Evaluation of Prolonged Walking in Persons with Multiple Sclerosis: Reliability of the Spatio-Temporal Walking Variables during the 6-Minute Walk Test
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
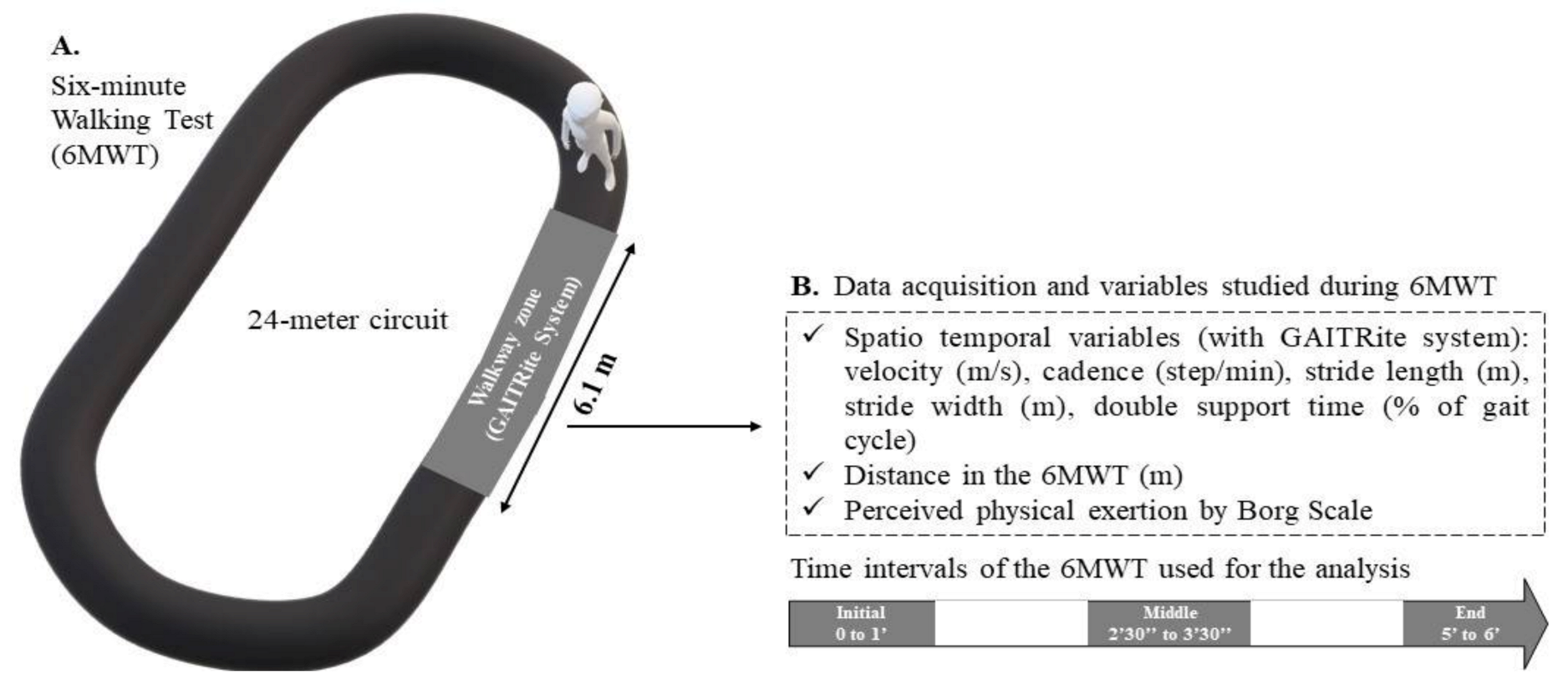
2.2. Procedures and Instrumentation
2.3. Statistical Analysis
3. Results
3.1. Characteristics of the Participants at Baseline
3.2. Test-Retest Reliability
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Compston, A.; Coles, A. Multiple sclerosis. Lancet 2008, 372, 1502–1517. [Google Scholar] [CrossRef]
- Noseworthy, J.; Paty, D.; Wonnacott, T.; Feasby, T.; Ebers, G. Multiple sclerosis after age 50. Neurology 1983, 33, 1537. [Google Scholar] [CrossRef] [PubMed]
- Dalgas, U.; Langeskov-Christensen, M.; Skjerbæk, A.; Jensen, E.; Baert, I.; Romberg, A.; Medina, C.S.; Gebara, B.; de Noordhout, B.M.; Knuts, K.; et al. Is the impact of fatigue related to walking capacity and perceived ability in persons with multiple sclerosis? A multicenter study. J. Neurol. Sci. 2018, 387, 179–186. [Google Scholar] [CrossRef] [PubMed]
- LaRocca, N.G. Impact of walking impairment in multiple sclerosis. Patient Patient-Cent. Outcomes Res. 2011, 4, 189–201. [Google Scholar] [CrossRef]
- Graham, J.E.; Ostir, G.V.; Fisher, S.R.; Ottenbacher, K.J. Assessing walking speed in clinical research: A systematic review. J. Eval. Clin. Pract. 2008, 14, 552–562. [Google Scholar] [CrossRef]
- Skjerbæk, A.G.; Boesen, F.; Petersen, T.; Rasmussen, P.V.; Stenager, E.; Nørgaard, M.; Feys, P.; Kjeldgaard-Jørgensen, M.L.; Hvid, L.G.; Dalgas, U. Can we trust self-reported walking distance when determining EDSS scores in patients with multiple sclerosis? The Danish MS hospitals rehabilitation study. Mult. Scler. 2018, 1352458518795416. [Google Scholar] [CrossRef]
- Brochet, B.; Bonnet, M.; Deloire, M.; Hamel, D.; Salort-Campana, E. Les troubles cognitifs au cours de la sclérose en plaques. Rev. Neurol. 2007, 163, 697–702. [Google Scholar] [CrossRef]
- Gijbels, D.; Dalgas, U.; Romberg, A.; de Groot, V.; Bethoux, F.; Vaney, C.; Gebara, B.; Medina, C.S.; Maamâgi, H.; Rasova, K. Which walking capacity tests to use in multiple sclerosis? A multicentre study providing the basis for a core set. Mult. Scler. J. 2012, 18, 364–371. [Google Scholar] [CrossRef]
- Motl, R.W.; Cohen, J.A.; Benedict, R.; Phillips, G.; LaRocca, N.; Hudson, L.D.; Rudick, R. Multiple Sclerosis Outcome Assessments Consortium, Validity of the timed 25-foot walk as an ambulatory performance outcome measure for multiple sclerosis. Mult. Scler. J. 2017, 23, 704–710. [Google Scholar] [CrossRef]
- Dalgas, U.; Severinsen, K.; Overgaard, K. Relations between 6 Minute Walking Distance and 10 Meter Walking Speed in Patients with Multiple Sclerosis and Stroke. Arch. Phys. Med. Rehabil. 2012, 93, 1167–1172. [Google Scholar] [CrossRef]
- Stellmann, J.P.; Neuhaus, A.; Götze, N.; Briken, S.; Lederer, C.; Schimpl, M.; Heesen, C.; Daumer, M. Ecological Validity of Walking Capacity Tests in Multiple Sclerosis. PLoS ONE 2015, 10, e0123822. [Google Scholar] [CrossRef]
- Moore, J.L.; Potter, K.; Blankshain, K.; Kaplan, S.L.; O’Dwyer, L.C.; Sullivan, J.E. A Core Set of Outcome Measures for Adults with Neurologic Conditions Undergoing Rehabilitation: A Clinical Practice Guideline. J. Neurol. Phys. Ther. 2018, 42, 174–220. [Google Scholar] [CrossRef]
- Bethoux, F.; Bennett, S. Evaluating walking in patients with multiple sclerosis: Which assessment tools are useful in clinical practice? Int. J. MS Care 2011, 13, 4–14. [Google Scholar] [CrossRef]
- Dalgas, U.; Kjølhede, T.; Gijbels, D.; Romberg, A.; Santoyo, C.; Noordhout, B.; Knuts, K.; Feys, P. Aerobic intensity and pacing pattern during the six-minute walk test in patients with multiple sclerosis. J. Rehabil. Med. 2014, 46, 59–66. [Google Scholar] [CrossRef]
- Goldman, M.D.; Marrie, R.A.; Cohen, J.A. Evaluation of the six-minute walk in multiple sclerosis subjects and healthy controls. Mult. Scler. J. 2008, 14, 383–390. [Google Scholar] [CrossRef]
- Cederberg, K.L.J.; Sikes, E.M.; Bartolucci, A.A.; Motl, R.W. Walking endurance in multiple sclerosis: Meta-analysis of six-minute walk test performance. Gait Posture 2019, 73, 147–153. [Google Scholar] [CrossRef]
- Motl, R.W.; Suh, Y.; Balantrapu, S.; Sandroff, B.M.; Sosnoff, J.J.; Pula, J.; Goldman, M.D.; Fernhall, B. Evidence for the different physiological significance of the 6- and 2-minute walk tests in multiple sclerosis. BMC Neurol. 2012, 12. [Google Scholar] [CrossRef]
- Socie, M.J.; Motl, R.W.; Sosnoff, J.J. Examination of spatiotemporal gait parameters during the 6-min walk in individuals with multiple sclerosis. Int. J. Rehabil. Res. 2014, 37, 311–316. [Google Scholar] [CrossRef]
- Plotnik, M.; Wagner, J.M.; Adusumilli, G.; Gottlieb, A.; Naismith, R.T. Gait asymmetry, and bilateral coordination of gait during a six-minute walk test in persons with multiple sclerosis. Sci. Rep. 2020, 10, 1–11. [Google Scholar] [CrossRef]
- Hadouiri, N.; Feuvrier, D.; Pauchot, J.; Decavel, P.; Sagawa, Y. Donor site morbidity after vascularized fibula free flap: Gait analysis during prolonged walk conditions. Int. J. Oral Maxillofac. Surg. 2018, 47, 309–315. [Google Scholar] [CrossRef]
- Beausoleil, S.; Miramand, L.; Turcot, K. Evolution of gait parameters in individuals with a lower-limb amputation during a six-minute walk test. Gait Posture 2019, 72, 40–45. [Google Scholar] [CrossRef]
- Givon, U.; Zeilig, G.; Achiron, A. Gait analysis in multiple sclerosis: Characterization of temporal–spatial parameters using GAITRite functional ambulation system. Gait Posture 2009, 29, 138–142. [Google Scholar] [CrossRef]
- Decavel, P.; Moulin, T.; Sagawa, Y. Gait tests in multiple sclerosis: Reliability and cut-off values. Gait Posture 2019, 67, 37–42. [Google Scholar] [CrossRef]
- Morrison, E.H.; Cooper, D.M.; White, L.J.; Larson, J.; Leu, S.-Y.; Zaldivar, F.; Ng, A.V. Ratings of Perceived Exertion During Aerobic Exercise in Multiple Sclerosis. Arch. Phys. Med. Rehabil. 2008, 89, 1570–1574. [Google Scholar] [CrossRef]
- Cleland, B.T.; Ingraham, B.A.; Pitluck, M.C.; Woo, D.; Ng, A.V. Reliability and Validity of Ratings of Perceived Exertion in Persons with Multiple Sclerosis. Arch. Phys. Med. Rehabil. 2016, 97, 974–982. [Google Scholar] [CrossRef]
- Guerra, E.; Di Cagno, A.; Mancini, P.; Sperandii, F.; Quaranta, F.; Ciminelli, E.; Fagnani, F.; Giombini, A.; Pigozzi, F. Physical fitness assessment in multiple sclerosis patients: A controlled study. Res. Dev. Disabil. 2014, 35, 2527–2533. [Google Scholar] [CrossRef]
- Angelini, L.; Hodgkinson, W.; Smith, C.; Dodd, J.M.; Sharrack, B.; Mazzà, C.; Paling, D. Wearable sensors can reliably quantify gait alterations associated with disability in people with progressive multiple sclerosis in a clinical setting. J. Neurol. 2020, 267, 2897–2909. [Google Scholar] [CrossRef] [PubMed]
- Thompson, A.J.; Banwell, B.L.; Barkhof, F.; Carroll, W.M.; Coetzee, T.; Comi, G.; Correale, J.; Fazekas, F.; Filippi, M.; Freedman, M.S.; et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol. 2018, 17, 162–173. [Google Scholar] [CrossRef]
- König, N.; Singh, N.; Von Beckerath, J.; Janke, L.; Taylor, W. Is gait variability reliable? An assessment of spatio-temporal parameters of gait variability during continuous overground walking. Gait Posture 2014, 39, 615–617. [Google Scholar] [CrossRef] [PubMed]
- Borg, G.A. Psychophysical bases of perceived exertion. Med. Sci. Sports Exerc. 1982, 14, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Walter, S.D.; Eliasziw, M.; Donner, A. Sample size and optimal designs for reliability studies. Stat. Med. 1998, 17, 101–110. [Google Scholar] [CrossRef]
- Kottner, J.; Audigé, L.; Brorson, S.; Donner, A.; Gajewski, B.J.; Hróbjartsson, A.; Roberts, C.; Shoukri, M.; Streiner, D.L. Guidelines for Reporting Reliability and Agreement Studies (GRRAS) were proposed. J. Clin. Epidemiol. 2011, 64, 96–106. [Google Scholar] [CrossRef]
- Atkinson, G.; Nevill, A.M. Statistical Methods for Assessing Measurement Error (Reliability) in Variables Relevant to Sports Medicine. Sports Med. 1998, 26, 217–238. [Google Scholar] [CrossRef]
- Langeskov-Christensen, D.; Feys, P.; Baert, I.; Riemenschneider, M.; Stenager, E.; Dalgas, U. Performed and perceived walking ability in relation to the Expanded Disability Status Scale in persons with multiple sclerosis. J. Neurol. Sci. 2017, 382, 131–136. [Google Scholar] [CrossRef]
- Vienne-Jumeau, A.; Quijoux, F.; Vidal, P.P.; Ricard, D. Value of gait analysis for measuring disease severity using inertial sensors in patients with multiple sclerosis: Protocol for a systematic review and meta-analysis. Syst. Rev. 2019, 8, 15. [Google Scholar] [CrossRef]
- Ngueleu, A.M.; Blanchette, A.K.; Maltais, D.; Moffet, H.; McFadyen, B.J.; Bouyer, L.; Batcho, C.S. Validity of Instrumented Insoles for Step Counting, Posture and Activity Recognition: A Systematic Review. Sensors 2019, 19, 2438. [Google Scholar] [CrossRef]
- Moumdjian, L.; Gervasoni, E.; Van Halewyck, F.; Eijnde, B.O.; Wens, I.; Van Geel, F.; Van Wijmeersch, B.; Feys, P.; Van Asch, P. Walking endurance and perceived symptom severity after a single maximal exercise test in persons with mild disability because of multiple sclerosis. Int. J. Rehabil. Res. 2018, 41, 316–322. [Google Scholar] [CrossRef]
- Pau, M.; Coghe, G.; Corona, F.; Marrosu, M.G.; Cocco, E. Effect of spasticity on kinematics of gait and muscular activation in people with Multiple Sclerosis. J. Neurol. Sci. 2015, 358, 339–344. [Google Scholar] [CrossRef]
- Liparoti, M.; della Corte, M.; Rucco, R.; Sorrentino, P.; Sparaco, M.; Capuano, R.; Minino, R.; Lavorgna, L.; Agosti, V.; Sorrentino, G.; et al. Gait abnormalities in minimally disabled people with Multiple Sclerosis: A 3D-motion analysis study. Mult. Scler. Relat. Disord. 2019, 29, 100–107. [Google Scholar] [CrossRef]
- Kervio, G.; Ville, N.; Carre, F. Le test de marche de 6 minutes chez le sujet sain: Reproductibilité et intensité relative. Sci. Sports 2003, 18, 40–42. [Google Scholar] [CrossRef]
| PwMS (n = 45) | Healthy Group (n = 24) | |
|---|---|---|
| Gender (female/male; N%) | 26/19; 57.8/42.2 | 14/10; 58.3/41.7 |
| Age (years) | 51.4 (12.3) | 51.3 (10.7) |
| Body mass (kg) | 75.5 (18.3) | 75.2 (13.7) |
| Height (m) | 1.7 (0.08) | 1.7 (0.08) |
| BMI (kg·m−2) | 26.2 (5.5) | 26.1 (3.9) |
| Tobacco exposition (y/n; N%) | 11/34; 24.4/75.6 | 5/19; 20.8/79.2 |
| Alcohol exposition (y/n; N%) | 4/41; 8.9/91.1 | 0/24; 0/100 |
| EDSS [4–6.5] | 4.5 [4–5] | NA NA |
| EDSS classes (N; %) | ||
| EDSS [4–5] | 28 (62.2%) | |
| EDSS [5–6] | 9 (20%) | |
| EDSS [6–7] | 8 (17.8%) | |
| Disease duration (years) | 17.4 (8.9) | NA |
| MS phenotype (N; %) | NA | |
| RR | 11 (24.4%) | |
| SP | 20 (44.4%) | |
| PP | 14 (31.1%) |
| PwMS (n = 45) | |||||||
|---|---|---|---|---|---|---|---|
| Variables | Test | 6MWT Intervals | ICC [95% CI] c | ||||
| Initial | Middle | End | Initial | Middle | End | ||
| Walking spatio-temporal a | |||||||
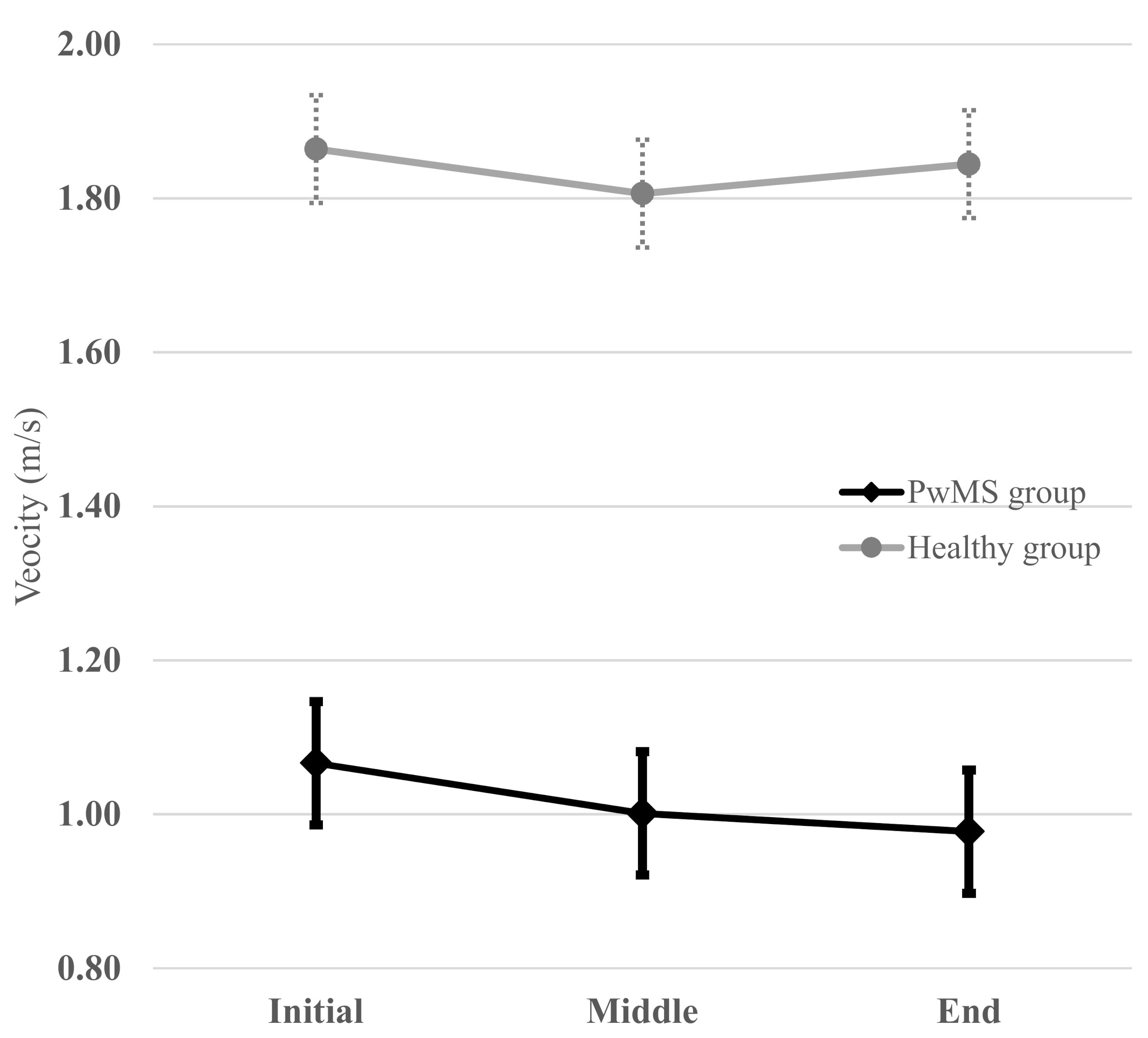
| Velocity (m/s) | Test | 1.03 (0.3) | 0.97 (0.3) | 0.96 (0.3) | 0.864 (0.698–0.933) | 0.879 (0.748–0.938) | 0.890 (0.810–0.938) |
| Retest | 1.10 (0.3) | 1.03 (0.3) | 0.99 (0.3) | ||||
| Cadence (step/min) | Test | 103.73 (13.5) | 99.64 (14.2) | 98.61 (14.4) | 0.874 (0.679–0.942) | 0.858 (0.696–0.929) | 0.873 (0.754–0.933) |
| Retest | 107.42 (13.4) | 103.18 (13.4) | 101.57 (13.4) | ||||
| Stride length (m) | Test | 1.18 (0.2) | 1.15 (0.2) | 1.16 (0.2) | 0.907 (0.812–0.952) | 0.919 (0.840–0.957) | 0.909 (0.841–0.949) |
| Retest | 1.22 (0.2) | 1.19 (0.2) | 1.16 (0.2) | ||||
| Stride width (m) | Test | 0.14 (0.04) | 0.16 (0.04) | 0.15 (0.04) | 0.919 (0.858–0.955) | 0.906 (0.835–0.947) | 0.846 (0.737–0.912) |
| Retest | 0.14 (0.04) | 0.15 (0.04) | 0.15 (0.04) | ||||
| Double support time (% GC) | Test | 32.83 (5.6) | 34.36 (6.1) | 34.75 (6.2) | 0.912 (0.836–0.952) | 0.889 (0.770–0.943) | 0.913 (0.840–0.952) |
| Retest | 32.19 (5.6) | 33.17 (5.8) | 33.91 (6.1) | ||||
| Borg scale (6–20) b | Test | 10 [9–11] | 12 [10–13] | 13 [12–15] | 0.322 (0.119–0.525) | 0.279 (0.105–0.454) | 0.376 (0.208–0.544) |
| Retest | 10 [9–10] | 11 [10–13] | 13 [12–14] | ||||
| 6MWT (m) a | Test | 349 (100) | 0.891 (0.791–0.942) | ||||
| Retest | 368 (102) | ||||||
| Healthy Group (n = 24) | |||||||
|---|---|---|---|---|---|---|---|
| Variables | Test | 6MWT intervals | ICC [95% CI] c | ||||
| Initial | Middle | End | Initial | Middle | End | ||
| Walking spatio-temporal a | |||||||
| Velocity (m/s) | Test | 1.81 (0.2) | 1.78 (0.2) | 1.80 (0.2) | 0.691 (0.232–0.874) | 0.775 (0.459–0.905) | 0.682 (0.290–0.862) |
| Retest | 1.94 (0.3) | 1.86 (0.3) | 1.92 (0.3) | ||||
| Cadence (step/min) | Test | 131.49 (8.3) | 129.48 (8.2) | 131.19 (10.1) | 0.624 (0.079–0.849) | 0.648 (0.180–0.852) | 0.616 (0.260–0.818) |
| Retest | 137.62 (11.7) | 134.84 (10.9) | 136.43 (12.6) | ||||
| Stride length (m) | Test | 1.65 (0.1) | 1.64 (0.1) | 1.64 (0.14) | 0.900 (0.728–0.960) | 0.946 (0.880–0.976) | 0.808 (0.551–0.918) |
| Retest | 1.68 (0.1) | 1.65 (0.1) | 1.68 (0.14) | ||||
| Stride width (m) | Test | 0.12 (0.02) | 0.12 (0.02) | 0.12 (0.02) | 0.896 (0.772–0.954) | 0.891 (0.767–0.951) | 0.767 (0.534–0.892) |
| Retest | 0.13 (0.02) | 0.12 (0.02) | 0.12 (0.03) | ||||
| Double support time (% GC) | Test | 23.12 (2.8) | 23.93 (2.9) | 23.56 (2.9) | 0.788 (0.527–0.907) | 0.788 (0.477–0.912) | 0.569 (0.201–0.791) |
| Retest | 22.25 (2.8) | 22.91 (2.8) | 21.84 (3.7) | ||||
| Borg scale (6–20) b | Test | 9 [6–10] | 9 [8–10] | 9 [8–10] | 0.788 (0.646–0.898) | 0.734 (0.543–0.925) | 0.786 (0.544–0.893) |
| Retest | 9 [6–9] | 9 [9–10] | 9 [9–10] | ||||
| 6MWT (m) a | Test | 630 (67) | 0.728 (0.350–0.886) | ||||
| Retest | 664 (88) | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hadouiri, N.; Monnet, E.; Gouelle, A.; Decavel, P.; Sagawa, Y. Evaluation of Prolonged Walking in Persons with Multiple Sclerosis: Reliability of the Spatio-Temporal Walking Variables during the 6-Minute Walk Test. Sensors 2021, 21, 3075. https://doi.org/10.3390/s21093075
Hadouiri N, Monnet E, Gouelle A, Decavel P, Sagawa Y. Evaluation of Prolonged Walking in Persons with Multiple Sclerosis: Reliability of the Spatio-Temporal Walking Variables during the 6-Minute Walk Test. Sensors. 2021; 21(9):3075. https://doi.org/10.3390/s21093075
Chicago/Turabian StyleHadouiri, Nawale, Elisabeth Monnet, Arnaud Gouelle, Pierre Decavel, and Yoshimasa Sagawa. 2021. "Evaluation of Prolonged Walking in Persons with Multiple Sclerosis: Reliability of the Spatio-Temporal Walking Variables during the 6-Minute Walk Test" Sensors 21, no. 9: 3075. https://doi.org/10.3390/s21093075
APA StyleHadouiri, N., Monnet, E., Gouelle, A., Decavel, P., & Sagawa, Y. (2021). Evaluation of Prolonged Walking in Persons with Multiple Sclerosis: Reliability of the Spatio-Temporal Walking Variables during the 6-Minute Walk Test. Sensors, 21(9), 3075. https://doi.org/10.3390/s21093075







