Nitrogen Dioxide Sensing Using Multilayer Structure of Reduced Graphene Oxide and α-Fe2O3
Abstract
1. Introduction
2. Materials and Methods
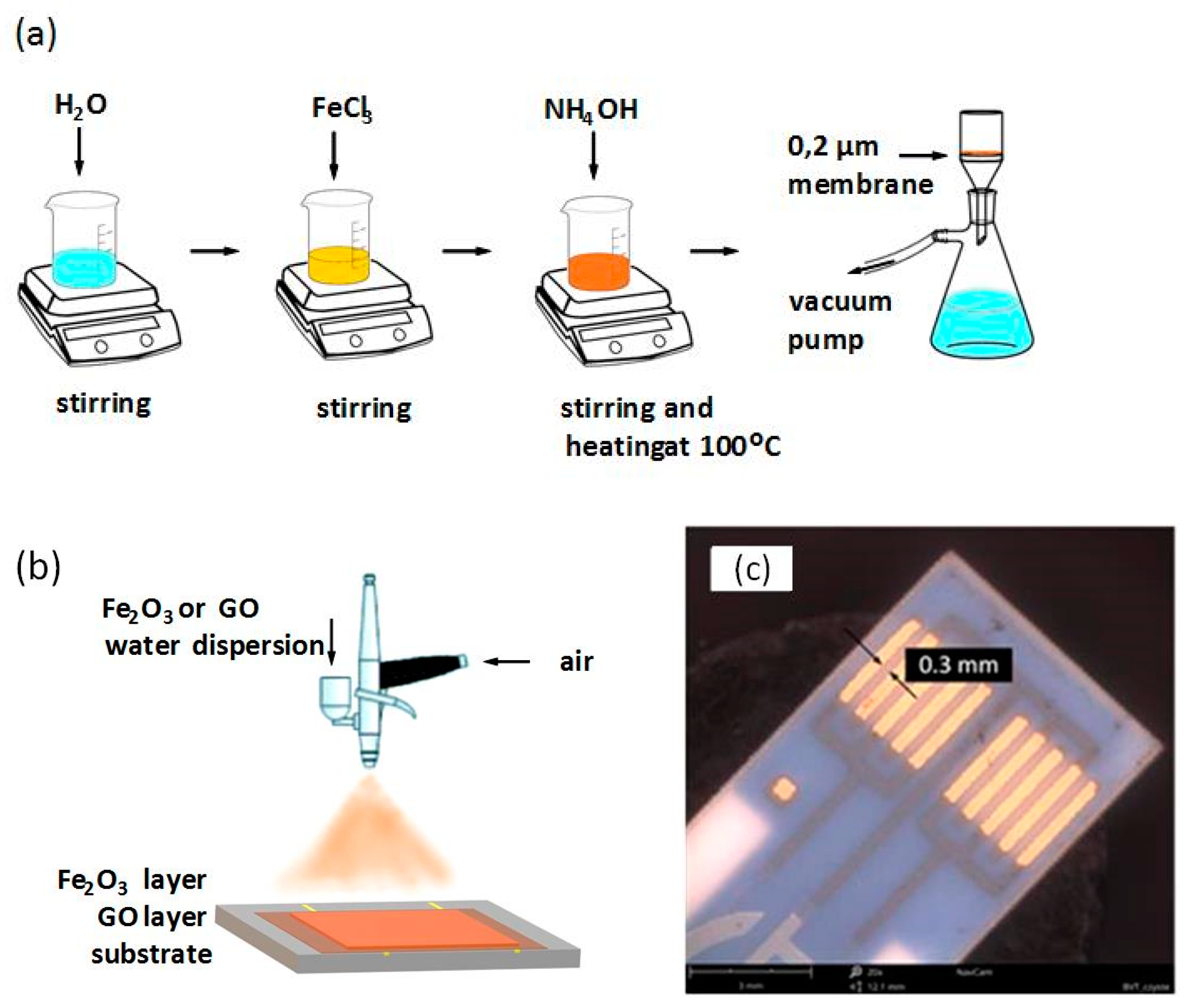
2.1. Sample Preparatiom
2.2. Electrical Measurements
3. Results and Discussion
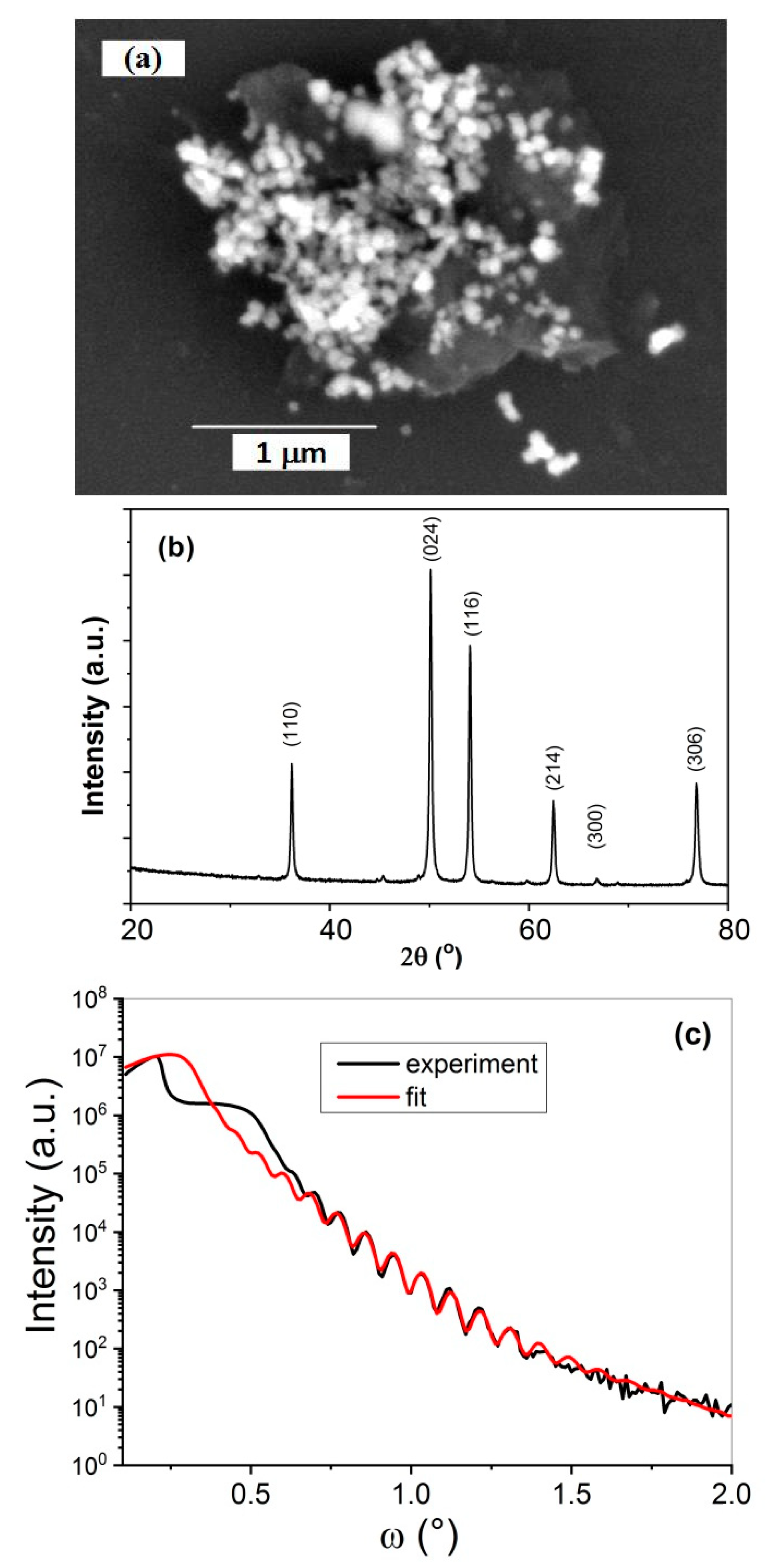
3.1. Structural and Morphological Characteristics
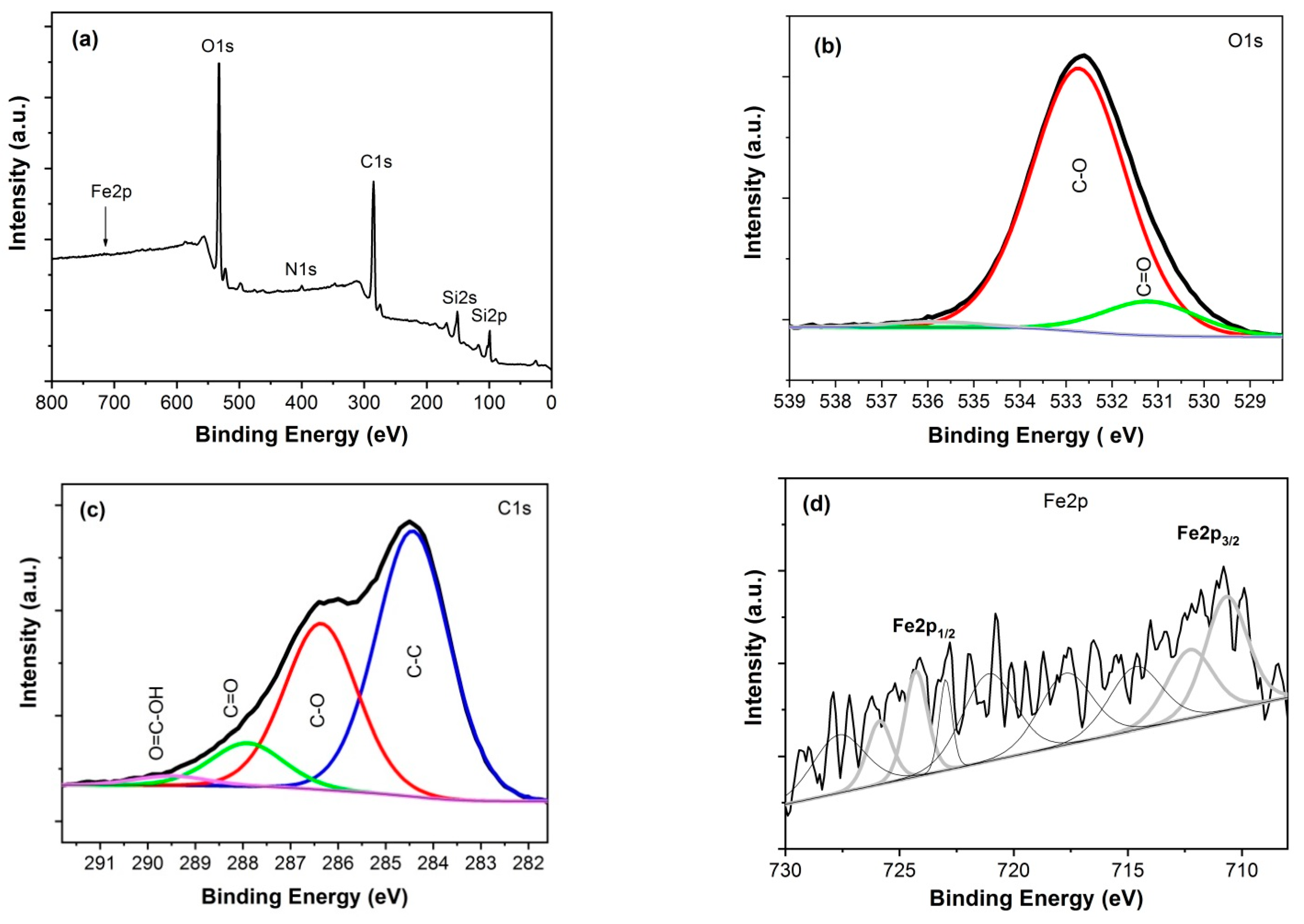
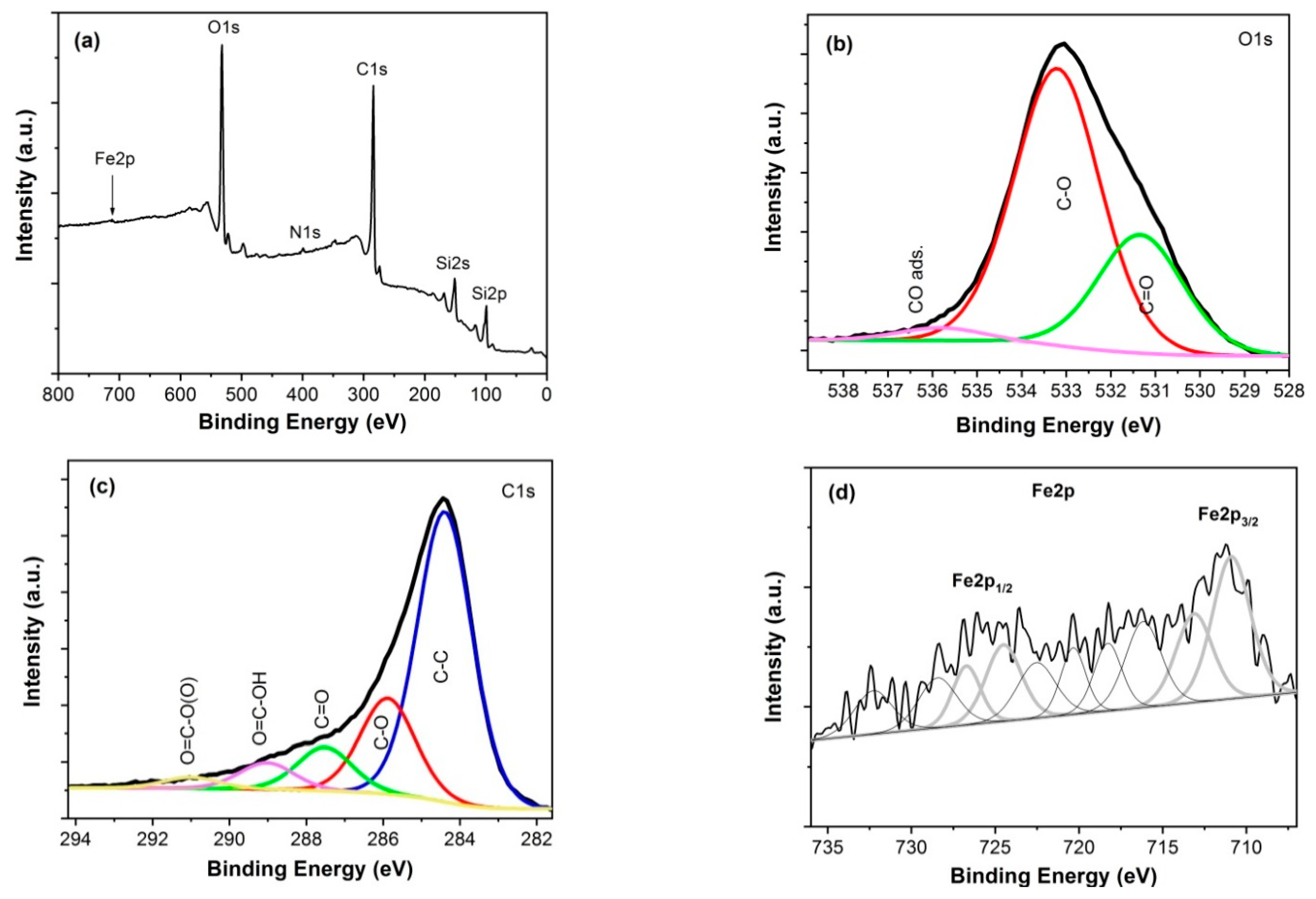
3.2. Chemical Composition
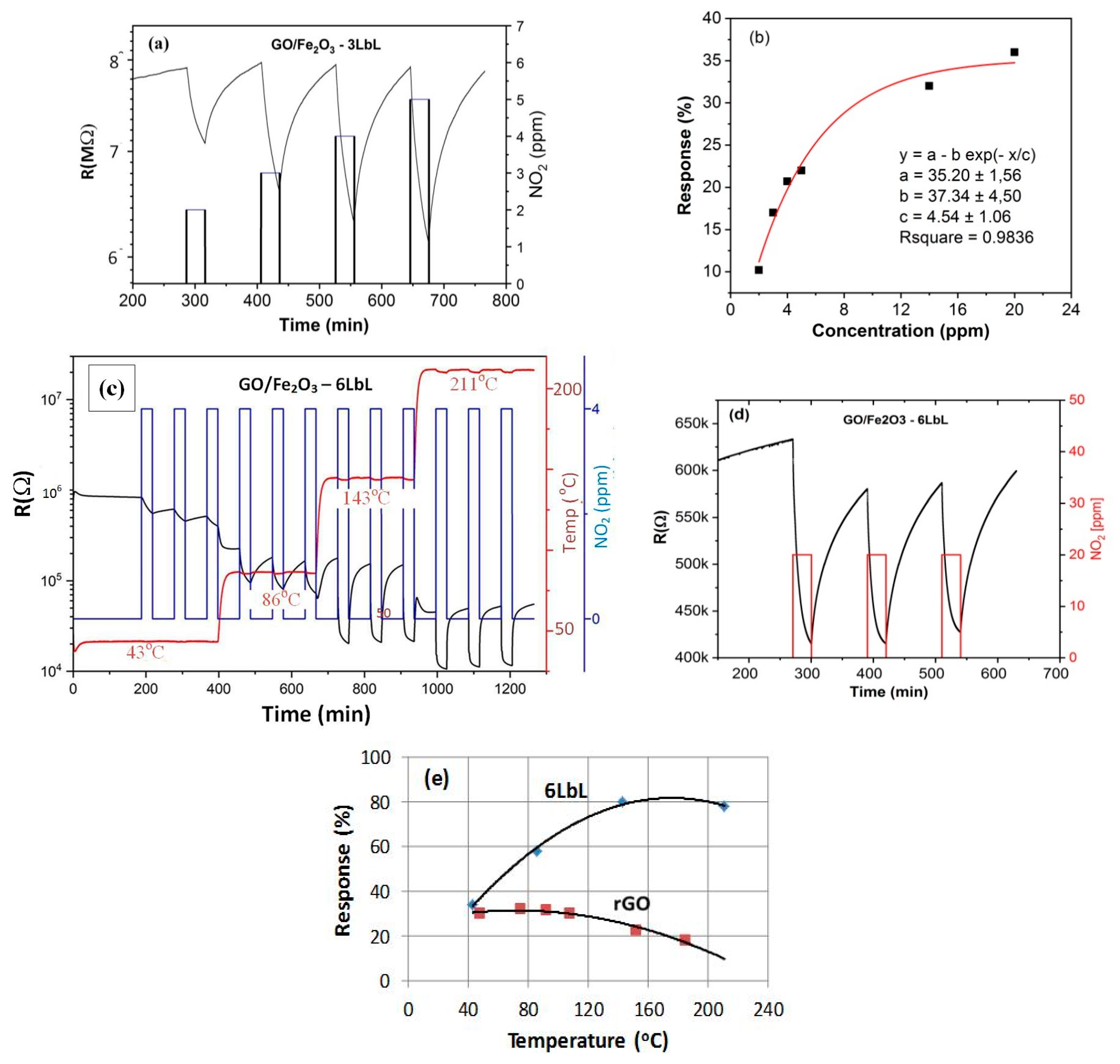
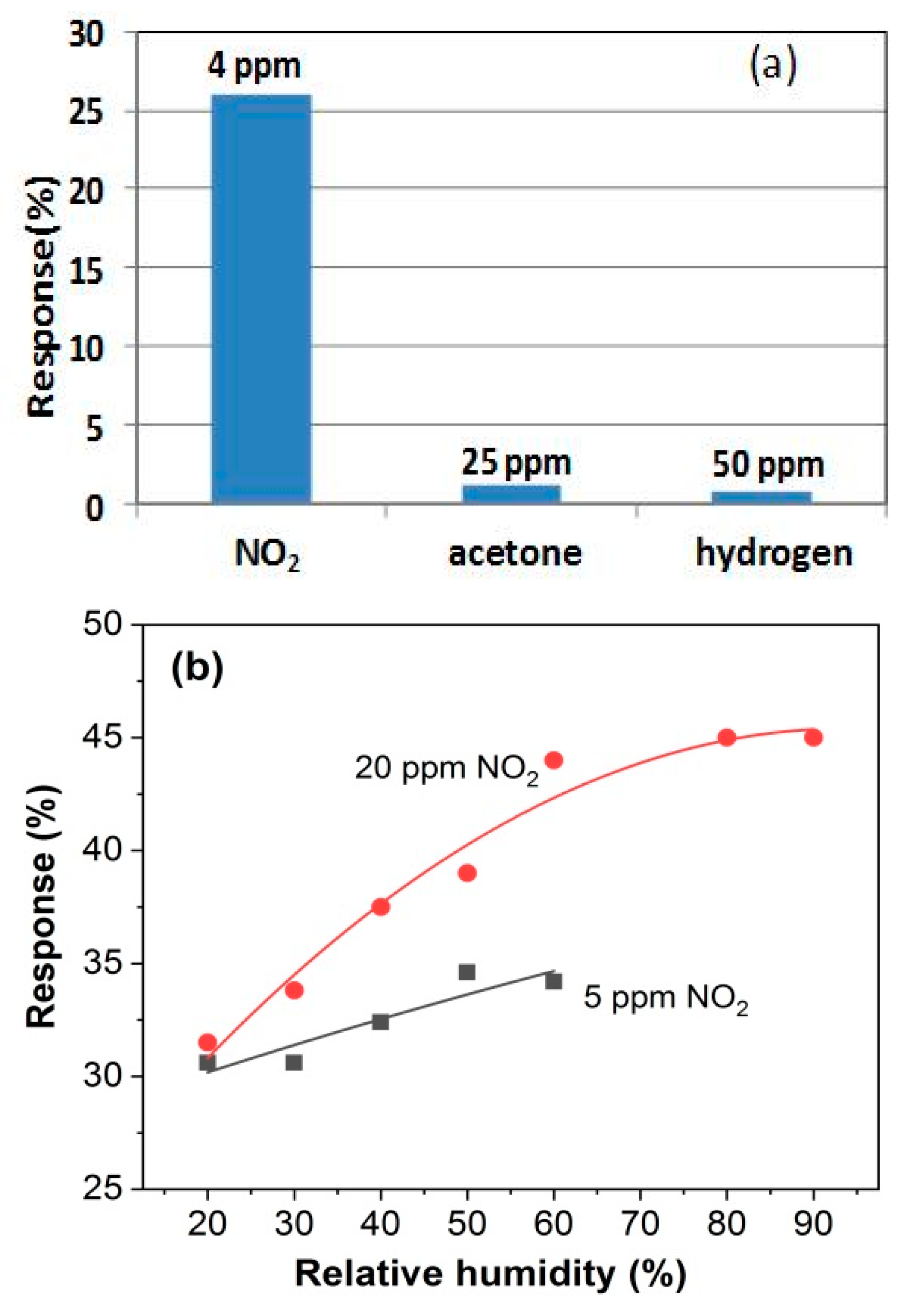
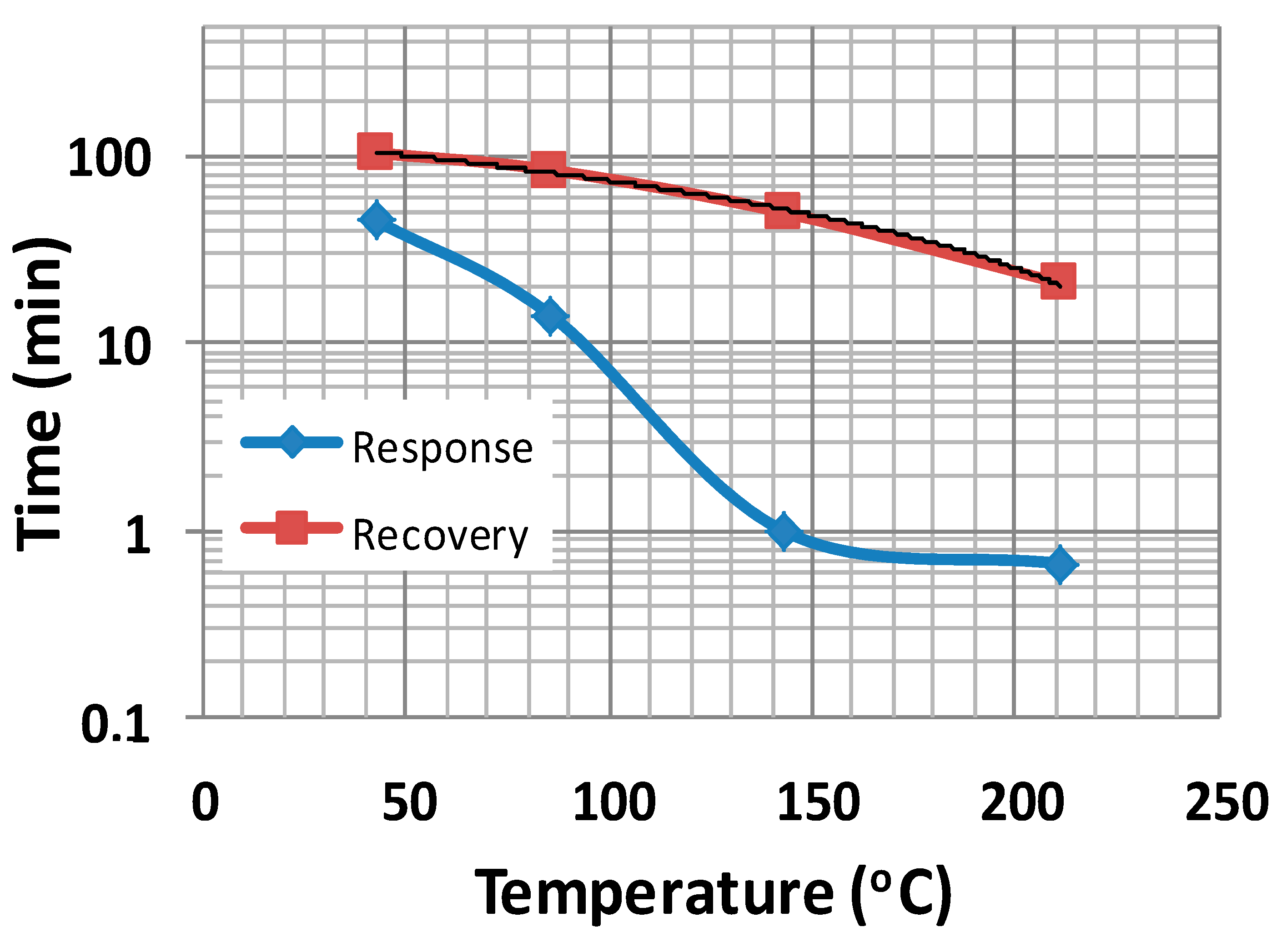
3.3. Gas Sensing Properties
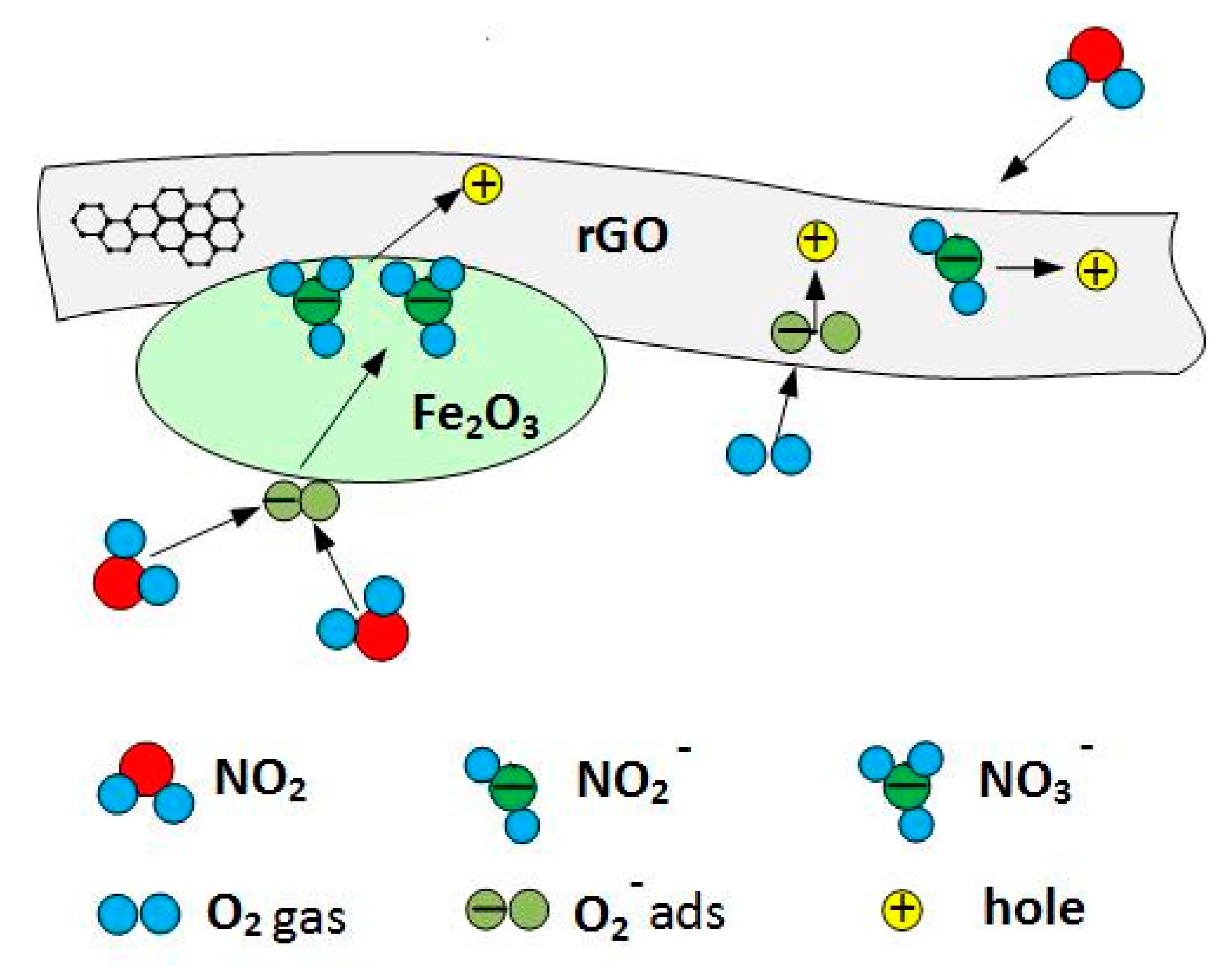
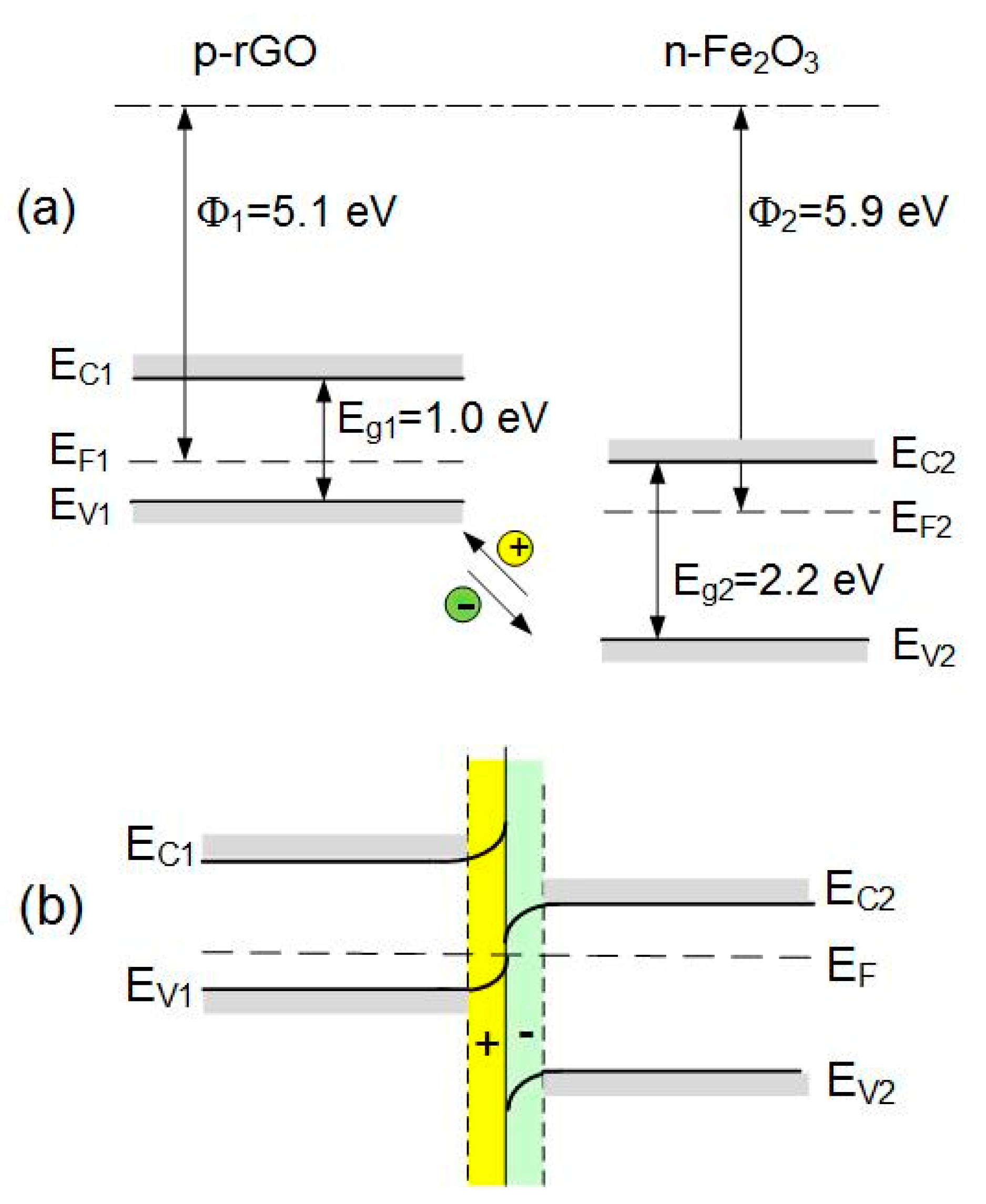
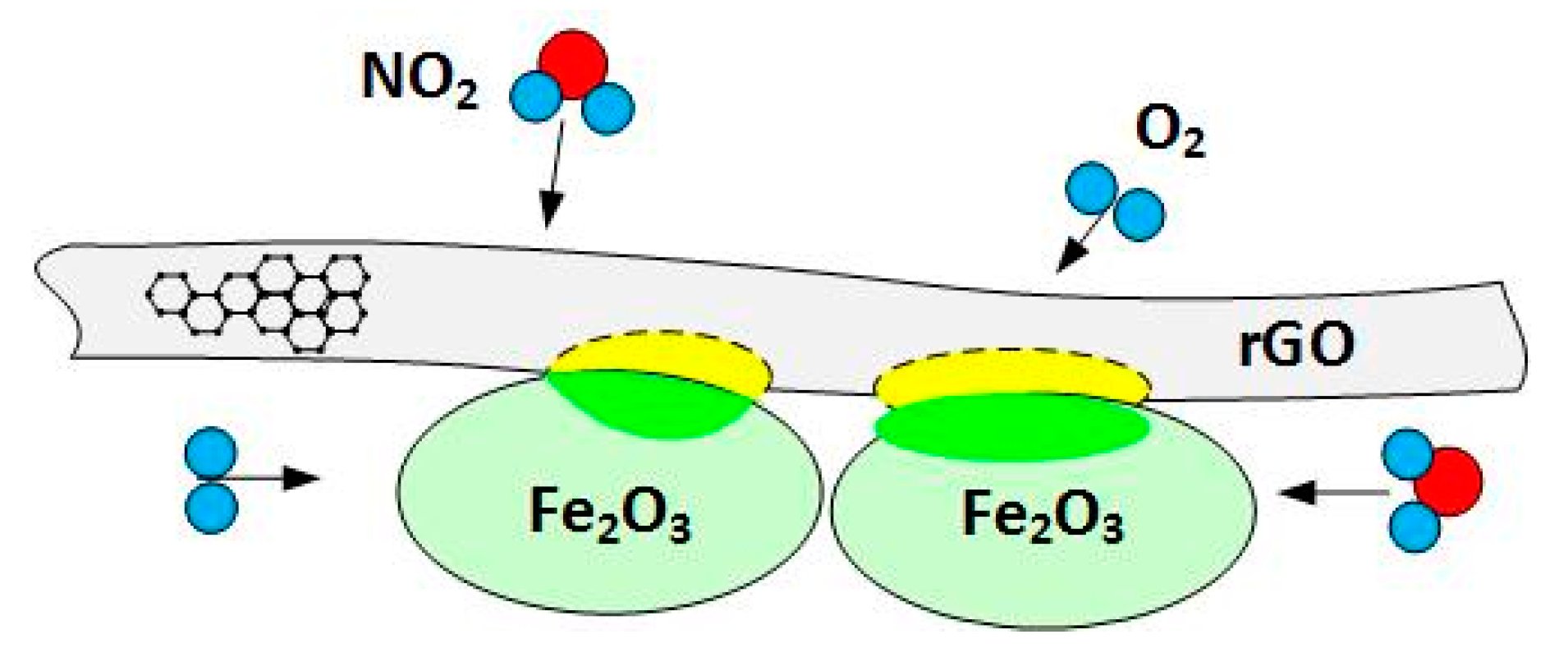
3.4. Gas Sensing Mechanism
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Geupel, A.; Schönauer, D.; Röder-Roith, U.; Kubinski, D.J.; Mulla, S.; Ballinger, T.H.; Chen, H.-Y.; Visser, J.H.; Moos, R. Inte-grating nitrogen oxide sensor: A novel concept for measuring low concentrations in the exhaust gas. Sens. Actuators B Chem. 2010, 145, 756–761. [Google Scholar] [CrossRef]
- Pal, N.; Murray, E. Dense LaSrMnO3 composite electrodes for NOx sensing. Sens. Actuators B Chem. 2018, 256, 351–358. [Google Scholar] [CrossRef]
- Liu, S.; Wang, Z.Y.; Zhang, Y.; Li, J.C.; Zhang, T. Sulfonated graphene anchored with tin oxide nanoparticles for detection of nitrogen dioxide at room temperature with enhanced sensitivity performances. Sens. Actuators B Chem. 2016, 228, 134–143. [Google Scholar] [CrossRef]
- Feng, Q.; Li, X.; Wang, J. Percolation effect of reduced graphene oxide (rGO) on ammonia sensing of rGO-SnO2 composite based sensor. Sens. Actuators B Chem. 2017, 243, 1115–1126. [Google Scholar] [CrossRef]
- Xiao, Y.; Yang, Q.; Wang, Z.; Zhang, R.; Gao, Y.; Sun, P.; Sun, Y.; Lu, G. Improvement of NO2 gas sensing performance based on discoid tin oxide modified by reduced graphene oxide. Sens. Actuators B Chem. 2016, 227, 419–426. [Google Scholar] [CrossRef]
- Punetha, D.; Kumar Pandey, S. Sensitivity Enhancement of Ammonia Gas Sensor Based on Hydrothermally Synthesized rGO/WO3 Nanocomposites. IEEE Sens. J. 2020, 20, 1738–1745. [Google Scholar] [CrossRef]
- Srivastava, S.; Jain, K.; Singh, V.N.; Singh, S.; Vijayan, N.; Dilawar, N.; Gupta, G.; Senguttuvan, T.D. Faster response of NO2 sensing in graphene–WO3 nanocomposites. Nanotechnology 2012, 23, 205501. [Google Scholar] [CrossRef]
- Galstyan, V.; Ponzoni, A.; Kholmanov, I.; Natile, M.M.; Comini, E.; Nematov, S.; Sberveglieri, G. Reduced Graphene Oxide–TiO2 Nanotube Composite: Comprehensive Study for Gas-Sensing Applications. ACS Appl. Nano Mater. 2018, 1, 7098–7105. [Google Scholar] [CrossRef]
- Lin, W.-D.; Liao, C.-T.; Chang, T.-C.; Chen, S.-H.; Wu, R.-J. Humidity sensing properties of novel graphene/TiO2 composites by sol–gel process. Sens. Actuators B Chem. 2015, 209, 555–561. [Google Scholar] [CrossRef]
- Gu, F.; Nie, R.; Han, D.; Wang, Z. In2O3–graphene nanocomposite based gas sensor for selective detection of NO2 at room temperature. Sens. Actuators B Chem. 2015, 219, 94–99. [Google Scholar] [CrossRef]
- Liu, S.; Yu, B.; Zhang, H.; Fei, T.; Zhang, T. Enhancing NO2 gas sensing performances at room temperature based on reduced graphene oxide-ZnO nanoparticles hybrids. Sens. Actuators B Chem. 2014, 202, 272–278. [Google Scholar] [CrossRef]
- Ugale, A.D.; Umarji, G.G.; Jung, S.H.; Deshpande, N.G.; Lee, W.; Cho, H.K.; Yoo, J. ZnO decorated flexible and strong graphene fibers for sensing NO2 and H2S at room temperature. Sens. Actuators B Chem. 2020, 308, 127690. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, T.; Zhao, C.; Han, T.; Fei, T.; Liu, S.; Lu, G. Rational synthesis of molybdenum disulfide nanoparticles decorated reduced graphene oxide hybrids and their application for high-performance NO2 sensing. Sens. Actuators B Chem. 2018, 260, 508–518. [Google Scholar] [CrossRef]
- Zhou, Y.; Liu, G.; Zhu, X.; Guo, Y. Ultrasensitive NO2 gas-sensing based on rGO/MoS2 nanocomposite film at low temper-ature. Sens. Actuators B Chem. 2017, 251, 280–290. [Google Scholar] [CrossRef]
- Hou, X.; Wang, Z.; Fan, G.; Ji, H.; Yi, S.; Li, T.; Wang, Y.; Zhang, Z.; Yuan, L.; Zhang, R.; et al. Hierarchical three-dimensional MoS2/GO hybrid nanostructures for trimethylamine-sensing applications with high sensitivity and selec-tivity. Sens. Actuators B Chem. 2020, 317, 128236. [Google Scholar] [CrossRef]
- Wang, X.; Gu, D.; Li, X.; Lin, S.; Zhao, S.; Kovalenko, V.V.; Gaskov, A.M. Reduced graphene oxide hybridized with WS2 nanoflakes based heterojunctions for selective ammonia sensors at room temperature. Sens. Actuators B Chem. 2019, 282, 290–299. [Google Scholar] [CrossRef]
- Wu, Z.; Chen, X.; Zhu, S.; Zhou, Z.; Yao, Y.; Quan, W.; Liu, B. Room Temperature Methane Sensor Based on Graphene Nanosheets/Polyaniline Nanocomposite Thin Film. IEEE Sens. J. 2012, 13, 777–782. [Google Scholar] [CrossRef]
- Gil Na, H.; Cho, H.Y.; Kwon, Y.J.; Kang, S.Y.; Lee, C.; Jung, T.K.; Lee, H.-S.; Kim, H.W. Reduced graphene oxide functionalized with Cu nanoparticles: Fabrication, structure, and sensing properties. Thin Solid Film. 2015, 588, 11–18. [Google Scholar] [CrossRef]
- Shafiei, M.; Spizzirri, P.G.; Arsat, R.; Yu, J.; du Plessis, J.; Dubin, S.; Ianer, R.B.; Kalantar-Zadeh, K.; Wlodarski, W. Platinum/graphene nanosheet/SiC contacts and their application for hydrogen gas sensing. J. Phys. Chem. C 2010, 114, 13796–13801. [Google Scholar] [CrossRef]
- Cho, B.; Yoon, J.; Hahm, M.G.; Kim, D.-H.; Kim, A.R.; Khang, Y.H.; Park, S.-W.; Lee, Y.-J.; Park, S.-G.; Kwon, J.-D.; et al. Graphene-based gas sensor: Metal decoration effect and application to a flexible device. J. Mater. Chem. C 2014, 2, 5280–5285. [Google Scholar]
- Kumar, R.; Varandani, D.; Mehta, B.R.; Singh, V.N.; Wen, Z.; Feng, X.; Müllen, K. Fast response and recovery of hydrogen sensing in Pd–Pt nanoparticle–graphene composite layers. Nanotechnology 2011, 22, 275719. [Google Scholar] [CrossRef] [PubMed]
- Drmosh, Q.A.; Hendi, A.H.; Hossain, M.K.; Yamani, Z.H.; Moqbel, R.A.; Hezam, A.; Gondal, M.A. UV-activated gold deco-rated rGO/ZnO heterostructured nanocomposite sensor for efficient room temperature H2 detection. Sens. Actuators B Chem. 2019, 290, 666–675. [Google Scholar] [CrossRef]
- Guo, W.; Zhou, Q.; Zhang, J.; Fu, M.; Radacsi, N.; Li, Y. Hydrothermal synthesis of Bi-doped SnO2/rGO nanocomposites and the enhanced gas sensing performance to benzene. Sens. Actuators B Chem. 2019, 299, 126959. [Google Scholar] [CrossRef]
- Sun, D.; Luo, Y.; Debliquy, M.; Zhang, C. Graphene-enhanced metal oxide gas sensors at room temperature: A review. Beilstein J. Nanotechnol. 2018, 9, 2832–2844. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhang, S.; Yang, Y.; Yu, H.; Dong, X. Highly sensitive H2S sensors based on metal-organic framework driven γ-Fe2O3 on reduced graphene oxide composites at room temperature. Sens. Actuators B Chem. 2020, 325, 128804. [Google Scholar] [CrossRef]
- Staszek, K.; Szkudlarek, A.; Kawa, M.; Rydosz, A. Microwave system with sensor utilizing GO-based gas-sensitive layer and its application to acetone detection. Sens. Actuators B Chem. 2019, 297, 126699. [Google Scholar] [CrossRef]
- Hjiri, M.; Aida, M.S.; Neri, G. NO2 Sensitive Sensor Based on α-Fe2O3 Nanoparticles Synthesized via Hydrothermal Technique. Sensors 2019, 19, 167. [Google Scholar] [CrossRef]
- Zhang, H.; Yu, L.; Li, Q.; Du, Y.; Ruan, S. Reduced graphene oxide/α-Fe2O3 hybrid nanocomposites for room temperature NO2 sensing. Sens. Actuators B Chem. 2017, 241, 109–115. [Google Scholar] [CrossRef]
- Zhang, B.; Liu, G.; Cheng, M.; Gao, Y.; Zhao, L.; Li, S.; Liu, F.; Yan, X.; Zhang, T.; Sun, P.; et al. The preparation of reduced graphene oxide-encapsulated α-Fe2O3 hybrid and its outstanding NO2 gas sensing properties at room temperature. Sens. Actuators B Chem. 2018, 261, 252–263. [Google Scholar] [CrossRef]
- Dong, Y.-L.; Zhang, X.-F.; Cheng, X.; Xu, Y.-M.; Gao, S.; Zhao, H.; Huo, L.-H. Highly selective NO2 sensor at room temperature based on nanocomposites of hierarchical nanosphere-like α-Fe2O3 and reduced graphene oxide. RSC Adv. 2014, 4, 57493–57500. [Google Scholar] [CrossRef]
- Song, H.; Xia, L.; Jia, X.; Yang, W. Polyhedral α-Fe2O3 crystals@RGO nanocomposites: Synthesis, characterization and ap-plication in gas sensing. J. Alloys Compd. 2018, 732, 191–200. [Google Scholar] [CrossRef]
- Guo, L.; Kou, X.; Ding, M.; Wang, C.; Dong, L.; Zhang, H.; Feng, C.; Sun, Y.; Gao, Y.; Sun, P.; et al. Reduced graphene oxide/α-Fe2O3 composite nanofibers for application in gas sensors. Sens. Actuators B Chem. 2017, 244, 233–242. [Google Scholar] [CrossRef]
- Liang, S.; Zhu, J.; Wang, C.; Yu, S.; Bi, H.; Liu, X.; Wang, X. Fabrication of α-Fe2O3@graphene nanostructures for enhanced gas-sensing property to ethanol. Appl. Surf. Sci. 2014, 292, 278–284. [Google Scholar] [CrossRef]
- Jia, X.; Lian, D.; Shi, B.; Dai, R.; Li, C.; Wu, X. Facile synthesis of α-Fe2O3@graphene oxide nanocomposites for enhanced gas-sensing performance to ethanol. J. Mater. Sci. Mater. Electron. 2017, 28, 12070–12079. [Google Scholar] [CrossRef]
- Li, B.; Li, M.; Meng, F.; Liu, J. Highly selective ethylene sensors using Pd nanoparticles and rGO modified flower-like hierarchical porous α-Fe2O3. Sens. Actuators B Chem. 2019, 290, 396–405. [Google Scholar] [CrossRef]
- Geng, W.; Ge, S.; He, X.; Zhang, S.; Gu, J.; Lai, X.; Wang, H.; Zhang, Q. Volatile Organic Compound Gas-Sensing Properties of Bimodal Porous α-Fe2O3 with Ultrahigh Sensitivity and Fast Response. ACS Appl. Mater. Interfaces 2018, 10, 13702–13711. [Google Scholar] [CrossRef]
- Sun, G.; Chen, H.; Li, Y.; Ma, G.; Zhang, S.; Jia, T.; Cao, J.; Wang, X.; Bala, H.; Zhang, Z. Synthesis and triethylamine sensing properties of mesoporous α-Fe2O3 microrods. Mater. Lett. 2016, 178, 213–216. [Google Scholar] [CrossRef]
- Wang, M.; Hou, T.; Shen, Z.; Zhao, X.; Ji, H. MOF-derived Fe2O3: Phase control and effects of phase composition on gas sensing performance. Sens. Actuators B Chem. 2019, 292, 171–179. [Google Scholar] [CrossRef]
- Stobinski, L.; Lesiak, B.; Malolepszy, A.; Mazurkiewicz, M.; Mierzwa, B.; Zemek, J.; Jiricek, P.; Bieloshapka, I. Graphene oxide and reduced graphene oxide studied by the XRD, TEM and electron spectroscopy methods. J. Electron Spectrosc. Relat. Phenom. 2014, 195, 145–154. [Google Scholar] [CrossRef]
- Pisarkiewicz, T.; Maziarz, W.; Małolepszy, A.; Stobiński, L.; Michoń, D.; Rydosz, A. Multilayer Structure of Reduced Graphene Oxide and Copper Oxide as a Gas Sensor. Coatings 2020, 10, 1015. [Google Scholar] [CrossRef]
- Yang, H.; Li, Y.; Yu, D.; Li, L. Seed/catalyst free growth and self-powered photoresponse of vertically aligned ZnO na-norods on reduced graphene oxide nanosheets. Cryst. Growth Des. 2016, 16, 4831–4838. [Google Scholar] [CrossRef]
- Huang, H.; Li, Z.; She, J.; Wang, W. Oxygen density dependent band gap of reduced graphene oxide. J. Appl. Phys. 2012, 111, 054317. [Google Scholar] [CrossRef]

| Sample | Chemical Bonds | ||||
|---|---|---|---|---|---|
| GO/Fe2O3 | C-C (284.4 eV) | C-O (286.4 eV) | C=O (287.9 eV) | O=C-OH (289.5 eV) | - |
| 54.9% | 34.2% | 8.9% | 2.0% | - | |
| Chemical Bonds | |||||
| rGO/Fe2O3 | C-C (284.4 eV) | C-O (285.9 eV) | C=O (287.5 eV) | O=C-OH (289.0 eV) | O=C-O(O) (291.0 eV) |
| 62.2% | 20.5% | 9.3% | 5.6% | 2.4% | |
| Temperature (°C) | Response Time tres (min) | Recovery Time trec (min) | trec/tres |
|---|---|---|---|
| 43 | 45 | 104 | 2.31 |
| 86 | 14 | 84 | 6 |
| 143 | 1 | 50 | 50 |
| 211 | 0.67 | 21 | 31.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pisarkiewicz, T.; Maziarz, W.; Małolepszy, A.; Stobiński, L.; Michoń, D.A.; Szkudlarek, A.; Pisarek, M.; Kanak, J.; Rydosz, A. Nitrogen Dioxide Sensing Using Multilayer Structure of Reduced Graphene Oxide and α-Fe2O3. Sensors 2021, 21, 1011. https://doi.org/10.3390/s21031011
Pisarkiewicz T, Maziarz W, Małolepszy A, Stobiński L, Michoń DA, Szkudlarek A, Pisarek M, Kanak J, Rydosz A. Nitrogen Dioxide Sensing Using Multilayer Structure of Reduced Graphene Oxide and α-Fe2O3. Sensors. 2021; 21(3):1011. https://doi.org/10.3390/s21031011
Chicago/Turabian StylePisarkiewicz, Tadeusz, Wojciech Maziarz, Artur Małolepszy, Leszek Stobiński, Dagmara Agnieszka Michoń, Aleksandra Szkudlarek, Marcin Pisarek, Jarosław Kanak, and Artur Rydosz. 2021. "Nitrogen Dioxide Sensing Using Multilayer Structure of Reduced Graphene Oxide and α-Fe2O3" Sensors 21, no. 3: 1011. https://doi.org/10.3390/s21031011
APA StylePisarkiewicz, T., Maziarz, W., Małolepszy, A., Stobiński, L., Michoń, D. A., Szkudlarek, A., Pisarek, M., Kanak, J., & Rydosz, A. (2021). Nitrogen Dioxide Sensing Using Multilayer Structure of Reduced Graphene Oxide and α-Fe2O3. Sensors, 21(3), 1011. https://doi.org/10.3390/s21031011







