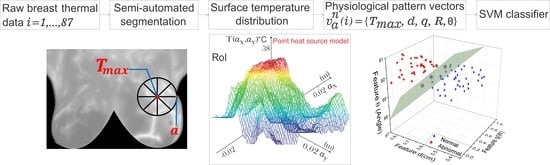
Highly Discriminative Physiological Parameters for Thermal Pattern Classification
Abstract
:1. Introduction
2. Materials and Methods
2.1. Image Database
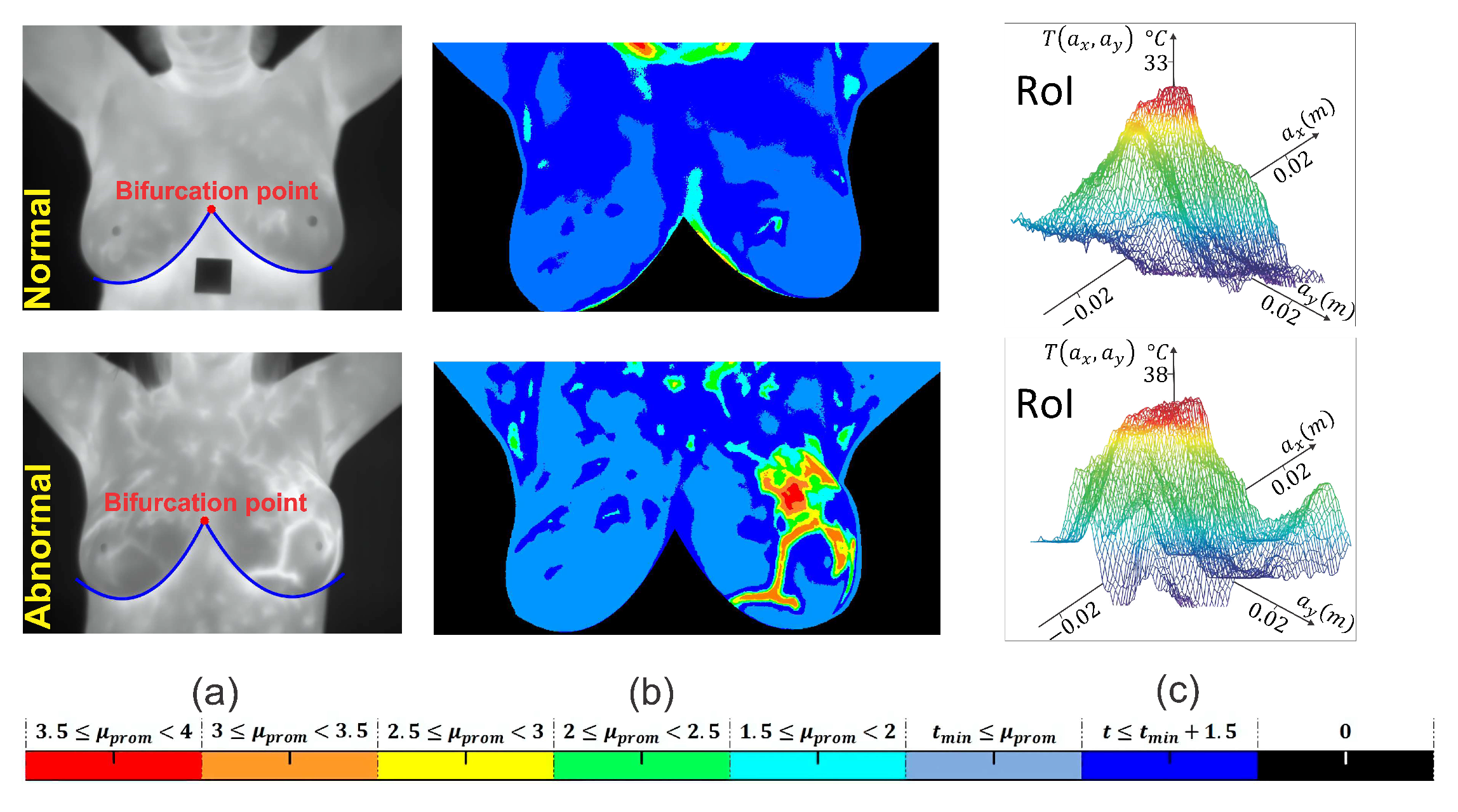
2.2. Segmentation of Breast Thermograms
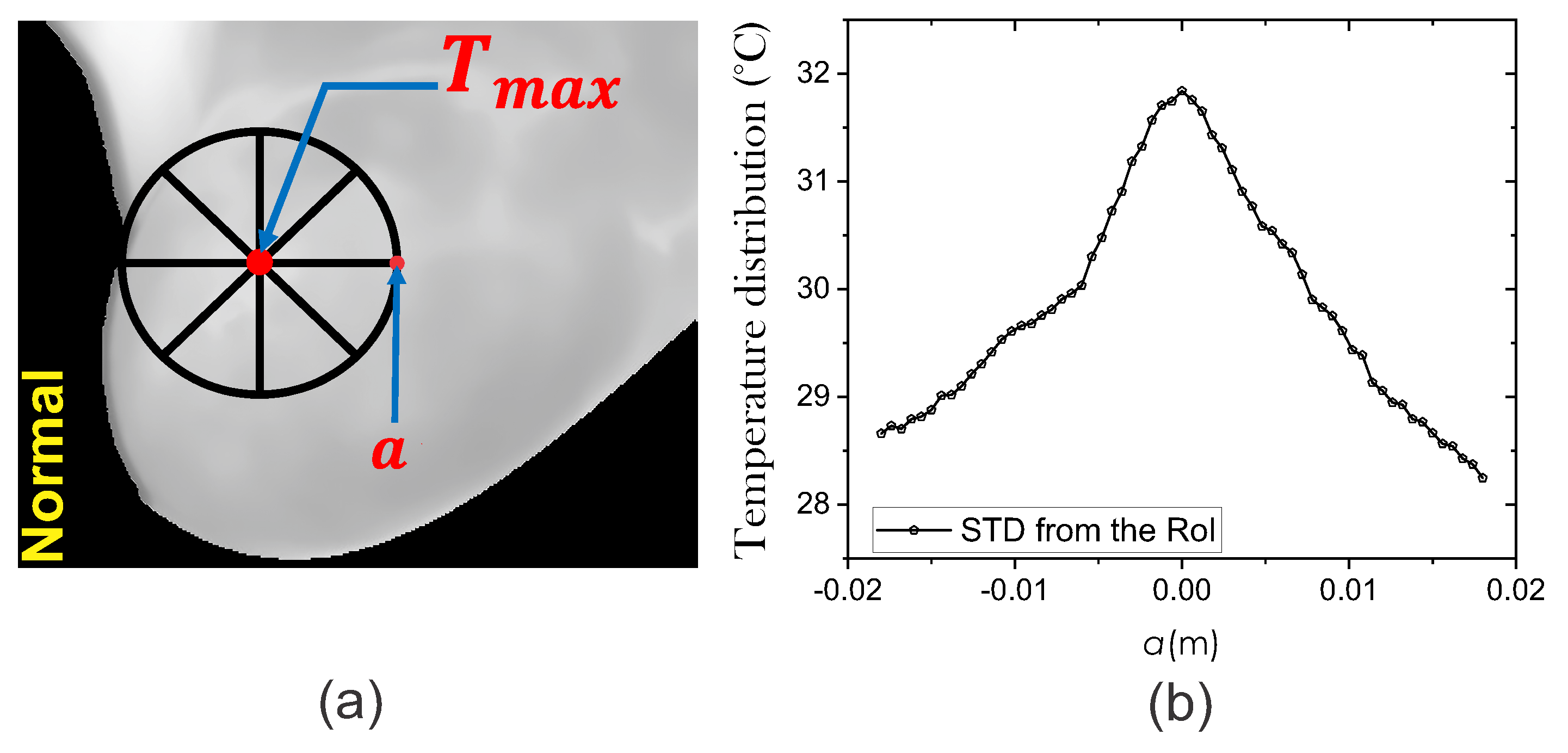
2.3. Heat Source Model: A Mathematical Review
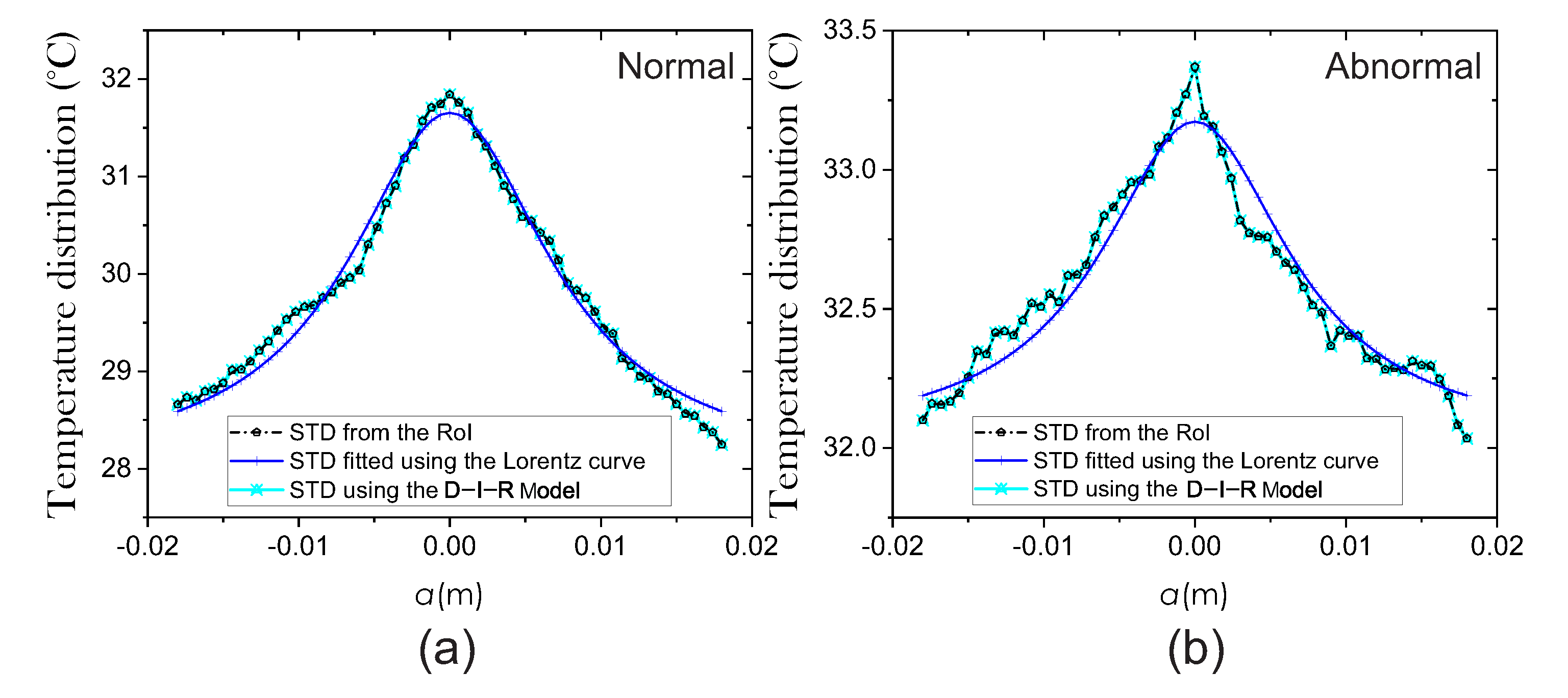
2.3.1. Fitting Method of Lorentz Curve
2.3.2. D-I-R Model
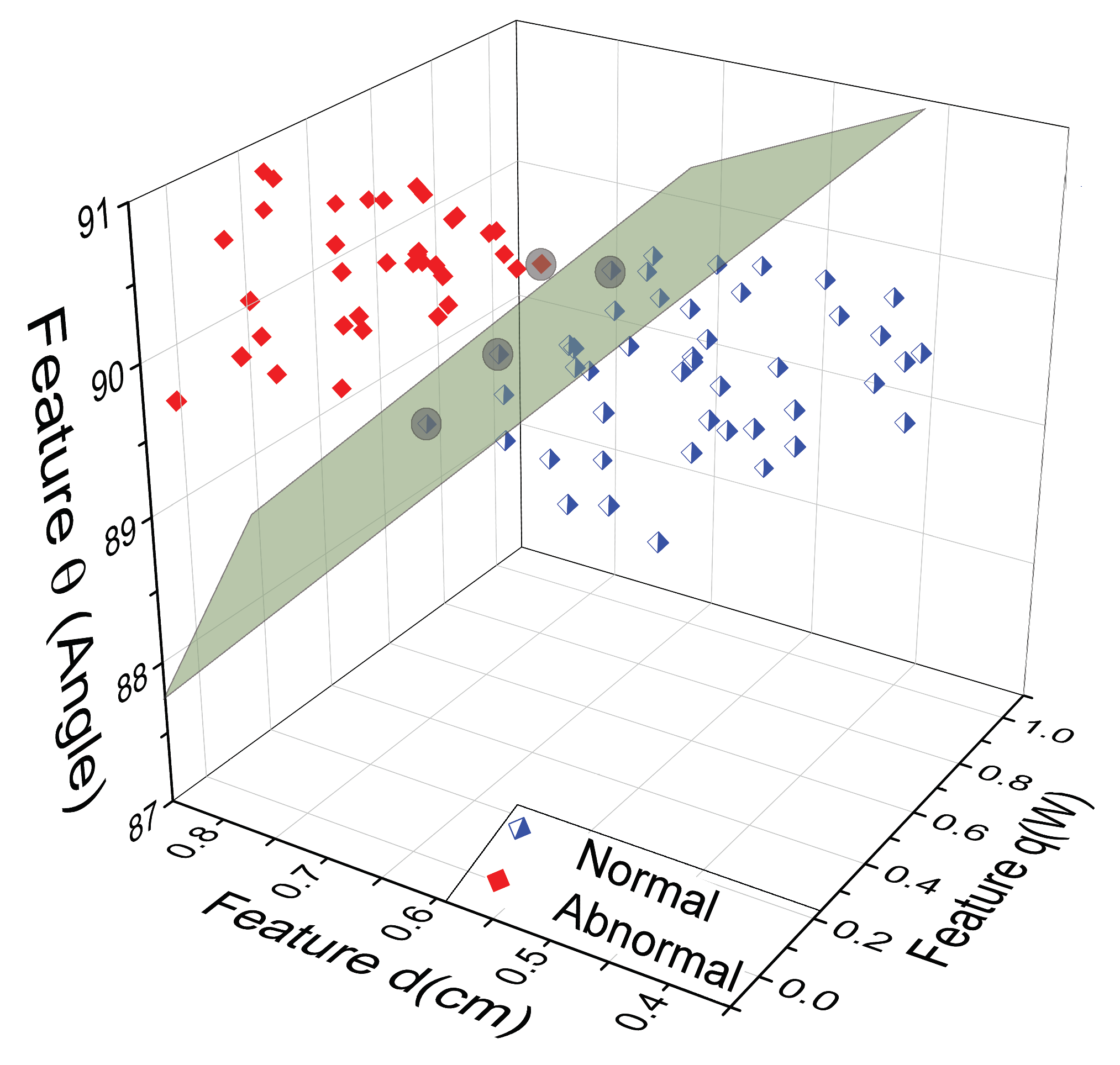
2.4. Thermal Pattern Classification Using SVM
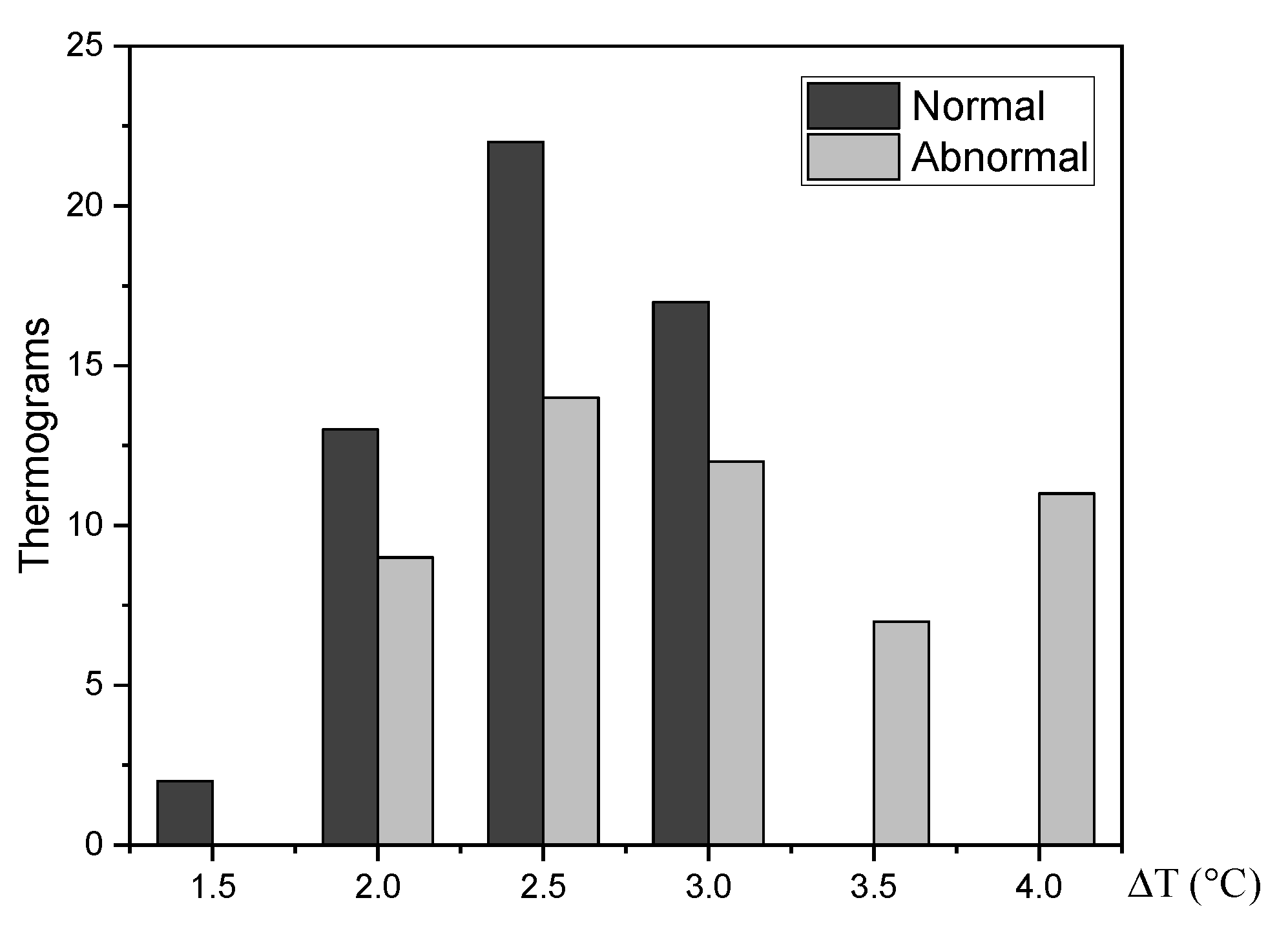
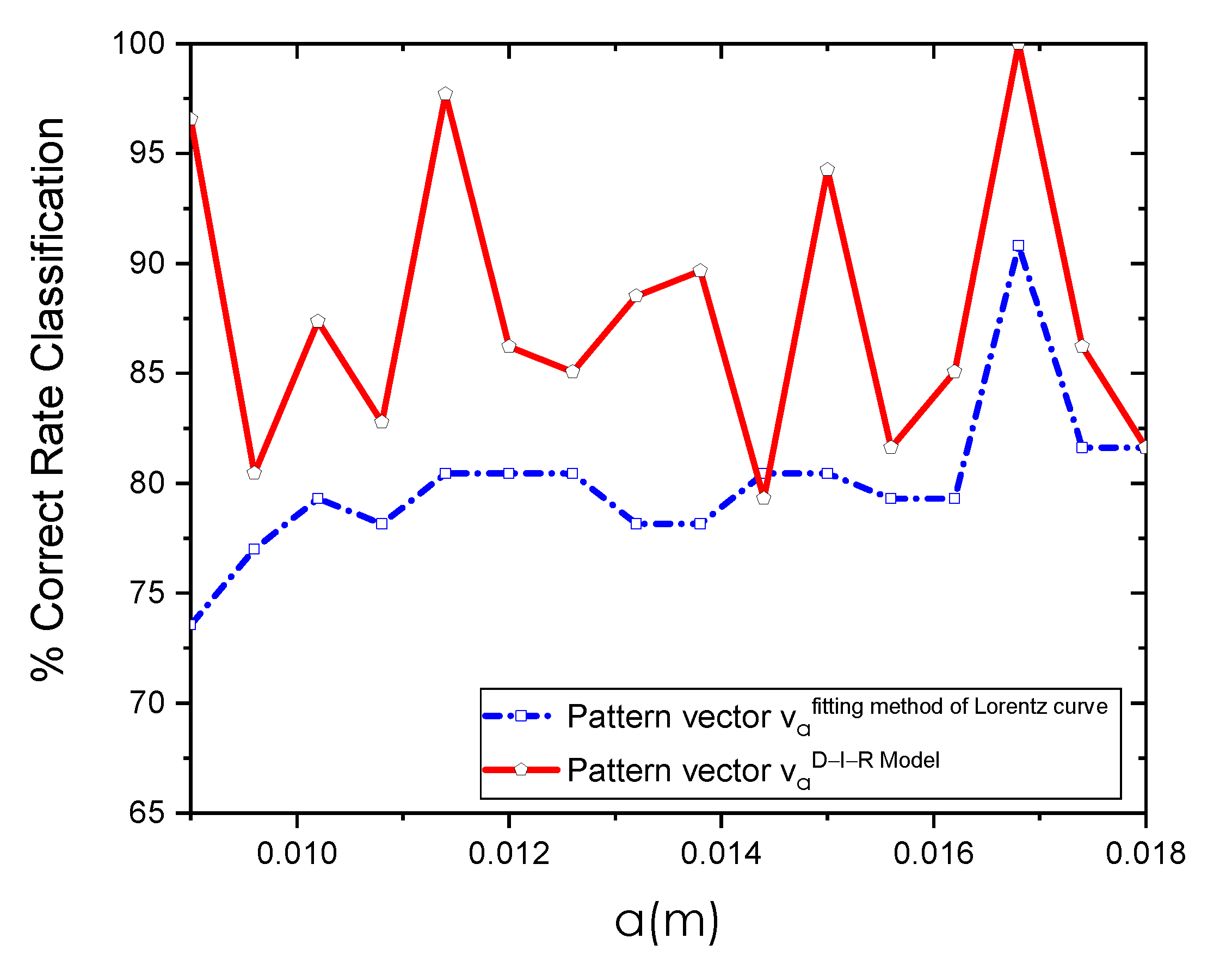
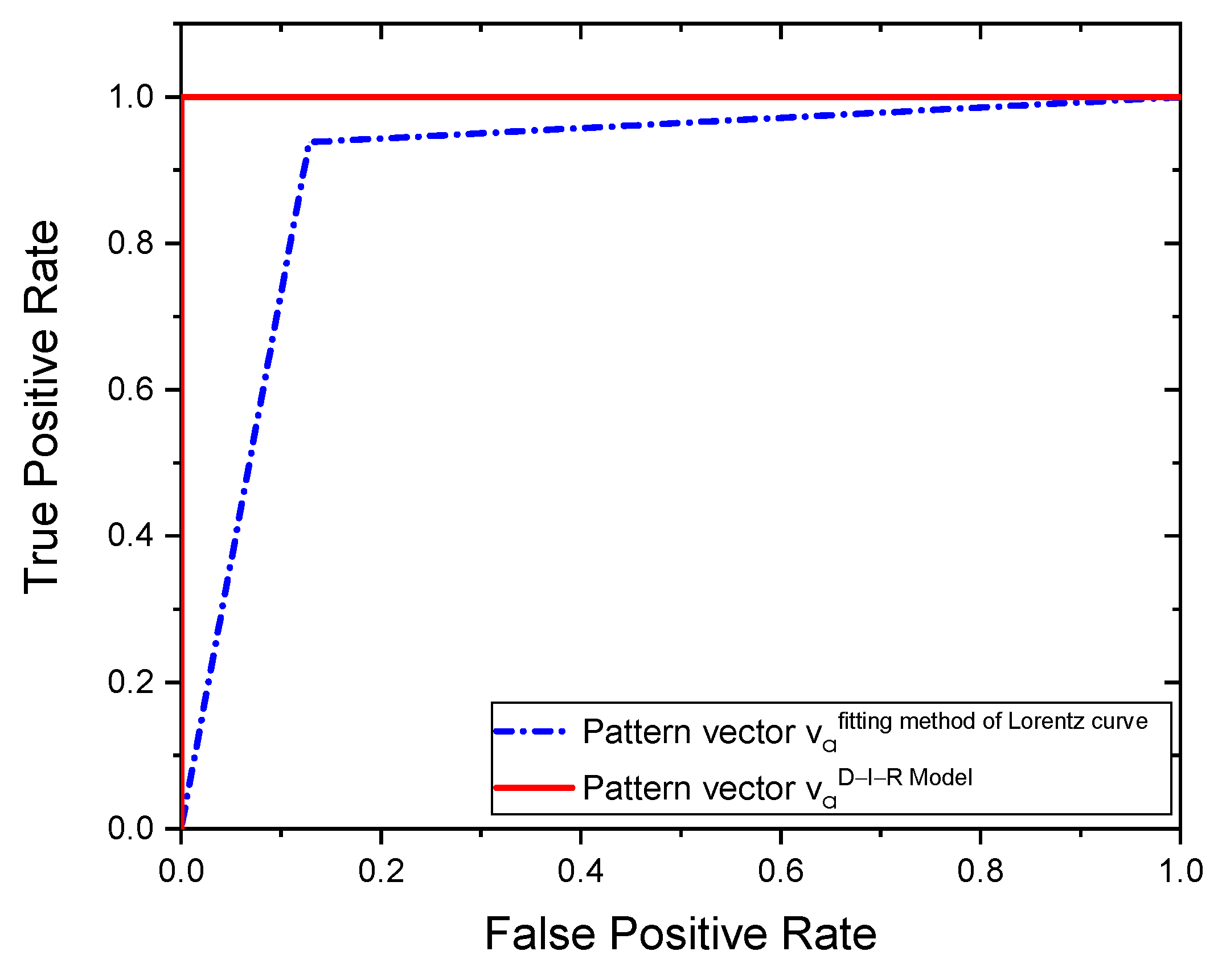
3. Results
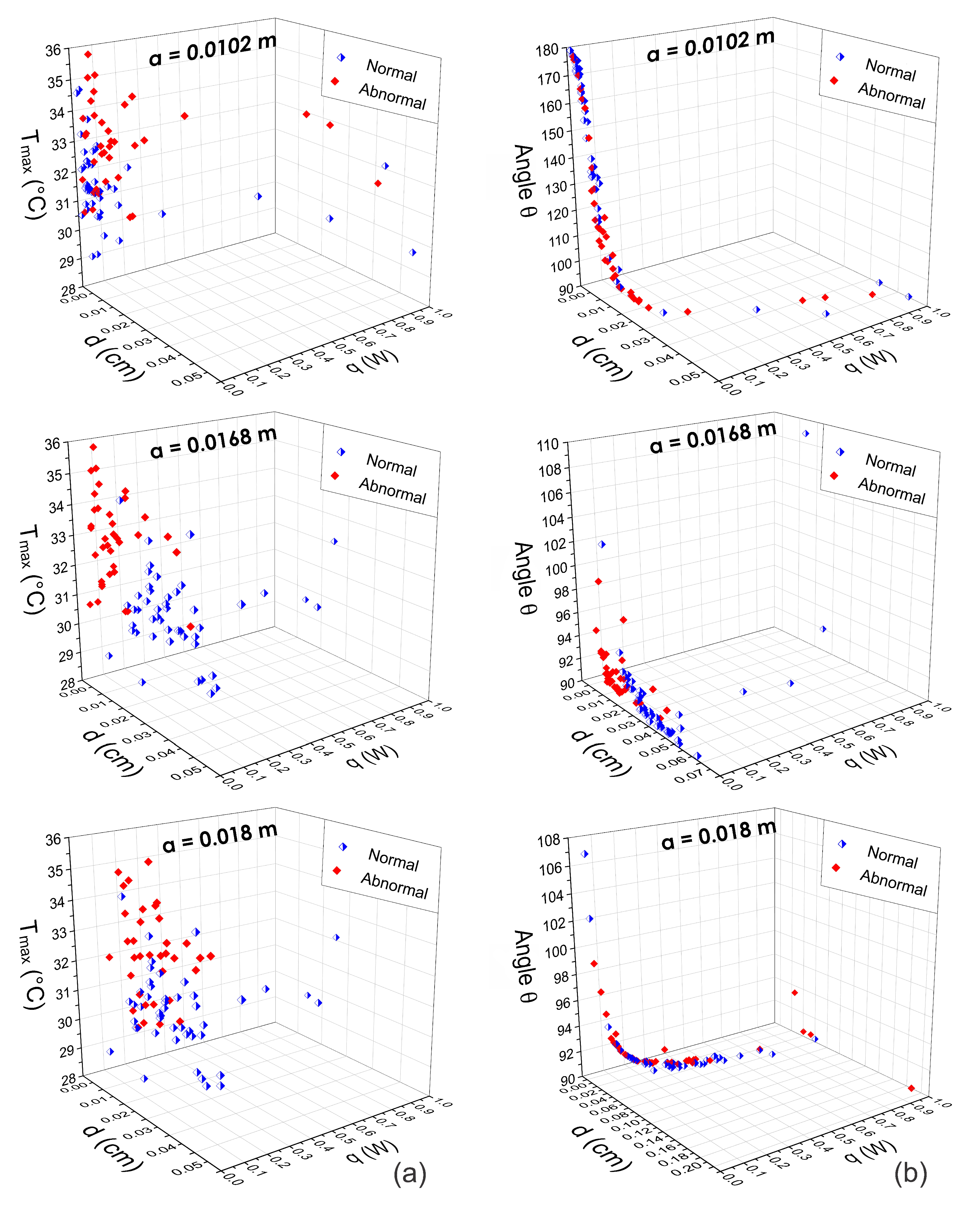
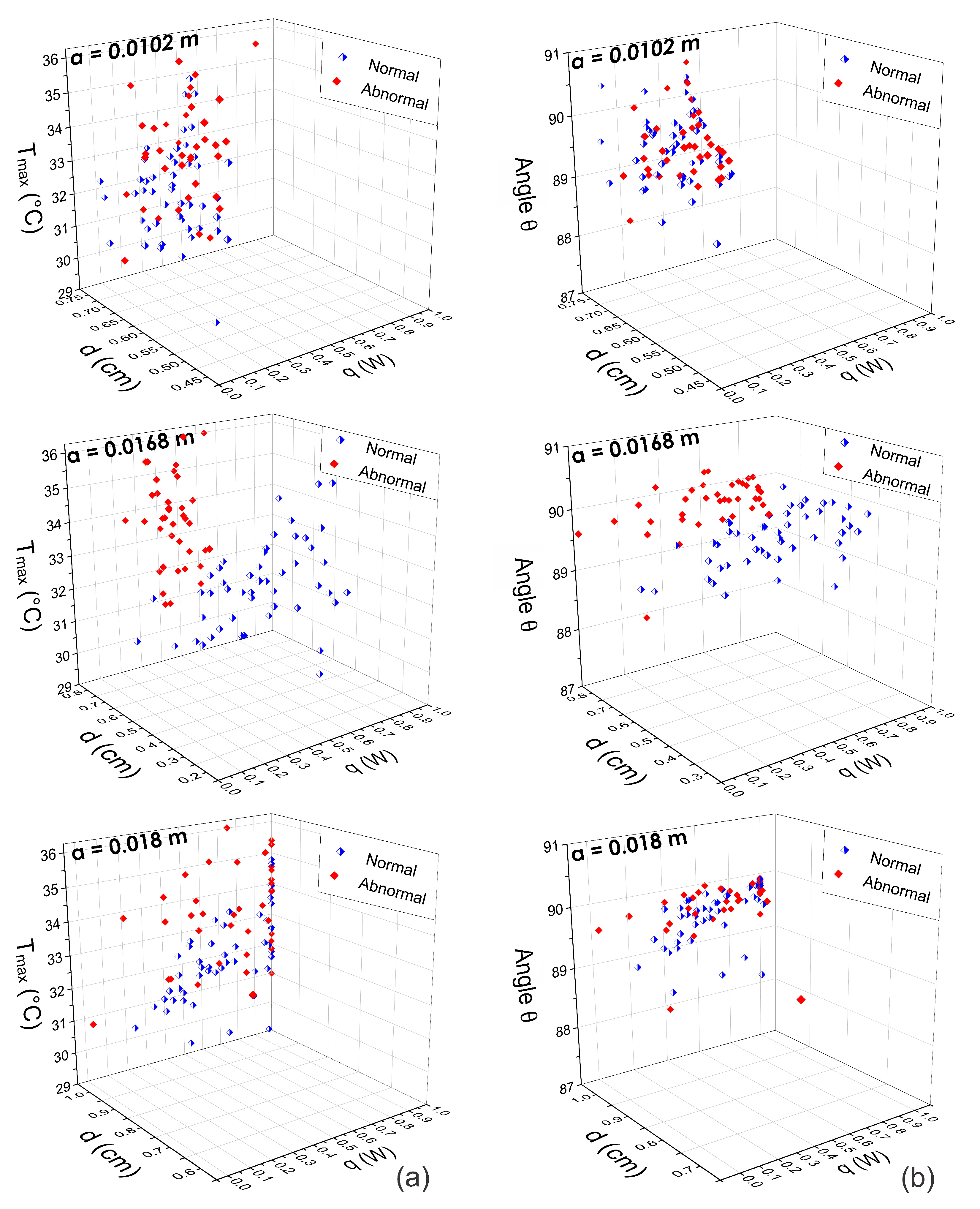
Extraction of the Input Heat Source Parameters
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fernández, C.I.; Bouzas, J.; Arnáiz, J.; Gómez, P.; Piñonosa, C.; García, M.; Sillero, M. Classification of factors influencing the use of infrared thermography in humans: A review. Infrared Phys. Technol. 2015, 71, 28–55. [Google Scholar] [CrossRef]
- Lawson, R.; Chughtai, M.S. Breast Cancer and Body Temperature. Can. Med. Assoc. J. 1963, 88, 68–70. [Google Scholar]
- Silva, L.F.; Saade, D.C.; Sequeiros, G.O.; Silva, A.C.; Paiva, A.C.; Bravo, R.S.; Conci, A. Database for Mastology Research with Infrared Image (DMR-IR). Available online: http://visual.ic.uff.br/dmi (accessed on 1 March 2021).
- Silva, L.F.; Saade, D.C.; Sequeiros, G.O.; Silva, A.C.; Paiva, A.C.; Bravo, R.S.; Conci, A. A new database for breast research with infrared image. J. Med. Imaging Health Inform. 2014, 4, 92–100. [Google Scholar] [CrossRef]
- Pennes, H.H. Analysis of tissue and arterial blood temperatures in the resting human forearm. J. Appl. Physiol. 1998, 85, 5–34. [Google Scholar] [CrossRef] [PubMed]
- Hossain, S.; Mohammadi, F.A.; Nejad, E.T. Neural network approach for the determination of heat source parameters from surface temperature image. In Proceedings of the 2011 24th Canadian Conference on Electrical and Computer Engineering (CCECE), Niagara Falls, ON, Canada, 8–11 May 2011; Volume 5, pp. 1109–1112. [Google Scholar]
- Chunfang, G.; Kaiyang, L.; Shaoping, Z. A Novel Approach of Analyzing the Relation between the Inner Heat Source and the Surface Temperature Distribution in Thermal Texture Maps. In Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Shanghai, China, 1–4 September 2005; pp. 623–626. [Google Scholar]
- Shi, G.; Han, F.; Wang, L.; Liang, C.; Li, K. Q-r curve of thermal tomography and its clinical application on breast tumor diagnosis. Biomed. Opt. Express 2015, 64, 1109–1123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shi, G.; Wang, L.; Han, F.; Liang, C.; Li, K. Diagnosis of breast tumor using thermal tomography q-r curve. J. Biomed. Opt. 2015, 20, 68001. [Google Scholar] [CrossRef] [PubMed]
- Rastgar-Jazi, M.; Mohammadi, F. Parameters sensitivity assessment and heat source localization using infrared imaging techniques. BioMed. Eng. Online 2017, 16, 113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Han, F.; Shi, G.; Liang, C.; Wang, L.; Li, k. A Simple and Efficient Method for Breast Cancer Diagnosis Based on Infrared Thermal Imaging. Cell Biochem. Biophys. 2015, 71, 491–498. [Google Scholar] [CrossRef]
- Minhua, Z.; Qian, C. Study of the Surface Temperature Distribution of the Tissue Affected by the Point Heat Source. In Proceedings of the 2007 1st International Conference on Bioinformatics and Biomedical Engineering, Wuhan, China, 6–8 July 2007; pp. 474–477. [Google Scholar]
- Zermeño, L.O.; Orozco, E.; Toxqui, C.; Padilla, A.; Mejias, Y.N. Caracterización de imágenes en la región espectral del infrarrojo para la detección de lesiones en mama. Rev. Inst. TecnolóGico Cd. JuáRez Acad. J. 2017, 6, 75–82. [Google Scholar]
- Cristianini, N.; Taylor, J.S. An Introduction to Support Vector Machines and Other Kernel-Based Learning Methods; Cambridge University Press: Cambridge, UK, 2000. [Google Scholar]
- Farooq, M.A.; Corcoran, P. Infrared Imaging for Human Thermography and Breast Tumor Classification using Thermal Images. In Proceedings of the 2020 31st Irish Signals and Systems Conference (ISSC), Letterkenny, Ireland, 11–12 June 2020; pp. 1–6. [Google Scholar]
- Silva, T.A.E.D.; Silva, L.F.D.; Muchaluat-Saade, D.C.; Conci, A. A computational method to assist the diagnosis of breast disease using dynamic thermography. Sensors 2020, 20, 3866. [Google Scholar] [CrossRef] [PubMed]
- FLIR Systems. FLIR SC620 Infrared Camera Datasheet FLIR Systems. Available online: http://w1.sayato.com/7040/file/FLIR%20SC620.pdf (accessed on 21 March 2020).
- Castro, R.; Toxqui, C.; Padilla, A.; Solís J., F.; Orozco E., E. Zernike moment invariants for hand vein pattern description from raw biometric data. J. Electron. Imaging 2019, 28, 053019. [Google Scholar]
- Sathish, D.; Kamath, S.; Prasad, K.; Kadavigere, R.; Roshan, J. Asymmetry analysis of breast thermograms using automated segmentation and texture features. Signal Image Video Process. 2017, 11, 745–752. [Google Scholar] [CrossRef]
- Garduño, R.M.A.; Vega, M.S.G.; Morales, H.L.A.; Osornio, R.R.A. Supportive Noninvasive Tool for the Diagnosis of Breast Cancer Using a Thermographic Camera as Sensor. Sensors 2017, 17, 497. [Google Scholar] [CrossRef] [Green Version]
- Alvarado, L.B.; Toxqui, C.; Hernández, J.A.; Padilla, A. Breast thermography: A non-invasive technique for the detection of lesions. In Proceedings of the Applications of Digital Image Processing XLI, San Diego, CA, USA, 20–23 August 2018; Volume 10752, p. 1075230. [Google Scholar]
- STD Plugin Computer Vision Laboratory. Available online: https://sites.google.com/view/lvc-upt/inicio (accessed on 1 October 2021).
- Cortes, C.; Vapnik, V. Support-vector networks. Mach. Learn. 1995, 20, 273–297. [Google Scholar] [CrossRef]
- Karatzoglou, A.; Meyer, D.; Hornik, K. Support vector machines in R. J. Stat. Softw. 2006, 15, 1–28. [Google Scholar] [CrossRef] [Green Version]
- Lin, W.; Yuan, X. Classification of in vivo autofluorescence spectra using support vector machines. J. Biomed. Opt. 2004, 9, 180–186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Castro, J.; Toxqui, C.; Manriquez, F.; Orozco, E.; Padilla, A.; Sánchez, J.J. Detecting Jaundice by using digital image processing. In Proceedings of the Three-Dimensional and Multidimensional Microscopy: Image Acquisition and Processing XXI, San Francisco, CA, USA, 1–6 February 2014; Volume 8949, p. 89491U. [Google Scholar]
- Steel, R.G.; Torrie, J.H. Principles and Procedures of Statistics with Special Reference to the Biological Sciences; McGraw Hill: New York, NY, USA, 1960; pp. 187–287. [Google Scholar]
- Devi, R.R.; Anandhamala, G.S. Analysis of breast thermograms using asymmetry in infra-mammary curves. J. Med. Syst. 2019, 43, 146. [Google Scholar] [CrossRef] [PubMed]
- Acharya, U.R.; Ng, E.Y.K.; Tan, J.H.; Sree, S.V.; Saade, D.M.; Conci, A. Thermography Based Breast Cancer Detection Using Texture Features and Support Vector Machine. J. Med. Syst. 2012, 36, 1503–1510. [Google Scholar] [CrossRef] [PubMed]
- Vartika, M.; Rath, S.K. Detection of breast cancer tumours based on feature reduction and classification of thermograms. Quant. Infrared Thermogr. J. 2020, 18, 300–313. [Google Scholar]
- Gogoi, U.R.; Majumdar, G.; Bhowmikl, M.K.; Ghoshl, A.K.; Bhattacharjee, D. Breast abnormality detection through statistical feature analysis using infrared thermograms. In Proceedings of the 2015 International Symposium on Advanced Computing and Communication (ISACC), Silchar, India, 14–15 September 2015; pp. 258–265. [Google Scholar]
- Suganthi, S.S.; Ramakrishnan, S. Analysis of breast thermograms using gabor wavelet anisotropy index. J. Med. Syst. 2014, 38, 101. [Google Scholar] [CrossRef] [PubMed]
- Alvarado, L.B.; Delgadillo, M.; Toxqui, C.; Padilla, A.; Castro, R.; Arreola, M.M. Fractal analysis for classification of breast lesions. In Proceedings of the Current Developments in Lens Design and Optical Engineering XX, San Diego, CA, USA, 12 August 2019; SPIE: Bellingham, WA, USA, 2019; Volume 11104. [Google Scholar]
- Resmini, R.; Silva, L.; Araujo, A.S.; Medeiros, P.; Saade, D.M.; Conci, A. Combining Genetic Algorithms and SVM for Breast Cancer Diagnosis Using Infrared Thermography. Sensors 2021, 21, 4802. [Google Scholar] [CrossRef] [PubMed]




| T (°C) | Surface temperature distribution. |
| (Kg/m3) | Biological tissue’s density. |
| c (J/Kg · °C) | Thermal capacity of biological tissue. |
| k (W/m/°C) | Heat conduction coefficient. |
| (Kg/m3 · s) | Blood perfusion rate. |
| (Kg/m3) | Blood density. |
| (J/Kg · °C) | Blood thermal capacity. |
| (°C) | Arterial blood temperature. |
| (W/m3) | Metabolic heat rate. |
| q (W) | Heat source intensity. |
| d (cm) | Heat source depth. |
| R (m) | Radius of spherical heat source. |
| a (m) | Distance from point to an arbitrary point on the body surface. |
| r (m) | Distance from point O to an arbitrary point on the body surface. |
| O | Point heat source position. |
| The hottest spot of the RoI. | |
| (°C) | Maximum temperature. |
| (W/m2 · °C) | Heat exchange coefficient. |
| (°C) | Ambient temperature. |
| , (degrees) | Spherical coordinates. |
| Mean temperature. | |
| Minimum temperature. |
| Image resolution | pixels |
| Pixel size | 45 μm |
| Sensor size | cm × cm |
| Standard temperature range | °C to °C |
| Sensitivity | <0.04 °C |
| Method | Breast Thermograms | R-Squared | CRC | AUC | Optimal Position of the RoI |
|---|---|---|---|---|---|
| D-I-R Model | 87 | 100% | 1 | m | |
| Fitting method of Lorentz curve | 87 | % | m |
| Method | Accuracy | Sensitivity | Specificity |
|---|---|---|---|
| D-I-R model | 100% | 100% | 100% |
| Fitting method of Lorentz curve | % | 87% | 97% |
| Authors | Segmentation | Features Extracted | CRC | Thermograms Number |
|---|---|---|---|---|
| Sathish et al. [19] | The breast is segmented. | Histogram and Gray Level Cooccurrence Matrix (GLCM) -based texture features. | 90% | 80 |
| R. Devi et al. [28] | The left and right breast are separated. | GLCM features and first-order histogram. | 95% | 60 |
| V. Mishra [30] | The breast is segmented. | Gray Level Run Length Matrix (GLRLM) and GLCM. | % | 56 |
| U. R. Gogoi [31] | The breast is segmented. | First-order statistical features. | — | 60 |
| S. S. Suganthi et al. [32] | The breast is segmented. | Anisotropy and orientation measures. | — | 20 |
| R. Resmini et al. [34] | The breast is segmented with different approaches (with and without armpits) to compose four experiments. | GLCM, Local Ternary Pattern, Daubechies Wavelet, Higuchi, Petrosian Fractal, Dimensions, and Hurst Coefficient. | % | 80 |
| Proposed approach | The breast is segmented with a well-defined RoI using SVM. | Physiological pattern vectors = { , q, d, R, }. | 100% | 87 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alvarado-Cruz, L.B.; Toxqui-Quitl, C.; Castro-Ortega, R.; Padilla-Vivanco, A.; Arroyo-Núñez, J.H. Highly Discriminative Physiological Parameters for Thermal Pattern Classification. Sensors 2021, 21, 7751. https://doi.org/10.3390/s21227751
Alvarado-Cruz LB, Toxqui-Quitl C, Castro-Ortega R, Padilla-Vivanco A, Arroyo-Núñez JH. Highly Discriminative Physiological Parameters for Thermal Pattern Classification. Sensors. 2021; 21(22):7751. https://doi.org/10.3390/s21227751
Chicago/Turabian StyleAlvarado-Cruz, Laura Benita, Carina Toxqui-Quitl, Raúl Castro-Ortega, Alfonso Padilla-Vivanco, and José Humberto Arroyo-Núñez. 2021. "Highly Discriminative Physiological Parameters for Thermal Pattern Classification" Sensors 21, no. 22: 7751. https://doi.org/10.3390/s21227751
APA StyleAlvarado-Cruz, L. B., Toxqui-Quitl, C., Castro-Ortega, R., Padilla-Vivanco, A., & Arroyo-Núñez, J. H. (2021). Highly Discriminative Physiological Parameters for Thermal Pattern Classification. Sensors, 21(22), 7751. https://doi.org/10.3390/s21227751