Application of a Platform for Gluten-Free Diet Evaluation and Dietary Advice: From Theory to Practice
Abstract
:1. Introduction
2. Materials and Methods
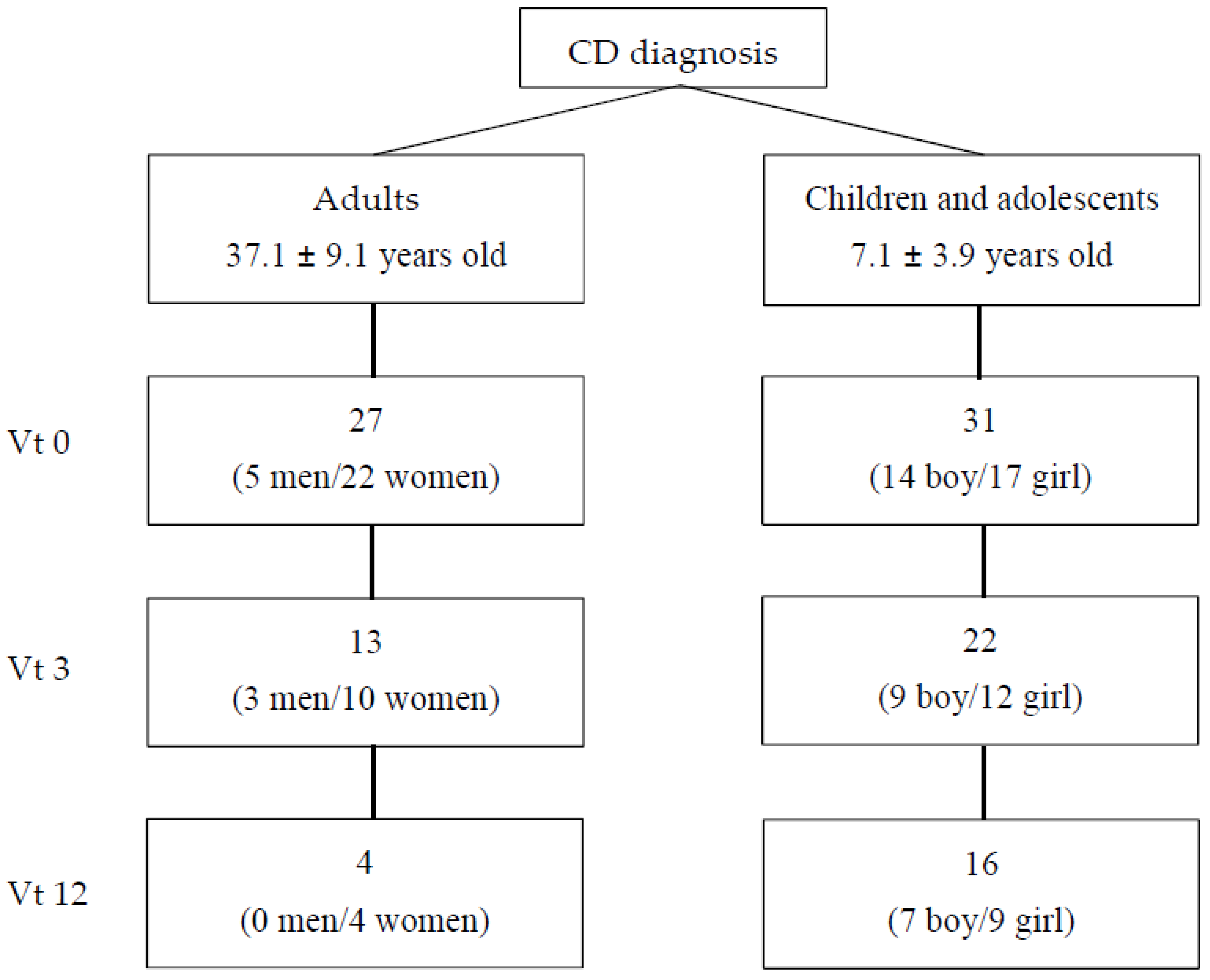
2.1. Participants and Procedure
2.2. Anthropometric Measurements
2.3. Body Composition and Energy Expenditure
2.4. Biochemical Data
2.5. Analysis of Symptoms Presence
2.6. Dietary Assessment and Counselling
2.7. Determination of FODMAP Daily Intake
2.8. Statistical Analysis
3. Results
3.1. Anthropometric and Biochemical Parameters over 1 Year of GFD in Adults and Children with CD
3.2. Energy, Macronutrient, Fibre and Cholesterol Intake of Adults, Children and Adolescents with CD
3.3. Food Frequency Consumption
3.4. Food Consumption According to NOVA System
3.5. GFP Contribution to the Total Energy Intake
3.6. FODMAP Consumption of Celiac Adults and Children
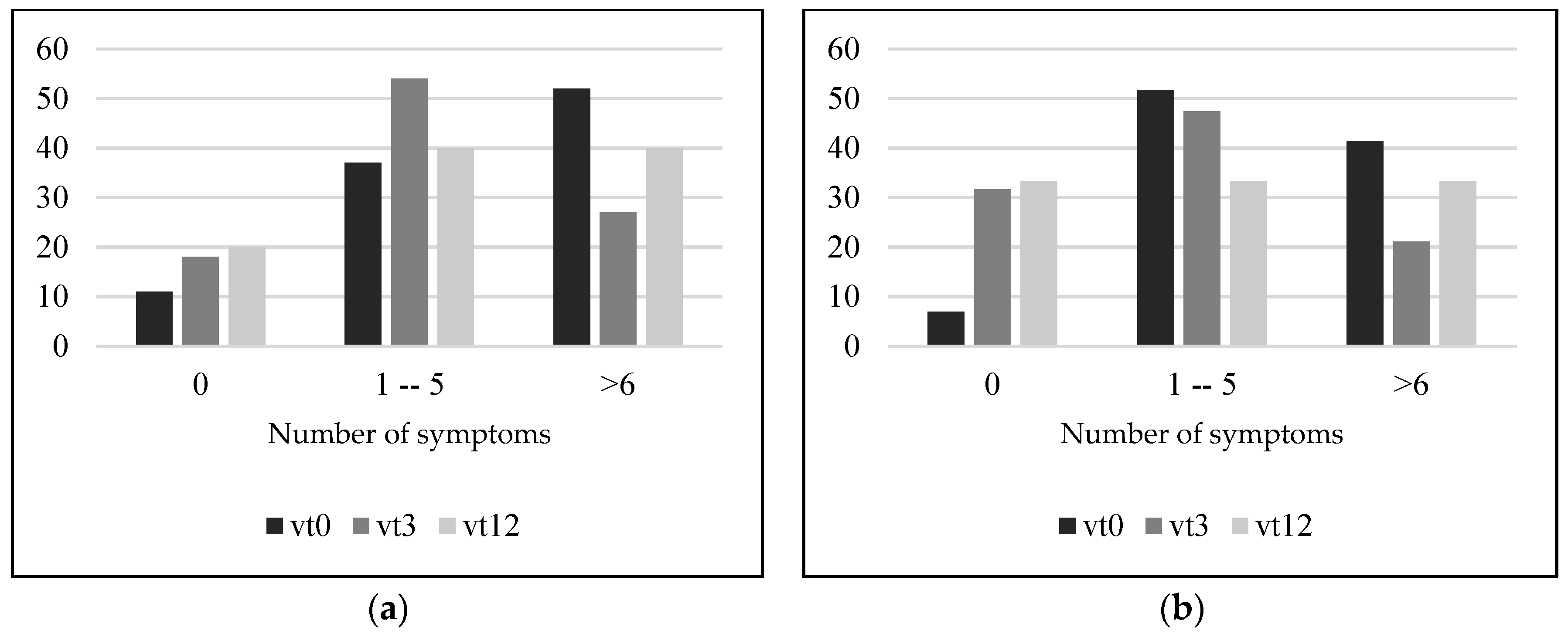
3.7. Presence of Gastrointestinal Symptom
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Hussain, M.; Al-Haiqi, A.; Zaidan, A.A.; Zaidan, B.B.; Kiah, M.L.M.; Anuar, N.B.; Abdulnabi, M. The landscape of research on smartphone medical apps: Coherent taxonomy, motivations, open challenges and recommendations. Comput. Meth. Programs Biomed. 2015, 122, 393–408. [Google Scholar] [CrossRef]
- Hutchesson, M.J.; Gough, C.; Müller, A.M.; Short, C.E.; Whatnall, M.C.; Ahmed, M.; Vandelanotte, C. eHealth interventions targeting nutrition, physical activity, sedentary behavior, or obesity in adults: A scoping review of systematic reviews. Obes. Rev. 2021, 22, e13295. [Google Scholar] [CrossRef] [PubMed]
- Robert, C.; Erdt, M.; Lee, J.; Cao, Y.; Naharudin, N.B.; Theng, Y.L. Effectiveness of eHealth nutritional interventions for middle-aged and older adults: Systematic review and meta-analysis. J. Med. Internet. Res. 2021, 23, e15649. [Google Scholar] [CrossRef] [PubMed]
- Churruca, I.; Miranda, J.; Lasa, A.; Bustamante, M.; Larretxi, I.; Simon, E. Analysis of body composition and food habits of spanish celiac women. Nutrients 2015, 7, 5515–5531. [Google Scholar] [CrossRef] [Green Version]
- Bardella, M.T.; Fredella, C.; Prampolini, L.; Molteni, N.; Giunta, A.M.; Bianchi, P.A. Body composition and dietary intakes in adult celiac disease patients consuming a strict gluten-free diet. Am. J. Clin. Nutr. 2000, 72, 937–939. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vici, G.; Belli, L.; Biondi, M.; Polzonetti, V. Gluten free diet and nutrient deficiencies: A review. Clin. Nutr. 2016, 35, 1236–1241. [Google Scholar] [CrossRef]
- Martin, J.; Geisel, T.; Maresch, C.; Krieger, K.; Stein, J. Inadequate nutrient intake in patients with celiac disease: Results from a German dietary survey. Digestion 2013, 87, 240–246. [Google Scholar] [CrossRef]
- Theethira, T.G.; Dennis, M. Celiac disease and the gluten-free diet: Consequences and recommendations for improvement. Dig. Diss. 2015, 33, 175–182. [Google Scholar] [CrossRef]
- Suárez-González, M.; Bousoño García, C.; Jiménez Treviño, S.; Iglesias Cabo, T.; Díaz Martín, J.J. Influence of nutrition education in paediatric coeliac disease: Impact of the role of the registered dietitian: A prospective, single-arm intervention study. J. Hum. Nutr. Diet. 2020, 33, 775–785. [Google Scholar] [CrossRef]
- Phillips, C.M.; Kesse-Guyot, E.; McManus, R.; Hercberg, S.; Lairon, D.; Planells, R.; Roche, H.M. High dietary saturated fat intake accentuates obesity risk associated with the fat mass and obesity—Associated gene in adults. J. Nutr. 2012, 142, 824–831. [Google Scholar] [CrossRef]
- Martín-Masot, R.; Nestares, M.T.; Diaz-Castro, J.; López-Aliaga, I.; Alférez, M.J.M.; Moreno-Fernandez, J.; Maldonado, J. Multifactorial etiology of anemia in celiac disease and effect of gluten-free diet: A comprehensive review. Nutrients 2019, 11, 2557. [Google Scholar] [CrossRef] [Green Version]
- Slavin, J.L. Dietary fiber and body weight. Nutrition 2005, 21, 411–418. [Google Scholar] [CrossRef]
- Ganji, R.; Moghbeli, M.; Sadeghi, R.; Bayat, G.; Ganji, A. Prevalence of osteoporosis and osteopenia in men and premenopausal women with celiac disease: A systematic review. Nutr. J. 2019, 18, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Capristo, E.; Malandrino, N.; Farnetti, S.; Mingrone, G.; Leggio, L.; Addolorato, G.; Gasbarrini, G. Increased serum high-density lipoprotein-cholesterol concentration in celiac disease after gluten-free diet treatment correlates with body fat stores. J. Clin. Gastroenterol. 2009, 43, 946–949. [Google Scholar] [CrossRef] [Green Version]
- Capristo, E.; Addolorato, G.; Mingrone, G.; De Gaetano, A.; Greco, A.V.; Tataranni, P.A.; Gasbarrini, G. Changes in body composition, substrate oxidation, and resting metabolic rate in adult celiac disease patients after a 1-y gluten-free diet treatment. Am. J. Clin. Nutr. 2000, 72, 76–81. [Google Scholar] [CrossRef]
- Capristo, E.; Mingrone, G.; Addolorato, G.; Greco, A.V.; Corazza, G.R.; Gasbarrini, G. Differences in metabolic variables between adult coeliac patients at diagnosis and patients on a gluten-free diet. Scand. J. Gastroenterol. 1997, 32, 1222–1229. [Google Scholar] [CrossRef]
- Corazza, G.R.; Di Sario, A.; Sacco, G.; Zoli, G.; Treggiari, E.A.; Brusco, G.; Gasbarrini, G. Subclinical coeliac disease: An anthropometric assessment. J. Intern. Med. 1994, 236, 183–187. [Google Scholar] [CrossRef] [PubMed]
- Pietzak, M.M. Follow-up of patients with celiac disease: Achieving compliance with treatment. Gastroenterology 2005, 128 (Suppl. S1), S135–S141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miranda, J.; Lasa, A.; Bustamante, M.A.; Churruca, I.; Simon, E. Nutritional differences between a gluten-free diet and a diet containing equivalent products with gluten. Plant Food Hum. Nutr. 2014, 69, 182–187. [Google Scholar] [CrossRef] [PubMed]
- Larretxi, I.; Txurruka, I.; Navarro, V.; Lasa, A.; Bustamante, M.; Fernández-Gil, M.D.P.; Simón, E.; Miranda, J. Micronutrient analysis of gluten-free products: Their low content is not involved in gluten-free diet imbalance in a cohort of celiac children and adolescent. Foods 2019, 8, 321. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lasa, A.; Larretxi, I.; Simón, E.; Churruca, I.; Navarro, V.; Martínez, O.; Bustamante, M.; Miranda, J. New software for gluten-free diet evaluation and nutritional education. Nutrients 2019, 11, 2505. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Larretxi, I.; Simon, E.; Benjumea, L.; Miranda, J.; Bustamante, M.A.; Lasa, A.; Eizaguirre, F.J.; Churruca, I. Gluten-free-rendered products contribute to imbalanced diets in children and adolescents with celiac disease. Eur. J. Nutr. 2019, 58, 775–783. [Google Scholar] [CrossRef]
- Zuccotti, G.; Fabiano, V.; Dilillo, D.; Picca, M.; Cravidi, C.; Brambilla, P. Intakes of nutrients in Italian children with celiac disease and the role of commercially available gluten-free products. J. Hum. Nutr. Diet. 2013, 26, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Cardo, A.; Churruca, I.; Lasa, A.; Navarro, V.; Vázquez-Polo, M.; Perez-Junkera, G.; Larretxi, I. Nutritional imbalances in adult celiac patients following a gluten-free diet. Nutrients 2021, 13, 2877. [Google Scholar] [CrossRef] [PubMed]
- Lanzini, A.; Lanzarotto, F.; Villanacci, V.; Mora, A.; Bertolazzi, S.; Turini, D.; Carella, G.; Malagoli, A.; Ferrante, G.; Cesana, B.M.; et al. Complete recovery of intestinal mucosa occurs very rarely in adult coeliac patients despite adherence to gluten-free diet. Aliment. Pharmacol. Ther. 2009, 29, 1299–1308. [Google Scholar] [CrossRef] [PubMed]
- Lebwohl, B.; Sanders, D.S.; Green, P.H.R. Coeliac disease. Lancet 2018, 391, 70–81. [Google Scholar] [CrossRef]
- Sharkey, L.M.; Corbett, G.; Currie, E.; Lee, J.; Sweeney, N.; Woodward, J.M. Optimising delivery of care in coeliac disease—Comparison of the benefits of repeat biopsy and serological follow-up. Aliment. Pharmacol. Ther. 2013, 38, 1278–1291. [Google Scholar] [CrossRef]
- Clifford, S.; Taylor, A.J.; Gerber, M.; Devine, J.; Cho, M.; Walker, R.; Stefani, I.; Fidel, S.; Drahos, J.; Leffler, D.A. Concepts and instruments for patient-reported outcome assessment in celiac disease: Literature review and experts’ perspectives. Value Health 2020, 23, 104–113. [Google Scholar] [CrossRef]
- Penny, A.H.; Baggus, M.R.E.; Rej, A.; Snowden, A.J.; Sanders, S.D. Non-responsive coeliac disease: A comprehensive review from the NHS England National Centre for refractory coeliac disease. Nutrients 2020, 12, 216. [Google Scholar] [CrossRef] [Green Version]
- Monzani, A.; Lionetti, E.; Felici, E.; Fransos, L.; Azzolina, D.; Rabbone, I.; Catassi, C. Adherence to the gluten-free diet during the lockdown for COVID-19 pandemic: A web-based survey of italian subjects with celiac disease. Nutrients 2020, 12, 3467. [Google Scholar] [CrossRef]
- Nachman, F.; Mauriño, E.; Vázquez, H.; Sfoggia, C.; Gonzalez, A.; Gonzalez, V.; del Campo, M.P.; Smecuol, E.; Niveloni, S.; Sugai, E.; et al. Quality of life in celiac disease patients: Prospective analysis on the importance of clinical severity at diagnosis and the impact of treatment. Dig. Liv. Dise. 2009, 41, 15–25. [Google Scholar] [CrossRef]
- Häuser, W.; Janke, K.-H.; Klump, B.; Gregor, M.; Hinz, A. Anxiety and depression in adult patients with celiac disease on a gluten-free diet. World J. Gastroenterol. 2010, 16, 2780–2787. [Google Scholar] [CrossRef]
- Barratt, S.M.; Leeds, J.S.; Sanders, D.S. Quality of life in coeliac disease is determined by perceived degree of difficulty adhering to a gluten-free diet, not the level of dietary adherence ultimately achieved. J. Gastrointest. Liver Dis. 2011, 20, 241–245. [Google Scholar]
- Czaja-Bulsa, G.; Bulsa, M. Adherence to gluten-free diet in children with celiac disease. Nutrients 2018, 10, 1424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodrigues, M.; Yonamine, G.H.; Satiro, C.A.F. Correction to: Rate and determinants of non-adherence to a gluten-free diet and nutritional status assessment in children and adolescents with celiac disease in a tertiary Brazilian referral center: A cross-sectional and retrospective study. BMC Gastroenterol. 2018, 18, 36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Newnham, E.D.; Shepherd, S.J.; Strauss, B.J.; Hosking, P.; Gibson, P.R. Adherence to the gluten-free diet can achieve the therapeutic goals in almost all patients with coeliac disease: A 5-year longitudinal study from diagnosis. J. Gastroenterol. Hepatol. 2016, 31, 342–349. [Google Scholar] [CrossRef] [PubMed]
- Mcintosh, K.; Reed, D.E.; Schneider, T.; Dang, F.; Keshteli, A.H.; De Palma, G.; Madsen, K.; Bercik, P.; Vanner, S. FODMAPs alter symptoms and the metabolome of patients with IBS: A randomised controlled trial. Gut 2017, 66, 1241–1251. [Google Scholar] [CrossRef]
- Chumpitazi, B.P.; Cope, J.L.; Hollister, E.B.; Tsai, C.M.; McMeans, A.R.; Luna, R.A.; Versalovic, J.; Shulman, R.J. Randomised clinical trial: Gut microbiome biomarkers are associated with clinical response to a low FODMAP diet in children with the irritable bowel syndrome. Aliment. Pharmacol. Ther. 2015, 42, 418–427. [Google Scholar] [CrossRef]
- Eswaran, S.L.; Chey, W.D.; Han-Markey, T.; Ball, S.; Jackson, K. A randomized controlled trial comparing the low FODMAP diet vs. modified nice guidelines in US adults with IBS-D. Am. J. Gastroenterol. 2016, 111, 1824–1832. [Google Scholar] [CrossRef]
- Staudacher, H.M.; Lomer, M.C.E.; Farquharson, F.M.; Louis, P.; Fava, F.; Franciosi, E.; Scholz, M.; Tuohy, K.M.; Lindsay, J.O.; Irving, P.M.; et al. A diet low in FODMAPs reduces symptoms in patients with irritable bowel syndrome and a probiotic restores bifidobacterium species: A randomized controlled trial. Gastroenterology 2017, 153, 936–947. [Google Scholar] [CrossRef] [Green Version]
- Sobradillo, B.; Aguirre, A.; Aresti, U.; Bilbao, A.; Fernández-Ramos, C.; Lizárraga, A.; Lorenzo, H.; Madariaga, L.; Rica, I.; Ruiz, I.; et al. Curvas y Tablas de Crecimiento. Estudios Longitudinal y Transversal; Fundación Faustino Orbegozo: Bilbao, Spain, 2004; pp. 1–31. [Google Scholar]
- Gallagher, D.; Heymsfield, S.B.; Heo, M.; Jebb, S.A.; Murgatroyd, P.R.; Sakamoto, Y. Healthy percentage body fat ranges: An approach for developing guidelines based on body mass index. Am. J. Clin. Nutr. 2000, 72, 694–701. [Google Scholar] [CrossRef]
- Lean, M.E.; Han, T.S.; Morrison, C.E. Waist circumference as a measure for indicating need for weight management. BMJ (Clin. Res. Ed.) 1995, 311, 158–161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moreno Aznar, L.; Zaragozano, J.F.; De Frenne, L.M.; Solana, C.F.; Martínez, G.R.; Chueca, A.S.; Sánchez, M.B. Distribución de la Grasa en Niños y Adolescentes de Ambos Sexos; Anales Españoles de Pediatría: Barcelona, Spain, 1998; Volume 49, pp. 135–139. [Google Scholar]
- Moreno Aznar, L.; Zaragozano, J.F.; Martínez, G.R.; Chueca, A.S.; Sánchez, M.B. Masa Grasa Corporal en Niños y Adolescentes de Sexo Masculino; Anales Españoles de Pediatría: Barcelona, Spain, 1999; Volume 51, pp. 629–632. [Google Scholar]
- Heymsfield, S.B.; Smith, R.; Aulet, M.; Bensen, B.; Lichtman, S.; Wang, J.; Pierson, R.N., Jr. Appendicular skeletal muscle mass: Measurement by dual-photon absorptiometry. Am. J. Clin. Nutri. 1990, 52, 214–218. [Google Scholar] [CrossRef] [PubMed]
- Ito, H.; Ohshima, A.; Ohto, N.; Ogasawara, M.; Tsuzuki, M.; Takao, K.; Hijii, C.; Tanaka, H.; Nishioka, K. Relation between body composition and age in healthy Japanese subjects. Eur. J. Clin. Nutr. 2001, 55, 462–470. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Health Organization. Waist Circumference and Waist-Hip Ratio: Report of a WHO Expert Consultation, Geneva, 8–11 December 2008; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Laurikka, P.; Salmi, T.; Collin, P.; Huhtala, H.; Mäki, M.; Kaukinen, K.; Kurppa, K. Gastrointestinal symptoms in celiac disease patients on a long-term gluten-free diet. Nutrients 2016, 8, 429. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hopman, E.G.D.; Koopman, H.M.; Wit, J.M.; Mearin, M.L. Dietary compliance and health-related quality of life in patients with coeliac disease. Eur. J. Gastroenterol. Hepatol. 2009, 21, 1056–1061. [Google Scholar] [CrossRef]
- Simrén, M.; Axelsson, J.; Gillberg, R.; Abrahamsson, H.; Svedlund, J.; Björnsson, E.S. Quality of life in inflammatory bowel disease in remission: The impact of IBS-like symptoms and associated psychological factors. Am. J. Gastroenterol. 2002, 97, 389–396. [Google Scholar] [CrossRef]
- Svedlund, J.; Sjödin, I.; Dotevall, G. GSRS—A clinical rating scale for gastrointestinal symptoms in patients with irritable bowel syndrome and peptic ulcer disease. Dig. Dis. Sci. 1988, 33, 129–134. [Google Scholar] [CrossRef]
- Olafsson, S.; Hatlebakk, J.G.; Berstad, A. Patients with endoscopic gastritis and/or duodenitis improve markedly following eradication of helicobacter pylori, although less so than patients with ulcers. Scand. J. Gastroenterol. 2002, 37, 1386–1394. [Google Scholar] [CrossRef]
- Hori, K.; Matsumoto, T.; Miwa, H. Analysis of the gastrointestinal symptoms of uninvestigated dyspepsia and irritable bowel syndrome. Gut Liver 2009, 3, 192–196. [Google Scholar] [CrossRef] [Green Version]
- Dimenäs, E.; Carlsson, G.; Glise, H.; Israelsson, B.; Wiklund, I. Relevance of norm values as part of the documentation of quality of life instruments for use in upper gastrointestinal disease. Scand. J. Gastroenterol. 1996, 31 (Suppl. S221), 8–13. [Google Scholar] [CrossRef] [PubMed]
- Russolillo, G.; Marques, I. Food Portion Sizes Album; Imagen Comunicación Multimedia: Madrid, Spain, 2008; p. 186. [Google Scholar]
- FESNAD. Dietary reference intakes (DRI) for spanish population. Act. Dietética 2010, 14, 196–197. [Google Scholar]
- SENC. Objetivos nutricionales para la población española. Consenso de la Sociedad Española de nutrición. Rev. Esp. Nutri. Comunitaria 2011, 17, 178–199. [Google Scholar]
- Lawrence, M.A.; Baker, P.I. Ultra-processed food and adverse health outcomes. Br. Med. J. 2019, 365, 12289. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, C.A.; Cannon, G.; Lawrence, M.; Costa Louzada, M.L.; Pereira Machado, P. Ultra-Processed Foods, Diet Quality, and Health Using the NOVA Classification System; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2019. [Google Scholar]
- Food Standards Australia New Zealand. Australian Food Composition Database—Release 1. Canberra: FSANZ. Available online: www.foodstandards.gov.au (accessed on 14 July 2020).
- Muir, J.G.; Shepherd, S.J.; Rosella, O.; Rose, R.; Barrett, J.S.; Gibson, P.R. Fructan and free fructose content of common Austral-ian vegetables and fruit. J. Agri. Food. Chem. 2007, 55, 6619–6627. [Google Scholar] [CrossRef] [PubMed]
- Biesiekierski, J.R.; Rosella, O.; Rose, R.; Liels, K.; Barrett, J.S.; Shepherd, S.J.; Gibson, P.R.; Muir, J.G. Quantification of fructans, galacto-oligosacharides and other short-chain carbohydrates in processed grains and cereals. J. Hum. Nutr. Diet. 2011, 24, 154–176. [Google Scholar] [CrossRef]
- Miranda, J.; Vázquez-Polo, M.; Perez-Junkera, G.; Fernández-Gil, M.D.P.; Bustamante, M.Á.; Navarro, V.; Simón, E.; Martínez, O. FODMAP intake in Spanish population: Open approach for risk assessment. Int. J. Environ. Res. Public Health 2020, 17, 5882. [Google Scholar] [CrossRef]
- Clark, R.; Johnson, R. Malabsorption syndromes. Nurs. Clin. N. Am. 2018, 53, 361–374. [Google Scholar] [CrossRef]
- Nolan, J.D.; Johnston, I.M.; Walters, J.R.F. Physiology of malabsorption. Surgery 2015, 33, 193–199. [Google Scholar]
- van der Heide, F. Acquired causes of intestinal malabsorption. Best Pract. Res. Clin. Gastroenterol. 2016, 30, 213–224. [Google Scholar] [CrossRef]
- González, T.; Larretxi, I.; Vitoria, J.C.; Castaño, L.; Simón, E.; Churruca, I.; Navarro, V.; Lasa, A. Celiac male’s gluten-free diet profile: Comparison to that of the control population and celiac women. Nutrients 2018, 10, 1713. [Google Scholar] [CrossRef] [Green Version]
- Mariani, P.; Viti, M.G.; Montuori, M.; La Vecchia, A.; Cipolletta, E.; Calvani, L.; Bonamico, M. The gluten-free diet: A nutritional risk factor for adolescents with celiac disease? J. Pediatr. Gastroenterol. Nutr. 1998, 27, 519–523. [Google Scholar] [CrossRef] [PubMed]
- Forchielli, M.L.; Fernicola, P.; Diani, L.; Scrivo, B.; Salfi, N.C.; Pessina, A.C.; Lima, M.; Conti, V.; Pession, A. Gluten-free diet and lipid profile in children with celiac disease: Comparison with general population standards. J. Ped. Gastroenterol. Nutr. 2015, 61, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Dieterich, W.; Zopf, Y. Gluten and FODMAPS-sense of a restriction/When is restriction necessary? Nutrients 2019, 11, 1957. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fry, L.; Madden, A.M.; Fallaize, R. An investigation into the nutritional composition and cost of gluten-free versus regular food products in the UK. J. Hum. Nutr. Diets. 2018, 31, 108–120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Larretxi, I.; Churruca, I.; Navarro, V.; Miranda, J.; Lasa, A.; Bustamante, M.Á.; Simon, E. Effect of analytically measured fiber and resistant starch from gluten-free products on the diets of individuals with celiac disease. Nutrition 2020, 70, 110586. [Google Scholar] [CrossRef]
- Allen, B.; Orfila, C. The availability and nutritional adequacy of gluten-free bread and pasta. Nutrients 2018, 10, 1370. [Google Scholar] [CrossRef] [Green Version]
- Cornicelli, M.; Saba, M.; Machello, N.; Silano, M.; Neuhold, S. Nutritional composition of gluten-free food versus regular food sold in the Italian market. Dig. Liver Dis. 2018, 50, 1305–1308. [Google Scholar] [CrossRef] [PubMed]
- Lionetti, E.; Antonucci, N.; Marinelli, M.; Bartolomei, B.; Franceschini, E.; Gatti, S.; Catassi, G.N.; Verma, A.K.; Monachesi, C.; Catassi, C. Nutritional status, dietary intake, and adherence to the mediterranean diet of children with celiac disease on a gluten-free diet: A case-control prospective study. Nutrients 2020, 12, 143. [Google Scholar] [CrossRef] [Green Version]
- Schnabel, L.; Kesse-Guyot, E.; Allès, B.; Touvier, M.; Srour, B.; Hercberg, S.; Buscail, C.; Julia, C. Association between ultraprocessed food consumption and risk of mortality among middle-aged adults in France. JAMA Intern. Med. 2019, 179, 490–498. [Google Scholar] [CrossRef]
- Griffin, J.; Albaloul, A.; Kopytek, A.; Elliott, P.; Frost, G. Effect of ultraprocessed food intake on cardiometabolic risk is mediated by diet quality: A cross-sectional study. BMJ Nutr. Prev. Health 2021, 4, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Santos, F.S.D.; Dias, M.D.S.; Mintem, G.C.; Oliveira, I.O.; Gigante, D.P. Food processing and cardiometabolic risk factors: A systematic review. Rev. Saude Publica 2020, 54, 70. [Google Scholar] [CrossRef] [PubMed]
- Andrade, C. Ultraprocessed food and cardiovascular risk: Estimating the number needed to harm in an unfamiliar situation. Indian J. Psychol. Med. 2019, 41, 501–502. [Google Scholar] [CrossRef] [PubMed]
- Anania, C.; Pacifico, L.; Olivero, F.; Perla, F.M.; Chiesa, C. Cardiometabolic risk factors in children with celiac disease on a gluten-free diet. World J. Clin. Pediatr. 2017, 6, 143–148. [Google Scholar] [CrossRef]
- Norsa, L.; Shamir, R.; Zevit, N.; Verduci, E.; Hartman, C.; Ghisleni, D.; Riva, E.; Giovannini, M. Cardiovascular disease risk factor profiles in children with celiac disease on gluten-free diets. World J. Gastroenterol. 2013, 19, 5658–5664. [Google Scholar] [CrossRef]
- Caio, G.; Volta, U.; Sapone, A.; Leffler, D.A.; De Giorgio, R.; Catassi, C.; Fasano, A. Celiac disease: A comprehensive current review. BMC Med. 2019, 17, 142. [Google Scholar] [CrossRef] [Green Version]
- Rajpoot, P.; Sharma, A.; Harikrishnan, S.; Baruah, B.J.; Ahuja, V.; Makharia, G.K. Adherence to gluten-free diet and barriers to adherence in patients with celiac disease. Indian J. Gastroenterol. 2015, 34, 380–386. [Google Scholar] [CrossRef]
- Pekki, H.; Kurppa, K.; Mäki, M.; Huhtala, H.; Sievänen, H.; Laurila, K.; Collin, P.; Kaukinen, K. Predictors and significance of incomplete mucosal recovery in celiac disease after 1 year on a gluten-free diet. Am. J. Gastroenterol. 2015, 110, 1078–1085. [Google Scholar] [CrossRef]
- Mooney, P.; Evans, K.; Singh, S.; Sanders, D. Treatment failure in coeliac disease: A practical guide to investigation and treatment of non-responsive and refractory coeliac disease. J. Gastrointest. Liver Dis. 2012, 21, 197–203. [Google Scholar]
- Stasi, E.; Marafini, I.; Caruso, R.; Soderino, F.; Angelucci, E.; Del Vecchio Blanco, G.; Paoluzi, O.A.; Calabrese, E.; Sedda, S.; Zorzi, F.; et al. Frequency and cause of persistent symptoms in celiac disease patients on a long-term gluten-free diet. J. Clin. Gastroenterol. 2016, 50, 239–243. [Google Scholar] [CrossRef]
- Ciacci, C.; Cirillo, M.; Cavallaro, R.; Mazzacca, G. Long-term follow-up of celiac adults on gluten-free diet: Prevalence and correlates of intestinal damage. Digestion 2002, 66, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Moya, D.A.; Nugent, C.A.; Baker, R.D.; Baker, S.S. Celiac disease nutritional status and poor adherence to follow-up. Clin. Pediatr. 2020, 59, 649–655. [Google Scholar] [CrossRef] [PubMed]
- Murray, J.A.; Watson, T.; Clearman, B.; Mitros, F. Effect of a gluten-free diet on gastrointestinal symptoms in celiac disease. Am. J. Clin. Nutr. 2004, 79, 669–673. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Norström, F.; Sandström, O.; Lindholm, L.; Ivarsson, A. A gluten-free diet effectively reduces symptoms and health care consumption in a Swedish celiac disease population. BMC Gastroenterol. 2012, 12, 125. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Silvester, J.A.; Comino, I.; Rigaux, L.N.; Segura, V.; Green, K.H.; Cebolla, A.; Weiten, D.; Dominguez, R.; Leffler, D.A.; Leon, F.; et al. Exposure sources, amounts and time course of gluten ingestion and excretion in patients with coeliac disease on a gluten-free diet. Aliment. Pharmacol. Ther. 2020, 52, 1469–1479. [Google Scholar] [CrossRef] [PubMed]
- Weynants, A.; Goossens, L.; Genetello, M.; De Looze, D.; Van Winckel, M. The long-term effect and adherence of a low fermentable oligosaccharides disaccharides monosaccharides and polyols (FODMAP) diet in patients with irritable bowel syndrome. J. Hum. Nutr. Diet. 2020, 33, 159–169. [Google Scholar] [CrossRef] [Green Version]
| Adults | Children and Adolescents | Adults vs. Children | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Recommended Intake * | vt0 | vt3 | vt12 | p Value | vt0 | vt3 | vt12 | p Value | p Value | |||||
| N | 27 | 13 | 2 | vt0 vs. vt3 | vt0 vs. vt12 | 22 | 10 | 4 | vt0 vs. vt3 | vt0 vs. vt12 | Vt0 | Vt3 | Vt12 | |
| Energy intake (kcal) | ±20% of EE | 1968.4 ± 607.4 | 2079.7 ± 474.0 | 1944.0 ± 97.7 | NS | NS | 1867.5 ± 517.4 | 2194.2 ± 393.5 | 1944.0 ± 97.7 | NS | NS | NS | NS | NS |
| Protein (%) | 12.5 | 16.3 ± 3.6 | 19.6 ± 3.6 | 19.8 ± 2.2 | NS | NS | 16.3 ± 3.7 | 20.3 ± 3.5 | 19.8 ± 2.0 | <0.05 | NS | NS | NS | NS |
| Lipids (%) | 32.5 | 41.1 ± 7.8 | 36.6 ± 6.7 | 40.6 ± 4.6 | 0.08 | NS | 41.3 ± 7.4 | 36.0 ± 7.3 | 40.6 ± 4.6 | NS | NS | NS | NS | NS |
| Saturated fatty acids (%) | <10 | 12.8 ± 6.8 | 12.1 ± 2.8 | 14.5 ± 2.6 | NS | NS | 12.0 ± 4.3 | 12.8 ± 2.9 | 14.5 ± 2.6 | NS | NS | NS | NS | NS |
| Carbohydrates (%) | 55 | 40.5 ± 8.3 | 42.5 ± 7.0 | 36.3 ± 7.1 | NS | NS | 40.2 ± 8.3 | 42.7 ± 8.0 | 36.3 ± 7.1 | NS | NS | NS | NS | NS |
| Simple sugars (%) | <10 | 4.3 ± 2.2 | 3.7 ± 1.2 | 4.1 ± 0.1 | NS | NS | 4.6 ± 2.3 | 3.8 ± 1.1 | 4.1 ± 0.1 | NS | NS | NS | NS | NS |
| Fibre (g) | 14 g/1000 kcal (25–35 g) | 21.6 ± 11.2 | 26.3 ± 11.6 | 25.3 ± 7.1 | 0.1 | NS | 19.2 ± 10.0 | 25.9 ± 9.3 | 25.3 ± 7.1 | 0.06 | NS | NS | <0.05 | NS |
| Cholesterol (mg) | <300 | 298.8 ± 132.3 | 302.7 ± 77.0 | 378.3 ± 125.2 | NS | NS | 296.3 ± 138.9 | 326.5 ± 65.5 | 378.3 ± 125.2 | NS | NS | NS | NS | NS |
| Adults | Children and Adolescents | Adults vs. Children | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Recommended Daily Portion Intake * | vt0 | vt12 | p Value | vt0 | vt12 | p Value | p Value | |||
| N | 22 | 4 | vt0 vs. vt12 | 31 | 16 | vt0 vs. vt12 | vt0 | vt12 | ||
| Daily consumption | Dairy | 2–4 | 2.6 ± 1.4 | 3.2 ± 0.3 | NS | 2.7 ± 1.3 | 2.5 ± 0.6 | NS | NS | NS |
| Cereals | 4–6 | 2.6 ± 1.4 | 2.7 ± 0.8 | NS | 3.8 ± 1.9 | 2.7 ± 1.5 | 0.098 | <0.05 | NS | |
| Vegetables | 2 | 1.3 ± 1.4 | 2.0 ± 1.3 | NS | 0.8 ± 0.5 | 0.9 ± 0.6 | NS | <0.05 | <0.05 | |
| Fruits | 3 | 2.3 ± 1.6 | 2.2 ± 1.5 | NS | 1.7 ± 1.3 | 2.0 ± 1.1 | NS | NS | NS | |
| Oils | 3–6 | 2.5 ± 1.2 | 2.7 ± 2.3 | NS | 2.1 ± 1.4 | 2.4 ± 1.3 | NS | NS | NS | |
| Weekly consumption | Meat | 3–4 | 6.2 ± 2.5 | 4.5 ± 1.3 | 0.1 | 7.2 ± 3.5 | 6.5 ± 1.4 | <0.01 | NS | NS |
| Fish | 3–4 | 3.5 ± 2.3 | 4.3 ± 1.4 | NS | 3.1 ± 1.6 | 3.4 ± 1.6 | NS | NS | NS | |
| Eggs | 3–4 | 2.9 ± 2.5 | 3.0 ± 1.4 | NS | 2.7 ± 1.2 | 2.8 ± 0.6 | NS | NS | NS | |
| Legumes | 2–4 | 1.9 ± 1.3 | 1.7 ± 1.2 | NS | 2.6 ± 1.2 | 2.5 ± 0.9 | NS | 0.06 | 0.1 | |
| Nuts | 3–7 | 2.6 ± 2.2 | 2.2 ± 1.4 | NS | 1.3 ± 2.1 | 1.1 ± 1.2 | NS | NS | NS | |
| Pastries | Ocassional (<1 portion/week) | 0.8 ± 0.9 | 0.3 ± 0.3 | NS | 1.3 ± 1.7 | 1.8 ± 2.2 | NS | <0.05 | NS | |
| Adults | p Value | Children and Adolescents | p Value | p Value (Adults vs. Children) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Vt0 | Vt3 | Vt12 | Vt0 vs. Vt3 | Vt0 vs. Vt12 | Vt0 | Vt3 | Vt12 | Vt0 vs. Vt3 | Vt0 vs. Vt12 | Vt0 | Vt3 | Vt12 | |
| n | 27 | 13 | 4 | 31 | 20 | 16 | |||||||
| G1 | 6.3 ± 2.4 | 7.2 ± 2.7 | 8.2 ± 1.8 | NS | 0.066 | 5.6 ± 1.9 | 6.8 ± 2.0 | 7.1 ± 2.3 | NS | 0.078 | <0.05 | <0.05 | NS |
| G2 | 2.8 ± 1.0 | 3.2 ± 1.2 | 3.8 ± 1.3 | NS | NS | 3.4 ± 1.3 | 4.4 ± 1.6 | 3.5 ± 1.0 | NS | NS | NS | 0.05 | NS |
| G3 | 1.4 ± 1.1 | 1.5 ± 0.9 | 1.8 ± 0.6 | NS | NS | 1.1 ± 0.8 | 1.1 ± 0.8 | 1.1 ± 0.9 | NS | NS | <0.05 | NS | NS |
| G4 | 5.0 ± 2.2 | 4.3 ± 1.8 | 3.7 ± 1.1 | <0.05 | 0.068 | 6.1 ± 2.1 | 5.9 ± 2.3 | 5.9 ± 1.8 | NS | <0.05 | NS | 0.068 | <0.05 |
| Adults | p Value | Children and Adolescents | p Value (Adults vs. Children) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Vt0 | Vt3 | Vt12 | Vt0 vs. Vt3 | Vt0 vs. Vt12 | Vt0 | Vt3 | Vt12 | Vt0 vs. Vt3 | Vt0 vs. Vt12 | Vt0 | Vt3 | Vt12 | |
| n | 27 | 13 | 4 | 25 | 21 | 14 | |||||||
| GFP E% | 23.7 ± 10.0 | 18.9 ± 6.1 | 21.4 ± 11.4 | <0.05 | 0.08 | 17.4 ± 12.6 | 18.8 ± 14.7 | 21.4 ± 10.7 | NS | NS | <0.05 | NS | NS |
| Adults | p Value | Children | p Value | Adults vs. Children | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Vt0 | Vt3 | Vt12 | Vt0 vs. Vt3 | Vt0 vs. Vt12 | Vt0 | Vt3 | Vt12 | Vt0 vs. Vt3 | Vt0 vs. Vt12 | Vt0 | Vt3 | Vt12 | |
| n | 25 | 12 | 4 | 30 | 21 | 11 | |||||||
| Fructose | 11.3 ±5.5 | 16.4 ± 12.3 | 14.7 ± 4.9 | 0.18 | NS | 10.2 ± 5.2 | 11.8 ± 6.3 | 13.2 ± 6.4 | NS | 0.06 | NS | NS | NS |
| Glucose | 9.5 ± 4.7 | 14.2 ± 9.5 | 12.1 ± 4.8 | <0.05 | NS | 8.6 ± 4.2 | 10.6 ± 5.3 | 9.9 ± 4.8 | NS | NS | NS | NS | NS |
| Fructans | 0.8 ± 1.1 | 0.8 ± 1.0 | 13.0 ± 1.0 | NS | NS | 0.5 ± 0.6 | 0.9 ± 1.1 | 0.3 ± 0.5 | 0.08 | NS | NS | NS | NS |
| Lactose | 10.4 ± 7.5 | 12.0 ± 8.9 | 13.0 ± 9.5 | NS | NS | 18.8 ± 10.8 | 13.8 ± 6.8 | 14.8 ± 8.6 | NS | NS | <0.05 | NS | NS |
| Inulin | 0.1 ± 0.2 | 0.4 ± 0.6 | 0.3 ± 0.5 | 0.1 | NS | 0.4 ± 0.3 | 0.2 ± 0.2 | 0.3 ± 0.4 | 0.055 | NS | <0.01 | NS | NS |
| Manitol | 0.1 ± 0.2 | 0.0 ± 0.0 | 0.1 ± 0.3 | NS | NS | 0.02 ± 0.1 | 0.01 ± 0.1 | 0.04 ± 0.2 | NS | NS | NS | NS | NS |
| Raffinose | NC | NC | NC | - | - | NC | NC | NC | - | - | - | - | - |
| Stachyose | 0.02 ± 0.1 | 0.0 ± 0.0 | 0.0 ± 0.0 | NS | NS | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | NS | NS | NS | NS | NS |
| Sorbitol | 0.4 ± 0.6 | 1.3 ± 1.9 | 1.4 ± 2.1 | <0.01 | NS | 1.0 ± 1.2 | 0.7 ± 0.7 | 0.8 ± 1.1 | NS | NS | <0.05 | NS | NS |
| TOTAL FODMAP | 32.8 ± 12.1 | 45.1 ± 26.8 | 42.3 ± 18.5 | <0.05 | NS | 39.4 ± 15.2 | 38.2 ± 11.4 | 41.8 ± 13.5 | NS | NS | 0.08 | NS | NS |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perez-Junkera, G.; Vázquez-Polo, M.; Eizagirre, F.J.; Benjumea, L.; Tutau, C.; Esteban, B.; Miranda, J.; Larretxi, I.; Navarro, V.; Churruca, I.; et al. Application of a Platform for Gluten-Free Diet Evaluation and Dietary Advice: From Theory to Practice. Sensors 2022, 22, 732. https://doi.org/10.3390/s22030732
Perez-Junkera G, Vázquez-Polo M, Eizagirre FJ, Benjumea L, Tutau C, Esteban B, Miranda J, Larretxi I, Navarro V, Churruca I, et al. Application of a Platform for Gluten-Free Diet Evaluation and Dietary Advice: From Theory to Practice. Sensors. 2022; 22(3):732. https://doi.org/10.3390/s22030732
Chicago/Turabian StylePerez-Junkera, Gesala, Maialen Vázquez-Polo, Francisco Javier Eizagirre, Laura Benjumea, Carlos Tutau, Blanca Esteban, Jonatan Miranda, Idoia Larretxi, Virginia Navarro, Itziar Churruca, and et al. 2022. "Application of a Platform for Gluten-Free Diet Evaluation and Dietary Advice: From Theory to Practice" Sensors 22, no. 3: 732. https://doi.org/10.3390/s22030732
APA StylePerez-Junkera, G., Vázquez-Polo, M., Eizagirre, F. J., Benjumea, L., Tutau, C., Esteban, B., Miranda, J., Larretxi, I., Navarro, V., Churruca, I., & Lasa, A. (2022). Application of a Platform for Gluten-Free Diet Evaluation and Dietary Advice: From Theory to Practice. Sensors, 22(3), 732. https://doi.org/10.3390/s22030732






