The Development of Digital Image Colorimetric Quantitative Analysis of Multi-Explosives Using Polymer Gel Sensors
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Materials
2.2. Preparation of Polymer Gel Sensor
2.3. Color Test of Explosives
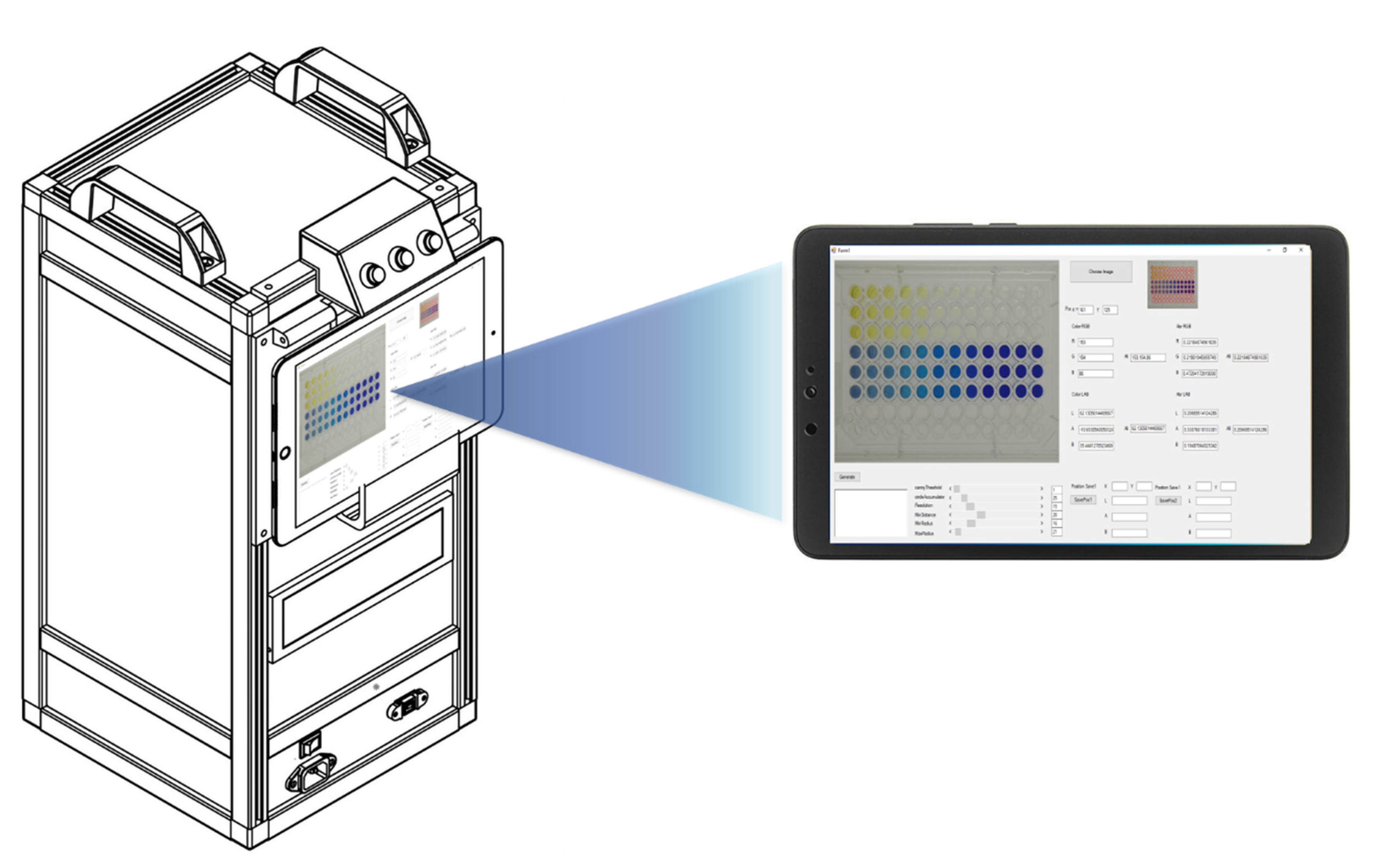
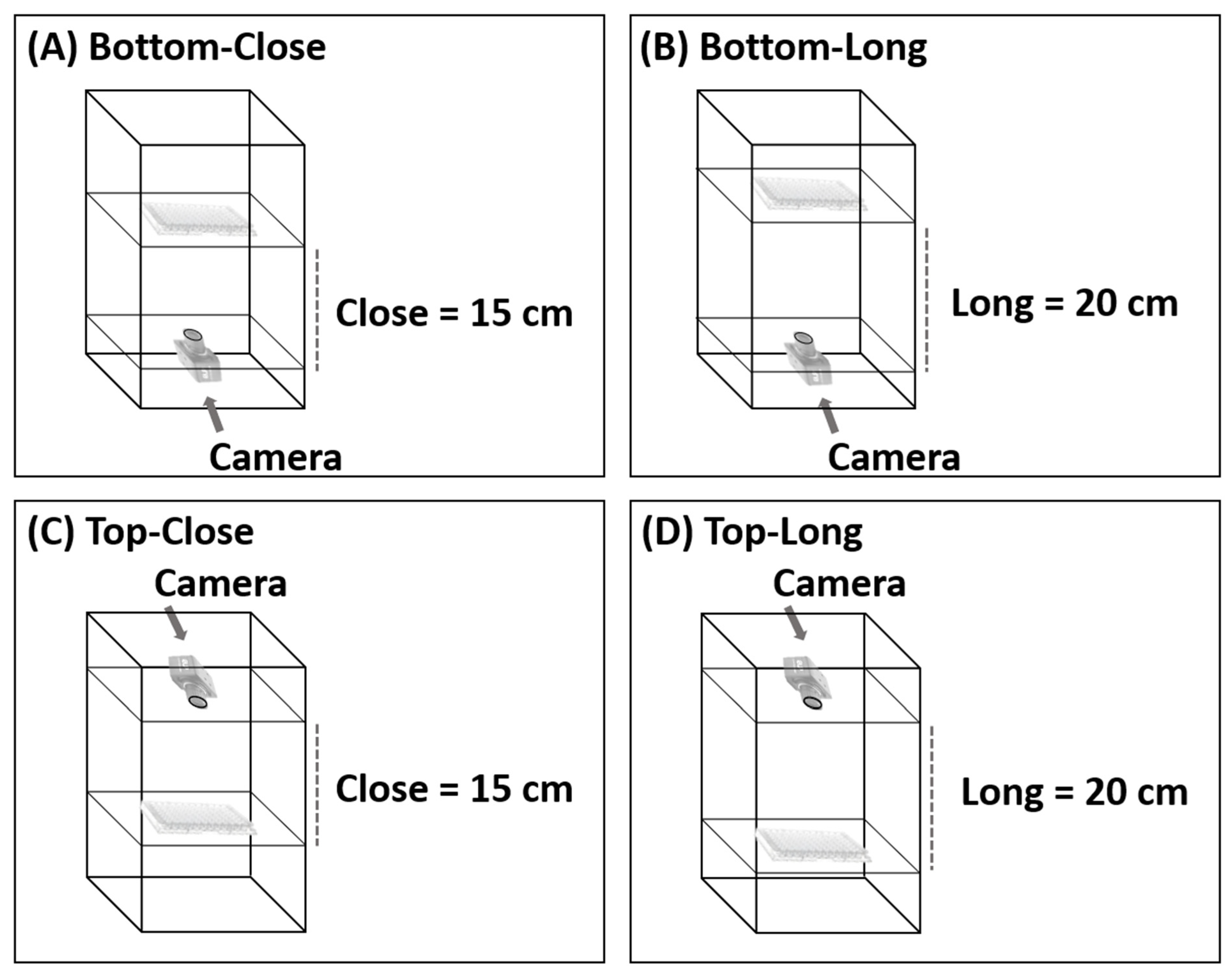
2.4. Photographic System
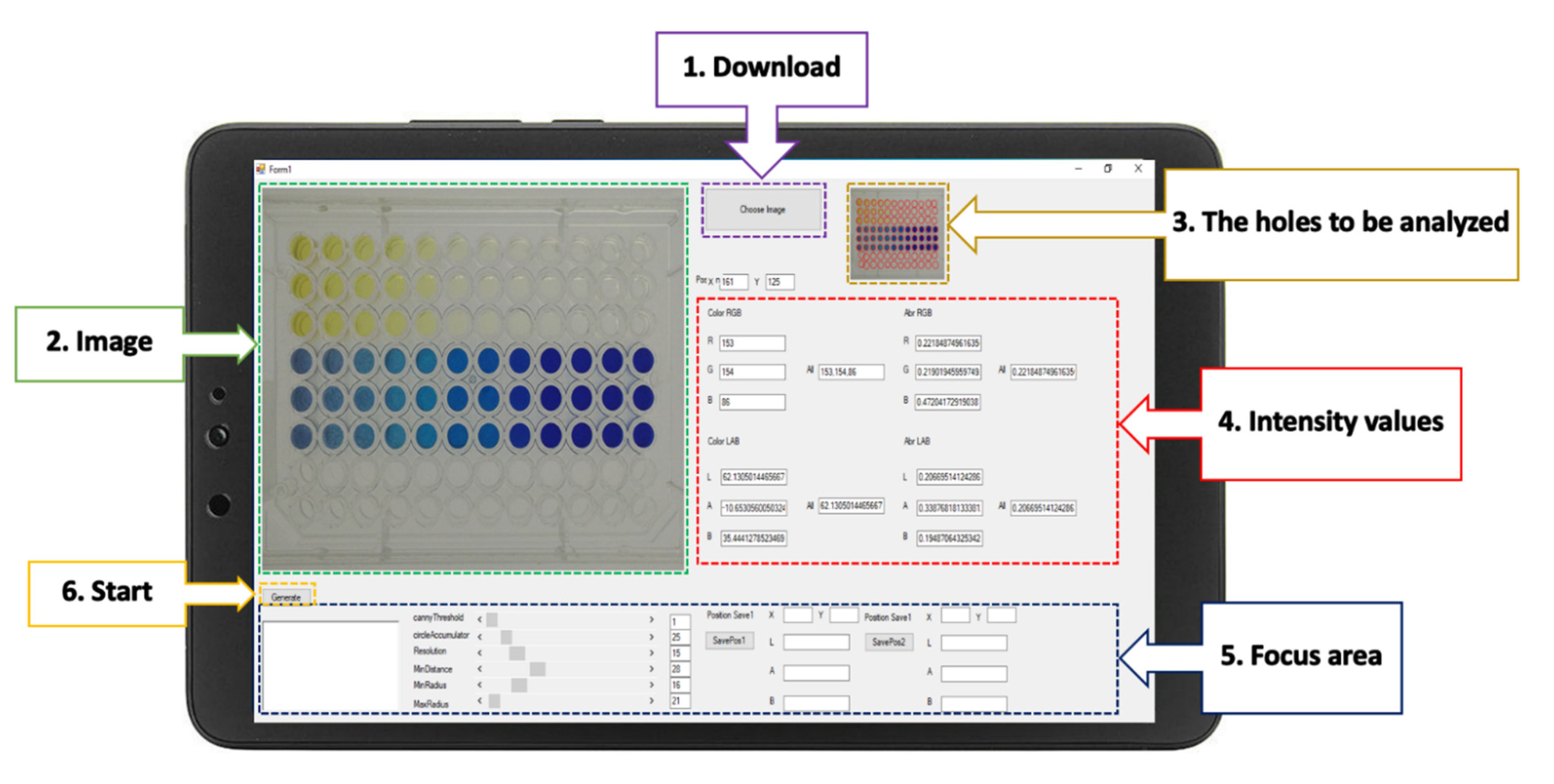
2.5. The RGB Analyzer Program
2.6. Analytical Performance and Method Validation
2.7. Interference Effects
2.8. Application for Multi-Explosives Sensing
3. Results and Discussion
3.1. Optimization of the Photographic System
3.2. Colorimetric Test of Explosives
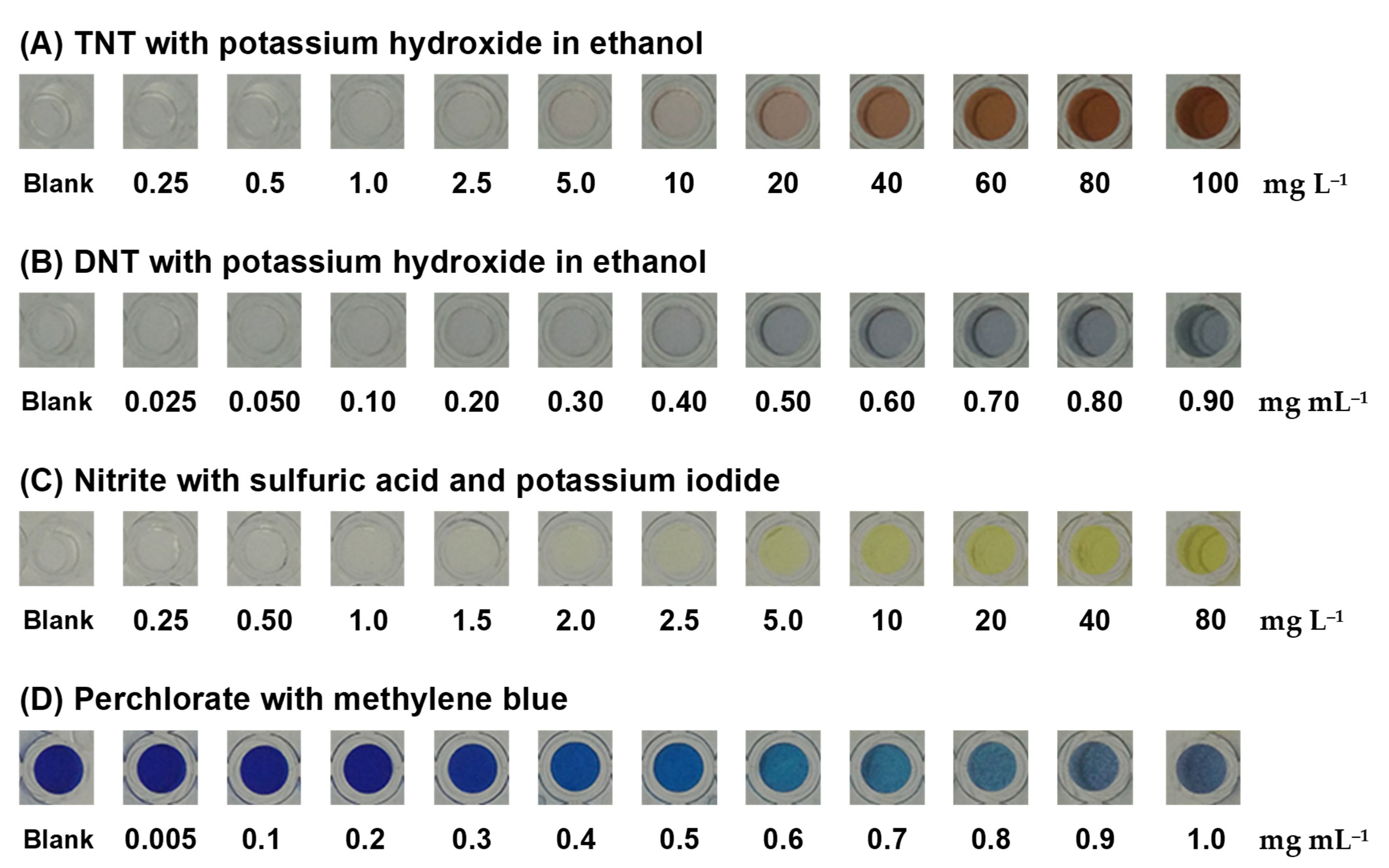
3.2.1. Colorimetric Test of TNT and DNT
3.2.2. Colorimetric Test of Nitrite
3.2.3. Colorimetric Test of Perchlorate
3.3. Digital Image Colorimetry for Explosives Quantification
3.4. Analytical Performance and Method Validation
3.5. Specificity
3.6. Interference
3.7. Analysis of Case Work Sample
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- DePalma, R.G.; Aharonson-Daniel, L. Blast Injuries. N. Engl. J. Med. 2005, 352, 1335–1342. [Google Scholar] [CrossRef]
- Sharma, M.; Kumar, S.; Jain, P. Importance of Forensic Investigation in Explosion: A Case study. J. Forensic Res. 2016, 7, 5. [Google Scholar]
- National Academies of Sciences, Engineering, and Medicine. Reducing the Threat of Improvised Explosive Device Attacks by Restricting Access to Explosive Precursor Chemicals; National Academies Press: Washington, DC, USA, 2018; p. 214. [Google Scholar]
- Boonpanya, J.; Chaikum, N.; Kontrong, P.; Wilairat, P.; Tiensuwan, M. Forensic Investigation of Explosion Cases in the Restive South. R. Thai Army Med. J. 2007, 60, 91–100. [Google Scholar]
- Steinfeld, J.I.; Wormhoudt, J. Explosives detection: A Challenge for Physical Chemistry. Annu. Rev. Phys. Chem. 1998, 49, 203–232. [Google Scholar] [CrossRef] [PubMed]
- Perr, J.M.; Furton, K.G.; Almirall, J.R. Gas chromatography positive chemical ionization and tandem mass spectrometry for the analysis of organic high explosives. Talanta 2005, 67, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Pinnaduwage, L.A.; Thundat, T.; Hawk, J.; Hedden, D.; Britt, P.; Houser, E.; Stepnowski, S.; McGill, R.; Bubb, D. Detection of 2,4-dinitrotoluene using microcantilever sensors. Sens. Actuators B Chem. 2004, 99, 223–229. [Google Scholar] [CrossRef]
- Hao, M.; Zeng, W.; Li, Y.-Q.; Wang, Z.-C. Three-dimensional graphene and its composite for gas sensors. Rare Met. 2021, 40, 1494–1514. [Google Scholar] [CrossRef]
- Liu, W.; Gu, D.; Zhang, J.-W.; Li, X.-G.; Rumyantseva, M.N.; Gaskov, A.M. ZnSe/NiO heterostructure-based chemiresistive-type sensors for low-concentration NO2 detection. Rare Met. 2020, 40, 1632–1641. [Google Scholar] [CrossRef]
- Zhao, Q.-N.; Zhang, Y.-J.; Duan, Z.-H.; Wang, S.; Liu, C.; Jiang, Y.-D.; Tai, H.-L. A review on Ti3C2Tx-based nanomaterials: Synthesis and applications in gas and humidity sensors. Rare Met. 2021, 40, 1459–1476. [Google Scholar] [CrossRef]
- Gaurav; Kaur, V.; Kumar, A.; Malik, A.K.; Rai, P.K. SPME-HPLC: A new approach to the analysis of explosives. J. Hazard. Mater. 2007, 147, 691–697. [Google Scholar] [CrossRef]
- Gaurav; Malik, A.K.; Rai, P.K. Development of a new SPME–HPLC–UV method for the analysis of nitro explosives on reverse phase amide column and application to analysis of aqueous samples. J. Hazard. Mater. 2009, 172, 1652–1658. [Google Scholar] [CrossRef]
- Sarazin, C.; Delaunay, N.; Varenne, A.; Vial, J.; Costanza, C.; Eudes, V.; Minet, J.-J.; Gareil, P. Identification and determination of inorganic anions in real extracts from pre- and post-blast residues by capillary electrophoresis. J. Chromatogr. A 2010, 1217, 6971–6978. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Liu, Z.; Sun, Y.; Zeng, L.; Mei, H.; Xu, J.; Guo, B.; Hao, H. Detection of Three Common Organic Explosives Using Capillary Electrophoresis. J. Mater. Sci. Chem. Eng. 2016, 4, 17–25. [Google Scholar] [CrossRef][Green Version]
- Johns, C.; Shellie, R.; Potter, O.G.; O’Reilly, J.W.; Hutchinson, J.P.; Guijt, R.; Breadmore, M.; Hilder, E.; Dicinoski, G.W.; Haddad, P.R. Identification of homemade inorganic explosives by ion chromatographic analysis of post-blast residues. J. Chromatogr. A 2008, 1182, 205–214. [Google Scholar] [CrossRef]
- Tyrrell, É.; Dicinoski, G.W.; Hilder, E.F.; Shellie, R.A.; Breadmore, M.C.; Pohl, C.A.; Haddad, P.R. Coupled reversed-phase and ion chromatographic system for the simultaneous identification of inorganic and organic explosives. J. Chromatogr. A 2011, 1218, 3007–3012. [Google Scholar] [CrossRef]
- Marinović, V.; Marinović, S.; Jovanović, M.; Jovanović, J.; Štrbac, S. The electrochemical reduction of trinitrotoluene on a platinum wire modified by chemisorbed acetonitrile. J. Electroanal. Chem. 2010, 648, 1–7. [Google Scholar] [CrossRef]
- Promsuwan, K.; Kanatharana, P.; Thavarungkul, P.; Limbut, W. Nitrite amperometric sensor for gunshot residue screening. Electrochim. Acta 2019, 331, 135309. [Google Scholar] [CrossRef]
- Arshad, A.; Wang, H.; Bai, X.; Jiang, R.; Xu, S.; Wang, L. Colorimetric paper sensor for sensitive detection of explosive nitroaromatics based on Au@Ag nanoparticles. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 206, 16–22. [Google Scholar] [CrossRef]
- Bagheri, N.; Khataee, A.; Hassanzadeh, J.; Habibi, B. Visual detection of peroxide-based explosives using novel mimetic Ag nanoparticle/ZnMOF nanocomposite. J. Hazard. Mater. 2018, 360, 233–242. [Google Scholar] [CrossRef]
- Thipwimonmas, Y.; Jaidam, J.; Samoson, K.; Khunseeraksa, V.; Phonchai, A.; Thiangchanya, A.; Chang, K.; Abdullah, A.; Limbut, W. A Simple and Rapid Spectrophotometric Method for Nitrite Detection in Small Sample Volumes. Chemosensors 2021, 9, 161. [Google Scholar] [CrossRef]
- Choodum, A.; Kanatharana, P.; Wongniramaikul, W.; NicDaeid, N. A sol–gel colorimetric sensor for methamphetamine detection. Sens. Actuators B Chem. 2015, 215, 553–560. [Google Scholar] [CrossRef]
- Kaewnu, K.; Samoson, K.; Thiangchanya, A.; Phonchai, A.; Limbut, W. A novel colorimetric indicator for ethanol detection in preserved baby mangoes. Food Chem. 2022, 369, 130769. [Google Scholar] [CrossRef]
- Arts, R.; Hartog, I.D.; Zijlema, S.; Thijssen, V.; Van Der Beelen, S.H.E.; Merkx, M.M. Detection of Antibodies in Blood Plasma Using Bioluminescent Sensor Proteins and a Smartphone. Anal. Chem. 2016, 88, 4525–4532. [Google Scholar] [CrossRef] [PubMed]
- Choodum, A.; Kanatharana, P.; Wongniramaikul, W.; NicDaeid, N. Using the iPhone as a device for a rapid quantitative analysis of trinitrotoluene in soil. Talanta 2013, 115, 143–149. [Google Scholar] [CrossRef]
- Choodum, A.; Boonsamran, P.; NicDaeid, N.; Wongniramaikul, W. On-site semi-quantitative analysis for ammonium nitrate detection using digital image colourimetry. Sci. Justice 2015, 55, 437–445. [Google Scholar] [CrossRef]
- Jirasek, A. Experimental investigations of polymer gel dosimeters. J. Phys. Conf. Ser. 2007, 56, 23. [Google Scholar] [CrossRef]
- Li, X.; Nakagawa, S.; Tsuji, Y.; Watanabe, N.; Shibayama, M. Polymer gel with a flexible and highly ordered three-dimensional network synthesized via bond percolation. Sci. Adv. 2019, 5, eaax8647. [Google Scholar] [CrossRef]
- Philippova, O.E.; Khokhlov, A.R. 1.13-Polymer Gels, in Polymer Science: A Comprehensive Reference; Matyjaszewski, K., Möller, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2012; pp. 339–366. [Google Scholar]
- Hassan, P.A.; Verma, G.; Ganguly, R. 1-Soft Materials—Properties and Applications, in Functional Materials; Banerjee, S., Tyagi, A.K., Eds.; Elsevier: London, UK, 2012; pp. 1–59. [Google Scholar]
- Zheng, J.; Zeng, R.; Zhang, F.; Kan, J. Effects of sodium carboxymethyl cellulose on rheological properties and gelation behaviors of sodium alginate induced by calcium ions. LWT 2019, 103, 131–138. [Google Scholar] [CrossRef]
- Capanema, N.S.V.; Mansur, A.A.; de Jesus, A.C.; Carvalho, S.M.; de Oliveira, L.C.; Mansur, H.S. Superabsorbent crosslinked carboxymethyl cellulose-PEG hydrogels for potential wound dressing applications. Int. J. Biol. Macromol. 2018, 106, 1218–1234. [Google Scholar] [CrossRef]
- Kono, H. Characterization and properties of carboxymethyl cellulose hydrogels crosslinked by polyethylene glycol. Carbohydr. Polym. 2014, 106, 84–93. [Google Scholar] [CrossRef]
- Choodum, A.; Malathong, K.; NicDaeid, N.; Limsakul, W.; Wongniramaikul, W. A cost effective hydrogel test kit for pre and post blast trinitrotoluene. Forensic Sci. Int. 2016, 266, 202–208. [Google Scholar] [CrossRef]
- Hassanzadeh, J.; Khataee, A.; Bagheri, N.; Lotfi, R. Sensitive chemiluminescence determination method for 2,4,6-trinitrotoluene based on the catalytic activity of amine-capped gold nanoparticles. New J. Chem. 2017, 41, 134–141. [Google Scholar] [CrossRef]
- AOAC. Appendix F: Guidelines for Standard Method Performance Requirements; AOAC: Rockville, MD, USA, 2016. [Google Scholar]
- Jenkins, T.F.; Walsh, M.E. Development of field screening methods for TNT, 2,4-DNT and RDX in soil. Talanta 1992, 39, 419–428. [Google Scholar] [CrossRef]
- Üzer, A.; Erçağ, E.; Apak, R. Selective colorimetric determination of TNT partitioned between an alkaline solution and a strongly basic Dowex 1-X8 anion exchanger. Forensic Sci. Int. 2008, 174, 239–243. [Google Scholar] [CrossRef]
- Erçağ, E.; Üzer, A.; Eren, Ş.; Sağlam, Ş.; Filik, H.; Apak, R. Rapid detection of nitroaromatic and nitramine explosives on chromatographic paper and their reflectometric sensing on PVC tablets. Talanta 2011, 85, 2226–2232. [Google Scholar] [CrossRef] [PubMed]
- Shanmugam, R.; Sornambikai, S.; Karthikeyan, N.S.; Sathiyanarayanan, K.; Kumar, A.S. A Simple Colorimetric Screening of Nitrite Using Iodide in an Acidic pH Solution (Invited article). Austin J. Anal. Pharm. Chem. 2014, 1, 2. [Google Scholar]
- Miura, Y.; Hamada, H. Ion chromatography of nitrite at the ppb level with photometric measurement of iodine formed by post-column reaction of nitrite with iodide. J. Chromatogr. A 1999, 850, 153–160. [Google Scholar] [CrossRef]
- Grgur, B.N.; Gvozdenović, M.; Stevanovic, J.; Jugovic, Z.K.; Trišović, T. Electrochemical oxidation of iodide in aqueous solution. Chem. Eng. J. 2006, 124, 47–54. [Google Scholar] [CrossRef]
- Rastegarzadeh, S.; Kalantaripour, M. Development of an optical redox chemical sensor for nitrite determination. Química Nova 2012, 35, 766–770. [Google Scholar] [CrossRef][Green Version]
- Peters, K.L.; Corbin, I.; Kaufman, L.M.; Zreibe, K.; Blanes, L.; McCord, B.R. Simultaneous colorimetric detection of improvised explosive compounds using microfluidic paper-based analytical devices (μPADs). Anal. Methods 2015, 7, 63–70. [Google Scholar] [CrossRef]
- Urbansky, E.T. Perchlorate Chemistry: Implications for Analysis and Remediation. Bioremediat. J. 1998, 2, 81–95. [Google Scholar] [CrossRef]
- Nabar, G.M.; Ramachandran, C.R. Quantitative Determination of Perchlorate Ion in Solution. Anal. Chem. 1959, 31, 263–265. [Google Scholar] [CrossRef]
- Urbansky, E.T. Quantitation of Perchlorate Ion: Practices and Advances Applied to the Analysis of Common Matrices. Crit. Rev. Anal. Chem. 2000, 30, 311–343. [Google Scholar] [CrossRef]
- Chemical Confirmation Tests. In Field Confirmation Testing for Suspicious Substances; CRC Press: Boca Raton, FL, USA, 2009; pp. 75–351.
- Li, G.; Yuan, R.; Yang, Z.; Huang, X. A yellow license plate location method based on RGB model of color image and texture of plate. In Proceedings of the Second Workshop on Digital Media and its Application in Museum & Heritages (DMAMH 2007), Chongqing, China, 10–12 December 2007. [Google Scholar]
- Bruce, P.; Minkkinen, P.; Riekkola, M.-L. Practical method validation: Validation sufficient for an analysis method. Microchim. Acta 1998, 128, 93–106. [Google Scholar] [CrossRef]


| Food Coloring | Relative Standard Deviation (n = 96) | |
|---|---|---|
| Microplate Spectrophotometer | Proposed Method | |
| Red | 1.78 | 3.24 |
| Green | 1.67 | 3.61 |
| Yellow | 2.04 | 3.27 |
| Black | 2.20 | 3.59 |
| Explosive Type | Relationships | Calibration Equation | Linear Range | r |
|---|---|---|---|---|
| TNT a | R and C | yR = −(0.51 ± 0.02)x + (149.6 ± 0.8) | 1.0–100 | −0.9965 |
| G and C | yG = −(1.58 ± 0.09)x + (153 ± 2) | 1.0–40 | −0.9937 | |
| B and C | yB = −(1.8 ± 0.1)x + (145 ± 3) | 1.0–60 | −0.9912 | |
| T and C | yT = −(4.2 ± 0.2)x + (451 ± 3) | 2.5–40 | −0.9980 | |
| DNT b | R and C | yR = −(63 ± 2)x + (149.3 ± 0.9) | 0.1–0.9 | −0.9977 |
| G and C | yG = −(52 ± 2)x + (149 ± 1) | 0.1–0.9 | −0.9930 | |
| B and C | yB = −(40 ± 1)x + (144.8 ± 0.7) | 0.1–0.9 | −0.9956 | |
| T and C | yT = −(155 ± 5)x + (443 ± 3) | 0.1–0.9 | −0.9967 | |
| Nitrite a | R and C | yR = −(1.6 ± 0.3)x + (164.9 ± 0.7) | 1.0–5.0 | −0.9589 |
| G and C | yG = −(1.6 ± 0.4)x + (167 ± 1) | 1.0–5.0 | −0.9132 | |
| B and C | yB = −(10.5 ± 0.3)x + (165.2 ± 0.7) | 1.0–5.0 | −0.9990 | |
| T and C | yT = −(13.6 ± 0.8)x + (497 ± 2) | 1.0–5.0 | −0.9954 | |
| Perchlorate b | R and C | yR = (122 ± 9)x − (46 ± 7) | 0.5–0.9 | 0.9914 |
| G and C | yG = (116 ± 24)x + (15 ± 10) | 0.2–0.6 | 0.9379 | |
| B and C | yB = (50 ± 12)x + (110 ± 5) | 0.2–0.6 | 0.9138 | |
| T and C | yT = (218 ± 32)x + (126 ± 17) | 0.3–0.7 | 0.9673 |
| Explosive Type | Relationships | Sensitivity | LOD |
|---|---|---|---|
| TNT | T and C | −4.2 ± 0.2 a′ | 2.28 a |
| DNT | T and C | −155 ± 5 b′ | 0.0523 b |
| Nitrite | T and C | −13.6 ± 0.8 a’ | 0.512 a |
| Perchlorate | R and C | 122 ± 9 b’ | 0.163 b |
| Explosive | Reagent Test | ||
|---|---|---|---|
| KOH | H2SO4/KI | Methylene Blue | |
| A | + | − | − |
| B | + | − | − |
| C | − | + | − |
| D | − | − | + |
| A + B | purple (mixed) | − | − |
| A + C | + | − | − |
| A + D | + | − | + |
| B + C | + | − | − |
| B + D | + | − | + |
| C + D | − | + | + |
| A + B + C | purple (mixed) | − | − |
| A + B + D | purple (mixed) | − | + |
| A + C + D | + | − | + |
| B + C + D | + | − | + |
| A + B + C + D | purple (mixed) | − | + |
| Interference Species | TNT (10 mg L−1) | DNT (0.5 mg mL−1) | Nitrite (NO2−) (2 mg L−1) | Perchlorate (ClO4−) (0.7 mg mL−1) | ||||
|---|---|---|---|---|---|---|---|---|
| Tolerance Ratio | %Relative Error | Tolerance Ratio | %Relative Error | Tolerance Ratio | %Relative Error | Tolerance Ratio | %Relative Error | |
| Urea | 600 | +2.95 | 500 | −1.73 | 5000 | +2.95 | 250 | −3.51 |
| Zn2+ | 400 | −4.76 | 500 | −2.41 | 5000 | +3.50 | 500 | −0.53 |
| Ba2+ | 200 | −2.83 | 50 | −0.71 | 5000 | −3.68 | 25 | −3.30 |
| Cu2+ | 80 | +2.46 | 5 | −4.23 | 400 | −1.67 | 500 | −2.34 |
| Pb2+ | 80 | −2.90 | 5 | −0.35 | 50 | −4.23 | 50 | −4.21 |
| Ni2+ | 50 | +0.37 | 25 | −4.89 | 5000 | −0.94 | 25 | −4.74 |
| Mg2+ | 50 | +0.19 | 500 | −0.53 | 5000 | −1.31 | 100 | −4.35 |
| Fe2+ | 20 | −4.53 | 5 | −4.08 | 150 | −4.60 | 100 | −4.89 |
| Sb3+ | 10 | +0.81 | 2 | +3.65 | 500 | −1.16 | 1 | −0.73 |
| SO42− | 400 | −4.76 | 500 | −2.41 | 5000 | +3.50 | 500 | −0.53 |
| Cl− | 200 | −2.83 | 500 | −0.53 | 5000 | −3.68 | 100 | −4.35 |
| NO3− | 80 | −2.90 | 5 | −0.35 | 50 | −4.23 | 50 | −4.21 |
| Sample | Explosives | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| TNT (mg L−1) | DNT (mg mL−1) | Nitrite (mg L−1) | Perchlorate (mg mL−1) | |||||||||
| Add | Found | Recovery | Add | Found | Recovery | Add | Found | Recovery | Add | Found | Recovery | |
| 1 | 10 | 9.8 ± 0.9 | 98 ± 4 | 0.25 | 0.25 ± 0.02 | 99 ± 3 | 1.5 | 1.6 ± 0.1 | 103.4 ± 0.6 | 0.55 | 0.61 ± 0.05 | 111 ± 4 |
| 15 | 12.4 ± 0.7 | 83 ± 3 | 0.30 | 0.30 ± 0.01 | 101 ± 2 | 2.0 | 2.0 ± 0.3 | 100 ± 4 | 0.60 | 0.59 ± 0.01 | 98 ± 1 | |
| 20 | 20.3 ± 0.6 | 102 ± 3 | 0.35 | 0.36 ± 0.01 | 103 ± 1 | 2.5 | 2.4 ± 0.4 | 98 ± 4 | 0.65 | 0.56 ± 0.00 | 87 ± 0 | |
| 25 | 22 ± 1 | 89 ± 5 | 0.40 | 0.40 ± 0.04 | 101 ± 5 | 3.0 | 3.1 ± 0.4 | 104 ± 5 | 0.70 | 0.58 ± 0.04 | 83 ± 3 | |
| 30 | 27.2 ± 0.6 | 91 ± 2 | 0.45 | 0.43 ± 0.02 | 97 ± 3 | 3.5 | 3.6 ± 0.2 | 102 ± 3 | 0.75 | 0.73 ± 0.04 | 97 ± 3 | |
| 2 | 10 | 8.7 ± 0.2 | 87 ± 1 | 0.25 | 0.25 ± 0.02 | 100 ± 2 | 1.5 | 1.5 ± 0.4 | 98 ± 4 | 0.55 | 0.65 ± 0.09 | 118 ± 3 |
| 15 | 14.5 ± 0.7 | 97 ± 3 | 0.30 | 0.31 ± 0.03 | 104 ± 3 | 2.0 | 2.0 ± 0.2 | 99 ± 2 | 0.60 | 0.61 ± 0.06 | 102 ± 4 | |
| 20 | 18 ± 1 | 90 ± 5 | 0.35 | 0.35 ± 0.07 | 99 ± 8 | 2.5 | 2.6 ± 0.3 | 105 ± 4 | 0.65 | 0.62 ± 0.04 | 96 ± 3 | |
| 25 | 23.0 ± 0.8 | 92 ± 4 | 0.40 | 0.39 ± 0.05 | 96 ± 6 | 3.0 | 3.1 ± 0.4 | 105 ± 4 | 0.70 | 0.59 ± 0.04 | 84 ± 3 | |
| 30 | 31.8 ± 0.6 | 106 ± 2 | 0.45 | 0.45 ± 0.03 | 99 ± 4 | 3.5 | 3.3 ± 0.1 | 94 ± 1 | 0.75 | 0.61 ± 0.01 | 81.5 ± 0.6 | |
| 3 | 10 | 8 ± 1 | 84 ± 5 | 0.25 | 0.25 ± 0.01 | 101 ± 2 | 1.5 | 1.6 ± 0.4 | 106 ± 5 | 0.55 | 0.62 ± 0.04 | 113 ± 3 |
| 15 | 13 ± 1 | 86 ± 5 | 0.30 | 0.31 ± 0.02 | 104 ± 2 | 2.0 | 2.2 ± 0.3 | 108 ± 2 | 0.60 | 0.64 ± 0.03 | 106 ± 2 | |
| 20 | 18.3 ± 0.3 | 91 ± 2 | 0.35 | 0.35 ± 0.04 | 101 ± 4 | 2.5 | 2.5 ± 0.1 | 100.2 ± 0.6 | 0.65 | 0.62 ± 0.02 | 95 ± 2 | |
| 25 | 22.5 ± 0.8 | 90 ± 4 | 0.40 | 0.39 ± 0.05 | 98 ± 5 | 3.0 | 2.9 ± 0.4 | 96 ± 4 | 0.70 | 0.59 ± 0.02 | 85 ± 2 | |
| 30 | 28.3 ± 0.7 | 94 ± 3 | 0.45 | 0.42 ± 0.05 | 92 ± 2 | 3.5 | 3.6 ± 0.3 | 102 ± 4 | 0.75 | 0.59 ± 0.01 | 79.3 ± 0.6 | |
| 4 | 10 | 9.6 ± 0.6 | 91 ± 3 | 0.25 | 0.25 ± 0.06 | 100 ± 7 | 1.5 | 1.4 ± 0.2 | 91 ± 3 | 0.55 | 0.62 ± 0.03 | 112 ± 2 |
| 15 | 15 ± 1 | 101 ± 6 | 0.30 | 0.31 ± 0.02 | 102 ± 3 | 2.0 | 2.0 ± 0.4 | 102 ± 4 | 0.60 | 0.64 ± 0.02 | 106 ± 1 | |
| 20 | 18.6 ± 0.2 | 93 ± 1 | 0.35 | 0.36 ± 0.02 | 104 ± 3 | 2.5 | 2.5 ± 0.3 | 99 ± 4 | 0.65 | 0.70 ± 0.1 | 107.0 ± 0.6 | |
| 25 | 24.0 ± 0.8 | 96 ± 4 | 0.40 | 0.39 ± 0.05 | 97 ± 7 | 3.0 | 3.0 ± 0.2 | 98 ± 2 | 0.70 | 0.72 ± 0.02 | 103 ± 2 | |
| 30 | 27.1 ± 0.2 | 90 ± 1 | 0.45 | 0.43 ± 0.06 | 96 ± 7 | 3.5 | 3.2 ± 0.1 | 90.8 ± 0.6 | 0.75 | 0.72 ± 0.06 | 95 ± 4 | |
| 5 | 10 | 9.5 ± 0.9 | 95 ± 4 | 0.25 | 0.25 ± 0.01 | 102 ± 2 | 1.5 | 1.3 ± 0.1 | 89 ± 2 | 0.55 | 0.60 ± 0.05 | 109 ± 3 |
| 15 | 15.5 ± 0.2 | 104 ± 1 | 0.30 | 0.30 ± 0.05 | 102 ± 6 | 2.0 | 1.9 ± 0.3 | 95 ± 4 | 0.60 | 0.59 ± 0.03 | 99 ± 2 | |
| 20 | 19 ± 1 | 95 ± 6 | 0.35 | 0.36 ± 0.03 | 104 ± 3 | 2.5 | 2.6 ± 0.3 | 104 ± 3 | 0.65 | 0.59 ± 0.02 | 92 ± 2 | |
| 25 | 23.6 ± 0.7 | 94 ± 3 | 0.40 | 0.41 ± 0.01 | 104 ± 2 | 3.0 | 3.1 ± 0.2 | 103 ± 3 | 0.70 | 0.66 ± 0.04 | 94 ± 3 | |
| 30 | 28.4 ± 0.9 | 94 ± 4 | 0.45 | 0.45 ± 0.04 | 100 ± 5 | 3.5 | 3.6 ± 0.3 | 104 ± 3 | 0.75 | 0.60 ± 0.08 | 81 ± 6 | |
| 6 | 10 | 10 ± 1 | 103 ± 5 | 0.25 | 0.24 ± 0.03 | 96 ± 3 | 1.5 | 1.5 ± 0.3 | 102 ± 3 | 0.55 | 0.60 ± 0.05 | 109 ± 3 |
| 15 | 13.0 ± 0.9 | 87 ± 4 | 0.30 | 0.31 ± 0.04 | 103 ± 5 | 2.0 | 1.9 ± 0.3 | 94 ± 3 | 0.60 | 0.64 ± 0.04 | 106 ± 3 | |
| 20 | 17.1 ± 0.9 | 85 ± 4 | 0.35 | 0.34 ± 0.03 | 96 ± 3 | 2.5 | 2.4 ± 0.2 | 96 ± 3 | 0.65 | 0.71 ± 0.05 | 109 ± 4 | |
| 25 | 22.6 ± 0.6 | 91 ± 3 | 0.40 | 0.40 ± 0.02 | 101 ± 3 | 3.0 | 3.0 ± 0.4 | 100 ± 5 | 0.70 | 0.80 ± 0.08 | 114 ± 6 | |
| 30 | 27.7 ± 0.6 | 92 ± 3 | 0.45 | 0.45 ± 0.02 | 100 ± 2 | 3.5 | 3.6 ± 0.2 | 102 ± 3 | 0.75 | 0.74 ± 0.09 | 99 ± 7 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thipwimonmas, Y.; Thiangchanya, A.; Phonchai, A.; Thainchaiwattana, S.; Jomsati, W.; Jomsati, S.; Tayayuth, K.; Limbut, W. The Development of Digital Image Colorimetric Quantitative Analysis of Multi-Explosives Using Polymer Gel Sensors. Sensors 2021, 21, 8041. https://doi.org/10.3390/s21238041
Thipwimonmas Y, Thiangchanya A, Phonchai A, Thainchaiwattana S, Jomsati W, Jomsati S, Tayayuth K, Limbut W. The Development of Digital Image Colorimetric Quantitative Analysis of Multi-Explosives Using Polymer Gel Sensors. Sensors. 2021; 21(23):8041. https://doi.org/10.3390/s21238041
Chicago/Turabian StyleThipwimonmas, Yudtapum, Adul Thiangchanya, Apichai Phonchai, Sittipoom Thainchaiwattana, Wachirawit Jomsati, Sunisa Jomsati, Kunanunt Tayayuth, and Warakorn Limbut. 2021. "The Development of Digital Image Colorimetric Quantitative Analysis of Multi-Explosives Using Polymer Gel Sensors" Sensors 21, no. 23: 8041. https://doi.org/10.3390/s21238041
APA StyleThipwimonmas, Y., Thiangchanya, A., Phonchai, A., Thainchaiwattana, S., Jomsati, W., Jomsati, S., Tayayuth, K., & Limbut, W. (2021). The Development of Digital Image Colorimetric Quantitative Analysis of Multi-Explosives Using Polymer Gel Sensors. Sensors, 21(23), 8041. https://doi.org/10.3390/s21238041





