IoT Based Smart Monitoring of Patients’ with Acute Heart Failure
Abstract
:1. Introduction
- Designed an efficient smart healthcare system based on IoT and cloud-based technologies to provide a timely health care service to heart-failure patients using a deep learning model;
- This is the first study to design a smart healthcare system to monitor heart failure patients using the Heart-failure-clinical-records-dataset;
- Performance of deep learning models is investigated in predicting the survival of heart patients;
- The performance of the proposed CNN deep learning model is compared with MLP, RNN, LSTM and ML-based algorithms trained on the same dataset.
2. Related Work
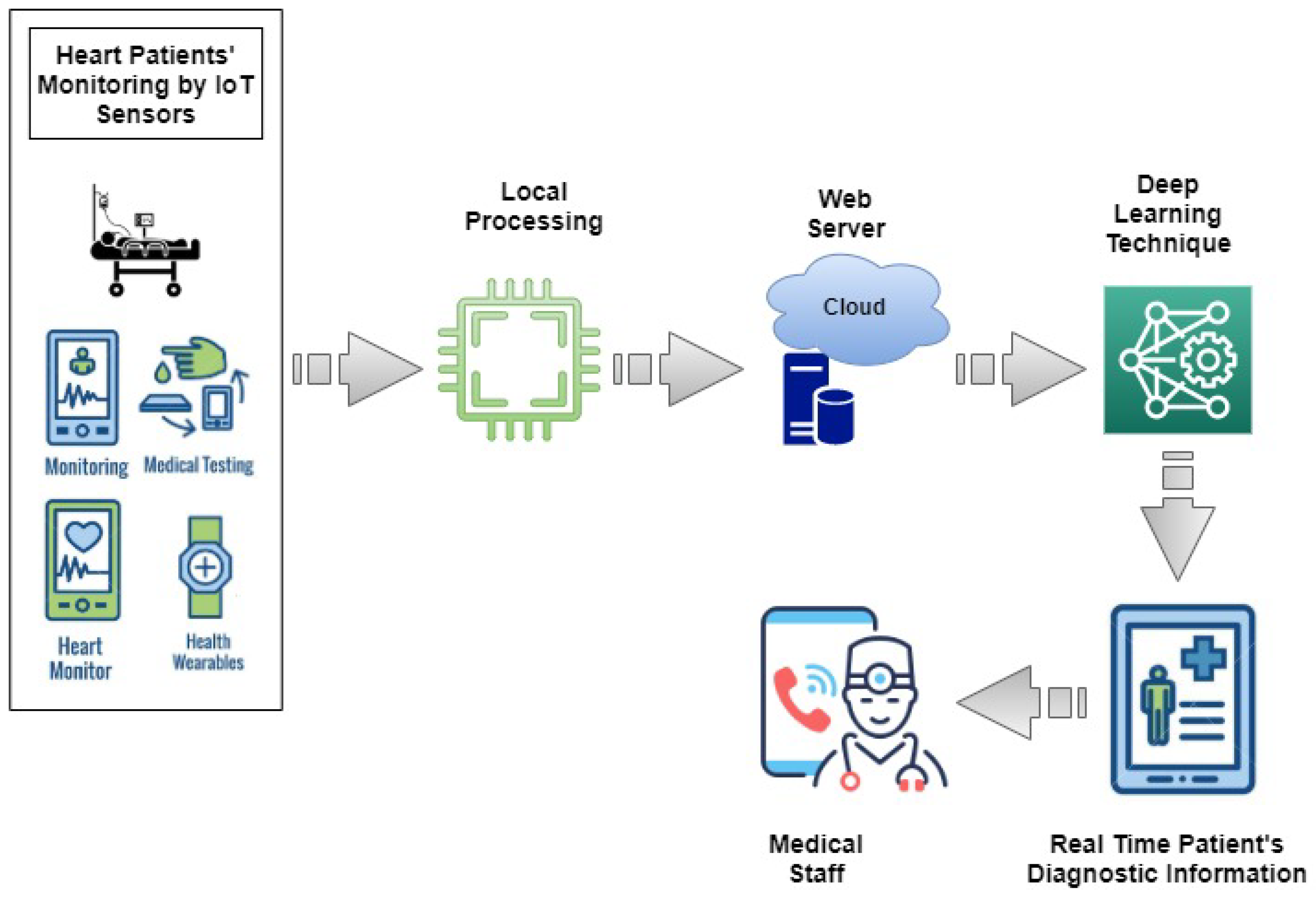
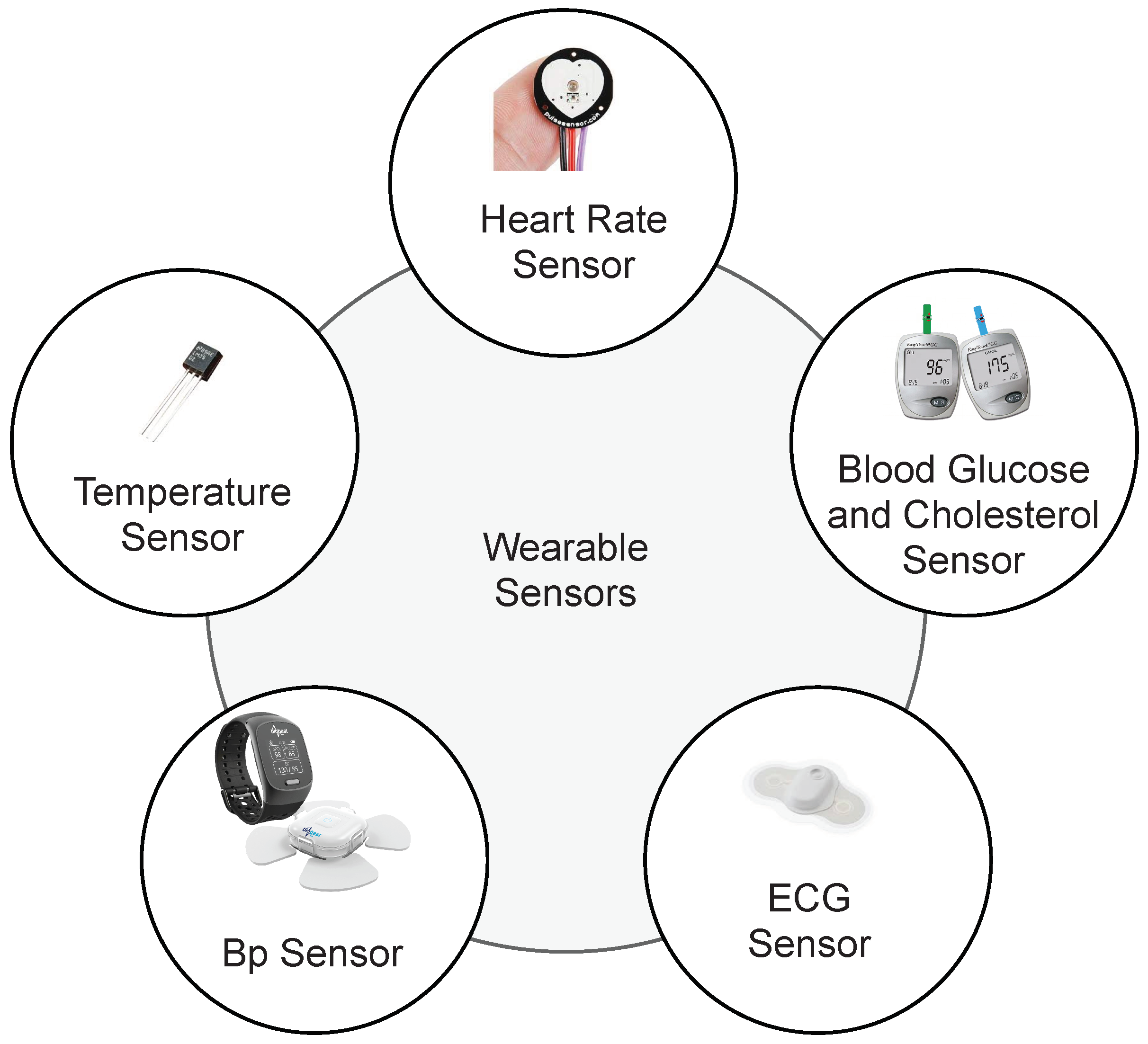
3. Smart Healthcare Framework
| Algorithm 1 The steps of the proposed IoT architecture based on the Deep Learning Model. |
| Read: The smart medical healthcare sensors data. Connect: Make connection to firebase database for transferring data. Authentication: Medical officer authentication. IF: Transfer==’successful’ 1. Transferring of smart medical healthcare sensors data using JSON dump method. 2. Already trained deep learning model make predictions on sensor data. 3. Based on predictions medical report of patient is generated with remarks as prediction. 4. Report is sent back from firebase cloud storage to medical officer device. Else: Transfer==’Unsuccessful’ 1. Smart medical healthcare sensors data is stored in the shared preference of the device. 2. Shared preference data is sent to firebase cloud storage whenever the connectivity is successful. |
4. Materials and Methods
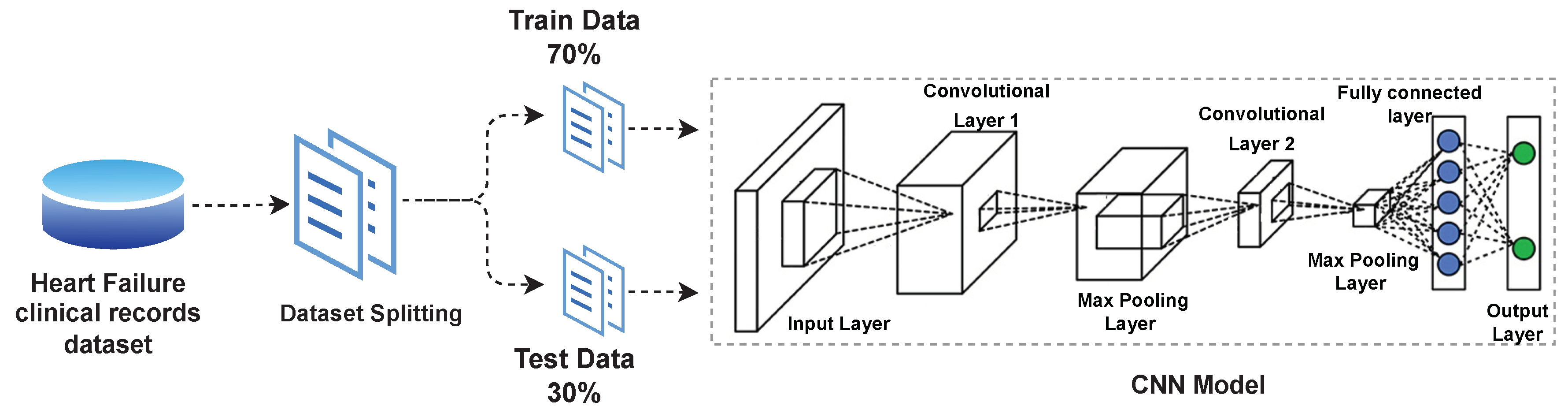
4.1. Dataset
4.2. Deep Leaning Models
4.2.1. Multilayer Perceptron Neural Network
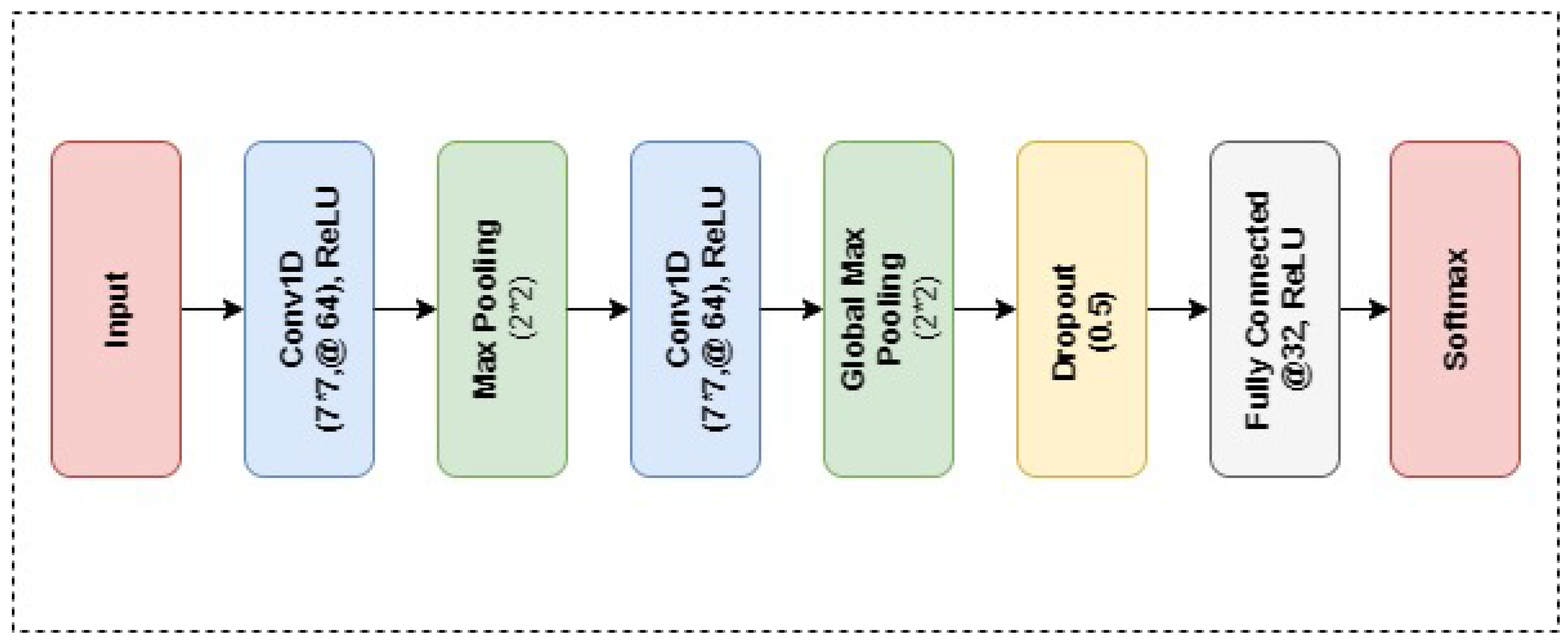
4.2.2. CNN
4.3. RNN
4.4. LSTM
5. Experimental Design
5.1. Experimental Details
5.2. Evaluation Measures
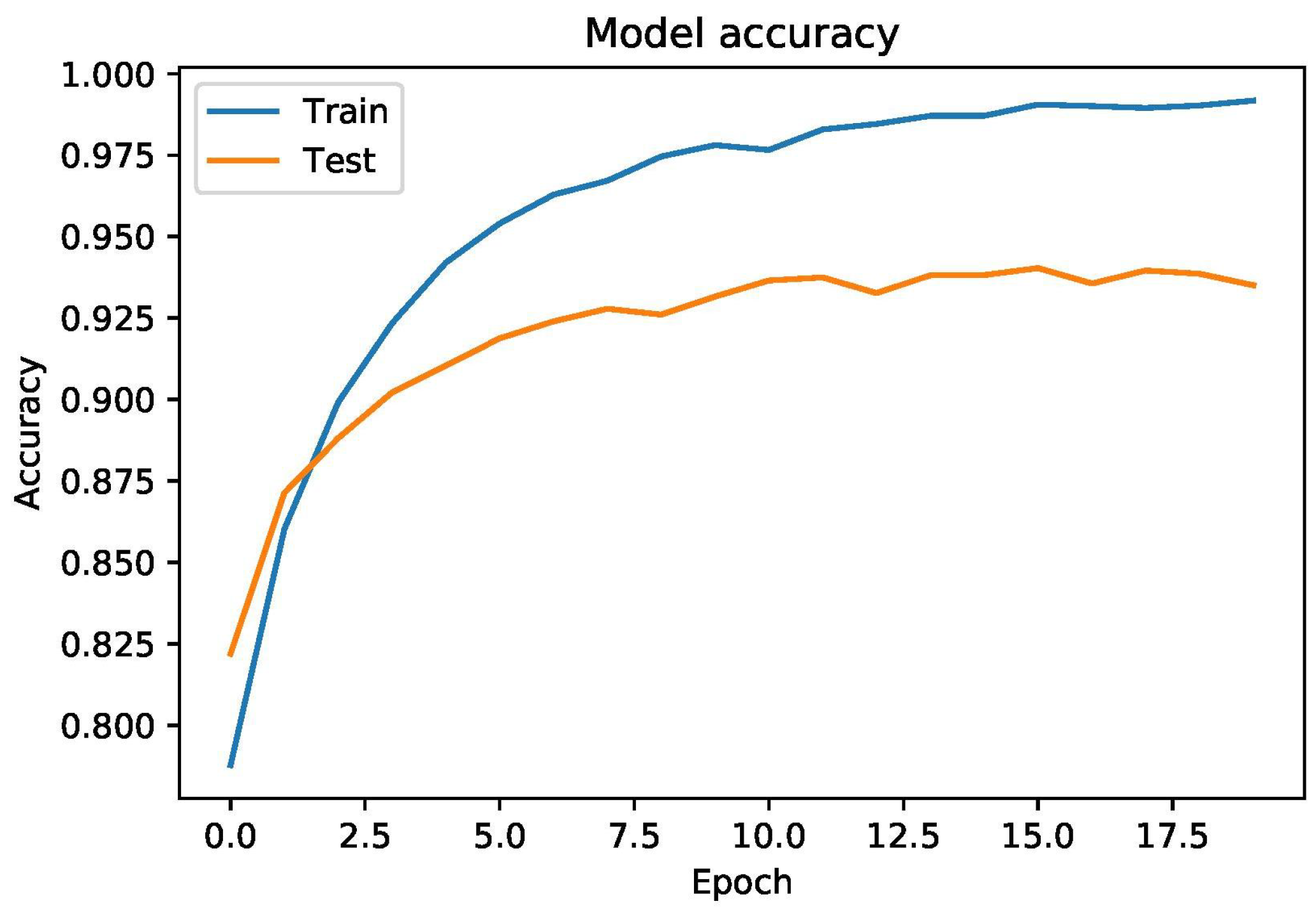
6. Results & Discussions
6.1. Results
6.1.1. Comparison with ML-Based Models
6.1.2. Comparison with Deep Transfer Learning Models
6.1.3. Validation of the Proposed Model
6.2. Discussions
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yuehong, Y.; Zeng, Y.; Chen, X.; Fan, Y. The internet of things in healthcare: An overview. J. Ind. Inf. Integr. 2016, 1, 3–13. [Google Scholar]
- Alhussein, M.; Muhammad, G.; Hossain, M.S.; Amin, S.U. Cognitive IoT-cloud integration for smart healthcare: Case study for epileptic seizure detection and monitoring. Mob. Networks Appl. 2018, 23, 1624–1635. [Google Scholar] [CrossRef]
- Hossain, M.S.; Muhammad, G. Emotion recognition using deep learning approach from audio–visual emotional big data. Inf. Fusion 2019, 49, 69–78. [Google Scholar] [CrossRef]
- Hossain, M.S.; Muhammad, G. Cloud-assisted industrial internet of things (iiot)–enabled framework for health monitoring. Comput. Netw. 2016, 101, 192–202. [Google Scholar] [CrossRef]
- Li, D.; Deng, L.; Gupta, B.B.; Wang, H.; Choi, C. A novel CNN based security guaranteed image watermarking generation scenario for smart city applications. Inf. Sci. 2019, 479, 432–447. [Google Scholar] [CrossRef]
- Castiglione, A.; Umer, M.; Sadiq, S.; Obaidat, M.S.; Vijayakumar, P. The Role of Internet of Things to Control the Outbreak of COVID-19 Pandemic. IEEE Internet Things J. 2021, 8, 16072–16082. [Google Scholar] [CrossRef]
- World Health Organization. Cardiovascular Diseases (CVDs). Available online: http://www.who.int/cardiovascular_diseases/en/ (accessed on 16 July 2019).
- Association, A.H. Classes of Heart Failure. Available online: https://www.heart.org/en/health-topics/heart-failure/what-is-heart-failure/classes-of-heart-failure (accessed on 11 August 2018).
- Arabasadi, Z.; Alizadehsani, R.; Roshanzamir, M.; Moosaei, H.; Yarifard, A.A. Computer aided decision making for heart disease detection using hybrid neural network-Genetic algorithm. Comput. Methods Programs Biomed. 2017, 141, 19–26. [Google Scholar] [CrossRef]
- Shalev-Shwartz, S.; Ben-David, S. Understanding Machine Learning: From Theory to Algorithms; Cambridge University Press: Cambridge, UK, 2014. [Google Scholar]
- Weng, S.F.; Reps, J.; Kai, J.; Garibaldi, J.M.; Qureshi, N. Can machine-learning improve cardiovascular risk prediction using routine clinical data? PLoS ONE 2017, 12, e0174944. [Google Scholar] [CrossRef] [Green Version]
- Ramalingam, V.; Dandapath, A.; Raja, M.K. Heart disease prediction using machine learning techniques: A survey. Int. J. Eng. Technol. 2018, 7, 684–687. [Google Scholar] [CrossRef] [Green Version]
- Ahmad, T.; Munir, A.; Bhatti, S.H.; Aftab, M.; Raza, M.A. Survival analysis of heart failure patients: A case study. PLoS ONE 2017, 12, e0181001. [Google Scholar] [CrossRef]
- Zahid, F.M.; Ramzan, S.; Faisal, S.; Hussain, I. Gender based survival prediction models for heart failure patients: A case study in Pakistan. PLoS ONE 2019, 14, e0210602. [Google Scholar] [CrossRef] [PubMed]
- Chicco, D.; Jurman, G. Machine learning can predict survival of patients with heart failure from serum creatinine and ejection fraction alone. BMC Med. Inform. Decis. Mak. 2020, 20, 16. [Google Scholar] [CrossRef] [PubMed]
- Ishaq, A.; Sadiq, S.; Umer, M.; Ullah, S.; Mirjalili, S.; Rupapara, V.; Nappi, M. Improving the Prediction of Heart Failure Patients’ Survival Using SMOTE and Effective Data Mining Techniques. IEEE Access 2021, 9, 39707–39716. [Google Scholar] [CrossRef]
- Hossain, M.S.; Muhammad, G. Emotion-aware connected healthcare big data towards 5G. IEEE Internet Things J. 2017, 5, 2399–2406. [Google Scholar] [CrossRef]
- Jain, P.; Joshi, A.M.; Mohanty, S.P. iGLU: An intelligent device for accurate noninvasive blood glucose-level monitoring in smart healthcare. IEEE Consum. Electron. Mag. 2019, 9, 35–42. [Google Scholar] [CrossRef]
- Samani, H.; Zhu, R. Robotic automated external defibrillator ambulance for emergency medical service in smart cities. IEEE Access 2016, 4, 268–283. [Google Scholar] [CrossRef]
- Stergiou, C.L.; Psannis, K.E.; Gupta, B.B. IoT-based Big Data secure management in the Fog over a 6G Wireless Network. IEEE Internet Things J. 2020. [Google Scholar] [CrossRef]
- Ghoneim, A.; Muhammad, G.; Amin, S.U.; Gupta, B. Medical image forgery detection for smart healthcare. IEEE Commun. Mag. 2018, 56, 33–37. [Google Scholar] [CrossRef]
- Gupta, B.; Quamara, M. An overview of Internet of Things (IoT): Architectural aspects, challenges, and protocols. Concurr. Comput. Pract. Exp. 2020, 32, e4946. [Google Scholar] [CrossRef]
- Melillo, P.; De Luca, N.; Bracale, M.; Pecchia, L. Classification tree for risk assessment in patients suffering from congestive heart failure via long-term heart rate variability. IEEE J. Biomed. Health Inform. 2013, 17, 727–733. [Google Scholar] [CrossRef]
- Guidi, G.; Pettenati, M.C.; Melillo, P.; Iadanza, E. A machine learning system to improve heart failure patient assistance. IEEE J. Biomed. Health Inform. 2014, 18, 1750–1756. [Google Scholar] [CrossRef] [PubMed]
- Parthiban, G.; Srivatsa, S. Applying machine learning methods in diagnosing heart disease for diabetic patients. Int. J. Appl. Inf. Syst. 2012, 3, 25–30. [Google Scholar] [CrossRef]
- Chapman, B.; DeVore, A.D.; Mentz, R.J.; Metra, M. Clinical profiles in acute heart failure: An urgent need for a new approach. ESC Heart Fail. 2019, 6, 464–474. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ansarullah, S.I.; Kumar, P. A Systematic Literature Review on Cardiovascular Disorder Identification using Knowledge Mining and Machine Learning Method. Int. J. Recent Technol. Eng. 2019, 7, 1009–1015. [Google Scholar]
- Shah, D.; Patel, S.; Bharti, S.K. Heart Disease Prediction using Machine Learning Techniques. SN Comput. Sci. 2020, 1, 1–6. [Google Scholar] [CrossRef]
- Mohan, S.; Thirumalai, C.; Srivastava, G. Effective heart disease prediction using hybrid machine learning techniques. IEEE Access 2019, 7, 81542–81554. [Google Scholar] [CrossRef]
- Geweid, G.G.; Abdallah, M.A. A New Automatic Identification Method of Heart Failure Using Improved Support Vector Machine Based on Duality Optimization Technique. IEEE Access 2019, 7, 149595–149611. [Google Scholar] [CrossRef]
- Manogaran, G.; Varatharajan, R.; Priyan, M. Hybrid recommendation system for heart disease diagnosis based on multiple kernel learning with adaptive neuro-fuzzy inference system. Multimed. Tools Appl. 2018, 77, 4379–4399. [Google Scholar] [CrossRef]
- Domingos, P. A few useful things to know about machine learning. Commun. ACM 2012, 55, 78–87. [Google Scholar] [CrossRef] [Green Version]
- Cawley, G.C.; Talbot, N.L. On over-fitting in model selection and subsequent selection bias in performance evaluation. J. Mach. Learn. Res. 2010, 11, 2079–2107. [Google Scholar]
- Yang, M.S.; Nataliani, Y. A feature-reduction fuzzy clustering algorithm based on feature-weighted entropy. IEEE Trans. Fuzzy Syst. 2017, 26, 817–835. [Google Scholar] [CrossRef]
- Chen, R.; Sun, N.; Chen, X.; Yang, M.; Wu, Q. Supervised feature selection with a stratified feature weighting method. IEEE Access 2018, 6, 15087–15098. [Google Scholar] [CrossRef]
- Liu, H.; Motoda, H. Feature Extraction, Construction and Selection: A Data Mining Perspective; Springer Science & Business Media: Berlin/Heidelberg, Germany, 1998; Volume 453. [Google Scholar]
- Singh, R.S.; Saini, B.S.; Sunkaria, R.K. Detection of coronary artery disease by reduced features and extreme learning machine. Clujul Med. 2018, 91, 166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yaghouby, F.; Ayatollahi, A.; Soleimani, R. Classification of cardiac abnormalities using reduced features of heart rate variability signal. In World Applied Sciences Journal; Citeseer: Princeton, NJ, USA, 2009. [Google Scholar]
- Asl, B.M.; Setarehdan, S.K.; Mohebbi, M. Support vector machine-based arrhythmia classification using reduced features of heart rate variability signal. Artif. Intell. Med. 2008, 44, 51–64. [Google Scholar] [CrossRef] [PubMed]
- Sodhro, A.H.; Sangaiah, A.K.; Sodhro, G.H.; Lohano, S.; Pirbhulal, S. An energy-efficient algorithm for wearable electrocardiogram signal processing in ubiquitous healthcare applications. Sensors 2018, 18, 923. [Google Scholar] [CrossRef] [Green Version]
- Muzammal, M.; Talat, R.; Sodhro, A.H.; Pirbhulal, S. A multi-sensor data fusion enabled ensemble approach for medical data from body sensor networks. Inf. Fusion 2020, 53, 155–164. [Google Scholar] [CrossRef]
- Al Rahhal, M.M.; Bazi, Y.; AlHichri, H.; Alajlan, N.; Melgani, F.; Yager, R.R. Deep learning approach for active classification of electrocardiogram signals. Inf. Sci. 2016, 345, 340–354. [Google Scholar] [CrossRef]
- Tama, B.A.; Im, S.; Lee, S. Improving an Intelligent Detection System for Coronary Heart Disease Using a Two-Tier Classifier Ensemble. Biomed Res. Int. 2020, 2020. [Google Scholar] [CrossRef]
- Baccouche, A.; Garcia-Zapirain, B.; Castillo Olea, C.; Elmaghraby, A. Ensemble Deep Learning Models for Heart Disease Classification: A Case Study from Mexico. Information 2020, 11, 207. [Google Scholar] [CrossRef] [Green Version]
- Monteriù, A.; Prist, M.R.; Frontoni, E.; Longhi, S.; Pietroni, F.; Casaccia, S.; Scalise, L.; Cenci, A.; Romeo, L.; Berta, R.; et al. A smart sensing architecture for domestic monitoring: Methodological approach and experimental validation. Sensors 2018, 18, 2310. [Google Scholar] [CrossRef] [Green Version]
- Asuncion, A.; Newman, D. UCI Machine Learning Repository; Irvine University of California: Irvine, CA, USA, 2007. [Google Scholar]
- Kocyigit, Y.; Alkan, A.; Erol, H. Classification of EEG recordings by using fast independent component analysis and artificial neural network. J. Med Syst. 2008, 32, 17–20. [Google Scholar] [CrossRef] [PubMed]
- Mandic, D.P. A generalized normalized gradient descent algorithm. IEEE Signal Process. Lett. 2004, 11, 115–118. [Google Scholar] [CrossRef]
- Krizhevsky, A.; Sutskever, I.; Hinton, G. ImageNet Classification with Deep Convolutional Neural Networks. Neural Inf. Process. Syst. 2012, 25. [Google Scholar] [CrossRef]
- Li, Q.; Cai, W.; Wang, X.; Zhou, Y.; Feng, D.D.F.; Chen, M. Medical image classification with convolutional neural network. In Proceedings of the 2014 13th International Conference on Control Automation Robotics and Vision, ICARCV 2014, Singapore, 10–12 December 2014; pp. 844–848. [Google Scholar] [CrossRef]
- Pereira, S.; Pinto, A.; Alves, V.; Silva, C. Brain Tumor Segmentation Using Convolutional Neural Networks in MRI Images. IEEE Trans. Med. Imaging 2016, 35, 1240–1251. [Google Scholar] [CrossRef] [PubMed]
- Bar, Y.; Diamant, I.; Wolf, L.; Lieberman, S.; Konen, E.; Greenspan, H. Chest pathology detection using deep learning with non-medical training. In Proceedings of the Proceedings—International Symposium on Biomedical Imaging, Brooklyn, NY, USA, 16–19 April 2015; pp. 294–297. [Google Scholar] [CrossRef] [Green Version]
- Luo, L.X. Network text sentiment analysis method combining LDA text representation and GRU-CNN. Pers. Ubiquitous Comput. 2019, 23, 405–412. [Google Scholar] [CrossRef]
- Song, S.; Huang, H.; Ruan, T. Abstractive text summarization using LSTM-CNN based deep learning. Multimed. Tools Appl. 2019, 78, 857–875. [Google Scholar] [CrossRef]
- Banerjee, I.; Ling, Y.; Chen, M.C.; Hasan, S.A.; Langlotz, C.P.; Moradzadeh, N.; Chapman, B.; Amrhein, T.; Mong, D.; Rubin, D.L.; et al. Comparative effectiveness of convolutional neural network (CNN) and recurrent neural network (RNN) architectures for radiology text report classification. Artif. Intell. Med. 2019, 97, 79–88. [Google Scholar] [CrossRef]
- Dai, L.; Sheng, B.; Wu, Q.; Li, H.; Hou, X.; Jia, W.; Fang, R. Retinal microaneurysm detection using clinical report guided multi-sieving CNN. In International Conference on Medical Image Computing and Computer-Assisted Intervention; Springer: New York, NY, USA, 2017; pp. 525–532. [Google Scholar]
- Lim, T.S.; Loh, W.Y.; Shih, Y.S. A comparison of prediction accuracy, complexity, and training time of thirty-three old and new classification algorithms. Mach. Learn. 2000, 40, 203–228. [Google Scholar] [CrossRef]
- Hay, A. The derivation of global estimates from a confusion matrix. Int. J. Remote Sens. 1988, 9, 1395–1398. [Google Scholar] [CrossRef]
- Simonyan, K.; Zisserman, A. Very Deep Convolutional Networks for Large-Scale Image Recognition. arXiv 2014, arXiv:1409.1556. International Conference on Learning Representations, 2015. [Google Scholar]
- Sattar, H.; Bajwa, I.S.; Amin, R.U.; Muhammad, J.; Mushtaq, M.F.; Kazmi, R.; Akram, M.; Ashraf, M.; Shafi, U. Smart wound hydration monitoring using biosensors and fuzzy inference system. Wirel. Commun. Mob. Comput. 2019, 2019. [Google Scholar] [CrossRef] [Green Version]
- Ullah, K.; Shah, M.A.; Zhang, S. Effective ways to use Internet of Things in the field of medical and smart health care. In Proceedings of the 2016 international conference on intelligent systems engineering (ICISE), Islamabad, Pakistan, 15–17 January 2016; pp. 372–379. [Google Scholar]
- Ruman, M.R.; Barua, A.; Rahman, W.; Jahan, K.R.; Roni, M.J.; Rahman, M.F. IoT based emergency health monitoring system. In Proceedings of the 2020 International Conference on Industry 4.0 Technology (I4Tech), Pune, India, 13–15 February 2020; pp. 159–162. [Google Scholar]
- Raj, C.; Jain, C.; Arif, W. HEMAN: Health monitoring and nous: An IoT based e-health care system for remote telemedicine. In Proceedings of the 2017 International Conference on Wireless Communications, Signal Processing and Networking (WiSPNET), Chennai, India, 22–24 March 2017; pp. 2115–2119. [Google Scholar]
- Tripathi, V.; Shakeel, F. Monitoring health care system using internet of things-an immaculate pairing. In Proceedings of the 2017 International Conference on Next Generation Computing and Information Systems (ICNGCIS), Jammu, India, 11–12 December 2017; pp. 153–158. [Google Scholar]
- Bashir, R.N.; Bajwa, I.S.; Shahid, M.M.A. Internet of Things and machine-learning-based leaching requirements estimation for saline soils. IEEE Internet Things J. 2019, 7, 4464–4472. [Google Scholar] [CrossRef]
- Reena, J.K.; Parameswari, R. A smart health care monitor system in IoT based human activities of daily living: A review. In Proceedings of the 2019 International Conference on Machine Learning, Big Data, Cloud and Parallel Computing (COMITCon), Faridabad, India, 14–16 February 2019; pp. 446–448. [Google Scholar]
- Saleem, K.; Bajwa, I.S.; Sarwar, N.; Anwar, W.; Ashraf, A. IoT healthcare: Design of smart and cost-effective sleep quality monitoring system. J. Sensors 2020, 2020. [Google Scholar] [CrossRef]
- Dwivedi, A.K. Performance evaluation of different machine learning techniques for prediction of heart disease. Neural Comput. Appl. 2018, 29, 685–693. [Google Scholar] [CrossRef]
- Vembandasamy, K.; Sasipriya, R.; Deepa, E. Heart diseases detection using Naive Bayes algorithm. Int. J. Innov. Sci. Eng. Technol. 2015, 2, 441–444. [Google Scholar]
| Reference | Method | Data | Findings | Limitation |
|---|---|---|---|---|
| [24] | Neural Network (NN), SVM, Fuzzy Genetic, CART and Random Forest | Database of heart failure patients 136 records from 90 patients, | CART proved as most appropriate in evaluating heart failure severity and its type | Proposed model did not generalize well due to small sample size. Accuracy result is quite low in severity assessment. |
| [28] | Naïve Bayes, KNN, Decision Tree, and Random Forest | Heart disease patient dataset consisting 303 instances obtained from UCI | This paper find the chance of heart disease in patients. | The authors considered 14 attributes out of 76 attributes and results could be improved by applying feature selection. |
| [29] | Naïve Bayes, KNN, Decision Tree, Random Forest and HRFLM model(combination of Random Forest and Linear Method) | Four datasets (Cleveland, Hungary, Switzerland, and the VA Long Beach) | The proposed hybrid model predicted heart disease better than machine learning models | More combination of models along with feature selection need to be explored. |
| [31] | Multiple Kernel Learning and Adaptive Neuro Fuzzy Inference System | KEGG Metabolic Reaction Network dataset. | Experiments have been applied in a two-fold approach in classifying patients into heart disease and healthy ones. | Very small number of features or parameters are considered in the experiment |
| [37] | Fisher ranking method, generalized discriminant analysis(GDA) | NSR-CAD and SR-CAD | Authors proposed noninvasive approach using (GDA) to automatically detect coronary artery disease using heart rate variability signals | Models need to train on a large dataset of heart rate variability signals for generalizability. |
| [43] | Ensemble of Random Forest, Gradient Boosting Machine, and XG Boost. | Z-Alizadeh Sani, Statlog, Cleveland, and Hungarian datasets | Used an ensemble model to detect coronary artery disease. | A stacked ensemble of three models also increased the complexity and the cost of the model. |
| [44] | Ensemble of BiLSTM, BiGRU and CNN | heart disease dataset | Ensemble learning framework using deep model was applied to deal with the problem of an imbalanced heart disease dataset. | The proposed approach has not tested on a benchmark dataset. |
| Sr No. | Attributes | Description | Range | Measured In |
|---|---|---|---|---|
| 1 | Time | Followup period | 4–285 | Days |
| 2 | Event (target) | If the patient died in the followup time | 0,1 | Boolean |
| 3 | Gender | Man or woman | 0,1 | Binary |
| 4 | Smoking | If the patient smokes | 0,1 | Boolean |
| 5 | Diabetics | If the patient has diabetics | 0,1 | Boolean |
| 6 | B.P | If the patient has blood pressure issue | 0,1 | Boolean |
| 7 | Anaemia | Decrease in red blood cell or haemoglobin | 0,1 | Boolean |
| 8 | Age | Age of the patient | 40–95 | Years |
| 9 | Ejection fraction | Percentage of blood leaving the heart at each concentration | 14–80 | Percentage |
| 10 | Sodium | Level of sodium in the blood | 114–148 | mEq/L |
| 11 | Creatinine | Level of creatinine in the blood | 0.50–9.40 | mg/dL |
| 12 | Platelets | Platelets in blood | 25.01–850.00 | kiloplatelets/mL |
| 13 | CPK (creatinine Phospho) | Level of CPK enzyme in the blood | 23-7861 | Mcg/L |
| Parameter | Value |
|---|---|
| Embedding dimension | 300 |
| Batch size | 256 |
| Pooling | 2 × 2 |
| No. of filters | 5 × 64 |
| Max_Sequence_length | 130 |
| Epochs | 25 |
| Optimizer | Adam |
| Function | Binary cross entropy |
| 1 | Accuracy = |
| 2 | Precision = |
| 3 | Recall = |
| 4 | F-Score = |
| Model | Accuracy | Precision | Recall | F-Score |
|---|---|---|---|---|
| CNN | 0.9289 | 0.94 | 0.94 | 0.94 |
| MLP | 0.9201 | 0.93 | 0.92 | 0.93 |
| RNN | 0.9001 | 0.88 | 0.90 | 0.89 |
| LSTM | 0.9169 | 0.92 | 0.92 | 0.92 |
| RF without SMOTE [16] | 0.8889 | 0.89 | 0.89 | 0.89 |
| ETC with SMOTE [16] | 0.9262 | 0.93 | 0.93 | 0.93 |
| Models | Accuracy |
|---|---|
| DT [16] | 0.8778 |
| AdaBoost [16] | 0.8852 |
| LR [16] | 0.8442 |
| SGD [16] | 0.5491 |
| RF [16] | 0.9188 |
| GBM [16] | 0.8852 |
| ETC [16] | 0.9262 |
| GNB [16] | 0.7540 |
| SVM [16] | 0.7622 |
| RNN | 0.9001 |
| LSTM | 0.9169 |
| MLP | 0.9201 |
| CNN | 0.9289 |
| Model | Accuracy | Precision | Recall | F-Score |
|---|---|---|---|---|
| VGG16 | 0.9129 | 0.90 | 0.92 | 0.91 |
| AlexNet | 0.9071 | 0.90 | 0.90 | 0.90 |
| CNN | 0.9289 | 0.94 | 0.94 | 0.94 |
| Model | Training Time |
|---|---|
| Proposed approach | 35 min |
| VGG16 | 39 min |
| AlexNet | 47 min |
| Fold Number | Accuracy | Precision | Recall | F-Score |
|---|---|---|---|---|
| 1st-Fold | 0.915 | 0.916 | 0.921 | 0.911 |
| 2nd-Fold | 0.912 | 0.907 | 0.922 | 0.926 |
| 3rd-Fold | 0.911 | 0.923 | 0.923 | 0.934 |
| 4th-Fold | 0.918 | 0.907 | 0.949 | 0.935 |
| 5th-Fold | 0.904 | 0.911 | 0.948 | 0.933 |
| 6th-Fold | 0.916 | 0.926 | 0.947 | 0.932 |
| 7th-Fold | 0.924 | 0.907 | 0.916 | 0.941 |
| 8th-Fold | 0.914 | 0.914 | 0.945 | 0.937 |
| 9th-Fold | 0.902 | 0.924 | 0.954 | 0.918 |
| 10th-Fold | 0.947 | 0.945 | 0.957 | 0.949 |
| Average | 0.9263 | 0.9281 | 0.9399 | 0.9340 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Umer, M.; Sadiq, S.; Karamti, H.; Karamti, W.; Majeed, R.; NAPPI, M. IoT Based Smart Monitoring of Patients’ with Acute Heart Failure. Sensors 2022, 22, 2431. https://doi.org/10.3390/s22072431
Umer M, Sadiq S, Karamti H, Karamti W, Majeed R, NAPPI M. IoT Based Smart Monitoring of Patients’ with Acute Heart Failure. Sensors. 2022; 22(7):2431. https://doi.org/10.3390/s22072431
Chicago/Turabian StyleUmer, Muhammad, Saima Sadiq, Hanen Karamti, Walid Karamti, Rizwan Majeed, and Michele NAPPI. 2022. "IoT Based Smart Monitoring of Patients’ with Acute Heart Failure" Sensors 22, no. 7: 2431. https://doi.org/10.3390/s22072431
APA StyleUmer, M., Sadiq, S., Karamti, H., Karamti, W., Majeed, R., & NAPPI, M. (2022). IoT Based Smart Monitoring of Patients’ with Acute Heart Failure. Sensors, 22(7), 2431. https://doi.org/10.3390/s22072431







