Will Your Next Therapist Be a Robot?—A Review of the Advancements in Robotic Upper Extremity Rehabilitation
Abstract
1. Introduction
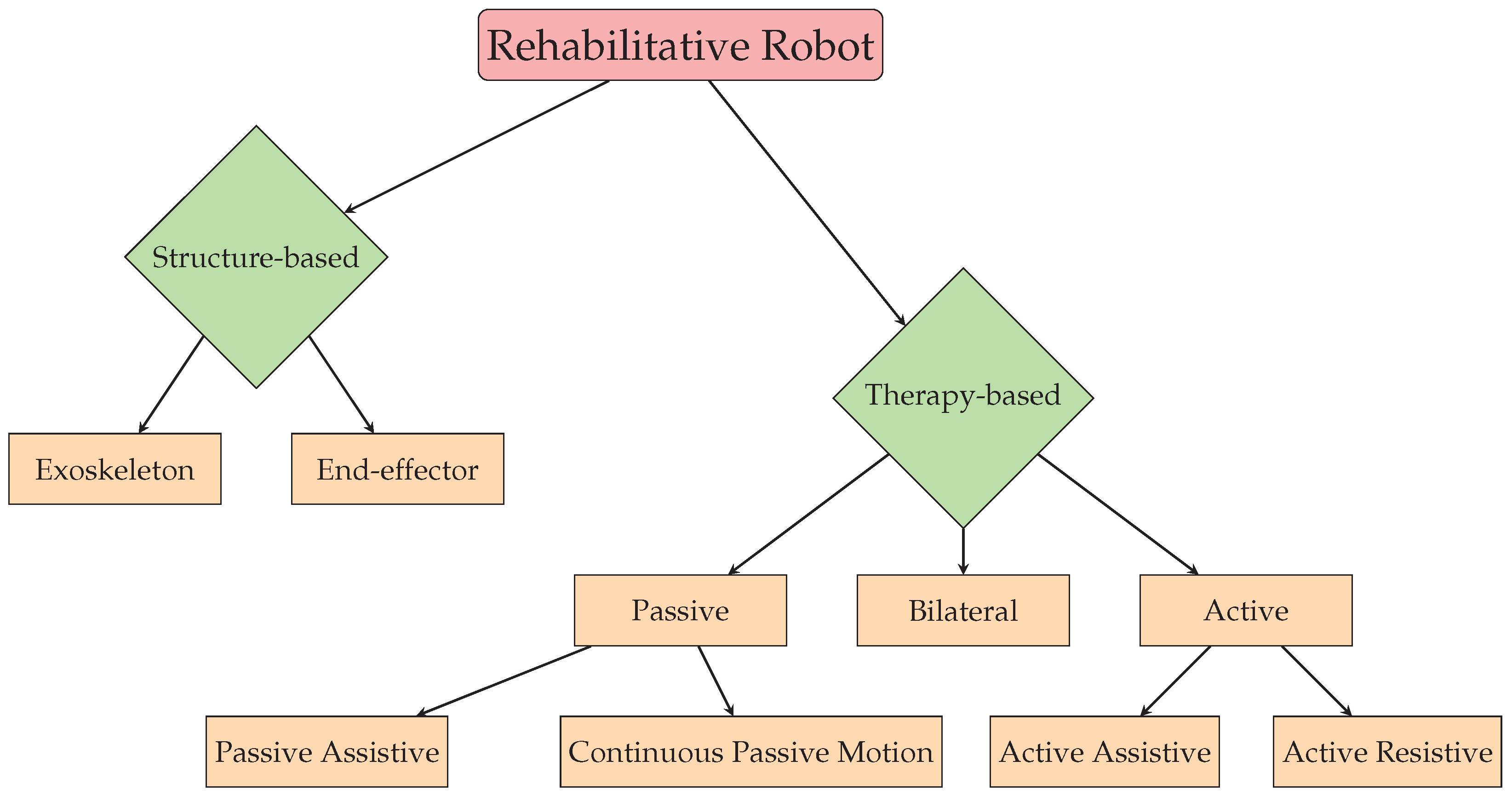
2. Classification of Rehabilitative Robots
2.1. Structure-Based Classification
2.1.1. End-Effector-Based Systems
2.1.2. Exoskeletons
- Harmony: The exoskeleton known as Harmony is equipped with a shoulder mechanism that follows the natural anatomy of the human body, allowing for unrestricted movement of all joints. It is capable of bearing the weight of the upper body and applying assisting force to help patients carry out desired movements [17].
- ANYexo: The ANYexo exoskeleton is a flexible and adaptable device with six degrees of freedom, intended for use on the upper limb. It is equipped with a series of elastic actuators that allow for low-impedance torque control. The device is primarily used as an experimental platform to test new hardware concepts and algorithms for autonomous therapy of patients with varying degrees of neural impairment. The aim of the device is to provide greater independence and functionality to individuals with arm impairments [18].
- MyoPro: (https://myomo.com/what-is-a-myopro-orthosis/, accessed on 25 April 2023) MyoPro is a powered upper limb orthosis developed by Myomo Inc. (Boston, MA, USA). The device is designed to help individuals with upper limb paralysis due to conditions such as stroke, spinal cord injury, or brachial plexus injury regain movement and function in their affected arm.
- Neofect Rapael Smart Glove: (https://www.neofect.com/us/smart-glove, accessed on 25 April 2023) Neofect Rapael Smart Glove is a wearable glove that uses sensors and haptic feedback to provide interactive training for individuals with hand weakness due to neurological conditions. The device is designed to help patients regain fine motor control and dexterity in their hands.
| End-Effector-Based Systems | Exoskeletons | |
|---|---|---|
| Advantages | Faster to set up | Increased gait transparency |
| Easier to manufacture | Isolated joint control | |
| Disadvantages | Less sophisticated | Expensive |
| Limited joint control | Not easily adjustable to different arm lengths | |
| Examples | [11,12,13,14,15,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38] | [19,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87] |
2.2. Therapy-Based Classification
2.2.1. Passive Therapy
2.2.2. Active Therapy
2.2.3. Bilateral
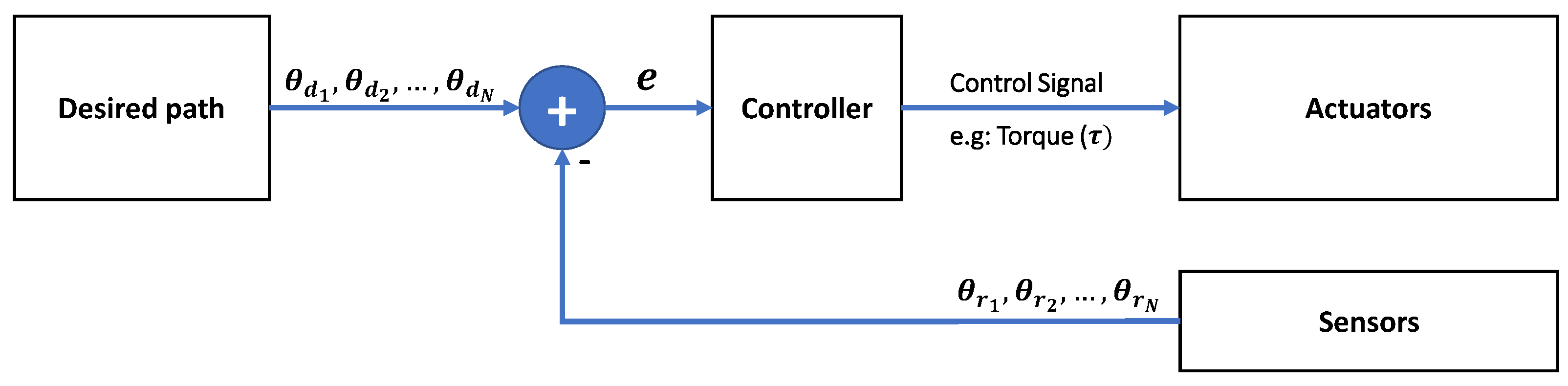
3. Control Strategies
3.1. Controller Input
3.2. Actuation
3.3. Controllers
3.3.1. PID Controllers
3.3.2. Robust Controllers
3.3.3. Adaptive Controllers
3.3.4. AI-Based Controllers
4. Motor Learning Strategies
4.1. Game Therapy
4.2. Virtual Reality
5. Clinical Trials
5.1. Quantifying Motor Recovery
5.1.1. Fugl-Meyer Assessment
5.1.2. Action Research Arm Test
5.1.3. Wolf Motor Function Test
5.1.4. Stroke Impact Scale
5.1.5. Barthel’s ADL Index
5.2. Trials
5.3. Evaluation of the Current State of Robotic Rehabilitation Systems: The Gaps, Challenges, and Requirements
- Cost: Many of the advanced robots used in rehabilitation are expensive, which limits their accessibility to patients;
- User-friendliness: Robots used in rehabilitation need to be easy to operate and require minimal training so that they can be used by patients with varying levels of physical and cognitive abilities;
- Adaptability: Robots need to be adaptable to various patient needs and abilities, which requires sophisticated algorithms and control systems;
- Safety: Robots must be safe to use, with built-in safety features to prevent accidents and injuries;
- Evidence-based: There is a need for more research to determine the effectiveness of robots in rehabilitation, and to identify the specific patient populations and conditions for which they are most useful;
- Ethical considerations: There are ethical considerations to be addressed, such as how to balance the benefits of using robots with the potential loss of human interaction and empathy.
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yedulla, N.R.; Koolmees, D.S.; Battista, E.B.; Raza, S.S.; Montgomery, Z.A.; Day, C.S. Upper-Extremity Injuries Are the 2nd Most Common Workplace Injuries from 1992 to 2018. Med. Stud. Res. Symp. 2020, 2. [Google Scholar]
- Longhi, M.; Merlo, A.; Prati, P.; Giacobbi, M.; Mazzoli, D. Instrumental indices for upper limb function assessment in stroke patients: A validation study. J. Neuroeng. Rehabil. 2016, 13, 52. [Google Scholar] [CrossRef] [PubMed]
- Beretta, E.; Cesareo, A.; Biffi, E.; Schafer, C.; Galbiati, S.; Strazzer, S. Rehabilitation of upper limb in children with acquired brain injury: A preliminary comparative study. J. Healthc. Eng. 2018, 2018, 1537170. [Google Scholar] [CrossRef] [PubMed]
- Afsar, S.I.; Mirzayev, I.; Yemisci, O.U.; Saracgil, S.N.C. Virtual reality in upper extremity rehabilitation of stroke patients: A randomized controlled trial. J. Stroke Cerebrovasc. Dis. 2018, 27, 3473–3478. [Google Scholar] [CrossRef]
- Karamians, R.; Proffitt, R.; Kline, D.; Gauthier, L.V. Effectiveness of virtual reality-and gaming-based interventions for upper extremity rehabilitation poststroke: A meta-analysis. Arch. Phys. Med. Rehabil. 2020, 101, 885–896. [Google Scholar] [CrossRef]
- Kaur, J.; Ghosh, S.; Sahani, A.K.; Sinha, J.K. Mental Imagery as a Rehabilitative Therapy for Neuropathic Pain in People with Spinal Cord Injury: A Randomized Controlled Trial. Neurorehabilit. Neural Repair 2020, 34, 1038–1049. [Google Scholar] [CrossRef]
- Noelker, L.S.; Browdie, R. Sidney Katz, MD: A New Paradigm for Chronic Illness and Long-Term Care. Gerontol. 2013, 54, 13–20. [Google Scholar] [CrossRef]
- Physiotherapy • fund OUR NHS, 2021. Available online: https://www.csp.org.uk/news/2016-07-19-almost-two-thirds-patients-muscular-dystrophy-cant-get-specialist-physio-finds-hard (accessed on 21 May 2023).
- Dobra, A. General classification of robots. Size criteria. In Proceedings of the 2014 23rd International Conference on Robotics in Alpe-Adria-Danube Region (RAAD), Smolenice, Slovakia, 3–5 September 2014; pp. 1–6. [Google Scholar] [CrossRef]
- Lo, H.S.; Xie, S.Q. Exoskeleton robots for upper-limb rehabilitation: State of the art and future prospects. Med. Eng. Phys. 2012, 34, 261–268. [Google Scholar] [CrossRef]
- Hogan, N.; Krebs, H.; Charnnarong, J.; Srikrishna, P.; Sharon, A. MIT-MANUS: A workstation for manual therapy and training. I. In Proceedings of the [1992] Proceedings IEEE International Workshop on Robot and Human Communication, Tokyo, Japan, 1–3 September 1992. [Google Scholar] [CrossRef]
- Krebs, H.; Hogan, N.; Aisen, M.; Volpe, B. Robot-aided neurorehabilitation. IEEE Trans. Rehabil. Eng. 1998, 6, 75–87. [Google Scholar] [CrossRef]
- Hesse, S.; Schulte-Tigges, G.; Konrad, M.; Bardeleben, A.; Werner, C. Robot-assisted arm trainer for the passive and active practice of bilateral forearm and wrist movements in hemiparetic subjects. Arch. Phys. Med. Rehabil. 2003, 84, 915–920. [Google Scholar] [CrossRef]
- Lum, P.S.; Burgar, C.G.; Shor, P.C.; Majmundar, M.; der Loos, M.V. Robot-assisted movement training compared with conventional therapy techniques for the rehabilitation of upper-limb motor function after stroke. Arch. Phys. Med. Rehabil. 2002, 83, 952–959. [Google Scholar] [CrossRef]
- Burgar, C.G.; Lum, P.S.; Shor, P.C.; Machiel Van der Loos, H.F. Development of robots for rehabilitation therapy: The Palo Alto VA/Stanford experience. J. Rehabil. Res. Dev. 2000, 37, 663–673. [Google Scholar]
- Xie, Q.; Meng, Q.; Zeng, Q.; Fan, Y.; Dai, Y.; Yu, H. Human-exoskeleton coupling dynamics of a multi-mode therapeutic exoskeleton for upper limb rehabilitation training. IEEE Access 2021, 9, 61998–62007. [Google Scholar] [CrossRef]
- Kim, B.; Deshpande, A.D. An upper-body rehabilitation exoskeleton Harmony with an anatomical shoulder mechanism: Design, modeling, control, and performance evaluation. Int. J. Robot. Res. 2017, 36, 414–435. [Google Scholar] [CrossRef]
- Zimmermann, Y.; Forino, A.; Riener, R.; Hutter, M. ANYexo: A versatile and dynamic upper-limb rehabilitation robot. IEEE Robot. Autom. Lett. 2019, 4, 3649–3656. [Google Scholar] [CrossRef]
- Rahman, M.H.; Rahman, M.J.; Cristobal, O.L.; Saad, M.; Kenné, J.P.; Archambault, P.S. Development of a whole arm wearable robotic exoskeleton for rehabilitation and to assist upper limb movements. Robotica 2015, 33, 19–39. [Google Scholar] [CrossRef]
- Rosati, G.; Gallina, P.; Masiero, S.; Rossi, A. Design of a new 5 d.o.f. wire-based robot for rehabilitation. In Proceedings of the 9th International Conference on Rehabilitation Robotics, ICORR 2005, Chicago, IL, USA, 28 June–1 July 2005; pp. 430–433. [Google Scholar] [CrossRef]
- Ju, M.S.; Lin, C.C.; Lin, D.H.; Hwang, I.S.; Chen, S.M. A rehabilitation robot with force-position hybrid fuzzy controller: Hybrid fuzzy control of rehabilitation robot. IEEE Trans. Neural Syst. Rehabil. Eng. 2005, 13, 349–358. [Google Scholar] [CrossRef]
- Takaiwa, M.; Noritsugu, T. Development of Wrist Rehabilitation Equipment Using Pneumatic Parallel Manipulator. In Proceedings of the 2005 IEEE International Conference on Robotics and Automation, Barcelona, Spain, 18–22 April 2005; pp. 2302–2307. [Google Scholar] [CrossRef]
- Mali, U.; Munih, M. HIFE-haptic interface for finger exercise. IEEE/Asme Trans. Mechatronics 2006, 11, 93–102. [Google Scholar] [CrossRef]
- Johnson, M.; Wisneski, K.; Anderson, J.; Nathan, D.; Smith, R. Development of ADLER: The Activities of Daily Living Exercise Robot. In Proceedings of the First IEEE/RAS-EMBS International Conference on Biomedical Robotics and Biomechatronics, BioRob 2006, Pisa, Italy, 20–22 February 2006; pp. 881–886. [Google Scholar] [CrossRef]
- Rosati, G.; Gallina, P.; Masiero, S. Design, Implementation and Clinical Tests of a Wire-Based Robot for Neurorehabilitation. IEEE Trans. Neural Syst. Rehabil. Eng. 2007, 15, 560–569. [Google Scholar] [CrossRef]
- Kung, P.C.; Ju, M.S.; Lin, C.C.K. Design of a forearm rehabilitation robot. In Proceedings of the 2007 IEEE 10th International Conference on Rehabilitation Robotics, Noordwijk, The Netherlands, 12–15 June 2007; pp. 228–233. [Google Scholar] [CrossRef]
- Song, R.; Tong, K.Y.; Hu, X.L.; Zheng, X. Myoelectrically Controlled Robotic System That Provide Voluntary Mechanical Help for Persons after Stroke. In Proceedings of the 2007 IEEE 10th International Conference on Rehabilitation Robotics, Noordwijk, The Netherlands, 12–15 June 2007; pp. 246–249. [Google Scholar] [CrossRef]
- Ellis, M.D.; Sukal, T.; DeMott, T.; Dewald, J.P. ACT3D exercise targets gravity-induced discoordination and improves reaching work area in individuals with stroke. In Proceedings of the 2007 IEEE 10th International Conference on Rehabilitation Robotics, Noordwijk, The Netherlands, 12–15 June 2007; pp. 890–895. [Google Scholar] [CrossRef]
- Sulzer, J.S.; Peshkin, M.A.; Patton, J.L. Design of a Mobile, Inexpensive Device for Upper Extremity Rehabilitation at Home. In Proceedings of the 2007 IEEE 10th International Conference on Rehabilitation Robotics, Noordwijk, The Netherlands, 12–15 June 2007; pp. 933–937. [Google Scholar] [CrossRef]
- Masia, L.; Krebs, H.I.; Cappa, P.; Hogan, N. Design, Characterization, and Impedance Limits of a Hand Robot. In Proceedings of the 2007 IEEE 10th International Conference on Rehabilitation Robotics, Noordwijk, The Netherlands, 12–15 June 2007; pp. 1085–1089. [Google Scholar] [CrossRef]
- Dovat, L.; Lambercy, O.; Gassert, R.; Maeder, T.; Milner, T.; Leong, T.C.; Burdet, E. HandCARE: A Cable-Actuated Rehabilitation System to Train Hand Function After Stroke. IEEE Trans. Neural Syst. Rehabil. Eng. 2008, 16, 582–591. [Google Scholar] [CrossRef]
- Scherer, R.; Pradhan, S.; Dellon, B.; Kim, D.; Klatzky, R.; Matsuoka, Y. Characterization of multi-finger twist motion toward robotic rehabilitation. In Proceedings of the 2009 IEEE International Conference on Rehabilitation Robotics, Kyoto, Japan, 23–26 June 2009; pp. 812–817. [Google Scholar] [CrossRef]
- Oda, K.; Isozumi, S.; Ohyama, Y.; Tamida, K.; Kikuchi, T.; Furusho, J. Development of isokinetic and iso-contractile exercise machine “MEM-MRB” using MR brake. In Proceedings of the 2009 IEEE International Conference on Rehabilitation Robotics, Kyoto, Japan, 23–26 June 2009; pp. 6–11. [Google Scholar] [CrossRef]
- Chang, J.J.; Tung, W.L.; Wu, W.L.; Huang, M.H.; Su, F.C. Effects of Robot-Aided Bilateral Force-Induced Isokinetic Arm Training Combined with Conventional Rehabilitation on Arm Motor Function in Patients With Chronic Stroke. Arch. Phys. Med. Rehabil. 2007, 88, 1332–1338. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.S.; Ju, M.S.; Lin, C.C.K. Improving Elbow Torque Output of Stroke Patients with Assistive Torque Controlled by EMG Signals. J. Biomech. Eng. 2004, 125, 881–886. [Google Scholar] [CrossRef] [PubMed]
- Freeman, C.T.; Hughes, A.M.; Burridge, J.H.; Chappell, P.H.; Lewin, P.L.; Rogers, E. A robotic workstation for stroke rehabilitation of the upper extremity using FES. Med. Eng. Phys. 2009, 31, 364–373. [Google Scholar] [CrossRef]
- Hesse, S.; Kuhlmann, H.; Wilk, J.; Tomelleri, C.; Kirker, S.G.B. A new electromechanical trainer for sensorimotor rehabilitation of paralysed fingers: A case series in chronic and acute stroke patients. J. Neuroeng. Rehabil. 2008, 5, 21. [Google Scholar] [CrossRef] [PubMed]
- Micera, S.; Carrozza, M.C.; Guglielmelli, E.; Cappiello, G.; Zaccone, F.; Freschi, C.; Colombo, R.; Mazzone, A.; Delconte, C.; Pisano, F.; et al. A Simple Robotic System for Neurorehabilitation. Auton. Robot. 2005, 19, 271. [Google Scholar] [CrossRef]
- Colombo, R.; Pisano, F.; Mazzone, A.; Delconte, C.; Micera, S.; Carrozza, M.C.; Dario, P.; Minuco, G. Design strategies to improve patient motivation during robot-aided rehabilitation. J. Neuroeng. Rehabil. 2007, 4, 3. [Google Scholar] [CrossRef]
- Bouzit, M.; Burdea, G.; Popescu, G.; Boian, R. The Rutgers Master II-new design force-feedback glove. IEEE/Asme Trans. Mechatronics 2002, 7, 256–263. [Google Scholar] [CrossRef]
- Kiguchi, K.; Esaki, R.; Tsuruta, T.; Watanabe, K.; Fukuda, T. An exoskeleton system for elbow joint motion rehabilitation. In Proceedings of the Proceedings 2003 IEEE/ASME International Conference on Advanced Intelligent Mechatronics (AIM 2003), Kobe, Japan, 20–24 July 2003; Volume 2, pp. 1228–1233. [Google Scholar] [CrossRef]
- Sarakoglou, I.; Tsagarakis, N.; Caldwell, D. Occupational and physical therapy using a hand exoskeleton based exerciser. In Proceedings of the 2004 IEEE/RSJ International Conference on Intelligent Robots and Systems (IROS) (IEEE Cat. No.04CH37566), Sendai, Japan, 28 September–2 October 2004; Volume 3, pp. 2973–2978. [Google Scholar] [CrossRef]
- Kline, T.; Kamper, D.; Schmit, B. Control system for pneumatically controlled glove to assist in grasp activities. In Proceedings of the 9th International Conference on Rehabilitation Robotics, ICORR 2005, Chicago, IL, USA, 28 June–1 July 2005; pp. 78–81. [Google Scholar] [CrossRef]
- Loureiro, R.; Belda-Lois, J.; Lima, E.; Pons, J.; Sanchez-Lacuesta, J.; Harwin, W. Upper limb tremor suppression in ADL via an orthosis incorporating a controllable double viscous beam actuator. In Proceedings of the 99th International Conference on Rehabilitation Robotics, ICORR 2005, Chicago, IL, USA, 28 June–1 July 2005; pp. 119–122. [Google Scholar] [CrossRef]
- Mulas, M.; Folgheraiter, M.; Gini, G. An EMG-controlled exoskeleton for hand rehabilitation. In Proceedings of the 9th International Conference on Rehabilitation Robotics, ICORR 2005, Chicago, IL, USA, 28 June–1 July 2005; pp. 371–374. [Google Scholar] [CrossRef]
- Sanchez, R.; Wolbrecht, E.; Smith, R.; Liu, J.; Rao, S.; Cramer, S.; Rahman, T.; Bobrow, J.; Reinkensmeyer, D. A pneumatic robot for re-training arm movement after stroke: Rationale and mechanical design. In Proceedings of the 9th International Conference on Rehabilitation Robotics, ICORR 2005, Chicago, IL, USA, 28 June–1 July 2005; pp. 500–504. [Google Scholar] [CrossRef]
- Wege, A.; Hommel, G. Development and control of a hand exoskeleton for rehabilitation of hand injuries. In Proceedings of the 2005 IEEE/RSJ International Conference on Intelligent Robots and Systems, Edmonton, AB, Canada, 2–6 August 2005; pp. 3046–3051. [Google Scholar] [CrossRef]
- Sasaki, D.; Noritsugu, T.; Takaiwa, M. Development of Active Support Splint driven by Pneumatic Soft Actuator (ASSIST). In Proceedings of the Proceedings of the 2005 IEEE International Conference on Robotics and Automation, Barcelona, Spain, 18–22 April 2005, pp.; pp. 520–525. [CrossRef]
- Winter, S.H.; Bouzit, M. Use of Magnetorheological Fluid in a Force Feedback Glove. IEEE Trans. Neural Syst. Rehabil. Eng. 2007, 15, 2–8. [Google Scholar] [CrossRef]
- Rocon, E.; Belda-Lois, J.M.; Ruiz, A.F.; Manto, M.; Moreno, J.C.; Pons, J.L. Design and Validation of a Rehabilitation Robotic Exoskeleton for Tremor Assessment and Suppression. IEEE Trans. Neural Syst. Rehabil. Eng. 2007, 15, 367–378. [Google Scholar] [CrossRef]
- Worsnopp, T.; Peshkin, M.; Colgate, J.; Kamper, D. An Actuated Finger Exoskeleton for Hand Rehabilitation Following Stroke. In Proceedings of the 2007 IEEE 10th International Conference on Rehabilitation Robotics, Noordwijk, The Netherlands, 12–15 June 2007; pp. 896–901. [Google Scholar] [CrossRef]
- Song, R.; Tong, K.y.; Hu, X.; Li, L. Assistive Control System Using Continuous Myoelectric Signal in Robot-Aided Arm Training for Patients After Stroke. IEEE Trans. Neural Syst. Rehabil. Eng. 2008, 16, 371–379. [Google Scholar] [CrossRef]
- Xing, K.; Xu, Q.; He, J.; Wang, Y.; Liu, Z.; Huang, X. A wearable device for repetitive hand therapy. In Proceedings of the 2008 2nd IEEE RAS EMBS International Conference on Biomedical Robotics and Biomechatronics, Scottsdale, AZ, USA, 19–22 October 2008; pp. 919–923. [Google Scholar] [CrossRef]
- Gopura, R.; Kiguchi, K. A human forearm and wrist motion assist exoskeleton robot with EMG-based Fuzzy-neuro control. In Proceedings of the 2008 2nd IEEE RAS EMBS International Conference on Biomedical Robotics and Biomechatronics, Scottsdale, AZ, USA, 19–22 October 2008; pp. 550–555. [Google Scholar] [CrossRef]
- Ding, M.; Ueda, J.; Ogasawara, T. Pinpointed muscle force control using a power-assisting device: System configuration and experiment. In Proceedings of the 2008 2nd IEEE RAS EMBS International Conference on Biomedical Robotics and Biomechatronics, Scottsdale, AZ, USA, 19–22 October 2008; pp. 181–186. [Google Scholar] [CrossRef]
- Nathan, D.E.; Johnson, M.J.; McGuire, J. Feasibility of integrating FES grasp assistance with a task-oriented robot-assisted therapy environment: A case study. In Proceedings of the 2008 2nd IEEE RAS EMBS International Conference on Biomedical Robotics and Biomechatronics, Scottsdale, AZ, USA, 19–22 October 2008; pp. 807–812. [Google Scholar] [CrossRef]
- Rotella, M.F.; Reuther, K.E.; Hofmann, C.L.; Hage, E.B.; BuSha, B.F. An orthotic hand-assistive exoskeleton for actuated pinch and grasp. In Proceedings of the 2009 IEEE 35th Annual Northeast Bioengineering Conference, Cambridge, MA, USA, 3–5 April 2009; pp. 1–2. [Google Scholar] [CrossRef]
- Vanderniepen, I.; Van Ham, R.; Van Damme, M.; Versluys, R.; Lefeber, D. Orthopaedic rehabilitation: A powered elbow orthosis using compliant actuation. In Proceedings of the 2009 IEEE International Conference on Rehabilitation Robotics, Kyoto, Japan, 23–26 June 2009; pp. 172–177. [Google Scholar] [CrossRef]
- Ertas, I.H.; Hocaoglu, E.; Barkana, D.E.; Patoglu, V. Finger exoskeleton for treatment of tendon injuries. In Proceedings of the 2009 IEEE International Conference on Rehabilitation Robotics, Kyoto, Japan, 23–26 June 2009; pp. 194–201. [Google Scholar] [CrossRef]
- Pylatiuk, C.; Kargov, A.; Gaiser, I.; Werner, T.; Schulz, S.; Bretthauer, G. Design of a flexible fluidic actuation system for a hybrid elbow orthosis. In Proceedings of the 2009 IEEE International Conference on Rehabilitation Robotics, Kyoto, Japan, 23–26 June 2009; pp. 167–171. [Google Scholar] [CrossRef]
- Rosati, G.; Cenci, S.; Boschetti, G.; Zanotto, D.; Masiero, S. Design of a single-dof active hand orthosis for neurorehabilitation. In Proceedings of the 2009 IEEE International Conference on Rehabilitation Robotics, Kyoto, Japan, 23–26 June 2009; pp. 161–166. [Google Scholar] [CrossRef]
- Chen, M.; Ho, S.; Zhou, H.F.; Pang, P.M.; Hu, X.; Ng, D.; Tong, K.Y. Interactive rehabilitation robot for hand function training. In Proceedings of the 2009 IEEE International Conference on Rehabilitation Robotics, Kyoto, Japan, 23–26 June 2009; pp. 777–780. [Google Scholar] [CrossRef]
- Gopura, R.A.R.C.; Kiguchi, K.; Li, Y. SUEFUL-7: A 7DOF upper-limb exoskeleton robot with muscle-model-oriented EMG-based control. In Proceedings of the 2009 IEEE/RSJ International Conference on Intelligent Robots and Systems, St. Louis, MO, USA, 10–15 October 2009; pp. 1126–1131. [Google Scholar] [CrossRef]
- Klein, J.; Spencer, S.; Allington, J.; Bobrow, J.E.; Reinkensmeyer, D.J. Optimization of a Parallel Shoulder Mechanism to Achieve a High-Force, Low-Mass, Robotic-Arm Exoskeleton. IEEE Trans. Robot. 2010, 26, 710–715. [Google Scholar] [CrossRef]
- Tong, K.; Ho, S.; Pang, P.; Hu, X.; Tam, W.; Fung, K.; Wei, X.; Chen, P.; Chen, M. An intention driven hand functions task training robotic system. In Proceedings of the 2010 Annual International Conference of the IEEE Engineering in Medicine and Biology, Buenos Aires, Argentina, 31 August–4 September 2010; pp. 3406–3409. [Google Scholar] [CrossRef]
- Ho, N.S.K.; Tong, K.Y.; Hu, X.L.; Fung, K.L.; Wei, X.J.; Rong, W.; Susanto, E.A. An EMG-driven exoskeleton hand robotic training device on chronic stroke subjects: Task training system for stroke rehabilitation. In Proceedings of the 2011 IEEE International Conference on Rehabilitation Robotics, Zurich, Switzerland, 29 June–1 July 2011; pp. 1–5. [Google Scholar] [CrossRef]
- Pignolo, L.; Dolce, G.; Basta, G.; Lucca, L.F.; Serra, S.; Sannita, W.G. Upper limb rehabilitation after stroke: ARAMIS a “robo-mechatronic” innovative approach and prototype. In Proceedings of the 2012 4th IEEE RAS EMBS International Conference on Biomedical Robotics and Biomechatronics (BioRob), Rome, Italy, 24–27 June 2012; pp. 1410–1414. [Google Scholar] [CrossRef]
- Lalitharatne, T.D.; Teramoto, K.; Hayashi, Y.; Tamura, K.; Kiguchi, K. EEG-based evaluation for perception-assist in upper-limb power-assist exoskeletons. In Proceedings of the 2014 World Automation Congress (WAC), Waikoloa, HI, USA, 3–7 August 2014; pp. 307–312. [Google Scholar] [CrossRef]
- Cozens, J. Robotic assistance of an active upper limb exercise in neurologically impaired patients. IEEE Trans. Rehabil. Eng. 1999, 7, 254–256. [Google Scholar] [CrossRef]
- Rosen, J.; Brand, M.; Fuchs, M.; Arcan, M. A myosignal-based powered exoskeleton system. IEEE Trans. Syst. Man, Cybern.- Part Syst. Humans 2001, 31, 210–222. [Google Scholar] [CrossRef]
- Cordo, P.; Lutsep, H.; Cordo, L.; Wright, W.G.; Cacciatore, T.; Skoss, R. Assisted Movement With Enhanced Sensation (AMES): Coupling Motor and Sensory to Remediate Motor Deficits in Chronic Stroke Patients. Neurorehabilit. Neural Repair 2008, 23, 67–77. [Google Scholar] [CrossRef]
- Fleischer, C.; Kondak, K.; Wege, A.; Kossyk, I. Research on Exoskeletons at the TU Berlin. In Advances in Robotics Research: Theory, Implementation, Application; Kröger, T., Wahl, F.M., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 335–346. [Google Scholar] [CrossRef]
- Fuxiang, Z. An Embedded Control Platform of a Continuous Passive Motion Machine for Injured Fingers. In Rehabilitation Robotics; Kommu, S.S., Ed.; IntechOpen: Rijeka, Croatia, 2007; Chapter 31. [Google Scholar] [CrossRef]
- Gupta, A.; O’Malley, M.K.; Patoglu, V.; Burgar, C. Design, Control and Performance of RiceWrist: A Force Feedback Wrist Exoskeleton for Rehabilitation and Training. Int. J. Robot. Res. 2008, 27, 233–251. [Google Scholar] [CrossRef]
- Hu, X.; Tong, K.; Song, R.; Zheng, X.; Lui, K.; Leung, W.; Ng, S.; Au-Yeung, S. Quantitative evaluation of motor functional recovery process in chronic stroke patients during robot-assisted wrist training. J. Electromyogr. Kinesiol. 2009, 19, 639–650. [Google Scholar] [CrossRef]
- Kiguchi, K.; Iwami, K.; Yasuda, M.; Watanabe, K.; Fukuda, T. An exoskeletal robot for human shoulder joint motion assist. IEEE/Asme Trans. Mechatronics 2003, 8, 125–135. [Google Scholar] [CrossRef]
- Kiguchi, K.; Rahman, M.H.; Sasaki, M.; Teramoto, K. Development of a 3DOF mobile exoskeleton robot for human upper-limb motion assist. Robot. Auton. Syst. 2008, 56, 678–691. [Google Scholar] [CrossRef]
- Lucas, L.; DiCicco, M.; Matsuoka, Y. An EMG-Controlled Hand Exoskeleton for Natural Pinching. J. Robot. Mechatronics 2004, 16. [Google Scholar] [CrossRef]
- Mavroidis, C.; Nikitczuk, J.; Weinberg, B.; Danaher, G.; Jensen, K.; Pelletier, P.; Prugnarola, J.; Stuart, R.; Arango, R.; Leahey, M.; et al. Smart portable rehabilitation devices. J. Neuroeng. Rehabil. 2005, 2, 18. [Google Scholar] [CrossRef]
- Nef, T.; Guidali, M.; Riener, R. ARMin III – arm therapy exoskeleton with an ergonomic shoulder actuation. Appl. Bionics Biomech. 2009, 6, 127–142. [Google Scholar] [CrossRef]
- Oldewurtel, F.; Mihelj, M.; Nef, T.; Riener, R. Patient-cooperative control strategies for coordinated functional arm movements. In Proceedings of the 2007 European Control Conference (ECC), Kos, Greece, 2–5 July 2007. [Google Scholar] [CrossRef]
- Ögce, F.; Özyalçin, H. Case study: A myoelectrically controlled shoulderelbow orthosis for unrecovered brachial plexus injury. Prosthetics Orthot. Int. 2000, 24. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, R.; Liu, J.; Rao, S.; Shah, P.; Smith, R.; Rahman, T.; Cramer, S.; Bobrow, J.; Reinkensmeyer, D. Automating Arm Movement Training Following Severe Stroke: Functional Exercises with Quantitative Feedback in a Gravity-Reduced Environment. IEEE Trans. Neural Syst. Rehabil. Eng. 2006, 14, 378–389. [Google Scholar] [CrossRef] [PubMed]
- Schabowsky, C.N.; Godfrey, S.B.; Holley, R.J.; Lum, P.S. Development and pilot testing of HEXORR: Hand EXOskeleton Rehabilitation Robot. J. Neuroeng. Rehabil. 2010, 7, 36. [Google Scholar] [CrossRef]
- Stein, J.; Narendran, K.; McBean, J.; Krebs, K.; Hughes, R. Electromyography-Controlled Exoskeletal Upper-Limb-Powered Orthosis for Exercise Training after Stroke. Am. J. Phys. Med. Rehabil. 2007, 86. [Google Scholar] [CrossRef]
- Turner, M.L.; Gomez, D.H.; Tremblay, M.R.; Cutkosky, M.R. Preliminary Tests of an Arm-Grounded Haptic Feedback Device in Telemanipulation. In Proceedings of the Dynamic Systems and Control. American Society of Mechanical Engineers, Anaheim, CA, USA, 15–20 November 1998. [Google Scholar] [CrossRef]
- Moubarak, S.; Pham, M.T.; Pajdla, T.; Redarce, T. Design Results of an Upper Extremity Exoskeleton. In Proceedings of the 4th European Conference of the International Federation for Medical and Biological Engineering, Antwerp, Belgium, 23–27 November 2008; Vander Sloten, J., Verdonck, P., Nyssen, M., Haueisen, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 1687–1690. [Google Scholar]
- Bressi, F.; Campagnola, B.; Cricenti, L.; Santacaterina, F.; Miccinilli, S.; Di Pino, G.; Bravi, M. Upper limb home-based robotic rehabilitation in chronic stroke patients: A pilot study. Front. Neurorobotics 2023, 17. [Google Scholar] [CrossRef]
- Khan, M.M.R.; Rahman, M.M.; De Caro, J.S.; Wang, I.; Rahman, M. An End-Effector Type Therapeutic Robot for Home-Based Upper Limb Rehabilitation. Arch. Phys. Med. Rehabil. 2022, 103, 146–147. [Google Scholar] [CrossRef]
- Hu, X.L.; Tong, R.K.y.; Ho, N.S.K.; Xue, J.j.; Rong, W.; Li, L.S.W. Wrist Rehabilitation Assisted by an Electromyography-Driven Neuromuscular Electrical Stimulation Robot after Stroke. Neurorehabilit. Neural Repair 2015, 29, 767–776. [Google Scholar] [CrossRef]
- Gassert, R.; Dietz, V. Rehabilitation robots for the treatment of sensorimotor deficits: A neurophysiological perspective. J. Neuroeng. Rehabil. 2018, 15, 46. [Google Scholar] [CrossRef]
- Proietti, T.; Crocher, V.; Roby-Brami, A.; Jarrassé, N. Upper-Limb Robotic Exoskeletons for Neurorehabilitation: A Review on Control Strategies. IEEE Rev. Biomed. Eng. 2016, 9, 4–14. [Google Scholar] [CrossRef]
- Kitago, T.; Krakauer, J.W. Motor learning principles for neurorehabilitation. In Neurological Rehabilitation; Elsevier: Amsterdam, The Netherlands, 2013; pp. 93–103. [Google Scholar] [CrossRef]
- Luft, A.R.; McCombe-Waller, S.; Whitall, J.; Forrester, L.W.; Macko, R.; Sorkin, J.D.; Schulz, J.B.; Goldberg, A.P.; Hanley, D.F. Repetitive bilateral arm training and motor cortex activation in chronic stroke: A randomized controlled trial. JAMA 2004, 292, 1853–1861. [Google Scholar] [CrossRef]
- Blanco Ortega, A.; Magadán Salazar, A.; Guzmán Valdivia, C.H.; Gómez Becerra, F.A.; Palacios Gallegos, M.J.; García Velarde, M.A.; Santana Camilo, J.A. CNC Machines for Rehabilitation: Ankle and Shoulder. Machines 2022, 10, 1055. [Google Scholar] [CrossRef]
- Trigili, E.; Crea, S.; Moisè, M.; Baldoni, A.; Cempini, M.; Ercolini, G.; Marconi, D.; Posteraro, F.; Carrozza, M.C.; Vitiello, N. Design and experimental characterization of a shoulder-elbow exoskeleton with compliant joints for post-stroke rehabilitation. IEEE/Asme Trans. Mechatronics 2019, 24, 1485–1496. [Google Scholar] [CrossRef]
- Rodríguez-León, J.F.; Castillo-Castañeda, E.; Aguilar-Pereyra, J.F.; Carbone, G. Experimental Characterization of A-AFiM, an Adaptable Assistive Device for Finger Motions. Machines 2022, 10, 280. [Google Scholar] [CrossRef]
- Seçkin, M.; Turan, N.Y. Rehabilitation glove device design. J. Eng. Technol. Appl. Sci. 2018, 3, 75–81. [Google Scholar] [CrossRef]
- Thalman, C.M.; Lam, Q.P.; Nguyen, P.H.; Sridar, S.; Polygerinos, P. A novel soft elbow exosuit to supplement bicep lifting capacity. In Proceedings of the 2018 IEEE/RSJ International Conference on Intelligent Robots and Systems (IROS), Madrid, Spain, 1–5 October 2018; pp. 6965–6971. [Google Scholar]
- Tiboni, M.; Borboni, A.; Faglia, R.; Pellegrini, N. Robotics rehabilitation of the elbow based on surface electromyography signals. Adv. Mech. Eng. 2018, 10, 1687814018754590. [Google Scholar] [CrossRef]
- Tiboni, M.; Legnani, G.; Lancini, M.; Serpelloni, M.; Gobbo, M.; Fausti, D. ERRSE: Elbow robotic rehabilitation system with an EMG-based force control. In Advances in Service and Industrial Robotics: Proceedings of the 26th International Conference on Robotics in Alpe-Adria-Danube Region, RAAD 2017, Turin, Italy, 21–23 June 2017; Springer: Berlin/Heidelberg, Germany, 2018; pp. 892–900. [Google Scholar]
- Tsabedze, T.; Trinh, J.; Alomran, A.; Clayton, J.; Zhang, J. Design and Characterization of AWARD: An Active Wearable Assistive and Resistive Device. In Proceedings of the 2022 IEEE/ASME International Conference on Advanced Intelligent Mechatronics (AIM), Sapporo, Japan, 11–15 July 2022; pp. 844–849. [Google Scholar]
- Zhang, L.; Guo, S.; Sun, Q. An assist-as-needed controller for passive, assistant, active, and resistive robot-aided rehabilitation training of the upper extremity. Appl. Sci. 2020, 11, 340. [Google Scholar] [CrossRef]
- Lin, C.H.; Su, Y.Y.; Lai, Y.H.; Lan, C.C. A spatial-motion assist-as-needed controller for the passive, active, and resistive robot-aided rehabilitation of the wrist. IEEE Access 2020, 8, 133951–133960. [Google Scholar] [CrossRef]
- Krebs, H.I.; Palazzolo, J.J.; Dipietro, L.; Ferraro, M.; Krol, J.; Rannekleiv, K.; Volpe, B.T.; Hogan, N. Rehabilitation Robotics: Performance-Based Progressive Robot-Assisted Therapy. Auton. Robot. 2003, 15, 7–20. [Google Scholar] [CrossRef]
- Krebs, H.I.; Volpe, B.T. Rehabilitation robotics. Handb. Clin. Neurol. 2013, 110, 283–294. [Google Scholar]
- Hogan, N. Impedance Control: An Approach to Manipulation: Part I—Theory. J. Dyn. Syst. Meas. Control 1985, 107, 1–7. [Google Scholar] [CrossRef]
- Lalitharatne, T.D.; Teramoto, K.; Hayashi, Y.; Kiguchi, K. Evaluation of perception-assist with an upper-limb power-assist exoskeleton using EMG and EEG signals. In Proceedings of the 11th IEEE International Conference on Networking, Sensing and Control, Miami, FL, USA, 7–9 April 2014; pp. 524–529. [Google Scholar] [CrossRef]
- Popovic, M.R.; Masani, K.; Micera, S. Functional Electrical Stimulation Therapy: Recovery of Function Following Spinal Cord Injury and Stroke. In Neurorehabilitation Technology; Springer International Publishing: New York, NY, USA, 2016; pp. 513–532. [Google Scholar] [CrossRef]
- Joyo, M.K.; Raza, Y.; Kadir, K.; Naidu, K.; Ahmed, S.F.; Khan, S. Firefly Optimised PID Control for Upper Extremity Rehabilitation Robot. In Proceedings of the 2019 IEEE International Conference on Smart Instrumentation, Measurement and Application (ICSIMA), Kuala Lumpur, Malaysia, 27–29 August 2019; pp. 1–5. [Google Scholar]
- Jagodnik, K.M.; Blana, D.; van den Bogert, A.J.; Kirsch, R.F. An optimized proportional-derivative controller for the human upper extremity with gravity. J. Biomech. 2015, 48, 3692–3700. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Iibuchi, K.; Kurosawa, K.; Hoshimiya, N. A method of multichannel PID control of two-degree-of-freedom wrist joint movements by functional electrical stimulation. Syst. Comput. Jpn. 2003, 34, 25–36. [Google Scholar] [CrossRef]
- Yu, W.; Rosen, J. A novel linear PID controller for an upper limb exoskeleton. In Proceedings of the 49th IEEE Conference on Decision and Control (CDC), Atlanta, GA, USA, 15–17 December 2010; pp. 3548–3553. [Google Scholar]
- Islam, M.R.; Rahmani, M.; Rahman, M.H. A novel exoskeleton with fractional sliding mode control for upper limb rehabilitation. Robotica 2020, 38, 2099–2120. [Google Scholar] [CrossRef]
- Rahmani, M.; Rahman, M.H. An upper-limb exoskeleton robot control using a novel fast fuzzy sliding mode control. J. Intell. Fuzzy Syst. 2019, 36, 2581–2592. [Google Scholar] [CrossRef]
- Babaiasl, M.; Goldar, S.N.; Barhaghtalab, M.H.; Meigoli, V. Sliding mode control of an exoskeleton robot for use in upper-limb rehabilitation. In Proceedings of the 2015 3rd RSI International Conference on Robotics and Mechatronics (ICROM), Tehran, Iran, 7–9 October 2015; pp. 694–701. [Google Scholar]
- Madoński, R.; Kordasz, M.; Sauer, P. Application of a disturbance-rejection controller for robotic-enhanced limb rehabilitation trainings. Isa Trans. 2014, 53, 899–908. [Google Scholar] [CrossRef]
- Aole, S.; Elamvazuthi, I.; Waghmare, L.; Patre, B.; Meriaudeau, F. Non-linear active disturbance rejection control for upper limb rehabilitation exoskeleton. Proc. Inst. Mech. Eng. Part J. Syst. Control. Eng. 2021, 235, 606–632. [Google Scholar] [CrossRef]
- Aole, S.; Elamvazuthi, I.; Waghmare, L.; Patre, B.; Bhaskarwar, T.; Meriaudeau, F.; Su, S. Active Disturbance Rejection Control Based Sinusoidal Trajectory Tracking for an Upper Limb Robotic Rehabilitation Exoskeleton. Appl. Sci. 2022, 12, 1287. [Google Scholar] [CrossRef]
- Li, H.; Cheng, L.; Li, Z.; Xue, W. Active disturbance rejection control for a fluid-driven hand rehabilitation device. IEEE/Asme Trans. Mechatronics 2020, 26, 841–853. [Google Scholar] [CrossRef]
- Xu, G.; Song, A.; Li, H. Adaptive impedance control for upper-limb rehabilitation robot using evolutionary dynamic recurrent fuzzy neural network. J. Intell. Robot. Syst. 2011, 62, 501–525. [Google Scholar] [CrossRef]
- Wu, Q.; Chen, B.; Wu, H. Adaptive admittance control of an upper extremity rehabilitation robot with neural-network-based disturbance observer. IEEE Access 2019, 7, 123807–123819. [Google Scholar] [CrossRef]
- Krakauer, J.W. Motor learning and consolidation: The case of visuomotor rotation. Adv. Exp. Med. Biol. 2009, 629, 405–421. [Google Scholar]
- Hallett, M.; Grafman, J. Executive Function and Motor Skill Learning. In International Review of Neurobiology; Schmahmann, J.D., Ed.; Academic Press: Cambridge, MA, USA, 1997; Volume 41, pp. 297–323. [Google Scholar] [CrossRef]
- Reis, J.; Schambra, H.M.; Cohen, L.G.; Buch, E.R.; Fritsch, B.; Zarahn, E.; Celnik, P.A.; Krakauer, J.W. Noninvasive cortical stimulation enhances motor skill acquisition over multiple days through an effect on consolidation. Proc. Natl. Acad. Sci. USA 2009, 106, 1590–1595. [Google Scholar] [CrossRef]
- Shmuelof, L.; Krakauer, J.W.; Mazzoni, P. How is a motor skill learned? Change and invariance at the levels of task success and trajectory control. J. Neurophysiol. 2012, 108, 578–594. [Google Scholar] [CrossRef]
- Nadel, L. Encyclopedia of Cognitive Science; Nature Publishing Group: London, UK, 2003. [Google Scholar]
- Patton, J.; Mussa-Ivaldi, F. Robot-assisted adaptive training: Custom force fields for teaching movement patterns. IEEE Trans. Biomed. Eng. 2004, 51, 636–646. [Google Scholar] [CrossRef]
- Pohl, P.S.; McDowd, J.M.; Filion, D.; Richards, L.G.; Stiers, W. Implicit learning of a motor skill after mild and moderate stroke. Clin. Rehabil. 2006, 20, 246–253. [Google Scholar] [CrossRef]
- Reinkensmeyer, D.J.; Pang, T.J.; Nessler, J.A.; Pang, C.T. Java Therapy: Web-Based Robotic Rehabilitation. In Proceedings of the 7th International Conference on Rehabilitation Robotics, ICORR 2001, Integration of Assistive Technology in the Information Age, Evry, France, 25–27 April 2001; Volume 9, pp. 66–71. [Google Scholar]
- Laffont, I.; Froger, J.; Jourdan, C.; Bakhti, K.; van Dokkum, L.E.; Gouaich, A.; Bonnin, H.Y.; Armingaud, P.; Jaussent, A.; Picot, M.C.; et al. Rehabilitation of the upper arm early after stroke: Video games versus conventional rehabilitation. A randomized controlled trial. Ann. Phys. Rehabil. Med. 2020, 63, 173–180. [Google Scholar] [CrossRef]
- Woo, L.K.; Beom, K.S.; Hwa, L.J.; Joung, L.S.; Wan, K.J. Effect of Robot-Assisted Game Training on Upper Extremity Function in Stroke Patients. Ann. Rehabil. Med. 2017, 41, 539–546. [Google Scholar] [CrossRef]
- Woo, L.K.; Beom, K.S.; Hwa, L.J.; Joung, L.S.; Wan, Y.S. Effect of Upper Extremity Robot-Assisted Exercise on Spasticity in Stroke Patients. Ann. Rehabil. Med. 2016, 40, 961–971. [Google Scholar] [CrossRef]
- Weiss, P.L.; Sveistrup, H.; Rand, D.; Kizony, R. Video capture virtual reality: A decade of rehabilitation assessment and intervention. Phys. Ther. Rev. 2009, 14, 307–321. [Google Scholar] [CrossRef]
- Levin, M.F.; Weiss, P.L.; Keshner, E.A. Emergence of Virtual Reality as a Tool for Upper Limb Rehabilitation: Incorporation of Motor Control and Motor Learning Principles. Phys. Ther. 2015, 95, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Triandafilou, K.M.; Tsoupikova, D.; Barry, A.J.; Thielbar, K.N.; Stoykov, N.; Kamper, D.G. Development of a 3D, networked multi-user virtual reality environment for home therapy after stroke. J. Neuroeng. Rehabil. 2018, 15, 88. [Google Scholar] [CrossRef] [PubMed]
- Thielbar, K.O.; Triandafilou, K.M.; Barry, A.J.; Yuan, N.; Nishimoto, A.; Johnson, J.; Stoykov, M.E.; Tsoupikova, D.; Kamper, D.G. Home-based Upper Extremity Stroke Therapy Using a Multiuser Virtual Reality Environment: A Randomized Trial. Arch. Phys. Med. Rehabil. 2020, 101, 196–203. [Google Scholar] [CrossRef]
- Laver, K.; Lange, B.; George, S.; Deutsch, J.; Saposnik, G.; Crotty, M. Virtual reality for stroke rehabilitation. Cochrane Database Syst. Rev. 2017, 11, CD008349. [Google Scholar] [CrossRef] [PubMed]
- Fugl-Meyer, A.R.; Jääskö, L.; Leyman, I.; Olsson, S.; Steglind, S. The post-stroke hemiplegic patient. 1. a method for evaluation of physical performance. Scand. J. Rehabil. Med. 1975, 7, 13–31. [Google Scholar]
- Gladstone, D.J.; Danells, C.J.; Black, S.E. The fugl-meyer assessment of motor recovery after stroke: A critical review of its measurement properties. Neurorehabilit. Neural Repair 2002, 16, 232–240. [Google Scholar] [CrossRef]
- Lyle, R.C. A performance test for assessment of upper limb function in physical rehabilitation treatment and research. Int. J. Rehabil. Res. 1981, 4, 483–492. [Google Scholar] [CrossRef]
- Wolf, S.L.; Lecraw, D.E.; Barton, L.A.; Jann, B.B. Forced use of hemiplegic upper extremities to reverse the effect of learned nonuse among chronic stroke and head-injured patients. Exp. Neurol. 1989, 104, 125–132. [Google Scholar] [CrossRef]
- Wolf, S.L.; Thompson, P.A.; Morris, D.M.; Rose, D.K.; Winstein, C.J.; Taub, E.; Giuliani, C.; Pearson, S.L. The EXCITE trial: Attributes of the Wolf Motor Function Test in patients with subacute stroke. Neurorehabilit. Neural Repair 2005, 19, 194–205. [Google Scholar] [CrossRef]
- Duncan, P.W.; Wallace, D.; Lai, S.M.; Johnson, D.; Embretson, S.; Laster, L.J. The Stroke Impact Scale Version 2.0. Stroke 1999, 30, 2131–2140. [Google Scholar] [CrossRef]
- Duncan, P.W.; Bode, R.K.; Min Lai, S.; Perera, S. Rasch analysis of a new stroke-specific outcome scale: The Stroke Impact Scale. Arch. Phys. Med. Rehabil. 2003, 84, 950–963. [Google Scholar] [CrossRef]
- Mahoney, F.I.; Barthel, D.W. Functional evaluation: The Barthel index. Md. State Med J. 1965, 14, 61–65. [Google Scholar]
- Collin, C.; Wade, D.T.; Davies, S.; Horne, V. The Barthel ADL Index: A reliability study. Int. Disabil. Stud. 1988, 10, 61–63. [Google Scholar] [CrossRef]
- Dijkers, M.P.; deBear, P.C.; Erlandson, R.F.; Kristy, K.; Geer, D.M.; Nichols, A. Patient and staff acceptance of robotic technology in occupational therapy: A pilot study. J. Rehabil. Res. Dev. 1991, 28, 33–44. [Google Scholar] [CrossRef]
- Rodgers, H.; Bosomworth, H.; Krebs, H.I.; van Wijck, F.; Howel, D.; Wilson, N.; Aird, L.; Alvarado, N.; Andole, S.; Cohen, D.L.; et al. Robot assisted training for the upper limb after stroke (RATULS): A multicentre randomised controlled trial. Lancet 2019, 394, 51–62. [Google Scholar] [CrossRef]
- Housman, S.J.; Scott, K.M.; Reinkensmeyer, D.J. A Randomized Controlled Trial of Gravity-Supported, Computer-Enhanced Arm Exercise for Individuals With Severe Hemiparesis. Neurorehabilit. Neural Repair 2009, 23, 505–514. [Google Scholar] [CrossRef]
- Lum, P.; Burgar, C.; Van der Loos, M.; Shor, P.; Majmundar, M.; Yap, R. The MIME robotic system for upper-limb neuro-rehabilitation: Results from a clinical trial in subacute stroke. In Proceedings of the 9th International Conference on Rehabilitation Robotics, ICORR 2005, Chicago, IL, USA, 28 June–1 July 2005; pp. 511–514. [Google Scholar] [CrossRef]
- Hesse, S.; Werner, C.; Pohl, M.; Rueckriem, S.; Mehrholz, J.; Lingnau, M. Computerized Arm Training Improves the Motor Control of the Severely Affected Arm After Stroke. Stroke 2005, 36, 1960–1966. [Google Scholar] [CrossRef]
- Reinkensmeyer, D.J.; Wolbrecht, E.T.; Chan, V.; Chou, C.; Cramer, S.C.; Bobrow, J.E. Comparison of Three-Dimensional, Assist-as-Needed Robotic Arm/Hand Movement Training Provided with Pneu-WREX to Conventional Tabletop Therapy ater Chronic Stroke. Am. J. Phys. Med. Rehabil. 2012, 91. [Google Scholar] [CrossRef]
- Klamroth-Marganska, V.; Blanco, J.; Campen, K.; Curt, A.; Dietz, V.; Ettlin, T.; Felder, M.; Fellinghauer, B.; Guidali, M.; Kollmar, A.; et al. Three-dimensional, task-specific robot therapy of the arm after stroke: A multicentre, parallel-group randomised trial. Lancet Neurol. 2014, 13, 159–166. [Google Scholar] [CrossRef]
- Lin, I.H.; Tsai, H.T.; Wang, C.Y.; Hsu, C.Y.; Liou, T.H.; Lin, Y.N. Effectiveness and Superiority of Rehabilitative Treatments in Enhancing Motor Recovery within 6 Months Poststroke: A Systemic Review. Arch. Phys. Med. Rehabil. 2019, 100, 366–378. [Google Scholar] [CrossRef]
- Riener, R.; Nef, T.; Colombo, G. Robot-aided neurorehabilitation of the upper extremities. Med. Biol. Eng. Comput. 2005, 43, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Hung, Y.H.; Chen, P.J.; Lin, W.Z. Design factors and opportunities of rehabilitation robots in upper-limb training after stroke. In Proceedings of the 2017 14th International Conference on Ubiquitous Robots and Ambient Intelligence (URAI), Jeju, Republic of Korea, 28 June–1 July 2017; pp. 650–654. [Google Scholar]
- Di Lazzaro, V.; Capone, F.; Di Pino, G.; Pellegrino, G.; Florio, L.; Zollo, L.; Simonetti, D.; Ranieri, F.; Brunelli, N.; Corbetto, M.; et al. Combining robotic training and non-invasive brain stimulation in severe upper limb-impaired chronic stroke patients. Front. Neurosci. 2016, 10, 88. [Google Scholar] [CrossRef] [PubMed]
- Rong, W.; Li, W.; Pang, M.; Hu, J.; Wei, X.; Yang, B.; Wai, H.; Zheng, X.; Hu, X. A Neuromuscular Electrical Stimulation (NMES) and robot hybrid system for multi-joint coordinated upper limb rehabilitation after stroke. J. Neuroeng. Rehabil. 2017, 14, 1–13. [Google Scholar] [CrossRef] [PubMed]








| Active Therapy | Passive Therapy | Bilateral Therapy | |
|---|---|---|---|
| Subtypes | Active Assistive | Passive Assistive | - |
| Active Resistive | Continuous Passive Motion | ||
| Advantages | - Efficient for advanced rehabilitation treatments | - Ideal for early stages of post-stroke symptoms | - Ideal for specific cases (e.g., Hemiplegia) |
| - Has feedback information | - Easy to implement | - Simplicity | |
| Disadvantages | - Needs patient interaction | - Does not get feedback from the patient | - Needs having some undamaged parts in the patient’s body |
| - Can be complex to design | - Needs to be tuned continuously | - Only used for a few specific cases (e.g., hemiplegia) | |
| Examples | [11,12,14,15,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,35,36,38,39,40,41,42,43,44,45,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,74,75,76,77,78,79,80,81,82,84,85,86] | [13,14,15,19,26,37,46,58,59,60,62,73,83] | [13,14,15,34,70] |
| Therapy | Movement | Paper | Year | DoF | Comment |
|---|---|---|---|---|---|
| CPM | Finger | [61,73], | 2007, 2009 | 4,1 | [59,73] are single-finger systems |
| Shoulder | [95] | 2022 | 2 | Provides the movements of dorsiflexion (DF), plantarflexion (PF), abduction (AB), and adduction (AD). | |
| PA | Sholder, Elbow, and Forearm | [46,83] | 2006, 2005 | 5,5 | [46,83] are Exoskeletons with Gravity Compensation technique |
| Sholder and Elbow | [96] | 2019 | 4 | Upper-limb neurorehabilitation and treatment of spasticity | |
| AA | Finger | [23,30,32,40,42,43,45,47,49,51,53,56,57,61,65,66,72,78,84,86,97,98] | 2022, 2018, 2011, 2010, 2010, 2009, 2009, 2009, 2009, 2008, 2008, 2007, 2007, 2007, 2006, 2005, 2005, 2005, 2004, 2004, 2002, 1998 | 4, 1, 5, 2, 10, 6, 1, 4, 20, 6, 2, 8, 5, 3, 2, 1, 2, 4, 3, 7, 5, 5 | Controlled Independently: [49,65,66,72,86,97]. Controlled Together: [30,32,43,56,98], Single Finger: [23,47,78]. The authors in [97] adopted four slider-crank mechanisms, each fixed, and with movement of one finger. In the reference [98]: A device consisting of a glove, a microcontroller, and a motor has been considered. |
| Elbow | [29,33,35,41,52,69,70,79,82,85,99] | 2018, 2009, 2008, 2007, 2007, 2005, 2004, 2003, 2001, 2000, 1999 | [29] is 3, others: 1 | [99]: The design consists of an array of pneumatically pressurized soft actuators, End-Effector systems: [29,33,35], others: Exoskeletons | |
| Wrist | [11,12,27,39,44,48,75] | 2009, 2007, 2007, 2005, 2005, 1992, 1992 | 1, 1, 1, 1, 1, 5, 5 | End-Effector systems: [11,12,27], others: Exoskeletons | |
| Shoulder | [76] | 2003 | 2 | Exoskeleton robot | |
| Shoulder and Elbow | [20,21,25,28,36,38,64] | 2010, 2009, 2009, 2007, 2007, 2005, 2005, 2005 | 4, 2, 4, 3, 3, 5, 2, 2 | All techniques have adopted Admittance Control | |
| Forearm and Wrist | [54] | 2008 | 3 | Exoskeleton robot | |
| Shoulder, Elbow, and Forearm | [24,67] | 2012,2006, | 6,3 | All techniques have adopted Admittance Control | |
| Shoulder, Elbow, Forearm, and Wrist | [63,68,80,81] | 2014, 2009, 2009, 2009 | 7,7,7,7 | All techniques have adopted Admittance Control | |
| AR | Forearm and Wrist | [50,74,100] | 2018, 2008, 2007 | 1, 4,3 | In the reference [100], an electromyography signal is used as an input to drive the joint movement [50] includes Elbow movement also |
| Elbow, Forearm, and Wrist | [50] | 2007 | 3 | Exoskeleton robot | |
| BT | Shoulder and Elbow | [34] | 2007 | 2 | End-Effector robot |
| Therapy | Movement | Paper | Year | DoF | Comment |
|---|---|---|---|---|---|
| CPM, AA | Finger | [59,62] | 2009, 2009 | 6,2 | [59] is a single-finger system |
| Elbow | [58,60] | 2009, 2009 | 1,1 | [58,60] are Exoskeletons | |
| Shoulder | [19] | 2015 | 7 | Exoskeleton robot | |
| Forearm | [26] | 2007 | 1 | End-Effector robot | |
| AA, AR | Elbow | [101] | 2018 | 1 | Surface EMG measurements are used to implement a force-based active and resistive control. |
| Finger | [102] | 2022 | 1 | The device was actuated by six twisted string actuators (TSAs) | |
| [31] | 2008 | 5 | Have used Admittance and Impedance Control | ||
| Elbow, Forearm, and Wrist | [55] | 2008 | 4 | Have used Admittance and Impedance Control | |
| BT, CPM | Forearm, and Wrist | [13] | 2003 | 1 | End-Effector robot |
| BT, AA | Elbow | [70] | 2001 | 1 | Exoskeleton robot |
| AR, BT, AA, CPM | Shoulder and Elbow | [14,15] | 2000, 2000 | 6,6 | Have used Admittance and Impedance Control |
| PA, AA, AR | Arm | [103] | 2020 | 6 | Tested with an elderly female participant |
| Wrist | [104] | 2020 | Up to 3 | Robot consists of Series elastic actuators with high torque-to-weight ratios |
| Robot | Advantages | Disadvantages |
|---|---|---|
| ETS-MARSE [19] | - Mimics natural human spine motion - Can be used for studying the biomechanics of the spine and testing spinal implants and surgical techniques | - Not designed for use in clinical rehabilitation settings - Expensive and complex to build and operate |
| Handcare [31] | - Designed specifically for hand and finger therapy - Lightweight and easy to use - Provides personalized and goal-oriented therapy | - Limited to hand and finger therapy only - Relatively new technology, may not be widely available |
| PNEU-WREX [46] | - Can assist with wrist and hand movements - Lightweight and easy to put on and take off - Has shown promise in improving upper limb function and independence | - Limited to upper limb therapy only - May not be suitable for individuals with severe upper limb impairments - Requires additional training for clinicians and therapists to use |
| surface Electro- MyoGraphy (sEMG) [76] | - Adaptive assistance that is natural and responsive to patient’s movements
- Fine-tuned adjustments based on patient’s needs - Can learn and adapt to patient’s needs over time. | - Requires sophisticated control algorithms and sensors
- May be expensive and complex to develop and maintain. |
| MyoPro (https://myomo.com/what-is-a-myopro-orthosis/, access on 25 April 2023) | - Provides powered assistance for upper limb movement - Easy to use | - Expensive - Limited evidence of efficacy |
| Rapael Smart Glove (https://www.neofect.com/us/smart-glove, access on 25 April 2023) | - Interactive training with haptic feedback - Wearable design | - Limited range of motion supported - Expensive |
| Harmony [17] | - Anatomically aligned shoulder mechanism - Unconstrained mobility of all joints - Supports body weight - Provides assistive force | - Expensive - Limited evidence of efficacy - Limited range of motion supported |
| ANYexo [18] | - Adaptable and customizable to different arm sizes and levels of assistance - Controlled by a smartphone app or joystick | - Experimental device not yet widely tested in clinical trials - Limited evidence of efficacy |
| Controller Input | Reference |
|---|---|
| Force/Torque | [13,14,15,19,21,36,39,79,80,81] |
| Optical Encoders | [14,15] |
| Position | [11,12,13,21,23,28,34,36,42,53] |
| Angular velocity | [11,12,33] |
| EMG | [27,35,41,45,52,58,60,62,63,65,66,68,70,72,75,76,78,82,85] |
| Joint angle | [26,27,29,42,43,47,48,51,59,64,69,72,86] |
| Cylinder pressure | [64] |
| Actuator | Reference |
|---|---|
| DC | [11,12,14,15,20,23,25,27,30,31,34,35,36,37,38,41,42,45,47,48,51,52,57,59,61,62,63,66,68,70,72,76,77,79,80,81,82,84,85,86,87] |
| AC | [19,21,26,29,58,83,84] |
| Hydraulic | [60] |
| Pneumatic | [22,40,43,46,53,64,78] |
| FES | [36,56] |
| Class | Technique | Paper | Year | Exp/Sim | Comment |
|---|---|---|---|---|---|
| PID | Optimized PID | [110] | 2019 | Sim | Controlling an exoskeleton of a three-DoF system designed to facilitate the movements of the elbow and the shoulder |
| [111] | 2015 | Sim | Controlling a musculoskeletal system based on a five-DoF arm model and 22 muscles | ||
| MIMO PID | [112] | 2003 | Exp | A trajectory control of a two-DoF wrist joint with neurologically intact subjects | |
| Linear PID | [113] | 2010 | Sim | EXO-UL7 robot | |
| Robust | Fractional SMC | [114] | 2020 | Sim | Design of a 7 DoF upper limb robotic exoskeleton (u-Rob) which was controlled using fractional SMC |
| Fuzzy SMC | [115] | 2019 | Sim | A seven-DoF upper-limb exoskeleton robot was controlled using Fuzzy SMC | |
| [116] | 2015 | Sim | A mechanical design of a new three-DOF exoskeleton robot for shoulder joint rehabilitation was also proposed. The parameters of the SMC controller were optimized using GA | ||
| Adaptive | ADRC | [117] | 2014 | Exp | The experiments were conducted on a model of a flexible joint robot, which imitates a real rehabilitation robot. |
| NLADRC | [118] | 2021 | Sim | NLADRC and NLESO were adopted to track a sinusoidal path for a two-link model of an upper limb rehabilitation exoskeleton. | |
| ADRC | [119] | 2022 | Sim | LESO and FTSTD techniques were adopted to estimate the status of the system and to reject the disturbances. | |
| ADRC | [120] | 2020 | Exp, Clinical | ADRC and RESO were utilized to control a proposed rehabilitation device made from elastomeric materials. | |
| AI-Based | EDRFNN model | [121] | 2011 | Sim | GA, HEP, and BP techniques were adopted to optimize the parameters of the model. |
| RBF NN | [122] | 2019 | Exp | The proposed control system contained a disturbance observer with a radial basis function network |
| Class | Advantages | Disadvantages | Papers |
|---|---|---|---|
| PID | - Simplicity | - Not optimal | [110,111,112,113] |
| - Process independent | - Suffer from derivative noise amplification | ||
| - Acceptable performance with tuned parameters | - Needs tuning | ||
| Robust control | - Advanced performance in the presence of bounded uncertainties and disturbances | - Cannot handle unbounded uncertainties and disturbances | [114,115,116] |
| - Relatively Simple | - More complex than PID | ||
| - Stability can be proved using Lyapunov theory | - Chattering (for SMC controllers) | ||
| Adaptive control | - Advanced performance in the presence of unbounded uncertainties and disturbances | - Not practical with large dimension systems | [117,118,119,120] |
| AI-Based control | - applicable to non-mathematical models | - Needs to be trained | [121,122] |
| - Efficient in predicting models | - Good predictions need large data | ||
| - Non-linear nature | - Overfitting problems |
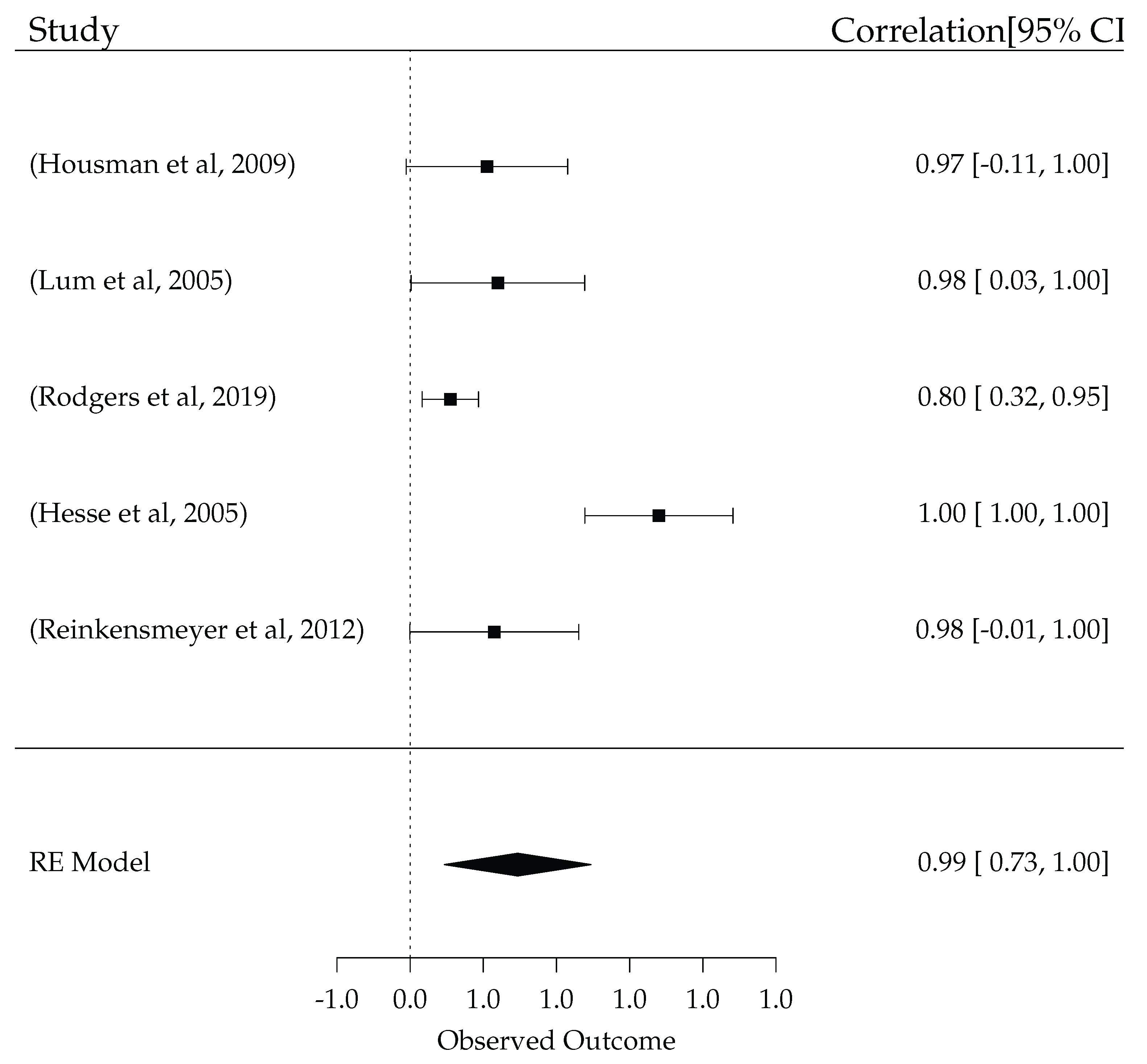
| Housman et al. [150] | Lum et al. [151] | Rodgers et al. [149] | Hesse et al. [152] | Reinkensmeyer et al. [153] | ||
|---|---|---|---|---|---|---|
| Study Duration (Weeks) | 8, s = 24 | 4, s = 15 | 12, s = 36 | 6, s = 30 | 8–9, s = 24 | |
| Follow-Up (Months) | 6 | 6 | 6 | 3 | 3 | |
| n of Sex(m/f) | Control | 7/7 | 4/2 | 101/153 | 12/10 | 12/1 |
| Experimental | 11/3 | 2/3 | 101/156 | 12/10 | 5/8 | |
| Age | Control | |||||
| Experimental | ||||||
| Stroke | Control | 8 ischemic, 5 hemorrhagic, 1 unknown | No Info. | 214 cerebral infarction, 38 primary intracerebral haemorrhage, 2 subarachnoid haemorrhage | No Info. | 4 ischemic, 6 hemorrhagic, 3 unknown |
| Experimental | 9 ischemic (1 with hemorrhagic conversion), 4 hemorrhagic, 1 unknown | No Info. | 197 cerebral infarction, 58 primary intracerebral haemorrhage, 2 subarachnoid haemorrhage | No Info. | 9 ischemic, 2 hemorrhagic, 3 unknown | |
| FMA-UE (out of 66) | ||||||
| Control | Baseline | |||||
| Change AS3 | ||||||
| Change AF4 | ||||||
| Experimental | Baseline | |||||
| Change AS3 | ||||||
| Change AF4 | ||||||
| Characteristics of experimental interventions used in clinical trials | ||||||
| Robot | [83] | [14,15] | [11] | [13] | [46] | |
| Robot Type | Exoskeleton | End-Effector | End-Effector | End-Effector | Exoskeleton | |
| Degrees of Freedom | 5 | 6 | 5 | 1 | 5 | |
| Control Strategem | Gravity Compensation | Admittance Control | Admittance Control | Admittance Control | Gravity Compensation | |
| Type of Therapy | Passive | Bilateral | Active | Bilateral | Passive | |
| Motor Learning Strategy | Assistance | Mirroring | Assistance | Mirroring | Assistance | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fareh, R.; Elsabe, A.; Baziyad, M.; Kawser, T.; Brahmi, B.; Rahman, M.H. Will Your Next Therapist Be a Robot?—A Review of the Advancements in Robotic Upper Extremity Rehabilitation. Sensors 2023, 23, 5054. https://doi.org/10.3390/s23115054
Fareh R, Elsabe A, Baziyad M, Kawser T, Brahmi B, Rahman MH. Will Your Next Therapist Be a Robot?—A Review of the Advancements in Robotic Upper Extremity Rehabilitation. Sensors. 2023; 23(11):5054. https://doi.org/10.3390/s23115054
Chicago/Turabian StyleFareh, Raouf, Ammar Elsabe, Mohammed Baziyad, Tunajjina Kawser, Brahim Brahmi, and Mohammad H. Rahman. 2023. "Will Your Next Therapist Be a Robot?—A Review of the Advancements in Robotic Upper Extremity Rehabilitation" Sensors 23, no. 11: 5054. https://doi.org/10.3390/s23115054
APA StyleFareh, R., Elsabe, A., Baziyad, M., Kawser, T., Brahmi, B., & Rahman, M. H. (2023). Will Your Next Therapist Be a Robot?—A Review of the Advancements in Robotic Upper Extremity Rehabilitation. Sensors, 23(11), 5054. https://doi.org/10.3390/s23115054