1. Introduction
The Track-Hold System (THS) project, developed in a healthcare facility and therefore in a controlled and protected healthcare environment, is carried out in the broader framework of Robotic-Assisted Therapy (RAT). RAT represents an advanced and innovative rehabilitation method, both motor and cognitive, and makes use of active, passive, and facilitating robotic devices. Sensors often equip robotic devices for detecting and tracking both voluntary and involuntary movements, and specially developed multimedia protocols to achieve the highest possible level of functional re-education.
Like conventional rehabilitation methods (e.g., Perfetti Method, Mirror Therapy, Biofeedback, etc.), the RAT requires close collaboration between the members of the Multidisciplinary Team, which in the RAT, besides the Doctor and the Therapist, requires the Physiologist and Biomedical and Computer Engineer [
1].
Track-Hold: A System for Robotic-Assisted Therapy
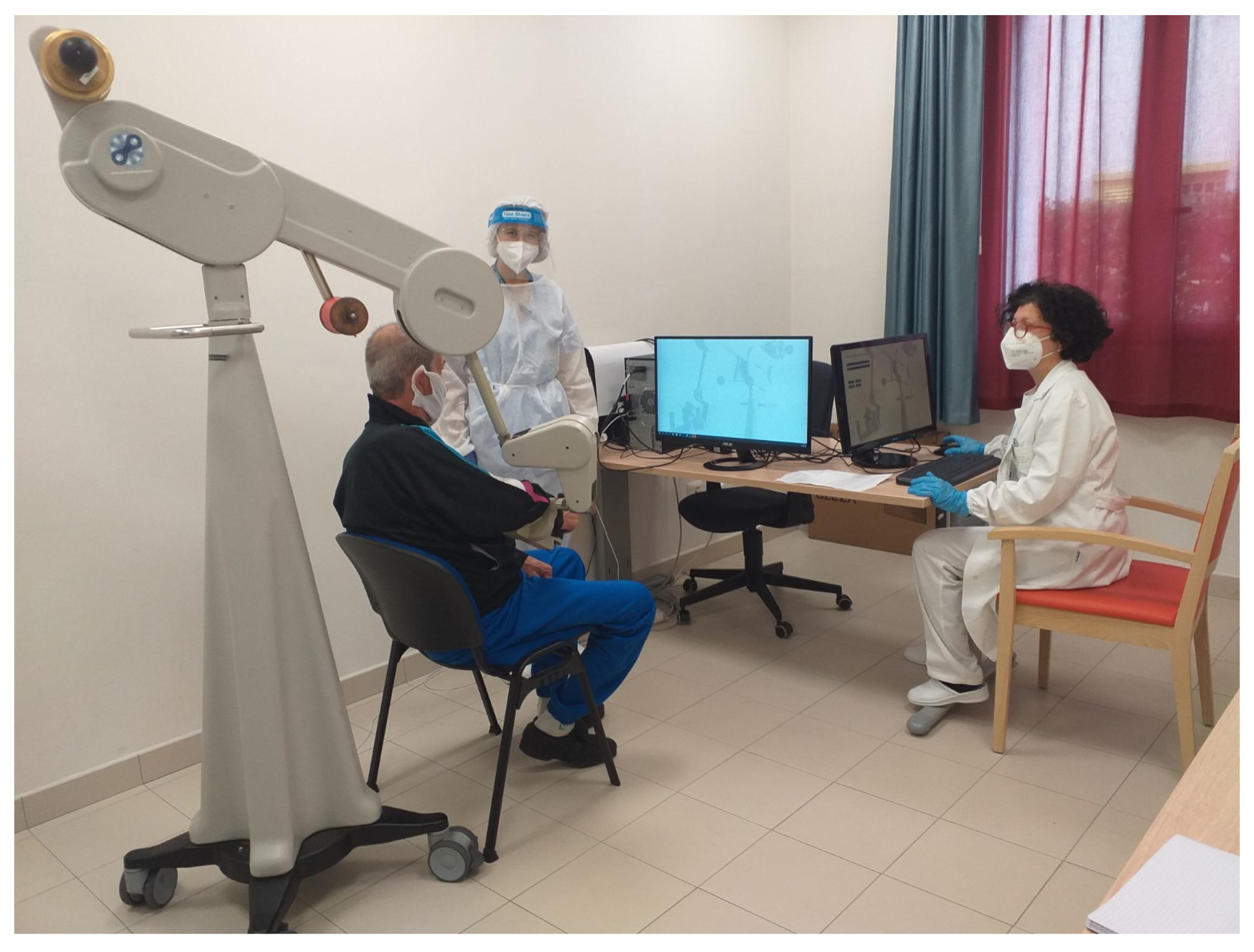
Track-Hold is a passive robotic arm capable of recording and facilitating the movements of the upper limbs. The robotic device was created in its mechanical and sensor components by the “Wearable Robotics” Laboratory, a spin-off of the Institute of Biorobotics of the Sant’Anna School of Advanced Studies (Pisa), and completed by an operational interface for its use in the clinical field, by the Institute of Science and Technology (ISTI) “Faedo” of the National Research Council (CNR) of Pisa, based on specialist clinical advice.
Thanks to a solution based on levers and weights, the arm allows to move a person’s upper limb with a push from the bottom up, varying the weight. Track-Hold records the movements of the limb of the subject under various conditions and multiple settings. Namely, Track-Hold settings permit the movement of the subject’s limb in the following conditions:
in the absence of gravitational force or with opposing gravitational pull (thus, the user to keep the arm in balance must apply a downward force);
in the presence of the natural force of gravitation;
in an intermediate condition.
The levers, the weights, and the limb constitute a set of scales that allow it to function even in equilibrium conditions. These characteristics, especially the functioning with the limb in weightlessness, make it suitable for the rehabilitation and passive training of subjects with motor difficulties of the upper limbs, resulting from previous ischaemic events, progressive neurological pathologies, and extreme loss of strength—reduction in muscle mass or, again, coordination or difficulty in organizing purposeful movements.
The execution of the limb movements of the patient to be re-educated is facilitated by the passive mechanics of the balance wheel, as the upward thrust allows the patient to move the limb with minimal muscular effort. The clinical evaluation made by the physician is used to adapt the amount of support, determining how much weight should be relieved. The device consists of arms connected by joints. Each of the joints is equipped with a patented precision angle sensor [
2].
The integrated Track-Hold technology allows recording the arm’s movements through a two-way association between the actions of the upper limb and those of the robotic arm. The recorded kinematic data are objective and can be used, among other things, to estimate improvements and progress of personalized therapeutic programs.
The manipulator of the device, consisting of a handle, is equipped with a pressure sensor that allows detecting when it is pressed with the hand (for example, by detecting the gesture of gripping objects). The sampling rate of the positions is 100 Hz, which is sufficient for correct motion detection. The device is connected to a computer via a USB interface.
Track-Hold has been equipped with an agent software that acts as an operational interface. CNR-ISTI Signals and Images laboratory (SI-Lab) set up the interface in the laboratory in collaboration with the Clinicians (this activity took place between July 2020 and October 2020). The operational interface offers a wide range of exercises that the patient must perform under the control of the operators.
The research work, which took place in the period indicated above, had among the main objectives the creation of special software which uses the potential of the device technology in response to the clinical-rehabilitation needs.
2. State of the Art in Robotic Assisted Therapy (RAT)
Robot Assisted Therapy (RAT) with both passive and active devices, is an innovative form of rehabilitation that enables highly repetitive, intensive, adaptive, and quantifiable physical training. It has been increasingly used to restore loss of motor function, mainly in stroke survivors suffering from an upper limb paresis. Multiple studies collated in a growing number of review articles showed the positive effects on motor impairment, less clearly on functional limitations [
1]. Recovering upper limb motor functions has important implications for improving the independence of patients after stroke and also in patients with tetraplegia caused by spinal cord injury, traumatic brain injury, and in patients with impaired function of motor neurons, that characterize certain neurological diseases, such as multiple sclerosis, cerebral palsy, Guillain-Barre syndrome, finally in patients affected by essential tremor, and Parkinson’s disease [
1,
3,
4].
In this context, Robotic Assisted Therapy (RAT) is an innovative approach to upper limb rehabilitation. The RAT involves intensive and repetitive exercise, with interactive and individualized practice, which could also imply the execution of oriented tasks or dual-task paradigms, as was shown in our preliminary work [
5].
The RAT encompasses technological solutions, such as computerized control systems, and mechanical devices, with both active and passive functioning, that could promote motor learning [
6].
Promoting processes of motor learning and cross-education from unaffected to affected upper limb [
7] is the most important goal of rehabilitation, because the recovery of motor functions requires neurophysiological mechanisms.
The devices that implement RAT, such as wearable sensors and free-contact systems for gesture tracking, are highly reliable to capture and measure kinematic and dynamic parameters of the upper limb, such as movement quality, speed, direction, strength and range of joints motion [
5,
8,
9].
Results of previous studies [
4,
10] showed that RAT could improve motor control, regulating muscle activation patterns, as required for the execution of aimed movements during daily activities. It has also been reported that these improvements of motor skills are increased both in the short and long term [
4].
A recent metanalysis [
11] shows that RAT, compared to the traditional rehabilitation approach, enhances, with prime evidence, arm motion and muscle strength, improving the motor ability of patients in daily activities, and finally, their quality of life.
Notably, it has been reported that RAT, with individualized and customized protocol of exercises, could improve in post-stroke patients also neurophysiological aspects of the upper limb, mainly enhancing shoulder and elbow range of motion [
12,
13,
14,
15,
16].
The most important paradigm of RAT is not only to maximize the number of task repetitions but also to increase patient attention and effort as well [
17].
Indeed, it is known that repetitive and monotonous exercises provide worse retention of motor skills, compared with alternate training [
18].
Furthermore, it has been speculated that RAT could decrease recovery of motor functions if it encourages assuaging since the patients could decrease effort and attention due to the use of adaptive algorithms, i.e., during training with active robotic devices [
19].
Indeed, clinical evidence shows that, since learning is error-based, a faster improvement of motor skills may be achieved when the rate of error is increased [
20].
Cognitive and motor rehabilitation after stroke and other neurological diseases is firmly based on intensive and repetitive training and task-specific learning for promoting neural reorganization and subsequent recovery [
21]. Conventional methods for cognitive and motor rehabilitation of upper limbs, such as paper and pencil tasks performing, still strive to accomplish this therapeutic aim [
22].
Moreover, studies with stroke survivors have shown a differential pattern of motor outcomes, depending on the cognitive domain impaired [
23].
Rehabilitation methods, based on robotic systems, implemented with exergames, provided positive results in patients with post-stroke disabilities of upper limb [
24].
Exergames, used as task-oriented motor functions, have been used in RAT to improve certain cognitive functions, promoting the activity of specific domains, especially those related to executive functions and attention, that regulate decision making, movement programming, and time reaction [
25].
As described above, state of the art indicates that the combination of RAT with exergames might have a beneficial outcome. In consideration of our previous work [
2] in which a specific suite of exergames was designed for use with the Track Hold System, in this paper, we extend the experimental results by proposing a pilot study and analyzing the impact of our methodology in improving upper limb motricity.
3. The Case Study
3.1. Design and Objectives
Our study was an extended case report with an enrollment of a small sample of patients.
The enrolled patients signed an informed consent drafted according to the guidelines of the local Ethics Committee, furthermore, the procedures were carried out in accordance with the provisions of the Declaration of Helsinki on clinical trials on humans.
SI-Lab staff, the practitioner in neuro-rehabilitation, and the physiotherapists operating in the ICARE Healthcare Facility of the former Tabarracci hospital (Viareggio) collaborated on the THScase study experimentation.
The case study mainly had the objective of developing a motor and cognitive rehabilitation protocol in subjects with post-stroke outcomes affecting the right upper limb, including strength deficits, tremor, incoordination, and motor apraxia.
3.2. Experimental Protocol
The protocol consisted of a 4-week training period for subjects with a motor deficit of the right upper limb; enrolled patients performed guided motor exercises with weight relief using the TrackHold device. Patients performed the proposed tasks following a criterion of progressive difficulty (achievement of a greater number of key points). Furthermore, based on the progressive recovery of muscle strength, weight relief was slowly reduced, clinically assessing each patient’s level of muscle strength session by session.
6. Conclusions and Future Works
Our preliminary results show that THS training could be effective to improve upper limb motricity in the acute and subacute post-stroke stage. Preliminary clinical results of the Track-Hold System demonstrated good patient compliance with this kind of robotic rehabilitation. The interfaces for users and operators made it possible to establish an effective recovery path for patients who were able to gradually resume (even if not completely) the functions of the upper limb. In the form of exergames, the guiding exercises of varying difficulty made it possible to emulate the gestures of daily activities. Following the exergames, the patients learned the lost movement capacity during the workouts; then, they have reproduced the movements in their daily acts, thus approaching the quality of life before the disease.
Further experimentation is necessary for a large sample of patients to confirm the effectiveness of this cognitive and motor rehabilitation approach.
This case study has shown that exercises carried out using a passive robotic device equipped with weight relief allow partial recovery of the functional capacity of the upper limb even three months after a stroke episode. The next step of this study involves a comparison between this rehabilitation approach and a clinical method that is currently used for the functional recovery of stroke survivors, namely Action Observation Therapy (AOT) [
30]. The AOT is mainly based on a process of movements learning: during the rehabilitation sessions, the patient tries to reproduce gestures performed by the physiotherapist, which are always representative of activities of daily life. The method described in the present work could offer advantages compared to AOT, in terms of the accuracy of monitoring and efficacy of the treatment [
30].