Comparison of the Performance of the Leap Motion ControllerTM with a Standard Marker-Based Motion Capture System
Abstract
1. Introduction
2. Materials and Methods
2.1. Subject Information
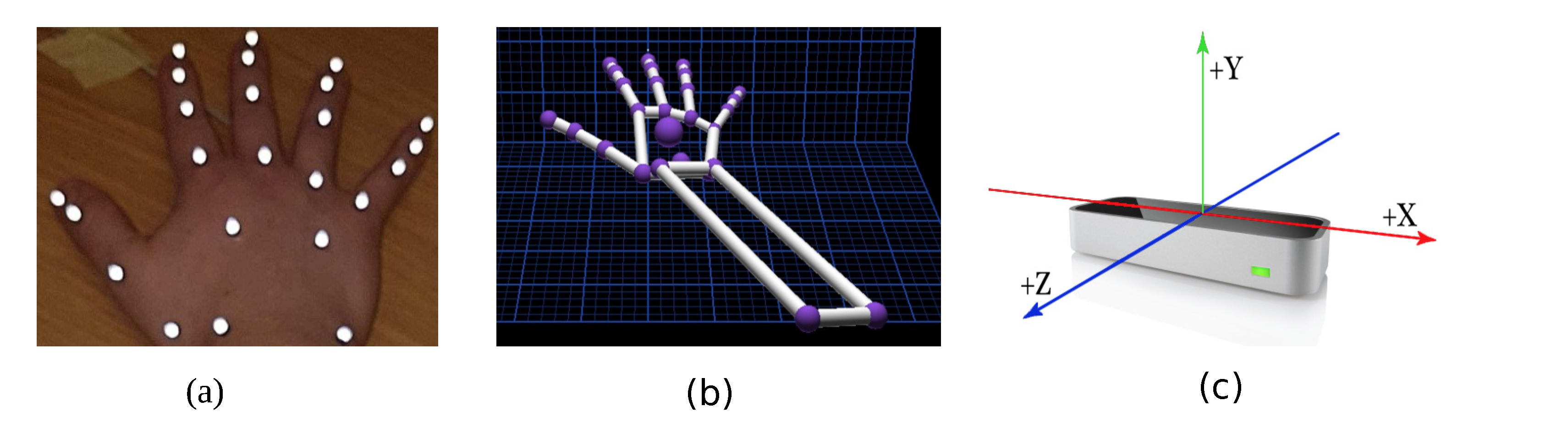
2.2. Marker-Based Motion Capture Technology
2.3. Leap Motion Controller™
2.4. Data Collection
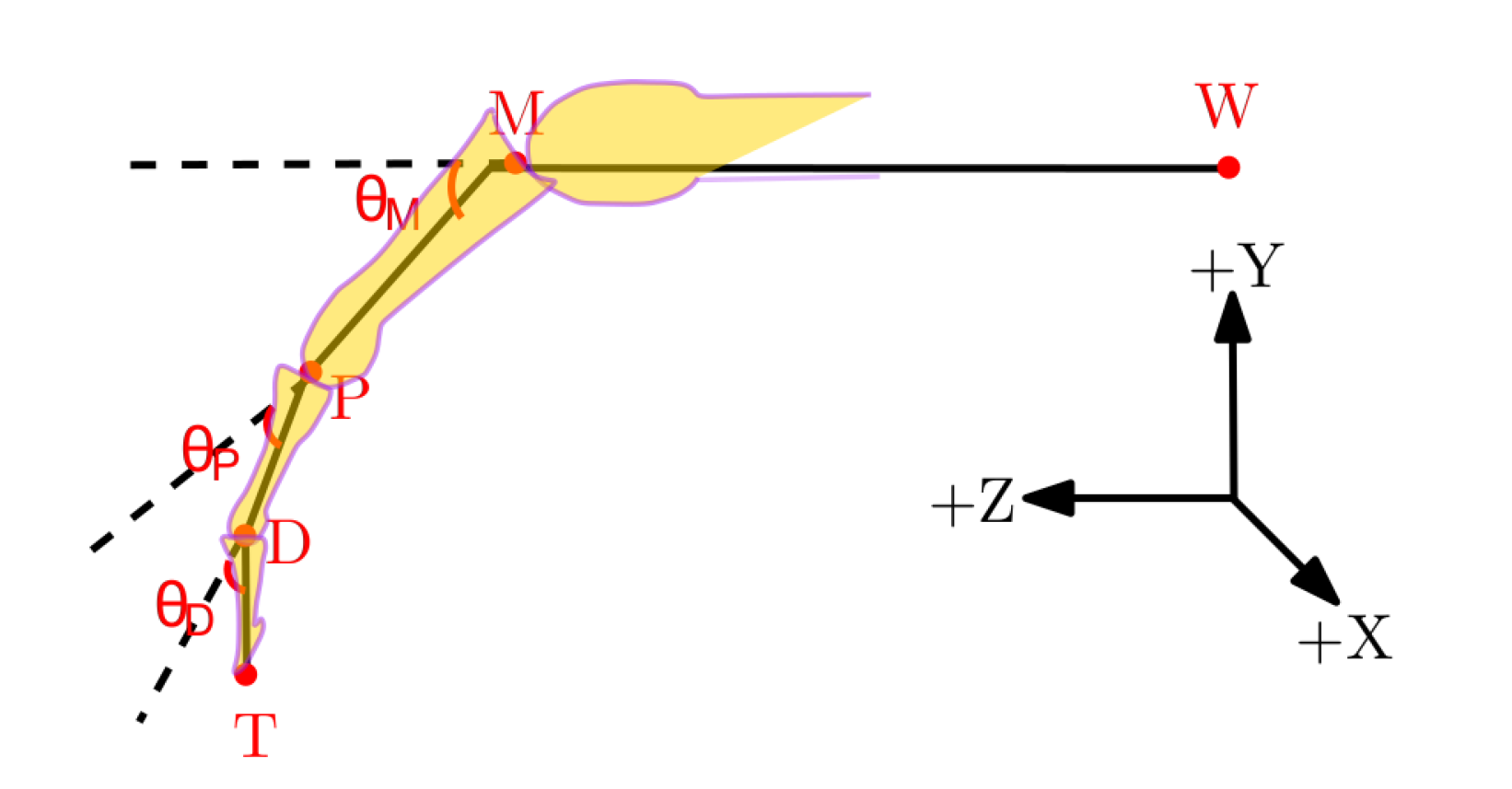
2.5. Data Processing
2.6. Data Analysis
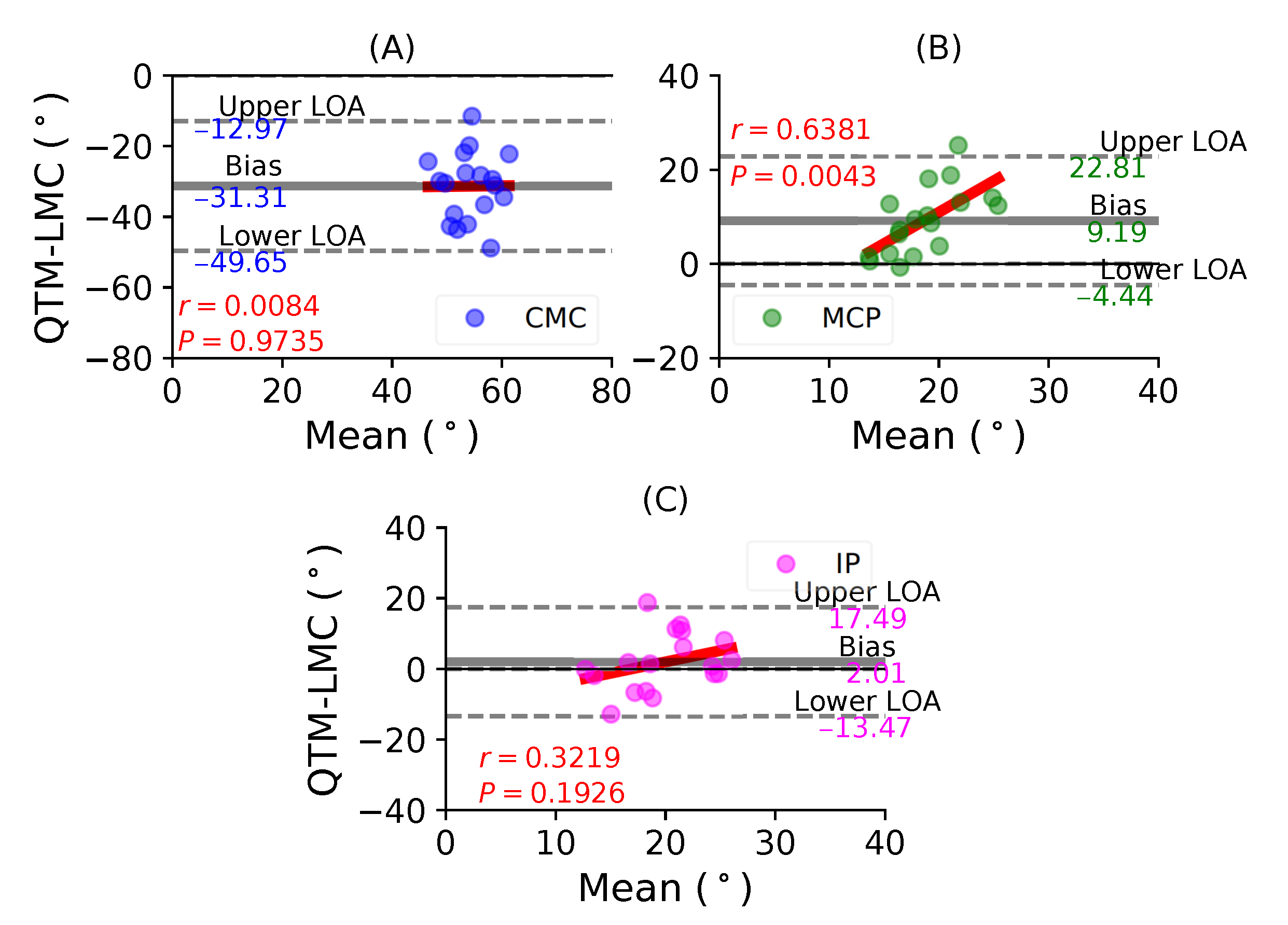
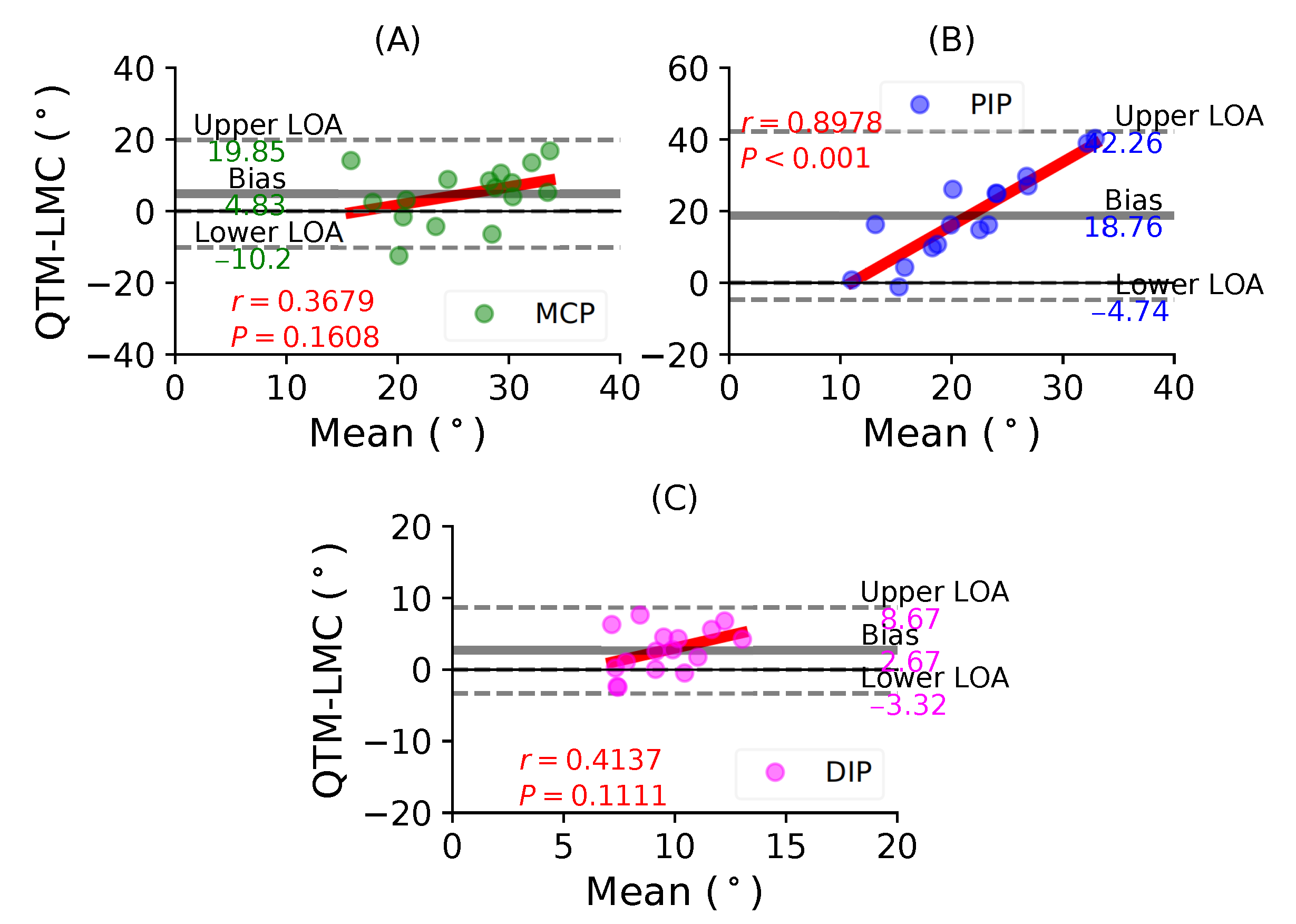
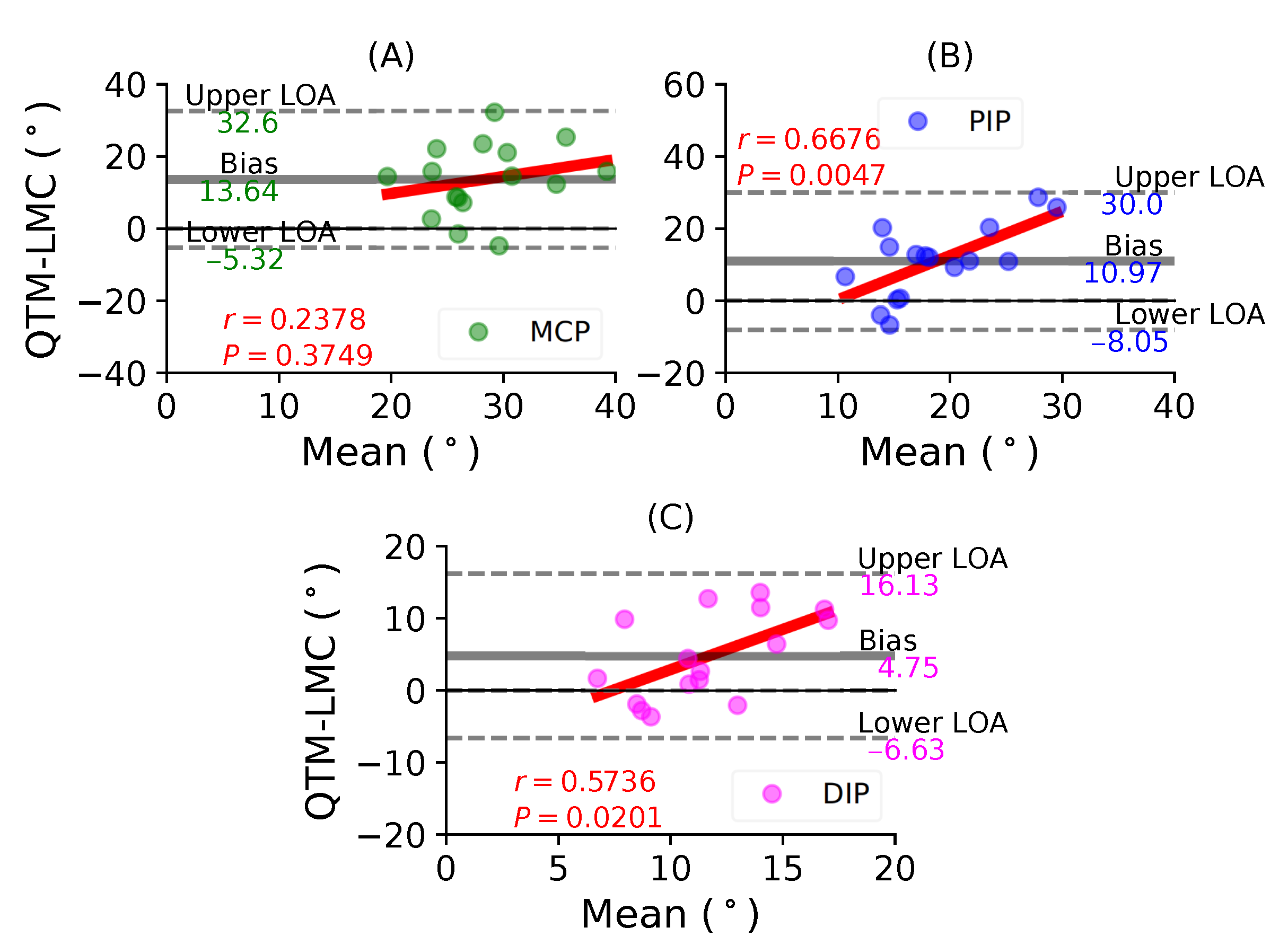
3. Results
Initial Analysis
- (1)
- The time recorded for the trials by the two systems varied. This shows the need to compare dynamic trials using techniques that will handle such time dependent data. In this paper, we used time normalisation to compare flexion trials between the LMC and QTM.
- (2)
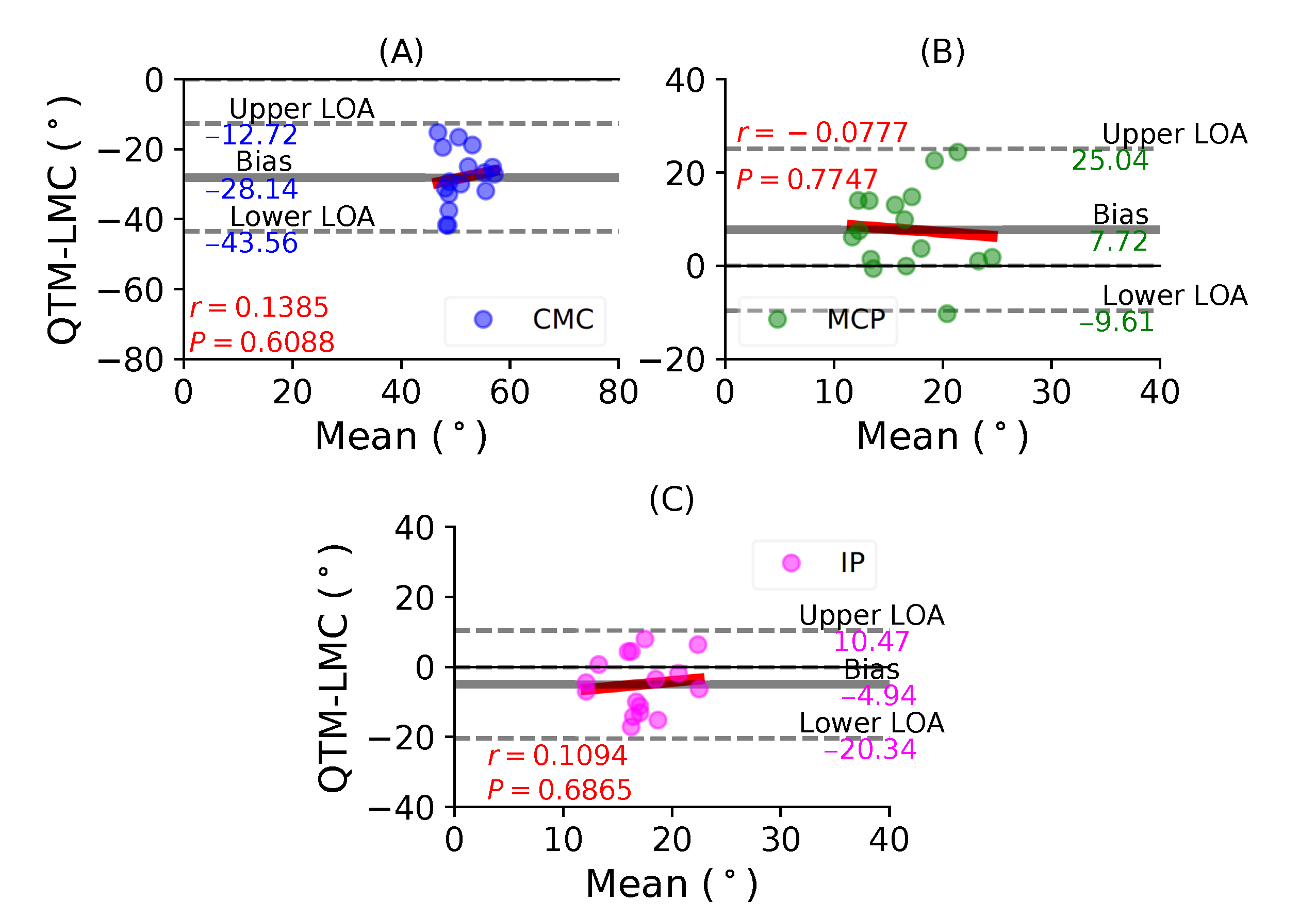
- Table 2 shows that the ROM calculation for each joint during the flexion trials was quite varied, and this information is not enough to conclusively say that LMC performed better or worse than the QTM system. Therefore, subsequent results compared the LMC and QTM with the help of BA plots.
4. Discussion
4.1. Why Is a Trend Observed between Two Measurement Techniques?
4.2. Repeatability
4.3. Clinical Recommendation
4.4. Study Limitations
4.5. Further Research
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| LMC | Leap Motion Controller™ |
| QTM | Qualisys Track Manager |
| BA | Bland–Altman |
| MCP | Metacarpophalangeal |
| PIP | Proximal interphalangeal joint |
| DIP | Distal interphalangeal joint |
| CMC | Carpometacarpal phalangeal joint |
| IP | Interphalangeal joint |
| W | Midpoint of the wrist joint |
| M | Midpoint of MCP joint |
| P | Midpoint PIP joint |
| D | Midpoint of DIP joint |
| T | Tip of the finger |
| ROM | Range of motion |
| CI | Confidence interval |
| LOA | Limits of agreement |
References
- Cheng, L.L.; Liu, H.B. Examples of Quadrocopter control on ROS. In Proceedings of the 2015 IEEE 9th International Conference on Anti-counterfeiting, Security, and Identification (ASID), Xiamen, China, 25–27 September 2015; pp. 92–96. [Google Scholar]
- Sarkar, A.; Patel, K.A.; Ram, R.G.; Capoor, G.K. Gesture control of drone using a motion controller. In Proceedings of the 2016 International Conference on Industrial Informatics and Computer Systems (CIICS), Dubai, United Arab Emirates, 13–15 March 2016; pp. 1–5. [Google Scholar]
- Fernandez, R.A.; Sanchez-Lopez, J.L.; Sampedro, C.; Bavle, H.; Molina, M.; Campoy, P. Natural user interfaces for human-drone multi-modal interaction. In Proceedings of the 2016 International Conference on Unmanned Aircraft Systems (ICUAS), Arlington, VA, USA, 7–10 June 2016; pp. 1013–1022. [Google Scholar]
- Partridge, R.W.; Brown, F.S.; Brennan, P.M.; Hennessey, I.A.; Hughes, M.A. The LEAPTM gesture interface device and take-home laparoscopic simulators: A study of construct and concurrent validity. Surg. Innov. 2016, 23, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Carrieri, M.; Petracca, A.; Lancia, S.; Basso Moro, S.; Brigadoi, S.; Spezialetti, M.; Ferrari, M.; Placidi, G.; Quaresima, V. Prefrontal cortex activation upon a demanding virtual hand-controlled task: A new frontier for neuroergonomics. Front. Hum. Neurosci. 2016, 10, 53. [Google Scholar] [CrossRef] [PubMed]
- Naidu, C.; Ghotkar, A. Hand gesture recognition using leap motion controller. Int. J. Sci. Res. (IJSR) ISSN (Online) 2016, 5, 2319–7064. [Google Scholar]
- Kim, S.; Park, S.; Lee, O. Development of a Diagnosis and Evaluation System for Hemiplegic Patients Post-Stroke Based on Motion Recognition Tracking and Analysis of Wrist Joint Kinematics. Sensors 2020, 20, 4548. [Google Scholar] [CrossRef]
- Fernández-González, P.; Carratalá-Tejada, M.; Monge-Pereira, E.; Collado-Vázquez, S.; Baeza, P.S.H.; Cuesta-Gómez, A.; Cano-de la Cuerda, R. Leap motion controlled video game-based therapy for upper limb rehabilitation in patients with Parkinson’s disease: A feasibility study. J. Neuroeng. Rehabil. 2019, 16, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bostanci, H.; Emir, A.; Tarakci, D.; Tarakci, E. Video game-based therapy for the non-dominant hand improves manual skills and grip strength. Hand Surg. Rehabil. 2019, 39, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Tarakci, E.; Arman, N.; Tarakci, D.; Kasapcopur, O. Leap Motion Controller–based training for upper extremity rehabilitation in children and adolescents with physical disabilities: A randomized controlled trial. J. Hand Ther. 2020, 33, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Kincaid, C.J.; Vaterlaus, A.C.; Stanford, N.R.; Charles, S.K. Frequency response of the leap motion controller and its suitability for measuring tremor. Med. Eng. Phys. 2019, 63, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Weichert, F.; Bachmann, D.; Rudak, B.; Fisseler, D. Analysis of the accuracy and robustness of the leap motion controller. Sensors 2013, 13, 6380–6393. [Google Scholar] [CrossRef] [PubMed]
- Bachmann, D.; Weichert, F.; Rinkenauer, G. Evaluation of the leap motion controller as a new contact-free pointing device. Sensors 2015, 15, 214–233. [Google Scholar] [CrossRef]
- Smeragliuolo, A.H.; Hill, N.J.; Disla, L.; Putrino, D. Validation of the Leap Motion Controller using markered motion capture technology. J. Biomech. 2016, 49, 1742–1750. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, P.; Johnson, P.W. Comparison of measurement accuracy between two types of wrist goniometer systems. Appl. Ergon. 2001, 32, 599–607. [Google Scholar] [CrossRef]
- Carter, T.I.; Pansy, B.; Wolff, A.L.; Hillstrom, H.J.; Backus, S.I.; Lenhoff, M.; Wolfe, S.W. Accuracy and reliability of three different techniques for manual goniometry for wrist motion: A cadaveric study. J. Hand Surg. 2009, 34, 1422–1428. [Google Scholar] [CrossRef] [PubMed]
- Niechwiej-Szwedo, E.; Gonzalez, D.; Nouredanesh, M.; Tung, J. Evaluation of the Leap Motion Controller during the performance of visually-guided upper limb movements. PLoS ONE 2018, 13, e0193639. [Google Scholar] [CrossRef] [PubMed]
- Tung, J.Y.; Lulic, T.; Gonzalez, D.A.; Tran, J.; Dickerson, C.R.; Roy, E.A. Evaluation of a portable markerless finger position capture device: Accuracy of the Leap Motion controller in healthy adults. Physiol. Meas. 2015, 36, 1025. [Google Scholar] [CrossRef] [PubMed]
- Trejo, R.L.; Vázquez, J.P.; Ramirez, M.L.; Corral, L.E.; Marquez, I.R. Hand goniometric measurements using leap motion. In Proceedings of the 2017 14th IEEE Annual Consumer Communications and Networking Conference (CCNC), Las Vegas, NV, USA, 8–11 January 2017; pp. 137–141. [Google Scholar]
- Nizamis, K.; Rijken, N.H.; Mendes, A.; Janssen, M.M.; Bergsma, A.; Koopman, B.F. A Novel Setup and Protocol to Measure the Range of Motion of the Wrist and the Hand. Sensors 2018, 18, 3230. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, C.D.; Notley, S.V.; Chappell, P.H.; Burridge, J.H.; Yule, V.T. Validation and application of a computational model for wrist and hand movements using surface markers. IEEE Trans. Biomed. Eng. 2008, 55, 1199–1210. [Google Scholar] [CrossRef]
- Leap Motion Controller Data Sheet. Available online: https://www.ultraleap.com/product/leap-motion-controller (accessed on 9 November 2020).
- Guzsvinecz, T.; Szucs, V.; Sik-Lanyi, C. Suitability of the Kinect sensor and Leap Motion controller—A literature review. Sensors 2019, 19, 1072. [Google Scholar] [CrossRef]
- Guna, J.; Jakus, G.; Pogačnik, M.; Tomažič, S.; Sodnik, J. An analysis of the precision and reliability of the leap motion sensor and its suitability for static and dynamic tracking. Sensors 2014, 14, 3702–3720. [Google Scholar] [CrossRef]
- Cook, J.R.; Baker, N.A.; Cham, R.; Hale, E.; Redfern, M.S. Measurements of wrist and finger postures: A comparison of goniometric and motion capture techniques. J. Appl. Biomech. 2007, 23, 70–78. [Google Scholar] [CrossRef] [PubMed]
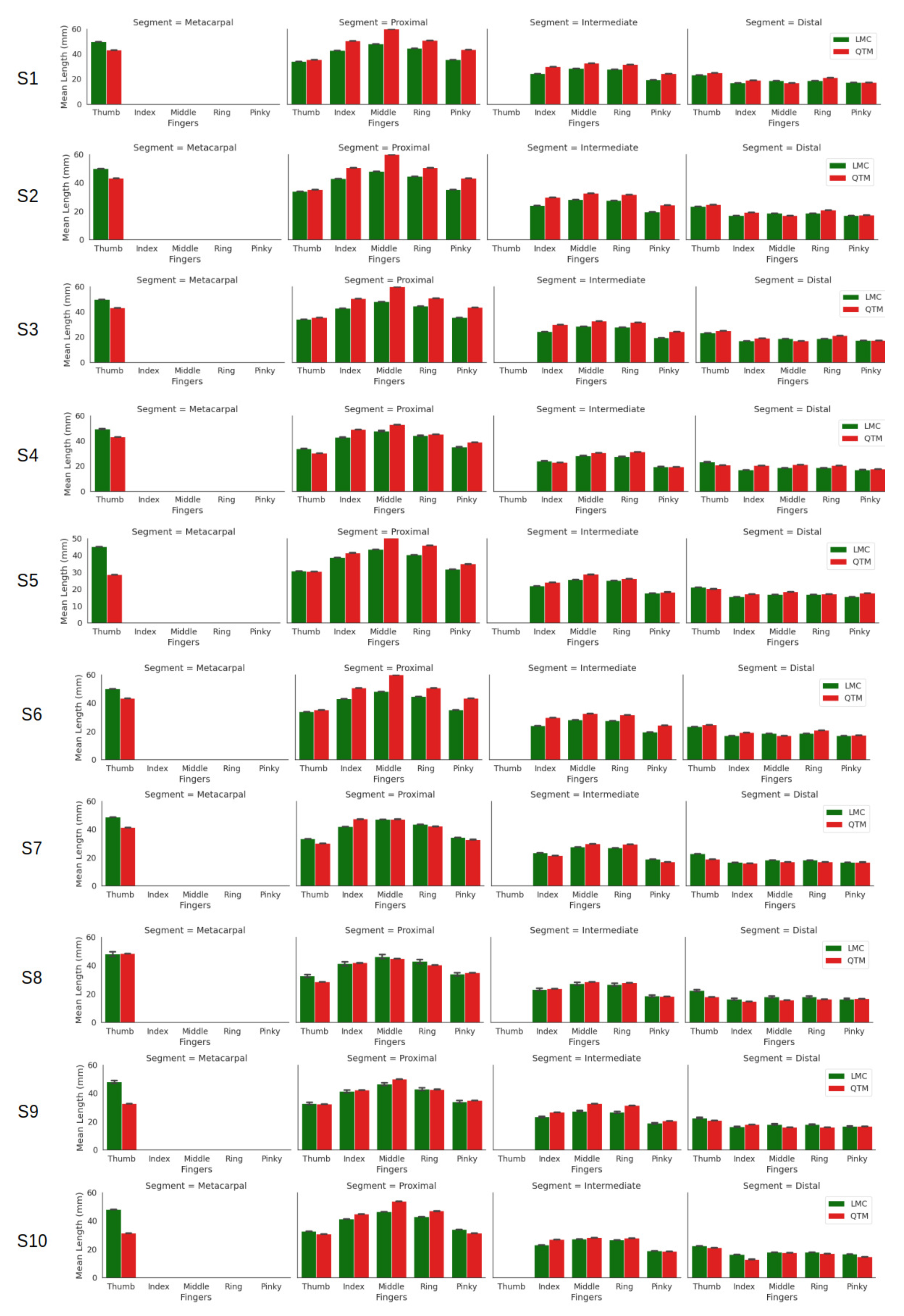



| Fingers | Segment | LMC (mm) | QTM (mm) |
|---|---|---|---|
| Thumb | Distal | 22.68 ± 10.61 | 19.27 ± 10.52 |
| Proximal | 33.06 ± 10.70 | 32.32 ± 11.65 | |
| Metacarpal | 48.40 ± 12.41 | 38.12 ± 11.15 | |
| Index | Distal | 18.99 ± 10.25 | 17.08 ± 9.72 |
| Intermediate | 23.42 ± 10.69 | 25.73 ± 12.14 | |
| Proximal | 39.17 ± 12.86 | 44.89 ± 13.19 | |
| Middle | Distal | 18.21 ± 12.08 | 17.71 ± 11.57 |
| Intermediate | 27.58 ± 11.51 | 30.21 ± 12.07 | |
| Proximal | 46.46 ± 13.24 | 50.59 ± 13.07 | |
| Ring | Distal | 18.10 ± 10.84 | 17.40 ± 10.75 |
| Intermediate | 26.87 ± 10.56 | 28.95 ± 10.94 | |
| Proximal | 43.33 ± 11.59 | 44.90 ± 10.90 | |
| Pinky | Distal | 16.67 ± 8.50 | 16.69 ± 8.65 |
| Intermediate | 18.96 ± 8.41 | 19.58 ± 7.96 | |
| Proximal | 34.28 ± 2.76 | 36.31 ± 3.58 |
| Fingers | Joint | LMC () | QTM () | QTM-LMC () |
|---|---|---|---|---|
| Thumb Abduction | IP | 31.26 | 37.75 | 6.49 |
| MCP | 41.38 | 49.65 | 8.27 | |
| CMC | 54.10 | 42.44 | –11.66 | |
| Thumb Flexion | IP | 43.86 | 72.15 | 28.29 |
| MCP | 43.16 | 62.47 | 19.31 | |
| CMC | 53.66 | 45.73 | –7.93 | |
| Index | DIP | 68.78 | 68.31 | –0.47 |
| PIP | 81.09 | 81.34 | 0.25 | |
| MCP | 107.86 | 86.66 | –21.2 | |
| Middle | DIP | 33.80 | 21.38 | –12.42 |
| PIP | 43.59 | 105.10 | 61.51 | |
| MCP | 72.36 | 92.64 | 20.28 | |
| Ring | DIP | 47.49 | 19.86 | –27.63 |
| PIP | 69.55 | 103.94 | 34.39 | |
| MCP | 78.89 | 83.39 | 4.5 | |
| Pinky | DIP | 41.16 | 50.32 | 9.16 |
| PIP | 57.35 | 94.68 | 37.33 | |
| MCP | 78.08 | 95.61 | 17.53 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ganguly, A.; Rashidi, G.; Mombaur, K. Comparison of the Performance of the Leap Motion ControllerTM with a Standard Marker-Based Motion Capture System. Sensors 2021, 21, 1750. https://doi.org/10.3390/s21051750
Ganguly A, Rashidi G, Mombaur K. Comparison of the Performance of the Leap Motion ControllerTM with a Standard Marker-Based Motion Capture System. Sensors. 2021; 21(5):1750. https://doi.org/10.3390/s21051750
Chicago/Turabian StyleGanguly, Amartya, Gabriel Rashidi, and Katja Mombaur. 2021. "Comparison of the Performance of the Leap Motion ControllerTM with a Standard Marker-Based Motion Capture System" Sensors 21, no. 5: 1750. https://doi.org/10.3390/s21051750
APA StyleGanguly, A., Rashidi, G., & Mombaur, K. (2021). Comparison of the Performance of the Leap Motion ControllerTM with a Standard Marker-Based Motion Capture System. Sensors, 21(5), 1750. https://doi.org/10.3390/s21051750






