Determination of Gas Permeation Properties in Polymer Using Capacitive Electrode Sensors
Abstract
:1. Introduction
2. Experimental Aspects
Sample Preparation and Gas Exposure Conditions
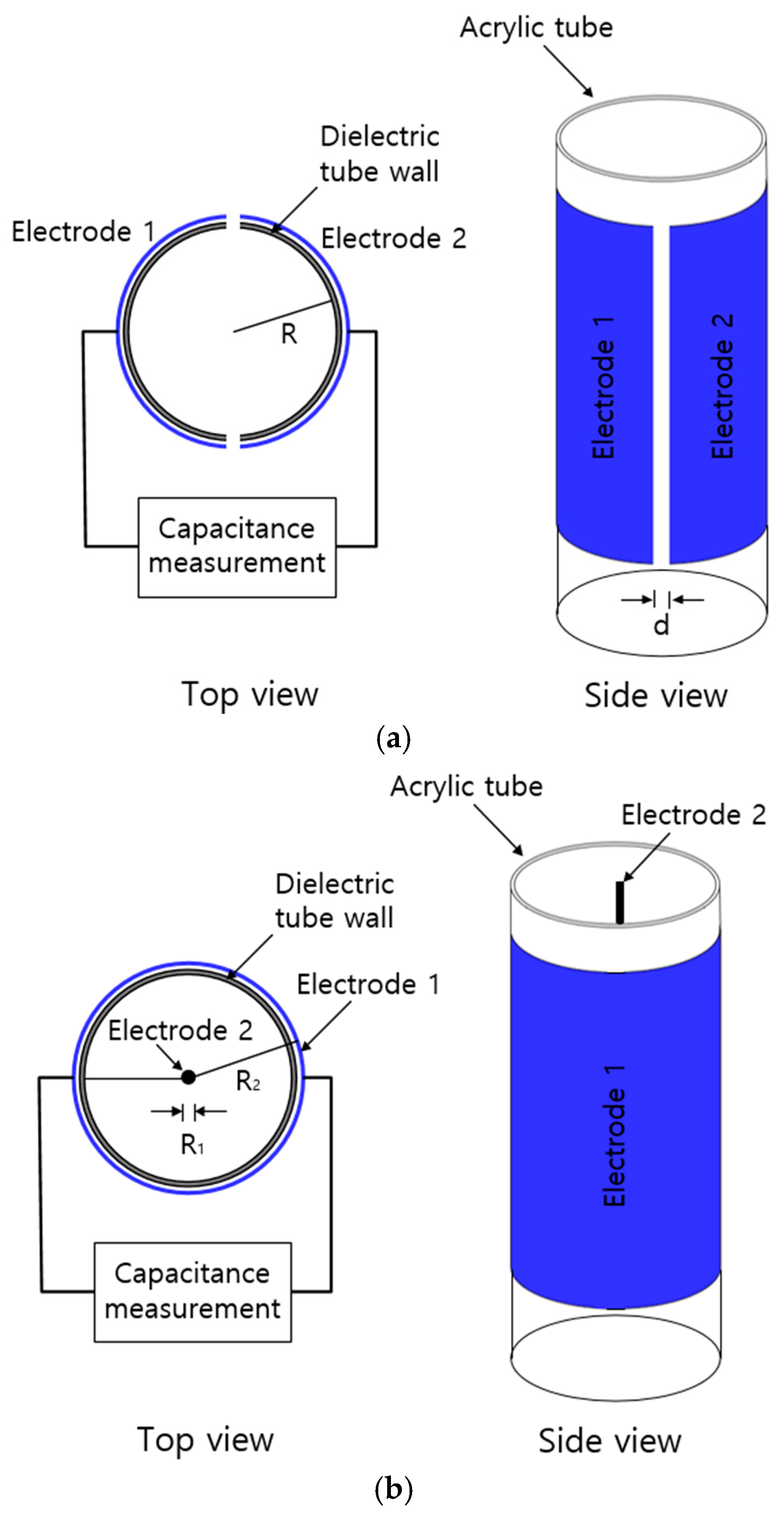
3. Two Types of Capacitor Electrodes to Measure the Water Level
3.1. Semi-Cylindrical Capacitor Electrode
3.2. Coaxial-Cylindrical Capacitor Electrode
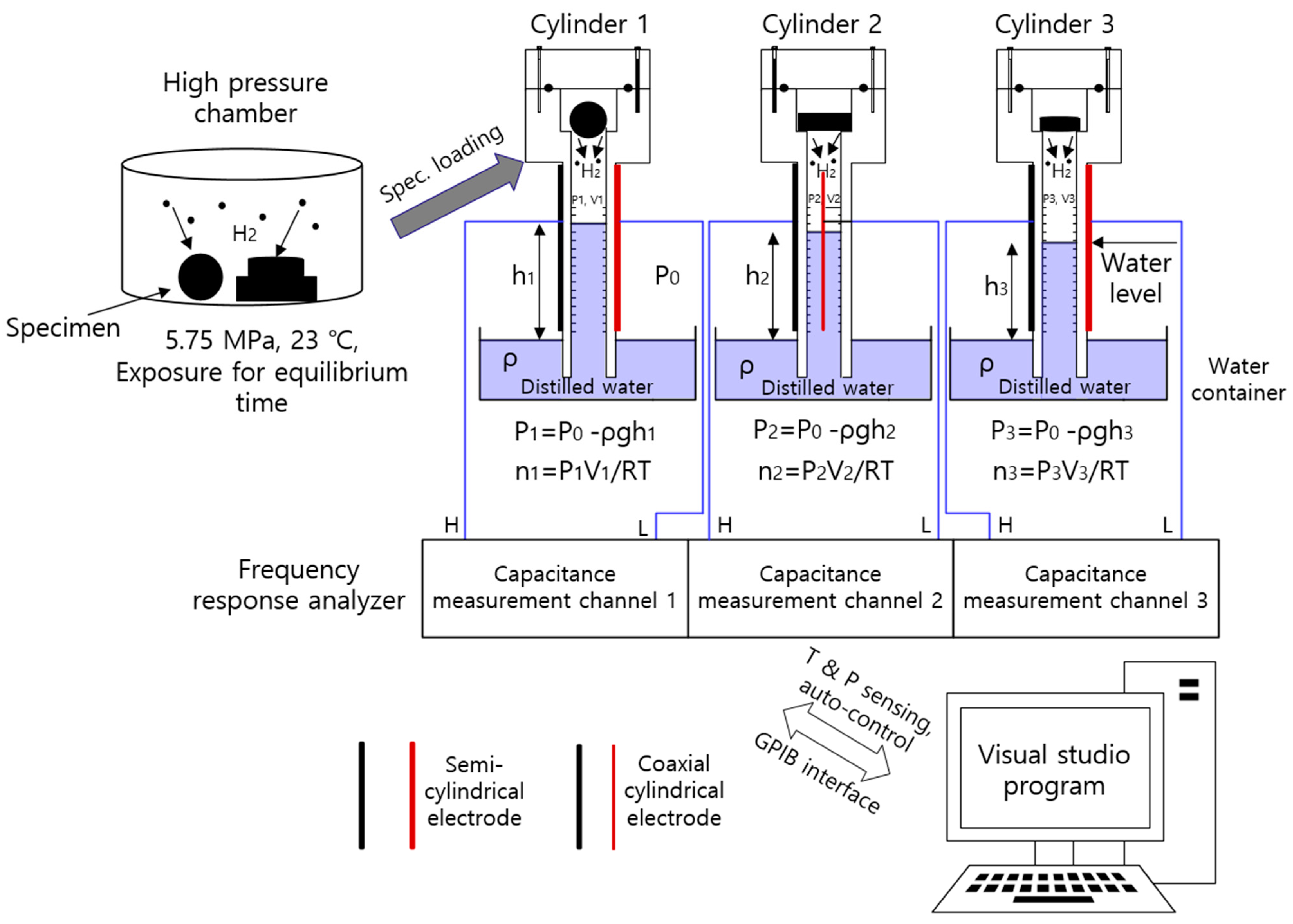
4. Volumetric Analysis Measurement System
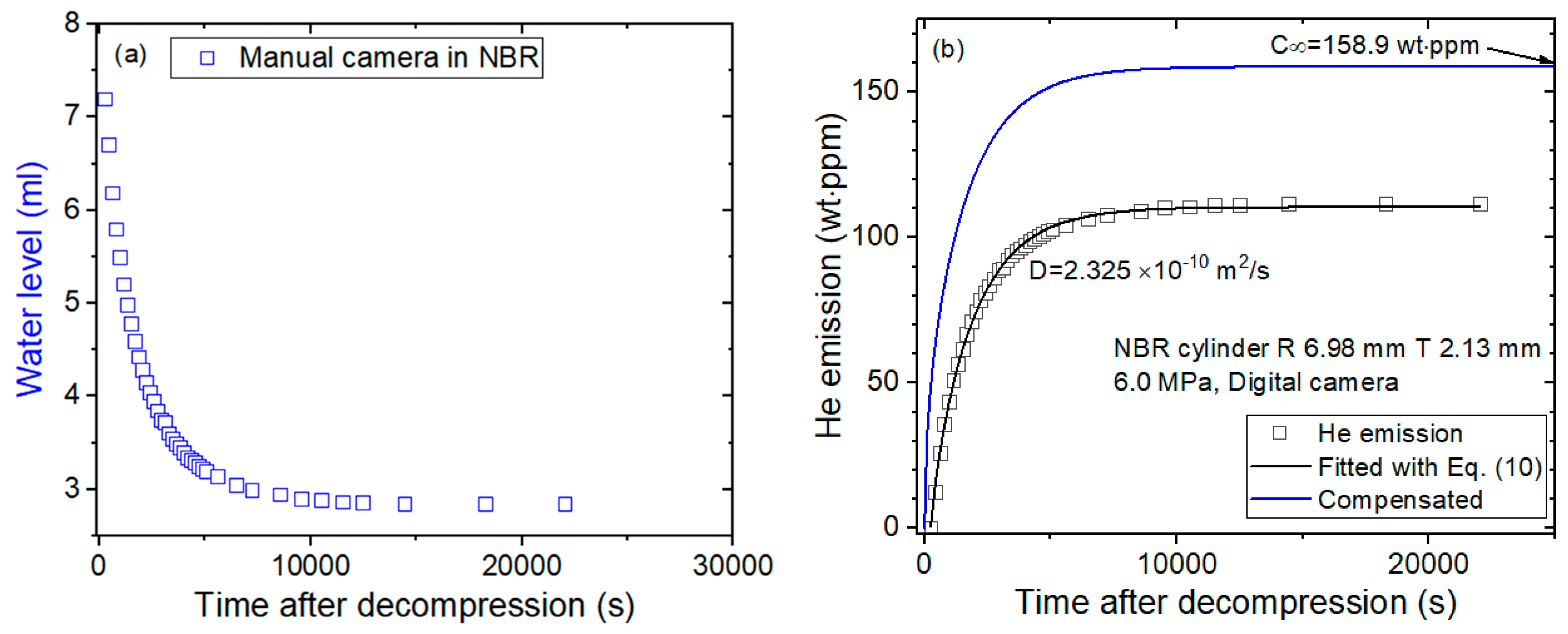
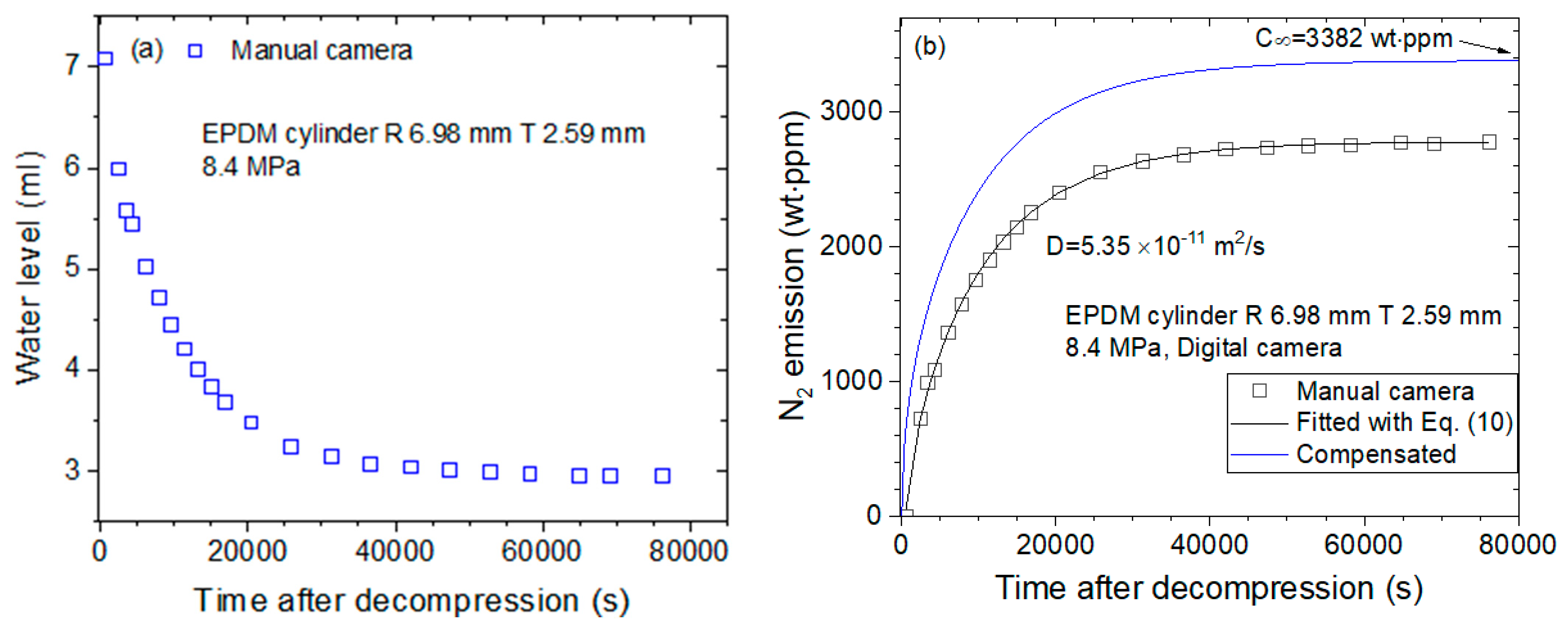
4.1. Volumetric Measurement of Emitted Gas
4.2. Time-Dependent Emitted Gas Concentration versus Specimen Shape
4.3. Diffusion Parameter Analysis through Programmed Capacitance Measurement
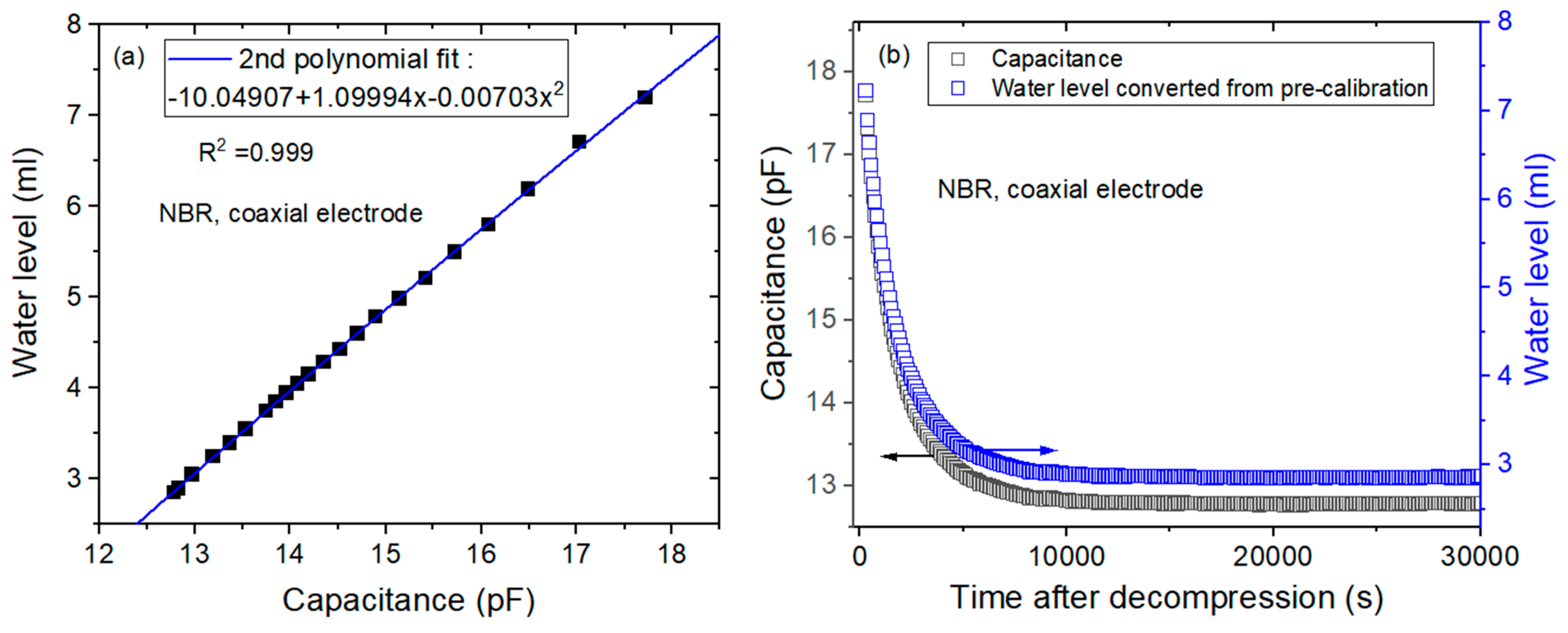
- (a)
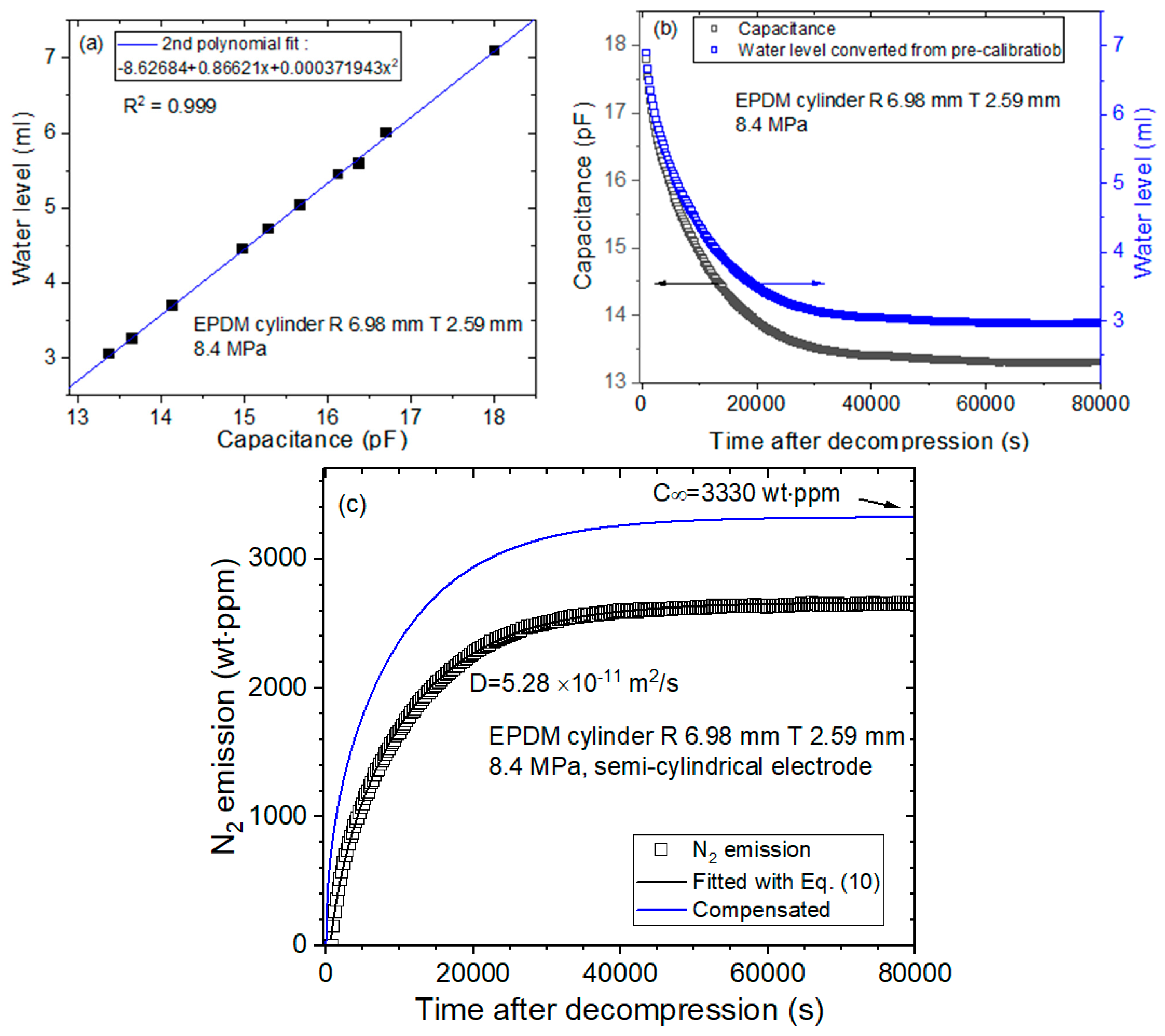
- To obtain the precalibration data, the user measures the water level versus the capacitance at the corresponding channel with decreasing water levels. Then, the 2nd polynomial equation related to the position of the water level and capacitance is obtained by quadratic regression, as shown in Figure 3a. The 2nd polynomial equation originates from Equation (4). The position of the water level is measured by a digital camera.
- (b)
- According to the precalibration data, the capacitance is transformed to the water level, as shown in Figure 3b. The black and blue squares correspond to the capacitance and position of the water level, respectively, versus the time elapsed.
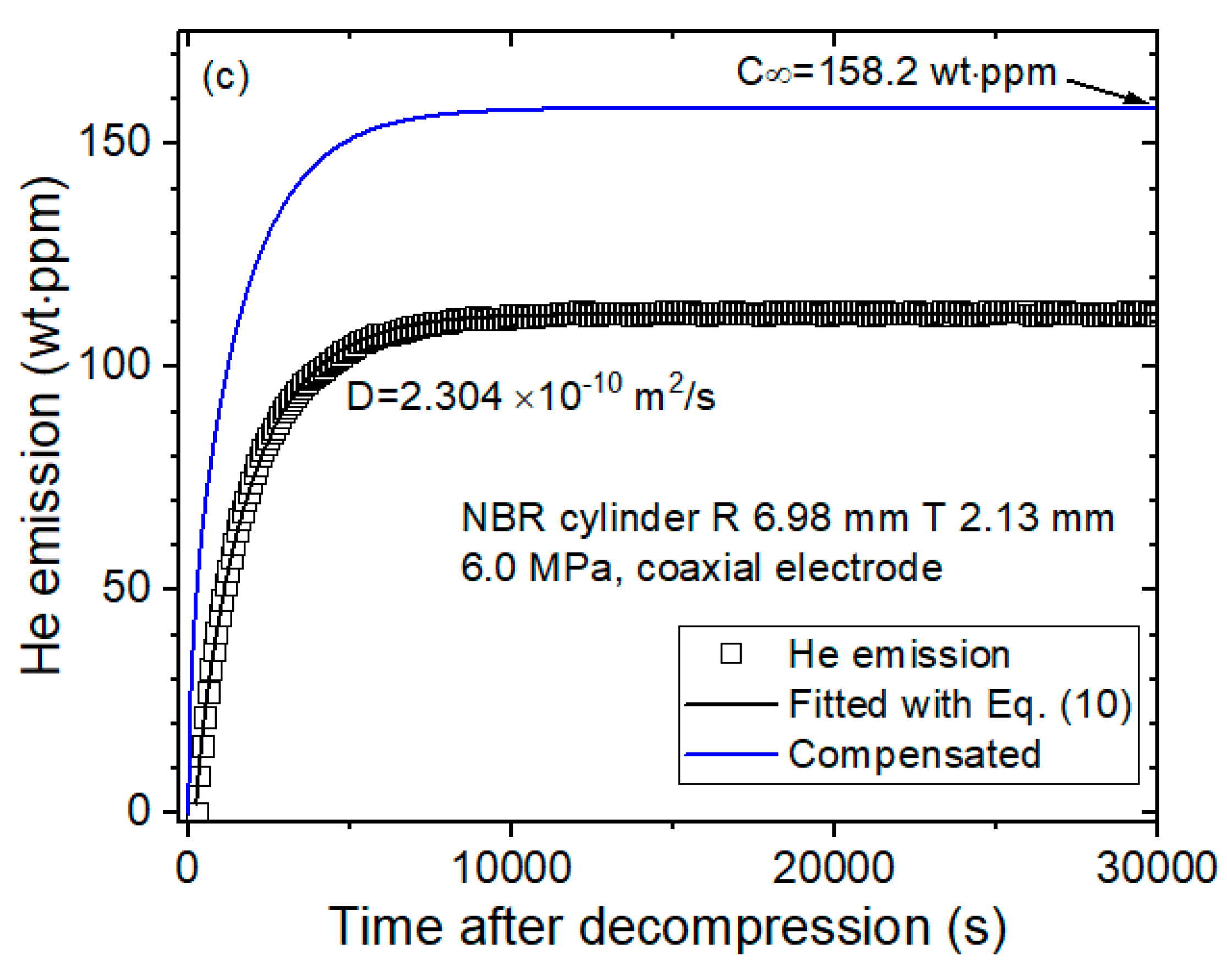
- (c)
- Last, the diffusion parameters D are determined using a diffusion analysis program by applying Equation (10) based on least-squares regression, as shown in Figure 3c.
5. Results and Discussion
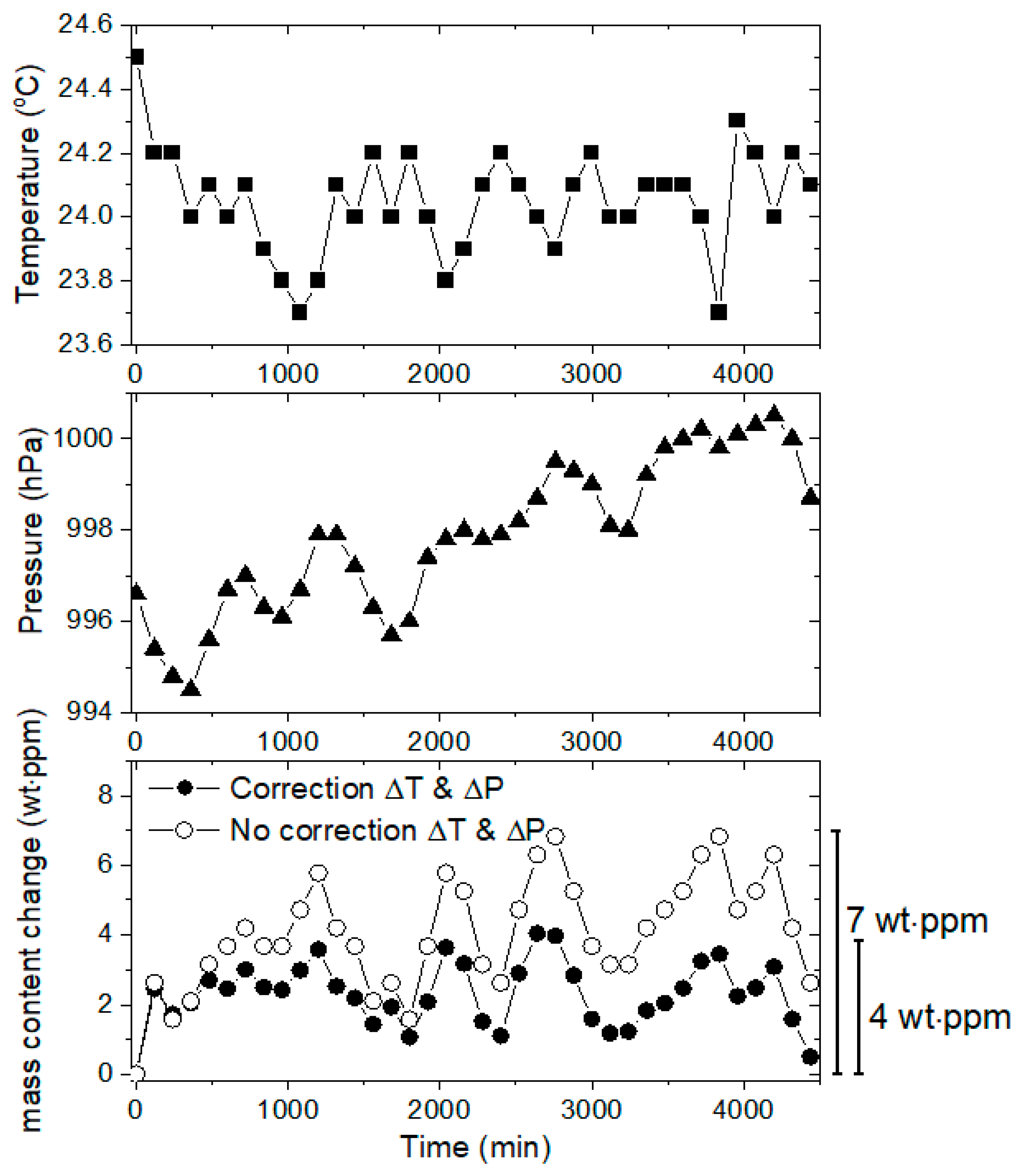
5.1. Stability Test of the Volumetric Measurement System
5.2. Pressure Dependence on the Permeation Parameter
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mitchell, M.; Link, R.; Severin, J.; Bix, L.; Linz, J.; Gilliland, D.; Lockhart, H. A new methodology for whole-package microbial challenge testing for medical device trays. J. Test. Eval. 2007, 35, 373–380. [Google Scholar] [CrossRef]
- Siracusa, V. Food packaging permeability behaviour: A report. Int. J. Polym. Sci. 2012, 2012, 1–11. [Google Scholar] [CrossRef]
- Tseng, H.H.; Lin, Z.Y.; Chen, S.H.; Lai, W.H.; Wey, M.Y. Reuse of reclaimed tire rubber for gas-separation membranes prepared by hot-pressing. J. Clean. Prod. 2019, 237, 117739. [Google Scholar] [CrossRef]
- Oriyama, T.; Yamamoto, T.; Yanagihara, Y.; Nara, K.; Abe, T.; Nakajima, K.; Aoyama, T.; Suzuki, H. Evaluation of the permeation of antineoplastic agents through medical gloves of varying materials and thickness and with varying surface treatments. J. Pharm. Health Care Sci. 2017, 3, 13. [Google Scholar] [CrossRef]
- George, S.; Ninan, K.; Thomas, S. Permeation of nitrogen and oxygen gases through styrene–butadiene rubber, natural rubber and styrene–butadiene rubber/natural rubber blend membranes. Eur. Polym. J. 2001, 37, 183–191. [Google Scholar] [CrossRef]
- BS EN ISO 2556; Plastics–Determination of the Gas Transmission Rate of Films and Thin Sheets under Atmospheric Pressure–Manometric Method. 2001. Available online: https://www.en-standard.eu/une-en-iso-2556-2001-plastics-determination-of-the-gas-transmission-rate-of-films-and-thin-sheets-under-atmospheric-pressure-manometric-method-iso-2556-1974 (accessed on 27 January 2022).
- Monson, L.; Moon, S.I.; Extrand, C. Gas permeation resistance of various grades of perfluoroalkoxy-polytetrafluoroethylene copolymers. J. Appl. Polym. Sci. 2009, 111, 141–147. [Google Scholar] [CrossRef]
- Minelli, M.; Sarti, G.C. Gas transport in glassy polymers: Prediction of diffusional time lag. Membranes 2018, 8, 8. [Google Scholar] [CrossRef] [Green Version]
- Mukaddam, M.; Litwiller, E.; Pinnau, I. Gas sorption, diffusion, and permeation in nafion. Macromolecules 2016, 49, 280–286. [Google Scholar] [CrossRef] [Green Version]
- Mamaliga, I.; Schabel, W.; Kind, M. Measurements of sorption isotherms and diffusion coefficients by means of a magnetic suspension balance. Chem. Eng. Process. Process Intensif. 2004, 43, 753–763. [Google Scholar] [CrossRef]
- Markočič, E.; Škerget, M.; Knez, Ž. Measurement of CO2 solubility and diffusivity in poly(l-lactide) and poly(d, l-lactide-co-glycolide) by magnetic suspension balance. J. Supercrit. Fluids 2008, 47, 296–301. [Google Scholar] [CrossRef]
- Liu, L.B.; Hashi, Y.; Qin, Y.P.; Zhou, H.X.; Lin, J.M. Development of automated online gel permeation chromatography-gas chromatograph mass spectrometry for measuring multiresidual pesticides in agricultural products. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2007, 845, 61–68. [Google Scholar] [CrossRef]
- Bemporad, D.; Essex, J.W.; Luttmann, C. Permeation of small molecules through a lipid bilayer: A computer simulation study. J. Phys. Chem. B 2004, 108, 4875–4884. [Google Scholar] [CrossRef]
- Barth, T.; Herbin, R.; Ohlberger, M. Finite volume methods: Foundation and analysis. In Encyclopedia of Computational Mechanics; Stein, E., Borst, R.D., Hughes, T.J.R., Eds.; John Wiley and Sons: New York, NY, USA, 2017. [Google Scholar]
- Jung, J.K.; Kim, I.G.; Kim, K.T.; Ryu, K.S.; Chung, K.S. Evaluation techniques of hydrogen permeation in sealing rubber materials. Polym. Test. 2021, 93, 107016. [Google Scholar] [CrossRef]
- Jung, J.K.; Kim, I.G.; Kim, K.T. Evaluation of hydrogen permeation characteristics in rubbery polymers. Curr. Appl. Phys. 2021, 21, 43–49. [Google Scholar] [CrossRef]
- Basel, I.I.; Wael, H.A. Innovative techniques for two-phase flow measurements. Recent Patents Electr. Eng. 2008, 1, 1–13. [Google Scholar] [CrossRef]
- Dangjuan, L.; Tang, L.; Shenjiang, W.; Huan, X.; Bin, W. The liquid level measurement of ultra low temperature cylinder based on the relative capacity method. In Proceedings of the 4th National Conference on Electrical, Electronics and Computer Engineering (NCEECE 2015), Xi’an, China, 12–13 December 2015; pp. 1061–1066. [Google Scholar]
- Crank, J. The Mathematics of Diffusion; Oxford University Press: Oxford, UK, 1975. [Google Scholar]
- Yang, Y.; Liu, S. Estimation and modeling of pressure-dependent gas diffusion coefficient for coal: A fractal theory-based approach. Fuel 2019, 253, 588–606. [Google Scholar] [CrossRef]
- Nelder, J.A.; Mead, R. A simplex method for function minimization. Comput. J. 1965, 7, 308–313. [Google Scholar] [CrossRef]
- Sander, R. Compilation of Henry’s law constants (Version 4.0) for water as solvent. Atmos. Chem. Phys. 2015, 15, 4399–4981. [Google Scholar] [CrossRef] [Green Version]
- Jung, J.K.; Kim, K.T.; Chung, K.S. Two volumetric techniques for determining the transport properties of hydrogen gas in polymer. Mater. Chem. Phys. 2022, 276, 125364. [Google Scholar] [CrossRef]
- Mehio, N.; Dai, S.; Jiang, D.E. Quantum mechanical basis for kinetic diameters of small gaseous molecules. J. Phys. Chem. A 2014, 118, 1150–1154. [Google Scholar] [CrossRef]
- Liu, Q.; Cai, Z. Study on the characteristics of gas molecular mean free pathin nanopores by molecular dynamics simulations. Int. J. Mol. Sci. 2014, 15, 12714–12730. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.G.; Kresse, I.; Xu, Z.K.; Springer, J. Effect of temperature and pressure on gas transport in ethyl cellulose membrane. Polymer 2001, 42, 6801–6810. [Google Scholar] [CrossRef]
- Shieh, J.J.; Chung, T.S. Gas permeability, diffusivity, and solubility of poly(4-vinylpyridine) film. J. Polym. Sci. Part B 1999, 37, 2851–2861. [Google Scholar] [CrossRef]
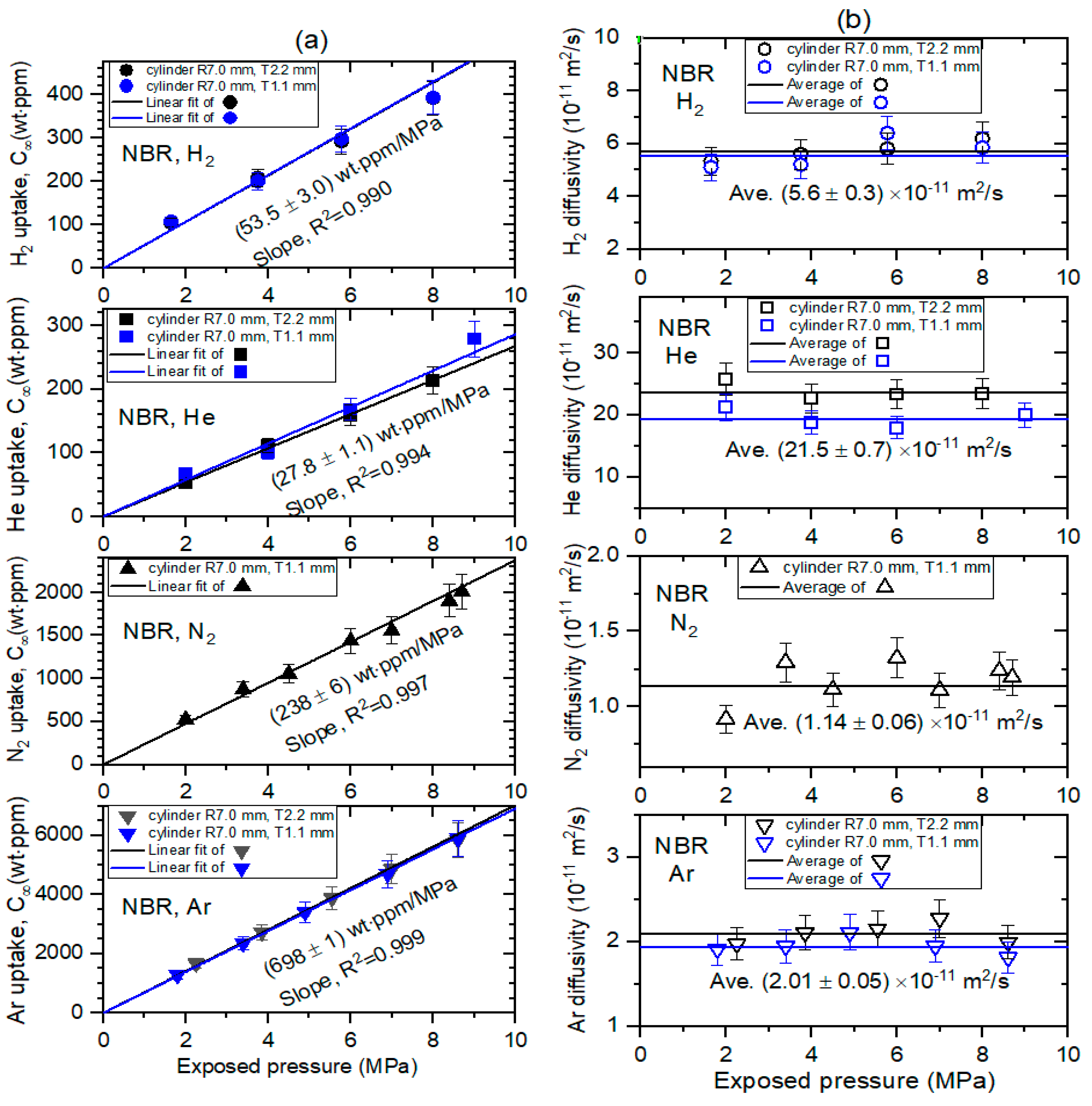


| Specimen | Solubility (mol/m3·MPa) | Diffusivity (×10−11 m2/s) | Permeability (mol/m·s·MPa, ×10−10) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| H2 | He | N2 | Ar | H2 | He | N2 | Ar | H2 | He | N2 | Ar | |
| NBR | 34.2 (35.3) | 8.96 | 11.0 | 22.5 | 5.60 (6.50) | 21.5 | 1.14 | 2.01 | 19.2 (22.8) | 19.3 | 1.25 | 4.53 |
| EPDM | 25.6 (26.2) [23] | 7.79 | 17.0 | 38.6 | 19.7 (24.1) [23] | 83.1 | 7.24 | 10.5 | 50.3 (63.1) [23] | 64.8 | 12.3 | 40.4 |
| Parameter | Coaxial-Cylindrical | Semi-Cylindrical |
|---|---|---|
| Sensitivity | ~3 pF/mL | ~1 pF/mL |
| Resolution | ~0.5 wt·ppm | ~2 wt·ppm |
| Stability | <10 wt·ppm | <15 wt·ppm |
| Detection range | ~max 1000 wt·ppm for H2 | ~max 1000 wt·ppm for H2 |
| Response time | <1 s | <1 s |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jung, J.; Kim, G.; Gim, G.; Park, C.; Lee, J. Determination of Gas Permeation Properties in Polymer Using Capacitive Electrode Sensors. Sensors 2022, 22, 1141. https://doi.org/10.3390/s22031141
Jung J, Kim G, Gim G, Park C, Lee J. Determination of Gas Permeation Properties in Polymer Using Capacitive Electrode Sensors. Sensors. 2022; 22(3):1141. https://doi.org/10.3390/s22031141
Chicago/Turabian StyleJung, Jaekap, Gyunghyun Kim, Gahyoun Gim, Changyoung Park, and Jihun Lee. 2022. "Determination of Gas Permeation Properties in Polymer Using Capacitive Electrode Sensors" Sensors 22, no. 3: 1141. https://doi.org/10.3390/s22031141
APA StyleJung, J., Kim, G., Gim, G., Park, C., & Lee, J. (2022). Determination of Gas Permeation Properties in Polymer Using Capacitive Electrode Sensors. Sensors, 22(3), 1141. https://doi.org/10.3390/s22031141






