Immunomagnetic Separation Improves the Detection of Mycobacteria by Paper-Based Lateral and Vertical Flow Immunochromatographic Assays
Abstract
:1. Introduction
2. Materials and Methods
2.1. Instrumentation and Materials
2.2. Chemicals and Biochemical
2.3. Oligonucleotides Sequences
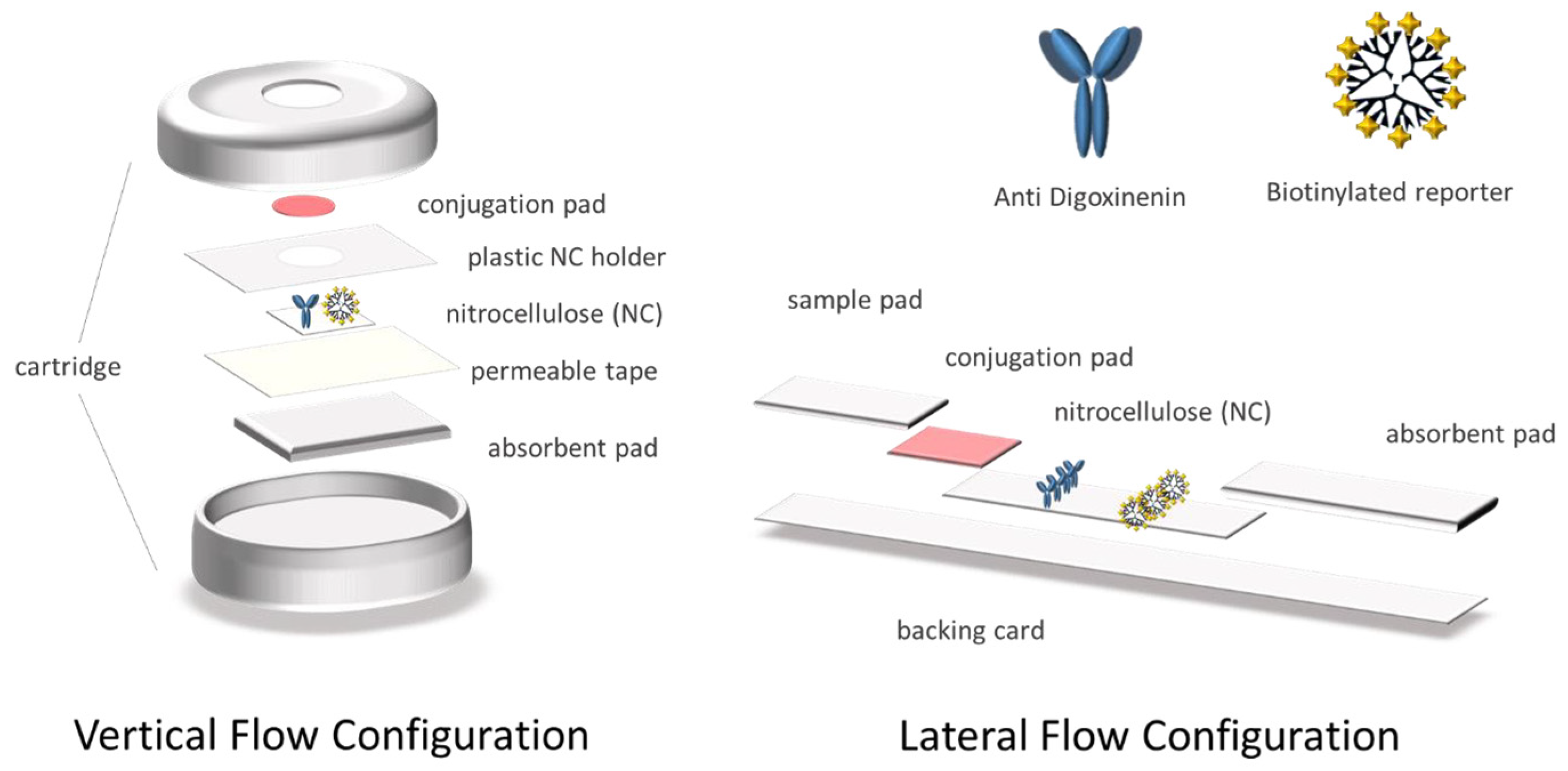
2.4. Preparation of the Devices in Vertical Flow Configuration
2.5. Preparation of Devices in Lateral Flow Configuration
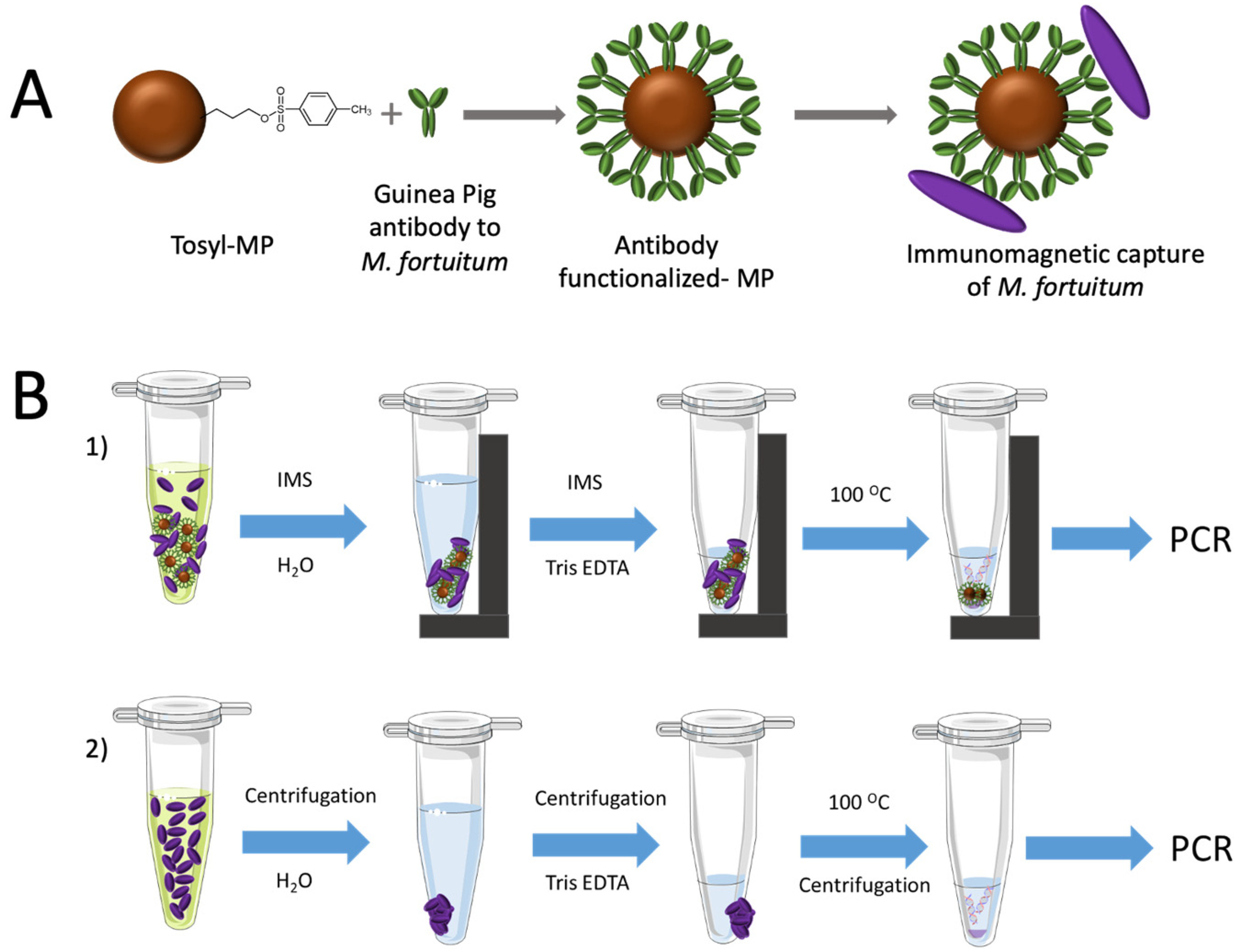
2.6. Covalent Immobilization of Antibodies on Magnetic Particles
2.7. Bacterial Strain and Culture
2.8. Immunomagnetic Separation and DNA Extraction
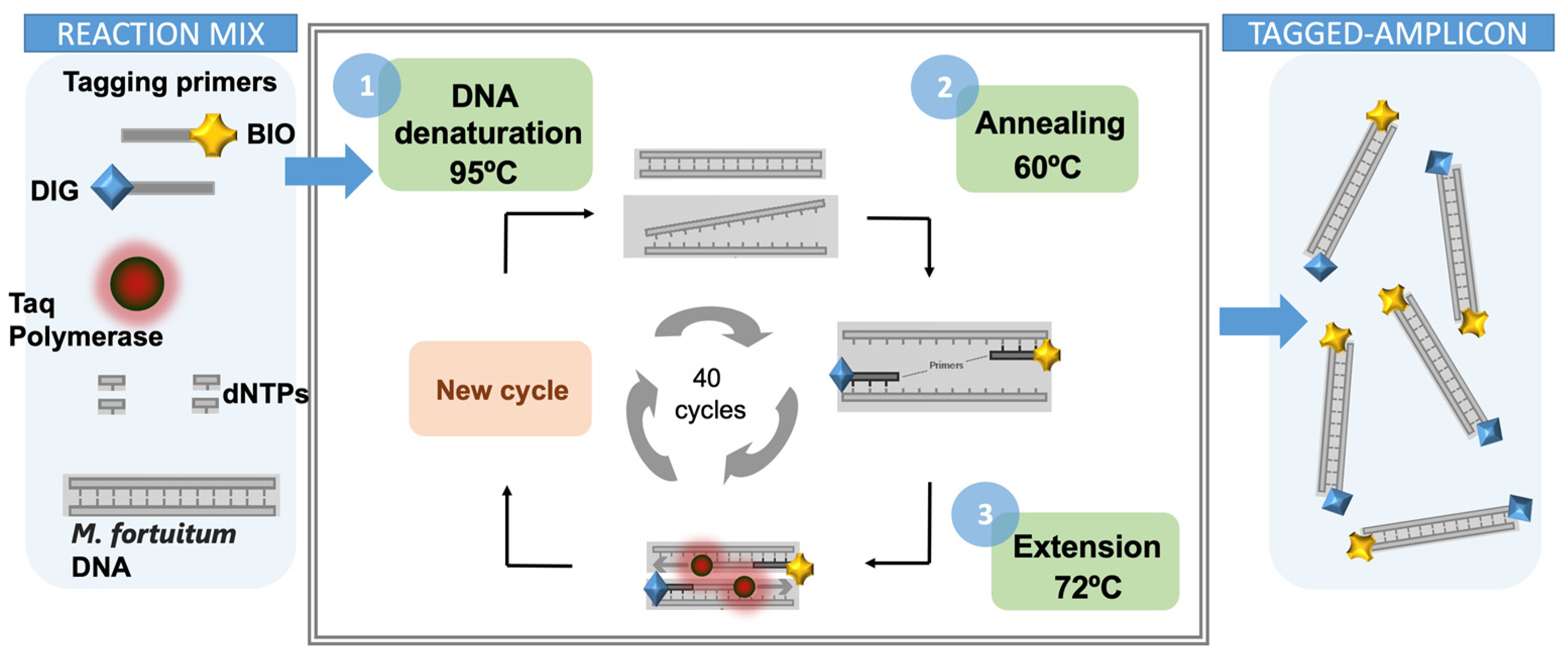
2.9. Double-Tagging PCR for the Amplification of Mycobacteria
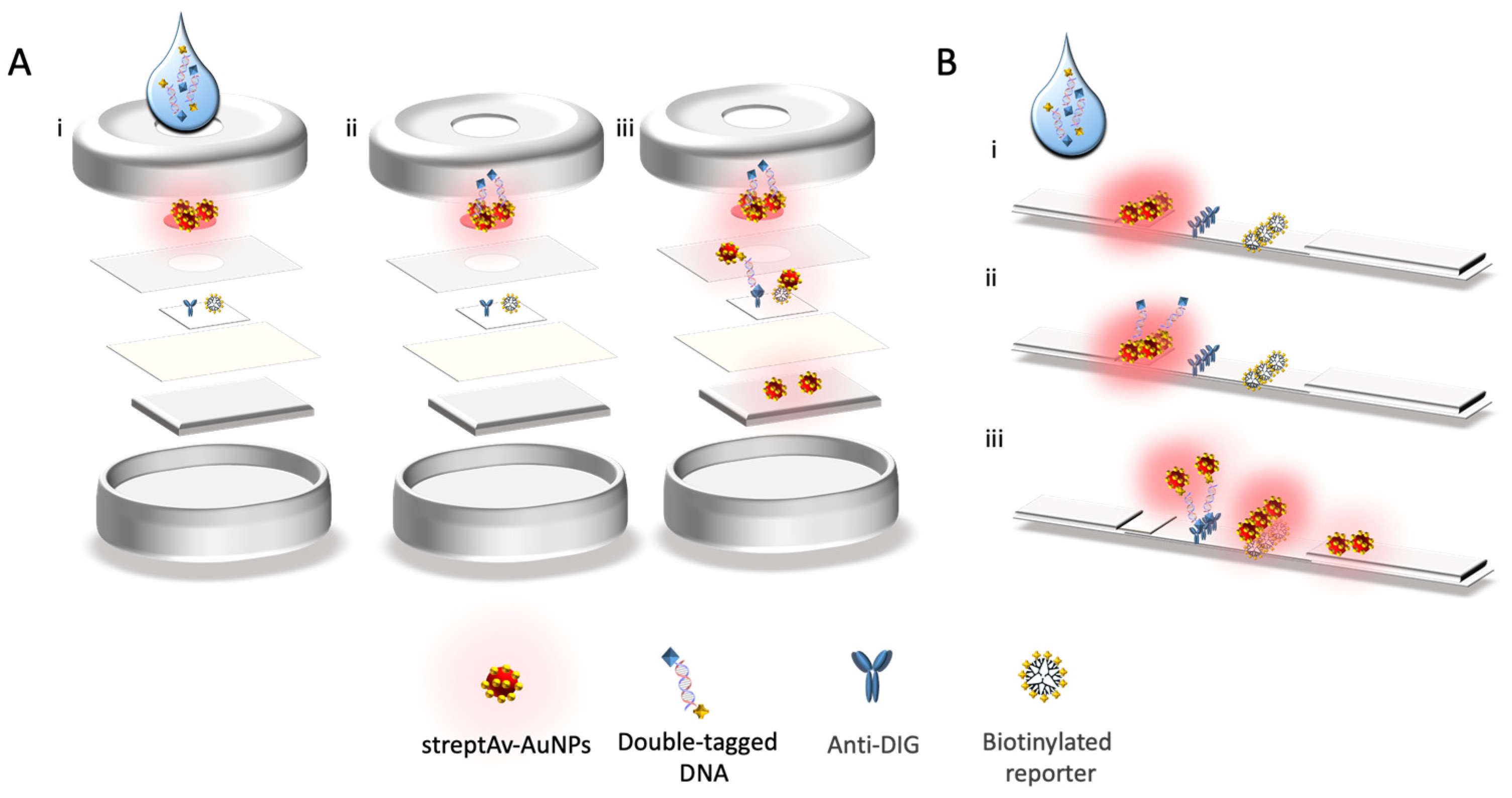
2.10. Nucleic Acid Vertical Flow
2.11. Nucleic acid Lateral Flow
2.12. Data Interpretation and Analysis
3. Results and Discussion
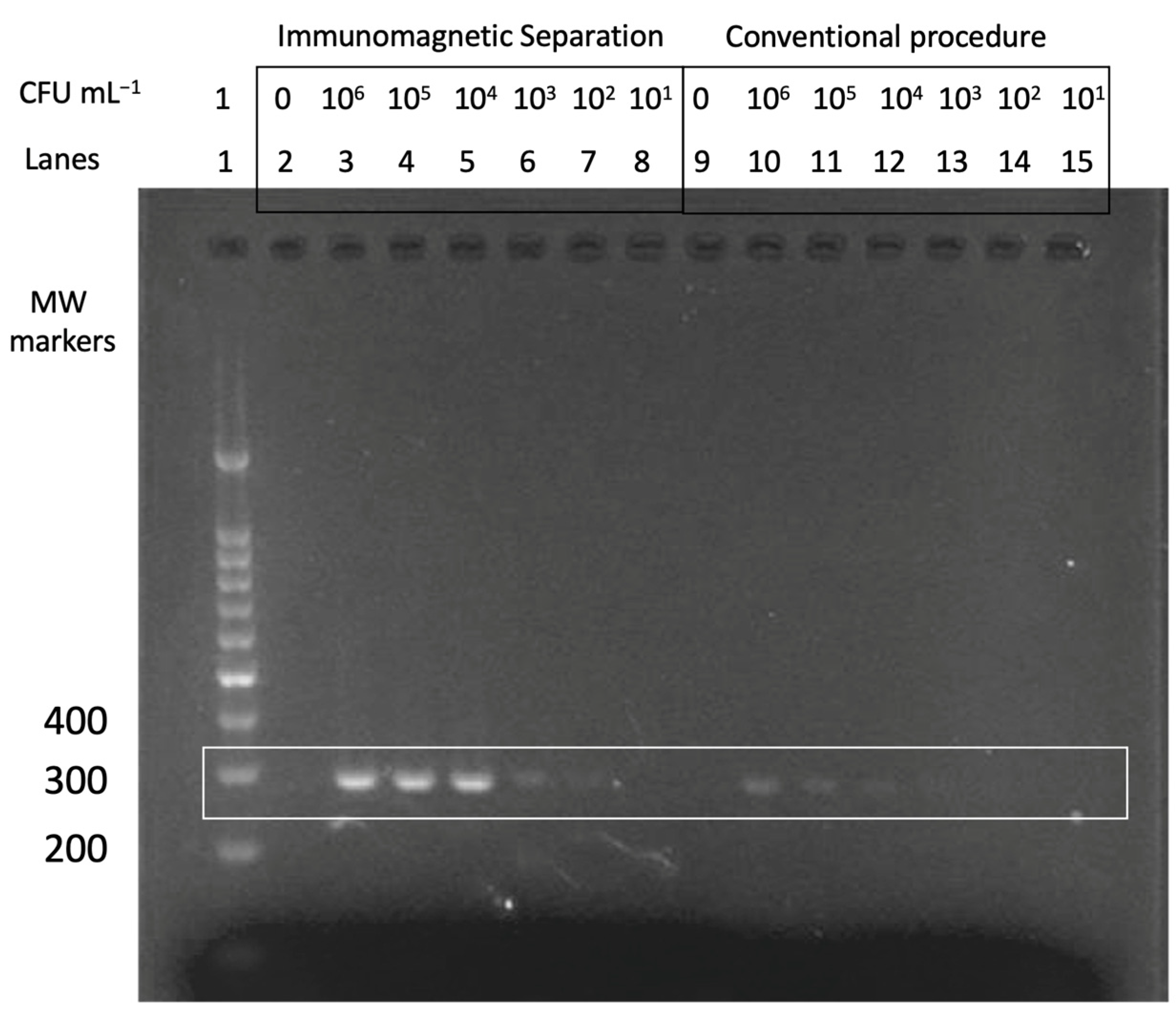
3.1. Double-Tagging PCR for the Amplification of Mycobacteria
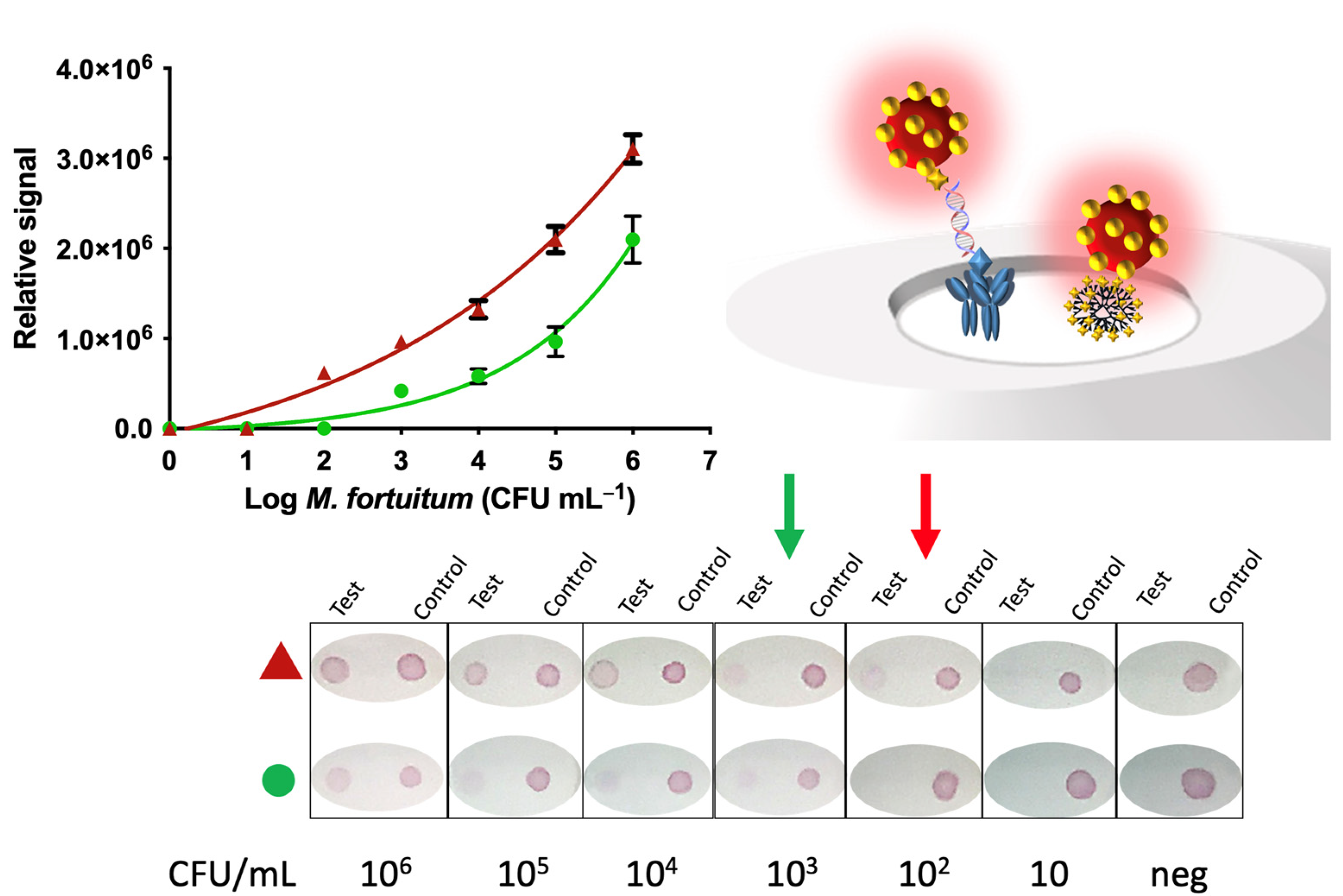
3.2. Nucleic Acid Vertical Flow
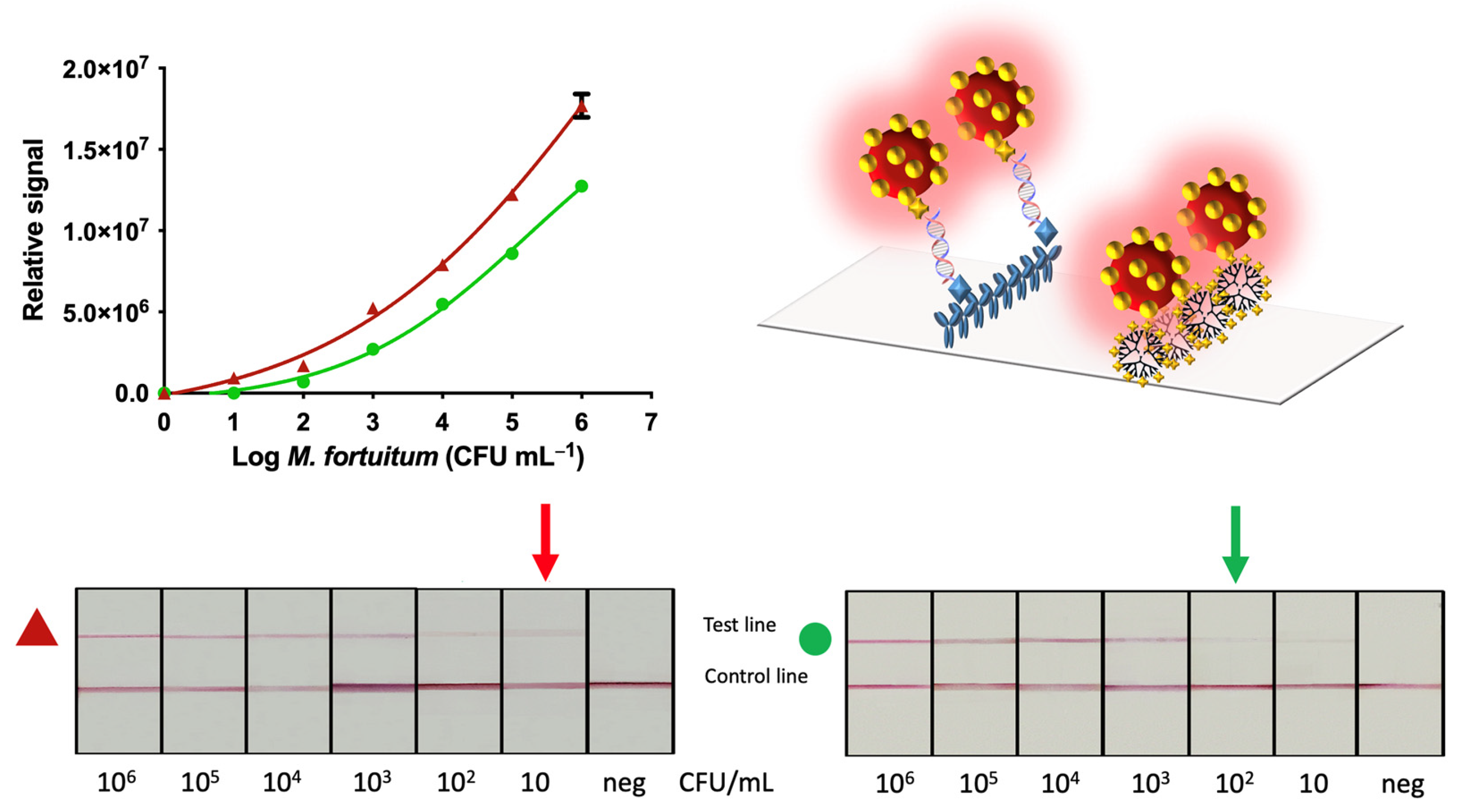
3.3. Nucleic Acid Lateral Flow Immunoassay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chen, H.; Liu, K.; Li, Z.; Wang, P. Point of care testing for infectious diseases. Clin. Chim. Acta 2019, 493, 138–147. [Google Scholar] [CrossRef]
- Primiceri, E.; Chiriacò, M.S.; Notarangelo, F.M.; Crocamo, A.; Ardissino, D.; Cereda, M.; Bramanti, A.P.; Bianchessi, M.A.; Giannelli, G.; Maruccio, G. Key enabling technologies for point-of-care diagnostics. Sensors 2018, 18, 3607. [Google Scholar] [CrossRef] [Green Version]
- Di Serio, F.; Lovero, R.; Leone, M.; De Sario, R.; Ruggieri, V.; Varraso, L.; Pansini, N. Integration between the Tele-Cardiology Unit and the central laboratory: Methodological and clinical evaluation of point-of-care testing cardiac marker in the ambulance. Clin. Chem. Lab. Med. 2006, 44, 768–773. [Google Scholar] [CrossRef]
- Schlacks, S.; Vishkautsan, P.; Butkiewicz, C.; Shubitz, L. Evaluation of a commercially available, point-of-care Coccidioides antibody lateral flow assay to aid in rapid diagnosis of coccidioidomycosis in dogs. Med. Mycol. 2019, 58, 328–332. [Google Scholar] [CrossRef] [PubMed]
- Na, J.Y. Point-of-care hemoglobin A1c testing in postmortem examination. Forensic Sci. Med. Pathol. 2018, 14, 322–326. [Google Scholar] [CrossRef] [PubMed]
- Cummins, B.; Ligler, F.S.; Walker, G.M. Point-of-Care Diagnostics for Niche Applications. Biotechnol. Adv. 2016, 34, 161–176. [Google Scholar] [CrossRef] [Green Version]
- Amerongen, A.; Veen, J.; Arends, H.; Koets, M. Lateral Flow Immunoassay. In Handbook of Immunoassay Technologies. Approaches, Performances, and Applications; Vashist, S., Luong, J., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 157–182. [Google Scholar]
- Anfossi, L.; Giovannoli, C.; Baggiani, C. Introductory Chapter: Rapid Test—Advances in Design, Formats, and Detection Strategies. In Rapid Test. Advances in Design, Format and Diagnostic Applications; Anfonssi, L., Ed.; IntechOpen: London, UK, 2018; p. 13. [Google Scholar]
- Singh, A.T.; Lantigua, D.; Meka, A.; Taing, S.; Pandher, M.; Camci-Unal, G. Paper-based sensors: Emerging themes and applications. Sensors 2018, 18, 2838. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Farrell, B. Evolution in Lateral Flow–Based Immunoassay Systems. In Lateral Flow Immunoassay; Wong, R., Tse, H., Eds.; Humana Press: Totowa, NJ, USA, 2009; pp. 1–33. [Google Scholar]
- Chen, P.; Gates-Hollingsworth, M.; Pandit, S.; Park, A.; Montgomery, D.; Aucoin, D.; Gu, J.; Zenhausern, F. Paper-based Vertical Flow Immunoassay (VFI) for detection of bio-threat pathogens. Talanta 2019, 191, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Posthuma-Trumpie, G.A.; Korf, J.; Van Amerongen, A. Lateral flow (immuno) assay: Its strengths, weaknesses, opportunities and threats. A literature survey. Anal. Bioanal. Chem. 2009, 393, 569–582. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miočević, O.; Cole, C.R.; Laughlin, M.J.; Buck, R.L.; Slowey, P.D.; Shirtcliff, E.A. Quantitative Lateral Flow Assays for Salivary Biomarker Assessment: A Review. Front. Public Health 2017, 5, 133. [Google Scholar] [CrossRef] [Green Version]
- Boum, Y.; Eyangoh, S.; Okomo, M.-C. Beyond COVID-19—Will self-sampling and testing become the norm? Lancet Infect. Dis. 2021, 9, 1194–1195. [Google Scholar] [CrossRef]
- Castilho, M.D.S.; Laube, T.; Yamanaka, H.; Alegret, S.; Pividori, M.I. Magneto Immunoassays for Plasmodium falciparum Histidine-Rich Protein 2 Related to Malaria based on Magnetic Nanoparticles. Anal. Chem. 2011, 83, 5570–5577. [Google Scholar] [CrossRef]
- Carinelli, S.; Martí, M.; Alegret, S.; Pividori, M.I. Biomarker detection of global infectious diseases based on magnetic particles. New Biotechnol. 2015, 32, 521–532. [Google Scholar] [CrossRef]
- Brandão, D.; Liébana, S.; Campoy, S.; Alegret, S.; Pividori, M.I. Immunomagnetic separation of Salmonella with tailored magnetic micro and nanocarriers. A comparative study. Talanta 2015, 143, 198–204. [Google Scholar] [CrossRef]
- Liébana, S.; Lermo, A.; Campoy, S.; Cortés, M.P.; Alegret, S.; Pividori, M.I. Rapid detection of Salmonella in milk by electrochemical magneto-immunosensing. Biosens. Bioelectron. 2009, 25, 510–513. [Google Scholar] [CrossRef]
- Liébana, S.; Lermo, A.; Campoy, S.; Barbé, J.; Alegret, S.; Pividori, M.I. Magneto immunoseparation of pathogenic bacteria and electrochemical magneto genosensing of the double-tagged amplicon. Anal. Chem. 2009, 81, 5812–5820. [Google Scholar] [CrossRef] [PubMed]
- Marks, M.; Millat-Martinez, P.; Ouchi, D.; Roberts, C.h.; Alemany, A.; Corbacho-Monné, M.; Ubals, M.; Tobias, A.; Tebé, C.; Ballana, E.; et al. Transmission of COVID-19 in 282 clusters in Catalonia, Spain: A cohort study. Lancet Infect. Dis. 2021, 21, 629–636. [Google Scholar] [CrossRef]
- Urdea, M.; Penny, L.A.; Olmsted, S.S.; Giovanni, M.Y.; Kaspar, P.; Shepherd, A.; Wilson, P.; Dahl, C.A.; Buchsbaum, S.; Moeller, G.; et al. Requirements for high impact diagnostics in the developing world. Nature 2006, 444 (Suppl. 1), 73–79. [Google Scholar] [CrossRef] [PubMed]
- Marx, V. PCR heads into the field. Nat. Methods. 2015, 12, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Pividori, M.I.; Merkoci, A.; Barbe, J.; Alegret, S. PCR-Genosensor Rapid Test for Detecting Salmonella. Electroanalysis 2003, 15, 1815–1823. [Google Scholar] [CrossRef]
- Lermo, A.; Campoy, S.; Barbé, J.; Hernández, S.; Alegret, S.; Pividori, M.I. In situ DNA amplification with magnetic primers for the electrochemical detection of food pathogens. Biosens. Bioelectron. 2007, 22, 2010–2017. [Google Scholar] [CrossRef] [PubMed]
- Lermo, A.; Zacco, E.; Barak, M.; Delwiche, M.; Campoy, S.; Barbé, J.; Alegret, S.; Pividori, M.I. Towards Q-PCR of pathogenic bacteria with improved electrochemical double-tagged genosensing detection. Biosens. Bioelectron. 2008, 23, 1805–1811. [Google Scholar] [CrossRef]
- Brandão, D.; Liébana, S.; Campoy, S.; Cortés, M.P.; Alegret, S.; Pividori, M.I. Simultaneous electrochemical magneto genosensing of foodborne bacteria based on triple-tagging multiplex amplification. Biosens. Bioelectron. 2015, 74, 652–659. [Google Scholar] [CrossRef]
- Vidal, L.; Ben Aissa, A.; Salabert, J.; Jara, J.J.; Vallribera, A.; Pividori, M.I.; Sebastian, R.M. Biotinylated phosphorus dendrimers as control line in nucleic acid lateral flow tests. Biomacromolecules 2020, 21, 1315–1323. [Google Scholar] [CrossRef] [PubMed]
- BenAissa, A.; Jara, J.J.; Sebastián, R.M.; Vallribera, A.; Campoy, S.; Pividori, M.I. Comparing nucleic acid lateral fl ow and electrochemical genosensing for the simultaneous detection of foodborne pathogens. Biosens. Bioelectron. 2017, 88, 265–272. [Google Scholar] [CrossRef]
- Porras, J.C.; Bernuz, M.; Marfa, J.; Pallares-Rusiñol, A.; Martí, M.; Pividori, M.I. Comparative Study of Gold and Carbon Nanoparticles in Nucleic Acid Lateral Flow Assay. Nanomaterials 2021, 11, 741. [Google Scholar] [CrossRef]
- Roshdi Maleki, M.; Moaddab, S.R.; Samadi Kafil, H. Hemodialysis waters as a source of potentially pathogenic mycobacteria (PPM). Desalin. Water Treat. 2019, 152, 168–173. [Google Scholar] [CrossRef]
- Brugnera, M.F.; Bundalian, R., Jr.; Laube, T.; Julián, E.; Luquin, M.; Zanoni, M.V.B.; Pividori, M.I. Magneto-actuated immunoassay for the detection of Mycobacterium fortuitum in hemodialysis water. Talanta 2016, 153, 38–44. [Google Scholar] [CrossRef] [Green Version]
- Richardson, E.T.; Samson, D.; Banaei, N. Rapid identification of Mycobacterium tuberculosis and nontuberculous mycobacteria by multiplex, real-time PCR. J. Clin. Microbiol. 2009, 47, 1497–1502. [Google Scholar] [CrossRef] [Green Version]
- Dilek, Ç.A.M. Lateral Flow Assay for Salmonella Detection and Potential Reagents. In New Insight into Brucella Infection and Foodborne Diseases; Ranjbar, M., Nojomi, M., Mascellino, M.T., Eds.; IntechOpen: London, UK, 2019. [Google Scholar]
- Tominaga, T. Rapid detection of coliform bacteria using a lateral flow test strip assay. J. Microbiol. Methods 2019, 160, 29–35. [Google Scholar] [CrossRef]
| Strain | Gene | Primer Sequence | Type | 5’ Label | Size (bp) |
|---|---|---|---|---|---|
| M. fortuitum | 16 s | ACTTGCGCTTCGTCCCTAT | Forward | Biotin | 287 |
| ACCACGCATTTCATGGTCT | Reverse | Digoxigenin |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ben Aissa, A.; Araújo, B.; Julián, E.; Boldrin Zanoni, M.V.; Pividori, M.I. Immunomagnetic Separation Improves the Detection of Mycobacteria by Paper-Based Lateral and Vertical Flow Immunochromatographic Assays. Sensors 2021, 21, 5992. https://doi.org/10.3390/s21185992
Ben Aissa A, Araújo B, Julián E, Boldrin Zanoni MV, Pividori MI. Immunomagnetic Separation Improves the Detection of Mycobacteria by Paper-Based Lateral and Vertical Flow Immunochromatographic Assays. Sensors. 2021; 21(18):5992. https://doi.org/10.3390/s21185992
Chicago/Turabian StyleBen Aissa, Alejandra, Barbara Araújo, Esther Julián, Maria Valnice Boldrin Zanoni, and María Isabel Pividori. 2021. "Immunomagnetic Separation Improves the Detection of Mycobacteria by Paper-Based Lateral and Vertical Flow Immunochromatographic Assays" Sensors 21, no. 18: 5992. https://doi.org/10.3390/s21185992
APA StyleBen Aissa, A., Araújo, B., Julián, E., Boldrin Zanoni, M. V., & Pividori, M. I. (2021). Immunomagnetic Separation Improves the Detection of Mycobacteria by Paper-Based Lateral and Vertical Flow Immunochromatographic Assays. Sensors, 21(18), 5992. https://doi.org/10.3390/s21185992







