Construction of a Ginseng Root-Meristem Sensor and a Sensing Kinetics Study on the Main Nitrogen Nutrients
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Instruments and Equipment
2.3. Pre-Treatment of Glassy Carbon Electrodes
2.4. Preparation of a Ginseng Root-Meristem Sensor
2.5. Measurement of the Ginseng Root-Meristem Sensor
3. Results and Discussion
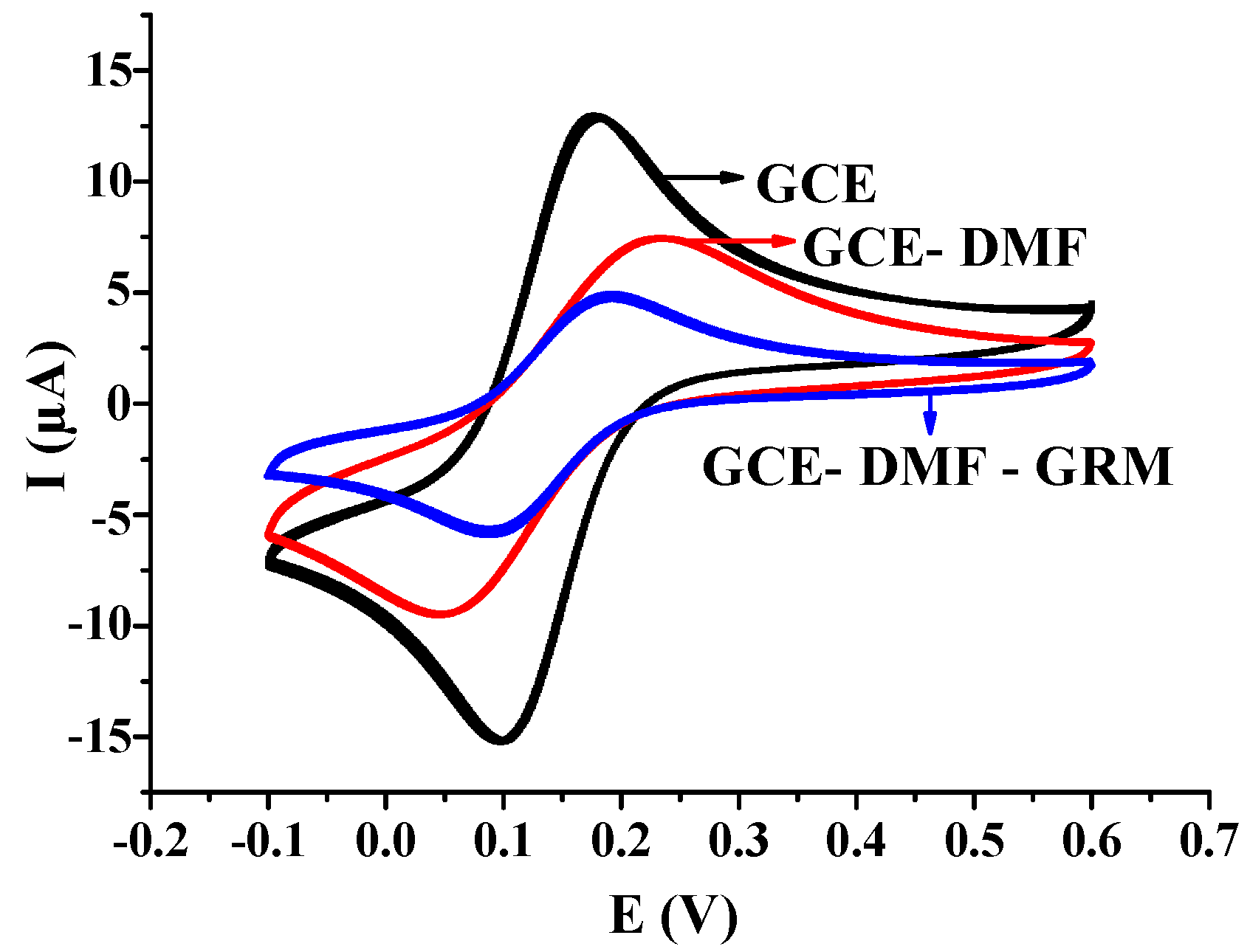
3.1. Electrode Pre-Treatment and Its Characterization
3.2. Assembly and Characterization of Sensitive Elements of the Ginseng Root Meristem
3.3. Potential Optimization of the Current–Time Measurement Method
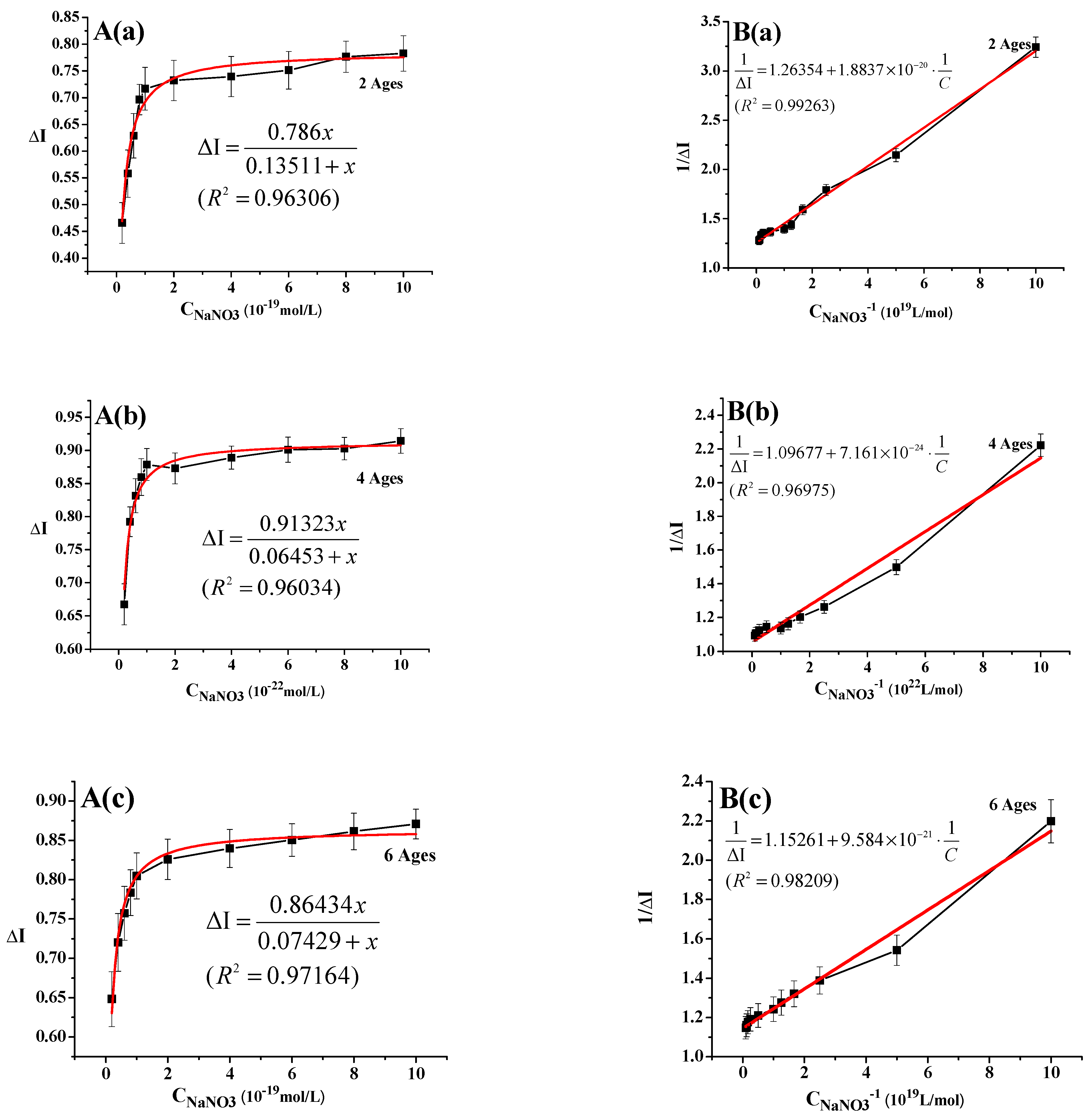
3.4. Determination of Five Types of Nitrogen Source Sensing Parameters by Ginseng Root-Meristem Sensors of Different Ages
3.5. Reproducibility and Stability of Ginseng Root-Meristem Sensors
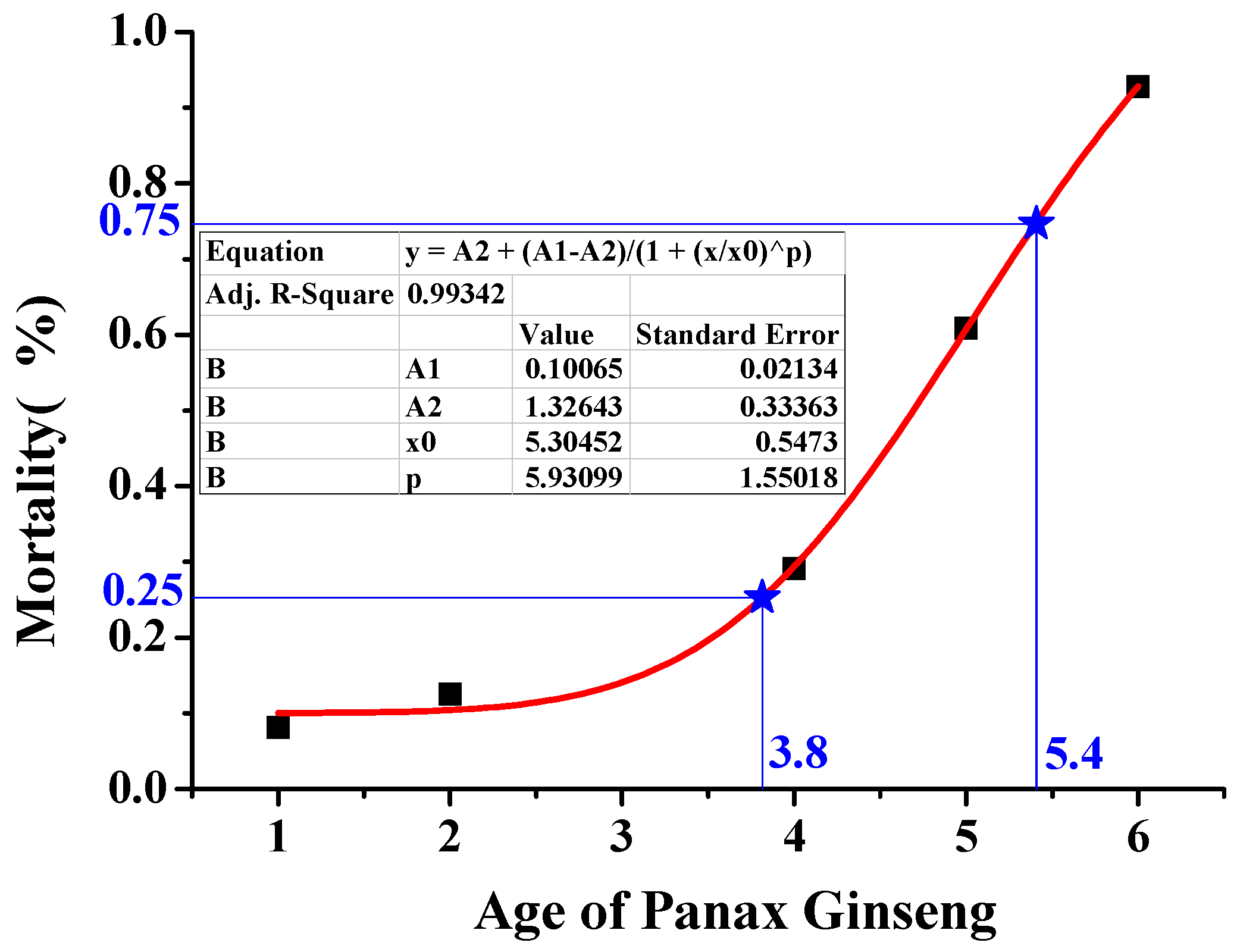
3.6. Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- ChapinIII, F.S.; Matson, P.A.; Vitousek, P.M.; Mooney, H.A. Carbon Input to Terrestrial Ecosystems. In Principles of Terrestrial Ecosystem Ecology, 2nd ed.; Springer-Verlag New York, Inc.: New York, NY, USA, 2012; pp. 110–112. [Google Scholar]
- Wei, L.; Qiao, L.; Pang, G.; Xie, J. A kinetic study of bitter taste receptor sensing using immobilized porcine taste bud tissues. Biosens. Bioelectron. 2017, 92, 74–80. [Google Scholar] [CrossRef]
- Pang, G.; Chen, Q.; Hu, Z.; Xie, J. Advances in Research on Taste Receptors and Application Prospects of Taste Sensors. Available online: http://www.cnki.com.cn/Article/CJFDTotal-SPKX201705051.htm (accessed on 1 January 2021).
- Lu, D.; Xu, Q.; Pang, G.; Lu, F. A novel electrochemical immunosensor based on Au nanoparticles and horseradish peroxidase signal amplification for ultrasensitive detection of a-fetoprotein. Biomed. Microdevices 2018, 20, 46.1–46.11. [Google Scholar] [CrossRef]
- Wei, L.; Wang, X.; Lu, D.; Li, Y.; Pang, G.; Xie, J. A novel staphylococcal enterotoxin q immunosensor prepared with self-assembly method based on horseradish peroxidase and double-layer gold nanoparticles. Food Anal. Methods 2017, 10, 892–899. [Google Scholar] [CrossRef]
- Lu, D.; Lu, F.; Pang, G. A Novel Tetrahydrocannabinol Electrochemical Nano Immunosensor Based on Horseradish Peroxidase and Double-Layer Gold Nanoparticles. Molecules 2016, 21, 1377. [Google Scholar] [CrossRef]
- Lu, D.; Pang, G.; Xie, J. A new phosphothreonine lyase electrochemical immunosensor for detecting Salmonella based on horseradish peroxidase/GNPs-thionine/chitosan. Biomed. Microdevices 2017, 19, 12. [Google Scholar] [CrossRef]
- Barre, A.; Hervé, C.; Lescure, B.; Rougé, P. Lectin Receptor Kinases in Plants. Crit. Rev. Plant Sci. 2002, 21, 379–399. [Google Scholar] [CrossRef]
- Wu, Q.; Michael, R.; Hiro, F.; David, J.; Nathan, M.S. Role of heterotrimeric Gα proteins in maize development and enhancement of agronomic traits. PLoS Genet. 2018, 14, e1007374. [Google Scholar] [CrossRef]
- Cao, Z.; Jin, H.; Xu, Y.; Song, X.; Lou, Z.; Cao, L.; Pan, X.; Zhang, D. Continuous Growing Ginseng in Aged Ginseng Field Experimental Report. J. Chin. Med. Mater. 2004, 27, 554–555. [Google Scholar]
- Shi, K.; Shiu, K.K. Scanning tunneling microscopic and voltammetric studies of the surface structures of an electrochemically activated glassy carbon electrode. Anal. Chem. 2002, 74, 879–885. [Google Scholar] [CrossRef]
- Gombotz, W.R.; Wee, S.F. Protein release from alginate matrices. Adv. Drug Deliv. Rev. 2012, 64, 194–205. [Google Scholar] [CrossRef]
- Zactiti, E.M.; Kieckbusch, T.G. Release of Potassium Sorbate from Active Films of Sodium Alginate Crosslinked with Calcium Chloride. Packag. Technol. Sci. 2009, 22, 349–358. [Google Scholar] [CrossRef]
- Reiss, M.; Heibges, A.; Metzger, J.; Hartmeier, W. Determination of BOD-values of starch-containing waste water by a BOD-biosensor. Biosens. Bioelectron. 1998, 13, 1083–1090. [Google Scholar] [CrossRef]
- Bierhalz, A.C.K.; Silva, M.A.D.; Braga, M.E.M.; Sousa, H.J.C.; Kieckbusch, T.G. Effect of calcium and/or barium crosslinking on the physical and antimicrobial properties of natamycin-loaded alginate films. LWT-Food Sci. Technol. 2014, 57, 494–501. [Google Scholar] [CrossRef]
- Rhim, J. Physical and mechanical properties of water resistant sodium alginate films. LWT Food Sci. Technol. 2004, 37, 323–330. [Google Scholar] [CrossRef]
- Huang, Y.; Lu, D.; Liu, H.; Liu, S.; Jiang, S.; Pang, G.; Liu, Y. Preliminary research on the receptor–ligand recognition mechanism of umami by an hT1R1 biosensor. Food Funct. 2019, 10, 1280–1287. [Google Scholar] [CrossRef]
- Nagaoka, T.; Yoshino, T. Surface properties of electrochemically pretreated glassy carbon. Anal. Chem. 1986, 58, 1037–1042. [Google Scholar] [CrossRef]
- Liu, K.H.; Tsay, Y.F. Switching between the two action modes of the dual-affinity nitrate transporter CHL1 by phosphorylation. EMBO J. 2003, 22, 1005–1013. [Google Scholar] [CrossRef]
- Filleur, S.; Dorbe, M.F.; Cerezo, M.; Orsel, M.; Granier, F.; Gojon, A.; Daniel-Vedele, F. An Arabidopsis T-DNA mutant affected in Nrt2 genes is impaired in nitrate uptake. FEBS Lett. 2001, 489, 2–3. [Google Scholar] [CrossRef]
- Li, W.; Wang, Y.; Okamoto, M.; Crawford, N.M.; Siddiqi, M.Y.; Glass, A.D.M. Dissection of the: Inducible high-affinity nitrate transporter gene cluster. Pract. Biochem. Coll. 2007, 13, 161–165. [Google Scholar]
- Pham, A.Q.; Cho, S.H.; Nguyen, C.T.; Stacey, G. Arabidopsis Lectin Receptor Kinase P2K2 is a second plant receptor for extracellular ATP and contributes to innate immunity. Plant Physiol. 2020, 183, 1364–1375. [Google Scholar] [CrossRef]
- Qiu, X.M.; Sun, Y.Y.; Ye, X.Y.; Li, Z.G. Signaling role of glutamate in plants. Front. Plant ENCE 2020, 10, 1743. [Google Scholar] [CrossRef] [PubMed]
- Ju, C.; Song, Y.; Kong, D. Arabidopsis GLR3.5-modulated seed germination involves GA and ROS signaling. Plant Signal. Behav. 2020, 15, 1729537. [Google Scholar]
- Forde, B.G.; Roberts, M.R. Glutamate receptor-like channels in plants: A role as amino acid sensors in plant defence? F1000prime Rep. 2014, 6, 37. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, G.; Huo, Z.; Xiao, Y.; Xiong, F.; Zhang, H.; Dai, Q. Effects of application of nitrogen combined with silicon on the photosynthesis and activities of nitrogen metabolic enzyme of rice leaf. J. Yangzhou Univ. 2010, 28, 491–496. [Google Scholar]
- Zhang, R.; Zhu, J.; Cao, H.; Xie, X.; Luo, Z. Isolation and characterization of LHT-type plant amino acid transporter gene from Panax ginseng Meyer. J. Ginseng Res. 2013, 37, 361–370. [Google Scholar] [CrossRef]
- Ensafi, A.A.; Rezaei, B.; Nouroozi, S. Simultaneous spectrophotometric determination of nitrite and nitrate by flow injection analysis. Anal. Sci. 2004, 20, 1749–1753. [Google Scholar] [CrossRef]
- Alonso, E.V.; Cordero, M.T.S.; Torres, M.P.C.; Torres, A.G.; Pavon, J.M.C. Indirect determination of nitrate by electrothermal atomic absorption spectrometry using an online cadmium microcolumn. Analyst 1998, 123, 1561–1564. [Google Scholar] [CrossRef]
- Guo, Y.X.; Zhang, Q.F.; Shangguang, X.; Zhen, G. Spectrofluorimetric determination of trace nitrite with o-phenylenediamine enhanced by hydroxypropyl-β-cyclodextrin. Spectrochim. Acta Part A 2013, 101, 107–111. [Google Scholar] [CrossRef]
- Yaqoob, M.; Folgado Biot, B.; Nabi, A.; Worsfold, P.J. Determination of nitrate and nitrite in freshwaters using flow injection with luminol chemiluminescence detection. Luminescence 2021, 27, 419–425. [Google Scholar] [CrossRef]
- Sun, B.; Fitch, P.G. Nitrate Ion-Selective Sensor Based on Electrochemically Prepared Conducting Polypyrrole Films. Electroanalysis 1997, 9, 6. [Google Scholar] [CrossRef]
- Lee, R.B. Sources of reductant for nitrate assimilation in non-photosynthetic tissue: A review. Plant. Cell Environ. 1980, 3, 65–90. [Google Scholar]
- Xu, Q.; Lu, D.; Pang, G. Comparative study of hGPR120 receptor self-assembled nano-gold sensor and tissue sensor. Sens. Actuators B Chem. 2020, 320, 128382. [Google Scholar] [CrossRef]
- Wauson, E.M.; Zaganjor, E.; Lee, A.Y.; Guerra, M.L.; Ghosh, A.B.; Bookout, A.L.; Chambers, C.P.; Jivan, A.; McGlynn, K.; Hutchison, M.R.; et al. The G Protein-Coupled Taste Receptor T1R1/T1R3 Regulates mTORC1 and Autophagy. Mol. Cell 2012, 47, 851–862. [Google Scholar] [CrossRef] [PubMed]
- Riera, C.E.; Dillin, A. Emerging role of sensory perception in aging and metabolism. Trends Endocrinol. Metab. 2016, 27, 294–303. [Google Scholar] [CrossRef]
- Taddese, B.; Upton, G.J.G.; Bailey, G.R.; Jordan, S.R.D.; Abdulla, N.Y.; Reeves, P.J.; Reynolds, C.A. Do plants contain g protein-coupled receptors? Plant. Physiol. 2014, 164, 287–307. [Google Scholar] [CrossRef]
- Plakidou-Dymock, S.; Dymock, D.; Hooley, R. A higher plant seven-transmembrane receptor that influences sensitivity to cytokinins. Curr. Biol. 1998, 8, 315–324. [Google Scholar] [CrossRef]
- Rogato, A.; Valkov, V.T.; Alves, L.M.; Apone, F.; Colucci, G.; Chiurazzi, M. Down-regulated lotus japonicus gcr1 plants exhibit nodulation signalling pathways alteration. Plant. ENCE 2016, 247, 71–82. [Google Scholar] [CrossRef]
- Jagdeep, K.; Swarup, R.C.; Anitha, V.; Laryssa, H.; Zach, R.; Rob, P.; Dylan, B.; Sona, P. Arabidopsis Type III Gγ Protein AGG3 Is a Positive Regulator of Yield and Stress Responses in the Model Monocot Setaria viridis. Front. Plant. ENCE 2018, 9, 109. [Google Scholar]
- Sun, S.; Wang, L.; Mao, H.; Shao, L.; Li, X.; Xiao, J.; Ouyang, Y.; Zhang, Q. A G-protein pathway determines grain size in rice. Nat. Commun. 2018, 9, 851. [Google Scholar] [CrossRef]
- Qian, S.; Zheng, D. Advances in Research on Taste Receptors and Application Prospects of Taste Sensors. Available online: http://www.cqvip.com/QK/83290X/20002/4717897.html (accessed on 1 January 2021).

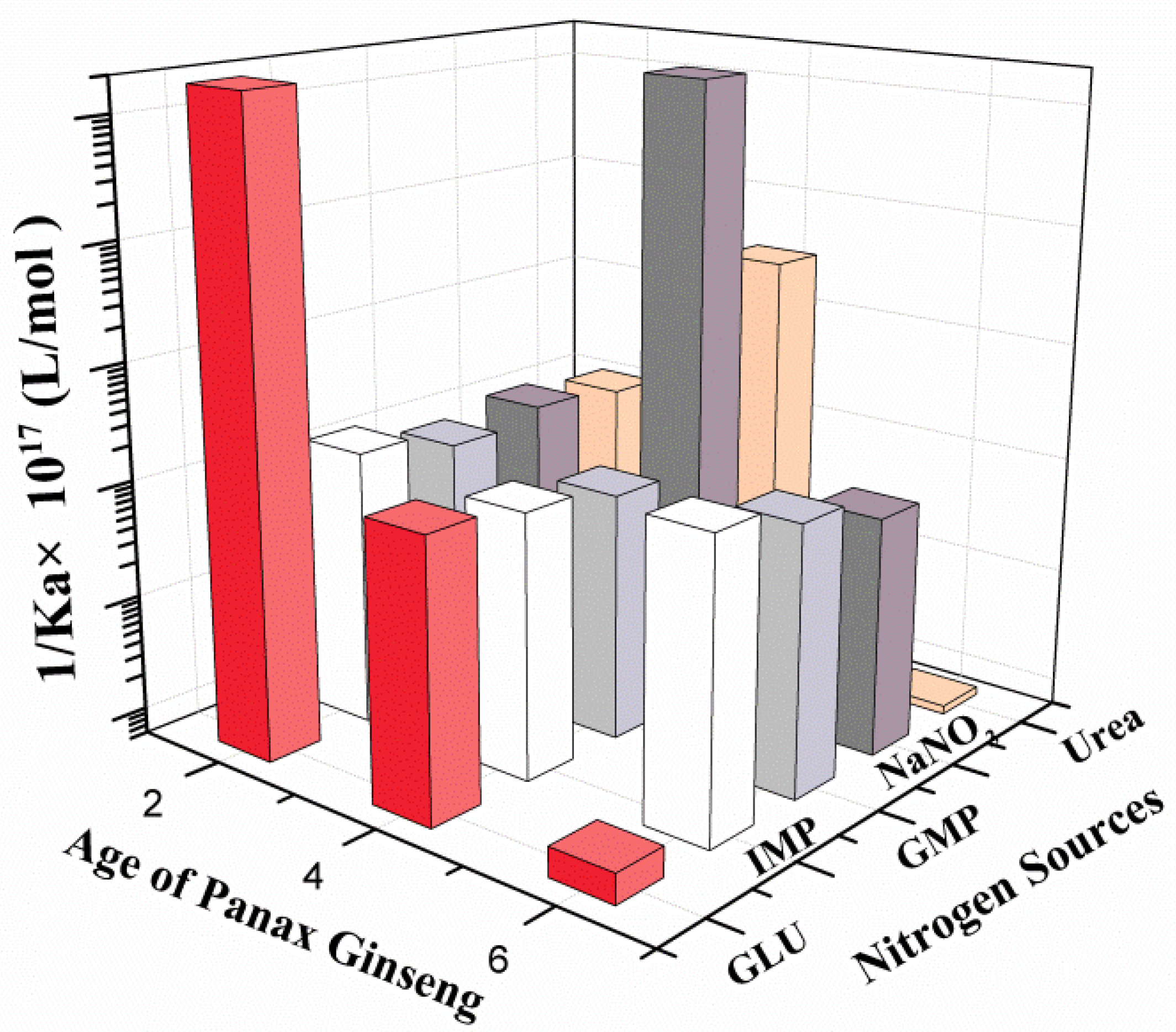
| Age of Ginseng (Years) | Ka × 10−21 (mol/L) | ||||
|---|---|---|---|---|---|
| IMP | GMP | Sodium Glutamate | NaNO3 | Urea | |
| Two | 5.024 | 8.339 | 927.7 | 14.91 | 1416 |
| Four | 9.035 | 12.44 | 6.980 | 0.006529 | 0.3333 |
| Six | 7.035 | 10.67 | 0.005451 | 8.315 | 9.970 |
| Method | Sample | Detection Target | Detection Limit (mol/L) | Reference |
|---|---|---|---|---|
| Catalytic-spectrophotometric | Water/Food | Nitrate | 4.838 × 10−4 | [28] |
| Atomic absorption spectroscopic | Water | Nitrate | 8.063 × 10−4 | [29] |
| Fluorescence-spectroscopic | Water/Soil/Forensic samples | Nitrate | 1.0 × 10−5 | [30] |
| Chemiluminescence | Water | Nitrate | 3.2 × 10−7 | [31] |
| Electrochemical conducting polypyrrole films | Nitrate | Nitrate | 4.7 × 10−5 | [32] |
| Electrochemicalroot-meristem sensor | Ginseng Root-Meristem | Pure solutions of various nutrients | 6.529 × 10−24 | This work |
| Age of Ginseng (Years) | Cardinality (Emergence of Seeding Rate) | Survival Number | Survival Rate | Mean | Mortality | References |
|---|---|---|---|---|---|---|
| One | 1128 | 1004 | 89.01% | 91.89% | 8.11% | [10] |
| 1128 | 1032 | 91.49% | ||||
| 1142 | 1087 | 95.18% | ||||
| Two | 1128 | 977 | 86.61% | 87.45% | 12.55% | |
| 1128 | 963 | 85.37% | ||||
| 1142 | 1032 | 90.37% | ||||
| Four | 16.8 (AVG) | 11.9 (AVG) | 70.83% | 70.83% | 29.17% | [42] |
| Five | 935 | 330 | 35.29% | 39.13% | 60.87% | [10] |
| 922 | 344 | 37.31% | ||||
| 922 | 413 | 44.79% | ||||
| Six | 935 | 49 | 5.24% | 7.24% | 92.76% | |
| 922 | 55 | 5.97% | ||||
| 922 | 97 | 10.52% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, S.; Lu, D.; Pang, G. Construction of a Ginseng Root-Meristem Sensor and a Sensing Kinetics Study on the Main Nitrogen Nutrients. Sensors 2021, 21, 681. https://doi.org/10.3390/s21030681
Wang S, Lu D, Pang G. Construction of a Ginseng Root-Meristem Sensor and a Sensing Kinetics Study on the Main Nitrogen Nutrients. Sensors. 2021; 21(3):681. https://doi.org/10.3390/s21030681
Chicago/Turabian StyleWang, Shiang, Dingqiang Lu, and Guangchang Pang. 2021. "Construction of a Ginseng Root-Meristem Sensor and a Sensing Kinetics Study on the Main Nitrogen Nutrients" Sensors 21, no. 3: 681. https://doi.org/10.3390/s21030681
APA StyleWang, S., Lu, D., & Pang, G. (2021). Construction of a Ginseng Root-Meristem Sensor and a Sensing Kinetics Study on the Main Nitrogen Nutrients. Sensors, 21(3), 681. https://doi.org/10.3390/s21030681







