Moosmann, M.; Villringer, A. Rolandic alpha and beta EEG rhythms’ strengths are inversely related to fMRI-BOLD signal in primary somatosensory and motor cortex. Hum. Brain Mapp. 2009, 30, 1168–1187. [Google Scholar] [CrossRef] [PubMed]
- Jurkiewicz, M.T.; Gaetz, W.C.; Bostan, A.C.; Cheyne, D. Post-movement beta rebound is generated in motor cortex: Evidence from neuromagnetic recordings. Neuroimage 2006, 32, 1281–1289. [Google Scholar] [CrossRef]
- Muhamed, A.F.; Pollick, F.; Vuckovic, A. Improving Motor Imagination with Support of Real-Time LORETA Neurofeedback. In Proceedings of the Sixth International Brain-Computer Interface Meeting: BCI Past, Present, and Future, Asilomar, CA, USA, 30 May–3 June 2016. [Google Scholar]
- Gardner, E.B. The neurophysiological basis of motor learning. Phys. Ther. 1967, 47, 1115–1122. [Google Scholar] [CrossRef]
- Mizuguchi, N.; Nakata, H.; Hayashi, T.; Sakamoto, M.; Muraoka, T.; Uchida, Y.; Kanosue, K. Brain activity during motor imagery of an action with an object: A functional magnetic resonance imaging study. Neurosci. Res. 2013, 76, 150–155. [Google Scholar] [CrossRef]
- Cassim, F.; Szurhaj, W.; Sediri, H.; Devos, D.; Bourriez, J.-L.; Poirot, I.; Derambure, P.; Defebvre, L.; Guieu, J.-D. Brief and sustained movements: Differences in event-related (de)synchronization (ERD/ERS) patterns. Clin. Neurophysiol. 2000, 111, 2032–2039. [Google Scholar] [CrossRef]
- Emmert, K.; Kopel, R.; Sulzer, J.; Brühl, A.B.; Berman, B.D.; Linden, D.E.J.; Horovitz, S.G.; Breimhorst, M.; Caria, A.; Frank, S.; et al. Meta-analysis of real-time fMRI neurofeedback studies using individual participant data: How is brain regulation mediated? Neuroimage 2016, 124 Pt A, 806–812. [Google Scholar] [CrossRef]
- Tariq, M.; Trivailo, P.M.; Simic, M. Mu-Beta event-related (de)synchronization and EEG classification of left-right foot dorsiflexion kinaesthetic motor imagery for BCI. PLoS ONE 2020, 15, e0230184. [Google Scholar] [CrossRef]
- Newton, J.M.; Dong, Y.; Hidler, J.; Plummer-D’Amato, P.; Marehbian, J.; Albistegui-DuBois, R.M.; Woods, R.P.; Dobkin, B.H. Reliable assessment of lower limb motor representations with fMRI: Use of a novel MR compatible device for real-time monitoring of ankle, knee and hip torques. Neuroimage 2008, 43, 136–146. [Google Scholar] [CrossRef]
- Choi, H.; Park, J.; Yang, Y.-M. Whitening Technique Based on Gram–Schmidt Orthogonalization for Motor Imagery Classification of Brain–Computer Interface Applications. Sensors 2022, 22, 6042. [Google Scholar] [CrossRef] [PubMed]
- Vučković, A.; Altaleb, M.K.H.; Fraser, M.; McGeady, C.; Purcell, M. EEG Correlates of Self-Managed Neurofeedback Treatment of Central Neuropathic Pain in Chronic Spinal Cord Injury. Front. Neurosci. 2019, 13, 762. [Google Scholar] [CrossRef] [PubMed]
- Putri, F.; Luca, I.S.; Pedro, J.A.G.; Ding, H.; Vučković, A. Winners and losers in brain computer interface competitive gaming: Directional connectivity analysis. J. Neural Eng. 2022, 19, 046037. [Google Scholar] [CrossRef]
- Esch, L.; Sun, L.; Klüber, V.; Lew, S.; Baumgarten, D.; Grant, P.E.; Okada, Y.; Haueisen, J.; Hämäläinen, M.S.; Dinh, C. MNE Scan: Software for real-time processing of electrophysiological data. J. Neurosci. Methods 2018, 303, 55–67. [Google Scholar] [CrossRef]
- Smetanin, N.; Volkova, K.; Zabodaev, S.; Lebedev, M.; Ossadtchi, A. NFBLab—A Versatile Software for Neurofeedback and Brain-Computer Interface Research. Front. Neuroinform. 2018, 12, 100. [Google Scholar] [CrossRef] [PubMed]
- Guarnieri, R.; Zhao, M.; Taberna, G.A.; Ganzetti, M.; Swinnen, S.P.; Mantini, D. RT-NET: Real-time reconstruction of neural activity using high-density electroencephalography. Neuroinformatics 2021, 19, 251–266. [Google Scholar] [CrossRef] [PubMed]
- Malouin, F.; Richards, C.L.; Durand, A.; Doyon, J. Clinical assessment of motor imagery after stroke. Neurorehabilit. Neural Repair. 2007, 22, 330–340. [Google Scholar] [CrossRef] [PubMed]

| BA | Condition | % Volume Active 8–15 Hz | % Volume Active 16–24 Hz | ||||||
|---|---|---|---|---|---|---|---|---|---|
| S1 | S2 * | S1 | S2 * | ||||||
| L | R | L | R | L | R | L | R | ||
| 1–3 | MI2 | 30 | 18 | 2 | 0 | 10 | 11 | 0 | 14 |
| MI3 | 28 | 24 | 1 | 6 | 8 | 14 | 4 | 24 | |
| 4 | MI2 | 24 | 17 | 4 | 1 | 6 | 8 | 0 | 16 |
| MI3 | 22 | 16 | 0 | 0 | 5 | 6 | 3 | 23 | |
| 5 | MI2 | 9 | 0 | 7 | 0 | 7 | 0 | 0 | 15 |
| MI3 | 10 | 2 | 5 | 6 | 9 | 0 | 10 | 25 | |
| 6 | MI2 | 28 | 24 | 2 | 0 | 6 | 11 | 0 | 0 |
| MI3 | 27 | 27 | 0 | 0 | 1 | 6 | 15 | 18 | |
| 7 | MI2 | 2 | 0 | 10 | 0 | 2 | 0 | 0 | 12 |
| MI3 | 5 | 0 | 11 | 4 | 2 | 0 | 4 | 19 | |
| 9 | MI2 | 47 | 51 | 0 | 0 | 15 | 11 | 0 | 0 |
| MI3 | 47 | 45 | 0 | 0 | 0 | 5 | 0 | 0 | |
| 10 | MI2 | 50 | 49 | 0 | 0 | 0 | 0 | 0 | 0 |
| MI3 | 50 | 50 | 0 | 0 | 1 | 0 | 0 | 0 | |
| 17 | MI2 | 0 | 0 | 46 | 6 | 0 | 0 | 0 | 0 |
| MI3 | 0 | 0 | 31 | 0 | 0 | 0 | 0 | 0 | |
| 24 | MI2 | 25 | 18 | 0 | 0 | 0 | 0 | 0 | 0 |
| MI3 | 18 | 11 | 0 | 0 | 0 | 0 | 6 | 14 | |
| 40 | MI2 | 41 | 9 | 11 | 0 | 20 | 2 | 0 | 31 |
| MI3 | 40 | 18 | 11 | 34 | 17 | 8 | 7 | 37 | |
| 43 | MI2 | 54 | 46 | 0 | 0 | 50 | 35 | 0 | 0 |
| MI3 | 54 | 4 | 0 | 0 | 42 | 0 | 0 | 0 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zulauf-Czaja, A.; Osuagwu, B.; Vuckovic, A. Source-Based EEG Neurofeedback for Sustained Motor Imagery of a Single Leg. Sensors 2023, 23, 5601. https://doi.org/10.3390/s23125601
Zulauf-Czaja A, Osuagwu B, Vuckovic A. Source-Based EEG Neurofeedback for Sustained Motor Imagery of a Single Leg. Sensors. 2023; 23(12):5601. https://doi.org/10.3390/s23125601
Chicago/Turabian StyleZulauf-Czaja, Anna, Bethel Osuagwu, and Aleksandra Vuckovic. 2023. "Source-Based EEG Neurofeedback for Sustained Motor Imagery of a Single Leg" Sensors 23, no. 12: 5601. https://doi.org/10.3390/s23125601
APA StyleZulauf-Czaja, A., Osuagwu, B., & Vuckovic, A. (2023). Source-Based EEG Neurofeedback for Sustained Motor Imagery of a Single Leg. Sensors, 23(12), 5601. https://doi.org/10.3390/s23125601






