Measuring Protons with Photons: A Hand-Held, Spectrophotometric pH Analyzer for Ocean Acidification Research, Community Science and Education
Abstract
1. Introduction
2. Materials and Methods
2.1. Spectrophotometric pH Chemistry
2.1.1. pH Chemistry
2.1.2. Meta–Cresol Purple Dye
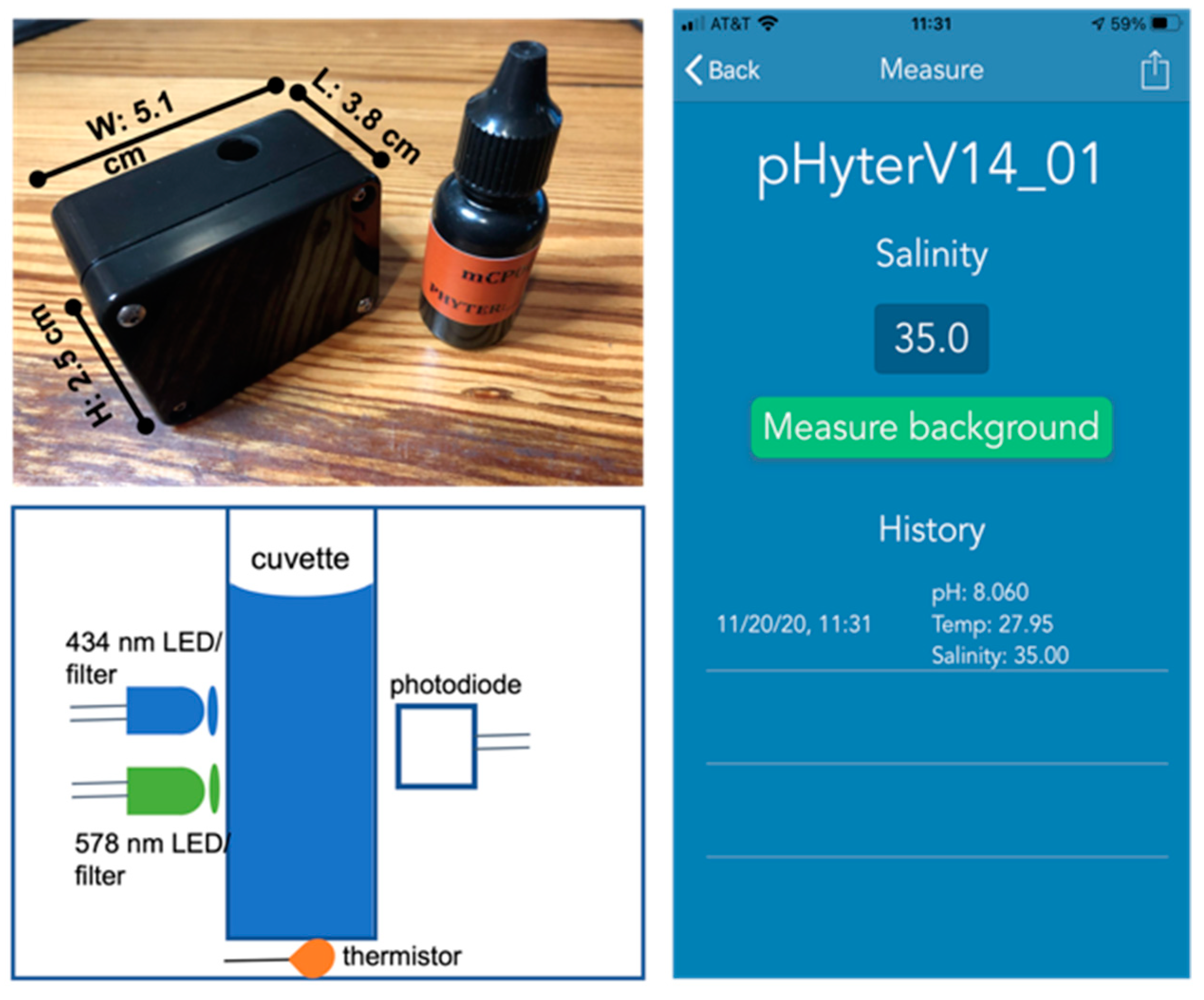
2.2. Instrument Design and Operation
2.2.1. pHyter Components
2.2.2. pHyter Operation
- (1).
- Prior to each measurement, the user can input the salinity of the solution into the UI, otherwise, it will default to 35. Salinity readings can be obtained from a simple, inexpensive hand-held refractometer, e.g., a Brix Refractometer B01LW4HHRC, for ~$20.
- (2).
- About 3 mL of sample solution is added to the sample cuvette, using a dropper or by submerging the pHyter directly in the water. Then the rubber stopper is placed on the sample cuvette and a blank (Iλ0) is measured at both wavelengths by clicking the “Measure Background” button on the smartphone UI (Figure 1). After a few seconds, the UI will read “Measure pH”, noting that the background was successfully collected.
- (3).
- The stopper is removed, and two drops of the indicator dye are added to the sample. The stopper is replaced, and the instrument is inverted 10 times slowly to mix the solution and dye. The transmitted intensity (Iλ) of the sample with the dye addition is then measured at both wavelengths by pressing the “Measure pH” button on the UI.
2.3. Laboratory Evaluation
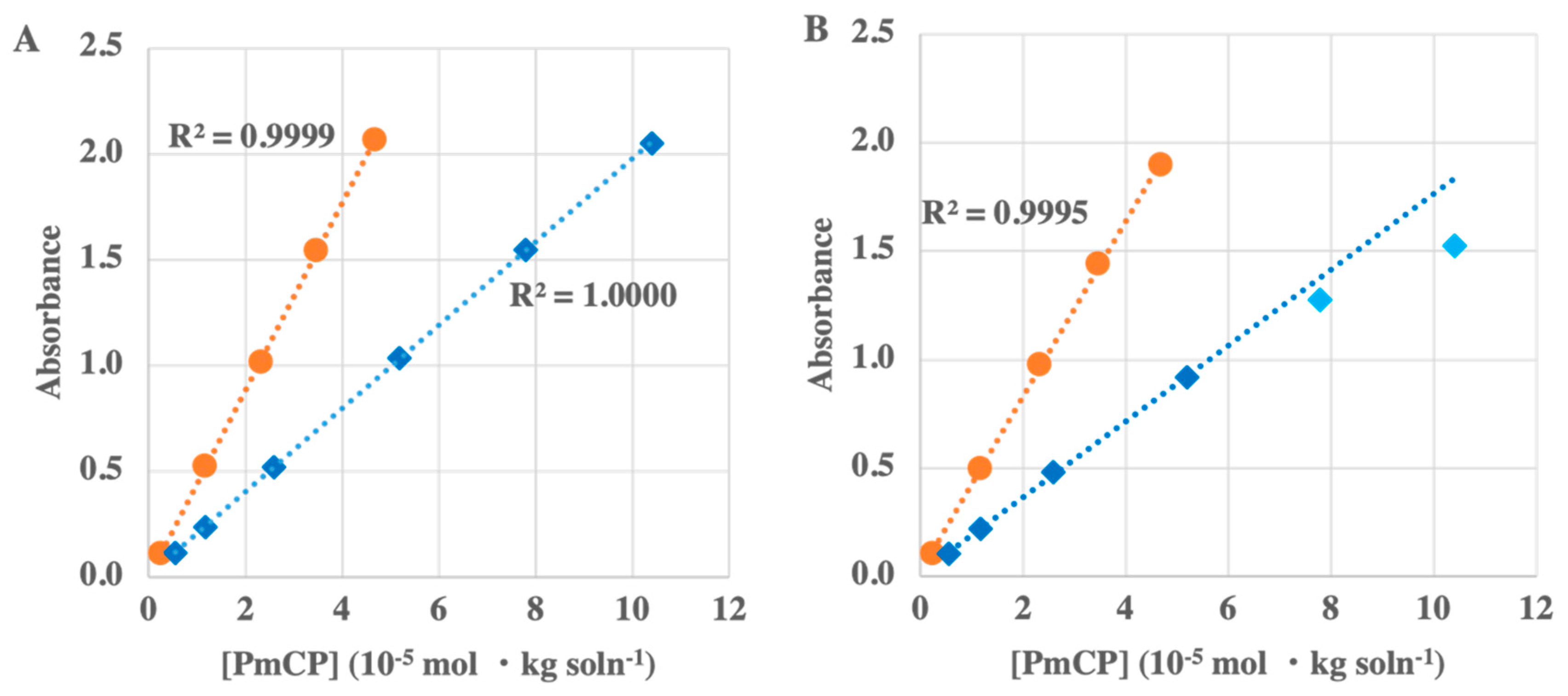
2.3.1. pHyter Linearity
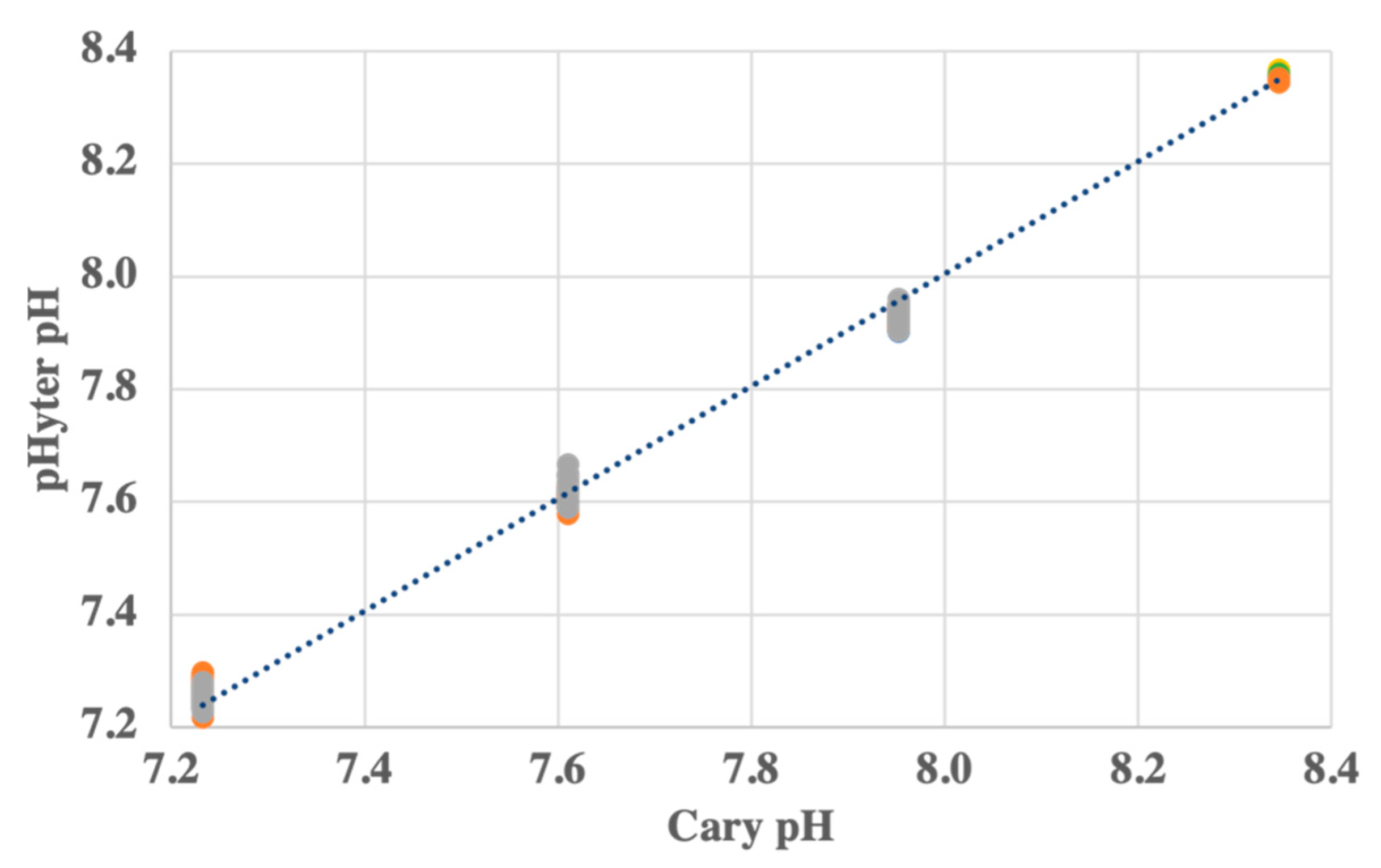
2.3.2. Evaluation of Precision and Accuracy
2.4. Field Verification
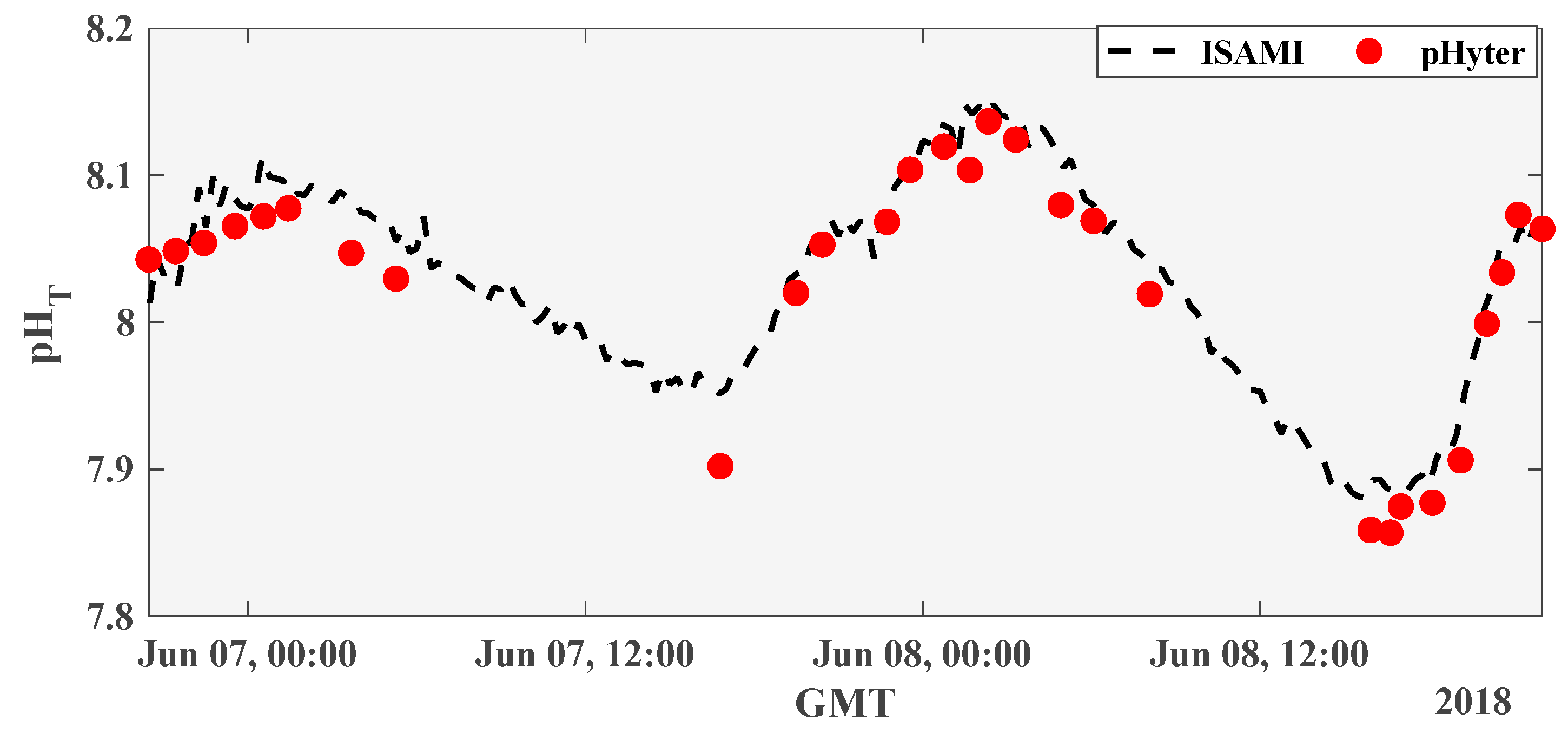
2.4.1. Field Measurements in Comparison to the iSAMI-pH
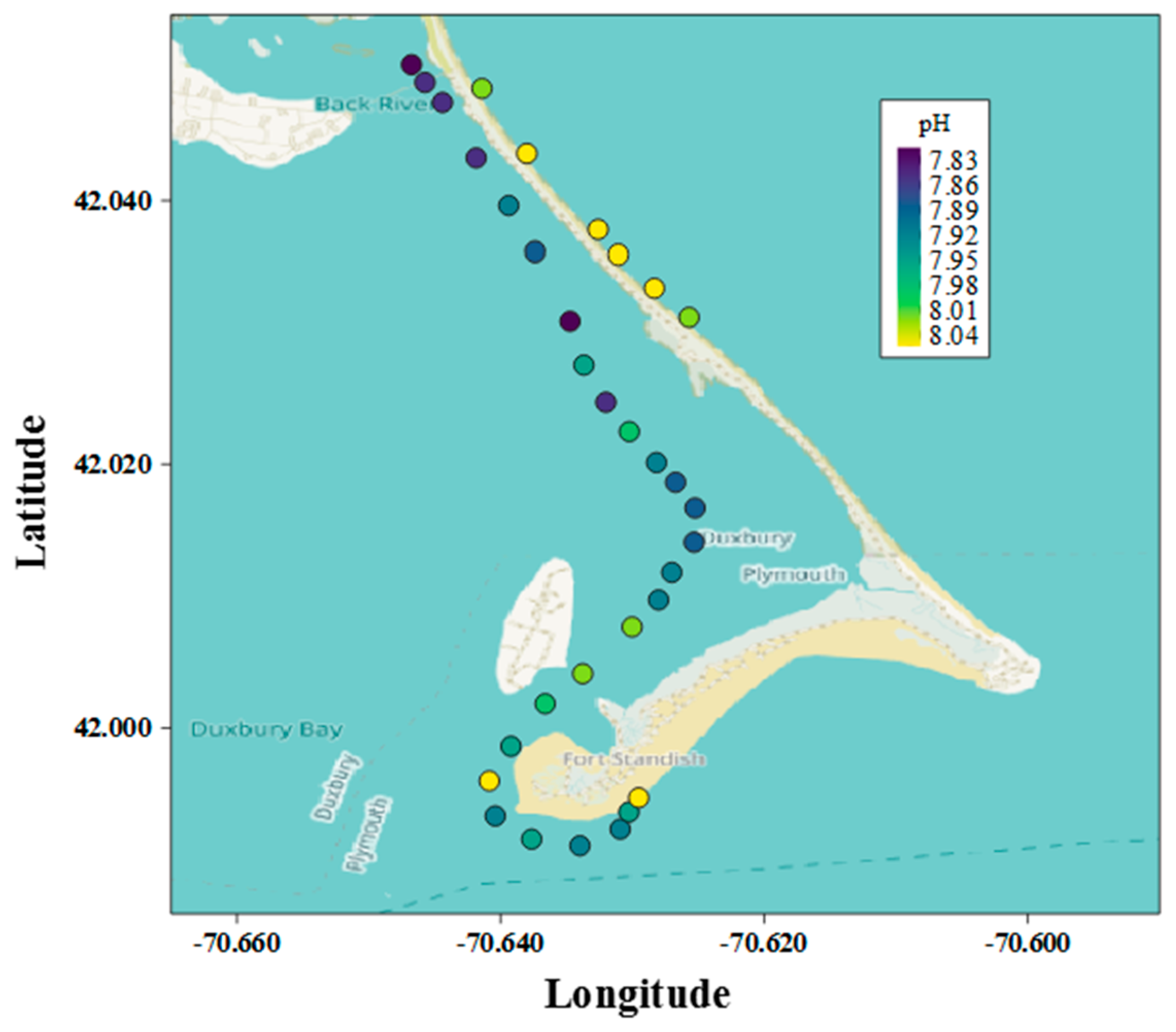
2.4.2. Spatial Field Application
3. Results and Discussion
3.1. Laboratory Evaluation
3.1.1. pHyter Linearity
3.1.2. pHyter Precision and Accuracy
3.2. Field Evaluation
3.2.1. Temporal Field Measurements in Comparison to the iSAMI-pH
3.2.2. Spatial Field Application
3.3. The pHyter in Community-Based Science, Education, and Outreach
3.4. The Future of the pHyter
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Keeling, R.F.; Keeling, C.D. Atmospheric Monthly in Situ CO2 Data-Mauna Loa Observatory, Hawaii. Available online: http://scrippsco2.ucsd.edu/data/atmospheric_co2/primary_mlo_co2_record (accessed on 29 December 2020).
- Sabine, C.L.; Feely, R.A.; Gruber, N.; Key, R.M.; Lee, K.; Bullister, J.L.; Wanninkhof, R.; Wong, C.S.; Wallace, D.W.R.; Tilbrook, B.; et al. The Oceanic Sink for Anthropogenic CO2. Science (1979) 2004, 305, 367–371. [Google Scholar] [CrossRef]
- Doney, S.C.; Fabry, V.J.; Feely, R.A.; Kleypas, J.A. Ocean Acidification: The Other CO2 Problem. Ann. Rev. Mar. Sci. 2009, 1, 169–192. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.I.; Mukherjee, K.; Shoukat, R.; Dong, H. A Review on PH Sensitive Materials for Sensors and Detection Methods. Microsyst. Technol. 2017, 23, 4391–4404. [Google Scholar] [CrossRef]
- Richier, S.; Fiorini, S.; Kerros, M.-E.; Von Dassow, P.; Gattuso, J.-P. Response of the Calcifying Coccolithophore Emiliania Huxleyi to Low PH/High PCO2: From Physiology to Molecular Level. Mar. Biol. 2011, 158, 551–560. [Google Scholar] [CrossRef]
- Hofmann, G.E.; Smith, J.E.; Johnson, K.S.; Send, U.; Levin, L.A. High-Frequency Dynamics of Ocean PH: A Multi-Ecosystem Comparison. PLoS ONE 2011, 6, 28983. [Google Scholar] [CrossRef]
- Ghoneim, M.T.; Nguyen, A.; Dereje, N.; Huang, J.; Moore, G.C.; Murzynowski, P.J.; Dagdeviren, C. Recent Progress in Electrochemical PH-Sensing Materials and Configurations for Biomedical Applications. Chem. Rev. 2019, 119, 5248–5297. [Google Scholar] [CrossRef]
- Olof, H.; Samuelsson, G.; Sjoberg, S.; Ingri, N. A Precise Potentiometric Method for Determination of Algal Activity in an Open CO2 System. Plant Cell Environ. 1983, 6, 195–201. [Google Scholar]
- Brehmer, P.; do Chi, T.; Laugier, T.; Galgani, F.; Laloë, F.; Darnaude, A.M.; Fiandrino, A.; Mouillot, D. Field Investigations and Multi-Indicators for Shallow Water Lagoon Management: Perspective for Societal Benefit. Aquat. Conserv. 2011, 21, 728–742. [Google Scholar] [CrossRef]
- Pauly, D.; Christensen, V.; Guenette, S.; Pitcher, T.J.; Sumaila, U.R.; Walters, C.J.; Watson, R.; Zeller, D. Towards Sustainability in World Fisheries. Nature 2002, 418, 689–695. [Google Scholar] [CrossRef]
- Bauer, J.E.; Cai, W.-J.; Raymond, P.A.; Bianchi, T.S.; Hopkinson, C.S.; Regnier, P.A.G. The Changing Carbon Cycle of the Coastal Ocean. Nature 2013, 504, 61–70. [Google Scholar] [CrossRef]
- Feely, R.A.; Sabine, C.L.; Hernandez-Ayon, J.M.; Ianson, D.; Hales, B. Evidence for Upwelling of Corrosive “Acidified” Water onto the Continental Shelf. Science 2008, 320, 1490–1492. [Google Scholar] [CrossRef] [PubMed]
- Cai, W.J.; Hu, X.; Huang, W.J.; Murrell, M.C.; Lehrter, J.C.; Lohrenz, S.E.; Chou, W.C.; Zhai, W.; Hollibaugh, J.T.; Wang, Y.; et al. Acidification of Subsurface Coastal Waters Enhanced by Eutrophication. Nat. Geosci. 2011, 4, 766–770. [Google Scholar] [CrossRef]
- Cornwall, C.E.; Hepburn, C.D.; Mcgraw, C.M.; Currie, K.I.; Pilditch, C.A.; Hunter, K.A.; Boyd, P.W.; Hurd, C.L. Diurnal Fluctuations in Seawater PH Influence the Response of a Calcifying Macroalga to Ocean Acidification. Proc. R. Soc. B Biol. Sci. 2013, 280, 20132201. [Google Scholar] [CrossRef] [PubMed]
- Yuqing, M.; Jianrong, C.; Keming, F. New Technology for the Detection of PH. J. Biochem. Biophys. Methods 2005, 63, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Martz, T.R.; Connery, J.G.; Johnson, K.S. Testing the Honeywell Durafet® for Seawater PH Applications. Limnol. Oceanogr. Methods 2010, 8, 172–184. [Google Scholar] [CrossRef]
- Clayton, T.D.; Byrne, R.H. Instruments and methods spectrophotometric Seawater PH Measurements: Total Hydrogen Ion Concentration Scale Calibration of m-Cresol Purple and at-Sea Results. Deep Sea Res. Part I Oceanogr. Res. Pap. 1993, 40, 2115–2129. [Google Scholar] [CrossRef]
- Friis, K.; Körtzinger, A.; Wallace, D.W.R. Spectrophotometric PH Measurement in the Ocean: Requirements, Design, and Testing of an Autonomous Charge-Coupled Device Detector System. Limnol. Oceanogr. Methods 2004, 2, 126–136. [Google Scholar] [CrossRef]
- Chierici, M.; Fransson, A.; Anderson, L.G. Influence of M-Cresol Purple Indicator Additions on the PH of Seawater Samples: Correction Factors Evaluated from a Chemical Speciation Model. Mar. Chem. 1999, 65, 281–290. [Google Scholar] [CrossRef]
- Robert-Baldo, G.L.; Morris, M.J.; Byrne, R.H. Spectrophotometric Determination of Seawater PH Using Phenol Red. Anal. Chem 1985, 57, 2564–2567. [Google Scholar] [CrossRef]
- Byrne, R.H.; Yao, W. Procedures for Measurement of Carbonate Ion Concentrations in Seawater by Direct Spectrophotometric Observations of Pb(II) Complexation. Mar. Chem. 2008, 112, 128–135. [Google Scholar] [CrossRef]
- Byrne, R.H.; Breland, J.A. High Precision Multiwavelength PH Determinations in Seawater Using Cresol Red. Deep Sea Res. Part A Oceanogr. Res. Pap. 1989, 36, 803–810. [Google Scholar] [CrossRef]
- Seidel, M.P.; DeGrandpre, M.D.; Dickson, A.G. A Sensor for in Situ Indicator-Based Measurements of Seawater PH. Mar. Chem. 2008, 109, 18–28. [Google Scholar] [CrossRef]
- Cullison Gray, S.E.; DeGrandpre, M.D.; Moore, T.S.; Martz, T.R.; Friederich, G.E.; Johnson, K.S. Applications of in Situ PH Measurements for Inorganic Carbon Calculations. Mar. Chem. 2011, 125, 82–90. [Google Scholar] [CrossRef]
- Korostynska, O.; Arshak, K.; Gill, E.; Arshak, A. Review on State-of-the-Art in Polymer Based PH Sensors. Sensors 2007, 7, 3027–3042. [Google Scholar] [CrossRef]
- DeGrandpre, M.D.; Spaulding, R.S.; Newton, J.O.; Jaqueth, E.J.; Hamblock, S.E.; Umansky, A.A.; Harris, K.E. Considerations for the Measurement of Spectrophotometric PH for Ocean Acidification and Other Studies. Limnol. Oceanogr. Methods 2014, 12, 830–839. [Google Scholar] [CrossRef]
- Byrne, R.H.; Mecking, S.; Feely, R.A.; Liu, X. Direct Observations of Basin-Wide Acidification of the North Pacific Ocean. Geophys. Res. Lett. 2010, 37. [Google Scholar] [CrossRef]
- Liu, X.; Patsavas, M.C.; Byrne, R.H. Purification and Characterization of Meta-Cresol Purple for Spectrophotometric Seawater Ph Measurements. Environ. Sci. Technol. 2011, 45, 4862–4868. [Google Scholar] [CrossRef]
- Yang, B.; Patsavas, M.C.; Byrne, R.H.; Ma, J. Seawater PH Measurements in the Field: A DIY Photometer with 0.01 Unit PH Accuracy. Mar. Chem. 2014, 160, 75–81. [Google Scholar] [CrossRef]
- Dickson, A.G. Standard Potential of the Reaction: AgCl(s) + 1 2H2(g) = Ag(s) + HCl(Aq), and the Standard Acidity Constant of the Ion HSO4- in Synthetic Sea Water from 273.15 to 318.15 K. J. Chem. Thermodyn. 1990, 22, 113–127. [Google Scholar] [CrossRef]
- Müller, J.D.; Rehder, G. Metrology of PH Measurements in Brackish Waters-Part 2: Experimental Characterization of Purified Meta-Cresol Purple for Spectrophotometric PHT Measurements. Front. Mar. Sci. 2018, 5, 177. [Google Scholar] [CrossRef]
- Douglas, N.K.; Byrne, R.H. Spectrophotometric PH Measurements from River to Sea: Calibration of MCP for 0 ≤ S ≤ 40 and 278.15 ≤ T ≤ 308.15 K. Mar. Chem. 2017, 197, 64–69. [Google Scholar] [CrossRef]
- Mosley, L.M.; Husheer, S.L.G.; Hunter, K.A. Spectrophotometric PH Measurement in Estuaries Using Thymol Blue and M-Cresol Purple. Mar. Chem. 2004, 91, 175–186. [Google Scholar] [CrossRef]
- Douglas, N.K.; Byrne, R.H. Achieving Accurate Spectrophotometric PH Measurements Using Unpurified Meta-Cresol Purple. Mar. Chem. 2017, 190, 66–72. [Google Scholar] [CrossRef]
- Patsavas, M.C.; Byrne, R.H.; Liu, X. Purification of Meta-Cresol Purple and Cresol Red by Flash Chromatography: Procedures for Ensuring Accurate Spectrophotometric Seawater PH Measurements. Mar. Chem. 2013, 150, 19–24. [Google Scholar] [CrossRef]
- Marion, G.M.; Millero, F.J.; Camões, M.F.; Spitzer, P.; Feistel, R.; Chen, C.T.A. PH of Seawater. Mar. Chem. 2011, 126, 89–96. [Google Scholar] [CrossRef]
- Müller, J.D.; Bastkowski, F.; Sander, B.; Seitz, S.; Turner, D.R.; Dickson, A.G.; Rehder, G. Metrology for PH Measurements in Brackish Waters-Part 1: Extending Electrochemical PHT Measurements of TRIS Buffers to Salinities 5-20. Front. Mar. Sci. 2018, 5, 176. [Google Scholar] [CrossRef]
- Takeshita, Y.; Warren, J.K.; Liu, X.; Spaulding, R.S.; Byrne, R.H.; Carter, B.R.; DeGrandpre, M.D.; Murata, A.; Watanabe, S. Consistency and Stability of Purified Meta-Cresol Purple for Spectrophotometric PH Measurements in Seawater. Mar. Chem. 2021, 236, 104018. [Google Scholar] [CrossRef]
- Bockmon, E.E.; Dickson, A.G. An Inter-Laboratory Comparison Assessing the Quality of Seawater Carbon Dioxide Measurements. Mar. Chem. 2015, 171, 36–43. [Google Scholar] [CrossRef]
- Takeshita, Y.; Martz, T.R.; Johnson, K.S.; Dickson, A.G. Characterization of an Ion Sensitive Field Effect Transistor and Chloride Ion Selective Electrodes for Ph Measurements in Seawater. Anal. Chem. 2014, 86, 11189–11195. [Google Scholar] [CrossRef]
- Easley, R.A.; Byrne, R.H. Spectrophotometric Calibration of PH Electrodes in Seawater Using Purified M-Cresol Purple. Environ. Sci. Technol. 2012, 46, 5018–5024. [Google Scholar] [CrossRef]
| Temperature (C) | Tris pH | pHyter 1 | pHyter 2 | pHyter 3 | pHyter 4 | pHyter 5 | Average Offset | Standard Deviation |
|---|---|---|---|---|---|---|---|---|
| 24.74 ± 0.09 | 8.107 ± 0.003 | 8.130 | 8.120 | 8.120 | 8.120 | 8.110 | +0.013 | ±0.007 |
| 22.09 ± 0.35 | 8.190 ± 0.011 | 8.230 | 8.220 | 8.230 | 8.200 | 8.190 | +0.024 | ±0.010 |
| 27.72 ± 0.19 | 8.016 ± 0.006 | 8.030 | 8.189 | 8.010 | 8.013 | N/A | +0.046 (+0.002) | ±0.086 (0.011) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pardis, W.; Grabb, K.C.; DeGrandpre, M.D.; Spaulding, R.; Beck, J.; Pfeifer, J.A.; Long, D.M. Measuring Protons with Photons: A Hand-Held, Spectrophotometric pH Analyzer for Ocean Acidification Research, Community Science and Education. Sensors 2022, 22, 7924. https://doi.org/10.3390/s22207924
Pardis W, Grabb KC, DeGrandpre MD, Spaulding R, Beck J, Pfeifer JA, Long DM. Measuring Protons with Photons: A Hand-Held, Spectrophotometric pH Analyzer for Ocean Acidification Research, Community Science and Education. Sensors. 2022; 22(20):7924. https://doi.org/10.3390/s22207924
Chicago/Turabian StylePardis, William, Kalina C. Grabb, Michael D. DeGrandpre, Reggie Spaulding, James Beck, Jonathan A. Pfeifer, and David M. Long. 2022. "Measuring Protons with Photons: A Hand-Held, Spectrophotometric pH Analyzer for Ocean Acidification Research, Community Science and Education" Sensors 22, no. 20: 7924. https://doi.org/10.3390/s22207924
APA StylePardis, W., Grabb, K. C., DeGrandpre, M. D., Spaulding, R., Beck, J., Pfeifer, J. A., & Long, D. M. (2022). Measuring Protons with Photons: A Hand-Held, Spectrophotometric pH Analyzer for Ocean Acidification Research, Community Science and Education. Sensors, 22(20), 7924. https://doi.org/10.3390/s22207924






