Step Length Is a Promising Progression Marker in Parkinson’s Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Individuals
2.2. Ethics
2.3. Quantitative Data
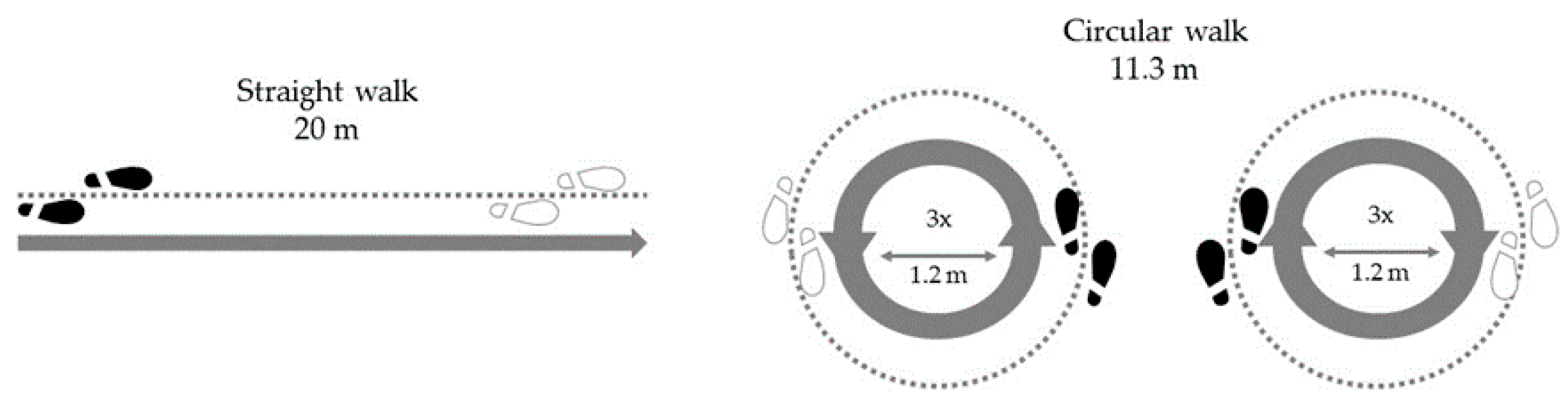
- Walking a 20 m track from a standing position on a wide (>2 m) corridor without obstacles [26]. The subjects walked at a self-selected walking speed. The exercise was completed when the participant had completely crossed the finish line.
- Circular walking around an area with diameter of 1.20 m three times at a self-selected speed, which corresponds to a walking distance of 11.3 m [7]. The start was carried out from a standing position initiated by the instructions of the examiner and the exercise was performed once clockwise (starting with the right leg) and once anticlockwise (starting with the left leg) [19].
2.4. Statistics
3. Results
3.1. Demographic and Clinical Parameters
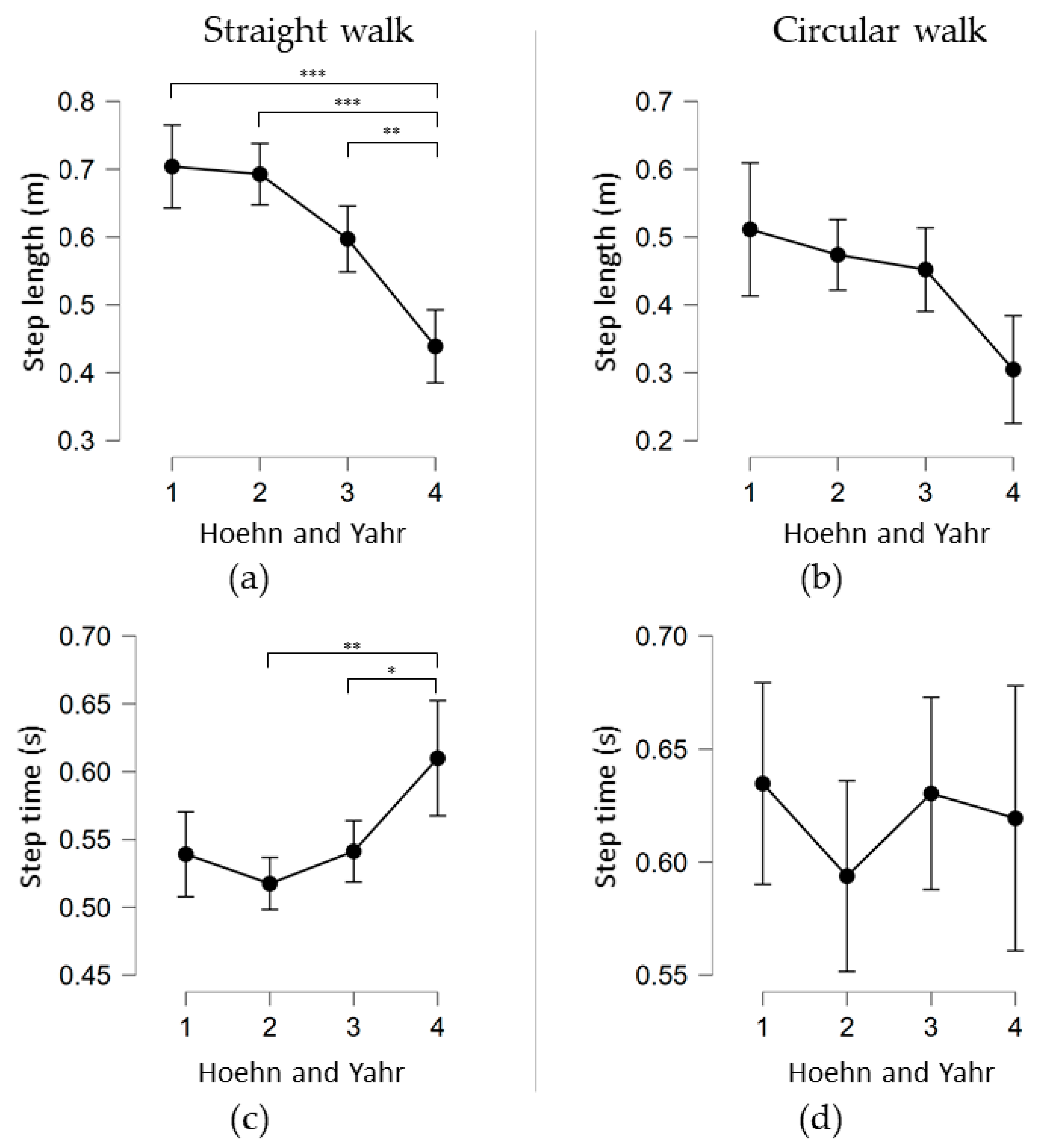
3.2. Straight Walk
3.3. Circular Walk
3.4. Total Task Time
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hatanaka, N.; Sato, K.; Hishikawa, N.; Takemoto, M.; Ohta, Y.; Yamashita, T.; Abe, K. Comparative Gait Analysis in Progressive Supranuclear Palsy and Parkinson’s Disease. Eur. Neurol. 2016, 75, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Goetz, C.G.; Tilley, B.C.; Shaftman, S.R.; Stebbins, G.T.; Fahn, S.; Martinez-Martin, P.; Poewe, W.; Sampaio, C.; Stern, M.B.; Dodel, R.; et al. Movement Disorder Society-Sponsored Revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): Scale presentation and clinimetric testing results. Mov. Disord. 2008, 23, 2129–2170. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Martin, P.; Skorvanek, M.; Rojo-Abuin, J.M.; Gregova, Z.; Stebbins, G.T.; Goetz, C.G. Validation study of the hoehn and yahr scale included in the MDS-UPDRS. Mov. Disord. 2018, 33, 651–652. [Google Scholar] [CrossRef]
- Toro, B.; Nester, C.; Farren, P. A review of observational gait assessment in clinical practice. Physiother. Theory Pract. 2003, 19, 137–149. [Google Scholar] [CrossRef]
- Curtze, C.; Nutt, J.G.; Carlson-Kuhta, P.; Mancini, M.; Horak, F.B. Levodopa Is a Double-Edged Sword for Balance and Gait in People With Parkinson’s Disease. Mov. Disord. 2015, 30, 1361–1370. [Google Scholar] [CrossRef]
- Schlachetzki, J.C.M.; Barth, J.; Marxreiter, F.; Gossler, J.; Kohl, Z.; Reinfelder, S.; Gassner, H.; Aminian, K.; Eskofier, B.M.; Winkler, J.; et al. Wearable sensors objectively measure gait parameters in Parkinson’s disease. PLoS ONE 2017, 12, e0183989. [Google Scholar] [CrossRef]
- Micó-Amigo, M.E.; Kingma, I.; Heinzel, S.; Rispens, S.M.; Heger, T.; Nussbaum, S.; van Lummel, R.C.; Berg, D.; Maetzler, W.; van Dieën, J.H. Potential Markers of Progression in Idiopathic Parkinson’s Disease Derived From Assessment of Circular Gait With a Single Body-Fixed-Sensor: A 5 Year Longitudinal Study. Front. Hum. Neurosci. 2019, 13, 59. [Google Scholar] [CrossRef]
- Hass, C.J.; Malczak, P.; Nocera, J.; Stegemöller, E.L.; Shukala, A.; Malaty, I.; Jacobson, C.E.; Okun, M.S.; McFarland, N. Quantitative Normative Gait Data in a Large Cohort of Ambulatory Persons with Parkinson’s Disease. PLoS ONE 2012, 7, e42337. [Google Scholar] [CrossRef]
- Wilson, J.; Alcock, L.; Yarnall, A.J.; Lord, S.; Lawson, R.A.; Morris, R.; Taylor, J.-P.; Burn, D.J.; Rochester, L.; Galna, B. Gait Progression Over 6 Years in Parkinson’s Disease: Effects of Age, Medication, and Pathology. Front. Aging Neurosci. 2020, 12, 320. [Google Scholar] [CrossRef]
- Herman, T.; Weiss, A.; Brozgol, M.; Giladi, N.; Hausdorff, J.M. Gait and balance in Parkinson’s disease subtypes: Objective measures and classification considerations. J. Neurol. 2014, 261, 2401–2410. [Google Scholar] [CrossRef] [PubMed]
- Bayle, N.; Patel, A.S.; Crisan, D.; Guo, L.J.; Hutin, E.; Weisz, D.J.; Moore, S.T.; Gracies, J.-M. Contribution of Step Length to Increase Walking and Turning Speed as a Marker of Parkinson’s Disease Progression. PLoS ONE 2016, 11, e0152469. [Google Scholar] [CrossRef]
- Del Din, S.; Elshehabi, M.; Galna, B.; Hobert, M.A.; Warmerdam, E.; Suenkel, U.; Brockmann, K.; Metzger, F.; Hansen, C.; Berg, D.; et al. Gait analysis with wearables predicts conversion to Parkinson disease. Ann. Neurol. 2019, 86, 357–367. [Google Scholar] [CrossRef]
- Ben Mansour, K.; Gorce, P.; Rezzoug, N. The Multifeature Gait Score: An accurate way to assess gait quality. PLoS ONE 2017, 12, e0185741. [Google Scholar] [CrossRef]
- van Schooten, K.S.; Pijnappels, M.; Lord, S.R.; van Dieën, J.H. Quality of Daily-Life Gait: Novel Outcome for Trials that Focus on Balance, Mobility, and Falls. Sensors 2019, 19, 4388. [Google Scholar] [CrossRef]
- Smulders, K.; Dale, M.L.; Carlson-Kuhta, P.; Nutt, J.G.; Horak, F.B. Pharmacological treatment in Parkinson’s disease: Effects on gait. Park. Relat. Disord. 2016, 31, 3–13. [Google Scholar] [CrossRef]
- Suppa, A.; Bologna, M.; Conte, A.; Berardelli, A.; Fabbrini, G. The effect of L-dopa in Parkinson’s disease as revealed by neurophysiological studies of motor and sensory functions. Expert Rev. Neurother. 2017, 17, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Warlop, T.; Detrembleur, C.; Buxes Lopez, M.; Stoquart, G.; Lejeune, T.; Jeanjean, A. Does Nordic Walking restore the temporal organization of gait variability in Parkinson’s disease? J. Neuroeng. Rehabil. 2017, 14, 17. [Google Scholar] [CrossRef]
- Kroneberg, D.; Elshehabi, M.; Meyer, A.-C.; Otte, K.; Doss, S.; Paul, F.; Nussbaum, S.; Berg, D.; Kühn, A.A.; Maetzler, W.; et al. Less Is More—Estimation of the Number of Strides Required to Assess Gait Variability in Spatially Confined Settings. Front. Aging Neurosci. 2019, 10, 435. [Google Scholar] [CrossRef]
- Heinzel, S.; Maechtel, M.; Hasmann, S.E.; Hobert, M.A.; Heger, T.; Berg, D.; Maetzler, W. Motor dual-tasking deficits predict falls in Parkinson’s disease: A prospective study. Parkinsonism Relat. Disord. 2016, 26, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Geritz, J.; Maetzold, S.; Steffen, M.; Pilotto, A.; Corrà, M.F.; Moscovich, M.; Rizzetti, M.C.; Borroni, B.; Padovani, A.; Alpes, A.; et al. Motor, cognitive and mobility deficits in 1000 geriatric patients: Protocol of a quantitative observational study before and after routine clinical geriatric treatment—The ComOn-study. BMC Geriatr. 2020, 20, 45. [Google Scholar] [CrossRef] [PubMed]
- Tomlinson, C.L.; Stowe, R.; Patel, S.; Rick, C.; Gray, R.; Clarke, C.E. Systematic review of levodopa dose equivalency reporting in Parkinson’s disease. Mov. Disord. 2010, 25, 2649–2653. [Google Scholar] [CrossRef] [PubMed]
- Nasreddine, Z.S.; Phillips, N.A.; Bãdirian, V.; Charbonneau, S.; Whitehead, V.; Collin, I.; Cummings, J.L.; Chertkow, H. The Montreal Cognitive Assessment, MoCA: A Brief Screening Tool For Mild Cognitive Impairment. J. Am. Geriatr. Soc. 2005, 53, 695–699. [Google Scholar] [CrossRef] [PubMed]
- Visser, M.; Leentjens, A.F.G.; Marinus, J.; Stiggelbout, A.M.; van Hilten, J.J. Reliability and validity of the Beck depression inventory in patients with Parkinson’s disease. Mov. Disord. 2006, 21, 668–672. [Google Scholar] [CrossRef]
- Heidenblut, S.; Zank, S. Entwicklung eines neuen Depressionsscreenings für den Einsatz in der Geriatrie: Die “Depression-im-Alter-Skala” (DIA-S). Z. Gerontol. Geriatr. 2010, 43, 170–176. [Google Scholar] [CrossRef]
- Donath, L.; Faude, O.; Lichtenstein, E.; Nuesch, C.; Mündermann, A. Validity and reliability of a portable gait analysis system for measuring spatiotemporal gait characteristics: Comparison to an instrumented treadmill. J. Neuroeng. Rehabil. 2016, 13. [Google Scholar] [CrossRef] [PubMed]
- Hobert, M.A.; Nussbaum, S.; Heger, T.; Berg, D.; Maetzler, W.; Heinzel, S. Progressive Gait Deficits in Parkinson’s Disease: A Wearable-Based Biannual 5-Year Prospective Study. Front. Aging Neurosci. 2019, 11, 22. [Google Scholar] [CrossRef] [PubMed]
- Pham, M.H.; Elshehabi, M.; Haertner, L.; Del Din, S.; Srulijes, K.; Heger, T.; Synofzik, M.; Hobert, M.A.; Faber, G.S.; Hansen, C.; et al. Validation of a Step Detection Algorithm during Straight Walking and Turning in Patients with Parkinson’s Disease and Older Adults Using an Inertial Measurement Unit at the Lower Back. Front. Neurol. 2017, 8, 457. [Google Scholar] [CrossRef] [PubMed]
- Maetzler, W.; Nieuwhof, F.; Hasmann, S.E.; Bloem, B.R. Emerging therapies for gait disability and balance impairment: Promises and pitfalls. Mov. Disord. 2013, 28, 1576–1586. [Google Scholar] [CrossRef]
| Author | Year | Cohort | H&Y Stage | Task | Main Findings |
|---|---|---|---|---|---|
| Herman et al. [10] | 2014 | 31 PwP-PIGD, 32 PwP-TD, cross-sectional | 1–4 | 30 m straight walk | Slower gait speed, shorter strides, less smoothness, and excessive instability in PwP-PIGD compared to PwP-TD. No association between step length and H&Y stages investigated. |
| Curtze et al. [5] | 2015 | 104 PwP cross-sectional | 2–4 | 7 m straight walk with turns | Levodopa intake improves arm swing range/velocity, gait velocity and stride length. No association between step length and H&Y stages investigated. |
| Hatanaka et al., [1] | 2016 | 124 PwP cross-sectional | 1–4 | 10 m straight walk | Decreased velocity and step length with increasing H&Y |
| Bayle et al. [11] | 2016 | 39 PwP cross-sectional | 2–4 | 5 m straight walk with turns | The contribution of step length and cadence to increased ambulation speed is age-invariant but a marker of PD. No association between step length and H&Y stages investigated. |
| Del Din et al. [12] | 2019 | 16 PD converter in a cohort of 696 healthy controls | - | 20 m straight walk | Higher step time variability and asymmetry of gait characteristics are associated with a shorter time to PD diagnosis. No association between step length and H&Y stages investigated. |
| Micò-Amigo et al. [7] | 2019 | 27 PwP longitudinal | 0–4 | 1.2 m circular walk (×3) | Number of steps, total task time, stride time variability, and stride regularity are potential PD progression markers. No association between step length and H&Y stages investigated. |
| Wilson et al. [9] | 2020 | 109 PwP longitudinal, 130 HC | 1–3 | 25 m oval circuit | Increased variability of swing time, step time and step width, and reduced swing time asymmetry specific to PD when compared to healthy older adults. No association between step length and H&Y stages investigated. |
| H&Y Stage | 1 | 2 | 3 | 4 |
|---|---|---|---|---|
| N (female) | 11 (5) | 26 (12) | 17 (9) | 14 (8) |
| Age (years) | 62 ± 10 | 62 ± 9 | 69 ± 5 | 76 ± 6 |
| Height (m) | 1.74 ± 0.11 | 1.74 ± 0.08 | 1.70 ± 0.10 | 1.70 ± 0.10 |
| MDS-UPDRS-III (0–132) | 13 ± 7 | 29 ± 11 | 34 ± 14 | 30 ± 13 |
| LEDD (mg/d) | 301 ± 187 | 368 ± 201 | 416 ± 282 | 608 ± 320 |
| On/Off (%) | 37/63 | 32/68 | 44/56 | 92/8 |
| MoCA (0–30) | 25.8 ± 3.0 | 26.3 ± 3.1 | 25.1 ± 3.5 | 23.7 ± 3.1 |
| PIGD (0–16) | 1.3 ± 1.0 | 2.4 ± 2.6 | 5.4 ± 1.9 | 8.5 ± 2.0 |
| Freezing (%) | 0 | 0 | 18 | 54 |
| Depressive symptoms (%) | 20 | 28 | 20 | 27 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Welzel, J.; Wendtland, D.; Warmerdam, E.; Romijnders, R.; Elshehabi, M.; Geritz, J.; Berg, D.; Hansen, C.; Maetzler, W. Step Length Is a Promising Progression Marker in Parkinson’s Disease. Sensors 2021, 21, 2292. https://doi.org/10.3390/s21072292
Welzel J, Wendtland D, Warmerdam E, Romijnders R, Elshehabi M, Geritz J, Berg D, Hansen C, Maetzler W. Step Length Is a Promising Progression Marker in Parkinson’s Disease. Sensors. 2021; 21(7):2292. https://doi.org/10.3390/s21072292
Chicago/Turabian StyleWelzel, Julius, David Wendtland, Elke Warmerdam, Robbin Romijnders, Morad Elshehabi, Johanna Geritz, Daniela Berg, Clint Hansen, and Walter Maetzler. 2021. "Step Length Is a Promising Progression Marker in Parkinson’s Disease" Sensors 21, no. 7: 2292. https://doi.org/10.3390/s21072292
APA StyleWelzel, J., Wendtland, D., Warmerdam, E., Romijnders, R., Elshehabi, M., Geritz, J., Berg, D., Hansen, C., & Maetzler, W. (2021). Step Length Is a Promising Progression Marker in Parkinson’s Disease. Sensors, 21(7), 2292. https://doi.org/10.3390/s21072292







